Abstract
Ecologists and physiologists have documented extensive variation in water use efficiency (WUE) in Arabidopsis thaliana, as well as association of WUE with climatic variation. Here, we demonstrate correlations of whole-plant transpiration efficiency and carbon isotope composition (δ13C) among life history classes of A. thaliana. We also use a whole-plant cuvette to examine patterns of co-variation in component traits of WUE and δ13C. We find that stomatal conductance (g s) explains more variation in WUE than does A. Overall, there was a strong genetic correlation between A and g s, consistent with selection acting on the ratio of these traits. At a more detailed level, genetic variation in A was due to underlying variation in both maximal rate of carboxylation (V cmax) and maximum electron transport rate (Jmax). We also found strong effects of leaf anatomy, where lines with lower WUE had higher leaf water content (LWC) and specific leaf area (SLA), suggesting a role for mesophyll conductance (g m) in variation of WUE. We hypothesize that this is due to an effect through g m, and test this hypothesis using the abi4 mutant. We show that mutants of ABI4 have higher SLA, LWC, and g m than wild-type, consistent with variation in leaf anatomy causing variation in g m and δ13C. These functional data also add further support to the central, integrative role of ABI4 in simultaneously altering ABA sensitivity, sugar signaling, and CO2 assimilation. Together our results highlight the need for a more holistic approach in functional studies, both for more accurate annotation of gene function and to understand co-limitations to plant growth and productivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The efficiency with which plants fix CO2 relative to their rate of H2O loss is called water use efficiency (WUE), and when high, WUE can mitigate the tradeoff between CO2 uptake and H2O loss. In C3 plants, low stomatal conductance (g s) minimizes water loss (transpiration, E) and can be a rapid and effective strategy; however, it results in reduced CO2 uptake (A) and growth (Schulze 1986; Geber and Dawson 1997; Condon et al. 2002). Genetically based variation in WUE has been documented in both crops and non-cultivated species (McKay et al. 2003; Hall et al. 2005). Physiologists are interested in intrinsic WUE (A/g s) as a tool for studying how the fundamental trade-off of losing water for gaining CO2 is regulated by stomatal and other physiological adjustments (Buckley and Mott 2002; Comstock 2002). Evolutionary biologists have studied variation in WUE as it is likely an important component of local adaptation (Donovan and Ehleringer 1994; Heschel et al. 2002; Geber and Griffen 2003; Caruso et al. 2005). Likewise, plant breeders have long considered WUE an important target (Passioura 1977).
WUE can be estimated in a variety of ways at various spatio-temporal scales, including with lysimeter studies, gas exchange measurements, or stable carbon isotope composition. Tissue carbon isotope composition is an increasingly popular approach, and its advantages include integration over long periods of gas exchange and development, amenability to high throughput sampling, relatively low cost, and high heritability. Stable carbon isotope composition of leaves (δ13C) (the ratio of the amount of 13C to 12C isotopes in a sample relative to a standard), provides a time-integrated estimate of intrinsic WUE (Farquhar et al. 1989; Dawson et al. 2002). In Arabidopsis thaliana (here after Arabidopsis), common garden experiments have identified substantial variation in δ13C among natural accessions and some of this variation likely represents local adaptation to climate (McKay et al. 2003, 2008; Juenger et al. 2005, 2010; Christman et al. 2008; Monda et al. 2011; Des Marais et al. 2012; Lasky et al. 2012). In addition, QTL have been identified for δ13C (Juenger et al. 2005; Masle et al. 2005; McKay et al. 2008).
In plant breeding, WUE is an important target of selection, although the complexity of the trait, and difficulty of phenotyping has prevented many breeding programs from attempting to select on WUE directly (Araus et al. 2002). Many studies have shown variation in δ13C among cultivars. In crops, one particularly successful example is an Australian wheat breeding program, where selection on δ13C in a greenhouse environment led to new varieties that had increased yield in semiarid rainfed conditions (Rebetzke et al. 2002). Conversely, in conditions where water is not limiting, selection for reduced WUE may lead to greater yields (Passioura 1977; Fischer et al. 1998).
Although it is heritable, appears to be under selection in nature, and may correlate with yield in C3 crops (Condon et al. 1987), the mechanistic basis of genetic variation in δ13C is still unclear. Variation in δ13C can be due to variation in photosynthetic biochemistry, conductance of CO2 to the leaf interior and chloroplast, or a combination of these (Seibt et al. 2008). Thus, similar leaf δ13C and similar WUE can evolve via mutations that cause low A with low conductance or mutations that cause high A with proportionally higher conductance (Farquhar et al. 1989). This is further complicated because conductance from ambient air to the interior of the leaf is influenced both by g s and additional variability of conductance into leaf mesophyll cells and chloroplasts (g m), which can change over the long-term with leaf morphology (von Caemmerer and Evans 1991; Evans et al. 1994, 2009; Tosens et al. 2012) and over the short-term through changes in protein-mediated chloroplast membrane permeability (Flexas et al. 2006; Uehlein et al. 2008; Heckwolf et al. 2011). When examining the combined effects of g s and g m, it is important to recognize that they operate in series rather than in parallel and that the regulation of g m is poorly understood. Within a genotype, g s and g m usually respond in a correlated way to environmental stimuli (Flexas et al. 2007, 2008; Warren 2008; Barbour et al. 2010) although, opposite responses have also been observed (Galle et al. 2012). Patterns of genetic covariation of g s and g m have not been investigated. However, it is known that variation in g m contributes to leaf carbon isotope discrimination, further increasing the importance of considering g s and g m in interpretations of δ13C (Warren and Adams 2006; Barbour et al. 2010).
