Abstract
Context
The roosting habits of many temperate zone bats are well documented at microhabitat scales, but fewer studies have included multi-scale assessments of landscape patterns in bat roost site selection.
Objectives
To identify and assess at the landscape-scale the location of spring and early season maternity roosts of female northern long-eared bats (Myotis septentrionalis) from 2015 to 2016 at Mammoth Cave National Park (MACA), Kentucky, USA.
Methods
We used mist-nets and radiotelemetry to catch and track bats to roost trees across the landscape of MACA. Data on roosting sites were evaluated using spatial point pattern analysis to examine distributional trends of roosts. A variety of spatial covariates were used to model the effect of landscape pattern, including: forest type, elevation, and proximity to hibernacula, water, and road corridors.
Results
Data indicate that roost locations of female northern long-eared bats in MACA were typically situated within 2000 m of known winter hibernacula, occurring more often at higher elevations in mesic upland deciduous forests, and in close proximity to water sources and roads. We present hypotheses to account for the patterns observed in relation to landscape features and habitat resources in the Park.
Conclusions
Our data indicate that a more comprehensive understanding of habitat requirements which includes empirically-based, landscape-scale patterns, and not solely considerations at stand or local levels, could lead to better informed management policies targeting conservation of maternity habitat of forest-dwelling bats, including the northern long-eared bat, a species in decline throughout much of its distribution in North America.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bats (Order: Chiroptera) constitute approximately one-fifth of all mammal species (Martin et al. 2011). They are broadly distributed, occupy a variety of feeding guilds, and may be the most abundant mammals on a local scale, especially in the tropics (Patterson et al. 2003; Gorresen et al. 2005). Anthropogenic forces worldwide, such as deforestation and fragmentation (Burgar et al. 2015; Toth et al. 2015; Rocha et al. 2017), urbanization and habitat loss (Lintott et al. 2015, 2016; Caryl et al. 2016), agricultural intensification (Azam et al. 2016; Cleary et al. 2016; Mendes et al. 2017), and alternate energy technologies (Peste et al. 2015; Ferri et al. 2016) are elevating the rates of species extinction and the loss of Chiropteran diversity. Globally, deforestation and fragmentation represent the most abrupt form of landscape change (Millennium Ecosystem Assessment 2005; Boughey et al. 2011). Unfortunately, an understanding of species-specific needs of bats at landscape levels, including responses to changes from anthropogenic forces, remains elusive.
The majority of studies on summer roosting ecology of bats in North America has focused on habitat conditions at the scale of the roost tree or surrounding forest stand (Lacki and Baker 2003; Kalcounis-Rüeppell et al. 2005; Barclay and Kurta 2007). Theoretical and empirical evidence suggests, however, that animal species rarely follow a linear association with gradients in habitat characteristics (Wiens 1989; Lord and Norton 1990; With and Crist 1995; Gorresen et al. 2005), and criteria that species use for habitat and resource selection likely vary by both landscape and proximal spatial scales. Use of multi-scale analyses in examining roost selection of North American bats has been achieved for foliage-roosting species (Veilleux et al. 2004; Limpert et al. 2007; Hein et al. 2008) and select Myotis species (Arnett and Hayes 2009; Lacki et al. 2010; Hammond et al. 2016; Jachowski et al. 2016), with a range of landscape patterns found to be beneficial depending on species and geographic location. Comparative studies are limited on landscape-level selection of tree roosts by the northern long-eared bat, Myotis septentrionalis (Pauli et al. 2015; Ford et al. 2016); a threatened species experiencing severe population declines across much of its distribution in North America (USDI 2015).
Spatial statistics of point patterns provide a rigorous format for describing distributions of species or any other spatially-temporally discrete events of interest (e.g., earthquake, fire ignition) and testing hypotheses about those distributions at larger spatial scales (Loosmore and Ford 2006; Law et al. 2009; Reiter and Anderson 2013). We employed spatial point pattern analysis to quantify patterns of spring and early maternity season roosts of adult, female northern long-eared bats at Mammoth Cave National Park, Kentucky, USA. Our objectives were to determine what landscape characteristics, if any, were important for roost selection of non-reproductive and reproductive (i.e., pregnant or lactating) female northern long-eared bats, and whether patterns in landscape characteristics helped explain outcomes for spatial locations of roosts of this species. Based on known patterns in roosting behavior of northern long-eared bats elsewhere in the distribution, we hypothesized these bats would roost within mesic upland deciduous forests (Foster and Kurta 1999; Menzel et al. 2002; Broders and Forbes 2004; Pauli et al. 2015), in close proximity to flyways and corridors such as water sources and roads (Henderson and Broders 2008; Perry et al. 2008); the latter presumably to enhance access to foraging sites elsewhere on the landscape. We also hypothesized that topography would influence the likelihood of roosting occurrences, as topographic features known to be important to female northern long-eared bats elsewhere include higher elevation sites and upper and mid-slope positions (Lacki and Schwierjohann 2001; Lacki et al. 2009; Krynak 2010). Finally, we hypothesized that female northern long-eared bats would roost near known winter hibernacula, especially during spring emergence, i.e., staging, when fat reserves are reduced and availability of insect prey remain at seasonal lows. Reproductive female bats are presumably more constrained by energy demands than male bats (Cryan et al. 2000; Willis and Wilcox 2014; Wilcox and Willis 2016), so it would be reasonable to assume that roost selection of female bats during staging would be consistent with minimizing movements and energy expenditures during an energetically challenging season of the year.
