Abstract
Context
Slash-and-burn agriculture generates landscape mosaics composed of different land uses. Ecological theory postulates that in these mosaics, the structure of species assemblages depends on both local- and landscape-scale factors, but their relative role remains poorly known.
Objectives
Understanding the relative importance of local and landscape variables for bird assemblages in landscape mosaics of the Mayan tropical forest, Mexico.
Methods
We evaluated the effect of two local variables (land use type and tree basal area) and two landscape variables (old-growth forest cover and human proximity) on bird abundance and diversity, separately assessing forest and non-forest birds. As species extirpation may accelerate when forest loss exceeds certain limits (extinction thresholds), we assessed forest loss effects with linear and nonlinear models.
Results
We recorded 1867 birds from 118 species in four land-use types (old-growth forest, secondary forest, burned lands, and agricultural lands). Land-use type and surrounding old-growth forest cover better predicted bird abundance and diversity than tree basal area and human proximity. Agricultural lands showed the lowest abundance and diversity of forest species, but the highest abundance and diversity of non-forest species. We found no support for the extinction threshold hypothesis. Yet, independently of the land use, old-growth forest loss decreased the diversity of forest species and increased the abundance and diversity of non-forest species. Human proximity also decreased total bird abundance, especially because of the loss of forest species.
Conclusions
Our findings highlight the key role played by old-growth forests for preserving bird assemblages in landscape mosaics exposed to slash-and-burn agriculture.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forest loss and degradation is reaching planetary limits that, if surpassed, will have catastrophic consequences for wildlife and humans (Rockström et al. 2009; Ripple et al. 2017). These threats are mainly caused by extensive agriculture (FAO 2022). Yet, small-scale slash-and-burn agriculture was responsible for 25% of tropical forest loss between 2001 and 2015 (Curtis et al. 2018), and is related to biodiversity loss and soil impoverishment (Pedroso et al. 2009). This farming method creates complex landscape mosaics composed of old-growth forest remnants, secondary forests, relatively small (0.5–5 ha) agricultural fields, and areas burned by accidental fires related to agriculture (Terán and Rasmussen 2009; Ellis et al. 2017). Nevertheless, our understanding of the ability of species to use these land-use types is limited, as ecological research has been focused on assessing biodiversity patterns in protected areas (Martin et al. 2012). Consequently, if we intend to design biodiversity-friendly landscapes, we need to improve our knowledge of the factors that shape biotic assemblages in these landscape mosaics at different scales (Melo et al. 2013; Arroyo‐Rodríguez et al. 2020).
This topic has gained increased attention in ecology and conservation biology (Tscharntke et al. 2005; Gonthier et al. 2014; Mendenhall et al. 2016; Yang et al. 2021; Bezerra et al. 2023; Carvalho et al. 2023), and several theoretical models have been proposed to predict wildlife responses to local and landscape factors (Tscharntke et al. 2012; Arroyo-Rodríguez et al. 2020). For example, the habitat amount hypothesis predicts that species density in a given site increases as the amount of available habitat in the surrounding landscape increases (Fahrig 2013). Although some studies support this prediction (Watling et al. 2020), others suggest that species density is weakly related to habitat amount (e.g. Martínez‐Ruiz et al. 2024). These contradictory results may result from the confounding effect of other local and landscape factors. For example, individuals in landscape mosaics can use resources not only from native habitats, but also from the surrounding anthropic matrix. This process is called “landscape supplementation” (Dunning et al. 1992) and can buffer some negative effects of habitat loss on wildlife (Gascon et al. 1999). Human proximity in the surrounding landscape can also be an important driver of wildlife populations, including birds (Ferreiro-Arias et al. 2024), as it promotes chronic disturbances, such as hunting, logging, and the invasion of feral pets, such as cats and dogs (Benítez-López et al. 2017; Nickel et al. 2020). While human activities may offer some resources (e.g. food) to wildlife, they also make animals more vulnerable to hunting or trafficking (Naughton‐Treves et al. 2003; Ferreiro-Arias et al. 2024). At the local scale, tree basal area can also be critical for biotic assemblages, as this variable is positively related to forest age, its conservation status, and the availability of key resources such as water, food, shelter, and appropriate nesting sites (Chapman et al. 1992; Arroyo-Rodríguez et al. 2007; Cudney‐Valenzuela et al. 2023).
These local and landscape factors can be particularly important for bird assemblages (Leyequien et al. 2007; Arriaga-Weiss et al. 2008; Haslem and Bennett 2008; Terraube et al. 2016; Macchi et al. 2020; Vargas-Cárdenas et al. 2022). Birds are usually negatively impacted by human disturbances, affecting their habitat use, foraging strategies and reproductive behavior (Price 2008). However, few studies have simultaneously assessed the effect of local and landscape variables on birds across complex landscape mosaics (but see Smith et al. 2001; Urquiza‐Haas et al. 2011; Macchi et al. 2020). Filling this knowledge gap is particularly important in the Mayan Forest of Mexico – a biodiversity hotspot that has been affected by slash-and-burn agriculture since before European colonization (De Albuquerque et al. 2015). Despite a long history of land-use change, the region has recently experienced a rapid expansion of the agricultural frontier and increasing occurrence of fire, which are causing a rapid loss of old-growth forests (Ellis et al. 2017; Pacheco et al. 2021; Global Forest Watch 2024). Therefore, identifying the response of birds to these land-use changes is critical to provide effective conservation guidelines to landscape managers and policymakers.
Here, we assessed the effect of two local (land use type and tree basal area) and two landscape factors (percentage of old-growth forest cover and human proximity) on the abundance and diversity of bird assemblages in the Mayan Forest, Yucatan, Mexico. We assessed these responses for the complete bird assemblage, and then separately for forest and non-forest birds. In particular, we addressed the following questions: (i) what are the most important local and landscape variables related to bird abundance and diversity?; (ii) how does each variable affect forest vs. non-forest birds? If a relationship exists; and (iii) what is the threshold value of forest cover below which the loss of individuals and/or species increases?
We hypothesized that land-use type and old-growth forest cover are the most important predictors because they determine habitat structure and the availability of resources for birds at local and landscape scales, respectively (e.g. Fahrig et al. 2011; Fahrig 2013). Furthermore, considering that structurally complex habitats provide a larger diversity of resources for birds (e.g. water, food, nesting and resting sites; Karr and Roth 1971; Terborgh 1987; Hanowski et al. 2005; Lee and Carroll 2018), we expect that the more heterogeneous land uses (old-growth and secondary forests) produce a greater abundance and diversity of birds, particularly of forest species, than structurally simpler habitats (burned and agricultural lands). Although expected to be weaker, the effect of tree basal area is likely positive for forest birds and negative for non-forest birds (Lynch and Whigham 1984). Similarly, human proximity probably has negative effects on forest species, but positive ones on non-forest species, as the latter group is composed of disturbance-adapted species (Blumstein 2013; Vargas-Cárdenas et al. 2022). Finally, following theory (Fahrig 2013) and empirical evidence (Macchi et al. 2020; Watling et al. 2020; Vargas-Cárdenas et al. 2022), we expect the diversity of forest birds to increase in sites surrounded by higher old-growth forest cover. Yet, such an effect could be nonlinear, as species loss usually accelerates in landscapes with < 30–40% of forest cover (Fahrig 2002; Swift and Hannon 2010; Morante-Filho et al. 2015; Macchi et al. 2019; Arroyo‐Rodríguez et al. 2021).
