Abstract
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) are a class of drugs with potent glucose-lowering activity. Additionally, some GLP-1 RAs have demonstrated cardiovascular and renal benefits. Current guidelines recommend their use in patients with type 2 diabetes (T2D) at high risk of or with established cardiovascular disease (CVD), regardless of glycaemic control, with lifestyle modification and metformin. However, several studies have recently highlighted the limited number of patients with T2D benefiting from these medications worldwide. Given the huge burden of CVD among patients with T2D, efforts should be made to bring clinical practice closer to expert guidelines. This review describes the current situation of GLP-1 RA use in Spain and the reasons behind the gap between guidelines and real-world practice and suggests possible solutions. Administrative issues, lack of awareness of the cardiovascular benefits, clinical inertia, rejection of injectable medication and costs could be some of the reasons for the current situation. Possible strategies that could help to close the gap include encouraging a multidisciplinary approach to the treatment of diabetes which involves cardiologists, endocrinologists, nephrologists, primary care providers and pharmacists; improved awareness of comorbidities and earlier evaluation and treatment or risks; and better education of healthcare providers regarding the cardioprotective benefits of these drugs.
Plain Language Summary
The glucagon-like peptide 1 receptor agonists (GLP-1 RAs) are a class of drugs that can be beneficial for patients with type 2 diabetes who are at high risk of cardiovascular complications, such as heart attacks. For this reason, the current clinical guidelines strongly recommend their use in these patients. Unfortunately, many patients with type 2 diabetes and high cardiovascular risk still do not benefit from these drugs. This review analyses the reasons for this situation in Spain, and proposes some possible solutions. The reasons for the low use of GLP-1 RAs could be related to doctors not updating a patient’s diabetes medicine as often as they should, lack of awareness about the cardiovascular benefits of these drugs, fear of medicines that involve needles, administrative issues, and costs. Some of the possible strategies to improve the use of GLP-1 RAs among patients with type 2 diabetes with high cardiovascular risk could be to foster greater cooperation among specialists, increase awareness of the need to treat cardiovascular risk in patients with diabetes, and better education of doctors regarding the benefits of these drugs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Some glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have demonstrated benefit for patients with type 2 diabetes (T2D) at risk of cardiovascular complications or with established cardiovascular disease. |
Despite clinical guidelines recommendations to use GLP-1 RAs in these patients, the use of these medications is still suboptimal in Spain and worldwide. |
The reasons for the limited use of GLP-1 RAs could be complex, and include clinical inertia, lack of awareness of the cardiovascular benefits by prescribing physicians, rejection of injectable medication by some patients, administrative issues, and higher costs compared with other drugs. |
It is a matter of urgency to implement the recommendations from current clinical guidelines that encourage the use of GLP-1 RAs in patients with T2D and high cardiovascular risk. |
Possible solutions include promoting physician education, a multidisciplinary approach to patient care, and increased awareness of the importance of treating comorbidities. |
Introduction
Type 2 diabetes (T2D) is a chronic metabolic disease with a huge clinical and economic impact globally. In Spain, the total direct costs of treating T2D have been estimated at €10 billion yearly [1]. Therefore, the development of novel treatments and the optimisation of available resources are key to diabetes management. Currently, in addition to glucose-lowering efficacy, factors such as weight control and impact on comorbidities are usually considered in the evaluation of any new glucose-lowering therapies [2]. Also, the rise in costs and the effort to individualise care for T2D has promoted a multifactorial approach that aims to control risk factors for diabetes-related complications, and especially cardiovascular disease (CVD). Approximately 32% of patients with T2D develop CVD, and it is their leading cause of mortality [3]. A recent study of patients with T2D in the region of Catalonia, Spain, revealed that 93% of patients were at high or very high cardiovascular (CV) risk [4]. For these reasons, and given the burden of CVD and the elevated costs of care, primary and secondary prevention of CVD among patients with T2D is a priority [5].
