Abstract
Purpose
We conducted a feasibility study to investigate the use of ketogenic diets (KDs) as an adjuvant therapy for patients with glioblastoma (GBM), investigating (i) trial feasibility; (ii) potential impacts of the trial on patients’ quality of life and health; (iii) patients’ perspectives of their decision-making when invited to participate in the trial and (iv) recommending improvements to optimize future phase III trials.
Methods
A single-center, prospective, randomized, pilot study (KEATING), with an embedded qualitative design. Twelve newly diagnosed patients with GBM were randomized 1:1 to modified ketogenic diet (MKD) or medium chain triglyceride ketogenic diet (MCTKD). Primary outcome was retention at three months. Semi-structured interviews were conducted with a purposive sample of patients and caregivers (n = 15). Descriptive statistics were used for quantitative outcomes and qualitative data were analyzed thematically aided by NVivo.
Results
KEATING achieved recruitment targets, but the recruitment rate was low (28.6%). Retention was poor; only four of 12 patients completed the three-month diet (MCTKD n = 3; MKD n = 1). Participants’ decisions were intuitive and emotional; caregivers supported diet implementation and influenced the patients’ decision to participate. Those who declined made a deliberative and considered decision factoring diet burden and quality of life. A three-month diet was undesirable to patients who declined and withdrew.
Conclusion
Recruitment to a KD trial for patients with GBM is possible. A six-week intervention period is proposed for a phase III trial. The role of caregivers should not be underestimated. Future trials should optimize and adequately support the decision-making of patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glioblastoma (GBM) is the commonest malignant primary brain tumor in adults, affecting 2–3 per 100,000 per year [1]. Even with maximal safe resection, radiotherapy and temozolomide [2, 3], the prognosis remains poor [4]. In the last 10 years, a series of large-scale clinical trials testing targeted therapies have all reported negative results [4,5,6] with no change in the current standard of care. Patients and caregivers often explore other alternative treatment options, including dietary changes such as the ketogenic diet (KD). Indeed, the James Lind Alliance Priority Setting Partnership identified that ‘the effect of lifestyle factors, including diet, on tumor growth’ to be a top 10 research priority for the neuro-oncology community [5].
KD is an ‘umbrella term’ used to describe high fat, low carbohydrate, adequate protein diets which promote the utilization of fat for energy, in the form of ketones. Initially hypothesized to work by exploiting the ‘Warburg effect’ [6,7,8,9], newer theories have proposed other mechanisms of action, with ketone bodies and medium chain triglyceride (MCT) fats playing a role in tumor metabolism, rather than or in addition to a reduction in glucose [10,11,12,13]. Animal models of glioma have shown that KD potentiates the effects of radiotherapy [14], reduce peri-tumoral edema [15] and reduce tumor angiogenesis [7]. In patients with gliomas, the evidence for KD is limited to case studies and single case reports [16,17,18,19,20,21,22]; all utilizing different KDs at different time points in the treatment pathway. No studies have been powered to assess efficacy.
Prior to designing and undertaking an adequately powered randomized control trial (RCT) investigating the efficacy of KDs in the therapeutic management of GBM, feasibility must be demonstrated [23,24,25]. We conducted the KEATING study to i) investigate protocol feasibility; ii) explore the potential impact of the study on patients’ quality of life and health; iii) explore patients’ perspectives of their decision-making when invited to participate in the study; and iv) optimize future phase III clinical trial design, whilst comparing two different KDs in an NHS setting.
Methods
Study design
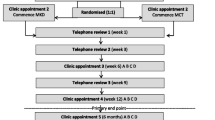
KEATING consisted of two parts; a pilot study and a qualitative study. To investigate the feasibility of KDs as an adjuvant therapy for patients with newly diagnosed GBM undergoing chemoradiotherapy a prospective, non-blinded, single-center, randomized pilot study was undertaken. Twelve patients were randomized in a 1:1 ration to either medium chain triglyceride ketogenic diet (MCTKD) or modified ketogenic diet (MKD). A three-month dietary intervention was planned (primary end point), following which patients could choose to continue with the diet for a total of 12 months (secondary end point). To explore the decision-making of patients’ invited to participate in KEATING a qualitative study was embedded, interviewing patients who participated and declined, along with their caregivers. Ethical approval was granted by North West-Greater Manchester West Research Ethics Committee (17/NW/0013). KEATING was registered with the International Standard Randomized Controlled Trial Number Registry (reference number 71665562) and ClinicalTrials.gov (reference number NCT03075514). The KEATING pilot study protocol has been published previously [26] (substantial amendments are detailed in the online resources).
