Abstract
An acylated flavonol glycoside, trans-tiliroside (1), is found in certain parts of different herbs, including the seeds of Rosa canina (Rosaceae). Previous studies on compound 1 have focused on triglyceride (TG) metabolism, including its anti-obesity and intracellular TG reduction effects. In the present study, the effects of compound 1 on cholesterol (CHO) metabolism were investigated using human hepatocellular carcinoma-derived HepG2 cells and mice. Compound 1 decreased CHO secretion in HepG2 cells, which was enhanced by mevalonate in a concentration-dependent manner and decreased the secretion of apoprotein B (apoB)-100, a marker of very low-density lipoprotein (VLDL). Compound 1 also inhibited the activity of microsomal triglyceride transfer proteins, which mediate VLDL formation from cholesterol and triglycerides in the liver. In vivo, compound 1 inhibited the accumulation of Triton WR-1339-induced TG in the blood of fasted mice and maintained low levels of apoB-100. These results suggest that compound 1 inhibits the secretion of CHO as VLDL from the liver and has the potential for use for the prevention of dyslipidemia.
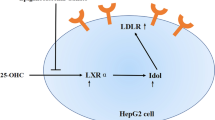
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dyslipidemia is a pathological condition in which blood levels of cholesterol (CHO), triglycerides (TG), and lipoproteins are elevated. It is an important risk factor for atherosclerotic cardiovascular disease (ASCVD), which is the most common cause of death not only in Japan but globally [1]. Low-density lipoprotein cholesterol (LDL-C), also known as the "bad cholesterol", plays an important role in transporting CHO and TG to the peripheral tissues. However, excess LDL-C activates the vascular endothelial cells and triggers inflammatory responses in macrophages [2]. These macrophages take up LDL, especially small dense LDL (sdLDL), and transform into foam cells, causing atherosclerosis [3]. Owing to this, reducing LDL-C levels is of primary importance for the prevention of ASCVD.
In circulating blood, CHO and TG form lipoprotein particles together with hydrophilic phospholipids and apoproteins. Apoproteins act as ligands for receptors expressed in various tissues and regulate the metabolic enzymes of lipoproteins [3]. Dietary CHO and TG absorbed from the small intestine are transported as chylomicrons by apoproteins, such as apoB-48, C-II, and E [3]. On the other hand, CHO and TG synthesized in the liver are secreted from the liver to blood as very low-density lipoprotein (VLDL) equipped with apoB-100 and E. VLDL is degraded by lipoprotein lipase (LPL) and converted to LDL via intermediate-density lipoprotein (IDL) [3]. Normally, VLDL is secreted as large VLDL1, composed mostly of TG, which is converted to small VLDL2 by LPL in the blood [4]. Normal LDL is produced from VLDL2, which is taken up via LDL receptors (LDLR) and metabolized in the liver. However, the increased influx of free fatty acids into the liver, accelerated fat synthesis, and suppressed degradation of apoB caused by insulin resistance (IR) results in increased selective production of VLDL1 [4]. As IR attenuates LPL activity, the conversion of VLDL1 to VLDL2 is also reduced, promoting sdLDL formation [4]. sdLDL appears to be more atherosclerosis-inducing because of its long residence time in the circulation, owing to its low affinity for the LDL receptor [5], susceptibility to oxidative modification [6], and tendency to permeate the vessel walls because of its small size [7]. Therefore, it has been reported that the number of sdLDL particles is a more accurate prediction of ASCVD risk than LDL-C [3, 8,9,10]. In addition, each lipoprotein particle carries one apoprotein molecule, and the amount of apoprotein reflects the number of lipoprotein particles [7]. Therefore, in addition to the level of LDL-C in the blood, the number of these particles can be determined by monitoring the concentration of apoB, which is carried by the LDLs.
Rose hip is a common name for the fruit of Rosaceae plants. The pericarps, seeds, and seed oil of Rosa canina and R. rubiginosa [11] are commercially available and used in jams, beverages, vitamin C supplements, cosmetic ingredients, and other products [12, 13]. Rose hip has traditionally been used as folk medicine in Europe for the treatment of rheumatism and osteoarthritis [14]. The acylated flavonol glycoside trans-tiliroside (1) (Fig. 1) contained in rose hip seeds has been reported to exhibit various pharmacological activities, including anti-inflammatory [15], antioxidant [16], anticancer [17, 18], tyrosinase inhibitory [19], and hepatoprotective [20] effects. We have previously reported that compound 1 potently suppresses body weight gain in mice [21, 22], increases mRNA expression of peroxisome proliferator-activated receptor α [21], which regulates lipid metabolism, and reduces intracellular TG and enhances glucose metabolism in human hepatocellular carcinoma-derived HepG2 cells [22, 23]. Studies on lipid metabolism of compound 1 have focused on the decrease in TG in the liver and muscle [21, 24], with few studies on CHO having been conducted.
In this study, we investigated the effects of compound 1 on the secretion of CHO, TG, and apoB-100 in HepG2 cells and mice. We also evaluated the inhibitory effects of nobiletin (2) [25,26,27], hesperidin (3) [28,29,30], and hesperetin (4) [31, 32] (Fig. 1), the citrus-derived components with reported anti-hyperlipidemic effects, on CHO secretion in HepG2 cells and compared their mode of action with that of compound 1.
