Abstract
Background and aims
There is limited information on combination of hepatic arterial infusion chemotherapy (HAIC) and systemic therapy for advanced hepatocellular carcinoma (Ad-HCC). We aim to compare the efficacy and safety of HAIC plus camrelizumab (a PD-1 inhibitor) and apatinib (an VEGFR-2 inhibitor) versus camrelizumab and apatinib for Ad-HCC.
Methods
From April 2019 to October 2022, 416 patients with Ad-HCC who received either HAIC plus camrelizumab and apatinib (TRIPLET protocol, n = 207) or camrelizumab and apatinib (C–A protocol, n = 209) were reviewed retrospectively. The propensity score matching (PSM) was used to reduce selective bias. Overall survival (OS) and progression-free survival (PFS) were compared using the Kaplan–Meier method with the log-rank test. Cox regression analyses of independent prognostic factors were evaluated.
Results
After PSM 1:1, 109 patients were assigned to two groups. The median OS of not reached in the TRIPLET group was significantly longer than that of 19.9 months in the C–A group (p < 0.001), while in the TRIPLET group, the median PFS of 11.5 months was significantly longer than that of 9.6 months in the C–A group (p < 0.001). Multivariate analyses showed that the factors significantly affected the OS were CTP grade, tumor number > 3, and TRIPLET treatment (p < 0.001). Grade 3/4 adverse events occurred at a rate of 82.1% vs. 71.3% in TRIPLET and C–A groups, respectively.
Conclusion
The TRIPLET protocol has promising survival benefits in the management of patients with Ad-HCC, with acceptable safety.
Trail registration: The study has been retrospectively registered at Chinese Clinical Trial Registry (https://www.chictr.org.cn/, ChiCTR2300075828).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the second most common malignant tumor and the fourth leading cause of cancer death, hepatocellular carcinoma (HCC) has an annual incidence of more than 840,000 new cases globally [1]. Unfortunately, most patients with HCC with liver cirrhosis are already in the advanced stage when they are initially diagnosed, at which point, they have lost the chance to obtain curative therapies [2]. More than 90% of HCC cases in Chinese patients are attributed to hepatitis B virus (HBV) infection, which is thought to be associated with the development of progressive disease and poorer prognoses, with a 5-year overall survival (OS) of only 10–18% [2,3,4].
Conventional TACE (cTACE) using lipiodol mixed with chemotherapeutics is recommended as the first-line treatment in intermediate-stage HCC [5]. However, a poorer objective response rate (ORR) is commonly found in cases of HCC with a high tumor burden [6]. Hepatic arterial infusion chemotherapy (HAIC) is a promising option that can provide sustained local high concentrations of chemotherapy agents into the tumor and has been shown to outperform the intravenous administration; consequently, HAIC has received extensive support for the effective and safe treatment of intermediate advanced-stage HCC. Zhao Ming et al. have reported that HAIC with the FOLFOX regimen (fluorouracil, leucovorin, and oxaliplatin) displayed an encouraging safety profile and antitumor activity for locally advanced HCC in an open-label, phase III trial [7]. Moreover, for patients with HCC with a mean tumor diameter of 11.2 cm, the median OS was 20.8 months, with an ORR reaching 35.4%. Moreover, in a randomized phase III trial, Shi Ming et al. showed that HAIC with the FOLFOX regimen yielded a higher median OS (23.1 months) and ORR (48%) than cTACE treatment in large HCC (largest diameter > 7 cm) in BCLC A or B stage [8].
In recent years, the efficacy of combining immune checkpoint inhibitors (ICIs) and multitargeted tyrosine kinase inhibitors (TKIs) has attracted increasing interest as an anti-cancer therapy [9,10,11,12]. Several active intrinsic immune evasion pathways, including overexpression of vascular endothelial growth factor (VEGF), have been linked to the development and progression of HCC. AntiVEGF agents reduce VEGFmediated immunosuppression and induce blood vessel normalization within the tumor and its microenvironment, which may enhance anti-PD-1 efficacy by reversing VEGFmediated immunosuppression and promoting Tcell infiltration in tumors [13]. An open-label, multicenter, phase 2 trial (RESCUE) using camrelizumab (a PD-1 inhibitor) along with apatinib (an VEGFR-2 inhibitor) as first-line therapy for treating patients with Ad-HCC presented at ESMO 2020 reported a median OS of 20.3 months (95% CI 15.0–NR) with an overall response rate of 34% per RECIST 1.1 and 46% per mRECIST [14]. Moreover, another open-label, multicenter, phase 3 trial (CARES-310) reported that camrelizumab plus ribonucleotide (apatinib) showed a statistically significant and clinically meaningful survival benefit compared with sorafenib for patients with unresectable HCC [15]. Based on the CARES-310 study, China National Medical Products Administration (NMPA) has approved apatinib plus camrelizumab as the first-line treatment of Ad-HCC. Different from Europe and America, the local hepatic treatments including transcatheter arterial chemoembolization (TACE) and radiotherapy were recommended for the treatment of Ad-HCC by China Liver Cancer Staging (CNLC) guideline due to the relatively heavy hepatic tumor burden. A prospective, phase II, open-label study on the TRIPLET protocol has demonstrated the ideal efficacy and relatively acceptable safety of the treatment (ORR per RECIST v1.1: 77.1%, mPFS: 10.38 months, no grade 5 AEs) [16].
