Abstract
The ionome—an important expression of the physiological state of organisms—is poorly known for mammals. The focus on particular tissues—such as liver, kidney, and bones—in biomonitoring of environmental pollution and potential deficiencies is based on widely held assumptions rather than solid knowledge of full mammalian ionomes. We examined the full ionome of Red deer (Cervus elaphus) and Wild boar (Sus scrofa), two commonly used mammals for biomonitoring, in a Dutch protected nature reserve (Veluwezoom). We used four individuals per species. We dissected 13 tissues and organs from each individuals (eight in total) of each species and measured 22 elemental concentrations in each. We assessed, for each element, how concentrations varied across tissues within and between individuals. Based on existing literature, we put our findings in the context of their function in the mammalian body. We found that the ionome was highly variable between as well as within the two species. For most elements, tissues containing the highest and lowest concentration differed between individuals. No single tissue accurately represented the accumulation of toxic elements or potential deficiencies in the bodies. Our assessment of the element’s biological roles revealed a serious lack of reference values. Our findings imply that analyses of commonly used tissues in biomonitoring do not necessarily capture bioaccumulation of toxins or potential deficiencies. We recommend establishing a centralized database of mammalian ionomes to derive reference values in future. To our knowledge, our study is one of the most complete assessments of mammalian ionomes to date.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The elemental composition of organisms—the ionome—is an important expression of their physiological state [1, 2]. Like ecological stoichiometry—the study of the balance of energy and chemical elements in ecological interactions [3,4,5] -, ionomics has been applied to population ecology to understand allocation and life history plasticity [6, 7] and population growth [8, 9]. The ionome relates to a wide variety of ecological processes, including foraging, scavenging and carrion decomposition [10, 11]. So far, ionomics and ecological stoichiometry have been widely applied to plants [e.g. 12,13,14,15], invertebrates [e.g. 16, 17], and fish [e.g. 18, 19]. Insights into the mammalian ionome, particularly large mammals, are limited.
The ionome reflects both the nutrient status and ecotoxic load of animals. While foraging across landscapes, large mammals accumulate a wide range of elemental nutrients in their bodies over their lifespan [20]. These include essential trace elements—e.g. cobalt (Co) and selenium (Se)—that are hard to gather for animals in sufficient amounts, especially in nutrient-poor areas. Large mammals may experience difficulties in acquiring sufficient amounts of essential elements, increasing the risk of deficiencies. This particularly applies to areas with unbalanced nutrient availability, for example due to decades of high nitrogen (N) deposition, which leads to acidified soils and leaching of cations to deeper horizons, hence increasing the risks of deficiencies for wildlife [21,22,23].
While foraging, large mammals may also take up potential ecotoxic elements, such as arsenic (As), cadmium (Cd) and lead (Pb). Large mammals, especially ungulates that are the main consumers of vegetation [e.g. 24], are therefore commonly used for biomonitoring of such ecotoxic elements [25]. Biomonitoring of environmental pollution—i.e. ecotoxicity—is mainly based on the assumption that ecotoxic elements would accumulate in liver, kidney, bones or hairs of wildlife, in order to be excreted from the body as fast as possible [e.g. 26,27,28]. These tissues are also most frequently used for assessing risks of elemental deficiencies of scarce elements [e.g. 29, 30]. However, it is uncertain that this assumption holds given the high variation of the mammalian ionome [e.g. 11, 31].
In Europe, ecotoxicity and potential deficiencies are commonly monitored by screening of particular tissues—mainly liver and kidney—of culled Red deer (Cervus elaphus) and Wild boar (Sus scrofa) [e.g.32,33,34,35,36,37]. For example, Nowakowska et al. [38] used liver and kidney samples of Wild boar to examine environmental levels of Se in Poland. Vikøren et al. [39] used the liver as an indication for the cupper (Cu), Co and Se concentration in wild Red deer in Norway. However, due to the lack of knowledge of the full mammalian ionome, it is uncertain whether liver and kidney are indeed the proper focal tissues for ecotoxic and scarce elements. The lack of extensive overviews of the full mammalian ionome also limit the interpretation of current ionomic studies [40].
For both ecotoxic and scarce elements, the distribution over the mammalian body thus remains poorly known and extensive overviews are missing [e.g. 41]. Most ionomic studies thus use an incomplete approach, which could unintendedly cause severe bias in the assessment of ecotoxicology and potential deficiencies in ionomic studies dealing with large mammals. Also, a systematic overview of the biological relevance of chemical elements for the mammalian body, as well as signs of deficiency or toxicity, is missing. This is needed to put existing and new ionomic insights in the context of their biological and physiological role.
Here, we aimed to gain more insight in the ionome of large mammals, particularly in how elements are distributed across the body. We measured the concentrations of 22 elements in 13 different tissues from four individuals of Red deer and four individuals of Wild boar (henceforth ‘deer’ and ‘boar’). These individuals were culled in a national park in The Netherlands, a mineral- and nutrient-poor environment that experienced decades of high N deposition, where deficiencies are likely to occur. We also tried to put our findings in the context of their function in the mammalian body, based on the literature.
Methods
Study Site and Species
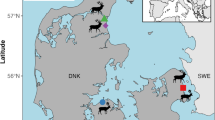
We focused on deer and boar in Veluwezoom National Park (henceforth ‘Veluwezoom’), the Netherlands (52°02’N, 6°01’E), a protected area of 5,000 ha situated on partly glacier deposits and partly on cover sands over these deposits, making the natural mineral availability limited to very scarce (mineral-poor cover sands). Veluwezoom is a former agro-silvopastoral landscape that became a national park in 1930 [42]. It contains a mosaic of dry grass-heathlands, pastures, abandoned crop fields, and woodland, grazed by free-ranging Scottish highland cattle (Bos taurus) and Icelandic horses (Equus ferus caballus)—introduced in the 1980s—as well as by Roe deer (Capreolus capreolus), Fallow deer (Dama dama), Red deer and Wild boar [43]. Due to surrounding highways former pollution (especially before 1980’s) with Pb might be possible. Samecka-Cymerman et al. [44] found elevated levels of Pb in bryophytes collected from Veluwezoom.
The Red deer is a herbivorous ruminant ungulate that is associated with woodland habitats [e.g. 45, 46]. Only the males have antlers, that cast in spring and regrow in late summer [e.g. 47]. Single-born calves are born in spring. The mating season (“rut”), which costs lots of energy, is from September to November with a peak in October [e.g. 48, 49].
The Wild boar is a monogastric, opportunistic omnivorous ungulate that is known for its highly plastic diet and their ability to adapt to diverse food and habitats [e.g. 50]. Reproduction is less seasonal and litter sizes range from one to twelve piglets, depending on many factors including the maternal body weight and summer temperature [e.g. 51].
Carcass Dissection
We used freshly culled carcasses that were obtained in the culling season 2019–2020 (culled between October 2019 and March 2020), and used four individuals of each species. Local hunters used Pb free RWS HIT ammunition. Culling at Veluwezoom is only used as an intervention against wildlife damage, i.e. culled individuals are not harvested but left to decompose in nature. For deer, we used two young females (‘RD1’ and ‘RD2’), one young male (‘RD3’), and one male calf (‘RD4’). For boar, we used two male piglets (‘WB1’ and ‘WB2’) and two female piglets (‘WB3’ and ‘WB4’). For each carcass, we dissected 13 tissues belonging to different organ systems [52]: bone; skin and hair; muscle; brain; eyes; lungs; heart; spleen; kidney; liver; pancreas; stomach (including rumen for deer); and intestines. We were able to dissect all these tissues from all the carcasses, except the pancreas for the male calf (RD4) and a male piglet (WB1).
No animals were killed for the purpose of our study. According to the Animal Welfare Officer of Wageningen University & Research, our study is not considered as experimentation on animals (Appendix 1), and therefore permitted under Dutch law.
The dissection procedure was performed in two steps. First, we dissected seven of the tissues—skin and hair, muscle, lungs, heart, spleen, kidney, and liver—in a self-made dissection room at Veluwezoom. We also collected the entire guts, head and the right hind leg that we needed to create tissue samples for the other six tissues. The carcass remains after dissection were returned to nature. Second, we further dissected the guts—pancreas, stomach, and intestines -, the head—brain and eyes -, and the hind leg—bone—in the dissection room of Wageningen Environmental Research. For the bone, we sawed a piece of bone from the lower leg, that we cleaned by boiling it a few minutes. Once dissected, we stored all the collected tissues in the freezer at minus 18 °C until we further processed them into homogeneous tissue samples.
For culling purposes only, the local game wardens occasionally provide mineral licks for the deer and corn for the boar. We analyzed these mineral licks—two different types—and the corn using the same procedure as described below since mineral licks and additional feeding might help ungulates to compensate for any deficiencies in their diet [e.g. 53]. However, since we can only speculate about the consequences for the ionome of deer and boar that we analyzed, we do not discuss the elemental composition of these salt licks and corn (Appendix 2), and potential effects on the ionome of deer and boar, in detail in this study.
Measurements
Each collected tissue was homogenized in the dissection room of Wageningen Environmental Research by grinding it with a blender. We stored about 15–25 g of the grinded tissue—three table spoons—in plastic bags. The tissue samples were frozen at minus 18 °C before we transferred them into plastic tubes for freeze-drying. The freeze-dried samples were transported to Radboud University, where we further prepared them for the chemical analysis.
We used a microwave digestion method with 5 mL 65% nitric acid (HNO3) and 2 mL 30% hydrogen peroxide (H2O2) to prepare the tissue samples for measuring the elemental concentrations with Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) and Inductively Coupled Plasma Mass Spectroscopy (ICP-MS). We measured 22 elemental concentrations for all the tissue samples. We used ICP-OES to measure 7 elements: calcium (Ca), potassium (K), magnesium (Mg), sodium (Na), phosphorus (P), sulfur (S), and silicon (Si). The other 15 elements were measured using ICP-MS: aluminum (Al), As, boron (B), Cd, Co, chromium (Cr), Cu, iron (Fe), manganese (Mn), molybdenum (Mo), nickel (Ni), Pb, Se, strontium (Sr), and zinc (Zn). We used the same devices as in Wenting et al. [11], meaning that the reported spike-and-recovery experiments also apply to this study. Correspondingly, the accuracy of these devices was guaranteed—besides using certified reference material for every microwave run—by using the following quality controls (QC): Multi element standard IV, Merck 1.11355; Phosphate standard, Merck 1.19898; Sulphate standard, Merck 1.19813; and Silicium standard, Merck 1.70236. The QC matrices were considered to correspond to the sample matrices since for both, any contamination of HNO3 and H2O2 was eliminated by using blanks (see for more details, including spike-and-recovery experiments, Wenting et al. [11]).
