Abstract
Context
Intensification of land-use caused a reduction of ecosystem heterogeneity and diversity, and subsequently led to dramatic decrease of biodiversity. Species depending on dynamic ecosystems are particularly affected from this trend of land-use intensification, landscape homogenization, and the optimization of land-use. Forest species suffer under the intensification of forest management, in the worst case transforming light and heterogeneous deciduous forests into species-poor intensively used deciduous forests optimized for wood production. This lead to the destruction of a mosaic consisting of various successional stages in parallel.
Objective
In this study we analyse the relevance of forest heterogeneity, forest disturbance and microhabitat preferences of egg oviposition and larval development for a highly endangered butterfly species, Euphydryas maturna. This butterfly species mainly occurs in light and moist deciduous forests, such as riparian forests along mountain streams in northern Austria.
Methods
We combine detailed field observations with high resolution aerial pictures taken with an Unmanned Aerial Vehicle (UAV) to build ensemble habitat suitability models from GAM, GBM, GLM, and Maxent models.
Results
We found that egg ovipositions take place exclusively on the tree species Fraxinus excelsior, preferably exposed to the south, partly shaded, and at medium height (3 m). Our habitat suitability models based on high resolution aerial pictures indicate that egg ovipositions are clustered and accumulate along forest edges and at sites with high forest heterogeneity.
Conclusion
Our study underlines the high relevance and importance of light deciduous forest structures with environmental dynamics creating the preconditions of specific microhabitat structures for endangered species, such as E. maturna. Our study shows that UAV-captured high precision aerial imagery are well suited to optimally connect two spatial scales, the ecosystem and microhabitat scale.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The transformation of natural habitats into anthropogenic landscapes such as intensively used pastures, agricultural fields, settlements and forests optimized for wood production caused a dramatic loss of habitats and biodiversity during the past decades (Maxwell et al. 2016). These land-use changes frequently lead to the destruction of heterogeneous and species rich ecosystems into homogenous and species-poor landscapes (Gámez-Virués et al. 2015). Apart from the complete loss of natural and near natural ecosystems, the remaining habitats frequently suffer under the deterioration of habitat quality through increased atmospheric nitrogen depositions and the influx of pesticides, among others (Thomas et al. 2016). This deterioration of habitat quality negatively impacts local plant and animal populations (Dennis and Eales 1997). Particularly sensitive species demanding specific habitat conditions suffer under reduced habitat quality, as documented for various butterfly species across Central Europe (Thomas et al. 2011; Thomas 2016; Habel et al. 2021).
A very relevant prerequisite to the existence of many plant and animal species are ecosystem dynamics and the co-existence of various successional stages (Kuuluvainen 2009). However, human demographic pressure and increasing life-standards caused a rising demand for more land to settle and to cultivate for the production of food and energy crops (Steffan-Dewenter et al. 2007). This trend also affects forests, which are frequently transformed from natural forest into forests for intense wood production (Kirby and Watkins 2015). This counteracts natural dynamics such as forest succession, natural rejuvenation, and the availability of ecotones (Skrajna 2020). As a result, dynamic environments exist today only sporadically. However, many species rely on such dynamic environments (Bubová et al. 2015). Last remnant ecosystems shaped by strong environmental dynamics can still be rarely found along non-regulated streams and rivers (Naiman et al. 1993; Ward et al. 1999; Trockner and Stanford 2002).
Forest biodiversity suffers in particular under management intensification across major parts of Central Europe (Seibold et al. 2015), turning light and heterogeneous deciduous forests into comparatively dark and uniform monocultures of coniferous trees (Naumov et al. 2018). However, most forest species rely on light forests and heterogeneous vegetation structures consisting of various (deciduous) trees species, shrubs, and herbs, and the co-existence of various successional stages (Kozák et al. 2021). A prominent example is the scarce fritillary butterfly Euphydryas maturna. This butterfly depends on light and humid deciduous forests with Fraxinus excelsior (Freese et al. 2006) and is usually found along transitions of forest and open land, along forest pastures, and in light forests creating mosaics of trees, shrubs and grassland (Fartmann et al. 2013). Such forest structures are known to be hotspots of biodiversity (Horák et al. 2018).
