Abstract
Urban development is thought to negatively affect most native mammals. Here we assess whether adjacent suburbia, fire regime and vegetation influence the activity of mammal species in a reserve in Hobart, Tasmania, Australia. We used multiple regression to analyse the relationship between distance from houses, fire history, vegetation structure and floristics, and mammal activity, derived from camera visits and signs. Animal diggings and visits by long-nosed potoroos, southern brown bandicoots and cats were greater close to houses, while visits by red-necked wallabies, rufous-bellied pademelons, brushtail possums and short-beaked echidnas were not. The structure of the vegetation, particularly related to shelter, was important in many models. Many models also included floristic ordination axes that reflected drainage, fire regime and distance from high-density housing. Positive relationships between the introduced predatory cat and some of its potential native prey animals may reflect the influences of close proximity to suburbia, such as elevated domestic mesopredator populations, and elevated resource availability in domestic gardens. Our results suggest that urban areas can have a valuable role in nature conservation, despite, or because of, their effects on remnant native ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Increasing urbanisation is one of the leading causes of global biodiversity loss and species extinction (Czech et al. 2000; Marzluff 2001; McKinney 2006). It can impact species through a range of processes including: loss, degradation and fragmentation of natural habitats; increases in exotic species; noise and light pollution; and altered hydrology. Impacts vary depending on the intensity of urbanisation (McKinney 2002, 2008), as in Melbourne, Victoria, Australia where suburbs (built up residential areas outside the Central Business District) with treed gardens have very different avifaunas to those less wooded (Conole and Kirkpatrick 2011). Urban areas, particularly in their suburbs, often contain remnant patches of seminatural habitats, parklands, sports fields, and private residential gardens (green spaces), all of which provide habitats for a range of species and may be important for species conservation. For example, in Australia, 39 threatened species (37 plants and 2 animals) are restricted to urban environments (Soanes and Lentini 2019; Soanes et al. 2020). Understanding the positive impacts of urbanisation on biodiversity may provide opportunities to protect species and habitats in novel environments and to engage people with nature (Soanes and Lentini 2019; Busschots et al. 2021; Spotswood et al. 2021). For example, the threatened eastern barred bandicoot (Perameles gunnii) is more common in exurban (residential areas outside suburbs) environments than in equivalent native vegetation (Daniels and Kirkpatrick 2012). The species has become a focus for urban habitat restoration programs (NRM 2021).
In a review of 787 papers, Shwartz et al. (2014) found that the importance of urban areas for general conservation is not convincingly supported by empirical research, with only a few studies showing these areas can support viable populations of rare or threatened species. Some studies (summarised in McKinney 2002) have reported higher species richness in some suburban areas than in more natural areas or more intensively modified parts of the city, a pattern consistent with the intermediate disturbance hypothesis. For example, Brady et al. (2011) found that native mammal species richness peaked at moderate levels of development intensity. Suburbs in a matrix of largely natural habitat may provide environmental heterogeneity, which is further enhanced by productive suburban gardens (Falk 1976; McKinney 2002). However, other studies have reported reduced species diversity in suburban and exurban areas (Marzluff 2001; Daniels and Kirkpatrick 2012). Globally most urban conservation studies have focussed on plants, invertebrate and birds (Shwartz et al. 2014). More studies on the impacts of urbanisation and urban remnant management on mammals are required. We know that urbanisation can cause a decrease in mammal species richness and abundance (Tait et al. 2005; van der Ree and McCarthy 2005; Ordenana et al. 2010; Daniels and Kirkpatrick 2012; Luck et al. 2013) as well as have adverse physiological consequences (Brearley et al. 2012). While urbanisation generally has adverse impacts on mammal populations, some species are more abundant in exurban environments than in similar native habitats (Ordenana et al. 2010; Daniels and Kirkpatrick 2012; Maclagan et al. 2018).
With urbanisation increasing globally there is a need to better understand factors that could help maintain and promote retention of native species or mitigate against negative factors. Habitat structure, vegetation cover, vegetation extent, urban intensity/housing density, native vegetation proximity/urban proximity, functional connectivity of habitat fragments, road density and access by domestic dogs have all been found to be important for determining presence and abundance/activity of mammal species in urban and peri-urban (on the margin of built up areas) environments (Baker and Harris 2007; FitzGibbon et al. 2007; Garden et al. 2007; Ordenana et al. 2010; Luck et al. 2013; Caryl et al. 2016; Bryant et al. 2017; Van Helden et al. 2020; Zungu et al. 2020). Where large remnants of native vegetation occur adjacent to, or enclosed by, urban development, management of potential threats to biodiversity such as fire and introduced plants can be difficult if maintenance of native mammal biodiversity is a primary aim. Planned fires to protect nearby houses from fires lit by arsonists can deleteriously affect habitats for mammals if undertaken too frequently or infrequently or if not enough unburnt habitat is retained.
The present paper provides a Tasmanian case study of the effects of vegetation, fire history and proximity to houses on the activity of the more common mammals of a peri-urban reserve. Our work is of particular interest in that Tasmania is one of the few places where the suite of rticial-weight-range mammals, susceptible to extinction through European fox (Vulpes vulpes) predation, survive.