Considering the success of these combined therapies, we designed a combination protocol by combining HAIC of FOLFOX with camrelizumab plus apatinib (TRIPLET protocol) for advanced HCC. In this study, we investigated the effectiveness and safety of the TRIPLET protocol and camrelizumab and apatinib treatment in Ad-HCC.
Materials and methods
The treatment decision was principally made by the patient and their family under the recommendations of interventional oncologists and surgeons. Informed consent was waived because of the retrospective nature of the study. Starting from the onset of combination treatment, all patients were routinely assessed for safety and treatment response. All patients were followed up for the evaluation of toxicity and treatment outcome under the approval of the hospital ethics committee of SYSUCC (B2023-411). This study was conducted in accordance with the principles of the Declaration of Helsinki.
Patients’ inclusion
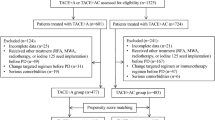
This retrospective study reviewed 922 consecutive patients with Ad-HCC who received combined therapy at three hospitals from April 1, 2019, to October 31, 2022. The inclusion criteria were as follows: (1) patients were aged 18–75 years; (2) the patients with HCC were clinically or pathologically confirmed to have BCLC Stage B or C HCC; (3) Eastern Cooperative Oncology Group performance status of 0 or 1; (4) adequate hepatic function with Child–Pugh A or B7; (5) at least one intrahepatic evaluable tumor; and (6) the patients received HAIC plus camrelizumab and apatinib (TRIPLET group) or camrelizumab and apatinib (C–A group). The exclusion criteria were as follows: (1) active or prior autoimmune disease; history of immunosuppressive agent use; (2) history of any other PD-L1/PD-1 antagonist treatment; (3) history of any local treatments including liver section, tumor ablation or transhepatic arterial chemoembolization; (4) HCC combined with other malignancies; (5) simultaneous treatment of TACE combined with HAIC; and (6) incomplete clinical and follow-up data.
The patients in the C–A group all met the following requirements: (1) at least one time camrelizumab injection; (2) at least 1 month apatinib treatment; (3) the injection of camrelizumab occurred the administration of apatinib; (4) during the apatinib plus camrelizumab treatment without any local treatments including ablation, TACE and HAIC. By contrast, the patients included in the TRIPLET group met the following criteria: (1) at least one time camrelizumab injection; (2) at least 1 month apatinib treatment; (3) at least 1 HAIC circle; (4) the injection of camrelizumab occurred during the administration of apatinib; (5) HAIC was performed during or in the previous week to the administration of apatinib; (6) during the apatinib plus camrelizumab treatment without any local treatments including ablation and TACE. All participating institutions strictly followed the inclusion and exclusion criteria to ensure consistency in the baseline characteristics of the population. The treatment regimen for patients would be recommended by the multi-disciplinary team (MDT) that consist of surgical oncologists, radiotherapist, diagnostic radiologist and interventional radiologist. Final decision-making was dominated together with patients and their family members according to patients’ willingness and economic condition.
Treatment regimen
For the HAIC procedure, a 5-French Yashiro or right hepatic catheter was inserted through the femoral artery with a 2.7-French microcatheter inside. The tip of the microcatheter was located in the tumor feeding artery on day 1 in every treatment cycle. The location of the tip was dependent on the arterial supply of the tumor identified by arteriography: the right/left hepatic artery for tumors in the right/left lobe, and the proper hepatic artery for tumors in two lobes. When the tumor accepted blood supply from extrahepatic arteries simultaneously, such as the celiac trunk or the superior mesenteric artery, the tip was located in the superior feeding artery and the sub-superior arteries were embolized. If necessary, the gastroduodenal artery was embolized using coil embolization. The administration of medication was initiated within 3 days of catheter insertion. The therapeutic scheme was a modified FOLFOX7 regimen, including oxaliplatin (85 mg/m2 infusion for 3 h on day 1), leucovorin (400 mg/m2 for 2 h from 4 to 5 h on day 1), and fluorouracil (2,500 mg/m2 continuous 46-h infusion on days 1 to 3). All chemotherapeutic agents were delivered via HAIC. The catheter and sheath were removed after the completion of HAIC and reinserted for the next HAIC cycle. The criteria for protocol discontinuation are described in Supplementary Information E1.1.