Participants
Patients were recruited from a single adult neuroscience center. Patients were eligible if they were ≥ 16 years, ECOG performance status 0–2, had histologic diagnosis of GBM (WHO grade IV [27] within last four months (biopsy of surgical resection), were planned to undergo radiotherapy and temozolomide chemotherapy [2]. Patients were not eligible if they had any prior use of a KD, kidney, liver or gallbladder dysfunction, Metabolic or eating disorder, Body mass index (BMI) ≤ 18.5 kg/m2, were taking weight loss medications, pregnant or breastfeeding, or had Medical conditions that may increase risks associated with KD.
Randomization
Patients were randomized to either MCT KD or MKD using 'sealedenvelope'™ randomization system and a permuted block randomization method, ensuring similar numbers in each group, at a ratio of 1:1. This was constructed and administered by the study statistician (CTS), who was not involved with recruiting patients, thus concealing the sequence of allocation. Patients were then informed of their dietary intervention group by telephone and initiated diet within five working days of consent.
Dietary intervention and procedures
Two KDs were included in KEATING; MCTKD and MKD. A comparison of the macronutrient content, example meal plan and monitoring requirements for each diet can be found in Online Resource 1, Table A. Patients and their caregiver (if present) received dietary education from the dietician and were provided with a bespoke seven-day meal plan, recipes, dietary information sheets and food diaries. MCT was provided as Betaquik® (Vitaflo International Ltd), a nutritional product available by prescription. Patients were reviewed by telephone at weeks one, three and nine, and in an outpatient’s clinic at weeks six and twelve. Patients who wished to continue with the diet were then reviewed at six, nine and 12 months. Urinary ketones were monitored twice daily for the first six weeks, then weekly thereafter using Ketostix® (Bayer, Germany). Blood ketones and glucose levels were monitored once weekly using GlucoMen Aero 2 K® home monitoring kit (Abbott Laboratories, UK). All surgical and oncological interventions were undertaken as per current standard of care [28].
Outcomes
The primary outcome for KEATING was to estimate retention rate at three months to inform sample size calculations for future definitive trials. Secondary outcomes included estimations of recruitment rates, enrolment rates, long term retention rates and to obtain data on dietary compliance through food diaries, ketosis through ketone diaries, dietetic time to complete intervention, protocol refinements, completeness of data, quality of life assessed using EORTC QLQ C30 and BN20 questionnaires, food acceptability assessed through questionnaire, gastrointestinal side effects graded using Common Terminology Criteria for Adverse Event (CTCAE) reporting, biochemical markers (renal, bone, liver function tests, fasting lipid and fasting glucose) and anthropometry (weight, BMI, hand grip strength, mid arm muscle circumference, free fat mass and waist circumference. All outcomes were assessed at three months and twelve months.
Pilot success for KEATING was graded using a predetermined traffic light system (≥ 75% to proceed, ≥ 50% required review and < 50% study closure), which considered recruitment success, retention rates, dietary acceptability, the commencement of diet pre chemoradiotherapy and extent of missing data [29]. Magnetic Resonance Imaging (MRI) was used to interpret tumor progression. Progression free survival (PFS) was defined as the time from date of surgery randomization to date of recurrence on MRI. Recurrence was defined by a Neuroradiologist using the Response Assessment in Neuro-Oncology (RANO) criteria [30]. Overall survival (OS) was defined as the time from date of surgery to date of death from any cause. Adverse events (AE) and serious adverse events (SAE) were also reported as per CONSORT guidelines for pilot and feasibility studies [31].
Statistical analysis
Previous feasibility work estimated recruitment targets of one patient per month [32], in keeping with National Institute for Health Research (NIHR) funded trials [33]. Twelve patients were to be recruited over 12 months. Descriptive statistical methods were used and PFS and OS were PFS and OS were assessed Kaplan–Meier survival curves. During the course of the pilot study it was clear that retention on diet was an issue and with agreement from the Trial Steering Committee, a sub analyses was introduced at week six, with a view to providing further information which would inform the design of later trials. This was not included in the original study protocol (for amendments see Online Resource 2).