Results
Effects of trans-tiliroside (1) and flavonoids (2–4) on CHO secretion in HepG2 cells
CHO biosynthesis in the liver is regulated by the rate-limiting enzyme, 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase, which catalyzes the synthesis of mevalonate, followed by several steps to produce CHO [33]. Chen et al. reported that the addition of mevalonate to incubation medium increased CHO production in HepG2 cells [34]; we used modified conditions in the present study. The CHO concentration in the medium was standardized based on the protein concentration measured after sonication of the cells. We observed that the addition of mevalonate to serum-free DMEM for 24 h significantly increased the amount of CHO secreted into the medium in HepG2 cells in a concentration-dependent manner, while cell viability decreased at higher concentration of mevalonate (Table S1). Thus, the concentration of mevalonate was optimized to 20 mM. As shown in Fig. 2 and Table S2, the addition of 20 mM mevalonate to serum-free DMEM for 24 h increased the amount of CHO secreted into the medium by approximately 1.3-fold. Compared to the addition of mevalonate alone, compound 1 and 2 significantly reduced the CHO concentration at 100 μM (80.8 ± 1.7% at 10 μM, 64.9 ± 0.5% at 30 μM, 27.7 ± 1.0% at 100 μM) and 30 μM (73.4 ± 2.3%), respectively. Interestingly, 100 μM of compound 1 reduced CHO production to approximately 40% of its basal level, regardless of the presence of mevalonate. Compound 3 showed no effect on CHO secretion, and compound 4 had only a slight inhibitory effect on CHO secretion (90.4 ± 2.2% at 30 μM, 89.6 ± 1.5% at 100 μM).
Effects of compounds 1–4 on CHO secretion in HepG2 cells. HepG2 cells were treated with 1–4 in 20 mM mevalonate-supplemented DMEM (FBS and phenol red-free) for 24 h. The CHO content in the medium was determined using the Amplex® Red assay. Each bar represents the mean ± S.E.M. (n = 4). Significantly different from the control, *p < 0.05, **p < 0.01 (Dunnett’s test)
Effects of trans-tiliroside (1) and nobiletin (2) on HepG2 cell viability
Next, the optimal concentrations of compounds 1 and 2 were determined. The WST-8 assay was used to determine the upper concentration limit at which compounds 1 and 2 did not affect the cells. HepG2 cells were cultured in DMEM supplemented with compounds 1 or 2 for 24 h, and the amount of soluble formazan produced 2 h after the addition of WST-8 was evaluated by measuring the absorbance at 450 nm. As shown in Fig. 3 and Table S3, absorbance significantly decreased to 74.9 ± 5.8% and 72.9 ± 3.7% at 300 μM and 100 μM, respectively, for compounds 1 and 2. Based on these results, the maximum concentration of compounds 1 and 2 was set at 100 μM and 30 μM, respectively, in subsequent experiments.
Effects of trans-tiliroside (1) and nobiletin (2) on apoB-100 secretion in HepG2 cells
For apoB-100 production, an indicator of VLDL particle count, the addition of mevalonate to the medium slightly increased its level but this was not statistically significant (Fig. 4 and Table S4). Compounds 1 and 2 showed a concentration-dependent inhibition of apoB-100 secretion compared with the addition of mevalonate alone. In particular, compound 2 showed a significant reduction, with apoB-100 production at 30 μM being 18.9 ± 1.2% that of mevalonate alone.
Effects of compounds 1 and 2 on apoB-100 secretion in HepG2 cells. HepG2 cells were treated with 1 or 2 in 20 mM mevalonate-supplemented DMEM (FBS-and phenol red-free) for 24 h. The apoB-100 content in medium were determined by ELISA using commercial kit. Each bar represents the mean ± S.E.M. (n = 4). Significantly different from the control, *p < 0.05, **p < 0.01 (Dunnett’s test)
Effects of trans-tiliroside (1) and nobiletin (2) on MTP activity
MTP, a microsomal triglyceride transfer protein, plays an important role in the secretion and assembly of lipoprotein-containing apoB in the liver and intestines [35]. MTP inhibition leads to hypolipidemic activity by preventing the secretion of apoB-containing lipoproteins [36]. Herein, MTP activity was measured as the change in fluorescence intensity due to TG transfer from donor particles containing fluorescently labeled TG in a self-quenched state to acceptor particles. Therefore, the fluorescence intensity increased as the TG was transferred. First, we examined whether each sample had a direct effect on HepG2-derived MTP (Table 1). The results revealed that compound 1 strongly inhibited MTP activity in a concentration-dependent manner and 2 had no effect on MTP activity. Table 1 also indicates the results of the evaluating MTP activity from mevalonate and sample-treated HepG2 cells. MTP activity was slightly increased in cells cultured in medium containing mevalonate. In contrast, MTP activity was not altered by pretreatment with compound 1. Interestingly, compound 2, which inhibits CHO and apoB-100 secretion from HepG2 cells as previously stated, accelerated MTP activity after 24 h of treatment.
Effects of trans-tiliroside (1) and nobiletin (2) on VLDL secretion in mouse liver
The most endogenous TGs and CHO secreted from the liver are as VLDL. As mentioned above, VLDL is converted to IDL when TG is degraded by LPL in the circulation. Therefore, the ability of the liver to secrete VLDL can be evaluated by inactivating LPL and measuring the changes in blood TG levels. In this study, we orally administered the test samples to mice for 7 days, followed by intraperitoneal administration of Triton WR-1339, an LPL inhibitor, to halt LPL activity, and measured blood TG levels over time. As a result, the blood TG concentration in the group that received 50 mg/kg of compound 2 was similar to that in the control group, while that in the group that received 50 mg/kg of 1 was significantly lower at 4 h after administration of Triton WR-1339 (Fig. 5a and Table S5). The area under the curve of blood TG concentration (AUC0–4) showed a tendency to decrease in the compound 1-administered group (Fig. 5b and Table S5). Furthermore, the concentration of apoB-100 was lower in the compound 1 group than in the control group (Fig. 5c and Table S6). No effect on the body weight of the mice was observed during the period of sample administration (Table S7).