Materials and methods
Study area
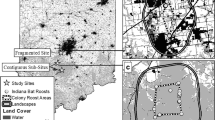
The study was located at Mammoth Cave National Park (MACA), situated within the Green River Valley in south central Kentucky, USA (Fig. 1). The Park is approximately 212 km2 and is positioned on a karst landscape recognized for the longest known cave system in the world. The limestone rocks beneath date to 325 million years ago during the Mississippian Period (Livesay 1953). Much of the landscape on and around the Park is pitted by depressions or sinkholes due to the karst topography, resulting in few surface streams other than the Green and Nolin Rivers. The Park ranges in elevation from 128 to 281-m above sea level, has a mean annual temperature of 14.9 °C, and an average annual rainfall of approx. 130 cm (U.S. Climate Data 2016).
The Mammoth Cave region is dominated by second-growth oak-hickory forest (USNPS 2016). The area is considered to be a transitional zone between open grasslands and oak-hickory forests to the west and mesophytic forests to the east. Likewise, the Park is situated between colder climates to the north and sub-tropical climates to the south. The different vegetation types create a mosaic of habitats across the Park that support a vast array of flora and fauna, including 43 mammal species (USNPS 2016). In 2002, a prescribed fire management plan was set in place at the Park to reduce fuel loads and restore the forest to pre-settlement conditions. Between 2002 and 2011, over 25% of the Park was burned with prescribed fire techniques (Lacki et al. 2014).
Capture and telemetry
Northern long-eared bats were captured from April to July, 2015 and 2016, using mist-nets measuring 6–18 m in length and stacked 6–9 m high (Avinet, Dryden, NY). Nets were placed at capture sites that included cave entrances, backcountry roads, and ephemeral ponds. Upon capture, the mass (g), right forearm length (mm), reproductive condition (females: pregnant, lactating or non-reproductive; males: scrotal or non-scrotal), Reichard’s wing index score (Reichard and Kunz 2009), sex, and age (Brunet-Rossinni and Wilkinson 2009) were collected for every individual. Adult females were grouped as non-reproductive if no evidence of pregnancy or lactation was visible; however, because all of these bats were captured in the post-hibernation staging period, many likely were reproductively active and would have demonstrated to be so if captured later in summer. Bats were identified to species and released at the site of capture. Myotis bats were banded with 2.9-mm bands provided by the Kentucky Department of Fish and Wildlife Resources. Adult, female northern long-eared bats receiving radio-transmitters were not banded to keep added weight < 5% of their body mass (Aldridge and Brigham 1988). All handling procedures included adherence to decontamination protocols laid out by the U.S. Fish and Wildlife Service (USFWS 2016).
Nineteen adult female northern long-eared bats and one juvenile male were captured and fitted with LB-2XT radio-transmitters (Holohil Systems, Ltd., Ontario, Canada) with surgical glue (Perma-Type Company, Inc., Plainville, CT) between the shoulder blades. Transmitters mass was ≤ 0.33 g to comply with the 5% rule (Aldridge and Brigham 1988). Bats were tracked daily for approximately 8-15 days or until the transmitter battery failed or fell off the bat. A 3-element yagi antenna (Wildlife Materials, Inc., Murphysboro, IL) and an Icom IC-R20 radio-receiver (Icom America, Inc., Kirkland, WA) were used to track bats. We located 69 roosts used by radio-tagged adult, female northern long-eared bats at MACA. For each roost identified, the coordinates were recorded with a Garmin GPS unit (Garmin International, Inc., Olathe, KS).
Modeling roosting habitat
Locations of spring and summer roost trees were geographically referenced using the UTM (Universal Transverse Mercator) Zone 16 N coordinate system. Spatial covariates (i.e., roadways, hydrology, and land cover) were mapped and processed using a geographic information system (ArcGIS 10.4.1, Redlands, CA). We obtained digital vegetation coverage, data for hydrology and roadways (L. Scoggins, MACA, U.S. National Park Service), and locations of known bat overwintering caves for analyses (R. Toomey, MACA, U.S. National Park Service). We used a point process modeling (PPM) approach to describe spring and summer roost locations based on an inhomogeneous Poisson process (Yang et al. 2007; Renner et al. 2015).
A spatial point pattern process (SPP) is a stochastic mechanism that generates a set of points in time and space which describe the locations of observed species or events (Law et al. 2009; Baddeley et al. 2015). Early ecological applications of SPP analysis were mainly to characterize spatial trend of points with first-order statistics for quantifying variations in expected density (also called intensity) of observed individuals across the sample space and to identify spatial interaction (i.e., clustering, regularity, and random) among points with second-order statistics such as Ripley’s K function (Perry et al. 2006; Law et al. 2009). Recent theoretical developments in SPP have provided a rigorous framework and versatile diagnostic tools to fit the observed point data to underlying point processes (e.g., Poisson, Cox, Strauss) (Baddeley et al. 2015). While methods for fitting spatial point pattern data are closely related to common regression models, PPMs have considerable potential in modeling presence-only data with various advantages, including (but not limited to) (1) explicit focus on where the points were observed, (2) clarity of model assumptions and tools for checking them, and (3) direct handle of spatial dependence between points (Renner et al. 2015). Kernel intensity estimation and Ripley’s K function were calculated to describe spatial patterns (i.e., clustering or regularity) of roosts at the Park (Yang et al. 2007).
