Abstract
Introduction
Major depressive disorder (MDD) is a debilitating and costly condition. This analysis characterized the health-related quality of life (HRQoL), health care resource utilization (HCRU), and costs between patients with versus without MDD, and across MDD severity levels.
Methods
The 2019 National Health and Wellness Survey was used to identify adults with MDD, who were stratified by disease severity (minimal/mild, moderate, moderately severe, severe), and those without MDD. Outcomes included HRQoL (Short Form-36v2 Health Survey, EuroQol Five-Dimension Visual Analogue Scale, utility scores), HCRU (hospitalizations, emergency department [ED] visits, health care provider [HCP] visits), and annualized average direct medical and indirect (workplace) costs. A subgroup analysis was conducted in participants with MDD and prior medication treatment failure. Participant characteristics and study outcomes were evaluated using bivariate analyses and multivariable regression models, respectively.
Results
Cohorts comprised 10,710 participants with MDD (minimal/mild = 5905; moderate = 2206; moderately severe = 1565; severe = 1034) and 52,687 participants without MDD. Participants with MDD had significantly lower HRQoL scores than those without (each comparison, P < 0.001). Increasing MDD severity was associated with decreasing HRQoL. Relative to participants without MDD, participants with MDD reported more HCP visits (2.72 vs 5.64; P < 0.001) and ED visits (0.18 vs 0.22; P < 0.001) but a similar number of hospitalizations. HCRU increased with increasing MDD severity. Although most patients with MDD had minimal/mild to moderate severity, total direct medical and indirect costs were significantly higher for participants with versus without MDD ($8814 vs $6072 and $5425 vs $3085, respectively, both P < 0.001). Direct and indirect costs were significantly higher across all severity levels versus minimal/mild MDD (each comparison, P < 0.05). Among patients with prior MDD medication treatment failure (n = 1077), increasing severity was associated with significantly lower HRQoL and higher total indirect costs than minimal/mild MDD.
Conclusion
These results quantify the significant and diverse burdens associated with MDD and prior MDD medication treatment failure.
Plain Language Summary
This study described the burdens associated with major depressive disorder. To accomplish this, we compared outcomes from a national health survey between patients who had a diagnosis of major depressive disorder and those who did not. Participants with major depressive disorder were further characterized by the severity of their symptoms. The first outcome was health-related quality of life and the second outcome was the amount of health visits, such as the number of hospitalizations, emergency department visits, and visits with health care providers. Finally, health care-related costs and workplace-related costs were evaluated. Survey participants with major depressive disorder had lower health-related quality of life scores compared with those without major depressive disorder. Increasing severity of major depressive disorder was linked with decreasing health-related quality of life. Participants with major depressive disorder also reported more health care provider and emergency department visits relative to participants without the disorder, although they both reported a similar number of hospitalizations. Both health care-related and workplace-related costs were higher in participants with major depressive disorder than in those without major depressive disorder, and costs were higher among participants with more severe symptoms compared with minimal/mild symptoms. Among participants who had major depressive disorder and reported that their current medication had replaced an old medication because of a lack of response, increasing major depressive disorder severity was associated with significantly lower health-related quality of life scores and higher total workplace-related costs versus minimal/mild major depressive disorder.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Major depressive disorder is a debilitating and costly condition |
This study aimed to characterize the effects of major depressive disorder on health-related quality of life, health care resource utilization, and direct medical and indirect costs |
Health-related quality of life was lower in those with major depressive disorder relative to those without, and health-related quality of life decreased with increasing severity of major depressive disorder |
Health care resource utilization and costs showed similar trends, with higher health care resource utilization and costs in those with major depressive disorder versus without; these outcomes also increased with increasing severity |
Finally, among patients reporting a prior major depressive disorder treatment failure, the significant association between increasing major depressive disorder severity with lower health-related quality of life and higher indirect costs was still observed |
Introduction
Major depressive disorder (MDD) is one of the most prevalent mental health conditions in the USA and affects an estimated 20.6% of adults during their lifetime [1]. Characterized by a persistent state of depressed mood and/or anhedonia [2], MDD is severely debilitating and remains among the most burdensome disorders worldwide [3, 4]. Furthermore, identifying effective treatment that also addresses patient-centered outcomes can be difficult, with one analysis finding approximately 50% of patients treated with antidepressants continued to experience “severely impaired” quality of life, despite experiencing response to treatment [5].
As the leading cause of disability worldwide, MDD contributes significantly to increased health care resource utilization (HCRU) and costs, as well as lost workplace productivity [4, 6,7,8]. For instance, one study found patients with MDD had at least two times the rate of emergency department (ED) visits and at least three times the rate of hospitalizations relative to patients without MDD [4, 9]. Furthermore, the economic burden of MDD in the USA is high and is increasing; from 2010 to 2018, annual overall costs for MDD, including direct, suicide-related, and workplace (indirect) costs, increased by 38% from $237 billion to $326 billion [10]. The largest growth in costs was attributed primarily to workplace costs, including absenteeism due to missed days of work and presenteeism due to lost productivity while at work, which increased from 48% of the annual economic burden in 2010 to 61% in 2018 [10]. Although direct costs comprise a relatively lower proportion of the economic burden of MDD, they still contribute substantially [10]. In a retrospective analysis of claims data and hospital admission records, Citrome et al. reported that the mean cost for MDD-related inpatient stays was approximately $6700 and over 5% of hospitalized patients were readmitted within 30 days following discharge [11]. Additionally, results from a retrospective claims-based analysis indicated that mean annual mental health-related costs were $3753 higher for patients with MDD than patients without MDD [9], demonstrating how medical costs can exacerbate the economic burden of patients with MDD [12].