Understanding the physiological basis of variation in δ13C and intrinsic WUE is important for improving plant productivity and understanding the evolution of wild species. Here, we report a series of experiments designed to investigate a mechanistic understanding of the physiological basis of variation in intrinsic WUE in Arabidopsis. At the coarse level, we can ask if variation in intrinsic WUE is primarily due to variation in A or g s. For example, threefold variation in g s and twofold variation in leaf N concentration among natural accessions of Arabidopsis suggest substantial variation in g s and A may separately or in concert be responsible for the observed variation in δ13C (Christman et al. 2008; Des Marais et al. 2012). Des Marais et al. (2012) found large differences in physiology between life history classes in Arabidopsis. Although, the Des Marais study focused on variation in gene expression, they also reported constitutive variation in leaf structural traits between life history classes. Winter annual types had higher intrinsic WUE. This is consistent with coordinated selection on WUE, A, and g s and life history observed in other species (Geber and Dawson 1997). Higher WUE was associated with lower leaf water content (LWC) and specific leaf area (SLA) (Des Marais et al. 2012). Taken together, these results suggest that increased leaf density is associated with higher photosynthetic capacity (Terashima et al. 2011), but may come at the cost of lower stomatal and mesophyll conductance to CO2 (Parkhurst and Mott 1990; Evans et al. 1994; Syvertsen et al. 1995; Kogami et al. 2001).
Studies in Arabidopsis have identified extensive natural variation in plant–water relations and gas exchange physiology (Juenger et al. 2005, 2010; Masle et al. 2005; Bouchabke et al. 2008; Christman et al. 2008; McKay et al. 2008; Monda et al. 2011; Des Marais et al. 2012; Pons 2012). The present study was undertaken to examine natural variation in leaf physiological traits that are the likely cause of the observed variation in δ13C and associated WUE parameters in natural accessions of Arabidopsis, and to determine if these traits vary independently or co-vary in a coordinated and predictable manner. First, we tested if the expected relationship between transpiration efficiency (shoot dry mass/transpiration; TE) and leaf δ13C was present in 96 natural accessions of Arabidopsis. In a smaller set of 18 natural accessions spanning the range of variation in δ13C, we measured rosette A, g s, and intercellular CO2 concentration (C i) and examined the relationship of C i and δ13C. To further characterize natural variation in A, we examined maximal carboxylation rate (V cmax) and photosynthetic electron transport rate (Jmax) in three accessions using photosynthetic carbon dioxide response curves (Sharkey et al. 2007). Additionally, we used gas exchange measurements coupled with online isotopic measurements to determine instantaneous carbon isotope discrimination using tunable diode laser spectroscopy (TDL) (Flexas et al. 2006; Barbour et al. 2007; Heckwolf et al. 2011) to estimate g m in stomatal regulation mutants to investigate the relationship of these mechanistically related traits (Warren et al. 2003; Yamori et al. 2006).
Materials and methods
δ13C and transpiration efficiency (Experiment 1)
Our first goal was to use a relatively high throughput approach to look for variation and co-variation across the species range. 96 natural accessions were selected from the native range of Arabidopsis to evaluate plant biomass production and water use (Nordborg et al. 2005). Individual plants were grown in 250-mL plastic cups, each filled with a standard mass of 1:1 fritted clay and Promix BT potting soil mix. We measured field capacity of the soil mix following a 24-h gravitational drain of saturated soil. Each cup was covered with parafilm and sealed with a plastic lid that had a 6-mm diameter hole. Two replicates of each of 96 ecotypes were planted and cold stratified in the dark for 7 days at 4 °C. Plants were grown in two independent growth chambers at 200 μmol m−2 s−1 PPFD in a randomized block design. Photoperiod was 12 h light/12 h dark and the temperature cycled 23/18 °C (light/dark). Every 2 days, each container was weighed and additional water was added with a syringe to bring the soil in each container to 90 % field capacity. Total transpiration (E total) was summed for the 35 days growing period for each experimental plant. Plants were harvested, and aboveground material was oven dried and weighed (DW). We assessed evaporative loss from the containers using “blanks” lacking an Arabidopsis plant. Total evaporation from the blank containers was <4 % of the average E total from pots in the experiment. Transpiration efficiency (TE) of each plant was calculated as DW/E total. Dried leaves were ground to a fine powder and δ13C was determined at the UC Davis Stable Isotope Facility (http://stableisotopefacility.ucdavis.edu/). When grown outside in free air, the use of carbon isotope discrimination, Δ, is preferred (Farquhar et al. 1982), but when growth chamber and greenhouse studies are included the value of air δ13C is uncertain and variable, thus requiring the use of leaf δ13C instead of Δ. Differences in δ13C within the same experiment indicate differences in intercellular CO2 concentration, but δ13C must be viewed with caution when comparing different experimental conditions.
Whole-shoot gas exchange (Experiment 2)
To follow up on the patterns from the 96 accessions, 18 natural accessions of Arabidopsis were used in whole-shoot gas exchange experiments to evaluate the physiological basis of variation in δ13C. Eleven of the accessions were spring annuals, and seven were winter annuals. Four replicates of each genotype were grown in a growth chamber in a randomized block design. Each plant was grown in a pot constructed from a 50-mL centrifuge tube with the bottom cut off and “planted” in a 164-mL Conetainer™ pots (Stuewe and Sons, Corvallis, OR) filled with a 1:1 mixture of potting mix (Sunshine mix, Sun Gro Horticulture, Bellevue, WA) and fritted clay. After planting, pots were cold stratified at 4 °C for 7 days, then transferred to a growth chamber. Photoperiod was 12 h with 350 μmol m−2 s−1 PPFD and temperature was cycled 23/20 °C (light/dark).
Instantaneous whole-canopy gas exchange rate was measured using a LI-6400 (Li-Cor Inc., Lincoln, NE, USA) with a custom-made whole-shoot Arabidopsis cuvette (Fig. 1). Cuvette PPFD was maintained at 350 μmol m−2 s−1 PPFD, CO2 was maintained at 400 μmol mol−1, and temperature and relative humidity were set to growth chamber conditions. Each block was measured on a different day, 28–31 days after sowing. Following measurements for each plant, leaf area was determined from digital photographs of the rosette using Scion Image (Scion Corporation, Frederick, MD, USA).