Methods
Study area
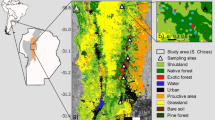
The study was conducted in the Mayan Forest of the Yucatan Peninsula, Mexico (Fig. 1), located in the municipalities of Chemax (Yucatan) and Lazaro Cardenas (Quintana Roo), encompassing approximately 15,000 hectares. The climate is warm (mean annual temperature = 24.9 to 26.1 °C) and subhumid, with annual precipitation ranging from 1220 to 1550 mm (INEGI 2017a, b). The area was originally covered by evergreen semideciduous tropical forest. However, the expansion of agriculture and the occurrence of fires over the past decade resulted in the loss of approximately 2700 ha of forest (18% of its original cover; Global Forest Watch 2024). Consequently, the region is currently comprised of a complex land use mosaic, including patches of old-growth forest, secondary forest, burned areas, and agricultural land (Fig. 1).
Location of the study region and sampled sites in the Yucatan Peninsula, Mexico. We sampled bird assemblages in five independent sites per land use type: old-growth forest (black triangles), secondary forest (black circles), burned lands (black squares), and agricultural land (‘milpa,’ white circles). In each site, we also quantified the percentage of old-growth forest cover across five concentric circular landscapes (radii 200–1000 m) (see an example in the upper right panel)
Land use classification and selection of study sites
To select our sampling sites, we first classified the land uses present in our study area. We developed a land-cover map in the JavaScript API Code Editor in the Google Earth Engine, using high-resolution satellite imagery from the Sentinel-2 Multispectral Instrument. These satellite images have a resolution of 10 m per pixel and are open access (Rapinel et al. 2019). We used a collection of satellite images from the 1st of January to the 31st of October 2021 to create the land-cover map. From these satellite images, we generated a mosaic (e.g. a fusion of several satellite images of the same area) and applied an atmospheric correction to produce a clean satellite image without cloud cover. We then used the random forest algorithm to delimit the different land-cover categories (Breiman 2001), which is one of the most effective machine learning algorithms available for digital classification (Pal 2005). We classified the land-cover classes as follows: (i) agricultural land; (ii) burned land; (iii) secondary forest; and (iv) old-growth forest. Agricultural lands consisted of traditional ‘milpa’, a Mesoamerican polyculture that includes maize, beans, and pumpkin. Burned lands resulted from recent (< 5 years) accidental fires related to agriculture. They consisted of young, regenerating trees, with a maximum height of 2–3 m, forming a dense thicket with some standing trees that survived the fire. Secondary forests are older forests (10–25 years old) that regenerated following the abandonment of lands previously dedicated to traditional agriculture. These forests exhibit a taller canopy (8–10 m high), with some emergent trees (up to 15 m high), mostly from light-demanding tree species, such as Cecropia peltata, Cochlospermum vitifolium, Guazuma ulmifolia, and Bursera simaruba. Finally, old-growth forests have not been used for agriculture or livestock for more than 40–50 years, consisting of trees above 10 m tall, with emergent trees reaching > 20–25 m (e.g. Ficus spp., Metopium brownei, Cordia alliodora, Enterolobium cyclocarpum, Cedrela odorata).
In order to train the random forest algorithm and accurately delimit the different land-cover classes, we collected a series of training points in the field in the northern region of the state of Quintana Roo. The location of each training point was determined with a Global Positioning System (GPS) device (model Garmin GPSMAP 64). These training points for each land-cover class allowed the classification program to recognize the different classes. 70% of the data was used to train the algorithm to recognize each land-cover class and the remaining 30% was used to test the model. The consumer and producer accuracy values for the agricultural lands layers were greater than 0.93 for each of the coverage layers, and the Kappa coefficient accuracy for the land-cover map that we produced was 0.89. This coefficient indicates that the map is of high quality and is a good representation of the satellite image used as a reference. Once this process was completed, we selected five bird sampling sites per land-cover class (4 classes × 5 sites = 20 sampling units). These sites were separated by at least 1 km to ensure independence of the samples (Bibby et al. 1992).
Bird sampling
We identified both resident and migrant bird species on each sample plot in two field campaigns, one conducted during the dry season (November–December 2022) and the other during the rainy season (May–June 2023). Birds were surveyed with 20-m radius point-counts (Bibby et al. 1992). We sampled three different points per sampling unit (20 sampling units × 3 points = 60 sampling points per field campaign), positioned in a triangular configuration with a 100-m distance separating them. These three points were sampled on different days. A total of 10 min was spent at each point-count, and all birds seen or heard within a 20-m radius were counted. This was done for four hours after sunrise, which occurred at 5:00 AM in the rainy season and 6:00 AM in the dry season. Birds were visually identified with the help of Celestron Nature DX 10 × 42 binoculars. To verify bird identification, all bird vocalizations (songs and calls) were digitally recorded with a Samsung S9 + built-in voice recorder. When possible, we also photographed the birds at each sampling point (Nikon P900 camera). In the few cases where the birds could not be identified in the field, we compared their songs and photographs with existing acoustic databases (Cornell Lab of Ornithology 2023; Xeno-canto 2023) and field guides (Van Perlo 2006; Howell and Webb 2010). We used the bird nomenclature proposed by the American Ornithological Society (Chesser et al. 2023).
Subsequently, we used the literature (Stotz et al. 1996; Bregman et al. 2014; Carrara et al. 2015; Billerman et al. 2022) to classify birds into two groups: forest birds (e.g. those that inhabit forest interior and secondary forests) and non-forest birds (e.g. those species that use a variety of habitats, including the above mentioned, open areas, and crops). We excluded aquatic and nocturnal birds because our methods were not designed to detect these groups.
Response variables
To circumvent the potential issues associated with pseudo-replication in our analyses, we summed all the data from the three points and both field campaigns (dry and rainy) to obtain an accumulated value of abundance and diversity per site. We measured bird diversity with Hill numbers of order 0 (0D, species richness) and 2 (2D, number of dominant species) (Jost 2006) using the ‘iNEXT’ package (Chao et al. 2014; Hsieh et al. 2022) for R (v.4.3.1). To assess whether 0D was correctly estimated in all sites, we calculated the sample coverage index proposed by Chao and Jost (2012). We found that sample coverage varied among sites (0.7–1.0). Therefore, we standardized the samples using the coverage-based extrapolation and rarefaction techniques suggested by Chao and Jost (2012). In particular, we used the estimateD function within the ‘iNEXT’ package to estimate the 0D within each site for samples of equal completeness. Such completeness differed slightly among bird groups, with values of 0.77, 0.79, and 0.78 for the complete bird assemblage, forest birds, and non-forest birds, respectively. All statistical analyses involving 0D were conducted using the estimated values of 0D to prevent any potential bias in the results due to differences in sample coverage among sites (Chao and Jost 2012).
