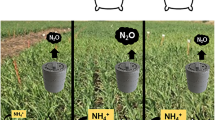
Abstract
Background and aims
Sorghum (Sorghum bicolor) is able to exude allelochemicals with biological nitrification inhibition (BNI) capacity. Therefore, sorghum might be an option as cover crop since its BNI ability may reduce N pollution in the following crop due to a decreased nitrification. However, BNI exudation is related to the physiological state and development of the plant, so abiotic stresses such as drought might modify the rate of BNI exudation. Hence, the objective was to determine the effect of drought stress on sorghum plants’ BNI release.
Methods
The residual effects of sorghum crops over ammonia-oxidizing bacteria (AOB) were monitored in a 3-year field experiment. In a controlled-conditions experiment, sorghum plants were grown under Watered (60% WFPS) or Moderate drought (30% WFPS) conditions, and fertilized with ammonium sulphate (A), ammonium sulphate + DMPP (A+D), or potassium nitrate (KNO3−). Soil mineral N was determined, and AOB populations were quantified. Additionally, plant biomass, isotopic discrimination of N and C, and photosynthetic parameters were measured in sorghum plants.
Results
In the driest year, sorghum was able to reduce the AOB relative abundance by 50% at field conditions. In the plant-soil microcosm, drought stress reduced leaf photosynthetic parameters, which had an impact on plant biomass. Under these conditions, sorghum plants exposed to Moderate drought reduced the AOB abundance of A treatment by 25% compared to Watered treatment.
Conclusion
The release of BNI by sorghum under limited water conditions might ensure high soil NH4+-N pool for crop uptake due to a reduction of nitrifying microorganisms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The availability of nitrogen (N) is the major limiting nutrient for crop growth (LeBauer and Treseder 2008). Although agriculture relies on the intensive use of N fertilizers to maximize crop yields, a great amount is lost as reactive N since crops cannot take the entire N applied and it cannot be retained by soils (Lassaletta et al. 2014). The main pathways for N losses that cause a negative environmental impact are through nitrate (NO3−) leaching, ammonia (NH3) volatilization, and emissions of nitrogenous gases such as nitric oxide (NO) and nitrous oxide (N2O) (Coskun et al. 2017). Nitrous oxide, one of the main greenhouse gasses (GHG) generated in upland agriculture (Syakila and Kroeze 2011) with a global warming potential (GWP) between 265 and 298 times higher than that of CO2 in a 100-year time horizon (IPCC 2014), is mainly generated by microbial nitrification and denitrification processes (Li et al. 2016). There are several approaches to reduce N losses derived from fertilization, e.g., the use of synthetic nitrification inhibitors (SNIs), such as 3,4-dimethylpyrazole phosphate (DMPP), when applying ammonium-based fertilizers (Huérfano et al. 2015, 2018). Unfortunately, the use of SNIs is not widely adopted by farmers due to having some disadvantages such as additional product and field application costs and low cost-effectiveness for farmers (Subbarao et al. 2006, 2013a, 2017). Notwithstanding these drawbacks, the use of crops with the capability of producing biological nitrification inhibitions (BNIs) has become in a promising option to alleviate N losses derived from nitrification.
The biological nitrification inhibition (BNI) was firstly described in 1966 in Hyparrhenia filipendula, but it was not termed as BNI until 2003 when Ishikawa et al. (2003) tried to describe the capacity of Brachiaria humidicola to inhibit the ammonium (NH4+) oxidation to NO3−. Moreover, the opportunity to exploit this strategy in agricultural systems to minimize the problem of N losses has gone unnoticed until recently (Subbarao and Searchinger 2021). This ability to produce BNIs is highlighted in the framework of sustainable agriculture based on the use of environmentally friendly agronomic practices to decrease pollution derived from the use of fertilizers (Subbarao et al. 2013a; Zhang et al. 2015). Therefore, the use of cover crops capable of producing BNIs represents another promising strategy to control nitrification and, thus, to increase the availability of N in the soil for the next crop while reducing N losses from the agrosystem (Karwat et al. 2019; Momesso et al. 2019). The ability to exude BNIs seems to be related to plants’ adaptability to low N environments (Subbarao et al. 2015). Regarding field crops, unlike crops adapted to high N input environments such as wheat (Triticum aestivum) and maize (Zea mays), the strongest BNI capacity is found in sorghum (Sorghum bicolor) since it is adapted to low N environments (Subbarao et al. 2007). Sorghum roots release two categories of BNIs, hydrophilic and hydrophobic, which may have complementary roles. The hydrophobic BNIs may remain close to the root systems, as they are strongly absorbed by soil mineral or organic particles, which may further increase their persistence (Dayan et al. 2010; Subbarao et al. 2013b). In contrast, hydrophilic BNIs are more likely to move out of the rhizosphere, which may enhance their capacity to suppress nitrification in bulk soil (Nardi et al. 2013; Subbarao et al. 2013b). In addition, BNIs from sorghum can be released until close to the physiological maturity of the crop (Sarr et al. 2021), which would ensure the availability of N during all the stages of crop development.
Increasing efforts are taken to identify and characterize BNI molecules, but the regulation of their synthesis and release is still barely understood. In general, BNI activity depends on the presence and direct contact of NH4+ in the root environment (Subbarao et al. 2017; Afzal et al. 2020). Nevertheless, the influence of soil physicochemical factors like texture, composition, and pH on the release of BNIs by roots has not yet been sufficiently studied. Moreover, the effectiveness of BNIs in a climate change scenario such as the increase of environmental temperature or water scarcity during plant growth remains to be evaluated. Therefore, the study of the impact of environmental conditions on the production of BNIs by crops is crucial to introduce this quality in agricultural operations such as crop rotation systems (Bozal-Leorri et al. 2023). Although drought periods happen naturally, it is estimated that their frequency and severity will increase in most of the world’s crop-producing regions as a result of climate change (Hochholdinger 2016; Daryanto et al. 2017). Drought stress is the abiotic factor that most limits the productivity of agrosystems, reducing significantly crop growth and yield (Fleury et al. 2010). Since BNI release is related to the physiological state and development of the plant (Sarr et al. 2021), stresses that affect crop growth might also limit or modify the rate of BNI exudation. To date, only Ghatak et al. (2021) have studied the effects of abiotic stresses on the BNI root exudation and its composition in pearl millet plants (Pennisetum glaucum). Indeed, these authors found a genotype-dependent enhancement of BNI activity after a defined period of drought stress.
The sorghum ability to produce BNIs (Subbarao et al. 2013b), and, more importantly, the fact of being a drought-tolerant crop (Hadebe et al. 2017) lead us to hypothesize the BNI release in sorghum could be stimulated by soil water scarcity, which will benefit the following crop since sorghum has been postulated as a potential catch crop or cover crop (Bozal-Leorri et al. 2021a, 2023). Therefore, the aim of this work was to determine the impact of drought stress on BNI activity of sorghum plants, and the effects of exuded BNIs on soil mineral N content and nitrifying microorganisms.
