Abstract
Context
Identifying how animals select habitat while navigating landscapes is important for understanding behavioral ecology and guiding management and conservation decisions. However, habitat selection may be spatially and temporally plastic, making it challenging to quantify how species use resources across space and time.
Objectives
We investigated how landscape context and dispersal shape habitat selection at multiple spatial scales in white-tailed deer (Odocoileus virginianus).
Methods
Using step-selection functions, we quantified habitat selection of landcover and topographic covariates at three spatial scales for juvenile males during three movement periods (before, during, after dispersal) in two regions of Missouri, USA—a fragmented, low forest cover region with rolling hills, and a forested, topographically variable region.
Results
Although selection for forest cover increased after dispersal in both regions, deer selected forest cover at smaller spatial scales in the fragmented, low forest cover region. This result indicates scale of selection was dependent on forest availability and configuration with deer likely perceiving landscapes differently across their distribution. Functional responses to topography differed in magnitude and direction between regions with deer avoiding roads and selecting valleys in the rolling hills region (especially during dispersal) while showing no response to roads and selecting for ridgelines (during dispersal) in the topographically variable region. This result suggests movement behavior is strongly dependent on topography.
Conclusions
Although deer may select similar habitats among regions, landscape context and movement period shape the scale, strength, and direction of selection. This result has important implications for how animals use landscapes across different regional contexts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat selection is a key behavioral process that shapes the movement of species within and across landscapes (Millspaugh and Marzluff 2001; Manly et al. 2002). Understanding this process is critical for guiding landscape-level management and conservation practices, particularly in the context of habitat preservation, gene flow, and disease transmission (Slarkin 1985; Cullingham et al. 2011; Walter et al. 2011; Holbrook et al. 2017). However, habitat selection and movement dynamics are often spatially and temporally plastic and depend upon the broader landscape context, particularly the availability and configuration of habitat (Levin 1992; Morales and Ellner 2002; Schick et al. 2008). For instance, animals often demonstrate a functional response of increasing selection as available habitat decreases, a pattern that is driven by broader regional patterns of landcover mosaics (Godvik et al. 2009; Roever et al. 2012). Additionally, the spatial configuration of landscape features can influence habitat selection depending on composition and level of fragmentation (Stubblefield et al. 2006; Radford and Bennett 2007). These patterns can further vary temporally as both resources and abiotic conditions change throughout the year (Holbrook et al. 2017). Collectively, these spatial and temporal factors underscore the need to assess habitat selection in a framework that is likewise spatially and temporally dynamic.
Habitat composition and configuration also can influence how animals perceive landscapes, which in turn can change the spatial scale at which individuals respond to a given habitat characteristic (Laforge et al. 2016). An individual’s perceptual range within a landscape partly defines the scale at which it responds to the environment, and such perceptions vary according to habitat type and topography (Olden et al. 2004). For example, prey species may perceive and respond to the landscape at smaller spatial scales in habitats with denser vegetation compared to more open areas where predator vigilance requires them to perceive the landscape at larger spatial scales (Jayakody et al. 2008; Laforge et al. 2016). Thus, when evaluating habitat selection it is important to consider the scale of effect for a given habitat covariate (Laforge et al. 2016; Heit et al. 2023).
The type of movement an animal exhibits may further modulate habitat selection and the scale of that selection (Killeen et al. 2014; McGarigal et al. 2016). Two of the most common movement types in motile animals are home range (“station-keeping”) and dispersal movements (Burt 1943; Schlägel et al. 2020). These movement types have distinct characteristics, with dispersal movements typically being faster and straighter than those in home ranges (Soulsbury et al. 2011; Moll et al. 2021). During dispersal, terrestrial animals often select habitat features that provide greater cover while allowing for ease of movement (Long et al. 2005; Cox and Kesler 2012). In more topographically complex areas, valleys are often used during dispersal since they are relatively easy to transverse (Puskas et al. 2010). Movement type may also change an individual’s perception of the landscape and in turn the scale at which it selects habitat (Lima and Zollner 1996; Nathan et al. 2008). For example, spatial memory within home ranges may allow individuals to select habitat at finer scales compared to dispersing or migrating individuals that may use landscape cues to select habitat at larger scales (Fagan et al. 2013).
We quantified how landscape context and dispersal shape seasonal habitat selection, and the spatial scale of that selection in a widespread and highly mobile ungulate, the white-tailed deer (Odocoileus virginianus; hereafter deer). Although both sexes and all demographic classes of deer may disperse (e.g., Nixon et al. 2007; Anderson et al. 2015; Lutz et al. 2015; Moll et al. 2021), juvenile males (~ 10–20 months of age) make up the greatest proportion of dispersing individuals (Long et al. 2008). Deer dispersals tend to be relatively brief and directional, but vary both seasonally and by landscape context (Long et al. 2010). For example, most juveniles disperse in the spring or fall, with spring dispersal driven by inbreeding avoidance (facilitated by adult female aggression) and fall dispersal by mate competition with adult males (Long et al. 2008). These seasonal dispersals often differ in length with longer dispersals in the spring relative to fall (Long et al. 2008). All dispersals differ from within-home range movements, which are typically slower, less directional, and centered on one or more core areas where food and cover is plentiful (Moll et al. 2021). Forest cover is one of the most important habitat variables affecting both within-home range (Heit et al. 2023) and dispersal movements in deer, with higher rates of dispersal and distance traveled in areas with less forest cover (Nixon et al. 2007; Diefenbach et al. 2008; Lutz et al. 2015). Additionally, during dispersal, topographic features such as rivers and roads act as semipermeable barriers that influence dispersal movements (Long et al. 2010; Moll et al. 2021). Although dispersal has been well studied in deer, it is unclear how habitat selection during dispersal compares to that of within-home range movements (but see Gilbertson et al. 2022; Hooven et al. 2023) and how spatial scale and landscape heterogeneity interact to modulate habitat selection.