Results
We present our results in a descriptive way due to the low sample sizes that we used, with four individuals of each species. First, we summarized the total concentrations per element per individual in a table (Table 1). This table revealed variation in the total concentrations that we measured, which may indicate variation amongst tissues as well. Second, we listed the highest and lowest concentration measured per element for deer (Table 2) and for boar (Table 3), including the tissues in which these were found. For most elements, the tissues containing the highest and lowest concentrations varied within and between the species. Third, we used wind rose diagrams, with log(y + 1)-scale, to visualize how the total elemental concentration—as noticed in Table 1—is distribution over the 13 tissues (Figs. 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22).
As we aimed to put our findings in the context of their biological function, we report them per element separately, including a description of the biological relevance of each element for the mammalian body, as well as signs of toxicity and deficiency. We discuss the elements in alphabetical order.
Aluminum (Al)
Although Al is the third most common element in the earth’s crust, it is poorly absorbed in the animal body and there is no clear role described for this element [54, 55]. Due to long-lasting acidification of the environment due to excess N deposition mainly from agriculture, industry and traffic, pH values dropped significantly below 3.5 [22], which is much lower than the given pH value of 4.2 below which Al becomes soluble from soil aluminum oxides and hydroxides [56] and is thus potentially taken up by the vegetation, and so by animals. Although Al toxicity is associated with malabsorption of e.g. P (Allen 1984, cited in [54]), it remains unclear which and how other elements interplay with it to cause toxicity [54]. Most described symptoms of Al toxicity include inappropriately decreased feelings of thirst, refusal to swallow, and movement disorders such as hypokinesia [e.g. 57, 58].
Thurston et al. [59] and Pérez-Granados and Vaquero [55] described that Al will be mainly accumulated in bone when renal function is compromised. However, although the digestive organs tended to be important target tissues for some individuals (Fig. 1a-h; Tables 2 and 3), we found no particular tissue that contained most of the Al for the individuals we measured.
Arsenic (As)
Contrary to Al, As is well absorbed but has no specific role in the animal body [54], although Frost et al. [60] described that As may function as antibiotic and anti-coccidial in swine and poultry. By our knowledge, it has not been used for that purpose in other animals [54]. Since it is well absorbed, As toxicity is likely to occur when food is contaminated with As [e.g. 61], for instance from Cu and Pb smelters [62]. When As exposure builds up slowly, animals may get used to it and ruminants may even develop a taste for it [63]. As appears to be most toxic in inorganic form [64, 65], and affects most organs, although kidney is mentioned as the most sensitive organ for As toxicity [66]. Commonly described symptoms of As toxicity include straining, abdominal pain, bone marrow depression with anemia, skin pigmentation changes, and diarrhea containing blood and mucus [e.g. 67,68,69].
Although the As concentration in WB2 was noticeable lower compared to the other boar, we found noticeably higher As concentrations in boar compared to deer (Table 1). It has been traditionally thought that As accumulates in the hairs [e.g. 70], however skin and hair did not contain the highest concentration for any individual (Fig. 2a-h; Tables 2 and 3). As was rather randomly scattered throughout the whole body for WB3 (Fig. 2g), and the guts appeared to be the main target tissues for the other individuals (Fig. 2a-h). We were not able to detect As in any other tissue than liver for RD3 (Fig. 2c; Table 2).
Boron (B)
B is an essential trace element that is important for numerous life functions, including bone density, wound healing, embryonic development and metabolism of sex steroids and vitamin D (e.g. 71,72,73]. Although symptoms of B toxicity are poorly understood in animals [e.g. 74], acute B toxicity has been described for humans, including symptoms as nausea, vomiting, diarrhoea, and lethargy [75]. Chronic B toxicity is associated with symptoms including weight loss, reduced reproduction, and decreased appetite (Hunt 1993, cited in Nielsen [74]). However, for both humans and animals, no critical intake values have been described, which also applies to potentially B deficiency [e.g. 71]. Signs of B deficiency, however, are correlated with low immune function and increased mortality risk due to high incidence of osteoporosis [73].
Although it has been suggested that B is mostly accumulated in bone, nails and hair [e.g. 76, 77], we found it scattered throughout the whole body (Fig. 3a-h). The B concentration was the lowest for RD4 (Table 1), which tended to accumulate more in bone, eyes, intestines and stomach (Fig. 3d). B was not detectable in the bone and heart of RD3 but was present in any other tissue (Fig. 3c). Moreover, WB3 was the only individual with the highest B concentration in bone (Table 3; Fig. 3g), while the B concentration was notably the highest in the stomach and intestines for WB4 (Table 3; Fig. 3h).
Calcium (Ca)
Ca has many functions in the animal body, including the formation of skeletal tissues, transmission of nervous tissue impulses, excitation of skeletal and cardiac muscle contraction, and blood clotting (e.g. 54, 78]. Excessive dietary intake is not associated with any specific signs of [54]. Ca toxicity—aka hypercalcemia—can manifest in many different forms, including renal stones, bone pain, gastrointestinal abdominal moans, neuromuscular psychic groans, and cardiovascular issues [e.g. 79]. Ca deficiency—aka hypocalcemia—is most dangerous for young animals, which leads to reduced mineralization of new bones and therefore reduced growth [54, 80]. Like a deficiency of vitamin D or P, a deficiency of Ca can also contribute to rickets [54, 80].
We found Ca in all body tissues for all individuals (Fig. 4a-h). NRC [54] described that about 98 percent of the total Ca pool is located in the bones. However, although we found the highest Ca concentration in bone for all individuals (Tables 2 and 3), we found that the Ca concentrations in bone contributed to 86 to 94 percent of the total Ca of deer (Tables 1 and 2), and 92 to 97 percent for boar (Tables 1 and 3). WB4 had the lowest Ca concentration in its body (Table 1), of which the concentration in bone was about 92 percent of the total Ca (Tables 1 and 3), and was the only boar with a total Ca concentration lower than 300,000 µg Kg−1 (Table 1). RD1 had the highest Ca concentration of the deer, followed by RD4 (Table 1). For these individuals, the percentage of Ca stored in bone was higher compared to RD2 and RD4 (Tables 1 and 2).
Cadmium (Cd)
Cd is a highly toxic heavy metal that has no essential function in any physiological and biochemical process [e.g. 54, 81,82,83,84,85]. It accumulates in kidney, causing renal damage, from where it is very poorly and slowly excreted [54]. It is antagonistic to Zn, Cu and Fe [54, 86]. Acute Cd toxicity appears to be rare, but chronic Cd toxicity may be expressed by disturbed renal function, altered reproduction, and damaged lung function [e.g. 87].
For all individuals, we indeed found the highest Cd concentrations in the kidney, but we also detected it in liver and, for some individuals, in pancreas (Fig. 5a-h). In one wild boar (WB1), however, we found Cd more scattered throughout the body (Fig. 5e), which might be due to a lower Se concentration (Table 1) as Se can act as antioxidant for both As and Cd toxicity [88].
Cobalt (Co)
Co is a scarce but essential trace element that is an important component of vitamin B12 [54]. Although Co toxicity is less plausible due to its scarcity, symptoms of Co toxicity include reduced feed intake, hyperchromemia and eventually anemia [e.g. 89, 90]. Co deficiency seems much more likely to occur and include symptoms as fatty degeneration of the liver, anemia with pale mucous membranes, and increased susceptibility to infections due to impaired neutrophil function [91,92,93].
Although ruminants seem to be more sensitive to Co deficiency than monogastric animals [54], we found higher Co concentrations for deer than for boar (Table 1). For boar, we found comparable concentrations as found by Gasparik et al. [94], although they only measured muscle, liver and kidney. For all deer, we found the highest concentrations in liver (Fig. 6a-d; Table 2), while the guts were more prominent for boar (Fig. 6e-h; Table 3). Overall, we found Co more scattered throughout the body than expected.
Chromium (Cr)
Although some forms of Cr are known as ecotoxic metals, Cr is an important trace element for normal glucose metabolism, especially when animals experience physiologic stress [e.g. 54]. Cr toxicity is primarily linked to hexavalent Cr (Cr6+) exposure, that passes the cell walls faster and is at least five times more toxic than other Cr forms, eventually causing pathologic changes in the DNA [e.g. 54, 95,96,97]. Symptoms of Cr deficiency include reduced insulin sensitivity and reduced growth [e.g. 98]. Other symptoms may include glucose intolerance, hunger hyperglycemia, neuropathy, and reduced muscle proportion [e.g. 99, 100].
Gasparik et al. [29] reported Cr concentrations in liver, kidney, and muscle of Red deer in Slovakia, and found comparable Cr concentrations between these tissues. However, we found that these tissues did not store the major Cr pool in the body (Fig. 7a-h). Cr was more randomly scattered throughout the whole body for RD1, RD3, RD4, WB1, WB2, and WB4 (Fig. 7c-f, h). Cr was predominantly found in the intestines of RD2 and WB3 (Fig. 7b, g), the individuals with the highest Se concentrations (Table 1).
Copper (Cu)
Cu is an essential element that is a component of many enzymes in a wide range of biochemical processes that include cellular respiration, free radical detoxification and iron transport [54]. Cu toxicity has been most described for ruminants, especially cattle [e.g. 54, 101]. Commonly described symptoms include lethargy, recumbency, pale mucous membranes, excessive thirst, and jaundice [e.g. 102]. Cu deficiency has been reported as ataxia in adult deer [e.g. 103,104,105]. For ungulates in general, Cu deficiency may manifest subclinical with low Cu concentrations in liver and serum but without any other signs of poor health [e.g. 106,107,108].
Traditionally, Cu concentrations are measured in the liver and kidney [e.g. 109]. Although we found the highest concentrations in liver for most of the deer (Table 2) and in kidney for some boar (Table 3), we found Cu present throughout the whole body (Fig. 8a-h). It has been described in cattle that Cu accumulates in the liver before toxicosis becomes evident [54]. This seems unlikely for the deer and boar that we measured due to the scarcity of Cu in nature [e.g. 110]. Furthermore, levels of Cu concentration that are considered as normal seem to be unclear or case specific as McCullough [111], for instance, reported Cu levels in liver between 84 and 142 ppm and considered these to be normal, which is up to 600 times higher than the concentrations we found (Tables 1, 2 and 3).
Iron (Fe)
Fe functions as a component of heme in hemoglobin and myoglobin and is therefore very important for all vital organs in the body [54]. When Fe uptake exceeds the binding capacity, free Fe may increase in the body, which is very reactive and can cause increased free radical production and oxidative stress, which increases the need of anti-oxidants [112]. This is associated with symptoms like diarrhea, reduced feed intake and weight gain [54]. Fe levels in water are believed to be higher than those in food items and should not exceed 0.3 mg Fe per liter for humans, while animals may be able to cope with higher concentrations [54]. Fe deficiency seems to be more likely to occur, of which a major symptom is hypochromic microcytic anemia, which is the result of improper hemoglobin production [54]. Another symptom can be increased morbidity and mortality due to depressed immune responses [113]. Generally, Fe deficiency is very rare due to the ubiquitous nature of Fe in the environment including soil contamination, and requirements decrease with increase of age [91].