Apatinib (Jiangsu Hengrui Medicine Co. Ltd), a selective VEGFR-2 inhibitor was continuously administered orally at the dose of 250 mg once a day. Camrelizumab, (Jiangsu Hengrui Medicine Co. Ltd), a programmed cell death protein-1 (PD-1) blocker was administered intravenously at the dose of 200 mg each 21-day. The apatinib and camrelizumab treatment would be interrupted when the occurrence of progressive disease or unacceptable toxicity. Dose reduction for apatinib and chemotherapy agents was allowed because of unacceptable toxicity. Meanwhile, if the patients were diagnosed with hepatitis B virus (HBV) infection, he or she would receive antiviral therapy: the oral administration of entecavir or tenofovir. In addition, serum HBV DNA test would be carried out to adjust the regimen of antiviral therapy.
Data collection and follow-up protocol
To maintain the balance between the TRIPLET and C–A groups and analyze the risk factors associated with survival outcomes, we collected 29 clinical variables related to the enrolled patients. The variable definitions are described in Supplementary Information E1.2. In this study, enrolled patients were censored at the last follow-up date (June 30, 2023). After a thorough TRIPLET procedure was accomplished, the serum alpha-fetoprotein (AFP) and dynamic contrast-enhanced images were examined again at 1–3-month intervals during TRIPLET and at approximately 3-month intervals in the first year and every 6-month intervals thereafter. If suspected metastasis was encountered, chest computed tomography (CT), whole-body bone scans, or positron emission tomography (PET)-CT were performed selectively. The post-study treatment in follow-up is shown in Table S1.
Outcomes and safety
The responses to combined therapy were assessed by dynamic contrast-enhanced CT or magnetic resonance imaging (MRI) by the investigator as per the modified RECIST (mRECIST), including complete response (CR), partial response (PR), stable disease (SD), and progression disease (PD), which was evaluated independently by two radiologists with 10 years of experience who were blinded to the combined therapy procedures at the time of data collection. In this study, the outcome measures were overall survival (OS) and progression-free survival (PFS). OS was calculated from the date of initial treatment to the date of death from any cause or the deadline for follow-up. The date from the first treatment to the date of PD or the end of follow-up was applied for the calculation of PFS. The other outcomes we assessed were the ORR and disease control rate (DCR). The ORR was defined as the percentage of patients with CR and PR lasting more than 4 weeks from the first radiological confirmation, while the DCR was defined as the percentage of patients with CR, PR, and SD.
Safety assessments were based on symptom, laboratory tests and vital signs during therapy and follow-ups. Outpatient and telephone follow-up by special clinical assistants every 3–6 months were finished. Adverse events (AEs) were evaluated on the basis of Common Terminology Criteria for Adverse Events v5.0. Assessments.
Statistical analysis
Statistical analysis was performed using SPSS version 25.0 (IBM Corp., NY, USA) and the R software version 4.2.2 (http://www.r-project.org/). The quantitative variables with mean ± standard deviation or median with range or interquartile range (IQR) were compared by the Student t-test. The qualitative variables with frequency were compared using the 2 test (or Fisher exact test, if applicable). The factors including vascular invasion, extrahepatic metastasis, CTP grade and BCLC stage which may influence the survival and tumor response were chosen to perform propensity score matching (PSM). We applied 1:1 PSM using a nearest-neighbor algorithm with the 0.03 caliper width to adjust the potential unbalance variables which may influence the patients’ prognosis in both treatment groups. The survival results were compared using the Kaplan–Meier method with log-rank test. Univariate analyses of independent prognostic factors were evaluated by means of the Cox regression model, and then multivariable analyses were applied with the factors showing significance in the univariate analyses.
All tests of significance were two-sided and a p < 0.05 was interpreted to carry statistical significance.
Results
Patients enrolled
Figure 1 demonstrates the enrollment pathways of patients with Ad-HCC who underwent combined therapy. Finally, 416 treatment-naïve patients with HCC (43 females and 373 males; mean age, 50.7 ± 10.1 years) were reviewed in the study. Among them, 207 patients were included in the TRIPLET (T) group, and 209 patients were included in the camrelizumab and apatinib (C–A) groups. The baseline characteristics stratified by combined therapy modality are outlined in Table 1. Overall, 88.2% patients in Barcelona Clinic Liver Cancer (BCLC) C stage, 86.2% had hepatitis B virus etiology, 28.3% had an AFP concentration of > 400 ng/ml. Several clinical variables including Child–Turcotte–Pugh (CTP) grade, tumors’ maximum diameter and number, AFP, vascular invasion, metastasis, BCLC stage, and ALBI grade have significantly different distribution between T group and C–A group (p < 0.05). After 1:1 PSM, the variables have no significant statistical difference.
