Abstract
Numerous studies have demonstrated the occurrence of pharmaceuticals in the natural environment, raising concerns about their impact on non-target organisms or human health. One region where little is known about the exposure and effects of pharmaceuticals in the environment is Iraq. Due to the high number of pharmaceuticals used by the public health sector in Iraq (hospitals and care centres) and distributed over the counter, there is a need for a systematic approach for identifying substances that should be monitored in the environment in Iraq and assessed in terms of environmental risk. In this study, a risk-based prioritization approach was applied to 99 of the most dispensed pharmaceuticals in three Iraqi cities, Baghdad, Mosul and Basrah. Initially, information on the amounts of pharmaceuticals used in Iraq was obtained. The top used medicines were found to be paracetamol, amoxicillin and metformin with total annual consumption exceeding 1000 tonnes per year. Predicted environmental concentrations (PECs) and predicted no-effect concentrations (PNECs), derived from ecotoxicological end-points and effects related to the therapeutic mode of action, were then used to rank the pharmaceuticals in terms of risks to different environmental compartments. Active pharmaceutical ingredients used as antibiotics, antidepressants and analgesics were identified as the highest priority in surface water, sediment and the terrestrial environment. Antibiotics were also prioritized according to their susceptibility to kill or inhibit the growth of bacteria or to accelerate the evolution and dissemination of antibiotic-resistant genes in water. Future work will focus on understanding the occurrence, fate and effects of some of highly prioritized substances in the environment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
It is estimated that more than 1500 active pharmaceutical ingredients (APIs) are currently in use. Following use, these compounds can be emitted into the natural environment e.g. via wastewater collection and treatment networks (Boxall et al. 2012; Ginebreda et al. 2010). The ongoing use of many of these APIs by society means that the active substances and their major metabolites will occur in the environment continuously (Monteiro and Boxall 2010).
For most pharmaceuticals in use, the evidence that they have deleterious effects on the natural environment is still limited and our knowledge of the fate of these pharmaceuticals in the environment is still deficient (Roos et al. 2012). This is partly due to the fact that the number of APIs in use is large and that experimental data on the environmental levels, fate and effects are available for only a small proportion of these substances. For example, the knowledge of environmental exposure to antibiotics which may lead to possible evolution and dissemination of antibiotic-resistant pathogens in bacteria is limited (Bengtsson-Palme and Larsson 2016). To experimentally assess the environmental risk of all APIs in use would be a challenge (Perazzolo et al. 2010). One solution is to use formalized prioritization procedures that identify those substances in use that pose the greatest risks towards the natural environment (Boxall et al. 2012). By using these approaches, experimental testing resources can then be focused on those substances that are likely to have the greatest impact.
Several studies have been recently performed that employ different approaches for ranking and assessing the risk posed by APIs to the environment. Most have focused on surface or drinking water and the risks to aquatic organisms or human health. These approaches have been applied in Switzerland (Perazzolo et al. 2010), USA (Kostich and Lazorchak 2008; Dong et al. 2013), France (Besse and Garric 2008), the UK (Boxall et al. 2003; Guo et al. 2016), South Korea (Kim et al. 2008) and Sweden (Roos et al. 2012). Many of these approaches use exposure and toxicological predictions so they can be readily applied to large numbers of compounds with limited data (Boxall et al. 2012).
Most prioritization studies have focused on North America and Western Europe, so our knowledge of priorities in other geographical areas such as Eastern Europe, Africa, Asia and South America is limited. This can be partly explained by the challenges in obtaining information on API usage in these regions. Moreover, although there are strong incentives to introduce the evaluation of an antibiotic to select for resistance into environmental risk assessment guidelines (Bengtsson-Palme and Larsson 2016), none of the previous prioritization approaches has attempted to assess the risk of antibiotics in the environment in terms of their potential to select for antimicrobial resistance.
In Iraq, there are no specific management guidelines for pharmaceuticals in the environment. Pharmaceuticals are freely available to everyone without any restriction and regulation or even without prescription, and there are many routes by which these substances are distributed to the population. One route is the public health sector which is represented by the Ministry of Health (MOH) via the state company for importation and distribution of drug and medical appliances (KIMADIA). The second source is the private sector (licensed and unlicensed low value manufacturers) which includes 23 manufacturing plants, importers and dispensers who supply the local markets with unknown quantities of pharmaceuticals. Additionally, all the locally produced and imported finished pharmaceuticals are not subjected to taxes in order to make them affordable for most of the population (USAID; 2007; EMRO WHO 2011; MOH 2011).
With a highly urbanized population, Iraq still has insufficient environmental management and suffers from poor and old water distribution systems and contaminated main water resources (UNEP 2003). Due to the absence of water quality regulations and the continuous discharges from industry and households via insufficient wastewater treatment plants (WWTPs), up to 19 % of the Iraqi population is exposed to unsafe water (UNEP 2003; USAID 2007). In addition, only 32 % of the population is served with wastewater treatment, meaning that a significant amount of untreated wastewater is released to the environment (COSIT 2012). Few studies to evaluate the quality of environmental systems in Iraq have been performed, and most that have been performed have focused on monitoring the occurrence of trace metals, polycyclic aromatic hydrocarbons (PAHs) and non-polar lipids in the aquatic environment (Abaychi and DouAbul 1985; Al-Saad 1987; Rushdi et al. 2014). The risks of emerging contaminants such as pharmaceuticals have been neglected.
The aim of the present study was therefore to establish the importance of API exposure as a pressure on the natural environment in Iraq and to identify APIs of most concern in local aquatic and terrestrial environments of the three main cities in the country (Baghdad, Mosul and Basrah), where only little is currently known about the exposure and effects of these substances. The prioritization approaches used to achieve this were based on the potential for APIs to enter the aquatic and terrestrial environments and their potential toxic effects on the ecosystems, bacterial community and human health.
Materials and methods
Prioritization approach
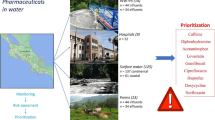
The prioritization approach is illustrated in Fig. 1 and involved the use of predicted environmental concentrations (PECs) and concentrations relating to different effect endpoints (i.e. predicted no-effect concentrations (PNECs), human plasma therapeutic concentrations (HTPCs), minimal inhibitory concentrations (MICs) and minimal selective concentrations (MSCs)) for each of the pharmaceuticals in aquatic and terrestrial systems. PECs and PNECs were then used to calculate risk characterization ratios (RCRs) for apical endpoints, secondary poisoning, toxicity to humans and antimicrobial resistance selection. Pharmaceuticals were then ranked based on their RCRs where compounds with the highest RCRs were considered the highest priority.