The treatment of MDD can be complex, and remission may be difficult to achieve [9]. While antidepressants are the first-line treatment for MDD recommended by the 2010 American Psychiatric Association (APA) guidelines [2], approximately one-third of patients with MDD experience inadequate response to antidepressant monotherapy [13]. Moreover, the likelihood of achieving remission decreases with each subsequent treatment step [13]. For example, in an analysis of patients requiring multiple treatment steps in the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, rates of remission decreased with each additional therapy change, including from 36.8% after the first line of therapy to 13.0% after the fourth line of therapy [13]. Consequences of not achieving remission from depression include a continuation of functional impairment, increased health care costs, and reduced health-related quality of life (HRQoL), emphasizing the importance of effective treatment early in the course of illness [14, 15].
While previous research has demonstrated the significant burden of MDD, further exploration is needed to understand the impact of increasing MDD severity and prior MDD medication treatment failure. The objective of this study was to further characterize the humanistic and economic burden of MDD using data from the 2019 National Health and Wellness Survey (NHWS). The incremental burden associated with increasing MDD severity was evaluated by comparing outcomes across patients with minimal/mild, moderate, moderately severe, and severe MDD. To explore the impact of prior MDD medication treatment failure on these outcomes, a subgroup analysis was conducted among patients who self-reported failure of at least one MDD medication.
Methods
Data Source
This study analyzed data from the 2019 NHWS, a nationally representative, self-administered, internet-based survey that is completed annually by approximately 75,000 US adults. Quota sampling is used to ensure national representativeness in terms of age, gender, race and ethnicity as determined by annual Census Bureau data. Participants are identified using opt-in survey panels, and demographics and health characteristics are assessed via a base questionnaire. Disease state-specific modules are then completed by participants to evaluate health conditions and behaviors using a variety of validated instruments. Of note, this study was exempt from ethics committee approval and institutional review because it is a retrospective analysis which used anonymized and de-identified data certified as fully compliant with US patient confidentiality requirements set forth in the Health Insurance Portability and Accountability Act of 1996. Allergan (prior to its acquisition by AbbVie) obtained permission to access and use the 2019 National Health and Wellness Survey data used in the analysis through licensing agreements.
Study Sample
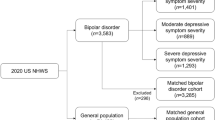
The MDD cohort consisted of adults (≥ 18 years of age) with a self-reported physician diagnosis of MDD who were not diagnosed with any type of bipolar disorder in the past 12 months; all participants with MDD were also required to screen negative for probable bipolar I disorder on the Mood Disorder Questionnaire (MDQ) [16]. Current MDD severity was then determined using Patient Health Questionnaire (PHQ-9) cutoff scores for minimal/mild (0–9), moderate (10–14), moderately severe (15–19), or severe (≥ 20) MDD [17]. Participants with MDD were compared with a cohort of participants who did not self-report any experience with either MDD or bipolar disorder in the previous 12 months and did not report a diagnosis of MDD or bipolar disorder at any time. Participants were excluded from both cohorts if they had a prior diagnosis of schizophrenia at any time.
A subgroup analysis was performed and included participants with MDD who reported a prior MDD medication treatment failure. These participants were identified from the survey as those who were currently taking a medication for MDD that replaced a prior MDD medication and self-reported “not responding to treatment” as one of the reasons for replacement, with no time limit specified. Participants in this subgroup analysis were also stratified by current disease severity via PHQ-9 scores.
Participant Demographics and Health Characteristics
Demographic information collected from the survey was self-reported and included age, sex, race and ethnicity, marital status, university education, annual household income, unemployment status, disability, health insurance type, alcohol use, exercise status, and current smoker status. Health characteristics, including time since MDD diagnosis, number of comorbid mental health diagnoses, and body mass index (BMI, kg/m2), were also collected. Furthermore, all participants were required to complete relevant instruments, including the Charlson Comorbidity Index (CCI, higher scores indicative of higher mortality risk) [18] and General Anxiety Disorder-7 (GAD-7, score 0–21; higher scores indicative of more severe anxiety) [19]. Participants also self-reported information regarding MDD treatment, including receipt of an MDD prescription, types of medications currently being used, and adherence (0–100 Visual Analogue Scale [VAS]).
Study Outcomes
Health-Related Quality of Life
HRQoL was assessed using the 36-item Short Form version 2 (SF-36v2) health survey physical and mental component summary scores and the EuroQol Five-Dimension Visual Analogue Scale (EQ-5D VAS). The SF-36v2 consists of 36 items assigned to eight physical and mental health domains. These domain subscales are constructed into physical and mental component summary scores ranging from 0 to 100, with higher scores indicating a more favorable health state [20]. Scores are then standardized to the normative mean value of 50 using software from Quality Metric [21]. The EQ-5D VAS is a self-assessment questionnaire using a VAS to rate the positive (well-being) and negative (illness) aspects of current health. Scores range from 0 to 100, with higher scores indicative of better overall HRQoL [22]. The average (SD) online-reported EQ-5D VAS score in the USA is 74.6 (18.7) [23]. Utility indices, including Short Form 6-Dimension (SF-6D) health state utility scores and EQ-5D-5 level (5L) utility scores, were used to enable an evaluation of health relative to values obtained from the US general population. These utility scores constitute a single-index, preference-based measure of health, as described elsewhere [24,25,26,27].
