Abstract
This study aimed to assess the effects of a 10-week multicomponent outdoor exercise program on hemodynamic, physical fitness and psychological variables in subjects with CVDs. Twenty participants (12 M and 8 F; age 68.5 ± 8.6 years; BMI 28.49 ± 4.98 kg/m2) suffering from CVDs participated in this randomized controlled study and were allocated into two groups: an experimental group (EG; 6 M, 5 F) who underwent a multicomponent training (i.e., cardiorespiratory, resistance, mobility and breathing exercises; 60’, 2d·wk-1), or a wait-list control group (CG; 6 M, 3 F) who did not engage in any PA. Hemodynamic parameters were assessed through resting heart rate (RHR) and peripheral blood pressure (P-SBP/P-DBP). Physical fitness was assessed via a 30″ chair stand test (30CST), timed up and go (TUG) test, handgrip strength (HGS) test, and 2’ step test (TMST). The health status and quality of life (QoL) were evaluated with short form-12 (SF-12) and World Health Organization QoL (WHOQoL-bref), respectively. After the intervention, EG showed significant reduction in hemodynamic parameters (RHR p < 0.001, ES = 0.98; P-SBP p < 0.001, ES = 0.53; P-DBP: p < 0.001, ES = 0.78), physical fitness (30CST p < 0.001, ES = 0.92; TUG p = 0.001, ES = 0.67; TMST p < 0.05, ES = 0.79) and physical component of SF-12 (PSC-12 p < 0.05, ES = 0.42) compared to CG; no significant changes were observed in HGS, QoL and mental health component of the SF-12 (p > 0.05). The findings showed significant improvements in hemodynamic parameters and physical fitness suggesting the effectiveness of the multicomponent outdoor exercise program in adults and elderly with stabilized CVDs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular diseases (CVDs) remain a significant global health concern, accounting for a substantial burden of morbidity and mortality. The prevalence of CVDs is consistently rising, and their impact on individuals' physical well-being and quality of life (QoL) is profound [1, 2]. Effective management of CVDs requires multifaceted interventions that address both the physiological and psychosocial dimensions of the disease [3].
Risk factors like obesity, high blood pressure, hypercholesterolemia, ageing, male sex and physical inactivity can contribute to CVDs [4, 5]. Physical activity (PA) has emerged as a crucial therapeutic strategy in the management and prevention of CVDs, contributing to improved cardiovascular function, physical fitness, and overall QoL [6]. Furthermore, PA has been shown to enhance cardiovascular fitness, reduce the risk of cardiovascular events, and improve various health markers in individuals with CVDs, independently of weight loss [7,8,9,10].
Exercise training, particularly aerobic and resistance exercises, has garnered attention as a non-pharmacological approach to managing CVDs. Studies have consistently demonstrated that aerobic training improves exercise tolerance, reduces resting heart rate (RHR), and enhances vascular function in individuals with CVDs [11,12,13,14]. Moreover, aerobic exercise positively influences lipid profiles, glucose metabolism, and body composition, contributing to the overall management of CVD risk factors. Resistance training can contribute to enhanced daily physical activities and reduced frailty, thereby supporting cardiovascular health [15,16,17]. Additionally, it may positively impact blood pressure regulation and endothelial function [18, 19].
The impaired blood flow and oxygen delivery associated with CVDs can impact the structural integrity and functional capacity of muscles, tendons, and ligaments that support joint mobility. Moreover, chronic inflammation characteristic of CVDs can promote joint inflammation, further compromising mobility [20]. Stretching exercises can reduce arterial stiffness and improve endothelial function [20,21,22,23].
The complexity of CDVs demands a holistic approach to exercise prescription. Multicomponent training protocols target multiple aspects of physical fitness, addressing not only cardiovascular endurance but also muscular strength, flexibility, and balance [24]. These components collectively can contribute to enhanced functional capacity and improve QoL [24]. However, the holistic impact of integrating these exercise modes into a unified multicomponent training protocol in CVDs field remains poorly explored.
While there is a growing body of literature examining the impact of indoor exercise interventions on CVDs management [25, 26], comparatively fewer studies have explored the potential of outdoor exercise programs in this context [27, 28]. Outdoor exercise provides exposure to natural elements, varied terrains, and a sense of novelty that could contribute to sustained engagement and potentially enhanced outcomes [29, 30].
Therefore, this study aimed to assess the effects of a structured multicomponent outdoor exercise program on the hemodynamic parameters, physical fitness, psychophysical health status and QoL of adults and elderly with stabilized CVDs. We hypothesized that the experimental group would experience reduction in hemodynamic parameters, improvement in cardiovascular fitness, muscular strength, mobility, psychophysical health status and overall QoL compared to the control group.
Materials and methods
Participants and study design
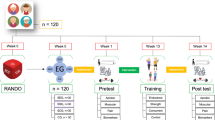
This study utilized a randomized experimental research design to compare the experimental and the waitlist control groups before the intervention and after 10 weeks. Eligible patients were randomly assigned to one of the two groups.
A waitlist control group consists of participants who do not receive the experimental treatment immediately but are scheduled to receive it after the active treatment group. This group serves two main purposes: it provides an untreated comparison for the active experimental group, helping researchers determine if the treatment had an effect by isolating the independent variable. It also offers the waitlisted participants the opportunity to receive the intervention later, making it an ethical alternative to a no-treatment control group in situations where denying treatment would be unethical.
Before entering the study, informed consent was obtained from each participant. The procedures followed were in accordance with the ethical standards of the Helsinki Declaration and approved by the Ethics Committee of Bari University (protocol code 0015637|16/02/23).
Participants were recruited through the ODV association “Amici di Cuore” based in Bari, Italy and a preliminary medical examination was performed before the start of the study (February 2023). After consent from the corresponding cardiologist, participants were considered eligible based on the following inclusion criteria: age between 45 and 80 years, presence of stabilized CVDs (cardiovascular conditions have reached a state of equilibrium where symptoms are managed and under control); sedentary lifestyle, subjects who have not followed the WHO guidelines for aerobic and resistance exercise in the last 3 months [35]. Additionally, participants were excluded based on the following criteria: presence of serious medical conditions (unstable coronary heart disease, decompensated heart failure, severe pulmonary hypertension) or acute onset that would prevent safe participation in physical activity according to American College of Sports Medicine (ACSM), American Heart Association (AHA) and European Society of Cardiology (ESC) guidelines [31,32,33,34]; smoking; anticipated absence of more than 1 week during the intervention period; lack of sports suitability confirmed by a cardiologist; presence of joint pain, dizziness, chest pain or angina during physical exercise; high peripheric blood pressure: PBP ≥ 160/100.