Effects of compounds 1 and 2 on plasma TG and apoB-100 levels in Triton WR-1339-treated mice. The mice were orally administered the sample solution for 7 days. On the sixth day, the mice were deprived of food after sample administration. After 18 h, Triton WR-1339 (400 mg/kg) was injected (i.p.) 1 h after the final administration of the sample. Blood samples were collected at 0, 1, 2, and 4 h after Triton WR-1339 injection. Plasma TG concentration (a) was measured calorimetrically, and the area under the curve (AUC0–4) (b) was calculated using the trapezoidal method. Plasma apoB-100 concentration at 4 h after Triton WR-1339 injection (c) was measured using a commercial ELISA kit. Each bar represents the mean ± S.E.M. (n = 10–11). Significantly different from the control, * p < 0.05, ** p < 0.01 (Dunnett’s test)
Discussion
The currently used drugs for dyslipidemia include HMG-CoA reductase inhibitors, anion exchange resins, CHO transporter inhibitors that inhibit CHO absorption from the small intestine, fibrates that enhance lipid metabolism, polyunsaturated fatty acids such as DHA/EPA, and proprotein convertase subtilisin/kexin type 9 inhibitors [37]. However, the treatment of dyslipidemia begins with lifestyle improvements, such as smoking cessation, reduced alcohol intake, and aerobic exercise, to control LDL-C and TG to appropriate levels. If lipid management is inadequate using the aforementioned strategies. Statins, HMG-CoA reductase inhibitors, are the most frequently used drugs for dyslipidemia. However, they are associated with a high incidence of statin-associated muscle symptoms such as rhabdomyolysis, which may contribute to decreased adherence [38]. Therefore, there is an increasing need for dietary supplements and nutraceuticals (nutraceuticals) as integrative therapy in combination with pharmaceuticals or as adjunctive therapy when LDL-C does not reach target levels with drug therapy [39].
Several food-derived ingredients have been reported to exhibit lipid-lowering effects, including red yeast rice, bergamot, and artichoke leaf extract [40]. In addition, there are several meta-analyses of clinical trials, including single compounds such as flavonoids and dietary fiber, as well as their combinations [40, 41]. Various pharmacological functions have been reported for flavonoids derived from citrus fruits, especially hesperidin (3) [28], a combination of hesperetin (4) combined with rutinose. Mono-glucosyl hesperidin is a compound in which one molecule of glucose is added to compound 3 to improve its solubility in water. It is used in Japan as a food for specified health uses (FOSHU), with the aim of reducing blood TG levels [42, 43]. However, under our test conditions, both compounds 3 and 4 showed weak effects on CHO secretion from HepG2 cells (Fig. 2).
In the present study, compound 1 inhibited CHO and apoB-100 secretion in HepG2 cells (Figs. 2 and 4). Compound 1 strongly inhibited MTP activity directly in a concentration-dependent manner, and pretreatment with 1 and mevalonate-containing medium showed little effect on MTP activity (Table 1), suggesting that 1 may not affect the expression of MTP. These results suggested that MTP suppression may be one of the mechanisms underlying the inhibitory effects of compound 1 on CHO secretion. Lomitapide, developed as an MTP inhibitor, is approved for the treatment of homozygous familial hypercholesterolemia (hoFH) and strongly reduces LDL-C levels in patients with FH [44, 45]. However, inhibition of VLDL secretion induces nonalcoholic fatty liver disease (NAFLD), and the overall inhibition of MTP may result in fatty liver or liver injury [46]. Selective MTP inhibitors for the small intestine, where the expression of MTP is as high as that in the liver, are also being developed [47, 48]. Lomitapide has also been reported to cause adverse effects on the liver, but in a study using Zucker fatty rats, it was reported to reduce fat accumulation in the liver when used in combination with triiodothyronine, which enhances fatty acid β-oxidation [49]. Compound 1 lowers hepatic fat content in KK-Ay mice [24], a mouse model of type 2 diabetes, and reduces TG accumulation in HepG2 cells [23], suggesting that even if MTP inhibition suppresses VLDL secretion from the liver, it may reduce the risk of developing hepatic steatosis. In addition, the decrease in the concentration of apoB-100 (Fig. 4) and the direct inhibitory effect on MTP (Table 1) were less potent than the decrease in the CHO concentration in the medium (Fig. 2). Therefore, intracellular TG reduction may have a greater effect on VLDL formation. This is supported by the fact that compound 1 inhibited not only the increased CHO production induced by mevalonate but also basal secretion, as shown in Fig. 2.
The effect of compound 1 on VLDL secretion in vivo was evaluated in a mouse model of hyperlipidemia induced by the intraperitoneal administration of Triton WR-1339. Triton WR-1339, a nonionic surfactant, is known to induce hypercholesterolemia [50,51,52,53]. Triton WR-1339 inhibits the activity of lipoprotein lipase, causing the accumulation of triglycerides and VLDL in the blood and stimulating HMG-CoA reductase activity, significantly increasing CHO synthesis in the liver [54]. This increase in TG levels is thought to be due to the secretion of VLDL in the liver. Compound 1 inhibited the increase in blood TG and apoB-100 levels after administration of Triton WR-1339 (Fig. 5), suggesting that compound 1 inhibited VLDL secretion from the liver in vivo, similar to the results obtained in HepG2 cells. During the administration of compound 1, the body weight of the mice did not change from that of the control (Table S7). The decrease in blood TG was not considered to be due to changes in body weight or food intake. Meanwhile, the bioavailability of compound 1 has been reported in a plant extract containing 1. The maximum plasma concentration (Cmax) of compound 1 was 21.34 ng/mL (approximately 35.9 nM) when 1 was administered at a dose equivalent to 20.4 mg/kg [55]. This concentration in plasma is much lower than the effective concentration of 1 in our study. In addition, the apparent permeability of compound 1 was reported to be low in the small intestinal Caco-2 cell model [56]. However, co-incubation with kaempferol increases transfer of compound 1 from the apical to the basolateral side [56]. On the other hand, Juárez Ramírez et al. reported that compound 1 is metabolized to phloroglucinol and p-hydroxyphenyl acetic acid through kaempferol in vivo [57]. The effect of compound 1 on VLDL secretion in this study may result from the enhancement of absorption of 1 by kaempferol produced in the gastrointestinal tract and the cumulated inhibitory effect on VLDL formation in the liver by continuous administration for 7 days. Further studies are needed considering the pharmacokinetics of compound 1. Other limitations of this study include a lack of experiments on the long-term administration of compound 1 and its effects on CHO-loaded animals. The determination of the preventive effect of compound 1 on dyslipidemia via suppression of VLDL secretion still remains an intriguing question.
