Abstract
How does tree species composition vary in relation to geographical and environmental gradients in a globally rare tropical/subtropical broadleaf dry forest community in the Caribbean? We analyzed data from 153 Forest Inventory and Analysis (FIA) plots from Puerto Rico and the U.S. Virgin Islands (USVI), along with 42 plots that we sampled in the Bahamian Archipelago (on Abaco and Eleuthera Islands). FIA data were collected using published protocols. In the Bahamian Archipelago, we recorded terrain and landscape variables, and identified to species and measured the diameter of all stems ≥5 cm at 1.3 m height in 10 m radius plots. All data were analyzed using clustering, ordination, and indicator species analysis at regional and local scales. Regionally, the largest cluster group included over half of all plots and comprised plots from all three island groups. Indicator species were native Bursera simaruba (Burseraceae) and Metopium toxiferum (Anacardiaceae). Species composition was similar to dry forests throughout the region based on published studies. Other groups we identified at the regional scale consisted of many Puerto Rico and USVI plots that were dominated by non-native species, documenting the widespread nature of novel ecosystems. At the local scale the Bahamian data clustered into two main groups corresponding largely to the two islands sampled, a pattern consistent with the latitudinal aridity gradient. Bahamian dry forests share previously undocumented compositional similarity with native-dominated dry forests found throughout the Caribbean, but they lack extensive post-disturbance novel dry forests dominated by non-native trees found in the Greater Antilles.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding regional variation in plant community composition is required in an era of global change because ecosystem processes and services depend on taxon-specific responses to climate change, land use change, novel species combinations, and altered disturbance regimes (Sala et al. 2000; Tylianakis et al. 2008). Describing plant communities also serves important purposes in conservation planning. Plant communities are used to set conservation targets for terrestrial ecosystems (Keith et al. 2013; Murdoch et al. 2010), and as a “coarse filter” (Noss 1987) for estimating patterns of biodiversity to assess gaps in conservation coverage (Butterfield et al. 1994). Major ongoing efforts aim to develop vegetation classifications using standardized protocols and quantitative methods (Franklin et al. 2012); a global classification does not yet exist, however, and there is great unevenness in the geographical distribution of quantitative plant community data (Dengler et al. 2011).
Janzen (1988) declared that tropical dry forests are the most threatened tropical forest ecosystems, a statement that remains valid (Gillespie et al. 2012; Miles et al. 2006). Patterns of diversity and composition of Neotropical dry forests have been broadly described (Gentry 1995), and ecological attributes of Antillean (Lugo et al. 2006) and south Florida, US (Gillespie 2006) dry forests have been reviewed. Although dry forests have been studied on individual islands within the Caribbean region, a comprehensive, quantitative analysis of Caribbean dry forest composition is lacking. Furthermore, dry forest communities in some parts of the Caribbean, such as the Bahamian Archipelago, are not well studied (Carey et al. 2014). The Bahamian Archipelago is divided politically into The Commonwealth of the Bahamas and The Turks and Caicos Islands.
The broadleaf forest found in the Bahamian Archipelago, known locally as “coppice” (Correll and Correll 1982), has been called dry evergreen forest (Smith and Vankat 1992), lowland tropical or subtropical seasonal evergreen forest (Areces-Mallea et al. 1999) or Caribbean dry forest (Dunphy et al. 2000). We use “dry forest” in this paper for simplicity. Bahamian dry forest is a globally rare and endangered ecosystem, supporting endemic and endangered species (WWF Worldwide Report 2001 http://worldwildlife.org/ecoregions/nt0203). It has been identified as an important conservation target, threatened by clearing and degradation (Sullivan-Sealey et al. 2002; Wunderle and Waide 1993). Little is known, however, about its relationship to other dry forests in the region, or compositional variation within the archipelago, where dry forest has been studied only within single islands (Currie et al. 2005a; Franklin and Steadman 2013; Freid and Kerwin 1998; Helmer et al. 2010; Smith and Vankat 1992).
This study begins to fill the gap in our knowledge of regional variation in Caribbean dry forest composition by addressing the following research questions: (1) What are the broad regional patterns of variation in Caribbean dry forest composition? (2) Is there local variation in dry forest composition within the less-studied Bahamian islands that is geographically or environmentally structured?
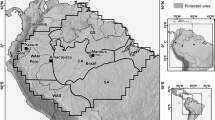
We addressed the first question by analyzing Forest Inventory and Analysis (FIA) data available from the U.S. Department of Agriculture (USDA) Forest Service for Puerto Rico and the U.S. Virgin Islands (USVI) in conjunction with comparable plot data we collected on Bahamian islands. These data range across two-thirds of the latitudinal extent and half of the longitudinal extent of the West Indies (Fig. 1). Results of this regional analysis are compared to published descriptions of dry forests elsewhere in the Caribbean. We expected that composition would vary among major island groups in our sample because of differences in island size and elevation, as well as geological substrate. We then addressed the second question by comparing our Bahamian dry forest data from two islands—Abaco and Eleuthera. These islands are separated by deep water, and together span more than 2° of latitude and nearly 2° of longitude, almost half of the linear extent of the archipelago. We expected to discover latitudinal variation in species composition related to the north–south gradient of increasing aridity and warmth.