Predictor variables
We considered four predictor variables that can be particularly important for birds and other vertebrate groups in complex landscape mosaics (Terborgh 1987; Fahrig 2013; Benítez-López et al. 2017; Lee and Carroll 2018). Two variables described the characteristics of sampling sites (local variables hereafter): (i) the land-use type (categorical variable) where the site is located; and (ii) the tree basal area (continuous variable) in each site. This latter variable was calculated by summing the basal area of all trees with a diameter at breast height (DBH) ≥ 10 cm recorded in a 20 m radius from the center of each sample site (i.e. sampled area per site = 0.1 ha). We selected this radius because it covered the area where birds were surveyed. We also measured two landscape variables, which described the spatial context surrounding the sites; (iii) the percentage of old-growth forest cover; and (iv) a human proximity index. As the effect of old-growth forest cover may depend on the scale at which this landscape variable is measured, we used a multi-scale approach to identify the “scale of effect”, i.e. the landscape size that yields the strongest association between forest cover and each response variable (Jackson and Fahrig 2015). In particular, we estimated forest cover with ArcGis 10.8 and the Patch Analyst extension (Rempel et al. 2012), considering landscapes of 200- to 1000-m radii (at 200 m intervals) from the center of each sampling site. Human proximity (HP) was assessed with the equation proposed by Scabin and Peres (2021):
where S represents the population size of the human settlements within a given radii (es = Estrella, co = Cocoyol, and bv = Buenavista), and d is the Euclidian distance from each sample site to the center of each human settlement. We calculated HP considering two scales (5- and 10-km radii), as (i) all human settlements were located > 5 km away; and (ii) local hunters informed us that they rarely go beyond 10 km. Population sizes were obtained from the Population and Housing Census 2020 (INEGI 2020).
Data analysis
To assess the effect of each predictor variable (i.e. percentage of old-growth forest cover, human proximity, tree basal area, and land use type) on each response (i.e. bird abundance, expected species richness, and number of dominant species) and bird group (i.e. complete assemblage, forest species, and non-forest species), we used generalized linear models (GLMs). As suggested for count variables (bird abundance) in GLMs, we used a Poisson error distribution, whereas continuous variables (expected species richness and number of dominant species) were used a Gaussian error distribution (Crawley 2012). We first identified the scale at which each landscape predictor (forest cover and human proximity) best predicted each response. To this end, we built simple GLMs and compared them using the Akaike Information Criterion corrected for small samples (AICc). For each predictor-response combination, the best model was the one with the lowest AICc value (Table S1). We then built the full (complete) model for each response variable, which included both landscape variables measured at their respective scale of effect (see Table S1). To assess possible multi-collinearity problems in these full models, we calculated the Variance Inflation Factor (VIF) of each predictor variable with the ‘car’ package (Fox and Weisberg 2019). As all VIF values were < 2, we assumed that there are no collinearity problems in our models (O’brien 2007). To identify the most important predictor variable of each response and bird group, we used an information-theoretic approach and a multi-model averaging approach (Burnham and Anderson 2002), with the ‘glmulti’ (Calcagno 2020), and ‘AICcmodavg’ packages (Mazerolle 2023). For simplicity (parsimony principle), we excluded possible interaction effects between predictors and assessed only isolated and combined (additive) effects. For each response variable, we constructed 25 models, which represented all combinations of predictor variables plus the null model. For each candidate model, we computed the AICc and Akaike weight (wi). To correct for overdispersion associated to count data, bird abundance was assessed with qAICc instead of AICc values. We calculated model-averaged parameter estimates (and associated unconditional variances) using the Akaike weights (wi) (Carrara et al. 2015). We also calculated the sum of Akaike weights (∑wi) for each predictor variable as a proxy of relative importance (Burnham and Anderson 2002). To be more conservative, we considered that a given variable was important for a given response if the following three criteria were met: (i) it showed a relatively high Σwi; (ii) the model-averaged parameter estimate was higher than its unconditional variance (i.e. it did not include zero); and (iii) it showed a relatively high goodness of fit (Burnham and Anderson 2002). Following Carrara et al. (2015), the goodness-of-fit of the models was estimated as the percentage of explained deviance by the complete model.
To assess potential extinction thresholds in the bird assemblages, we assessed the linear and nonlinear effect of old-growth forest cover on bird abundance and diversity. The linear model was a GLM, and the nonlinear model was a piecewise regression model built with the ‘segmented’ package (Muggeo 2008). Again, forest cover effects were measured across five scales, so we compared 11 models per response (5 linear models, 5 nonlinear models, and the null model) to identify which had the highest empirical support (i.e. the lowest AICc value).
Results
Overview
In total, we recorded 1867 birds of 118 species (31 families). Seventy-two species (61%) were non-forest birds; the rest (39%) were forest birds. Bird abundance per site ranged from 50 to 145 individuals (mean ± SD, 95 ± 25 individuals), and species richness ranged from 29 to 47 species (37 ± 5 species). Considering the total number of species per land-use type, burned lands (82 species), agricultural lands (78), and secondary forests (74) showed more species than old-growth forests (63), but this pattern was driven by the relatively high number (and percentage) of non-forest birds in agricultural and burned lands. In contrast, forest birds were mainly recorded in old-growth and secondary forests (Figure S1). In general, most species were recorded in two or more land-use types (shared distribution), and the percentage of species restricted to one land-use type was similarly low in all land uses (10–14.8%; Figure S1). The species with the highest number of individuals were Rufous-browed Peppershrike (Cyclarhis gujanensis; n = 114), Yucatan Jay (Cyanocorax yucatanicus; 85), Olive-throated Parakeet (Eupsittula nana; 79), and Melodious Blackbird (Dives dives; 70). Yet, each land-use type was dominated by different species, with old-growth and secondary forests being dominated by C. gujanensis, and agricultural lands by D. dives (Fig. 2).
Effects of local and landscape variables on bird abundance and diversity
The importance of each predictor variable depended on the response variable and bird group, but in general, old-growth forest cover and land-use type predicted the observations better than tree basal area and human proximity (Fig. 3). Regardless of the bird group, old-growth forest cover was negatively related to bird abundance, but positively related to the diversity (0D and 2D) of forest species (Table 1). Land-use type was particularly important for forest and non-forest species (Fig. 3), as agricultural lands showed the lowest abundance and diversity of forest species and the highest abundance of non-forest birds (Fig. 4). Tree basal area mainly predicted abundance patterns (Fig. 3), as more individuals, especially those belonging to non-forest species, were recorded in sites with a lower basal area (i.e. more open habitats; Table 1). We also found that species richness of the complete bird assemblage tended to increase in sites with higher tree basal area. Finally, although more weakly, human proximity also played a role in shaping bird assemblages, especially forest species, which tended to decrease in abundance and diversity in sites with higher human proximity (Fig. 3; Table 1).
