Abstract
Purpose of the Review
The purpose of this paper is to review the current knowledge regarding imaging inflammation in cardiac sarcoidosis (CS).
Recent Findings
Noninvasive imaging methods like cardiac magnetic resonance (CMR) and 18-fluorodeoxyglucose (18FDG) positron-emission tomography-computed tomography (PET-CT) scans have emerged as the most important modalities in diagnosing and monitor therapy efficacy in CS. The differentiation between an active and a chronic, silent clinical state is crucial for decision making. T1- and T2-mapping by CMR provide a unique opportunity to discriminate normal and diseased myocardium by detecting myocardial edema and fibrosis. While T1-mapping is sensitive to fibrosis and edema, T2 is primarily influenced by tissue water content. Increased 18-fluorodeoxyglucose (18FDG) uptake in positron-emission tomography-computed tomography (PET-CT) scan is a hallmark feature of CS and defines active inflammation. Therefore, the hybrid application of both CMR and 18FDG-PET-CT scans has evolved as the standard procedure to detect CS and monitor the therapeutic response.
Summary
Imaging inflammation in CS is basically performed by CMR T2-mapping and 18FDG uptake in PET-CT scan. Both are reliable methods in identifying active CS and follow up therapy and can be used synergistically. Additionally CMR provides further prognostic insights by late gadolinium enhancement that increases the risk for malignant arrhythmia and for relapse after therapy weaning.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sarcoidosis is a systemic inflammatory disease, and cardiac involvement is the most frequent cause of death from this condition [1]. While the majority of cases with systemic sarcoidosis show a self-limiting process that only affects the lung and the lymph nodes, cardiac involvement always requires high-dose immunosuppressive therapy [2]. About 5% of cases show symptomatic cardiac sarcoidosis (CS), but autopsy studies suggest that in up to 25% there is a silent cardiac involvement with unknown significance [3, 4].
Myocarditis is defined by an immigration of immune cells and activation of different cytokines, leading to myocyte necrosis and apoptosis as well as fibrosis of the surrounding interstitium [5,6,7]. Most frequently, myocarditis is triggered by viral infection with cardiotropic agents, which either directly induce myocyte death or effect a cytokine-mediated activation of the immune system [7, 8]. Moreover, systemic inflammatory diseases like sarcoidosis or rheumatological diseases (e.g. systemic lupus erythematosus, systemic sclerosis, and myositis) frequently involve the myocardium and provoke myocardial inflammation and remodeling processes [7,8,9,10]. The hallmark of CS is the formation of noncaseating granuloma and an overlap of acute, potentially reversible inflammation and irreversible fibrotic remodeling, which result in heart failure, arrhythmia, and sudden cardiac death [2, 11].
The multifarious origins of myocardial inflammation emphasize the importance of both detecting active inflammation and differentiating its etiology. However, a single, noninvasive imaging modality to differentiate these entities is still not available. In this review we focus on the importance of multimodal imaging to diagnose sarcoidosis. We provide information on the identification of active and chronic inflammation and illuminate the current state of the art for monitoring CS during medical therapy.
Multimodal Imaging to Identify Myocardial Inflammation
Sarcoidosis can affect almost every organ and has a different clinical presentation, from asymptomatic incidental findings to sudden cardiac death. The diagnosis of isolated CS can be challenging because of the unspecific clinical presentation [12]. As of today some diagnostic recommendations still require histological confirmation [13, 14]. However, the focal character of the disease results in a high sampling error, so that CS could be missed without adding other diagnostic modalities [12, 15]. Therefore, the Japanese Circulation Society (JCS) guidelines and the Heart Rhythm Society (HRS) consensus paper raised the importance of noninvasive imaging tools like cardiac magnetic resonance (CMR) and 18-fluorodeoxyglucose (18FDG) positron-emission tomography-computed tomography (PET-CT) [13, 14]. According to the HRS guidelines, however, a definite diagnosis of CS is only possible with extracardiac histopathological proof of noncaseating granuloma [13].
