Abstract
The Atlantic blue crab Callinectes sapidus (Decapoda, Portunidae) Rathbun, 1896 is native to the east coasts of North and South America and has recently expanded its distribution in the non-native range into the Gulf of Cadiz (SW Iberian Peninsula, Europe). Considering the impacts caused by this invasive species in numerous estuarine ecosystems and its generalist feeding behavior, this study aims to provide the first account of the Atlantic blue crab diet on the East Atlantic coast. We studied the species’ feeding habits using stomach content analyses to predict food web interactions and putative impacts. Samples were obtained in the Guadalquivir estuary (SW Spain, Europe), which was colonized in 2017. The main food items identified on their stomach were, fish (49.9%), mollusks (44.4%) and crabs (32.3%). They also consumed plant material (27.2%), and the sediment (32.3%) in their digestive tract was likely the result of secondary ingestion. The Atlantic blue crab exhibited the same omnivorous behavior as in the native area. There was no sexual variation in diet composition or feeding activity in general, but there was a seasonal variation in the diet composition of females. The decrease of the caramote prawn Penaeus kerathurus (Forskål 1775) observed in the Guadalquivir estuary since 2021 is likely not due to the Atlantic blue crab because they seldomly eat this prey. Overall, our study provides clear baseline information to expand the knowledge about the ecological roles of the Atlantic blue crab in non-native ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive species disrupt the functioning of ecosystems and the trophic processes in which they rank the most significant disruptors of established ecological networks (Wainright et al. 2021). The invasive Atlantic blue crab Callinectes sapidus Rathbun 1896, (Decapoda, Portunidae), native to the east coast of North and South America, has rapidly dispersed along the western Euro-African area since the mid-2010s (Mancinelli et al. 2021; Encarnação et al. 2021; González-Ortegón et al. 2022; Chairi and González-Ortegón 2022) and is known for multiple ecological impacts in areas of the Mediterranean Sea where they have been established for a long time (Mancinelli et al. 2017b).
The Atlantic blue crab is common on muddy and sandy bottoms from inter-tidal to subtidal areas up to 90-m depth (Van Engel 1958; Harris 1982; Hines et al. 1990). It tolerates a broad range of environmental conditions, including freshwater, but tends to complete its life cycle in coastal and estuarine habitats (Onofri et al. 2008; Nehring 2011). Mature adults usually mate in the low-salinity waters of upper estuaries, while females spawn offshore where the salinity regime favors larvae survival (Meise and Stehlik 2003), so males and females have, at times, different spatial distributions (Millikin and Williams 1984). The capacity for overwintering has been reported to influence their latitudinal range limits and distribution (Norse 1977). During wintertime, food becomes scarce, a challenge that can be addressed by migrating or reducing physiological activity (Hines et al. 1990; Conover 1992).
In the non-native area, the Atlantic blue crab inhabits coastal waters mainly associated with estuaries (Castejón and Guerao 2013; Mancinelli et al. 2021) and lagoons with freshwater inputs (Mancinelli et al. 2017c). The populations established along the Mediterranean Sea have supported profitable fisheries for the last 50 years (Ayas and Ozogul 2011; Kevrekidis et al. 2013). In the Gulf of Cadiz (SW Iberian Peninsula, Europe) and associated estuarine ecosystems, the Atlantic blue crab abundance increased substantially, and its distribution rapidly expanded since they were first detected in the region (Mancinelli et al. 2017a, c; Morais et al. 2019; González-Ortegón et al. 2020; Encarnação et al. 2021).
The increasing population of the Atlantic blue crab in the Mediterranean basin impacted established communities (Onofri et al. 2008; Marchessaux et al. 2023), including the displacement and extirpation by predation of the Japanese common clam Ruditapes philippinarum (A. Adams and Reeve 1850) (Fuentes et al. 2019; Clavero et al. 2022) and caramote prawn Penaeus kerathurus (Forskål 1775) (Guijarro-García et al. 2019), and predation and competition with the Mediterranean green crab Carcinus aeustuarii Nardo 1847 (Kampouris et al. 2019). Despite the negative impacts on established communities and fisheries in the Mediterranean Sea (Mancinelli et al. 2017b; Öndes and Gökce 2021), the impact of the Atlantic blue crab in the recently colonized areas of the Gulf of Cadiz is incipient. Morais et al. (2019) pointed out that this invasive crab is a potential competitor with native carcinofauna because of overlapping niches and functions in the ecosystem. Spanish fishers in the Gulf of Cadiz are particularly concerned about the decrease of the caramote prawn in local ports (e.g., Sanlucar de Barrameda, Andalusia, Spain) since they attribute its decrease to the establishment of the Atlantic blue crab in the Guadalquivir estuary (SW Spain, Europe).
Studying the diet of non-indigenous species helps predict food web interactions (Chaffai et al. 2020) and recognize the potential impacts on newly invaded areas (Triay-Portella et al. 2022). Stomach content analysis is a universal method for detailed exploration of diet composition and feeding ecology as it allows reasonably accurate quantification of a species’ diet in a full array of habitats (Hyslop 1980; Hines 2007; Triay-Portella et al. 2022). In the native area, the Atlantic blue crab is a generalist predator and an omnivore (Hines et al. 1990; Dittel et al. 2006), mostly considered as actively preying on fish, worms, mollusks, crustaceans, and even conspecifics (Darnell 1959; Millikin and Williams 1984; Clark et al. 1999; Mascaro et al. 2003). The Atlantic blue crab can also eat plant material (Van Engel 1958) and ingest sediment and detritus, but in lower quantities (Darnell 1959). An increase in the ingestion of sediments and plant material has been observed in Atlantic blue crabs inhabiting contaminated areas, as their predatory capacity is constrained by these contaminants (Reichmuth 2009; Belgrad and Griffen 2016). The Atlantic blue crab occasionally presents a scavenger behavior upon dead fish, prawns, and crabs (Van Engel 1958; Scherer et al. 2017). In the Mediterranean basin, Atlantic blue crabs show similar feeding habits as in their native area, feeding mainly on mollusks (Pla et al. 2018; Rady et al. 2018; Gil 2018; Prado et al. 2021), fish, and crabs (Carrozzo et al. 2014). The Atlantic blue crab diet is influenced by environmental conditions, such as salinity and temperature (Seitz et al. 2011; Mancinelli et al. 2017c), seasonal variation in food items (Laughlin 1982; Haefner 1990; Hsueh et al. 1992), and energy requirements (Rosas et al. 1994). Males and females exhibit similar diets when inhabiting the same habitat (Hsueh et al. 1992).