An a priori power analysis [36] with an assumed type I error of 0.05 and a type II error rate of 0.20 (80% statistical power) has calculated that 8 participants per group would be sufficient to observe medium “Time × Group” interaction effects. However, 20 participants (12 males and 8 females, 68.5 ± 8.6 years) among those that have been contacted (n = 28; 15 males, 13 females) met all the above-mentioned eligibility criteria. The participants were allocated into two groups: an experimental group (EG n = 11, 6 males and 5 females) who underwent progressive outdoor multicomponent training (60 min, 2d·wk-1) or a waitlist control group (CG n = 9, 6 males and 3 females) who did not engage in any structured physical activity during the intervention period. Among the CVDs diagnosed are included: hypertension (n = 14), valvular heart disease (n = 1), aortic valve dis-ease (n = 1), atrial fibrillation (n = 1) and previous myocardial infarction (n = 3). All participants were advised to maintain all their prescribed medications during the intervention. None of the participants followed a specific food plan. The study was carried out between the months of February and May 2023. All participants completed the study. Figure 1 shows the eligibility assessment of the participants.
Testing procedures
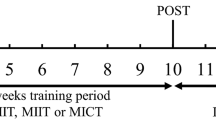
Data were collected and recorded at week 1 (Baseline) and after week 10 (Post-test), three days after the last session in the same place (indoor setting). First, the anthropometric measurements were collected. Body height (in cm to the nearest 0.1 cm) was measured using a SECA® stadiometer, and body mass (in kg to the nearest 0.1 kg) was measured using a SECA® digital scale (0–200 kg, accuracy of 0.1 kg). The subjects were barefooted and wore light clothing during the measurements. Body mass index (BMI) was calculated as body weight (kg) divided by the square of body height (m2).
Second, RHR and peripheral blood pressure (systolic blood pressure: P-SBP; diastolic blood pressure: P-DBP) were measured, by medical staff, using the Sphygmocor XCEL (AtCor Medical, Itasca, IL, USA) automated oscillometric device. After a 5-min rest, a blood pressure cuff was applied to the participant’s left arm, positioned over the brachial artery, while they were seated. The device took three consecutive measurements of brachial pressure, with a 2-min break between each reading. The aver-age of the measurements taken was adopted.
Finally, the following measures were collected: (1) Physical Fitness: 30-s chair stand (30CST) test, Timed Up and Go (TUG) Test, Handgrip Strength (HGS) test and 2-Minute Step Test (TMST); (2) Psychological: Short Form Survey (SF-12) and World Health Organization Quality of Life–Bref (WHOQoL-BREF). All participants were trained in an outdoor green space located inside a sporting club (Angiulli Gymnastics Club, Bari, Italy). One week before the pre-test, two familiarization sessions were held. Initial and final test measurements were made at the same time of day and under the same experimental and treatment conditions. All measurements were performed and supervised by the same Adapted Physical Activity (APA) Specialists, who were blinded to treatment allocations.
30-s chair stand (30CST) test
This test is one of the most important functional evaluation clinical tests because it measures lower body strength and relates it to the most demanding daily life activities (e.g., climbing stairs, getting out of a chair or bathtub or rising from a horizontal position) [37]. It is also able to assess functional fitness levels and the fatigue effect caused by the number of sit-to-stand repetitions (ICC = 0.95). It consists of standing up and sitting down from a chair as many times as possible (n) within 30 s. A standard chair (with a seat height of 42 cm) without armrests was used. Initially, the participants were seated on the chair with their back in an upright position. They were instructed to look straight for-ward and to rise after the “1, 2, 3, go” command at their preferred speed with their arms folded across their chest [38].
Timed up and go (TUG) test
This test is the one most used to assess the mobility of subjects. It assesses several aspects related to mobility such as static/dynamic balance and gait speed, along with lower limb strength. TUG results are predictors of several outcomes (i.e., falls, quality of life and difficulty in performing daily activities) [39,40,41]. Furthermore, it appears to be the best test of physical function in the prediction of cardiovascular disease in older adults [42] and the reliability of the test was found to be very high (ICC = 0.90) [43]. Subjects wear their regular footwear and can use a walking aid if needed (no one used it). Begin by having the patient sit back in a standard chair (height of 42 cm) with armrests and identify a line 3 m on the floor. Subjects were instructed to stand up from the chair, at the signal, and walk towards the line, at a self-selected speed, turn, walk towards the chair, and sit down again. At the go signal, timing starts and ends (s) when the subject sits back down [44].
Handgrip strength (HGS) test
The handgrip strength test is a practical and validated instrument for assessing the maximum voluntary strength of the extrinsic and intrinsic muscles of the hand. Its results are an indicator of clinical conditions such as sarcopenia and correlate with general muscle strength (ICC dominant = 0.97; ICC undominant = 0.98). It may be a predictor of the incidence of chronic diseases, independence in daily life and nutritional status [45]. Grip strength was measured with a mechanic Smedley hand dynamometer (GIMA, Milan, Italy). The participants were positioned sitting in a straight-backed chair with feet on the floor, shoulder adducted with 0 degrees flexion, elbow flexed to 90 degrees and forearm in a neutral position; participants were instructed to hold the dynamometer and squeeze it as hard as possible for 5 s. The measurement was performed three times with an interval of 30 s between measurements and 60 s before evaluating the other hand. The mean of the two measurements, expressed in kilograms (Kg), was used for the analysis.
2-min step test (TMST)
The TMST aims to assess subjective aerobic capacity, a fundamental component of physical fitness, which has been studied and utilized in different populations (e.g. hyper-tension, stroke, heart failure) [46]. The TMST is often used as an alternative to a 6-min walking test, we have used it due to limited space available (i.e., indoor corridor of the necessary length) (ICC = 0.90). Subjects standing near a wall measured the height of the iliac crest and the patella and placed a marker on the wall halfway between the two. At the starting signal, the subjects began stepping in place by raising their knees to the height of the mark on the wall, as many times as possible during the 2 min. The number of times the right knee reaches the mark represents the test score (n).
