Abstract
Context
Large carnivores have faced severe extinction pressures throughout Europe during the last centuries, where human-induced disturbances reached unprecedented levels. In the late twentieth century, the Cantabrian brown bear population was on the verge of extinction, due to poaching. Yet, the end of the last century was a turning point for this population. Presence data on the western Cantabrian subpopulation was collected since the beginning of the century and insights provided by this long-term monitoring may be useful for brown bear conservation.
Objectives
Here, we aim to: (i) identify the landscape features relevant to bears’ recovery; and (ii) understand if and how the landscape use patterns by bears changed over time.
Methods
We tested the influence of landscape structure (i.e., composition and configuration) on bear occurrence patterns using MAXENT in three periods representative of land cover change.
Results
Despite variation across the 19-year monitoring period, brown bears were more often detected near broad-leaf forests and bare rock areas and at lower to intermediate altitudes, but avoided arable lands, permanent crops, and burnt areas. Human population density or distance to roads—often used for modelling habitat suitability for Cantabrian brown bears—were not identified as relevant variables for this brown bear subpopulation. Artificial areas were identified as relevant landscape features, but not as disturbance.
Conclusions
These findings reinforce the importance of preserving bears’ native habitats and provide new insights, namely on the use of humanized landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Humans have been critical in the decline of many European large carnivore populations over the past centuries (Ripple et al. 2014). Habitat loss, fragmentation and direct persecution have been the most noticeable impacts of human activities (Ray, Hunter, & Zigouris, 2005). During the twentieth century, suitable habitats for large mammals, such as the European brown bear (Ursus arctos), wolf (Canis lupus), wolverine (Gulo gulo), Iberian (Lynx pardinus) and Eurasian lynx (L. lynx), have become scarcer, scattered, and more disconnected (Swenson et al. 2000; Chapron et al. 2014). Human direct persecution, is also an important driver of carnivore population decline and contraction of distribution ranges (Ray et al. 2005; Ripple et al. 2014). This is the case of the brown bear, which was almost extirpated from Western Europe, and suffered a substantial decline in Eastern and Northern Europe (Penteriani and Melletti 2020; Wiegand et al. 1998). As a result, their survival relied on a delicate balance between resource exploitation on a humanized landscape (Clevenger et al. 1987) and seeking refuge from human-induced mortality (Naves et al. 2003).
Swenson et al (2000) reported twelve extant brown bear populations; from these, six were distributed in Western Europe and considered very small (with less than 100 individuals). The critical status of these six populations was aggravated by their isolation from possible source and stable populations (Swenson et al. 2000; Zedrosser et al. 2001; Pérez et al. 2010). Small and isolated populations are more vulnerable to extinction as a consequence of stochastic events (Zedrosser et al. 2011). In Spain, two of these small populations remain—in the Cantabrian and the Pyrenean mountains. If the Pyrenean brown bear population was reinforced with translocated individuals (Parres et al. 2020), the Cantabrian population (consisting of two spatially segregated nuclei, Western and Eastern of the Cantabrian Mountains) survived a near-extinction scenario during the late twentieth century, without any direct conservation action to increase the population size, being considered one of the most endangered bear populations in the world (Méndez et al. 2014; Penteriani et al. 2020). This population was threatened by poaching, increased landscape and genetic fragmentation, and lack of connectivity (Méndez et al. 2014; Penteriani et al. 2020).
The beginning of the twenty-first century was a turning point for the Cantabrian population, particularly for the Western population nuclei (Méndez et al. 2014; Gonzalez et al. 2016), where a consistent increase in the number of females with cubs was detected between 2003 and 2014 (Gonzalez et al. 2016; Lamamy et al. 2019) associated with an increase in the area of occurrence in both nuclei (Díaz-Fernández et al. 2023). Bidirectional migration and gene flow were reported between Western and Eastern Cantabrian mountains, with earlier reports of a dominant eastward migration (Pérez et al. 2010; Gonzalez et al. 2016) and, more recently, a dominant westward migration pattern (Gregório et al. 2020). While it is not clear if habitat differences are the single driver of population and migration patterns in the Cantabrian brown bear (Naves et al. 2003; Lamamy et al. 2019), identifying the most suitable habitat during the recovery of the western subpopulation may provide valuable information to the future conservation, not only of this subpopulation, but to the population of the Cantabrian bear as a whole.
Bearing this in mind, our goal is to use brown bear presence records available for a 19-year period (2000–2018) to test: (i) the relevance of distinct landscape structure (i.e. composition—type of landscape patches—and configuration—spatial characteristics of patches) (Farina 2000) in explaining bear occurrence patterns in the western Cantabrian brown bear subpopulation; and (ii) whether and—if so—how landscape composition relevance changed over 19 years, a period during which bear population size and distribution range increased in this nucleus. It is often assumed in studies focused on identifying suitable areas for brown bears in the Cantabrian Mountains that this species tends to avoid humanized elements (roads, settlements) of the landscape, as well as more densely populated areas (Clevenger et al. 1990; Naves et al. 2003; Mateo-Sánchez et al. 2016). Therefore, we hypothesize that the brown bears in the study area tend to occur in areas more distant from these humanized elements of the landscape. On the other hand, given the historical presence of humans and the brown bear in Europe (Zedrosser et al. 2011), the Cantabrian brown bears may have evolved to balance the exploitation of resources and the search for refuge, despite the human presence in the landscape (Ordiz et al. 2011). For this reason, we hypothesise that this species may favour the use of landscape features associated with refuge and food resources over the avoidance of features related to human disturbance (Fernandéz et al. 2012). Finally, because individuals may use habitat differently during different stages of population expansion (Wydeven et al. 2009; Clavero et al. 2010; Hemmingmoore et al. 2020)—and given both the Cantabrian brown bear population expansion (Palomero et al. 2007; Gonzalez et al. 2016; Lamamy et al. 2019) and the long period analysed—we hypothesize the factors that most conditioned the presence of the brown bear in the region may have varied over the last 19 years.