Antitumor activity
The antitumor activity comparison between the T and C–A groups is shown in Table 2. After PSM 1:1, the optimal ORR in T group was higher significantly than those in C–A group (69.7% vs. 38.5%, p < 0.001). 12.1% (25/207) HCC patients occurred downstage after TRIPLET, showing significantly higher than that (4.5%) in C–A group (p < 0.001). Moreover, the conversion surgery rate in the T group was significantly higher than that in the C–A group (p < 0.001). Notably, curative surgical resection after TRIPLET was conducted in 54 patients (26.2%, 54/207) in the T group and 16 patients (7.7%, 16/209) in the C–A group, owing to intrahepatic tumor shrinkage which made the patients meet the Milan criteria.
Oncological outcome comparison
The median follow-up duration for the T and C–A groups was 22.8 months (IQR: 8.2–61.3 months) and 24.5 months (IQR: 6.4–48.8 months), respectively. In the crude Kaplan–Meier analyses, significant differences were observed with regard to PFS (median PFS: T, 11.8 months vs C–A, 5.8 months; p < 0.001; Fig. 2A) and OS (median OS: T, Not reached vs C–A, 20.9 months; p < 0.001; Fig. 2B). The PSM adjusted Kaplan–Meier analyses also showed that significant differences were observed with regard to PFS (median PFS: T, 11.5 months vs C–A, 5.6 months; p < 0.001; Fig. 2C) and OS (median OS: T, Not reached vs C–A, 19.9 months; p = 0.001; Fig. 2D).
Kaplan–Meier curves of cumulative 2-year overall survival rate and 2-year progression-free survival rate in two different groups. a Cumulative 2-year progression-free survival rate before propensity score matching; b cumulative 2-year overall survival rate before propensity score matching; c cumulative 2-year progression-free survival rate after propensity score matching; d cumulative 2-year overall survival rate after propensity score matching
Risk factors for survival outcomes
The risk factors for OS and PFS were assessed by univariate and multivariate analyses prior to the PSM (Table 3). In the univariate analyses, CTP-B [hazard ratio (HR): 2.86; 95% confidence interval (CI) 1.67–5.87; p < 0.001], tumor number > 3 (HR: 1.39; 95% CI 1.04–1.85; p = 0.024), metastasis (HR: 10.44; 95% CI 1.07–1.93; p = 0.017), and camrelizumab plus apatinib (HR: 1.79; 95% CI 1.33–2.40; p < 0.001) were significant factors for poor OS. Multivariate analyses showed that the factors that significantly affected the OS rate were CTP-B (HR: 4.73; 95% CI 2.50–8.94; p < 0.001), tumor number > 3 (HR: 1.72; 95% CI 1.28–2.32; p < 0.001), and camrelizumab plus apatinib (HR: 2.34; 95% CI 1.64–3.32; p < 0.001). In the univariate analyses, ECOG-PS 1 (HR: 1.53; 95% CI 1.14–2.06; p = 0.005), tumor number > 3 (HR: 1.30; 95% CI 1.03–1.66; p = 0.029), metastasis (HR: 1.50; 95% CI 1.17–1.91; p = 0.001), and camrelizumab plus apatinib (HR: 1.83; 95% CI: 1.43–2.35; p < 0.001) were significant factors for poor PFS. Multivariate analyses showed that tumor number > 3 (HR: 1.56; 95% CI 1.22–2.01; p < 0.001) and camrelizumab plus apatinib (HR: 1.96; 95% CI 1.44–2.65; p < 0.001) significantly affected the PFS rate.
Subgroup analysis
Prior to the PSM, the subgroup analyses of OS and PFS based on clinical variables using forest plots are shown in Fig. 3A and B, which showed that TRIPLET provided a clinical benefit for OS and PFS, outperforming camrelizumab and apatinib in most clinical variables. These results suggested that the TRIPLET appeared to particularly benefit patients with age ≤ 65 years, male, HBV, CTP A, 1–3 tumor number, BCLC C grade (HR: 1.34; 95% CI 1.17–1.54, p < 0.001), > 5 cm tumor diameter, without metastasis and ALBI grade 1 based on the OS and PFS. These results provide an interventional treatment strategy for patients with different clinical factors.
