Abstract
Purpose of the Review
Atrial fibrillation (AF) is the most commonly encountered arrhythmia in clinical practice with constantly rising prevalence due to an overall increased ageing population with multiple co-morbidities. Since AF predisposes patients to debilitating and life-threatening complications such as stroke and coronary artery disease (CAD), much emphasis has been placed recently on early recognition and management of the disease to prevent complications. Hence, the focus of this review is deliberate on the potential risks and benefits of screening initiatives for AF and their ultimate impact on healthcare systems the associated disease burden.
Recent Findings
Due to lack of clarity on the matter, implementation of national screening programmes for AF are not currently recommended by health authorities in the UK and USA. Nevertheless, recent multicentre studies such as AF-SCREEN and STROKESTOP have concluded that select groups may benefit from screening for this disease. Research in the use of electrocardiographs (ECGs) and wearable smart devices for early detection of AF has also demonstrated a marked decrease in AF-associated thromboembolic events
Summary
Although there is a dire need for further robust clinical trials to investigate various screening modalities and develop a robust criterion for AF screening, current literature supports the implementation of a national screening programme to combat the rate of AF-associated complications which continue to challenge the healthcare system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Opinion Statement
The pathological progression of atrial fibrillation (AF) naturally predisposes patients to a significantly increased risk of thromboembolic events which could be life-threatening or, at the very least, debilitating. The level of medical and social care required for patients suffering such events puts a high degree of strain on the healthcare system. Since AF can also present in an episodic or paroxysmal manner, it can often be very difficult for patients and healthcare professionals to recognise the symptoms and ascertain a valid diagnosis. This means that AF, in a large proportion of patients, is often not identified until they present with a serious thromboembolic event. Hence, the need for early recognition of AF and appropriate interventions to prevent the complications associated with this disease is becoming increasingly apparent. One potential approach to early recognition of AF is through a national screening programme for high-risk groups within the population. Although a growing body of research is indicating that implementation of an AF screening programme is justified, there is currently no nationally recognised screening initiative for AF in the UK or USA. To combat this, our review explores a variety of screening modalities for AF such as 12-lead ECGs and photoplethysmography (PPG) based devices all of which are inexpensive and easily accessible for patients and practitioners. We continue to highlight the potential risk and benefits of a national screening programme of AF on patients and the healthcare system at large. Finally, building on the growing belief that a preventative approach to managing AF-associated complications will provide the best health outcomes for patients, our review further explores the implications of a proposed national AF screening programme on future clinical practice in this field.
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia in western countries. It affects over 1.5 million people in the UK and around 37.6 million individuals globally, as of 2019 [1, 2]. Due to the rapidly aging population and increased numbers of risk factors such as hypertension and diabetes, the prevalence of AF is expected to quickly rise over the coming years [3].
The pathophysiology underlying is complex and multi-factorial, but the prevailing consensus is that AF arises due to a triad of structural, electrical, and autonomic remodelling [4]. This facilitates ectopic firing in an atrium which can stabilize re-entry. The pulmonary veins are thought to be the main source of ectopic firing, primarily due to impaired Ca2+ handling. This is mediated by excessive Ca2+ release from the sarcoplasmic reticulum and altered ion channel function. This, coupled with the presence of an abnormal gap junction protein in AF myocytes, causes non-uniform electrical conduction and therefore arrhythmias. These triggers are important for initiation, but equally, an atrium which stabilizes re-entry is critical in AF pathogenesis [5]. This is facilitated by electrophysiological and structural abnormalities such as atrial fibrosis and shorter depolarisation times.
Due to the commonly encountered paroxysmal, asymptomatic, or mildly symptomatic nature of AF presentation within the population, a significant number of patients with AF remain undiagnosed. In England, it is estimated that around 30% of the patients living with AF are undiagnosed. Additionally, only 13% of those newly diagnosed with AF were treated with antiarrhythmic medications within the first month after diagnosis (in accordance with national guidelines), and only 16% of patients were receiving anticoagulation therapy at the time of the initial diagnosis [6]. Early detection of AF should, therefore, allow for prompt initiation of therapy with anticoagulants and appropriate medical or surgical interventions, ultimately reducing the risk of complications associated with this disorder [7].
National screening and early intervention programs have the potential to improve quality of care and reduce the morbidity and mortality associated with AF. The efficacy of national screening and intervention programmes is evident in the impact of such initiatives for diseases such as diabetes, prostate cancer, and breast cancer to name a few. Recent studies have reported a 39% reduction in breast cancer-related mortality as a direct result of the national health service (NHS) breast cancer screening programme [8]. Currently, there no national screening programme in the UK for AF, as per NICE recommendations. The current models of detecting AF patient populations have been based on risk factors associated with the disease. These include risk prediction tools such as Cohorts for aging and research in genomic epidemiology for AF (CHARGE-AF) and the CHA2DS2VASc scoring [9]. One particular drawback of these tools is that they were developed in the United States and therefore may not be as efficiently applicable to the United Kingdom or any other populations with differing demographics.
Screening programmes are an effective way of identifying high-risk individuals, prior to the development of AF or its complications. It can thus be stipulated that a lack of national screening for this disease are obstacles to effective management. Ultimately, early identification and management of AF will lead to better outcomes in AF associated morbidity and mortality. The aim of this review is to therefore explore the potential implications of an AF screening programme, both positive and negative, on its diagnosis, management, patient outcomes, and the cardiovascular services as a whole.
