Abstract
Context
Conservation of endangered species necessitates an in-depth understanding of their ecological requirements. Particularly in landscape ecology, the behavioural tendencies of threatened butterfly species in Gotland, a biodiversity-rich island in the Baltic Sea, become crucial.
Objectives
The primary aim of this study was to elucidate the movement patterns of three threatened butterfly species—Euphydryas aurinia, Parnassius apollo, and Phengaris arion—in Gotland and to identify the influence of specific land characteristics on these patterns.
Methods
Our study, conducted from 2017 to 2020 across 60 km2 in Gotland, involved detailed capture-mark-recapture (CMR) efforts of 29,584 captures including 16,223 unique butterflies. We investigate the departure and arrival events of butterflies, specifically focusing on the associations between movements when individuals leave or enter a hectare grid different from their previously recorded location and key landscape features: open vegetated land, ground moisture, and forest cover. We model landscape features to examine the interplay between these and butterfly movement patterns, providing insights into preferred landscape features and conservation strategies.
Results
Among the 4821 arrivals and 5083 departures documented, the species exhibited differential responses to the evaluated habitat features. Both E. aurinia and P. apollo displayed a positive density-dependent dispersal, while P. arion’s movements were not significantly associated with any of the examined habitat features. Landscape properties like open vegetated land and ground moisture index statistically influenced the likelihood of arrival and departure.
Conclusions
The study accentuates the relationship between land cover and the behavioural tendencies of the subject butterfly species. It has broader implications for the targeted habitat management strategies that would benefit threatened butterfly populations in Gotland.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over recent decades, butterflies have become pivotal indicators in ecology and environmental adaptation studies (Warren et al. 2021; Sunde et al. 2023). Often associated with sunny, transient habitats, butterflies are vulnerable to climatic and land-use shifts, especially in northern Europe’s fragmented landscapes (Kindvall et al. 2022). Many butterfly species are declining primarily due to habitat degradation, emphasising the need for robust conservation strategies (Warren et al. 2021). Protecting species in semi-natural grasslands becomes challenging when reliant on age-old land-use practices and threatened by intensive livestock grazing (Ellis et al. 2012; Kindvall et al. 2022). Many butterflies exist within metapopulations, distinct units interlinked by dispersal (Hanski 1998; Saccheri et al. 1998; Ranius et al. 2011). Their constant movement, driven more by daily needs than intrinsic dispersal motives, underscores the nuanced nature of their behaviour (van Dyck and Baguette 2005). Current understanding hinges on studies of fragmented populations, including isolated and migratory species (Shreeve 1995; Hanski et al. 2011). However, regions with abundant suitable habitats often remain under-researched due to the challenges they present, such as the difficulty in delineating habitats, the huge resources required to study large and mobile populations over time, problems in separating routine movements from dispersal, and the complexity involved in designing and conducting studies that can effectively capture the spatiotemporal dynamics of these ecosystems. We must also focus on more widespread areas sustaining endangered species and populations to enhance our understanding of biodiversity dynamics.
Existing literature has revealed multiple factors influencing butterfly habitat selection and movement, including individual attributes (e.g., age and sex), habitat features such as nectar availability and vegetation, and the spatial configuration of the landscape (Warren 1987; Wiens 1989; Hsiung et al. 2018). Increasing studies have focused on animal movements in relation to land cover (Loos et al. 2015; Brown et al. 2017), and the findings offer landscape ecologists insights into animal perceptions of environmental variance and the resulting macro-level effects (Turlure et al. 2011). Additionally, the density of conspecifics has been identified as important, often influencing dispersal (Enfjäll and Leimar 2005). While previous studies offer critical insights into these relationships, they often neglect the potential differential impacts of land cover variables on patterns of departures from and arrivals in sites (Kareiva et al. 1990; Baguette et al. 1998; Cowley et al. 2001; Ims and Andreassen 2005). Understanding the interplay between land cover, population densities and the movement dynamics of endangered butterflies is crucial for devising sound conservation strategies. Therefore, this study’s focus on dissecting these relationships is not merely additive to the field; it is pivotal for crafting more effective, evidence-based conservation measures that could determine the survival of endangered species within rapidly changing ecosystems.
In this study, we offer an in-depth examination and comparison of movement patterns in three declining and locally red listed butterfly species (Maes et al. 2019): Marsh Fritillary (Euphydryas aurinia, Rottemburg, 1775; Nymphalidae), Apollo (Parnassius apollo, Linneaus, 1758; Papilionidae), and Large Blue (Maculinea/Phengaris arion Linneaus, 1758; Lycaenidae). Our study area in the northern part of the island of Gotland in the Baltic Sea is known for its extensive suitable habitats, which support some of the world’s highest population densities for these species, at least in favourable years (Franzén et al. 2024; Sunde et al. 2024). This unique setting provides an unparalleled opportunity to study the dynamics of these butterfly species in conditions where their populations can thrive.