The Caribbean study region showing the Bahamian Archipelago (The Bahamas + Turks and Caicos Islands), Cuba, Jamaica, Hispaniola, and Puerto Rico (Greater Antilles), the U.S. Virgin Islands, and the southern portion of Florida, US. Inset shows with the number of vegetation plots for locations on Abaco and Eleuthera. Plots are clustered and cannot be discerned at this scale. For example, 8 plots were located in a large patch of forest on east-central Abaco in the area known as Wilson City
Materials and methods
Study area
FIA data came from the USVI and the Commonwealth of Puerto Rico (the main island of Puerto Rico and its outlying islands), and published vegetation studies also provided descriptions of dry forest on the large islands of the Greater Antilles (Jamaica, Cuba, Puerto Rico and Hispaniola), and in south Florida, US (Fig. 1). Dry forests of Puerto Rico and throughout the Greater Antilles tend to occur at low elevations and often on limestone (Lugo et al. 2006). The USVI are volcanic, low, hilly islands at the eastern margin of the Greater Antilles. FIA dry forest plots were located near the southwestern (leeward) coast of the island of Puerto Rico and nearby islands of Mona, Vieques, and Culebra, and at lower elevations of St. Croix, St. John and St. Thomas islands in the USVI. Climate data for the locations of the dry forest plots were extracted from WorldClim (worldclim.org; accessed 22 Sep 2014); temperatures for those locations range from an average high of 31 °C in Aug to average low of 20 °C in Jan, with average annual precipitation ~1200 mm (range 900–1900 mm). Although some error is deliberately added to the reported locations of FIA plots owing to privacy regulations (O’Connell et al. 2014)), this merely added a small amount of uncertainty to our descriptive climate statistics. Many of the dry forests found today in Puerto Rico and the USVI are dominated by young small-diameter trees. Furthermore, naturalized, introduced species are an important component of forests regenerating on abandoned cropland and pastures (Brandeis and Turner 2013a, b; Molina Colón et al. 2011).
The Bahamian Archipelago consists of limestone from indurated calcareous sand deposited on massive, shallow carbonate banks (Sealey 1994). Abaco (also called Great Abaco), which lies on the Little Bahama Bank, is the second largest island in the archipelago (1244 km2), has a maximum elevation of 44 masl, and extends ~120 km N–S (Fig. 1). Climate data were not available for Abaco but at nearby Freeport, Grand Bahama (26º30′N) temperatures range from an average high of 30 °C in Aug to average low of 18 °C in Jan, with annual precipitation ~1450 mm. Abaco at 26–27ºN experiences more winter cold fronts than Eleuthera (Sealey 1994). The most extensive vegetation community on Abaco is pine woodland (Areces-Mallea et al. 1999), dominated by Pinus caribaea var. bahamensis, endemic to the Bahamian Archipelago and found on only six of its islands (Sullivan-Sealey et al. 2002). Dry forest occupies about 10 % of the land area of Abaco. Eleuthera (457 km2, maximum elevation 60 masl) lies on the Great Bahama Bank, is about 120 km long NW–SE, and in places is <1.5 km wide (Fig. 1). Climate data were not available for Eleuthera but at nearby Nassau, New Providence (25º4′N) temperatures range from an average high of 32 °C in Aug to average low of 21 °C in Jan, with ~1400 mm annual precipitation. Upland habitats on Eleuthera are dominated by broadleaf dry forest and scrub. Abaco and Eleuthera, lying on different Bahamian Banks, would have each been part of much larger islands and closer together when these shallow banks were exposed at lower sea-levels during much of the Pleistocene (Steadman and Franklin 2015).
Bahamian dry broadleaf woody vegetation is subdivided into “short” or “low” (<4.6 m) scrub versus “tall” or “high” (>4.6 m) forest that differs primarily in stature, because of variation in site quality or time since disturbance (Currie et al. 2005b), and into coastal (“whiteland”) versus inland (“blackland”) forest. Coastal broadleaf woody vegetation occurs on sands near the coast and tends to be short. Inland broadleaf woody vegetation is found on limestone with greater soil development, is typically taller, and differs somewhat in species composition from the coastal form (Correll and Correll 1982; Smith and Vankat 1992). Our study focuses on inland, tall forest, although these forms grade into one another in many locations. Bahamian dry forest is considered to be resilient to hurricanes that occur frequently there (Morrison and Spiller 2008), although anthropogenic disturbances include clearing, grazing, and fire (Franklin and Steadman 2013; Larkin et al. 2012; Miller 2007; Sullivan-Sealey et al. 2002).
Data collection
Forest plot data for Puerto Rico (sampled between 2001 and 2009) and the USVI (2004–2009) were acquired from the USDA Forest Service’s Forest Inventory and Analysis (FIA) Program (http://www.fia.fs.fed.us/). FIA plots from Puerto Rico have been used to develop biomass and volume equations for dry forest (Brandeis et al. 2006) and those from the USVI have been used in a study of bird habitat on St. John (Steadman et al. 2009). We extracted data for 184 plots classified as “subtropical dry forest.” Of these, 31 plots were excluded from our analyses (leaving 153) because their multivariate distance from another plot was zero, precluding some multivariate analyses. These were plots with only a few trees, all of a single species. For example, 25 of the excluded plots contained only a single species of a non-native leguminous tree. In FIA plots, dbh (diameter at breast height) is measured at 1.37 m height on the trunk and the height of each tree is measured. Trees ≥12.5 cm dbh are sampled in 4 clustered circular subplots of 168 m2 each (total sampling area 672 m2) whose centers are 36.6 m apart; trees 2.5 to <12.5 cm dbh are sampled in 4 circular microplots of 13.46 m2 (total sampling area 53.84 m2) nested within subplots (Gray et al. 2012; O’Connell et al. 2014)). We extracted data only for trees ≥ 5.0 cm dbh for comparability with our Bahamian data.