Pathophysiology
Despite the prevalence of AF, its concrete cause remains a matter of debate. However, numerous triggers have been identified including atrial ischemia, inflammation, haemodynamic stress, etc. [10]. These can in turn result in direct pathological alterations and remodelling of cardiac electrophysiology in three broad ways (Fig. 1) [11].
Pathophysiology of AF. The pathological processes leading to AF can be categorised into structural, autonomic, and electrical remodelling. Structural remodelling, caused by inflammation and oxidative stress, involves fibrogenesis in atrial myocardium, enlargement of the atria and valvular defects all of which impact the linear conduction of the electrical impulse to the atrioventricular node resulting in re-entry circuits. Autonomic remodelling entails increased sympathetic innervation and vagal activity, both of which alter the heart rate via increased afterdepolarisation. Finally, the mechanisms underlining electrical remodelling include a rise in intracellular Ca2+, dysfunctional ion channels and disruption of gap-junctions between cardiomyocytes. These result in arrhythmogenic action potentials which lead to AF. Original figure created with Biorender.com
Trigger and Maintenance
One phenomenon which may trigger AF the formation of an abnormal impulse being generated from an ectopic focus. Here, the depolarisation would not be governed by the SAN and therefore manifests in a tachyarrhythmia [12, 13]. A proposed electro-pathological role for ectopic foci triggering AF is calcium-channel dysfunction [14]. An increased efflux of calcium during diastole results in shortened action potential duration [15]. Consequently, this dysfunctional calcium handling can cause afterdepolarisations manifesting in paroxysms of AF. Conversely, another explanation for the increased afterdepolarisation may be the increased sympathetic stimulation [16]. Nevertheless, such paroxysmal tachycardia eventually begets further electrical remodelling. A feature of this is shortened refractory period and conduction delays allowing quicker recovery of cardiomyocytes for subsequent excitation creating re-entry circuits to maintain AF [17]. If this AF terminates within 7 days, it is classified as paroxysmal, else it is persistent.
Persistence of AF
The transition from paroxysmal to persistent AF occurs as a result of pathological structural and electrical remodelling of atrial tissues [18, 19]. One structural remodelling culpable for atrial substrate development is perhaps secondary to atrial myopathy. Progression from paroxysmal to persistent AF has demonstrated a preferential increase in myopathic substrate activity as opposed to pulmonary veins foci [20]. The second structural remodelling is atrial scarring/fibrosis, triggered by inflammation, oxidative stress, and aging [21, 22]. This creates regions of slowed conduction affecting the physiological electrical pathway [23]. Such heterogeneity in conduction velocities generates the ideal environment for re-entry circuit, aiding in the persistence of AF.
Management
Management of AF homes around two main methods of control: rate and rhythm control. These consist of various pharmacological and electro-physiotherapy strategies, with rhythm control also incorporating more invasive ablation techniques. A summary of current evidence-based management for AF is provided in Fig. 2. Recently, there has been a focus on controlling risk factors via medical therapy to reduce the risk of AF complications. These primarily include stroke and coronary artery disease (CAD). AF poses a great risk towards vascular disease, which are the biggest contributors to mortality worldwide [24].
Summary Atrial Fibrillation Management. This diagram illustrates the various management options available for AF patients, including conservative, medical and surgical rhythm, and rate control. AADs: Antiarrhythmic drugs, CCBs: Calcium-channel blockers. Original figure created with Biorender.com
Risk Factor Modification (RFM)
Primary prevention of AF relates to reducing risk factors which could eventually predispose patients to AF via pharmacological agents and lifestyle measures. These include hypertension, diabetes, obstructive sleep apnoea, hypercholesterolemia etc. Many emerging randomised control trials (RCTs) are urgently looking at RFM as an initial strategy to control the prevalence of debilitating complications of AF. A recent study found a significant reduction in AF recurrence in patients who participated in an alcohol abstinence programme [25]. Furthermore, the ARREST-AF study investigated the effect of RFM in patients receiving catheter ablation (CA). Participants who engaged with RFM demonstrated improved outcomes of ablation including AF frequency, duration, and symptoms [26]. This suggests that RFM plays a major role in improving outcomes for not only AF but also procedural outcomes.
Currently, the most common stroke prevention strategy in patients presenting with AF involves anticoagulation, offered based on a CHA2DS2-VASc score [27]. This score was designed to account for bleeding risk in AF patients. The scoring system incorporates common risk factors for stroke [28]. Currently, apixaban, edoxaban, dabigatran and rivaroxaban are licensed anticoagulants for stroke prevention in AF. Vitamin K antagonists such as warfarin may be indicated if these are contraindicated, given that INR is monitored and kept between 2–3 [29]. These have been estimated to reduce stroke risk and mortality by 64% and 26% respectively [30].
Acute Atrial Fibrillation
Acute management for AF encompasses rate and rhythm control. Pharmacological rate control can be achieved with beta-blockers and rate-limiting calcium channel blockers such as diltiazem [31]. The choice of drug varies depending on patient characteristics [10]. Whether rhythm control serves a greater purpose than rate control remains undetermined. Subsequent sub-analysis of the CASTLE-AF study revealed no significant difference between rate and rhythm control in terms of cardiovascular outcomes post-CA in AF [19].
