Abstract
Background and aims
The ability of grass mulches to inhibit weed performance has been linked to their limitations on nitrogen availability to the weeds. Fungal translocation of N from the soil to the surface mulch has been confirmed in laboratories, but this mechanism has not been documented under field conditions.
Methods
Experiments used 15N (NH4)2SO4 , 99.7 at.%, which was uniformly injected below the soil surface at a rate of 1 mg 15 N kg−1 soil. Some plots were treated with a fungicide (Captan) every 2 weeks after injection, while others were not treated. Nitrogen transfer was monitored by measuring levels in surface residue, soybean tissue, and extractable soil inorganic N pools.
Results
Despite the N release from the cereal rye (Secale cereale L.) tissues ranging from 15 to 50 kg N ha−1, there was a detectable increase in 15N enrichment of 10–15 % in the cereal rye tissue. Six weeks after injection, tissue from the plots not treated with fungicide contained 36 % more 15 N.
Conclusions
The increased 15N enrichment in the cereal rye mulch supports laboratory observations that soil inorganic N is translocated into surface mulch via fungal mechanisms. These findings illustrate microbial-mediated sinks for nitrogen in cereal rye mulches in no-till soybean production systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
To manage weed pressures, organic soybean production relies on costly and labor-intensive production practices that utilize frequent tillage and cultivation techniques such as blind cultivation; between-row cultivation; and expensive, low-efficacy organic herbicides. Organic soybean systems that employ intensive cultivation practices can substantially reduce weed-driven yield loss (Smith et al. 2011; Reberg-Horton et al. 2012; Wells et al. 2013). However, these practices come at an environmental and economic cost. Excessive soil cultivation can compact the sub-soil layer, increasing water and wind erosion, increasing soil organic matter decomposition, and causing higher labor and energy inputs (Bernstein et al. 2011).
The time, labor, and economic costs of production have prompted interest in reducing tillage in organic soybean production. A cover crop-based approach to producing organic soybeans without tillage is being developed to address these costs. In this case, a grass cover crop is roll-crimped, producing weed-suppressive mulch that soybean cash crops are planted into. Coupling a grass cover crop with a legume cash crop (i.e., cereal rye and soybeans) can produce yields equivalent to those from conventional no-till soybean production (Smith et al. 2011; Reberg-Horton et al. 2012; Mirsky et al. 2013). Current research suggests that the success of this system depends on maximizing cover crop biomass production (Smith et al. 2011; Wells et al. 2013).
It is understood that cover crop mulches suppress weeds physically and chemically (Teasdale and Mohler, 2000; Reberg-Horton et al. 2005; Reberg-Horton et al. 2012). Physically, cover crop mulches provide a restrictive barrier that reduces temperatures and light at the soil surface, thus inhibiting the germination and emergence of small-seeded summer annual weeds. Cover crop mulches also suppress the germination of weeds through the release of phytotoxic allelopathic chemicals (Reberg-Horton et al. 2005).
To achieve maximum cereal rye biomass production in sites where N carryover from previous crops is minimal in lighter-textured soil systems, producers apply composted manures both before fall planting and in the spring before jointing to deliver N fertility. Manure rates are determined in concert with tissue testing to ensure optimal nutrient conditions throughout the growing season. As a result, many producers in the Southeast can achieve 9000–10,000 kg ha−1 dry matter over the season (Smith et al. 2011; Mirsky et al. 2013).
While physical factors, and to a far lesser extent allelopathy, appear to be the principal factors driving weed suppression via mulches, nutrient availability may also play an important role. Wells et al. (2013) observed low soil inorganic N levels under a cover crop-based, no-till soybean crop; there appeared to be a relationship between weed performance and low soil inorganic N levels. In these systems, plant-available N wasn’t just lower at the beginning of the soybean season — N release was also suppressed throughout the season (Wells et al. 2013). The lower plant-available N in this high-residue system suggests that nitrogen limitation may enhance overall weed control.
The nutritive control mechanism, which refers to suppression induced by low N availability, has not been well studied in roll-crimped, cover crop-mulched systems (Mirsky et al. 2013; Wells et al. 2013). What has been studied are the effects of incorporating high C:N residues with tillage and the subsequent microbial N immobilization—a process in which N is sequestered by microbes and incorporated into proteins and other organic N constituents (Schomberg et al. 1994; Recous et al. 1995; Burgess et al. 2002; Jin et al. 2008). Research evaluating high C/N cover crop surface residue impacts on N immobilization in agricultural systems is sparse (Marinari et al. 2010). Nitrogen immobilization effects have been reported when high C/N ratio wheat straw residues (ranging from 40 to 80:1) were applied to soil surfaces (Holland and Coleman 1987; Schwendener et al. 2005).
The mechanism of N transfer from soil to surface residues was directly confirmed by (Frey et al. 2000). Frey et al. (2000) demonstrated a 52 to 86 % reduction in total N transferred from mineral soil to wheat surface residues when fungal populations were inhibited by fungicides. Similar N immobilization is likely to occur in agricultural systems with high C/N surface residues. Determining the extent to which fungal-mediated N translocation occurs would help explain the magnitude of microbial competitive nutritive weed suppression mechanisms.