Methods
Study area
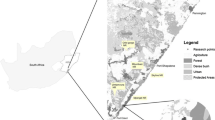
The study was conducted in a small conservation reserve (Peter Murrell State Reserve and Conservation Area; 277 ha; 43°00’ S, 147°18’ E) comprising eucalypt forest, eucalypt woodland (mainly black peppermint Eucalyptus amygdalina) and heathland that is isolated from other areas of native vegetation by development, including adjacent residential suburbs (Fig. 1). The study area is described in detail by Kirkpatrick (1999) and Driessen et al. (2010).
Experimental design
This study used a design previously established to investigate the response of mammals to planned burns, comprising four fire-management blocks (each 17–20 ha) (Driessen and Jarman 2010, 2014; Driessen et al. 2021). The transects were located to adequately cover the area of the fire-management blocks, with spacing taking into account the home ranges of the medium-sized and small-sized mammals that were the target of the fire study. Two transects, 100 m apart, were established in each block and each transect consisted of 10 stations (marked by a steel stake and used for vegetation and fauna surveys) each ~ 50 m apart (Fig. 1). At the time of our surveys in 2018, block AB had been subject to a moderate intensity (500–3000 kWm− 1) planned burn in 2010, and block IJ and the eastern 60% of block CD had been subject to a moderate intensity planned burn in December 2013. Most of the reserve (94%) was burnt by an intense fire in January 1988 (30 years prior to our study).
Data collection
Camera-trapping took place in seven-day sessions at four stations per transect; every second station from the east or south except for the last station (total 32 stations, see Fig. 1); one session in autumn and one in spring 2018. We used the same camera stations in each seven-day session. KeepGuard KG 680 V cameras were attached to small trees and angled down towards a PVC bait container (peanut butter, rolled oats and sesame oil) and pegged into the ground. Heights of cameras above ground (0.5-1 m) and distances to the bait stations (1.2-2 m) were as standardized as natural variations in tree-trunk shape and ground micro-topography allowed. By aiming the cameras towards the bait container we minimised possible variation in detection due to variation in camera height and distance between camera and bait station (McIntyre et al. 2020). Cameras, with a minimum triggering interval of 30 s and high-level sensitivity, took single-frame images, with date and time recorded on each. The cameras use infrared illumination at night and have a 50° field of view and a trigger time of 1 s. KeepGuard KG 680 V cameras have been shown to perform as well as or better than some other commonly used cameras (e.g., Reconyx H600) (Driessen et al. 2017) and no performance issues occurred with the cameras during their deployment for this study. All images were assessed for the presence of an animal and in most images the species was identified. Our metric for mammal activity was the number of visits by a species rather than the number of images (Jarman and Driessen 2019). We defined a visit as one or more images of a species with no interval between animal images greater than 5 min. Our choice of 5 min as the maximum allowable within-visit interval was based on our analyses of inter-image durations in large samples of timed images of long-nosed potoroos (Potorous tridactylus) (Jarman and Driessen 2019) and was appropriate for other species recorded in the study area (P. Jarman and M. Driessen, unpublished data).
In autumn 2018, all observed vascular plant taxa were recorded in 80 10-by-10 m plots centred on each of the 80 stations. The tape that defined the borders of each plot was used as an intercept line. The outline intercept cover was measured to the nearest cm for shrubs, graminoids (all monocotyledonous plants), ferns, herbs, bryophytes, lichen, bare ground (clear to the sky), rock and litter. A Bitterlich wedge count from the middle of the quadrat was undertaken by species to determine basal area. Slope (degrees), aspect (degrees) and topographic position (lower, upper, ridge and valley) were also recorded. The distance in metres to the nearest house and the nearest high-density housing (250–1000 houses/km2) was estimated using Google maps. Scats of macropods (red-necked wallaby (Notamacropus rufogriseus) and rufous-bellied pademelon (Thylogale billardierii)), and possums (brushtail (Trichosurus vulpecula) and ring-tailed (Pseudocheirus peregrinus)) were counted for the whole plot of all stations, as were all marsupial and monotreme diggings.
In autumn 2018, an index of horizontal vegetation density was recorded for all stations using a 100-cm-tall by 50-cm-wide cover board marked with 200 5-by-5-cm squares. The cover board was placed on the ground 5 m to the north, and then to the south, of each station and the numbers of squares obscured by vegetation between 0 and 50 cm and between 50 and 100 cm above the ground were recorded by an observer standing at the station. The average of the north and south scores provided an index for the station of vegetation density between 0 and 50 cm and between 50 and 100 cm.
Fire history data since 1988 was obtained from the Peter Murrell State Reserve and Conservation Area Fire Management Plan (PWS 2006) as well as a fire mapping database maintained by the Tasmanian Parks and Wildlife Service. From this information the times since last fire in 2018 and number of fires between 1988 and 2018 were determined for each station.
Variables used in the analyses are summarised in Table 1 together with minimum, maximum and mean values (± 95% confidence intervals) for the 80 stations.
Statistical analysis
All analyses were undertaken in Minitab18, except for ordination of the vegetation data.