Understanding the movement patterns of butterflies has long been a cornerstone of conservation biology and landscape ecology, reflecting broader concerns about biodiversity, habitat fragmentation, and ecological networks (Baguette et al. 2013). Research has traditionally distinguished between two primary types of movements: routine, localised movements within habitat patches, often driven by immediate needs such as foraging, mating, and thermoregulation. (Stevens et al. 2010), and dispersal movements, which involve longer distances and are crucial for gene flow, population dynamics, and the colonisation of new habitats (Clobert et al. 2001; Bowler and Benton 2005). While much of the butterfly movement ecology literature focuses on the former, emphasising daily activities within specific habitat patches (Warren et al. 2001) (Dover and Settele 2009), there is a growing recognition of the need to better understand the triggers and consequences of dispersal movements. These are especially relevant in the context of environmental change and habitat fragmentation, which increasingly challenge the persistence of butterfly populations (Schtickzelle et al. 2006).
Our study aims to bridge this gap by examining butterflies' departure and arrival events across hectare grids, moving beyond the confines of localised movements to explore the factors influencing dispersal and longer-distance movements. Specifically, we investigate if land cover properties trigger butterflies to leave a particular one-hectare grid and arrive in another. Our core aim was to discern whether departures from and arrivals in hectare grids of these species correlate with local population density and land cover variables, including open vegetated land, ground moisture index, and forest cover. To this end, we assessed the relationships between land cover, hectare grid-level population density, and whether the proportion of butterflies departing and arriving in these hectare grids differed from grids where they were previously recorded.
Materials and methods
Description of the study area
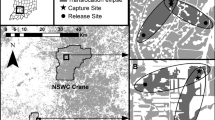
Our study encompasses a 60 km2 region near Slite on Gotland Island, Sweden (midpoint: 57°69′ N, 18°69′ E) (Fig. 1). This area, one of Europe’s last refuges for these species, is distinguished by its calcareous bedrocks and vast natural habitats. These support sustainable populations of the butterfly species we investigated, including the Marsh Fritillary, Apollo, and Large Blue. So far, these geological and ecological features have created a unique sanctuary for these species, offering them a stronghold against the global trend of habitat degradation and population declines (Franzén et al. 2024). The climate is temperate, with cool summers averaging 16.6 °C in July, cold winters dipping to − 2.1 °C in February, and an average annual precipitation of 524 mm (Persson 2015). The heterogeneous environment featuring 15 habitats per the Habitats Directive (Kindvall et al. 2022), has seen intensified livestock grazing since 2000 (Kindvall et al. 2022). Old forests, primarily pine, are scattered throughout the region. The western and northeastern borders are marked by agricultural fields commonly treated with pesticides and fertilisers.
A red dot denotes the location of the study area—the spatial distribution of hectare grids with captured target species on Gotland, southeastern Sweden. Panel A depicts the geographic dispersion of Marsh Fritillary (Euphydryas aurinia), Panel B of Apollo (Parnassius apollo), and Panel C of Large Blue (Phengaris arion). In each panel, the top sub-panel illustrates the number of departures per grid, while the bottom sub-panel delineates the number of arrivals per hectare grid. The colour gradient, transitioning from blue to purple, quantitatively represents the range of recorded departures and arrivals, with purple indicating higher numbers. Photographs Markus Franzén
Description of studied species
We examined three butterfly species: E. aurinia, P. apollo, and P. arion. These species face rapid declines globally and significant conservation challenges at regional and local levels in Europe. They are protected within the European Union (EU) under the Habitats Directive (Council Directive 92/43/EEC). Parnassius apollo is near threatened in Europe, P. arion is endangered, and E. aurinia is red-listed in several European countries (Maes et al. 2019). Notably, they can be locally abundant in areas of Gotland, enabling in-depth population and movement analysis (Franzen et al. 2022; Franzén et al. 2022, 2024; Johansson et al. 2020; Sunde et al. 2024).
Phengaris arion, a blue butterfly spanning 32–42 mm (Fig. 1), is native to the western Palaearctic. Thriving in dry calcareous grasslands and Alvar in our study region, this univoltine butterfly is active from July to August. The larvae, feeding on Thymus serpyllum, are adopted by Myrmica ants, feeding parasitically on ant broods (Thomas and Wardlaw 1992; Eliasson et al. 2005).
Parnassius apollo, a white butterfly in the Papilionidae family with a 73–87 mm wingspan (Fig. 1), populates areas similar to P. arion. Preferring areas with bare rocks, it thrives on open alvar terrain in our study area. P. apollo has experienced a notable decline since the 1950s, primarily due to habitat loss and environmental changes. Its adult flight period occurs from June to August and visits flowering Thymus serpyllum and Centaurea scabiosa, while the larvae feed on Sedum album from April to June (Eliasson et al. 2005).
Euphydryas aurinia, an orange-to-brown butterfly with a 33–48 mm wingspan (Fig. 1), ranges from northern Africa to China. It occurs on fens and ungrazed grasslands in our study area and is active from May to June. The larvae feed on Succisa pratensis, creating silk shelters and hibernating in vegetation, resuming feeding in spring (Eliasson et al. 2005).