Cuvette used for whole-plant gas exchange measurements. The cuvette is mounted on the LI-6400 IRGA and cuvette control system (gold-plated panel, fan and aluminum box, upper photograph). This system allows accurate, rapid measurement of CO2 (A) and H2O (E) exchange of whole shoots of Arabidopsis plants. The whole-plant cuvette incorporates a leaf temperature thermocouple that interfaces directly with the LI-6400. Intrinsic WUE (A/g s), stomatal conductance (g s), internal CO2 concentration (C i), and other variables can be calculated from these measurements. All interior surfaces are Teflon coated or Ni-plated, the cuvette has extremely low leak rates when operated in lab conditions with high external CO2, and the circular design provides excellent mixing using the LI-6400 fans. Plants can be rapidly changed using multiple inserts (lower photo)
A:C i responses were measured for three accessions (Tsu-1, SQ-8, and Kas-1) which differed in A and δ13C. Cuvette conditions were the same as above, but light was increased to 1,000 μmol m−2 s−1 PPFD. Photosynthetic carbon dioxide response curves were measured on four rosettes of each accession. The number of replications of A:C i measurements were limited by chamber environment equilibration time at each CO2 set point. The least squares iterative curve-fitting procedure (Sharkey et al. 2007) model was used to fit Farquhar et al.’s (1980) biochemical model of photosynthesis and obtain maximal carboxylation rate (V cmax) and maximal photosynthetic electron transport rate (Jmax).
Leaf water content (Experiment 3)
39 natural accessions from the native range of Arabidopsis previously used in Mckay et al. (2003) were measured for LWC and leaf δ13C. Four replicates of each ecotype were grown in a greenhouse at UC Davis in a randomized block design. Seeds were sown in 250-mL pots in peat-based potting mix with slow-release fertilizer and vernalized at 4 °C for 5 days. Day length was extended to 16 h using supplemental lighting at 350 μmol m−2 s−1 PPFD. Greenhouse mean relative humidity and air temperature were 44 % and 23 °C, respectively. Shoots were harvested at the initiation of flowering and shoot fresh weight (FW) was determined, leaf area was determined from scans of dissected rosettes using Scion Image (Scion Corporation, Frederick, MD, USA), and shoots were dried and weighed (DW). Entire dried shoots were ground and processed for carbon isotope analysis at the UC Davis Stable Isotope Facility (http://stableisotopefacility.ucdavis.edu/). LWC (%) was calculated as 100 × (FW − DW)/DW.
Mesophyll conductance (Experiment 4)
Arabidopsis seeds of ecotype Columbia and the abi4 mutant provided by the Arabidopsis Biological Resource Center (Columbus, OH, USA) were used for leaf mesophyll conductance to CO2 (g m) experiments. Seven replicates of each genotype were grown in a growth chamber in a randomized block design. Photoperiod was 12 h with 350 μmol m−2 s−1 PPFD and temperature was cycled 23/20 °C (light/dark). A LI-6400 (Li-Cor Inc., Lincoln, NE, USA) with whole-shoot Arabidopsis cuvette (Fig. 1) was coupled with online isotopic measurements of CO2 entering and leaving the shoot chamber to determine instantaneous carbon isotope discrimination and g m using TDL (Flexas et al. 2006; Barbour et al. 2007; Heckwolf et al. 2011). Calculations for g m were based on whole-shoot gas exchange measurements at 350, 700, and 175 (μmol m−2 s−1) PPFD using the slope-based approach given in Evans et al. (1986). Shoots were harvested after gas exchange, leaf area was determined from rosette photographs using Scion Image (Scion Corporation, Frederick, MD, USA), and shoots were dried and weighed (DW). LWC (%) was calculated as above and SLA was calculated as rosette area/DW.
Statistical analysis
We analyzed phenotypic data for physiological traits using standard fixed effect ANOVAs with the Proc GLM in SAS (SAS Institute 1999). We estimated correlations among physiological traits as the standard Pearson product-moment correlation between genotype means.
In the case of the TE experiment, we analyzed phenotypic data for physiological traits using a linear mixed model analysis with the Proc Mixed procedure in SAS (SAS Institute 1999). We fit a model including accessions as a random effect and chamber, experiment, and their interaction as fixed effects. The variance component for the random effect was estimated using restricted maximum likelihood (REML) and assessments of significance were based on likelihood ratio tests (Little et al. 1996). We obtained empirical best linear unbiased predictors (BLUPs) associated with the random effects and consider these breeding values for each accessions. BLUPs are robust estimates of the impact of a particular accession on the measured trait while controlling for the fixed effects (chamber and experimental run). For TE, we fit a model that included both chamber and experimental run as a fixed effect. For δ13C, we fit a simpler model including accession as a random variable and experimental run as a fixed effect. In this case, factors associated with chamber could not be included because replicates within each experimental run were pooled for mass spectroscopy analysis. All subsequent analyses involving TE and δ13C rely on BLUP estimates. The TE and δ13C values were normally distributed and residuals from analyses did not exhibit heteroscedasticity.
We estimated broad-sense heritability by computing the ratio V G/V P, where V G equals the among-accession variance component and V P equals the total phenotypic variance for the study phenotypes. We estimated genetic correlations (r G) among TE and δ13C as the standard Pearson product-moment correlation between genotype means or BLUPs.