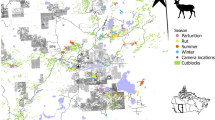
We analyzed habitat selection and movement attributes of male juvenile deer in two study areas in Missouri, USA during three different periods—before, during, and after dispersal (Fig. 1a, b). We took advantage of natural variation in both landcover and topography between the two study areas to examine how landscape context shapes habitat selection. One study area was in an agricultural region with rolling hills and low forest cover, and the other in a forested region with considerable topographic variation (Fig. 1). We used step-selection functions to quantify habitat selection using high-resolution data from three landscape cover variables (forest, forest edge, cropland; Fig. 1c–e) and three topographic variables (topographic position index, distance to water, and distance to roads; Fig. 1f–h) at three spatial scales. We hypothesized that the strength and scale of habitat selection would be dependent on movement period and would vary between seasons and by study area (Table 1). Specifically, we predicted that during a dispersal, deer would select resources at larger spatial scales since they would have less spatial memory, or familiarity with their surroundings (Fagan et al. 2013). Additionally, we predicted that during dispersal, deer would show stronger selection for landcover variables that provide cover (e.g., forest, forest edge) and topographical features that help facilitate and direct movement (e.g., valleys and water ways) than either before or after dispersal and that areas with greater mortality risk (i.e., roads) would be avoided (Table 1). We also predicted that agriculture would be avoided in the spring, when it provides little cover, compared to the fall when agricultural crops provide both cover and food (Gilbertson et al. 2022). However, during late spring, agriculture can provide substantial forage in this study area, so an alternative prediction was that selection for agriculture would be positive across seasons. Lastly, we expected faster and more direct movements during dispersal compared to home range movements (Killeen et al. 2014; Moll et al. 2021).
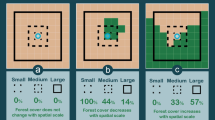
Locations a of the two study areas (North and South) in Missouri, USA, b example of a white-tailed deer (N17014) with locations before, during, and after a spring dispersal, and c–e landcover and f–h topographic characteristics for each study area shown as mean values (± 95% confidence intervals) and square satellite view insets (10 × 10 km near center of study area). Values for means were extracted from 1000 random locations (1 km2 area) within each study area and compared with t-tests. Within insets, habitat characteristics are shown in white except for topographic relief which is elevation (low = black, high = white). Forest, forest edge, cropland, topographic relief, and stream density differed between study areas (asterisks denotes α < 0.05), whereas road density was similar (P = 0.135). The amount of grassland was similar (t = 0.01, P = 0.99) between the North (25.8%) and South (25.7%) but was not included in habitat selection models due to multicollinearity with other variables (see Habitat selection analysis in Methods). Deer silhouette by Gabriela Palomo-Munoz (https://creativecommons.org/licenses/by-nc/3.0/)
Methods
Study system
Our study took place in the northwestern and south-central portions of Missouri (hereafter North and South, respectively), which are over 300 km apart and differ considerably in both landcover and topographical features (Fig. 1a, c–h). Landcover in the North consisted of highly fragmented forest patches (19% of landscape) in a mosaic of grasslands (26%) and cultivated croplands (51%) (Fig. 1c–e). Comparatively, landcover in the South consisted of more contiguous forest (72%) with interspersed grassland patches (26%) and almost no cropland (< 1%) (Fig. 1c–e). The fragmented nature of forest patches in the North resulted in a higher forest edge density (Fig. 1d). Forests in both study areas were primarily composed of oak (Quercus spp.) and hickory (Carya spp.) with the South also having smaller amounts shortleaf pine (Pinus echinata) (Wright et al. 2019). Grasslands were used for cattle grazing or hay production in both study areas and corn and soybeans were the primarily row crops in the North (Wright et al. 2019).
Topography in the North was characterized by rolling hills whereas the South (in the Ozark Highlands region) had steep hills and valleys with nearly twice the local relief (Fig. 1f). Streams and waterways were abundant in both regions with the North having slightly higher stream density than the South (Fig. 1g). Although road density was similar between study areas, flatter areas in the North accommodated a gridded road system whereas the hillier areas in the South had a more variable road layout that generally followed the terrain (Fig. 1h).
Both study areas had deer hunting seasons during fall with an archery season extending from mid-September through mid-January and a primary firearms season in mid-November. Additionally, there was an antlerless-only firearm season in early December, youth firearm seasons in late October and late November, and an alternative methods season in late December/early January. Hunting pressure is high with over 500,000 permit holders in the state and around 300,000 deer harvested annually (Keller et al. 2017).
Deer collaring and dispersals
Deer were captured using modified Clover traps or rocket nets between January and March of 2015–2019 and were chemically immobilized for collaring (see Wright et al. 2019 for details). Each individual was assigned a unique deer ID number and fitted with an Iridium global positioning system (GPS) radio‐collar (model G2110E, 825 g; Advanced Telemetry Systems, Isanti, Minnesota, USA) that recorded locations every 5 h. During handling, deer were aged based on tooth eruption and wear patterns as follows: fawn (6 months), yearling (1.5 yr), and adult (> 1.5 yr) (Severinghaus 1949). All trapping and immobilization protocols were approved by the University of Missouri Institutional Animal Care and Use Committee (protocol number 8216).
To avoid differences in habitat selection and movement that may vary among sexes and demographic groups, our analyses included only juvenile males (i.e., fawns at capture and those dispersing between ~ 10–20 months of age) which comprise the majority of dispersing deer within a population (Gilbertson et al. 2022). We defined a dispersal as a permanent emigration from one home range to a new range, such that ranges do not overlap (Diefenbach et al. 2008; Haus et al. 2019). We visually screened deer locations to identify individuals with multiple location clusters, signifying a potential dispersal event using QGIS (v. 3.28.1; QGIS Development Team 2022). We defined a ‘dispersal window’ as the time period between when individuals exited a cluster of locations and established to another cluster.