The first step in the management of any patient acutely presenting with AF is to treat the underlying cause. These include alcohol use, thyrotoxicosis, hypovolaemia e.g., sepsis, myocarditis, pneumonia, hypertension, and electrolyte disturbances [32]. The basis of the acute management of AF without a reversible cause depends on the haemodynamic stability of the patient. If the patient is hemodynamically unstable, the aim is to appropriately resuscitate the patient followed by direct-current (DC) cardioversion to restore sinus rhythm [31]. In cases where DC cardioversion is unsuccessful, chemical cardioversion with amiodarone or flecainide IV infusion is possible. However, flecainide is contraindicated in ischaemic heart disease due to its negative inotropic effects elicited on the myocardium, increasing the risk of a non-fatal cardiac arrest [33].
Similarly, if the patient is stable and within the 48-h window of onset, they are eligible for DC cardioversion or chemical cardioversion. However, if the patient has had AF for more than 48 h, only rhythm or rate control are appropriate. If rhythm control is chosen, patients must receive anticoagulation for 3–4 weeks before elective cardioversion to reduce thromboembolism risk [34]. Moreover, transoesophageal echocardiography (TEE) may be performed to screen for any left atrial appendage thrombus where a dislodged emboli can cause an ischaemic event with DC cardioversion when sinus rhythm is restored [35]. TEE has been increasingly used as a screening tool in AF management as it allows risk stratification of patients undergoing cardioversion. In the Assessment of Cardioversion using Transoesophageal Echocardiography (ACUTE) study, randomised patients undergoing TEE with anticoagulation demonstrated a significantly lower composite endpoint of major and minor bleeding compared to conventional therapy alone [36].
Chronic Atrial Fibrillation
The main goal of chronic AF management is rate control, as well as anticoagulation to decrease the long-term stroke risk. Current evidence recommends aiming for a ventricular rate of 90 bpm using rate-limiting medication [37]. However, diltiazem is contraindicated in heart failure [38]. Instead, digoxin is used first-line and is preferred over bisoprolol if the patient has evidence of heart failure. Despite this, there is a lack of evidence for long-term rate control. Rhythm control is appropriate for younger, symptomatic patients known to have congestive heart failure, although evidence for this is debatable. The evidence for rate and rhythm control differs between the types of AF. For instance, it is well known that CA plays a key role in the treatment of paroxysmal AF for symptomatic or frequent episodes, however, the evidence for persistent AF remains limited [39]. In paroxysmal AF, a ‘pill in the pocket’ may be offered if the AF is infrequent and there is no past LV dysfunction [40]. Medications which may be trialled include sotalol or flecainide PRN.
CA is reserved as a rhythm control strategy for patients who do not respond to pharmacological medical therapy with anti-arrhythmic drugs (AAD) [10]. CA is a class IIa recommendation for rhythm control for the management of AF in patients with heart failure (HF). The primary focus of CA is to disrupt the electrical pathways which trigger AF. Primarily, CA is used to reduce the burden of AF by pulmonary vein isolation (PVI). PVI is a technique where radiofrequency is used to electrically isolate the pulmonary veins to reduce AF triggers by destroying the foci. The recommendation for CA is heavily based on AATAC and CASTLE-AF trials [41, 42]. Both demonstrated a reduced rate of mortality and hospitalisation with CA, two major burdens of AF. It is postulated that CA improves left ventricular function, as suggested by the CAMTAF trial. Ablation is an option if antiarrhythmic drugs (AADs) are not successful, currently in 1–2% of patients with AF [43].
For several years, many RCTs have investigated alternative strategies adjunctive to PVI. For persistent AF, the evidence-based management utilising CA is not as strong compared to paroxysmal AF. Therefore, research into emerging therapies is exploring new technology and imaging that identify areas of fibrosis to ablate beyond the pulmonary veins. The ERASE-AF trial investigated substrate modification (SM) as an additional ablation strategy in persistent AF [44]. The ERASE-AF trial established PVI + SM as a superior method to PVI alone in terms of a significant reduction in AF recurrence. However, a high rate of complications in the cohort undergoing adjunctive SM raises a safety concern for this procedure. Therefore, further studies investigating the safety of these novel techniques are imperative to introduce them into clinical practice to increase freedom from AF.
Screening
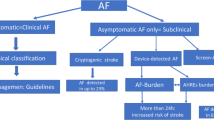
Screening aims to identify specific treatable diseases prior to the pathological manifestation of the disease [45, 46]. In the case of AF, screening aims to identify asymptomatic AF prior to adverse events such as stroke and heart failure [47]. Screening programmes are implemented nationally with the aim of health promotion, prevention, and early disease detection [48]. For a screening test to be used the test must have a high specificity for disease and be cost-effective in the long run [46]. Some of the main modalities utilised in AF screening are discussed below.
ECG
ECGs are the gold standard for the diagnosis of atrial fibrillation, ECGs detect both symptomatic and asymptomatic disease [49]. Suggesting when used for screening ECGs have high diagnostic accuracy in patients with persistent AF. A challenge is encountered in patients with paroxysmal AF where ECGs can fail to pick up paroxysmal AF [50]. When considering the feasibility of ECG use for AF screening ECGs are readily available, easily used and economically sustainable. ECGs can be performed in primary and secondary care settings allowing for both opportunistic and scheduled testing [51, 52]. ECGs testing can allow early detection of AF and early intervention in identifying patients who may benefit from rate control, rhythm control and anticoagulation [53].