Health Care Resource Utilization and Costs
To determine HCRU, participants were prompted to recall the number of health care provider (HCP) visits, ED visits, and hospitalizations they had over the prior 6-month period. Direct medical costs were then annualized by doubling the self-reported number of HCRU visits for each participant and multiplying the number of visits by its average unit cost according to the 2018 Medical Expenditure Panel Survey [28,29,30]. Indirect costs were calculated for employed participants using results from the self-reported Work Productivity and Activity Impairment questionnaire [31], which prompts participants to recall the impact that health had on their work during the last 7 days. Annualized indirect costs, including mean cost of absenteeism, mean cost of presenteeism, and total average indirect costs (absenteeism plus presenteeism costs) were then calculated by multiplying the number of lost work hours by age- and gender-stratified median 2018 wages reported by the Bureau of Labor and Statistics [32]. Additionally, average total costs (average direct medical costs plus average indirect costs) were calculated and annualized to 12 months.
Statistical Analysis
Raw NHWS data were analyzed with chi-square tests and analysis of variance (ANOVA) to compare demographic characteristics, socioeconomic status, general health characteristics, and health conditions and behaviors of participants with versus without MDD and across the MDD severity cohorts. Multivariable regression models were used to compare HRQoL, HCRU, average annualized direct medical, indirect, and total costs for participants with MDD versus those without MDD and between participants with minimal/mild, moderate, moderately severe, or severe MDD. In the severity analyses, the minimal/mild MDD cohort was used as the reference group. All multivariable analyses controlled for potential confounders, including sex, age, race and ethnicity, education, employment status, annual household income, insurance status, marital status, smoking history, CCI, and BMI. Additional covariates were used when comparing severity groups to control for different disease and treatment statuses, including total number of comorbid mental health conditions, current MDD medication use (yes, no), and early-onset MDD diagnosis status (diagnosed between the ages of 12–24 years, diagnosed after age 25 years, unknown). The analysis was repeated in the subgroup of patients with prior MDD medication treatment failure.
Results
Participant Demographic and Health Characteristics
Out of 74,994 survey respondents, a total of 10,710 participants had a self-reported diagnosis of MDD and were compared with 52,687 participants who did not self-report a diagnosis of MDD or bipolar disorder. Of the participants with MDD, 5905 (55%) were classified in the minimal/mild cohort, 2206 (21%) in the moderate cohort, 1565 (15%) in the moderately severe cohort, and 1034 (10%) in the severe cohort. Participants with MDD had a mean (SD) duration of MDD of 13.8 (10.9) years.
Demographic and health characteristics of participants with versus without MDD are shown in Table 1. On average, the cohort of participants with MDD was younger than the cohort without MDD (44.4 vs 49.4 years, P < 0.05) and included more female participants (70.6% vs 53.1%, P < 0.05). Additionally, MDD was consistently associated with poorer socioeconomic status. For example, a significantly higher proportion of participants with MDD relative to those without MDD were single, unemployed, had less than a university education, reported long- or short-term disability, and had an annual household income of less than $25,000 per year (each comparison, P < 0.05). MDD was also associated with poorer general health. Relative to participants without MDD, those with MDD reported a greater comorbidity burden as measured by the CCI, a greater level of anxiety as measured by the GAD-7, a greater number of mental health diagnoses, and greater average BMI; they were also more likely to currently smoke and less likely to exercise (each comparison, P < 0.05).
Furthermore, socioeconomic status and general health decreased with increasing MDD severity (Supplementary Table 1). For instance, relative to participants with minimal/mild MDD, those with severe MDD were more likely to be unemployed (43.0% vs 52.6%), have an annual household income of less than $25,000 per year (19.5% vs 33.9%), report short- or long-term disability (6.3% vs 15.8%), have less than a university education (40.5% vs 58.5%), and be uninsured (8.0% vs 15.4%; each comparison, P < 0.05). Additionally, compared with the minimal/mild MDD cohort, participants with severe MDD reported a higher BMI (29.6 vs 30.8, P < 0.05), a higher number of comorbid mental health diagnoses (1.3 vs 2.3, P < 0.05), a higher comorbidity burden as measured by the CCI (0.61 vs 0.79, P < 0.05), and a greater level of anxiety as measured by the GAD-7 (4.6 vs 15.1, P < 0.05). They were also more likely to currently smoke (16.7% vs 26.3%, P < 0.05) and less likely to exercise (36.6% vs 48.3%, P < 0.05).
Analysis of MDD treatment revealed approximately 60% of participants with MDD were currently using a prescribed medication for treatment. Of these participants, over 80% were on a selective serotonin reuptake inhibitor (SSRI) or serotonin and norepinephrine reuptake inhibitor (SNRI). The second most common prescription was for bupropion (21%), followed by atypical antipsychotics/antipsychotic combination pills (8%). Of those not currently on medication for MDD, 63% had previously been prescribed a treatment. In the overall MDD cohort, the mean (SD) medication adherence was 87.3% (22.9) as measured on a 0–100 VAS. Participants in the minimal/mild MDD cohort had a longer duration of medication use than participants with severe MDD (81.8 months vs 59.6 months, P < 0.05) and higher medication adherence as measured on a 0−100 VAS (90.0% vs 82.1%, P < 0.05); current use of a prescription medication and type of medication used generally did not significantly differ across MDD severity levels.
Impact of MDD on Health-Related Quality of Life, Health Care Resource Utilization, and Cost Outcomes
Health-Related Quality of Life
Relative to participants without MDD, participants with MDD reported significantly lower HRQoL, with lower scores on both the mental and physical components of the SF-36v2 and EQ-5D VAS (Fig. 1a). Additionally, MDD severity was strongly associated with HRQoL—as depression severity increased, HRQoL decreased as shown by significantly lower scores in SF-36v2 (both mental and physical components) and EQ-5D VAS scores across the moderate, moderately severe, and severe MDD cohorts versus the minimal/mild MDD cohort (Fig. 1b).