We found Fe in all body tissues that we measured (Fig. 9a-h). For both species, we found the highest concentrations in lungs—a vital organ (Reece et al. 2011)—and the lowest concentrations in bones (Tables 2 and 3). However, we cannot rule out that this is due to the gunshot.
Potassium (K)
K is one of the most abundant elements in the body and is important for many life functions including maintaining osmotic pressure, acid–base regulation, nerve impulse transport and muscle contraction [54]. Under natural conditions, K toxicity seems unlikely to occur [54] and it is not well defined which dietary K concentration may lead to toxicity [114]. It has been suggested that K toxicity can cause cardiac arrest [115]. A daily intake of 0.06 to 0.15 percent K of the total food intake has been reported as too low for dairy cattle [116, 117]. Signs of K deficiency include reduced feed and water intake, weight loss, loss of hair flossiness, and decreased pliability of the skin [e.g. 54].
K is thus needed in the entire body and, as such, we found it in all tissues that we measured (Fig. 10a-h). For deer, spleen tended to store the highest concentrations of K (Table 2), while we did not identify a specific tissue for boar (Table 3). Bone, and skin and hair appeared to store the lowest K concentrations for both species (Tables 2 and 3).
Magnesium (Mg)
Similar to K, Mg is an essential element that is needed for enzymatic reactions vital to every major metabolic pathway, normal nerve conduction, muscle function and bone mineral formation [54]. Animals may suffer from skeletal abnormalities when they consume excessive amounts of Mg in their diets [e.g. 118], which is unlikely to occur since most animals are able to excrete large amounts of Mg via urine [54]. Symptoms of Mg deficiency—which is often described for livestock, despite being the fourth most abundant cation in the world—have been extensively described and include muscle twitches, tremors, osteoporosis, and cramps [e.g. 119,120,121].
We found Mg throughout the whole body, with no particular target tissue (Fig. 11a-h). Mg was detectable in all tissues, except the intestines of RD3 (Fig. 11d), which we would attribute to a measurement error since this is the only missing Mg concentration in our data. We found rather similar Mg concentrations for deer and boar, ranging from 19,319 to 36,420 µg Kg−1 (Table 1).
Manganese (Mn)
Mn is an essential trace element that is important for the forming of connective tissue, bones, blood clotting and sex hormones [54]. Mn toxicity has been widely described [e.g. 122, 123], and can be associated with many symptoms including Parkinsonism, bradykinesia, tremor, impaired postural reflexes and dystonia [123, 124]. Other symptoms include, especially for ruminants, reduced food intake and growth [125]. It has been shown that rats and humans that suffered from Fe deficiency experienced increased Mn absorption [e.g. 126], and vice versa [e.g. 127, 128]. Although Mn deficiency is unlikely to occur since Mn is available in nearly all food items [129], Mn deficiency reveals most likely in the form of skeletal abnormalities [e.g. 123, 130], like enlarged joints, deformed legs with thickened and shortened long bones, and overall lameness in pigs, ruminants and poultry [131].
Mentioned target tissues for Mn include skeleton, liver and hair [132], or brain and bone [e.g. 133,134,135]. Mn has been traditionally measured in tissues as liver, kidney and muscle [e.g. 29, 30]. We found the highest concentrations in the guts for most individuals (Fig. 12a-h). We found the highest concentration in bone only for WB3 (Tables 1 and 3; Fig. 12g). Bone stored the least Mn for most of the other individuals (Tables 2 and 3). We found overall higher Mn concentrations for deer than for boar, respectively ranging from 2,785 to 5,462 µg Kg−1 and from 698 to 1,902 µg Kg−1 (Table 1).
Molybdenum (Mo)
As an essential trace element, Mo is a component of many enzymes throughout the body, including enzymes found in milk (Mills and Davis 1987, cited in [54]). Mo is antagonistic to Cu, implying that Mo toxicity can occur in the form of Cu deficiency [e.g. 54, 136, 137]. Ruminants would be more sensitive to Mo toxicity than monogastric animals [137]. Mo toxicity can cause diarrhea, anorexia, depigmentation of hair, neurological disturbances and premature death [138]. Although naturally occurring Mo deficiency has never been demonstrated in free-living animals, Mo deficiency can be the result of low Mo levels in soil, plants, drinking water and other food items [e.g. 139]. Mo is known for its anticarcinogen properties, low concentrations being associated with oesophageal cancer in particular [e.g. 139,140,141].
Mo concentrations are traditionally measured in liver and kidney [e.g. 136, 139, 142]. However, we found no specific tissue that stored the majority of the Mo pool in the body (Fig. 13a-h). The eyes turned out to have the major content for WB3 (Fig. 13g), while Mo had been hardly detected in this tissue for most of the other individuals (Fig. 13a-d, f, h). We found slightly higher Mo concentrations in deer than in boar, ranging from 47.30 to 59.71 µg Kg−1 and 35.63 to 59.42 µg Kg−1, respectively (Table 1).
Sodium (Na)
Na is an essential macro element that is important for life functions including controlling blood pressure, blood volume and water balance [54]. A proper Na and K balance is required for heart function and nerve impulse conduction [e.g. 54], and it is a major component of salts in saliva to buffer acid from ruminal fermentation [143]. When the Na concentration in the blood is too high, which can be a result of dehydration, animals can suffer from hypernatremia, manifesting in symptoms like excessive thirst or lethargy [e.g. 144, 145]. Excessive Na levels would be first detectable in the brain [e.g. 146, 147]. Animals that suffer from Na deficiency are described to have an intense craving for salt, that they show by chewing and licking various objects [e.g. 54, 148].
We detected Na in all tissues that we measured (Fig. 14a-h). The highest Na concentrations were found in the eyes of boar (Table 3), and in the eyes and brain of deer (Table 2). We found the lowest concentrations in muscle, skin and hair (Tables 2 and 3). Overall, the deer tented to have slightly higher Na concentrations compared to the boar, ranging from 145,511 to 156,760 µg Kg−1 and 117,545 to 158,860 µg Kg−1, respectively (Table 1).
Nickel (Ni)
The best described functions of the essential trace element Ni include increasing hormonal activity, lipid metabolism, and urease activity [e.g. 12, 54, 149]. One of the best described forms for Ni toxicity is found in its carcinogenic effects [e.g. 150, 151]. Other toxic effects are genotoxic, immunological, endocrine, neurogenic, cardiovascular, gastrointestinal, musculoskeletal, dermal and metabolic [150], although it remains unclear when Ni accumulation would become a problem for wildlife [152]. Naturally occurring Ni deficiency is rare due to the extremely low intake requirements [153,154,155]. Signs of Ni deficiency include depressed growth, lower reproduction, lower plasma glucose, or altered distribution of e.g. Fe, Cu, Ca, and Zn [156].
Although it has been suggested that Ni is, once ingested, distributed mostly to kidney, bone and lungs [e.g. 54], we found it more randomly throughout the body (Fig. 15a-h). For some individuals—RD2, RD3, RD4, WB2, and WB3—we indeed found relatively high concentrations in lungs (Tables 2 and 3; Fig. 15b-d, f-g). We found remarkably high concentrations of Ni in the pancreas of RD1 (Table 2; Fig. 15a), the intestines of RD2 and WB1 (Tables 2 and 3; Fig. 15b, e), and the spleen of WB4 (Table 3; Fig. 15h).
Phosphorous (P)
P has more known functions in the animal body than any other element and is located in all body cells where it is involved in nearly all energy transactions [e.g. 54, 119]. When P is excessive in the diet for a long period, it can cause problems of Ca metabolism [e.g. 54]. This is most likely to occur in monogastric animals since ruminants can tolerate a wider Ca:P ratio [54]. P deficiency seems most likely to occur when animals forage on P poor soils [e.g. 157, 158]. General signs of P deficiency include weight loss, stiff joints and muscular weakness [e.g. 54, 159]. Other symptoms can be the desire to eat wood, bones, rocks and other materials [e.g. 160, 161].
We found P in every tissue and none of the tissues in particular (Fig. 16a-h). Most individuals had the highest P concentrations in bone and the lowest concentrations in skin and hair, or eyes (Tables 2 and 3). We found similar concentrations for deer and boar, ranging from 273,821 to 376,183 µg Kg−1 and from 223,948 to 399,327 µg Kg−1, respectively (Table 1).
Lead (Pb)
Pb is the most common cause of toxicoses in animals [162]. There is no evidence for its essentiality [e.g. 54, 162]. Often described symptoms of Pb toxicity include disturbed muscular coordination, reduced cognitive performance and anemia [e.g. 162, 163]. Pb disturbs the balance between functions of other metals including Cu and Zn [164].
Accordingly other heavy metals, the liver and kidney are traditionally considered as the target organs for Pb [e.g. 30, 164, 165]. It is also believed to accumulate in bone [e.g. 86, 166]. We found bone as the major storage pool of Pb for RD1, WB1, WB2 and WB3 (Fig. 17a, e–g), whereas we found the highest concentrations in the digestive system for RD2 and WB4 (Fig. 17b, h). For RD4 and WB3, Pb was more scattered throughout the body (Fig. 17d, g), which was also the case to a lesser extent for WB1 (Fig. 17e). RD3 was the only individual with the highest Pb concentration in the lungs (Fig. 17c), with considerably higher concentrations compared to any of the other individuals (Tables 1, 2 and 3). This might be a sign of toxicosis, as the lungs are one of the vital organs [52], although reference values are missing to validate this presumption.
Sulfur (S)
S is an essential element that has many functions in the animal body, including forming several amino acids, and promoting DNA fixation and the antioxidant systems [e.g. 54, 167]. It is present in every body cell [e.g. 168]. S toxicity is most likely to manifest neurologic transmissions, causing acute symptoms including blindness, muscle twitches and recumbency [169]. Other symptoms include severe enteritis, peritoneal effusion, and petechial hemorrhages in especially kidney [170]. It may occur that S toxicity can be smelled in the breath [54]. Symptoms of S deficiency in ruminants include reduced food intake, weight and hair loss, overall weakness and death, which are all signs of digestive tract or metabolism problems [168, 171]. Since S deficiency has been mostly studied in ruminants, it is unclear whether monogastric animals experience similar symptoms.