Opportunistic Screening
Opportunistic screening aims to identify AF during routine encounters with healthcare professionals such as appointments with general practitioners and routine follow-up appointments [54]. Opportunistic screening can be undertaken in the form of pulse palpation, and opportunistic ECGs [55]. Various healthcare professionals within the multidisciplinary team (MDT) could be trained to carry out opportunistic testing. Opportunistic testing can identify AF without the need for specifically scheduled appointments, further reducing the cost [56, 57]. Challenges presented by opportunistic testing include discrepancies in provider knowledge, compliance, and availability of time during healthcare encounters. Further research is required to identify provider willingness to provide opportunistic testing, provider knowledge of opportunistic testing and the possibility of implementing quotas to promote opportunistic testing.
Photoplethysmography (PPG)
PPG-based devices such as smartwatches or fitness trackers harness sensory technology to detect inconsistencies in heart rhythm. These have grown in popularity with the general population independent of the potential health screening benefits [58]. A community-based study aimed at assessing the feasibility of PPG devices in the detection of AF indicated a 98% accuracy and 90% specificity of PGG devices in identifying AF within a community with 1000 individual populations [59]. Furthermore, systematic reviews aiming to identify PPG-based devices' accuracy found a pooled sensitivity and specificity of 94% and 96% receptively [60].
These studies suggest that PPG-based devices have the potential to accurately detect AF. As the potential to integrate PPG devices into routine healthcare screening is emerging, further research is required to evaluate the economic feasibility and practicality of PPG-based screening programmes.
Benefits vs Risks - “To Screen or Not to Screen…”
The primary rationale behind a potential screening programme for AF is the prompt identification of asymptomatic individuals before a thromboembolic event occurs. It is evident in literature that currently established UK national screening programmes have significantly reduced overall morbidity and mortality as well as having eased the financial and practical burden on the healthcare system in providing long-term chronic care [8, 61, 62]. Thus, it is logical to question whether a future screening programme for AF would yield similar results in health outcomes for AF patients given the severity of adverse health events associated with untreated or undiagnosed AF. Current guidance from the AF-SCREEN international collaboration recommends that all individuals above the age of 65 with health characteristics suggestive of an increased stroke risk [63]. The guidance, however, also appreciates that screening for AF at any level must be linked to a specified diagnosis and treatment pathway for it to be effective.
The implementation of a national screening programme for any condition requires a thorough evaluation of the risks associated with the condition against the costs of screening and the potential for benefits in both the short and long term. Conducting this process at a national level across multiple jurisdictions and collaborating with all relevant authorities, organisations and stakeholders makes it all the more challenging [64]. This section of the review will, therefore, aim to shed light on some of the benefits and risks associated with early national screening for AF as well as propose some suggestions on the way forward in tackling this disease.
Benefits
Although the implementation of a national screening programme for AF is not currently recommended by the UK national screening committee (UKNSC) on the basis that it is not yet clear whether screening will provide more benefit to patients with AF in comparison to the current approach, many large multi-centre trials are currently underway to highlight the potential benefits of a national screening programme for AF. Nevertheless, there are lessons to be learnt based on the evidence from various regional screening programmes. One particular multi-centre randomised control trial conducted in Sweden (STROKESTOP) showed a substantial improvement in standard of care for elderly patients with AF as a result of screening [65]. This in addition to other studies conducted across Europe deem AF screening to be a cost-effective and efficient way to improve health outcomes and reduce the risk of comorbidities associated with this condition [66]. The question, however, still remains whether similar outcomes can be practically and logistically replicated in a resource-restricted UK healthcare system.
The primary concern for all healthcare professionals involved in AF management is this associated increase in stroke risk. In addition to the five-fold increased risk of developing a stroke, patients with AF also carry a 40–90% greater risk of mortality from any subsequent thromboembolic events compared to those without AF [67]. Furthermore, the use of oral anticoagulation in patients diagnosed with AF significantly decrease in the risk of developing stroke as well as its associated mortality by 64% and 25% respectively [68]. This leaves the significant proportion of the population with undiagnosed AF at a sustained greater risk of stroke and its associated mortality making the need for a national screening programme to recognise and treat these patients even more apparent.
Another significant benefit of screening for AF is the range of modalities through which AF could be screened for. AF screening can be performed opportunistically at regular health checks or systematically across both primary and secondary care services. Additionally, the recent rise in the development of inexpensive screening devices for AF, such as wearable heart rate monitors and health watches could make the process more cost effective than it is currently projected to be [69, 70]. This is further supported by a recent study conducted in Hampshire, UK, comparing the safety and efficacy of such new inexpensive devices in screening with AF the gold-standard 12-lead ECG, reported that such consumer devices can be used to detect AF with a high accuracy in accordance with the National Institute of Clinical Excellence (NICE) guidelines [71]. This has the potential to not only reduce the potential costs of establishing a national screening programme but also pave the way for a new and more practical approach to screening for health conditions such as AF.
Risks
Whilst it is clear that a national screening programme for AF would have a number of potential benefits, there are also some important risks and limitations that need to be considered. Some of these risks apply to all screening modalities, whilst others are relevant to specific modalities.
As with all screening tests, there is a risk of false positive and false negative results [50, 72]. In the case of false positives, AF screening may lead to patients being commenced on anticoagulants, thus increasing their risk of haemorrhages [50]. False negative results give patients false reassurance and delay prompt treatment, exposing them to the risks of complications such as stroke and heart failure.
In addition, a national AF screening programme would require commitment of time and resources. With the NHS already battling long waiting lists and resource shortages, investing significant resources into a national screening programme may not be currently feasible and may not be the most cost-effective utilisation of resources [72].