Relative importance (i.e. sum of Akaike weights) of each predictor variable obtained through multimodel comparison of generalized linear models for bird abundance and diversity of order 0 (0D, species richness) and 2 (2D, number of dominant species), for the complete bird assemblage, and for forest and non-forest species. The landscape predictors (i.e. old-growth forest cover and human proximity index) were included at their respective scales of effect (Table S1). The percentage of deviance explained by these four predictor variables is indicated within parentheses for each response variable
Linear vs nonlinear effects of old-growth forest cover
We found that linear models better predicted the observations than nonlinear ones (Fig. 5). In particular, we assessed 9 study cases (i.e. 3 response variables in 3 bird groups), and in most of them (5 of 9 cases, 56%) the linear models showed higher empirical support (i.e., lower AICc) than nonlinear models (Table S2). The linear models indicated that forest loss proportionally decreases the diversity (0D and 2D) of forest species but increases the abundance and diversity of non-forest species (Fig. 5). We only found two cases in which nonlinear models predicted the bird data better than linear models, and indicated that total bird abundance, and particularly the abundance of forest species, reached their maximum values in intermediately deforested landscapes (30–40% of old-growth forest cover; Fig. 5).
Best models describing the effect of landscape old-growth forest cover on bird abundance, species richness, and number of dominant species, separately assessed for the complete bird assemblage, forest, and non-forest species. When nonlinear models (e.g. piecewise regression) best predicted the data, we indicate the breaking point (vertical dotted line) and 95% confidence interval (shaded area). Note that the scale (landscape radii, in meters) at which forest cover was measured differs among response variables and bird groups (see details in Table S3)
Discussion
To the best of our knowledge, ours is the first study to assess the isolated and combined (additive) effect of two local (site-scale) and two landscape variables on the structure of bird assemblages in complex landscape mosaics exposed to slash-and-burn agriculture. We separately assessed forest and non-forest birds to identify the most important drivers of each bird group, and assessed whether forest loss effects are linear (proportional) or nonlinear, to evaluate the extinction threshold hypothesis (Swift and Hannon 2010). This research provides new knowledge on the factors that shape biotic assemblages in these landscape mosaics at different scales, which is a critical step if we are to design biodiversity-friendly landscapes (Melo et al. 2013; Arroyo-Rodríguez et al. 2020). This topic is not trivial, as the expansion of slash-and-burn agriculture during the last decades was responsible for 25% of tropical forest loss, and is threatening biodiversity worldwide (Pedroso et al. 2009; Curtis et al. 2018).
Our findings indicate that, as expected, local land-use type and surrounding old-growth forest cover are important predictors of the structure of bird assemblages. Forest and non-forest birds exhibited opposing responses to these predictors, with the diversity of forest species reaching its peak in sites located in old-growth and secondary forests, while the diversity of non-forest species peaked in treeless areas (i.e. burned and agricultural lands; Fig. 4). Furthermore, independently of the land-use type, the loss of old-growth forest cover in the surrounding landscape decreased the diversity of forest birds but increased the diversity of non-forest species (Fig. 5). Nevertheless, such responses were proportional (linear) to forest loss, suggesting that there are no extinction thresholds in the study landscapes. As discussed below, these findings have important ecological and applied implications.
The relatively high importance of land-use type and old-growth forest cover for birds is not surprising. There is evidence from other ecosystems that these two variables influence the structure of habitats and the availability of resources at local and landscape scales (Fahrig et al. 2011; Fahrig 2013). For instance, structurally complex habitats, such as old-growth and secondary forests, can provide a greater quantity and variety of resources (e.g. water, food, nesting sites) for forest species (Karr and Roth 1971; Terborgh 1987; Díaz et al. 2005; Hanowski et al. 2005; Kirk et al. 2012; Lee and Carroll 2018; Cudney-Valenzuela et al. 2023). This can explain why the diversity of this bird group peaked in old-growth and secondary forests and was positively related to old-growth forest cover in the surrounding landscape. Yet, by definition, non-forest species are able to use resources from a variety of habitats, including anthropogenic land uses. Consequently, they can thrive and proliferate in human-modified land covers (Bregman et al. 2014; Morante-Filho et al. 2015; Carrara et al. 2015; Macchi et al. 2020; Billerman et al. 2022; Vargas-Cárdenas et al. 2022), explaining why the diversity of non-forest birds increased in more deforested landscapes, especially in treeless land covers, such as burned and agricultural lands. Indeed, the abundance of non-forest birds was strongly and negatively related to local tree basal area, thereby indicating their dominance in treeless sites. Following the 'winner-loser paradigm' (Macchi et al. 2020; Filgueiras et al. 2021), these results suggest that forest species are negatively impacted by human disturbances (loser species), and are being gradually replaced by non-forest species (winner species).
Importantly, these winner-loser replacements caused compensatory dynamics, as the extirpation of forest species was to some degree compensated by the colonization of non-forest species. This pattern was particularly noticeable when assessing bird responses to old-growth forest loss, allowing the maintenance of total abundance and diversity in the complete bird assemblage (Fig. 5). Such compensatory dynamics have been previously documented in studies of birds (Supp and Ernest 2014; Carrara et al. 2015; Morante-Filho et al. 2015; Vargas-Cárdenas et al. 2022) and other vertebrate groups (e.g. Russildi et al. 2016), and highlight the importance of separately evaluating forest and non-forest species to better understand the impact of land-use change on biological assemblages. It is important to note that had we evaluated only the complete assemblage, we would have concluded that forest loss has a weak effect on birds. However, this would have overlooked the fact that forest species are critically impacted by old-growth forest loss.
A somewhat unexpected outcome was the relatively high abundance of forest birds in burned lands. These areas may be structurally simple when compared to old-growth and secondary forests, but they have a dense understory where forest birds can find refuge and a variety of foods (e.g. insects, fruits, flowers). Moreover, the burned lands we studied also have some scattered trees that survived the fire, as well as a few standing dead trees that can be used by forest birds as nesting sites and perches for resting. The importance of these natural structures for forest birds is increasingly evident in restoration projects, as they can attract forest birds, thereby promoting seed dispersal to restored areas (e.g. Alencar and Guilherme 2020). In this sense, the high abundance of forest (and non-forest) species in burned lands could be considered good news, as it suggests that birds are visiting these areas, and in large numbers, potentially contributing to their recovery. This would be an interesting avenue for future research.
Our findings also suggest that secondary forests have a high conservation value. The abundance and diversity of birds were similarly high in old-growth and secondary forests, both when considering the complete bird assemblage, and forest birds alone. Secondary forests shared 54 species with old-growth forests and were dominated by forest species such as the Yucatan Jay (Cyanocorax yucatanicus, a species endemic to the Yucatan peninsula) and the Thicket tinamou (Crypturellus cinnamomeus). Our findings support previous studies on the topic (Dunn 2004; Dent and Wright 2009; Vargas-Daza et al. 2023) and can be explained by two conditions that have been demonstrated to increase the conservation value of secondary forests (reviewed by Melo et al. 2013; Arroyo-Rodríguez et al. 2017): (i) we studied relatively old (10–25 years) secondary forests; and (ii) the region still maintains a relatively high proportion of old-growth forests. Regarding the first point, forest age is important because secondary forests increase their structural complexity over time, which contributes to the accumulation of more bird species (Dunn 2004; Santamaria-Rivero et al. 2016; Acevedo-Charry and Aide 2019). However, such accumulation depends on the colonization of species from neighboring old-growth forests, which represent the main source of forest species in human-modified landscapes (Melo et al. 2013; Arroyo-Rodríguez et al. 2017). Consequently, the conservation value of secondary forests is context-dependent, as these conditions could soon change if slash-and-burn agriculture and associated fires continue to expand in the region (Ellis et al. 2017; Pacheco et al. 2021; Global Forest Watch 2024).