The objective of this study was to characterize, in situ, the magnitude of the fungal-mediated N transfer, from the soil-N pool to the surface of the cereal rye cover crop mulch. We hypothesize that fungal-mediated N transfer does occur under field conditions, and that greater 15N enrichment will be detected in the rye surface mulches not treated with fungicides.
Materials and methods
Study location and soil properties
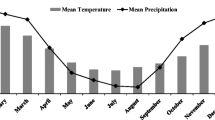
In 2010, two independent experiments were conducted at both the Caswell Research Farm (KINS) in Kinston, NC (35.273206° N, −77.623816° W), and USDA-ARS Beltsville Agricultural Research Center (BARC) in Beltsville, MD (39.03269° N, −76.928143° W). The soil types at KINS were a Johns loamy sand (fine-loamy over sandy or sandy-skeletal, siliceous, semiactive, thermic Aquic Hapludults) with 0 to 2 % slope and a Kenansville loamy sand (loamy, siliceous, subactive, thermic Arenic Hapludults) with 0 to 3 % slope. At BARC the soil types were a Codorus and Hatboro (mica-bearing loamy alluvium), with 0 to 2 % slope. Daily weather information (e.g., air temperature and precipitation) was tracked at both locations (Fig. 1).
Experimental design and macro system descriptions
We utilized two study designs because they have different strengths and weaknesses. A single injection time (i.e., a pulse) is typical in much of the ecological literature and is far easier to implement. That was one method used. However, with only one injection time, a large rain can end the study by moving nitrogen too deep in the soil profile. Furthermore, over a long enough period, the isotope changes pools multiple times, confusing the interpretation of current exchange rates amongst N sinks. Therefore, we also included a staggered injection strategy (i.e., a series of pulse injections) to mitigate the risk of losing the entire study to a heavy rain and to provide more insight into the exchange rates of the nitrogen isotope among N sinks.
Both experiments utilized factorial arrangements comprised of (F) with fungicide and (NF) without fungicide, as well as sampling times measured in days after soybean planting (DAP). The Staggered Injection Study (StIS) was a randomized complete block design, and the Single Injection Study (SIS) was a split-plot design with main plots arranged in a randomized complete block design. In the SIS study, the main plot factors consisted of fungicide treatments and the split-plot factor was sampling time. In the StIS experiment, sampling time was crossed with the fungicide treatments. Each experiment had four replications.
Prior to fall drilling of cereal rye (Secale cereale L. ‘Rymin’ or ‘Aroostook’), the entire field at both locations was disked and field-cultivated to incorporate any existing vegetation from the previous maize crop. Lime, P, and K were broadcast according to soil test results before cereal rye planting (Table 1). The cereal rye cover crop was sown during mid-October for all treatments using a grain drill with 13-cm row spacing and 2.54-cm depth at 134 kg ha−1. In the fall, approximately 30 kg N ha−1 of urea ammonium nitrate (UAN) was applied to the cereal rye cover crop to ensure adequate fall tillering and optimal growth in the spring.
The microplots for StIS and SIS consisted of 50 steel boxes that were 0.58 m2 (0.76 m × 0.76 m × 0.42 m) and were 6.5 mm thick. Before the microplots were installed, the cereal rye cover crop was terminated via a mower conditioner haybine in early June (Feeke’s growth stage 11.1 at KINS and 11.4 at BARC). The cut cereal rye was removed from the experiment area, where it was air-dried for one week. Following the removal of the cereal rye cover crop, the 50 steel boxes were installed directly into the cereal rye cover crop stubble to a depth of 40 cm. Soybeans (Glycine max L. ‘Pioneer 95Y40), Maturity Group V, were hand sown at a rate of 370,650 live seed ha−1 with 76.2 cm row spacing (approximately 30 plants m−1). Following soybean planting, cereal rye biomass was applied to each microplot, at 8000 kg ha−1 dry matter. The cereal rye biomass was separated over the soybean furrow to ensure maximum soybean emergence and then thinned to 30 plants m−1 (i.e., 23 soybean plants per microplot). The weed species at both sites was redroot pigweed (Amaranthus retroflexus L.), and microplots were managed weed-free throughout the season with two broadcast applications of glyphosate.
Single injection study
Temporal distribution of N was evaluated in the SIS through a single pulse enrichment of 15N-enriched (NH4)2SO4 solution (99.7 at.%) at a rate of 1 mg 15 N kg−1 soil uniformly injected across the microplot. After the soybeans were no-till planted into cereal rye stubble, the previously harvested and dried cereal rye mulch was placed on the microplots. Enriched 15N (NH4)2SO4 solution (99.7 at.%) was uniformly injected in 5 ml increments across the microplot via a 6-gauge needle to a depth of 4.4 cm below the soil surface (Fig. 2). To ensure uniform distribution of the 15N tracer, the microplots were spatially divided into 25 grids of 231 cm2 where a total of 75 injections, three per grid, delivered 375 ml of 15N enriched solution for both experiments (Fig. 2).