The developments of prioritization approach of pharmaceuticals in the environment in Iraq. RCR risk characterization ratios, PECsw, PNECsw predicted environmental concentration and predicted no-effect concentration in surface water, PECsludge predicted environmental concentration in sludge compartment, PECsed, PNECsed predicted environmental concentration and predicted no-effect concentration in sediment compartment, WWTPwastewater treatment plant, BCF fish bioconcentration factor, BMF biomagnification factor, PECbiota predicted environmental concentration in biota (e.g. fish), PNEChuman predicted no-effect concentration in humans from drinking water and fishery products consumption, PECsoil predicted environmental concentration in soil, FSSPC fish steady state plasma concentration, H T PC human therapeutic plasma concentration, PECwarthworm predicted environmental concentration in earthworm, PNECearthworm predicted no-effect concentration in earthworm, PNECmammal predicted no effect concentration in mammal, MIC minimal inhibitory concentration, PNECresistance predicted no-effect concentration for antibiotics resistance selection, MSC minimal selective concentration
Data collection
Usage data
Data on the consumption of pharmaceuticals for hospitals and primary care centres in Iraq in 2014 were obtained from the state company KIMADIA (Kimadia, access 2014). To obtain the total amount of pharmaceuticals consumed, concentrations of active ingredient in packaging units (i.e. blister, bottle, etc.) were converted into mass units. Vitamins, medical supplements, electrolytes and vaccines were excluded which reduced the list of APIs to 99 compounds. In the case of combined medicines, only individual active ingredients were considered and summed up to calculate the weight of pharmaceutical compound.
Information is scarce on the use of over-the-counter pharmaceuticals in Iraq. However, research by the Center of Market Research and Consumer Protection at the University of Baghdad (Mohammed et al. 2009) indicates that over-the-counter usage can contribute 68 % of the total usage of pharmaceuticals in Iraq. Therefore, to obtain a total pharmaceutical usage in Iraq (for both hospitals and primary care centres and over the counter), the results of the analysis of the KIMADIA data were multiplied by a factor of 3.125. Some APIs, such as cancer treatments or those used in surgical procedures in hospitals, were not corrected (multiplied by the factor) as they would not be distributed over the counter. The final usage data are provided in the supporting information (Table S1).
Effects data and physico-chemical properties
To estimate the environmental risk posed by the pharmaceuticals to aquatic and terrestrial ecosystems in Iraq, data on toxicity of the APIs to algae, daphnia, fish and earthworms was used. The data collection included acute and chronic ecotoxicity endpoints (typically the most sensitive LC/EC50 value). These data were obtained from the peer-reviewed literature, grey literature and available online databases (e.g. Swedish voluntary environmental classification of pharmaceuticals at www.fass.se). As experimental ecotoxicity data were not available for a large number of the pharmaceuticals, estimation tools, such as Quantitative Structure-Property Relationships (QSAR) used in the Organisation for Economic Co-operation and Development (OECD QSAR, 2013) Toolbox and the Ecological Structure Activity Relationship ECOSAR (USEPI 4.1) software, were used to fill data gaps (Guo et al. 2016). The database present in the QSAR Toolbox was used to identify experimental data for molecules deemed ‘similar’ to each of the individual pharmaceutical with no data. Then, within the software, a relationship was built to allow an estimation of the ecotoxicological endpoint for the query molecule. Regarding human and mammalian toxicity effects from oral exposure, endpoints such as acceptable daily intake (ADI) values and median lethal dose (LD50) for rat/mouse were used (Technical Guidance for Deriving Environmental Quality Standards EC 2011, Carvalho et al. 2015; Guo et al. 2016). The HTPCs available in peer-reviewed publications were used in the fish plasma model. Finally, for terrestrial toxicity, earthworm acute toxicity (14-day LC50 in mM kg−1 dry soil) was predicted using the QSAR available in ECOSAR for compounds with no experimentally determined earthworm ecotoxicity data. Due to the absence of experimentally determined effects of antibiotics in complex microbial communities, the theoretical MICs, MSCs and PNECs selective resistance calculated by Bengtsson-Palme and Larsson (2016) were used.
Physico-chemical properties required for predicting the fate and behavior of pharmaceuticals in the environment were collated from published articles and open resources. DrugBank, NCCOS (2014) was used to obtain acid dissociation constants (pKa), and the CODATA (2014) database was used to obtain octanol-water partition coefficients (Kow). As there was a lack of experimental data on organic carbon partition coefficient (Koc) for the APIs, for compounds where experimental Koc data were not available, we used the estimation model developed by Franco and Trapp (2008). Excretion profiles for pharmaceuticals were obtained from the peer-reviewed literature, databases or pharmaceutical safety data sheets (i.e. MEDSAFE, Pfizer).
Wastewater generation and dilution factor
Information on wastewater disposal for the main highly urbanized cities in Iraq (Baghdad, Mosul and Basrah) was collected. The daily generated wastewater discharges are 1.6 million m3/day in Baghdad, 0.5 million m3/day in Mosul and 0.331 million m3/day in Basrah (COSIT 2014). These data were used to calculate the wastewater generated per inhabitant (Supporting Information, Equation S1).
It is difficult to determine the dilution factor (DF) in countries with none or very scarce hydrological information like Iraq. For this purpose, we therefore used two dilution factors of 10 and 40 which had been estimated based on a national scale for Iraq by Keller et al. (2014). The percentage of wastewater treatment efficiency will also be important for the calculation of exposure concentrations in surface water so information was also collected on the percentage connectivity to wastewater treatment plants for the three cities. Data on the population, wastewater per capita, wastewater treatment percentage and dilution factors for the cities under study is provided in Table S3 in the supporting information.