In the Bahamian Archipelago, we established 20 plots on Abaco and 22 plots on Eleuthera. Plot locations were geographically stratified in order to sample all major patches of inland dry forest on Abaco and to span the extent of Eleuthera (Fig. 1). Random sampling was not possible because access was challenged by rugged terrain, lack of trails, and land ownership. For efficiency, plots often were located in nearby pairs (>500 m apart) or, in the case of Abaco, on transects associated with bird surveys (Franklin and Steadman 2013). Sampling took place on 7 Jan and 6–9 May 2009; 9 Jan, 12–17 Mar, 4–6 May, and 30 Nov-3 Dec 2012; and 5–10 Dec 2013. We did not observe any large scale canopy gaps created by the hurricanes that affected the islands during this time frame.
Bahamian plots were 10 m radius (314 m2), a plot size adequate for forest community analysis (Otypková and Chytry 2006). Within each plot, the diameters of all stems ≥ 5 cm diameter were measured at 1.3 m height on the trunk (“dbh”). Basal area per tree was summed to yield species basal area. Trees were identified to the greatest level of taxonomic resolution possible. A few individuals not in fruit or flower could not be identified to species level, and were identified to genus or family level or left as unknowns in our analyses (Appendix S2). Tree height was measured using an Impulse laser rangefinder (Laser Technology Inc., Centennial, CO, US) based on the average height of five overstory trees, chosen evenly among all directions from plot center. Slope angle and aspect were measured with a clinometer and compass, and percent rock exposed in the plot was estimated visually. Plot locations were recorded with a Garmin Global Positioning System (GPSMAP 60CSx, Garmin International Inc., Olathe, KS). Plot elevation and distance from the nearest road and coastline were measured using Google Earth Pro (Google Inc., Menlo Park, CA). Plot and environmental data are provided in Supplemental Material Appendices S1 and S2 and archived in VegBank (www.vegbank.org). While FIA plots differed from the Bahamian plots in plot size and (very slightly) height of dbh measurement, the multivariate analysis methods we used are based on relative species importance and are robust to plot-size differences far greater than those found in our study (Otypková and Chytry 2006).
Data analysis
Analyses of species composition were based on patterns of relative abundance described by species Importance Value (average of relative basal area and relative density per plot). Analyses were also conducted using relative basal area for comparison. Clustering, based on Jaccard distance and average linkage (Peet and Roberts 2013), was calculated from the plot-by-species Importance Value matrix in order to detect subgroups of plots sharing distinct assemblages of species. Permutational Multivariate Analysis of Variance or PERMANOVA (Anderson 2001), a non-parametric test based on permuting observations to obtain a probability of difference among groups, was used to evaluate the significance of compositional differences among groups defined by clustering. Indicator Species analysis (Dufrêne and Legendre 1997) was used to identify species that have strong association (high relative abundance within the group) and fidelity (occurring in many plots within but few outside the group) with groups defined by clustering. Unconstrained ordination with Non-metric Multidimensional Scaling (NMDS) (Clarke 1993) was used to visualize continuous variation in species composition among plots and the distinction among groups identified by clustering. Detrended correspondence analysis was run in parallel, with the results examined for congruence (Økland 1996). This sequence of clustering and ordination was carried out twice—first for the 153 FIA plots together with the 42 Bahamian plots in order to analyze regional variation in forest composition, and then for the Bahamian plots alone in order to examine local gradients of composition within the Bahamian Archipelago.
Additionally, Canonical Correspondence Analysis (CCA), a constrained ordination method (ter Braak 1987), was conducted for the 42 Bahamian plots in order to identify local scale compositional variation explained by environmental variables. These variables described geographical location (latitude, longitude), terrain (slope, aspect, percent rock), landscape (distance from coast, distance from road), and stand structure (average tree height). Statistical analyses were conducted using R (R Development Core Team 2012); vegan (Oksanen et al. 2011) was used for PERMANOVA, NMDS, DCA and CCA, and labdsv (Roberts 2012) was used for Indicator Species analysis. Nomenclature follows Correll and Correll (1982) for Bahamian species, with updates from Acevedo-Rodríguez and Strong (2012), and for FIA data, the USDA, NRCS PLANTS database (2014).
Results
Regional patterns
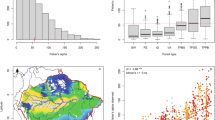
Summaries of the regional plot data are given in Table 1. Over 75 % of FIA dry forest plots in Puerto Rico and USVI were found below 100 masl. Multivariate analyses that used relative basal area versus Importance Value yielded almost identical results and so only those based on Importance Value are reported here. The combined FIA and Bahamian plots included 193 taxa. Based on clustering the regional data and retaining 13 clusters (PERMANOVA R = 0.073, P = 0.001 based on 999 permutations), the largest group comprised more than half of all plots, including all 42 Bahamian plots, almost half of the Puerto Rican plots (45 of 91), and one quarter (16 of 62) of the plots from USVI (Group 1; Table 2). Indicator species (Indicator Species Value > 0.48 and P ≤ 0.2) for this group were Bursera simaruba and Metopium toxiferum, with Coccoloba diversifolia also abundant (Table 2). Although the NMDS plot suggests some differences in dry forest composition among the Bahamian Archipelago, Puerto Rico and USVI (Fig. 2), the plots in this large group are much more similar to each other than they are to other dry forest plots in the FIA sample.