CMR
The renewed European Society of Cardiology (ESC) guidelines raised CMR to a level I recommendation in the differential diagnosis of cardiomyopathies and heart failure [16]. In cases of suspected CS, CMR provides the possibility to identify structural changes like regional aneurysms, regional or global fibrosis, and myocardial edema and overcomes the sampling error of endomyocardial biopsy (EMB). Further, CMR has better diagnostic power than traditional diagnostic tools like electrocardiography and echocardiography [17•]. The updated Lake Louise Criteria (LLC) from 2018 require the presence of increased native T1 or late gadolinium enhancement (LGE) as indicative of myocardial damage plus increased T2 or pathological T2-weighted images to detect active inflammation [16, 18]. In a meta-analysis of 33 studies with 1997 patients, including 687 with CS, Aitken et al. found a higher sensitivity (95% vs. 84%, p = 0.002) of CMR compared with 18FDG PET-CT but no difference in specificity [19].
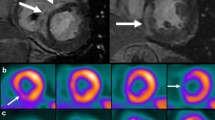
There are some indicative imaging features of CS, including basal septum aneurysms, but CS can also mimic other cardiomyopathies such as arrhythmogenic ventricular dysplasia, ischemic heart disease (ICM), myocarditis, giant cell myocarditis (GCM), or even amyloidosis [20,21,22, 23•, 24]. In particular, the presentation of GCM and CS can be similar in CMR. In a group of biopsy-confirmed GCM and CS patients, Bibbio et al. found no difference in CMR appearance for these two entities, whereas the clinical presentation for GCM was more acute [25]. Even after matching for ejection fraction (EF), sex, and age there were no relevant differences aside from soft morphological features [23•]. Figure 1 shows a case where CS mimics progressed ICM. Based on the pathological angiogram the patient was recommended for coronary artery bypass surgery. The diffuse, partly transmural and subendocardial distribution and the perfusion defect in connection with the angiogram led to the wrong diagnosis of ICM. The lack of clinical improvement after revascularization, recurrent malignant ventricular arrhythmia (VA), and graft failure led to questioning of the initial diagnosis.
A patient with cardiac sarcoidosis was referred for coronary artery bypass surgery because of progressed coronary three-vessel disease. External cardiovascular magnetic resonance yielded diffuse late gadolinium enhancement (a, b) in nearly all supply areas and ventricular thrombus (white arrow). (c) Perfusion imaging showed diffuse perfusion deficit in first pass, leading to the misdiagnosis of ischemic cardiomyopathy (*). Coronary angiography with multiple stenotic lesions in all 3 coronary vessels (d-f)
In the following section we discuss data on the different CMR techniques that are part of the LLC and their relative diagnostic merit.
Late Gadolinium Enhancement
Focal changes in myocardial architecture like replacement fibrosis, inflammation, and any form of extracellular expansion can cause LGE in CMR [26]. CS can present as any other cardiomyopathy or inflammatory heart disease in LGE. The LGE pattern most suspect for CS is an intense, partially mass-like subepicardial or midventricular distribution predominantly affecting the septum; however, there can be any other form mimicking an infarct-like lesion or just a discreet focal finding [27]. Therefore, LGE alone cannot confirm the diagnosis of CS. Figure 2 shows three different typical presentations of CS in LGE from our cohort. However the majority of cases the LGE pattern is quite characteristic.
Three presentations of cardiac sarcoidosis in late gadolinium enhancement. Typical mass-like distribution with right ventricular involvement in the short-axis (a, white arrow) and 4-chamber view (b) with basal septum aneurysm and irreversible replacement fibrosis (*). Weak gadolinium enhancement with complete remission after immunosuppressive therapy in the short-axis view (c, white star)
The presence of LGE indicates myocardial damage but does not inform about current inflammatory activity. There is no single LGE criterion that can distinguish between active focal inflammation or a chronic irreversible process. LGE can appear in the acute phase due to necrosis and edema but also in the chronic setting by replacement fibrosis. However, LGE can provide essential prognostic information. The amount of LGE is one of the most important prognosticators for death or VA in CS [28,29,30,31]. In a meta-analysis of 13 studies with 1318 patients, Stevenson et al. demonstrated that patients with LGE showed a significantly higher risk of VA and all-cause mortality [32••]. In a group of 50 patients who underwent CMR and fasting 18FDG PET-CT, Gowani et al. even showed that LGE was a better prognosticator for VA than 18FDG uptake in PET-CT scans [33•]. Pathological 18FDG uptake yields no additional information to help predict VA [33•]. This is very plausible, as LGE delineates the arrhythmogenic substrate, similar to what it shows in ICM.