Methods
Study area
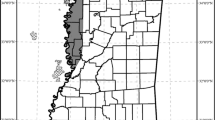
The Cantabrian Mountains run parallel to the Atlantic coast of northern Spain and exhibit a mild and humid climate throughout the year (Ortega Villazán and Morales Rodríguez 2015). The mountains extend for 300km, throughout the provinces of Asturias, Cantabria, León, Palencia, and Galicia and occupy an area of approximately 18.000 km2 (Clevenger et al. 1990).
The mountain range is covered by forests and woodlands mostly composed of oaks (Quercus petraea, Q. pyrenaica, Q. rotundifolia), beech (Fagus sylvatica), birch (Betula celtiberica), holly (Ilex aquifolium), and ash (Sorbus spp.) (Díaz González and Penas 2017). Subalpine shrubland (Juniperus communis, Vaccinium uliginosum, V. myrtillus, Arctostaphylos uva-ursi) dominates mountain areas above the treeline (Díaz González and Penas 2017). In areas converted into pastures, is frequent the occurrence of Genista spp., Cytisus spp., Erica spp., and Calluna spp. (Naves et al. 2003, 2006).
The main economic activities across the bear’s range are agriculture, livestock farming, timber harvesting, mining, and tourism (Naves et al. 2003; Blanco-Fontao et al. 2011). Human population and road density are generally low within the brown bear range in this region (Clevenger et al. 1990; Naves et al. 2003). Anthropic landscape modifications in the last four centuries have altered the composition and distribution of the native flora and fauna in this area, with hardwood forests being converted into pastures for livestock grazing (Clevenger et al. 1990). In more recent years, woody vegetation increased in semi-natural open systems and agriculture expanded in the most suitable areas (García-Llamas et al. 2019).
Data collection
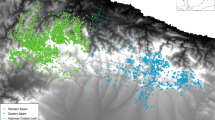
Brown bear presence data were collected in an area of approximately 3.500 km2, within the range of the western Cantabrian Mountains brown bear subpopulation range (Fig. 1). Bear presence data collection in the study area was done by FAPAS (Fondo para la Protección de los Animales Salvajes; Spain, by the same two observers throughout the whole period), under a long-term monitoring program through indirect methods (signs of presence, such as tracks, denning sites, scats and marked trees), by monitoring 97 transects every year, from 2000 to 2018 (Figure A.1). Sampling effort was not systematically recorded but has been increasing over the years, accompanying brown bear expansion—targeting more intensively the areas of known bear distribution and of likely expansion and the seasons of higher bear activity (e.g., there were more frequent surveys in spring than winter).
A wide diversity of signs was consistently recorded throughout the years, totalizing 7179 presence signs found in 19 years. The number of signs ranged between 115—588 annually (averaging 378 ± 159 signs detected per year). Bear presence records comprised bear footprints (39.7% of the data); marked trees (32.5% of the data); scats (23.5% of the data); hive attacks (1.7% of the data); and denning sites (0.57% of the data). Many signs were found in combination, in the same location and date. In these cases, different signs were combined in a single presence point.
Variables were selected to be representative of three groups of predictors identified as highly influential in shaping the bear distribution, and were related to three main ecological drivers of presence: disturbance (Naves et al. 2003; Mateo-Sánchez et al. 2016); food resources (Clevenger et al. 1990; Mateo-Sánchez et al. 2016); and refuge (Naves et al. 2003; Mateo-Sánchez et al. 2016) (Table 1). Altitude was also considered a likely predictor of brown bear presence in all three groups of predictor variables, given that: (i) human settlements are often at lower altitudes; (ii) food resources are more often abundant at lower (when provided by human activities, such as agriculture) and intermediate altitudes (Posillico et al. 2004); and (iii) refuge (i.e., areas inaccessible to humans and/or natural areas rich in vegetation that provides cover) is often found at intermediate to higher altitudes. Thus, we considered that the preference for different altitudinal ranges would provide support to a more important role of each of the ecological drivers, depending on the resulting altitudinal values for which bears’ occurrence probability is higher, in case this predictor emerges as relevant. The same approach was used for the landscape composition, since it may be an important driver of habitat selection (Mateo Sánchez et al. 2014). All variables were defined according to the source terminology and the respective concepts are summarised in Table A.1.
As presence data was collected throughout almost two decades, ecological modelling was performed using the environmental data that temporally better matched each presence data period, to overcome any bias linked to the interannual variation of those variables. Given the wide timeframe of data collection, to account for land cover variation and more accurately model bear presence, we grouped presence data by year periods that best represented the land cover changes over the years. As a result, for each set of years (2000–2010; 2011–2016; 2017–2018) different versions of the Corine Land Cover were used (2006, 2012 and 2018 versions, respectively). Similarly, for the population density, we used the 2006 version to characterize the first set of years (2000–2010), and the 2011 version to characterize the remaining set of years (2011–2016 and 2017–2018). For other variables, such as “Altitude” or “Distance to roads”, we used the most updated information available (Tables A.2 and A.3).