In this study we analyse ecosystem structures and microhabitat preferences for oviposition of the scarce fritillary E. maturna, across sparse deciduous riparian forests along the highly dynamic Taugl River in northern Austria (close to the village Hintersee). We assessed caterpillar nests and habitat parameters (manually) across our study region and took high-resolution aerial photographs with a UAV. We combined these datasets to calculate habitat suitability models to project the occurrence of larvae of this butterfly and to quantify ecosystem structures and microhabitat preferences. Based on our data and results we will answer the following questions:
-
1.
Which ecosystem structures are preferably used for oviposition of E. maturna?
-
2.
Which microhabitat conditions are of relevance for oviposition and successful larval development?
-
3.
What conservation conclusions can be derived from our findings for this butterfly species, and for forest biodiversity in general?
-
4.
What are the advantages and limitations of using high-resolution aerial imagery through drones?
Material and methods
Study species
The scare fritillary butterfly, E. maturna, is a typical continental woodland butterfly and occurs across the Palaearctic region (Kudrna et al. 2011). This butterfly species is mainly found in open, sunny, humid, and sheltered gaps of open, sparse deciduous woodlands (Freese et al. 2006; Dolek et al. 2013), being on the wing mainly in June, in one generation (Ebert and Rennwald 1991). Oviposition usually takes place in batches (one batch consists out of about 250 eggs) (Eliasson and Shaw 2003) mainly on Fraxinus excelsior (with some exceptions observed, e.g. Ligustrum vulgare in eastern Austria, see Straka 2014, or different species of herbs in Sweden, see Eliasson 2001b). Females mostly prefer small trees and oviposit at heights between 1.5 and 3 m above the ground, along forest edges, at forest clearings and along road verges (Gros 2002; Dolek et al. 2006). Mortality of larvae increases under full sun (Dolek et al. 2006) and with increasing duration of sunshine (Weidemann 1985; Ebert and Rennwald 1991; Wahlberg 1998, 2000, 2001; Eliasson 2001a). Preference of sites for oviposition not only depends on habitat structures, but also on female behaviour. Studies showed that the more nests of larvae in a tree the higher is the survival probability of larvae. In consequence, females often oviposit in trees where already eggs have been layed by other females (Dolek et al. 2013). This behaviour might be related with parasitoid pressure survival probabilities. Pre-hibernation mortality rates are very high due to various parasitoids (Dolek et al. 2006; Freese et al. 2006). Thus, population fluctuations of E. maturna are very high (Dolek et al. 2006; John et al. 2021). The silk web around the larvae may act as a shelter against predators and parasitoids (Fitzgerald 1993). Early larval stages hibernate and complete its development in the following spring (first on various herbs, shrub and tree species, later on Fraxinus excelsior after the opening of the leaf buds, see Gros 2002; Dolek et al. 2006). Due to intensification in forestry, open-canopy woodlands become very scarce. Typical habitats are early successional stages of deciduous woodland, historically maintained by coppicing and forest pastures. Due to changes in management as well as habitat destruction, this butterfly species is ranked among the most threatened European butterfly species (van Swaay and Warren 1999; van Swaay et al. 2010). Specific and locally adapted conservation management (e.g. coppicing of forest stands, creation of early-successional stages in deciduous and humid forests) support this butterfly, among many other taxa (Gros 2008; Fartmann et al. 2013; Dolek et al. 2018; Vrabec et al. 2019; Bubová et al. 2015).
Data collection
We assessed sites with egg ovipositions of E. maturna based on the sightings of caterpillar nests during June and July 2021. For each of these caterpillar nests, we recorded the following parameters: Exact geographic location (GPS coordinates, with a handheld GPS device, Trimble Juno SB, accuracy ± 3 m); height of Fraxinus excelsior trees (the single tree species on which caterpillar nests could be found, classified into < 5 m, 5–10 m, 10–15 m, > 15 m); height of caterpillar nests (m); cardinal direction; and shading (grouped into the categories completely shaded, partly shaded, predominantly unshaded).
Aerial pictures
The study area was mapped with a UAV equipped with a multispectral camera system. For the purpose of detailed mapping, the following requirements were considered for flight planning. Based on local relief and flight restrictions a flight height of 80 m above ground level (AGL) was used and resulted in a geometric resolution of 7 cm ground sampling distance for the final orthomosaic. The used camera sensor system, a multispectral MicaSense RedEdge-MX Dual, was configured with a front and lateral overlap of 75% to successfully create the orthomosaic. As flight platform we used the quadrocopter DJI Inspire 2 (https://www.dji.com/de/inspire-2). Flight route planning was done using DJI Pilot app on the remote controller with precompiled KML files of the target ground coverage. Sensor system details were saved to DJI Pilot app, flight route planning was automatically calculated and checked with the settings of the MicaSense RedEdge MX Dual web configuration concerning capture mode, front overlap and AGL.
