Abstract
A loss of skeletal muscle mass and an increase in intramuscular fat are known to occur as we enter middle and older age, but the expected changes or normative values have remained unknown. The primary reason for this is that imaging studies are difficult and expensive to conduct, and consequently, the sample sizes have remained small. The development of the UK Biobank which provides access to a large magnetic resonance imaging (MRI) data set of more than 50,000 participants provides an opportunity to finally address this question of normative values for each age group. The study’s primary aim was to determine the age-related changes in thigh muscle composition (e.g., thigh fat-free muscle volume and intramuscular fat) between the ages of 45 and 84 years. The second aim was to analyse associations between thigh fat-free muscle volume and intramuscular fat with lifestyle behaviours (smoking, alcohol consumption, and physical activity), leg pain, and bone mineral density. Fifty thousand three hundred thirty-two participants were included in the study. Total fat-free thigh muscle declined between the ages of 45 and 84 years, while intramuscular fat of the thigh continued to increase. The changes were stable between these age groups. The mean volume of fat-free muscle ranged from 11.16 (SD: 1.40) to 13.26 L (SD: 1.85) in adult males and 7.60 (SD: 0.97) to 8.80 L (SD 1.29) in females between the ages of 45 and 84 years. For intramuscular fat, the change among women was from 6.94% (SD: 1.59) in the 45 to 54 years age bracket to 8.83% (SD: 1.92) in the 75 to 84 age bracket, while for men, it was 5.83% (SD: 1.30) in the 45 to 54 age bracket to 7.85% (SD 1.89) in the 75 to 84 age bracket. The total fat-free muscle volume and intramuscular fat percentage provided can be used for the purpose of reference standards or normative values for adults in the age groups provided. Fat-free muscle and intramuscular fat were found to be associated with a range of health, activity, and leg pain outcomes, and these should be investigated in a follow-up longitudinal imaging study.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
This progressive loss of muscle mass is ubiquitous across all mammalian species and common in adults aged 65 years and older [1, 2]. Recent analyses suggest that muscle loss is a natural phenomenon of ageing [3]; while others suggest that muscle loss is a disease due to its association with health-related outcomes [4, 5]. Intramuscular fat has also been considered an important marker of health, however, like muscle mass, its impact on health-related outcomes remains unclear [6, 7]. Muscle mass and intramuscular fat have been found previously to be associated with a range of pain and health conditions [8,9,10,11,12]. Unfortunately, these studies are limited by smaller sample sizes with imprecise estimates and low statistical power.
To increase our understanding of changes in muscle-mass (specifically fat-free muscle) and intramuscular fat as we age, studies with large sample sizes are needed. The UK Biobank offers a unique opportunity to overcome the limitations of the previous studies of fat-free muscle and intramuscular fat by providing access to the imaging of over 50,000 participants [13]. This dataset far exceeds the sample size of previous imaging studies and creates the opportunity to contribute to the development of a normative reference standard for muscle composition that could allow adults to compare their muscle composition to others of the same age bracket.
One major consideration for researchers using the UK Biobank is which muscle group to select when identifying muscle composition. For this study, the thigh was selected for identifying fat-free muscle volume and intramuscular fat as the thigh is able to provide reliable estimates with the available imaging data in the UK Biobank, and its precedent as a representative region of muscle composition in a person [10, 11].
Given the focus of previous studies [8,9,10,11,12] on the association of fat-free muscle and intramuscular fat on health outcomes, a secondary aim was to analyse any associations between a range of behavioural outcomes (smoking use and alcohol consumption), self-reported physical function (i.e., walking, duration of strenuous activity), the clinical outcomes of bone mineral density, and leg pain with muscle composition. The primary aim of the study was to present the mean total fat-free muscle volume and intramuscular fat between the ages of 45 and 84 years to generate sex-related normative values and reference standards for the community.