Non-metric global multidimensional scaling ordination in DECODA (Minchin 1990) with the default options was used to reduce the plant species presence/absence data to four variables. The four-dimensional ordination solutions had a minimum stress of 13.4%. The first axis discriminated assemblages of dry heathy vegetation, characterised by such species as Bossiaea cinerea, Allocasuarina monilifera and Epacris impressa, from other types of assemblages (Table 2). The heathy extreme was on steeper slopes (Table 3). The second axis discriminated species assemblages characteristic of heaths on poorly-drained ground from the rest. The species of poorly-drained ground included Gymnoschoenus sphaerocephalus, Leptocarpus tenax and Restio monocephalus (Table 2). The scores on this axis were also predicted by slope, with the positive values associated with species of the poorly-drained ground associated with gentler slopes (Table 3). The scores on axis 3 were strongly correlated with graminoid cover, with high cover of Gahnia radula and Hypolaena fastigiata at the positive end of the axis. (Table 2). The scores on this axis were also correlated with the two distance from houses variables (Table 3). The scores on the fourth axis discriminated recently burned vegetation from less recently burned vegetation, with strong correlations with time since last fire and number of fires, as well as distance from high-density housing (Table 3).
Multiple regression analysis, guided by the ‘Best Subsets’ routine in Minitab, was undertaken with each of the taxa, and indicators of taxa, for which quantitative data were available as dependent variables (Table 1). The maximum number of predictor variables was determined by the sample size, with at least 10 samples per variable being used to avoid overfitting. The selected best model was that which had the highest adjusted r2 and significant (P < 0.05) slopes on each variable. Where possible, our analyses remove collinearities from predictive equations. However, the precision of measurement can affect the relative strength of explanatory variables, thereby influencing inclusion, so we have been careful to assess the logic of the variables included in our models.
Cat (Felis catus) and short-beaked echidna (Tachyglossus aculeatus) camera visits were too infrequent to treat as continuous variables, so were treated as presence/absence. One-way ANOVA was used to determine whether animal observations and some environmental variables varied by the time since the last fire, the presence/absence of cats and the presence/absence of echidnas. Pearson product moment correlation coefficient was used to determine the significance of linear relationships between continuous variables.
Results
Overview
A total of 1,795 visits to camera by mammals was recorded comprising 8 native species and 3 non-native species. The native species were rufous-bellied pademelon (707 visits), long-nosed potoroo (657), brushtail possum (218), red-necked wallaby (115), southern brown bandicoot (Isoodon obesulus) (82), short-beaked echidna (12), ringtail possum (3) and Tasmanian devil (Sarcophilus harrisii) (1). The non-native species were house mouse (Mus musculus) (72), cat (10) and dog (Canis familiaris) (2).
There were sufficient data to develop multiple regression models for the long-nosed potoroo, southern brown bandicoot, red-necked wallaby, rufous-bellied pademelon, and brushtail possum visits, macropod scat, possum scat and total diggings density. The best models had R2 values varying from 6.1% for possum scat density to 73.5% for southern brown bandicoot visits (Table 4). The data for cats and short-beaked echidnas could not be modelled beyond one predictor because of the low numbers of visits. There were no significant relationships for the house mouse.
Distance to houses
The distance to high-density housing had negative coefficients in the models for southern brown bandicoot visits, long-nosed potoroo visits and total diggings density (Table 4). Cat visits occurred closer to high-density housing (mean distance to high-density housing for: cat present = 380 m, cat absent = 765 m; F = 17.811,30, P < 0.001).
Fire regime
The number of years since the last fire had positive coefficients in the models for southern brown bandicoot visits and total diggings density, and negative coefficients in the models for red-necked wallaby visits and macropod scat density (Table 4). There was a positive coefficient for number of fires in the southern brown bandicoot visit model (Table 4).
The role of vegetation and topography
The total basal area of trees had a positive coefficient in the model for possum scat density (Table 4). The basal area of the most common tree, Eucalyptus amygdalina, had positive coefficients in the models for the southern brown bandicoot visits and total diggings density and a negative coefficient in the model for macropod scat density (Table 4). Vegetation density at 0–50 cm had a negative coefficient in the total diggings density model, while vegetation density at 50–100 cm had positive coefficients in the models for the red-necked wallaby and rufous-bellied pademelon visits. Fern cover had negative coefficients in the red-necked wallaby and brushtail possum visits models, graminoid cover had a positive coefficient in the macropod scat density model and shrub cover had a positive coefficient in the long-nosed potoroo visits model. The scores on axis 2 of the floristic ordination had a negative coefficient in the long-nosed potoroo visits model, indicating an association with plant communities of well-drained ground (Table 3). The scores on axis 4 of the ordination had negative coefficients in the models for brushtail possum visits and total diggings density (Table 4), indicating their preference for communities of recently burned ground (Table 3). The model for total diggings also included a positive coefficient for slope (Table 4).
The places that cats visited had lower graminoid cover than elsewhere (27% versus 57%, F = 4.691,30, P = 0.038), and lower scores on MDS4.3 (F = 4.731,30, P = 0.038), which reinforced the other relationship with a vegetation attribute in that axis three was heavily positively loaded on graminoid cover (Table 3).
The strongest relationship for the presence/absence of short-beaked echidna visits was with the score on axis 2 of the ordination (F = 8.921,30, P = 0.008), which indicated that echidnas preferentially visited better-drained ground on steeper slopes (mean = 4o versus 2o) (F = 4.441,30, P = 0.044). Shrub cover was less where echidnas visited than elsewhere (mean = 10 versus 29%, F = 5.311,30, P = 0.028).