Materials and methods
Field experiment
The field experiment was conducted in three different fields from Garinoain, Northern Spain (42° 35’ N, 1° 40’ W and 532 m above sea level) during three successive summer seasons 2017, 2018, and 2019. Soil characteristics of the upper horizon of the three fields are compiled in Supplementary Tables 1, and daily air mean temperature and accumulated precipitation of the three summer seasons are presented in Supplementary Fig. 1. Sorghum (Sorghum bicolor var. PR88P68 Pioneer Corteva Agriscience) was sown in no-till conditions at a rate of 15 kg seeds ha− 1 in May of each year after the previous hairy vetch (Vicia villosa Roth) winter cover crop that was halted with glyphosate and left on the soil surface. The experiment consisted of two randomized blocks with three replications and two treatments (5 m x 5 m plots) in each block: (1) uncultivated soil after the termination of the precedent crop (Fallow) and (2) sorghum without fertilizer application (Sorghum). For fallow plots, sorghum plants were desiccated with RoundUp (a glyphosate-based herbicide; 36% w/v. Monsanto) one month after sorghum sowing, according to manufacture recommendations in no-till systems from this region, 1.5 L ha− 1. The use of glyphosate was chosen because application at higher rates than in this experiment has been reported to have no effect on nitrifying bacteria (Allegrini et al. 2017; Zabaloy et al. 2017), and so it was observed in previous works from our group (Bozal-Leorri et al. 2021a, 2023).
Greenhouse experiment
The second experiment was carried out in microcosms in a greenhouse under a daily regimen of 14/10 h day/night cycle with an average day/night temperature and relative humidity of 25/18°C and 50/60%, respectively. The soil was collected in June 2019, from a 0–30 cm layer of clay loam soil in a wheat field (Supplementary Table 1) in Arkaute (Alava, Spain; 42° 51’ N, 2° 37’ W and 530 m above sea level). Roots and stones were removed and the soil was passed through a 5 mm sieve. Soil was mixed with sand in 3:1 soil:sand (v:v) proportion to increase soil porosity. After this, it was air-dried, homogenised and kept at 4 °C until the start of the experiment. In order to reactivate soil microorganisms, pots were supplied with 86 mg of N in the form of ammonium sulphate ((NH4)2SO4), an equivalent dose to 15 kg N ha− 1, and soil was rehydrated with deionised water up to 50% water filled pore space (WFPS). WFPS was calculated as in Linn and Doran (1984) following the equation:
Particle density was assumed to be 2.65 Mg m− 3 and soil bulk density was determined in the laboratory, resulting in a value of 1.31 Mg m− 3.
A trifactorial experimental design (presence/absence of sorghum plant, water regimen, and type of fertilization) with three replications was implemented in thirty-six 1.35 L pots. After soil activation, eighteen microcosms were planted with 4 seedlings of sorghum (Sorghum bicolor var. PR88P68 Pioneer Corteva Agriscience) per microcosm and the other eighteen microcosms were kept with only soil. Sorghum seeds were previously germinated on a tray with perlite:vermiculite (1:3) mixture at 20 °C for 6 days. All 36 microcosms were watered for 15 days after sorghum transplanting to maintain soil WFPS. On the 15th day of watering, two groups of 9 microcosms were established within each group of microcosms with soil and with soil and plant. One group held “Watered” regimen and the other group held the “Moderate drought” regimen. At the same time, each group was randomly divided into 3 groups of three microcosms corresponding to three different fertilizer treatments. The fertilizer treatments were: ammonium sulphate (A), ammonium sulphate combined with DMPP (A+D), and potassium nitrate (KNO3−). Nitrogen was applied in an equivalent dose to 195 kg N ha− 1, which was achieved by adding 1.726 g of potassium nitrate (δ 15 N value of 16.5) or 1.128 g of ammonium sulphate, alone or mixed with DMPP (δ 15 N value of -1.2 and − 0.9 respectively) (EuroChem Agro Iberia S.L.); DMPP content represented 0.8% of the applied NH4+-N. In order to achieve a homogeneous distribution of nitrogen in the soil, fertilizers were dissolved in deionised water, ready to be added to the corresponding treatments by pipetting. After treatment application, “Watered” pots increased their WFPS up to 60%. On the other hand, “Moderate drought” pots decreased to 30% of WFPS by stopping the watering until they reached the calculated weight for that moisture level. All of them were watered every two days in order to maintain each WFPS for another 60 days.
Plant biomass and isotopic discrimination
Above plant biomass production in the microcosm experiment was measured at 60 days post-fertilization as dry weight (DW) from sorghum plants at vegetative stage 4 (Vanderlip 1993). To do so, one sorghum plant per microcosm was dried at 80 °C in a circulation oven for 72 h until a constant DW was reached.
For a deeper understanding of plant N source acquisition and drought effect, N and C isotopic composition in leaves of sorghum was determined by an elemental analyzer (FlashEA1112 ThermoFinnigan) coupled to a mass spectrometer (DELTAplus Finnigan MAT) in the Unidade de Técnicas Instrumentais de Análise, Servizos de Apoio á Investigación (SAI), Universidade da Coruña. The absorption of different sources of N by the plant changes the δ 15 N values (Werner and Schmidt 2002). On the other hand, the C isotopic signature has been widely used as indicator of plant’s water use efficiency and drought stress (Eggels et al. 2021). The values of the isotopic ratio were expressed as δ15N and δ13C, in parts per thousand (‰) relative to atmospheric N2 and VPDB (Vienna Pee Dee Belemmite), respectively. The isotope composition values δ (‰) were obtained by the following equation:
where Rsample is the 15 N/14 N or 13 C/12 C ratio of the plant sample and Rstandard is the 15 N/14 N ratio of the atmospheric N2 or the 13 C/12 C ratio of VPDB.
Leaf determinations
Leaf gas-exchange parameters were conducted in the totally expanded upper leaf using a Li-COR 6400XP portable photosynthesis system (LI-COR Inc., Lincoln, Nebraska, USA). The rate of CO2 assimilation (AN), stomatal conductance (gs), and intercellular CO2 (Ci) parameters were determined under light-saturated conditions with a photosynthetic photon flux density (PPFD) of 1000 µmol m− 2 s− 1 at 25 °C and with a CO2 concentration of 400 ppm. The maximum quantum of PSII (Fv/Fm) was measured under light-adapted conditions in the centre of the youngest fully developed leaf with a mini-PAM (miniaturized pulse amplitude–modulated photosynthesis yield analyser).
Geochemical analysis
Soil NH4+ and NO3− contents were determined the day after sorghum harvest for field experiment while those of greenhouse experiment were made 30 days post-fertilization. Three soil subsamples of 3 cm diameter × 0.3 m depth for the field experiment and 1.5 cm diameter × 10 cm depth for the microcosms experiment were taken. Plant debris and stones from soil subsamples were removed and finally, they were homogenized. Then 100 g were mixed with 200 mL 1 M KCl and shaken for one hour at 165 rpm. The soil solution was filtered twice through Whatman n°1 filter papers (GE Healthcare) and Sep-Pak Classic C18 Cartridges 125 Å pore size (Waters) to remove big soil particles and organic matter, respectively. The filtered solution was used to determine the content of NH4+, using the Berthelot method (Patton and Crouch 1977), and NO3−, as described by Cawse (1967).