Inhomogeneous Poisson point process models were used to fit the observed roost tree location data, which assume that expected number of roost trees per unit area varies spatially and roost trees are independent of each other at the scale of our investigation. It has been shown that Poisson point process model is equivalent to the popular entropy-based MAXENT model but with a transparent modeling structure (Renner and Warton 2013). We examined a variety of spatial covariates with transformations in the inhomogeneous Poisson models including elevation, southwestness, distance to water, distance to roads, distance to winter hibernacula, and proportion of mesic upland deciduous forest. Residual analysis for the spatial point processes and Akaike’s Information Criterion (AIC) methods were used to select variables with a backward-stepwise model selection to find the best fit model for the data (Yang et al. 2007).
Spatial covariates were chosen for analysis, in part, on the basis of prior research indicating patterns of habitat selection by northern long-eared bats. For example, the digital vegetation layer contained nine different vegetation classes. The ‘mesic upland deciduous’ vegetation class was chosen based off prior studies that indicated use of this habitat type by the northern long-eared bat (Foster and Kurta 1999; Menzel et al. 2002; Broders and Forbes 2004; Henderson and Broders 2008). We used a moving window analysis (30 × 30 m cell size) to determine the proportion of mesic upland deciduous forest within a neighborhood (i.e., the window) for every location within the Park. The purpose of moving window analysis was to create a GIS variable that could describe local-scale vegetation composition and transform the categorical vegetation class GIS variable into a continuous variable. The continuous vegetation variable was then examined in spatial point pattern modeling to quantify vegetation effects on roosting locations. Proximities to road and water were determined by calculating the Euclidian distance from each cell (30-m resolution) to the nearest road or water source, a function provided by the ArcGIS Spatial Analyst tool. We used a digital elevation model (DEM) published by the Kentucky Geological Survey (Kentucky Geological Survey 1998). Slope and aspect were calculated from DEM data with the surface analysis provided by the ArcGIS Spatial Analyst tool. Calculated aspect azimuths were later transformed into southwestness using the equation (cos(aspect)-225) to change the circular aspect to a linear gradient for indexing potential incident solar radiation (southwestness). We used a Euclidian distance function to determine distances from roost locations to the nearest known winter bat caves. A lurking plot variable was used to identify non-linear or spatial trends in the point processes (Baddeley and Turner 2005). First, the cumulative residual is plotted against a select spatial covariate. Then noticeable trends in the lurking variable plot are accounted for and appropriately modified for that spatial covariate. Eventually, selected covariates were plotted in R with a polynomial function up to the power of two based on the final model coefficients to model the marginal effects of the variables on roost likelihood (Yang et al. 2015). All the analyses were conducted in the R software environment with the spatstat package (Baddeley and Turner 2005).
Results
The non-parametric kernel density estimation showed a high concentration of roosts in the northwest section of the Park, indicating the roost occurrence pattern was not completely random (Fig. 2). The minimum distance between any two roosts was 4.24 m, with an average nearest neighbor distance of 108 m ± 12.4(SE), and a maximum distance of 381 m. The estimated K function was larger than theoretical complete spatial randomness (CSR), indicating the locations of roosts on the landscape exhibited a spatial clustering pattern (Fig. 3). Regardless, strong spatial dependence could be due to either bat behavior, i.e., fission–fusion, or to association with clustering of environmental factors (i.e., vegetation type, elevation, etc.) on the landscape.
The inhomogeneous Poisson process model demonstrated that spatial clustering of roosts could be accounted for by environmental heterogeneity. The null model (homogeneous Poisson) assumes that roost locations are equally likely across the landscape. The cumulative Pearson residuals were plotted against the seven spatial covariates (i.e., distance to road, distance to water, distance to winter caves, slope, elevation, southwestness, and proportion of mesic upland deciduous forest) and the two Cartesian coordinates (x and y) for the null model. The cumulative Pearson residuals for the predicted random values exceeded the observed roost occurrence values, suggesting that the null model underestimated roost likelihood at this scale.
Lurking variables plotted against the null model of roost occurrence on the Park landscape indicated clear systematic patterns (Fig. 4). Female northern long-eared bats selected roost locations within approximately 500 m of known roadways (Fig. 4a), 800 m of water sources (Fig. 4b), and between approximately 2000 and 4000 m away from known winter bat hibernacula (Fig. 4c). Female bats avoided potential roost locations at elevations between 198 and 259 m (Fig. 4d). A lurking plot of the proportion of mesic upland deciduous forest with the 900-m moving window analysis of vegetation cover type revealed the cumulative residuals of the null hypothesis were smaller than expected for areas where the proportion of mesic upland deciduous forest was less than 80% (Fig. 4e). This suggests that female northern long-eared bats in MACA had a strong preference towards mesic upland deciduous forests. The last two variable plots, slope and southwestness, exhibited empirical curves of cumulative Pearson residuals within the two-standard-deviation error bounds, suggesting bats did not preferentially choose roost sites by aspect or slope position.