Results and discussion
Variation in TE and δ13C
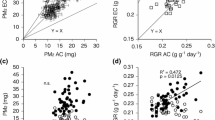
The 96 natural accessions of Arabidopsis in experiment 1 (Table 1) exhibited considerable variation in time-integrated measures of water use efficiency, i.e., whole-plant TE and δ13C. We observed a 3.33 g kg−1 and 5.12 ‰ range of variation in TE and δ13C among accessions, respectively, (TE mean = 2.02 ± 0.28 g kg−1) (δ13C mean = −30.64 ± 0.90 ‰). In both cases, we observed significant broad-sense heritability (TE, H 2 = 0.09, accession P = 0.031; δ13C, H 2 = 0.667, accession P = 0.001). For the experiment 1, we found replication block, growth chamber, and their interaction were significant sources of environmental variation in TE (in all cases, P < 0.005). Likewise, we found that the replication block was a significant source of environmental variation for δ13C (P < 0.0001). Despite the low heritability of the TE data, our experimental design and analysis allowed us to estimate breeding values as BLUPs. Spring accessions fit the expected positive relationship between TE and δ13C (r 2G = 0.265, P < 0.0001, Fig. 2). The winter annuals had greater intrinsic WUE as indicated by δ13C than the spring annuals, but this was not related to TE (r 2G = 0.011, P = 0.531, Fig. 2). Together these data suggest that variation in δ13C is likely due to stomatal limitations (on C i) in the spring accessions, but in winter accessions, other mechanisms (like g m) not affecting water loss may be leading to variation in δ13C (Seibt et al. 2008). Alternatively, variation in root carbon allocation unaccounted for in TE may explain the observed pattern in winter accessions. In principle, the greater belowground allocation in winter accessions could result in lower TE without affecting δ13C, but this hypothesis remains to be tested.
Relationships of transpiration efficiency (TE) and leaf carbon isotope composition (δ13C) among 96 natural accessions of Arabidopsis thaliana. Symbols represent best linear unbiased predictors (BLUPs) associated with breeding values for each accession (see text). Open and filled symbols represent spring and winter accession means, respectively. Lines represent linear regression; r 2 and P values are given
Variation in components of WUE
The 18 natural accessions of Arabidopsis in experiment 2 were selected to represent a wide range of intrinsic WUE as indicated by δ13C (Table 1). Whole-plant gas exchange measurements in a custom cuvette (Fig. 1) showed that these lines also exhibit considerable variation in whole rosette A and g s in a common environment (Fig. 3). Accession mean whole rosette A ranged between 10 and 16 μmol m−2 s−1, but the heritability was not significantly different from zero (P = 0.137). g s showed significant genetic variation, ranging between 0.17 and 0.45 mol m−2 s−1 with a heritability of H 2 = 0.33 (accession P value = 0.002). In addition, g s was a better predictor of variation in δ13C than A. We found a significant negative correlation between δ13C and g s among accessions (r 2 = 0.40, P = 0.0027), and a weaker correlation between δ13C and A (r 2 = 0.25, P = 0.036). In general, the high conductance lines had low intrinsic WUE, as indicated by δ13C, but there was a wide range of δ13C in the low conductance lines, suggesting additional sources of variation. The expected negative correlation between δ13C and g s was largely caused by the spring accessions. The winter accessions tended to show the opposite pattern (not significant), with the exception of Tamm-2, an accession from Finland that had the highest g s of all.
Relationships between assimilation (A), stomatal conductance (g s), and leaf carbon isotope composition (δ13C) at 350 μmol photons m−2 s−1 from whole-shoot gas exchange of 18 accessions of Arabidopsis selected from the larger panel of accessions to represent extremes in δ13C. Open and filled symbols represent spring and winter accession means, respectively. Lines represent linear regression; r 2 and P values are given
Despite the lack of heritability of A and the weak correlation of A with δ13C, we did find a significant positive correlation between g s and A among accessions (r 2 = 0.78, P = 0.00001). This is consistent with the optimization of stomatal regulation to maximize carbon gain while minimizing the water loss (Katul et al. 2010). Accessions that have high conductance should be under selection for increased biochemical capacity (Bloom et al. 1985). Although, it is not formally stated, such optimality approaches interpret consistent patterns of correlation in physiological traits (Reich et al. 1997) as evidence of selection optimizing their ratios or covariances (Donovan et al. 2011). Under such a scenario, selection would favor mutations that lead to a co-limitation of g s and RuBP utilization and regeneration.
In general, winter Arabidopsis accessions had lower g s and A than spring Arabidopsis accessions. Across accessions there was large variation in C i /C a, but it was only weakly related to δ13C (Fig. 4). No consistent difference in C i /C a was seen between the winter and spring annuals.
Relationship between the ratio of intercellular to atmospheric partial pressure CO2 (C i/C a) at 350 μmol photons m−2 s−1 and carbon isotope composition (δ13C). Open and filled symbols represent spring and winter accession means, respectively. Line represents linear regression; r 2 and P values are given
The overall finding of experiment 2 was that accessions with low g s and high δ13C had lower A compared to low δ13C accessions. Overall, these data are consistent with large effects of g s on δ13C, but the weaker correlation of C i and δ13C suggest a more complex mechanism than predicted by theory. To better understand processes limiting photosynthesis in Arabidopsis accessions, we conducted detailed CO2 response curves of assimilation for low and high WUE spring accessions Tsu-1 and SQ-8 and high WUE winter accession Kas-1. Maximum carboxylation rate of rubisco (V cmax) was higher in low WUE Tsu-1 (δ13C = −29.7) than Sq-8 (δ13C = −28.6) (P = 0.01), as expected (Fig. 5). Similar, maximal photosynthetic electron transport (Jmax) was also higher in Tsu-1 than Sq-8 or Kas-1 (δ13C = −28.8) (P = 0.002, P = 0.002).