Collection of butterfly occupancy and movement data
Our study utilised a systematic grid-based approach, covering the entire designated area. We overlaid a fishnet grid, each square measuring 100 × 100 m2, across the study area, resulting in 3430-hectare grids subjected to butterfly surveys. The surveys spanned different years for each species for logistical reasons: E. aurinia in 2017 across 1330 grids, P. apollo in 2019 across 2359 grids, and P. arion in 2020 over 2256 grids. The timing of our fieldwork was meticulously planned to coincide with the flight periods of the respective species. The survey for E. aurinia lasted 33 days in 2017, P. apollo spanned 52 days in 2019, and P. arion lasted 26 days in 2020. Surveys were conducted daily between 8 a.m. and 6 p.m. during suitable weather by up to ten trained personnel daily. Upon each butterfly’s capture, it was individually marked and identified by species and sex. The capture location and timestamp were directly and automatically recorded into our database through the Field Maps application (ESRI). We avoided surveys during rain or temperatures below 14 °C. Each hectare grid was systematically checked daily or every other day, and we employed a rotation strategy to ensure varied personnel surveyed grids at different times. In total, 14, 18, and nine field workers assisted with the E. aurinia, P. apollo, and P. arion surveys.
Land cover data extraction
Utilising the Swedish land cover database (Anonymous 2020), we procured data on three crucial land cover variables: forest cover, vegetated open land coverage, and the ground moisture index. For each hectare grid, we calculated the proportion of forest cover and vegetated open land and the average ground moisture index to be employed in subsequent modelling.
Assessing density-dependent movements: proportions of departures (emigration) and arrivals (immigration) per hectare grid
We defined two primary metrics per hectare grid to evaluate density dependence in butterfly movement: ‘arrivals’ and ‘departures’. The departure fraction for grid “1” was calculated by dividing the number of butterflies leaving grid “1” by the overall recaptures in grid “1”. Similarly, the arrival fraction for grid “1” was calculated by dividing the number of recaptures of butterflies not originally marked in grid “1” but found there later (immigration) by the overall recaptures in grid “1”.
Statistical analysis
Analysis of land cover and density-dependent movements
To assess potential land cover and density-dependent butterfly movements within hectare grids, we utilised Generalised Linear Models (GLMs) implemented through the glm function in the stats package, part of R’s core distribution. These models examined the relationship between the proportions of departures and arrivals (examined separately) and the four continuous predictors: forest cover vegetated open land coverage, ground moisture index, and population density (quantified as the number of individuals per visit) within the hectare grid. Two GLMs were conducted for each species—one for departures and one for arrivals. Models were manually selected based on the Akaike Information Criterion (AIC), with the final model boasting the lowest AIC presented. Given overdispersion and the proportions of departures (emigration) and arrivals (immigration) as dependent variables, a quasibinomial distribution was applied. This distribution corrects variance, making more precise statistical inferences. (Dunn et al. 2018). Visualisation tools from the ggplot2 and ggeffects packages facilitated model interpretation by presenting predicted probabilities for different population densities and capturing events and species (Lüdecke 2018). All statistical analyses were performed using R version 4.3.1 (R Core Team 2023).
To specifically investigate the dynamics of arrivals and departures associated with longer-distance movements, we refined our dataset to include only those events that surpassed species-specific movement thresholds. These thresholds were established based on the average movement distances for Euphydryas aurinia (942 m), Parnassius apollo (735 m), and Phengaris arion (594 m), respectively. This filtration allowed us to isolate and examine the subset of movements that constitute significant (longer-term) dispersal events rather than shorter routine, localised foraging, sun basking or mating activities. Subsequent analyses utilised Generalized Linear Models (GLMs) as described above to assess the influence of grid characteristics and population densities on these arrivals and departures that resulted from long-distance movement events.
Results
Departure and arrival patterns
The uniquely marked individuals per species included 10,161 E. aurinia in 2017, 5902 P. apollo in 2019, and 160 P. arion in 2020. Table 1 presents the density of butterflies (average individuals marked per visit per grid), recapture rates, arrivals, and departures.
Movements in relation to land cover
For P. apollo, we identified a negative correlation between departures from the hectare grid and forest cover and a positive correlation with the ground moisture index (Fig. 2; Table 2). Similarly, the pattern for arrivals of P. apollo mirrored the findings for departures to forest cover and ground moisture index (Fig. 2; Table 2). For E. aurinia, an increase in the proportion of vegetated ‘other open land’ was correlated with an increased frequency of departures from the grids and a similarly positive relationship with the ground moisture index (Fig. 2; Table 2). These correlations were reciprocated in the frequency of arrivals, with significant values for vegetated ‘other open land’ and ground moisture index (Fig. 2; Table 2). In P. arion, neither arrivals nor departures from the grids were significantly associated with any of the three habitat features that we investigated (Fig. 2; Table 2). None of the examined factors statistically significantly influenced longer-distance arrivals or departures among the studied butterfly species.
Model coefficients representing the estimates of different predictors ± SE for the three butterfly species. Each point denotes the estimate for a predictor, with the colour indicating the specific butterfly species: E. aurinia (red), P. apollo (black), and P. arion (blue). The associated error bars signify the standard error of these estimates. Predictors are displayed on the y-axis, while the x-axis represents the estimated values. Circles = departures, and triangles = arrivals
Density-dependent movements
Both E. aurinia and P. apollo showed strong positive trends for arrivals and departures in relation to population density, as displayed in Table 2. However, P. arion also leaned towards similar trends, although it did not reach traditional levels of statistical significance. Despite this, including these variables improved the model’s overall fit, according to the AIC. Arrivals and departures filtered to only include longer-distance movements were not significantly related to population density.