Methods
Study participants
This study analysed the currently available imaging data of 50,332 subjects from the UK Biobank, acquired between 2014 and 2022, with patient meta-data obtained through application number 86983. The UK Biobank is a large prospective study of over 500,000 volunteers between 40 and 84 years across the UK, collecting data on genetics, biochemistry, health and lifestyle, and medical records [13]. A subset of individuals also volunteered for the whole-body imaging study, with a planned 100,000 participants. The study received ethical approval from the North West Multi-Centre Research Ethics Committee (REC reference: 16/NW/0274) and was conducted in accord with the principles of the Declaration of Helsinki. All participants provided written informed consent for data collection, analysis, and record linkage. The study followed the STROBE reporting guidelines.
Participants from the UK Biobank meeting the inclusion and none of the exclusion criteria were included.
Inclusion criteria was as follows:
-
An adult (female or male) who had enrolled in and completed data collection for the UK Biobank.
-
Had imaging data available (the initial imaging assessment (Instance 2) of the UK Biobank.
Exclusion criteria was as follows:
-
Did not have data available on the primary and secondary outcomes at the initial imaging assessment follow-up.
Imaging protocol
The UK Biobank MRI protocol examined in this study is listed as UK Biobank field 20,201 and detailed in supplementary documentation. All participants were scanned in a Siemens MAGNETOM Aera 1.5-T MRI scanner (Siemens Healthineers, Erlangen, Germany) at one of six centres. Volumetric, co-aligned images of water and fat signal were acquired with a 6-min dual-echo Dixon Vibe protocol with TR = 6.69, TE 1 and 2 = 2.39/4.77 ms, and flip angle 10° [14, 15]. Each whole-body data set consists of 6 scan positions covering the neck, thorax, abdomen, pelvis, and thighs. The image resolution varied between stations, with a typical grid of 224 × 174 × 44 and voxel size of 2.232 × 2.232 × 4.5 mm. Full-body DEXA (General Electric Lunar iDXA, Madison, WI) is also completed with staff trained to standardise assessment and using previously validated methods [16].
Image analysis
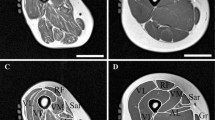
Derived measures of body composition were developed by AMRA™ Profiler (Advanced MR Analytics, Linköping, Sweden), based on volumetric multi-atlas segmentations [14, 17]. These included abdominal subcutaneous adipose tissue (ASAT), visceral adipose tissue (VAT), and posterior and anterior thigh muscle mass (right and left) and measured in litres as described in Borga et al. [17]. The total abdominal adipose tissue (TAAT) is separated into intra-abdominal (IAAT) and abdominal subcutaneous (ASAT) adipose tissue. The volume of the thigh muscles is measured in litres as described in Karlsson et al. [18]. The muscle volume is defined as the lean muscle volume, being the volume enclosing the muscle subtracted by the volume of adipose tissue within the same volume. The anterior thigh muscle group comprised of the quadriceps femoris and sartorius. The posterior thigh muscle is comprised of the hamstring, gluteus, iliacus, and adductor.
Exposures
Fat-free muscle volume was defined as the volume of all voxels with fat fraction < 50% in the thighs. Intramuscular fat was defined as the mean fat fraction in the ‘viable muscle tissue’ of the right and left anterior thighs. These definitions have been used in previous UK Biobank publications [19].
Outcomes
The primary outcomes were (a) the total thigh fat-free muscle volume and (b) intramuscular fat of participants characterised by age (45–54, 55–64, 65–74, and 75–84) from fat–water Dixon MRI. Secondary outcomes included associations for total thigh fat-free muscle volume or intramuscular fat with bone mineral density (total, legs), smoking status, alcohol consumption, leg pain, and physical activity. Leg pain included eight variables: leg pain on walking, leg pain when standing still or sitting, leg pain in calf/calves, leg pain when walking uphill or hurrying, leg pain when walking normally, leg pain when walking ever disappears while walking, leg pain on walking: action taken, and leg pain on walking: effect of standing still. Physical activity included eight duration-related variables (duration of heavy Do-It-Yourself (DIY), duration of light DIY, duration of moderate activity, duration of other exercises, duration of strenuous sports, duration of vigorous activity, duration of walks, and duration walking for pleasure) and six frequency-related variables (frequency of heavy DIY in last 4 weeks, frequency of light DIY in last 4 weeks, frequency of other exercises in last 4 weeks, frequency of stair climbing in last 4 weeks, frequency of strenuous sports in last 4 weeks, and frequency of walking for pleasure in last 4 weeks) (see Supplementary Table 1A for full list).