The MicaSense camera system was chosen, because it simultaneously captures 10 spectral bands (with central wavelength in nm and band width in brackets: Coastal blue 444(28), blue 475(32), green 531(14), green 560(27), red 650(16), red 668(14), red edge 705(10), red edge 717(12), red edge 740(18), NIR 842(57)). The arrangement of the spectral bands follows the well-known satellite platforms LANDSAT and Sentinel-2 and thus allows the calculation of several vegetation indices, which can be used by subsequent habitat suitability modelling on coarser scales, e.g. from local to regional or even national scale. To ensure data quality and usability before and after the flight, images of a spectral reference plate were recorded to consider the influence of radiation and changes during the flight when processing the data. Irradiance values were recorded during flight mission through the Downwelling Light Sensor of the sensor system (Mamaghani and Salvaggio 2019).
Based on the captured imagery, a digital surface model and a multispectral orthomosaic were calculated. Georeferencing was done using meta-data of imagery and manual referencing of prominent terrain points, which were equally spread across the study area. Thus, the resulting high resolution multispectral orthomosaic was exported as a GeoTIFF file with 10 bands for further processing in QGIS software. QGIS version 3.4.5 was used to prepare the data for habitat suitability modelling (HSM) and besides the multispectral orthomosaic, the commonly used vegetation index NDVI. A digital surface model (DSM) was exported from Agisoft Metashape Professional as GeoTIFF file and then was used to generate derived variables for the HSMs using QGIS. The variables aspect, topographic position index (TPI), terrain ruggedness index (TRI), and roughness (Wilson et al. 2007) were considered potentially relevant to describe the preferred habitat of the species. The TPI is calculated as the difference between the value of a cell and the mean value of its eight surrounding cells. The TRI is the mean of the absolute differences between the value of a cell and the value of its eight surrounding cells. Roughness is the maximum value of a cell and its eight surrounding cells minus the respective minimum value. Before calculating these variables, the resolution of the DSM was decreased, by building the mean of 20 × 20 pixels (1.4 m × 1.4 m) in each horizontal and vertical direction. This way the variables captured topographical variation introduced by whole trees, shrubs, and similar, instead of single branches or even leaves. The resolution of all variables was subsequently rescaled such that one pixel represented the mean of 5 × 5 pixels of the original resolution, which corresponds to pixel sizes of 35 × 35 cm.
Habitat suitability modelling
To avoid possible effects of inter-correlation, we calculated pairwise Pearson correlation coefficients among all variables. In case of |r|> 0.7 (Dormann et al. 2013), only the biologically putatively more relevant variables were used for modelling. Habitat suitability models were then inferred based on spectral bands 4 (green@560 nm) and 9 (red edge@740 nm), the NDVI, aspect, TPI, and roughness. We calculated ensemble models of habitat suitability with presence only data using the biomod2 package version 3.5.1 in R (Thuiller et al. 2013), using GAM, GBM, GLM, and the built-in Maxent algorithms. Eighty percent of the presence records were randomly chosen to build the models, while the remaining 20% were used for model evaluations by the relative operating curve (ROC). Three sets of 1000 pseudo-absences were generated using the disk-method and dis-allowing pseudo-absence points to be closer than 3 m to presence points. We ran three repeats for evaluation to make up 36 single models (three repeats × four model algorithms × three pseudo-absence sets). Variable importance was estimated in three permutations. biomod2 implements the evaluation strip method (Elith et al. 2005), which permits a direct comparison of predicted responses across all model algorithms used. All model predictions were scaled for better comparability. Single models were considered for the ensemble model if ROC scores were > 0.7. Final ensemble models were calculated as weighted mean of single models, where weightings of single models were determined by the area under the receiver operator curve (ROC).