Subgroup analysis of two different two groups. a Forest plot for overall survival; b Forest plot for progression-free survival. ECOG-PS: Eastern Cooperative Oncology Group; HBV: hepatitis B virus; CTP: Child–Turcotte–Pugh; BCLC: Barcelona Clinic Liver Cancer; HCC: hepatocellular carcinoma; AFP: alpha-fetoprotein
Safety
The adverse event (AE) comparison between the TRIPLET and C–A groups is shown in Table 4. Before PSM, the incidence of patients with AE was 82.3% in the TRIPLET group, which was significantly higher than that in the C–A group (71.3%) (p = 0.01). The most common AE was elevated AST in C–A group as well as abdominal pain in the TRIPLET groups. The incidence of patients with grade 3–4 AEs in total was 82.1% in the TRIPLET group, which was higher than that in the C–A group at 71.3% (p = 0.01). Moreover, the frequencies of grade 3–4 vomit, abdominal pain, elevated ALT, elevated AST, diarrhea, neutropenia, thrombocytopenia and fatigue were significantly higher in the TRIPLET group than in the C–A monotherapy group. Furthermore, abdominal pain was observed in most of patients in the TRIPLET group after oxaliplatin injection, although the pain was relieved when the injection of oxaliplatin was stopped instantly. Following PSM, there remained no significant difference between the two groups in any grade AE (TRIPLET vs. C–A, 79.8% vs. 60.6%, p = 0.08). Meanwhile, the incidence of grade 3–4 AEs in the TRIPLE group was higher than that in the C–A group (78.0% vs. 64.2%, p < 0.001). No grade 5 AEs were observed in the study.
Discussion
This retrospective cohort study demonstrated that the combined treatment protocol of HAIC plus camrelizumab and apatinib (TRIPLET) is both safe and feasible. In this study, TRIPLET provided a significant OS benefit compared with camrelizumab and apatinib. CARES-301 trial had revealed camrelizumab combined with apatinib was an effective first-line treatment option for uHCC, while the ORR was only 25% and lack of evidence for patients with high tumor burden [15]. Previously, some triple combination treatment protocols have also been registered and are recruiting eligible patients for enrollment. Beyond LEAP-002, the LEAP-012 trial assessed the safety and efficacy of TACE plus lenvatinib plus pembrolizumab in participants with incurable/non-metastatic HCC compared with TACE alone [17]. In addition, the EMERALD-1 global study aimed to evaluate TACE plus durvalumab plus bevacizumab therapy in patients with locoregional HCC compared with TACE plus durvalumab or TACE alone [18]. In this study, we chose HAIC instead of TACE because the standard operating procedure could be standardized and was technically easy to repeat. Furthermore, HAIC avoids most of the uncertain factors affecting TACE, including the lack of standard medication usage, distinctive operating skills, and different operator habits. Therefore, HAIC-oriented combined therapy has the potential to be popularized with a standard dose regimen.
In this study, the ORR, PFS, and OS of TRIPLET treatment outperformed those of current TKI or ICI monotherapy [19], including lenvatinib (ORR in 18.8% and PFS in 7.4 months) in the REFLECT trial [20], nivolumab (ORR in 15% and PFS in 3.7 months) in the CheckMate 459 trial [21], and pembrolizumab (ORR in 18.3% and PFS in 3.0 months) in the KEYNOTE-240 trial [22]. In addition to its superiority to mono-agent therapies, TRIPLET treatment was considered superior to two-agent combined therapies. Compared with the landmark IMbrave-150 trial, which has changed the first-line recommendation of BCLC guidelines, the results from this TRIPLET treatment showed a median PFS of 12.0 months with an ORR of 67.2% as per mRECIST, which is significantly higher than the ORR of 33.2% as per mRECIST and the median PFS of 6.8 months reported by the IMbrave-150 trial [9]. The advantage of TRIPLET is also evident compared with the KEYNOTE-524, LEAP-002 and ORIENT-32 trial, in which lenvatinib and pembrolizumab, as well as sintilimab and bevacizumab-biosimilar were combined for patients with unresectable HCC. Both the estimated median PFS (KEYNOTE-524: 9.7 months, LEAP-002: 7.4 months, ORIENT-32: 4.6 months) and ORR (KEYNOTE-524: 46.3% per mRECIST, LEAP-002: 40.8%, ORIENT-32: 24.3%) of the above trials were lower than those of TRIPLET treatment [10, 12, 23].
In most cases, the patients with high tumor burden have shorter survival and the main cause of death in HCC is intrahepatic tumor progression. The IMbrave-150 study showed that the mOS of patients with high risk (VP4 portal vein invasion, and/or bile duct invasion and/or tumor occupancy of ≥ 50% of liver) is only 7.6 m, which is far shorter than non-high risk group [11]. Combination with HAIC may reduce the tumor burden and prolong survival. In the treatment decision-making of our study, most patients with high tumor burden were recommended local treatment combination with systematic treatment by MDT based on the Guidelines for Diagnosis and Treatment of Primary Liver Cancer in China (2022 Edition), so the tumor burden and AFP level have not been chosen to perform PSM. Even though the TRIPLET group has the lager tumor burden than C–A group, HAIC combined with camrelizumab and apatinib is still associated with better efficacy.