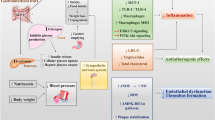
The glucagon-like peptide 1 receptor agonists (GLP-1 RAs) are a class of drugs with potent glucose-lowering effects [6]. Clinical studies have demonstrated that GLP-1 RAs can lower glycated haemoglobin (HbA1c) by more than 1.0% from baseline values with a reduced risk of hypoglycaemia compared with sulfonylurea and insulin [7]. In addition, some GLP-1 RAs have beneficial effects in other conditions frequently associated with diabetes, such as overweight and obesity, hypertension, hyperlipidaemia, and most importantly atherosclerotic CVD [6, 8, 9]. The accumulated evidence shows that some GLP-1 RAs can reduce the occurrence of major adverse CV events, CV mortality, and all-cause mortality risk, without significant known safety concerns [8, 10, 11]. The basis for the CVD effects of GLP-1 RAs is not fully understood, but there is strong evidence supporting clinically relevant anti-inflammatory and antioxidant effects of these drugs [12]. Although GLP-1 RAs are generally recommended for secondary prevention in patients with T2D and established CVD or at high CV risk, for dulaglutide an added benefit in early primary prevention has been demonstrated [13, 14].
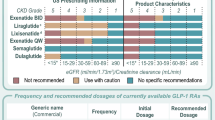
Current guidelines suggest use of GLP-1 RAs with proven CV benefit (liraglutide, subcutaneously administered semaglutide and dulaglutide) in patients at high risk or with established atherosclerotic CVD, regardless of glycaemic control, with lifestyle modification and independent of the use of metformin [7, 13, 15, 16]. Apart from the CVD benefit, other guidelines also focus on weight reduction and restrict their use exclusively to patients with high body mass index (BMI) [17]. Yet other guidelines offer a combined approach and advise use of GLP-1 RAs in patients with CVD and/or high BMI [2, 18,19,20]. Guidelines in Spain underscore that the adequate treatment of T2D requires a simultaneous approach to HbA1c, overweight/obesity, and other CV risk factors such as hypertension, dyslipidaemia or smoking, and highlight that the presence of CVD is a determinant factor for treatment selection [2, 21, 22].
Given the complexity of possible treatment pathways, it was of interest to analyse the available evidence from real-world clinical practice in Spain with respect to GLP-1 RA use. Therefore, we reviewed all observational studies with data on this topic published from 2016 to 2021. A second objective was to assess the extent to which the use of GLP-1 RAs is in line with guideline recommendations, to obtain information on possible reasons for mismatches, and to evaluate possible strategies or recommendations to bridge the gap between guidelines and clinical practice regarding use of GLP-1 RA. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
How Are GLP-1 RAs Being Used in Spain?
GLP-1 RAs are infrequently prescribed among patients with diabetes worldwide. In the USA, it has been estimated that approximately only 1–5% of people with diabetes are prescribed GLP-1 RAs, despite the potential benefits of these drugs in patients with diabetes and CVD [23,24,25,26,27]. A database study in Spain showed that only 3.2% of patients with T2D started treatment with a GLP-1 RA (dulaglutide, exenatide, liraglutide) [28]. A study from Catalonia, in the northeast of Spain, showed that, despite a prevalence of CVD of 23.2% in the population with T2D, only 1.4% were prescribed GLP-1 RAs [27].
Several studies carried out in Spain provide a description of the real-world demographic and clinical characteristics of the population that initiate GLP-1 RA treatments (Table 1). The cross-sectional CHADIG study was the first to assess the characteristics of patients initiating GLP-1 RAs, concluding that these drugs were usually combined with oral glucose-lowering drugs approximately a decade after T2D diagnosis, and that patients were relatively young, with poor glycaemic control, and a high BMI [29]. More recent studies agree that patients starting GLP-1 RAs usually have a mean age of approximately 60 years, a BMI > 35 kg/m2 and HbA1c > 8.1% [28, 30, 31]. Additionally, these studies showed that the time since diagnosis of diabetes to initiation of GLP-1 RAs was greater than 9 years, reflecting long-standing and probably poorly controlled diabetes [28, 32]. This is also reflected in the fact that GLP-1 RAs are often used in combination with other glucose-lowering medication such as metformin, insulin, sodium-glucose cotransporter 2 (SGLT2) inhibitors, and sulfonylureas [28], suggesting that they are used in later phases of diabetes and not mainly after metformin, as suggested by guidelines [2, 18,19,20].
Real-world studies show that most patients initiating GLP-1 RAs present with one or more comorbidities. Interestingly, only 21.8% of the patients presented with established CVD (macrovascular) when initiating GLP-1 RAs [28], suggesting that the presence of CVD is not a main criterion for their use. In a study of patients in the region of Catalonia, 33.6% presented with CVD [30]. This is within the range of the prevalence of CVD in the general T2D population, which ranges from 6.9% to 40.8% [5]. In addition, most patients have high or very high CV risk [4].