Opportunistic screening is the most simple and cost-effective screening modality for AF but has a number of limitations [54]. Firstly, it relies on the healthcare professional carrying out the screening technique (e.g. pulse palpation) correctly and being able to accurately identify any abnormalities which may be suggestive of AF [50]. Individuals who are apparently healthy and do not commonly present to healthcare professionals are less likely to be tested, and so an opportunistic programme may fail to screen for AF in these individuals. Pulse palpation is not a very reliable method of screening for AF, with studies showing it has a low specificity for detecting AF in asymptomatic patients [73]. Opportunistic screening is also unlikely to identify cases of paroxysmal AF [74].
Screening modalities such as ECG and PPG also come with certain risks and limitations. Simple ECGs are unlikely to identify some forms of AF, such as paroxysmal AF. Identification of paroxysmal AF therefore requires extended continuous ECG monitoring, which can cause skin irritation and may not be tolerated by some patients [50]. PPG is shown to have a high specificity and sensitivity in identifying AF, and is particularly beneficial for detecting paroxysmal AF, but is reliant on use of wearable devices with an in-built ECG monitor [74]. As the target population for AF screening is likely to be over-65 s, the technology required for PPG screening is unlikely to be in widespread use amongst those who are at most risk for developing AF. The use of PPG in a national screening programme, though effective, would therefore come at significant cost [57].
Implications for Clinical Practice
Opportunistic screening is perhaps the most common and the most cost-effective method of diagnosing AF [54]. However, given that it is mostly conducted as a one-off event, it’s utility in diagnosing paroxysmal AF remains questionable. Usually, this screening is composed of pulse palpation and/or ECG. Whilst in the past, pulse palpation yielded high diagnostic value for AF, it now seems to be in decline with the specificity and sensitivity dropping well below 90% [73, 75]. A potential explanation for which is the reduced physical patient contact with the advent of virtual appointments as well as the health practitioner’s knowledge of pulse morphologies. Consequently, further research assessing this knowledge is imperative to increase the uptake of opportunistic screening with pulse palpation. On the contrary, ECG remains the gold standard diagnostic tool for AF [10]. Not only is it the most sensitive method, it is also relatively cheaper when compared to other options such as PPG. Therefore, implementing this in general care of patients would be relatively inexpensive owing to its availability in the primary and secondary sector [76]. However, establishing which patients' group would benefit the most form such screening remains unanswered, warranting further research.
Although limited in numbers, some studies have demonstrated a high diagnostic accuracy for AF using PPG technology, e.g., smart watches [77,78,79]. The aggregate result from such trials makes a case for potentially using smartwatches to diagnose AF in the near future - particularly paroxysmal AF. However, it should be noted that these studies are not without their limitations. The primary limitation is the lack of technological standardisation. For example, whilst the WATCH-AF trial utilised commercially available smart watches for the PPG data accusation, others used bespoke wearable devices [77,78,79]. These devices can be extremely expensive therefore the financial strain on the healthcare system for merely screening AF cannot be neglected. Consequently, it is perhaps more financially viable for healthcare systems to utilise data from individual’s personal wearable devices. Thus, the field would benefit greatly by more trials employing the aforementioned study design.
Conclusion
AF is a common cardiac arrhythmia, the prevalence of which is expected to rise over the coming years. Patients with undiagnosed AF may experience delays detecting and starting treatment for their condition, thus putting them at greater risk of complications such as stroke and CAD. The rising costs associated with AF, along with the high proportion of undiagnosed patients, suggests that it may be a good candidate for a national screening programme. AF as a condition fulfils many of the criteria for screening laid out by the World Health Organisation. Several screening modalities have been tested. Current literature supports the implementation of a national screening programme for AF allowing early detection and management of AF. Thus, reducing the rate of associated complications and could potentially reduce the burden on the healthcare system. However, further research and consideration is needed into factors such as the criteria for screening, the frequency of testing and the overall cost–benefit analysis of a screening programme.
Data Availability
No datasets were generated or analysed during the current study.
References
Disease GBD, Injury I, Prevalence C. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789–858.
England PH. Atrial Fibrillation prevalence estimates. 2019.
Chung SC, Sofat R, Acosta-Mena D, Taylor JA, Lambiase PD, Casas JP, et al. Atrial fibrillation epidemiology, disparity and healthcare contacts: a population-wide study of 5.6 million individuals. Lancet Reg Health Eur. 2021;7:100157.
Kornej J, Benjamin EJ, Magnani JW. Atrial fibrillation: global burdens and global opportunities. Heart. 2021.
Iwasaki YK, Nishida K, Kato T, Nattel S. Atrial fibrillation pathophysiology: implications for management. Circulation. 2011;124(20):2264–74.
Sekelj S, Sandler B, Johnston E, Pollock KG, Hill NR, Gordon J, et al. Detecting undiagnosed atrial fibrillation in UK primary care: Validation of a machine learning prediction algorithm in a retrospective cohort study. Eur J Prev Cardiol. 2021;28(6):598–605.
Linz D, Hermans A, Tieleman RG. Early atrial fibrillation detection and the transition to comprehensive management. Europace. 2021;23(23 Suppl 2):ii46-ii51.
Maroni R, Massat NJ, Parmar D, Dibden A, Cuzick J, Sasieni PD, et al. A case-control study to evaluate the impact of the breast screening programme on mortality in England. Br J Cancer. 2021;124(4):736–43.
Christophersen IE, Yin X, Larson MG, Lubitz SA, Magnani JW, McManus DD, et al. A comparison of the CHARGE-AF and the CHA2DS2-VASc risk scores for prediction of atrial fibrillation in the Framingham Heart Study. Am Heart J. 2016;178:45–54.
Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomstrom-Lundqvist C, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021;42(5):373–498.
Fuster V, Harrington RA, Narula J, Eapen ZJ. Hurst's The Heart, 14e. New York, NY: McGraw-Hill Education. 2017.
Haissaguerre M, Jais P, Shah DC, Takahashi A, Hocini M, Quiniou G, et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998;339(10):659–66.
Chen SA, Hsieh MH, Tai CT, Tsai CF, Prakash VS, Yu WC, et al. Initiation of atrial fibrillation by ectopic beats originating from the pulmonary veins: electrophysiological characteristics, pharmacological responses, and effects of radiofrequency ablation. Circulation. 1999;100(18):1879–86.
Nattel S, Maguy A, Le Bouter S, Yeh YH. Arrhythmogenic ion-channel remodeling in the heart: heart failure, myocardial infarction, and atrial fibrillation. Physiol Rev. 2007;87(2):425–56.
Nattel S, Heijman J, Zhou L, Dobrev D. Molecular basis of atrial fibrillation pathophysiology and therapy: A translational perspective. Circ Res. 2020;127(1):51–72.
Allessie MA, Boyden PA, Camm AJ, Kleber AG, Lab MJ, Legato MJ, et al. Pathophysiology and prevention of atrial fibrillation. Circulation. 2001;103(5):769–77.
Goette A, Honeycutt C, Langberg JJ. Electrical remodeling in atrial fibrillation. Time course and mechanisms Circulation. 1996;94(11):2968–74.
Nattel S, Dobrev D. Controversies about atrial fibrillation mechanisms: Aiming for order in chaos and whether it matters. Circ Res. 2017;120(9):1396–8.
Noseworthy PA, Van Houten HK, Gersh BJ, Packer DL, Friedman PA, Shah ND, et al. Generalizability of the CASTLE-AF trial: Catheter ablation for patients with atrial fibrillation and heart failure in routine practice. Heart Rhythm. 2020;17(7):1057–65.
Kottkamp H. Human atrial fibrillation substrate: towards a specific fibrotic atrial cardiomyopathy. Eur Heart J. 2013;34(35):2731–8.
Akoum N, Daccarett M, McGann C, Segerson N, Vergara G, Kuppahally S, et al. Atrial fibrosis helps select the appropriate patient and strategy in catheter ablation of atrial fibrillation: a DE-MRI guided approach. J Cardiovasc Electrophysiol. 2011;22(1):16–22.
Lubbers ER, Price MV, Mohler PJ. Arrhythmogenic substrates for atrial fibrillation in obesity. Front Physiol. 2018;9:1482.
Ma J, Chen Q, Ma S. Left atrial fibrosis in atrial fibrillation: Mechanisms, clinical evaluation and management. J Cell Mol Med. 2021;25(6):2764–75.
Barnes RW. Vascular holism: the epidemiology of vascular disease. Ann Vasc Surg. 1995;9(6):576–82.
Voskoboinik A, Kalman JM, De Silva A, Nicholls T, Costello B, Nanayakkara S, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382(1):20–8.
Pathak RK, Middeldorp ME, Lau DH, Mehta AB, Mahajan R, Twomey D, et al. Aggressive risk factor reduction study for atrial fibrillation and implications for the outcome of ablation: the ARREST-AF cohort study. J Am Coll Cardiol. 2014;64(21):2222–31.
Papakonstantinou PE, Tsioufis K. Optimizing anticoagulation management in atrial fibrillation: Beyond the guidelines. How and for whom? J Cardiovasc Pharmacol. 2023;81(6):397–9.
Chao TF, Liu CJ, Chen SJ, Wang KL, Lin YJ, Chang SL, et al. Atrial fibrillation and the risk of ischemic stroke: does it still matter in patients with a CHA2DS2-VASc score of 0 or 1? Stroke. 2012;43(10):2551–5.
Chen A, Stecker E, B AW. Direct oral anticoagulant use: a practical guide to common clinical challenges. J Am Heart Assoc. 2020;9(13):e017559.
Hart RG, Pearce LA, Aguilar MI. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med. 2007;146(12):857–67.
NICE. Atrial fibrillation: diagnosis and management. Atrial fibrillation: diagnosis and management. National institute for health and care excellence: Guidelines. London2022.
Staerk L, Sherer JA, Ko D, Benjamin EJ, Helm RH. Atrial fibrillation: Epidemiology, pathophysiology, and clinical outcomes. Circ Res. 2017;120(9):1501–17.
Andrikopoulos GK, Pastromas S, Tzeis S. Flecainide: Current status and perspectives in arrhythmia management. World J Cardiol. 2015;7(2):76–85.
Luca F, Giubilato S, Di Fusco SA, Piccioni L, Rao CM, Iorio A, et al. Anticoagulation in atrial fibrillation cardioversion: What is crucial to take into account. J Clin Med. 2021;10(15).
Yingchoncharoen T, Jha S, Burchill LJ, Klein AL. Transesophageal Echocardiography in Atrial Fibrillation. Card Electrophysiol Clin. 2014;6(1):43–59.
Asher CR, Klein AL, trial A. Transesophageal echocardiography to guide cardioversion in patients with atrial fibrillation: ACUTE trial update. Card Electrophysiol Rev. 2003;7(4):387–91.