Discussion
Departure and arrival patterns of two of the three butterfly species studied were intricately tied to land cover characteristics and local population densities. The dichotomy between within-site and between-site movements suggests distinct ecological and behavioural drivers at play. Within-site movements, predominantly within the same hectare grid (< 100 m), are closely tied to daily survival strategies, such as temperature regulation, foraging for nectar, and seeking host plants, reflecting localised resource utilisation (Warren et al. 2001; Dover and Settele 2009). In contrast, between-site movements here defined as moving from and to another hectare grid, although less frequent, suggest a response to land cover properties, possibly driven by the need for suitable breeding sites, avoidance of predation, or adaptation to environmental changes (Schtickzelle et al. 2006). Although our study did not find significant determinants for long-distance movements, this aspect remains crucial for understanding metapopulation dynamics and gene flow. The lack of significant results may be attributed to the relative rarity of long-distance dispersal events, which are often stochastic and influenced by factors beyond the scope of our study design (Kindvall 1999; Hanski 1994, 1998; Hanski et al. 2011). Landscape barriers, weather conditions, and individual variation in dispersal capacity could all contribute to the complexity of long-distance movements (Alerstam et al. 2003). Future research following each movement of individual butterflies may be necessary to elucidate patterns in long-distance dispersal for these species.
When considering these species, conservation strategies must account for the nuanced interplay between habitat attributes and local butterfly abundances. For P. apollo, the evident disinclination towards tree-covered areas aligns well with its established preference for open terrains replete with bare rocks (Nakonieczny et al. 2007; Sunde et al. 2024). The species has been noted for its selective habitat choices, primarily seeking areas with minimal vegetation, a behavioural trait potentially driven by evolutionary and ecological pressures (Fred et al. 2006). Euphydryas aurinia, on the other hand, displayed a strong inclination towards vegetated open lands, supporting findings from previous studies which suggest this species thrives in such habitats due to the availability of specific resources or potential mating opportunities (Warren et al. 1994; Wahlberg et al. 2002). The ground moisture index also played a pivotal role, which is unsurprising given the butterfly’s predilection for rich fens and ungrazed grasslands, habitats often associated with higher moisture levels (Botham et al. 2011). Lastly, the P. arion exhibited patterns subtly influenced by population densities rather than explicit land cover types. This could imply a more complex behavioural framework, possibly driven by interspecific interactions, competition, or other ecological dynamics not directly evaluated in this study (Hayes 2015; Vilbas et al. 2015).
Parnassius apollo and E. aurinia arrived and departed from grids with certain land cover properties. These movements in butterfly species are not merely a linear trajectory from unsuitable to suitable habitats; their activities seem to be influenced by a complex matrix of factors. The decision to depart from or arrive in a particular area could be driven by the heterogeneity of microhabitats within a larger ‘suitable’ habitat. For instance, butterflies may continually seek optimal microsites within a favourable landscape to maximise access to resources such as nectar, mates, or oviposition sites (Hanski 1994; Schultz 2001). Moreover, these butterflies' movements might be intricately tied to their behavioural ecology. Territoriality, mate-seeking behaviours, and avoiding intraspecific competition could all play pivotal roles in their frequent movements (Baguette et al. 1996; Brunzel 2002). It is also worth considering that some areas, while rich in resources, might also be hotspots for predators or parasitoids, prompting butterflies to move frequently to balance resource acquisition with predation risk (Kruess and Tscharntke 2000; Eliasson and Shaw 2003). Furthermore, butterflies' cognitive abilities should be considered. They may engage in exploratory flights within suitable habitats to gather information and reevaluate the quality of their surroundings (Dukas 1998; Couto et al. 2023). In doing so, they continually optimise their choices, even within a habitat broadly categorised as ‘suitable’. Lastly, environmental cues, including temperature, humidity, and light fluctuations, could prompt temporary movements even within favourable habitats. Such transient departures and arrivals might be strategies to optimise thermoregulation or avoid transient adverse conditions (Porter 1982, 1984; Kuussaari et al. 1996; Braem and Van Dyck 2021; Franzen et al. 2022).
Forested areas are characterised by dense tree canopies, which can significantly reduce sunlight penetration to the forest floor. The resultant cool, shaded microclimate is typically less hospitable to many butterfly species, particularly those adapted to open habitats (Dennis et al. 2004). The diminished sunlight also translates to fewer flowering plants, which are primary nectar sources for butterflies (Clausen et al. 2001). Consequently, the negative association we observed between forest cover and butterfly arrivals and departures aligns with the understanding that dense forests are less preferred if not outright avoided, due to their suboptimal conditions (Franzén and Nilsson 2008; Sunde et al. 2024). Further, in stark contrast to forests, open vegetated areas offer abundant sunlight, facilitating the growth of many flowering plants. Such landscapes become veritable oases for butterflies, providing them with a rich supply of nectar—a crucial energy source (Tscharntke et al. 2002). Our findings of a positive relationship between open vegetated land and butterfly movements echo this ecological reality. The open land offers sustenance and promotes mate-finding and oviposition activities due to increased butterfly visibility (Thomas et al. 1996; Konvicka and Kuras 1999).
