Abstract
Purpose
Thyroid transcription factor‐1 (TTF‐1) assessed by immunohistochemistry (IHC) is a specific biomarker for lung adenocarcinoma, and is commonly used to confirm the pulmonary origin of neuroendocrine tumours (NET). The majority of the available data suggest that TTF-1 is favourable prognostic biomarker for lung adenocarcinomas, whereas its role is more conflicting for lung NET. The main aim of this multicenter retrospective study was to investigate the potentially relevant associations between TTF-1 biomarker and clinical and pathological features of the study population, as well as determine TTF-1 prognostic effect on the clinical outcome of the patients.
Methods
A multicentre retrospective study was conducted on 155 surgically-removed lung NET, with available IHC TTF-1 assessment.
Results
Median age was 59.5 years (range 13–86), 97 patients (62.6%) were females, 31 cases (20%) were atypical carcinoids, 4 (2.6%) had TNM stage IV. Mitotic count ≥2 per 10 high-power field was found in 35 (22.6%) subjects, whereas necrosis was detected in 20 patients (12.9%). TTF-1 was positive in 78 cases (50.3%). The median overall survival was 46.9 months (range 0.6–323) and the median progression-free survival was 39.1 months (range 0.6–323). Statistically significant associations were found between (1) TTF-1 positivity and female sex (p = 0.007); and among (2) TTF-1 positivity and the absence of necrosis (p = 0.018).
Conclusions
This study highlights that TTF-1 positivity differs according to sex in lung NET, with a more common TTF-1 positive staining in female. Moreover, TTF-1 positivity correlated with the absence of necrosis. These data suggest that TTF-1 could potentially represent a gender-related biomarker for lung NET.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Thyroid transcription factor‐1 (TTF‐1) is a 38‐kD transcription factor that is normally expressed in adult thyroid and lung tissue [1, 2], with a relevant role in both thyroid and lung differentiation, development, and functional maintenance [3]. In the context of lung cancer, specifically non-small cell lung carcinoma (NSCLC), TTF-1 is expressed in nearly 75% of lung adenocarcinoma, whereas lung squamous cell carcinoma does not express TTF-1. Thereby TTF‐1, assessed by immunohistochemistry (IHC), is considered a specific diagnostic biomarker for lung adenocarcinoma [4]. In addition, in the neuroendocrine tumours (NET) setting, TTF-1 is commonly used to confirm the pulmonary origin of the tumour [5, 6], both for typical carcinoid, TC, as well as for atypical carcinoid, AC.
Available evidences suggest a specificity of this biomarker for the differential diagnosis of NET from different primary sites, but at the same time highlight that TTF-1 presents an extremely heterogeneous expression in lung NET [7, 8]. Several studies have evaluated the prognostic role of TTF-1 in NSCLC [9], suggesting that TTF-1 is favourable prognostic biomarker for lung adenocarcinomas [10]. Unfortunately, its role is more conflicting for lung NET [11, 12]. This issue is of note, in the setting of lung NET, given that established prognostic factors, beyond the histological subtype of TC (associated to a better prognosis) vs. AC and the pathological TNM stage, are missing. In this context, in a previous work by our group we postulated a role for primary lung NET laterality [13]. In that retrospective multicentre analysis, tumours located in the left lung clearly presented a higher biological aggressiveness (expressed as higher proliferation index, as mitotic count and Ki-67, more common presence of necrosis and higher tumour grade). A clinical impact of these differences at the pathological level was further confirmed by univariable analysis and Cox regression-based multivariable model. In addition, we demonstrated different angiogenic pattern, in terms of micro vessel density (MVD) by CD34 immunohistochemical (IHC) staining and hypoxia, according to lung NET laterality [14], with right tumours presenting higher angiogenesis rates, and left tumours associated more commonly with hypoxia. In this study, TTF-1-negative cases presented a lower OS, with a 1-year OS rate of 90.0% vs. 100% for TTF-1-positive cases. Notably, the difference resulted as more evident when comparing 10-years OS rates, with 60% for TTF-1 negative cases vs. 100% for TTF-1 positive cases. However, the impact on OS was not statistically significant in the Cox regression analysis (p = 0.468, HR: 0.14, 95% CI 0.001–1414.981) [14], confirming the controversial literature evidences about a possible prognostic role of TTF-1 for lung NET.