Kierdorf et al. [172] found S in higher concentrations in deer’s antlers compared to pedicles. We found overall slightly higher S concentrations in deer compared to boar (Table 1). Skin and hair seems to contain the highest S concentrations for deer (Table 2), while we did not find any specific tissue for boar (Table 3). As S is part of every body cell, we found it distributed throughout the whole body with no particular target tissue (Fig. 18a–h).
Selenium (Se)
Se has antioxidant properties when supplied in low concentrations [e.g. 88]. It is important in several enzymes, helps to make DNA, and protects against cell damage and infections (e.g. [54]). The soil is the best Se source for all life forms, although the Se concentration in the soil does not seem to be the best indicator of Se availability for animals [173, 174]. Se toxicity is most commonly known in the form of alkali disease—aka selenosis, severely damaged hooves [e.g. 54, 174]. Se deficiency can cause white muscle disease or nutritional muscular dystrophy [e.g. 54], often resulting in death.
Kidney or liver are most commonly used as bioindicator of Se in the environment [e.g. 38, 39, 175, 176]. We found the highest Se concentration in kidney for all individuals (Tables 2 and 3), while it was also found in nearly all other tissues that we analyzed in lower concentrations (Tables 2 and 3; Fig. 19a-h). We found the highest concentrations in boar and the lowest concentrations in deer (Table 1).
Silicon (Si)
Si is only found in very trace amounts in animal bodies [54]. It has a role in connective tissue and healing from injuries [e.g. 54, 55, 177, 178]. Since Si is easily excreted, Si toxicity is unlikely to occur [179]. Si deficiency may lead to delays in growth, bone deformations and abnormal skeletal development [177]. It affects connective tissue metabolism and thus organic bone formation [180].
Although we found a lot of variation in the total Si concentration among individuals (Table 1) and it was scattered throughout all tissues we measured (Fig. 20a-h), Si slightly tended to be most concentrated in the digestive system—intestines or pancreas—for some individuals (Tables 2 and 3). Moreover, Bellés et al. [181] described that Si prevents Al absorption and reduces Al concentrations in tissues including brain, liver, bone, kidney, and spleen. We found, however, the highest Al concentrations in the individuals with the highest Si concentrations—RD2, RD4, and WB4 -, while for none of these individuals the highest Si concentrations were measured in brain, liver, bone, kidney, or spleen (Fig. 1b, d, h; Tables 1, 2 and 3).
Strontium (Sr)
Sr can be seen as the chemical analog of Ca, and as such, its major role is found in the formation and breakdown of bony material [e.g. 182,183,184]. Bony material is most commonly used to measure the Sr concentration in animals [185]. Sr toxicity seems unlikely to occur since dietary Sr can vary widely without any toxic symptoms [186]. It has been mentioned that high dietary Sr increased the risk of P deficiency [187]. Although Sr has never been shown to be an essential element, Sr has been shown to promote bone Ca and to reduce fracture rate in osteoporotic patients [186]. This might imply that osteoporosis may be related to Sr deficiency, although reference values to investigate this presumption are missing [186].
We found indeed the highest Sr concentrations in the bones of all individuals (Tables 2 and 3; Fig. 21a-h). Skibniewski et al. [188] used muscle as model tissue, however muscle belonged to the tissues with lowest Sr concentration in our analysis (Fig. 21a-h), which was for RD3 and RD4 even the tissue with the lowest Sr concentration (Table 2).
Zinc (Zn)
As a component of many enzymes, Zn affects the metabolism of carbohydrates, proteins, lipids, and nucleic acids, and it helps in regulating hormones and the immune system [e.g. 12, 54, 189]. Zn salts have been shown to protect against different forms of toxicity, including Cu toxicity [190,191,192]. Zn toxicity is most likely to occur in the form of Cu deficiency since Zn and Cu are antagonistic to each other [54, 193]. Excessive Zn uptake can also give symptoms including epigastric pain, lethargy, and fatigue [194]. Zn deficiency include symptoms as reduced feed intake and reduced growth [54].
Although most studies measure Zn in the liver, kidney, or muscle (e.g. 24, 175, 188, 189), we found Zn scattered throughout the whole body, with no particular tissue as major storage pool (Fig. 22a–h). Overall, deer tended to have slightly higher Zn concentrations compared to boar (Table 1).
Discussion and Conclusions
In this study, we aimed to gain insights in the ionome of large mammals, by measuring 22 chemical elements across 13 tissues of two ungulate species (deer and boar), and evaluating how these elements are distributed over the body. We used four individuals of deer and boar that lived in a nutrient-poor Dutch National Park (Veluwezoom), where deficiencies are most likely to occur. We found that de ionome was highly variable between and within the two species. For most elements, tissues having the highest and lowest concentration differed between individuals (Tables 2 and 3). No single tissue accurately represented the accumulation of toxic or scarce elements in the bodies. These findings imply that analyses of elemental concentrations in single tissues do not necessarily reflect bioaccumulation of toxins or deficiencies of scarce elements.
We attempted to put our findings in the context of the biological and physiological role of the elements, and noticed that the lack of reference values per element per species indeed limited our understanding and the interpretation of the measured concentrations. Such reference values are required to determine any toxicities or deficiencies. The signs of toxicities or deficiencies are element-specific [e.g. 39, 195,196,197], and can be even species-specific [e.g. 197,198,199]. Since the margins per element for wild deer and boar remain unknown, we were unable to assess toxicities or deficiencies. Therefore, the elemental concentrations that we reported here must be seen as a first step to enlarge the comprehension of the elemental composition of wild living deer and boar.
Our study showed that at the individual level most elements are rather scattered throughout the whole body. Also elements that seem to have a target tissue, e.g. kidney for Se (Fig. 19a-h), appeared to be more scattered than expected. Thus, when focusing on only a few samples of tissues traditionally mentioned as storage pools for particular elements, there is a high risk of underestimation or missing crucial information, which may lead to wrong conclusions. Moreover, we encourage further studies to collect samples of individuals in the shortest time interval, largest sample sizes, and most complete ionomes as possible.
We see four possible limitations of our study. First, the sample size per species was low given the high variability of elemental concentrations that we found within and between the species. Many more individuals would probably be needed to attain stable averages, if possible, of elemental concentrations. Second, the individuals we dissected were obtained from regular culling. This might introduce some unintended bias due to potential harvest bias in terms of sex, age and condition [e.g. 40, 200, 201]. Third, salt licks and corn—for the deer and boar, respectively—were provided occasionally to facilitate culling. Although we measured these salt licks and corn for the same elements as we did for the tissue samples that we analyzed (Appendix 2), we are only able to speculate about the effect of these supplements on the elemental concentrations we that found. For instance, the mineral licks contained more Co and Cu than the corn (Appendix 2). Since we found higher Co and Cu concentrations in deer compared to boar (Table 1), it might be possible that the mineral licks contributed to these higher concentrations, although any evidence is missing [e.g. 135]. We assume that these supplements attracted the individuals equally. Fourth, we did not include the antlers of male deer in our analysis. Therefore, we cannot address their potential function of yearly shedding in reducing the ecotoxic burden in male deer. We propose to include this as an extra tissue in future studies examining the full ionome of deer. We do not believe, however, that any of the above-mentioned limitations affect the conclusions of our study.
We encourage other scientists to execute extensive surveys of the elemental composition of wild living animals, including as much information possible about these animals and the circumstances they encountered during their lives. This will not only improve the physiological understanding of trace elements in the animal body, but also enables us to link ionomic insights to ecological processes. We propose to install an international database where all the measured elemental concentrations can be uploaded per tissue and species combination, including the sex, status (e.g. pregnancy, lactating) and age of the individuals, to enlarge the current knowledge and to potentially approach reference values in the future.
Data Availability
The complete dataset will be accessible through Figshare: https://doi.org/10.6084/m9.figshare.23633997.