Short form survey (SF-12)
The SF-12 is a shortened version questionnaire of SF-36, consisting of 12 items assessing physical (PCS-12) and mental (MCS-12) health [47, 48]. Designed as a general measure of health, it can be used with the general population (Cronbach’s α = 0.91). It comprises eight domains such as physical function, role-physical, bodily pain, general health, vitality, social functioning, role-emotional and mental health. PCS and MCS were computed using the scores of 12 questions ranging from 0 to 100, where zero indicates the lowest level of health and 100 indicates the highest level of health.
World health organization quality of life–bref (WHOQoL-BREF)
The WHOQoL-BREF is a questionnaire, developed as a short version of the WHOQOL-100, available in multiple languages and in this study, the Italian version was used to assess the quality of life [49]. This self-administered questionnaire presents 26 items concerning the perception of individual health and well-being over the past 2 weeks. Scored in four domains: Domain 1: Physical health (Cronbach’s α = 0.80), Domain 2: Psychological well-being (Cronbach’s α = 0.75), Domain 3: Social relations (Cronbach’s α = 0.65) and Domain 4: Environment health (Cronbach’s α = 0.73) with all facet items scored as part of their hypothesized domain [50]. Zero points represent the worst possible state of health, while 100 points represent the best possible state of health regarding the respective domain.
Exercise intervention
Before starting the training session, blood pressure and RHR were measured. For patient safety, the exercise session was only performed if P-SBP was between 110 and 180 mmHg and/or P-DBP between 50 and 100 mmHg and, also, RHR between 50 and 100 bpm. Sessions were performed within two small groups (one of six and one of five participants). These groups were closely supervised by exercise professionals (i.e., APA specialists) to ensure the participants' safety, maintain the appropriate level of intensity, and correct exercise techniques. To monitor and adjust the training intensity (internal load) as the sessions progressed, the Borg Rating of Perceived Exertion (RPE) scale (6–20) [51] was employed at the end of each set of aerobic and resistance exercises, to adjust the load in Borg = 13 to 15 points [32], considering that the higher the number chosen, the more intense the exercise session was. The participants were prior familiarized with the scale. This approach evaluates an individual's overall perception of the training load, considering both central factors, like lung ventilation, and peripheral factors, involving muscles and joints.
The 10-week study period followed the initial data collection, with the EG performing an intervention program consisting of twice-per-week (Monday and Wednesday) exercise sessions lasting 60 min each, usually performed from 3:30 to 4:30 p.m. Every single exercise session included an initial phase of muscle activation through a 10-min warm-up (brisk walk) to increase heart rate, improve muscle blood flow, and prepare the main joints for the subsequent work phase, a 40-min main exercise period (aerobic exercise, mobility exercises and resistance training), and a 10-min cooldown period (breathing and stretching exercises).
During the main exercise period, cardiorespiratory training consisted of progressive aerobic exercises: controlled and rhythmic jumping jacks, step-ups on a sturdy platform (such as a low step or a stable surface), standing knee raises (alternating legs), brisk side steps or lateral leg raises. The exercises were performed at an intensity ensuring that the perceived exertion (RPE) stayed between 13 and 15 points on the Borg Scale (6–20). Finally, conclude with 3 min of light walking to facilitate recovery for the next phase of training. The main goal of this phase was to maintain a consistent exercise duration of ~ 15 min while gradually intensifying the exercise stimuli.
Mobility training consisted of specific exercises (thoracic extensions, cat to cows, overhead reach with stick and hips active internal rotation) targeting the main joint, per-formed maximally (1–3 sets) but avoiding pain. Duration was gradually increased from 30 to 60 s per repetition, repeating one to three times. Participants were provided with rest intervals of 30–60 s between sets and exercises.
Resistance training consisted of exercises targeting various muscle groups: quadriceps (seated leg extension with anklet weight/half squat with chair), biceps brachii (unilateral curl with dumbbell), shoulder (shoulder press with dumbbell), triceps brachii (French press with dumbbell), pectoralis major (dumbbell chest press/dumbbell flyes), latissimus dorsi (dumbbell rows). The resistance training program adhered to the principle of gradually progressive load. Initially, participants performed a set of 10–15 repetitions, which progressed to three sets of 10–15 repetitions. Adjustments to the load were made to ensure that the perceived exertion (RPE) stayed between 13 and 15 points on the Borg Scale (6–20). Throughout the protocol, participants were provided with rest intervals of 60–120 s between sets and exercises to promote recovery. To prevent premature muscle fatigue, the exercises were carried out using an alternating training method based on muscle groups (upper muscle exercises were performed on Monday, and lower muscle exercises were performed on Wednesday). To avoid breath holding and any compromise in circulatory the significance of correct breathing was underscored.
The Cool down period consisted of breathing and stretching exercises. Stretching was performed maximally on all major muscle groups (1–3 sets per muscle group) avoiding joint pain. Duration was gradual from 10 to 30 s per stretch, repeating one to three times for a total of 60 s per stretch.
Statistical analysis
All statistical analyses were conducted using the JASP software v. 0.17.2.1 [52]. The Shapiro–Wilk test was used to test the normality of all variables. Levene’s test was used to check the homogeneity of variances between groups. One-way ANOVA was used to compare groups at pre-test for anthropometric characteristics and all outcome measures. A two-way ANOVA (group (intervention/control) × time (pre/post-intervention)) with repeated measures was conducted to examine the effects of the intervention on all dependent variables. When ‘Time x Group’ interactions reached the level of significance, group-specific post hoc tests (i.e., Tukey’s test) were conducted to identify the significant comparisons. Partial eta squared (η2p) was used to estimate the magnitude of the difference with-in each group and interpreted using the following criteria: small (η2p < 0.06), medium (0.06 ≤ η2p < 0.14), and large (η2p ≥ 0.14) effect size (ES). ES for the post hoc pairwise comparisons was determined by Cohen’s d, calculated as post-training mean minus pre-training mean divided by pooled SD before and after training and interpreted as small (0.20 ≤ d < 0.50), moderate (0.50 ≤ d < 0.79) and large (d ≥ 0.80) ES [53]. An alpha level of p ≤ 0.05 was considered statistically significant.
Results
All the EG participants completed the intervention, and no adverse effects were detected over the ten weeks. All patients attended at least 85% of training sessions. Table 1 shows the descriptive data of the study participants.
No statistically significant difference was found between the groups at pre-test regarding age, anthropometric characteristics, and all outcome measures (p > 0.05).