Dispersal events in deer generally take place in either spring or fall, with proximate causes of dispersal, along with habitat quality, differing between seasons (Long et al. 2008). Applying season designations similar to Gilbertson et al. (2022), we classified dispersal events as occurring during spring (April 1–July 31) or fall (September 1–December 31). Within a season, we modeled home ranges before and after dispersal using kernel density estimation (95% adaptive kernel, reference bandwidth) in R (R Core Team 2021) with default setting in the amt package (Signer et al. 2019). For home range estimates we excluded points that fell within a two-day buffer on either side of the dispersal window to avoid atypical movements that could occur either before a dispersal or when initiating a new home range following a dispersal. We used points that fell within the home range polygon for modeling habitat selection before and after a dispersal. We only retained deer that had at least 50 points within a home range (Seaman et al. 1999). Using a one-day buffer on either side of the dispersal window, we identified locations associated with dispersal as points that fell between the last point within the pre-dispersal home range and the first point within the post-dispersal home range. This allowed us to exclude points associated with exploratory movements which occasionally occurred before a dispersal. Some dispersals took place in a short timeframe; we only modeled habitat selection during the dispersal period for deer that had at least nine locations (i.e., dispersing for at least 45 h), which we considered the minimum acceptable sample size for step-selection analysis (see below). For seven individuals, dispersals took place over a longer period where they moved repeatedly back and forth between the two home ranges. For these deer we did not analyze any locations associated with the dispersal but did analyze their before and after dispersal locations to compare habitat selection in pre- and post-dispersal home ranges.
Habitat selection covariates
We collected landscape covariates that we hypothesized could shape habitat selection or movement of deer (Table 1) including four variables related to landscape cover (forest, forest edge, cropland, and grassland; Fig. 1c–e) and three related to topography and linear features (topographic position index, distance to water, and distance to roads; Fig. 1f–h). We characterized landscape cover using Dynamic World land use land cover (LULC) classifications, which are based on machine learning of 10 m Sentinal-2 imagery (Brown et al. 2022). We used Google Earth Engine (Gorelick et al. 2017) to generate a composite Dynamic World LULC at a 10 m pixel resolution based on imagery between June 1, 2015 (earliest imagery available) through Dec. 31, 2019 (end of study period), taking dominant LULC for each pixel. From the Dynamic World LULC we used ‘trees’ to represent forest, ‘crops’ to represent cropland, and ‘grass’ to represent grassland. We calculated forest edge density (m/ha) using ‘forest’ relative to other landcover types (with ‘built’ LUCL classification removed) with the function ‘lsm_l_ed’ in the landscapemetrics R package (version 1.5.6; Hesselbarth et al. 2019). To quantify topographic position, we used a Digital Elevation Model (DEM; 10 m pixel resolution) from the United States Geological Survey National Elevation Dataset (USGS, 2022a) to create a topographic position index (TPI) in QGIS. Pixel values were based on a neighborhood radius of 5 cells with negative values indicating valleys, values near zero indicating flats or continuous slopes, and positive values indicating ridges or hills. To represent water, we used the high-resolution shapefiles of streams (flowlines), rivers, and waterbodies in the National Hydrography Dataset (scale of 1:24,000, USGS 2022b) to build a raster (10 m pixel resolution) with cells representing distance to nearest water feature. For roads, we used both paved and unpaved roads from the U.S. Census TIGER county-level road shapefiles (U.S. Census Bureau 2019) to generate a distance to nearest road raster (10 m pixel resolution).
Habitat selection analysis
We determined habitat selection using a step-selection function analysis which compares observed steps (pairs of consecutive GPS points) to available steps. We generated available steps associated with an observed GPS location as the start point of the step and a random end point based on changes in distance moved (step length) and bearing (turning angle) (Thurfjell et al. 2014). For each observed step of an individual deer during each movement period (before, during, or after dispersal), we generated nine available steps by fitting a gamma distribution to the observed step lengths and the von Mises distribution to the turn angles using the ‘amt’ R package (Thurfjell et al. 2014; Signer et al. 2019). Because deer respond to landscape features at different spatial scales (Heit et al. 2023), we used observed and available locations (end point in step) to extract each landscape cover covariate (as a proportion; forest edge as a density) and topographic covariate (as mean cell values) at three spatial scales (30 m, 90 m, and 270 m radii) using the raster R package (version 3.5.21; Hijmans 2022).
We evaluated habitat selection among movement periods by fitting step-selection functions as generalized linear mixed models (GLMMs) using the ‘glmmTMB’ function in the glmmTMB R package (version 1.1.5; Brooks et al. 2017). Following Muff et al. (2020), we included step-specific fixed intercepts along with deer‐specific random slopes for each movement (step length and turning angle) and habitat covariate to account for individual variation in habitat selection. Before fitting full models, we first determined the appropriate spatial scale for each habitat covariate within a study area and movement period by fitting models that contained movement covariates and a single habitat covariate at each of the three spatial scales and selected the scale with the lowest Akaike Information Criterion (AIC) score (the scale of effect). When models with different scales had delta AIC values < 2, we used the smaller spatial scale which often has the greatest functional response (Laforge et al. 2016). We then checked for collinearity (r > 0.6) among habitat covariates. In the South, forest and grassland were highly correlated (r = 0.95), and in the North, forest, grassland, and cropland were not highly correlated (r < 0.55) but had high variance inflation factors (≥ 3). Consequently, we removed grassland from models in both study areas which reduced all correlations and variance inflation factors to acceptable levels (Zuur et al. 2010; Dormann et al. 2013). Due to cropland being absent in the South, we separately fit final GLMMs for both study areas. Models included each three-way interaction between season (spring, fall), movement period (before, during, and after dispersal), and a habitat covariate or movement parameter. We centered and scaled all continuous covariates prior to analysis. In all models, we included natural log of step length and cosine of turning angle to reduce bias in the parameter estimates (Forester et al. 2009). Within a season and habitat covariate, we also tested for differences among movement periods with post hoc comparisons using estimated marginal means and Bonferroni corrections in the emmeans R package (version 1.7.5, Lenth 2018). Additionally, to more fully assess effect sizes of significant variables in the GLMMs, we plotted relative selection strength following Avgar et al. (2017) and used a smoothed function (a generalized additive model with k = 3) fit to model-predicted selection at available points to visualize changes in selection across the value range for each covariate.