Results
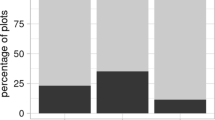
In total we recorded 296 nests of E. maturna, which were distributed to 170 F. excelsior trees throughout the study area (Supplement Table S1). Most of them were exposed to the south, south-west, or south-east, at a mean height of 3 m (ranging from 1–8 m) (Fig. 1). The vast majority of nests was found at partly shaded branches, as opposed to fully shaded and fully exposed places (Fig. 2). Young trees smaller than 10 m in height were preferentially used for oviposition (Fig. 3). The habitat suitability model clearly favoured forest edges with southern, south-western, or south-eastern expositions, as suitable nesting-habitats, whereas it did not matter whether the edge was adjacent to the river or the surrounding meadows (Fig. 4). Likewise, somewhat isolated trees within the forest were determined to be suitable. The model thus well reflected the occurrences of caterpillar nests of E. maturna throughout the area, as also indicated by the evaluation metrics (Supplement Table S2). Topographical variables had the highest influence on the model according to the variable response plots (Fig. 5). Values around zero were favoured for the topographical position index (TPI), in combination with the aspect reflecting the favouring of trees with open space to the south, including forest edges. In the present case, high TPI values either indicate tree-tops that protrude from the surrounding terrain or such positions that are surrounded by higher trees (at a resolution of 1.4 m; see methods section), which were both modelled to be unsuitable. The model excluded flat regions like the river and surrounding meadows by the terrain roughness, which needed to be above five for 50% habitat suitability, meaning a minimum height difference of 5 m was needed within a square of 4.2 × 4.2 m (3 × 3 pixels á 1.4 m edge length). The mandatory tree species was likely incorporated in the model by the NDVI and the two spectral bands (Fig. 5).
Study area and habitat suitability model. a Aerial photograph of the study site as taken by the UAV. Open circles indicate records of nests of E. maturna that were used to model habitat suitability. b Projection of the inferred habitat suitability model to the study area and c to an enlarged section from the centre of the area. Pixels with warmer colours at the southern margin of forest edges and more isolated trees indicate better condition for E. maturna as inferred by the model
Discussion
We found that topographical variables had the highest influence on the model according to the variable response plots. Values around zero were favoured for the topographical position index, in combination with the aspect reflecting the favouring of trees with open space to the south, including forest edges. This is also confirmed by the manually mapped information on oviposition sites of E. maturna, which are mainly restricted to sunny spots of deciduous forest mostly along forest edge habitats, such as close to the River Taugl, in the transition area between forest and adjacent meadows and along forest roads, with heterogeneous and light vegetation structures. Our findings are in line with previous studies showing that nests of larvae of E. maturna are mainly in light forest structures of early successional stage, along forest edges with smooth forest-openland ecotones (Gros 2002). Furthermore, our results underline that the accumulation of eggs and larvae concentrates at spots being protected from the wind, such as forest clearings in the forest interior, or in indentations at the edge of the forest (see also Gros 2002). Due to the intensification in forestry, light, sparse, species-rich and structure-rich mixed deciduous forests become transformed into monocultures of trees. With the combination of very detailed data recorded manually in the field through observations and the larger scale ecosystem information recorded using UAV, it is possible to very precisely project detailed information onto the landscape and to identify areas of high conservation value (see Habel et al. 2016, 2019; Leitao & Santos 2019).
Apart of these ecosystem structures, further microhabitat preconditions are of relevance for successful larval development of E. maturna. Our habitat suitability model clearly favoured forest edges with southern, south-western, or south-eastern expositions as suitable nesting habitats, whereas it did not matter whether the edge was adjacent to the river or the surrounding meadows. Also isolated trees within the forest, e.g. forest glades, were determined to be suitable. Our results using habitat suitability models (based on accurate aerial imagery) are consistent with previous studies that clearly state that egg oviposition of this butterfly species usually occurs at forest edges, or in sparse forest clearings, and is mostly in a southerly orientation (Gros 2002). This might be that eggs and subsequently larvae are exposed to the sun (already during morning), and thus warm up more quickly and thus there are higher rates of successful larval development (Gros 2002). In addition to sky orientation, there was also a clear preference of oviposition on medium height (mean height of 3 m) on Fraxinus excelsior trees (assessed manually in our study), which matches well with earlier observations in another E. maturna population in northern Austria (average 3.2 m, Gros 2002). At this height, there might exist a very specific microclimate, with presumably high levels of humidity (close proximity to the ground), but is in parallel well exposed to solar radiation (and thus high temperatures). In addition to ecosystem structures and microclimatic conditions, further behavioural factors may drive the accumulation of egg oviposition at specific sites. Studies found that females of E. maturna lay more eggs at sites that have already been visited by other females and where egg deposition has already occurred. Such a behaviour and subsequent mass ovipositions may reduce parasitic pressure (Freese et al. 2006; Dolek et al. 2006, 2013).