Considering previous observations made in the native and non-native ranges, we hypothesized that, in the Guadalquivir estuary, the Atlantic blue crab (1) has feeding habits similar to those observed in the native area, (2) their diet composition and feeding activity will change between seasons but not sex, (3) the feeding activity will be more intense in summer, and (4) the caramote prawn will be a preferred prey and thus explaining why their population has been declining. To test these four hypotheses, we aimed to describe the feeding habits of mature male and female Atlantic blue crabs by analyzing their stomach contents, comparing their diet between seasons and sex, and assessing a putative impact upon the caramote prawn.
Material and Methods
Study Area, Sampling, and Laboratory Procedures
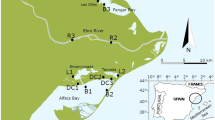
The Guadalquivir estuary (SW Spain, Europe) extends for 110 km and has a maximum width of 800 m at the river mouth (Fig. 1). At the river mouth, water temperature ranges from 11.7 ± 0.3 °C in January to 25.5 ± 0.3 °C in July–August, and the annual average water temperature is 19.0 ± 1.5 °C (González-Ortegón et al. 2015a). The tidal range is 3.5 m, and salinity intrusion extends up to 50 km upstream (Drake et al. 2002). The Alcala del Río dam, located 110 km from the river mouth, partially controls the estuary’s tidal regime and salinity fluctuations (Fernández-Delgado et al. 2007). Annual river flow varies between 40 and 400 m3 s−1, while the river discharge averages 31 hm3 in dry years and 18.862 hm3 in wetter years.
Atlantic blue crab specimens were collected at the mouth of the Guadalquivir estuary (36°47′38.8″N, 6°22′33.1″W) (Fig. 1) using a seine net (3–5-cm mesh, 3-m length × 0.7-m width) pulled by a boat. Samples were frozen at −20 °C until laboratory analyses. Sampling occurred during three seasons — summer (July, August, and September), fall (October and November), and winter (December and March) — in 2020 and 2021. No specimens were collected during spring despite the multiple attempts to collect Atlantic blue crabs. Specimens were sexed by inspecting the pleon’s morphology, the carapace width (CW) and length (CL) were measured with a Vernier caliper (± 0.1 mm), and the total weight was determined with a Kern scale (± 0.01 g).
The stomach contents of 155 Atlantic blue crabs were inspected under a stereomicroscope, and food items were identified with the support of identification guides (Campbell 1982). Prey was identified to the class or order level due to the advanced digestion state of prey. So, we often relied exclusively on hard structures to identify prey.
Characterization of Diet and Feeding Activity
To characterize the diet of the Atlantic blue crab in the Guadalquivir estuary, we calculated the relative presence (RP) and frequency of occurrence (FO), while feeding activity was estimated with the response variables fullness state (FS), fullness index (FI), and digestion rate (DR). For these analyses, we assigned each prey or food item into nine categories: fish, mollusks, crabs, cephalopods, worms, caramote prawn, other arthropods, plant material, and sediment (Fig. 2). Sediment is not regarded as prey but as a secondary food item in the diet.
Photographs of structures from the main taxonomic groups found in the stomach contents of Atlantic blue crabs Callinectes sapidus Rathbun 1896 collected in 2020 and 2021 in the Guadalquivir estuary (SW Spain, Europe). A Shell hinge (mollusks), B ring teeth (cephalopods), C sediments, D chitinous limbs (crabs), E vertebra (fish), F setae (worms), G plant or algal tissue (plant material), H chitinous pleonal segment of caramote prawn Penaeus kerathurus (Forskål 1775), and I chitinous structure of an insect (other arthropods)
RP is quantified as the percentage abundance of prey items concerning a known surface, providing a qualitative assessment of the presence of each item in the stomach contents (Hines et al. 1990). The RP of each item in the stomach contents was assigned to each stomach using the visual method (Haram and Jones 1971). This method visually estimates deviation and is calculated from the total number of stomachs with food items. The total stomach content was spread in a Petri dish, and the percentage of prey abundance was estimated relative to the total surface of the Petri dish. The sum of each food item in each stomach was 100%. FO corresponds to the number of stomachs containing a given food item and is then expressed as a percentage (%) of the total analyzed stomachs.
RP and FO were initially calculated using data from 129 non-empty stomachs. However, due to the high abundance of non-identified organic items, we calculated the RP and FO indexes using data from stomachs with at least an identifiable food item (animal, plant, or sediment) to obtain more meaningful results. So, 99 stomachs with at least one food item were considered in our data analysis, 74 samples from females (summer = 46, fall = 7, winter = 21) and 25 from males (summer = 9, fall = 8, winter = 8).
The FS was assessed visually and consisted of six levels according to the quantity of food in the stomach (Hines 2007; Rady et al. 2018) — State 0: empty stomach; State 1: food occupies less than 5% of the stomach; State 2: food occupies between 5 and 35% of the stomach; State 3: food occupies between 35 and 65% of the stomach; State 4: food occupies between 65 and 90% of the stomach; and State 5: food occupies 90 to 100% of the stomach. The FI was estimated with Eq. 1 (Hyslop 1980):
The FI was calculated using specimens with all the legs, carapace, and chelas (83 out of 155 individuals) to avoid biases introduced by missing body structures.
The DR refers to the varying degrees or stages of how the stomach contents were found digested or broken down within the stomach itself, and it was determined visually with a subsample of 99 stomachs containing any identified food item. The levels of the stomach content digestibility were defined according to Buckland et al. (2017) with slight modifications: Level 1: non-digested and identifiable food item; Level 2: digested identifiable food item; Level 3: identifiable biological structures linked to organic digested matter; Level 4: identifiable biological inorganic structures unlinked to organic digested matter; and Level 5: unidentifiable digested contents.