Hemodynamic parameters
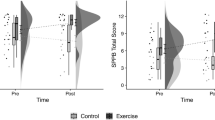
Two-way ANOVA with repeated measures found significant ‘Time x Group’ interaction effects in: RHR (F = 23.189, p < 0.001, η2p = 0.563, large ES), P-SBP (F = 18.856, p < 0.001, η2p = 0.512, large ES), P-DBP (F = 27.950, p < 0.001, η2p = 0.608, large ES).
In the EG, the post hoc analyses revealed a significant improvement in the score from pre- to post-intervention for RHR (t = 6.287, p < 0.001, d = 0.981, large ES), P-SBP (t = 5.798, p < 0.001, d = 0.526, moderate ES), P-DBP (t = 7.023, p < 0.001, d = 0.779, moderate ES).
Physical fitness parameters
Two-way ANOVA with repeated measures found significant ‘Time x Group’ interaction effects in: 30CST (F = 18.907, p < 0.001, η2p = 0.512, large ES), TUG (F = 14.468, p = 0.001, η2p = 0.446, large ES), MST (F = 7.522, p < 0.05, η2p = 0.295, large ES).
In the EG, the post hoc analyses revealed a significant improvement in the score from pre- to post-intervention for 30CST (t = −4.209, p < 0.01, d = 0.921, large ES), TUG (t = 3.934, p < 0.05, d = 0.667, moderate ES), TMST (t = -3.078, p < 0.05, d = 0.793, moderate ES).
No significant interaction was observed in the HGS (p > 0.05).
Psychological parameters
Two-way ANOVA with repeated measures found significant ‘Time x Group’ interaction effects in physical component of SF-12 (PSC-12 F = 5.543, p < 0.05, η2p = 0.235, large ES).
In the EG, the post hoc analyses revealed a significant improvement in the score from pre- to post-intervention for PCS-12 (t = −3.004, p < 0.05, d = 0.417, small ES).
No significant interaction was observed in the QoL and mental health component of the SF-12 (p > 0.05).
No significant changes were found in the CG (p > 0.05). Pre- and post-intervention outcomes for all the variables considered and the statistical analysis results are shown in Table 2.
Discussion
This study aimed to assess the effects of a multicomponent outdoor exercise program on the hemodynamic parameters, physical fitness, psychophysical health status and QoL of adults and elderly with stabilized CVDs. The study revealed significant improvements in peripheral blood pressure (P-SBP and P-DBP) supporting the previously reported results of other combined training protocols in hypertensive subjects [18, 54]. Whereas Schroeder et al. [55] observed a reduction in P-DBP, but not in P-SBP, after 8 weeks of combined exercise training in previously sedentary adults with elevated blood pressure/hypertension and overweight/obese. The authors point to the short duration of the intervention as a possible cause of this contradiction. Many works observe a reduction in blood pressure after interventions lasting at least 12 weeks [56, 57]. Our results show that a 10-week outdoor multicomponent training protocol may be sufficient to improve blood pressure in adults and elderly with stabilized CVDs. Although some studies report an association between sunlight exposure, stimulating vitamin D production, and reduced blood pressure in vitamin D-insufficiency subjects [58, 59], more recent evidence does not support these observations, finding any such improvement following vitamin D supplementation [60, 61]. However, exposure to solar ultraviolet radiation could affect blood pressure, independent of vitamin D production, by increasing the availability of nitric oxide, a vasodilator, leading to blood pressure reduction [62, 63]. Consequently, the outdoor environment may have contributed to this outcome. Similarly, in the EG, we found a significant reduction, in RHR that appears to be inversely correlated with life expectancy and positively correlated with cardiovascular and all causes of mortality [64]. These results are in line with previous studies of healthy older adults [14] or those with medical conditions [65].
Furthermore, the results of our work show improvement in several fitness parameters among EG participants. Loss of muscle mass and strength was associated with increased arterial stiffness and subsequent higher blood pressure [66, 67]. Significant improvement in muscle strength was observed in EG in our study, while, although the increase in muscle mass could not be measured, the lack of change in BMI values could also be a consequence of a reduction in fat mass with a concomitant increase in lean mass [68,69,70]. Specifically, the improvement noted in lower limb strength could be also important in decreasing the risk of falls and muscle injuries and better performance in daily activities, in adults and the elderly [71]. These results are in line with previous research [15, 72].
Reduced functional mobility has been associated with the occurrence of future cardiovascular and all causes of mortality [42]. The TUG is the validated test most used to assess physical mobility, gait speed and balance, used as a predictor of falls and evaluation of dynamic balance, specifically in older adults and the elderly [73, 74]. Furthermore, the TUG test has been associated with future cardiovascular mortality in women and in subjects without obesity, diabetes, or cigarette smoking [42]. A recent meta-analysis showed that multicomponent training could be an optimal strategy to improve functional mobility, even more efficient than strength training only, in older adults [21]. Similarly, our results showed a significant decrease in time in the TUG test in the EG after 10 weeks of multicomponent training.
Contrary to what has been observed in other studies [8], our study did not find statistically significant pre/post-changes in handgrip strength values in the EG compared to CG. This could be due to the shorter duration and frequency of our protocol (2d·wk-1 for 10 weeks), compared with previous studies such as that of Kambic et al. [8] (3d·wk-1 for 12 weeks). However, it should be noted that a meta-analysis aiming to evaluate the actual transfer effect of different types of exercise on handgrip strength showed only small effects, in healthy older adults [75].
Notably, we observed a significant improvement in aerobic capacity, which is important for individuals with CVDs contributing to better disease management. This finding supports previous research [8, 18, 76], which highlights the effectiveness of combined training in improving cardiovascular function in this population. Specifically, considering that aerobic training is the most common exercise for aerobic capacity and cardiovascular improvements [77, 78], a good portion of these previous work’s training volume was composed of aerobic training, from 25 [18] to 45 [8] or 50 min [76]. In our protocol, the time of each component of the main exercise period was roughly equally distributed (~ 15') and despite the shorter duration of the aerobic component, compared to previous studies, our work showed similar results.
Despite improvements in parameters related to physical performance found in our study, the QoL outcomes, contrary to what we expected, shown no significant changes in EG, but just an improvement in the physical component (PCS) of the SF-12 used to assess perceived physical and mental health status, and thus Health-related QoL. While the observed result assumes low practical relevance (small ES), this is consistent with the improvements seen at the physical level.