Relationships between dependent variables
Brushtail possum and red-necked wallaby visits were positively correlated (r = 0.352, d.f. = 30, p = 0.048). Total diggings density was positively correlated with both southern brown bandicoot visits (r = 0.496, d.f. = 30, P = 0.004) and short-beaked echidna visits (r = 0.472, d.f. = 30, P = 0.006). The presence of cats was positively associated with visits of long-nosed potoroos (F = 5.31,30, P = 0.028) and southern brown bandicoots (F = 29.31,30, P < 0.001), but negatively associated with macropod scat density (F = 4.41,30, P = 0.045). Both southern brown bandicoots and cats decrease rapidly in visits with distance from high-density housing (Fig. 2).
Relationship between southern brown bandicoot visits and distance to the nearest high-density housing. Sites with both southern brown bandicoot and cat visits are marked with a triangle. The quadratic regression fit (SBB visits = 15.88–0.03389 Distance to nearest high-density housing + 0.000018 Distance to nearest high-density housing2) and 95% confidence intervals are shown. R2(adj) = 33%, F(2, 29) = 8.78, P = 0.001. All sites visited by cats were also visited by southern brown bandicoots
Discussion
Within a remnant, native vegetation reserve, we investigated the relationship between adjacent suburban environment and the activity of native and introduced mammals taking into account recent fire regimes and habitat variation.
Suburban influences
The human threat hypothesis suggests that population performance declines with increasing human dominance (Lawson et al. 2008). Thus, we might have expected decreased mammal activity with increased proximity to suburbia. However, the assumption that human-modified or “novel” habitats are poorer in quality to remnant habitats has been questioned (Maclagan et al. 2018; Soanes and Lentini 2019), and for some species abundance is greater in novel habitats than in remnant habitats (Daniels and Kirkpatrick 2012; Maclagan et al. 2018). We found no evidence of decreased mammal activity with increasing proximity to houses, but we did find that the activity of the southern brown bandicoot, the long-nosed potoroo and the cat increased closer to high-density housing. For the southern brown bandicoot this is consistent with other studies on this species (Daniels and Kirkpatrick 2012; Bryant et al. 2017; Maclagan et al. 2018), whereas the relationships between long-nosed potoroos and urban environments have not been previously studied. Not surprisingly, cats were associated strongly with high-density housing; the surprise was their positive associations with long-nosed potoroos and southern brown bandicoots. Both native mammals were more widespread than the cat in the study area, but the numbers of visits per site were higher where cats occurred, which was higher close to high-density housing than otherwise. Given that the direct effects of the cat on such mammals is almost certain to be negative, through the disease toxoplasmosis and direct predation, alternative explanations for the association between these species are: (1) that the habitat for these mammals in the reserve is better close to high-density housing than elsewhere and the cats are domestic, fed by the householders; (2) that the habitat is better near the high-density housing and the cats are attracted by the high density of prey; (3) that the small mammals have elevated populations close to high-density housing because they utilise domestic gardens as well as the ecosystems in the reserve, as do the domestic cats. The first two hypotheses are not consistent with the strong effect of proximity to high-density housing in the same multiple regression models as other environmental variables. This variable, thus, has an effect independent of the other variables included in the models, and is more explanatory than them. The third hypothesis is consistent with observations of bandicoots occurring in exurban gardens (Daniels and Kirkpatrick 2012) and with the high productivity associated with the elevated fertility and moisture availability of suburban gardens (Falk 1976; Bidwell et al. 2006; Bell et al. 2011).
The resources created by gardeners may have a fecundity and survival effect that outweighs mortality from high numbers of domesticated mesopredators. Gardeners also provide a complex environment within which predation may be less effective than in some native vegetation. Thus, high mesopredator abundances and high mammal abundances are likely to be independently determined by the actions of householders in maintaining domestic animals and creating and maintaining gardens rich in food resources for native animals. The models suggest that elevated populations of mammals may also partially depend on dense sheltering growth in the native vegetation near the gardens, indicating the desirability, for conservation of native mammals, of a low fire frequency (15–25-year intervals). Mosaic burning is desirable as several native species appear to benefit from recent burning.
Fire regime influences
Fire regimes can be an important influence on mammal community assemblages in many ecosystems around the world (e.g., Whelan et al. 2002; Fontaine and Kennedy 2012). For protected areas adjacent to suburban areas, fire management is a critical issue for land managers because of the risk of bushfire to human life and property and this can sometimes be at odds with management for conservation (Driscoll et al. 2016; Ryan et al. 2020). Within our study area, fire is primarily used as a tool to manage risk to human life and property and therefore has potential to impact on the persistence of mammal species that occur therein. Time since last fire featured in the models associated with southern brown bandicoot and red-necked wallaby activity (visits, diggings and scats). The negative effect of recent fire on southern brown bandicoot visits and total diggings density is consistent with that found for diggings of mainland southern brown bandicoots (Claridge and Barry 2000) and may reflect a lack of shelter and food resources. However, other studies have found limited effect on their activity following fire (Shan et al. 2006; Hope 2012; Driessen et al. 2021). The presence of cats at our study site may have played a role in our study as Arthur et al. (2012) suggested that cat predation may retard southern brown bandicoot recovery after fire. The positive association of southern brown bandicoots with areas of the reserve that had been burnt several times suggests that burning may be a positive influence for this species over the longer term; but this requires further investigation. In contrast to southern brown bandicoots, red-necked wallaby activity (visits and macropod scats) was greater in recently burnt areas of the reserve, consistent with two previous studies (Southwell and Jarman 1987; Driessen et al. 2021), and most likely reflecting a tendency of these macropods to feed on fresh growth in recently burnt areas (Southwell and Jarman 1987; Kirkpatrick et al. 2011, 2016; Styger et al. 2011). However, Dixon et al. (2019) found that red-necked wallabies occurred more frequently on long-unburnt than more recently burnt subalpine forests and woodlands.