Moist conditions are generally favourable for butterflies for several reasons. First, damp environments can support a diverse range of plant species, enhancing habitat quality (Maclean et al. 2015). Moreover, moist areas can also serve as ‘mud-puddling’ sites, where butterflies extract essential minerals from damp soils—a behaviour observed especially in males (Molleman 2010). Our findings suggest a complex relationship between ground moisture and butterfly movement that varies among species. While some butterflies prefer arid habitats, ground moisture may serve as a proxy for other crucial variables like floral abundance or microhabitat structures (Cabral et al. 2017; Franzen et al. 2022). Our results indicate that moisture conditions indirectly influence habitat selection and movement patterns of E. aurinia and P. apollo, whose larval food plants and ant nests, respectively, are affected by soil moisture (Thomas et al. 1989; Fred et al. 2006; Franzén et al. 2022; Kindvall et al. 2022).
As a mobile species and avid flower visitor (Franzén et al. 2024), P. apollo's departures and arrivals may be driven by the pursuit of abundant floral resources or favorable microclimatic conditions for thermoregulation in areas of higher ground moisture (Fred et al. 2010; Franzen et al. 2022). Intriguingly, P. arion diverged from the behavioural trends exhibited by the other butterfly species under consideration. While variables such as forest cover, open vegetated land, and ground moisture index were less impactful, population density emerged as a salient predictor for the movements of P. arion. This divergence could potentially be attributed to the smaller sample size and fewer individual specimens studied, thereby reducing statistical power to detect nuanced habitat preferences (Thomas et al. 2009). Additionally, the populations of these species are recovering from a recent drought, which could be a confounding variable influencing movement patterns. It is plausible that P. arion's ecological requirements (depends on certain nectar plants and host ants) or behaviours predispose it to be more sensitive to conspecific density over broader habitat characteristics. The ecological specificity of P. arion may render it more responsive to variations in conspecific density rather than to general habitat features. This interpretation aligns with the findings of Thomas et al. (2011), who observed that P. arion exhibits a heightened sensitivity to conspecific presence, potentially influencing its dispersal and habitat utilisation patterns more than the physical attributes of the environment. Moreover, Osváth-Ferencz et al. (2017) state that the unique life-cycle dependencies and social interactions within P. arion populations accentuate the role of density dynamics over environmental heterogeneity in determining their spatial distribution. Given the limited dataset for this species, we advocate for further extensive studies to elucidate the specific habitat preferences of P. arion.
Our study unearthed a consistent trend of positive density-dependent mobility across all three butterfly species, resonating with patterns identified in various flora and fauna studies (Rodrigues and Johnstone 2014). The annals of ecological research have documented both positive (Nowicki and Vrabec 2011) and negative density-dependent mobilities (Gilbert and Singer 1973; Brown and Ehrlich 1980; Ims and Andreassen 2005; Støen et al. 2006; Konvicka et al. 2012). Intriguingly, the same species, such as the E. aurinia and Glanville fritillary butterfly, Melitaea cinxia, have demonstrated contrasting mobility depending on circumstances (Kuussaari et al. 1996; Enfjäll and Leimar 2005; Johansson et al. 2022). A particularly captivating observation from our study is the propensity of these butterflies to leave high-density regions, only to later settle in areas of similar density. This challenges the conventional ecological wisdom, which posits that species migrate from areas of high to low density to mitigate competition for resources (Solomon 1949). Instead, our data suggests that these butterflies might be navigating between densely populated grids in pursuit of prime habitats (Dunning et al. 1992), evading predators (Lima and Dill 1990), or responding to intrinsic territorial and behavioural impulses (Bowler and Benton 2005). Future endeavours in this domain should delve deeper into individual butterfly movement patterns and map resource availability (Dennis et al. 2003), and explore potential predation threats.
Conclusion and future directions
In synthesising these findings, it becomes clear that while overarching patterns can be discerned, each species exhibits unique responses to the environment. Such distinctions underscore the importance of tailored conservation strategies. When working towards conserving these endangered species, it is imperative to consider both the broader landscape changes and the intricate species-specific behaviours in response to their immediate surroundings (Jones & Harrison, 2015; Smith et al., 2010).
Data availability statement
The datasets generated during and analysed during the current study are available from the corresponding author upon reasonable request.