Statistical analysis
For the primary outcome, the baseline characteristics were tabulated with means and standard deviations presented for continuous variables. For the categorical variables, the frequency of responses, the percentage of the cohort, and the percentage of total responses are presented.
For the secondary outcome, we examined the association between muscle composition (total thigh fat-free muscle volume and intramuscular fat) and all above mentioned outcomes through a stepped modelling framework: model 1, unadjusted analyses; model 2, analyses adjusted for age and sex (see Tables 3 and 4). Logistic regression was used when the outcome was binary (e.g., smoking status) and the corresponding estimates were reported as odds ratio (OR) and 95% confidence interval (CI); linear regression was used when the outcome was continuous and the corresponding estimates were reported as mean difference (MD) and 95% CI; ordinal logistic regression was used when the outcome was ordinal (e.g., frequency of walking for pleasure in last 4 weeks) and the corresponding estimates were reported as proportional OR and 95% CI. All statistical analyses were performed in R, version 4.2.2 (R Group for Statistical Computing).
Patient and public involvement
No patients or public involvement was present in our study.
Results
The study included 50,332 participants from the UK Biobank study who consented and underwent imaging that included the thigh. Baseline characteristics of the participants are outlined in Table 1, with the full table provided in supplementary material (see Table 1a).
In both females and males, the peak volume of thigh fat-free muscle was in the 45 to 54 age group (8.80 L in females and 13.26 L in males) and continued a linear decline, with the lowest volumes recorded in the 75 to 84 age group (7.60 L in females and 11.16 L in males). Increasing age was related to increasing intramuscular fat, with the lowest intramuscular fat recorded in the 45 to 54 age group for both females and males (6.94% in females and 5.83% in males) and highest recorded in the 75 to 84 age group (8.83% in females and 7.85% in males) (Table 2, Fig. 1).
Association between fat-free muscle volume and outcomes
Smoking and physical activity
Table 3 identifies the association between fat-free muscle volume and selected outcomes. Participants who smoked were less likely to have higher fat-free muscle volume (OR 0.92, 95% CI 0.88 to 0.96). Participants who reported longer durations of vigorous activity (MD 1.40, 95% CI 1.02 to 1.78), spent longer time walking for pleasure (OR 1.05, 95% CI 1.04 to 1.07), spent longer time doing other exercises (OR 1.09, 95% CI 1.07 to 1.12), spent longer time doing light (OR 1.02, 95% CI 1.00 to 1.04) and heavy DIY activities (OR 1.05, 95% CI 1.03 to 1.07), more frequently doing other exercises (OR 1.10, 95% CI 1.08 to 1.12), more frequently climbing a flight of stairs (OR 1.01, 95% CI 1.00 to 1.02), and more frequently doing strenuous sports (OR 1.11, 95% CI 1.07 to 1.15) were more likely to have higher fat-free muscle volume.
Leg pain
People that reported stopping or slowing down when they get pain during walking (OR 0.92, 95% CI 0.87 to 0.98), getting pain when they walk uphill or hurry (OR 0.92, 95% CI 0.88 to 0.95), and getting pain in their calves (OR 0.92, 95% CI 0.88 to 0.96) were less likely to have higher fat-free muscle volume. People reporting pain in either leg on walking were associated with lower fat-free muscle volume (OR 1.05, 95% CI 1.03 to 1.07) (see Table 3A in supplementary for full list).