While fire regime variables were in the models for total diggings density, macropod scat density, red-necked wallaby visits and southern brown bandicoot visits, the effects of fire regime on vegetation structure is also likely to be important for those species with shrub cover or vegetation density in their models (Monamy and Fox 2000). The pademelon and potoroo fit this description. The opposite responses to time since fire shown by red-necked wallaby and brushtail possums is striking. Such differences between species in response to fire regime and vegetation have been widely observed in non-urban areas (Catling et al. 2001; Lindenmayer et al. 2008; Arthur et al. 2012).
Brushtail possum visits were positively associated with recent fire (axis 4), which is consistent with an increase in activity in forest and woodlands following cool burns (Driessen et al. 1991, 2021) and wildfire (Lindenmayer et al. 2008) but not with abundance after wildfire in the study by Catling et al. (2001).
In peri-urban areas fire management to reduce fuels is a priority for fire managers and is complex and challenging. Bioturbation by wildlife as a result of foraging activities can assist with reducing fuel loads (Ryan et al. 2020). Management practices should facilitate maintenance of these populations and protection of life and property. Scale of fire is also important. Here, we investigated low-moderate severity fires, which aim to mitigate against larger-scale and or intense fires to reduce impacts on adjacent human life and property and to avoid adverse biodiversity outcomes. For example, Ramalho et al. (2018) used population viability analysis models based on field collected data to demonstrate that isolated metapopulations of the southern brown bandicoot were predicted to be highly sensitive to fire, with fires that burned large areas having greater negative impacts on population abundance than small fires. Fire management that limits the size of unplanned fires and promotes a mosaic of vegetation within and between remnant habitats will help protect human life and property as well as biodiversity. However, appropriate fire mosaics in peri-urban environments and the appropriate means for achieving them require further investigation.
Vegetation influences
In addition to proximity to houses and fire regime, variables associated with vegetation were also important in determining variation in mammal species activity within our habitat remnant. The positive effect of Eucalyptus amygdalina basal area in the model for the southern brown bandicoot visits is consistent with the year-round importance of fungi in their diet (Quin 1985; Mallick et al. 1998) and the importance of trees in supporting soil fungi (Johnson 1994; McMullan-Fisher et al. 2010). However, no such relationship was recorded for fungivorous long-nosed potoroos, which occurred widely throughout the reserve. The association of the long-nosed potoroo with high shrub cover and good soil drainage in its best model is consistent with previous descriptions of its habitat (Seebeck et al. 1989; Bennett 1993; Norton et al. 2010), although, at the local scale, Bennett (1993) also found that the long-nosed potoroo was not strongly correlated with any structural feature of the vegetation. It was not surprising that possum scat density increased as total basal area of trees increased, given the importance of trees for both ringtail and brushtail possums. Brushtail possums feed on the ground to some extent (Kerle 1984; MacLennan 1984), explaining the negative association with fern cover in their best model and their positive association with macropod scat density. The model for macropod scat density may reflect the importance of graminoids as a food source for macropods in the area and a preference for open vegetation for feeding (McArthur et al. 2000), including recently burned areas as previously mentioned. However, the models for red-necked wallaby visits, as well as rufous-bellied pademelon visits, included a positive influence of vegetation density 50–100 cm above the ground, seemingly contradicting the preference for openness implied by the macropod scat density model. However, this relationship reflects the well-known tendency of both wallabies and pademelons to shelter in dense vegetation during daylight hours (Le Mar and McArthur 2005; While and McArthur 2005). The avoidance of dense fern cover indicated by the red-necked wallaby visit model could reflect the association of Pteridium esculentum with low graminoid cover.
Caveats
While the reserve supports many mammal species, some of which appear to benefit from the adjacent suburb, there are several species that may already have been disadvantaged by the encroachment of suburbia, its associated disturbances and the increasing isolation of the reserve. Tasmanian devils, eastern quolls (Dasyurus viverrinus), eastern bettongs (Bettongia gaimardi) and the platypus (Ornithorhynchus anatinus) have been recorded from the area in the past but are uncommon or absent now (Driessen and Jarman 2010; Driessen et al. 2021).
Conclusions
We conclude that the effects of adjacent suburbs on native mammals in the reserve were positive or neutral, depending on taxon. The reserve is isolated from other areas of native vegetation by suburbia and farmland, so could theoretically be subject to loss of species that require larger areas of habitat to maintain viable populations, accounting for the apparent absence of species found in the areas in the past. Within the reserve, fire and vegetation structural variables were most associated with variation in mammal visits and signs, reinforcing the importance of continuing to maintain a diversity of fire ages in different vegetation types.
Data Availability
Data and material are available from the authors upon written request.
Code Availability
Not applicable.