Lurking variable plots of a distance to road, b distance to water, c distance to winter caves, d elevation (DEM), e proportion of vegetation code 3 (mesic upland deciduous), f slope, and g southwestness (aspect) for the null model of roost occurrence on the landscape at Mammoth Cave National Park, Kentucky, USA. Solid lines indicate empirical curves of cumulative Pearson residuals. Shaded areas denote two-standard-deviation error bounds
A full range of alternative models were considered that included all possible combinations of the potential spatial covariates examined. The full model (AIC = 2113.2) predictors included distance to road, distance to water, distance to winter caves, elevation, southwestness, slope, and mesic upland deciduous forest. Due to most continuous variables displaying curvilinear relationships, the full model also included second order transformations for all covariates except mesic upland deciduous cover type and southwestness. The best fit model possessed an AIC = 2100.3 (Table 1). The second order distance to water, first order distance to roads, and both first and second order distances to winter caves were retained in the best fit model (P < 0.01). The second order elevation and first order mesic upland deciduous forest cover type were also retained but with a larger P value (P < 0.1). Slope and southwestness were excluded during the model selection process.
The best fit models generated a prediction map of likely roosting locations of female northern long-eared bats at MACA. Dark blue areas on the left top panel of the map indicated areas the model predicted to have the highest likelihood of roost occurrence on the landscape (Fig. 5). However, the cumulative sum of raw residuals did not fit within the two-standard-deviation error bounds in the northwest portion of the Park (in red), meaning the model did not account for all variations in the data.
Effects of likelihood of roost occurrence within the Park were plotted against three variables: elevation, distance to water and distance to winter caves (Fig. 6). Elevation appears to have a positive association with probability of roost location, suggesting higher elevation areas are preferred habitats of female northern long-eared bats during staging and the early maternity season. Distance to water demonstrated a monotonically decreasing pattern indicating that probability of roost location decreases as distance to the nearest water source increases. The distance to known overwintering hibernacula within the Park shows an inflection point at approximately 2000 m. Bats appeared to select roosts farther away from winter caves up to the inflection point. Beyond the inflection point distance, the likelihood of roosts occurring on the landscape dropped significantly.
Discussion
Studies have demonstrated that responses of bats to spatial structure of habitats is highly dependent on focal scale (Gorresen et al. 2005; Perry et al. 2008; O’Keefe et al. 2009). Proximity to water sources, foraging areas, and topography (i.e., slope position, elevation, aspect) can all potentially affect roost selection by bats at the landscape scale (Perry et al. 2008). Multi-scale patterns in roost selection of other bat species in southeastern North America have demonstrated eastern red bats (Lasiurus borealis) to favor mature streamside management areas within intensively managed pine plantations (Elmore et al. 2005), and to select roosting sites near open, urban land and water compared with random sites (Limpert et al. 2007). Distance to corridors and mature pine forest was associated with roost selection in Seminole bats (L. seminolus; Hein et al. 2008), and tri-colored bats preferentially selected riparian and upland forests over bottomland habitats (Veilleux et al. 2004).
We observed that during the spring and early maternity seasons, roosts of female northern long-eared bats were spatially clustered on the landscape, consistent with patterns expected for bats which form roost-networks during the summer maternity season (Garroway and Broders 2007; Johnson et al. 2012). We identified several environmental factors that served as important determinants of spatial locations for these bats in MACA, with our results corroborating findings that demonstrated female northern long-eared bats to choose roosts in proximity to roads (Perry et al. 2008; Pauli et al. 2015). Roads have been directly associated with flight corridors and improved access to suitable areas for foraging by bats (Limpens and Kapteyn 1991; Walsh and Harris 1996). Our findings also indicated a strong positive correlation of roosts of northern long-eared bats with available sources of water. Empirical evidence has demonstrated the importance of nearby water sources in roost tree selection by several other temperate-zone bat species (Kalcounis-Rüeppell et al. 2005; Limpert et al. 2007; Perry et al. 2008).
Female northern long-eared bats at MACA had a strong preference toward selection of roosts within the vegetation cover type described as mesic upland deciduous forest. The lurking variable plot of the proportion of mesic upland deciduous forest, combined with the moving window analysis, indicated that habitats where these bats chose roost trees at MACA, on average, had up to 80% of habitat patches in the mesic upland deciduous forest cover type. These results are consistent with findings elsewhere across the distribution of the species that demonstrated preference for roosts in deciduous trees within relatively contiguous forests (Foster and Kurta 1999; Menzel et al. 2002; Henderson and Broders 2008; Pauli et al. 2015). We also observed northern long-eared bats preferentially choosing roosts in higher elevation habitats that are away from the cold bottomlands, and suggest that roosts situated at higher elevation sites reflected the needs of adult females to inhabit structures possessing warmer microclimates. Warmer roosting microclimates could potentially reduce the cost of maintaining normothermic body temperatures during the early reproductive season, and presumably help facilitate parturition, lactation and the development of young. Landscape-scale selection of roosts at high elevations has also been demonstrated for the Indiana bat in southern Appalachian Mountains (Hammond et al. 2016).