Although impoverished compared to undisturbed forests, bird assemblages in agricultural lands (traditional ‘milpa’) still maintain a considerable abundance and diversity of birds. In fact, many forest species, such as Patagioenas flavirostris, Amazona xantholora, Cyanocorax yucatanicus and C. yncas, were recorded in agricultural lands. This pattern can be related to several complementary, but non-exclusive factors. For example, the studied agricultural lands are relatively small (1–10 ha), have low-external inputs (e.g. agrochemicals), and are located in a highly forested region with low human population density—attributes that can make agricultural lands more biodiversity-friendly (e.g. Mendenhall et al. 2016; Bisht et al. 2020). Also, birds have a relatively high vagility, which may contribute to the spillover of individuals across land-use types, especially from forested to agricultural lands (Tscharntke et al. 2005, 2012). Therefore, again, the conservation role of these agricultural lands is contingent upon the specific landscape-scale context and is likely to vary depending on the future management of these landscapes.
As expected, tree basal area and human proximity were relatively less important for birds. Yet, in agreement with previous studies (James and Wamer 1982; Askins et al. 1987; Jayson and Mathew 2003; Jankowski et al. 2013) tree basal area was positively related to species diversity, probably because increased tree basal area adds structural complexity to the habitats and determines the conservation value of the forest and the availability of resources (e.g. water, food, and shelter for wildlife; Chapman et al. 1992; Arroyo-Rodríguez et al. 2007; Cudney‐Valenzuela et al. 2023). However, bird abundance related negatively (not positively) to this tree basal area, probably because of the high abundance of non-forest birds in treeless land uses. Regarding the effect of human proximity, our findings align with expectations, as this predictor was negatively related to bird abundance. Although this effect was independent of the bird group, it was stronger in forest birds. This pattern may relate to chronic disturbances (e.g. hunting, logging, and feral pets) in sites located close to human settlements (Thiollay 1999; Pautasso 2007; Benítez-López et al. 2017; LaManna and Martin 2017; Ferreiro-Arias et al. 2024). However, as hunting pressure can vary among species, and is usually stronger in large-bodied species (Ferreiro-Arias et al. 2024), additional species-specific studies are needed to better understand the effect of human proximity on the studied species.
Collectively, our findings suggest that old-growth forests play a pivotal role in the conservation of tropical birds in heterogeneous landscape mosaics of the Mayan Forest in Mexico. In this region, slash-and-burn agriculture has created heterogeneous landscape mosaics composed of different land uses, most of which still maintain a large number of tropical birds. However, such conservation value largely depends on the percentage of old-growth forests in the surrounding landscape, as it can facilitate the cross-habitat spillover of birds, especially from natural to anthropogenic land uses. In this sense, placing our findings within the land sharing/sparing model, our study concurs with previous studies suggesting that most species will do better if slash-and-burn agriculture is practiced on as small an area as possible while preserving (sparing) as much old-growth forest cover as possible in the surrounding landscape (reviewed by Phalan 2018). Importantly, the replacement of forest species (losers) by non-forest species (winners) in more disturbed sites contributed to maintaining a high number of species. Yet, such compensatory dynamics were not strong enough to prevent the loss of species in agricultural lands, where we found the lowest species diversity. Therefore, in agreement with Gibson et al. (2011) and others (e.g. Fahrig 2013; Arroyo-Rodríguez et al. 2020; Watling et al. 2020), our results support the idea that old-growth forests are irreplaceable for preserving tropical biodiversity, in general, and forest species in particular. In this sense, it is worth highlighting that most of the lands in this region are owned by local communities (locally known as ‘ejidos’), and therefore, their future will depend on landowner activities and decisions in the mid and long terms. Thus, it may be necessary to implement incentives, such as payment for environmental services, to promote the establishment of new forest reserves, which remain quite scarce in the region. Furthermore, as low-paying nonfarming occupations are known to be associated with forest loss in Mesoamerican reserves (Auliz-Ortíz et al. 2024), forest loss could perhaps be prevented if we provide nonfarming livelihood opportunities (e.g. ecotourism) to local communities. Taken together, such simple actions could have profound positive effects on birds, contributing to maintaining their key ecological roles (e.g. seed dispersal and pest control).
References
Acevedo-Charry O, Aide TM (2019) Recovery of amphibian, reptile, bird, and mammal diversity during secondary forest succession in the tropics. Oikos 128:1065–1078. https://doi.org/10.1111/oik.06252
Alencar L, Guilherme E (2020) Artificial perches for the supply of seeds in a fragmented landscape in southwest Brazilian Amazon. Rev Bras Bot. 43:1013–1023. https://doi.org/10.1007/s40415-020-00662-z
Arriaga-Weiss SL, Calmé S, Kampichler C (2008) Bird communities in rainforest fragments: guild responses to habitat variables in Tabasco, Mexico. Biodivers Conserv 17:173–190. https://doi.org/10.1007/s10531-007-9238-7
Arroyo-Rodríguez V, Mandujano S, Benítez-Malvido J, Cuende-Fanton C (2007) The influence of large tree density on howler monkey (Alouatta palliata mexicana) presence in very small rain forest fragments. Biotropica 39:760–766. https://doi.org/10.1111/j.1744-7429.2007.00330.x
Arroyo-Rodríguez V, Melo FPL, Martínez-Ramos M et al (2017) Multiple successional pathways in human-modified tropical landscapes: new insights from forest succession, forest fragmentation and landscape ecology research. Biol Rev 92:326–340. https://doi.org/10.1111/brv.12231
Arroyo-Rodríguez V, Fahrig L, Tabarelli M et al (2020) Designing optimal human-modified landscapes for forest biodiversity conservation. Ecol Lett 23:1404–1420. https://doi.org/10.1111/ele.13535
Arroyo-Rodríguez V, Fahrig L, Watling JI et al (2021) Preserving 40% forest cover is a valuable and well-supported conservation guideline: reply to Banks‐Leite et al. Ecol Lett 24:1114–1116. https://doi.org/10.1111/ele.13689