Ordination diagram showing the distribution of 153 plots from the Bahamian Archipelago (Bah), Puerto Rico (PR), and U.S. Virgin Islands (VI) on first two dimensions (NMDS1, NMDS2) derived from unconstrained Non-metric Multidimensional Scaling (NDMS; stress = 27.0 %). Symbols indicate the island groups. Ellipses indicate the 95 % confidence ellipse for the two largest groups (Groups 1 and 3; see Table 1). Note the main group (Group 1) with plots from all three regions (see Table 1)
Group 3 (55 plots from Puerto Rico and USVI; Fig. 2) was dominated by Leucaena leucocephala and Prosopis pallida, both non-native and naturalized disturbance indicators, and Group 12 (13 plots from USVI) was dominated by Melicoccus bijugatus (an Indicator Species), also non-native, naturalized and considered invasive (Table 2). Group 12 was also dominated by Trema micrantha, a small shade-intolerant tree, and included Tamarindus indica, a planted and naturalized fruit tree. This group appears to represent forests with a legacy of human activity (clearing, agriculture). All other groups identified by clustering included less than 10 plots (five of them with only one plot), and therefore are outliers of unique species composition, or perhaps forest types that have not been adequately sampled. Notably, as with Groups 3 and 12, 10 of these remaining 24 plots were dominated by non-native, potentially invasive leguminous trees (Acacia farnesiana, Pithecellobium dulce) that usually establish in thickets and near roads.
Local patterns
Average linkage clustering of the local data (42 Bahamian plots), retaining eight clusters, resulted in significantly different groups (PERMANOVA R = 0.102, P = 0.001 based on 999 permutations). Two large clusters accommodated most plots from Abaco or Eleuthera. Group B1 included 13 of 20 plots from Abaco, whereas Group B3 contained 17 of 22 plots from Eleuthera (and 2 from Abaco). The six additional “clusters” each included only one to three plots from Abaco or Eleuthera with distinctive species composition (Fig. 3).
Dry forest on both Abaco (Group B1) and Eleuthera (Group B3) was dominated by Coccoloba diversifolia, with Metopium toxiferum, Sideroxylon foetidissimum, Bursera simaruba, Exothea paniculata and Guapira obtusata also dominant or abundant (Table 3). The Group B1 plots were distinguished from those of Group B3 by greater abundance of M. toxiferum and S. foetidissimum, and the presence of Swietenia mahagoni. Group B3 also had four minor species that were absent from Group B1 (Table 3).
Other groups identified by clustering included only one to three plots of distinctive species composition, such as three Abaco plots dominated by Eugenia axillaris (Group B2; Fig. 3), a rocky hilltop Eleuthera plot dominated by Guaiacum sanctum and Lysiloma latisilliquum (B5), an Eleuthera plot distinguished by the palm Pseudophoenix sargentii (B7), and another Eleuthera plot (B8) with a diverse set of indicator species (Schaefferia frutescens, Stenostomum lucida, Erythroxylum areolatum, Guettarda krugii and Bourreria succulenta).
The first two axes of the constrained ordination, CCA, carried out for the Bahamian data had eigenvalues of 0.3706 and 0.2406 (variance explained 0.0915 and 0.0594), suggesting that the first axis represents a fairly strong gradient and the second axis a somewhat weaker one. For comparison, unconstrained ordination, DCA, had eigenvalues of 0.4762 and 0.2992. CCA axes 1 and 2 were positively correlated with latitude, distance from roads, and tree height, and negatively correlated with longitude, elevation, and slope (Table 4). Rockiness was negatively correlated with CCA1 and positively with CCA2. Group B1 plots were at higher latitude and more negative longitude (north and west, because we used the convention of negative longitude values west of Greenwich), at greater distances from roads, and with taller canopies (Figs. 3 and 4). Group B3 sites were at somewhat higher elevations, on steeper, rockier slopes, and at greater longitude and lower latitude (further east and south; Appendix S1).
Ordination diagram showing the distribution of 42 plots from Abaco and Eleuthera on first two dimensions (CCA1, CCA2) based on constrained ordination using Canonical Correspondence Analysis (CCA). Symbols indicate the 8 groups (Grp B#) of plots defined by clustering (see text). 95 % confidence ellipses are shown for Grp B1 and Grp B3. Vectors show the correlation of environmental variables with constrained ordination axes, vector length indication strength of correlation (shown for variables with P = 0.001; defined in Table 3)
Discussion
Regional patterns
We expected to see variation in composition among the island groups analyzed in this study because of differences in island size, elevation, and substrate; instead we found remarkable similarities in native-dominated Caribbean dry forest at the regional level when data from the Bahamian Archipelago, Puerto Rico, and USVI were analyzed. Most plots fell into a single region-wide dry forest community type dominated by native species Bursera simaruba and Metopium toxiferum. A geographically structured compositional difference is the dominance of Coccoloba diversifolia in the Bahamian Archipelago and its absence from most FIA dry forest plots in Puerto Rico and the USVI. Interestingly, C. diversifolia was recorded frequently in other FIA plots from Puerto Rico and the USVI classified as subtropical moist forest, whereas C. microstachya was recorded frequently in FIA dry forest plots, suggesting niche displacement for these congeners (Silvertown 2004).