Abundance of nitrifying bacteria in soil
Quantitative polymerase chain reaction (qPCR) was used to quantify the abundance of nitrifying genes. Soil DNA was isolated from the same samples used for geochemical determinations (at the end of the sorghum crop for the field experiment, and 30 days post-fertilization for greenhouse experiment). DNA was extracted from 0.25 g of dry soil using the PowerSoil DNA Isolation Kit (Quiagen) including the modifications described in Harter et al. (2014). Extracted DNA concentration and quality were determined spectrophotometrically (NanoDrop® 1000, Thermo Scientific). Quantification of total bacteria abundance (16 S rRNA) and functional marker genes involved in nitrification (bacterial amoA) were amplified by qPCR using SYBR® Premix Ex TaqTM II (Takara-Bio Inc.) and gene-specific primers in a StepOne PlusTM Real-Time PCR System (Torralbo et al. 2017). Data analysis was carried out by StepOnePlusTM Software 2.3 (Thermo Scientific). Standard curves were prepared from serial dilutions of linearized plasmids with insertions of the target gene ranging from 107 to 102 gene copies µL− 1. The amoA relative abundance was calculated following this equation:
where the absolute abundance of the 16 S rRNA and amoA genes (copy number of target gene per gram of dry soil) were calculated according to a modified equation detailed in Behrens et al. (2008):
Statistical analysis
Data was analysed with IBM SPSS v. 24.0 statistical software (IBM Corp. Armonk, NY, USA). Comparisons of two soil conditions (fallow and sorghum crop) from the field experiment were made using the Mann-Whitney U test. Leaf gas-exchange and Fv/Fm parameters were subject to a two-way (water regimen, W; and fertilizer treatment, F) analysis of variance. The results of soil mineral N, microbial quantification, aboveground biomass, and leaf δ15N and δ13C were analysed by one-way ANOVA using Duncan’s multiple range test for separation of means between treatments. The Mann–Whitney U test was used to compare the effect of the absence or presence of sorghum plant within the same treatment. p-values < 0.05 were considered to be statistically significant differences.
Results
Three-year comparison of fallow and sorghum field
Cumulative precipitations during the early development of the sorghum crop (May to July) were similar (Supplementary Fig. 1), but the rain events from July to the end of the crop made a great difference between the three years. The amoA relative abundance was reduced by 50% in the soil with sorghum crop compared to soil with fallow in 2017 (Fig. 1a). The accumulated precipitation at the end of the crop was 120 L m− 2 since it was a dry summer compared to years 2018 and 2019, with 240 and 200 L m− 2, respectively (Fig. 1a). In turn, the growth of nitrifying bacteria during these two years was not statistically affected by the presence of sorghum in relation to fallow soil. However, even though it was not statistically significant, the amoA relative abundance in the sorghum crop was reduced a 16% regarding the fallow in the year 2019. In addition, the presence of the sorghum crop reduced significantly soil N content compared to fallow in all three years (Fig. 1b). Although no differences were observed in soil NH4+ content between soil with fallow and soil that held sorghum (Fig. 1b), the effect of sorghum presence did reduce soil NO3− content by more than 40%, 90% and 68% in soil that held sorghum compared to fallow soil (Fig. 1b) in years 2017, 2018 and 2019, respectively. The lower soil NO3− content from the soil with fallow detected in year 2019 compared to the previous years could be due to a lower soil organic matter (Supplementary Table 1).
Ammonia-oxidizing bacteria (AOB) relative abundance (measured as the relative abundance of gene amoA) at the end of sorghum crop and accumulated precipitation during sorghum development (a) and soil mineral nitrogen at the end of sorghum crop (b) during three years of campaign. The Mann-Whitney U test was used for the comparison between fallow and sorghum plots. Significant differences at p < 0.05 are marked with a letter
Effect of moderate drought conditions
Sorghum plants reached the highest aboveground biomass under KNO3− treatment from Watered regimen, while A and A+D treatment presented similar aboveground biomass (Fig. 2a). Fertilizer treatment had no effect on aboveground biomass under Moderate drought regimen. Nevertheless, the water regimens affected the sorghum plants growth since plants under Watered regimen presented 2–5 times the aboveground biomass than those of Moderate Drought regimen. The application of fertilizers, as well as the water regimens, affected the leaf δ 15 N values. Sorghum plants fertilized with NO3− presented the highest δ 15 N, whereas A+D treatment had the lowest, regardless of the water availability (Fig. 2b). However, A treatment showed lower leaf δ 15 N values in Moderate drought regimen than in Watered regimen. Under Watered regimen, KNO3− treatment showed the most negative leaf δ 13 C values, whilst A and A+D treatment had similar values (Fig. 2c). No effect of fertilization was found on leaf δ 13 C values of Moderate Drought regimen, but this regimen presented less negative leaf δ 13 C values than Watered regimen. Fertilizer application had not a very strong effect on leaf gas exchange parameters in either Watered regimen or Moderate drought regimen (Fig. 3). Nevertheless, strong differences were found comparing both watered regimens. The net photosynthetic rate decreased by an average of 47% on treatments from Moderate drought regimen in respect to Watered regimen (Fig. 3a). Similarly, the stomatal conductance of treatments from Moderate drought regimen was 48% lower compared to those of Watered regimen (Fig. 3b). On the contrary, the intercellular CO2 increased in treatments from Moderate drought regimen, being 43% higher compared to Watered regimen (Fig. 3c). Since photosynthesis was decreased in Moderate drought regimen, akin results were found in the maximum quantum of PSII. Although fertilizer application did not affect Fv/Fm, Moderate drought regimen reduced it by 12% compared to Watered regimen (Fig. 3d).
Dry aboveground biomass of sorghum (Sorghum bicolor) plants (a) and leaf determination of δ15N (b) and δ13C (c). Pots were fertilized with ammonium sulphate (A); ammonium sulphate + DMPP (A+D) and potassium nitrate (KNO3−). Significant differences between treatments are marked with a lowercase letter (Duncan Test; p < 0.05; n = 3)
Net photosynthetic rate (a), stomatal conductance (b), intercellular CO2 mole fraction (c) and maximum quantum of PSII (d) of sorghum leaves during 45 days post-fertilization. Pots were fertilized with ammonium sulphate (A); ammonium sulphate + DMPP (A+D) and potassium nitrate (KNO3−). Statistical analysis was made through the Mann-Whitney U test (p < 0.05; n = 3) for the comparison between watered and Moderate drought regimens of each parameter at 0 day post-fertilization (DPF); ANOVA (p < 0.05; n = 3) for the comparison of all treatments from both water regimens at 15, 30, and 45 DPF; and to analyse the effect of water regimen (W), fertilizer treatment (F) and their interaction (WxF) was made through analysis of variance (two-way ANOVA; significant differences are marked with an asterisk [*] when p < 0.05 and double asterisk [**] when p < 0.01)
In microcosms of soil without plant, the NH4+ added in A treatment was not kept in the soil and presented similar soil NH4+ content than KNO3− treatment, whereas the greatest soil NH4+ content was found in A+D treatment (Fig. 4a). In the absence of plant, soil NH4+ content was not affected by the water regimen. In microcosms of soil with plant, A+D treatment also showed the highest soil NH4+ content, and soil NH4+ content of A treatment from Watered regimen was similar to that of KNO3− treatment. However, under Moderate drought regimen, A treatment presented greater soil NH4+ content than KNO3− treatment. In addition, A and A+D treatments from Moderate drought regimen kept higher soil NH4+ content than Watered regimen when plants were present. Regarding soil NO3− content, KNO3− treatment presented the highest values in pots of soil without plant (Fig. 4b). The addition of the synthetic nitrification inhibitor reduced the generation of NO3−, showing the lowest soil NO3− content. As with the soil NH4+ content, the water regimen did not affect the soil NO3− content when the plant is absent. On the other hand, when the plant was present, KNO3− treatment also showed the highest soil NO3− content. Nevertheless, A and A+D treatments did not present any differences in soil NO3− content. In this case, the water regimen affected the soil NO3− content in A and KNO3− treatment, where Moderate drought regimen showed higher values than those of Watered regimen. The presence of the plant greatly modified the soil mineral N since the lowest values of soil NH4+ content and soil NO3− content were found in the microcosm of soil with plant.