Association between intramuscular fat and outcomes
Smoking and physical activity
Table 4 identified associations between intramuscular fat and selected outcomes. Participants who smoked were found to be more likely to have higher intramuscular fat (OR 1.16, 95% CI 1.13 to 1.20). People who reported shorter durations of walks (MD − 0.95, 95% CI − 1.38 to − 0.52), moderate activity (MD − 1.19, 95% CI − 1.63 to − 0.74), and vigorous activity (MD − 1.53, 95% CI − 1.86 to − 1.20) were associated with lower odds of increased intramuscular fat. Spending less time: walking for pleasure (OR 0.87, 95% CI 0.86 to 0.88), doing other exercises (OR 0.92, 95% CI 0.91 to 0.94), doing light (OR 0.98, 95% CI 0.97 to 0.99) and heavy (OR 0.96, 95% CI 0.94 to 0.98) DIY, climbing a flight of stairs (OR 0.86, 95% CI 0.85 to 0.87), and less frequently doing strenuous sports (OR 0.85, 95% CI 0.82 to 0.89) were also associated with lower odds of higher intramuscular fat.
Leg pain
Participants who reported pain in either leg on walking (OR 1.28, 95% CI 1.26 to 1.30), stopping or slowing down when getting pain during walking (OR 1.22, 95% CI 1.18 to 1.27), experiencing pain that usually continues for more than 10 min while walking if they stand still (OR 1.07, 95% CI 1.04 to 1.10), getting pain when they walk at an ordinary pace on the level (OR 1.11, 95% CI 1.08 to 1.14), getting pain when they walk uphill or hurry (OR 1.15, 95% CI 1.12 to 1.18), getting pain in their calves (OR 1.07, 95% CI 1.04 to 1.10), and getting pain when they are standing still or sitting (OR 1.06, 95% CI 1.03 to 1.10) were associated with higher intramuscular fat. People reporting the disappearance of pain when they walk were less likely to have higher intramuscular fat (OR 0.85, 95% CI 0.82 to 0.88) (see Table 4A in supplementary for full list).
Discussion
In this large database study (n = 50,332), we found that total fat-free thigh muscle declined between the ages of 45 and 84 years, while intramuscular fat of the thigh continued to increase. The changes were stable and linear between these age groups. The mean volume of fat-free muscle ranged from 11.2 to 13.3 L in adult males and 7.6 to 8.8 L in females between the ages of 45 and 84 years. For intramuscular fat, the change among women was from 6.94% in the 45 to 54 years age bracket to 8.83% in the 75 to 84 age bracket, while for men it was 5.83% in the 45 to 54 age bracket to 7.85% in the 75 to 84 age bracket. The results demonstrate that on average men experience a greater loss of fat-free muscle compared with women between the ages of 45 to 54 and 75 to 84 (15.83% reduction for men and 13.63% reduction for women). Men also experience a greater increase in intramuscular fat compared with women between the ages of 45 to 54 and 75 to 84. Our results are supported by previous studies [20], including those that have used older versions of UK Biobank with smaller samples (i.e., 14,148 participants compared with our study of 50,332 participants) [20].
A key advantage of our study is the ability to generate very precise results due to the large sample size. The results can therefore be considered as reference value for individuals comparing their fat-free muscle and intramuscular fat values with others in their age bracket. Reference values for muscle composition serve the benefit of allowing the community to identify what changes an individual may expect as they age and to avoid being classified as having ‘abnormal changes’. Reference values are commonly in use with blood and urine tests among others, but have not yet been established for muscle composition.
Our secondary aim to analyse associations between muscle composition and selected outcomes found significant but often small associations. Interestingly, leg pain outcomes had inconsistent associations with total fat-free muscle volume, but were strongly associated with intramuscular fat, warranting further investigation. The studies should include longitudinal follow-up and should consider using available automated segmentation algorithms, which can provide time-saving measures without sacrificing measurement accuracy. Given the burden of leg pain among adults 45 years and older is more than 43% [21], targeted treatments to reduce its prevalence remain a priority [22, 23], and increased understanding of any causal relationship is worthy of investigation. Previous associations have already been reported between intramuscular fat with other conditions, such as fibromyalgia [8], lumbar spinal stenosis in those with achondroplasia [24], cervical myelopathy [9], whiplash [25], neck pain [26], and heart failure [10].