Comparison with published descriptions of dry forest communities provides additional evidence for region-wide compositional similarities (Table 5). Greater Antillean dry forests, especially those on limestone substrates, have similar species composition to our Group 1 forests, although when found at higher elevations those forests tend to be more diverse and taller, with greater basal area. A mature dry forest at ~100 masl on limestone in Puerto Rico, in the well-studied Guánica Biosphere Reserve (Molina Colón and Lugo 2006), was dominated by Coccoloba diversifolia, and shared other species with our Group 1. Of the limestone dry forests surveyed in Jamaica by Kelly et al. (1988), their Round Hill sites at ~300 masl (annual precipitation ~1200 mm) had different dominants, but nonetheless shared many species and genera with the plots analyzed in our study (Table 5). A mature dry forest on limestone was described by Roth (1999) in the Cordillera Central of the Dominican Republic (Hispaniola) at ~300–500 masl (1000 mm strongly seasonal annual rainfall). The most abundant species featured those that dominated our Group 1 (Bursera simaruba, Coccoloba diversifolia and so forth) as well as some not found in the Bahamian Archipelago (Oxandra lanceolata; Table 5). In Cuba, seasonal evergreen forest on limestone-derived soils is similar in composition to these other dry forests and to our Group 1, although it lacks Coccoloba diversifolia; instead it is co-dominated by Sideroxylon foetidissimum, S. salicifolium, Roystonea regia, and Bursera simaruba (Table 5), with many other associated tree species (Areces-Mallea et al. 1999; Borhidi 1991). The absence of Swietenia mahagoni from Greater Antillean dry forests on islands where it is native may be a result of historic selective logging (Rodan et al. 1992). S. mahagoni is introduced and naturalized in Puerto Rico and the USVI.
Other studies have noted floristic similarities among dry forests on limestone in the Greater Antilles (Trejo-Torres and Ackerman 2002). Gillespie (2005) also recorded many of the important species identified in our analyses in subtropical dry forest sites on mainland southern Florida and the Florida Keys (Table 5). Taken together, our quantitative analysis and comparison with published studies suggest a region-wide dry forest type for south Florida, the Bahamian Archipelago and the Greater Antilles. Comparable plot data from throughout the region, as well as from dry forest in the Lesser Antilles, Central America and northern South America, would allow a more detailed quantitative classification of variation within the region to be used as a basis for conservation planning.
Notably, the other two dry forest groups identified in our region-wide analysis were dominated by non-native species. An extensive group found in Puerto Rico and the USVI is dominated by non-native Leucaena leucocephala and Prosopis pallida, and one found only in the USVI is dominated by non-native Melicoccus bijugatus (Table 1). Tropical dry forests are particularly susceptible to invasion by non-native species owing to extensive human disturbance including land clearing and fire (Fine 2002). These novel ecosystems (Lugo and Helmer 2004; Martinuzzi et al. 2013; Molina Colón et al. 2011) with a legacy of human activity are widespread in Puerto Rico and the USVI but not on Bahamian islands. These two groups, as well as many of the remaining dry forest plots that did not cluster into large groups, were dominated by naturalized species considered to be invasive (Rojas-Sandoval and Acevedo-Rodríguez 2015) and constitute post-disturbance communities (Lugo et al. 2006). This finding reinforced concerns about the conservation status of dry forest (Janzen 1988; Gillespie et al. 2012).
Local patterns
At the local scale on Bahamian islands, we found some evidence for the expected variation in species composition correlated with the north to south gradient of increasing aridity and warmth. Some species recorded in plots on Eleuthera but not Abaco (Guaiacum sanctum, Pithecellobium keyense) are abundant in the dry forests of the warmer, more arid southern Bahamian islands (JF, EHF and DWS, personal observation). Most species characterizing these groups were found on both islands, however, and the subtle differences between them were mainly in patterns of species dominance (Table 2). The taller canopies recorded in plots on Abaco may indicate that dry forest, restricted to limestone ridges on this pine island, occurs in protected and productive sites, while on Eleuthera dry forest is the dominant terrestrial plant community and was sampled across a wide range of site conditions. Differences in canopy height and composition may also be related to disturbance history (Larkin et al. 2012), although dry forest on both islands is subjected to similar natural and human disturbance regimes, and plots were located in tall, closed canopy forests, e.g. mid- to late-successional.
Published studies on dry forest composition in the Bahamian Archipelago can be used to more fully describe local patterns of variation. Previous studies, focused on single islands, described forest communities with varying combinations of the same dominant species found in this study. On North Andros (Great Bahama Bank), Smith and Vankat (1992) quantitatively defined three inland dry forest communities that, as in our findings, were distinguished only by relative abundance of the dominant species C. diversifolia, M. toxiferum, Exothea paniculata and/or B. simaruba. They found the compositional gradient to be weakly related to elevation (while ours was not). E. H. Freid (unpublished data) sampled dry forest at Clifton National Heritage Park on New Providence. The forest there was dominated by Coccoloba sp. and Nectandra coriacea, with greater abundance of B. simaruba nearer the coast. Subdominants included Eugenia axillaris, E. foetida, Tabebuia bahamensis and S. foetidissimum. This forest is somewhat more similar to Group B1 (Abaco) than to B3 (mostly Eleuthera) in spite of being on the Great Bahama Bank.