Although several studies have suggested that physical activity in outdoor settings, such as green and blue spaces, can improve psychophysiological well-being [29, 79], and the high adherence to the protocol could also be a consequence of the outdoor environment, increasing the sense of novelty and reducing friction to participation, our study only assessed an outdoor intervention group and did not compare it with an indoor group. Future studies comparing the same intervention protocol carried out in two different settings, outdoor and indoor, would be appropriate in order to draw objective and nonspeculative conclusions.,.
To the best of our knowledge, this is the first study aiming to assess the effects of outdoor multicomponent training on the hemodynamic parameters, physical fitness, psychophysical health status and QoL of adults and elderly with stabilized CVDs. However, this study has some limitations that should be considered. The relatively small sample size might limit the broader applicability of the results. Without long-term follow-up evaluation, it cannot be determined how sustainable the observed improvements are over time. It is also important to note that this study did not investigate the influence of nutrition, which is a significant factor in managing CVDs.
Conclusions
The findings suggest that a 10-week multicomponent outdoor exercise protocol is effective in improving hemodynamic parameters and physical fitness in adults and elderly with stabilized CVDs, without adverse effects.
However, although the exercise intervention led to significant physical and physiological improvements, handgrip strength and QoL were not improved suggesting that these factors may take longer to reach the statistical significance. Only the physical component (PCS) of the SF-12 has shown significant improvement, albeit with low practical relevance (small ES). Further randomized controlled trials with a larger sample size are needed to strengthen these findings.
Data availability
No datasets were generated or analysed during the current study.
References
Saglietto A, Manfredi R, Elia E, D’Ascenzo F, De Ferrari GM, Biondi-Zoccai G, Munzel T (2021) Cardiovascular disease burden: Italian and global perspectives. Minerva Cardiol Angiol. https://doi.org/10.23736/S2724-5683.21.05538-9
Townsend N, Kazakiewicz D, Lucy Wright F, Timmis A, Huculeci R, Torbica A, Gale CP, Achenbach S, Weidinger F, Vardas P (2022) Epidemiology of cardiovascular disease in Europe. Nat Rev Cardiol 19:133–143. https://doi.org/10.1038/s41569-021-00607-3
Adams ML, Grandpre J, Katz DL, Shenson D (2020) Cognitive impairment and cardiovascular disease: a comparison of risk factors, disability, quality of life, and access to health care. Public Health Rep 135:132–140. https://doi.org/10.1177/0033354919893030
Kraus WE, Powell KE, Haskell WL, Janz KF, Campbell WW, Jakicic JM, Troiano RP, Sprow K, Torres A, Piercy KL (2019) Physical activity, all-cause and cardiovascular mortality, and cardiovascular disease. Med Sci Sports Exerc 51:1270–1281. https://doi.org/10.1249/MSS.0000000000001939
Tian D, Meng J (2019) Exercise for prevention and relief of cardiovascular disease: prognoses, mechanisms, and approaches. Oxid Med Cell Longev 2019:1–11. https://doi.org/10.1155/2019/3756750
Stewart J, Manmathan G, Wilkinson P (2017) Primary prevention of cardiovascular disease: a review of contemporary guidance and literature. JRSM Cardiovasc Dis 6:204800401668721. https://doi.org/10.1177/2048004016687211
Alves AJ, Viana JL, Cavalcante SL, Oliveira NL, Duarte JA, Mota J, Oliveira J, Ribeiro F (2016) Physical activity in primary and secondary prevention of cardiovascular disease: overview updated. WJC 8:575. https://doi.org/10.4330/wjc.v8.i10.575
Kambic T, Šarabon N, Lainscak M, Hadžić V (2022) Combined resistance training with aerobic training improves physical performance in patients with coronary artery disease: a secondary analysis of a randomized controlled clinical trial. Front Cardiovasc Med 9:909385. https://doi.org/10.3389/fcvm.2022.909385
Nasi M, Patrizi G, Pizzi C, Landolfo M, Boriani G, Dei Cas A, Cicero AFG, Fogacci F, Rapezzi C, Sisca G et al (2019) The role of physical activity in individuals with cardiovascular risk factors: an opinion paper from italian society of cardiology-emilia romagna-marche and SIC-sport. J Cardiovasc Med 20(10):631–639. https://doi.org/10.2459/JCM.0000000000000855
Soares-Miranda L, Siscovick DS, Psaty BM, Longstreth WT, Mozaffarian D (2016) Physical activity and risk of coronary heart disease and stroke in older adults: the cardiovascular health study. Circulation 133:147–155. https://doi.org/10.1161/CIRCULATIONAHA.115.018323
Ashor AW, Lara J, Siervo M, Celis-Morales C, Mathers JC (2014) Effects of exercise modalities on arterial stiffness and wave reflection: a systematic review and meta-analysis of randomized controlled trials. PLoS ONE 9:e110034. https://doi.org/10.1371/journal.pone.0110034
Brito LC, Peçanha T, Fecchio RY, Pio-Abreu A, Silva G, Mion-Junior D, Halliwill JR, Forjaz CLM (2021) Comparison of morning versus evening aerobic-exercise training on heart rate recovery in treated hypertensive men: a randomized controlled trial. Blood Press Monit 26:388–392. https://doi.org/10.1097/MBP.0000000000000545
Pedralli ML, Eibel B, Waclawovsky G, Schaun MI, Nisa-Castro-Neto W, Umpierre D, Pescatello LS, Tanaka H, Lehnen AM (2018) Effects of exercise training on endothelial function in individuals with hypertension: a systematic review with meta-analysis. J Am Soc Hypertens 12:e65–e75. https://doi.org/10.1016/j.jash.2018.09.009
Reimers A, Knapp G, Reimers C-D (2018) Effects of exercise on the resting heart rate: a systematic review and meta-analysis of interventional studies. JCM 7:503. https://doi.org/10.3390/jcm7120503
Islami F, Saghebjoo M, Kazemi T, Hedayati M (2021) Gym and home-based combined training in men with primary hypertension: are they equally effective on functional fitness profile, body composition components, and biochemical parameters of hypertension? Clin Exp Hypertens 43:758–771. https://doi.org/10.1080/10641963.2021.1960365