Therefore, the main aim of this multicentre and retrospective study was to evaluate the potential value of TTF-1 for lung NET, assessing its potentially relevant associations with key clinical and pathological variables and evaluating its prognostic role in a selected and homogeneous population of surgically resected lung NET.
Materials and methods
A multicentre retrospective study was performed including patients with a confirmed histological diagnosis of lung NET classified as TC (NET G1) or AC (NET G2) according to WHO 2022 classification [15], who were diagnosed at the study Institutions. The resected lung tissues were fixed in a 10% neutral buffered formalin solution and the specimens were set in paraffin and sliced (2-µm-thick for each section). Each included case was stained with haematoxylin–eosin, chromogranin A, Synaptophysin, TTF-1 and Ki-67. IHC marker expression was quantified by expression intensity (weak, moderate, strong) and the percentage of IHC marker-positive tumour cells in fields of view of 200-fold magnification by experienced pathologist of each centre. Chromogranin A and synaptophysin were considered positive if >90% of the neoplastic cells exhibited at least moderate staining intensity. TTF-1 was scored positive if >10% of the nuclei of tumour cells were positive and the staining intensity was moderate or strong. The Ki-67 index was obtained by counting the positive tumour cells in areas of higher nuclear labelling (so-called hotspots) and was expressed as a percentage.
Inclusion criteria, beyond the diagnosis, were: (1) the surgical removal of the primary tumour, (2) patients with available IHC assessment for TTF-1. We chose these criteria to (1) ensure a homogeneous population and, also, comparable tumoral samples for histopathological evaluation, (2) study specifically the TTF-1 significance as biomarker for lung NET, given the conflicting data available in literature. After patients’ selection, we carefully collected their relevant clinical and pathological data in a dedicated database.
Descriptive statistical analyses were performed on the overall collected data. Univariable analysis by Chi-square test and multivariable analysis using a logistic regression model were applied to investigate the association among TTF-1 expression and demographic and clinical factors considered relevant for lung NET (i.e. sex, age, smoking history, disease stage, tumour grade, Ki-67 value and tumour subtype). Univariable and multivariable Cox regression analysis was performed to evaluate the prognostic role of TTF-1 on the patients’ outcome (i.e. progression-free survival and overall survival) together with other relevant factors., All statistical analyses were performed using IBM-SPSS version 25 (IBM Corporation, New York, United States of America). p values < 0.05 were considered statistically significant.
Results
Study population
Patients’ characteristics are summarised in Table 1.
Overall, we included 155 surgically removed lung NET. Median age was 59.5 years (13–86), N = 97 (62.6%) were females, N = 40 (25.8%) were smokers, N = 31 (20%) were ACs, N = 85 (54.8%) were centrally located, N = 89 (57.4%) were located in the right lung, N = 4 patients (2.6%) presented a TNM stage IV at the diagnosis. Mitotic count was ≥2 per 10 high-power field (HPF) in N = 35 cases (22.6%), necrosis was present in N = 20 (12.9%). Grade 1 was reported in 103 cases (66.5%), whereas Ki-67 was >20% in N = 5 (3.2%). TTF-1 was positive in 78 cases (50.3%), Chromogranin A in 139 (89.7%) and Synaptophysin in 137 (88.4%), respectively. The most common type of surgery was lobectomy in 91 patients (58.7%). Median overall survival (OS) was 46.9 months (0.6–323), median progression-free survival (PFS) was 39.1 months (0.6–323).
Specifically, as detailed above, we focused on TTF-1 positive cases. Comparison among the prevalence and distribution of selected clinical and pathological features among the entire study population and the subgroup of TTF-1 positive patients are reported in Fig. 1. All the collected data of TTF-1 positive cases are detailed in Supplementary Material 1.