Although several geographical and environmental variables measured for our Bahamian plots were correlated with species composition, only rockiness correlated with composition independently of the geographical factors that differed systematically between the two islands. Distance from the coast was not correlated with species composition, but this may have been a result of the sampling focused on the inland “blackland” form of dry forest.
Vegetation classification for conservation planning
These striking similarities and overlap in dominant species composition in our regional-scale analysis suggest that a more comprehensive, quantitative region-wide treatment of Caribbean dry forest is needed. In addition to geographical variation in community composition, temporal dynamics (succession following disturbance) also need to be better understood (Franklin 2007; Larkin et al. 2012). The Nature Conservancy’s Guide to Caribbean Vegetation Types (Areces-Mallea et al. 1999) was developed to establish regional conservation priorities. While this classification is comprehensive in its treatment of rain forest, it did not describe the Greater Antillean dry forests of Hispaniola and Jamaica summarized in Table 5. Furthermore, it included only a single dry forest Alliance for the Bahamian Archipelago (Reynosia septentrionalis—Sideroxylon americanum—Pithecellobium keyense—Jacquinia keyensis Forest), synonymized with “whiteland” (e.g., coastal) coppice (Correll and Correll 1982); no inland Bahamian dry forest was described by Areces-Mallea et al. (1999).
Our study begins to address this lack of quantitative understanding of Caribbean dry forest communities, although we note that data are lacking from the southeastern half of the Bahamian Archipelago, a gap that we hope to fill. More comprehensive analysis of dry forest associations at both regional and local scales could be accomplished by collaboration among vegetation scientists working in the Caribbean (for an example, see Franklin et al. 2013). Such analyses could help define Caribbean dry forest types at the mid-level of established vegetation classification hierarchies; e.g., our regional Group 1 may contribute to vegetation class descriptions at the Macrogroup level of the U.S. National Vegetation Classification System (Franklin et al. 2012). Local analyses will help to define classes at lower levels in a hierarchy, such as Alliances or Associations (e.g., our groups B1 and B3), that form the basis of conservation planning. A regionally coherent classification is needed to guide conservation management of this widespread yet threatened forest community in an era of global change.
References
Acevedo-Rodríguez P, Strong MT (2012) Catalogue of seed plants of the West Indies. Smithson Contribut Bot 98:1–1192
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austr Ecol 26:32–46
Areces-Mallea AE, Weakley AS, Li X, Sayre RG, Parrish JD, Tipton CV, Boucher T (1999) A guide to Caribbean vegetation types: preliminary classification system and descriptions. Nature Conservancy, Washington
Borhidi A (1991) Phytogeography and vegetation ecology of Cuba. Akademiai Kiado, Budapest
Brandeis TJ, Turner JA (2013a) Puerto Rico’s forests, 2009. Resour Bull. SRS-191. U.S. Department of Agriculture Forest Service, Southern Research Station, Asheville
Brandeis TJ, Turner JA (2013b) U.S. Virgin Islands’ forests, 2009. Resour Bull. SRS-196. U.S. Department of Agriculture Forest Service, Southern Research Station, Asheville
Brandeis TJ, Delaney M, Parresol BR, Royer L (2006) Development of equations for predicting Puerto Rican subtropical dry forest biomass and volume. Forest Ecol Manage 233:133–142
Butterfield BR, Csuti B, Scott JM (1994) Modeling vertebrate distribution for gap analysis. In: Miller RI (ed) Mapping the diversity of nature. Chapman and Hall, London, pp 53–68
Carey E, Gape L, Manco BN, Hepburn D, Smith R, Knowles L, Knowles D, Daniels M, Vincent M, Freid E, Jestrow B, Griffith MP, Calonje M, Meerow A, Stevenson D, Francisco-Ortega J (2014) Plant conservation challenges in the Bahama Archipelago. Bot Rev 80:265–282
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. J Ecol 18:117–143
Correll DS, Correll HB (1982) Flora of the Bahama archipelago. A.R.G. Gantner Verlag K.-G., Fl-9490 Vaduz
Currie D, Wunderle JM, Ewert DN, Anderson MR, Davis A, Turner J (2005a) Habitat distribution of birds wintering in Central Andros, The Bahamas: implications for management. Caribb J Sci 41:75–87
Currie D, Wunderle JM, Ewert DN, Davis A, McKenzie Z (2005b) Winter avian distribution and relative abundance in six terrestrial habitats on southern Eleuthera, The Bahamas. Caribb J Sci 41:88–100
Dengler J, Jansen F, Glockler F, Peet RK, De Caceres M, Chytry M, Ewald J, Oldeland J, Lopez-Gonzalez G, Finckh M, Mucina L, Rodwell JS, Schaminee JHJ, Spencer N (2011) The Global Index of Vegetation Plot Databases (GIVD): a new resource for vegetation science. J Veg Sci 22:582–597