Soil mineral nitrogen in form of NH4+ (a) and NO3− (b) on pots with soil and pots with soil and plant at 30 days post-fertilization. Pots were fertilized with ammonium sulphate (A); ammonium sulphate + DMPP (A+D) and potassium nitrate (KNO3−). Significant differences between treatments from pots with soil are marked with a lowercase letter. Significant differences between treatments from pots with soil and plant are marked with a capital letter. For both ANOVA, the Duncan Test was used (p < 0.05; n = 3). The Mann-Whitney U test was used for the comparison between the absence or presence of plant within the same fertilization treatment. Significant differences at p < 0.05 are marked with an asterisk (*) only in the “Soil + Plant” treatment
The total bacteria abundance (measured as the abundance of 16 S rRNA gene) was affected neither by the fertilizer treatment nor by the water regimen in the microcosms of soil without plant (Supplementary Fig. 2a). However, the presence of plants altered the total bacteria abundance. Under Watered regimen, A treatment showed the highest 16 S rRNA gene abundance; while in Moderate drought regimen, A and A+D treatments were the ones with higher values. Water regimen also had no effect on the total bacteria abundance in microcosms of soil with plants. Nevertheless, A treatment from microcosms of soil with plant from both water regimens presented higher 16 S rRNA gene abundance than A treatment from microcosms of soil without plant. Fertilization with NH4+ highly increased the amoA gene abundance in both microcosms of soil without plant and soil with plant (Supplementary Fig. 2b). However, the observed changes in the total abundance of bacteria could mask the real power of sorghum inhibition; thus, the relative abundance of amoA gene is presented (Fig. 5). The application of DMPP in the microcosms of soil diminished the amoA relative abundance up to levels of KNO3− treatment, which means a 78% and 81% reduction compared to A treatment in Watered and Moderate drought regimens, respectively. In microcosms of soil with plant, A treatment had the highest amoA relative abundance, but its abundance was 25% lower under Moderate drought compared to Watered regimen. A+D treatment decreased nitrifying-gene abundance by 85% compared to A treatment in Watered regimen, whereas the reduction in Moderate drought regimen was 64%. The presence of sorghum plant affected amoA relative abundance of A treatment from both water regimens. While the amoA relative abundance of A treatment was reduced by 25% in watered regimen when sorghum plants were present in the soil compared to when they were not, it was also decreased by 59% in Moderate drought regimen.
Ammonia oxidizing bacteria (AOB) relative abundance (measured as the relative abundance of gene amoA) at 30 days post-fertilization. Pots were fertilized with ammonium sulphate (A); ammonium sulphate + DMPP (A+D) and potassium nitrate (KNO3−). Significant differences between treatments from pots with soil are marked with a lowercase letter. Significant differences between treatments from pots with soil and plant are marked with a capital letter. For both ANOVA, the Duncan Test was used (p < 0.05; n = 3). The Mann-Whitney U test was used for the comparison between the absence or presence of plant within the same fertilization treatment. Significant differences at p < 0.05 are marked with an asterisk (*) only in the “Soil + Plant” treatment
Discussion
One of the possibilities to achieve suitable use of nitrogen (N) could be a proper crop rotation (Macdonald et al. 2005; Moreau et al. 2012). The heat tolerance and drought resistance that sorghum presents (Smith and Frederiksen 2000; Hadebe et al. 2017) make it a good option as a catch crop for the summer season in a crop rotation. The use of this kind of crop absorbs the exceeding soil N from precedent crop, reducing the N losses through NO3− leaching (Askegaard et al. 2005; Berntsen et al. 2006). This is in line with our results since in the three years of experiment, soil NO3− content from soil that held sorghum presented a decrease between 44% and 92% compared to soil with fallow (Fig. 1b). In this way, the use of sorghum reduces the amount of NO3− that can be leached during heavy rain events from the autumn-winter period (Gabriel et al. 2012), which happens before the following wheat crop develops a high N uptake capacity. Moreover, the substrate for denitrifying bacteria that usually produces greenhouse gases, such as N2O (Smith et al. 2008), is also reduced. On the other hand, sorghum is also an interesting crop due to its allelopathy. Although firstly was used as a weed control alternative (Purvis et al. 1985), now it is studied for its potential BNI role. The inhibition mediated by BNIs is time dependant, but also highly conditioned by the environment, and hence the nitrification inhibition under field conditions was only detected during the first year of our experiment (Fig. 1a). The field experiment took place in three close locations with similar physicochemical soil properties (Supplementary Table 1) as it can be noticed by the lack of effect over the relative amoA abundance from the soil with fallow among the three years. Therefore, soil properties cannot be the reason why nitrification was not reduced in soil with sorghum. In addition, BNI exudation is dependent on the physiological state and development of the plant (Sarr et al. 2021), but also on the soil water status and temperature. Thus, both biotic and abiotic stresses that affect crop growth or nutrient uptake might modify the rate of BNI exudation. The growing period of the year 2017 was drier compared to the years 2018 and 2019 (Fig. 1a), ensuring an aerobic soil condition that favour nitrification. However, the amoA relative gene abundance of soil that held sorghum presented a reduction of 50% compared to fallow soil. On the contrary, the years 2018 and 2019 were characterized by higher soil moisture and reduced soil air pores that might reduce nitrification processes (Menéndez et al. 2012; Barrena et al. 2017; Torralbo et al. 2017). Although sorghum was not able to significantly inhibit soil nitrification under these conditions, soil nitrification was reduced by 16% in the year 2019 compared to soil with fallow, when accumulated precipitation was average compared to the previous two years. Therefore, BNI activity in sorghum plants may be related to water availability, increasing the BNI capacity when sorghum is under drought stress. Hence, more experiments at different times during the sorghum and the following crop periods should be carried out to confirm this hypothesis, since sorghum could be a good candidate as a catch crop by reducing soil NO3− content and affecting the nitrifying populations for the following crop.