Movement analysis
To more fully understand how movement parameters changed before, during, and after a dispersal, we further assessed changes in turning angle and speed. We used the amt R package to extract turning angle, distance, and time for each step. We compared turning angles among movement periods using analysis of variance for circular data by applying the function ‘aov.circular’ in the circular R package (version 0.4.95; Agostinelli and Lund 2022). We calculated speed (m/h) by dividing step length by elapsed time and compared it among movement periods within a study area and season using linear mixed effects models in the “lme4” R package (version 1.1.30; Bates et al. 2015) followed by post hoc comparisons with estimated marginal means and Bonferroni corrections using the emmeans R package. We included deer ID and year as random effects for this analysis.
All statistical analyses were performed in R version 4.0.4 (R Core Team 2021). Variance around means is presented as ± 1 SE unless otherwise noted. We considered variables significant at α < 0.05. Additionally, given relatively small sample sizes for dispersal locations (see Results), and less power to detect differences, we also highlight trends (0.05 < α < 0.1) for dispersals.
Results
Across the five-year study period, 104 out of 622 collared deer dispersed, including 7.9% of juvenile females (6 of 76), 0.6% of adult females (1 of 178), 41.7% of juvenile males (80 of 192), and 6.6% of adult males (17 of 259) (note that some individuals lived multiple years and therefore the sum of demographic bins exceeds the number of total deer). For juvenile males, three exhibited both distinct spring and fall dispersal events and these individuals were subsequently used for both spring and fall dispersal analyses. We acknowledge these as exceptions to the traditional definition of dispersals, but these movements were clearly not migratory in nature and thus we considered them unique, dispersal-like movements. For juvenile males, observations were distributed (number of deer [GPS locations] for the spring and fall, respectively) in the North before (22 [6,550]; 22 [4,464]), during (8 [221]; 11 [219]), and after (22 [6,454]; 23 [11,247]) a dispersal and in the South before (6 [1,662]; 29 [5,816]), during (4 [88]; 9 [380]), and after (7 [1,842]; 28 [10,856]) a dispersal (Table 2). For juvenile males, dispersal metrics were similar on average between study areas with distances ranging from 1.1 to 52.1 km and taking between < 5 h to 15 days to complete (Table 2). On average, spring dispersals took place in late May and early June and fall dispersals took place in mid-October (Table 2).
Habitat selection
Sample size of deer (4 to 29) and GPS locations (88 to over 10,000) varied among seasons, movement periods, and study areas (Table 2).
The scale of effect for habitat covariates (as determined by univariate models) differed both among movement periods and among study areas (Fig. 2). In particular, deer in the South tended to select habitat covariates at larger spatial scales than deer in the North, especially for forest, forest edge, and topographic position index (Fig. 2).
Best scale (radius) for habitat covariates in each study area as determined by AIC. Filled shapes indicate significant (α < 0.05) habitat selection within a habitat covariate and movement period in at least one season within GLMMs (see Fig. 3). Note that during a dispersal there were several scales within 2 ΔAIC for some habitat covariates; although these are indicated in the figure, the smallest scale was used in models
Selection patterns for landscape cover variables were similar between study areas, whereas selection patterns for topographic features varied between the two study areas (Fig. 3, Fig. 4; full model results including all parameter estimates and P-values are in Supporting Information, Table S1). We note that relatively small sample sizes during the spring season in the South may have limited our ability to detect differences (Table 2). In both study areas, and across seasons and movement periods, deer selected forest and forest edge, and tended to increase selection for forest after a dispersal, whereas selection for edge decreased in the spring and stayed consistent (North) or increased (South) in the fall. In the North, cropland generally had little effect on habitat selection regardless of season or movement period. Despite less topographic relief in the North, deer selected for valleys (negative values) across movement periods and showed particularly strong selection for valleys during dispersal (Fig. 4). Comparatively, in the South, deer selection for valleys before a dispersal but selected for ridges (positive values) during dispersal, particularly in the fall. In the North, deer selected for areas farther away from water before a dispersal and areas closer to water after a dispersal in the spring, and in the South, deer selected areas closer to water before a dispersal in the fall. Additionally, during fall dispersals, deer in both study areas showed a trend of selection for areas closer to water. In the North, deer selected areas farther from roads both during and after dispersal events, whereas deer in the South showed little selection toward roads except before a dispersal in the spring.
Results of GLMMs for the North and South study areas (Missouri, USA) indicating movement responses (step length and turning angle) and habitat selection (all other covariates) during dispersal movement periods and between seasons. Shapes are coefficient estimates and lines are 95% confidence intervals. Filled shapes indicate significant (α < 0.05) movement responses and habitat selection within a movement period. Additionally, trends (0.05 < α < 0.10) are indicated with a light blue for dispersals. Within a study area, season, and covariate, movement periods connected by vertical brackets are significantly different as indicated by post hoc tests with Bonferroni correction (full model results are in Supporting Information, Table S1)
Movement
Turning angles differed among movement periods in both study areas and seasons with movements during dispersals having turning angles centered on 0 (indicating straight-line movements) whereas before and after dispersal movements tended to be more evenly distributed with slight peaks closer to 180°, indicating back-tracking movements (Fig. 1b, Fig. 5a). Speed also varied by movement period, but patterns differed between spring and fall (Fig. 5b). In the spring, deer moved over twice as fast during a dispersal compared to either before or after the dispersal. Although deer moved faster during a dispersal in the fall, movements after a dispersal were significantly faster than before a dispersal.
Differences in a distribution of turning angles and b mean movement speed (± 95% confidence intervals) among movement periods (before, during, and after a dispersal event) and between study areas and seasons. Inset P-values from analysis of variance for circular data indicate significant differences in mean direction of movements among movement periods. For movement speed, within a study area and season, movement periods with the same letters are not significantly different, whereas movement periods with different letters are significantly different as indicated by linear mixed effects models and post-hoc tests with P-values adjusted with Bonferroni correction (full model results are in Supporting Information, Table S2)
Discussion
We observed similar habitat selection patterns for landcover variables (related to forest cover and configuration) between study areas, but the spatial scale at which forest was selected depended on availability and landscape configuration. Comparatively, selection for topographical features (topographic position index, waterways, roads), and the strength of that selection, differed by study area and movement period. Overall, season had a relatively small influence on habitat selection but did influence speed of movements. Together, these results suggest that although deer selected similar habitat covariates between our two study areas, the scale, strength, and direction of selection was shaped by both landscape context and movement period. This finding has important implications for how deer use the landscape in different regions which can ultimately influence population-level processes such as disease transmission and geneflow patterns.