According to our modelling results, oviposition mostly occurred along forest edges and forest roads. Such forest clearings are comparatively humid and frequently characterized by intense sun-exposure, and are at the same time comparatively windless (Weidemann 1985). Strong environmental dynamics create numerous early successional stages in ecosystems, and thus enable the growth of herbs and diverse flowering plants, shrubs and diverse pioneer tree species, as well as young Fraxinus excelsior trees, which is of high relevance for the successful larval development of E. maturna (Gros 2002). Sunny sites in the forest and a smooth transition of the forest into adjacent openland habitats are essential—not only for E. maturna, but for a large number of animal and plant species (Kozel et al. 2021). For example, numerous species depend on the combination of sunshine and a moist forest climate, or feed on and develop on herbs growing in open disturbance areas in or along the forest. Thus, the detected habitats for E. maturna development generally represent valuable structures for a variety of now rare animal and plant species (Vojta and Drhovská 2012; Hartel et al. 2014). Prescribed forest grazing may be an efficient management strategy to promote forest biodiversity adapted to light, moist heterogeneous deciduous forests.
Conclusion
Our study supports the relevance of sparse deciduous forests that provide valuable moist and sunny habitats through moderate disturbance (Kirby et al. 1995). The identification of such valuable ecosystems and specific ecosystem structures and its projection across landscapes is possible using high-resolution aerial imagery and modelling approaches (Perepenova and Skalos 2019). The high resolution of recent camera systems (e.g., submeter resolution) already makes it possible to determine not only vegetation composition (distinguishing among plant species), but also study accurate vegetation topographies and structural diversity for forests and landscapes (Marvin et al. 2016; Librán-Embid et al. 2020). Nevertheless, the investigation of details on the occurrence, ecology and behaviour of species is essential to provide the basis for the calculation of the models, and to correctly interpret the results derived from habitat suitability models.
References
Bubová T, Vrabec V, Kulma M, Nowicki P (2015) Land management impacts on European butterflies of conservation concern: a review. J Insect Conserv 19:805–821.
Dennis RLH, Eales HT (1997) Patch occupancy in Coenonympha tullia (Muller, 1764) (Lepidoptera: Satyrinae): habitat quality matters as much as patch size and isolation. J Insect Conserv 1:167–176.
Dolek M, Freese-Hager A, Cizek O, Gros P (2006) Mortality of early instars in the highly endangered butterfly Euphydryas maturna (Linnaeus, 1758) (Nymphalidae). Nota Lepidopterol 29:221–224
Dolek M, Freese-Hager A, Geyer A et al (2013) Multiple oviposition and larval feeding strategies in Euphydryas maturna (Linné, 1758) (Nymphalidae) at two disjoint European sites. J Insect Conserv 17:357–366.
Dolek M, Kőrösi Á, Freese-Hager A (2018) Successful maintenance of Lepidoptera by government-funded management of coppiced forests. J Nat Conserv 43:75–84.
Dormann CF, Elith J, Bacher S et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46.
Ebert G, Rennwald E (1991) Die schmetterlinge Baden-Württembergs. Eugen Ulmer, Stuttgart
Eliasson CU (2001a) Studier av boknätfjärilen (Euphydryas maturna) (Lepidoptera: Nymphalidae) i Västmanland 2 - fenologi, protandri, könskvot och parningslek. Entomologisk Tidskrift 122:153–167
Eliasson CU (2001b) Studier av boknätfjärilen Euphydryas maturna (Lepidoptera: Nymphalidae) förekomst och biologi i Västmanland. Entomologisk Tidskrift 112:113–124
Eliasson CU, Shaw MR (2003) Prolonged life cycles, oviposition sites, foodplants and Cotesia parasitoids of Melitaeini butterflies in Sweden. Oedippus 21:1–52
Elith J, Ferrier S, Huettmann F, Leathwick J (2005) The evaluation strip: a new and robust method for plotting predicted responses from species distribution models. Ecol Model 186:280–289.
Fartmann T, Müller C, Poniatowski D (2013) Effects of coppicing on butterfly communities of woodlands. Biol Cons 159:396–404.