Several studies have addressed the effects of drought stress on sorghum plants (Saini and Westgate 1999; Hadebe et al. 2017; Prasad et al. 2021). However, there is not much knowledge of how stress affects the regulation of metabolites present in root exudates, such as BNIs. To our knowledge, only Ghatak et al. (2021) studied the BNI root exudation and its composition of pearl millet plants (Pennisetum glaucum L.) under different drought stresses. These authors found a genotype-dependent enhancement of BNI activity after a defined period of drought stress. In climate change scenarios where aridity is increasing in several areas of the globe (Greve et al. 2019), it can be of importance to investigate the effects of drought stress on the BNI exudation capacity from other types of plants. Because of the environmental conditions seemed to condition BNI release, sorghum plants were exposed to different water stress under controlled conditions, which allowed us to investigate in more detail the effects on potential BNI exudation by means of soil nitrification inhibition. As expected, sorghum aboveground biomass was greatly reduced under moderate drought conditions (Fig. 2a) indicating that dry matter accumulation, which is the result of photosynthesis and nutrient uptake from the soil, was seriously affected (Hasan et al. 2017). Regardless of N fertilization, sorghum plants from Moderate drought regimen showed a 47% reduction in net photosynthesis as a consequence of a low stomata conductance. During the early stages of drought stress (10 days post-fertilization), stomata regulated the amount of water transpired while maintained similar net photosynthetic rate, gs and Ci that before the imposition of the water deprivation. However, both net photosynthetic rate and gs decreased but Ci gradually increased as long as drought stress progressed, which may indicate other factors rather than stomatal closure might be the main responsible of the reduction in photosynthetic rate. Similar results have been observed in naked oat (Zhang et al. 2022). In addition, the reduction of net CO2 assimilation, but the increase of Ci might indicate an imbalance in the photochemical activity of PSII, leading to an overexcitation and subsequent photoinhibitory damage of the PSII reaction centre (Kaiser 1987; Meng et al. 2016). This coincides with our results where Moderate drought stress made 12% lower the sorghum leaves Fv/Fm from all fertilized treatments compared to those of Watered regimen (Fig. 3d). Although non-stomatal processes such us damages in chloroplast might have an impact on decreasing photosynthetic rates under drought stress, the main cause of reduced photosynthesis under drought stress in C4 plants is generally a stomatal closure (Chaves et al. 2011), which present a close correlation between stomatal conductance and photosynthetic rate in sweet sorghum plants under drought conditions (Tingting et al. 2010). In line with these results, we also found that sorghum plants from Moderate drought regimen presented a 48% reduction in stomatal conductance (Fig. 3b). Furthermore, the C isotopic signature has been frequently used as an indicator for water use and drought stress of plants (Eggels et al. 2021). Under water deficit conditions, plants do not discriminate against the uptake of the heavier C isotope (13 C) due to closing stomata, leading to an enrichment of 13 C in biomass that increases the δ 13 C value (Farquhar et al. 1982, 1989). Our results from the microcosm experiment are in line with that since sorghum plants from Moderate drought regimen showed higher δ 13 C values compared to those of Watered regimen (Fig. 2c).
The inhibition of nitrification withholds the oxidation of NH4+ into NO3− in the soil (Clough et al. 2020). For this reason, microcosms of soil with plant from Moderate drought presented higher soil NH4+ contents (Fig. 4a) and lower amoA relative abundance (Fig. 5) than that of Watered regimen, which implies a possible signal of inhibited nitrification through BNIs (Subbarao et al. 2017). However, higher soil NH4+ and NO3− content can be noticed in all treatments from Moderate drought regimen compared to Watered regimen (Fig. 4) accompanied by a reduction in dry biomass (Fig. 2a). Therefore, the difference in soil NH4+ content may not be fully attributed to nitrification inhibition since plants under drought stress might reduce water and nutrient uptake that show low plant N content (Ogbaga et al. 2016; Sun et al. 2020). To further characterize plant N acquisition N isotopic composition from sorghum leaves was determined. Since the plant δ 15 N value tend to be similar to that of the acquired N source, changes in δ 15 N values could indicate the type of N source that the plant had during sorghum plants from this treatment its development (Werner and Schmidt 2002). Plants under ammonium nutrition often present low δ 15 N values (Ariz et al. 2011). It is known that drought produce changes in the soil pH (Wang et al. 2021) and this, in turn, modifies the NH3/NH4+ balance, which could affect the plant δ 15 N. However, Ariz et al. (2018) described a depletion of δ 15 N values when NH4+ was the N source in a wide range of external pH, indicating its independence of NH3 formation in solution. Then, any change in NH3/NH4+ balance should not affect the plant to present low values of leaf δ 15 N when NH4+ is the source of N. Nonetheless, the δ15N values observed in A treatment from Watered regimen, which were not so distant from those of KNO3− treatment (Fig. 2b), could suggest that the main source of N while plants were growing was the NO3− formed during nitrification since this process enriches the soil δ 15 N (Delwiche and Stein 1970; Herman and Rundel 1989; Jones and Dalal 2017). Nevertheless, A treatment from Moderate drought regimen showed similar δ 15 N values to A+D treatment, indicating that sorghum plants from this treatment may had ammonium nutrition rather than nitric nutrition. This preference for NH4+ uptake could be a result of higher BNI activity and a reduction of nitrifiers (Fig. 5). In this sense, low leaf δ15 N values are also reported as an indicator of BNI activity and reduced soil microbial nitrification (Jones and Dalal 2017; Karwat et al. 2018). The application of the synthetic nitrification inhibitor maintained the amoA relative abundance at the level of KNO3− treatment where no NH4+ was added (Fig. 5). These results are in line with other studies where DMPP showed great inhibition of nitrifying population (Barrena et al. 2017; Torralbo et al. 2017; Bozal-Leorri et al. 2021b; Corrochano-Monsalve et al. 2021a). On the other hand, nitrifiers from the A treatment experienced a huge increase after the application of NH4+-based fertilizer, especially in microcosms of soil without plant from Moderate drought regimen (Fig. 5). The low WFPS from Moderate drought regimen was a more aerobic environment where nitrifiers could carry on the nitrification process (Arp and Stein 2003). Nevertheless, the amoA relative abundance of A treatment from Moderate drought regimen was reduced by 25% compared to Watered treatment when sorghum plants were present and by 59% compared to A treatment of pots of soil without plant from Moderate drought regimen (Fig. 5). It is generally accepted that the uptake of nutrients by crop plants is reduced in dry-soil conditions (Ogbaga et al. 2016), but plants may also increase the production of root exudates in order to increase chelation and uptake of nutrients (Henry et al. 2007). The increase of root exudates is an extra C supply that can be used by heterotrophic microorganisms, to increase its abundance (Surey et al. 2020). This matches with the increased total bacterial abundance found in A treatment from Moderate drought regimen (Supplementary Fig. 2a). However, nitrifying microorganisms were decreased (Fig. 5). Based on our results, we suggest that the exudation of BNI molecules is also raised within the increment of root exudations, indicating that sorghum has the potential to increase BNI release under limited water conditions to ensure high soil NH4+-N pool for crop uptake due to a reduction of nitrifying microorganisms.
Conclusion
Sorghum crop could be a good option as a catch crop to reduce potential N losses through NO3− leaching because the presence of sorghum reduced the soil NO3− content between 44% and 92% compared to fallow soils. Furthermore, the presence of sorghum reduced the amoA relative gene abundance by 50% presumably due to its BNI exudation ability. However, BNI activity in sorghum plants may be related to water availability since they only reduced the amoA relative abundance during the driest year. Under moderate drought conditions, sorghum plants presented a great reduction of aboveground biomass together with reduced photosynthetic rate, stomatal conductance, Fv/Fm, and less negative δ 13 C values. In this way, lower δ15 N values detected in A treatment from Moderate drought regimen than those of Watered regimen indicated that sorghum plants from this treatment had more ammonium nutrition. Finally, sorghum plants had the potential to reduce amoA gene relative abundance by 25% under Moderate drought regimen compared to Watered treatment and by 59% regarding microcosms without plants. Moderate drought conditions under controlled conditions confirmed the observed reduction of amoA relative abundance in the year 2017, which presumably might have happened through the increase of root exudates like BNIs caused by lower water availability.