Heist EK, Mansour M, Ruskin JN. Rate control in atrial fibrillation: targets, methods, resynchronization considerations. Circulation. 2011;124(24):2746–55.
Jandali MB. Safety of intravenous diltiazem in reduced ejection fraction heart failure with rapid atrial fibrillation. Clin Drug Investig. 2018;38(6):503–8.
Ha ACT, Wijeysundera HC, Qiu F, Henning K, Ahmad K, Angaran P, et al. Differences in healthcare use between patients with persistent and paroxysmal atrial fibrillation undergoing catheter-based atrial fibrillation ablation: A population-based cohort study from Ontario, Canada. J Am Heart Assoc. 2021;10(1):e016071.
Camm AJ, Savelieva I. Some patients with paroxysmal atrial fibrillation should carry flecainide or propafenone to self treat. BMJ. 2007;334(7594):637.
Di Biase L, Mohanty P, Mohanty S, Santangeli P, Trivedi C, Lakkireddy D, et al. Ablation versus amiodarone for treatment of persistent atrial fibrillation in patients with congestive heart failure and an implanted device: Results from the AATAC multicenter randomized trial. Circulation. 2016;133(17):1637–44.
Marrouche NF, Brachmann J, Andresen D, Siebels J, Boersma L, Jordaens L, et al. Catheter Ablation for Atrial Fibrillation with Heart Failure. N Engl J Med. 2018;378(5):417–27.
Hunter RJ, Berriman TJ, Diab I, Kamdar R, Richmond L, Baker V, et al. A randomized controlled trial of catheter ablation versus medical treatment of atrial fibrillation in heart failure (the CAMTAF trial). Circ Arrhythm Electrophysiol. 2014;7(1):31–8.
Huo YGT, Schönbauer R, et al. Low-voltage myocardium-guided abblation of persistent atrial fibrillation. NEJM Evidence. 2022;1(11):10.
Eriksen CU, Rotar O, Toft U, Jorgensen T. What is the effectiveness of systematic population-level screening programmes for reducing the burden of cardiovascular diseases? Copenhagen: WHO Health Evidence Network Synthesis Reports; 2021.
Dobrow MJ, Hagens V, Chafe R, Sullivan T, Rabeneck L. Consolidated principles for screening based on a systematic review and consensus process. CMAJ. 2018;190(14):E422–9.
Nesheiwat Z, Goyal A, Jagtap M. Atrial fibrillation. StatPearls. Treasure Island (FL) 2023.
Maxim LD, Niebo R, Utell MJ. Screening tests: a review with examples. Inhal Toxicol. 2014;26(13):811–28.
Cai W, Chen Y, Guo J, Han B, Shi Y, Ji L, et al. Accurate detection of atrial fibrillation from 12-lead ECG using deep neural network. Comput Biol Med. 2020;116:103378.
Engdahl J, Rosenqvist M. Large-scale screening studies for atrial fibrillation - is it worth the effort? J Intern Med. 2021;289(4):474–92.
Schnabel RB, Wallenhorst C, Engler D, Blankenberg S, Pfeiffer N, Spruenker NA, et al. Refined atrial fibrillation screening and cost-effectiveness in the German population. Heart. 2022;108(6):451–7.
Ghazal F, Aronsson M, Al-Khalili F, Rosenqvist M, Levin LA. Cost-effectiveness of screening for atrial fibrillation in a single primary care center at a 3-year follow-up. Scand Cardiovasc J. 2022;56(1):35–41.
Schnabel RB, Marinelli EA, Arbelo E, Boriani G, Boveda S, Buckley CM, et al. Early diagnosis and better rhythm management to improve outcomes in patients with atrial fibrillation: the 8th AFNET/EHRA consensus conference. Europace. 2023;25(1):6–27. Findings from this study reiterate that implimenting new evidence-based approaches to screening for AF can greatly improve health outcomes for patients.
Fay MR, Fitzmaurice DA, Freedman B. Screening of older patients for atrial fibrillation in general practice: Current evidence and its implications for future practice. Eur J Gen Pract. 2017;23(1):246–53.
Fitzmaurice DA, Hobbs FD, Jowett S, Mant J, Murray ET, Holder R, et al. Screening versus routine practice in detection of atrial fibrillation in patients aged 65 or over: cluster randomised controlled trial. BMJ. 2007;335(7616):383.
Jones ID, Lane DA, Lotto RR, Oxborough D, Neubeck L, Penson PE, et al. Supermarket/hypermarket opportunistic screening for atrial fibrillation (SHOPS-AF): A mixed methods feasibility study protocol. J Pers Med. 2022;12(4).
Kamel Boulos MN, Haywood G. Opportunistic atrial fibrillation screening and detection in “self-service health check-up stations”: a brief overview of current technology potential and possibilities. Mhealth. 2021;7:12.
Raja JM, Elsakr C, Roman S, Cave B, Pour-Ghaz I, Nanda A, et al. Apple Watch, Wearables, and Heart Rhythm: where do we stand? Ann Transl Med. 2019;7(17):417.
Mc MD, Chong JW, Soni A, Saczynski JS, Esa N, Napolitano C, et al. PULSE-SMART: Pulse-Based Arrhythmia Discrimination Using a Novel Smartphone Application. J Cardiovasc Electrophysiol. 2016;27(1):51–7.
Tieleman RG, Plantinga Y, Rinkes D, Bartels GL, Posma JL, Cator R, et al. Validation and clinical use of a novel diagnostic device for screening of atrial fibrillation. Europace. 2014;16(9):1291–5.