Stomach content analyses in portunids are limited when quantifying the prey items (Williams 1981). For example, to estimate the FO some authors exclude stomachs with FS ≤ 3 (Reichmuth et al. 2009). In the case of the RP index, the total food item abundance adjustment based on the FS (Hines et al. 1990; Rady et al. 2018) is done to normalize the relative importance of prey for the total diet (Williams 1981).
To ensure comparability with other studies, we corrected the RP using the FS as a weighting factor by multiplying the RP of each food item in each stomach by levels of punctuation related to its FS as defined by Wear and Haddon (1987) (see Rady et al. 2018). The FS punctuation depends on the stomach’s relative degree of fullness, as follows: FS 0 = 0.00 points, FS 1 = 0.02 points, FS 2 = 0.25 points, FS 3 = 0.50 points, FS 4 = 0.75 points, and FS 5 = 1 point.
Selectivity Index
The Food Item Selectivity Index (Li) for an item (i) was estimated using the linear food selection model proposed by Strauss (1979). For a determined type of item, this model aims to describe and quantify the relationship between ri (percentage of abundance of a food item in the stomach) and pi (percentage of abundance of the same food item in the environment) (Eq. 2):
ri was determined using the mean and standard deviation of RP index across all stomachs analyzed. pi was inferred using the means and standard deviations of temporal series data representing the percentage abundance of different groups of benthic and nektonic fauna sampled regularly since 1997 in the Guadalquivir estuary (Drake et al. 2002; González-Ortegón et al. 2015a). The mean of the temporal series of a series of faunal species abundance in the Guadalquivir estuary showed that the most abundant groups, in terms of percentage in the community, were other arthropods (93.7%), fish (4.5%), and crabs and other decapods (1.9%) for the nektonic taxa, and worms (98.9%) and mollusks (0.3%) for benthic taxa. The Li estimation was limited to available data on local food items. The available local benthic and nektonic fauna were grouped into five categories: other arthropods (mostly Mysidacea), crabs and other decapods, fish, worms, and mollusks.
Data Analysis
We used a non-metric multidimensional scaling (MDS) ordination with the Bray–Curtis similarity measurement for diet composition data (RP and FO) and Euclidian distance similarity for feeding activity data (FS, FI, and DR) to assess seasonal and sexual differences in the diet composition. The relative importance of seasonal and sexual variability in the feeding habits was estimated by calculating the average similarity of samples. Diet composition and feeding activity were estimated (a) for the monthly samples to study seasonal changes (summer, fall, and winter) and (b) between females and males to determine differences between sex. A PERMANOVA analysis with covariate inclusion was performed for diet composition to assess size (CW) effects (ANCOVA) and tested using 9999 permutations. For feeding activities, a two-way crossed ANOSIM analysis was conducted to assess differences between seasons and sex using 9999 permutations. These analyses were done with PRIMER 7 (Plymouth Routines in Multivariate Ecological Research) (Clarke and Gorley 2006).
A Mantel test was done in R v4.3.8 (R core Team 2020), to assess the correlation between diet composition measured as RP%, with and without considering the FS of the stomach using vegan package v 2.6–4 (Oksanen et al. 2022).
Results
Diet Composition
A total of 155 specimens (112 females and 43 males) were collected during this study, 81 in 2020 and 74 in 2021. A total of 85 specimens were collected during summer, 28 during fall, and 42 during winter. The size of females ranged between 124.0- and 210.0-mm CW and 53.5- and 84.7-mm CL, while males ranged between 67.0 and 210.0 mm CW and 32.0 and 92.0 mm CL. The total weight of females ranged between 107.32 and 353.6 g, while the weight of males ranged between 19.8 and 590.56 g.
Of the 155 stomachs collected, 16.8% (n = 26) were empty. Among the remaining 129 non-empty stomachs, 76.7% (n = 99) contained at least one food item, 6.2% (n = 8) uniquely contained fishing net debris, 9.3% (n = 12) uniquely contained non-identified organic matter, and 7.8% (n = 10) exclusively had non-identified material. Of the 129 non-empty stomachs, the relative FO was 76.7% for food items (n = 99), 78.3% for non-identified organic material (n = 101), and 30.2% for fishing net debris (n = 39). Of the 129 non-empty stomachs, the RP for food items was 48.9 ± 35.3%, 36.7 ± 31.5% for non-identified organic material, and 13.7 ± 27.8% for fishing net debris, which was the most abundant inorganic item.
The RP in the stomachs with at least one food item (99 stomachs) indicated that fish was the most abundant food item (19.1 ± 26.3%), while the caramote prawn had the lowest value (0.1 ± 1.0%). Similarly, the FO in those 99 stomachs exhibited a similar pattern to the RP. On average, FO showed 49.4% for fish (n = 49) and 1.0% for the caramote prawn (n = 1) (Table 1).
ANCOVA analysis for the RP and FO did not indicate size effects and differences in diet between sex or seasons (Table 2). However, ANCOVA and pair-wise comparisons showed that females’ RP and FO values varied between summer and winter (pair-wise p (MC) = 0.006) (Fig. 3; Table 2). For females, sediment had higher RP values in winter (23.1 ± 23.1%) compared to summer (11.2 ± 21.5%), and fish had higher RP values in summer (25.8 ± 30.1%) compared to winter (6.3 ± 16.6%) (Fig. 3).
Box-and-whisker plots for the relative presence (RP) of each food item in the stomachs of Atlantic blue crabs Callinectes sapidus Rathbun, 1896 (all specimens (n = 99), females (n = 74), males (n = 25)) collected in the Guadalquivir estuary in 2021 and 2022 in three different seasons (summer, fall, and winter). Legend: Art = other arthropods, Cep = cephalopods, Cra = crabs, Fis = fish, Mol = mollusks, Pke = Penaeus kerathurus, Sed = sediment, Pla = plant material, Wor = worms
Regarding inorganic items, the abundance of fishing net debris varied between sex and among seasons, with the highest RP found in winter in males (35.8 ± 46.2%) and the lowest in females also during winter (5.2 ± 14.3%).