Askins RA, Philbrick MJ, Sugeno DS (1987) Relationship between the regional abundance of forest and the composition of forest bird communities. Biol Conserv 39:129–152. https://doi.org/10.1016/0006-3207(87)90030-9
Auliz-Ortiz DM, Benítez-Malvido J, Arroyo-Rodríguez V et al (2024) Underlying and proximate drivers of biodiversity changes in Mesoamerican biosphere reserves. Proc Natl Acad Sci USA 121(6):e2305944121. https://doi.org/10.1073/pnas.2305944121
Benítez-López A, Alkemade R, Schipper AM et al (2017) The impact of hunting on tropical mammal and bird populations. Science 356:180–183. https://doi.org/10.1126/science.aaj1891
Bezerra JS, Arroyo-Rodríguez V, Dupuy JM, Leal IR, Tabarelli M (2023) Negative impact of slash-and-burn agriculture on the seed rain in a tropical dry forest. For Ecol Manage 531:120821. https://doi.org/10.1016/j.foreco.2023.120821
Bibby CJ, Burgess ND, Hill DA (1992) Bird census techniques. San Diego Academic Press, London
Billerman S, Keeney B, Rodewald P, Schulenberg T (2022) Birds of the World. In: Mobley JA (ed) Comprehensive life histories for all bird species and families. Cornell Lab of Ornithology. Marshall cavendish Corporation, New york
Bisht IS, Rana JC, Yadav R, Ahlawat SP (2020) Mainstreaming agricultural biodiversity in traditional production landscapes for sustainable development: the Indian scenario. Sustainability 12:10690. https://doi.org/10.3390/su122410690
Blumstein DT (2013) Attention, habituation, and antipredator behaviour. In: Gil D, Brumm H (eds) Avian urban ecology, 1st edn. Oxford University Press, Oxford, pp 41–53
Bregman TP, Sekercioglu CH, Tobias JA (2014) Global patterns and predictors of bird species responses to forest fragmentation: implications for ecosystem function and conservation. Biol Conserv 169:372–383. https://doi.org/10.1016/j.biocon.2013.11.024
Breiman L (2001) Random forests. Mach Learn 45:5–32. https://doi.org/10.1023/A:1010933404324
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Calcagno V (2020) glmulti: Model Selection and Multimodel Inference Made Easy. R package version 1.0.8. https://CRAN.R-project.org/package=glmulti
Carrara E, Arroyo-Rodríguez V, Vega-Rivera JH et al (2015) Impact of landscape composition and configuration on forest specialist and generalist bird species in the fragmented Lacandona rainforest, Mexico. Biol Conserv 184:117–126. https://doi.org/10.1016/j.biocon.2015.01.014
Carvalho RL, Andresen E, Arroyo-Rodríguez V et al (2023) Biodiversity in landscape mosaics: the roles of local land use and the surrounding landscape on dung beetle assemblages. J Appl Ecol 60:1647–1658. https://doi.org/10.1111/1365-2664.14440
Chao A, Jost L (2012) Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533–2547. https://doi.org/10.1890/11-1952.1
Chao A, Gotelli NJ, Hsieh TC et al (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67. https://doi.org/10.1890/13-0133.1
Chapman CA, Chapman LJ, Wangham R et al (1992) Estimators of fruit abundance of tropical trees. Biotropica 24:527–531. https://doi.org/10.2307/2389015
Chesser R, Billerman K, Burns K, et al (2023) AOU Checklist of North and Middle American birds. Check-list of North American Birds. https://checklist.americanornithology.org/taxa. Accessed 30 Nov 2023
Cornell Lab of Ornithology (2023) Merlin Bird ID - Free, instant bird identification help and guide for thousands of birds. Cornell Lab of Ornithology. https://merlin.allaboutbirds.org. Accessed 30 Nov 2023
Crawley MJ (2012) The R book. Wiley, New York
Cudney-Valenzuela SJ, Arroyo-Rodríguez V, Morante-Filho JC et al (2023) Tropical forest loss impoverishes arboreal mammal assemblages by increasing tree canopy openness. Ecol Appl 33:e2744. https://doi.org/10.1002/eap.2744
Curtis PG, Slay CM, Harris NL et al (2018) Classifying drivers of global forest loss. Science 361:1108–1111. https://doi.org/10.1126/science.aau3445
De Albuquerque FS, Benito B, Beier P et al (2015) Supporting underrepresented forests in Mesoamerica. Nat Conservacao 13:152–158. https://doi.org/10.1016/j.ncon.2015.02.001
Dent DH, Wright JS (2009) The future of tropical species in secondary forests: a quantitative review. Biol Conserv 142:2833–2843. https://doi.org/10.1016/j.biocon.2009.05.035
Díaz IA, Armesto JJ, Reid S et al (2005) Linking forest structure and composition: avian diversity in successional forests of Chiloé Island, Chile. Biol Conserv 123:91–101. https://doi.org/10.1016/j.biocon.2004.10.011
Dunn RR (2004) Recovery of faunal communities during tropical forest regeneration. Conserv Biol 18:302–309. https://doi.org/10.1111/j.1523-1739.2004.00151.x
Dunning JB, Danielson BJ, Pulliam HR (1992) Ecological processes that affect populations in complex landscapes. Oikos 65:169. https://doi.org/10.2307/3544901
Ellis EA, Romero Montero JA, Hernández Gómez IU (2017) Deforestation processes in the state of Quintana Roo, Mexico: the role of land use and community forestry. Trop Conserv Sci. https://doi.org/10.1177/1940082917697259
Fahrig L (2002) Effect of habitat fragmentation on the extinction threshold: a synthesis. Ecol Appl 12:346–353. https://doi.org/10.2307/3060946
Fahrig L (2013) Rethinking patch size and isolation effects: the habitat amount hypothesis. J Biogeogr 40:1649–1663. https://doi.org/10.1111/jbi.12130
Fahrig L, Baudry J, Brotons L, Burel FG, Crist TO, Fuller RJ, Sirami C, Siriwardena GM, Martin JL (2011) Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol Lett 14:101–112. https://doi.org/10.1111/j.1461-0248.2010.01559.x
FAO (2022) The State of the World’s Forests 2022: Forest pathways for green recovery and building inclusive, resilient and sustainable economies. Food and Agriculture Organization of the United Nations, Rome
Ferreiro-Arias I, Santini L, Sagar HS et al (2024) Drivers and spatial patterns of avian defaunation in tropical forests. Divers Distrib. https://doi.org/10.1111/ddi.13855
Filgueiras BKC, Peres CA, Melo FPL, Leal IR, Tabarelli M (2021) Winner–loser species replacements in human-modified landscapes. Trends Ecol Evol 36:545–555. https://doi.org/10.1016/j.tree.2021.02.006
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd edn. Thousand Oaks, Sage
Gascon C, Lovejoy TE, Bierregaard RO Jr et al (1999) Matrix habitat and species richness in tropical forest remnants. Biol Conserv 91:223–229. https://doi.org/10.1016/S0006-3207(99)00080-4
Gibson L, Lee TM, Koh LP, Brook BW, Gardner BJ, Peres CA, Bradshaw CJA, Laurance WF, Lovejoy TE, Sodhiet NS (2011) Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 478:378–383. https://doi.org/10.1038/nature10425