References
Alerstam T, Hedenström A, Åkesson S (2003) Long-distance migration: evolution and determinants. Oikos 103(2):247–260
Anonymous (2020) Nationella marktäckedata 2018 basskikt: produktbeskrivning [National Land-Cover Data 2018 Basic Layer: Product Description]. Version 2.2; 7 July 2020. Swedish Environmental Protection Agency
Baguette M, Convie I, Neve G (1996) Male density affects female spatial behaviour in the butterfly Proclossiana eunomia. Acta Oecol-Int J Ecol 17(3):225–232
Baguette M, Vansteenwegen C, Convie I, Neve G (1998) Sex-biased density-dependent migration in a metapopulation of the butterfly Proclossiana eunomia. Acta Oecol-Int J Ecol 19(1):17–24
Baguette M, Blanchet S, Legrand D, Stevens VM, Turlure C (2013) Individual dispersal, landscape connectivity and ecological networks. Biol Rev 88(2):310–326
Botham M, Ash D, Aspey N et al (2011) The effects of habitat fragmentation on niche requirements of the marsh fritillary, Euphydryas aurinia,(Rottemburg, 1775) on calcareous grasslands in southern UK. J Insect Conserv 15(1–2):269–277
Bowler DE, Benton TG (2005) Causes and consequences of animal dispersal strategies: relating individual behaviour to spatial dynamics. Biol Rev 80(2):205–225
Braem S, Van Dyck H (2021) On the role of direct sunlight for egg-laying behaviour: lessons and ideas from the Speckled wood. Ecol Entomol 46(4):800–806
Brown IL, Ehrlich PR (1980) Population biology of the checkerspot butterfly, Euphydryas chalcedona structure of the Jasper Ridge colony. Oecologia 47:239–251
Brown LM, Fuda RK, Schtickzelle N et al (2017) Using animal movement behavior to categorize land cover and predict consequences for connectivity and patch residence times. Landscape Ecol 32:1657–1670
Brunzel S (2002) Experimental density-related emigration in the cranberry fritillary Boloria aquilonaris. J Insect Behav 15(6):739–750
Cabral I, Keim J, Engelmann R, Kraemer R, Siebert J, Bonn A (2017) Ecosystem services of allotment and community gardens: a Leipzig, Germany case study. Urban for Urban Green 23:44–53
Clausen HD, Holbeck HB, Reddersen J (2001) Factors influencing abundance of butterflies and burnet moths in the uncultivated habitats of an organic farm in Denmark. Biol Cons 98(2):167–178
Clobert J, Danchin E, Dhondt AA, Nichols J (2001) Dispersal. Oxford University Press; 1st edition, Oxford, p. 480
Couto A, Young FJ, Atzeni D et al (2023) Rapid expansion and visual specialisation of learning and memory centres in the brains of Heliconiini butterflies. Nat Commun 14(1):4024
Cowley MJR, Thomas CD, Roy DB et al (2001) Density-distribution relationships in British butterflies. I. The effect of mobility and spatial scale. J Anim Ecol 70(3):410–425
Dennis RLH, Shreeve TG, van Dyck H (2003) Towards a functional resource-based concept for habitat: a butterfly biology viewpoint. Oikos 102(2):417–426
Dennis RLH, Hodgson JG, Grenyer R, Shreeve TG, Roy DB (2004) Host plants and butterfly biology. Do host-plant strategies drive butterfly status? Ecol Entomol 29(1):12–26
Dover J, Settele J (2009) The influences of landscape structure on butterfly distribution and movement: a review. J Insect Conserv 13(1):3–27
Dukas R (1998) Cognitive ecology: the evolutionary ecology of information processing and decision making. University of Chicago Press
Dunn PK, Smyth GK, Dunn PK, Smyth GK (2018) Chapter 9: models for proportions: binomial GLMs. In Peter K. Dunn, Gordon K. Smyth, Generalized linear models with examples in R. Springer, New York.
Dunning JB, Danielson BJ, Pulliam HR (1992) Ecological processes that affect populations in complex landscapes. Oikos 169–175
Eliasson CU, Ryrholm N, Holmér M, Gilg K, Gärdenfors U (2005) Nationalnyckeln till Sveriges flora och fauna. Fjärilar: Dagfjärilar. Hesperidae—Nymphalidae. ArtDatabanken, SLU, Uppsala
Eliasson CU, Shaw MR (2003) Prolonged life cycles, oviposition sites, foodplants and Cotesia parasitoids of Melitaeini butterflies in Sweden. Oedippus 21:1–52
Ellis S, Bourn NA, Bulman CR (2012) Landscape-scale conservation for butterflies and moths: lessons from the UK. Butterfly Conservation Wareham
Enfjäll K, Leimar O (2005) Density-dependent dispersal in the Glanville fritillary. Melitaea Cinxia Oikos 108(3):465–472
Franzén M, Nilsson SG (2008) How can we preserve and restore species richness of pollinating insects on agricultural land? Ecography 31:698–708
Franzén M, Francioli Y, Askling J, Kindvall O, Johansson V, Forsman A (2022) Differences in phenology, daily timing of activity, and associations of temperature utilization with survival in three threatened butterflies. Sci Rep 12(1):1–13
Franzen M, Francioli Y, Askling J, Kindvall O, Johansson V, Forsman A (2022) Yearly weather variation and surface temperature drives the spatiotemporal dynamics of a threatened butterfly and its host plant. Front Ecol Evolut 10. https://doi.org/10.3389/fevo.2022.917991
Franzén M, Johansson H, Askling J et al (2024) Long‐distance movements, large population sizes and density‐dependent dispersal in three threatened butterfly species. Insect Conservation and Diversity. Online early. https://doi.org/10.1111/icad.12766