A recent survey on the use of glucose-lowering medication in primary care in Spain (CONTROVERTI2) showed that the use of GLP-1 RAs is associated with patients with T2D and obesity, while patients with established CVD are preferentially treated with SGLT2 inhibitors [33]. However, a national level study of elderly patients with T2D admitted to hospital and at very high CV risk concluded that most were not being treated with these drugs [34].
In summary, the data from recent years in Spain show that GLP-1 RAs are mainly used in patients with high BMI (> 35 kg/m2), HbA1c levels above 8%, with comorbidities and a long history of diabetes, but not especially in patients with established atherosclerotic CVD or high CV risk. This suggests that in Spain, there is a gap between the guidelines that propose the use of GLP-1 RAs in patients with established atherosclerotic CVD/high CV risk and real-world clinical practice, which shows that they are used in patients with T2D and obesity with long-standing and poorly controlled diabetes.
These results agree with another retrospective study in Italy showing that patients initiating GLP-1 RAs presented with an HbA1c of 8.3% and a BMI of 34 kg/m2 at baseline. In addition, the data showed there were significant increases in the last 10 years in age at baseline, diabetes duration and CVD for these patients [35].
Potential Reasons That Limit the Use of GLP-1 RAs Based on Their CV Benefit
Characteristics of the Spanish National Health System
Despite the evidence of CV benefit with the use of GLP-1 RAs included in Spanish guidelines, this benefit was not considered in reimbursement decisions, which took place before the publication of the landmark studies showing CV benefit of GLP-1 RAs. Currently in Spain, a high BMI (≥ 30 kg/m2) is an essential factor for reimbursement. The reason for recommending the use of GLP-1 RAs to patients with high BMI was based on models developed by the National Institute for Health and Care Excellence in the UK, for some specific GLP-1 RAs (exenatide and liraglutide) that were then generalised for the drug class [36]. It has been argued that restricting GLP-1 RA therapy to patients with BMI > 35 kg/m2 based exclusively on weight loss may be counterproductive if glycemic efficacy is not maintained [36], and it could set unrealistic goals for both prescribing physicians and patients [32]. Although weight loss should be an objective in patients with obesity and diabetes, it should not be a determinant for the prescription of GLP-1 RAs. The benefits of glucose control and reduction of CV risk with GLP-1 RAs have also been proven in non-obese patients with T2D [16, 21, 37]. However, although the limitation by the Spanish National Health Service could affect the use of GLP-1 RAs, it may not be the major reason for the majority of patients, as other countries without the BMI limitation have similar prescription rates as Spain [26, 38, 39].
Clinical Inertia
It is possible that lack of physician familiarity with GLP-1 RAs, or clinical inertia, are partly responsible for the reduced use of GLP-1 RAs. Therapeutic inertia has been shown to be a significant barrier to glycaemic control, ultimately contributing to long-term complications in high-risk patients [40, 41]. Currently, the focus of diabetes treatment has shifted from strict glycaemic control to a more comprehensive approach that considers prevention or control of CVD, kidney disease or other diabetes complications [13, 42]. Accordingly, a recent survey suggested that one of the most influential clinical factors in the choice of glucose-lowering treatment in Spain is the presence of CVD (the other being high HbA1c) [33]. The current guidelines recommend SGLT2 inhibitors or GLP-1 RAs for the management of patients with T2D and established atherosclerotic CVD, while SGLT2 inhibitors are preferred for patients at risk of heart failure [2]. However, a Delphi study found that, although SGLT2 inhibitors are the treatment of choice in patients with T2D and heart failure or uncontrolled hypertension, no consensus was reached regarding the preferential use of SGLT2 inhibitors or GLP-1 RAs in patients with established CVD. This study also revealed that management of patients with T2D in primary or secondary CV prevention is highly heterogeneous in real-world clinical practice in Spain [43]. Another recent Delphi study also showed that the presence of CVD or high CV risk was a more influential factor for the choice of glucose-lowering treatment than baseline HbA1c levels [44]. Although the Delphi surveys support a treatment choice based also on CV criteria, it seems that this opinion is not being reflected in the use of GLP-1 RAs in real practice.
Injectable Medication
To date, the GLP-1 RAs that have shown CV benefit have subcutaneous administration and it is possible that patients reject the concept of injectable therapy when they first encounter it. GLP-1 RAs are recommended before insulin initiation as the first injectable therapy [13, 16]. However, insulin is still often the first injectable therapy in Spanish patients with T2D, suggesting that the route of administration would not be a strong barrier [28]. Also, weekly administration is available for GLP-1 RAs, which may be preferable to daily insulin and improve adherence to treatment [45, 46]. Studies have shown that the type of injection device and needle size are important attributes driving patient preferences for specific GLP-1 RAs. Understanding these preferences could help improve patient adherence and clinical outcomes [47].