Maximum carboxylation rate of rubisco (V cmax) and maximal photosynthetic electron transport (Jmax) obtained from photosynthetic carbon dioxide response curves in three accessions (Tsu-1, Sq-8, and Kas-1) which differed in A. Each bar represents the mean ± SE (n = 4) for each accession. Letters represent significant differences among accessions. Genotype F-ratio = 12.14 and P = 0.0078 for V cmax. Genotype F-ratio = 11.01 and P = 0.0098 for Jmax
The major biochemical limitations to photosynthesis, V cmax and Jmax, appeared optimized to accessions’ C i as indicated by δ13C. V cmax and Jmax were lower in low g s, high WUE accessions operating at lower C i. The higher ratio of V cmax to Jmax in Kas-1 compared to Sq-8 suggests a lack of limitation by Jmax under the low g s typical of Kas-1. Simultaneous changes in V cmax and Jmax are consistent with a limitation of photosynthesis by RuBP utilization and regeneration (Farquhar and Sharkey 1982). Likewise, proportional changes in components of photosynthetic apparatus and g s suggest acclimation of these processes are closely coupled (Cowan 1986).
Variation in structure
In experiment 3, we examined 39 natural accessions of Arabidopsis for variation in δ13C and LWC (Table 1). We found a significant negative correlation between δ13C and LWC among accessions (r 2 = 0.6, P < 0.0001). Spring accessions tended to have higher LWC and lower WUE, as indicated by δ13C, than winter accessions. Accession differences in LWC most likely result from the effect of mesophyll cell wall thickness on leaf density and not differences in water potential as plants in experiment 3 were not water stressed (Garnier and Laurent 1994; Evans et al. 1994). Leaf anatomical traits such as leaf and cell wall thickness, surface area of mesophyll cells exposed to internal air spaces, and the location of chloroplasts within those cells was initially shown to correlate with g m several decades ago (von Caemmerer and Evans 1991; Evans et al. 1994). In particular, mesophyll cell wall thickness was shown to negatively affect g m. Therefore, high LWC accessions should have thinner mesophyll cell walls resulting in high g m and more negative δ13C (Evans et al. 1994), which is consistent with our data. These ideas have been revisited recently and the importance of the cell wall properties (thickness and water content) and the coverage of air exposed surfaces of mesophyll cells by chloroplasts is receiving more attention (Evans et al. 2009; Tholen and Zhu 2011; Tosens et al. 2012). Direct measurement of leaf thickness and density may explain some of the variation in g m and δ13C among plants with similar LWC values (Fig. 6). Alternatively, variation in COO-porin content or activity could be responsible for the g m and δ13C variation in plants with LWC. Recent studies have found a significant role for chloroplast membrane CO2 transporting aquaporins (COO-porin) has been demonstrated and provides a clearly heritable mechanism for both rapid and sustained adjustment of g m (Flexas et al. 2006; Uehlein et al. 2008, 2012; Heckwolf et al. 2011). We have found strong correlations between LWC, A, and g s, so focusing on plants with similar LWC should limit the influence of those factors on variation in δ13C and increase the relative influence of g m from cell wall properties or COO-porin content or activity on δ13C variation.
The ABI4 transcription factor causes changes in leaf anatomy and mesophyll conductance
To further test for a causal effect of leaf anatomy on gas exchange (experiment 4 in Table 1), we used abi4, a mutant of locus AT2G40220, which is an AP2/ERF transcription factor (TF). ABI4 is closely related to the DREB2 TFs and the mutant was initially described as ABA insensitive based on a germination screen (Finkelstein 1994). Subsequent work has shown that the transcript is expressed in seedlings (Soderman et al. 2000) and fully developed rosette leaves (Finkelstein et al. 1998). In addition to its key role in ABA signaling, further characterization of this transcription factor has proposed a large and diverse set of functions including sugar signaling and response (Husijer et al. 2000; Bossi et al. 2009), and root development (Signora et al. 2001; Shkolnik-Inbar and Bar-Zvi 2011). There are hundreds of loci whose expression is altered in the ABI4 mutant (Kerchev et al. 2011). Given that it is a transcription factor, this is not surprising, but does illustrate the challenge of functional annotation of such pleiotropic loci.
abi4 had higher SLA and LWC than wildtype, revealing a novel effect of this TF on leaf anatomy. In addition, abi4 had increased g m and more negative δ13C, consistent with the idea that SLA causes variation in δ13C via effects on g m (Fig. 7). The correlation of SLA, A, and g s with LWC helps to explain why LWC is strongly correlated with leaf gas exchange, i.e., LWC appears to be an inverse proxy for cell wall thickness. When taken together, our data show that Arabidopsis leaves trade-off high WUE for low A, by trading off leaf anatomy based diffusional CO2 limitation with water loss through stomata. Essentially, plants with the highest A achieve this via the combination of high g s and thin leaves (high SLA). High g s keeps C i high and the thin leaves have cells with thin walls. Thin walls increase g m and keeps CO2 concentration at the sites of carboxylation (C c) high (Evans et al. 1994). Conversely, when photosynthesis is directly limited by the combination of cool winter temperatures and high light through effects on electron transport, then low g s would be selected for to improve WUE. We hypothesize that thicker leaves would provide more internal shading and more efficient light use, further decreasing g m and C c explaining the winter annual phenotype.
Although, a few of the AP2/ERF transcription factors in Arabidopsis have been the subject of detailed study, there are 122 of these loci in Arabidopsis (Nakano et al. 2006) and much remains unknown about their function. Recent studies have revealed increasingly complex roles for members of this transcription factor family. For example, a recent study identified eight AP2/ERFs induced by photorespiration (Foyer et al. 2012). This, combined with the known roles of ABI4 in sugar signaling to photosynthesis including repression of RBCS (Van Oosten et al. 1997; Teng et al. 2008), and our results showing effects on leaf density and g m, are expanding this picture.
Conclusions
Detailed measurements on a diverse set of accessions detail the traits underlying natural variation in intrinsic WUE and carbon isotope composition. Previous studies have shown that spring accessions have lower intrinsic WUE than accessions with winter life histories. Proportional changes in A, g s, V cmax, and Jmax suggest acclimation of these processes are closely coupled. We also show strong covariation between LWC and δ13C, where spring annuals tend to have higher LWC and lower intrinsic WUE. We hypothesize that this is due to an effect through g m, and test this hypothesis using the abi4 mutant. The abi4 mutant shows increased SLA and reduced g m compared to the wildtype, consistent with the pattern of covariance found in the natural accessions.