HRQoL of a participants with MDD versus without MDD and b across MDD severity levels. ***P < 0.001 vs participants without MDD; †††P < 0.001 vs the minimal/mild MDD cohort; higher scores indicate better HRQoL. aEstimates shown were derived from separate models predicting SF-36v2 MCS, SF-36v2 PCS, and EQ-5D VAS scores. Model covariates for comparing to the general population a included sex, age, race and ethnicity, education, employment status, income, insurance status, marital status, smoking history, CCI, and BMI, while severity comparisons b included additional covariates relating to early-onset MDD diagnosis status, number of comorbid mental health conditions, and current medication use for MDD. bSE values are labeled in the figure rather than shown as error bars given the small scale. BMI body mass index, CCI Charlson Comorbidity Index, EQ-5D VAS EuroQol Five-Dimension Visual Analogue Scale, HRQoL health-related quality of life, MCS mental component summary, MDD major depressive disorder, Mod Severe moderately severe, PCS physical component summary, SE standard error, SF-36v2 Short Form 36v2 Health Survey
Health utility scores followed a similar pattern to the SF-36v2 and EQ-5D VAS. The average SF-6D health state utility score for participants with MDD was significantly lower than those without MDD (0.64 vs 0.75, P < 0.001), and the average EQ-5D-5L utility score was significantly lower for participants with MDD (0.74) relative to patients without MDD (0.86; P < 0.001). Furthermore, utility scores decreased with increasing MDD disease severity. The SF-6D health state utility scores decreased across minimal/mild (0.66), moderate (0.59), moderately severe (0.56), and severe MDD (0.52; each comparison vs mild MDD cohort, P < 0.001). Additionally, the average EQ-5D-5L utility scores were 0.77, 0.71, 0.65, and 0.57 for patients with minimal/mild, moderate, moderately severe, and severe MDD, respectively (each comparison vs minimal/mild MDD cohort, P < 0.001).
Health Care Resource Utilization
Relative to participants without MDD, participants with MDD reported a significantly greater number of HCP visits and ED visits during the 6 months prior to survey completion; the number of hospitalizations was similar (Fig. 2a). Increasing MDD severity was also associated with greater HCRU. Compared with the minimal/mild MDD cohort, participants with moderate, moderately severe, or severe MDD had a greater average number of HCP visits (minimal/mild = 5.47, moderate = 6.05, moderately severe = 6.51, severe = 6.91), ED visits (minimal/mild = 0.26, moderate = 0.33, moderately severe = 0.37, severe = 0.46), and hospitalizations (minimal/mild = 0.01, moderate = 0.17, moderately severe = 0.18, severe = 0.22) in the past 6 months. All HCRU differences between the minimal/mild cohort and the moderate, moderately severe, and severe cohorts were significant (each comparison, P < 0.05; Fig. 2b).
HCRU of a participants with MDD versus without MDD and b across MDD severity levels. ***P < 0.001 vs participants without MDD; †P < 0.05, ††P < 0.01, †††P < 0.001 vs the minimal/mild MDD cohort. aEstimates shown were derived from separate models predicting the number of HCP visits, ED visits, and hospitalizations. Model covariates for comparing to the general population a included sex, age, race and ethnicity, education, employment status, income, insurance status, marital status, smoking history, CCI, and BMI, while severity comparisons b included additional covariates relating to early-onset MDD diagnosis status, number of comorbid mental health conditions, and current medication use for MDD. bSE values are labeled in the figure rather than shown as error bars given the small scale. BMI body mass index, CCI Charlson Comorbidity Index, ED emergency department, HCP health care provider, HCRU health care resource utilization, MDD major depressive disorder, Mod Severe moderately severe, SE standard error
Average Direct Medical Costs
Annualized mean total direct medical costs were significantly higher for participants with MDD ($8814) than for participants without MDD ($6072; P < 0.001; Fig. 3a). Participants with versus without MDD also had significantly higher annualized mean direct medical costs for HCP visits ($3571 vs $1662; P < 0.001), ED visits ($559 vs $366; P < 0.001), and hospitalizations ($4408 vs $3495; P < 0.01). Furthermore, increasing severity of MDD was associated with incrementally higher annualized mean total direct medical costs, with significant differences versus the minimal/mild MDD cohort for all severity levels (each comparison, P < 0.05; Fig. 3b). The average costs of HCP and ED visits increased with increasing MDD severity (HCP costs: minimal/mild = $3414; moderate = $3811; moderately severe = $4083; severe = $4352; each comparison vs minimal/mild MDD cohort, P < 0.01; ED costs: minimal/mild = $553; moderate = $711; moderately severe = $775; severe = $1063; each comparison vs minimal/mild MDD cohort, P < 0.01). While the average cost attributable to hospitalizations was numerically higher in the more severe groups, the differences were not statistically significant (minimal/mild = $3927; moderate = $5121; moderately severe = $5187; severe = $6398, each comparison vs minimal/mild MDD cohort, P > 0.05).