Feeding Activity and Stomach Correction
The FS of most stomachs was in States 1 (30%) and 2 (20%), meaning that the amount of food in the stomachs of these individuals was reduced to around 25% or below 5%. There were no differences in the FS between seasons (R = 0.03; p = 0.10) and sex (R = −0.002; p = 0.48). The frequency distribution of each stomach FS at each season was similar with a slight tendency to a higher FS in winter and fall (Fig. 4). Nevertheless, the FI did not present differences between seasons (R = 0.04; p = 0.17) or sex (R = −0.08; p = 0.95), as well as the DR (factor season: R = 0.02; p = 0.12 and sex: R = 0.05; p = 0.13) (Table 3).
The Mantel test revealed a correlation between the RP index with the FS correction and the RP index without the FS correction (R = 0.895; p = 0.0001). The FS correction slightly favored the RP for fish in males throughout the year and for other arthropods and sediments in males during winter. However, it also reduced the RP for plant material and sediments in females.
Selectivity Index
The Atlantic blue crab population from the Guadalquivir estuary focused on preying upon fish (14.6%), mollusks (11.2%) and crabs and other decapods (9.9%), and less so on other arthropods (−92.8%) and worms (−97.5%) as disclosed by the Li.
Discussion
The diet of the Atlantic blue crab in the Guadalquivir estuary relied mainly on fish and mollusks, disclosing its potential impact on local ecosystems. Despite its omnivorous behavior, mirroring that used in its native habitat, we found that the female’s diet varied seasonally. Contrary to the hypothesized, the decline in the caramote prawn population is unlikely due to predation by the Atlantic blue crab. In the following sections, we highlight why and how this study offers valuable baseline data to elucidate the ecological roles of the Atlantic blue crab in non-native ecosystems.
The Atlantic Blue Crab Diet
The Atlantic blue crab in the Guadalquivir estuary showed a high consumption and selectivity for fish, mollusks, and crabs, as shown by other portunid crabs (Williams 1981). The low consumption of worms, cephalopods, other arthropods, and plant material is similar to what has been documented in the native area (Hines 2007; Reichmuth et al. 2009). The caramote prawn was among the less relevant prey. The Li showed that fish, the most frequently occurring food items in the ecosystem, was the most consumed prey.
Females consumed more fish (25.8 ± 30.0%) in summer, compared to winter (6.4 ± 16.6%), while in winter, higher amounts of sediment (23.1 ± 23.1%) were found in the stomach contents of females compared to summer (11.2 ± 21.5%). By contrast, the diet of males did not vary throughout the year. The ingestion of sediment is associated with consuming mollusks and crabs because both prey types bury themselves (Dunnington 1956; Rakocinski et al. 2003). Consequently, sediments can be ingested unintentionally rather than being consumed intentionally (Reichmuth et al. 2009). This suggests that females may switch their diet during the winter because they are more sensitive to low water temperature (Bauer and Miller 2010) and overwinter in the mouth of the estuary to increase survival, while males remain in upper estuarine areas (Laughlin 1982; Schaffner and Diaz 1988; Hines et al. 1990; Hines 2007). There is also the possibility that in coastal fishing areas, Atlantic blue crabs find alternative prey all year round in ghost nets (Hines 2007; Mancinelli et al. 2017b; Marchessaux et al. 2023). Although we observed more fishing net debris in males during winter than in males and females collected in any other season, the male sample size during winter was limited (n = 12), so no firm conclusions can be made.
Mollusks are a common prey and, in some situations, they can be more frequent than fish (Hines 2007; Rady et al. 2018). The most representative groups are gastropods and bivalves of multiple families (Van Engel 1958; Seed 1980; Micheli 1995; Wright et al. 1996; Brown et al. 2011). Our data suggests that Atlantic blue crab preys more on mollusks during the fall season, accounting for 23.1 ± 21.2% of the diet, in terms of RP. This pattern also corresponds to the seasonal variation observed in the native area (Tagatz 1968; Dugas et al. 1974; Laughlin 1982; Eggleston 1990), probably coinciding with the recruitment season. When smaller size classes of mollusks are available, Atlantic blue crabs may feed more on this prey because this size ratio decreases claw damage, as reported in both native and invaded areas (Micheli 1995; Prado et al. 2020).
We did not observe significant effects of size on diet, or at least within the examined size range. Preying on fish and mollusks seems common for bigger individuals, both in non-native (Freire 1996; Tadi-Beni et al. 2023) and native areas (Laughlin 1982; Fitz and Wiegert 1991; Hsueh et al. 1992). Other portunids also exhibit the same tendency (Hamida et al. 2019; Tadi-Beni et al. 2023). This suggests that the large sampled crabs (67–210 mm) might have led to an increased relevance of fish and mollusks in their diet, compared to other prey such as crabs, more commonly found in smaller specimens (< 67 mm) (Hines 2007) but which we did not test.
Despite the size interaction with diet composition found in other studies, mollusks are the most abundant and frequent prey for the Atlantic blue crab and other portunids (Chande and Mgaya 2004) in non-native areas (Rady et al. 2018), contrary to our results. Our results revealed a higher presence of fish in the diet of the Atlantic blue crab in the Guadalquivir estuary. This variation in diet is characteristic of omnivore portunids, and it could be induced by differences in prey availability between study areas (Freire 1996) or by inter-specific mechanisms to avoid competition (Wu and Shin 1997). Therefore, although the Atlantic blue crab is a generalist and opportunistic species, it can select predation strategies according to prey availability or energy requirements (Seitz et al. 2011).
Fullness State Correction and Feeding Activity
The reliance of the Atlantic blue crab population from the Guadalquivir estuary on three prey types, i.e., fish, mollusks, and crabs, likely explains the similar results for the RP and FO indexes, even when the FS correction is applied. Similarities between both approaches suggest that utilizing all stomach contents, even if they are at State 3 or lower, could be a better choice to analyze the diet. Collecting a large sample size for this study was challenging and could have limited the robustness of data analyses. Nonetheless, the findings about the most common taxa seem undisputed, considering the recorded high values.