Katz A (2019) Brief periods of resistance training enhance insulin sensitivity in humans. Exp Physiol 104:458–459. https://doi.org/10.1113/EP087607
Paquin J, Lagacé J-C, Brochu M, Dionne IJ (2021) Exercising for insulin sensitivity – is there a mechanistic relationship with quantitative changes in skeletal muscle mass? Front Physiol 12:656909. https://doi.org/10.3389/fphys.2021.656909
Oliveira SND, Moro ARP, Domingues WJR, Bezerra EDS (2018) <b>Effects of concurrent training with self-selected intensity on the physical fitness of hypertensive individuals. Acta Sci Health Sci 40:35739. https://doi.org/10.4025/actascihealthsci.v40i1.35739
Trevizani GA, Seixas MB, Benchimol-Barbosa PR, Vianna JM, Da Silva LP, Nadal J (2018) Effect of resistance training on blood pressure and autonomic responses in treated hypertensives. J Strength Cond Res 32:1462–1470. https://doi.org/10.1519/JSC.0000000000001995
Kato M, Nihei Green F, Hotta K, Tsukamoto T, Kurita Y, Kubo A, Takagi H (2020) The efficacy of stretching exercises on arterial stiffness in middle-aged and older adults: a meta-analysis of randomized and non-randomized controlled trials. IJERPH 17:5643. https://doi.org/10.3390/ijerph17165643
Lemos ECWM, Guadagnin EC, Mota CB (2020) Influence of strength training and multicomponent training on the functionality of older adults: systematic review and meta-analysis. Rev bras cineantropom desempenho hum 22:e60707. https://doi.org/10.1590/1980-0037.2020v22e6070
Nishiwaki M, Yonemura H, Kurobe K, Matsumoto N (2015) Four weeks of regular static stretching reduces arterial stiffness in middle-aged men. Springerplus 4:555. https://doi.org/10.1186/s40064-015-1337-4
Suwa M, Imoto T, Kida A, Yokochi T, Iwase M, Kozawa K (2018) Association of body flexibility and carotid atherosclerosis in Japanese middle-aged men: a cross-sectional study. BMJ Open 8:e019370. https://doi.org/10.1136/bmjopen-2017-019370
Bouaziz W, Lang PO, Schmitt E, Kaltenbach G, Geny B, Vogel T (2016) Health benefits of multicomponent training programmes in seniors: a systematic review. Int J Clin Pract 70:520–536. https://doi.org/10.1111/ijcp.12822
Fiuza-Luces C, Santos-Lozano A, Joyner M, Carrera-Bastos P, Picazo O, Zugaza JL, Izquierdo M, Ruilope LM, Lucia A (2018) Exercise benefits in cardiovascular disease: beyond attenuation of traditional risk factors. Nat Rev Cardiol 15:731–743. https://doi.org/10.1038/s41569-018-0065-1
Gronek P, Wielinski D, Cyganski P, Rynkiewicz A, Zając A, Maszczyk A, Gronek J, Podstawski R, Czarny W, Balko S et al (2020) A review of exercise as medicine in cardiovascular disease: pathology and mechanism. Aging Dis 11:327. https://doi.org/10.14336/AD.2019.0516
Aguiar LT, Nadeau S, Britto RR, Teixeira-Salmela LF, Martins JC, Samora GAR, Da Silva Júnior JA, Faria CDCDM (2020) Effects of aerobic training on physical activity in people with stroke: a randomized controlled trial. NRE 46(3):391–401. https://doi.org/10.3233/NRE-193013
Nagyova I, Jendrichovsky M, Kucinsky R, Lachytova M, Rus V (2020) Effects of nordic walking on cardiovascular performance and quality of life in coronary artery disease. Eur J Phys Rehabil Med. https://doi.org/10.23736/S1973-9087.20.06120-1
Marini S, Mauro M, Grigoletto A, Toselli S, Maietta Latessa P (2022) The effect of physical activity interventions carried out in outdoor natural blue and green spaces on health outcomes: a systematic review. IJERPH 19:12482. https://doi.org/10.3390/ijerph191912482
Thompson Coon J, Boddy K, Stein K, Whear R, Barton J, Depledge MH (2011) Does participating in physical activity in outdoor natural environments have a greater effect on physical and mental wellbeing than physical activity indoors? A Syst Rev Environ Sci Technol 45:1761–1772. https://doi.org/10.1021/es102947t
Billinger SA, Arena R, Bernhardt J, Eng JJ, Franklin BA, Johnson CM, MacKay-Lyons M, Macko RF, Mead GE, Roth EJ et al (2014) Physical activity and exercise recommendations for stroke survivors: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 45:2532–2553. https://doi.org/10.1161/STR.0000000000000022
Gary L (2021) American College of Sports Medicine (ACSM) ACSM’s Guidelines for Exercise Testing and Prescription; 10th ed. Wolters Kluwer Health: USA, 1 ISBN 978–1–4963–3906–5
Pelliccia A, Sharma S, Gati S, Bäck M, Börjesson M, Caselli S, Collet J-P, Corrado D, Drezner JA, Halle M et al (2021) 2020 ESC guidelines on sports cardiology and exercise in patients with cardiovascular disease. Eur Heart J 42:17–96. https://doi.org/10.1093/eurheartj/ehaa605
Williams MA, Haskell WL, Ades PA, Amsterdam EA, Bittner V, Franklin BA, Gulanick M, Laing ST, Stewart KJ (2007) Resistance exercise in individuals with and without cardiovascular disease: 2007 update: a scientific statement from the american heart association council on clinical cardiology and council on nutrition, physical activity, and metabolism. Circulation 116:572–584. https://doi.org/10.1161/CIRCULATIONAHA.107.185214
Bull FC, Al-Ansari SS, Biddle S, Borodulin K, Buman MP, Cardon G, Carty C, Chaput J-P, Chastin S, Chou R et al (2020) World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br J Sports Med 54:1451–1462. https://doi.org/10.1136/bjsports-2020-102955
Kang H (2021) Sample size determination and power analysis using the G*Power software. J Educ Eval Health Prof 18:17. https://doi.org/10.3352/jeehp.2021.18.17
Macfarlane DJ, Chou KL, Cheng YH, Chi I (2006) Validity and normative data for thirty-second chair stand test in elderly community-dwelling Hong Kong Chinese. Am J Hum Biol 18:418–421. https://doi.org/10.1002/ajhb.20503
Rikli RE, Jones CJ (2013) Development and validation of criterion-referenced clinically relevant fitness standards for maintaining physical independence in later years. Gerontologist 53:255–267. https://doi.org/10.1093/geront/gns071