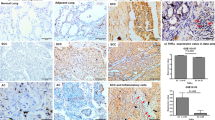
Key features: all vs. TTF-1 positive cases. In this graph are compared the distributions of all lung NET included in the study vs. TTF-1 positive cases in selected clinical and pathological variables (as sex, central vs. peripheral location of the primary tumour, tumour laterality, histopathological subtype of TC vs. AC, TNM stage, mitotic count, presence of necrosis and tumour grade G3 vs. G1 and G2). Significant (p < 0.05) associations are marked with an asterisk (*)
Associations
Statistically significant associations were found between (1) TTF-1 positivity and female sex (p = 0.007); and among (2) TTF-1 positivity and the absence of necrosis (p = 0.018). No other relevant associations were detected between TTF-1 and the remaining clinical or pathological variables. Notably, female sex correlates with the absence of necrosis (p = 0.002). Results of the multivariable analysis by the logistic regression model to take into account other potentially confounding variables confirmed the association between TTF-1 positivity and female sex (p = 0.042) (Table 2). Two exemplificative cases of TC, one female with TTF-1 positive tumour and a male with TTF-1 negative tumour, are depicted in Fig. 2.
Two exemplificative cases of lung NET comparing TTF-1 expression according to sex. Male patient (A, B) shows a well differentiated morphological pattern with the absence of necrosis, TC (A) with negative staining for TTF-1 (B). Female patient (C, D) with diagnosis of TC (C) but positive TTF-1 IHC staining (D). A, C Haematoxylin and eosin stain,10X, and (B, D) TTF-1 immunohistochemical stain, 10X
Of note, any difference in TTF-1 expression according to the laterality of the primary tumour was observed. TTF-1 positive cases were equally distributed in right and left lung (TTF-1 positive lung NET: 46 right-located and 32 left-located; TTF-1 negative tumours: 43 right-located and 34 left-located; p = 0.694).
Impact on survival
Although the statistical significance was not reached (p = 0.609 for OS and p = 0.368 for PFS), a favourable trend in OS and PFS was found for TTF-1 positivity, with an estimated OS of 126 months for TTF-1 negative cases and 200 months for positive cases; in terms of PFS the estimated months for TTF-1 negative cases was 107 vs. 169 for positive ones. In the multivariable analysis, tumour stage emerged as the unique factor with independent prognostic impact (p = 0.004) on OS (Table 3); however, a trend for TTF-1 expression, age, and histological subtype effects was detected.
Discussion
Unambiguous evidence supports the relevance of gender difference in oncology, from the diagnosis to the response to treatments and treatments’ side-effects prolife [16,17,18]. Overall, sex significantly influences the clinical and pathological features of cancer patients. These include disparities in incidence and mortality rates, clinical presentations including age, screening participation rates, site, stage and treatment utilisation, histopathology (including genetic and molecular features) and survival [19,20,21]. Environmental and behavioural factors (e.g. smoking habit or metabolic syndrome onset) play a key role in this context [22,23,24]. Moreover, biological (e.g. sex hormones) features have been showed to contribute to the differential risk in several tumour types [25, 26]. In the field of NET, indeed, the majority of available data arise from studies on gastro-entero-pancreatic (GEP) neuroendocrine neoplasms (NEN) [27]. A large population study, including 15,202 patients with pancreatic neuroendocrine tumours from The National Cancer Database (NCDB), suggested that men had more frequently tumours >2 cm, and poorly or undifferentiated tumours if compared to women [28]. Notably, no significant differences were found in the rates of lymph node involvement and metastatic recurrence after the surgical removal of the primary tumour. At the molecular level, MEN1 and DAXX mutations resulted more common in males and TP53 mutations in females, respectively. However, these data lacked to be confirmed at the multivariable analysis. Data of