Qualitative study
Due to poor recruitment in the early stages of the KEATING study, we proceeded to amend the protocol to embed a qualitative component to explore patients’ decision-making about KEATING. Participants for the qualitative study were a purposively sampled sub-set of patients and their caregivers, who had been approached to participate in KEATING [34, 35]. Sampling was informed by the review of screening logs maintained as part of KEATING and aimed to include both those who consented and declined, those randomized to MCTKD and those to MKD. Patients were interviewed retrospectively, up to three months after being approached about KEATING. Adequate sample size was determined using the ‘information power’ concept [36, 37]. The interviews were conversational, patient-centered, topic guided (see Online Resource 3, Table A) and iterative. The topic guide was devised by two members of the research team (KM, GC). The researcher conducting the interviews had a dual role (dietician and qualitative researcher), therefore, interviews were reflexive and conducted in a gentle, sensitive and non-judging manner, to make the experience as comfortable as possible for patients. Interviews were audio-recorded and transcribed. Analysis drew on the Braun and Clarke thematic approach to identify patterns of meaning within the data [38]. KM lead a process of iterating between the developing analysis and new data (familiarization). Other members of the qualitative study team (BY and GC) read a sub-set of transcripts and developed the analysis by periodic discussion. Integration between KEATING and the embedded qualitative study took place after individual analysis had occurred. Integration was conducted by three authors (KM, GC, BY).
Results
KEATING participant characteristics
Between 1st April 2017 and 8th February 2018 we assessed 57 patients for eligibility. Fifteen were ineligible (26.3%), 30 declined (52.6%) and 12 (21.1%) were randomized. Of those recruited eight were male and four female, with a median age of 57 years (44–66 years). Figure 1 shows the patient flow through the study and Table 1 presents the demographic and clinical characteristics of patients who were randomized.
Primary outcome: retention at three months
Of the 12 patients randomized in KEATING (n = 6 MCTKD; n = 6 MKD), two withdrew prior to commencing the diet (n = 1 MCTKD; n = 1 MKD). Reasons for withdrawal were non-dietary related SAE (n = 1) and patient change of mind (n = 1). Of the 10 patients who commenced diet, six withdrew before reaching the three month primary end point (n = 2 MCTKD; n = 4 MKD). The median duration until discontinuing the MCTKD was 38 days (36–40 days; n = 2) and for MKD was 39.5 days (32–49 days; n = 4).
Secondary outcomes: protocol feasibility
Recruitment
Twelve patients were recruited over 12 months from a sample of 42 eligible patients, achieving a recruitment rate of 28.6% (or 21% of the overall screened population).
Long term retention
Of the 12 patients randomized in KEATING, four continued with their KD after month three (n = 3 MCTKD; n = 1 MKD). One patient (MCTKD group) then stopped at month six due to gastrointestinal side effects. In total, three patients completed the 12 month intervention period (n = 2 MCTKD; n = 1 MKD). These patients all chose to continue with their KD after completing the study.
Level of ketosis
During the first six weeks, 79.7% of MCTKD (n = 3) and 79.3% of MKD (N = 3) recordings were within the desired level of ≥ 4 mmol/L. Those who withdrew from the study reported lower urinary and serum ketone levels than those who stayed on diet up to month 12. The median level of urinary ketosis for each patient for their duration on diet is shown in online resource 3, figure A.
Secondary outcomes: impact of the study on patients’ health
Quality of life
At baseline, there was little difference between the Global Health Status (GHS) of those patients who went on to withdraw and those who continued with their diet and were retained within the study, in either dietary group (withdrew MCTKD 72.2 ± 20.7 [n = 3]; retained MCTKD 75 ± 6.8 [n = 3]; withdrew MKD 70 ± 13.8 [n = 5]; retained MKD 80 ± 0 [n = 1]).
The GHS of those who withdrew from the study at week six, fell below the brain cancer reference value in both the MCTKD and MKD groups (withdrew MCTKD 41.7 ± 0 [n = 1]; withdrew MKD 50 ± 0 [n = 2]). For those who continued with their diet and were retained within KEATING, GHS improved for the patient following MKD and reduced for those patients following MCTKD. In both groups the GHS remained above the brain cancer reference value (retained MCTKD week six 66.7 ± 0 [n = 3]; retained MCTKD month three 66.7 ± 13.6 [n = 3]; retained MCTKD month 12 66.7 ± 8.4 [n = 2]; retained MKD 100 ± 0 [n = 1] from week six onwards) (see online resource 3, figure B).