References
Arthur AD, Catling PC, Reid A (2012) Relative influence of habitat structure, species interactions and rainfall on the post-fire population dynamics of ground-dwelling vertebrates. Austral Ecol 37:958–970. https://doi.org/10.1111/j.1442-9993.2011.02355x
Baker PJ, Harris S (2007) Urban mammals: what does the future hold? An analysis of the factors affecting patterns of use of residential gardens in Great Britain. Mamm Rev 37:297–215
Bell JNB, Power SA, Jarraud N, Agrawal M, Davies C (2011) The effects of air pollution on urban ecosystems and agriculture. Int J Sustain Dev World Ecol 18:226–235
Bennett AF (1993) Microhabitat use by the long-nosed potoroo, Potorous tridactylus, and other small mammals in remnant forest vegetation of south-western Victoria. Wildl Res 20:267–285
Bidwell S, Attiwill PM, Adams MA (2006) Nitrogen availability and weed invasion in a remnant native woodland in urban Melbourne. Austral Ecol 31:262–270
Brady MJ, McAlpine CA, Miller CJ, Possingham HP, Baxter GS (2011) Mammal responses to matrix development intensity. Austral Ecol 36:35–45
Brearley G, McAlpine C, Bell S, Bradley A (2012) Influence of urban edges on stress in an arboreal mammal: a case study of squirrel gliders in southeast Queensland, Australia. Landsc Ecol 27:1407–1419
Bryant GL, Kobryn HT, Hardy GES, Fleming PA (2017) Habitat islands in a sea of urbanisation. Urban Forestry & Urban Greening 28:131–137. https://doi.org/10.1016/j.ufug.2017.10.016
Busschots MB, Close PG, Van Helden BE, Speldewinde PC (2021) Is the presence of a threatened arboreal mammal in residential areas related to remnant habitats? Austral Ecol 46:181–185. https://doi.org/10.1111/aec.12953
Caryl FM, Lumsden LF, van der Ree R, Wintle BA (2016) Functional responses of insectivorous bats to increasing housing density support “land-sparing’ rather than “land-sharing’ urban growth strategies. J Appl Ecol 53:191–201. https://doi.org/10.1111/1365-2664.12549
Catling PC, Coops NC, Burt RJ (2001) The distribution and abundance of ground-dwelling mammals in relation to time since wildfire and vegetation structure in south-eastern Australia. Wildl Res 28:555–564. https://doi.org/10.1071/wr00041
Claridge AW, Barry SC (2000) Factors influencing the distribution of medium-sized ground-dwelling mammals in southeastern mainland Australia. Austral Ecol 25:676–688. https://doi.org/10.1046/j.1442-9993.2000.01068.x
Conole LE, Kirkpatrick JB (2011) Functional and spatial differentiation of urban bird assemblages at the landscape scale. Landsc Urban Plann 100:11–23
Czech B, Krausman PR, Devers PK (2000) Economic associations among causes of species endangerment in the United States. Bioscience 50:593–601
Daniels GD, Kirkpatrick JB (2012) The influence of landscape context on the distribution of flightless mammals in exurban developments. Landsc Urban Plann 104:114–123
Dixon KM, Cary GJ, Renton M, Worboys GL, Gibbons P (2019) More long-unburnt forest will benefit mammals in australian subalpine forests and woodlands. Austral Ecol 44:1150–1162
Driessen MM, Jarman P (2010) The response of mammal populations to fire in the Peter Murrell reserves: initial survey. Tasman Nat 132:96–100
Driessen MM, Jarman PJ (2014) Comparison of camera trapping and live trapping of mammals in tasmanian coastal woodland and heathland. In: Meek PD, Flemming PJS, Ballard G, Banks P, Claridge AW, Sanderson J, Swann D (eds) Camera trapping: Wildlife Management and Research. CSIRO Publishing, Collingwood, Victoria, pp 253–262
Driessen MM, Taylor RJ, Hocking GJ (1991) Trends in abundance of three marsupial species after fire. Aust Mammal 14:121–124
Driessen MM, Forster L, Bonham K, Throssell A, Rutherford R (2010) Biodiversity monitoring in the Peter Murrell reserves by the Tasmanian Field Naturalist Club. Tasman Nat 132:90–95
Driessen MM, Jarman PJ, Visoiu M, Dewar E (2021) Mammal responses to moderate-intensity planned burning in a small, isolated woodland reserve. Wildl Res 48:561–576. https://doi.org/10.1071/WR20165
Driscoll DA, Bode M, Bradstock RA, Keith DA, Penman TD, Price OF (2016) Resolving future fire management conflicts using multicriteria decision making. Conserv Biol 30:196–205. https://doi.org/10.1111/cobi.12580
Falk JH (1976) Energetics of a suburban lawn. Ecology 57:141–150. https://doi.org/10.2307/1936405
FitzGibbon SI, Putland DA, Goldizen AW (2007) The importance of functional connectivity in the conservation of a ground-dwelling mammal in an urban australian landscape. Landsc Ecol 22:1513–1525. https://doi.org/10.1007/s10980-007-9139-x
Fontaine JB, Kennedy PL (2012) Meta-analysis of avian and small-mammal response to fire severity and fire surrogate treatments in U.S. fire-prone forests. Ecol Appl 22:1547–1561
Garden JG, McAlpine CA, Possingham HP, Jones DN (2007) Habitat structure is more important than vegetation composition for local-level management of native terrestrial reptile and small mammal species living in urban remnants: a case study from Brisbane, Australia. Austral Ecol 32:669–685. https://doi.org/10.1111/j.1442-9993.2007.01750.x
Hope B (2012) Short-term response of the long-nosed bandicoot, Perameles nasuta, and the southern brown bandicoot, Isoodon obesulus, to low-intensity prescribed fire in heathland vegetation. Wildl Res 39:731–744. https://doi.org/10.1071/WR12110
Jarman PJ, Driessen MM (2019) Quantitative interpretation of images of long-nosed potoroos at baited camera-traps: defining a ‘visit’. Aust Mammal 41:147–149. https://doi.org/10.1071/AM17057