Data analysis
Prior to the model building, the selected variables were tested to assess multicollinearity. We considered that none of the variables was strongly correlated as the pairwise correlation Pearson coefficients were all lower than |0.7| (Dormann et al. 2007).
Due to the lack of standardization in data collection in time and space, we considered that the most robust approach for modelling bear presence was to use a presence-only method, based on a Maximum Entropy approach, through a simple general-purpose machine learning method (Phillips et al. 2006). This approach was implemented using the MAXENT software (Version 3.4.1) (Phillips et al. 2006), which models species geographic distribution with presence-only data and typically outperforms other methods based on predictive accuracy (Phillips et al. 2006; Merow et al. 2013). To further account for the sampling bias and reduce the spatial aggregation of detection points (Fourcade et al. 2014), the annual data was overlapped with a 100 m resolution grid (used as the sampling unit), and the cells with at least one bear sign were considered as a bear presence cell in a particular year. For each presence cell, the centroid coordinate was retrieved, which was used simultaneously to calculate the distance to the different landscape components used as configuration metrics in the models, and as the presence points used in the model analysis.
Analyses were two-folded. First, we analysed data per year and later we grouped the presence data into three consecutive and contiguous periods and replicated the analyses. Thus, nineteen models were generated (one run for each year of data collected). These were used to compare the evolution of the Cantabrian brown bear western subpopulation distribution over the 19 years of the study, its most important drivers, and to detail the variation within three multi-year periods. Second, three multi-year models were generated, corresponding to the previously grouped data: 2000–2010; 2011–2016; 2017–2018 (one run for each group). The annual models are included as supplementary information and we will base our main discussion on the multi-year trends.
For each generated model, MAXENT extracted 10,000 background points. This is large enough to represent the distribution of environmental conditions in the study area and assure increased predictive performance (Phillips and Dudík, 2008). Parameter settings were left to Maxent’s default (Table A.4), as Phillips and Dudík (2008) suggested these are well suited for a wide range of presence-only datasets. The evaluation of the models was based on 30-fold cross-validation and, hence, the fit of each model to the presence data distribution was assessed using the AUC (area under the ROC curve) (Merow et al. 2013). For each model, we weighted each variable contribution (i.e., a measure of the amount of model variation each variable explains) (Bradie and Leung 2017) and permutation importance (i.e., a measure of the dependence of the model on each variable) (Breiman 2001). These parameters were used to calculate the average, maximum, and minimum variable contribution over time, and respective permutation importance. We established a threshold to select the most relevant variables (i.e., the variables that contribute the most to explaining bear occurrence probability). The variables contributing, on average, more than 5% to the multi-year models, and more than 10% at least in one of these periods were used further for comparison and discussion.
Finally, three habitat suitability projections were generated in MAXENT (one for each multi-year model). Hence, habitat suitability was compared over time, by performing raster calculations between these three projections. The variation in habitat suitability was assessed for the entire 19-year period (2000–2018, as the variation between the first and last projection) and between the intermediate projection (2011–2016) and the two contiguous models.
Results
None of the model’s candidate covariables was correlated, and therefore all were used in the modelling procedure. All generated models produced consistently high AUC values (AUC > 0.7), indicating a high performance in discriminating presence points from background points (Manel et al. 2001). While the AUC value of the multi-year models was 0.97 for all three periods (AUC[2000–2010]) = 0.9693; AUC[2011–2016]) = 0.9676; AUC[2017–2018]) = 0.9712), the annual models varied between 0.75, in 2000, and 0.99, in 2002 (Table B.1). As the model produced for the year 2000 showed a relatively lower AUC value than that of all the other annual models (AUC > 0.97), this model was not included in the calculation of the most significant variables. Reporting of results and discussion will proceed based on the multi-year models’ results. Annual models may be consulted in the supplementary material (Appendix B).
Relevant landscape drivers
We identified eight environmental variables (broad-leaf forests, arable lands, bare rocks, inland wetlands, altitude, permanent crops, artificial areas, and burnt areas) that contributed: (i) on average, more than 5% to the multi-year models; and (ii) 10% or more at least in one of the three periods (9.6% and 9.9% in the case of the distance to permanent crops and altitude, respectively). Despite some variation across the 19-year period, five landscape features had a consistent role in explaining brown bear presence patterns (percent contribution always above 5% in all multi-year models): broad-leaf forests; arable lands; bare rocks; inland wetlands; and altitude (Tables 2 and A.5).
Brown bears were more often detected near broad-leaf forests and areas with bare rocks and at lower to intermediate altitudes. Conversely, they avoided arable lands, permanent crops, and burnt areas. In addition, two variables (inland wetlands and artificial areas) showed non-linear relations with bears’ presence probability: bears select intermediate distances to these landscape features (Fig. 2).