Almost all physical activity outcomes were associated with small but significant increases in total fat-free muscle volume and decreased intramuscular fat. Both increased duration of moderate and vigorous activity were associated with increased total fat-free muscle volume and decreased intramuscular fat, with the change being larger in vigorous activity. These findings are consistent with existing literature which promotes physical activity for increasing fat-free muscle and reducing intramuscular fat [27]. The impact of self-reported versus objective capture of physical activity remains unknown, but future studies would benefit from including both measures. Limitations are present in this review. The population within the UK Biobank is not ethnically diverse, which limits the generalisability of the results beyond citizens of the UK [28]. The composition of the thigh muscle relied upon the resolution of the imaging. Given the volume of imaging stored in the UK Biobank, the resolution of the imaging could be lower than in other smaller studies. The UK Biobank undertook a series of initiatives to ensure the quality of the imaging, however, such as extensive training of radiology staff, imaging quality checks, and a large data storage centre [15]. For the secondary aims, outcomes such as physical activity were self-reported, which, although common in studies, may not be representative of true function for that individual [29, 30]. The study also looked at associations, which can be impacted by confounders, such as age, sex at birth, and other life-expected comorbidities, including, but not limited to, diabetes, cardiovascular disease, neurodegenerative disorders, mental health conditions, or early life adversity. We aimed to address this limitation by including an additional analysis adjusted for age and sex, but this falls short of attempting to adequately capture the complexities of the human experience [31, 32].
Conclusions
This study found between 45 and 84 years of age fat-free muscle continued to decline, while the magnitude of intramuscular fat increased. The mean fat-free muscle volume and intramuscular fat percentage identified in this large study support an initial reference standard for normative values. Reference standards or normative value standards can provide a comparative measure controlled for age and sex and other potentially confounding factors. Most physical activity and leg pain outcomes were associated with both fat-free muscle and intramuscular fat, and these should be further investigated in a longitudinal study, potentially with the large UK Biobank data.
Data availability
This research has been conducted using the UK Biobank Resource under Project ID: 86983. Requests to access the data should be made via application directly to the UK Biobank, https://www.ukbiobank.ac.uk.
References
Franzon K, Zethelius B, Cederholm T, Kilander L. The impact of muscle function, muscle mass and sarcopenia on independent ageing in very old Swedish men. BMC Geriatr. 2019;19(1):153. https://doi.org/10.1186/s12877-019-1142-y.
Dent E, Morley JE, Cruz-Jentoft AJ, et al. International clinical practice guidelines for sarcopenia (ICFSR): screening, diagnosis and management. J Nutr Health Aging. 2018;22(10):1148–61. https://doi.org/10.1007/s12603-018-1139-9.
Haase CB, Brodersen JB, Bulow J. Sarcopenia: early prevention or overdiagnosis? BMJ. 2022;376: e052592. https://doi.org/10.1136/bmj-2019-052592.
Tarantino G, Sinatti G, Citro V, Santini SJ, Balsano C. Sarcopenia, a condition shared by various diseases: can we alleviate or delay the progression? Intern Emerg Med. 2023;18(7):1887–95. https://doi.org/10.1007/s11739-023-03339-z.
Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31. https://doi.org/10.1093/ageing/afy169.
Lube J, Cotofana S, Bechmann I, et al. Reference data on muscle volumes of healthy human pelvis and lower extremity muscles: an in vivo magnetic resonance imaging feasibility study. Surg Radiol Anat. 2016;38(1):97–106. https://doi.org/10.1007/s00276-015-1526-4.
Marcon M, Berger N, Manoliu A, et al. Normative values for volume and fat content of the hip abductor muscles and their dependence on side, age and gender in a healthy population. Skeletal Radiol. 2016;45(4):465–74. https://doi.org/10.1007/s00256-015-2325-z.
Gerdle B, Dahlqvist Leinhard O, Lund E, et al. Fibromyalgia: associations between fat infiltration, physical capacity, and clinical variables. J Pain Res. 2022;15:2517–35. https://doi.org/10.2147/JPR.S376590.