the Surveillance, Epidemiology and End Results Research (SEER) registry, based on 43,751 patients with GEP-NETs, demonstrated with multivariable analyses a prognostic value (in terms of OS) for sex with women associated to better outcomes (p < 0.001) [29]. In this analysis the 3-year survival rates resulted 84.6% and 87.7% and the 5-years survival rates of 80% vs. 84% for male and female, respectively. For lung NET a sex-difference has been described, with, even in this case, more favourable trends in female in a previous work by our group [13]. According to the Cox-univariate regression model a significant impact on patients’ outcome for sex was demonstrated, with male sex associated with dismal PFS (p < 0.0001) and OS (p < 0.0001). These data have been further confirmed by data coming from SEER registry, where female sex was associated with better OS compared with male sex (p < 0.001) [30]. Furthermore, few works suggest biological differences in terms of tumour aggressiveness in relation to patients’ sex [13]. Female sex has been associated with a more indolent disease, both considering a lower tumour stage (specifically, negative nodal status vs. positive) and also with regards to pathological features, as lower tumour grade (G1-2 vs. G3), lower Ki67 index and reduced mitotic count. In addition, a sex imbalance of the histological subtype (TC vs. AC) in males and females has been reported [31]. However, a sex-related distribution of the main IHC NET biomarkers (Chromogranin A, NSE and, specifically, TTF-1) has never been reported in literature. A retrospective study including 11 carcinoid tumorlets (TLs), 36 TC, 17 AC and 16 large cell neuroendocrine carcinomas (LCNECs) showed a more common positive TTF-1 IHC staining in LCNECs (5 of 6 positive cases), followed by TLs (4 of 8) respect to AC (1 of 4), and TC (0 of 10) [32]. Interestingly, in this study the percentage of female was higher in these two categories, LCNEC and TLs, whereas both TC and AC were well-balanced among male and female. In the current work we observed a statistically significant association between the IHC positivity for TTF-1 and female lung NET patients (p = 0.007). This result deserves further studies to confirm a potential biological significance of TTF-1 expression in connection with the sex of lung NET patients, with potentially relevant implications in the diagnostic work-up of these tumours.
TTF-1+ alveolar type II epithelial cells have been demonstrated to be the major source of vascular endothelial growth factor (VEGF) in the lung [33, 34]. At the molecular level, TTF-1 has been postulated to positively regulate VEGF expression and the major signalling receptor for VEGF as VEGFR2 lung cancer epithelial cells [34, 35]. TTF-1, indeed, has been suggested to reprogramme lung cancer secreted proteome into an antiangiogenic state. Interestingly, TTF-1 has been assessed as a potential predictive factor for antiangiogenic treatment in non-squamous NSCLC [36]. In this study, the 92 TTF-1-positive patients presented higher response rates (51.4% vs. 27.3%, p = 0.027) and PFS (216 days vs. 137 days, p = 0.012) in the group treated with the antiangiogenic bevacizumab to standard chemotherapy, whereas in TTF-1-negative patients no clinical benefit was obtained by the combination therapy (chemotherapy plus bevacizumab). Unfortunately, data about VEGF expression and TTF-1 in lung NET are lacking. In a previous work by our group, we demonstrated a significant association between the absence of expression of the TTF-1 and the presence of hypoxia (in 14/16, 87.5%, of TTF-1-negative cases, p = 0.012). Among hypoxia-negative cases, 11/13 (84.6%) were TTF-1 positive, whereas among hypoxia-positive cases, 10/24 expressed TTF-1 [14]. In the present study we detected a statistically significant correlation among TTF-1 positivity and the absence of necrosis (p = 0.018). Taken all together this data, it is possible to hypothesise that TTF-1 may be positively linked to increased angiogenesis, and associated with lower hypoxia and the absence of necrosis in lung neoplasms, potentially including lung NET.