Top variables’ effects across the three multi-year models (broad-leaf forests, arable lands, bare rocks, inland wetlands, altitude, permanent crops, artificial areas, and burnt areas). The average of the effects over time is represented on the left of the respective effects plot, with a black line. The cloglog output on the y-axis gives the probability of occurrence between 0 and 1
Variables’ contribution over time
Overall, the distance to broadleaf forests is the variable that contributed the most to explaining bear presence probability (an average of 20%), except during the first 10 years of monitoring, when the distance to bare rocks was more important (18% during 2000–2010). The main changes from the 1st to the 2nd period include the contribution increase of the distance to broad leaf forests (+ 12%), but also the decrease of the contribution of the distance of artificial areas (− 7%), which seem to play a less important role over time. During this trend, an inversion of the effect of this variable occurred: bears shifted from selecting closer distances to artificial areas to occurring further from these areas. From the 2nd to 3rd period, the most significant changes include an increase in the distance to burnt areas contribution to the models (+ 10%) and a decrease in the distance to bare rocks (− 6%) (Fig. 3 and Table A.6).
Predicted suitable areas for bear presence and changes in habitat suitability
The MAXENT output generated a prediction of the most suitable areas for bear occurrence in each of the three monitoring periods (Fig. 4), which were used to calculate the evolution in habitat suitability over time. From the first period (2000–2010) to the second (2011–2016), there was a general increase in the areas with suitable habitats. However, habitat suitability apparently decreased in the most recent years (2017–2018) – a result we should interpret with caution, as this period is relatively shorter and includes fewer presence records (Fig. 5).
Evolution of bear habitat suitability over the 19-year period in the Cantabrian mountains. Raster calculation derived from the MAXENT projection for each multi-year model. The variation in habitat suitability was assessed for the entire 19-year period (2000–2018, as the variation between the first and last projection) and between the intermediate projection (2011–2016) and the two contiguous models
Discussion
Recovery of large carnivores throughout Europe was anchored on the change of conservation laws and policies, to facilitate coexistence and improve population status, in combination with conservation actions focused on habitat management (Chapron et al. 2014), which rely on solid knowledge about habitat use by these species. Here, taking advantage of a large dataset of presence records of an endangered brown bear population, we identified the most relevant landscape features for the western Cantabrian nuclei throughout a 19-year period. We highlighted variations in drivers’ importance that can help understand bear population dynamics since the early stages of the brown bear recovery in the Western Cantabrian region. All the models present high performance, suggesting they could effectively distinguish the presence records from the background points. Thus, we were able to provide relevant insights, derived from a long-term monitoring programme targeting brown bear conservation.
In line with previous studies, we confirmed that Cantabrian brown bears are adapted to human presence, balancing their dietary needs with the safety provided by the available habitat in the Western Cantabrian region (Lamamy et al. 2019; Zarzo-Arias et al. 2019; Pratzer et al. 2023). We were able to identify a handful of habitat features that had a major role in shaping brown bear distribution for the last two decades: broad-leaf forests, arable lands, bare rocks, inland waters and altitude. In earlier years of monitoring, bear presence in the close vicinity of artificial areas was very frequent and decreased over time, while in most recent years the burnt areas might have been an important constraint to brown bear distribution in the Cantabrian mountains, as will be discussed in more detail ahead.
Living in human-dominated areas might drive species to establish a delicate balance between finding refuge or food resources. According to our results, broad-leaf forests promote bears’ presence throughout the years, which might be linked to the species' resource needs (e.g., food and, to a smaller extent, refuge) during most of the year (Lamamy et al. 2019; Zarzo-Arias et al. 2019). In winter, some individuals might avoid hibernation and remain active during this cold season, a behaviour that can derive from the mild winters of the region that allow animals to continue using and finding food, in forested areas (Swenson et al. 2000). This is also confirmed by a total of 806 presence records (11% of the total sample size) encountered consistently during the 19 winter seasons—every year, frequent brown bear detections were recorded between December and February. Bare rocks offer important refuge areas since caves or natural cavities are the structures most used as den sites in the Cantabrian Mountains (Naves and Palomero 1993), which reinforces the detected importance of such areas for bears throughout the monitored period. Bare rocks are usually located in areas that are less accessible to humans and that have more often higher snow cover, fitting the necessary conditions for undisturbed hibernation periods (Evans et al. 2016; Chirichella et al. 2019; Lamamy et al. 2019; González-Bernardo et al. 2020). Den disturbance can lead to the premature ending of hibernation, which can exponentially increase cub mortality (Naves and Palomero 1993; Linnell et al. 2001; González-Bernardo et al. 2020).
However, our results also show that bears tend to prefer lower to intermediate altitudes, where conditions are more adequate for finding food resources, and human presence is more abundant, a pattern that contrasts with what was described, for example, in the Italian population (Posillico et al. 2004). These results suggest that habitat selection by bears in the Cantabrian mountains is dependent upon a complex dynamic between accessing natural food resources (in broad-leaved forests or, in earlier years, in the close surroundings of humanized areas—here represented by artificial areas—most important drivers of this population occurrence. These results confirm our initial hypothesis that the Cantabrian brown bears favour landscape features associated with refuge and food resources rather than avoiding human disturbance, in a landscape that has been shaped by humans for centuries.