Habitat selection and scale
Deer consistently selected for forest across movement periods and seasons. Forests provide food (browse and mast) and cover during movement and rest periods (Beier and McCullough 1990; Stewart et al. 2011; Gilbertson et al. 2022). Although we did not detect stronger selection for forest during dispersal compared to home range movements, deer tended to select more forest cover after a dispersal compared to before. This result suggests that deer are actively seeking forested post-natal home ranges that provide forage and cover. Although the effect size and direction of selection for forest appeared to be similar between study areas, we found that the spatial scale of this selection differed (Figs. 2, 3, 4). In the North, where forest was relatively scarce, we observed selection at smaller spatial scales compared to the South where forest was more abundant. This difference in scale of effect may relate to both availability and configuration, where the North had less forest and was fragmented, often composed of narrow tracts (Fig. 1c, d) causing deer to select forest at smaller spatial scales as they track forest cover. We also found that the scale at which deer selected forest was smaller than the scale at which they selected forest edge. This difference in apparent perception may be due to more predator vigilance at forest edges (Jayakody et al. 2008), which often have higher densities of mesopredators such as coyotes (Kays et al. 2008). Overall, the spatial scale of effect for landcover variables was generally small (i.e., 30 m radius). This scale is considerably smaller than many studies use for deer and was only possible given the high-resolution, 10-m landcover data available with Dynamic World LULC (Brown et al. 2022). Future studies of habitat selection in deer, and other large mammals, may benefit from analyzing a variety of scales, including smaller spatial scales than have traditionally been considered.
Topographic features such as rivers and valleys can help direct movement in dispersing animals and can bound the home range of some individuals during-day to-day movements (Long et al. 2010; Clements et al. 2011; Gilbertson et al. 2022). We found that deer in both study areas selected areas closer to streams and rivers but only consistently during fall dispersal. Waterways may provide a relatively easy pathway to follow and the thicker vegetation, often found along riparian areas, may provide cover during the fall hunting season when deer seek additional concealment (Lone et al. 2015). Compared to other topographic and linear features, topographic position index had the strongest effect on habitat selection during dispersal movements, although patterns differed between the two study areas (Figs. 3 and 4). In the rolling hills of the North, deer selected valleys, whereas in the more topographically variable South, deer selected ridgelines. These differences may be attributed to landscape context and how deer perceive their environment. In the North, it is unlikely that the rolling hills impede movement compared to other regions where more rugged terrain with steep slopes necessitate ungulates to use valleys for movement corridors (Dussault et al. 2007; Killeen et al. 2014). However, given limited forest cover in the North study area, deer may use other features for cover such as valleys (or depressions in the landscape) that help reduce visibility as they move through open areas. Indeed, visual assessments of dispersal points in the North revealed that deer often selected low areas while moving through agricultural fields and grasslands. In addition to potential foraging opportunities, this may also explain why we did not see strong selection against agriculture in the spring. Valleys were also important during home range movements, but the magnitude of selection was considerably less than during the dispersal period.
Interestingly, although deer in the South selected valleys during home range movements, they selected ridgelines during dispersal, contrary to our expectations. The high percent of forest cover in the South likely had sufficient cover for concealment during dispersal events and home range activities. Therefore using ridgelines for dispersal may help reduce energy expenditure (compared to using steeper slopes; e.g., Killeen et al. 2014), while providing a large viewshed of the surrounding area to select the movement path (Olden et al. 2004). In agreement with this interpretation, we also found that during dispersal, deer in the South selected topographic position index at a larger scale than in the north (Fig. 2). This result suggests that deer are perceiving the landscape at larger extents, which may be facilitated by more expansive viewsheds along ridgelines. In both study areas the confidence intervals around selection for topographic position index during dispersal were large, which is likely a consequence of limited sample size coupled with individualistic habitat selection (Hooven et al. 2023).
We predicted that deer would avoid roads during both home range and dispersal movements. However, we found differential responses to roads between the study areas with deer in the agricultural North avoiding roads (particularly during and after dispersal) and deer in the more forested South showing little selection toward roads. Peterson et al. (2017) found similar results in Wisconsin where deer in an agricultural region were less likely to cross roads compared to deer in a forested region. Although deer are generally thought to avoid roads due to higher risk of mortality from vehicular collisions (Long et al. 2010), it appears that landscape context plays an important role in structuring this selection. In our study, road density was similar between the two study areas, but configuration differed dramatically (Fig. 1h). This configuration may be important as roads in the North generally followed a grided system making them easier to predict whereas roads in the South generally followed the topography of the landscape (e.g., ridgelines), making them less predictable and potentially occurring along paths that deer were selecting for during dispersal. Alternatively, roads that intersect forest may not be perceived as an obstacle compared to roads that pass through more open areas.
Movement
We expected that dispersal movements would be faster and more directional than movements within home ranges. Indeed, we found that deer tended to move on more directed paths and moved about twice as fast during a dispersal compared to within home ranges either before or after a dispersal. This pattern has previously been found in a variety of birds and mammals (Delgado et al. 2009; Soulsbury et al. 2011; Killeen et al. 2014), including white-tailed deer (Moll et al. 2021). Theoretical models indicate that moving in a straight-line search is more efficient at finding open territories (Zollner and Lima 1999), and thus may be the most energetically efficient way to disperse when selecting a new home range. Comparatively, spatial memory within the home range, as deer use resources, generates shorter non-directional movements (Van Moorter et al. 2009). Additionally, the faster movements that we observed during dispersal may reduce contact with conspecifics when moving through the home ranges of other individuals (Killeen et al. 2014). During home range movements, we found that speed was similar before and after a dispersal in the spring, but was faster following a dispersal in the fall. This increased speed in the fall may be because juvenile males are searching for mating opportunities (Schultz and Johnson 1992). Alternatively, food limitations later in the season may also increase speed as individuals move more across the landscape to find resources.