The survival benefit observed in this study may be due to the synergistic antitumor effects of the chemical agents (HAIC of FOLFOX), antiangiogenic agents, and PD-1 inhibitors. Instead of intravenous chemotherapy, HAIC injects chemical reagents directly into the tumor. Oxaliplatin can induce immunogenic cell death by releasing tumor antigens, transporting CRT to the cell surface, and secreting HMGB1 and ATP [24,25,26]. These molecules related to cell death bind to their respective receptors and support the evolution of tumor-specific CD8+ T cells. Indeed, combined antiangiogenic and anti-PD-1/PD-L1 therapy has been shown to elicit T-cell function and drive tumor cells to activate immune checkpoints, thereby generating greater antitumor immunity than anti-PD-1 treatment alone [13]. In addition, the low dose of apatinib therapy (250 mg daily) used in this study has been proven to induce prolonged vascular normalization, thereby reducing tumor hypoxia and acidosis and improving the anti-cancer activity of infiltrating immune cells [27].
During the median follow-up of 25.2 months, we observed increased ORR in the T group compared with those in the C–A group. However, the DCRs were similar between the two groups. In the T group, PD was observed in 71 patients (34.3%), 60.6% (43/71) of whom showed extrahepatic metastasis (EHM). As a result, we considered EHM in patients to indicate a tendency toward PD because HAIC is highly selective for intrahepatic tumors, and some patients with EHM did not achieve no evidence of disease (NED). However, multivariate analysis using the Cox regression model did not identify EHM as an independent prognostic factor for PFS, possibly because of the small sample size of this study. However, for patients who achieved a CR, the duration of the CR has not been reached, indicating satisfactory efficacy prolongation. One patient, who had evaluated as CR after 2 HAIC circles, died from decompensated liver function-related upper gastrointestinal bleeding after three cycles of TRIPLET treatment and subsequently refusing treatment. We also performed sub-analyses for OS and PFS based on common clinical variables. The results indicated that the patients with HCC who received TRIPLET obtained better tumor control and survival benefit than those who received camrelizumab and apatinib in most clinical variables, including ≤ 65 years old, male, ECOG-PS 0, CTP stage A, > 5 cm tumor diameter, and BCLC stage C. Moreover, radiological features, including pseudo-capsulated and infiltrative, are key indicators for combination therapy selection. Wu et al. also provided evidence that HAIC can obtain better outcomes than TACE for infiltrative HCC.
In terms of safety, the T and C–A groups showed some differences in the frequencies and severity of adverse events. The frequencies of both total AEs and grade 3–4 AEs were higher in the T group than those in the C–A group regardless of PSM. Except abdominal pain, the first and second most common grade 3–4 AEs were elevated AST and thrombocytopenia in the T group, which can be explained by chemotherapy-related liver damage and myelosuppression. Moreover, the increased frequencies of abdominal pain, nausea, vomiting, and diarrhea may be caused by chemotherapy, especially for drug diversion to the gastrointestinal tract or cholecyst. Therefore, we sometimes performed gastroduodenal artery embolization during HAIC to reduce drug diversion. Paradoxically, hypertension was the most frequently occurring AE in the C–A group, which was found to a greater extent than that in the T group. One possible explanation for this is that many patients in the T group were treated with chemotherapy, and the side effects, such as fatigue and anemia, counteracted the hypertension caused by the C–A regimen. Even so, we demonstrate that the safety of the TRIPLET scheme in treating advanced HCC was acceptable and similar to the findings of existing studies [16, 28,29,30,31].
This study has several limitations that warrant discussion. First, as a retrospective study, the patients were consecutively enrolled, which may have introduced patient selection bias, so some baseline characteristics of patients between two group is different. Second, no biomarker analysis was performed to determine the patients who would benefit most from the TRIPLET combination therapy. Third, the mean follow-up duration in TRIPLET group was not long enough.
Conclusion
TRIPLET has more promising survival benefits than apatinib and camrelizumab in the management of Ad-HCC, as well as showing tolerable safety. The efficacy and safety of TRIPLET still need to be confirmed by prospective phase III study in the future.
Data availability
The datasets supporting the conclusions of this manuscript are available in the Sun Yat-sen University Cancer Center Research Data Deposit repository (https://www.researchdata.org.cn/).