CS frequently involves the right ventricle (RV) [34]. A simple but relevant advantage of CMR over echocardiography is the possibility to display the RV in three dimensions. CMR provides a functional, anatomical, and LGE assessment of the whole RV wall. Velangi et al. found reduced RV function in 12.1% and RV LGE in 5.5% with different prognostic implications. RV dysfunction is a good predictor of all-cause-mortality, whereas RV LGE is predictive of VA and sudden cardiac death (SCD) [34]. In a meta-analysis of 8 studies analyzing the influence of RV EF and RV LGE on outcome in a total of 899 patients, Wang et al. found both RVEF and RV LGE are predictive of adverse outcome, and, further, RV LGE is helpful in identifying patients at risk of SCD [35•].
Tissue Characterization by T1 and T2 Mapping and ECV
T1 and T2 mapping are excellent methods of tissue characterization as they provide both diagnostic and prognostic information [36,37,38]. While native T1 time is sensitive to fibrosis and water, T2 time is an indicator of myocardial edema [36, 39,40,41,42]. An excellent review article by Haaf et al. summarizes the influence of different histological changes in acute myocarditis and in cases of storage diseases on the T1 time. Almost any change in myocardial architecture leads to a prolongation of T1 time. Even deposits of glycosphingolipids and iron in Fabry disease and hemochromatosis result in shortened T1 time [37]. T1 and T2 times are early indictors of heart disease and can be increased before functional limitations of the ventricle or focal fibrosis become evident [36, 43]. Elevated T1 and T2 relaxation time in patients with known extracardiac sarcoidosis also seem to predict an early cardiac involvement [44••, 45,46,47]. In 53 biopsy-confirmed CS patients Puntman et al. found higher T1 and T2 times in addition to structural and functional changes compared with healthy controls and irrespective of symptoms, time since diagnosis, and age [44••]. Even in the absence of LGE and preserved EF patients with sarcoidosis show higher T1 values than healthy controls [48]. Both studies conclude that T1 mapping is the best parameter to differentiate between normal and diseased myocardium [44••, 48]. We recently found that the quantification of regional extracellular volume (ECV) by a combination of pre- and post-contrast T1 mapping and the hematocrit can distinguish CS from other inflammatory cardiomyopathy [49]. In 33 LGE- and PET-positive patients ECV was more specific than regional or global native T1 and T2 mapping alone. Further, in our cohort EMB did not yield any histological evidence of CS [49]. Nevertheless, the effect of fibrosis or myocardial edema on the T1 prolongation cannot be discriminated.
Detection of edema is of particular importance for the identification of active inflammation. This can be achieved in two different ways: as T2 mapping is specifically sensitive to the water content of the tissue, either increased signal intensity is delineated on T2w turbo spin echo sequences, ideally in relation to the signal of skeletal muscle, or the T2 time is measured directly by parametric techniques. T2 can be considered as a physical biomarker: the higher the T2 time, the higher the water content of the tissue [50, 51]. T2 mapping sequences are a quantitative method and have been shown to be superior to T2-weighted turbo spin echo sequences for the detection of inflammation [41]. Spieker et al. found that elevated T2 time is associated with the presence of inflammatory cells in EMB in dilated cardiomyopathy [41]. By adding T2 mapping to the JCS recommendations, the diagnostic accuracy for active CS can be improved [52••]. Although studies addressing T2 mapping as a follow-up parameter during treatment of CS are sparse, the importance of this parameter has been demonstrated for other inflammatory heart diseases [53, 54]. In a small case series of 8 patients, including 6 undergoing immunosuppressive therapy, Crouser et al. showed alterations in the T2 signal during follow-up, which supports the reproducibility of this parameter [55]. Therefore, we recommend both T1 and T2 mapping for diagnostic considerations. Selectively elevated T1 time and normal T2 time indicate fibrotic remodeling, whereas an elevation of both parameters is suggestive of myocardial inflammation.