The feeding activity of the Atlantic blue crab was similar throughout the year in the Guadalquivir estuary (Table 3), which does not align with observations made in colder ecosystems in the native area where feeding ceases during winter (Hines 2007). Thus, the Atlantic blue crab could impact the ecological resources of the Guadalquivir estuary and fishing activities throughout the year.
Impacts on the Caramote Prawn
Local fishers expressed concern about the declining abundance of the caramote prawn in the Guadalquivir estuary over the last 3 years, attributing it to the presence of the Atlantic blue crab. While the caramote prawn generally exhibited relatively high dominance values in summer due to migrations from the sea into the estuary, this species was among the less frequent prey of the Atlantic blue crab. Indeed, it only appeared in the stomach of one female sampled in the summer. It is noteworthy that the abundance of the caramote prawn is not consistent throughout the year, as environmental factors dictate variations in the population size (González-Ortegón et al. 2023). Other explanations for the decline of the caramote prawn should be considered, such as overfishing, decrease of their prey, and/or competition for space and food with the Atlantic blue crab or other species. Additionally, changes in the prevailing environmental conditions in the estuary, including salinity and reduced river flow due to drought, impact fitness (González-Ortegón et al. 2015b) and may disrupt the energetic balance and physiological regulation (González-Ortegón et al. 2006; Ahmed and Diana 2015).
Conclusions
The Atlantic blue crab population from the Guadalquivir estuary showed an omnivore and generalist feeding behavior as observed in other native and non-native areas. The most consumed and selected food items were fish, mollusks, and crabs, with fish having a higher importance when compared with other areas. Although the diet composition of females showed seasonal variation adapted to the seasonal availability of prey, feeding activity was similar between seasons and sex. Diet composition index correction by the FS did not have an effect on the results, which suggests that using all stomachs would not bias diet composition as the most common taxa seem undisputed. The caramote prawn Penaeus kerathurus has been declining in the Guadalquivir estuary since 2021, and it is unlikely that the Atlantic blue crab is responsible for that, as they rarely preyed on it. Nevertheless, the Atlantic blue crab established numerous trophic interactions with native biota during its adult life in the Guadalquivir estuary, with a relatively high frequency of fishing nets in the stomachs highlighting putative negative impacts on fishing activities.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ahmed, N., and S.D. James. 2015. Coastal to inland: Expansion of prawn farming for adaptation to climate change in Bangladesh. Aquaculture Reports 2: 67–76.
Ayas, D., and Y. Ozogul. 2011. The effects of sex and seasonality on the metal levels of different muscle tissues of mature Atlantic blue crabs (Callinectes sapidus) in Mersin Bay, north-eastern Mediterranean. International Journal of Food Science and Technology 46 (10): 2030–2034.
Bauer, L.J., and T.J. Miller. 2010. Spatial and interannual variability in winter mortality of the blue crab (Callinectes sapidus) in the Chesapeake Bay. Estuaries and Coasts 33: 678–687.
Belgrad, B.A., and B.D. Griffen. 2016. The influence of diet composition on fitness of the blue crab. Callinectes Sapidus. Plos One. 11 (1): e0145481.
Brown, K.M., B. Aronhime, and X. Wang. 2011. Predatory blue crabs induce byssal thread production in hooked mussels. Invertebrate Biology 130 (1): 43–48.
Buckland, A., R. Baker, N. Loneragan, and M. Sheaves. 2017. Standardising fish stomach content analysis: The importance of food item condition. Fisheries Research 196: 126–140.
Castejón, D., and G. Guerao. 2013. A new record of the American blue crab, Callinectes sapidus Rathbun, 1896 (Decapoda: Brachyura: Portunidae), from the Mediterranean coast of the Iberian Peninsula. BioInvasions Records 2 (2): 141–143.
Carrozzo, L., L. Potenza, P. Carlino, M.L. Costantini, L. Rossi, and G. Mancinelli. 2014. Seasonal abundance and trophic position of the Atlantic blue crab Callinectes sapidus Rathbun 1896 in a Mediterranean coastal habitat. Rendiconti Lincei 25: 201–208.
Campbell, A.C. 1982. The hamlyn guide to the flora and fauna of the Mediterranean Sea, illustrated by Roger Gorring and James Nicholls. London: Hamlyn.
Chaffai, A., W. Rjiba-Bahri, A. Abidi, F. Denis, and J.B. Souissi. 2020. Trophic habits of the invasive crab Libinia dubia H. Milne Edwards. 1834 from the Gulf of Gabès (Tunisia). Mediterranean Marine Science 21 (2): 420–432.
Chairi, H., and E. González-Ortegón. 2022. Additional records of the blue crab Callinectes sapidus Rathbun, 1896 in the Moroccan Sea. Africa. Bioinvasions Records 11 (3): 776–784.
Chande, A.I., and Y.D. Mgaya. 2004. Food habits of the blue swimming crab Portunus pelagicus along the Coast of Dar es Salaam, Tanzaniajpeg. Western Indian Ocean Journal of Marine Science 3 (1): 37–42.
Clark, M.E., T.G. Wolcott, D.L. Wolcott, and A.H. Hines. 1999. Intraspecific interference among foraging blue crabs Callinectes sapidus: Interactive effects of predator density and food item patch distribution. Marine Ecology Progress Series 178: 69–78.
Clarke, K.R., and R.N. Gorley. 2006. PRIMER v.6: User manual/tutorial. PRIMER-E.
Clavero, M., N. Franch, R. Bernardo-Madrid, V. López, P. Abelló, J.M. Queral, and G. Mancinelli. 2022. Severe, rapid and widespread impacts of an Atlantic blue crab invasion. Marine Pollution Bulletin 176: 113479.
Conover, D.O. 1992. Seasonality and the scheduling of life history at different latitudes. Journal of Fish Biology 41 (sB): 161–178.
Darnell, R.M. 1959. Studies of the life history of the blue crab (Callinectes sapidus Rathbun) in Louisiana waters. Transactions of the American Fisheries Society 88 (4): 294–304.