(Left) Injection of 15(NH4)2SO4 (99.7 at.%) via a 6-ga needle to a depth of 4.4 cm below the soil surface. (Right) Each microplot was divided into a five-by-five grid of 231 cm2 per grid. Each grid received three injections of 15N enriched solution for a total of 75 injections that delivered 375 ml per microplot. This approach was used for both the staggered and single injection studies
Following the enrichment in the SIS, (0 DAP), the cereal rye tissue was collected from the 0 DAP microplots (i.e., whole plot, no subsampling) and the level of 15N enrichment in the cereal rye tissue was assessed. After the cereal rye biomass was removed according to the assigned time interval, soils were sampled and tested for 15N. Soil and cereal rye tissue were sampled at soybean planting and again four and eight weeks after the enrichment. The rye mulch collected from the entire microplot was dried in a forced-air oven at 60 °C for two weeks. After drying, the entire rye sample was milled. This was the material analyzed for 15N, total C and N, and fungal qPCR. Soybean tissue was sampled four and eight weeks after planting and was processed like the rye tissues were.
Staggered injection study
Spatial distribution of N was evaluated in the StIS through four injection intervals of 15N-enriched (NH4)2SO4 solution (99.7 at.%) at a rate of 1 mg 15 N kg−1 soil. The same injection protocol described in the SIS experiment was used in the StIS experiment (Fig. 2). For each microplot under StIS (i.e. experimental unit), there was only one 15N injection event occurring two weeks before destructive harvesting of all soil and plant parameters. Injections of 15N began at soybean planting, and were repeated in a different microplot every two weeks over an eight-week period (i.e., t = 0, 2, 4, and 6). No StIS microplot (i.e experimental unit) received only one injection event. The injections occurred every two weeks to avoid label loss due to a significant rain and allowed for higher resolution, albeit ephemeral, on 15N fate. Following the procedure outlined in the SIS experiment, 15N was injected below the soil surface to avoid capillary movement of label to the surface residues.
Fungicide treatments
Half the plots received fungicide treatments and others did not. Following the soybean planting, cereal rye biomass application, and 15N injection, all plots received 0.6 cm of water as simulated rainfall for each sampling and for each 15N injection interval throughout the study. For the fungicide treatments, Captan (50 W wettable powder, 48.9 % a.i., 4.6 % N) was delivered in the 0.6 cm of simulated rainfall at a rate of 9 kg ha−1. This was equivalent to 4.4 kg ha−1 a.i. to each of the ‘fungicide-treated’ plots starting at the initial 15N injection and planting of soybeans (i.e., t = 0). The fungicide application continued for six weeks, on two-week intervals, for a total of 17.6 kg ha−1 a.i. microplot−1. The goals of the fungicide applications were similar to those reported by (Frey et al. 2000), that is, to reduce fungal populations while having minimal impacts on nontarget organisms (Ingham and Coleman 1984; Ingham et al. 1986; Beare et al. 1992). Fungicide application rates were chosen to not exceed 1 mg N kg−1 soil, to minimize perturbation of the N status in the soil system.
Soil and plant measurements
For each sampling interval, the following soil and crop parameters were examined to monitor 15N movement: cereal rye mulch residue, soybean plant tissues, and soil extractable inorganic N. Cereal rye biomass and soybean tissues (i.e., above-ground plant tissue) were collected from the entire microplot and air-dried at 60 °C for 72 h. After drying, weights were recorded, plant tissues were finely milled (i.e., ball-ground), and tissues were analyzed for total N and C by a PerkinElmer (Norwalk, CT) Model 2400 CHN elemental analyzer (Tu et al. 2006; Wells et al. 2013). Ball-ground plant tissue was then packed into 9 × 5 mm tin capsules and sent to the Stable Isotope Facility at the University of California at Davis to be analyzed for N isotope ratios using a PDZ Europa ANCA-GSL elemental analyzer interfaced to a PDZ Europa 20–20 isotope ratio mass spectrometer (Sercon Ltd., Cheshire, UK).
Soil inorganic N was monitored by collecting 16 destructively harvested, equally spaced, (7.6 cm dia.) soil cores 10 cm deep. Bulk density and soil moisture were also measured during each sampling interval. Soil samples were homogenized, wet sieved (4 mm) to remove any plant material and rocks, and placed in cold (4 °C) storage for transport. Twenty grams of field moist-sieved soil was extracted in 50 ml of 0.5 M K2SO4 and shaken for 30 min on an orbital shaker (Hart et al. 1994; Frey et al. 2000). After samples had been shaken, the soil suspension was filtered with preleached filter papers (100 ml 0.5 M K2SO4) (Fisher Scientific Filter Paper Grade Q2). The filtered soil extractions were analyzed for ammonium and nitrate on a Quick Chem 8000 Lachat (Keeney and Nelson 1982). To determine the 15N enrichment in the soil, samples were diffused on to filter paper acid traps according to protocols outlined by Stark and Hart (Stark and Hart 1996). Diffused samples were transferred into 9 × 5 mm tin capsules and sent to the University of California at Davis to be analyzed for N isotope ratios using a PDZ Europa ANCA-GSL elemental analyzer interfaced to a PDZ Europa 20–20 isotope ratio mass spectrometer (Sercon Ltd., Cheshire, UK) (Brooks et al. 1989; Stark and Hart 1996; Frey et al. 2000).