Finally, in the present work we investigated the prognostic value of TTF-1 in our lung NET population. According to available evidences, TTF-1 positivity is considered an established positive prognostic factor for lung adenocarcinomas [10]. In lung NET field, more conflicting data have been reported. In a retrospective series of 370 lung NET, a difference in IHC positivity for TTF‐1 was found between patients with higher or lower Ki-67 [11]. Overall, a positive staining for TTF-1 was detected in 49 (17.1%) of the included lung NET, with TTF‐1 positivity in 30 (13.0%) of the low Ki‐67 group of patients and in 19 (34.5%) cases of the high Ki‐67 group. The second group (with higher Ki-67) was associated to a worse prognosis (p < 0.0001). Also, TTF-1 positivity correlated with a reduced survival outcome (p = 0.03). In a retrospective study of 34 lung NET treated with peptide receptor radionuclide therapy with (177) Lu-DOTATATE (Lu-PRRT), survival outcomes in terms of PFS were better in TTF-1 negative cases if compared to TTF-1 positive ones (26.3 vs. 7.2 months, respectively, p = 0.0009) [37]. However, these data have not been confirmed in subsequent works [12, 38]. A retrospective analysis of 108 lung NET lacked to demonstrate a correlation between TTF-1 positivity and patient outcomes [12,]. In another study, TTF-1 was positive in 78% of the 133 lung NET cases but was not associated with patients’ survival [38]. In the present study, in line with the available literature data, no significant association among TTF-1 expression and patient survival was observed (according to univariable and also multivariable model), despite a favourable trend for TTF-1 positive cases was noticed. Further prospective studies are encouraged to determine if this biomarker has a prognostic relevance for lung NET.
Conclusions
TTF-1 is a well-known biomarker for lung neoplasms, above all for adenocarcinomas. For lung NET, the diagnostic value of is TTF-1 debated, despite its widespread use in the clinical practice. TTF-1’s role as a prognostic biomarker for lung NET is still uncertain, and also in our analysis a not significant trend has been detected. TTF-1 negativity resulted associated with the presence of necrosis, in analogy with the few available literature data that have showed a higher expression of TTF-1 in conditions of higher angiogenesis and lower hypoxia. Notably, our study provides, to the best of our knowledge, the first evidence of a correlation among TTF-1 positivity and female sex, suggesting a potential biological and clinical relevance of this observation. Further studies with larger and independent patients’ populations are needed to confirm TTF-1 role as a gender-related biomarker for lung NET.
Data availability
All data generated or analysed during this study are included in this article. Further enquiries can be directed to the corresponding author.
References
K. Ikeda, J.C. Clark, J.R. Shaw-White, M.T. Stahlman, C.J. Boutell, J.A. Whitsett, Gene structure and expression of human thyroid transcription factor‐1 in respiratory epithelial cells. J. Biol. Chem. 270, 8108–8114 (1995)
L. Guan, X. Zhao, L. Tang, J. Chen, J. Zhao, M. Guo et al. Thyroid transcription factor-1: structure, expression, function and its relationship with disease. Biomed. Res. Int. 2021, 9957209 (2021). https://doi.org/10.1155/2021/9957209