Dittel, A.I., C.E. Epifanio, and M.L. Fogel. 2006. Trophic relationships of juvenile blue crabs (Callinectes sapidus) in estuarine habitats. Hydrobiologia 568: 379–390.
Drake, P., A.M. Arias, F. Baldó, J.A. Cuesta, A. Rodríguez, A. Silva-Garcia, and C. Fernández-Delgado. 2002. Spatial and temporal variation of the nekton and hyperbenthos from a temperate European estuary with regulated freshwater inflow. Estuaries 25: 451–468.
Dugas, R.J., J.W. Tarver, and L.S. Nutwell. 1974. The mollusk communities of Lake Pontchartrain and Maurepas, Louisiana. Louisiana Wildlife Fisheries Commission 10: 13.
Dunnington, E.A. 1956. Blue crabs observed to dig soft shell clams for food. Maryland Tidewater News 12 (12): 1–4.
Eggleston, D.B. 1990. Foraging behavior of the blue crab, Callinectes sapidus, on juvenile oysters, Crassostrea virginica: Effects of food item density and size. Bulletin of Marine Science 46 (1): 62–82.
Encarnação, J., V. Baptista, M.A. Teodósio, and P. Morais. 2021. Low-cost citizen science campaign effectively monitors the rapid expansion of a marine invasive species. Frontiers in Environmental Science 9: 752705.
Fernández-Delgado, C., F. Baldó, C. Vilas, D. García-González, J.A. Cuesta, E. González-Ortegón, and P. Drake. 2007. Effects of the river discharge management on the nursery function of the Guadalquivir river estuary (SW Spain). Hydrobiologia 587: 125–136.
Fitz, H.C., and R.G. Wiegert. 1991. Utilization of the intertidal zone of a salt marsh by the blue crab Callinectes sapidus: density, return frequency, and feeding habits. Marine Ecology Progress Series 76: 249–260.
Freire, J. 1996. Feeding ecology of Liocarcinus depurator (Decapoda: Portunidae) in the Ria de Arousa (Galicia, north-west Spain): Effects of habitat, season and life history. Marine Biology 126: 297–311.
Fuentes, M.A., L. Torrent, S. Barrera, and D. Boix. 2019. Rapid invasion of the American blue crab Callinectes sapidus Rathbun, 1896 in the North-East of the Iberian Peninsula. BioInvasions Record 8 (1): 113–118.
Gil, A. 2018. Análisis de la dieta de Callinectes sapidus (Rathbun, 1896) en ambientes recientemente invadidos del Golfo de Valencia. Valencia: Universitat Politècnica de València.
González-Ortegón, E., E. Pascual, J.A. Cuesta, and P. Drake. 2006. Field distribution and osmoregulatory capacity of shrimps in a temperate European estuary (SW Spain). Estuarine, Coastal and Shelf Science 67 (1–2): 293–302.
González-Ortegón, E., F. Baldó, A. Arias, J.A. Cuesta, C. Fernández-Delgado, C. Vilas, and P. Drake. 2015a. Freshwater scarcity effects on the aquatic macrofauna of a European Mediterranean-climate estuary. Science of the Total Environment 503: 213–221.
González-Ortegón, E., M.E.M. Walton, B. Moghaddam, C. Vilas, A. Prieto, H.A. Kennedy, J.P. Cañavate, and L. Le Vay. 2015b. Flow regime in a restored wetland determines trophic links and species composition in the aquatic macroinvertebrate community. Science of the Total Environment 503: 241–250.
González-Ortegón, E., S. Jenkins, B.S. Galil, P. Drake, and J.A. Cuesta. 2020. Accelerated invasion of decapod crustaceans in the southernmost point of the Atlantic coast of Europe: A non-natives’ hot spot? Biological Invasions 22: 3487–3492.
González-Ortegón, E., S. Berger, J. Encarnação, H. Chairi, P. Morais, M.A. Teodosio, and J.A. Cuesta. 2022. Free pass through the pillars of Hercules? Genetic and historical insights into the recent expansion of the Atlantic blue crab Callinectes sapidus to the west and the east of the Strait of Gibraltar. Frontiers in Marine Science 9: 918026.
González-Ortegón, E., G.F. de Carvalho-Souza, C. Vilas, F. Baldó, and J.A. Cuesta. 2023. Trends in the decapod crustacean community at the southernmost estuary of the Atlantic coast of Europe. Scientific Reports 13 (1): 22857.
Guijarro-García, E., M. Vivas, E. García, E. Barcala, M. Trives, and A. Muñoz. 2019. Atlantic blue crab (Callinectes sapidus Rathbun, 1896) in a protected coastal lagoon in SESpain. Gualtar (Braga): XX Iberian Symposium on Marine Biology Studies(SIEBM XX) Sep 9-12.
Haram, O.J., and J.W. Jones. 1971. Some observations on the food of the gwyniad Coregonus clupeoides pennantii Valenciennes of Llyn Tegid (Lake Bala), North Wales. Journal of Fish Biology 3 (3): 287–295.
Harris, R.E. 1982. Life history, ecology and stock assessment of the blue crab Callinectes sapidus of the United States Atlantic Coast- a review. In Proceedings of the Blue Crab Colloquium, ed. H.M. Perry and W.A. Van Engels, 13–14. Ocean Springs, Miss: Gulf States Marine Fisheries Commission.
Hamida, O.B.A.B.H., N.B.H. Hamida, R. Ammar, H. Chaouch, and H. Missaoui. 2019. Feeding habits of the swimming blue crab Portunus segnis (Forskål, 1775) (Brachyura: Portunidae) in the Mediterranean. Journal of the Marine Biological Association of the United Kingdom 99 (6): 1343–1351.
Haefner, P.A., Jr. 1990. Morphometry and size at maturity of Callinectes ornatus (Brachyura, Portunidae) in Bermuda. Bulletin of Marine Science 46 (2): 274–286.
Hines, A.H. 2007. Ecology of juvenile and adult blue crabs. Biology of the blue crab. In The blue crab: Callinectes sapidus, ed. V.S. Kennedy and L.E. Cronin, 565–654. College Park, Maryland: Maryland Sea Grant College.