DNA extraction and fungal ITS1 region primers performed on cereal rye surface residues
Rye mulch from all mulch treatments and dates was subsampled from the milled tissues. Twenty grams of ball-ground rye material was subsampled for metagenomic DNA extractions. Metagenomic DNA was extracted from 1 g of dried and ground cereal rye mulch using the PowerPlant DNA Isolation Kit (MoBio Laboratories, Solana Beach, CA) according to the manufacturer’s instructions. DNA concentration was determined using a NanoDrop 2000 spectrophotometer at 260 nm (Thermo Fisher Scientific, Pittsburgh, PA). DNA extracts were diluted to 10 ng μl−1 and stored at −20 °C for further analyses.
The fungal ITS1 region of the nuclear rDNA gene cluster was amplified from metagenomic DNA isolated from the cereal rye mulch using primers ITS1F (5′-CTTGGTCATTTAGAGGAAGTAA-3′) and ITS4R (5′-TCCTCCGCTTATTGATATGC-3′) (White et al. 1990; Gardes and Bruns 1993). Quantitative PCR (qPCR) was carried out on a Stratagene Mx3000p qPCR System (Agilent Technologies, Santa Clara, CA) using SYBR green detection. Each 20 μl reaction contained 10 μl of 2X Sso Fast EvaGreen Supermix (Bio-Rad Laboratories, Hercules, CA), 400 nM forward primer, 400 nM reverse primer, and 50 ng of total template DNA (except for standards and nontemplate controls). Thermal cycling conditions were as follows: 1 cycle of 98 °C for 3 mins., 40 cycles of 98 °C for 15 s., and 65 °C for 30 s., followed by a continuous melting curve of 30 s. at 65 °C to 95 °C to assess qPCR product purity. Product specificity was confirmed by agarose gel electrophoresis (1.5 % w/v gel containing GelStar). All field samples were run as analytical triplicates in the qPCR step. The mean value of the triplicates was reported as nanogram (ng) of ITS1 fungal gene and plotted against the time course of the experiment.
Statistical analysis
The two experimental designs were a randomized complete block design (StIS) and a split-plot design (SIS) with four replications. A combined analysis was attempted for both locations, but significant treatment interactions by location prevented us from pooling soil inorganic N data (StIS only) and cereal rye biomass data. However, soil inorganic N (SIS only) and soybean tissue data were pooled over both sites (Steel et al. 1996). All parameters were analyzed using PROC MIXED. Treatments were modeled as fixed effects, and blocks and environments as random effects (SAS 2011). The lack of homogeneous variance associated with soybean tissue and fungal DNA extractions data was corrected via a logarithmic transformation (Steel et al. 1996). Transformed means were back-transformed for presentation, and all means were separated using the Tukey-Kramer HSD method (P < 0.05) (Steel et al. 1996; SAS 2011).
Results
Surface residue N fluctuation
Cereal rye mulch N release was observed in the StIS over the eight-week duration of the study at Beltsville Agricultural Research Center (BARC), and Caswell Research Farm (KINS), totaling 15 and 50 kg N ha−1, respectively (Fig. 3). The analysis of the rye tissue from the SISt=0 plots confirmed that the 15N was confined below the surface, which ruled out anthropogenic 15N enrichment of the rye surface residues. There were no detectable differences in cereal rye tissue N (kg ha−1) between those treated and not treated with fungicide (Fig. 3A and B). There was observable 15N enrichment of the cereal rye residues over time. Six weeks after planting, there was a significant increase (P < 0.01) in 15N enrichment of the cereal rye biomass compared to the initial sampling interval at both BARC and KINS, 0.02 and 0.03 kg 15 N ha−1, respectively (Fig. 3C and D). The fungicidal treatments had minimal effect on reducing the 15N enrichment associated with the cereal rye biomass. However, at KINS only, at 6 WAP, greater 15N enrichment was detected in the cereal rye residues from plots not treated with fungicides (P < 0.1), indicating that the fungicides only partially controlled the upper movement of N (Fig. 3D).