References
Lahner B, Gong J, Mahmoudian M, Smith EL, Abid KB, Rogers EE, Guerinot ML, Harper JF, Ward JM, McIntyre L, Schroeder JI, Salt DE (2003) Genomic scale profiling of nutrient and trace elements in Arabidopsis thaliana. Nat Biotechnol 21:1215–1221. https://doi.org/10.1038/nbt865
Salt DE, Baxter I, Lahner B (2008) Ionomics and the study of the plant ionome. Annu Rev Plant Biol 59:709–733. https://doi.org/10.1146/annurev.arplant.59.032607.092942
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the Biology of elements from molecules to the biosphere. Princeton University Press
Elser J (2006) Biological stoichiometry: a chemical bridge between ecosystem ecology and evolutionary biology. Am Nat 168:S25–S35. https://doi.org/10.1086/509048
Raubenheimer D, Simpson SJ, Mayntz D (2009) Nutrition, ecology and nutritional ecology: toward an integrated framework. Funct Ecol:4–16. https://doi.org/10.1111/j.1365-2435.2009.01522.x
Jeyasingh PD, Weider LJ (2005) Phosphorus availability mediates plasticity in life-history traits and predator–prey interactions in Daphnia. Ecol Lett 8:1021–1028. https://doi.org/10.1111/j.1461-0248.2005.00803.x
Jeyasingh PD, Goos JM, Thompson SK, Godwin CM, Cotner JB (2017) Ecological stoichiometry beyond redfield: an ionomic perspective on elemental homeostasis. Front Microbiol 8:722. https://doi.org/10.3389/fmicb.2017.00722
Ågren GI (2004) The C: N: P stoichiometry of autotrophs–theory and observations. Ecol Lett 7:185–191. https://doi.org/10.1111/j.1461-0248.2004.00567.x
Vrede T, Dobberfuhl DR, Kooijman SALM, Elser JJ (2004) Fundamental connections among organism C: N: P stoichiometry, macromolecular composition, and growth. Ecology 85:1217–1229. https://doi.org/10.1890/02-0249
Ayotte JB, Parker KL, Arocena JM, Gillingham MP (2006) Chemical composition of lick soils: functions of soil ingestion by four ungulate species. J Mammal 87:878–888. https://doi.org/10.1644/06-MAMM-A-055R1.1
Wenting E, Siepel H, Jansen PA (2020) Stoichiometric variation within and between a terrestrial herbivorous and a semi-aquatic carnivorous mammal. J Trace Elem Med Biol 62:126622. https://doi.org/10.1016/j.jtemb.2020.126622
Hänsch R, Mendel RR (2009) Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, cl). Curr Opin Plant Biol 12:259–266. https://doi.org/10.1016/j.pbi.2009.05.006
Sha Z, Oka N, Watanabe T, Tampubolon BD, Okazaki K, Osaki M, Shinano T (2012) Ionome of soybean seed affected by previous cropping with mycorrhizal plant and manure application. J Agric Food Chem 60:9543–9552. https://doi.org/10.1021/jf3024744
Watanabe T, Maejima E, Yoshimura T, Urayama M, Yamauchi A, Owadano M, Okada R, Osaki M, Kanayama Y, Shinano T (2016) The ionomic study of vegetable crops. PLoS One 11:e0160273. https://doi.org/10.1371/journal.pone.0160273
Asis CA, Meschiari L, McConchie C (2017) Ionome balance analysis of mango fruit from orchard with and without resin canal discolouration. In XII International Mango Symposium 1244 (pp. 221–228). https://doi.org/10.17660/ActaHortic.2019.1244.33
Filipiak M, Kuszewska K, Asselman M, Denisow B, Stawiarz E, Woyciechowski M, Weiner J (2017) Ecological stoichiometry of the honeybee: Pollen diversity and adequate species composition are needed to mitigate limitations imposed on the growth and development of bees by pollen quality. PLoS One 12:e0183236. https://doi.org/10.1371/journal.pone.0183236
Filipiak M, Filipiak ZM (2022) Application of ionomics and ecological stoichiometry in conservation biology: nutrient demand and supply in a changing environment. Biol Cons 272:109622. https://doi.org/10.1016/j.biocon.2022.109622
Yoshida S, Date Y, Akama M, Kikuchi J (2014) Comparative metabolomic and ionomic approach for abundant fishes in estuarine environments of Japan. Sci Rep 4:7005. https://doi.org/10.1038/srep07005
Du F, Wang L, Yang Z, Liu P, Li D (2019) Ionomic profile and arsenic speciation in Semisulcospira cancellata, a freshwater shellfish from a mine-impacted river in China. Environ Sci Pollut Res 26:10148–10158. https://doi.org/10.1007/s11356-019-04489-4
Doughty CE, Roman J, Faurby S, Wolf A, Haque A, Bakker ES, Malhi Y, Dunning JB Jr, Svenning J-C (2016) Global nutrient transport in a world of giants. Proc Natl Acad Sci 113:868–873. https://doi.org/10.1073/pnas.1502549112
Bobbink R, Braun S, Nordin A, Power S, Schütz K, Strengbom J, Weijters M, Tomassen H (2010) Review and revision of empirical critical loads and dose-response relationships. In: Bobbink R, Hettelingh J-P (eds) UNECE Workshop on Review and Revision of Empirical Critical Loads and Dose-response Relationships. CCE, Noordwijkerhout, p 244
Siepel H, Vogels J, Bobbink R, Bijlsma RJ, Jongejans E, de Waal R, Weijters M (2018) Continuous and cumulative acidification and N deposition induce P limitation for the micro-arthropod soil fauna of mineral-poor dry heathlands. Soil Biol Biochem 119:128–134. https://doi.org/10.1016/j.soilbio.2018.01.025
Siepel H, Bobbink R, van de Riet BP, van den Burg AB, Jongejans E (2019) Long-term effects of liming on soil physico-chemical properties and micro-arthropod communities in Scotch pine forest. Biol Fertil Soils 55:675–683. https://doi.org/10.1007/s00374-019-01378-3
Reimoser F, Putman R (2011) Impacts of wild ungulates on vegetation: costs and benefits. Ungulate management in Europe: problems and practices. Cambridge University press, Cambridge, 144–191
Kuiters AT (1996) Accumulation of cadmium and lead in red deer and wild boar at the Veluwe, the Netherlands. Vet Q 18:134–135. https://doi.org/10.1080/01652176.1996.9694715
Castellino N, Aloj S (1964) Kinetics of the distribution and excretion of lead in the rat. Br J Ind Med 21:308. https://doi.org/10.1136/oem.21.4.308
Fassett DW (1975) Cadmium: biological effects and occurrence in the environment. Annu Rev Pharmacol 15:425–435
Vahter M, Concha G (2001) Role of metabolism in arsenic toxicity. Pharmacol Toxicol: MiniReview 89:1–5. https://doi.org/10.1111/j.1600-0773.2001.890101.x
Gasparik J, Massanyi P, Slamecka J, Fabis M, Jurcik R (2004) Concentration of selected metals in liver, kidney, and muscle of the red deer (Cervus elaphus). J Environ Sci Health, Part A 39:2105–2111. https://doi.org/10.1081/ESE-120039378
Malmsten A, Dalin AM, Pettersson J, Persson S (2021) Concentrations of cadmium, lead, arsenic, and some essential metals in wild boar from Sweden. Eur J Wildl Res 67:1–8. https://doi.org/10.1007/s10344-021-01460-y
Ma S, Lee SG, Kim EB, Park TJ, Seluanov A, Gorbunova V, Buffelstein R, Seravalli J, Gladyshev VN (2015) Organization of the mammalian ionome according to organ origin, lineage specialization, and longevity. Cell Rep 13:1319–1326. https://doi.org/10.1016/j.celrep.2015.10.014
Ellen G, van Loon JW, Tolsma K (1989) Copper, chromium, manganese, nickel and zinc in kidneys of cattle, pigs and sheep and in chicken livers in the Netherlands. Zeitschrift für Lebensmittel-Untersuchung und Forschung 189:534–537. https://doi.org/10.1007/BF01274272
Sobańska MA (2005) Wild boar hair (Sus scrofa) as a non-invasive indicator of mercury pollution. Sci Total Environ 339:81–88. https://doi.org/10.1016/j.scitotenv.2004.07.018
Yarsan E, Yipel M, Dikmen B, Altıntaş L, Ekici H, Köksal A (2014) Concentrations of essential and non-essential toxic trace elements in wild boar (Sus Scrofa L., 1758) tissues from southern Turkey. Bull Environ Contam Toxicol 92:10–14. https://doi.org/10.1007/s00128-013-1134-0
Cygan-Szczegielniak D, Stanek M, Stasiak K, Roślewska A, Janicki B (2018) The content of mineral elements and heavy metals in the hair of red deer (Cervus elaphus L.) from Selected Regions of Poland. Folia Biologica (Kraków) 66:133–142. https://doi.org/10.3409/fb_66-3.14
Oropesa AL, Ramos A, Gómez LJ (2022) Toxic and essential metal levels in the hair of red deer (Cervus elaphus) and wild boar (Sus scrofa) for monitoring the contamination in protected areas of South-Western Spain. Environ Sci Pollut Res 29:27430–27442. https://doi.org/10.1007/s11356-021-16439-0
Squadrone S, Robetto S, Orusa R, Griglione A, Falsetti S, Paola B, Abete MC (2022) Wildlife hair as bioindicators of metal exposure. Biol Trace Elem Res 1–8. https://doi.org/10.1007/s12011-021-03074-6
Nowakowska E, Pilarczyk B, Pilarczyk R, Tomza-Marciniak A, Bakowska M, Marciniak A (2016) Wild boars (Sus scrofa) as bioindicators of environmental levels of selenium in Poland. Pol J Vet Sci 19:4. https://doi.org/10.1515/pjvs-2016-0100
Vikøren T, Bernhoft A, Waaler T, Handeland K (2005) Liver concentrations of copper, cobalt, and selenium in wild Norwegian red deer (Cervus elaphus). J Wildl Dis 41:569–579. https://doi.org/10.7589/0090-3558-41.3.569
Wenting E, Siepel H, Christerus M, Jansen PA (2023) Ionomic variation among tissues in fallow deer (Dama dama) by sex and age. Biol Trace Elem Res, 1–15. https://doi.org/10.1007/s12011-023-03724-x
Amici A, Danieli PP, Russo C, Primi R, Ronchi B (2012) Concentrations of some toxic and trace elements in wild boar (Sus scrofa) organs and tissues in different areas of the Province of Viterbo, Central Italy. Ital J Anim Sci 11:e65. https://doi.org/10.4081/ijas.2011.e65
Kuiters AT (2005) Transition of an abandoned Dutch agrosilvopastoral landscape to'new wilderness' by extensive grazing with free-ranging cattle and horses. In Silvopastoralism and sustainable land management. Proceedings of an international congress on silvopastoralism and sustainable management held in Lugo, Spain, April 2004 (pp. 148–149). Wallingford UK: CABI Publishing
Bruinderink GG, Lammertsma DR (2001) Terreingebruik en gedrag van runderen, pony's, edelherten, reeën en wilde zwijnen in het Nationaal Park Veluwezoom van de Vereniging Natuurmonumenten (No. 343). Alterra
Samecka-Cymerman A, Kolon K, Kempers A, Jansen J, Boonen B (2005) Bioaccumulation of elements in bryophytes from Serra da Estrela, Portugal and Veluwezoom, the Netherlands (9 pp). Environ Sci Pollut Res 12:71–79. https://doi.org/10.1065/espr2004.10.219
Kamler JF, Jedrzejewski W, Jedrzejewska B (2008) Home ranges of red deer in a European old-growth forest. Am Midl Nat 159:75–82. https://doi.org/10.1674/0003-0031(2008)159[75:HRORDI]2.0.CO;2
Bywater KA, Apollonio M, Cappai N, Stephens PA (2010) Litter size and latitude in a large mammal: the wild boar Sus scrofa. Mammal Rev 40:212–220. https://doi.org/10.1111/j.1365-2907.2010.00160.x
Mitchell B, Parish T (1970) Are hummels bigger than stags with normal antlers. Deer 2:521–522
Clutton-Brock TH, Guinness FE, Albon SD (1983) The costs of reproduction to red deer hinds. J Anim Ecol, 367–383. https://doi.org/10.2307/4560
Alves J, da Silva AA, Soares AM, Fonseca C (2013) Sexual segregation in red deer: is social behaviour more important than habitat preferences? Anim Behav 85:501–509. https://doi.org/10.1016/j.anbehav.2012.12.018
Ballari SA, Barrios-García MN (2014) A review of wild boar S us scrofa diet and factors affecting food selection in native and introduced ranges. Mammal Rev 44:124–134. https://doi.org/10.1111/mam.12015
Frauendorf M, Gethöffer F, Siebert U, Keuling O (2016) The influence of environmental and physiological factors on the litter size of wild boar (Sus scrofa) in an agriculture dominated area in Germany. Sci Total Environ 541:877–882. https://doi.org/10.1016/j.scitotenv.2015.09.128
Reece JB, Urry LA, Cain ML, Wasserman SA, Minorsky PV, Jackson RB (2014) Campbell biology, vol 9. Pearson, Boston
Klaus G, Schmidg B (1998) Geophagy at natural licks and mammal ecology: a review. Mammalia 62:481–497. https://doi.org/10.1515/mamm.1998.62.4.482b
NRC (2001) Nutrient requirements of dairy cattle: 2001, National Academic Press
Pérez-Granados AM, Vaquero MP (2002) Silicon, aluminium, arsenic and lithium: essentiality and human health implications. J Nutr Health Aging 6:154–162
De Graaf MC, Bobbink R, Verbeek PJ, Roleofs JG (1997) Aluminium toxicity and tolerance in three heathland species. Water Air Soil Pollut 98:229–239. https://doi.org/10.1007/BF02047036
Hewitt CD, Savory J, Wills MR (1990) Aspects of aluminum toxicity. Clin Lab Med 10:403–422. https://doi.org/10.1016/S0272-2712(18)30576-6
Yellamma K, Saraswathamma S, Kumari BN (2010) Cholinergic system under aluminium toxicity in rat brain. Toxicol Int 17:106. https://doi.org/10.4103/0971-6580.72682
Thurston H, Gilmore GR, Swales JD (1972) Aluminium retention and toxicity in chronic renal failure. Lancet 299:881–883. https://doi.org/10.1016/s0140-6736(72)90743-x
Frost DV, Overby LR, Spruth HC (1955) Studies with arsanilic acid and related compounds. J Agric Food Chem 3:235–243
Ventura-Lima J, Bogo MR, Monserrat JM (2011) Arsenic toxicity in mammals and aquatic animals: a comparative biochemical approach. Ecotoxicol Environ Saf 74:211–218. https://doi.org/10.1016/j.ecoenv.2010.11.002
Lillie RJ (1970) Arsenic. Air pollutants affecting the performance of domestic animals—A literature review. Agricultural Handbook No. 380, Government Printing Office, U.S. Department of Agriculture, Washington, D.C.