Food acceptability
Food acceptability reduced from baseline in both groups. The lowest food acceptability scores were recorded at week six of following the diet (baseline MCTKD 60.7 ± 10.5 [n = 6]; baseline MKD 54.3 ± 6.2 [n = 6]; week six MCTKD 42 ± 8.9 [n = 4]; week six MKD 43.5 ± 12.8 [n = 4]). Food acceptability then improved between week six and three months (MCTKD 49 ± 2.9 [n = 3]; MKD 58 [n = 1]), but reduced slightly before the end of the study (MCTKD 47.5 ± 6.5 [n = 2]; MKD 53 [n = 1]).
Adverse and serious adverse events
There were five adverse events and three serious adverse events. Adverse events were due to hypokalemia (n = 2, CTCAE grade 1), hypernatremia (n = 1, CTCAE grade 1), hypocalcaemia (n = 1, not classified as adjusted calcium > 2 mmol/L) and a partial seizure (n = 1, CTCAE 1). Serious adverse events were due to post-operative wound infection (n = 1, CTCAE grade 3, resulting in withdrawal from the assigned dietary intervention), seizure (n = 1, CTCAE grade 2) and back pain (n = 1, CTCAE grade 2), none of which were related to the dietary intervention.
Survival analysis
The median time to progression was 14.4 weeks (SE 14.6; 95% CI 0–42.9 weeks). Median overall survival was 67.3 weeks (SE 6.2; 95% CI 55–79.6 weeks). Survival analysis is illustrated in Fig. 2.
Additional outcomes
Additional outcome reporting as per the KEATING protocol can be located in online resource 3 for the following outcomes: enrolment of participants prior to, during and post chemoradiotherapy commencement; dietary compliance; dietary adjustments required to achieve ketosis; dietetic time required for dietary interventions; gastrointestinal side effects; changes to biomarkers; anthropometric changes; and determining pilot success.
Qualitative study
Participant characteristics
Fifteen patients and their caregivers were invited to be interviewed. Between January and April 2018, 10 patients and five of their caregivers, all of whom were white British, were interviewed (Table 2). All participants were interviewed separately except one patient and one relative who were interviewed jointly. Individual interviews lasted for an average (median) of 44 min (36–62 min) and the dyad interview lasted 65 min.
Integrated results of KEATING and the embedded qualitative study
Table 3 integrates the findings of the KEATING and the embedded qualitative study, using an adapted triangulation protocol [39]. Throughout the table, we present patients’ verbatim quotes in speech marks, with ellipses indicating missing text and square brackets indicating replacement or explanatory text. To preserve anonymity, all patients are identified by the patient’s KEATING study screening log number (e.g. T01) and caregivers by an associated number (e.g. T01/R). We highlighted if the results of the two studies converge, are complementary, are contradiction, or are silent [39]. Further detailed analysis of the qualitative study can be found via online resource 4.
Discussion
This randomized, pilot study with an embedded qualitative design was designed to explore the feasibility of KD trials for patients with glioblastoma, with a view to recommending improvements to optimize the design of future phase III trials.
KEATING recruited to time and target, despite an initial slow start. The recruitment rate (28.6% of the eligible population) was much lower than NIHR HTA funded oncology clinical trials (50 to 89%) [33], but in keeping with recent survey data from the National Brain Tumour Society, with 21% of patients with brain tumors participating in clinical trials [40, 41]. Screening log data at the start of KEATING revealed that patients were declining to participate due to (i) not wanting to participate in research; (ii) the burden of dietitian visits; and (iii) the burden of KD. During the qualitative study those patients who declined to participate in KEATING identified their quality of life as an important factor in decision-making, and this aspect was not detected in the screening log data or in previous surveys regarding participation barriers [40]. These patients also spoke of the role of caregivers in influencing their decision to participate or not in KEATING, an aspect highlighted in trials elsewhere [41]. In contrast, patients who consented to KEATING made an intuitive and emotional decision, later reflecting that this decision was based upon quantity rather than quality of life. This optimism surrounding longevity of life is often used as a coping strategy by patients and may not invalidate their informed consent. There is currently little guidance offered by the Health Research Authority (HRA) Good Clinical Practice guidelines [42] regarding this matter, thus clinicians should continue to use their clinical judgement when assessing patients’ informed consent to participate in trials. Our findings are in keeping with recent publications highlighting the need for improved recruitment strategies and decisional support in neuro-oncology populations [40, 41].