Due to MACA’s karst topography, location of known overwintering hibernacula was a novel landscape feature not previously examined in any other landscape scale study on bats. The elevation of the water table in MACA, which is roughly the elevation of the Green River, corresponds with the elevation where many cave entrances occur (DiPietro 2013). As the Green River cuts downslope into the landscape, active cave formation drops to lower levels, leaving dry caves higher up in elevation. The uppermost passages of Mammoth Cave are located between 174 and 210 m in elevation, with the oldest and largest cave openings occurring at ground level or 227 m (DiPietro 2013). Female northern long-eared bats appeared to select roost locations increasingly farther away from known overwintering caves up to 2000 m in distance; however, no female bat chose a roost location beyond the 2000 m distance threshold from any known winter hibernaculum. We suggest two possibilities for this pattern. First, the relatively close proximity to overwintering caves allows for conspecifics to remain in contact and eventually regroup after the hibernation period in spring (i.e., staging behavior), facilitating formation of local summer maternity colonies. Second, as hypothesized, by minimizing distances moved from hibernacula in early spring, when temperatures are cooler on average and availability of insect prey less predictable, adult female bats are limiting energy expenditures to help maintain a positive energy balance at a time when many are pregnant and allocating energetic resources to the developing fetus.
Maintaining a buffer of at least 2000 m surrounding known overwintering caves of the northern long-eared bat would help to ensure the continued availability of suitable roosting sites for the species throughout the Park. The northern long-eared bat has declined significantly enough across its range over the past decade for the species to be added as threatened under the Endangered Species Act (USDI 2015). It is presently unknown whether these population trends will lead to permanent and lasting reductions in the abundance of this species. Thus, sustaining maternity habitats of the northern long-eared bat at landscape scales in geographic locations where they are still known to occur is imperative to conservation efforts for the recovery of the species across its distribution.
Assessing the status of a species requires an understanding of the basic biology, ecology, population size and trends over time (Alberta Sustainable Resource Development and Alberta Conservation Association 2009). Ultimately, more information about the basic ecology of bats is needed to effectively conserve them, with access to shelter, food, and water resources necessary to secure the survival of bat populations globally (Fenton and Simmons 2015). Our research used a point process pattern analysis to examine landscape-level patterns in roost tree selection of adult female northern long-eared bats at MACA. We found that during the spring staging and early maternity seasons, roosts of these bats were spatially clustered on the landscape, with landscape features including elevation, and distances to roads, water, and overwintering hibernacula being important determinants of spatial location of these roosts. For at least MACA, preferred roost tree locations of female northern long-eared bats are situated within 2000 m of known winter hibernacula at higher elevation sites supporting mesic upland deciduous forests. Management of habitats at the stand level remains fundamental in fostering increases in bat colony numbers during the maternity season (O’Donnell 2000; Willis and Brigham 2004; Garroway and Broders 2008); however, our findings also confirm the importance of larger spatial scales when developing long-term planning efforts for maternity habitat of the northern long-eared bat.
Bat populations in eastern North America are continually being threatened by anthropogenic forces and, now with the onset of the fungal (Pseudogymnoascus destructans) disease white-nose syndrome (WNS), face even greater challenges to survival moving forward (Blehert et al. 2009; USFWS 2017). It is presently unclear just how these impacts will interact synergistically to produce population-level effects, or whether declines in bat populations will continue and lead to permanent and lasting shifts in species relative abundance (Moosman et al. 2013; Reynolds et al. 2016; Thalken et al. 2018). Regardless, habitat needs of bats at the landscape scale, especially maternity habitat, should remain a top conservation priority and our analyses indicate that landscape features are important to location of maternity roosts of northern long-eared bats. We encourage further data collection on roost selection of forest-dwelling bats in eastern North America to facilitate management and recovery efforts of bat species, especially those affected by WNS.