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366
Dunphy B, Murphy P, Lugo AE (2000) The tendency for trees to be multiple-stemmed in tropical and subtropical dry forests: studies of Guánica forest, Puerto Rico. Trop Ecol 41:161–168
Fine PV (2002) The invasibility of tropical forests by exotic plants. J Trop Ecol 18:687–705
Franklin J (2007) Recovery from clearing, cyclone and fire in rain forests of Tonga, South Pacific: vegetation dynamics 1995–2005. Austr Ecol 32:789–797
Franklin J, Steadman DW (2013) The winter bird communities in pine woodland versus broadleaf forest on Abaco, The Bahamas. Caribb Nat 3:1–18
Franklin S, Faber-Langendoen D, Jennings M, Keeler-Wolf T, Loucks O, Peet R, Roberts D, McKerrow A (2012) Building the United States national vegetation classification. Annali di Botanica 2:1–9
Franklin J, Keppel G, Webb EL, Rey SJ, Seamon JO, Wiser SK, Drake DR, Steadman DW (2013) Dispersal limitations, speciation, environmental filtering and niche differentiation influence forest tree communities in West Polynesia. J Biogeogr 40:988–999
Freid EH, Kerwin MA (1998) Flora and vegetation of Bell Island (Exuma Chain), Bahamas. In: Wilson TK (ed) Seventh Symposium on the Natural History of the Bahamas. Bahamian Field Station Ltd, San Salvador, pp 41–52
Gentry A (1995) Diversity and floristic composition of Neotropical dry forests. In: Bullock SH, Mooney HA, Medina E (eds) Seasonally dry tropical forests. Cambridge University Press, Cambridge, pp 146–194
Gillespie TW (2005) Predicting woody-plant species richness in tropical dry forests: a case study from south Florida, USA. Ecol Appl 15:27–37
Gillespie TW (2006) Diversity, biogeography and conservation of woody plants in tropical dry forest of south Florida. In: Pennington RT, Lewis GP, Ratter JA (eds) Neotropical savannas and seasonally dry forests: Biodiversity, biogeography and conservation. CRC Press, Boca Raton, pp 383–394
Gillespie TW, Lipkin B, Sullivan L, Benowitz DR, Pau S, Keppel G (2012) The rarest and least protected forests in biodiversity hotspots. Biodivers Conserv 21:3597–3611
Gray AN, Brandeis TJ, Shaw JD, McWilliams WH, Miles PD (2012) Forest inventory and analysis database of the United States of America (FIA). Veg Databases for the 21st Century Biodiv Ecol 4:255–264
Helmer EH, Ruzyki TS, Wunderle JM Jr, Vogesser S, Ruefenacht B, Kwit C, Brandeis TJ, Ewert DN (2010) Mapping tropical dry forest height, foliage height profiles and disturbance type and age with a time series of cloud-cleared Landsat and ALI image mosaics to characterize avian habitat. Remote Sens Environ 114:2457–2473
Janzen DH (1988) Tropical dry forests. National Academy Press, Washington
Keith DA, Rodriguez JP, Rodriguez-Clark KM, Nicholson E, Aapala K, Alonso A, Asmussen M, Bachman S, Basset A, Barrow EG (2013) Scientific foundations for an IUCN Red List of ecosystems. PLoS ONE 8:e62111
Kelly D, Tanner E, Dickinson T, Goodfriend G, Fairbairn P, Kapos V (1988) Jamaican limestone forests: floristics, structure and environment of three examples along a rainfall gradient. J Trop Ecol 4:121–156
Larkin CC, Kwit C, Wunderle JM, Helmer EH, Stevens MHH, Roberts MT, Ewert DN (2012) Disturbance type and plant successional communities in Bahamian dry forests. Biotropica 44:10–18
Lugo AE, Helmer E (2004) Emerging forests on abandoned land: Puerto Rico’s new forests. Forest Ecol Manage 190:145–161
Lugo AE, Medina E, Trejo-Torres JC, Helmer E (2006) Botanical and ecological basis for the resilience of Antillean Dry Forests. In: Pennington RT, Lewis GP, Ratter JA (eds) Neotropical savannas and seasonally dry forests. CRC Press, Boca Raton, pp 359–381
Martinuzzi S, Lugo AE, Brandeis TH, Helmer EH (2013) Case study: geographic distribution and level of novelty of Puerto Rican forests. In: Hobbs RJ, Higgs ES, Hall CM (eds) Novel ecosystems: Intervening in the new ecological world order. John Wiley & Sons. Ltd., Chichester, pp 81–87
Miles L, Newton AC, DeFries RS, Ravilious C, May I, Blyth S, Kapos V, Gordon JE (2006) A global overview of the conservation status of tropical dry forests. J Biogeogr 33:491–505
Miller AC (2007) Fire history of Caribbean Pine (Pinus caribaea var. bahamensis (Griseb.) W.H. Barrett & Golfari) forests on Abaco Island, The Bahamas. Masters Thesis. Geography. University of Tennessee, Knoxville, p 92
Molina Colón S, Lugo AE (2006) Recovery of a subtropical dry forest after abandonment of different land uses. Biotropica 38:354–364
Molina Colón S, Lugo AE, Ramos González OM (2011) Novel dry forests in southwestern Puerto Rico. Forest Ecol Manage 262:170–177
Morrison LW, Spiller DA (2008) Patterns and processes in insular floras affected by hurricanes. J Biogeogr 35:1701–1710