Compound 2 is a polymethoxyflavone with multiple hydroxyl groups substituted with methoxyl groups and is a flavonoid unique to citrus fruits. Flavonoids from citrus fruits have been reported to reduce the risk of CVD [58,59,60], with compound 2 having been shown to reduce total CHO, TG, and LDL levels in the blood [61]. In the present study, compound 2 strongly inhibited the secretion of CHO and apoB-100 in HepG2 cells but had no direct effects on MTP activity (Figs. 2 and 4, and Table 1). On the contrary, compound 2 enhanced MTP activity by pretreatment with 2 and mevalonate-containing medium for 24 h (Table 1). Compound 2 has been reported to enhance expression and activity of the LDLR, and decrease expression and activity of MTP in HepG2 cells [25, 26]. Our results suggest that compound 2 does not directly affect MTP activity in HepG2 cells, but may increase MTP expression under certain conditions. Furthermore, no effect was observed on blood TG elevation or apoB-100 concentration after Triton WR-1339 administration (Fig. 5). In a previous study that used LDLR–deficient mice, compound 2 significantly decreased blood lipid levels and suppressed TG secretion [26]. However, it did not affect the expression of MTP or Acyl-CoA: diacylglycerol acyltransferase, which is involved in the synthesis of TG, with a mechanism of action that mainly decreases the amount of TG in the liver due to increased β-oxidation [26]. This study was based on the long-term administration of compound 2 in a mixed diet. The estimated dose of compound 2 that calculated from its ratio in the diet (0.3%) and the average mouse food intake was 5–10 times higher than the dose in this study (50 mg/kg). Therefore, the reason why we could not confirm the TG secretion inhibitory effect of compound 2 may be explained by the insufficient dosage for induction of its hypolipidemic effect. The low bioavailability of compound 2 may also be another reason. Compound 2 is hardly soluble in water, but evaluation by the Parallel Artificial Membrane Permeation Assay shows high membrane permeability in the pH of 4.0 and 7.0, consistent pH in the gastrointestinal tract [62]. However, in a pharmacokinetic study in rats, the Cmax was 1.78 µg/mL (approximately 4.4 µM) when 50 mg/kg of compound 2 was administered [62]. Therefore, it is unlikely that 30 µM, the effective concentration in present study, will be reached in vivo.
Conclusion
In conclusion, trans-tiliroside (1) reduced the CHO content in the medium after culturing HepG2 cells in a mevalonate-containing medium. The concentration of apoB-100, the apoprotein that is a component of VLDL, along with CHO and TG, was reduced by administration of compound 1. In addition, compound 1 inhibited MTP activity in HepG2 cells. In vivo, compound 1 lowered blood TG and apoB-100 concentration in Triton WR-1339-induced hyperlipidemic mice. These results suggest that compound 1 inhibits CHO secretion as VLDL in the liver by inhibiting MTP activity and has potential for use for the prevention of dyslipidemia, although further experiments, such as continuous administration studies, should be conducted.
Materials and methods
Materials
Trans-tiliroside (1) was isolated from the seeds of Rosa canina. 1H- and 13C-NMR spectra are available in Supplementary Information (Figures S1 and S2). Nobiletin (2), dimethyl sulfoxide (DMSO), and (±)-3-hydroxy-3-methyl-5-pentanolide ((±)-mevalonolactone) were purchased from FUJIFILM Wako Pure Chemical Co. (Osaka, Japan). Hesperidin (3) and hesperetin (4) were purchased from Funakoshi Co., Ltd. (Tokyo, Japan). Triton WR-1339 was purchased from Sigma-Aldrich Co., LLC. (St. Louis, MO, USA). (±)-Mevalonolactone is described as mevalonate in the text because it is readily converted to mevalonate in water.
Cell culture
HepG2 cells (RCB1648) purchased from Riken Cell Bank (Tsukuba, Japan) were maintained in Minimum Essential Medium Eagle (MEM, FUJIFILM Wako) containing 10% fetal bovine serum (FBS, Thermo Fisher Scientific Inc., Waltham, MA, USA), 1% MEM nonessential amino acids (FUJIFILM Wako), 100 units/mL penicillin G, and 100 μg/mL streptomycin (FUJIFILM Wako) at 37 °C under a humidified atmosphere containing 5% CO2. Each sample was prepared in DMSO to achieve a final concentration of 0.5%.
Determination on CHO excretion in HepG2 cell containing media
HepG2 cells were seeded into a 48-well tissue culture plate (105 cells/well in 150 μL/well MEM) and cultured for 24 h. The medium was replaced with 150 μL/well of FBS-and phenol red-free DMEM (Sigma-Aldrich) containing mevalonate and a test sample. Twenty-four hours later, the supernatant was collected following determination of CHO concentrations using the Amplex® Red Cholesterol Assay Kit (Thermo Fisher Scientific) according to the manufacturer’s instruction. Briefly, 50 μL of the supernatant was transferred into a 96-well black plate, then 50 μL of the working solution containing 300 μM Amplex® Red reagent, 0.2 U/mL cholesterol esterase, 2 U/mL cholesterol oxidase, and 2 U/mL horseradish peroxidase was added, and incubated for 30 min at 37 °C. Fluorescence intensity was measured using a microplate reader (SH-9000Lab, Hitachi High-Tech Corp., Tokyo, Japan) at excitation and emission wavelengths of 560 and 590 nm, respectively. The cells were homogenized by sonication and the protein concentration was determined using a BCA protein assay kit (FUJIFILM Wako). The CHO content was standardized according to the protein concentration. Aliquots of the collected medium were used to determine apoB-100 levels.
Cell viability
Cell Counting Kit-8 (WST-8) (Dojindo, Kumamoto, Japan) was used to evaluate cell viability according to the manufacturer’s instructions. Briefly, HepG2 cells were inoculated in a 96-well tissue culture plate (104 cells/well in 150 μL/well in MEM). The cells were cultured until 90% confluence, and the medium was replaced with Dulbecco’s modified Eagle’s medium (DMEM, low glucose, FUJIFILM Wako) containing the test sample. Twenty-four hours later, 5 μL/well of WST-8 was added to each well and incubated for 2 h. The optical density was then measured using a microplate reader (Multiskan Sky, Thermo Fisher Scientific) at a test wavelength of 450 nm. Cell viability was calculated using the following equation:
where A is the absorbance of the test sample and B is the absorbance of the control.