Koo S, Neilson LJ, Von Wagner C, Rees CJ. The NHS Bowel Cancer Screening Program: current perspectives on strategies for improvement. Risk Manag Healthc Policy. 2017;10:177–87.
O’Dowd EL, Lee RW, Akram AR, Bartlett EC, Bradley SH, Brain K, et al. Defining the road map to a UK national lung cancer screening programme. Lancet Oncol. 2023;24(5):e207–18.
Freedman B, Camm J, Calkins H, Healey JS, Rosenqvist M, Wang J, et al. Screening for Atrial Fibrillation: A Report of the AF-SCREEN International Collaboration. Circulation. 2017;135(19):1851–67.
Austoker J. Organisation of a national screening programme. Br Med Bull. 1991;47(2):416–26.
Svennberg E, Friberg L, Frykman V, Al-Khalili F, Engdahl J, Rosenqvist M. Clinical outcomes in systematic screening for atrial fibrillation (STROKESTOP): a multicentre, parallel group, unmasked, randomised controlled trial. Lancet. 2021;398(10310):1498–506. Findings from this study recognised the safety and a net benefit of screening for AF in older populations.
Jacobs MS, Kaasenbrood F, Postma MJ, van Hulst M, Tieleman RG. Cost-effectiveness of screening for atrial fibrillation in primary care with a handheld, single-lead electrocardiogram device in the Netherlands. Europace. 2018;20(1):12–8.
Jatau AI, Bereznicki LR, Wimmer BC, Bezabhe WM, Peterson GM. Improving knowledge and early detection of atrial fibrillation through a community-based opportunistic screening program: What's your beat? Int J Environ Res Public Health. 2022;19(11).
Healey JS, Connolly SJ, Gold MR, Israel CW, Van Gelder IC, Capucci A, et al. Subclinical atrial fibrillation and the risk of stroke. N Engl J Med. 2012;366(2):120–9.
Willits I, Keltie K, Craig J, Sims A. WatchBP Home A for opportunistically detecting atrial fibrillation during diagnosis and monitoring of hypertension: a NICE medical technology guidance. Appl Health Econ Health Policy. 2014;12(3):255–65.
Giles D, Draper N, Neil W. Validity of the Polar V800 heart rate monitor to measure RR intervals at rest. Eur J Appl Physiol. 2016;116(3):563–71.
Lown M, Yue AM, Shah BN, Corbett SJ, Lewith G, Stuart B, et al. Screening for atrial fibrillation using economical and accurate technology (From the SAFETY Study). Am J Cardiol. 2018;122(8):1339–44.
Welton NJ, McAleenan A, Thom HH, Davies P, Hollingworth W, Higgins JP, et al. Screening strategies for atrial fibrillation: a systematic review and cost-effectiveness analysis. Health Technol Assess. 2017;21(29):1–236.
Cooke G, Doust J, Sanders S. Is pulse palpation helpful in detecting atrial fibrillation? A systematic review J Fam Pract. 2006;55(2):130–4.
Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur J Cardiothorac Surg. 2016;50(5):e1–88.
Baneras J, Pariggiano I, Rodenas-Alesina E, Oristrell G, Escalona R, Miranda B, et al. Optimal opportunistic screening of atrial fibrillation using pulse palpation in cardiology outpatient clinics: Who and how. PLoS ONE. 2022;17(4): e0266955.
Compiet SAM, Willemsen RTA, Konings KTS, Stoffers H. Competence of general practitioners in requesting and interpreting ECGs - a case vignette study. Neth Heart J. 2018;26(7–8):377–84.
Dorr M, Nohturfft V, Brasier N, Bosshard E, Djurdjevic A, Gross S, et al. The WATCH AF Trial: SmartWATCHes for Detection of Atrial Fibrillation. JACC Clin Electrophysiol. 2019;5(2):199–208.
Proesmans T, Mortelmans C, Van Haelst R, Verbrugge F, Vandervoort P, Vaes B. Mobile phone-based use of the photoplethysmography technique to detect atrial fibrillation in primary care: Diagnostic accuracy study of the fibricheck app. JMIR Mhealth Uhealth. 2019;7(3):e12284.
Osca Asensi J, Izquierdo de Francisco MT, Cano Perez O, Tello Sancho, de Carranza MJ, Alberola Rubio J, Planells Palop C, et al. The RITHMI study: diagnostic ability of a heart rhythm monitor for automatic detection of atrial fibrillation. Rev Esp Cardiol (Engl Ed). 2021;74(7):602–7.
Acknowledgements
An early version of this review was submitted to Authorea, a pre-print server, in October 2023 at the following URL: (https://www.authorea.com/users/683475/articles/678493-screening-for-atrial-fibrillation-risks-benefits-and-implications-on-future-clinical-practice). DOI: https://doi.org/10.22541/au.169869663.34838140/v1.
Funding
None.
Author information
Authors and Affiliations
Contributions
M.H.I. directed the project, wrote and reviewed the main manuscript, provided overall support to authors responded to reviewers comments, A.M.S., J.T.J., A.R.Z.A., A.K. and M.K. contributed to the manuscript write-up and review, S.A.Z. devised the project, provided overall supervision and reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ilyas, M.H., Sharih, A.M., Tukur Jido, J. et al. Screening for Atrial Fibrillation: Risks, Benefits, and Implications On Future Clinical Practice. Curr Treat Options Cardio Med 26, 233–242 (2024). https://doi.org/10.1007/s11936-024-01047-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11936-024-01047-1