Exposure assessment
Predicted environmental concentrations of APIs on the usage list were calculated in aquatic systems (surface water and sediment) and terrestrial systems according to the Guideline on the Environmental Risk Assessment of Medicinal Products for Human Use (EMEA 2006) and the Technical Guidance Document on Risk Assessment part II (TGD 2003) with some modifications to be fitted to the case of Iraq. In surface water, PECsw values for APIs were calculated using the following equation (Eq.1):
where PECSW is the PEC of an API in surface water. Subinhab is the consumed amount of pharmaceuticals per inhabitant in Iraq per day (mg/inh/day) and was calculated based on annual pharmaceutical consumption (kg year−1) and using the population of Iraq (34.2 million), (Eq. S2, Supporting Information); DF is the dilution factor of 10 and 40 and WWinhab is the daily amount of wastewater per inhabitant in either Baghdad, Mosul or Basrah. Fexc is the fraction of parent ingredients excreted unchanged via human metabolism. Sludgeinhab [kg/inh/day] is the mass of waste sludge per inhabitant per day, which is 0.074 (EC 2001); Koc is the organic carbon partitioning coefficient determined experimentally or estimated according to Franco and Trapp (2008) for ionisable chemicals using Kow and pKa; and focsludge is the fraction of sludge organic carbon and was assumed to be 0.326 (Struijs et al. 1991).
The assumption of removal by adsorption was just used in the case of Baghdad because of the absence of wastewater treatment in both Mosul and Basrah. For Mosul, there are no wastewater treatment plants in the city while for Basrah the efficiency percentage of wastewater treatment in existing WWTPs is zero (COSIT 2014).
For sediment, the standard algorithms in the TGDII (2003) was used to estimate concentrations of the APIs in terms of wet weight (ww) (PECsed_ww), and since the final PECsed was calculated in terms of dry weight, a conversion step was applied to determine PECsed on a dry weight basis (Carvalho et al. 2015) (Eqs. S2 to S5 in Supporting Information).
For the calculation of the PECbiota, the following equation was used.
where BCF is the bioconcentration factor for biota (e.g. fish) which was retrieved when available or calculated according to Fick et al. (2010) (Eq. S7, Supporting Information). Default biomagnification factor (BMF) values were retrieved from technical guidance document (EC 2001).
Data on measured levels of pharmaceuticals in fish plasma following exposure via water are still scarce (Fick et al. 2010). As an indicator of specific drugs’ potential to cause adverse pharmacological effects at certain concentrations, the fish steady state plasma concentration (FSSPC) resulting from exposure via surface water was calculated (Eq. S8 in Supporting Information). Predictions were based on estimations of the partitioning of an API between the aqueous phase and arterial blood in the fish (Pblood/water) (Eqs. S9 and S10 in Supporting Information). This partition coefficient was initially estimated based on the log KOW of the API, and this was subsequently combined with the PECsw to estimate the FSSPC.
PECsoil was calculated since a PECsludge had been calculated using algorithms described in the TGD (2003). To estimate the concentration of an API in earthworms (PECearthworm), the concentration in the earthworms on a wet weight basis (C earthworm) was calculated using an estimate of the concentration in porewater (Cporewater) from PECsoil by considering the partitioning behaviour of substances between the soil and aqueous phase (Eqs. S11 to S13 in Supporting Information). The BCF for earthworms was calculated according to the approach in the TGD (2003).
Hazard characterization
In order to calculate PNECs for toxicity to surface water organisms, effects data were divided by a relevant assessment factor (AF), i.e. acute QSAR data =1000; acute experimental data =100; chronic QSAR data =100 and chronic experimental data =10, (TGD 2003). In instances where more than one ecotoxicological value was found, the most sensitive endpoint was used for the generation of the PNEC. PNECs for earthworms were obtained by dividing the 14-day LC50 value by an AF of 1000. PNECs for mammals were obtained by dividing median lethal doses for mouse or rat by an AF of 100. PNECs for resistance were obtained from MSCs using an AF of 10. AFs were not used for the estimation of concentrations causing mode of action-based effects (using the HTPC) or for the MICs for microbes. Specific equations are provided in the Supporting Information (Eqs. S9–S11).
Results and discussion
Experimental data availability
Experimental acute ecotoxicological data were only available for 51 of the 99 APIs under consideration. Chronic ecotoxicity endpoints were only available for 21 compounds so the ecotoxicity values of the others were estimated using the QSAR Toolbox and the ECOSAR software. In terms of data on mammalian safety, data were available on the toxicity of 72 compounds, 87 had an ADI and 88 had a HTPC. Experimental bioconcentration factors in fish (BCFfish) were only available for two compounds (diclofenac and naproxen). Experimental organic carbon partition coefficient (Koc) values were only available for 21 pharmaceuticals (Table 1).
RCR lists of APIs in different systems
The top ranked APIs with RCR >0.1, derived from the different prioritizations for the aquatic environments in the three cities under consideration and at two dilution factors, are presented in Tables 2 and 3 for surface water and Table 4 for sediment. The compounds on the top of the prioritization list with an RCR ≥1 according to PECsw and acute ecotoxicological endpoint were amoxicillin, azithromycin, cefalexine, valproic acid, erythromycin, paracetamol and clarithromycin in Mosul and Basrah. In Baghdad, only five compounds had an RCR ≥1 (amoxicillin, clarithromycin, azithromycin, valproic acid and paracetamol). This difference between the cities is due to the absence of wastewater treatment processes in Mosul and Basrah and hence that no removal of APIs by adsorption on sludge will occur in these cities. When chronic effects were considered, at the lower dilution factor, six compounds had RCR values ≥1 for all cities i.e. amoxicillin, clarithromycin, diclofenac, miconazole nitrate and mefenamic acid. At the higher dilution rate, only two compounds (amoxicillin and clarithromycin) had an RCR ≥1 (Table 3). All other pharmaceuticals had a risk score <0.1 (Table S3, Supporting Information).
When the potential impact of subtle pharmacological effects were considered by comparing the human therapeutic concentration in plasma to estimated levels in fish plasma, using a dilution factor of 10, phenylephrine, atorvastatin and mebeverine showed RCR values >1 in all three cities. Additionally, amitriptyline and mefenamic acid had an RCR ≥1 In Mosul and Basrah. Using the higher dilution factor, only phenylephrine showed RCR >1 in Baghdad and Mosul whereas phenylephrine and atorvastatin exceeded an RCR of 1 in Basrah (Table 3).
Assessment of human exposure from consumption of fish products showed that phenylephrine and atorvastatin had an RCR >1 in all cities when a DF of 10 was used and only phenylephrine (RCR >1) when the DF of 40 was used. For human exposure via drinking water, tramadol HCL was the highest ranked compound (with an RCR between 0.1 and 1 while for the rest of pharmaceuticals the RCR was below 0.1.