Data availability
Raw data used to generate the presented results are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Abbreviations
- AOB:
-
Ammonia-oxidizing bacteria
- BNI:
-
Biological nitrification inhibition
- BNIs:
-
Biological nitrification inhibitors
- DMPP:
-
3,4-dimethylpyrazole phosphate
- SNIs:
-
Synthetic nitrification inhibitors
- WFPS:
-
Water filled pore space
References
Afzal MR, Zhang M, Jin H, Zhang M, Ding M, Hu J, Zhu Y, Wang G, Raza S, Zeng H, Gao X, Subbarao GV (2020) Post-translational regulation of plasma membrane H+-ATPase is involved in the release of biological nitrification inhibitors from sorghum roots. Plant Soil 450:357–372. https://doi.org/10.1007/s11104-020-04511-6
Allegrini M, Gomez EDV, Zabaloy MC (2017) Repeated glyphosate exposure induces shifts in nitrifying communities and metabolism of phenylpropanoids. Soil Biol Biochem 105:206–215. https://doi.org/10.1016/j.soilbio.2016.11.024
Ariz I, Boeckstaens M, Gouveia C, Martins AP, Sanz-Luque E, Fernández E, Soveral G, Wiren N von, Marini AM, Aparicio Tejo PM, Cruz C (2018) Nitrogen isotope signature evidences ammonium deprotonation as a common transport mechanism for the AMT-Mep-Rh protein superfamily. Sci Adv 4. https://doi.org/10.1126/sciadv.aar3599
Ariz I, Cruz C, Moran JF, González-Moro MB, García-Olaverri C, González-Murua C, Martins-Louçao MA, Aparicio-Tejo PM (2011) Depletion of the heaviest stable N isotope is associated with NH4+/NH3 toxicity in NH4+-fed plants. BMC Plant Biol 11:83. https://doi.org/10.1186/1471-2229-11-83
Arp DJ, Stein LY (2003) Metabolism of inorganic N compounds by ammonia-oxidizing bacteria. Crit Rev Biochem Mol Biol 38:471–495. https://doi.org/10.1080/10409230390267446
Askegaard M, Olesen JE, Kristensen K (2005) Nitrate leaching from organic arable crop rotations: effects of location, manure and catch crop. Soil Use Manag 21:181–188. https://doi.org/10.1111/j.1475-2743.2005.tb00123.x
Barrena I, Menéndez S, Correa-Galeote D, Vega-Mas I, Bedmar EJ, Gonzalez-Murua C, Estavillo JM (2017) Soil water content modulates the effect of the nitrification inhibitor 3,4-dimethylpyrazole phosphate (DMPP) on nitrifying and denitrifying bacteria. Geoderma 303:1–8. https://doi.org/10.1016/j.geoderma.2017.04.022
Behrens S, Azizian MF, McMurdie PJ, Sabalowsky A, Dolan ME, Semprini L, Spormann AM (2008) Monitoring abundance and expression of “Dehalococcoides” species chloroethene-reductive dehalogenases in a tetrachloroethene-dechlorinating flow column. Appl Environ Microbiol 74:5695–5703. https://doi.org/10.1128/AEM.00926-08
Berntsen J, Olesen JE, Petersen BM, Hansen EM (2006) Long-term fate of nitrogen uptake in catch crops. Eur J Agron 25:383–390. https://doi.org/10.1016/j.eja.2006.07.006
Bozal-Leorri A, Corrochano-Monsalve M, Arregui LM, Aparicio-Tejo PM, González-Murua C (2021a) Biological and synthetic approaches to inhibiting nitrification in non-tilled Mediterranean soils. Chem Biol Technol Agric 8:1–12. https://doi.org/10.1186/s40538-021-00250-7
Bozal-Leorri A, González-Murua C, Marino D, Aparicio-Tejo PM, Corrochano-Monsalve M (2021b) Assessing the efficiency of dimethylpyrazole-based nitrification inhibitors under elevated CO2 conditions. Geoderma 400:115–160. https://doi.org/10.1016/j.geoderma.2021.115160
Bozal-Leorri A, Corrochano-Monsalve M, Arregui LM, Aparicio-Tejo PM, González-Murua C (2023) Evaluation of a crop rotation with biological inhibition potential to avoid N2O emissions in comparison with synthetic nitrification inhibition. J Environ Sci 127:222–233. https://doi.org/10.1016/j.jes.2022.04.035
Cawse P (1967) The determination of nitrate in soil solutions by ultraviolet spectrophotometry. Analyst 92:311–315. https://doi.org/10.1039/AN9679200311
Chaves MM, Costa JM, Saibo NJM (2011) Recent advances in photosynthesis under drought and salinity. Adv Bot Res 57:49–104. https://doi.org/10.1016/B978-0-12-387692-8.00003-5
Clough TJ, Cardenas LM, Friedl J, Wolf B (2020) Nitrous oxide emissions from ruminant urine: science and mitigation for intensively managed perennial pastures. Curr Opin Environ Sustain 47:21–27. https://doi.org/10.1016/j.cosust.2020.07.001
Corrochano-Monsalve M, González-Murua C, Bozal-Leorri A, Lezama L, Artetxe B (2021) Mechanism of action of nitrification inhibitors based on dimethylpyrazole: A matter of chelation. Sci Total Environ 752:141885–141896. https://doi.org/10.1016/j.scitotenv.2020.141885
Coskun D, Britto DT, Shi W, Kronzucker HJ (2017) How plant root exudates shape the nitrogen cycle. Trends Plant Sci 22:661–673. https://doi.org/10.1016/j.tplants.2017.05.004
Daryanto S, Wang L, Jacinthe PA (2017) Global synthesis of drought effects on cereal, legume, tuber and root crops production: a review. Agri Water Manag 179:18–33. https://doi.org/10.1016/j.agwat.2016.04.022
Dayan FE, Rimando AM, Pan Z, Baerson SR, Gimsing AL, Duke SO (2010) Sorgoleone Phytochem 71:1032–1039. https://doi.org/10.1016/j.phytochem.2010.03.011
Delwiche CC, Steyn PL (1970) Nitrogen isotope fractionation in soils and microbial reactions. Environ Sci Technol 4:929–935. https://doi.org/10.1021/es60046a004
Eggels S, Blankenagel S, Schön CC, Avramova V (2021) The carbon isotopic signature of C4 crops and its applicability in breeding for climate resilience. Theor Appl Genet 134:1663–1675. https://doi.org/10.1007/s00122-020-03761-3
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Biol 40:503–537
Farquhar GD, O’Leary MH, Berry JA (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Funct Plant Biol 9:121–137
Fleury D, Jefferies S, Kuchel H, Langridge P (2010) Genetic and genomic tools to improve drought tolerance in wheat. J Exp Bot 61:3211–3222. https://doi.org/10.1093/jxb/erq152
Gabriel JL, Muñoz-Carpena R, Quemada M (2012) The role of cover crops in irrigated systems: Water balance, nitrate leaching and soil mineral nitrogen accumulation. Agric Ecosys Environ 155:50–61. https://doi.org/10.1016/j.agee.2012.03.021
Ghatak A, Schindler F, Bachmann G, Engelmeier D, Bajaj P, Brenner M, Subbarao GV, Weckwerth W (2021) Root exudation of contrasting drought-stressed pearl millet genotypes conveys varying biological nitrification inhibition (BNI) activity. Biol Fertil Soils 1–16. https://doi.org/10.1007/s00374-021-01578-w
Greve P, Roderick ML, Ukkola AM, Wada Y (2019) The aridity index under global warming. Environ Res Lett 14:124006–124018. https://doi.org/10.1088/1748-9326/ab5046
Hadebe ST, Modi AT, Mabhaudhi T (2017) Drought tolerance and water use of cereal crops: a focus on sorghum as a food security crop in sub-saharan Africa. J Agron Crop Sci 203:177–191. https://doi.org/10.1111/jac.12191