Determination of apoB-100 by ELISA
Determination of apoB-100 in medium or mouse plasma was performed using commercial sandwich ELISA kits and an APOB ELISA Kit (Proteintech Group, Inc., Rosemont, IL, USA) for medium, and a Mouse Apo B ELISA Kit (Abcam plc, Cambridge, UK) for mouse plasma, according to the manufacturer’s instructions. The samples were diluted with the dilution buffer provided in the kit (medium: 3 × ; mouse plasma: 65,000 ×).
Determination of MTP activity
MTP activity was measured using an MTP Activity Assay Kit (Sigma-Aldrich) according to the manufacturer’s instructions. A protein homogenate containing MTP was prepared from HepG2 cells. The cells were grown in a 75 cm2 cultivation flask in MEM until they reached 90% confluence. Twenty-four hours before harvesting, the medium was replaced with fresh DMEM with or without the test sample and mevalonate (20 mM). HepG2 cells were washed thrice with PBS (−) and collected using a cell scraper. After centrifugation, PBS (−) was discarded and cells were suspended in native buffer of the Minute Total Protein Extraction Kit (Invent Biotechnologies, Inc., Plymouth, MN, USA) with protease inhibitors. The cells were disrupted by centrifugation in a shredder column, and the protein concentration in the homogenate was quantified using the BCA method. Cell homogenate (100 μg of protein), sample solution, donor particle, and acceptor particle were mixed in a 96-well black plate and incubated at 25 °C for 24 h. The increase in fluorescence was measured using a microplate reader at excitation and emission wavelengths of 465 and 535 nm, respectively.
Animals
Male ddY mice were purchased from Kiwa Laboratory Animal Co., Ltd. (Wakayama, Japan). The animals were housed at 23 ± 2 °C and fed a standard chow (MF, Oriental Yeast Co., Ltd., Tokyo, Japan) and supplied filtered tap water. The mice were used 1 week after acclimation. The Kindai University Committee for the Care and Use of Laboratory Animals approved the experimental protocol (KAPR-2023-005).
Determination of liver triglyceride (TG) secretion in a hyperlipidemic mouse model
Male ddY mice (9 weeks old) were divided into three groups and orally administered the test sample (50 mg/kg) suspended in 5% (w/v) acacia solution for 7 days. The administration volume was 10 mL/kg body weight. On the sixth day of administration, mice were fasted for 18 h. One hour after sample administration on the seventh day, 400 mg/kg of Triton WR-1339, an LPL inhibitor, which was prepared with saline, was administered intraperitoneally. Blood samples were collected into polyethylene tubes (1.5 mL) from the infraorbital venous plexus under isoflurane anesthesia before (0 h) and 1, 2, and 4 h after Triton WR-1339 administration. The blood samples were centrifuged, and plasma TG concentration was determined enzymatically using LabAssay™ Triglyceride (FUJIFILM Wako). Plasma aliquots were used for apoB measurements (vide supra).
Statistical analysis
Values are expressed as mean ± standard error of the mean (S.E.M.). For statistical analysis, one-way analysis of variance followed by Dunnett’s test was performed using JMP® 9.0.2 (SAS Institute Inc., NC, USA). Statistical significance was set at p < 0.05.
References
Liu T, Zhao D, Qi Y (2022) Global trends in the epidemiology and management of dyslipidemia. J Clin Med 11:6377. https://doi.org/10.3390/jcm11216377
Moore K, Sheedy F, Fisher E (2013) Macrophages in atherosclerosis: a dynamic balance. Nat Rev Immunol 13:709–721. https://doi.org/10.1038/nri3520
Qiao YN, Zou YL, Guo SD (2022) Low-density lipoprotein particles in atherosclerosis. Front Physiol 13:931931. https://doi.org/10.3389/fphys.2022.931931
Packard CJ, Demant T, Stewart JP, Bedford D, Caslake MJ, Schwertfeger G, Bedynek A, Shepherd J, Seidel D (2000) Apolipoprotein B metabolism and the distribution of VLDL and LDL subfractions. J Lipid Res 41:305–318. https://doi.org/10.1016/S0022-2275(20)32065-4
Hurt-Camejo E, Camejo G, Sartipy P (2000) Phospholipase A2 and small, dense low-density lipoprotein. Curr Opin Lipidol 11:465–471. https://doi.org/10.1097/00041433-200010000-00004
Alsaweed M (2021) Oxidative modification of lipoproteins: a potential role of oxidized small dense LDL in enhanced atherogenicity. J Pharm Res Int 33:118–140. https://doi.org/10.9734/JPRI/2021/v33i47A32997
Sniderman AD, Thanassoulis G, Glavinovic T, Navar AM, Pencina M, Catapano A, Ference BA (2019) Apolipoprotein B particles and cardiovascular disease: a narrative review. JAMA Cardiol 4:1287–1295. https://doi.org/10.1001/jamacardio.2019.3780
Stalenhoef AF, de Graaf J (2008) Association of fasting and nonfasting serum triglycerides with cardiovascular disease and the role of remnant-like lipoproteins and small dense LDL. Curr Opin Lipidol 19:355–361. https://doi.org/10.1097/MOL.0b013e328304b63c
Austin MA, Breslow JL, Hennekens CH, Buring JE, Willett WC, Krauss RM (1988) Low-density lipoprotein subclass patterns and risk of myocardial infarction. JAMA 260:1917–1921. https://doi.org/10.1001/jama.1988.03410130125037