Lower humidity and precipitation levels during the Cantabrian mountains’ summer period (Lamamy et al. 2019) might contribute to the selection of areas at intermediate distances to inland water areas by brown bears. However, unlike in other regions of the species’ distribution (Ansari and Ghoddousi 2018), our results suggest that the Cantabrian brown bears are not heavily constrained by the distance to water bodies. Moreover, wetlands may also be associated with important sources of food, as European brown bear populations, including the Cantabrian brown bear, have an omnivorous diet, dominated by plant material (e.g., berries and fruits) (Cicnjak et al. 1987; Clevenger et al. 1992; Naves et al. 2006).
Although they show this frugivorous behaviour, in the earlier monitoring years, our results show avoidance of large agricultural areas (i.e., permanent crops and arable lands) contrasting a higher occurrence near human settlements (i.e., artificial areas). While permanent crops and arable lands can provide important food resources (e.g., grains, grass, and fruits), these results are consistent with bears avoiding intensively managed agricultural areas while exploiting small crops and fruit trees, which are often located within the vicinity of settlements (Viñas 2010)—and not explicitly represented in our models due to their small size (though being associated to artificialized areas). This increased risk of exposure to humans in areas rich in food resources (Zajec et al. 2005; Martin et al. 2012; Lamamy et al. 2019) seems to rule the non-linear effect of artificialized areas (e.g., settlements) highlighted by our models’ results, since, even in later years, several presence records were registered in areas at close to intermediate distances to settlements. Regardless, artificial areas seemed to lose relevance over time, in brown bears' occurrence patterns, accompanied by the displacement out of these areas. This trend is consistent with the decrease in the dependence on orchards close to the villages over time. Not only the abundance of this food resource has decreased due to the abandonment of villages, but also the availability of natural food resources improved as natural forests recovered, both due to rural exodus (MacDonald et al. 2000). This confirms our hypothesis that the factors that most conditioned the presence of the brown bear in the region have varied over the last 19 years, in light of the inversion in the population trend—from decline to population expansion.
The contributions of permanent crops and inland wetlands have also decreased over time. In the case of permanent crops, this decrease was accompanied by the bear's occurrence optima starting to appear at smaller distances over time, with the opposite trend for inland wetlands. Both these variables occur only in the periphery of our presence records but given the current expansion of the population distribution range, we advise that, as bears get closer to these landscape features, this apparent avoidance pattern should be monitored in more detail.
We did not find enough evidence to support our initial hypothesis that brown bears in the study area tend to occur in areas more distant from humanized elements of the landscape. Particularly, human population density and distance to roads were not identified as relevant ecological drivers representative of disturbances, contrasting previous studies (Clevenger et al. 1990; Naves et al. 2003; Mateo-Sánchez et al. 2016). Additionally, given the previously discussed role of artificial areas, this scenario reinforces the hypothesis that large carnivore conservation should consider the complementary role of anthropic systems (e.g., agriculture patches instead of intensively managed agricultural areas), as long as the conditions for human-wildlife coexistence are guaranteed (López-Bao et al. 2015; Chapron and López-Bao 2016), such as the availability of refuge (e.g., bare rocks).
This analysis provides critical information to policymakers and managers to pursue effective conservation actions for this species, particularly for the Cantabrian mountains population. Namely, our results reinforce the importance of preserving bears’ native habitats (most importantly broadleaf forests). Future conservation actions must also address the challenge of preventing fire events, which has recently become a problem for this population, possibly due to the increase in intentionally burned areas for pasture and game management. This may become an even greater threat if burned areas encroach into brown bear’s distribution range, particularly into areas of reproduction. Our results show that bears appear not to avoid humanized elements of the landscape and occupy intermediate altitudes. Thus, we are confident that, instead of a generalized avoidance of human presence, bears might perceive only certain anthropogenic activities as disturbances and might have adapted their behaviour to coexistence. This view is supported by the avoidance of intensively managed agricultural areas. Therefore, we suggest that alternative proxies of disturbance—such as quantitative and/or spatial proxies to specific human activities—such as poaching or touristic pressure—should be integrated into future ecological modelling analyses. Additionally, using smaller scales and individual tracking, both temporally and spatially, is of utmost importance to gain relevant insights into the segregation of bears with human presence and activities at specific times of significant disturbance.
Human-dominated landscapes are highly dynamic, and therefore long-term studies are crucial, since different stages in the recovery of wildlife populations in social-ecological systems may require different habitat characteristics and management strategies. Therefore, a far-reaching approach, such as long-term monitoring, is mandatory to understand the dynamics of landscape change and the species’ response to those alterations.
Data availability
Raw data used in this manuscript pertains to georeferenced presence locations of an endangered and highly persecuted brown bear population. Therefore, making raw location data available might pose serious risk to this brown bear population. Approximate location data (annual presence points in a 100 m resolution grid), used as sampling units for the analysis conducted in this paper are available upon request to the authors.