The retention rate in KEATING was lower than anticipated. Out of the 12 patients randomized, 10 commenced KD and only four met the primary endpoint of three-month dietary intervention (1 KD, 3 MKD). Cancer trials in general report median retention rates of 89% (IQR 79–97% with valid primary outcome data at follow up) [33] and previous KD studies for patients with GBM report retention rates of 50 to 100%, with retention determined at eight weeks [19], three months [18, 32] and the point of tumor progression [17]. However, patients in these previous KD studies self-selected to try the diet, mainly at recurrence or post-treatment, creating an optimistic bias in retention, when compared to the general unselected GBM population approached for KEATING. Those who withdrew from KEATING did so either after randomization but prior to commencing the diet (n = 2) or after following the KD for approximately six weeks (MCTKD median 38 days [36 to 40 days], n = 2); MKD 39.5 days [32 to 49 days], n = 4), during which time patients were undergoing radiotherapy and concomitant temozolomide chemotherapy. Patients reported their reasons for withdrawal to be related to dietary burden and side effects, in particular nausea, which could have been related to the chemotherapy. Those continued on their assigned KD were generally younger patients with more favorable prognostic features (MGMT methylated; IDH-1 mutant) and this may also have influenced their ability to stay on the study and implement the diet.
The reasons for poor retention on diet were explored in our qualitative study. Those who withdrew spoke of finding their low urinary and blood ketones to be ‘demoralizing’, feeling that the diet was not working, and withdrawing due to the negative effect this feeling had on their quality of life. This was confirmed in the quality of life data as patients who withdrew reported their global health status (GHS) to be below the brain cancer reference value at week six of the study. We appreciate multiple factors can affect the quality of life for these patients and whilst ketones are used to monitor the diet, urinary ketones not always robust markers of compliance and can be effected by hydration levels and the use of dexamethasone. Therefore low ketones may demoralize patients, even when they appear to be following the diet robustly.
For those patients who continued to participate in the trial to 12 months, GHS reduced within the MCTKD group and improved in the MKD group. However, during the qualitative interviews, both groups reported to experiencing a ‘fantastic quality of life’ describing the diet as offering a sense of ‘control’ whilst receiving their tumor treatment. Although the EORTC QLQC30 and BN20 questionnaires are validated for patients with glioblastoma, they are time consuming to complete and some questions are not relevant for patients following KDs. It may be beneficial for future KD trials to reduce the length of the questionnaire and therefore patient burden, focusing particularly on GHS, as these questions provided the most insight in KEATING.
During interviews, patients reported several motivational factors for continuing with the diet, including through online blogs of long term glioblastoma survivors, positive MRI results and high ketone levels, using these as a means of validating their decision to stay on diet. This corroborates the findings from KEATING since high ketones indicted compliance with KD. Patients with higher ketones stayed in the study, whilst those with lower ketones withdrew early, at around week six. Furthermore, a pilot study for KD in patients with other advanced cancers (breast, ovarian, lung, gastrointestinal), also experienced similar retention rates to KEATING (retention rate 31%, n = 5 of 16), in a trial which permitted a more liberal KD (70 g of carbohydrates per day) and where all food provision was provided [43]. Thus, a more flexible dietary approach may not be the simple solution.
The high withdrawal rates in KEATING suggest that a three-month KD intervention may be too long for most patients. Our qualitative study also highlighted that those who withdrew considered the three-month intervention to be undesirable, an opinion also reflected by those who declined, further corroborating findings from KEATING. A shorter, six-week intervention, is likely to be more tolerable and acceptable to patients. This could be offered alongside radiotherapy and concomitant chemotherapy, which coincides with the proposed optimal time for the diet derived from animal model data [14]. Offering the diet at the same hospital site as the radiotherapy would aid a timely start of the diet. Whilst patients reported ‘feeling free again’ once the diet was discontinued, it is important to note, that this may not necessarily equate to dietary acceptability outside of a clinical trial. In a future post-trial environment, information regarding the efficacy of the diet may be available, which could alter patients’ willingness to engage.
The qualitative study also highlighted caregivers to be key in supporting patients to implement the diet. The role of caregivers, both in the decision-making of patients and the ongoing support offered, were aspects that were underappreciated in KEATING. A recent KD study for patients with Alzheimer’s disease, also highlighted dietary and caregiver burden to be influential over patient withdrawal [44].