Previous separate studies in Arabidopsis have addressed variation in δ13C, plant–water relations, leaf anatomy, and photosynthetic capacity and limitations, including g m. Here, we use a whole canopy approach to examine variation and covariation in all of these components. As predicted by optimality, these traits are not independent, but instead covary as would be expected if selection and photosynthetic acclimation favors states of colimitation. In addition, we show that perturbation of a single transcription factor leads to this trait covariance. This emphasizes the need for whole plant approaches and high dimensional phenotyping to accurately annotate the gene function.
References
Araus JL, Slafer GA, Reynolds MP, Royo C (2002) Plant breeding and drought in C3 cereals: what should we breed for? Ann Bot 89:925–940
Barbour MM, McDowell NG, Tcherkez G, Bickford CP, Hanson DT (2007) A new measurement technique reveals rapid post-illumination changes in the carbon isotope composition of leaf-respired CO2. Plant Cell Environ 30:469–482
Barbour MM, Warren CR, Farquhar GD, Forrester G, Brown H (2010) Variability in mesophyll conductance between barley genotypes, and effects on transpiration efficiency and carbon isotope discrimination. Plant Cell Environ 33:1176–1185
Bloom AJ, Chapin FS III, Mooney HA (1985) Resource limitation in plants: an economic analogy. Annu Rev Ecol Syst 16:363–392
Bossi F, Cordoba E, Dupre P, Mendoza MS, Roman CS, Leon P (2009) The Arabidopsis ABA-INSENSITIVE (ABI) 4 factor acts as a central transcription activator of the expression of its own gene, and for the induction of ABI5 and SBE2.2 genes, during sugar signaling. Plant J 59:359–374
Bouchabke O, Chang F, Simon M, Voisin R, Pelletier G, Durand-Tardif M (2008) Natural variation in Arabidopsis thaliana as a tool for highlighting differential drought responses. PLoS One 3:e1705
Buckley TN, Mott KA (2002) Dynamics of stomatal water relations during the humidity response: implications of two hypothetical mechanisms. Plant Cell Environ 25:407–419
Caruso CM, Maherali H, Mikulyuk A, Carlson K, Jackson RB (2005) Genetic variance and covariance for physiological traits in Lobelia: are there constraints on adaptive evolution? Evolution 59:826–837
Christman MA, Richards JH, McKay JK, Stahl EA, Juenger TE, Donovan LA (2008) Genetic variation in Arabidopsis thaliana for night-time leaf conductance. Plant Cell Environ 31:1170–1178
Comstock JP (2002) Hydraulic and chemical signalling in the control of stomatal conductance and transpiration. J Exp Bot 53:195–200
Condon AG, Richards RA, Farquhar GD (1987) Carbon isotope discrimination is positively correlated with grain-yield and dry-matter production in field-grown wheat. Crop Sci 27:996–1001
Condon AG, Richards RA, Rebetzke GJ, Farquhar GD (2002) Improving intrinsic water-use efficiency and crop yield. Crop Sci 42:122–131
Cowan IR (1986) Economics of carbon fixation in higher plants. In: Givnish TJ (ed) On the economy of plant form and function. Cambridge U Press, Cambridge, pp 133–170
Dawson TE, Mambelli S, Plamboeck AH, Templer PH, Tu KP (2002) Stable isotopes in plant ecology. Annu Rev Ecol Syst 33:507–559
Des Marais DL, McKay JK, Richards JH, Sen S, Wayne T, Juenger TE (2012) Physiological genomics of response to soil drying in diverse Arabidopsis accessions. Plant Cell 24:893–914
Donovan LA, Ehleringer JH (1994) Potential for selection on plants for water-use efficiency as estimated by carbon isotope discrimination. Amer J Bot 81:927–935
Donovan LA, Maherali H, Caruso CM, Huber H, de Kroon H (2011) The evolution of the worldwide leaf economics spectrum. Trends Ecol Evo 26:88–95
Evans JR, Sharkey TD, Berry JA, Farquhar GD (1986) Carbon isotope discrimination measured concurrently with gas exchange to investigate CO2 diffusion in leaves of higher plants. Aust J Plant Physiol 13:281–292
Evans JR, von Caemmerer S, Setchell BA, Hudson GS (1994) The relationship between CO2 transfer conductance and leaf anatomy in transgenic tobacco with a reduced content of rubisco. Aust J Plant Physiol 21:475–495
Evans JR, Kaldenhoff R, Genty B, Terashima I (2009) Resistances along the CO2 diffusion pathway inside leaves. J Exp Bot 60:2235–2248
Farquhar GD, Sharkey TD (1982) Stomatal conductance and photosynthesis. Annu Rev Plant Physiol 33:317–345
Farquhar GD, von Caemmerer S, Berry JA (1980) A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149:78–90
Farquhar GD, O’Leary MH, Berry JA (1982) On the relationship between carbon isotopic discrimination and intercellular carbon dioxide concentration in leaves. Aust J Plant Physiol 9:121–137
Farquhar GD, Ehleringer JR, Hubrick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Physiol 40:503–537
Finkelstein RR (1994) Mutations at 2 new Arabidopsis ABA response loci are similar to the abi3 mutations. Plant J 5:765–771
Finkelstein RR, Wang ML, Lynch TJ, Rao S, Goodman HM (1998) The Arabidopsis abscisic acid response locus ABI4 encodes an APETALA2 domain protein. Plant Cell 10:1043–1054
Fisher RA, Rees D, Sayre KD, Larque Saavedra A (1998) Wheat yield progress associated with higher stomatal conductance and photosynthetic rate, and cooler canopies. Crop Sci 38:1467–1475