Global Forest Watch (2024) Yucatán, Mexico deforestation rates and statistics. Global Forest Watch. https://www.globalforestwatch.org/dashboards/country/MEX/31?category=land-cover. Accessed 05 Apr 2024
Gonthier DJ, Ennis KK, Farinas S et al (2014) Biodiversity conservation in agriculture requires a multi-scale approach. Proc R Soc B 281:20141358. https://doi.org/10.1098/rspb.2014.1358
Hanowski J, Danz N, Lind J, Niemi G (2005) Breeding bird response to varying amounts of basal area retention in riparian buffers. J Wild Manage 69:689–698. https://doi.org/10.2193/0022-541X(2005)069[0689:BBRTVA]2.0.CO;2
Haslem A, Bennett AF (2008) Birds in agricultural mosaics: the influence of landscape pattern and countryside heterogeneity. Ecol Appl 18:185–196. https://doi.org/10.1890/07-0692.1
Howell N, Webb S (2010) A guide to the birds of Mexico and northern Central America. Oxford University Press, Oxford
Hsieh TC, Ma KH, Chao A (2022) iNEXT: Interpolation and extrapolation for species diversity. In: R package version 3.0.0. http://chao.stat.nthu.edu.tw/wordpress/software_download/
INEGI (2017a) Anuario estadístico y geográfico de Quintana Roo 2017. Instituto Nacional de Estadística y Geografía, Mexico City
INEGI (2017b) Anuario estadístico y geográfico de Yucatán 2017. Instituto Nacional de Estadística y Geografía, Mexico City
INEGI (2020) Censo de Población y Vivienda 2020. Instituto Nacional de Estadística y Geografía, Mexico City
Jackson HB, Fahrig L (2015) Are ecologists conducting research at the optimal scale? Global Ecol Biogeogr 24:52–63. https://doi.org/10.1111/geb.12233
James FC, Wamer NO (1982) Relationships between temperate forest bird communities and vegetation structure. Ecology 63:159–171. https://doi.org/10.2307/1937041
Jankowski JE, Merkord CL, Rios WF et al (2013) The relationship of tropical bird communities to tree species composition and vegetation structure along an Andean elevational gradient. J Biogeogr 40:950–962. https://doi.org/10.1111/jbi.12041
Jayson EA, Mathew DN (2003) Vertical stratification and its relation to foliage in tropical forest birds in Western Ghats (India). Acta Ornithol 38:111–116. https://doi.org/10.3161/068.038.0207
Jost L (2006) Entropy and diversity. Oikos 113:363–375. https://doi.org/10.1111/j.2006.0030-1299.14714.x
Karr JR, Roth RR (1971) Vegetation structure and avian diversity in several new world areas. Am Nat 105:423–435
Kirk DA, Welsh DA, Baker JA et al (2012) Avian assemblages differ between old-growth and mature white pine forests of Ontario, Canada: a role for supercanopy trees? Avian Conserv Ecol. 7:art4. https://doi.org/10.5751/ACE-00503-070104
LaManna JA, Martin TE (2017) Logging impacts on avian species richness and composition differ across latitudes and foraging and breeding habitat preferences. Biol Rev 92:1657–1674. https://doi.org/10.1111/brv.12300
Lee M, Carroll JP (2018) Effects of patch size and basal area on avian taxonomic and functional diversity in pine forests: Implication for the influence of habitat quality on the species–area relationship. Ecol Evol 8:6909–6920. https://doi.org/10.1002/ece3.4208
Leyequien E, Verrelst J, Slot M et al (2007) Capturing the fugitive: applying remote sensing to terrestrial animal distribution and diversity. Int J Appl Earth Obs 9:1–20. https://doi.org/10.1016/j.jag.2006.08.002
Lynch JF, Whigham DF (1984) Effects of forest fragmentation on breeding bird communities in Maryland, USA. Biol Conserv 28:287–324. https://doi.org/10.1016/0006-3207(84)90039-9
Macchi L, Baumann M, Bluhm H et al (2019) Thresholds in forest bird communities along woody vegetation gradients in the South American Dry Chaco. J Appl Ecol 56:629–639. https://doi.org/10.1111/1365-2664.13342
Macchi L, Decarre J, Goijman AP et al (2020) Trade-offs between biodiversity and agriculture are moving targets in dynamic landscapes. J Appl Ecol 57:2054–2063. https://doi.org/10.1111/1365-2664.13699
Martin LJ, Blossey B, Ellis E (2012) Mapping where ecologists work: biases in the global distribution of terrestrial ecological observations. Front Ecol Environ 10:195–201. https://doi.org/10.1890/110154
Martínez-Ruiz M, Arroyo-Rodríguez V, Arasa-Gisbert R et al (2024) Maintenance of different life stages of old-growth forest trees in deforested tropical landscapes. Ecology. https://doi.org/10.1002/ecy.4273
Mazerolle M (2023) AICcmodavg: Model selection and multimodel inference based on (Q)AIC(c). R package version 2.3.3. https://cran.r-project.org/package=AICcmodavg
Melo FPL, Arroyo-Rodríguez V, Fahrig L et al (2013) On the hope for biodiversity-friendly tropical landscapes. Trends Ecol Evol 28:462–468. https://doi.org/10.1016/j.tree.2013.01.001
Mendenhall CD, Shields-Estrada A, Krishnaswami AJ, Daily GC (2016) Quantifying and sustaining biodiversity in tropical agricultural landscapes. Proc Natl Acad Sci USA 113:14544–14551. https://doi.org/10.1073/pnas.1604981113
Morante-Filho JC, Faria D, Mariano-Neto E, Rhodes J (2015) Birds in anthropogenic landscapes: the responses of ecological groups to forest loss in the Brazilian Atlantic Forest. PLoS ONE 10:e0128923. https://doi.org/10.1371/journal.pone.0128923
Muggeo V (2008) segmented: an R package to fit regression models with broken-line relationships. R News, 8/1, 20–25. https://cran.r-project.org/doc/Rnews/
Naughton-Treves L, Mena JL, Treves A et al (2003) Wildlife survival beyond park boundaries: the impact of slash-and-burn agriculture and hunting on mammals in Tambopata, Peru. Conserv Biol 17:1106–1117. https://doi.org/10.1046/j.1523-1739.2003.02045.x
Nickel BA, Suraci JP, Allen ML, Wilmers CC (2020) Human presence and human footprint have non-equivalent effects on wildlife spatiotemporal habitat use. Biol Conserv 241:108383. https://doi.org/10.1016/j.biocon.2019.108383
O’brien RM, (2007) A caution regarding rules of thumb for variance inflation factors. Qual Quant 41:673–690. https://doi.org/10.1007/s11135-006-9018-6
Pacheco P, Mo K, Dudley N, Shapiro A, Aguilar-Amuchastegui N, Ling PY, Anderson C, Marx A (2021) Frentes de deforestación: Causas y respuestas en un mundo cambiante. World Wildlife Fund, Gland
Pal M (2005) Random forest classifier for remote sensing classification. Int J Remote Sens 26:217–222. https://doi.org/10.1080/01431160412331269698
Pautasso M (2007) Scale dependence of the correlation between human population presence and vertebrate and plant species richness. Ecol Lett 10:16–24. https://doi.org/10.1111/j.1461-0248.2006.00993.x
Pedroso N, Adams C, Rui M (2009) Slash-and-burn agriculture: a system in transformation. In: Lopes P, Begossi A (eds) Current trends in human ecology. Cambridge Scholars Publishing, Newcastle upon Tyne