The predicted concentrations for amoxicillin in all cities when DF = 10 was used were close to the MICs, and the RCRs were between 1 to 10, suggesting that concentration could be high enough to inhibit growth of or kill bacteria. Amoxicillin and metronidazole were on the top list of antibiotics identified as a risk for selection for bacterial resistance (RCR >10), with a further seven APIs having RCR values between 1 and 10 (Tables 2 and 3).
The highest ranked APIs based on acute effect in sediment organisms were amoxicillin, erythromycin, azithromycin, ciprofloxacin, valproic acid and paracetamol in all cities with RCR >1 (Table 4). Ciprofloxacin was dropped off the top priority list when a DF of 40 was applied in Mosul and Basrah and also paracetamol in Baghdad. The highest ranked compounds based on chronic endpoints were amoxicillin, clarithromycin, diclofenac, miconazole nitrate and mefenamic acid at DF = 10 and only amoxicillin showed RCR >10 in Basrah at DF = 40.
In soil, theophylline was ranked highest priority based on the effect on lower trophic level organisms (earthworm). Based on the potential for secondary poisoning in the aquatic environment (i.e. risk to mammalian predators), only phenylephrine had an RCR >1 for all the city scenarios. For secondary poisoning in the terrestrial environments (i.e. earthworm-eating birds and mammals), the highest ranked compound was atropine with an RCR between 0.1 and 1 (Table 5).
Comparison of ranking outcomes
Generally, the outcome of the risk-based prioritization showed that the majority of the top ranked pharmaceuticals were antibiotics. Based on all risk comparisons, a final list of 23 compounds (amoxicillin, amitriptyline, ampicillin, atorvastatin, azithromycin, cefalexine, ceftriaxone sodium, ciprofloxacin, clarithromycin, diclofenac, erythromycin, ibuprofen, valproic acid, mebeverine, mefenamic acid, metronidazole, miconazole nitrate, olanzapine, omeprazole paracetamol, phenylephrine, theophylline, trimethoprim) which had an RCR >1 for at least one endpoint or compartment was generated. Interestingly, the results of the current prioritization approach agreed with previously published prioritization studies from other countries. Amoxicillin, the compound with the highest score in this study, was also ranked the top veterinary medicine with high hazard to aquatic organisms in the UK and Korea (Boxall et al. 2003; Kim et al. 2008). Clarithromycin and azithromycin where found alongside amoxicillin on the top priority list in France (Besse and Garric 2008). Paracetamol, amoxicillin and azithromycin were ranked as highly prescribed pharmaceuticals of concern in the USA whereas ciprofloxacin was identified as posing a risk toward aquatic organisms and humans (Dong et al. 2013). Paracetamol, mefenamic acid, amoxicillin, ciprofloxacin, erythromycin and valproic acid were prioritized as highest environmental risk in Switzerland (Perazzolo et al. 2010). A prioritization study performed by Roos et al. (2012) showed amitriptyline, paracetamol, diclofenac and valproic acid to be the highest ranked compounds in one or more comparison studies in Sweden while no antibiotics from this study were found in the ranking lists. Paracetamol ranked the 2nd in terms of usage volume in Sweden while in Iraq it was found to be 1st on the prioritization list. Diclofenac showed a risk score of 0.013 which is equal to the one reported in the UK by Ashton et al. (2004). On the other hand, this compound showed a higher risk score (1–10) in Iraq when chronic ecotoxicological endpoints were used. A recent risk-based prioritization study in the UK has shown most of the antibiotics in our list (amoxicillin, azithromycin, ciprofloxacin, clarithromycin and atorvastatin) to have risk scores greater than 1 in one or more of the risk comparisons proposed (Guo et al. 2016). Amitriptyline was ranked as high priority compound when the potential impact of subtle pharmacological effects was considered by comparing the HTPC to estimated levels in fish in the same study. Miconazole was ranked as one of the priority substances used as herd treatment that is moderately used and metabolized (Boxall et al. 2003). It was also found on the top ranking list of pharmaceuticals according to the fish plasma model (Roos et al. 2012). Theophylline showed low risk score in aquatic system, and this agrees with a ranking score of 0.015 in surface water reported by Huschek et al. (2004); while in this study the RCR of theophylline toward terrestrial lower trophic levels was >1 followed by omeprazole and olanzapine. Omeprazole was ranked 19th and 22nd in terms of number of prescribed pharmaceuticals in the prioritization studies in the USA and Sweden (Dong et al. 2013; Roos et al. 2012). No previous prioritization study has ranked phenylephrine as a compound of concern. To our knowledge and after reviewing the literature, antibiotics have not been previously prioritized in surface water in terms of their impact on bacterial community or the susceptibility to pose bacterial resistance.
Pharmaceuticals of concern on the top of priority lists
Antibiotics are often ranked as the highest priority compounds in risk characterization exercises. Recently, the awareness of the risks of antibiotics in the environment has been raised. For example, the European Environmental Quality Directive has added four antibiotics to the watch list of the Water Framework Directive (Carvalho et al. 2015). All of the added antibiotics (azithromycin, erythromycin, ciprofloxacin and clarithromycin) are ranked as high risk compounds in our priority list. Antibiotics are structurally diverse and do not share a common mode of action (Sanderson et al. 2004), and very low concentrations of antibiotics can be considered extremely harmful to organisms and high concentrations of antibiotics in sediment inhibit the growth of bacteria (Kümmerer 2009a, b).