Clarke EGC, Clarke ML (1975) Veterinary toxicology, 3rd edn. Williams & Wilkins Co., Baltimore
Shi H, Shi X, Liu KJ (2004) Oxidative mechanism of arsenic toxicity and carcinogenesis. Mol Cell Biochem 255:67–78. https://doi.org/10.1023/B:MCBI.0000007262.26044.e8
Valko MMHCM, Morris H, Cronin MTD (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208. https://doi.org/10.2174/0929867053764635
Cohen SM, Arnold LL, Eldan M, Lewis AS, Beck BD (2006) Methylated arsenicals: the implications of metabolism and carcinogenicity studies in rodents to human risk assessment. Crit Rev Toxicol 36:99–133. https://doi.org/10.1080/10408440500534230
Selby LA, Case AA, Osweiler GD, Hayes HM Jr (1977) Epidemiology and toxicology of arsenic poisoning in domestic animals. Environ Health Perspect 19:183–189. https://doi.org/10.1289/ehp.7719183
Hutton JT, Christians BL (1983) Sources, symptoms, and signs of arsenic poisoning. J Fam Pract 17:423–426
Hall AH (2002) Chronic arsenic poisoning. Toxicol Lett 128:69–72. https://doi.org/10.1016/S0378-4274(01)00534-3
Hindmarsh JT (2002) Caveats in hair analysis in chronic arsenic poisoning. Clin Biochem 35:1–11. https://doi.org/10.1016/S0009-9120(01)00282-X
Goldbach HE, Wimmer MA (2007) Boron in plants and animals: is there a role beyond cell-wall structure? J Plant Nutr Soil Sci 170:39–48. https://doi.org/10.1002/jpln.200625161
Pizzorno L (2015) Nothing boring about boron. Integrative Medicine: A Clinician’s Journal 14:35
Abdelnour SA, Abd El-Hack ME, Swelum AA, Perillo A, Losacco C (2018) The vital roles of boron in animal health and production: a comprehensive review. J Trace Elem Med Biol 50:296–304. https://doi.org/10.1016/j.jtemb.2018.07.018
Nielsen FH (1997) Boron in human and animal nutrition. Plant Soil 193:199–208. https://doi.org/10.1023/A:1004276311956
Linden CH, Hall AH, Kulig KW, Rumack BH (1986) Acute ingestions of boric acid. J Toxicol Clin Toxicol 24:269–279. https://doi.org/10.3109/15563658608992592
Ku WW, Chapin RE, Moseman RF, Brink RE, Pierce KD, Adams KY (1991) Tissue disposition of boron in male Fischer rats. Toxicol Appl Pharmacol 111:145–151. https://doi.org/10.1016/0041-008X(91)90143-3
Uluisik I, Karakaya HC, Koc A (2018) The importance of boron in biological systems. J Trace Elem Med Biol 45:156–162. https://doi.org/10.1016/j.jtemb.2017.10.008
Simon MJK, Beil FT, Rüther W, Busse B, Koehne T, Steiner M, Pogoda P, Ignatius A, Oheim R (2014) High fluoride and low calcium levels in drinking water is associated with low bone mass, reduced bone quality and fragility fractures in sheep. Osteoporos Int 25:1891–1903. https://doi.org/10.1007/s00198-014-2707-4
Carroll MF, Schade DS (2003) A practical approach to hypercalcemia. Am Fam Physician 67:1959–1966
Schenck PA, Chew DJ, Nagode LA, Rosol TJ (2006) Disorders of calcium: hypercalcemia and hypocalcemia. Fluid, electrolyte, and acid-base disorders in small animal practice 4:120–194
Patra RC, Swarup D, Sharma MC, Naresh R (2006) Trace mineral profile in blood and hair from cattle environmentally exposed to lead and cadmium around different industrial units. J Vet Med Ser A 53:511–517. https://doi.org/10.1111/j.1439-0442.2006.00868.x
Yu H, Wang J, Fang W, Yuan J, Yang Z (2006) Cadmium accumulation in different rice cultivars and screening for pollution-safe cultivars of rice. Sci Total Environ 370:302–309. https://doi.org/10.1016/j.scitotenv.2006.06.013
El-Sharaky AS, Newairy AA, Badreldeen MM, Eweda SM, Sheweita SA (2007) Protective role of selenium against renal toxicity induced by cadmium in rats. Toxicology 235:185–193. https://doi.org/10.1016/j.tox.2007.03.014
Swarup D, Naresh R, Varshney VP, Balagangatharathilagar M, Kumar P, Nandi D, Patra RC (2007) Changes in plasma hormones profile and liver function in cows naturally exposed to lead and cadmium around different industrial areas. Res Vet Sci 82:16–21. https://doi.org/10.1016/j.rvsc.2006.05.002
Djukić-Ćosić D, Jovanović MĆ, Bulat ZP, Ninković M, Maličević Ž, Matović V (2008) Relation between lipid peroxidation and iron concentration in mouse liver after acute and subacute cadmium intoxication. J Trace Elem Med Biol 22:66–72. https://doi.org/10.1016/j.jtemb.2007.09.024
Bridges CC, Zalups RK (2005) Molecular and ionic mimicry and the transport of toxic metals. Toxicol Appl Pharmacol 204:274–308. https://doi.org/10.1016/j.taap.2004.09.007
Rani A, Kumar A, Lal A, Pant M (2014) Cellular mechanisms of cadmium-induced toxicity: a review. Int J Environ Health Res 24:378–399. https://doi.org/10.1080/09603123.2013.835032
Zwolak I (2020) The role of selenium in arsenic and cadmium toxicity: an updated review of scientific literature. Biol Trace Elem Res 193:44–63. https://doi.org/10.1007/s12011-019-01691-w
Ely RE, Dunn KM, Huffman CF (1948) Cobalt toxicity in calves resulting from high oral administration. J Anim Sci 7:239–246
Keener HA, Percival GP, Mobbow KS, Etus GH (1949) Cobalt tolerance in young dairy cattle. J Dairy Sci 32:527–533
Underwood EJ (1981) The mineral nutrition of livestock, 2nd edn. Commonwealth Agricultural Bureaux, Slough
MacPherson A, Gray D, Mitchell GBB, Taylor CN (1987) Ostertagia infection and neutrophil function in cobalt-deficient and cobalt-supplemented cattle. Br Vet J 143:348–353. https://doi.org/10.1016/0007-1935(87)90069-8
Paterson JE, MacPherson A (1990) The influence of a low cobalt intake on the neutrophil function and severity of Ostertagia infection in cattle. Br Vet J 146:519–530. https://doi.org/10.1016/0007-1935(90)90055-8
Gasparik J, Dobias M, Capcarova M, Smehyl P, Slamecka J, Bujko J, Gasparik J Jr (2012) Concentration of cadmium, mercury, zinc, copper and cobalt in the tissues of wild boar (Sus scrofa) hunted in the western Slovakia. J Environ Sci Health, Part A 47:1212–1216. https://doi.org/10.1080/10934529.2012.672065
Jennette KW (1979) Chromate metabolism in liver microsomes. Biol Trace Elem Res 1:55–62. https://doi.org/10.1007/BF02783843
Alexander J (1993) Toxicity versus essentially of chromium. Scand J Work Environ Health 19:126–127
Ryberg D, Alexander J (1990) Mechanisms of chromium toxicity in mitochondria. Chem Biol Interact 75:141–151. https://doi.org/10.1016/0009-2797(90)90114-3
Lindemann MD (1996) Organic chromium–the missing link in farm animal nutrition. Feeding Times 1:8
Anderson RA (1994) Stress effects on chromium nutrition of humans and farm animals. Biotechnology in the feed industry 267
Pechova A, Pavlata L (2007) Chromium as an essential nutrient: a review. Vet Med 52:1
Todd JR (1969) Chronic copper toxicity of ruminants. Proc Nutr Soc 28:189–198. https://doi.org/10.1079/PNS19690037
Suciu I, Prodan L, Lazar V (1981) Research on copper poisoning. Med Lav 72:190–197
Barlow RM, Butler EJ, Purves D (1964) An ataxic condition in red deer (Cervus elaphus). J Comp Pathol 74:519–529. https://doi.org/10.1016/S0368-1742(64)80058-8
Terlecki S, Done JT, Clegg FG (1964) Enzootic ataxia of red deer. Br Vet J 120:311–321
Reid TC, McAllum HJF, Johnstone PD (1980) Liver copper concentrations in red deer (Cervus elaphus) and wapiti (C canadensis) in New Zealand. Res Vet Sci 28:261–262. https://doi.org/10.1016/S0034-5288(18)32760-7
Bingley JB, Anderson N (1972) Clinically silent hypocuprosis and the effect of molybdenum loading on beef calves in Gippsland, Victoria. Aust J Agric Res 23:885–904. https://doi.org/10.1071/AR9720885
Thornton I, Kershaw GF, Davies MK (1972) An investigation into copper deficiency in cattle in the Southern Pennines. I. Identification of suspect areas using geochemical reconnaissance followed by blood copper surveys. J Agric Sci 78:157–163. https://doi.org/10.1017/S0021859600087839
Gogan PP, Jessup DA, Akenson M (1989) Copper deficiency in tule elk at Point Reyes, California. Rangeland Ecol Manag/J Range Manag Arch 42:233–238
Handeland K, Viljugrein H, Lierhagen S, Opland M, Tarpai A, Vikøren T (2017) Low copper levels associated with low Carcass Weight in Wild Red Deer (Cervus elaphus) in Norway. J Wildl Dis 53:176–180. https://doi.org/10.7589/2016-02-037
López-Alonso M, Miranda M (2020) Copper supplementation, a challenge in cattle. Animals 10:1890. https://doi.org/10.3390/ani10101890
McCullough DR (1969) The tule elk: its history, behavior, and ecology. Univ Calif Publ Zool 88:1–209
Halliwell B (1987) Oxidants and human disease: Some new concepts. FASEB J 1:358–364. https://doi.org/10.1096/fasebj.1.5.2824268
Möllerberg L, Moreno-Lopez J (1975) The response of normal and iron anemic calves to nasal infection with an attenuated strain of parainfluenza-3 virus. Acta Vet Scand 16:186
Ward GM (1966) Potassium metabolism of domestic ruminants—a review. J Dairy Sci 49:268–276
Ward GM (1966) Oral potassium chloride fatal to a cow. J Am Vet Med Assoc 148:543–544