Harter J, Krause HM, Schuettler S, Ruser R, Fromme M, Scholten T, Kappler A, Behrens S (2014) Linking N2O emissions from biochar-amended soil to the structure and function of the N-cycling microbial community. ISME J 8:660–674. https://doi.org/10.1038/ismej.2013.160
Hasan SA, Rabei SH, Nada RM, Abogadallah GM (2017) Water use efficiency in the drought-stressed sorghum and maize in relation to expression of aquaporin genes. Biol Plant 61:127–137. https://doi.org/10.1007/s10535-016-0656-9
Henry A, Doucette W, Norton J, Bugbee B (2007) Changes in crested wheatgrass root exudation caused by flood, drought, and nutrient stress. J Environ Qual 36:904–912. https://doi.org/10.2134/jeq2006.0425sc
Herman DJ, Rundel PW (1989) Nitrogen isotope fractionation in burned and unburned chaparral soils. Soil Sci Soc Am J 53:1229–1236. https://doi.org/10.2136/sssaj1989.03615995005300040040x
Hochholdinger F (2016) Untapping root system architecture for crop improvement. J Exp Bot 67:4431–4434. https://doi.org/10.1093/jxb/erw262
Huerfano X, Fuertes-Mendizabal T, Dunabeitia MK, Gonzalez-Murua C, Estavillo JM, Menendez S (2015) Splitting the application of 3,4-dimethylpyrazole phosphate (DMPP): influence on greenhouse gases emissions and wheat yield and quality under humid Mediterranean conditions. Eur J Agron 64:47–57. https://doi.org/10.1016/j.eja.2014.11.008
Huerfano X, Estavillo JM, Fuertes-Mendizabal T, Torralbo F, Gonzalez-Murua C, Menendez S (2018) DMPSA and DMPP equally reduce N2O emissions from a maize-ryegrass forage rotation under Atlantic climate conditions. Atmos Environ 187:255–265. https://doi.org/10.1016/j.atmosenv.2018.05.065
IPCC (Intergovernmental Panel on Climate Change, The United Nations) (2014) Synthesis report summary chapter for policymakers. IPCC, p 31
Ishikawa T, Subbarao GV, Ito O, Okada K (2003) Suppression of nitrification and nitrous oxide emission by the tropical grass Brachiaria humidicola Roots: the dynamic interface between plants and the Earth. Springer, Dordrecht, pp 413–419. https://doi.org/10.1007/978-94-017-2923-9_40
Jones AR, Dalal RC (2017) Enrichment of natural 15 N abundance during soil N losses under 20 years of continuous cereal cropping. Sci Total Environ 574:282–287. https://doi.org/10.1016/j.scitotenv.2016.08.192
Kaiser WM (1987) Effects of water deficit on photosynthetic capacity. Physiol Plant 71:142–149. https://doi.org/10.1111/j.1399-3054.1987.tb04631.x
Karwat H, Egenolf K, Nuñez J, Rao I, Rasche F, Arango J, Cadisch G (2018) Low 15 N natural abundance in shoot tissue of Brachiaria humidicola is an indicator of reduced N losses due to biological nitrification inhibition (BNI). Front Microbiol 9:2383. https://doi.org/10.3389/fmicb.2018.02383
Karwat H, Sparke MA, Rasche F, Arango J, Nuñez J, Rao IM, Moreta D, Cadisch G (2019) Nitrate reductase activity in leaves as a plant physiological indicator of in vivo biological nitrification inhibition by Brachiaria humidicola Plant Physiol Biochem 137:113–120. https://doi.org/10.1016/j.plaphy.2019.02.002
Lassaletta L, Billen G, Grizzetti B, Anglade J, Garnier J (2014) 50 year trends in nitrogen use efficiency of world cropping systems: the relationship between yield and nitrogen input to cropland. Environ Res Lett 9:105011–105021. https://doi.org/10.1088/1748-9326/9/10/105011
LeBauer DS, Treseder KK (2008) Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89:371–379. https://doi.org/10.1890/06-2057.1
Li X, Sørensen P, Olesen JE, Petersen SO (2016) Evidence for denitrification as main source of N2O emission from residue-amended soil. Soil Biol Biochem 92:153–160. https://doi.org/10.1016/j.soilbio.2015.10.008
Linn DM, Doran JW (1984) Effect of water-filled pore space on carbon dioxide and nitrous oxide production in tilled and nontilled soils. Soil Sci Soc Am J 48:1267–1272. https://doi.org/10.2136/sssaj1984.03615995004800060013x
Macdonald AJ, Poulton PR, Howe MT, Goulding KWT, Powlson DS (2005) The use of cover crops in cereal-based cropping systems to control nitrate leaching in SE England. Plant Soil 273:355–373. https://doi.org/10.1007/s11104-005-0193-3
Menéndez S, Barrena I, Setien I, González-Murua C, Estavillo JM (2012) Efficiency of nitrification inhibitor DMPP to reduce nitrous oxide emissions under different temperature and moisture conditions. Soil Biol Biochem 53:82–89. https://doi.org/10.1016/j.soilbio.2012.04.026
Meng LL, Song JF, Wen J, Zhang J, Wei JH (2016) Effects of drought stress on fluorescence characteristics of photosystem II in leaves of Plectranthus scutellarioides Photosynthetica 54:414–421. https://doi.org/10.1007/s11099-016-0191-0
Momesso L, Crusciol CA, Soratto RP, Vyn TJ, Tanaka KS, Costa CH, Cantarella H (2019) Impacts of nitrogen management on no-till maize production following forage cover crops. Agron J 111:639–649. https://doi.org/10.2134/agronj2018.03.0201
Moreau P, Ruiz L, Raimbault T, Vertès F, Cordier MO, Gascuel-Odoux C, Durand P (2012) Modeling the potential benefits of catch-crop introduction in fodder crop rotations in a Western Europe landscape. Sci Total Environ 437:276–284. https://doi.org/10.1016/j.scitotenv.2012.07.091
Nardi P, Akutsu M, Pariasca-Tanaka J, Wissuwa M (2013) Effect of methyl 3-4-hydroxyphenyl propionate, a Sorghum root exudate, on N dynamic, potential nitrification activity and abundance of ammonia-oxidizing bacteria and archaea. Plant Soil 367:627–637. https://doi.org/10.1007/s11104-012-1494-y
Ogbaga CC, Stepien P, Dyson BC, Rattray NJ, Ellis DI, Goodacre R, Johnson GN (2016) Biochemical analyses of sorghum varieties reveal differential responses to drought. PLoS ONE 11:e0154423
Patton CJ, Crouch SR (1977) Spectrophotometric and kinetics investigation of the Berthelot reaction for the determination of ammonia. Anal Chem 49:464–469. https://doi.org/10.1371/journal.pone.0154423
Prasad VB, Govindaraj M, Djanaguiraman M, Djalovic I, Shailani A, Rawat N, Prasad PV (2021) Drought and high temperature stress in sorghum: physiological, genetic, and molecular insights and breeding approaches. Int J Mol Sci 22:9826. https://doi.org/10.3390/ijms22189826
Purvis CE, Jessop RS, Lovett JV (1985) Selective regulation of germination and growth of annual weeds by crop residues. Weed Res 25:415–421. https://doi.org/10.1111/j.1365-3180.1985.tb00664.x
Saini HS, Westgate ME (1999) Reproductive development in grain crops during drought. Adv Agron 68:59–96. https://doi.org/10.1016/S0065-2113(08)60843-3