Murdoch W, Ranganathan J, Polasky S, Regetz J (2010) Using return on investment to maximize conservation effectiveness in Argentine grasslands. Proc Nat Acad Sci USA 107:20855–20862
Noss RF (1987) From plant communities to landscapes in conservation inventories: a look at The Nature Conservancy (USA). Biol Conserv 41:11–37
O’Connell BM, LaPoint EB, Turner JA, Ridley T, Pugh SA, Wilson AM, Waddell KL, Conkling BL (2014) The Forest Inventory and Analysis Database: Database Description and User Guide Version 6.0 for Phase 2. U.S. Department of Agriculture Forest Service, Raleigh, p 624
Økland RH (1996) Are ordination and constrained ordination alternative or complimentary strategies in general ecological studies? J Veg Sci 7:289–292
Otypková Z, Chytry M (2006) Effects of plot size on the ordination of vegetation samples. J Veg Sci 17:465–472
Oksanen J, Guillaume Blanchet F, Kindt R, Legendre P, O’Hara RB, Simpson GL, Solymos P, H. SMH, Wagner H (2011) vegan: Community Ecology Package. R package version 1.17-11. http://CRAN.R-project.org/package=vegan
Peet RK, Roberts DW (2013) Classification of natural and semi-natural vegetation. In: van der Maarel E, Franklin J (eds) Vegetation Ecology. Wiley-Blackwell, Chichester, pp 28–70
Roberts DW (2012) Labdsv: ordination and multivariate analysis for ecology. R package version 1.5-0. http://CRAN.R-project.org/package=labdsv
Rodan BD, Newton AC, Verissimo A (1992) Mahogany conservation: status and policy initiatives. Environ Conserv 19:331–338
Rojas-Sandoval J, Acevedo-Rodríguez P (2015) Naturalization and invasion of alien plants in Puerto Rico and the Virgin Islands. Biol Invasions 17:149–163
Roth LC (1999) Anthropogenic change in subtropical dry forest during a century of settlement in Jaiqui Picado, Santiago Province, Dominican Republic. J Biogeogr 26:739–759
Sala OE, Chapin FS, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774
Sealey N (1994) Bahamian landscapes, an introduction to the geography of the Bahamas. Media Publishing, Nassau
Silvertown J (2004) Plant coexistence and the niche. Trends Ecol Evol 19:605–611
Smith IK, Vankat JL (1992) Dry evergreen forest (coppice) communities of North Andros Island, Bahamas. Bull Torrey Bot Club 119:181–191
Steadman DW, Franklin J (2015) Changes in an insular bird community since the late Pleistocene. J Biogeogr 42:426–438
Steadman DW, Montambault JR, Robinson SK, Oswalt SN, Brandeis TJ, Londono G, Reetz MJ, Schlesky WM, Wright NA, Martinez AE, Smith J (2009) Relative abundance, habitat and long-term population changes of wintering and resident landbirds on St. John, U.S. Virgin Islands. Wilson J Ornithol 121:41–53
Sullivan-Sealey K, Brunnick B, Harzen S, Luton C, Nero V, Flowers L (2002) An ecoregional plan for the Bahamian Archipelago. Taras Oceanographic Foundation, Jupiter, FL
R Development Core Team (2012) R: a language and environment for statistical computing. http://www.R-project.org. R Foundation for Statistical Computing, Vienna
ter Braak CJF (1987) The analysis of vegetation-environment relationships by canonical correspondence analysis. Vegetatio 69:69–77
Trejo-Torres JC, Ackerman JD (2002) Composition patterns of Caribbean limestone forests: are parsimony, classification, and ordination analyses congruent? Biotropica 34:502–515
Tylianakis JM, Didham RK, Bascompte J, Wardle DA (2008) Global change and species interactions in terrestrial ecosystems. Ecol Lett 11:1351–1363
USDA, NRCS (2014) The PLANTS Database. National plant data team, Greensboro, NC 27401-4901 USA. http://plants.usda.gov
Wunderle JM Jr, Waide RB (1993) Distribution of overwintering Nearctic migrants in the Bahamas and Greater Antilles. Condor 95:904–933
Acknowledgments
This research was sponsored by the U.S. National Science Foundation (Grant BCS-1118340 to JF and BCS-1118369 to DWS) and Arizona State University (JF). For permissions and cooperation on Abaco and Eleuthera, we thank The Bahamas National Trust, The National Museum of The Bahamas (Antiquities, Monuments and Museums Corporation), Friends of the Environment, the Leon Levy Native Plant Preserve, C. Adair, M. and N. Albury, E. Carey, M. Daniels, A. and S. Knowles, D. Knowles, P. Maillis, O. Patterson, K. Tinker, and K. Williams. B. Trapido-Lurie produced Fig. 1. The USDA Forest Service (USFS) FIA program for Puerto Rico and the U.S. Virgin Islands is jointly funded by USFS-Southern Research Station-FIA and the USFS International Institute of Tropical Forestry.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Lesley Rigg.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Franklin, J., Ripplinger, J., Freid, E.H. et al. Regional variation in Caribbean dry forest tree species composition. Plant Ecol 216, 873–886 (2015). https://doi.org/10.1007/s11258-015-0474-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-015-0474-8