Pradhan K, Hemken RW (1968) Potassium depletion in lactating dairy cows. J Dairy Sci 51:1377–1381. https://doi.org/10.3168/jds.S0022-0302(68)87198-X
Mallone P, Beede DK, Schneider PL, Caputo SJ, Wilcox CJ (1982) Acute response of lactating Holstein cows to dietary potassium deficiency. J Dairy Sci 61:112
Lee SR, Britton WM, Rowland GN (1980) Magnesium toxicity: bone lesions. Poult Sci 59:2403–2411. https://doi.org/10.3382/ps.0592403
Davis GK (1968) Mineral elements in the nutrition of larger mammals. Am Zool 8:169–174. https://doi.org/10.1093/icb/8.1.169
Rude RK, Gruber HE (2004) Magnesium deficiency and osteoporosis: animal and human observations. J Nutr Biochem 15:710–716. https://doi.org/10.1016/j.jnutbio.2004.08.001
Rude RK, Singer FR, Gruber HE (2009) Skeletal and hormonal effects of magnesium deficiency. J Am Coll Nutr 28:131–141. https://doi.org/10.1080/07315724.2009.10719764
Keen CL, Lonnerdal B, Hurley LS (1984) Biochemistry of the essential ultratrace elements. Biochem Elem 3:89–132
Finley JW, Davis CD (1999) Manganese deficiency and toxicity: are high or low dietary amounts of manganese cause for concern? BioFactors 10:15–24. https://doi.org/10.1002/biof.5520100102
Calne DB, Chu NS, Huang CC, Lu CS, Olanow W (1994) Manganism and idiopathic parkinsonism: similarities and differences. Neurology 44:1583–1586. https://doi.org/10.1212/wnl.44.9.1583
Jenkins KJ, Hidiroglou M (1991) Tolerance of the preruminant calf for excess manganese or zinc in milk replacer. J Dairy Sci 74:1047–1053. https://doi.org/10.3168/jds.S0022-0302(91)78254-4
Thomson ABR, Olatunbosun D, Valberg LS (1971) Interrelation of intestinal transport system for manganese and iron. J Lab Clin Med 78:642–655. https://doi.org/10.5555/uri:pii:0022214371901004
Davis CD, Ney DM, Greger JL (1990) Manganese, iron and lipid interactions in rats. J Nutr 120:507–513. https://doi.org/10.1093/jn/120.5.507
Davis CD, Wolf TL, Greger JL (1992) Varying levels of manganese and iron affect absorption and gut endogenous losses of manganese by rats. J Nutr 122:1300–1308. https://doi.org/10.1093/jn/122.6.1300
Pennington JAT, Young BE, Wilson DB, Johnson RD, Vanderveen JE (1986) Mineral content of foods and total diets: the selected minerals in food surveys. J Am Diet Assoc 86:876–891. https://doi.org/10.1016/S0002-8223(21)04040-2
Nielsen FH (2020) Manganese, molybdenum, boron, silicon, and other trace elements. In Present knowledge in nutrition (pp. 485–500). Academic Press
Strause LB, Hegenauer J, Saltman P (1986) Effects of long-term dietary manganese and copper deficiency on rat skeleton. J Nutr 116:135–141. https://doi.org/10.1093/jn/116.1.135
Black JR, Ammerman CB, Henry PR (1985) Effects of high dietary manganese as manganese oxide or manganese carbonate in sheep. J Anim Sci 60:861–866. https://doi.org/10.2527/jas1985.603861x
Rahil-Khazen R, Bolann BJ, Myking A, Ulvik RJ (2002) Multi-element analysis of trace element levels in human autopsy tissues by using inductively coupled atomic emission spectrometry technique (ICP-AES). J Trace Elem Med Biol 16:15–25. https://doi.org/10.1016/S0946-672X(02)80004-9
O’Neal SL, Zheng W (2015) Manganese toxicity upon overexposure: a decade in review. Curr Environ Health Rep 2:315–328. https://doi.org/10.1007/s40572-015-0056-x
Tajchman K, Ukalska-Jaruga A, Bogdaszewski M, Pecio M, Janiszewski P (2021) Comparison of the accumulation of macro-and microelements in the bone marrow and bone of wild and farmed red deer (Cervus elaphus). BMC Vet Res 17:1–11. https://doi.org/10.1186/s12917-021-03041-2
Pitt M, Fraser J, Thurley DC (1980) Molybdenum toxicity in sheep: Epiphysiolysis, exotoses and biochemical changes. J Comp Pathol 90:567–576. https://doi.org/10.1016/0021-9975(80)90105-X
Vyskočil A, Viau C (1999) Assessment of molybdenum toxicity in humans. J Appl Toxicol 19:185–192. https://doi.org/10.1002/(SICI)1099-1263(199905/06)19:3%3c185::AID-JAT555%3e3.0.CO;2-Z
Anke M, Seifert M, Arnhold W, Anke S, Schäfer U (2010) The biological and toxicological importance of molybdenum in the environment and in the nutrition of plants, animals and man: part V: essentiality and toxicity of molybdenum. Acta Aliment 39:12–26. https://doi.org/10.1556/aalim.39.2010.1.2
Pandey R, Kumar R, Singh SP, Srivastava SP (2002) Molybdenum in rat tissue. Hum Exp Toxicol 21:33–35. https://doi.org/10.1191/0960327102ht203oa
Luo XM, Wei HJ, Yang SP (1983) Inhibitory effects of molybdenum on esophageal and forestomach carcinogenesis in rats. J Natl Cancer Inst 71:75–80. https://doi.org/10.1093/jnci/71.1.75
Komada H, Kise Y, Nakagawa M, Yamamura M, Hioki K, Yamamoto M (1990) Effect of dietary molybdenum on esophageal carcinogenesis in rats induced by N-methyl-N-benzylnitrosamine. Can Res 50:2418–2422
Grace ND, Wilson PR, Quinn AK (2005) Impact of molybdenum on the copper status of red deer (Cervus elaphus). N Z Vet J 53:137–141. https://doi.org/10.1080/00480169.2005.36491
Blair-West JR, Brodie A, Coghlan JP, Denton DA, Flood C, Goding JR, Scoggins BA, Tait JF, Tait SAS, Wintour EM, Wright RD (1970) Studies on the biosynthesis of aldosterone using the sheep adrenal transplant: effect of sodium depletion on the conversion of corticosterone to aldosterone. J Endocrinol 46:453–476. https://doi.org/10.1677/joe.0.0460453
Hiyama TY, Matsuda S, Fujikawa A, Matsumoto M, Watanabe E, Kajiwara H, Niimura F, Noda M (2010) Autoimmunity to the sodium-level sensor in the brain causes essential hypernatremia. Neuron 66:508–522. https://doi.org/10.1016/j.neuron.2010.04.017
Noda M, Hiyama TY (2015) Sodium sensing in the brain. Pflugers Arch - Eur J Physiol 467:465–474. https://doi.org/10.1007/s00424-014-1662-4
McKinley MJ, Denton DA, Weisinger RS (1978) Sensors for antidiuresis and thirst - osmoreceptors or CSF sodium detectors? Brain Res 141:89–103. https://doi.org/10.1016/0006-8993(78)90619-4
Denton DA, McKinley MJ, Weisinger RS (1996) Hypothalamic integration of body fluid regulation. Proc Natl Acad Sci 93:7397–7404. https://doi.org/10.1073/pnas.93.14.73
Babcock SM (1905) The addition of salt to the ration of dairy cows. Wis Agr Expt Sta Rpt
Kumar S, Trivedi AV (2016) A review on role of nickel in the biological system. Int J Curr Microbiol Appl Sci 5:719–727. https://doi.org/10.20546/ijcmas.2016.503.084
Das KK, Reddy RC, Bagoji IB, Das S, Bagali S, Mullur L, Khodnapur JP, Biradar MS (2019) Primary concept of nickel toxicity–an overview. J Basic Clin Physiol Pharmacol 30:141–152. https://doi.org/10.1515/jbcpp-2017-0171
Genchi G, Carocci A, Lauria G, Sinicropi MS, Catalano A (2020) Nickel: Human health and environmental toxicology. Int J Environ Res Public Health 17:679. https://doi.org/10.3390/ijerph17030679
McIlveen WD, Negusanti JJ (1994) Nickel in the terrestrial environment. Sci Total Environ 148:109–138. https://doi.org/10.1016/0048-9697(94)90390-5
Schroeder HA, Mitchener M, Nason AP (1974) Life-term effects of nickel in rats: survival, tumors, interactions with trace elements and tissue levels. J Nutr 104:239–243. https://doi.org/10.1093/jn/104.2.239
Cartañá J, Arola L, Mas A (1991) Effects of acute nickel toxicity upon plasma and liver metal homeostasis as a function of sex. Toxicology 69:133–141
Phipps T, Tank SL, Wirtz J, Brewer L, Coyner A, Ortego LS, Fairbrother A (2002) Essentiality of nickel and homeostatic mechanisms for its regulation in terrestrial organisms. Environ Rev 10:209–261. https://doi.org/10.1139/a02-009
Nielsen FH (1993) Is nickel nutritionally important? Nutr Today 28:14–19
Brooks HV, Cook TG, Mansellf GP, Walker GA (1984) Phosphorus deficiency in a dairy herd. N Z Vet J 32:174–176. https://doi.org/10.1080/00480169.1984.35113
Elliott HA, O’connor GA, Brinton S (2002) Phosphorus leaching from biosolids-amended sandy soils. J Environ Qual 31:681–689. https://doi.org/10.2134/jeq2002.6810
Fuller TJ, Carter NW, Barcenas CAMILO, Knochel JP (1976) Reversible changes of the muscle cell in experimental phosphorus deficiency. J Clin Investig 57:1019–1024
Theiler PJ, Green A, du Toit HH (1924) Phosphorus in the livestock industry. J Dep Agricult 8:460–504
Karn JF (2001) Phosphorus nutrition of grazing cattle: a review. Anim Feed Sci Technol 89:133–153. https://doi.org/10.1016/S0377-8401(00)00231-5
Neathery MW, Miller WJ (1975) Metabolism and toxicity of cadmium, mercury, and lead in animals: a review. J Dairy Sci 58:1767–1781. https://doi.org/10.3168/jds.S0022-0302(75)84785-0
Wani AL, Ara A, Usmani JA (2015) Lead toxicity: a review. Interdiscip Toxicol. 8: 55–64. Go to PubMed
Bąkowska M, Pilarczyk B, Tomza-Marciniak A, Udała J, Pilarczyk R (2016) The bioaccumulation of lead in the organs of roe deer (Capreolus capreolus L.), red deer (Cervus elaphus L.), and wild boar (Sus scrofa L.) from Poland. Environ Sci Pollut Res 23:14373–14382. https://doi.org/10.1007/s11356-016-6605-5