Ekström H, Dahlin-Ivanoff S, Elmståhl S (2011) Effects of walking speed and results of timed get-up-and-go tests on quality of life and social participation in elderly individuals with a history of osteoporosis-related fractures. J Aging Health 23:1379–1399. https://doi.org/10.1177/0898264311418504
Kojima G, Masud T, Kendrick D, Morris R, Gawler S, Treml J, Iliffe S (2015) Does the timed up and go test predict future falls among british community-dwelling older people? Prospective cohort study nested within a randomised controlled trial. BMC Geriatr 15:38. https://doi.org/10.1186/s12877-015-0039-7
Wennie Huang W-N, Perera S, VanSwearingen J, Studenski S (2010) Performance measures predict onset of activity of daily living difficulty in community-dwelling older adults: performance measures predict onset of adl difficulty. J Am Geriatr Soc 58:844–852. https://doi.org/10.1111/j.1532-5415.2010.02820.x
Son KY, Shin DW, Lee JE, Kim SH, Yun JM, Cho B (2020) Association of timed up and go test outcomes with future incidence of cardiovascular disease and mortality in adults aged 66 years: korean national representative longitudinal study over 5.7 years. BMC Geriatr. https://doi.org/10.1186/s12877-020-01509-8
De Buyser SL, Petrovic M, Taes YE, Toye KRC, Kaufman J-M, Goemaere S (2013) Physical function measurements predict mortality in ambulatory older men. Eur J Clin Invest 43:379–386. https://doi.org/10.1111/eci.12056
Steffen TM, Hacker TA, Mollinger L (2002) Age- and gender-related test performance in community-dwelling elderly people: six-minute walk test, berg balance scale, timed up & go test, and gait speeds. Phys Ther 82:128–137. https://doi.org/10.1093/ptj/82.2.128
Lee SY (2021) Handgrip strength: an irreplaceable indicator of muscle function. Ann Rehabil Med 45:167–169. https://doi.org/10.5535/arm.21106
Bohannon RW, Crouch RH (2019) Two-minute step test of exercise capacity: systematic review of procedures, performance, and clinimetric properties. J Geriat Phys Ther 42:105–112. https://doi.org/10.1519/JPT.0000000000000164
Kodraliu P, Mosconi N, Groth G, Carmosino G (2001) Subjective health status assessment: evaluation of the Italian version of the SF-12 health survey results from the MiOS project. J Epidemiol Biostat. https://doi.org/10.1080/135952201317080715
Ware JE, Kosinski M, Keller SD (1996) A 12-item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med Care 34:220–233. https://doi.org/10.1097/00005650-199603000-00003
Girolamo GD, Rucci P, Scocco P, Becchi A, Coppa F, D’Addario A, Daru E, Leo DD, Galassi L, Mangelli L et al (2000) Quality of life assessment: validation of the Italian version of the WHOQOL-brief. Epidemiol Psichiatr Soc 9:45–55. https://doi.org/10.1017/S1121189X00007740
Skevington SM, Lotfy M, O’Connell KA (2004) The World Health Organization’s WHOQOL-BREF quality of life assessment: psychometric properties and results of the International field trial. A report from the WHOQOL group. Qual Life Res 13(2):299–310. https://doi.org/10.1023/B:QURE.0000018486.91360.00
Borg GA (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14:377–381
JASP Team JASP (Version 0.17.3) (2023)
Cohen J (1992) A power primer. Psychol Bulletin 112:155–159. https://doi.org/10.1037/0033-2909.112.1.155
Lima LG, Bonardi JTM, Campos GO, Bertani RF, Scher LML, Moriguti JC, Ferriolli E, Lima NKC (2017) Combined aerobic and resistance training: are there additional benefits for older hypertensive adults? Clinics 72:363–369. https://doi.org/10.6061/clinics/2017(06)06
Schroeder EC, Franke WD, Sharp RL, Lee D (2019) Comparative effectiveness of aerobic, resistance, and combined training on cardiovascular disease risk factors: a randomized controlled trial. PLoS ONE 14:e0210292. https://doi.org/10.1371/journal.pone.0210292
Cornelissen VA, Fagard RH, Coeckelberghs E, Vanhees L (2011) Impact of resistance training on blood pressure and other cardiovascular risk factors: a meta-analysis of randomized. Controlled Trials Hypertens 58:950–958. https://doi.org/10.1161/HYPERTENSIONAHA.111.177071
Sousa N, Mendes R, Abrantes C, Sampaio J, Oliveira J (2013) A randomized 9-month study of blood pressure and body fat responses to aerobic training versus combined aerobic and resistance training in older men. Exp Gerontol 48:727–733. https://doi.org/10.1016/j.exger.2013.04.008
Machado CDS, Venancio VP, Aissa AF, Hernandes LC, Mello MBD, Lama JECD, Marzocchi-Machado CM, Bianchi MLP, Antunes LMG (2016) Vitamin D3 deficiency increases DNA damage and the oxidative burst of neutrophils in a hypertensive rat model. Mutat Res/Genetic Toxicol Environ Mutagen 798–799:19–26. https://doi.org/10.1016/j.mrgentox.2016.01.005
Martins D, Wolf M, Pan D, Zadshir A, Tareen N, Thadhani R, Felsenfeld A, Levine B, Mehrotra R, Norris K (2007) Prevalence of cardiovascular risk factors and the serum levels of 25-Hydroxyvitamin D in the United States: data from the third National health and nutrition examination survey. Arch Intern Med 167:1159. https://doi.org/10.1001/archinte.167.11.1159
Pilz S, Gaksch M, Kienreich K, Grübler M, Verheyen N, Fahrleitner-Pammer A, Treiber G, Drechsler C, Hartaigh BÓ, Obermayer-Pietsch B et al (2015) Effects of vitamin D on blood pressure and cardiovascular risk factors: a randomized controlled trial. Hypertension 65(6):1195–1201. https://doi.org/10.1161/HYPERTENSIONAHA.115.05319
Zhang D, Cheng C, Wang Y, Sun H, Yu S, Xue Y, Liu Y, Li W, Li X (2020) Effect of vitamin D on blood pressure and hypertension in the general population: an update meta-analysis of cohort studies and randomized controlled trials. Prev Chronic Dis 17:190307. https://doi.org/10.5888/pcd17.190307
Weller RB (2016) Sunlight has cardiovascular benefits independently of vitamin D. Blood Purif 41:130–134. https://doi.org/10.1159/000441266
Weller RB, Wang Y, He J, Maddux FW, Usvyat L, Zhang H, Feelisch M, Kotanko P (2020) Does incident solar ultraviolet radiation lower blood pressure? JAHA 9:e013837. https://doi.org/10.1161/JAHA.119.013837