References
Alberta Sustainable Resource Development and Alberta Conservation Association (2009) Status of the northern myotis (Myotis septentrionalis) in Alberta: Update 2009. Alberta Sustainable Resource Development. Wildlife Status Report No. 3 (Update 2009). Edmonton
Aldridge HDJN, Brigham RM (1988) Load carrying and maneuverability in an insectivorous bat: a test of the 5% “rule” of radio-telemetry. J Mammal 69:379–382
Arnett EB, Hayes JP (2009) use of conifer snags as roosts by female bats in western Oregon. J Wildl Manag 73:214–225
Azam C, Le Viol I, Julien J, Bas Y, Kerbiriou C (2016) Disentangling the relative effect of light pollution, impervious surfaces and intensive agriculture on bat activity with a national-scale monitoring program. Landscape Ecol 31:2471–2483
Baddeley A, Rubak E, Turner R (2015) Spatial point patterns: methodology and applications with R. CRC Press, Boca Raton
Baddeley A, Turner R (2005) Spatstat: an R package for analyzing spatial point patterns. J Stat Softw 12:1–42
Barclay RMR, Kurta A (2007) Ecology and behavior of bats roosting in tree cavities and under bark. In: Lacki MJ, Hayes JP, Kurta A (eds) Bats in forests: conservation and management. Johns Hopkins University Press, Baltimore, MD, pp 17–59
Blehert DS, Hicks AC, Behr M, Meteyer CU, Berlowski-Zier BM, Buckles EL, Coleman JTH, Darling SR, Gargas A, Niver R, Okoniewski JC, Rudd RJ, Stone WB (2009) Bat white-nose syndrome: an emerging fungal pathogen? Science 323:227
Boughey KL, Lake IR, Haysom KA, Dolman PM (2011) Effects of landscape-scale broadleaved woodland configuration and extent on roost location for six bat species across the U.K. Biol Conserv 144:2300–2310
Broders HG, Forbes G (2004) Interspecific and intersexual variation in roost-site selection of northern long-eared and little brown bats in the Greater Fundy National Park ecosystem. J Wildl Manag 68:602–610
Brunet-Rossinni AK, Wilkinson GS (2009) Methods for age estimation and the study of senescence in bats. In: Kunz TH, Parsons S (eds) Ecological and behavioral methods for the study of bats, 2nd edn. Johns Hopkins University Press, Baltimore, MD, pp 315–325
Burgar JM, Craig MD, Stokes VL (2015) The importance of mature forest as bat roosting habitat within a production landscape. For Ecol Manag 356:112–123
Caryl FM, Lumsden LF, van der Ree R, Wintle BA (2016) Functional responses of insectivorous bats to increasing housing density support “land-sparing’ rather than “land-sharing’ urban growth strategies. J Appl Ecol 53:191–201
Cleary KA, Waits LP, Finegan B (2016) Agricultural intensification alters bat assemblage composition and abundance in a dynamic Neotropical landscape. Biotropica 48:667–676
Cryan PM, Bogan MA, Altenbach JS (2000) Effect on elevation on distribution of female bats in the Black Hills, South Dakota. J Mammal 811:719–725
DiPietro JA (2013) Landscape evolution in the United States: an introduction to the geography, geology, and natural history. Elsevier, Waltham, MA
Elmore LW, Miller DA, Vilella FJ (2005) Foraging area size and habitat use by eastern red bats (Lasiurus borealis) in an intensively managed pine landscape in Mississippi. Am Midl Nat 153:405–417
Fenton MB, Simmons NB (2015) Bats: a world of science and mystery. University Chicago Press, Chicago, IL
Ferri V, Battisti C, Soccini C (2016) Bats in a Mediterranean mountainous landscape: does wind farm repowering induce changes at assemblage and species levels? Environ Manag 57:1240–1246
Ford WM, Silvis A, Rodrigue JL, Kniowski AB, Johnson JB (2016) Deriving habitat models for northern long-eared bats from historical detection data: a case study using the Fernow Experimental Forest. J Fish Wildl Manag 7:86–98
Foster R, Kurta A (1999) Roosting ecology of the northern bat (Myotis septentrionalis) and comparisons with the endangered Indiana bat (Myotis sodalis). J Mammal 80:659–672
Garroway CJ, Broders HG (2007) Nonrandom association patterns at northern long-eared bat maternity roosts. Can J Zool 85:956–964
Garroway CJ, Broders HG (2008) Day roost characteristics of northern long-eared bats (Myotis septentrionalis) in relation to female reproductive status. Ecoscience 15:89–93
Gorresen PM, Willig MR, Strauss RE (2005) Multivariate analysis of scale-dependent associations between bats and landscape structure. Ecol Appl 15:2126–2136
Hammond KR, O’Keefe JM, Aldrich SP, Loeb SC (2016) A presence-only model of suitable roosting habitat for the endangered Indiana bat in the southern Appalachians. PLoS ONE 11(4):e0154464
Hein CD, Castleberry SB, Miller KV (2008) Sex-specific summer roost site selection by Seminole bats in response to landscape-level forest management. J Mammal 89:964–972
Henderson LE, Broders HG (2008) Movements and resources selection of the northern long-eared myotis (Myotis septentrionalis) in a forest-agriculture landscape. J Mammal 89:952–963
Jachowski DS, Rota CT, Dobony CA, Ford WM, Edwards JW (2016) Seeing the forest through the trees: considering roost-site selection at multiple spatial scales. PLoS ONE 11(3):e0150011