Paliwal M, Weber KA 2nd, Smith AC, et al. Fatty infiltration in cervical flexors and extensors in patients with degenerative cervical myelopathy using a multi-muscle segmentation model. PLoS ONE. 2021;16(6): e0253863. https://doi.org/10.1371/journal.pone.0253863.
Huynh K, Ayers C, Butler J, et al. Association between thigh muscle fat infiltration and incident heart failure: the health ABC study. JACC Heart Fail. 2022;10(7):485–93. https://doi.org/10.1016/j.jchf.2022.04.012.
Martel-Duguech L, Alonso-Jimenez A, Bascunana H, et al. Thigh muscle fat infiltration is associated with impaired physical performance despite remission in Cushing’s syndrome. J Clin Endocrinol Metab 2020;105(5). https://doi.org/10.1210/clinem/dgz329.
Visser M, Goodpaster BH, Kritchevsky SB, et al. Muscle mass, muscle strength, and muscle fat infiltration as predictors of incident mobility limitations in well-functioning older persons. J Gerontol A Biol Sci Med Sci. 2005;60(3):324–33. https://doi.org/10.1093/gerona/60.3.324.
Sudlow C, Gallacher J, Allen N, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12(3): e1001779. https://doi.org/10.1371/journal.pmed.1001779.
West J, Dahlqvist Leinhard O, Romu T, et al. Feasibility of MR-based body composition analysis in large scale population studies. PLoS ONE. 2016;11(9): e0163332. https://doi.org/10.1371/journal.pone.0163332.
Littlejohns TJ, Holliday J, Gibson LM, et al. The UK Biobank imaging enhancement of 100,000 participants: rationale, data collection, management and future directions. Nat Commun. 2020;11(1):2624. https://doi.org/10.1038/s41467-020-15948-9.
Rothney MP, Martin FP, Xia Y, et al. Precision of GE Lunar iDXA for the measurement of total and regional body composition in nonobese adults. J Clin Densitom. 2012;15(4):399–404. https://doi.org/10.1016/j.jocd.2012.02.009.
Borga M, Thomas EL, Romu T, et al. Validation of a fast method for quantification of intra-abdominal and subcutaneous adipose tissue for large-scale human studies. NMR Biomed. 2015;28(12):1747–53. https://doi.org/10.1002/nbm.3432.
Karlsson A, Rosander J, Romu T, et al. Automatic and quantitative assessment of regional muscle volume by multi-atlas segmentation using whole-body water-fat MRI. J Magn Reson Imaging. 2015;41(6):1558–69. https://doi.org/10.1002/jmri.24726.
Linge J, Petersson M, Forsgren MF, Sanyal AJ, Dahlqvist LO. Adverse muscle composition predicts all-cause mortality in the UK Biobank imaging study. J Cachexia Sarcopenia Muscle. 2021;12(6):1513–26. https://doi.org/10.1002/jcsm.12834.
Dai L, Huang XY, Lu YQ, et al. Defining reference values for body composition indices by magnetic resonance imaging in UK Biobank. J Cachexia Sarcopenia Muscle. 2023;14(2):992–1002. https://doi.org/10.1002/jcsm.13181.
Lucas JW CE, Bose J. Back, lower limb, and upper limb pain among U.S. adults, 2019. National Center for Health Statistics 2021. https://doi.org/10.15620/cdc:107894.
Jin Y, Guo C, Abbasian M, Abbasifard M, Abbott JH, Abdullahi A, Abedi A, Abidi H, Abolhassani H, Abu-Gharbieh E, Aburuz S. Global pattern, trend, and cross-country inequality of early musculoskeletal disorders from 1990 to 2019, with projection from 2020 to 2050. Med. 2024. https://doi.org/10.1016/j.medj.2024.04.009.
Vollset SE, Ababneh HS, Abate YH, Abbafati C, Abbasgholizadeh R, Abbasian M, Abbastabar H, Abd Al Magied AH, Abd ElHafeez S, Abdelkader A, Abdelmasseh M. Burden of disease scenarios for 204 countries and territories, 2022–2050: a forecasting analysis for the global burden of disease study 2021. The Lancet. 2024;403(10440):2204–56. https://doi.org/10.1016/S0140-6736(24)00685-8.