Johnson CN (1994) Distribution of feeding activity of the Tasmanian Bettong (Bettongia gaimardi) in relation to vegetation patterns. Wildl Res 21:249–255. https://doi.org/10.1071/wr9940249
Kerle JA (1984) Variation in the ecology of trichosurus: its adaptive significance. In: Smith AP, Hume ID (eds) Possums and gliders. Australian Mammal Society, Sydney, pp 115–128
Kirkpatrick JB (1999) The characteristics and management problems of the vegetation and flora of the Huntingfield area, southern Tasmania. Pap Proc R Soc Tasman 133:103–113
Kirkpatrick JB, Marsden-Smedley JB, Leonard SWJ (2011) Influence of grazing and vegetation type on post-fire flammability. J Appl Ecol 48:642–649. https://doi.org/10.1111/j.1365-2664.2011.01962.x
Kirkpatrick JB, Marsden-Smedley JB, Di Folco M-B, Leonard SWJ (2016) Influence of grazing and vegetation type on post-fire floristic and lifeform composition in Tasmania. Plant Ecol 217:57–69
Lawson DM, Lamar CK, Schwartz MW (2008) Quantifying plant population persistence in human-dominated landscapes. Conserv Biol 22:922–928. https://doi.org/10.1111/j.1523-1739.2008.00936.x
Le Mar K, McArthur C (2005) Comparison of habitat selection by two sympatric macropods, Thylogale billardierii and Macropus rufogriseus rufogriseus, in a patchy eucalypt-forestry environment. Austral Ecol 30:674–683
Lindenmayer DB et al (2008) Contrasting mammal responses to vegetation type and fire. Wildl Res 35:395–408. https://doi.org/10.1071/wr07156
Luck GW, Smallbone L, Threlfall C, Law B (2013) Patterns in bat functional guilds across multiple urban centres in south-eastern Australia. Landsc Ecol 28:455–469. https://doi.org/10.1007/s10980-012-9842-0
Maclagan SJ, Coates T, Ritchie EG (2018) Don’t judge habitat on its novelty: assessing the value of novel habitats for an endangered mammal in a peri-urban landscape. Biol Conserv 223:11–18. https://doi.org/10.1016/j.biocon.2018.04.022
MacLennan DG (1984) The feeding behaviour and activity patterns of the brushtail possum, Trichosurus vulpecula, in an open eucalypt woodland in southeat Queensland. In: Smith AP, Hume ID (eds) Possums and gliders. Australian Mammal Society, Sydney, pp 155–161
Mallick SA, Driessen MM, Hocking GJ (1998) Biology of the southern brown bandicoot (Isoodon obesulus) in south-eastern Tasmania. I Diet Aust Mammal 20:331–338
Marzluff JM (2001) Worldwide urbanization and its effects on birds. In: Marzluff JM, Bowman R, Donnelly R (eds) Avian Ecology in an Urbanizing World. Kluwer Academic Publishers, Norwell, MA, pp 19–47
McArthur C, Goodwin A, Turner S (2000) Preferences, selection and damage to seedlings under changing availability by two marsupial herbivores. For Ecol Manage 139:157–173
McIntyre T, Majelantle TL, Slip DJ, Harcourt RG (2020) Quantifying imperfect camera-trap detection probabilities: implications for density modelling. Wild Res 47:177–185. https://doi.org/10.1071/WR19040
McKinney ML (2002) Urbanization, biodiversity, and conservation. Bioscience 52:883–890
McKinney ML (2006) Urbanization as a major cause of biotic homogenization. Biol Conserv 127:247–260
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11:161–176
McMullan-Fisher SJM, Kirkpatrick JB, May TW, Pharo EJ (2010) Surrogates for macrofungi and mosses in reservation planning. Conserv Biol 24:730–736
Minchin PR (1990) DECODA users manual. Research School of Pacific Studies. Australian National University, Canberra
Monamy V, Fox BJ (2000) Small mammal succession is determined by vegetation density rather than time elapsed since disturbance. Austral Ecol 25:580–587
Norton MA, Claridge AW, French K, Prentice A (2010) Population biology of the long-nosed potoroo (Potorous tridactylus) in the Southern Highlands of New South Wales. Aust J Zool 58:362–368. https://doi.org/10.1071/ZO10075
Ordenana MA et al (2010) Effects of urbanization on carnivore species distribution and richness. J Mammal 91:1322–1331. https://doi.org/10.1644/09-mamm-a-312.1
Quin DG (1985) Observations on the diet of the southern-brown bandicoot, Isoodon obesulus (Marsupialia: Peramelidae), in southern Tasmania. Aust Mammal 11:15–25
Ramalho CE, Ottewell KM, Chambers BK, Yates CJ, Wilson BA, Bencini R, Barrett G (2018) Demographic and genetic viability of a medium-sized ground-dwelling mammal in a fire prone, rapidly urbanizing landscape. PLoS ONE 13. https://doi.org/10.1371/journal.pone.0191190
Ryan CM, Hobbs RJ, Valentine LE (2020) Bioturbation by a reintroduced digging mammal reduces fuel loads in an urban reserve. Ecol Appl 30:1–12
Seebeck JH, Bennett AF, Scotts DJ (1989) Ecology of the Potoroidae — A Review. In: Grigg GC, Jarman PJ, Hume I (eds) Kangaroos, Wallabies and rat-kangaroos. Surrey Beatty and Sons Pty Ltd, New South Wales, Australia, pp 67–88
Shan Y, Paull D, McKay RI (2006) Machine learning of poorly predictable ecological data. Ecol Model 195:129–138. https://doi.org/10.1016/j.ecolmodel.2005.11.015
Shwartz A, Turbé A, Julliard R, Simon L, Prévot A-C (2014) Outstanding challenges for urban conservation research and action. Global Environ Change 28:39–49
Soanes K, Lentini PE (2019) When cities are the last chance for saving species. Front Ecol Environ 17:225–231. https://doi.org/10.1002/fee.2032
Soanes K, Threlfall CG, Ramalho CE, Bekessy S, Fuller RA, Garrard GE, Ikin K, Kendal D, Lee KE, Mumaw L, Rowe R, Shanahan DF, Valentine LE, Williams NSG, Parris KM, Lentini PE (2020) Conservation opportunities for threatened species in urban environments. Report prepared by the Clean Air and Urban Landscapes Hub and Threatened Species Recovery Hub. Australia.)