Conclusion
Dispersal is a key life history event which has received considerable theoretical and empirical study in terms of variation in habitat selection before and after dispersal events (Stamps et al. 2005; Day et al. 2019). Although there has recently been more effort devoted toward understanding how animals select and move through their environment during dispersal (e.g., Killeen et al. 2014; O’Neill et al. 2020; Frankish et al. 2022; Thorsen et al. 2022; Orgeret et al. 2023), there are still large knowledge gaps, even for well-studied species such as white-tailed deer. Our study found nuanced ways in which deer alter habitat selection and movement during dispersal; in particular showing stronger selection for waterways and topographical features. These results are in line with deer dispersing in Wisconsin that selected for areas near rivers and also another ungulate (elk; Cervus elaphus) which demonstrated a different selection response to topography during dispersal (Killeen et al. 2014; Gilbertson et al. 2022). However, habitat selection during dispersal is likely highly species specific with avian and terrestrial carnivores often showing less responsiveness to environmental gradients during dispersal movements (O’Neill et al. 2020; Thorsen et al. 2022; Orgeret et al. 2023). Such differences in how animals move through the landscape has potential implications for how animals perceive the landscape during this life history event. A better understanding of habitat selection during dispersal can help inform landscape connectivity planning (e.g., managing specifically for habitat selected during dispersal) and has implications for broad-scale landscape connectivity in the face functional fragmentation due to anthropogenic development and climate change.
References
Agostinelli C, Lund U (2022) R package “circular”: Circular Statistics (version 0.4–95). https://r-forge.r-project.org/projects/circular/.
Anderson CW, Nielsen CK, Schauber EM (2015) Survival and dispersal of white-tailed deer in the agricultural landscape of east-central Illinois. Wildl Biol Pract 11:26–41
Avgar T, Lele SR, Keim JL, Boyce MS (2017) Relative selection strength: quantifying effect size in habitat-and step-selection inference. Ecol Evol 7:5322–5330
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Beier P, McCullough DR (1990) Factors influencing white-tailed deer activity patterns and habitat use. Wildl Monogr 109:3–51
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400
Brown CF, Brumby SP, Guzder-Williams B, Birch T, Hyde SB, Mazzariello J, Czerwinski W, Pasquarella VJ, Haertel R, Ilyushchenko S et al (2022) Dynamic World, Near real-time global 10 m land use land cover mapping. Sci Data 9:251
Burt WH (1943) Territoriality and home range concepts as applied to mammals. J Mamm 24:346
Carson RG, Peek JM (1987) Mule deer habitat selection patterns in northcentral Washington. J Wildl Manag 51:46–51
Clements GM, Hygnstrom SE, Gilsdorf JM, Baasch DM, Clements MJ, Vercauteren KC (2011) Movements of white-tailed deer in riparian habitat: implications for infectious diseases. J Wildl Manag 75:1436–1442
Cox AS, Kesler DC (2012) Prospecting behavior and the influenceof forest cover on natal dispersal in aresident bird. Behav Ecol 23:1068–1077
Cullingham CI, Merrill EH, Pybus MJ, Bollinger TK, Wilson GA, Coltman DW (2011) Broad and fine-scale genetic analysis of white-tailed deer populations: estimating the relative risk of chronic wasting disease spread. Evol Appl 4:116–131
Day CC, McCann NP, Zollner PA, Gilbert JH, MacFarland DM (2019) Temporal plasticity in habitat selection criteria explains patterns of animal dispersal. Behav Ecol 30:528–540
Delgado MM, Penteriani V, Nams VO, Campioni L (2009) Changes of movement patterns from early dispersal to settlement. Behav Ecol Sociobiol 64:35–43
Diefenbach DR, Long ES, Rosenberry CS, Wallingford BD, Smith DR (2008) Modeling distribution of dispersal distances in male white-tailed deer. J Wildl Manag 72:1296–1303
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46
Dussault C, Ouellet J-P, Laurian C, Courtois R, Poulin M, Breton L (2007) Moose movement rates along highways and crossing probability models. J Wildl Manag 71:2338–2345
Fagan WF, Lewis MA, Auger-Méthé M, Avgar T, Benhamou S, Breed G, LaDage L, Schlägel UE, Tang W, Papastamatiou YP et al (2013) Spatial memory and animal movement. Ecol Lett 16:1316–1329
Forester JD, Im HK, Rathouz PJ (2009) Accounting for animal movement in estimation of resource selection functions: sampling and data analysis. Ecology 90:3554–3565
Frankish CK, Manica A, Clay TA, Wood AG, Phillips RA (2022) Ontogeny of movement patterns and habitat selection in juvenile albatrosses. Oikos 2022:e09057
Gilbertson MLJ, Ketz AC, Hunsaker M, Jarosinski D, Ellarson W, Walsh DP, Storm DJ, Turner WC (2022) Agricultural land use shapes dispersal in white-tailed deer (Odocoileus virginianus). Mov Ecol 10:1–18
Godvik IMR, Loe LE, Vik JO, Veiberg V, Langvatn R, Mysterud A (2009) Temporal scales, trade-offs, and functional responses in red deer habitat selection. Ecology 90:699–710
Gorelick N, Hancher M, Dixon M, Ilyushchenko S, Thau D, Moore R (2017) Google earth engine: planetary-scale geospatial analysis for everyone. Remote Sens Environ 202:18–27
Gould JH, Jenkins KJ (1993) Seasonal use of conservation reserve program lands by white-tailed deer in east-central South Dakota. Wildl Soc Bull 1973–2006(21):250–255
Grovenburg TW, Jacques CN, Klaver RW, Jenks JA (2011) Drought effect on selection of conservation reserve program grasslands by white-tailed deer on the Northern Great Plains. Am Midl Nat 166:147–162
Haus JM, Webb SL, Strickland BK, Rogerson JE, Bowman JL (2019) Land use and dispersal influence mortality in white-tailed deer. J Wildl Manag 83:1185–1196
Heit DR, Millspaugh JJ, McRoberts JT, Wiskirchen KH, Sumners JA, Isabelle JL, Keller BJ, Hildreth AM, Montgomery RA, Moll RJ (2023) The spatial scaling and individuality of habitat selection in a widespread ungulate. Landsc Ecol 38:1–15
Hesselbarth MHK, Sciaini M, With KA, Wiegand K, Nowosad J (2019) landscapemetrics: an open-source R tool to calculate landscape metrics. Ecography 42:1648–1657
Hijmans RJ (2022) raster: Geographic Data Analysis and Modeling. R package version 3.5–21. https://CRAN.R-project.org/package=raster.