References
Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. https://doi.org/10.3322/caac.21660
El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557–2576. https://doi.org/10.1053/j.gastro.2007.04.061
Qu LS, Jin F, Huang XW, Shen XZ. High hepatitis B viral load predicts recurrence of small hepatocellular carcinoma after curative resection. J Gastrointest Surg. 2010;14(7):1111–1120. https://doi.org/10.1007/s11605-010-1211-1
Zhou L, Wang SB, Chen SG, Qu Q, Rui JA. Risk factors of recurrence and poor survival in curatively resected hepatocellular carcinoma with microvascular invasion. Adv Clin Exp Med. 2020;29(7):887–892. https://doi.org/10.17219/acem/76750
Reig M, Forner A, Rimola J, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76(3):681–693. https://doi.org/10.1016/j.jhep.2021.11.018
Yasui Y, Tsuchiya K, Kurosaki M, et al. Up-to-seven criteria as a useful predictor for tumor downstaging to within Milan criteria and Child-Pugh grade deterioration after initial conventional transarterial chemoembolization. Hepatol Res. 2018;48(6):442–450. https://doi.org/10.1111/hepr.13048
Lyu N, Wang X, Li JB, et al. Arterial chemotherapy of oxaliplatin plus fluorouracil versus sorafenib in advanced hepatocellular carcinoma: a biomolecular exploratory, randomized, phase III trial (FOHAIC-1). J Clin Oncol. 2022;40(5):468–480. https://doi.org/10.1200/JCO.21.01963
Li QJ, He MK, Chen HW, et al. Hepatic arterial infusion of oxaliplatin, fluorouracil, and leucovorin versus transarterial chemoembolization for large hepatocellular carcinoma: a randomized phase III trial. J Clin Oncol. 2022;40(2):150–160. https://doi.org/10.1200/JCO.21.00608
Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894–1905. https://doi.org/10.1056/NEJMoa1915745
Ren Z, Xu J, Bai Y, et al. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomised, open-label, phase 2–3 study. Lancet Oncol. 2021;22(7):977–990. https://doi.org/10.1016/S1470-2045(21)00252-7
Finn RS, Qin S, Ikeda M, et al. IMbrave150: Updated efficacy and safety by risk status in patients (pts) receiving atezolizumab (atezo) + bevacizumab (bev) vs sorafenib (sor) as first-line treatment for unresectable hepatocellular carcinoma (HCC). Conf Abstr Cancer Res. 2021. https://doi.org/10.1158/1538-7445.AM2021-CT009
Finn RS, Kudo M, Merle P, et al. LBA34 Primary results from the phase III LEAP-002 study: lenvatinib plus pembrolizumab versus lenvatinib as first-line (1L) therapy for advanced hepatocellular carcinoma (aHCC). Conf Abstr Ann Oncol. 2022;33:S1401. https://doi.org/10.1016/j.annonc.2022.08.031
Yi M, Jiao D, Qin S, Chu Q, Wu K, Li A. Synergistic effect of immune checkpoint blockade and anti-angiogenesis in cancer treatment. Mol Cancer. 2019;18(1):60. https://doi.org/10.1186/s12943-019-0974-6
Xu J, Shen J, Gu S, et al. Camrelizumab in combination with apatinib in patients with advanced hepatocellular carcinoma (RESCUE): a nonrandomized, open-label. Phase II Trial Clin Cancer Res. 2021;27(4):1003–1011. https://doi.org/10.1158/1078-0432.CCR-20-2571
Qin S, Chan SL, Gu S, et al. Camrelizumab plus rivoceranib versus sorafenib as first-line therapy for unresectable hepatocellular carcinoma (CARES-310): a randomised, open-label, international phase 3 study. Lancet. 2023. https://doi.org/10.1016/S0140-6736(23)00961-3
Zhang TQ, Geng ZJ, Zuo MX, et al. Camrelizumab (a PD-1 inhibitor) plus apatinib (an VEGFR-2 inhibitor) and hepatic artery infusion chemotherapy for hepatocellular carcinoma in Barcelona Clinic Liver Cancer stage C (TRIPLET): a phase II study. Signal Transduct Target Ther. 2023;8(1):413. https://doi.org/10.1038/s41392-023-01663-6
Llovet JM, Vogel A, Madoff DC, et al. Randomized phase 3 LEAP-012 study: transarterial chemoembolization with or without lenvatinib plus pembrolizumab for intermediate-stage hepatocellular carcinoma not amenable to curative treatment. Cardiovasc Intervent Radiol. 2022;45(4):405–412. https://doi.org/10.1007/s00270-021-03031-9
Sangro B, Kudo M, Qin S, et al. P-347 A phase 3, randomized, double-blind, placebo-controlled study of transarterial chemoembolization combined with durvalumab or durvalumab plus bevacizumab therapy in patients with locoregional hepatocellular carcinoma: EMERALD-1. Conf Abstr Ann Oncol. 2020;31:S202–S203. https://doi.org/10.1016/j.annonc.2020.04.429