Flexas J, Bota J, Galmes J, Medrano H, Ribas-Carbo M (2006) Keeping a positive carbon balance under adverse conditions: responses of photosynthesis and respiration to water stress. Physiol Plant 127:343–352
Flexas J, Diaz-Espejo A, Galmes J, Kaldenhoff R, Medrano H, Ribas-Carbo M (2007) Rapid variations of mesophyll conductance in response to changes in CO2 concentration around leaves. Plant Cell Environ 30:1284–1298
Flexas J, Ribas-Carbo M, Diaz-Espejo A, Galmes J, Medrano H (2008) Mesophyll conductance to CO2: current knowledge and future prospects. Plant Cell Environ 31:602–621
Foyer CH, Neukermans J, Queval G, Noctor G, Harbinson J (2012) Photosynthetic control of electron transport and the regulation of gene expression. J Exp Bot 63:1637–1661
Gallé A, Lautner S, Flexas J, Ribas-Carbo M, Hanson D, Roesgen J, Fromm J (2012) Photosynthetic responses of soybean (Glycine max L.) to heat-induced electrical signaling are predominantly governed by modifications of mesophyll conductance for CO2. Plant Cell Environ 36:542–552
Garnier E, Laurent G (1994) Leaf anatomy, specific mass and water-content in congeneric annual and perennial grass species. New Phytol 128:725–736
Geber MA, Dawson TE (1997) Genetic variation in stomatal and biochemical limitations to photosynthesis in the annual plant, Polygonum arenastrum. Oecologia 109:535–546
Geber MA, Griffin LR (2003) Inheritance and natural selection on functional traits. Int J Plant Sci 164:S21–S42
Hall NM, Griffiths H, Corlett JA, Jones HG, Lynn J, King GJ (2005) Relationships between water-use traits and photosynthesis in Brassica oleracea resolved by quantitative genetic analysis. Plant Breeding 124:557–564
Heckwolf M, Pater D, Hanson DT, Kaldenhoff R (2011) The Arabidopsis thaliana aquaporin AtPIP1; 2 is a physiologically relevant CO2 transport facilitator. Plant J 67:795–804
Heschel MS, Donohue K, Hausmann NJ, Schmitt J (2002) Population differentiation and natural selection for water-use efficiency in Impatiens capensis (Balsaminaceae). Int J Plant Sci 163:907–912
Husijer C, Kortsee A, Pego J, Weisbeek P, Wisman E, Smeekens S (2000) The Arabidopsis SUCROSE UNCOUPLED-6 gene is identical to ABSCISIC ACID INSENSITIVE-4: involvement of abscisic acid in sugar responses. Plant J 23:577–585
Juenger TE, McKay JK, Hausmann N, Keurentjes JJB, Sen S, Stowe KA, Dawson TE, Simms EL, Richards JH (2005) Identification and characterization of QTL underlying whole-plant physiology in Arabidopsis thaliana: δ13C, stomatal conductance and transpiration efficiency. Plant Cell Environ 28:697–708
Juenger TE, Sen S, Bray E, Stahl E, Wayne T, McKay J, Richards JH (2010) Exploring genetic and expression differences between physiologically extreme ecotypes: comparative genomic hybridization and gene expression studies of Kas-1 and Tsu-1 accessions of Arabidopsis thaliana. Plant Cell Environ 33:1268–1284
Katul G, Manzoni S, Palmroth S, Oren R (2010) A stomatal optimization theory to describe the effects of atmospheric CO2 on leaf photosynthesis and transpiration. Ann Bot 105:431–442
Kerchev PI, Pellny TK, Vivancos PD, Kiddle G, Hedden P, Driscoll S, Vanacker H, Verrier P, Hancock RD, Foyer CH (2011) The transcription factor ABI4 is required for the ascorbic acid-dependent regulation of growth and regulation of jasmonate-dependent defense signalling pathways in Arabidopsis. Plant Cell 23:3319–3334
Kogami H, Hanba YT, Kibe T, Terashima I, Masuzawa T (2001) CO2 transfer conductance, leaf structure and carbon isotope composition of Polygonum cuspidatum leaves from low and high altitudes. Plant, Cell Environ 24:529–538
Lasky JR, Des Marais DL, McKay JK, Richards JH, Juenger TE, Keitt TH (2012) The role of geography, climate and phenology in explaining characterizing genomic variation of Arabidopsis thaliana: the roles of geography and climate. Mol Ecol 12:5512–5529
Littell RC, Milliken GA, Stroup WW, Wolfinger RD (1996) SAS system for mixed models. SAS Institute Inc, Cary, p 633
Masle J, Gilmore SR, Farquhar GD (2005) The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature 436:866–870
McKay JK, Richards JH, Mitchell-Olds T (2003) Genetics of drought adaptation in Arabidopsis thaliana: I. Pleiotropy contributes to genetic correlations among ecological traits. Mol Ecol 12:1137–1151
McKay JK, Richards JH, Nemali KS, Sen S, Mitchell-Olds T, Boles S, Stahl EA, Wayne T, Juenger TE (2008) Genetics of drought adaptation in Arabidopsis thaliana II. QTL analysis of a new mapping population, Kas-1 × Tsu-1. Evolution 62:3014–3026
Monda K, Negi J, Iio A, Kusumi K, Kojima M, Hashimoto M, Sakakibara H, Iba K (2011) Environmental regulation of stomatal response in the Arabidopsis Cvi-0 ecotype. Planta 234:555–563
Nakano T, Suzuki K, Fujimura T, Shinshi H (2006) Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol 140:411–432
Nordborg M, Hu TT, Ishino Y, Jhaveri J, Toomajian C, Zheng H, Bakker E, Calabrese P, Gladstone J, Goyal R, Jakobsson M, Kim S, Morozov Y, Padhukasahasram B, Plagnol V, Rosenberg NA, Shah C, Wall JD, Wang J, Zhao K, Kalbfleisch T, Schulz V, Kreitman M, Bergelson J (2005) The pattern of polymorphism in Arabidopsis thaliana. PLoS Biol 3:e196