Fred MS, OïHara RB, Brommer J (2006) Consequences of the spatial configuration of resources for the distribution and dynamics of the endangered Parnassius apollo butterfly. Biol Cons 130:183–192
Fred MS, OïHara RB, Brommer J (2010) Consequences of the spatial configuration of resources for the distribution and dynamics of the endangered Parnassius apollo butterfly. Biol Cons 130:183–192
Gilbert LE, Singer MC (1973) Dispersal and gene flow in a butterfly species. Am Nat 107(953):58–72
Hanski I (1994) A practical model of metapopulation dynamics. J Anim Ecol 63(1):151–162
Hanski I (1998) Metapopulation dynamics. Nature 396(6706):41–49
Hanski I, Mononen T, Ovaskainen O (2011) Eco-evolutionary metapopulation dynamics and the spatial scale of adaptation. Am Nat 177(1):29–43
Hayes MP (2015) The biology and ecology of the large blue butterfly Phengaris (Maculinea) arion: a review. J Insect Conserv 19(6):1037–1051
Hsiung AC, Boyle WA, Cooper RJ, Chandler RB (2018) Altitudinal migration: ecological drivers, knowledge gaps, and conservation implications. Biol Rev 93(4):2049–2070
Ims RA, Andreassen HP (2005) Density-dependent dispersal and spatial population dynamics. Proc R Soc B 272(1566):913–918
Johansson V, Kindvall O, Askling J, Franzén M (2020) Extreme weather affects colonization–extinction dynamics and the persistence of a threatened butterfly. J Appl Ecol 57(6):1068–1077
Johansson V, Kindvall O, Askling J, Säwenfalk DS, Norman H, Franzén M (2022) Quick recovery of a threatened butterfly in well-connected patches following an extreme drought. Insect Conserv Divers 15:572–582
Kareiva P, Mullen A, Southwood R (1990) Population dynamics in spatially complex environments: theory and data [and discussion]. Philos Trans R Soc B 330(1257):175–190
Kindvall O (1999) Dispersal in a metapopulation of the bush cricket, Metrioptera bicolor (Orthoptera: Tettigoniidae). J Anim Ecol 68(1):172–185
Kindvall O, Franzén M, Askling J, Forsman A, Johansson V (2022) Subsidized Common Agricultural Policy grazing jeopardizes the protection of biodiversity and Natura 2000 targeted species. Anim Conserv 25:597–607
Konvicka M, Kuras T (1999) Population structure, behaviour and selection of oviposition sites of an endangered butterfly, Parnassius mnemosyne, in Litovelske Pomoravi, Czech Republic. J Insect Conserv 3(3):211–223
Konvicka M, Zimmermann K, Klimova M, Hula V, Fric Z (2012) Inverse link between density and dispersal distance in butterflies: field evidence from six co-occurring species. Popul Ecol 54:91–101
Kruess A, Tscharntke T (2000) Species richness and parasitism in a fragmented landscape: experiments and field studies with insects on Vicia sepium. Oecologia 122(1):129–137
Kuussaari M, Nieminen M, Hanski I (1996) An experimental study of migration in the Glanville fritillary butterfly Melitaea cinxia. J Anim Ecol 65(6):791–801
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68(4):619–640
Loos J, Kuussaari M, Ekroos J et al (2015) Changes in butterfly movements along a gradient of land use in farmlands of Transylvania (Romania). Landsc Ecol 30:625–635
Lüdecke D (2018) ggeffects: tidy data frames of marginal effects from regression models. J Open Sour Softw 3(26):772
Maclean IM, Hopkins JJ, Bennie J, Lawson CR, Wilson RJ (2015) Microclimates buffer the responses of plant communities to climate change. Glob Ecol Biogeogr 24(11):1340–1350
Maes D, Verovnik R, Wiemers M et al (2019) Integrating national Red Lists for prioritising conservation actions for European butterflies. J Insect Conserv 23(2):301–330
Molleman F (2010) Puddling: from natural history to understanding how it affects fitness. Entomol Exp Appl 134(2):107–113
Nakonieczny M, Kedziorski A, Michalczyk K (2007) Apollo butterfly (Parnassius apollo L.) in Europe–its history, decline and perspectives of conservation. Funct Ecosyst Commun 1(1):56–79
Nowicki P, Vrabec V (2011) Evidence for positive density-dependent emigration in butterfly metapopulations. Oecologia 167(3):657
Osváth-Ferencz M, Bonelli S, Nowicki P et al (2017) Population demography of the endangered large blue butterfly Maculinea arion in Europe. J Insect Conserv 21:411–422
Persson G (2015) Sveriges klimat 1860–2014: underlag till dricksvattenutredningen. SMHI
Porter K (1982) Basking behaviour in larvae of the butterfly Euphydryas aurinia. Oikos 38:308–312
Porter K (1984) Sunshine, sex-ratio and behaviour of Euphydryas aurinia larvae. In: Vane-Wright RI, Ackery PR (eds) The biology of butterflies. Academic Press, London, pp 309–311
R Core Team (2023) R: a language and environment for statistical. R version 4.3.0, Vienna
Ranius T, Nilsson SG, Franzén M (2011) How frequent is metapopulation structure among butterflies in grasslands? Occurrence patterns in a forest-dominated landscape in southern Sweden. Ecoscience 18:138–144
Rodrigues AM, Johnstone RA (2014) Evolution of positive and negative density-dependent dispersal. Proc Royal Soc B: Biol Sci 281(1791):20141226
Saccheri I, Kuussaari M, Kankare M, Vikman P, Fortelius W, Hanski I (1998) Inbreeding and extinction in a butterfly metapopulation. Nature 392(6675):491–494