Costs of GLP-1 RA Treatment
While older oral glucose-lowering medications (i.e. metformin, biguanides, sulfonylureas, glinides, thiazolidinediones or α-glucosidase inhibitors) are relatively inexpensive, the newest agents approved for diabetes (i.e. GLP-1 RAs, dipeptidyl peptidase 4 inhibitors and SGLT2 inhibitors) are comparatively more costly. However, the largest part of the cost of diabetes management is consultations, hospitalisations and the treatment of diabetes-related complications, especially if CVD develops [1, 5]. Cost–utility analyses of dulaglutide and other GLP-1 RAs have shown them to be a cost-effective treatment option as add-on therapy to metformin or directly compared to insulin in several countries [48,49,50]. Several comparative pharmacoeconomic analyses have been recently carried out in Spain regarding GLP-1 RAs [51,52,53,54,55,56].
Important differences and variabilities in the uptake of new glucose-lowering medicines in European countries have been observed. The high cost gap between old and new antidiabetic medicines and their financial burdens could affect market uptake and consequently patient access [57]. However, there are no studies showing that costs of GLP-1 RA treatment could strongly influence prescription of this group of drugs [57].
Other Reasons
As is the case with other chronic diseases, most T2D treatments often have low adherence and persistence. In this regard, observational studies in Spain also have shown that, even after a first prescription of GLP-1 RAs, adherence to treatment is low and discontinuation is high [28, 32], with a mean GLP-1 RA treatment duration of 1 year [28]. It was speculated that early discontinuation rates could be due to gastrointestinal side effects that commonly appear at the beginning of GLP-1 RA treatment [28]. However, a study showed that gastrointestinal adverse events were self‐reported by a low proportion of patients (14.96%), most of them by the first follow‐up visit [32].
Some guidelines recommend discontinuation if no beneficial response is observed within 6 months of therapy initiation. Efficacy has been defined as a combined reduction of at least 1.0% in HbA1c levels and 3% in initial weight [17]. Similar advice is found in regional health services in Spain [30, 32]. In Spain, the proportion of patients who discontinued treatment in an observational study largely matched with the proportion of patients not meeting the efficacy requirements as per the proposed definition (37.07%) [32]. It has been argued that requiring more than a 1% HbA1c reduction with GLP-1 RA treatment lacks justification because the HbA1c reduction is closely associated with baseline HbA1c [36]. It has been proposed that treatment withdrawal should be judged from a patient-centred approach, as about 80% of the patients who potentially could benefit from these treatments could not reach those targets [31, 36]. Also, these criteria for discontinuation were proposed prior to studies demonstrating CV benefit for many patients and should be revised in light of the new evidence.
Towards a New Focus of the Therapeutic Role of GLP-1 RAs in Spain
As described, the limitations in the implementation of therapies involving GLP-1 RAs could have patient, clinician and system-level origins. The existence of major gaps in the implementation of effective, evidence-driven therapies in patients with T2D and CVD has been highlighted in other countries [25, 26]. Adoption of these therapies could save lives by decreasing major atherosclerotic CV events and disease progression [26]. Recent reports have highlighted the urgency of implementing guidelines to ensure that patients with T2D and high cardiorenal risk benefit from GLP1 RAs and SGLT2 inhibitors [25, 58].
A multidisciplinary approach to the treatment of diabetes could improve the treatment of patients with cardiorenal risk [58]. Both the American Diabetes Association/European Association for the Study of Diabetes consensus report and the European Society of Cardiology guidelines urge cardiologists, endocrinologists, nephrologists, primary care providers and pharmacists to implement the use of GLP-1 RAs and SGLT2 inhibitors in clinical practice [13, 59]. For patients at risk, treatment at early stages of the disease is beneficial and should be encouraged [60]. As diabetes therapies are constantly improving, continued medical education of healthcare providers on their relative benefits should be a priority to overcome clinical inertia. Management of diabetes should change from a ‘glucocentric’ view to one in which the individual risks of the patient are taken into consideration. Likewise, a more detailed evaluation of the clinical histories of patients and their evolution could help improve personalised treatments and adapt them to emerging comorbidities.