S. Kimura, Y. Hara, T. Pineau, P. Fernandez-Salguero, C.H. Fox, J.M. Ward et al. The T/ebp null mouse: thyroid‐specific enhancer‐binding protein is essential for the organogenesis of the thyroid, lung, ventral forebrain, and pituitary. Genes Dev. 10, 60–69 (1996)
O. Kaufmann, M. Dietel, Thyroid transcription factor-1 is the superior immunohistochemical marker for pulmonary adenocarcinomas and large cell carcinomas compared to surfactant proteins A and B. Histopathology 36, 8–16 (2000). https://doi.org/10.1046/j.1365-2559.2000.00801.x
E.Z. Du, P. Goldstraw, J. Zacharias, O. Tiffet, P.J. Craig, A.G. Nicholson et al. TTF-1 expression is specific for lung primary in typical and atypical carcinoids: TTF-1-positive carcinoids are predominantly in peripheral location. Hum. Pathol. 35, 825–831 (2004). https://doi.org/10.1016/j.humpath.2004.02.016
A. Saqi, D. Alexis, F. Remotti, G. Bhagat, Usefulness of CDX2 and TTF-1 in differentiating gastrointestinal from pulmonary carcinoids. Am. J. Clin. Pathol. 123, 394–404 (2005). https://doi.org/10.1309/UKN6-PVRK-XHG4-22DA
C. Zhang, L.A. Schmidt, K. Hatanaka, D. Thomas, A. Lagstein, J.L. Myers, Evaluation of napsin A, TTF-1, p63, p40, and CK5/6 immunohistochemical stains in pulmonary neuroendocrine tumours. Am. J. Clin. Pathol. 142, 320–324 (2014). https://doi.org/10.1309/AJCPGA0IUA8BHQEZ
A. Srivastava, J.L. Hornick, Immunohistochemical staining for CDX-2, PDX-1, NESP-55, and TTF-1 can help distinguish gastrointestinal carcinoid tumours from pancreatic endocrine and pulmonary carcinoid tumours. Am. J. Surg. Pathol. 33, 626–632 (2009). https://doi.org/10.1097/PAS.0b013e31818d7d8b
N.H. Au, M. Cheang, D.G. Huntsman, E. Yorida, A. Coldman, W.M. Elliott et al. Evaluation of immunohistochemical markers in non‐small cell lung cancer by unsupervised hierarchical clustering analysis: a tissue microarray study of 284 cases and 18 markers. J. Pathol. 204, 101–109 (2004)
T. Berghmans, M. Paesmans, C. Mascaux, B. Martin, A.P. Meert, A. Haller et al. Thyroid transcription factor 1—a new prognostic factor in lung cancer: a meta‐analysis. Ann. Oncol. 17, 1673–1676 (2006)
G. Centonze, P. Maisonneuve, M. Simbolo, V. Lagano, F. Grillo, A. Fabbri et al. Lung carcinoid tumours: histology and Ki-67, the eternal rivalry. Histopathology 82, 324–339 (2023). https://doi.org/10.1111/his.14819
V.E. Georgakopoulou, E. Zygouris, C. Damaskos, A. Pierrakou, P. Papalexis, N. Garmpis et al. Prognostic value of the immunohistochemistry markers CD56, TTF-1, synaptophysin, CEA, EMA and NSE in surgically resected lung carcinoid tumours. Mol. Clin. Oncol. 16, 31 (2022). https://doi.org/10.3892/mco.2021.2464
A. La Salvia, I. Persano, A. Siciliani, M. Verrico, M. Bassi, R. Modica et al. Prognostic significance of laterality in lung neuroendocrine tumours. Endocrine 76, 733–746 (2022). https://doi.org/10.1007/s12020-022-03015-w
A. La Salvia, R. Carletti, M. Verrico, T. Feola, G. Puliani, M. Bassi et al. Angioside: the role of angiogenesis and hypoxia in lung neuroendocrine tumours according to primary tumour location in left or right parenchyma. J. Clin. Med. 11, 5958 (2022). https://doi.org/10.3390/jcm11195958
G. Rindi, H. Moch, W.G. McCluggage et al. Neuroendocrine neoplasms, non-endocrine organs. in WHO Classification of Tumours Endocrine and Neuroendocrine Tumours, 5th edn. ed. by WHO Classification of Tumours Editorial Board (International Agency for Research on Cancer (IARC), Lyon France, 2022).