Mikhailidis DP, Elisaf M, Rizzo M, Berneis K, Griffin B, Zambon A, Athyros V, de Graaf J, März W, Parhofer KG, Rini GB, Spinas GA, Tomkin GH, Tselepis AD, Wierzbicki AS, Winkler K, Florentin M, Liberopoulos E (2011) European panel on low density lipoprotein (LDL) subclasses: a statement on the pathophysiology, atherogenicity and clinical significance of LDL subclasses: executive summary. Curr Vasc Pharmacol 9:531–532. https://doi.org/10.2174/157016111796642698
Espinoza T, Valencia E, Quevedo R, Díaz O (2016) Physical and chemical properties importance of Rose hip (R. canina, R. rubiginosa): a review. Sci Agropecu 7:67–78. https://doi.org/10.17268/sci.agropecu.2016.01.07
Cohen M (2012) Rosehip—an evidence based herbal medicine for inflammation and arthritis. Aust Fam Physician 41:495–498
Patel S (2017) Rose hip as an underutilized functional food: evidence-based review. Trends Food Sci Technol 63:29–38. https://doi.org/10.1016/j.tifs.2017.03.001
Mármol I, Sánchez-de-Diego C, Jiménez-Moreno N, Ancín-Azpilicueta C, Rodríguez-Yoldi MJ (2017) Therapeutic applications of rose hips from different Rosa species. Int J Mol Sci 18:1137. https://doi.org/10.3390/ijms18061137
Corrêa WR, Serain AF, Aranha Netto L, Marinho JVN, Arena AC, de Santana F, Aquino D, Kuraoka-Oliveira ÂM, Júnior AJ, Bernal LPT, Kassuya CAL, Salvador MJ (2018) Anti-inflammatory and antioxidant properties of the extract, tiliroside, and patuletin 3-O-β-D-glucopyranoside from Pfaffia townsendii (Amaranthaceae). Evid Based Complement Alternat Med 30:6057579. https://doi.org/10.1155/2018/6057579
Sala A, Recio MC, Schinella GR, Máñez S, Giner RM, Cerdá-Nicolás M, Rosí JL (2003) Assessment of the anti-inflammatory activity and free radical scavenger activity of tiliroside. Eur J Pharmacol 461:53–61. https://doi.org/10.1016/s0014-2999(02)02953-9
Radziejewska I, Supruniuk K, Tomczyk M, Izdebska W, Borzym-Kluczyk M, Bielawska A, Bielawski K, Galicka A (2022) p-Coumaric acid, kaempferol, astragalin and tiliroside influence the expression of glycoforms in AGS gastric cancer cells. Int J Mol Sci 23:8602. https://doi.org/10.3390/ijms23158602
Han R, Yang H, Lu L, Lin L (2021) Tiliroside as a CAXII inhibitor suppresses liver cancer development and modulates E2Fs/Caspase-3 axis. Sci Rep 11:8626. https://doi.org/10.1038/s41598-021-88133-7
Lu YH, Chen J, Wei DZ, Wang ZT, Tao XY (2009) Tyrosinase inhibitory effect and inhibitory mechanism of tiliroside from raspberry. J Enzyme Inhib Med Chem 24:1154–1160. https://doi.org/10.1080/14756360802694252
Matsuda H, Ninomiya K, Shimoda H, Yoshikawa M (2002) Hepatoprotective principles from the flowers of Tilia argentea (linden): structure requirements of tiliroside and mechanisms of action. Bioorg Med Chem 10:707–712. https://doi.org/10.1016/s0968-0896(01)00321-2
Ninomiya K, Matsuda H, Kubo M, Morikawa T, Nishida N, Yoshikawa M (2007) Potent anti-obese principle from Rosa canina: structural requirements and mode of action of trans-tiliroside. Bioorg Med Chem Lett 17:3059–3064. https://doi.org/10.1016/j.bmcl.2007.03.051
Morikawa T (2023) Pharmaceutical food science: search for bio-functional molecules obtained from natural resources to prevent and ameliorate lifestyle diseases. Chem Pharm Bull 71:756–765
Morikawa T, Nagatomo A, Oka T, Miki Y, Taira N, Shibano-Kitahara M, Hori Y, Muraoka O, Ninomiya K (2019) Glucose tolerance-improving activity of helichrysoside in mice and its structural requirements for promoting glucose and lipid metabolism. Int J Mol Sci 20:6322. https://doi.org/10.3390/ijms20246322
Goto T, Teraminami A, Lee JY, Ohyama K, Funakoshi K, Kim YI, Hirai S, Uemura T, Yu R, Takahashi N, Kawada T (2012) Tiliroside, a glycosidic flavonoid, ameliorates obesity-induced metabolic disorders via activation of adiponectin signaling followed by enhancement of fatty acid oxidation in liver and skeletal muscle in obese-diabetic mice. J Nutr Biochem 23:768–776. https://doi.org/10.1016/j.jnutbio.2011.04.001
Morin B, Nichols LA, Zalasky KM, Davis JW, Manthey JA, Holland LJ (2008) The citrus flavonoids hesperetin and nobiletin differentially regulate low density lipoprotein receptor gene transcription in HepG2 liver cells. J Nutr 138:1274–1281. https://doi.org/10.1093/jn/138.7.1274
Mulvihill EE, Assini JM, Lee JK, Allister EM, Sutherland BG, Koppes JB, Sawyez CG, Edwards JY, Telford DE, Charbonneau A, St-Pierre P, Marette A, Huff MW (2011) Nobiletin attenuates VLDL overproduction, dyslipidemia, and atherosclerosis in mice with diet-induced insulin resistance. Diabetes 60:1446–1457. https://doi.org/10.2337/db10-0589
Lin Y, Vermeer MA, Bos W, van Buren L, Schuurbiers E, Miret-Catalan S, Trautwein EA (2011) Molecular structures of citrus flavonoids determine their effects on lipid metabolism in HepG2 cells by primarily suppressing apoB secretion. J Agric Food Chem 59:4496–4503. https://doi.org/10.1021/jf1044475
Khorasanian AS, Fateh ST, Gholami F, Rasaei N, Gerami H, Khayyatzadeh SS, Shiraseb F, Asbaghi O (2023) The effects of hesperidin supplementation on cardiovascular risk factors in adults: a systematic review and dose-response meta-analysis. Front Nutr 10:1177708. https://doi.org/10.3389/fnut.2023.1177708