Hines, A.H., A.M. Haddon, and L.A. Wiechert. 1990. Guild structure and foraging impact of blue crabs and epibenthic fish in a subestuary of Chesapeake Bay. Marine Ecology Progress Series 67: 105–126.
Hsueh, P.W., J.B. McClintock, and T.S. Hopkins. 1992. Comparative study of the diets of the blue crabs Callinectes similis and C. sapidus from a mud-bottom habitat in Mobile Bay, Alabama. Journal of Crustacean Biology 12 (4): 615–619.
Hyslop, E.J. 1980. Stomach contents analysis—a review of methods and their application. Journal of Fish Biology 17 (4): 411–429.
Kampouris, T.E., J.S. Porter, and W.G. Sanderson. 2019. Callinectes sapidus Rathbun, 1896 (Brachyura: Portunidae): An assessment on its diet and foraging behaviour, Thermaikos Gulf, NW Aegean Sea, Greece: Evidence for ecological and economic impacts. Crustacean Research 48: 23–37.
Kevrekidis, K., C. Antoniadou, K. Avramoglou, J. Efstathiadis, and C. Chintiroglou. 2013. Population structure of the blue crab Callinectes sapidus in Thermaikos Gulf (Methoni Bay), 113–116. Thessaloniki, Greece: Proceedings of the 15th Pan-Hellenic Congress of Ichthyologists 10–13 October.
Laughlin, R.A. 1982. Feeding habits of the blue crab, Callinectes sapidus Rathbun, in the Apalachicola estuary. Florida. Bulletin of Marine Science 32 (4): 807–822.
Mancinelli, G., P. Chainho, L. Cilenti, S. Falco, K. Kapiris, G. Katselis, and F. Ribeiro. 2017a. On the Atlantic blue crab (Callinectes sapidus Rathbun 1896) in southern European coastal waters: Time to turn a threat into a resource? Fisheries Research 194: 1–8.
Mancinelli, G., P. Chainho, L. Cilenti, S. Falco, K. Kapiris, G. Katselis, and F. Ribeiro. 2017b. The Atlantic blue crab Callinectes sapidus in southern European coastal waters: Distribution, impact and prospective invasion management strategies. Marine Pollution Bulletin 119 (1): 5–11.
Mancinelli, G., M.T. Guerra, K. Alujević, D. Raho, M. Zotti, and S. Vizzini. 2017c. Trophic flexibility of the Atlantic blue crab Callinectes sapidus in invaded coastal systems of the Apulia region (SE Italy): A stable isotope analysis. Estuarine, Coastal and Shelf Science 198: 421–431.
Mancinelli, G., R. Bardelli, and A. Zenetos. 2021. A global occurrence database of the Atlantic blue crab Callinectes sapidus. Scientific Data 8 (1): 111.
Marchessaux, G., M.C. Mangano, S. Bizzarri, C. M’Rabet, E. Principato, N. Lago, and G. Sarà. 2023. Invasive blue crabs and small-scale fisheries in the Mediterranean Sea: local ecological knowledge, impacts and future management. Marine Policy 148: 105461.
Mascaro, M., L.E. Hidalgo, X. Chiappa-Carrara, and N. Simoes. 2003. Size-selective foraging behaviour of blue crabs, Callinectes sapidus (Rathbun), when feeding on mobile food item: Active and passive components of predation. Marine and Freshwater Behaviour and Physiology 36 (3): 143–159.
Meise, C.J., and L.L. Stehlik. 2003. Habitat use, temporal abundance variability, and diet of blue crabs from a New Jersey estuarine system. Estuaries 26: 731–745.
Micheli, F. 1995. Behavioural plasticity in food item-size selectivity of the blue crab Callinectes sapidus feeding on bivalve food item. Journal of Animal Ecology 64 (1): 63–74.
Millikin, M.R., and A.B. Williams. 1984. Synopsis of biological data on the blue crab, Callinectes sapidus Rathbun (No. 138). National Oceanic and Atmospheric Administration (NOAA). NOAA Technical Report NMFS 1, FAO Fisheries Synopsis 138: 39.
Morais, P., M. Gaspar, E. Garel, V. Baptista, J. Cruz, I. Cerveira, and M. Teodosio. 2019. The atlantic blue crab Callinectes sapidus Rathbun, 1896 expands its non-native distribution into the ria formosa lagoon and the guadiana estuary (SW-Iberian Peninsula, Europe). BioInvasions Records 8 (1): 123–133.
Nehring, S. 2011. Invasion history and success of the American Blue Crab Callinectes sapidus in European and adjacent waters. In In the wrong place - alien marine crustaceans: distribution, biology and impacts, ed. B.S. Galil, P.F. Clark, and J.T. Carlton, 607–624. Netherlands: Springer.
Norse, E.A. 1977. Aspects of the zoogeographic distribution of Callinectes (Brachyura: Portunidae). Bulletin of Marine Science 27 (3): 440–447.
Oksanen, J., G.L. Simpson, F.G. Blanchet, R. Kindt, P. Legendre, P.R. Minchin, R.B. O’Hara, P. Solymos, M.H.H. Stevens, E. Szoecs, H. Wagner, M. Barbour, M. Bedward, B. Bolker, D. Borcard, G. Carvalho, M. Chirico, M.D. Caceres, S. Durand, and J. Weedon. 2022. Vegan: community ecology package (2.6–4).
Öndes, F., and G. Gökce. 2021. Distribution and fishery of the invasive blue crab (Callinectes sapidus) in Turkey based on local ecological knowledge of fishers. Journal of Anatolian Environmental and Animal Sciences 6 (3): 325–332.
Onofri, V., J. Dulčić, A. Conides, S. Matić-Skoko, and B. Glamuzina. 2008. The occurrence of the blue crab, Callinectes sapidus Rathbun, 1896 (Decapoda, Brachyura, Portunidae) in the eastern Adriatic (Croatian coast). Crustaceana 81 (4): 403–409.
Pla, M., S. Quiñonero, J. Hernández, J. Velázquez, P. Risueño, and J. López. 2018. Predation of the blue crab Callinectes sapidus Rathbun, 1896 on freshwater bivalves (Unionidae & Corbiculidae) in eastern Iberian Peninsula. Folia Conchyliologica 47: 3–9.