The occurrence and diverse effects of some of the highly ranked APIs have been reported. Although ciprofloxacin, a fluoroquinolone antibiotic, is highly removed in WWTPs, a concentration of 3.8 μg L−1 was detected in wastewater effluent in Australia (Watkinson et al. 2007) and much higher concentrations of 6.5 and 14.0 mg L−1 from two lakes and pharmaceutical production effluent in India, respectively (Fick et al. 2009). Ciprofloxacin showed luminescence inhibition to Vibrio fisheri at 5 mg L−1 of 30-min EC50 (Hernando et al. 2007) and shows high toxicity toward cyanobacteria (Microcystis aeruginosa) with an EC50 of 0.005 mg L−1 with growth inhibition as the endpoint (Halling-Sorensen 2000). In a recent study, ciprofloxacin exposure resulted in growth inhibition of algae (Pseudokirchneriella subcapitata) at a 96-h EC50 of 4.83 mg L−1 (Martins et al. 2012). Erythromycin is frequently detected in water around the world with concentrations between 0.13 and 0.89 μg L−1 (Meinertz et al. 2011; Hernando et al. 2006). It was found to be toxic to algae using chronic tests with a reported EC50 between 0.01 and 0.1 mg L−1 while ecotoxicological results showed that acute toxicity was in the range of 10–30 mg L−1 for algae, daphnia and bacteria (Isidori et al. 2005). Clarithromycin, a derivative of erythromycin, was detected in concentrations between 0.01 and 0.54 μg L−1 in different countries and has been shown to inhibit the growth of algae and cyanobacteria with EC50 values of 0.0371 and 0.0121 mg L−1, respectively (Baumann et al. 2014). The PECsw of amoxicillin, a β-lactam antibiotic, in Iraqi cities was very high and ranged from 0.6 to 24.0 μg L−1. This concentration is extremely high in comparison to levels <0.001 μg L−1 detected in other countries such as in Italy (Castiglioni et al. 2004). It shows high toxicity to blue-green algae (cyanobacteria) with a reported 96-h EC50 of 0.00222 mg L−1 (Fass.se) and is known to cause hepatocyte cytotoxicity as side effect to rainbow trout with a 24-h EC50 >182.7 mg L−1 (Laville et al. 2004).
In addition to direct toxicological risks, the occurrence of antibiotics raises concerns in terms of the promotion of antibiotic resistance in bacteria in environment, which could subsequently make antibiotics ineffective in terms of treatment for both humans and animals since aquatic ecosystems are a recognized reservoir for antibiotic-resistant bacteria (Santos et al. 2010; Kostich et al. 2014; Ågerstrand et al. 2015). Interestingly, the occurrence of antibiotic resistance in the environment is not on the main list of priorities that should be addressed by guidelines for the environmental risk assessment of medicinal products for both human and veterinary use in the European Union [European Medicines Agency (EMEA) 2006; 2008]. Studies from different parts of the world have highlighted the fact that resistant strains of bacteria occur in the environment. For example, in Slovakia, the occurrence of resistance to different antibiotics (erythromycin, clarithromycin, azithromycin, ciprofloxacin, trimethoprim) in coliforms and streptococci from WWTPs sludge was studied (Birošová et al. 2014). In Canada, isolated Escherichia coli retrieved from different sites and aquatic ecosystem compartments (biofilms, sediment and water) showed high frequency of resistance to ampicillin and ciprofloxacin (Maal-Bared et al. 2013). In Brazil, three strains of Salmonella from water samples of a shrimp farm exhibited multiresistance to ampicillin, tetracycline, oxytetracycline and nitrofurantoin (Carvalho et al. 2013). Recently, a study of tetracyclines, sulfonamides and (fluoro)quinolones in sediment and water samples in Guangdong, China, indicated that fish ponds are reservoirs of antimicrobial resistance genes and the presence of potential resistant and pathogen-associated taxonomic groups in fish ponds might imply the potential risk to human health (Xiong et al. 2015).
Two non-steroidal anti-inflammatory drugs (NSAIDs) were identified as high priority APIs i.e. diclofenac and mefenamic acid. In 2013, the European Directive identified diclofenac, alongside two synthetic hormones, as pollutants that should be included in the Water Framework Directive Watch List (Carvalho et al. 2015). van den Brandhof and Montforts (2010) reported the effect of diclofenac on growth retardation in zebrafish after exposure to concentrations >1.5 mg L−1. Hoeger et al. (2005) and Schwaiger et al. (2004) documented that diclofenac has the potential to cause histopathological damage to tissues (kidney) in fish at concentrations close to those regularly found in surface waters. Mefenamic acid showed a maximum PECsw (1.2 μg L−1) which is higher than the reported levels (0.20-0.34 μg L−1) in the UK by Roberts and Thomas (2006). Ecotoxicological effect of mefenamic acid in chronic toxicity tests to Daphnia magna and Moina macrocopa showed significant changes in reproduction (number of young per adult) after the exposure to 1.0 and 0.25 mg L−1 of mefenamic acid, respectively (Jung Collard et al. 2013). The top used compound in Iraq is paracetamol. In our study, the maximum PECsw for paracetamol in Iraqi cities was 23.99 μg L−1 in Basrah which is two times higher than the concentration obtained from a study by Jones et al. (2002) who found the maximum PEC in English rivers to be 11.96 μg L−1 and more than two orders of magnitude higher than the concentration of 0.11 μg L−1 which was detected in 24 % of the rivers in the USA (Kolpin et al. 2002). In terms of ecotoxicological effect, Galus et al. (2013) found that embryonic mortality of zebrafish was raised after exposure to paracetamol at the level of ≥0.5 μg L−1. Very limited studies have been performed on ecotoxicity of valproic acid toward environmental organisms. Herrmann (1993) carried out a prescreen test to investigate the possible hazard posed to humans using zebra fish exposure to valproic acid and revealed that exposure caused retardation and interruption of development. The cholesterol-lowering agent atorvastatin was reported to affect lemna (Lemna gibba) by decreasing pigment content at EC50 0.17 mg L−1 (Brain et al. 2004). It was also found to inhibit growth of Hyalella azteca with LC50 values ranging from 1.30 to 3.56 mg L−1 and Chironomus tentans with LC50 values ranging from 3.94 to 16.42 mg L−1 (Dussault et al. 2008). Amitriptyline was identified as a high priority list due to its potential to elicit subtle effect in fish in the current study. It has previously been reported to pose a risk to surface waters and show toxicity to fish and daphnia, EC50 = 0.78 mg L−1 (Kasprzyk-Hordern 2010). In lower trophics, amitriptyline was reported to inhibit the growth of a macrophyte Lemna minor with a 7-day EC50 of 1.69 mg L−1 (Ågerstrand and Rudén 2010).
Ibuprofen is predicted to occur in Iraqi surface water at concentrations of 0.13–0.8 μg L−1 and sediment at concentrations of 3.0–20 μg Kg−1. The log Kow of 3.73 and low solubility suggest the low mobility of ibuprofen in water and affinity to adsorb to sediment (Bouissou-Schurtz et al. 2014). Ibuprofen was detected at a concentration of 1.3 μg L−1 in surface water in Switzerland (Tixier et al. 2003) and 0.15–3.96 μg L−1 in the influent and effluent wastewater in Sweden (Bendz et al. 2005). It was found that exposure to chronic low levels of ibuprofen alters the pattern of reproduction of Japanese medaka, Oryzias latipes, and may produce sex-specific responses in teleosts (Flippin et al. 2007). Ibuprofen at a concentration slightly higher than 0.2 μg L−1 is able to significantly increase both genetic and cellular damage in freshwater bivalve Dreissena polymorpha (Parolini et al. 2011).