Cereal rye surface residue N flux as impacted by fungicide applications. Staggered Injection Study: (a and b) show N (kg ha−1) in rye residue tissue. (c and d) show 15N (kg ha−1) in rye residue tissue. N levels were measured in cereal rye-mulched soybean microplots at Beltsville, MD and Kinston, NC (2010) — both those treated and not treated with fungicide. Time after soybean planting significantly impacted (P ≤ 0.01) N and 15N concentrations in rye tissues (a – d). Fungicide treatment and the interaction with time were not significant (a – d). Error bars indicate ±1 standard error. Asterisks indicate significant differences between treatments at a given time interval as determined by pre-planned contrast (*: P ≤ 0.05, **: P ≤ 0.01)
Similar trends of increased 15N enrichment in the cereal rye surface residues were observed in both the SIS and the StIS experiments (Fig. 4). In addition to the 15N enrichment of the rye surface residues across both sites, in the StIS experiment, at 6 WAP, there was evidence of fungal transport of N into the cereal rye surface residues (P < 0.05). The plots not treated with fungicide experienced nearly a 36 % increase in 15 N enrichment above the plots treated with fungicide (Fig. 4B).
Single Injection Study. a, Single Injection Study and (b), Staggered Injection Study. This Fig. shows 15N enrichment in cereal rye residue tissue from soybean microplots mulched with cereal rye at Beltsville, MD and Kinston, NC (2010). Half the plots were treated with fungicide and others were not. Time after soybean planting (A, time = 0, 4, and 8 weeks after enrichment and soybean planting; B, time = 0, 2, 6, and 8 weeks) did significantly impact (P ≤ 0.001) 15N at.% enrichment of the rye tissue (a and b). The application of fungicide and the interaction with time was not significant. Error bars indicate ±1 standard error. Asterisks indicate significant differences between treatments at a given time interval as determined by pre-planned contrast (*: P ≤ 0.05, **: P ≤ 0.01)
Fungal DNA quanta in rye surface residues
Quantification of the fungal ITS1 DNA at both locations was greater in the plots not treated with fungicide than in the fungicide-treated plots, except for 8 WAP at Beltsville and 2, 4 WAP at Kinston (Fig. 5). The increase in fungal DNA was greatest in the plots not treated with fungicide six weeks after planting, which corresponds to the largest N transfer to the cereal rye residues (Figs. 3, 4, and 5).
Staggered Injection Study: Fungal quanta measured in cereal rye tissue from cereal rye-mulched soybean crops at Beltsville, MD (a) and Kinston, NC (b) (2010). Half of the plots were treated with fungicide and others were not. Fungicide treatments and time after soybean planting significantly (P ≤ 0.05) impacted the quantity of gene ITS1 detected in the rye surface mulch at both locations. The interaction of fungicide and time was only significant (P ≤ 0.05) at Beltsville. Asterisks indicate significant differences between treatments at a given time as determined by pre-planned contrast (*: P ≤ 0.05, **: P ≤ 0.01)
Soybean contribution to N constraints
Even though soybean plants biologically fix, on average, 50–60 % of assimilated N from the air, they remained a considerable sink for soil-derived N in the microplots (Fig. 6) (Salvagiotti et al. 2008). Between the six- and eight-week sampling intervals, soybean demand for soil-derived N increased by nearly 30 %, and during this time 0.15 kg 15 N ha−1 was removed from the soil by the soybeans. This reduced the extractable inorganic soil 15N in the SIS at Beltsville and Kinston by approximately 90 %, along with a 60 % reduction in extractable soil inorganic N (Figs. 6 and 7).
Staggered Injection Study: Plant tissue N (a) and 15N (b) enrichment from soybeans grown in cereal rye-mulched microplots at Beltsville, MD and Kinston, NC (2010). The combined data are from cereal rye-mulched microplots at Beltsville, MD and Kinston, NC (2010). Soybean 15N from air only and total 15N from air and soil were derived using widely accepted approaches for estimating (Salvagiotti et al. 2008). Error bars indicate ±1 standard error
Single Injection Study: Extractable soil inorganic N (kg ha−1) (a) and 15N (kg ha−1) (b) measured in soil under a soybean crop mulched with cereal rye at Beltsville, MD and Kinston, NC (2010). Half of the plots were treated with fungicide and others were not. Fungicide treatment did not significantly impact extractable soil inorganic N (a and b). However, time after soybean planting was significant (p ≤ 0.05, a and b). Error bars indicate ±1 standard error
Extractable inorganic soil N
Extractable soil inorganic N decreased in the StIS over the eight-week duration at Beltsville (4 kg N ha−1) and after the fourth week at Kinston (6 kg N ha−1) (Fig. 8). Fungicide treatments had a limited effect on soil-extractable N for all plots, with the exception of the 2 WAP interval at Beltsville (Fig. 8). During the 2 WAP time frame at Beltsville, plots not treated with fungicide had 2 and 0.07 kg ha−1 greater extractable soil inorganic N and 15N when compared to the fungicide-treated plots (Fig. 8A and C). There was no separation in the soil-extractable inorganic N or 15N between the plots treated and not treated with fungicide during the first two-week time interval at Kinston (Fig. 8). However, at Kinston during 4 WAP there was a 2 and 0.10 kg ha−1 increase in extractable soil inorganic N and 15N, respectively (Fig. 6). The increase in extractable soil inorganic N during this time was the result of a nearly 40 kg ha−1 N release from the cereal rye surface residue (Fig. 3). Despite the N release from the cereal rye, extractable N continued to decrease throughout the season—except for the previously mentioned interval at Kinston — and was below 4 and 7 kg N ha−1 by 8 WAP (Fig. 8A and B).