Holbrook JD, Squires JR, Olson LE, DeCesare NJ, Lawrence RL (2017) Understanding and predicting habitat for wildlife conservation: the case of Canada lynx at the range periphery. Ecosphere 8:e01939
Hooven ND, Springer MT, Nielsen CK, Schauber EM (2023) Influence of natal habitat preference on habitat selection during extra-home range movements in a large ungulate. Ecol Evol 13:e9794
Jayakody S, Sibbald AM, Gordon IJ, Lambin X (2008) Red deer Cervus elephus vigilance behaviour differs with habitat and type of human disturbance. Wildl Biol 14:81–91
Kämmerle J-L, Brieger F, Kröschel M, Hagen R, Storch I, Suchant R (2017) Temporal patterns in road crossing behaviour in roe deer (Capreolus capreolus) at sites with wildlife warning reflectors. PLoS ONE 12:e0184761
Kays RW, Gompper ME, Ray JC (2008) Landscape ecology of eastern coyotes based on large-scale estimates of abundance. Ecol Appl 18:1014–1027
Keller BJ, Batten J, Wiskirchen KH, Hildreth AM, Cordell K (2017) 2017 Missouri deer season summary & population status report Missouri. USA Missouri Department of Conservation, Jefferson City
Kie JG, Evans CJ, Loft ER, Menke JW (1991) Foraging behavior by mule deer: the influence of cattle grazing. J Wildl Manag 55:665–674
Killeen J, Thurfjell H, Ciuti S, Paton D, Musiani M, Boyce MS (2014) Habitat selection during ungulate dispersal and exploratory movement at broad and fine scale with implications for conservation management. Mov Ecol 2:1–13
Laforge MP, Brook RK, van Beest FM, Bayne EM, McLoughlin PD (2016) Grain-dependent functional responses in habitat selection. Landsc Ecol 31:855–863
Lenth RV (2018) Emmeans: estimated marginal means, aka least-squares means. R Package Package Version 1(1):2
Levin SA (1992) The problem of pattern and scale in ecology: the Robert H. MacArthur Award Lecture. Ecology 73:1943–1967
Lima SL, Zollner PA (1996) Towards a behavioral ecology of ecological landscapes. Trends Ecol Evol 11:131–135
Lone K, Loe LE, Meisingset EL, Stamnes I, Mysterud A (2015) An adaptive behavioural response to hunting: surviving male red deer shift habitat at the onset of the hunting season. Anim Behav 102:127–138
Long ES, Diefenbach DR, Rosenberry CS, Wallingford BD, Grund MD (2005) Forest cover influences dispersal distance of white-tailed deer. J Mammal 86:623–629
Long ES, Diefenbach DR, Rosenberry CS, Wallingford BD (2008) Multiple proximate and ultimate causes of natal dispersal in white-tailed deer. Behav Ecol 19:1235–1242
Long ES, Diefenbach DR, Wallingford BD, Rosenberry CS (2010) Influence of roads, rivers, and mountains on natal dispersal of white-tailed deer. J Wildl Manag 74:1242–1249
Lutz CL, Diefenbach DR, Rosenberry CS (2015) Population density influences dispersal in female white-tailed deer. J Mamm 96:494–501
Manly BFL, McDonald L, Thomas DL, McDonald TL, Erickson WP (2002) Resource selection by animals: statistical design and analysis for field studies, 2nd edn. Kluwer Press, New York
McGarigal K, Wan HY, Zeller KA, Timm BC, Cushman SA (2016) Multi-scale habitat selection modeling: a review and outlook. Landsc Ecol 31:1161–1175
Millspaugh J, Marzluff JM (2001) Radio tracking and animal populations. Academic Press, San Diego
Moll RJ, McRoberts JT, Millspaugh JJ, Wiskirchen KH, Sumners JA, Isabelle JL, Keller BJ, Montgomery RA (2021) A rare 300 kilometer dispersal by an adult male white-tailed deer. Ecol Evol 11:3685–3695
Morales JM, Ellner SP (2002) Scaling up animal movements in heterogeneous landscapes: the importance of behavior. Ecology 83:2240–2247
Muff S, Signer J, Fieberg J (2020) Accounting for individual-specific variation in habitat-selection studies: efficient estimation of mixed-effects models using Bayesian or frequentist computation. J Anim Ecol 89:80–92
Nathan R, Getz WM, Revilla E, Holyoak M, Kadmon R, Saltz D, Smouse PE (2008) A movement ecology paradigm for unifying organismal movement research. Proc Natl Acad Sci 105:19052–19059
Nixon CM, Mankin PC, Etter DR, Hansen LP, Brewer PA, Chelsvig JE, Esker TL, Sullivan JB (2007) White-tailed deer dispersal behavior in an agricultural environment. Am Midl Nat 157:212–220
O’Neill HMK, Durant SM, Woodroffe R (2020) What wild dogs want: habitat selection differs across life stages and orders of selection in a wide-ranging carnivore. BMC Zool 5:1–11
Olden JD, Schooley RL, Monroe JB, Poff NL (2004) Context-dependent perceptual ranges and their relevance to animal movements in landscapes. J Anim Ecol 73:1190–1194
Orgeret F, Grüebler MU, Scherler P, van Bergen VS, Kormann UG (2023) Shift in habitat selection during natal dispersal in a long-lived raptor species. Ecography 2023:e06729
Peterson BE, Storm DJ, Norton AS, Van Deelen TR (2017) Landscape influence on dispersal of yearling male white-tailed deer. J Wildl Manag 81:1449–1456
Puskas RB, Fischer JW, Swope CB, Dunbar MR, McLean RG, Root JJ (2010) Raccoon (Procyon lotor) movements and dispersal associated with ridges and valleys of Pennsylvania: implications for rabies management. Vector-Borne Zoonotic Dis 10:1043–1048