Johnson JB, Ford WM, Edwards JW (2012) Roost networks of northern myotis (Myotis septentrionalis) in a managed landscape. For Ecol Manag 266:223–231
Kalcounis-Rüppell MC, Psyllakis JM, Brigham RM (2005) Tree roost selection by bats: an empirical synthesis using meta-analysis. Wildl Soc Bull 33:1123–1132
Kentucky Geological Survey (1998) Elevation data. Web. http://www.uky.edu/KGS/gis/ElevationData.html. Accessed 1 April 2015
Krynak TJ (2010) Bat habitat use and roost tree selection for northern long-eared myotis (Myotis septentrionalis) in north-central Ohio. MS thesis. John Carroll University, Cleveland, OH
Lacki MJ, Baker MD (2003) A prospective power analysis and review of habitat characteristics used in studies of tree-roosting bats. Acta Chiropterol 5:199–208
Lacki MJ, Baker MD, Johnson JS (2010) Geographic variation in roost-site selection of long-legged myotis in the Pacific Northwest. J Wildl Manag 74:1218–1228
Lacki MJ, Cox DR, Dodd LE, Dickinson MB (2009) Response of northern bats (Myotis septentrionalis) to prescribed fires in eastern Kentucky forests. J Mammal 90:1165–1175
Lacki MJ, Dodd LE, Skowronski NS, Dickinson MB, Rieske LK (2014) Fire management and habitat quality for endangered bats in Kentucky’s Mammoth Cave National Park during the swarming and staging periods: predator-prey interactions and habitat use of bats threatened by white-nose syndrome. USDA Joint Fire Science Program, Final Report No.: 10-1-06-1
Lacki MJ, Schwierjohann JH (2001) Day-roost characteristics of northern bats in mixed mesophytic forest. J Wildl Manag 65:482–488
Law R, Illian J, Burslem DF, Gratzer G, Gunatilleke CVS, Gunatilleke IAUN (2009) Ecological information from spatial patterns of plants: insights from point process theory. J Ecol 97(4):616–628
Limpens HJGA, Kapteyn K (1991) Bats, their behavior and linear landscape elements. Myotis 29:63–71
Limpert DL, Birch DL, Scott MS, Andre M, Gillam E (2007) Tree selection and landscape analysis of eastern red bat day roosts. J Wildl Manag 71:478–486
Lintott PR, Barlow K, Bunnefeld N, Briggs P, Gajas Roig C, Park KJ (2016) Differential responses of cryptic bat species to the urban landscape. Ecol Evolut 6:2044–2052
Lintott PR, Bunnefeld N, Minderman J, Fuentes-Montemayor E, Mayhew RJ, Olley L, Park KJ (2015) Differential responses to woodland character and landscape context by cryptic bats in urban environments. PLoS ONE 10(5):e0126850
Livesay A (1953) Geology of the Mammoth Cave National Park area. Kentucky Geology Survey, University of Kentucky 7:1–40
Loosmore NB, Ford ED (2006) Statistical inference using the G or K point pattern spatial statistics. Ecology 87(8):1925–1931
Lord JM, Norton DA (1990) Scale and the spatial concept of fragmentation. Conserv Biol 4:197–202
Martin RE, Pine RH, DeBlase AF (2011) A manual of mammalogy: with keys to families of the world. Waveland Press, Long Grove, IL
Mendes ES, Fonseca C, Marques SF, Maia D, Ramos Pereira MJ (2017) Bat richness and activity in heterogeneous landscapes: guild-specific and scale-dependent? Landscape Ecol 32:295–311
Menzel MA, Owen SF, Ford WM, Edwards JW, Wood PB, Chapman BR, Miller KV (2002) Roost tree selection by northern long-eared bat (Myotis septentrionalis) maternity colonies in an industrial forest of the central Appalachian Mountains. For Ecol Manag 155:107–114
Millennium Ecosystem Assessment (2005) Ecosystems and Human Well-being: Biodiversity Synthesis. World Resources Institute, Washington, DC
Moosman PR Jr, Veilleux JP, Pelton GW, Thomas HH (2013) Changes in capture rates in a community of bats in New Hampshire during the progression of white-nose syndrome. Northeast Nat 20:552–558
O’Donnell CFJ (2000) Cryptic local populations in a temper- ate rainforest bat Chalinolobus tuberculatus in New Zealand. Anim Conserv 3:287–297
O’Keefe JM, Loeb SC, Lanham JD, Hill HS Jr (2009) Macrohabitat factors affect day roost selection by eastern red bats and eastern pipistrelles in the southern Appalachian Mountains, USA. For Ecol Manag 257:1757–1763
Patterson BD, Willig MR, Stevens RD (2003) Trophic strategies, niche partitioning, and patterns of ecological organization. In: Kunz TH, Fenton MB (eds) Bat ecology. University of Chicago Press, Chicago, IL, pp 536–579
Pauli BP, Badin HA, Haulton GS, Zollner PA, Carter TC (2015) Landscape features associated with the roosting habitat of Indiana bats and northern long-eared bats. Landscape Ecol 30:2015–2029
Perry GL, Miller BP, Enright NJ (2006) A comparison of methods for the statistical analysis of spatial point patterns in plant ecology. Plant Ecol 187(1):59–82
Perry RW, Thill RE, Leslie DM (2008) Scale-dependent effects of landscape structure and composition on diurnal roost selection of forest bats. J Wildl Manag 72:913–925
Peste F, Paula A, da Silva LP, Bernardino J, Pereira P, Mascarenhas M, Costa H, Vieira J, Bastos C, Fonseca C, Ramos Pereira MJ (2015) How to mitigate impacts of wind farms on bats? A review of potential conservation measures in the European context. Environ Impact Assess Rev 51:10–22
Reichard JD, Kunz TH (2009) White-nose syndrome inflicts lasting injuries to the wings of little brown myotis (Myotis lucifugus). Acta Chiropterol 11:457–464