Kumar R, Akhtar F, Rizvi SI (2021) Protective effect of hesperidin in Poloxamer-407 induced hyperlipidemic experimental rats. Biol Futur 72:201–210. https://doi.org/10.1007/s42977-020-00053-1
Wang X, Hasegawa J, Kitamura Y, Wang Z, Matsuda A, Shinoda W, Miura N, Kimura K (2011) Effects of hesperidin on the progression of hypercholesterolemia and fatty liver induced by high-cholesterol diet in rats. J Pharmacol Sci 117:129–138. https://doi.org/10.1254/jphs.11097fp
Shi L, Zou M, Zhou X, Wang S, Meng W, Lan Z (2023) Comparison of protective effects of hesperetin and pectolinarigenin on high-fat diet-induced hyperlipidemia and hepatic steatosis in Golden Syrian hamsters. Exp Anim 72:123–131. https://doi.org/10.1538/expanim.22-0115
Wilcox LJ, Borradaile NM, de Dreu LE, Huff MW (2001) Secretion of hepatocyte apoB is inhibited by the flavonoids, naringenin and hesperetin, via reduced activity and expression of ACAT2 and MTP. J Lipid Res 42:725–734. https://doi.org/10.1016/S0022-2275(20)31634-5
Luo J, Yang H, Song BL (2020) Mechanisms and regulation of cholesterol homeostasis. Nat Rev Mol Cell Biol 21:225–245. https://doi.org/10.1038/s41580-019-0190-7
Chen Y, Xu SC, Duan RD (2015) Mevalonate inhibits acid sphingomyelinase activity, increases sphingomyelin levels and inhibits cell proliferation of HepG2 and Caco-2 cells. Lipids Health Dis 14:130. https://doi.org/10.1186/s12944-015-0137-8
Gordon DA, Jamil H (2000) Progress towards understanding the role of microsomal triglyceride transfer protein in apolipoprotein-B lipoprotein assembly. Biochim Biophys Acta 1486:72–83. https://doi.org/10.1016/s1388-1981(00)00049-4
Lilly SM, Rader DJ (2007) New targets and emerging therapies for reducing LDL cholesterol. Curr Opin Lipidol 18:650–655. https://doi.org/10.1097/MOL.0b013e3282f169c6
Karantas ID, Okur ME, Okur NÜ, Siafaka PI (2021) Dyslipidemia management in 2020: an update on diagnosis and therapeutic perspectives. Endocr Metab Immune Disord Drug Targets 21:815–834. https://doi.org/10.2174/1871530320666200810144004
Cheeley MK, Saseen JJ, Agarwala A, Ravilla S, Ciffone N, Jacobson TA, Dixon DL, Maki KC (2022) NLA scientific statement on statin intolerance: a new definition and key considerations for ASCVD risk reduction in the statin intolerant patient. J Clin Lipidol 16:361–375. https://doi.org/10.1016/j.jacl.2022.05.068
Cheung B (2022) The intersection of the two complimentary fields of preventive cardiology and integrative cardiology: a fellow’s voice. Am J Prev Cardiol 12:100433. https://doi.org/10.1016/j.ajpc.2022.100433
Cheung B, Sikand G, Dineen EH, Malik S, Barseghian El-Farra A (2023) Lipid-lowering nutraceuticals for an integrative approach to dyslipidemia. J Clin Med 12:3414. https://doi.org/10.3390/jcm12103414
Cicero AFG, Colletti A, Bajraktari G, Descamps O, Djuric DM, Ezhov M, Fras Z, Katsiki N, Langlois M, Latkovskis G, Panagiotakos DB, Paragh G, Mikhailidis DP, Mitchenko O, Paulweber B, Pella D, Pitsavos C, Reiner Ž, Ray KK, Rizzo M, Sahebkar A, Serban MC, Sperling LS, Toth PP, Vinereanu D, Vrablík M, Wong ND, Banach M (2017) Lipid-lowering nutraceuticals in clinical practice: position paper from an International Lipid Expert Panel. Nutr Rev 75:731–767. https://doi.org/10.1093/nutrit/nux047
Miwa Y, Yamada M, Sunayama T, Mitsuzumi H, Tsuzaki Y, Chaen H, Mishima Y, Kibata M (2004) Effects of glucosyl hesperidin on serum lipids in hyperlipidemic subjects: preferential reduction in elevated serum triglyceride level. J Nutr Sci Vitaminol (Tokyo) 50:211–218. https://doi.org/10.3177/jnsv.50.211
Miwa Y, Mitsuzumi H, Sunayama T, Yamada M, Okada K, Kubota M, Chaen H, Mishima Y, Kibata M (2005) Glucosyl hesperidin lowers serum triglyceride level in hypertriglyceridemic subjects through the improvement of very low-density lipoprotein metabolic abnormality. J Nutr Sci Vitaminol (Tokyo) 51:460–470. https://doi.org/10.3177/jnsv.51.460
Blom DJ, Raal FJ, Santos RD, Marais AD (2019) Lomitapide and mipomersen-inhibiting microsomal triglyceride transfer protein (MTP) and apoB100 synthesis. Curr Atheroscler Rep 21:48. https://doi.org/10.1007/s11883-019-0809-3
Stefanutti C (2020) Lomitapide-a microsomal triglyceride transfer protein inhibitor for homozygous familial hypercholesterolemia. Curr Atheroscler Rep 22:38. https://doi.org/10.1007/s11883-020-00858-4
Koo SH (2013) Nonalcoholic fatty liver disease: molecular mechanisms for the hepatic steatosis. Clin Mol Hepatol 19:210–215. https://doi.org/10.3350/cmh.2013.19.3.210
Mera Y, Kawai T, Ogawa N, Odani N, Sasase T, Miyajima K, Ohta T, Kakutani M (2015) JTT-130, a novel intestine-specific inhibitor of microsomal triglyceride transfer protein, ameliorates lipid metabolism and attenuates atherosclerosis in hyperlipidemic animal models. J Pharmacol Sci 129:169–176. https://doi.org/10.1016/j.jphs.2015.10.004