Fitzgerald TD (1993) Social caterpillars. In: Stamp NE, Casey TM (eds) Caterpillars: ecological and evolutionary constraints on foraging. Chapman and Hall, New York, pp 372–403
Freese A, Benes J, Bolz R et al (2006) Habitat use of the endangered butterfly Euphydryas maturna and forestry in Central Europe. Anim Conserv 9:388–397.
Gámez-Virués S, Perović DJ, Gossner MM et al (2015) Landscape simplification filters species traits and drives biotic homogenization. Nat Commun 6:8568.
Gros P (2002) Habitatmanagement FFH-relevanter Tagfalterarten: Grundlagestudie für die Entwicklung eines Artenschutzprogrammes zur Förderung von Metapopulationssystemen des Eschen-Scheckenfalters (Euphydryas maturna Linnaeus, 1758) (Lepidoptera: Nymphalidae). University of Salzburg, Salzburg
Gros P (2008) Butterfly species of the Habitat Directive (EEC 1992/43, Annex II/IV) in the Natura 2000 Conservation Area “Untersberg-Vorland”: monitoring results for 2008. – Final report. Nature Conservation Department of the Province of Salzburg, pp 1–22
Habel JC, Segerer A, Ulrich W, Torchyk O, Weisser WW, Schmitt T (2016) Butterfly community shifts over two centuries. Conserv Biol 30:754–762
Habel JC, Trusch R, Schmitt T, Ochse M, Ulrich W (2019) Long-term large-scale decline in relative abundances of butterfly and burnet moth species across south-western Germany. Sci Rep 9:14921
Habel JC, Teucher M, Gros P et al (2021) Land use and climate change affects butterfly diversity across northern Austria. Landscape Ecol 36:1741–1754.
Hartel T, Hanspach J, Abson DJ et al (2014) Bird communities in traditional wood-pastures with changing management in Eastern Europe. Basic Appl Ecol 15:385–395.
Horák J, Pavlíček J, Kout J, Halda JP (2018) Winners and losers in the wilderness: response of biodiversity to the abandonment of ancient forest pastures. Biodivers Conserv 27:3019–3029.
John V, Pavlicko A, Vrabec V, Rybova V, Andres M, Konvicka M (2021) Cyclic abundance fluctuations in a completely isolated population of Euphydrias maturna. Nota Lpidopterologica 44:213–222
Kirby KJ, Watkins C (eds) (2015) Europe’s changing woods and forests: from wildwood to cultural landscapes. CAB International, Boston, MA
Kirby KJ, Thomas RC, Key RS et al (1995) Pasture-woodland and its conservation in Britain. Biol J Lin Soc 56:135–153.
Kozák D, Svitok M, Wiezik M et al (2021) Historical disturbances determine current taxonomic, functional and phylogenetic diversity of saproxylic beetle communities in temperate primary forests. Ecosystems 24:37–55.
Kozel P, Sebek P, Platek M et al (2021) Connectivity and succession of open structures as a key to sustaining light-demanding biodiversity in deciduous forests. J Appl Ecol 58:2951–2961.
Kudrna O (2011) Distribution atlas of butterflies in Europe. Gesellschaft für Schmetterlingsschutz e.V, Halle
Kuuluvainen T (2009) Forest management and biodiversity conservation based on natural ecosystem dynamics in Northern Europe: the complexity challenge. Abio 38:309–315
Leitão PJ, Santos MJ (2019) Improving models of species ecological niches: a remote sensing overview. Front Ecol Evol 7:9.
Librán-Embid F, Klaus F, Tscharntke T, Grass I (2020) Unmanned aerial vehicles for biodiversity-friendly agricultural landscapes - a systematic review. Sci Total Environ 732:139204.
Mamaghani S (2019) Multispectral sensor calibration and characterization for sUAS remote sensing. Sensors 19:4453.
Marvin DC, Koh LP, Lynam AJ et al (2016) Integrating technologies for scalable ecology and conservation. Global Ecol Conserv 7:262–275.
Maxwell SL, Fuller RA, Brooks TM, Watson JEM (2016) Biodiversity: the ravages of guns, nets and bulldozers. Nature 536:143–145.
Naiman RJ, Decamps H, Pollock M (1993) The role of riparian corridors in maintaining regional biodiversity. Ecol Appl 3:209–212.
Naumov V, Manton M, Elbakidze M et al (2018) How to reconcile wood production and biodiversity conservation? The Pan-European boreal forest history gradient as an “experiment.” J Environ Manage 218:1–13.