Limitation of the method
Knowledge about usage data is essential to establish a priority list of pharmaceuticals of most concern. In Iraq, it was difficult to obtain the consumption amount of all pharmaceuticals from the ministry of health list due to absence of a governmental statistical data and it is sometimes considered confidential. Moreover, it was not possible to quantify the usage data of over-the-counter (OTC) pharmaceuticals. Therefore, an accurate quantification approach of OTC usage should be a future priority. The project did not consider veterinary pharmaceuticals, but this use pattern could also be an important contributor to the environment, particularly for antibiotic compounds.
Another restraint which increases the uncertainty is the limited availability of ecotoxicological endpoints and the high dependence on the prediction of effects and properties. For example, the practice of using ECOSAR to extrapolate ecotoxicity data may not be appropriate since this software was developed to assess toxicity of compounds other than pharmaceuticals. Physico-chemical properties were also limited; for instance, Koc which was used to estimate adsorption during wastewater treatment was calculated by an empirical estimation model (Franco and Trapp 2008) due to absence of experimental values for all the pharmaceuticals on the list. Moreover, bioconcentration factors for worm (BCFworm) was predicted according to the TGD 2003 to allow the secondary poisoning assessment of pharmaceuticals in the terrestrial compartment due to limited availability of experimental data. This estimation is usually higher than the experimentally obtained BCF values (TGD 2003). Therefore, an improvement in the accuracy of BCFworm estimation in soil warrants further consideration.
Conclusion
An approach has been developed for prioritizing substances that may pose a risk to the aquatic and terrestrial systems in Iraq. Pharmaceutical usage data has been used together with information on the physico-chemical properties, patient metabolism and wastewater treatment removal in this practice to predict API concentrations. The ranking has been performed by comparing these concentrations to a range of experimental and estimated ecotoxicological endpoints including non-standard endpoints such as the potential for subtle pharmacological effects, secondary poisoning and the impact on human via consuming fishery products and drinking water. Dilution factor was found to play an important role to reduce the risk suspected to be posed toward environmental organisms by pharmaceuticals, and results of this study showed that the release of pharmaceuticals to the aquatic environment represents a significant environmental threat, especially when DF is low.
Twenty-three APIs including antibiotics, analgesics, antiepileptics, anti-hypercholesterolemia and anti-asthma have been identified as high priority substances. The study indicates that antibiotics are the pharmaceutical class of most concern with annual consumption of these molecules in Iraq up to 420 t year−1. Risks of pharmaceutical compounds in drinking water to human health are low with the exception of tramadol when no WWTP connectivity exists. Large numbers of pharmaceuticals considered in this study could be removed during wastewater treatment, and their risk towards environment will be highly reduced when a proper removal mechanism is used, but in our study case, the removal by this method is neglected due to the absence or inefficient operation of WWTPs in Iraq. Further evaluation is recommended to assess whether these compounds could indeed pose a risk to the environment as individuals or in a mixture since a broad range of different substances is used simultaneously in humans in any given area.
References
Abaychi JK, Douabul AAZ (1985) Trace metals in Shatt Al-Arab River, Iraq. Water Res 19:457–462
Ågerstrand M, Rudén C (2010) Evaluation of the accuracy and consistency of the Swedish Environmental Classification and Information System for pharmaceuticals. Sci Total Environ 408:2327–2339
Ågerstrand M, Berg C, Björlenius B et al (2015) Improving environmental risk assessment of human pharmaceuticals. Environ Sci Technol 49:5336–5345
Al-Saad HT (1987) Distribution of polycyclic aromatic hydrocarbons (PAH) in surficial sediment from the Shatt al-Arab River and the NW region of the Arabian Gulf. Mar Pollut Bull 18:248–251
Ashton D, Hilton M, Thomas KV (2004) Investigating the environmental transport of human pharmaceuticals to streams in the United Kingdom. Sci Total Environ 333:167–184
Baumann M, Weiss K, Maletzki D et al (2014) Aquatic toxicity of the macrolide antibiotic clarithromycin and its metabolites. Chemosphere 120C:192–198
Bendz D, Paxéus NA, Ginn TR, Loge FJ (2005) Occurrence and fate of pharmaceutically active compounds in the environment, a case study: Höje River in Sweden. J Hazard Mater 122:195–204
Bengtsson-Palme J, Larsson DGJ (2016) Concentrations of antibiotics predicted to select for resistant bacteria: proposed limits for environmental regulation. Environ Int 86:140–149
Besse JP, Garric J (2008) Human pharmaceuticals in surface waters. Implementation of a prioritization methodology and application to the French situation. Toxicol Lett 176:104–123
Birošová L, Mackuľak T, Bodík I et al (2014) Pilot study of seasonal occurrence and distribution of antibiotics and drug resistant bacteria in wastewater treatment plants in Slovakia. Sci Total Environ 490:440–444
Bouissou-Schurtz C, Houeto P, Guerbet M et al (2014) Ecological risk assessment of the presence of pharmaceutical residues in a French national water survey. Regul Toxicol Pharmacol 69:296–303
Boxall A, Rudd M et al (2012) Pharmaceuticals and personal care products in the environment: what are the big questions? Environ Health Perspect 9:1221–1229
Boxall ABA, Fogg LA, Kay P et al (2003) Prioritisation of veterinary medicines in the UK environment. Toxicol Lett 142:207–218