In summary, CMR provides an opportunity for detailed characterization of tissues of the whole myocardium that overcomes the issue of sampling error of EMB. T1 and T2 mapping are excellent methods for the discrimination of diseased and normal myocardium, the identification of remodeling, and even detection of subtle inflammation. T2 mapping has been identified as a suitable parameter for monitoring progression in many inflammatory diseases such as myocarditis or systemic sclerosis and is therefore an essential component in monitoring immunosuppressive therapy of CS patients. LGE adds insights regarding the underlying process of different cardiomyopathies and adds prognostic information to the diagnostic data. However, there is room for improvements, particularly for device-implanted patients who represent not an inconsiderable proportion. In the future, wideband sequences could overcome the problem of off-resonance artefacts and could make the modality suitable for this particular cohort.
18FDG PET-CT
A high glucose metabolism is a hallmark of inflammatory processes and sarcoid granuloma. Macrophages show high metabolic activity and therefore high 18FDG uptake. 18FDG PET-CT uses this feature to visualize pathological 18FDG uptake not just in the heart but in all potentially affected organs and to confirm active myocardial inflammation [56]. To suppress the physiological glucose metabolism in the heart, a fasting period is necessary to improve diagnostic accuracy [57, 58]. Further, coronary artery disease needs to be excluded because ischemia can also result in abnormal 18FDG uptake. At our center we use a fasting period of 18 h with a last meal that is low in carbohydrates. Before 18FDG application all patients are prepared with 50 I.E./kg bodyweight heparin to suppress the tracer uptake. In a meta-analysis of 7 studies with 164 patients the accuracy of 18FDG PET-CT for detecting CS was determined to have a sensitivity of 89% and a specificity of 78% [59]. All 18FDG PET-CT scans should be interpreted qualitatively and quantitatively by an experienced examiner [60]. Quantitative parameters like the maximum standard uptake value (SUVmax) can improve the assessment of therapy response, but they are not validated for prognostic information [61, 62]. However, qualitative PET-CT findings appear to add a prognostic value. Pathological tracer uptake is associated with a higher risk of VA or death [63].
A combination of CMR and fasting PET-CT scans is a widely used approach to confirm CS and to discriminate an acute from a chronic condition [49, 64]. In a recently published study Greulich et al. highlighted the role of a hybrid method of CMR and 18FDG PET-CT for the differentiation of active and chronic CS [65••]. In a cohort of 43 patients with histologically confirmed extracardiac sarcoidosis the authors found that a combined approach of CMR mapping method and PET-CT scan can increase the accuracy for diagnosing active CS. They showed that even in the absence of LGE, increased T1 and T2 mapping values can indicate active inflammation that can be confirmed by PET-CT with crucial therapeutic implications [65••]. 18FDG uptake in the atria can be a predictor of the development of atrial fibrillation during follow-up. In a multimodal approach in 118 CS patients without atrial fibrillation at initial diagnosis, Niemelä et al. found atrial inflammation and atrial enlargement as independent predictors of future atrial fibrillation [66•].
Figure 3 shows a comparison of CMR and PET-CT findings in a young male patient presenting with sudden cardiac death and provides an example of the additive information that can be obtained from multimodal imaging CS of myocardial inflammation.