References
Ansari HM, Ghoddousi A (2018) Water availability limits brown bear distribution at the southern edge of its global range. Ursus 29(1):13–24
Blanco-Fontao B, Quevedo M, Obeso JR (2011) Abandonment of traditional uses in mountain areas: typological thinking versus hard data in the Cantabrian Mountains (NW Spain). Biodivers Conserv 20(5):1133–1140
Bradie J, Leung B (2017) A quantitative synthesis of the importance of variables used in MaxEnt species distribution models. J Biogeogr 44(6):1344–1361
Breiman L (2001) Random forests. Mach Learn 45(1):5–32
Chapron G, López-Bao JV (2016) Coexistence with large carnivores informed by community ecology. Trends Ecol Evol 31(8):578–580
Chapron G, Kaczensky P, Linnell JDC, von Arx M, Huber D, Andrén H, López-Bao JV, Adamec M, Álvares F, Anders O, Balčiauskas L, Balys V, Bedő P, Bego F, Blanco JC, Breitenmoser U, Brøseth H, Bufka L, Bunikyte R, Ciucci P, Dutsov A, Engleder T, Fuxjäger C, Groff C, Holmala K, Hoxha B, Iliopoulos Y, Ionescu O, Jeremić J, Jerina K, Kluth G, Knauer F, Kojola I, Kos I, Krofel M, Kubala J, Kunovac S, Kusak J, Kutal M, Liberg O, Majić A, Männil P, Manz R, Marboutin E, Marucco F, Melovski D, Mersini K, Mertzanis Y, Mysłajek RW, Nowak S, Odden J, Ozolins J, Palomero G, Paunović M, Persson J, Potočnik H, Quenette P-Y, Rauer G, Reinhardt I, Rigg R, Ryser A, Salvatori V, Skrbinšek T, Stojanov A, Swenson JE, Szemethy L, Trajçe A, Tsingarska-Sedefcheva E, Váňa M, Veeroja R, Wabakken P, Wölfl M, Wölfl S, Zimmermann F, Zlatanova D, Boitani L (2014) Recovery of large carnivores in Europe’s modern human-dominated landscapes. Science 346(6216):1517–1519
Chirichella R, Mustoni A, Zibordi F, Armanini M, Caliari A, Apollonio M (2019) Rent a room in the Alps: winter den site preferences of native and reintroduced brown bears. Mamm Res 64(2):213–222
Cicnjak L, Huber D, Roth HU, Ruff RL, Vinovrski Z (1987) Food habits of brown bears in Plitvice lakes national park Yugoslavia. Bears: Their Biolo Manag 7:221–226
Clavero M, Hermoso V, Brotons L, Delibes M (2010) Natural, human and spatial constraints to expanding populations of otters in the Iberian Peninsula. J Biogeogr 37(12):2345–2357
Clevenger AP, Purroy FJ, de Buruaga MS, Purdoy F, de Burguaga MS (1987) status of the brown bear in the Cantabrian Mountains Spain. Bears: Their Biol Manag 7:1–8
Clevenger AP, Purroy FJ, Pelton MR (1990) Movement and activity patterns of a european brown bear in the Cantabrian Mountains Spain. Bears: Their Biol Manag 8:205–211
Clevenger AP, Purroy FJ, Pelton MR (1992) Food Habits of Brown Bears (Ursus arctos) in the Cantabrian Mountains Spain. J Mamm 73(2):415–421
Díaz González TE, Penas Á (2017) The high mountain area of Northwestern Spain: the cantabrian range, the Galician-Leonese mountains and the bierzo trench. In: Loidi J (ed) The vegetation of the iberian peninsula, vol 1. Springer International Publishing, Cham, pp 251–321
Díaz‐Fernádez M, Naves J, Revilla E (2023) Conservation implications of range dynamics in endangered populations: an example with brown bears. Conser Sci Pract 5(3):e12894. https://doi.org/10.1111/csp2.12894
Dormann CF, McPherson JM, Araújo MB, Bivand R, Bolliger J, Carl G, Davies RG, Hirzel A, Walter Jetz W, Kissling D, Kühn I, Ohlemüller R, Peres-Neto PR, Reineking B, Schröder B, Schurr FM, Wilson R (2007) Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography 30(5):609–628
Evans AL, Singh NJ, Friebe A, Arnemo JM, Laske TG, Fröbert O, Swenson JE, Blanc S (2016) Drivers of hibernation in the brown bear. Front Zool 13(1):7
Farina A (2000) Landscape ecology in action kluwer academic. The Netherlands, Dordrecht
Fernández N, Selva N, Yuste C, Okarma H, Jakubiec Z (2012) Brown bears at the edge: modeling habitat constrains at the periphery of the Carpathian population. Biol Conserv 153:134–142. https://doi.org/10.1016/j.biocon.2012.04.013
Fourcade Y, Engler JO, Rödder D, Secondi J (2014) Mapping species distributions with MAXENT using a geographically biased sample of presence data: a performance assessment of methods for correcting sampling bias. PLoS ONE 9(5):e97122
García-Llamas P, Geijzendorffer IR, García-Nieto AP, Calvo L, Suárez-Seoane S, Cramer W (2019) Impact of land cover change on ecosystem service supply in mountain systems: a case study in the Cantabrian Mountains (NW of Spain). Reg Environ Change 19(2):529–542
Gonzalez EG, Blanco JC, Ballesteros F, Alcaraz L, Palomero G, Doadrio I (2016) Genetic and demographic recovery of an isolated population of brown bear Ursus arctos L. 1758. PeerJ 4:e1928
González-Bernardo E, Russo LF, Valderrábano E, Fernández Á, Penteriani V (2020) Denning in brown bears. Ecol Evol 10(13):6844–6862
Gregório I, Barros T, Pando D, Morante J, Fonseca C, Ferreira E (2020) Paths for colonization or exodus? New insights from the brown bear (Ursus arctos) population of the Cantabrian Mountains. PLoS ONE 15(1):e0227302