Aune D, Sen A, ó’Hartaigh B, Janszky I, Romundstad PR, Tonstad S, Vatten LJ (2017) Resting heart rate and the risk of cardiovascular disease, total cancer, and all-cause mortality–a systematic review and dose–response meta-analysis of prospective studies. Nutr Metab Cardiovasc Dis 27(6):504–517. https://doi.org/10.1016/j.numecd.2017.04.004
Huang G, Shi X, Davis-Brezette JA, Osness WH (2005) Resting heart rate changes after endurance training in older adults: a meta-analysis. Med Sci Sports Exerc 37:1381–1386. https://doi.org/10.1249/01.mss.0000174899.35392.0c
Son W-M, Sung K-D, Bharath LP, Choi K-J, Park S-Y (2017) Combined exercise training reduces blood pressure, arterial stiffness, and insulin resistance in obese prehypertensive adolescent girls. Clin Exp Hypertens 39:546–552. https://doi.org/10.1080/10641963.2017.1288742
Zhang Y, Miyai N, Abe K, Utsumi M, Uematsu Y, Terada K, Nakatani T, Takeshita T, Arita M (2021) Muscle mass reduction, low muscle strength, and their combination are associated with arterial stiffness in community-dwelling elderly population: the wakayama study. J Hum Hypertens 35:446–454. https://doi.org/10.1038/s41371-020-0355-z
Barakat C, Pearson J, Escalante G, Campbell B, De Souza EO (2020) Body recomposition: can trained individuals build muscle and lose fat at the same time? Strength Cond J 42:7–21. https://doi.org/10.1519/SSC.0000000000000584
Lee S, Kuk JL (2013) Changes in fat and skeletal muscle with exercise training in obese adolescents: comparison of whole-body mri and dual energy X-ray absorptiometry: changes in adipose tissue and skeletal muscle with exercise. Obesity 21:2063–2071. https://doi.org/10.1002/oby.20448
Sabag A, Najafi A, Michael S, Esgin T, Halaki M, Hackett D (2018) The compatibility of concurrent high intensity interval training and resistance training for muscular strength and hypertrophy: a systematic review and meta-analysis. J Sports Sci 36:2472–2483. https://doi.org/10.1080/02640414.2018.1464636
Pedralli ML, Marschner RA, Kollet DP, Neto SG, Eibel B, Tanaka H, Lehnen AM (2020) Different exercise training modalities produce similar endothelial function improvements in individuals with prehypertension or hypertension: a randomized clinical trial. Sci Rep 10:7628. https://doi.org/10.1038/s41598-020-64365-x
Figueroa A, Park SY, Seo DY, Sanchez-Gonzalez MA, Baek YH (2011) Combined resistance and endurance exercise training improves arterial stiffness, blood pressure, and muscle strength in postmenopausal women. Menopause 18:980–984. https://doi.org/10.1097/gme.0b013e3182135442
Nightingale CJ, Mitchell SN, Butterfield SA (2019) Validation of the timed up and go test for assessing balance variables in adults aged 65 and older. J Aging Phys Act 27:230–233. https://doi.org/10.1123/japa.2018-0049
Northgraves MJ, Hayes SC, Marshall P, Madden LA, Vince RV (2016) The test-retest reliability of four functional mobility tests in apparently healthy adults. IES 24:171–179. https://doi.org/10.3233/IES-160614
Labott BK, Bucht H, Morat M, Morat T, Donath L (2019) Effects of exercise training on handgrip strength in older adults: a meta-analytical review. Gerontology 65:686–698. https://doi.org/10.1159/000501203
Sardeli AV, Gáspari AF, Dos Santos WM, De Araujo AA, De Angelis K, Mariano LO, Cavaglieri CR, Fernhall B, Chacon-Mikahil MPT (2022) Comprehensive time-course effects of combined training on hypertensive older adults: a randomized control trial. IJERPH 19:11042. https://doi.org/10.3390/ijerph191711042
Sardeli AV, Gáspari AF, Chacon-Mikahil MP (2018) Acute, short-, and long-term effects of different types of exercise in central arterial stiffness: a systematic review and meta-analysis. J Sports Med Phys Fitness. https://doi.org/10.23736/S0022-4707.17.07486-2
Sardeli AV, Griffith GJ, Dos Santos MVMA, Ito MSR, Nadruz W, Chacon-Mikahil MPT (2020) Do baseline blood pressure and type of exercise influence level of reduction induced by training in hypertensive older adults? A meta-analysis of controlled trials. Exp Gerontol 140:111052. https://doi.org/10.1016/j.exger.2020.111052
Song C, Ikei H, Igarashi M, Takagaki M, Miyazaki Y (2015) Physiological and psychological effects of a walk in urban parks in fall. IJERPH 12:14216–14228. https://doi.org/10.3390/ijerph121114216
Funding
Open access funding provided by Università degli Studi di Bari Aldo Moro within the CRUI-CARE Agreement. Open access funding provided by Università degli Studi di Bari Aldo Moro within the CRUI-CARE Agreement. This research received no external funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, L.P. and G.G.; methodology, L.P, G.G. and S.C.; software, S.M.; validation, G.G., M.M.C., and S.M.; formal analysis, S.M. and M.M.C.; investigation, L.P. and S.M; resources, S.C., S.M and F.F.; data curation, G.G., L.P. and S.M.; writing—original draft preparation, L.P. and G.G.; writing—review and editing, L.P., G.G. and F.F.; visualization, S.M. and M.M.C.; supervision, S.C., G.G. and F.F. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Ethical approval
The study was conducted in accordance with the Declara-tion of Helsinki and approved by the Ethics Committee of Bari University (protocol code 0015637|16/02/23).
Informed consent
Informed consent was obtained from all subjects involved in the study.
I also declare that the Informed Consent of the subjects participating in this study was obtained following the procedures established by the Ethics Committee no. 7595 of 03/28/2023 (Ethical Committee of the University Polyclinic – Policlinico Bari).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Poli, L., Mazić, S., Ciccone, M.M. et al. A 10-week multicomponent outdoor exercise program improves hemodynamic parameters and physical fitness in cardiovascular disease adult and elderly patients. Sport Sci Health (2024). https://doi.org/10.1007/s11332-024-01251-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11332-024-01251-3