Phalan BT (2018) What have we learned from the land sparing-sharing model. Sustainability 10:1760. https://doi.org/10.3390/su10061760
Price M (2008) The impact of human disturbance on birds: a selective review. In: Lunney D, Munn A, Meikle W (eds) Too close for comfort: contentious issues in human-wildlife encounters. Royal Zoological Society of New South Wales, Sidney
Rapinel S, Mony C, Lecoq L et al (2019) Evaluation of sentinel-2 time-series for mapping floodplain grassland plant communities. Remote Sens Environ 223:115–129. https://doi.org/10.1016/j.rse.2019.01.018
Rempel RS, Kaukinen D, Carr AP (2012) Patch analyst and patch grid. Centre for Northern Forest Ecosystem Research, Ontario
Ripple WJ, Wolf C, Newsome TM et al (2017) World scientists’ warning to humanity: a second notice. Bioscience 67:1026–1028. https://doi.org/10.1093/biosci/bix125
Rockström J, Steffen W, Noone K et al (2009) A safe operating space for humanity. Nature 461:472–475. https://doi.org/10.1038/461472a
Russildi G, Arroyo-Rodríguez V, Hernandez-Ordõnez O, Pineda E, Reynoso VH (2016) Species- and community-level responses to habitat spatial changes in fragmented rainforests: assessing compensatory dynamics in amphibians and reptiles. Biodivers Conserv 25:375–392. https://doi.org/10.1007/s10531-016-1056-3
Santamaria-Rivero W, Leyequien E, Hernandez-Stefanoni JL, Wood P (2016) Influence of landscape structure and forest age on the richness and abundance of different bird feeding guilds and forest-dependent birds in a seasonal dry tropical forest of Yucatan, Mexico. Trop Ecol 57:313–332
Scabin AB, Peres CA (2021) Hunting pressure modulates the composition and size structure of terrestrial and arboreal vertebrates in Amazonian forests. Biodivers Conserv 30:3613–3632. https://doi.org/10.1007/s10531-021-02266-9
Smith AL, Ortiz JS, Robertson RJ (2001) Distribution patterns of migrant and resident birds in successional forests of the Yucatan Peninsula, Mexico. Biotropica 33:153–170. https://doi.org/10.1111/j.1744-7429.2001.tb00165.x
Stotz DF, Fitzpatrick JW, Parker TA, Moskovits DK (1996) Neotropical birds: ecology and conservation. University of Chicago Press, Chicago
Supp SA, Ernest AKM (2014) Species-level and community-level responses to disturbance: a cross-community analysis. Ecology 95:1717–1723. https://doi.org/10.1890/13-2250.1
Swift TL, Hannon SJ (2010) Critical thresholds associated with habitat loss: a review of the concepts, evidence, and applications. Biol Rev 85:35–53. https://doi.org/10.1111/j.1469-185X.2009.00093.x
Terán S, Rasmussen C (2009) La milpa de los mayas: la agricultura de los mayas prehispánicos y actuales en el noreste de Yucatán. Universidad Nacional Autónoma de México, Ciudad de México
Terborgh JW (1987) Habitat selection in Amazonian birds. In: Cody M (ed) Habitat selection in birds, 1st edn. Academic Press, London, pp 311–338
Terraube J, Archaux F, Deconchat M et al (2016) Forest edges have high conservation value for bird communities in mosaic landscapes. Ecol Evol 6:5178–5189. https://doi.org/10.1002/ece3.2273
Thiollay J-M (1999) Responses of an avian community to rain forest degradation. Biodivers Conserv 8:513–534. https://doi.org/10.1023/A:1008912416587
Tscharntke T, Klein AM, Kruess A et al (2005) Landscape perspectives on agricultural intensification and biodiversity—ecosystem service management. Ecol Lett 8:857–874. https://doi.org/10.1111/j.1461-0248.2005.00782.x
Tscharntke T, Tylianakis JM, Rand TA et al (2012) Landscape moderation of biodiversity patterns and processes—eight hypotheses. Biol Rev 87:661–685. https://doi.org/10.1111/j.1469-185X.2011.00216.x
Urquiza-Haas T, Peres CA, Dolman PM (2011) Large vertebrate responses to forest cover and hunting pressure in communal landholdings and protected areas of the Yucatan Peninsula, Mexico. Anim Conserv 14:271–282. https://doi.org/10.1111/j.1469-1795.2010.00426.x
Van Perlo B (2006) Birds of Mexico and Central America. Princeton University Press, Princeton
Vargas-Cárdenas F, Arroyo-Rodríguez V, Morante-Filho JC et al (2022) Landscape forest loss decreases bird diversity with strong negative impacts on forest species in a mountain region. Persp Ecol Conserv 20:386–393. https://doi.org/10.1016/j.pecon.2022.10.001
Vargas-Daza AM, Betancurt-Grisales JF, Castaño-Villa GJ, Fontúrbel FE (2023) The importance of old secondary forests for understory birds in the tropical Andes. Global Ecol Conserv 47:e02658. https://doi.org/10.1016/j.gecco.2023.e02658
Watling JI, Arroyo-Rodríguez V, Pfeifer M et al (2020) Support for the habitat amount hypothesis from a global synthesis of species density studies. Ecol Lett 23:674–681. https://doi.org/10.1111/ele.13471
Xeno-canto (2023) Xeno-canto: sharing wildlife sounds from around the world. https://xeno-canto.org/. Accessed 30 Nov 2023
Yang S, Li C, Lou H et al (2021) Role of the countryside landscapes for sustaining biodiversity in karst areas at a semi centennial scale. Ecol Ind 123:107315. https://doi.org/10.1016/j.ecolind.2020.107315
Acknowledgements
This paper serves as a fulfillment of ALMP for obtaining a M.Sc. degree in the Posgrado en Ciencias Biológicas (Ecology field of knowledge) UNAM. We are indebted to don Pedro Poot Cupul for his invaluable field assistance. We also thank Severo Pool Caamal, Vidal Mazum, Gal Moas, the staff of the Yaakunah K’aax Reserve, and landowners from Buenavista, Cocoyol, and Palomares for allowing us to collect data on their properties and for their help in the field.
Funding
We thank the Consejo Nacional de Humanidades, Ciencia y Tecnología of Mexico (CONAHCYT) for funding and supporting this research through a graduate scholarship to ALMP. This research was funded by the Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT), DGAPA-UNAM (IN-206123). ALMP obtained a thesis research grant from the François Vuilleumier Fund (Neotropical Ornithological Society).
Author information
Authors and Affiliations
Contributions
VAR and ALMP developed the idea of the study, with support from JCMF and JS. VAR obtained the financial support. ALMP collected and analyzed the data with guidance from all coauthors. EJPR digitized the map and calculated the landscape variable. All authors made substantial contributions to the intellectual content, interpretation and editing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martínez-Penados, A.L., Arroyo-Rodríguez, V., Morante-Filho, J.C. et al. Old-growth forests are critical to safeguard tropical birds in complex landscape mosaics exposed to slash-and-burn agriculture. Landsc Ecol 39, 118 (2024). https://doi.org/10.1007/s10980-024-01919-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10980-024-01919-8