Annualized direct medical costs of a participants with MDD versus without MDD and b across MDD severity levels. **P < 0.01, ***P < 0.001 vs participants without MDD; †P < 0.05, ††P < 0.01, †††P < 0.001 vs the minimal/mild MDD cohort. aEstimates shown were derived from separate models predicting the cost of HCP visits, ED visits, hospitalizations, and total costs; therefore, individual component costs may not add up to total costs. Further, model covariates for comparing to the general population a included sex, age, race and ethnicity, education, employment status, income, insurance status, marital status, smoking history, CCI, and BMI, while severity comparisons b included additional covariates relating to early-onset MDD diagnosis status, number of comorbid mental health conditions, and current medication use for MDD. BMI body mass index, CCI Charlson Comorbidity Index, ED emergency department, HCP health care provider, MDD major depressive disorder, Mod Severe moderately severe
Average Indirect Costs
Participants with MDD incurred significantly greater annualized mean total indirect costs versus participants without MDD ($5425 vs $3085; P < 0.001; Fig. 4a). The average costs of absenteeism and presenteeism were also significantly higher for participants with versus without MDD. Compared with participants in the minimal/mild MDD cohort, participants in the moderate, moderately severe, and severe MDD cohorts had significantly greater annualized mean total indirect costs (minimal/mild = $4490; moderate = $6537; moderately severe = $7438; severe = $8797; each comparison vs minimal/mild MDD cohort, P < 0.001; Fig. 4b). Increasing MDD severity was also associated with higher average costs of absenteeism (minimal/mild = $1201; moderate = $1802; moderately severe = $2138; severe = $3233) and presenteeism (minimal/mild = $3225; moderate = $4662; moderately severe = $5222; severe = $5461) relative to those of the minimal/mild MDD cohort (each comparison vs minimal/mild MDD cohort, P < 0.001).
Annualized indirect costs of a participants with MDD versus without MDD and b across MDD severity levels. ***P < 0.001 vs participants without MDD; †††P < 0.001vs the minimal/mild MDD cohort. aEstimates shown were derived from separate models predicting the cost of absenteeism, the cost of presenteeism, and total costs; therefore, individual component costs may not add up to total costs. Further, model covariates for comparing to the general population a included sex, age, race and ethnicity, education, employment status, income, insurance status, marital status, smoking history, CCI, and BMI, while severity comparisons b included additional covariates relating to early-onset MDD diagnosis status, number of comorbid mental health conditions, and current medication use for MDD. BMI body mass index, CCI Charlson Comorbidity Index, MDD major depressive disorder, Mod Severe moderately severe
Total Costs
Annualized mean total costs (direct medical plus indirect costs) were significantly higher for participants with MDD versus without MDD ($14,658 vs $9091; P < 0.001). Increasing MDD severity was also associated with increasing mean total costs, with significantly higher total mean costs in the moderate ($17,314), moderately severe ($18,263), and severe ($20,901) MDD cohorts than in the minimal/mild MDD cohort ($12,756; each comparison, P < 0.001).
Subgroup Analysis of Participants with Prior MDD Medication Treatment Failure
The prior MDD medication treatment failure subgroup analysis included 1077 participants with MDD who indicated that their current medication had replaced a prior medication because of non-response. Of these participants, severity cohorts were as follows: minimal/mild, n = 517 (48%); moderate, n = 244 (23%); moderately severe, n = 187 (17%); and severe, n = 129 (12%) (Table 2).
Table 3 summarizes the HRQoL, HCRU, and cost data across severity levels of participants with a prior MDD medication treatment failure. Observed trends were similar to those in the main analysis. Participants with severe MDD reported significantly more hospitalizations than those with minimal/mild MDD (0.22 vs 0.10, P < 0.05). Annualized mean total direct medical costs were significantly greater for participants in the severe prior MDD medication treatment failure cohort ($15,420) versus the minimal/mild prior MDD medication treatment failure cohort ($9852; P < 0.05). Furthermore, annualized mean total indirect costs were significantly greater for all severity levels relative to minimal/mild MDD. Costs associated with absenteeism were only significantly greater in the severe MDD cohort versus the minimal/mild MDD cohort, while costs associated with presenteeism were significantly greater among all MDD severity cohorts compared with those in the minimal/mild MDD cohort.
Discussion
This large, cross-sectional, nationally representative survey study highlights the substantial and diverse burdens that patients with MDD experience and reveals that these burdens increase incrementally with MDD severity. Participants with MDD had significantly lower HRQoL and more HCRU relative to participants without MDD, as well as higher average direct, indirect, and total annualized costs. Similar results were observed with increasing MDD severity, as participants with moderate, moderately severe, and severe MDD reported significantly lower HRQoL and higher HCRU and average annual direct, indirect, and total costs than those with minimal/mild MDD. Among participants with a prior MDD medication treatment failure, increased MDD severity was associated with significantly lower HRQoL scores and higher annualized mean total indirect costs compared with minimal/mild MDD. The HRQoL, HCRU, and economic burdens observed in this study are significant and demonstrate a critical unmet need for more effective treatment of MDD.
Many participants with MDD in this study reported poorer socioeconomic status, based on factors such as disability, unemployment, low annual household income, and less formal education, relative to participants without MDD. Moreover, increasing MDD severity was also associated with decreasing socioeconomic status, suggesting that individuals with lower socioeconomic status may not only be more likely to have MDD but also suffer from a higher severity of MDD. These results are consistent with previous studies indicating that individuals with more years of education and higher annual household income have a lower risk of MDD onset compared with individual who have lower socioeconomic status [33, 34]. While MDD does not necessarily cause lower socioeconomic status, it is feasible that individuals with a lower socioeconomic status may suffer from systemic barriers that prevent access to care and treatment, therefore leading to higher rates of the disorder.