KEATING had several limitations. The return rate of food and ketone diaries was low at 12 months, subsequently affecting the analysis. This is a common problem in dietary intervention trials, given the time commitment required to return diaries. All accounts of dietary intake were also self-reported and at risk of reporter bias. The sample size for the qualitative was small and we cannot be certain that saturation was achieved. Nevertheless, drawing on the concept of ‘information power’ in qualitative research [36], this study had a well specified aim, and it has provided insights that will be valuable in informing a future phase III trial. Some patients were also interviewed up to three months after their decision about KEATING. They may have found it difficult to accurately recall their decision-making process, particularly given the nature of their condition and the numerous other decisions they will likely have had to make regarding their care and treatment.
In order to optimize protocol feasibility and patient experience future trials should consider the following suggestions:
-
To assess effectiveness in a phase III trial a six–week diet intervention period would be deliverable.
-
To optimize recruitment and retention a longitudinal, prospective, qualitative study, which focuses on patient and caregivers understanding and decision-making in the context of trial participation should be embedded within KD trials.
-
Future phase III trials would benefit from an internal pilot to further test the recommendations derived from KEATING, focusing on stop/ go criteria for staged recruitment, retention at 6-weeks and commencement of diet prior to chemoradiotherapy.
In conclusion, recruitment of patients with GBM to a KD trial is possible. To assess efficacy in a phase III clinical trial, a six-week intervention period is proposed. The role of caregivers in the patients’ decision-making process and in supporting patients to implement KDs should not be underestimated.
Data availability
The datasets generated and analysed during the current study are available at the University of Liverpool repository https://doi.org/10.17638/datacat.liverpool.ac.uk/692
References
Radhakrishnan K, Mokri B, Parisi JE, O’Fallon WM, Sunku J, Kurland LT (1995) The trends in incidence of primary brain tumors in the population of rochester, minnesota. Ann Neurol 37(1):67–73
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996. https://doi.org/10.1056/NEJMoa043330
Weller M, van den Bent M, Hopkins K, Tonn JC, Stupp R, Falini A et al (2014) EANO guideline for the diagnosis and treatment of anaplastic gliomas and glioblastoma. Lancet Oncol 15(9):e395–403
Krex D, Klink B, Hartmann C, Von Deimling A, Pietsch T, Simon M et al (2007) Long-term survival with glioblastoma multiforme. Brain 130(10):2596–2606
Macdonald L (2015) Top 10 priorities for clinical research in primary brain and spinal cord tumours: Final report of the James Lind Alliance Priority Setting Partnership in Neuro-Oncology. www.neuro-oncology.org
Seyfried TN, Kiebish MA, Marsh J, Shelton LM, Huysentruyt LC, Mukherjee P (2011) Metabolic management of brain cancer. Biochim Biophys Acta 1807(6):577–594
Zhou W, Mukherjee P, Kiebish M, Markis W, Mantis J, Seyfried T (2007) The calorically restricted ketogenic diet, an effective alternative therapy for malignant brain cancer. Nutr Metab 4:5
Tisdale MJ, Brennan RA (1983) Loss of acetoacetate coenzyme A transferase activity in tumours of peripheral tissues. Br J Cancer 47(2):293–297
Allen BG, Bhatia SK, Anderson CM, Eichenberger-Gilmore JM, Sibenaller ZA, Mapuskar KA et al (2014) Ketogenic diets as an adjuvant cancer therapy: History and potential mechanism. Redox Biol 2:1
Otto C, Kaemmerer U, Illert B, Muehling B, Pfetzer N, Wittig R et al (2008) Growth of human gastric cancer cells in nude mice is delayed by a ketogenic diet supplemented with omega-3 fatty acids and medium-chain triglycerides. BMC Cancer 8:122
Hao G-W, Chen Y-S, He D-M, Wang H-Y, Wu G-H, Zhang B (2015) Growth of human colon cancer cells in nude mice is delayed by ketogenic diet with or without omega-3 fatty acids and medium-chain triglycerides. Asian Pacific J Cancer Prev 16(5):2061–2068
Martuscello RT, Vedam-Mai V, Mccarthy DJ, Schmoll ME, Jundi MA, Louviere CD et al (2015) Cancer Therapy: Preclinical A Supplemented High-Fat Low-Carbohydrate Diet for the Treatment of Glioblastoma. Clin Cancer Res 22(10):2482–2495
Aminzadeh-Gohari S, Feichtinger RG, Vidali S, Locker F, Rutherford T, O’Donnel M et al (2017) A ketogenic diet supplemented with medium-chain triglycerides enhances the anti-tumor and anti-angiogenic efficacy of chemotherapy on neuroblastoma xenografts in a CD1-nu mouse model. Oncotarget 8(39):64728–64744
Abdelwahab MG, Fenton KE, Preul MC, Rho JM, Lynch A, Stafford P et al (2012) The ketogenic diet is an effective adjuvant to radiation therapy for the treatment of malignant glioma. PLoS ONE 7(5):e36197. https://doi.org/10.1371/journal.pone.0036197
Woolf EC, Scheck AC (2015) The ketogenic diet for the treatment of malignant glioma. J Lipid Res 56(1):5–10. https://doi.org/10.1194/jlr.R046797
Martin-McGill KJ, Srikandarajah N, Marson AG, Tudur Smith C, Jenkinson MD (2018) The role of ketogenic diets in the therapeutic management of adult and paediatric gliomas: a systematic review. CNS Oncol 7(2):CNS17.