Multimodal imaging in a 32-year-old patient presenting with sudden cardiac death during a soccer match. a 4-chamber and (b) short-axis view with diffuse late gadolinium enhancement distribution and right ventricular involvement (white arrow). Only global T1 values (c) were mildly elevated. T2 values (d) were neither globally nor regionally increased. 18-fluorodeoxyglucose (18FDG) positron-emission tomography-computed tomography (PET-CT) shows 18FDG uptake extended far beyond LGE-positive regions (e, f)
To summarize, 18FDG PET-CT scans are suitable for the identification of active inflammation but provide limited information about chronic irreversible disease status. 18FDG PET-CT is not able to reliably detect fibrotic remodeling areas whose presence is a significant prognosticator. The preparation of the patient for 18FDG PET-CT can affect the results and the comparability of the examinations [58], but to date there are no precise recommendations for the best dietary approach to optimize the scans. We recommend a controlled fasting period within an inpatient stay to ensure the suppression of physiological glucose uptake and to avoid false positive results. Furthermore, a lack of 18FDG-uptake only indicates the absence of active inflammation; it does not rule out chronic fibrotic remodeling in healed CS which is still associated with a relevant increased risk for malignant VA and SCD. Therefore, both methods – PET and CMR – are indispensable for the identification of CS and the assessment of activity status.
Monitoring Inflammation during Immunosuppressive Therapy in CS
CS always requires high-dose immunosuppressive therapy with glucocorticoids and optionally methotrexate or azathioprine as well as standard heart failure therapy [67]. As concerns still exist about the optimal dose and period of immunosuppressive therapy, many centers have their own follow-up regimen, which often makes comparability between studies difficult. CS is often characterized clinically by high-grade AV-block and malignant VA. Therefore, many patients are provided with a pacemaker or defibrillator. The electronic device influences in particular the image quality of CMR or even makes its application impossible. In such cases 18FDG PET-CT is an important alternative, especially for detecting disease recurrence under therapy [60, 68]. A reduction of 18FDG uptake seems to be associated with an improvement of EF [69]. The optimal timepoint for imaging follow-up is still a matter of debate, but a period of more than 5 months seems to be enough to reflect medically induced changes [70]. In a cohort of 68 patients Arps et al. investigated imaging parameters for the detection of a relapse [71•]. They found that about 50% of patients suffered disease recurrence, and the initial amount of LGE and isolated CS were relevant predictors.
In our center we use a multimodal approach consisting of clinical parameters, laboratory findings, and imaging modalities. In cases of suspected CS in CMR imaging, a fasting 18FDG PET-CT scan is always obtained to quantify disease activity status. As a follow-up tool we prefer CMR, if applicable. CMR does not entail exposure to radiation, which makes a serial application feasible. For interpretation of inflammation, we use the development of global and regional T1 and T2 mapping values and the amount of LGE. For the assessment of heart failure, we rely on functional and morphological parameters like EF and end-diastolic and end-systolic volumes. In cases having inconclusive CMR results, or if CMR is inapplicable or leaves any doubt about CS activity status, we add 18FDG PET-CT for further decision making. The imaging information is always put into context together with all collected parameters, and every case is discussed in an interdisciplinary conference for rare heart diseases before introduction, change, or termination of therapy. Figures 4 and 5 depict two cases in which CMR and 18FDG PET-CT were used to confirm treatment response.
Multimodal imaging in a 32-year-old patient presenting with sudden cardiac death during a soccer match. a 4-chamber and (b) short-axis view with diffuse late gadolinium enhancement distribution and right ventricular involvement (white arrow). Only global T1 values (c) were mildly elevated. T2 values (d) were neither globally nor regionally increased. 18-fluorodeoxyglucose (18FDG) positron-emission tomography-computed tomography (PET-CT) shows 18FDG uptake extended far beyond LGE-positive regions (e, f)
40-year old male patient with elevated cardiac markers, conduction disturbance and atypical pectangina. a and (b) with regional significantly elevated T2- (upper limit 40 ms) and T1-values (upper limit 1171 ms) and (c) dull late gadolinum enhancement (LGE, white arrow). cardiac sarcoidosis was confirmed by 18-fluorodeoxyglucose (18FDG) positron-emission tomography-computed tomography (PET-CT) (d-f) show normalized relaxationtimes and complete remission of LGE 6 months under therapy. No additional PET-CT scan was necessary. Still conduction disturbance was persistent
Perspectives for Imaging for Therapy Monitoring
Serial CMR is a reliable and sensitive tool to monitor the course of inflammation during therapy and avoids radiation exposure. However, in patients with implanted devices 18FDG PET-CT is indispensable. In serial 18FDG PET-CT a change of at least 20% in uptake regions and/or SUVmax are the recommended threshold for indicating therapy response [72]. Alterations of SUVmax in serial measurements do not correlate with prognosis and clinical outcome.