Hemmingmoore H, Aronsson M, Åkesson M, Persson J, Andrén H (2020) Evaluating habitat suitability and connectivity for a recolonizing large carnivore. Biol Cons 242:108352
Lamamy C, Bombieri G, Zarzo-Arias A, González-Bernardo E, Penteriani V (2019) Can landscape characteristics help explain the different trends of Cantabrian brown bear subpopulations? Mamm Res 64(4):559–567
Linnell JDC, Swenson JE, Anderson R (2001) Predators and people: conservation of large carnivores is possible at high human densities if management policy is favourable. Anim Conserv Fórum 4(4):345–349
López-Bao JV, Kaczensky P, Linnell JDC, Boitani L, Chapron G (2015) Carnivore coexistence: wilderness not required. Science 348(6237):871–872
MacDonald D, Crabtree JR, Wiesinger G et al (2000) Agricultural abandonment in mountain areas of Europe: environmental consequences and policy response. J Environ Manage 59:47–69. https://doi.org/10.1006/jema.1999.0335
Manel S, Williams HC, Ormerod SJ (2001) Evaluating presence–absence models in ecology: the need to account for prevalence. J Appl Ecol 38(5):921–931
Martin J, Revilla E, Quenette P-Y, Naves J, Allainé D, Swenson JE (2012) Brown bear habitat suitability in the Pyrenees: transferability across sites and linking scales to make the most of scarce data. J Appl Ecol 49(3):621–631
Mateo Sánchez MC, Cushman SA, Saura S (2014) Scale dependence in habitat selection: the case of the endangered brown bear (Ursus arctos) in the Cantabrian Range (NW Spain). Int J Geogr Inf Sci 28(8):1531–1546
Mateo-Sánchez MC, Gastón A, Ciudad C, García-Viñas JI, Cuevas J, López-Leiva C, Fernández-Landa A, Algeet-Abarquero N, Marchamalo M, Fortin M-J, Saura S (2016) Seasonal and temporal changes in species use of the landscape: how do they impact the inferences from multi-scale habitat modeling? Landscape Ecol 31(6):1261–1276
Méndez TP, Naves J, Vázquez JF, Fernández-Gil A, Seijas J, Albornoz J, Revilla E, Delibes M, Domínguez A (2014) Estimating the population size of the endangered Cantabrian brown bear through genetic sampling. Wildl Biol 20(5):300–309
Merow C, Smith MJ, Silander JA Jr (2013) A practical guide to MaxEnt for modeling species’ distributions: what it does, and why inputs and settings matter. Ecography 36(10):1058–1069
Naves J, Wiegand T, Revilla E, Delibes M (2003) Endangered species constrained by natural and human factors: the case of brown bears in Northern Spain. Conserv Biol 17(5):1276–1289
Naves J, Fernández-Gil A, Rodríguez C, Delibes M (2006) Brown bear food habits at the border of its range: a long-term study. J Mammal 87(5):899–908
Naves J, Palomero G (1993) Ecología de la hibernación del oso en la Cordillera Cantábrica. El oso pardo en España, Instituto Nacional para la Conservación de la Naturaleza, Madrid, pp 147–181
Ordiz A, Støen O-G, Delibes M, Swenson JE (2011) Predators or prey? Spatio-temporal discrimination of human-derived risk by brown bears. Oecologia 166(1):59–67
Ortega Villazán MT, Morales Rodríguez C (2015) El clima de la Cordillera Cantábrica castellano-leonesa: diversidad, contrastes y cambios. https://doi.org/10.14198/INGEO2015.63.04
Palomero G, Ballesteros F, Nores C, Blanco JC, Herrero J, García-Serrano A (2007) Trends in number and distribution of brown bear females with cubs-of-the-year in the Cantabrian Mountains, Spain. Ursus 18(2):145–157
Parres A, Palazón S, Afonso I, Quenette P-Y, Batet A, Camarra J-J, Garreta X, Gonçalves S, Guillén J, Mir S, Jato R, Rodríguez J, Sentilles J, Xicola L, Melero Y (2020) Activity patterns in the reintroduced Pyrenean brown bear population. Mamm Res 65:435–444. https://doi.org/10.1007/s13364-020-00507-w
Penteriani V, Melletti M (2020) Bears of the world: ecology, conservation and management. Cambridge University Press, Cambridge
Penteriani V, Karamanlidis AA, Ordiz A, Ciucci P, Boitani L, Bertorelle G, Zarzo-Arias A, Bombieri G, González-Bernardo E, Morini P, Pinchera F, Fernández N, Mateo-Sánchez MC, Revilla E, Hernando M, d. G., Mertzanis, Y., & Melletti. (2020) Bears in human-modified landscapes: the case studies of the Cantabrian, Apennine, and Pindos Mountains. Cambridge University Press, Cambridge
Pérez T, Naves J, Vázquez JF, Seijas J, Corao A, Albornoz J, Domínguez A (2010) Evidence for improved connectivity between Cantabrian brown bear subpopulations. Ursus 21(1):104–108
Phillips SJ, Dudík M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Eco graphy 31(2):161–175
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190(3):231–259
Posillico M, Meriggi A, Pagnin E, Lovari S, Russo L (2004) A habitat model for brown bear conservation and land use planning in the central Apennines. Biol Cons 118(2):141–150