Rieger J, Bähr O, Maurer GGD, Hattingen E, Franz K, Brucker D et al (2014) ERGO: A pilot study of ketogenic diet in recurrent glioblastoma. Int J Oncol 45(6):1843–1852. https://doi.org/10.3892/ijo.2014.2382
Schwartz K, Chang HT, Nikolai M, Pernicone J, Rhee S, Olson K et al (2015) Treatment of glioma patients with ketogenic diets: report of two cases treated with an IRB-approved energy-restricted ketogenic diet protocol and review of the literature. Cancer Metab 3(3):1–10
Nebeling LC, Miraldi F, Shurin S, Lerner E (1995) Effects of a ketogenic diet on tumor metabolism and nutritional status in pediatric oncology patients: two case reports. J Am Coll Nutr 14(2):202–208
Champ CE, Palmer JD, Volek JS, Werner-Wasik M, Andrews DW, Evans JJ et al (2014) Targeting metabolism with a ketogenic diet during the treatment of glioblastoma multiforme. J Neurooncol 117(1):125–131. https://doi.org/10.1007/s11060-014-1362-0
Strowd RE, Cervenka MC, Henry BJ, Kossoff EH, Hartman AL, Blakeley JO (2015) Glycemic modulation in neuro-oncology: experience and future directions using a modified Atkins diet for high-grade brain tumors. Neuro-Oncol Pract 2(3):127–136. https://doi.org/10.1093/nop/npv010
Zuccoli G, Marcello N, Pisanello A, Servadei F, Vaccaro S, Mukherjee P et al (2010) Metabolic management of glioblastoma multiforme using standard therapy together with a restricted ketogenic diet: case Report. Nutr Metab (Lond) 7(33):1–7. https://doi.org/10.1186/1743-7075-7-33
Moore C, Carter RE, Neitert PJ, Stewart PW (2012) Recommendations for planning pilot studies in clinical and translational research. Clin Transl Sci. 4(5):332–337
Eldridge SM, Lancaster GA, Campbell MJ, Thabane L, Hopewell S, Coleman CL et al (2016) Defining feasibility and pilot studies in preparation for randomised controlled trials: development of a conceptual framework. PLoS ONE 11(13):1–22
National Institute for Health Research. Feasibility and Pilot studies. 2019. https://www.nihr.ac.uk/funding-and-support/documents/funding-for-research-studies/research-programmes/PGfAR/FeasibilityandPilot studies.pdf. Accessed on 06 Mar 2019
Martin-McGill KJ, Marson AG, Tudur Smith C, Jenkinson MD (2017) Ketogenic diets as an adjuvant therapy in glioblastoma (the KEATING trial): study protocol for a randomised pilot study. Pilot Feasibility Stud 3(1):67. https://doi.org/10.1186/s40814-017-0209-9
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK et al (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131(6):803–820
Jenkinson M. Walton Centre / Clatterbridge Centre for Oncology guidelines for high grade glioma management. Merseyside and Cheshire Neuro-Oncology Clinical Network Group. Liverpool, UK; 2011. https://www.nwcscnsenate.nhs.uk/files/7114/1232/7814/High_grade_glioma_guidelines_November_2013.pdf
Avery KNL, Williamson PR, Gamble C, O’Connell Francischetto E, Metcalfe C, Davidson P et al (2017) Informing efficient randomised controlled trials: exploration of challenges in developing progression criteria for internal pilot studies. BMJ Open 7(2):e013537
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28(11):1963–1972
Eldridge SM, Chan CL, Campbell MJ, Bond CM, Hopewell S, Thabane L et al (2010) CONSORT statement: extension to randomised pilot and feasibility trials. BMJ 2016:355
Martin-McGill KJ, Marson AG, Tudur Smith C, Jenkinson MD (2018) The modified ketogenic diet in adults with glioblastoma: an evaluation of feasibility and deliverability within the national health service. Nutr Cancer 70(4):643–649
Walters SJ, Bonacho I, Henriques-Cadby A, Bortolami O, Flight L, Hind D et al (2017) Recruitment and retention of participants in randomised controlled trials: a review of trials funded and published by the United Kingdom Health Technology Assessment Programme. BMJ Open 7:e015276