Pereponova A, Skaloš J (2019) Spatio-temporal dynamics of wood-pastures in lowland and highland landscapes across Czechia. Reg Environ Change 19:267–278.
Seibold S, Brandl R, Buse J et al (2015) Association of extinction risk of saproxylic beetles with ecological degradation of forests in Europe: beetle extinction and forest degradation. Conserv Biol 29:382–390.
Skrajna T (2020) Impact of agriculture intensification on the floristic diversity of the forest-field ecotone. Pol J Ecol 68:47.
Steffan-Dewenter I, Kessler M, Barkmann J et al (2007) Tradeoffs between income, biodiversity, and ecosystem functioning during tropical rainforest conversion and agroforestry intensification. Proc Natl Acad Sci 104:4973–4978.
Straka U (2014) Zur Ökologie des Eschenscheckenfalters (Euphydryas maturna) im niederösterreichischen Weinviertel. Beiträge Zur Entomofaunistik 14:107–137
Thomas JA (2016) Butterfly communities under threat. Science 353:216–218.
Thomas JA, Simcox DJ, Hovestadt T (2011) Evidence based conservation of butterflies. J Insect Conserv 15:241–258.
Thuiller W, Münkemüller T, Lavergne S, Mouillot D, Mouguet N, Schiffers K, Gravel D (2013) A road map for integrating eco-evolutionary processes into biodiversity models. Ecol Lett 16:94–105
Tockner K, Stanford JA (2002) Riverine flood plains: present state and future trends. Envir Conserv 29:308–330.
van Swaay C, Cuttelod A, Collins S et al (eds) (2010) European red list of butterflies. Publications Office of the European Union, Luxembourg
Van Swaay C, Warren M (1999) Red Data Book of European Butterflies (Rhopalocera). Nature and Environment. Council of Europe Publishing, Strasbourg
Vojta J, Drhovská L (2012) Are abandoned wooded pastures suitable refugia for forest species? J Veg Sci 23:880–891.
Vrabec V, Bubová T, Kulma M et al (2019) How Euphydryas maturna survived extinction in the Czech Republic: status of a relic population after intensive conservation management. J Insect Conserv 23:393–403.
Wahlberg N (1998) The life history and ecology of Euphydryas maturna (Nymphalidae: Melitaeini) in Finland. Nota Lepidopterologica 21:154–169
Wahlberg N (2000) Comparative descriptions of the immature stages and ecology of five Finnish melitaeine butterfly species (Lepidoptera: Nymphalidae). Entomol Fennica 11:167–174
Wahlberg N (2001) On the status of the scarce fritillary Euphydryas maturna (Lepidoptera: Nymphalidae) in Finland. Entomol Fennica. https://doi.org/10.33338/ef.84127
Ward JV, Tockner K, Schiemer F (1999) Biodiversity of floodplain river ecosystems: ecotones and connectivity1. Regul Rivers: Res Mgmt 15:125–139.
Weidemann HJ (1985) Zum Einfluss veränderter Bewirtschaftungsweisen auf bestandsbedrohte Tagfalterarten: maivogel (Euphydryas maturna) und „Storchschnabel-Bläuling“ (Eumedonia eumedon) in Franken. Bericht Naturforschende Gesellschaft Bamberg 1985:99–136
Wilson MFJ, O’Connell B, Brown C et al (2007) Multiscale terrain analysis of multibeam bathymetry data for habitat mapping on the continental slope. Mar Geodesy 30:3–35.
Acknowledgements
We hereby confirm, that: The work is all original research carried out by the authors. All authors agree with the contents of the manuscript and its submission to the journal. No part of the research has been published in any form elsewhere, unless it is fully acknowledged in the manuscript. The research featured in this manuscript does not relate to any other manuscript. The manuscript is not being considered for publication elsewhere while it is being considered for publication in this journal. Any research in the paper not carried out by the authors is fully acknowledged in the manuscript. All sources of funding are acknowledged in the manuscript, and authors have declared any direct financial benefits that could result from publication. All appropriate ethics and other approvals were obtained for the research.
Funding
Open access funding provided by Paris Lodron University of Salzburg. The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Habel, J.C., Teucher, M., Gros, P. et al. The importance of dynamic open-canopy woodlands for the conservation of a specialist butterfly species. Landsc Ecol 37, 2121–2129 (2022). https://doi.org/10.1007/s10980-022-01472-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-022-01472-2