Fredwall SO, Linge J, de Vries O, et al. Fat infiltration in the thigh muscles is associated with symptomatic spinal stenosis and reduced physical functioning in adults with achondroplasia. Orphanet J Rare Dis. 2023;18(1):35. https://doi.org/10.1186/s13023-023-02641-5.
Elliott JM, Smith AC, Hoggarth MA, et al. Muscle fat infiltration following whiplash: a computed tomography and magnetic resonance imaging comparison. PLoS ONE. 2020;15(6): e0234061. https://doi.org/10.1371/journal.pone.0234061.
Snodgrass SJ, Stanwell P, Weber KA, et al. Greater muscle volume and muscle fat infiltrate in the deep cervical spine extensor muscles (multifidus with semispinalis cervicis) in individuals with chronic idiopathic neck pain compared to age and sex-matched asymptomatic controls: a cross-sectional study. BMC Musculoskelet Disord. 2022;23(1):973. https://doi.org/10.1186/s12891-022-05924-3.
Ogawa M, Tanaka N, Yoshiko A, Oshida Y, Koike T, Akima H. Relationship between physical activity time and intramuscular adipose tissue content of the thigh muscle groups of younger and older men. Sci Rep. 2021;11(1):19804. https://doi.org/10.1038/s41598-021-99126-x.
Fry A, Littlejohns TJ, Sudlow C, et al. Comparison of sociodemographic and health-related characteristics of UK biobank participants with those of the general population. Am J Epidemiol. 2017;186(9):1026–34. https://doi.org/10.1093/aje/kwx246.
Anderson DB, Mobbs RJ, Smith ZA, De Luca K, Sabet T, Van Gelder JM. Importance of valid, reliable, and responsive outcome measures for lumbar spinal stenosis. Spine J. 2022. https://doi.org/10.1016/j.spinee.2022.11.014.
Chakravorty A, Mobbs RJ, Anderson DB, et al. The role of wearable devices and objective gait analysis for the assessment and monitoring of patients with lumbar spinal stenosis: systematic review. BMC Musculoskelet Disord. 2019;20(1):288. https://doi.org/10.1186/s12891-019-2663-4.
Mittinty MM, Lee JY, Walton DM, El-Omar EM, Elliott JM. Integrating the gut microbiome and stress-diathesis to explore post-trauma recovery: an updated model. Pathogens 2022;11(7). https://doi.org/10.3390/pathogens11070716.
Walton DM, Elliott JM. A new clinical model for facilitating the development of pattern recognition skills in clinical pain assessment. Musculoskelet Sci Pract. 2018;36:17–24. https://doi.org/10.1016/j.msksp.2018.03.006.
Acknowledgements
The authors would like to thank all participants.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions No funding was provided for this manuscript, but access to the UK Biobank was provided by James M Elliott who used monies from his own research funds. This research was conducted using the UK Biobank resource (Project ID: 86983).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
UK Biobank has approval from the North West Multi-Centre Research Ethics Committee (REC reference 16/NW/0274). This study did not involve recontacting participants, and no separate ethics approval was required.
Competing interests
David Anderson is a senior editor for PM&R and receives payment for this role. Ken Weber reports that financial support was provided by the National Institutes of Health, and has received honourary payments from Norwegian Chiropractic Association and MGH. James Elliott reports a relationship with Orofacial Therapeutics LP that includes consulting or advisory. James Elliott reports a relationship with Medbridge that includes speaking and lecture fees. Lingxiao Chen is funded by the International Postdoctoral Exchange Fellowship Program (Talent-Introduction Program, YJ20220294) and the China Postdoctoral Science Foundation under Grant Number 2024M751834. Aaron Beach, Henry Feng, Marnee McKay, Zachary Smith, Evert Wesselink reported no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Anderson, D.B., Beach, A.J., Chen, L. et al. What is normal age-related thigh muscle composition among 45- to 84-year-old adults from the UK Biobank study. GeroScience (2024). https://doi.org/10.1007/s11357-024-01304-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11357-024-01304-y