Brain RA, Johnson DJ, Richards SM et al (2004) Microcosm evaluation of the effects of an eight pharmaceutical mixture to the aquatic macrophytes Lemna gibba and Myriophyllum sibiricum. Aquat Toxicol 70:23–40
Carvalho RN, Ceriani L, Ippolito A (2015) Development of the first Watch List under the Environmental Quality Standards Directive water policy. Available from: http://publications.jrc.ec.europa.eu /repository/bitstream/JRC95018/lbna27142enn.pdf. Accessed 20 November 2015
Castiglioni S, Fanelli R, Calamari D et al (2004) Methodological approaches for studying pharmaceuticals in the environment by comparing predicted and measured concentrations in River Po, Italy. Regul Toxicol Pharmacol 39:25–32
Carvalho FCT, Sousa O V, Carvalho EMR, et al. (2013) Antibiotic Resistance of Salmonella spp. Isolated from Shrimp Farming Freshwater Environment in Northeast Region of Brazil. J Pathog. 2013:685193. doi: 10.1155/2013/685193
CNC-CODATA - LogKOW database. http://codata.ca/eng/resources/logkow.html. Accessed 12 September 2014
Dong Z, Senn DB, Moran RE, Shine JP (2013) Prioritizing environmental risk of prescription pharmaceuticals. Regul Toxicol Pharmacol 65:60–67
Drugbank (2014) Open Data Drug and Drug Target Database. http://www.drugbank.ca. Accessed 21 October 2014
Dussault EB, Balakrishnan VK, Sverko E et al (2008) Toxicity of human pharmaceuticals and personal care products to benthic invertebrates. Environ Toxicol Chem 27:425–432
EC (2001) Organic contaminants in sewage sludge for agricultural use. European Commission. Brussels, Belgium, 1–73
EC. European Commission (2003) Technical Guidance Document on Risk Assessment (TGD II) 1–328
EMEA (2006) Guideline on the environmental risk assessment of medicinal products for human use.Committee for Medicinal Products for Human Use.EMEA/CHMP/SWP/4447/00. (December), 1–12
EMEA (2008) Revised guidline on environmental impact assessment for veterinary medicinal products. EMEA/CVMP/ERA/418282/2005-Rev.1. (November), 1–65.European Commission, 2011. Common implementation strategy for the Water Framework Directive (2000/60/EC). Guidance Document No. 27. Technical guidance for deriving Environmental Quality Standards
FASS.se. Swedish Environmental Classification of Pharmaceuticals Database. http://www.fass.se/. Accessed 22 November 2015
Fick J, Soderstrom H, Lindberg RH, Phan C, Tysklind M, Larsson DGJ (2009) Contamination of surface, ground and drinking water from pharmaceutical production. Environ Toxicol Chem 28:2522–2527
Fick J, Lindberg RH, Tysklind M, Larsson DGJ (2010) Predicted critical environmental concentrations for 500 pharmaceuticals. Regul Toxicol Pharmacol 58:516–523
Flippin JL, Huggett D, Foran CM (2007) Changes in the timing of reproduction following chronic exposure to ibuprofen in Japanese medaka, Oryzias latipes. Aquat Toxicol 81:73–78
Franco A, Trapp S (2008) Estimation of the soil-water partition coefficient normalized to organic carbon for ionizable organic chemicals. Environ Toxicol Chem 27:1995–2004
Galus M, Jeyaranjaan J, Smith E et al (2013) Chronic effects of exposure to a pharmaceutical mixture and municipal wastewater in zebrafish. Aquat Toxicol 132–133:212–222
Ginebreda A, Muñoz I, de Alda ML et al (2010) Environmental risk assessment of pharmaceuticals in rivers: relationships between hazard indexes and aquatic macroinvertebrate diversity indexes in the Llobregat River (NE Spain). Environ Int 36:153–162
Government of Iraq, central statistics organization COSIT (2012) Environmental Statistics of Iraq for 2011 (in Arabic). http://cosit.gov.iq/en/env-stats/envstat. Accessed 1 April 2014
Government of Iraq, central statistics organization COSIT (2014) Environmental Statistics of Iraq for 2013 (in English). http://cosit.gov.iq/en/env-stats/envstat. Accessed 15 December 2014
Guo J, Sinclair CJ, Selby K, Boxall ABA (2016) Toxicological and ecotoxicological risk-based prioritization of pharmaceuticals in the natural environment. Environ Toxicol Chem 9999:doi: 10.1002/etc.3319
Halling-Sorensen B (2000) Algal toxicity of antibacterial agents used in intensive farming. Chemosphere 40:731–739
Hernando MD, De Vettori S, Martinez Bueno MJ, Fernandez-Alba A (2007) Toxicity evaluation with Vibrio fischeri test of organic chemicals used in aquaculture. Chemosphere 68:724–730
Hernando MD, Mezcua M, Fernández-Alba AR, Barceló D (2006) Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments. Talanta 69:334–342
Herrmann K (1993) Effects of the anticonvulsant drug valproic acid and related substances on the early development of the zebrafish (Brachydanio rerio). Toxicol in Vitro 7:41–54
Hoeger B, Kollner B, Dietrich DR, Hitzfeld B (2005) Water-borne diclofenac affects kidney and gill integrity and selected immune parameters in brown trout (Salmo trutta f. fario). AquatTox icol 75:53–64
Huschek G, Hansen PD, Maurer HH et al (2004) Environmental risk assesssment of medicinal products for human use according to European Commission recommendations. Environ Toxicol 19:226–240
Isidori M, Lavorgna M, Nardelli A et al (2005) Toxic and genotoxic evaluation of six antibiotics on non-target organisms. Sci Total Environ 346:87–98
Jones OAH, Voulvoulis N, Lester JN (2002) Aquatic environmental assessment of the top 25 English prescription pharmaceuticals. Water Res 36:5013–5022
Jung Collard HR, Ji K, Lee S et al (2013) Toxicity and endocrine disruption in zebrafish (Danio rerio) and two freshwater invertebrates (Daphnia magna and Moina macrocopa) after chronic exposure to mefenamic acid. Ecotoxicol Environ Saf 94:80–86
Kasprzyk-Hordern B (2010) Pharmacologically active compounds in the environment and their chirality. Chem Soc Rev 39:4466–4503
Keller VDJ, Williams RJ, Lofthouse C, Johnson AC (2014) Worldwide estimation of river concentrations of any chemical originating from sewage-treatment plants using dilution factors. Environ Toxicol Chem 33:447–452
Kim Y, Jung J, Kim M et al (2008) Prioritizing veterinary pharmaceuticals for aquatic environment in Korea. Environ Toxicol Pharmacol 26:167–176