Bryman A (2016) Mixed methods research: combining quanitative and qualitative research. In: Social research methods. 5th ed. Oxford University Press, Oxford, pp 635–687
Emmel N (2013) Sampling and choosing cases in qualitative research : a realist approach, 1st edn. Mental Health Foundation, London, pp 33–44
Malterud K, Siersma VD, Guassora AD (2016) Sample size in qualitative interview studies: guided by information power. Qual Health Res 26(13):1753–1760. https://doi.org/10.1177/1049732315617444
Sim J, Saunders B, Waterfield J, Kingstone T (2018) Can sample size in qualitative research be determined a priori? Int J Soc Res Methodol 21(5):619–634. https://doi.org/10.1080/13645579.2018.1454643
Braun V, Clarke V (2006) Using thematic analysis in psychology. Qual Res Psychol 3(2):77–101
O’Cathain A (2018) A practical guide to using qualitative research with randomized controlled trials, 1st edn. Oxford University Press, Oxford
Bates AJ, Couillard SA, Arons DF, Yung WKA, Vogelbaum M, Wen PY et al (2017) Hout 15. Brain tumor patient and caregiver survey on clinical trials: identifying attitudes and barriers to patient participation. Neuro Oncol 19(suppl_6):vi109–vi109
Lee EQ, Chukwueke UN, Hervey-Jumper SL, de Groot JF, Leone JP, Armstrong TS et al (2019) Barriers to accrual and enrollment in brain tumor trials. Neuro Oncol 21(9):1100–1117. https://doi.org/10.1093/neuonc/noz104/5513026
Health Research Authority. Good Clinical Practice. 2018. https://www.hra.nhs.uk/planning-and-improving-research/policies-standards-legislation/good-clinical-practice/. Accessed 05 Aug 2019
Schmidt M, Pfetzer N, Schwab M, Strauss I, Kämmerer U (2011) Effects of a ketogenic diet on the quality of life in 16 patients with advanced cancer: a pilot trial. Nutr Metab (Lond) 8(1):54. https://doi.org/10.1186/1743-7075-8-54
Taylor MK, Sullivan DK, Mahnken JD, Burns JM, Swerdlow RH (2018) Feasibility and efficacy data from a ketogenic diet intervention in Alzheimer’s disease. Alzheimer’s Dement Transl Res Clin Interv 4:28–36. https://doi.org/10.1016/j.trci.2017.11.002
Acknowledgements
The authors would like to thank the patients and caregivers who took part in the study for sharing their experiences and Mr Paul Brennan, Trial Steering Committee Chair and Dr Matthew Williams, Trial Steering Committee external participant.
Funding
KJM received a PhD studentship from Vitaflo (International) Ltd. MDJ and AGM received salary costs from Vitaflo (International) Ltd, via University of Liverpool.
Author information
Authors and Affiliations
Contributions
KJM: Contributed to the conception and design of the project, the analysis and interpretation of data and the drafting of the manuscript. MDJ, AGM, CTS: Contributed to the conception and design of the project, the analysis and interpretation of data and the review of the manuscrip. MGC, BY: Contributed to the design of the qualitative embedded study, analysis and the review of the manuscript. SJM: Contributed to the data analysis and review of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
KJM received a PhD studentship from Vitaflo (International) Ltd.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standard of the institutional and nation research committee (North West-Greater Manchester West Research Ethics Committee [17/NW/0013]) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
M. Gemma Cherry and Michael D. Jenkinson are joint last author.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martin-McGill, K.J., Marson, A.G., Tudur Smith, C. et al. Ketogenic diets as an adjuvant therapy for glioblastoma (KEATING): a randomized, mixed methods, feasibility study. J Neurooncol 147, 213–227 (2020). https://doi.org/10.1007/s11060-020-03417-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-020-03417-8