J.B. Rubin, The spectrum of sex differences in cancer. Trends Cancer 8, 303–315 (2022). https://doi.org/10.1016/j.trecan.2022.01.013
A.D. Wagner, S. Oertelt-Prigione, A. Adjei, T. Buclin, V. Cristina, C. Csajka et al. Gender medicine and oncology: report and consensus of an ESMO workshop. Ann. Oncol. 30, 1914–1924 (2019). https://doi.org/10.1093/annonc/mdz414
C. Marosi, Gender aspects of treatment and drug related toxicity in medical oncology. Wien. Med. Wochenschr. 156, 534–540 (2006). https://doi.org/10.1007/s10354-006-0347-9
M.C.S. Wong, J. Huang, V. Lok, J. Wang, F. Fung, H. Ding, Z.J. Zheng, Differences in incidence and mortality trends of colorectal cancer worldwide based on sex, age, and anatomic location. Clin. Gastroenterol. Hepatol. 19, 955–966.e61 (2021). https://doi.org/10.1016/j.cgh.2020.02.026
H. Fajkovic, J.A. Halpern, E.K. Cha, A. Bahadori, T.F. Chromecki, P.I. Karakiewicz, E. Breinl, A.S. Merseburger, S.F. Shariat, Impact of gender on bladder cancer incidence, staging, and prognosis. World J. Urol. 29, 457–463 (2011). https://doi.org/10.1007/s00345-011-0709-9
G.R.C. Clark, C.G. Fraser, J.A. Strachan, R.J.C. Steele, Sex variation in colorectal cancer mortality: trends and implications for screening. Eur. J. Public Health 33, 331–335 (2023). https://doi.org/10.1093/eurpub/ckad029
C. Stapelfeld, C. Dammann, E. Maser, Sex-specificity in lung cancer risk. Int. J. Cancer 146, 2376–2382 (2020). https://doi.org/10.1002/ijc.32716
P. Wele, X. Wu, H. Shi, Sex-dependent differences in colorectal cancer: with a focus on obesity. Cells 11, 3688 (2022). https://doi.org/10.3390/cells11223688
K. Esposito, P. Chiodini, A. Colao, A. Lenzi, D. Giugliano, Metabolic syndrome and risk of cancer: a systematic review and meta-analysis. Diabetes Care 35, 2402–2411 (2012). https://doi.org/10.2337/dc12-0336
T.J. Key, V. Beral, Sex hormones and cancer. IARC Sci. Publ. 116, 255–269 (1992)
J.H. Lin, E. Giovannucci, Sex hormones and colorectal cancer: what have we learned so far? J. Natl Cancer Inst. 102, 1746–1747 (2010). https://doi.org/10.1093/jnci/djq444
G. Muscogiuri, L. Barrea, T. Feola, M. Gallo, E. Messina, M.A. Venneri et al. NIKE (Neuroendocrine Tumours, Innovation in Knowledge and Education) Group. Pancreatic Neuroendocrine Neoplasms: does sex matter? Trends Endocrinol. Metab. 31, 631–641 (2020). https://doi.org/10.1016/j.tem.2020.02.010
J.A. Greenberg, N.A. Ivanov, C.E. Egan, Y.J. Lee, R. Zarnegar, T.J. Fahey 3rd et al. Sex-based clinicopathologic and survival differences among patients with pancreatic neuroendocrine tumours. J. Gastrointest. Surg. 26, 2321–2329 (2022). https://doi.org/10.1007/s11605-022-05345-6
Z. Xu, L. Wang, S. Dai, M. Chen, F. Li, J. Sun et al. Epidemiologic trends of and factors associated with overall survival for patients with gastroenteropancreatic neuroendocrine tumours in the United States. JAMA Netw. Open 4, e2124750 (2021). https://doi.org/10.1001/jamanetworkopen.2021.24750
O. Abdel-Rahman, S. Ghosh, N. Fazio, Sex-based differences in the outcomes of patients with lung carcinoids. J. Comp. Eff. Res. 11, 523–531 (2022). https://doi.org/10.2217/cer-2021-0205
C. Kesrouani, C. Ghorra, M. Rassy, H.R. Kourie, J. Kattan, Distribution and characteristics of pulmonary neuroendocrine tumors: single institution experience in Lebanon. Asian Pac. J. Cancer Prev. 17(5), 2579–2581 (2016)