Kolpin DW, Furlong ET, Meyer MT (2002) Pharmaceuticals, Hormones, and Other Organic Wastewater Contaminants in U.S. Streams, 1999–2000: A National Reconnaissance. Environ Sci Technol 36: 1202–1211
Kostich MS, Lazorchak JM (2008) Risks to aquatic organisms posed by human pharmaceutical use. Sci Total Environ 389:329–339
Kostich MS, Batt AL, Lazorchak JM (2014) Concentrations of prioritized pharmaceuticals in effluents from 50 large wastewater treatment plants in the US and implications for risk estimation. Environ Pollut 184:354–359
Kümmerer K (2009a) Antibiotics in the aquatic environment—a review—part I. Chemosphere 75:417–434
Kümmerer K (2009b) Antibiotics in the aquatic environment—a review—part II. Chemosphere 75:435–441
Laville N, Ait-Aissa S, Gomez E, Casellas C, Porcher JM (2004) Effects of human pharmaceuticals on cytotoxicity, EROD activity and ROS production in fish hepatocytes. Toxicology 196:41–55
Maal-Bared R, Bartlett KH, Bowie WR, Hall ER (2013) Phenotypic antibiotic resistance of Escherichia coli and E. coli O157 isolated from water, sediment and biofilms in an agricultural watershed in British Columbia. Sci Total Environ 443:315–23
Martins N, Pereira R, Abrantes N et al (2012) Ecotoxicological effects of ciprofloxacin on freshwater species: data integration and derivation of toxicity thresholds for risk assessment. Ecotoxicology 21:1167–1176
Meinertz JR, Schreier TM, Bernardy JA (2011) Chronic toxicity of erythromycin thiocyanate to Daphnia magna in a flow-through, continuous exposure test system. Bull Environ Contam Toxicol 87:621–625
Ministry of health (MOH) report, government of Iraq (2011) Iraq Pharmaceutical Country Profile. http://www.who.int/medicines/areas/coordination/Iraq_PSCPNarrativeQuestionnaire_01022012.pdf
EMRO WHO, Government T, Joint WHO, Review P (2011) WHO EMRO | The Government of Iraq and WHO Joint Program Review Mission (JPRM), the first round dis. http://www.emro.who.int /pdf/iraq-press-releases/2010/thegovernmentofiraqandwhojointprogramreviewmissionjprm.pdf?ua = 1
Mohammed S, Jasim K, Hussain W (2009) Medicine consumption edification in the frame work of Iraqi consumer protection. Iraqi J Market Res Cosimer Protection 1:48–69 (in Arabic)
Monteiro SC, Boxall ABA (2010) Occurrence and fate of human pharmaceuticals in the environment. Rev Environ Contam Toxicol, Vol 202. Springer, New York
Organisation for Economic Co-operation and Development (2013) The OECD QSAR Toolbox.. Available from: http:// www.oecd.org/chemicalsafety/risk-assessment/theoecdqsartoolbox. Accessed 10 January 2015
Parolini M, Binelli A, Provini A (2011) Chronic effects induced by ibuprofen on the freshwater bivalve Dreissenapolymorpha. Ecotoxicol Environ Saf 74:1586–1594
Perazzolo C, Morasch B, Kohn T et al (2010) Occurrence and fate of micropollutants in the Vidy Bay of Lake Geneva, Switzerland. Part I: priority list for environmental risk assessment of pharmaceuticals. Environ Toxicol Chem 29:1649–1657
Roberts P, Thomas K (2006) The occurrence of selected pharmaceuticals in wastewater effluent and surface waters of the lower Tyne catchment. Sci Total Environ 356:143–153
Roos V, Gunnarsson L, Fick J et al (2012) Prioritising pharmaceuticals for environmental risk assessment: towards adequate and feasible first-tier selection. Sci Total Environ 421–422:102–110
Rushdi AI, DouAbul AAZ, Simoneit BRT et al (2014) Nonpolar lipid tracers in sediments from the Shatt al-Arab River of Iraq and the northwestern Arabian Gulf. Arab J Geosci 7:5495–5508
Sanderson H, Johnson DJ, Reitsma T et al (2004) Ranking and prioritization of environmental risks of pharmaceuticals in surface waters. Regul Toxicol Pharmacol 39:158–183
Santos LHMLM, Araújo AN, Fachini A et al (2010) Ecotoxicological aspects related to the presence of pharmaceuticals in the aquatic environment. J Hazard Mater 175:45–95
Schwaiger J, Ferling H, Mallow U, Wintermayr H, Negele RD (2004) Toxic effects of the non-steroidal anti-inflammatory drug diclofenac part 1: histopathological alterations and bioaccumulation in rainbow trout. Aquat Toxicol 68:141–150
Struijs J, Stoltenkamp J, van de Meent DA (1991) spreadsheet based box model to predict the fate of xenobiotics in a municipal wastewater treatment plant. Water Res 25:891–900
Tixier C, Singer HP, Oellers S, Muller SR (2003) Occurrence and fate of carbamazepine, clofibric acid, diclofenac, ibuprofen, ketoprofen, and naproxen in surface waters. Environ Sci Technol 37:1061–1068
UNEP (2003) Environment in Iraq: UNEP Progress Report. Geneva, Switzerland: United Nations Environment Programme, 20 October 2003. http://www.unep.org/disastersandconflicts/portals/155disastersandconflicts /docs/iraq /Iraq_pccleanup_report.pdf
USAID (2007) Iraq private sector growth and employment generation: Pharmaceutical and medical products in Iraq. https://archive.org pharmaceutical and medical products in Iraq. Accessed 10 December 2014
van den Brandhof E-J, Montforts M (2010) Fish embryo toxicity of carbamazepine, diclofenac and metoprolol. Ecotoxicol Environ Saf 73:1862–6
Watkinson AJ, Murby EJ, Costanzo SD (2007) Removal of antibiotics in conventional and advanced wastewater treatment: implications for environmental discharge and wastewater recycling. Water Res 41:4164–4176
Xiong W, Sun Y, Zhang T et al (2015) Antibiotics, antibiotic resistance genes, and bacterial community composition in fresh water aquaculture environment in China. Microb Ecol 70:425–432. doi:10.1007/s00248-015-0583-x
Acknowledgments
The authors would like to thank the Ministry of Higher Education and Scientific Research (MOHESR), Iraq, for funding the Ph.D. work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Ester Heath
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 61 kb).
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Al-Khazrajy, O.S.A., Boxall, A.B.A. Risk-based prioritization of pharmaceuticals in the natural environment in Iraq. Environ Sci Pollut Res 23, 15712–15726 (2016). https://doi.org/10.1007/s11356-016-6679-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6679-0