Reiter ME, Andersen DE (2013) Evidence of territoriality and species interactions from spatial point-pattern analyses of subarctic nesting geese. PLoS ONE 8:e81029
Renner IW, Elith J, Baddeley A, Fithian W, Hastie T, Phillips SJ, Popovic G, Warton DI (2015) Point process models for presence-only analysis. Methods Ecol Evol 6(4):366–379
Renner IW, Warton DI (2013) Equivalence of MAXENT and Poisson point process models for species distribution modeling in ecology. Biometrics 69(1):274–281
Reynolds RJ, Powers KE, Orndorff W, Ford WM, Hobson CS (2016) Changes in rates of capture and demographics of Myotis septentrionalis (Northern Long-eared Bat) in western Virginia before and after onset of white-nose syndrome. Northeast Nat 23:195–204
Rocha R, Lopez-Baucells A, Farneda FZ, Groenenberg M, Bobrowiec PED, Cabeza M, Palmeirim JM, Meyer CFJ (2017) Consequences of a large-scale fragmentation experiment for Neotropical bats: disentangling the relative importance of local and landscape-scale effects. Landsc Ecol 32:31–45
Thalken MM, Lacki MJ, Johnson JS (2018) Shifts in the assemblage of foraging bats at Mammoth Cave National Park following arrival of white-nose syndrome. Northeast Nat 25:202–214
Toth CA, Cummings G, Dennis TE, Parsons S (2015) Adoption of alternative habitats by a threatened, “obligate” forest-dwelling bat in a fragmented landscape. J Mammal 96:927–937
US Climate Data (2016) Climate- Mammoth Cave National Park, Kentucky. http://www.usclimatedata.com/climate/mammoth-cave/kentucky/united-states/usky1113. Accessed 1 Sept 2016
US Department of the Interior (2015) Final register for endangered and threatened wildlife and plants; threatened species status for the northern long-eared bat with 4(d) rule. Fish and Wildlife Service. vol 80. No. 63
US Fish and Wildlife Service (2016) White-nose syndrome decontamination protocol Version 4.12.2016. Web. https://www.whitenosesyndrome.org/sites/default/files/resource/national_wns_decon_protocol_04.12.2016.pdf. Accessed 25 Oct 2016
US Fish and Wildlife Service (2017) Bats affected by WNS. Web. https://whitenosesyndrome.org/about/bats-affected-wns. Accessed 24 March 2017
US National Park Service (2016) Learn about the park: Nature. Web. https://www.nps.gov/maca/learn/nature/index.htm. Accessed 20 Oct 2016
Veilleux JP, Whitaker JO Jr, Veilleux SL (2004) Reproductive stage influences roost use by tree roosting eastern pipistrelles, Pipistrellus subflavus. Ecosicence 11:249–256
Walsh AL, Harris S (1996) Foraging habitat preferences of verspertilionid bats in Britain. J Appl Ecol 33:508–518
Wiens JA (1989) Spatial scaling in ecology. Funct Ecol 3:385–397
Wilcox A, Willis CKR (2016) Energetic benefits of enhanced summer roosting habitat for little brown bats (Myotis lucifugus) recovering from white-nose syndrome. Conserv Physiol 4:1–7
Willis CKR, Brigham RM (2004) Roost switching, roost sharing, and social cohesion: forest-dwelling big brown bats, Eptesicus fuscus, conform to fission–fusion model. Anim Behav 68:495–505
Willis CKR, Wilcox A (2014) Hormones and hibernation: possible links between hormone systems, winter energy balance and white-nose syndrome in bats. Horm Behav 66:66–73
With KA, Crist TO (1995) Critical thresholds in species’ responses to landscape structure. Ecology 76:2446–2459
Yang J, He HS, Shifley SR, Gustafson EJ (2007) Spatial patterns of modern period human-caused fire occurrence in Missouri Ozark highlands. For Sci 53:1–15
Yang J, Weisberg PJ, Dilts TE, Loudermilk EL, Scheller RM, Stanton A, Skinner C (2015) Predicting wildfire occurrence distribution with spatial point process models and its uncertainty assessment: a case study in the Lake Tahoe Basin, USA. Int J Wildl Fire 24:38–390
Acknowledgements
We thank the National Park Service, the Walt Disney Foundation, and the University of Kentucky, College of Agriculture, for funding support. We are appreciative of R. Toomey (Mammoth Cave National Park), S. Thomas (National Park Service), and L. Dodd (Eastern Kentucky University) for assistance during the planning and implementation of this study. We are grateful to all the field technicians including: E. Stanmyer, B. Daly, T. Walters, M. Barnes, E. Lee, J. Ayres, H. Dykes, S. Zumdick, Z. Hackworth, M. McKenna, E. Kester, S. Fulton, and Z. Fry. All animal handling procedures used were approved by the University of Kentucky under IACUC Assurance No.: A3336-01. Data collection was supported through permits from the Kentucky Department of Fish and Wildlife Resources (SC1611176; SC1511245) and the U.S. Fish and Wildlife Service (TE38522A-1). The information reported in this paper (No. 17-09-048) is part of a project of the Kentucky Agricultural Experiment Station and is published with the approval of the Director.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Thalken, M.M., Lacki, M.J. & Yang, J. Landscape-scale distribution of tree roosts of the northern long-eared bat in Mammoth Cave National Park, USA. Landscape Ecol 33, 1103–1115 (2018). https://doi.org/10.1007/s10980-018-0659-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-018-0659-3