The results of this analysis are in agreement with previous research on the HRQoL burdens associated with MDD. For example, it has been observed that patients with MDD commonly experience significant impairments in HRQoL, which worsens with increased disease severity [4, 7, 35, 36]. Utilizing a larger, nationwide patient population, with more recent survey data, our results further support these findings, with significantly lower levels of HRQoL seen in participants with MDD versus without MDD; increasing MDD severity was also strongly associated with declining HRQoL. Deficits in HRQoL can increase the risk of relapse and therefore direct and indirect costs [5]. Because of its effect on health and costs, understanding the effect of MDD on patient-centered outcomes, such as HRQoL, is vital.
The HCRU and economic burdens demonstrated in our study are also supported by previous analyses. For example, in an analysis that used IBM® MarketScan® US commercial claims data, Zhu and colleagues observed higher economic burden and greater workplace impairment in patients with MDD compared with a population without MDD [9]. While Zhu et al. did not analyze outcomes across different severity levels, they did find MDD to be associated with an average of 11.0 more all-cause HCRU visits per year and $6809 higher annual mean direct medical costs compared with a non-MDD comparison population. Similarly, participants with MDD in our analysis reported over two times the number of HCP visits as participants without MDD, and annualized total mean direct costs were about 45% higher among the MDD cohort. Furthermore, another study analyzed economic burden and workplace impairment across MDD severity using 2001–2002 data and showed that both treatment costs and lost productivity costs increased significantly with increased severity [37]. Our analysis, which used a more recent dataset, found similar results; however, our results showed that both total direct and indirect costs increased significantly in the moderate, moderately severe, and severe MDD cohorts compared with the minimal/mild MDD cohort. Finally, a recently published study by Jain et al. analyzing survey respondents with versus without an MDD diagnosis also reported that MDD was associated with lower HRQoL and higher HCRU and costs compared with respondents without MDD [4]. While similar in design, our trial used a more recent dataset (2019 NHWS vs 2017 NHWS) and had a larger comparison population (n = 52,687 vs n = 30,478) and MDD population (n = 10,710 vs n = 8853) than the analysis by Jain et al. We also analyzed direct medical costs, further building upon Jain et al.’s findings focused on indirect costs. Moreover, it should be noted that Jain et al. evaluated severity groups against participants with minimal depressive symptoms (0–4 on PHQ-9) as a comparator as opposed to our analysis which used minimal/mild MDD (0–9 on PHQ-9) as a comparator. Because our comparator group had an average PHQ-9 score of 4.9, above the threshold for minimal symptoms, the significant differences seen in our severity analysis are even more remarkable than previous studies that compared outcomes to a population with minimal symptoms only. Together, these results demonstrate that patients with MDD have significant humanistic and economic burdens that generally increase with increasing severity.
In contrast to our findings, both Zhu et al. and Jain et al. found increasing hospitalizations associated with MDD, while our analysis found no significant difference in rates of hospitalization. While the exact reasons for this discrepancy are unclear, it may be due in part to differences in study design. For example, Zhu et al. collected data via a claims database analysis as opposed to patient-reported surveys which may have accounted for the discrepancy in results [9]. Furthermore, while Jain et al. had a similar study design as our analysis, it only evaluated patients between the ages of 18 and 64 [4], while our evaluation included patients over 65. As older individuals may have increased rates of hospitalization [38], the difference in age demographics in our patient population relative to Jain et al. could have affected our results.
Prior studies, such as that by Greenberg et al., reported that indirect costs constitute the majority of the annual economic burden of MDD in the USA. In our study, the total cost of care attributable to direct vs indirect costs was different, likely because of varying recall periods used to calculate direct and indirect costs. Direct costs were determined from a recall of HCRU visits over a period of 6 months, while indirect costs were determined from the WPAI with a recall period of 7 days which was then extrapolated to annual costs. However, presenteeism and absenteeism for patients with MDD may differ depending on how well the patient is managed and may vary throughout the year. Therefore, it is likely that the indirect costs in our study are underestimated.
Our study builds upon previously published results by describing HRQoL, HCRU, and cost burdens in a population that has experienced inadequate response to prior MDD treatment. Similar to the overall MDD population, findings from our subgroup analysis showed that increasing MDD severity in participants with a prior MDD medication treatment failure was associated with significantly decreased HRQoL scores. Additionally, while the analysis of participants with a prior MDD treatment failure was designed to compare different severity groups with the mild subgroup rather than the overall MDD population, point estimates for indirect costs were unexpectedly lower for participants with a prior MDD medication treatment failure relative to the overall MDD population. However, the point estimates for direct costs and total costs (indirect + direct) were generally higher compared to the overall MDD population, suggesting that high direct costs outweighed the low indirect costs and contributed to high overall costs. This is consistent with a claims-based analysis by Arnaud et al., which indicated that patients who completed treatment with a single line of antidepressant treatment had lower direct costs compared with patients who used two or more lines of treatment, although this analysis also found employment-related costs to be lower [39]. High direct costs are potentially due to patients with prior treatment failures experiencing treatment gaps leading to the use of more health care resources, as evident in higher rates of health care provider visits. Furthermore, previous analyses have also found early treatment nonresponse predicts poor outcomes in patients with MDD and therefore may contribute to higher direct costs [40]. The reason for the numerically lower indirect costs versus the overall MDD population among patients with prior MDD medication treatment failure is unknown. However, the timing of prior medication treatment failure was unaccounted for and therefore comparisons between these populations may be less meaningful than comparisons between severity groups. Nevertheless, our results highlight the significant HRQoL and indirect cost burdens associated with prior MDD medication treatment failure on patients and employers. These findings are further problematic when put into the context that the likelihood of achieving remission decreases with each subsequent treatment step [13], supporting the need for early and effective MDD treatment.