Pratzer M, Nill L, Kuemmerle T, Zurell D, Fandos G (2023) Large carnivore range expansion in Iberia in relation to different scenarios of permeability of human-dominated landscapes. Divers Distrib 29(1):75–88
Ray JC, Hunter L, Zigouris J (2005) Setting conservation and research priorities for larger African carnivores. Wildlife Conservation Society New York, Bronx
Ripple WJ, Estes JA, Beschta RL, Wilmers CC, Ritchie EG, Hebblewhite M, Berger J, Elmhagen B, Letnic M, Nelson MP, Schmitz OJ, Smith DW, Wallach AD, Wirsing AJ (2014) Status and ecological effects of the world’s largest carnivores. Science 343(6167):1241484
Rodríguez C, Naves J, Fernández-Gil A, Obeso JR, Delibes M (2007) Long-term trends in food habits of a relict brown bear population in northern Spain: the influence of climate and local factors. Environ Conserv 34(1):36–44
Stenset NE, Lutnæs PN, Bjarnadóttir V, Dahle B, Fossum KH, Jigsved P, Johansen T, Neumann W, Opseth O, Rønning O, Steyaert SMJG, Zedrosser A, Brunberg S, Swenson JE (2016) Seasonal and annual variation in the diet of brown bears (Ursus arctos) in the boreal forest of southcentral Sweden. Wildl Biol 22(3):107–116
Swenson JE, Ambarlı H, Arnemo JM, Baskin L, Ciucci P, Danilov PI, Danilov PI, Delibes M, Elfström M, Evans AL, Groff C, Hertel AG, Huber D, Jerina K, Karamanlidis AA, Kindberg J, Kojola I, Krofel M, Kusak J, Mano T, Melletti M, Mertzanis Y, Ordiz A, Palazón S, Parchizadeh J, Penteriani V, Quenette P-Y, Sergiel A, Selva N, Seryodkin I, Skuban M, Stayeart SMJG, Stoen O-G, Tirronen KF, Zedrosser A (2020) Brown bear (Ursus arctos; Eurasia). In: Bears of the world (pp. 139–161). Cambridge University Press
Viñas CD (2010) Recent changes in the rural landscapes of the Cantabrian Mountains. In: Robinson G, Molinero F, Guerra JC (eds) Proceedings of the III anglo-spanish rural geography conference. Royal Geo Soc Canter. pp. 49–80. https://www.scopus.com/authid/detail.uri?authorId=23052855000
Wiegand T, Naves J, Stephan T, Fernandez A (1998) Assessing the risk of extinction for the brown bear (Ursus arctos) in the Cordillera Cantabrica Spain. Ecol Monogr 68(4):539–570
Wydeven AP, Van Deelan TR, Heske E (2009) Recovery of gray wolves in the Great Lakes region of the United States. Springer, Berlin
Zajec P, Roth H et al (2005) The return of brown bear to Switzerland-suitable habitat distribution, corridors and potential conflicts. KORA Report(28).
Zarzo-Arias A, Penteriani V, Delgado MD, Peón Torre P, García-González R, Mateo-Sánchez MC, Vázquez García P, Dalerum F (2019) Identifying potential areas of expansion for the endangered brown bear (Ursus arctos) population in the Cantabrian Mountains (NW Spain). PLoS ONE 14(1):e0209972
Zedrosser A, Dahle B, Swenson JE, Gerstl N (2001) Status and management of the brown bear in Europe. Ursus 12:9–20
Zedrosser A, Steyaert SMJG, Gossow H, Swenson JE (2011) Brown bear conservation and the ghost of persecution past. Biol Cons 144(9):2163–2170
Acknowledgements
We acknowledge financial support by FCT to CESAM (UIDP/50017/2020+UIDB/50017/2020+ LA/P/0094/2020), cE3c (UIDB/00329/2020) and CHANGE (LA/P/0121/2020) through national funds and the co-funding by the FEDER, within the PT2020 Partnership Agreement and Compete 2020. Dário Hipólito was supported by a PhD grant from Fundação para a Ciência e Tecnologia (SFRH/BD/144437/2019), co-financed by the European Social Fund POPH-QREN program. João Carvalho and Rita T. Torres were supported by a research contract (CEECIND/01428/2018, 2021.00690.CEECIND, respectively) from FCT. Eduardo Ferreira is funded by national funds (OE) through FCT in the scope of framework contract foreseen in the article 23, of the Decree-Law 57/2016. We thank Nuno Negrões for the relevant insights provided during the conceptualization of this paper.
Funding
Open access funding provided by FCT|FCCN (b-on).
Author information
Authors and Affiliations
Contributions
CN, DH, DP and EF conceived and design the study; AH, JRM and RH performed the sampling; AH, JRM, CN and DP curated the raw data; AH, JRM, DP and RH provided detailed context in field data interpretation; CN, DH, EF, JC, LMR and RTT discussed and contributed to data analysis strategy; CN and DH performed the data analysis; EF supervised the study; All authors contributed discussion during manuscript preparation; RH and CF contributed with infrastructure and funding; CN, DH and EF wrote the manuscript; All authors read, commented on and approved the successive versions of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nogueira, C., Hipólito, D., Pando, D. et al. Brown bear habitat use patterns across 19 years of population expansion in the Cantabrian Mountains. Landsc Ecol 39, 163 (2024). https://doi.org/10.1007/s10980-024-01943-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10980-024-01943-8