Kottferová J, Koréneková B (1998) Distribution of Cd and Pb in the tissues and organs of free-living animals in the territory of Slovakia. Bull Environ Contam Toxicol 60:171–176
Schroeder HA, Tipton IH (1968) The human body burden of lead. Arch Environ Health: An International Journal 17:965–978. https://doi.org/10.1080/00039896.1968.10665354
Mukwevho E, Ferreira Z, Ayeleso A (2014) Potential role of sulfur-containing antioxidant systems in highly oxidative environments. Molecules 19:19376–19389. https://doi.org/10.3390/molecules191219376
Qi K, Owens FN, Lu CD (1994) Effects of sulfur deficiency on performance of fiber-producing sheep and goats: a review. Small Rumin Res 14:115–126. https://doi.org/10.1016/0921-4488(94)90101-5
Coghlin CL (1944) Hydrogen sulfide poisoning in cattle. Can J Comp Med 8:111–113
Bird PR (1970) Sulphur metabolism and excretion studies in ruminants. III. The effect of sulphur intake on the availability of copper in sheep. Proc Aust Soc Anim Prod 8:212–218
Kincaid WB, Nash TH (1988) Detection of a sulfur dioxide signal in a tree-ring record: a case study from Trail, British Columbia, Canada. GeoJournal 17:189–192. https://doi.org/10.1007/BF02432921
Kierdorf U, Stoffels D, Kierdorf H (2014) Element concentrations and element ratios in antler and pedicle bone of yearling red deer (Cervus elaphus) stags—a quantitative X-ray fluorescence study. Biol Trace Elem Res 162:124–133. https://doi.org/10.1007/s12011-014-0154-x
Elrashidi MA, Adriano DC, Lindsay WL (1989) Solubility, speciation, and transformations of selenium in soils. Selenium in Agriculture and the Environment 23:51–63. https://doi.org/10.2136/sssaspecpub23.c3
Oldfield JE (2002) A brief history of selenium research: From alkali disease to prostate cancer (from poison to prevention). In American Society of Animal Science, 1–4
Lazarus M, Orct T, Blanuša M, Vicković I, Šoštarić B (2008) Toxic and essential metal concentrations in four tissues of red deer (Cervus elaphus) from Baranja, Croatia. Food Addit Contam 25:270–283. https://doi.org/10.1080/02652030701364923
Pilarczyk B, Balicka-Ramisz A, Ramisz A, Adamowicz E, Pilarczyk R, Tomza-Marciniak A, Bąkowska M (2009) Selenium concentration in liver and kidney of free living animals (roe and red deer) from West Pomerania (Poland). Eur J Wildl Res 55:279–283. https://doi.org/10.1007/s10344-008-0247-y
Carlisle EM (1986) Silicon as an essential trace element in animal nutrition. Silicon Biochem 703:123
Nielsen FH (1999) Ultratrace elements. In: Sadler MJ, Strain JJ, Caballero B (eds) Encyclopedia of human nutrition. Academic Press, San Diego, pp 1884–1897
Vasanthi N, Saleena LM, Raj SA (2012) Silicon in day today life. World Appl Sci J 17:1425–1440
Hott M, de Pollak C, Modrowski D, Marie PJ (1993) Short-term effects of organic silicon on trabecular bone in mature ovariectomized rats. Calcif Tissue Int 53:174–179. https://doi.org/10.1007/BF01321834
Bellés M, Sánchez DJ, Gómez M, Corbella J, Domingo JL (1998) Silicon reduces aluminium accumulation in rats: relevance to the aluminium hypothesis of Alzheimer disease. Alzheimer Dis Assoc Disord 12:83–87
Schönhofer F, Tataruch F, Friedrich M (1994) Strontium-90 in antlers of red deer: an indicator of environmental contamination by strontium-90. Sci Total Environ 157:323–332. https://doi.org/10.1016/0048-9697(94)90596-7
Strandberg M, Strandgaard H (1995) 90Sr in antlers and bone of a Danish roe deer population. J Environ Radioact 27:65–74. https://doi.org/10.1016/0265-931X(94)00035-U
Baeza A, Vallejo I, Guillén J, Salas A, Corbacho JA (2011) Antlers of Cervus elaphus as biomonitors of 90Sr in the environment. J Environ Radioact 102:311–315. https://doi.org/10.1016/j.jenvrad.2010.12.008
Demesko J, Markowski J, Demesko E, Słaba M, Hejduk J, Minias P (2019) Ecotype variation in trace element content of hard tissues in the European roe deer (Capreolus capreolus). Arch Environ Contam Toxicol 76:76–86. https://doi.org/10.1007/s00244-018-0580-4
Nielsen SP (2004) The biological role of strontium. Bone 35:583–588. https://doi.org/10.1016/j.bone.2004.04.026
Jones JH (1938) The metabolism of calcium and phosphorus as influenced by the addition to the diet of salts of metals which form insoluble phosphates. Am J Physiol-Legacy Content 124:230–237
Skibniewski M, Skibniewska EM, Kośla T (2015) The content of selected metals in muscles of the red deer (Cervus elaphus) from Poland. Environ Sci Pollut Res 22:8425–8431. https://doi.org/10.1007/s11356-014-4007-0
Papachristodoulou C, Stamoulis K, Tsakos P, Vougidou C, Vozikis V, Papadopoulou C, Ioannides K (2015) Liver concentrations of copper, zinc, iron and molybdenum in sheep and goats from Northern Greece, determined by energy-dispersive X-ray fluorescence spectrometry. Bull Environ Contam Toxicol 94:460–467. https://doi.org/10.1007/s00128-015-1496-6
Suttle NF, Mills CF (1966) Studies of the toxicity of copper to pigs: 1. Effects of oral supplements of zinc and iron salts on the development of copper toxicosis. Br J Nutr 20:135–148. https://doi.org/10.1079/BJN19660018
Bremner I, Young BW, Mills CF (1976) Protective effect of zinc supplementation against copper toxicosis in sheep. Br J Nutr 36:551–561. https://doi.org/10.1079/BJN19760108
Allen JG, Masters HG, Peet RL, Mullins KR, Lewis RD, Skirrow SZ, Fry J (1983) Zinc toxicity in ruminants. J Comp Pathol 93:363–377. https://doi.org/10.1016/0021-9975(83)90024-5
Miller WJ, Amos HE, Gentry RP, Blackmon DM, Durrance RM, Crowe CT, Fielding AS, Neathery MW (1989) Long-term feeding of high zinc sulfate diets to lactating and gestating dairy cows. J Dairy Sci 72:1499–1508. https://doi.org/10.3168/jds.S0022-0302(89)79260-2
Fosmire GJ (1990) Zinc toxicity. Am J Clin Nutr 51:225–227. https://doi.org/10.1093/ajcn/51.2.225
Van Metre DC, Callan RJ (2001) Selenium and vitamin E. Vet Clin N Am Food Anim Pract 17:373–402. https://doi.org/10.1016/S0749-0720(15)30034-7
Wilson PR, Grace ND (2001) A review of tissue reference values used to assess the trace element status of farmed red deer (Cervus elaphus). N Z Vet J 49:126–132. https://doi.org/10.1080/00480169.2001.36219
Barboza PS, Rombach EP, Blake JE, Nagy JA (2003) Copper status of muskoxen: a comparison of wild and captive populations. J Wildl Dis 39:610–619. https://doi.org/10.7589/0090-3558-39.3.610
Flynn A, Franzmann AW, Arneson PD, Oldemeyer JL (1977) Indications of copper deficiency in a subpopulation of Alaskan moose. J Nutr 107:1182–1189. https://doi.org/10.1093/jn/107.7.1182
Arnhold W, Anke M, Goebel S (2002) The copper, zinc and manganese status in opossum and gray fox. Z Jagdwiss 48:77–86. https://doi.org/10.1007/BF02192395
Lombardini M, Rosin AV, Murru M, Cinerari CE, Meriggi A (2014) Reproductive and demographic parameters in Sardinian wild boar, Sus scrofa meridionalis. Folia Zoologica 63:301–307. https://doi.org/10.25225/fozo.v63.i4.a10.2014
Gürtler RE, Rodríguez-Planes LI, Gil G, Izquierdo VM, Cavicchia M, Maranta A (2018) Differential long-term impacts of a management control program of axis deer and wild boar in a protected area of north-eastern Argentina. Biol Invasions 20:1431–1447. https://doi.org/10.1007/s10530-017-1635-6
Acknowledgements
We are indebted to the managers and hunters of National Park Veluwezoom for supplying us with the culled animals for analysis, and allowing us to create a dissection room to dissect the animals on-site. Furthermore we thank the technicians of the General Instruments of Radboud University (Paul van der Ven and Sebastian Krosse) for their help in the analysis of the elements. We also thank Fleur van Beek and Jaap van Dorst for their help with the dissections.
Author information
Authors and Affiliations
Contributions
Elke Wenting: Conceptualization, Methodology, Formal analysis, Investigation, Resources, Data Curation, Writing—Original Draft, Writing—Review & Editing, Visualization, Project administration; Henk Siepel: Conceptualization, Methodology, Writing—Original Draft, Visualization; Patrick A. Jansen: Conceptualization, Writing—Original Draft.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Conflicts of Interest
No actual or potential conflicts of interest are declared by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wenting, E., Siepel, H. & Jansen, P.A. Variability of the Ionome of Wild Boar (Sus scrofa) and Red Deer (Cervus elaphus) in a Dutch National Park, with Implications for Biomonitoring. Biol Trace Elem Res 202, 2518–2546 (2024). https://doi.org/10.1007/s12011-023-03879-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03879-7