Limitations
Results of this study should be interpreted within the context of its limitations, including the potential for selection bias since the NHWS is an online survey available only to those with internet access. Furthermore, all data were self-reported and not verified via other data sources, such as electronic medical records or physician reports. Moreover, causality between diagnoses and health characteristics could not be determined, as NHWS data are cross-sectional. Additionally, the recall period for HCRU was the 6-month period prior to survey completion, during which a participant’s depression severity may have shifted. Also, as a result of the nature of a one-time survey of the general population, the recall period for the WPAI was 7 days which was then extrapolated to 12 months. This assumed work productivity was unchanged over 12 months, which may not have been the case for all employed participants. However, as this was a cross-sectional analysis focusing on between-group differences, any systematic over- or underestimation would be evenly distributed across respondents and therefore have minimal impact on the results. Some results could also have been influenced by variables that were not collected by the survey. For example, common comorbidities associated with MDD, such as substance use disorder, were not considered in our study though they could impact participant health characteristics. Additionally, since the timing of prior MDD medication treatment failure was unavailable and therefore unaccounted for, the results may underrepresent the burden of patients with a recent MDD medication treatment failure. Finally, it should also be noted that PHQ-9 scores of 0–4 are usually indicative of no/minimal depressive symptoms; however, we included participants with PHQ-9 scores of 0–9 in the minimal/mild subgroup to avoid excluding any participants from our comparison population. Therefore, although all participants were required to have an MDD diagnosis, it is feasible that some participants had minimal symptoms while others had mild symptoms.
Conclusions
The present study highlights the diverse burdens associated with MDD that can be significant and far-reaching. A systematic pattern of worsened HRQoL, as well as increased HCRU and costs, was seen for participants with MDD relative to participants without MDD. Additionally, increased MDD severity was generally associated with increased burdens. These same trends were observed in participants with prior MDD medication treatment failure, emphasizing the importance of early, appropriate MDD treatment. These results underscore the need for efficacious MDD treatment regimens to minimize disease severity and decrease the associated humanistic and economic burdens.
Data Availability
The NHWS and its annual data are proprietary to Cerner Enviza and, thus, are not publicly available without a licensing agreement.
References
Hasin DS, Sarvet AL, Meyers JL, et al. Epidemiology of adult DSM-5 major depressive disorder and its specifiers in the United States. JAMA Psychiat. 2018;74(4):336–46.
Gelenberg AJ, Freeman MP, Markowitz JC, et al. Practice guidelines for the treatment of patients with major depressive disorder. Providence: American Psychiatric Association; 2010.
Otte C, Gold SM, Penninx BW, et al. Major depressive disorder. Nat Rev Dis Primers. 2016;2:16065.
Jain S, Gupta S, Li VW, Suthoff E, Arnaud A. Humanistic and economic burden associated with depression in the United States: a cross-sectional survey analysis. BMC. 2022;22(1):542.
IsHak WW, Mirocha J, James D, et al. Quality of life in major depressive disorder before/after multiple steps of treatment and one-year follow-up. Acta Psychiatr Scand. 2015;131(1):51–60.
Egede LE. Major depression in individuals with chronic medical disorders: prevalence, correlates and association with health resource utilization, lost productivity and functional disability. Gen Hosp Psychiatry. 2007;29(5):409–16.
Beck A, Crain AL, Solberg LI, et al. Severity of depression and magnitude of productivity loss. Ann Fam Med. 2011;9(4):305–11.
World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates. (Geneva: Licence: CC BY-NC-SA 3.0 IGO.). 2017.
Zhu L, Ferries E, Suthoff E, Namjoshi M, Bera R. Economic burden and antidepressant treatment patterns among patients with major depressive disorder in the United States. J Manag Care Spec Pharm. 2022;28(11-a Suppl):S2–13.
Greenberg PE, Fournier A, Sisitsky T, et al. The economic burden of adults with major depressive disorder in the United States. Pharmacoeconomics. 2021;39:653–65.
Citrome L, Jain R, Tung A, Landsman-Blumberg PB, Kramer K, Ali S. Prevalence, treatment patterns, and stay characteristics associated with hospitalizations for major depressive disorder. J Affect Disord. 2019;249:378–84.
Cutler AJ, Keyloun KR, Higa S, et al. Annual costs among patients with major depressive disorder and the impact of key clinical events. J Manag Car Spec Pharm. 2022;28(12):1335–43.
Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905–17.
Thase M, Fava M, Zimmerman M, Culpepper L. Managing drepression in primary care: achieving remission. J Clin Psychiatry. 2006;8(2):88–97.
Sobocki P, Ekman M, Agren H, Runeson B, Jonsson B. The mission is remission: health economic consequences of achieving full remission with antidepressant treatment for depression. Int J Clin Pract. 2006;60(7):791–8.
Hirschfeld RM, Williams JB, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry. 2000;157(11):1873–5.
Levis B, Benedetti A, Thombs BD. Accuracy of Patient Health Questionnaire-9 (PHQ-9) for screening to detect major depression: individual participant data meta-analysis. BMJ. 2019;12:365.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
Spitzer RL, Kroenke K, Williams JB, Löwe B. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092–7.
Ware JE Jr, Sherbourne C. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473–83.
Quality Metric. Score range for the SF-36v2 Health Survey and SF-12v2 Health Surveys. Data sheet. https://www.qualitymetric.com/score-range-data-sheet/. Accessed 18 Jan 2024.
EuroQol Group. EuroQol—a new facility for the measurement of health-related quality of life. Health Policy. 1990;16:199–208.
Jiang R, Janssen MFB, Pickard AS. US population norms for the EQ-5D-5L and comparison of norms from face-to-face and online samples. Qual Life Res. 2021;30(3):803–16.