Southwell CJ, Jarman PJ (1987) Macropod studies at Wallaby Creek. 3. The effect of fire on pasture utilization by macropodids and cattle. Wildl Res 14:117–124. https://doi.org/10.1071/WR9870117
Spotswood EN, Beller EE, Grossinger R, Grenier JL, Heller NE, Aronson MFJ (2021) The Biological Deserts Fallacy: cities in their landscapes contribute more than we think to Regional Biodiversity. Bioscience 71:148–160. https://doi.org/10.1093/biosci/biaa155
Styger JK, Kirkpatrick JB, Marsden-Smedley JON, Leonard SWJ (2011) Fire incidence, but not fire size, affects macropod densities. Austral Ecol 36:679–686. https://doi.org/10.1111/j.1442-9993.2010.02203.x
Tait C, Daniels CB, Hill RS (2005) Changes in species assemblages within the Adelaide metropolitan area, Australia, 1836–2002. Ecol Appl 15:346–359
van der Ree R, McCarthy MA (2005) Inferring persistence of indigenous mammals in response to urbanisation. Anim Conserv 8:309–319. https://doi.org/10.1017/s1367943005002258
Van Helden BE, Close PG, Steven R (2020) Mammal conservation in a changing world: can urban gardens play a role? Urban ecosystems. https://doi.org/10.1007/s11252-020-00935-1
Whelan RJ, Rodgerson L, Dickman CR, Sutherland EF (2002) Critical life cycles of plants and animals: developing a process-based understanding of population changes in fire-prone landscapes. In: Bradstock RA, Williams JE, Gill AM (eds) Flammable Australia. Cambridge University Press, Cambridge, pp 94–124
While GM, McArthur C (2005) Foraging in a risky environment: a comparison of Bennett’s wallabies Macropus rufogriseus rufogriseus (Marsupialia: Macropodidae) and red-bellied pademelons Thylogale billiardierii (Marsupialia: Macropodidae) in open habitats. Austral Ecol 30:756–764
Zungu MM, Maseko MST, Kalle R, Ramesh T, Downs CT (2020) Factors affecting the occupancy of forest mammals in an urban-forest mosaic in EThekwini Municipality, Durban, South Africa. Urban Forestry & Urban Greening 48. https://doi.org/10.1016/j.ufug.2019.126562
NRM (2021) Urban Refuges — Design your garden for eastern barred bandicoots. Natural Resource Management North, Launceston,Tasmania
Acknowledgements
We thank the Tasmanian Parks and Wildlife Service for their support for this work. We thank Jayne Balmer, Margaret Brock, Peter Cusick, Elise Dewar, Peter Feil, Rosemary Gales, Jeremy Hood, Adam Landell, Lindsay Mitchell, Shannon Troy, and Micah Visoiu for their assistance with the project. This study followed the Standard Operating Procedures for Camera-trapping of Wild Tasmanian Mammals 2013 by the Department of Primary Industries, Parks, Water and Environment. This study was partly funded by the Tasmanian Government.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Contributions
Jamie Kirkpatrick designed the vegetation, diggings and scat data collection, collected the vegetation data and undertook most of the analysis and writing up.
Michael Driessen was the major designer of the larger project, collected much of the mammal and environmental data and contributed substantially to the writing of the paper.
Peter Jarman contributed to the design of the project, collected much of the mammal data and contributed to the writing of the paper.
Lauren Jakob collected the diggings and scat data and contributed to the writing of the paper.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
The authors declare that they have no conflict of interest.
Ethics approval
This study followed the Standard Operating Procedures for Camera-trapping of Wild Tasmanian Mammals 2013 by the Department of Primary Industries, Parks, Water and Environment.
Consent to participate
Not applicable.
Consent for publication
was obtained from the Tasmanian Government. All authors consent for the publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kirkpatrick, J.B., Driessen, M.M., Jarman, P.J. et al. Influences of adjacent suburbia, fire regimes and vegetation on the mammals of a peri-urban reserve. Urban Ecosyst 26, 905–916 (2023). https://doi.org/10.1007/s11252-023-01355-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-023-01355-7