Prado, P., A. Peñas, C. Ibáñez, P. Cabanes, L. Jornet, N. Álvarez, and N. Caiola. 2020. Food item size and species preferences in the invasive blue crab, Callinectes sapidus: Potential effects in marine and freshwater ecosystems. Estuarine, Coastal and Shelf Science 245: 106997.
Prado, P., C. Ibáñez, L. Chen, and N. Caiola. 2021. Feeding habits and short-term mobility patterns of blue crab, Callinectes sapidus, across invaded habitats of the Ebro Delta subjected to contrasting salinity. Estuaries and Coasts 45 (3): 839–855.
R Core Team. 2020. R: A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.R-project.org/.
Rady, A., W.S. Sallam, N.E.I. Abdou, and A.A.M. El-Sayed. 2018. Food and feeding habits of the blue crab, Callinectes sapidus (Crustacea: Decapoda: Portunidae) with special reference to the gastric mill structure. Egyptian Journal of Aquatic Biology and Fisheries 22 (5): 417–431.
Rakocinski, C.F., H.M. Perry, M.A. Abney, and K.M. Larsen. 2003. Soft-sediment recruitment dynamics of early blue crab stages in Mississippi Sound. Bulletin of Marine Science 72 (2): 393–408.
Reichmuth, J.M., R. Roudez, T. Glover, and J.S. Weis. 2009. Differences in food item capture behavior in populations of blue crab (Callinectes sapidus Rathbun) from contaminated and clean estuaries in New Jersey. Estuaries and Coasts 32: 298–308.
Rosas, C., E. Lazaro-Chavez, and F. Bückle-Ramirez. 1994. Feeding habits and food niche segregation of Callinectes sapidus, C. rathbunae, and C. similis in a subtropical coastal lagoon of the gulf of mexico. Journal of Crustacean Biology 14 (2): 371–382.
Schaffner, L.C., and R.J. Diaz. 1988. Distribution and abundance of overwintering blue crabs, Callinectes sapidus, in the lower Chesapeake Bay. Estuaries 11: 68–72.
Scherer, A.E., M.M. Garcia, and D.L. Smee. 2017. Predatory blue crabs induce stronger nonconsumptive effects in eastern oysters Crassostrea virginica than scavenging blue crabs. PeerJ 5: e3042.
Seed, R. 1980. Predator-food item relationships between the mud crab Panopeus herbstii, the blue crab, Callinectes sapidus and the Atlantic ribbed mussel Geukensia (= Modiolus) demissa. Estuarine and Coastal Marine Science 11 (4): 445–458.
Seitz, R.D., K.E. Knick, and M. Westphal. 2011. Diet selectivity of juvenile blue crabs (Callinectes sapidus) in Chesapeake Bay. Integrative and Comparative Biology 51 (4): 598–607.
Strauss, R.E. 1979. Reliability estimates for Ivlev’s electivity index, the forage ratio, and a proposed linear index of food selection. Transactions of the American Fisheries Society 108 (4): 344–352.
Tadi-Beni, F., J. Pazooki, M. Safaie, and F. Nazari. 2023. Diets of Portunus segnis (Forskål, 1775) (Decapoda, Portunidae) from the Persian Gulf. Iran. Crustaceana. 96 (2): 103–112.
Tagatz, M.E. 1968. Biology of the blue crab, Callinectes sapidus Rathbun, in the St. John’s River. Florida. Fishery Bulletin 67: 17–32.
Triay-Portella, R., J.A. Martín, L. Luque, and J.G. Pajuelo. 2022. Relevance of feeding ecology in the management of invasive species: Food item variability in a novel invasive crab. Estuarine, Coastal and Shelf Science 274: 107949.
Van Engel, W.A. 1958. The blue crab and its fishery in Chesapeake Bay. Part 1. Reproduction, early development, growth and migration. Commercial Fisheries Review 20 (6): 6–17.
Wainright, C.A., C.C. Muhlfeld, J.J. Elser, S.L. Bourret, and S.P. Devlin. 2021. Species invasion progressively disrupts the trophic structure of native food webs. Proceedings of the National Academy of Sciences 118 (45): e2102179118.
Wear, R.G., and M. Haddon. 1987. Natural diet of the crab Ovalipes catharus (Crustacea, Portunidae) around central and northern New Zealand. Marine EcologyProgress Series 35: 39–49.
Williams, M.J. 1981. Methods for analysis of natural diet in portunid crabs (Crustacea: Decapoda: Portunidae). Journal of Experimental Marine Biology and Ecology 52 (1): 103–113.
Wright, R.A., L.B. Crowder, and T.H. Martin. 1996. Selective predation by blue crabs on the gastropod, Bittium varium: Confirmation from opercula found in the sediments. Estuaries 19: 75–81.
Wu, R.S.S., and P.K.S. Shin. 1997. Food segregation in three species of portunid crabs. Hydrobiologia 362: 107–113.
Acknowledgements
We are grateful to all the fishermen and colleagues, especially to Gustavo F. de carvalho-Souza, Antonio Moreno, Gala González, Miguel Sánchez-G Hernández, Victor Acosta, and Enrique Garcia-Raso, for helping during sampling, laboratory work, and corrections. We want to express our gratitude to Dr. Pedro Morais for his outstanding work as an associate editor and to the two anonymous reviewers who provided valuable suggestions for refining this final version.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This work was funded by the Spanish Ministerio de Ciencia e Innovación through InvBlue project number PID2019-105978RA-I00, while the first author was supported by a Ph.D. grant (PRE2020-096343) from the Spanish Ministerio de Ciencia, Innovación y Universidades.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Pedro Morais
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ortega-Jiménez, E., Cuesta, J.A., Laiz, I. et al. Diet of the Invasive Atlantic Blue Crab Callinectes sapidus Rathbun, 1896 (Decapoda, Portunidae) in the Guadalquivir Estuary (Spain). Estuaries and Coasts 47, 1075–1085 (2024). https://doi.org/10.1007/s12237-024-01344-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-024-01344-9