The treatment of T2D requires long-term commitments and substantial changes in lifestyle, for which patient involvement is essential. Patient-centred approaches, which emphasise patient choice and shared decision-making in diabetes management, have been a focus of disease management [7]. In this regard, patients will need to be educated on the benefits of adherence to medication, and in the need to control comorbidities. Bridging these gaps will require the concerted action of patient associations, healthcare providers, regulators, professional societies and payers.
References
Mata-Cases M, Casajuana M, Franch-Nadal J, et al. Direct medical costs attributable to type 2 diabetes mellitus: a population-based study in Catalonia, Spain. Eur J Health Econ. 2016;17:1001–10.
Reyes-García R, Moreno-Pérez O, Tejera-Pérez O, et al. Sociedad Española de Endocrinología y Nutrición. Documento de abordaje integral de la DM2. Grupo de trabajo de diabetes de la SEEN. 2019. https://www.seen.es/docs/apartados/791/Abordaje%20Integral%20DM2_SEEN_2019_OCT_ISBN%20.pdf. Accessed 24 Oct 2021.
Einarson TR, Acs A, Ludwig C, Panton UH. Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc Diabetol. 2018;17:83.
Cebrián-Cuenca AM, Mata-Cases M, Franch-Nadal J, Mauricio D, Orozco-Beltrán D, Consuegra-Sánchez L. Half of patients with type 2 diabetes mellitus are at very high cardiovascular risk according to the ESC/EASD: data from a large Mediterranean population. Eur J Prev Cardiol. 2022;28:e32–4.
Artime E, Romera I, Díaz-Cerezo S, Delgado E. Epidemiology and economic burden of cardiovascular disease in patients with type 2 diabetes mellitus in Spain: a systematic review. Diabetes Ther. 2021;12:1631–59.
Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes—state-of-the-art. Mol Metab. 2021;46: 101102.
Davies MJ, Aroda VR, Collins BS, et al. Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia. 2022;65(12):1925–66.
Bethel MA, Patel RA, Merrill P, et al. Cardiovascular outcomes with glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes: a meta-analysis. Lancet Diabetes Endocrinol. 2018;6:105–13.
Romera I, Cebrián-Cuenca A, Álvarez-Guisasola F, Gomez-Peralta F, Reviriego J. A review of practical issues on the use of glucagon-like peptide-1 receptor agonists for the management of type 2 diabetes. Diabetes Ther. 2019;10:5–19.
Kristensen SL, Rørth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776–85.
Marsico F, Paolillo S, Gargiulo P, et al. Effects of glucagon-like peptide-1 receptor agonists on major cardiovascular events in patients with type 2 diabetes mellitus with or without established cardiovascular disease: a meta-analysis of randomized controlled trials. Eur Heart J. 2020;41:3346–58.
Bray JJH, Foster-Davies H, Salem A, et al. Glucagon-like peptide-1 receptor agonists improve biomarkers of inflammation and oxidative stress: a systematic review and meta-analysis of randomised controlled trials. Diabetes Obes Metab. 2021;23:1806–22.
Buse JB, Wexler DJ, Tsapas A, et al. 2019 update to: management of hyperglycemia in type 2 diabetes, 2018: a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2020;43:487–93.
Das SR, Everett BM, Birtcher KK, et al. 2020 expert consensus decision pathway on novel therapies for cardiovascular risk reduction in patients with type 2 diabetes: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2020;76:1117–45.
Grupo de Trabajo de diabetes, prediabetes y enfermedades cardiovasculares de la European Society of Cardiology (ESC) y la European Association for the Study of Diabetes (EASD). Guía ESC 2019 sobre diabetes, prediabetes y enfermedad cardiovascular, en colaboración con la European Association for the Study of Diabetes (EASD). Rev Esp Cardiol. 2020;73(5):404.e1-404.e59.
American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2021. Diabetes Care. 2021;44:S111–24.
National Institute for Health and Care Excellence. Clinical guideline NG28. Type 2 diabetes in adults: management. [last updated 16 December 2019]. 2015. https://www.nice.org.uk/guidance/ng28. Accessed 22 Oct 2021.
Fundación redGDPS. Guía de diabetes tipo 2 para clínicos: recomendaciones de la redGDPS. 2018 https://www.redgdps.org/gestor/upload/colecciones/Guia%20DM2_web.pdf. Accessed 26 Oct 2021.