Stefanini B, Ielasi L, Chen R, et al. TKIs in combination with immunotherapy for hepatocellular carcinoma. Expert Rev Anticancer Ther. 2023;23(3):279–291. https://doi.org/10.1080/14737140.2023.2181162
Kudo M, Finn RS, Qin S, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391(10126):1163–1173. https://doi.org/10.1016/S0140-6736(18)30207-1
Yau T, Park JW, Finn RS, et al. Nivolumab versus sorafenib in advanced hepatocellular carcinoma (CheckMate 459): a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2022;23(1):77–90. https://doi.org/10.1016/S1470-2045(21)00604-5
Zhu AX, Finn RS, Edeline J, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19(7):940–952. https://doi.org/10.1016/S1470-2045(18)30351-6
Finn RS, Ikeda M, Zhu AX, et al. Phase Ib study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma. J Clin Oncol. 2020;38(26):2960–2970. https://doi.org/10.1200/JCO.20.00808
Zhu H, Shan Y, Ge K, Lu J, Kong W, Jia C. Oxaliplatin induces immunogenic cell death in hepatocellular carcinoma cells and synergizes with immune checkpoint blockade therapy. Cell Oncol. 2020;43(6):1203–1214. https://doi.org/10.1007/s13402-020-00552-2
Srivastava S, Furlan SN, Jaeger-Ruckstuhl CA, et al. Immunogenic chemotherapy enhances recruitment of CAR-T cells to lung tumors and improves antitumor efficacy when combined with checkpoint blockade. Cancer Cell. 2021;39(2):193-208.e10. https://doi.org/10.1016/j.ccell.2020.11.005
Tesniere A, Schlemmer F, Boige V, et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene. 2010;29(4):482–491. https://doi.org/10.1038/onc.2009.356
Zhao S, Ren S, Jiang T, et al. Low-Dose apatinib optimizes tumor microenvironment and potentiates antitumor effect of PD-1/PD-L1 blockade in lung cancer. Cancer Immunol Res. 2019;7(4):630–643. https://doi.org/10.1158/2326-6066.CIR-17-0640
He MK, Liang RB, Zhao Y, et al. Lenvatinib, toripalimab, plus hepatic arterial infusion chemotherapy versus lenvatinib alone for advanced hepatocellular carcinoma. Ther Adv Med Oncol. 2021;13:17588359211002720. https://doi.org/10.1177/17588359211002720
Liu BJ, Gao S, Zhu X, et al. Real-world study of hepatic artery infusion chemotherapy combined with anti-PD-1 immunotherapy and tyrosine kinase inhibitors for advanced hepatocellular carcinoma. Immunotherapy. 2021;13(17):1395–1405. https://doi.org/10.2217/imt-2021-0192
Mei J, Tang YH, Wei W, et al. Hepatic arterial infusion chemotherapy combined with PD-1 inhibitors plus lenvatinib versus PD-1 inhibitors plus lenvatinib for advanced hepatocellular carcinoma. Front Oncol. 2021;11:618206. https://doi.org/10.3389/fonc.2021.618206
Lai Z, He M, Bu X, et al. Lenvatinib, toripalimab plus hepatic arterial infusion chemotherapy in patients with high-risk advanced hepatocellular carcinoma: a biomolecular exploratory, phase II trial. Eur J Cancer. 2022;174:68–77. https://doi.org/10.1016/j.ejca.2022.07.005
Acknowledgements
We are thankful for the support of the patients in this study. We are grateful Miss. Xiao-Ting Bei for her assistance with the clinical management of the patients.
Funding
None.
Author information
Authors and Affiliations
Contributions
Yuzhe Cao, Mengxuan Zuo, Yi Yang, Peng Song, Chao An and Wang Li participated equally in conceptualization. Yuzhe Cao, Mengxuan Zuo and Yi Yang made great contribution in the in the data curation, formal analysis, methodology and investigation. Guanglei Zheng, Da Li, Hongyan Shao and Qiaoyun Ma have participated in the investigation as well and the validation. The project administration and supervision were accomplished by Peng Song, Chao An and Wang Li. Mengxuan Zuo, Yuzhe Cao and Yi Yang had finished the original draft. Peng Song, Chao An and Wang Li have been responsible for the review and editing. All the authors have provided final approval of the manuscript version to be submitted for publication.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest to declare.
Ethical approval
Informed consent was waived due to the retrospective nature of the analysis. The protocol of this study (SL-B2023-411-02) was reviewed and approved by the Sun Yat-sen University Cancer Center Ethics Committee (Approval Number: B2023-411-01).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zuo, M., Cao, Y., Yang, Y. et al. Hepatic arterial infusion chemotherapy plus camrelizumab and apatinib for advanced hepatocellular carcinoma. Hepatol Int (2024). https://doi.org/10.1007/s12072-024-10690-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12072-024-10690-6