Rowen D, Brazier J, Roberts J. Mapping SF-36 onto the EQ-5D index: how reliable is the relationship? Health Qual Life Outcomes. 2009. https://doi.org/10.1186/1477-7525-7-27.
Brazier JE, Mulhern BJ, Bjorner JB, et al. Developing a New Version of the SF-6D Health State Classification System From the SF-36v2: SF-6Dv2. Med Care. 2020;58(6):557–65.
University of Sheffield. SF-6D & SF-6Dv2—Calculating QALYs from the SF-36, SF-12 and the standalone SF-6Dv2. https://licensing.sheffield.ac.uk/product/SF-6D. Accessed 18 Jan 2024.
Quality Metric. SF-6D Health Utility. https://www.qualitymetric.com/health-surveys/sf-6d-health-utility-2/. Accessed 18 Jan 2024.
Medical Expenditure Panel Survey. 2018 Emergency Room Visits. Rockville: Agency for Healthcare Research and Quality Center for Financing, Access, and Cost Trends; 2020. MEPS HC-206E.
Medical Expenditure Panel Survey. 2018 Hospital Inpatient Stays. Rockville: Agency for Healthcare Research and Quality Center for Financing, Access, and Cost Trends; 2020. MEPS HC-206D.
Medical Expenditure Panel Survey. 2018 Outpatient Department Visits. Rockville: Agency for Healthcare Research and Quality Center for Financing, Access, and Cost Trends; 2020. MEPS HC-206F.
Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4(5):353–65.
United States Bureau of Labor Statistics, United States Department of Labor. Highlights of women’s earnings in 2018. Division of Information and Marketing Services; 2019. Report No 1083.
Hoveling LA, Liefbroer AC, Scweren LJS, Bültmann U. Socioeconomic differences in major depressive disorder onset among adults are partially explained by lifestyle factors: a longitudinal analysis of the Lifelines Cohort Study. J Affect Disord. 2022;314:309–17.
Cai J, Wei Z, Chen M, et al. Socioeconomic status, individual behaviors and risk for mental disorders: a Mendelian randomization study. Eur Psychiatry. 2022;65(1):e28.
Trivedi MH, Rush AJ, Wisniewski SR, et al. Factors associated with health-related quality of life among outpatients with major depressive disorder: a STAR*D report. J Clin Psychiatry. 2006;67(2):185–95.
Ishak WW, Balayan K, Bresee C, et al. A descriptive analysis of quality of life using patient-reported measures in major depressive disorder in a naturalistic outpatient setting. Qual Life Res. 2013;22(3):585–96.
Birnbaum HG, Kessler RC, Kelley D, Ben-Hamadi R, Joish VN, Greenberg PE. Employer burden of mild, moderate, and severe major depressive disorder: mental health services utilization and costs, and work performance. Depress Anxiety. 2010;27(1):78–89.
Centers for Disease Control and Prevention. Persons with hospital stays in the past year, by selected characteristics: United States, selected years 1997–2018. 2019. https://www.cdc.gov/nchs/data/hus/2019/040-508.pdf. Accessed 20 Dec 2023.
Arnaud A, Suthoff E, Tavares RM, Zhang X, Ravindranath AJ. The increasing economic burden with additional steps of pharmacotherapy in major depressive disorder. Pharmacoeconomics. 2021;39(6):691–706.
Nakajima S, Suzuki T, Watanabe K, Kashima H, Uchida H. Accelerating response to antidepressant treatment in depression: a review and clinical suggestions. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(2):259–64.
Medical Writing and Editorial Assistance
The authors would like to thank Amanda Harrington, PhD, and Sara Higa, PharmD, MS, formerly of AbbVie, for their contributions to this study. Writing and editorial assistance were provided to the authors by Samantha Watry, PharmD, and Susan Bartko-Winters, PhD, of Prescott Medical Communications Group (Chicago, IL) and funded by AbbVie.
Funding
AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship. AbbVie also funded the Rapid Service and Open Access fees.
Author information
Authors and Affiliations
Contributions
Larry Culpepper, Ashley Martin, Nadia Nabulsi, and Mousam Parikh all participated in the writing, editing, and critical revision for intellectual content, and the approval of the final version of this manuscript. All authors met ICMJE authorship criteria and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of Interest
Larry Culpepper has served as an advisor or consultant for AbbVie Pharmaceuticals, Eisai Pharmaceuticals, Takeda, and Supernus Pharmaceuticals; owns stock in M-3 Information, LLC, and has received royalties from UpToDate and Oxford University Press and receives payment from Physicians Postgraduate Press as Editor in Chief of the Primary Care Companion for CNS Diseases. Ashley Martin was an employee of Cerner Enviza at the time of this study, which was funded by AbbVie to perform the analysis. She is currently employed at BluePath Solutions (Los Angeles, CA). Nadia Nabulsi and Mousam Parikh are employees of AbbVie and may hold stock.
Ethical Approval
This study was exempt from Ethics Committee approval and institutional review because it is a retrospective analysis which used anonymized and de-identified data certified as fully compliant with US patient confidentiality requirements set forth in the Health Insurance Portability and Accountability Act of 1996. Allergan (prior to its acquisition by AbbVie) obtained permission to access and use the 2019 National Health and Wellness Survey data used in the analysis through licensing agreements.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Culpepper, L., Martin, A., Nabulsi, N. et al. The Humanistic and Economic Burden Associated with Major Depressive Disorder: A Retrospective Cross-Sectional Analysis. Adv Ther 41, 1860–1884 (2024). https://doi.org/10.1007/s12325-024-02817-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-024-02817-w