Kim E, Campbell S, Schueller O, Wong E, Cole B, Kuo J, Ellis J, Ferkany J, Sweetnam P (2011) A small-molecule inhibitor of enterocytic microsomal triglyceride transfer protein, SLx-4090: biochemical, pharmacodynamic, pharmacokinetic, and safety profile. J Pharmacol Exp Ther 337:775–785. https://doi.org/10.1124/jpet.110.177527
Patel V, Joharapurkar A, Kshirsagar S, Patel M, Patel H, Savsani H, Jain M (2022) Microsomal triglyceride transfer protein inhibitor lomitapide-induced liver toxicity is ameliorated by Triiodothyronine treatment following improved bile homeostasis and β-oxidation. Toxicol Appl Pharmacol 434:115825. https://doi.org/10.1016/j.taap.2021.115825
Otway S, Robinson DS (1967) The use of a non-ionic detergent (Triton WR 1339) to determine rates of triglyceride entry into the circulation of the rat under different physiological conditions. J Physiol 190:321–332. https://doi.org/10.1113/jphysiol.1967.sp008211
Harnafi H, Serghini Caid H, el Houda BN, Aziz M, Amrani S (2008) Hypolipemic activity of polyphenol-rich extracts from Ocimum basilicum in Triton WR-1339-induced hyperlipidemic mice. Food Chem 108:205–212. https://doi.org/10.1016/j.foodchem.2007.10.062
Zarzecki MS, Araujo SM, Bortolotto VC, de Paula MT, Jesse CR, Prigol M (2014) Hypolipidemic action of chrysin on Triton WR-1339-induced hyperlipidemia in female C57BL/6 mice. Toxicol Rep 1:200–208. https://doi.org/10.1016/j.toxrep.2014.02.003
Bouhlali EDT, Hmidani A, Bourkhis B, Khouya T, Harnafi H, Filali-Zegzouti Y, Alem C (2020) Effect of Phoenix dactylifera seeds (dates) extract in triton WR-1339 and high fat diet induced hyperlipidaemia in rats: a comparison with simvastatin. J Ethnopharmacol 259:112961. https://doi.org/10.1016/j.jep.2020.112961
Janicki BW, Aron SA (1962) Effect of triton WR-1339 on lipoprotein lipase of guinea pig plasma. Proc Soc Exp Biol Med 109:507–509. https://doi.org/10.3181/00379727-109-27250
Zan T, Piao L, Wei Y, Gu Y, Liu B, Jiang D (2018) Simultaneous determination and pharmacokinetic study of three flavonoid glycosides in rat plasma by LC-MS/MS after oral administration of Rubus chingii Hu extract. Biomed Chromatogr 32:e4106. https://doi.org/10.1002/bmc.4106
Luo Z, Morgan MR, Day AJ (2015) Transport of trans-tiliroside (kaempferol-3-β-D-(6″-p-coumaroyl-glucopyranoside) and related flavonoids across Caco-2 cells, as a model of absorption and metabolism in the small intestine. Xenobiotica 45:722–730. https://doi.org/10.3109/00498254.2015.1007492
Juárez Ramírez VA, Jiménez-Beltrán MI, Zamilpa A, Herrera-Ruiz M, Abarca-Vargas R, Lombardo-Earl G, Tortoriello J, Jiménez-Ferrer E (2017) Pharmacokinetic study of biotransformation products from an anxiolytic fraction of Tilia americana. Molecules 22:1260. https://doi.org/10.3390/molecules22081260
Joshipura KJ, Hu FB, Manson JE, Stampfer MJ, Rimm EB, Speizer FE, Colditz G, Ascherio A, Rosner B, Spiegelman D, Willett WC (2001) The effect of fruit and vegetable intake on risk for coronary heart disease. Ann Intern Med 134:1106–1114. https://doi.org/10.7326/0003-4819-134-12-200106190-00010
Sánchez Macarro M, Martínez Rodríguez JP, Bernal Morell E, Pérez-Piñero S, Victoria-Montesinos D, García-Muñoz AM, Cánovas García F, Castillo Sánchez J, López-Román FJ (2020) Effect of a combination of citrus flavones and flavanones and olive polyphenols for the reduction of cardiovascular disease risk: an exploratory randomized, double-blind, placebo-controlled study in healthy subjects. Nutrients 12:1475. https://doi.org/10.3390/nu12051475
Victoria-Montesinos D, Abellán Ruiz MS, Luque Rubia AJ, Guillén Martínez D, Pérez-Piñero S, Sánchez Macarro M, García-Muñoz AM, Cánovas García F, Castillo Sánchez J, López-Román FJ (2021) Effectiveness of consumption of a combination of citrus fruit flavonoids and olive leaf polyphenols to reduce oxidation of low-density lipoprotein in treatment-naïve cardiovascular risk subjects: a randomized double-blind controlled study. Antioxidants (Basel) 10:589. https://doi.org/10.3390/antiox10040589
Kurowska EM, Manthey JA (2004) Hypolipidemic effects and absorption of citrus polymethoxylated flavones in hamsters with diet-induced hypercholesterolemia. J Agric Food Chem 52:2879–2886. https://doi.org/10.1021/jf035354z
Singh SP, Wahajuddin TD, Patel K, Jain GK (2011) Permeability determination and pharmacokinetic study of nobiletin in rat plasma and brain by validated high-performance liquid chromatography method. Fitoterapia 82:1206–1214. https://doi.org/10.1016/j.fitote.2011.08.010
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
This work was supported by the JSPS KAKENHI, Japan [Grant Numbers 22K06688 (T.M.), 18K06726 (T.M.)].
Author information
Authors and Affiliations
Contributions
Conceptualization, AN and TM; methodology, investigation, and writing, AN; review and editing, YM, TM, supervision, KM and HK. All authors have read and approved the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nagatomo, A., Kohno, M., Kawakami, H. et al. Inhibitory effect of trans-tiliroside on very low-density lipoprotein secretion in HepG2 cells and mouse liver. J Nat Med 78, 180–190 (2024). https://doi.org/10.1007/s11418-023-01756-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-023-01756-0