Staggered Injection Study: Extractable soil inorganic N (a and b) and 15N (c and d) measured from soil under a soybean crop mulched with cereal rye at Beltsville, MD and Kinston, NC (2010). Half of the plots were treated with fungicide and others were not. Fungicide treatment was not significant (a – d). Time after soybean planting did significantly (P ≤ 0.001) impact extractable soil inorganic N and 15N for both locations. The interaction of fungicide and time was only significant (P ≤ 0.01) at Beltsville (a). Error bars indicate ±1 standard error. Asterisks indicate significant differences between treatments at a given time interval as determined by pre-planned contrast (*: P ≤ 0.05, **: P ≤ 0.01)
Similar soil N status was observed via the 15N tracer at both Beltsville and Kinston (Fig. 8C and D). The extractable soil inorganic 15N enrichment in the StIS was far lower than the 1.77 kg 15 N ha−1 injected. However, soil sampled from the SIS study two hours after injection recovered only 0.6 kg 15 N ha−1 in the extractable soil N pool (Fig. 7B). Even though recovery of the 15N tracer was lower than expected in the SIS experiment, the concentration of the tracer was high enough to be detected in the soil at 4 WAP (Fig. 7B).
Discussion
The difference in N release from the cereal rye surface residue observed at BARC and KINS was likely due to differences in residue quality between BARC and KINS at the deployment of the study. At BARC, the cereal rye was harvested (i.e., haybined) at the hard kernel stage (Feeke’s growth stage 11.4), whereas at KINS, the cereal rye was just entering the soft dough stage (Feeke’s growth stage 11.1). As a result, the initial carbon to N ratios of the cereal rye residue was far greater at BARC (70:1) than at KINS (43:1). The lower C:N ratio in the cereal rye residues at KINS were likely less resistant to decomposition, so the residues released considerably more N to the soil system than the more recalcitrant residues from BARC (Fig. 3).
Contrary to what Frey et al. (2000) observed, there was no significant reduction in decomposition from the fungicide-treated cereal rye residues. Whether treated or untreated, plots at BARCt=0 (54 kg ha−1) and KINSt=0 (138 kg ha−1) lost nearly 40 % of their initial nitrogen content, suggesting minimal effect of fungicide in altering the decomposition processes in the cereal rye residues (Fig. 3). Using the approach as described by Frey et al. (2000) and the equation developed by Aber and Melillo (Aber and Melillo 1982), net N immobilization into the cereal rye surface mulch was determined by regressing the percentage of initial rye residue remaining vs. the percentage of the rye residue N using the regression parameters in the following equation:
Net N immobilization in the rye surface residues at BARC was 1.9 kg N ha−1. The reduced N immobilization observed at BARC, when compared to the 15–18 kg N ha−1 N immobilized as reported by other studies, could be attributed to the increased duration of previous studies (4 months longer) (Beare et al. 1992; Frey et al. 2000). The delayed application of both wheat straw and 15N tracers ensured that the fungal hyphal networks were well established, thus maximizing the amount of 15N transferred during the study (Beare et al. 1992; Frey et al. 2000). Despite the large N release from the cereal rye residues, there was an observed increase of 15N enrichment (atom% 15N) associated with the cereal rye residues. The increase in 15N enrichment indicates bilateral movement of N between the mulch and the mineral soil (Fig. 3).
A combined analysis of 15N atom % enrichment across site showed considerable enrichment of 15N in excess of background δ 15N enrichment in the cereal rye residues (Fig. 4). Since each plot received 0.6 cm of simulated rainfall after injection, it is unlikely that the increase in 15N enrichment in the cereal rye residue was a result of contamination during injection of 15N below the soil surface or by capillary wicking. Even though upward movement of 15N was observed, it is unclear whether the N transfer to the mulch is a function of volatile soil-sourced NOx, which is then dry deposited into the mulch layer (Goulding et al. 1998), or whether the nitrogen transfer is primarily mediated via fungal translocation. Additional mechanisms of 15N upward transfer such as NH3(g) volatilization followed by re-condensation or fungal and bacterial capture would have been low, given the lower soil pH values (Table 1). Physical mechanisms for upper movement via earthworms are not likely since earthworm activity was not observed (i.e., casting).
The qPCR analysis indicated increased fungal DNA in the plots not treated with fungicide for all time points at BARC (P < 0.05) and six and eight weeks after planting at KINS (P < 0.05), when compared to the plots treated with fungicide (Fig. 5). The detection of ITS1 gene signals in the plots that had fungicide treatment could have been due to an insufficient application rate of the fungicide. The application rate was lowered to avoid perturbation to the soil N status. We must also consider that the cereal rye mulch is field-grown, so it may contain both surface-dwelling as well as endophytic fungal species that also would be detectable by the qPCR technique used in this study. Studies have reported successful suppression of saprophytic fungi via Captan while minimizing effects on off-target populations (Ingham and Coleman 1984; Beare et al. 1992; Frey et al. 2000). However, since Captan contains approximately 4.67 % N, the prescribed rates of Captan specified by these studies could perturb the N cycling in low-N environments, and at those prescribed rates could have influenced the degree of N cycling in our microplots. Furthermore, it is plausible that microbes used Captan N and therefore decreased the overall magnitude of the fungal translocation of soil-derived N to the cereal rye surface residues.