Álvarez-Guisasola F, Orozco-Beltrán D, Cebrián-Cuenca AM, et al. Manejo de la hiperglucemia con fármacos no insulínicos en pacientes adultos con diabetes tipo 2. Atención Primaria. 2019;51:442–51.
Sociedad Española de Medicina Interna (SEMI). Recomendaciones para el tratamiento farmacológico de la DM2 (actualización de 2021). 2021. https://www.fesemi.org/sites/default/files/documentos/906.pdf. Accessed 25 Oct 2021.
Gómez Huelgas R, Gómez Peralta F, Carrillo Fernández L, et al. Position statement of the SEMI, SED, redGDPS, SEC, SEEDO, SEEN, SEMERGEN y SEMFYC. Rev Clin Esp (Barc). 2015;215:505–14.
Mata-Cases M, Artola Menéndez S, Díez-Espino J, Ezkurra Loiola P, Franch Nadal J, García Soidán FJ. Actualización de 2020 del algoritmo de tratamiento de la hiperglucemia en la diabetes mellitus tipo 2 de la redGDPS. Diabetes Práctica. 2020;11:41–76.
Vaduganathan M, Patel RB, Singh A, et al. Prescription of glucagon-like peptide-1 receptor agonists by cardiologists. J Am Coll Cardiol. 2019;73:1596–8.
Dave CV, Schneeweiss S, Wexler DJ, Brill G, Patorno E. Trends in clinical characteristics and prescribing preferences for SGLT2 inhibitors and GLP-1 receptor agonists, 2013–2018. Diabetes Care. 2020;43:921–4.
Draznin B, Hirsch IB. Time to follow the evidence: glycemic control and cardiovascular benefits of new diabetes medications. Am J Med. 2021;134:420–2.
Nelson AJ, Ardissino M, Haynes K, et al. Gaps in evidence-based therapy use in insured patients in the United States with type 2 diabetes mellitus and atherosclerotic cardiovascular disease. J Am Heart Assoc. 2021;10: e016835.
Mata-Cases M, Franch-Nadal J, Real J, Vlacho B, Gómez-García A, Mauricio D. Evaluation of clinical and antidiabetic treatment characteristics of different sub-groups of patients with type 2 diabetes: data from a Mediterranean population database. Prim Care Diabetes. 2021;15:588–95.
Norrbacka K, Sicras-Mainar A, Lebrec J, et al. Glucagon-like peptide 1 receptor agonists in type 2 diabetes mellitus: data from a real-world study in Spain. Diabetes Ther. 2021;12:1535–51.
Conget I, Mauricio D, Ortega R, Detournay B, CHADIG Study investigators. Characteristics of patients with type 2 diabetes mellitus newly treated with GLP-1 receptor agonists (CHADIG Study): a cross-sectional multicentre study in Spain. BMJ Open. 2016;6:e010197.
Franch-Nadal J, Mata-Cases M, Ortega E, et al. Glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes: prescription according to reimbursement constraints and guideline recommendations in Catalonia. J Clin Med. 2019;8:E1389.
Mata-Cases M, Franch-Nadal J, Ortega E, et al. Glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes: real-world evidence from a Mediterranean area. Curr Med Res Opin. 2019;35:1735–44.
Tofé S, Argüelles I, Mena E, et al. Real-world GLP-1 RA therapy in type 2 diabetes: a long-term effectiveness observational study. Endocrinol Diabetes Metab. 2019;2: e00051.
Obaya Rebollar JC, Miravet Jiménez S, Aranbarri Osoro I, Carramiñana Barrera FC, García Soidán FJ, Cebrián Cuenca AM. Management of patient profiles with type2 diabetes mellitus in primary care in Spain: CONTROVERTI2 program. SEMERGEN. 2021;S1138–3593(21):00225–32.
Ena J, Carretero-Gómez J, Zapatero-Gaviria A, et al. Uso de terapia antihiperglucemiante con beneficio cardiovascular en pacientes con diabetes tipo 2 que requieren hospitalización: un estudio transversal. Rev Clin Esp. 2021;221:517–28.
Fadini GP, Frison V, Rigato M, et al. Trend 2010–2018 in the clinical use of GLP-1 receptor agonists for the treatment of type 2 diabetes in routine clinical practice: an observational study from Northeast Italy. Acta Diabetol. 2020;57:367–75.
Thong KY, Gupta PS, Cull ML, et al. GLP-1 receptor agonists in type 2 diabetes—NICE guidelines versus clinical practice. Br J Diabetes. 2014;14:52.
Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019;394:121–30.
Morieri ML, Avogaro A, Fadini GP. Long-acting injectable GLP-1 receptor agonists for the treatment of adults with type 2 diabetes: perspectives from clinical practice. Diabetes Metab Syndr Obes. 2020;13:4221–34.
Fernando K, Bain SC, Holmes P, Jones PN, Patel DC. Glucagon-like peptide 1 receptor agonist usage in type 2 diabetes in primary care for the UK and beyond: a narrative review. Diabetes Ther. 2021;12:2267–88.
Khunti K, Gomes MB, Pocock S, et al. Therapeutic inertia in the treatment of hyperglycaemia in patients with type 2 diabetes: a systematic review. Diabetes Obes Metab. 2018;20:427–37.
Schernthaner G, Shehadeh N, Ametov AS, et al. Worldwide inertia to the use of cardiorenal protective glucose-lowering drugs (SGLT2i and GLP-1 RA) in high-risk patients with type 2 diabetes. Cardiovasc Diabetol. 2020;19:185.
Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35:1364–79.
Morillas C, Escalada J, Palomares R, Bellido D, Gómez-Peralta F, Pérez A. Treatment of type 2 diabetes by patient profile in the clinical practice of endocrinology in Spain: Delphi study results from the Think Twice program. Diabetes Ther. 2019;10:1893–907.
López de la Torre M, Botana López M, Rozas Moreno P, Merino Torres J, Escalada San Martín J, Pérez Maraver M. Abordaje en la práctica clínica de endocrinólogos españoles de los aspectos cardiovasculares y renales del paciente con diabetes mellitus tipo 2: resultados de un estudio Delphi. Endocrinol Diabetes y Nutr. 2021;68:111–2.
Polonsky WH, Henry RR. Poor medication adherence in type 2 diabetes: recognizing the scope of the problem and its key contributors. Patient Prefer Adherence. 2016;10:1299–307.
Polonsky WH, Arora R, Faurby M, Fernandes J, Liebl A. Higher rates of persistence and adherence in patients with type 2 diabetes initiating once-weekly vs daily injectable glucagon-like peptide-1 receptor agonists in US clinical practice (STAY Study). Diabetes Ther. 2022;13:175–87.
Thieu VT, Robinson S, Kennedy-Martin T, Boye KS, Garcia-Perez L-E. Patient preferences for glucagon-like peptide-1 receptor–agonist treatment attributes. Patient Prefer Adherence. 2019;13:561–76.
Pollock RF, Norrbacka K, Cameron C, Mancillas-Adame L, Jeddi M. A cost-utility analysis of dulaglutide versus insulin glargine as third-line therapy for type 2 diabetes in Canada. J Comp Eff Res. 2019;8:229–40.
Torre E, Bruno GM, Di Matteo S, et al. Economic evaluation of dulaglutide vs traditional therapies: implications of the outcomes of the Rewind study. Clinicoecon Outcomes Res. 2020;12:177–89.
Yang C-Y, Chen Y-R, Ou H-T, Kuo S. Cost-effectiveness of GLP-1 receptor agonists versus insulin for the treatment of type 2 diabetes: a real-world study and systematic review. Cardiovasc Diabetol. 2021;20:21.
Dilla T, Alexiou D, Chatzitheofilou I, Ayyub R, Lowin J, Norrbacka K. The cost-effectiveness of dulaglutide versus liraglutide for the treatment of type 2 diabetes mellitus in Spain in patients with BMI ≥ 30 kg/m2. J Med Econ. 2017;20:443–52.
Mezquita Raya P, Blasco FJA, Hunt B, et al. Evaluating the long-term cost-effectiveness of fixed-ratio combination insulin degludec/liraglutide (IDegLira) for type 2 diabetes in Spain based on real-world clinical evidence. Diabetes Obes Metab. 2019;21:1349–56.
Vidal J, Malkin SJP, Hunt B, Martín V, Hallén N, Javier OF. The short-term cost-effectiveness of once-weekly semaglutide versus once-daily sitagliptin and once-weekly dulaglutide for the treatment of patients with type 2 diabetes: a cost of control analysis in Spain. Diabetes Ther. 2020;11:509–21.
Martín V, Vidal J, Malkin SJP, Hallén N, Hunt B. Evaluation of the long-term cost-effectiveness of once-weekly semaglutide versus dulaglutide and sitagliptin in the Spanish setting. Adv Ther. 2020;37:4427–45.