E.Z. Du, P. Goldstraw, J. Zacharias, O. Tiffet, P.J. Craig, A.G. Nicholson, N. Weidner, E.S. Yi, TTF-1 expression is specific for lung primary in typical and atypical carcinoids: TTF-1-positive carcinoids are predominantly in peripheral location. Hum. Pathol. 35, 825–831 (2004). https://doi.org/10.1016/j.humpath.2004.02.016
X. Zeng, S.E. Wert, R. Federici, K.G. Peters, J.A. Whitsett, VEGF enhances pulmonary vasculogenesis and disrupts lung morphogenesis in vivo. Dev. Dyn. 211, 215–227 (1998). 10.1002/(SICI)1097-0177(199803)211:3<215:AID-AJA3>3.0.CO;2-K
Y. Maeda, V. Davé, J.A. Whitsett, Transcriptional control of lung morphogenesis. Physiol. Rev. 87, 219–244 (2007). https://doi.org/10.1152/physrev.00028.2006
L.W. Wood, N.I. Cox, C.A. Phelps, S.C. Lai, A. Poddar, C. Talbot Jr et al. Thyroid transcription factor 1 reprograms angiogenic activities of secretome. Sci. Rep. 6, 19857 (2016). https://doi.org/10.1038/srep19857
A. Takeuchi, T. Oguri, Y. Yamashita, K. Sone, S. Fukuda, O. Takakuwa et al. TTF-1 expression predicts the merit of additional antiangiogenic treatment in non-squamous non-small cell lung cancer. Anticancer Res. 38, 5489–5495 (2018). https://doi.org/10.21873/anticanres.12882
A. Ianniello, M. Sansovini, S. Severi, S. Nicolini, C.M. Grana, K. Massri et al. Peptide receptor radionuclide therapy with (177)Lu-DOTATATE in advanced bronchial carcinoids: prognostic role of thyroid transcription factor 1 and (18)F-FDG PET. Eur. J. Nucl. Med. Mol. Imaging 43, 1040–1046 (2016). https://doi.org/10.1007/s00259-015-3262-8
T. Vesterinen, S. Mononen, K. Salmenkivi, H. Mustonen, J. Räsänen, J.A. Salo et al. Clinicopathological indicators of survival among patients with pulmonary carcinoid tumor. Acta Oncol. 57, 1109–1116 (2018). https://doi.org/10.1080/0284186X.2018.1441543
Funding
Open access funding provided by Università degli Studi di Roma La Sapienza within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Individual contributions to the paper (according to CRediT roles): A.L.S., A.S., M.R., M.V., R.B., G.P., R.M., I.Z., I.P., G.F., M.B., M.M., S.B., E.G., M.I., F.P., M.P.B., and A.F. Conceptualisation: A.L.S. and A.F. Data curation: A.L.S., A.S., M.R., M.V., R.B., G.P., R.M., I.Z., I.P., G.F., M.B., and M.M. Formal analysis: A.L.S., E.G., and F.P. Methodology: A.L.S., A.S., G.F., S.B., and M.P.B. Project administration: G.F., M.M., E.G., M.I., F.P., M.P.B., and A.F. Software: A.S., M.R., R.B., I.Z., and M.B. Supervision: A.F. Validation: A.F. Visualisation: A.L.S., A.S., M.R., M.V., R.B., G.P., R.M., I.Z., I.P., G.F., M.B., M.M., S.B., E.G.M., and M.I. Roles/Writing—original draft: A.L.S., G.F., and A.F. Writing—review, editing and approving the final version of the manuscript: A.L.S., A.S., M.R., M.V., R.B., G.P., R.M., I.Z., I.P., G.F., M.B., M.M., S.B., E.G., M.I., F.P., M.P.B., and A.F.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study complies with the guidelines for human studies and it was conducted ethically in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Sapienza University of Rome (reference number 5917).
Informed consent
All patients gave written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
La Salvia, A., Siciliani, A., Rinzivillo, M. et al. Thyroid transcription factor‐1 expression in lung neuroendocrine tumours: a gender-related biomarker?. Endocrine 83, 519–526 (2024). https://doi.org/10.1007/s12020-023-03542-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03542-0