It is possible that Captan reduced soil-extractable inorganic N in the fungicide plots by limiting N cycling and enhancing bacterial N immobilization of dissolved organic C originating from both the above-ground cereal rye residue and from Captan, which contains 36 % carbon. Captan has been shown to augment the soil N status by reducing the microbial communities responsible for capturing N and by providing digestible carbon that can be immobilized into the bacterial biomass (Martínez-Toledo et al. 1998; Megadi et al. 2010a; Megadi et al. 2010b). It is possible that the 56 kg ha−1 N release from the cereal rye at Kinston when compared to Beltsville during the first month of the study (Fig. 3) masked any negative effects Captan had on both N cycling and bacterial N immobilization, thereby increasing the extractable soil inorganic N enrichment (Fig. 8D). The sharp reductions in extractable soil inorganic 15N observed in both experiments (Fig. 7) were immobilized via the intense microbial competition for N in the low N environment, or leached below the sampling depth (Kessavalou et al. 1996; Dou et al. 2007).
Unlike many of the studies that have evaluated N transfer from soil to surface residues either in grass or forest ecosystems (Schimel and Firestone 1989; Hart et al. 1994), this study evaluated N status associated with high biomass cereal rye cover crop residues in the presence of soybeans. Nitrogen net immobilization, at least at BARC (1.9 kg N ha−1), accounted for approximately 20 % of the total N release from the cereal rye surface mulches (14 kg N ha−1, Fig. 3), where a relatively large amount of N entered the soil. Since soybeans are the primary sink for N, and low N environments are not detrimental to soybean yields (Wells et al. 2013), the low soil N status in these systems could be leverage for increased weed control, especially when managing for herbicide resistance.
Conclusion
In conclusion, this body of work supports our hypothesis of upward movement of N from mineral soil to a high C:N residue cover crop mulch. Although the fungicide effects were small, 1.9 kg N ha−1 was transported into the cereal rye residue at BARC, directly contributing to the reduction of available N in the system. Even though there was observed release of cereal tissue N at both BARC (14 kg N ha−1) and KINS (56 kg N ha−1) over the duration of the study, the N release from the rye surface mulch had minimal impact on the extractable soil inorganic N. These results confirm that the extreme competition for N in high C:N soil systems and the observed N immobilization via fungal transport or some other means is over and above the N immobilized by the soil microbial community. The two study designs—multiple isotope injection times versus a single one—yielded similar results. The similarities suggest either method is acceptable for future studies.
References
Aber JD, Melillo JM (1982) Nitrogen immobilization in decaying hardwood leaf litter as a function of initial nitrogen and lignin content. Can J Bot 60:2263–2269
Beare MH, Parmelee RW, Hendrix PF, et al. (1992) Microbial and faunal interactions and effects on litter nitrogen and decomposition in agroecosystems. Ecol Monogr 62:569–591
Bernstein ER, Posner JL, Stoltenberg DE, Hedtcke JL (2011) Organically managed no-tillage rye-soybean systems: agronomic, economic, and environmental assessment. Agron J 103:1169–1179
Brooks PD, Stark JM, McInteer BB, Preston T (1989) Diffusion method to prepare soil extracts for automated nitrogen-15 analysis. Soil Sci Soc Am J 53:1707–1711
Burgess MS, Mehuys GR, Madramootoo CA (2002) Nitrogen dynamics of decomposing corn residue components under three tillage systems. Soil Sci Soc Am J 66:1350–1358
Dou FG, Wright AL, Hons FM (2007) Depth distribution of soil organic C and N after long-term soybean cropping in Texas. Soil Tillage Res 94:530–536
Frey SD, Elliott ET, Paustian K, Peterson GA (2000) Fungal translocation as a mechanism for soil nitrogen inputs to surface residue decomposition in a no-tillage agroecosystem. Soil Biol Biochem 32:689–698
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes - application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Goulding KW, Bailey NJ, Bradbury NJ, Hargreaves P, Howe M, Murphy DV, Poulton PR, Willison TW (1998) Nitrogen deposition and its contribution to nitrogen cycling and associated soil processes. New Phytol 139(1):49–58May 1
Hart SC, Stark JM, Davidson EA (1994) Nitrogen mineralization, immobilization, and nitrification. In: Weaver RW (ed) Methods of Soil Analysis. Part 2: Microbiological and Biochemical Properties. Soil Science Society of America Inc., Madison, WI, pp 985–1018