Schtickzelle N, Mennechez G, Baguette M (2006) Dispersal depression with habitat fragmentation in the bog fritillary butterfly. Ecology (washington) 87(4):1057–1065
Schultz CB (2001) Restoring resources for an endangered butterfly. J Appl Ecol 38(5):1007–1019
Shreeve TG (1995) Butterly mobility. In: Pullin AS (ed) Ecology and conservation of butterflies. Chapman and Hall, London
Solomon ME (1949) The natural control of animal populations. J Anim Ecol 18:1–35
Stevens VM, Turlure C, Baguette M (2010) A meta-analysis of dispersal in butterflies. Biol Rev (cambridge) 85(3):625–642
Støen O-G, Zedrosser A, Sæbø S, Swenson JE (2006) Inversely density-dependent natal dispersal in brown bears Ursus arctos. Oecologia 148:356–364
Sunde J, Askling J, Kindvall O, Johansson V, Franzén M (2024) Negative impacts of future forest succession on three threatened butterfly species. Biodivers Conserv 33, 2885–2910. https://doi.org/10.1007/s10531-024-02892-z
Sunde J, Franzén M, Betzholtz P-E et al (2023) Century-long butterfly range expansions in northern Europe depend on climate, land use and species traits. Commun Biol 6(1):601
Thomas JA, Wardlaw JC (1992) The capacity of a Myrmica ant nest to support a predacious species of Maculinea butterfly. Oecologia 91(1):101–109
Thomas JA, Elmes GW, Wardlaw JC, Woyciechowski M (1989) Host specificity among Maculinea butterflies in Myrmica ant nests. Oecologia 79(4):452–457
Thomas CD, Singer MC, Boughton DA (1996) Catastrophic extinction of population sources in a butterfly metapopulation. Am Nat 148(6):957–975
Thomas JA, Simcox DJ, Clarke RT (2009) Successful conservation of a threatened Maculinea butterfly. Science 325(5936):80–83
Thomas J, Simcox D, Hovestadt T (2011) Evidence based conservation of butterflies. J Insect Conserv 15(1–2):241–258
Tscharntke T, Steffan-Dewenter I, Kruess A, Thies C (2002) Contribution of small habitat fragments to conservation of insect communities of grassland-cropland landscapes. Ecol Appl 12(2):354–363
Turlure C, Baguette M, Stevens VM, Maes D (2011) Species-and sex-specific adjustments of movement behavior to landscape heterogeneity in butterflies. Behav Ecol 22(5):967–975
van Dyck H, Baguette M (2005) Dispersal behaviour in fragmented landscapes: Routine or special movements? Basic Appl Ecol 6:535–545
Vilbas M, Teder T, Tiitsaar A, Kaasik A, Esperk TJIC, Diversity (2015) Habitat use of the endangered parasitic butterfly Phengaris arion close to its northern distribution limit. 8(3):252–260
Wahlberg N, Klemetti T, Hanski I (2002) Dynamic populations in a dynamic landscape: the metapopulation structure of the marsh fritillary butterfly. Ecography 25(2):224–232
Warren MS (1987) The ecology and conservation of the heath fritillary butterfly, Mellicta-Athalia. 1. Host selection and phenology. J Appl Ecol 24(2):467–482
Warren MS, Munguira ML, Ferrin J (1994) Notes on the distribution, habitats and conservation of Euphydryas aurinia (Rottemburg) (Lepidoptera: Nymphalidae) in Spain. Entomologists Gazette 45:5–12
Warren MS, Hill JK, Thomas JA et al (2001) Rapid responses of British butterflies to opposing forces of climate and habitat change. Nature 414(6859):65–69
Warren MS, Maes D, van Swaay CAM et al (2021) The decline of butterflies in Europe: Problems, significance, and possible solutions. Proc Natl Acad Sci 118(2):e2002551117
Wiens JA (1989) Spatial scaling in ecology. Funct Ecol 3(4):385–397
Acknowledgements
Anders Birgersson, Andreas Friedrich, Anna Hassel, Bafraw Karimi, Caspar Ström, Daniela Polic, Demieka Säwenfalk, Emma Drotz, Hannah Norman, Jan Högvall, Jesper Wadstein, Johan Stenberg, Jonas Lundquist, Judith Askling, Julia Ödéhn, Junia Birgersson, Lovisa Johansson, Martin Lindner, Patrick Gant, Petter Drotz, Sara Nyberg, Staffan Nilsson, Stina Juhlin, Tove Rönnbäck and Veronika Kraft assisted in collecting data in the field. We are grateful to one anonymous reviewer for comments on the manuscript. The provincial government of Gotland provided the necessary permits for the study.
Funding
Open access funding provided by Linköping University. The study was funded by Heidelberg Materials AB, The Swedish Research Council, Formas (Grant to M.F. and A.F. Dnr. 2018-02846), Swedish National Research Programme on Climate (Grant to M.F., J.S., V.J and A.F. Dnr. 2021-02142), Stiftelsen Oscar och Lili Lamms Minne (Grant to VJ Dnr. FO2020-0023), and Carl Trygger foundation (Grant to MF).
Author information
Authors and Affiliations
Contributions
MF, JA, OK, AF, and VJ conceived the study and coordinated the fieldwork. JA, MF, and OK collected the data. MF and JS analysed the data. MF, JS, and AF interpreted the data/results and wrote the first draft. All authors commented on the manuscript and approved the final version before submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Franzén, M., Askling, J., Kindvall, O. et al. Landscape properties and density dependence shape the movement patterns of three threatened butterflies. Landsc Ecol 39, 160 (2024). https://doi.org/10.1007/s10980-024-01963-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10980-024-01963-4