Capel M, Ciudin A, Mareque M, Rodríguez-Rincón RM, Simón S, Oyagüez I. Cost-effectiveness analysis of exenatide versus GLP-1 receptor agonists in patients with type 2 diabetes mellitus. Pharmacoecon Open. 2020;4:277–86.
Gorgojo-Martínez JJ, Malkin SJP, Martín V, Hallén N, Hunt B. Assessing the cost-effectiveness of a once-weekly GLP-1 analogue versus an SGLT-2 inhibitor in the Spanish setting: once-weekly semaglutide versus empagliflozin. J Med Econ. 2020;23:193–203.
Mardetko N, Nabergoj Makovec U, Locatelli I, Janez A, Kos M. Uptake of new antidiabetic medicines in 11 European countries. BMC Endocr Disord. 2021;21:127.
Marx N, Davies MJ, Grant PJ, et al. Guideline recommendations and the positioning of newer drugs in type 2 diabetes care. Lancet Diabetes Endocrinol. 2021;9:46–52.
Cosentino F, Grant PJ, Aboyans V, et al. 2019 ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2020;41:255–323.
Mosenzon O, Del Prato S, Schechter M, et al. From glucose lowering agents to disease/diabetes modifying drugs: a «SIMPLE» approach for the treatment of type 2 diabetes. Cardiovasc Diabetol. 2021;20:92.
Moreno-Fernandez J, Garcia-Seco JA, Seco Segura AM, Garcia-Seco F, Rozas Moreno PJ, Aguirre SCM. Liraglutide vs. lixisenatide in obese type 2 diabetes mellitus patients: what effect should we expect in routine clinical practice? Prim Care Diabetes. 2020;14:68–74.
Díaz-Trastoy O, Villar-Taibo R, Sifontes-Dubón M, et al. GLP1 receptor agonist and SGLT2 inhibitor combination: an effective approach in real-world clinical practice. Clin Ther. 2020;42:e1-12.
Carretero Gómez J, Arévalo Lorido JC, Gómez Huelgas R, et al. Combination therapy with glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors in older patients with type 2 diabetes: a real-world evidence study. Can J Diabetes. 2019;43:186–92.
Alonso-Troncoso I, Carollo-Limeres C, Rios-Prego M, Guler I, Cadarso-Suárez C, F-Mariño A. Liraglutide in a real-world setting: joint modeling of metabolic response, prediction of efficacy, and cardiovascular risk. Endocrinol Diabetes Nutr (Engl Ed). 2019;66:376–84.
Gorgojo-Martínez JJ, Gargallo-Fernández MA, Brito-Sanfiel M, Lisbona-Catalán A. Real-world clinical outcomes and predictors of glycaemic and weight response to exenatide once weekly in patients with type 2 diabetes: the CIBELES project. Int J Clin Pract. 2018;72: e13055.
Bellido D, Abellán P, Ruiz Palomar JM, et al. Intensification of basal insulin therapy with lixisenatide in patients with type 2 diabetes in a real-world setting: the BASAL-LIXI study. Curr Ther Res Clin Exp. 2018;89:37–42.
Arévalo-Lorido JC, Gómez JC, Huelgas RG, et al. Lowering blood pressure with the combination of a sodium-glucose cotransporter 2 inhibitor and a glucagon-like peptide-1 receptor agonist in type 2 diabetic patients: a clinical evidence. High Blood Press Cardiovasc Prev. 2018;25:417–20.
Acknowledgements
Funding
This work and the journal’s Rapid Service Fee were funded by Eli Lilly and Company.
Medical Writing, Editorial and Other Assistance
Medical writing assistance was provided by Francisco López de Saro and Sheridan Henness (Rx Communications, Mold, UK), funded by Lilly and Company.
Authorship
All authors met the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the study conception and design. All authors contributed to literature search and analysis, and contributed to the writing of the manuscript. All authors read and approved the final manuscript.
Disclosures
Irene Romera and Miriam Rubio-de Santos are employees and minor shareholders of Eli Lilly and Company. Sara Artola and Carmen Suárez Fernández have no conflicts of interest to declare. Ignacio Conget is a member of the Steering Committee of the REWIND trial.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no data sets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Romera, I., Rubio-de Santos, M., Artola, S. et al. GLP-1 RAs in Spain: A Short Narrative Review of Their Use in Real Clinical Practice. Adv Ther 40, 1418–1429 (2023). https://doi.org/10.1007/s12325-023-02442-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-023-02442-z