QGIS Development Team (2022) QGIS Geographic Information System. QGIS Association
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. version 4.0.4. http://www.R-project.org
Radford JQ, Bennett AF (2007) The relative importance of landscape properties for woodland birds in agricultural environments. J Appl Ecol 44:737–747
Roever CL, Van Aarde RJ, Leggett K (2012) Functional responses in the habitat selection of a generalist mega-herbivore, the African savannah elephant. Ecography 35:972–982
Schick RS, Loarie SR, Colchero F, Best BD, Boustany A, Conde DA, Halpin PN, Joppa LN, McClellan CM, Clark JS (2008) Understanding movement data and movement processes: current and emerging directions. Ecol Lett 11:1338–1350
Schlägel UE, Grimm V, Blaum N, Colangeli P, Dammhahn M, Eccard JA, Hausmann SL, Herde A, Hofer H, Joshi J et al (2020) Movement-mediated community assembly and coexistence. Biol Rev 95:1073–1096
Schultz SR, Johnson MK (1992) Breeding by male white-tailed deer fawns. J Mammal 73:148–150
Seaman DE, Millspaugh JJ, Kernohan BJ, Brundige GC, Raedeke KJ, Gitzen RA (1999) Effects of sample size on kernel home range estimates. J Wildl Manag 63:739
Severinghaus CW (1949) Tooth development and wear as criteria of age in white-tailed deer. J Wildl Manag 13:195–216
Signer J, Fieberg J, Avgar T (2019) Animal movement tools (amt): R package for managing tracking data and conducting habitat selection analyses. Ecol Evol 9:880–890
Slarkin M (1985) Gene flow in natural populations. Annu Rev Ecol Syst 16:393–430
Soulsbury CD, Iossa G, Baker PJ, White PCL, Harris S (2011) Behavioral and spatial analysis of extraterritorial movements in red foxes (Vulpes vulpes). J Mammal 92:190–199
Stamps JA, Krishnan VV, Reid ML (2005) Search costs and habitat selection by dispersers. Ecology 86:510–518
Stewart KM, Bowyer RT, Weisberg PJ (2011) Spatial use of landscapes. In: Hewitt DG (ed) Biology and management of white-tailed deer. CRC Press, Boca Raton
Stubblefield CH, Vierling KT, Rumble MA (2006) Landscape-scale attributes of elk centers of activity in the central Black Hills of South Dakota. J Wildl Manag 70:1060–1069
Thorsen NH, Hansen JE, Støen O-G, Kindberg J, Zedrosser A, Frank SC (2022) Movement and habitat selection of a large carnivore in response to human infrastructure differs by life stage. Mov Ecol 10:52
Thurfjell H, Ciuti S, Boyce MS (2014) Applications of step-selection functions in ecology and conservation. Mov Ecol 2:1–12
U.S. Census Bureau. 2019 (2019) TIGER/Line Shapefiles (machinereadable data files)
USGS (U.S. Geological Survey) (2022a) 3D Elevation Program 1/3 Arc Second resolutionl. https://www.usgs.gov/the-national-map-data-delivery
USGS (U.S. Geological Survey) (2022b) National Hydrography Dataset (ver. USGS National Hydrography Dataset Best Resolution (NHD) - Missouri). https://www.usgs.gov/national-hydrography/national-hydrography-dataset
Van Moorter B, Visscher D, Benhamou S, Börger L, Boyce MS, Gaillard J-M (2009) Memory keeps you at home: a mechanistic model for home range emergence. Oikos 118:641–652
Volk MD, Kaufman DW, Kaufman GA (2007) Diurnal activity and habitat associations of white-tailed deer in tallgrass prairie of eastern Kansas. Trans Kans Acad Sci 110:145–154
Walter WD, Baasch DM, Hygnstrom SE, Trindle BD, Tyre AJ, Millspaugh JJ, Frost CJ, Boner JR, VerCauteren KC (2011) Space use of sympatric deer in a riparian ecosystem in an area where chronic wasting disease is endemic. Wildl Biol 17:191–209
Williamson SJ, Hirth DH (1985) An evaluation of edge use by white-tailed deer. Wildl Soc Bull 13:252–257
Wright CA, Mcroberts JT, Wiskirchen KH, Keller BJ, Millspaugh JJ (2019) Landscape-scale habitat characteristics and neonatal white-tailed deer survival. J Wildl Manag 83:1401–1414
Zollner PA, Lima SL (1999) Search strategies for landscape-level interpatch movements. Ecology 80:1019–1030
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14
Acknowledgements
We thank E. Flinn for project development and B. Keller, V. Vanderwerken, W. Dooling, and C. Wright for support with animal capture and field work. We thank S. Mackersie for help with categorizing deer dispersals.
Funding
This work was supported by the Missouri Department of Conservation Cooperative Agreement 369, the University of Montana, and the University of New Hampshire.
Author information
Authors and Affiliations
Contributions
All authors contributed to conceptualization and design of the study. JJM, JTM, KHW, JAS, JLI, and BJK facilitated funding, planning, and execution of field data collection. RBS analyzed the data and wrote the first draft of the manuscript with input from RJM. All authors contributed to revisions and gave final approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stephens, R.B., Millspaugh, J.J., McRoberts, J.T. et al. Scale-dependent habitat selection is shaped by landscape context in dispersing white-tailed deer. Landsc Ecol 39, 84 (2024). https://doi.org/10.1007/s10980-024-01879-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10980-024-01879-z