Parkhurst DF, Mott KA (1990) Intercellular diffusion limits to CO2 uptake in leaves. Plant Physiol 94:1024–1032
Passioura JB (1977) Grain yield, harvest index, and water use of wheat. J Aust I Agr Sci 43:117–120
Pons TL (2012) Interaction of temperature and irradiance effects on photosynthetic acclimation in two accessions of Arabidopsis thaliana. Photosynth Res 113:207–219
Rebetzke GJ, Condon AG, Richards RA, Farquhar GD (2002) Selection for reduced carbon isotope discrimination increases aerial biomass and grain yield of rainfed bread wheat. Crop Sci 42:739–745
Reich PB, Walters MB, Ellsworth DS (1997) From tropics to tundra: global convergence in plant functioning. Proc Natl Acad Sci USA 94:13730–13734
SAS Institute (1999) SAS/STAT user’s guide. SAS Institute Inc., Cary
Schulze E-D (1986) Carbon dioxide and water exchange in response to drought in the atmosphere and in the soil. Annu Rev Plant Physiol 37:247–274
Seibt U, Rajabi A, Griffiths H, Berry JA (2008) Carbon isotopes and water use efficiency: sense and sensitivity. Oecologia 155:441–454
Sharkey TD, Bernacchi CJ, Farquhar GD, Singsaas EL (2007) Fitting photosynthetic carbon dioxide response curves for C3 leaves. Plant Cell Environ 30:1035–1040
Shkolnik-Inbar D, Bar-Zvi D (2011) Expression of ABSCISIC ACID INSENSITIVE 4 (ABI4) in developing Arabidopsis seedlings. Plant Signal Behav 6:694–696
Signora L, De Smet I, Foyer CH, Zhang H (2001) ABA plays a central role in mediating the regulatory effects of nitrate on root branching in Arabidopsis. Plant J 28:655–662
Söderman EM, Brocard IM, Lynch TJ, Finkelstein RR (2000) Regulation and function of the Arabidopsis ABA-insensitive4 gene in seed and abscisic acid response signaling networks. Plant Physiol 124:1752–1765
Syvertsen JP, Lloyd J, Mcconchie C, Kriedemann PE, Farquhar GD (1995) On the relationship between leaf anatomy and CO2 diffusion through the mesophyll of hypostomatous leaves. Plant, Cell Environ 18:149–157
Teng S, Rognoni S, Bentsink L, Smeekens S (2008) The Arabidopsis GSQ5/DOG1 Cvi allele is induced by the ABA-mediated sugar signalling pathway, and enhances sugar sensitivity by stimulating ABI4 expression. Plant J 55:372–381
Terashima I, Hanba YT, Tholen D, Niinemets U (2011) Leaf functional anatomy in relation to photosynthesis. Plant Physiol 155:108–116
Tholen D, Zhu XG (2011) The mechanistic basis of internal conductance: a theoretical analysis of mesophyll cell photosynthesis and CO2 diffusion. Plant Physiol 156:90–105
Tosens T, Niinemets U, Vislap V, Eichelmann H, Diez PC (2012) Developmental changes in mesophyll diffusion conductance and photosynthetic capacity under different light and water availabilities in Populus tremula: how structure constrains function. Plant Cell Environ 35:839–856
Uehlein N, Otto B, Hanson DT, Fischer M, McDowell N, Kaldenhoff R (2008) Function of Nicotiana tabacum aquaporins as chloroplast gas pores challenges the concept of membrane CO2 permeability. Plant Cell 20:648–657
Uehlein N, Otto B, Eilingsfeld A, Itel F, Meier W, Kaldenhoff R (2012) Gas-tight triblock-copolymer membranes are converted to CO2 permeable by insertion of plant aquaporins. Sci Rep 2:538
Van Oosten JJM, Gerbaud A, Huijser C, Dijkwel PP, Chua NH, Smeekens SCM (1997) An Arabidopsis mutant showing reduced feedback inhibition of photosynthesis. Plant J 12:1011–1020
Von Caemmerer S, Evans JR (1991) Determination of the average partial-pressure of CO2 in chloroplasts from leaves of several C3 plants. Aust J Plant Physiol 18:287–305
Warren CR (2008) Does growth temperature affect the temperature responses of photosynthesis and internal conductance to CO2? A test with Eucalyptus regnans. Tree Physiol 28:11–19
Warren CR, Adams MA (2006) Internal conductance does not scale with photosynthetic capacity: implications for carbon isotope discrimination and the economics of water and nitrogen use in photosynthesis. Plant Cell Environ 29:192–201
Warren CR, Dreyer E, Adams MA (2003) Photosynthesis-Rubisco relationships in foliage of Pinus sylvestris in response to nitrogen supply and the proposed role of Rubisco and amino acids as nitrogen stores. Trees 17:359–366
Yamori W, Noguchi K, Terashima I (2006) Mechanisms of temperature acclimation of photosynthesis. Plant Cell Physiol 47:S4
Acknowledgments
We thank P Rispin for help in completing the TE experiment. This research is supported by NSF grants DEB-1022196 and DEB-0618302 to JKM, DEB-0618347 to TEJ, IOS-0719118 to DTH, DEB-0618294 to JHR, USDA NIFA 2007-35100-18379 to TEJ, and NIH-NCRR P20RR18754. Support from the California and Colorado Agricultural Experiment Stations is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Easlon, H.M., Nemali, K.S., Richards, J.H. et al. The physiological basis for genetic variation in water use efficiency and carbon isotope composition in Arabidopsis thaliana . Photosynth Res 119, 119–129 (2014). https://doi.org/10.1007/s11120-013-9891-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9891-5