Sarr PS, Nakamura S, Ando Y, Iwasaki S, Subbarao GV (2021) Sorgoleone production enhances mycorrhizal association and reduces soil nitrification in sorghum. Rhizosphere 17:100283–100295. https://doi.org/10.1016/j.rhisph.2020.100283
Smith CW, Frederiksen RA (eds) (2000) Sorghum: origin, history, technology, and production, vol 2. Wiley, Hoboken
Smith P, Martino D, Cai Z, Gwary D, Janzen H, Kumar P, Smith J (2008) Greenhouse gas mitigation in agriculture. Philos Trans R Soc B: Biol Sci 363:789–813. https://doi.org/10.1098/rstb.2007.2184
Subbarao GV, Ito O, Sahrawat KL, Berry WL, Nakahara K, Ishikawa T, Watanabe T, Suenaga K, Rondon M, Rao IM (2006) Scope and strategies for regulation of nitrification in agricultural systems—challenges and opportunities. Crit Rev Plant Sci 25:303–335. https://doi.org/10.1080/07352680600794232
Subbarao GV, Rondon M, Ito O, Ishikawa T, Rao IM, Nakahara K, Lascano C, Berry WL (2007) Biological nitrification inhibition (BNI)—is it a widespread phenomenon? Plant Soil 294:5–18. https://doi.org/10.1007/s11104-006-9159-3
Subbarao GV, Sahrawat KL, Nakahara K, Rao IM, Ishitani M, Hash CT, Lata JC (2013a) A paradigm shift towards low-nitrifying production systems: the role of biological nitrification inhibition (BNI). Ann Bot 112:297–316. https://doi.org/10.1093/aob/mcs230
Subbarao GV, Nakahara K, Ishikawa T, Ono H, Yoshida M, Yoshihashi T, Zhu Y, Zakir HAKM, Desphande SP, Hash CT, Sahrawat KL (2013b) Biological nitrification inhibition (BNI) activity in sorghum and its characterization. Plant Soil 366:243–259. https://doi.org/10.1007/s11104-012-1419-9
Subbarao GV, Yoshihashi T, Worthington M, Nakahara K, Ando Y, Sahrawat KL, Braun HJ (2015) Suppression of soil nitrification by plants. Plant Sci 233:155–164. https://doi.org/10.1016/j.plantsci.2015.01.012
Subbarao GV, Arango J, Kishii M, Hooper AM, Yoshihashi T, Ando Y, Nakahara K, Desphande S, Ortiz-Monasterio I, Ishitani M, Peters M, Chirinda N, Wollenberg L, Lata JC, Gerard B, Tobita S, Rao IM, Braun HJ, Kommerell V, Tohme J, Iwanaga M (2017) Genetic mitigation strategies to tackle agricultural GHG emissions: the case for biological nitrification inhibition technology. Plant Sci 262:165–168. https://doi.org/10.1016/j.plantsci.2017.05.004
Subbarao GV, Searchinger TD (2021) Opinion: a “more ammonium solution” to mitigate nitrogen pollution and boost crop yields. Proc Natl Acad Sci 118:e2107576118. https://doi.org/10.1073/pnas.2107576118
Sun Y, Liao J, Zou X, Xu X, Yang J, Chen HY, Ruan H (2020) Coherent responses of terrestrial C:N stoichiometry to drought across plants, soil, and microorganisms in forests and grasslands. Agric For Meteorol 292:108104–108112. https://doi.org/10.1016/j.agrformet.2020.108104
Surey R, Lippold E, Heilek S, Sauheitl L, Henjes S, Horn MA, Mueller CW, Merbach I, Kaiser K, Böttcher J, Mikutta R (2020) Differences in labile soil organic matter explain potential denitrification and denitrifying communities in a long-term fertilization experiment. App Soil Ecol 153:103630–103640. https://doi.org/10.1016/j.apsoil.2020.103630
Syakila A, Kroeze C (2011) The global nitrous oxide budget revisited. Greenh Gas Meas Manag 1:17–26. https://doi.org/10.3763/ghgmm.2010.0007
Tingting X, Peixi SU, Lishan S (2010) Photosynthetic characteristics and water use efficiency of sweet sorghum under different watering regimes. Pak J Bot 42:3981–3994
Torralbo F, Menéndez S, Barrena I, Estavillo JM, Marino D, González-Murua C (2017) Dimethyl pyrazol-based nitrification inhibitors effect on nitrifying and denitrifying bacteria to mitigate N2O emission. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-14225-y
Vanderlip RL (1993) How a sorghum plant develops. Kansas State University. S-3. http://www.bookstore.ksre.ksu.edu
Wang C, Sun Y, Chen HY, Yang J, Ruan H (2021) Meta-analysis shows non-uniform responses of above-and belowground productivity to drought. Sci Total Environ 782:146901. https://doi.org/10.1016/j.scitotenv.2021.146901
Werner RA, Schmidt HL (2002) The in vivo nitrogen isotope discrimination among organic plant compounds. Phytochem 61:465–484. https://doi.org/10.1016/S0031-9422(02)00204-2
Zabaloy MC, Allegrini M, Tebbe DA, Schuster K, Gomez EDV (2017) Nitrifying bacteria and archaea withstanding glyphosate in fertilized soil microcosms. App Soil Ecol 117:88–95. https://doi.org/10.1016/j.apsoil.2017.04.012
Zhang M, Fan CH, Li QL, Li B, Zhu YY, Xiong ZQ (2015) A 2-yr field assessment of the effects of chemical and biological nitrification inhibitors on nitrous oxide emissions and nitrogen use efficiency in an intensively managed vegetable cropping system. Agric Ecosyst Environ 201:43–50. https://doi.org/10.1016/j.agee.2014.12.003
Zhang X, Liu W, Lv Y, Li T, Tang J, Yang X, Bai J, Jin X, Zhou H (2022) Effects of drought stress during critical periods on the photosynthetic characteristics and production performance of Naked oat (Avena nuda L.). Sci Rep 12:1–11. https://doi.org/10.1038/s41598-022-15322-3
Acknowledgements
The authors thank for the technical and human support of phytotron and greenhouse provided by SGIker (UPV/EHU/ ERDF, EU).
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This project was funded by the Spanish Government (RTI2018-094623-B-C21 and C22 MCIU/AEI/FEDER, UE) and by the Basque Government (IT-932-16; IT-1560-22). Dr Adrián Bozal-Leorri held a grant from the Basque Government (PRE-2020-2-0142). Dr. Fernando Torralbo was supported by the European Union – Next Generation EU program. Authors thank the Jesus de Gangoiti Barrera Foundation for the financial support.
Author information
Authors and Affiliations
Contributions
ABL conducted the experiments and was the main contributor in the data processing and interpretation, also writing the manuscript. LMA prepared the experimental field site, sowed the sorghum crop and participated in formal analysis, writing, reviewing and editing. FT participated in the field experiment and in formal analysis, writing, reviewing and editing. MBGM reviewed the discussion section after journal’s revision. PAT and CGM supervised all phases of data analysis and interpretation and reviewed the entire manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Elizabeth M Baggs.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bozal-Leorri, A., Arregui, L.M., Torralbo, F. et al. Soil moisture modulates biological nitrification inhibitors release in sorghum plants. Plant Soil 487, 197–212 (2023). https://doi.org/10.1007/s11104-023-05913-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-05913-y