Holland EA, Coleman DC (1987) Litter placement effects on microbial and organic matter dynamics in an agroecosystem. Ecology 62:425–433
Ingham ER, Coleman DC (1984) Effects of streptomycin, cycloheximide, Fungizone, Captan, carbofuran, cygon, and PCNB on soil microorganisms. Microb Ecol 10:345–358
Ingham ER, Cambardella C, Coleman DC (1986) Manipulation of bacteria, fungi and protozoa by biocides in lodgepole pine forest soil microcosms: effects on organism interactions and nitrogen mineralization. Can J Soil Sci 66:261–272
Jin K, Sleutel S, Neve S, et al. (2008) Nitrogen and carbon mineralization of surface-applied and incorporated winter wheat and peanut residues. Biol Fertil Soils 44:661–665
Keeney DR, Nelson DW (1982) Nitrogen - inorganic forms. Methods Soil Anal Part 2 Chem Microbiol Prop 643–698
Kessavalou A, Qian JH, Kettler TA, et al. (1996) ) Bromide and nitrogen-15 tracers of nitrate leaching under irrigated corn in central Nebraska [electronic resource]. J Environ Qual 25:1008–1014. doi:10.113/16843
Marinari S, Lagomarsino A, Moscatelli MC, et al. (2010) Soil carbon and nitrogen mineralization kinetics in organic and conventional three-year cropping systems. Soil Tillage Res 109:161–168
Martínez-Toledo MV, Salmerón V, Rodelas B, et al. (1998) Effects of the fungicide Captan on some functional groups of soil microflora. Appl Soil Ecol 7:245–255
Megadi VB, Tallur PN, Hoskeri RS, et al. (2010a) Biodegradation of pendimethalin by Bacillus circulans. Indian J Biotechnol 9:173–177
Megadi VB, Tallur PN, Mulla SI, Ninnekar HZ (2010b) Bacterial degradation of fungicide Captan. J Agric Food Chem 58:12863–12868
Mirsky SB, Ryan MR, Teasdale JR, et al. (2013) Overcoming weed management challenges in cover crop-based organic rotational no-till soybean production in the eastern US. Weed Technol 27:193–203
Reberg-Horton SC, Burton JD, Danehower DA, et al. (2005) Changes over time in the allelochemical content of ten cultivars of rye (Secale cereale L.). J Chem Ecol 31:179–193
Reberg-Horton SC, Grossman JM, Kornecki TS, et al. (2012) Utilizing cover crop mulches to reduce tillage in organic systems in the southeastern USA. Renew Agric Food Syst 27:41–48
Recous S, Robin D, Darwis D, Mary B (1995) Soil inorganic N availability: effect on maize residue decomposition. Soil Biol Biochem 27:1529–1538
Salvagiotti F, Cassman KG, Specht JE, et al. (2008) Nitrogen uptake, fixation and response to fertilizer N in soybeans: a review. Field Crops Res 108:1–13
SAS (2011) Base SAS 9.3 Procedures guide: Statistical Procedures. SAS Institute Inc., Cary, NC
Schimel JP, Firestone MK (1989) Nitrogen incorporation and flow through a coniferous forest soil profile. Soil Sci Soc Am J 53:779–784
Schomberg HH, Steiner JL, Unger PW (1994) Decomposition and nitrogen dynamics of crop residues: residue quality and water effects. Soil Sci Soc Am J 58:372–381
Schwendener CM, Lehmann J, de Camargo PB, et al. (2005) Nitrogen transfer between high- and low-quality leaves on a nutrient-poor Oxisol determined by 15N enrichment. Soil Biol Biochem 37:787–794
Smith AN, Reberg-Horton S, Place GT, et al. (2011) Rolled rye mulch for weed suppression in organic no-tillage soybeans. Weed Sci 59:224–231
Stark JM, Hart SC (1996) Diffusion technique for preparing salt solutions, Kjeldahl digests, and persulfate digests for nitrogen-15 analysis. Soil Sci Soc Am Soil Sci Soc Am J 60:1846–1855
Steel RG, Torrie JH, Dickey DA (1996) Principles and procedures of statistics: A biometric approach, 3rd edn. McGraw-Hill, New York
Teasdale JR, Mohler CL (2000) The quantitative relationship between weed emergence and the physical properties of mulches [electronic resource]. Weed Sci 48:385–392. doi:10.1614/0043-1745(2000)048[0385:TQRBWE]2.0.CO;2 and http://hdl.handle.net/10113/42808
Tu C, Booker FL, Watson DM, et al. (2006) Mycorrhizal mediation of plant N acquisition and residue decomposition: impact of mineral N inputs. Glob Change Biol 12:793–803
Wells MS, Reberg-Horton SC, Smith AN, Grossman JM (2013) The reduction of plant-available nitrogen by cover crop mulches and subsequent effects on soybean performance and weed interference. Agron J 105:539–545
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a guide to method and applications. Academic Press, Inc., New York, pp 315–322
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Leslie A. Weston.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Wells, M.S., Reberg-Horton, S.C., Mirsky, S.B. et al. In situ validation of fungal N translocation to cereal rye mulches under no-till soybean production. Plant Soil 410, 153–165 (2017). https://doi.org/10.1007/s11104-016-2989-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-2989-8