There are still gaps in our knowledge concerning the best therapeutic approach, the optimal corticosteroid dose and therapy period, follow-up imaging settings and period, and indicators for escalating or deescalating immunosuppressive therapy. As a consequence, heart centers use their own strategy, which makes studies difficult to compare and to translate to clinical practice. An expert consensus-based, standardized follow-up approach that includes clinical and imaging parameters should be implemented, and multicenter studies will be necessary to significantly improve the management of the rare but life-threatening disease of CS.
Conclusion
Due to the multifaceted clinical appearance and imaging features of CS, a multimodal approach for diagnosis and follow-up is mandatory. According to the current state of knowledge, a combination of CMR and fasting 18FDG PET-CT is the favorable method for the diagnosis and identification of active inflammation. T1 and T2 mapping sequences provide a unique opportunity to quantify and track fibrosis and inflammation before any structural changes are observable. In addition, 18FDG PET-CT detects metabolically active areas, which is a fundamental feature in inflammatory processes. Finally, the evaluation of CS always requires an interdisciplinary approach for best decision making.
Abbreviations
- CMR:
-
Cardiac magnetic resonance
- CS:
-
Cardiac sarcoidosis
- ECV:
-
Extracellular volume
- EF:
-
Ejection fraction
- EMB:
-
Endomyocardial biopsy
- 18FDG:
-
18-Fluorodeoxyglucose
- GCM:
-
Giant cell myocarditis
- HRS:
-
Heart Rhythm Society
- ICM:
-
Ischemic heart disease
- JCS:
-
Japanese Circulation Society
- LGE:
-
Late gadolinium enhancement
- LV:
-
Left ventricle
- PET-CT:
-
Positron-emission tomography-computed tomography
- RV:
-
Right ventricle
- SCD:
-
Sudden cardiac death
- VA:
-
Ventricular arrhythmia
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ekström K, Lehtonen J, Nordenswan H-K, et al. Sudden death in cardiac sarcoidosis: an analysis of nationwide clinical and cause-of-death registries. Eur Heart J. 2019;40:3121–8. https://doi.org/10.1093/eurheartj/ehz428.
Birnie DH, Nery PB, Ha AC, Beanlands RSB. Cardiac Sarcoidosis. J Am Coll Cardiol. 2016;68:411–21. https://doi.org/10.1016/j.jacc.2016.03.605.
Patel MR, Cawley PJ, Heitner JF, et al. Detection of Myocardial Damage in Patients with Sarcoidosis. Circulation. 1969;2009:120. https://doi.org/10.1161/CIRCULATIONAHA.109.851352.
Perry A, Vuitch F. Causes of death in patients with sarcoidosis. A morphologic study of 38 autopsies with clinicopathologic correlations. Arch Pathol Lab Med. 1995;119:167–72.
Tschöpe C, Cooper LT, Torre-Amione G, Van Linthout S. Management of Myocarditis-Related Cardiomyopathy in Adults. Circ Res. 2019;124:1568–83. https://doi.org/10.1161/CIRCRESAHA.118.313578.
Leone O, Veinot JP, Angelini A, et al. 2011 consensus statement on endomyocardial biopsy from the Association for European Cardiovascular Pathology and the Society for Cardiovascular Pathology. Cardiovasc Pathol. 2012;21:245–74. https://doi.org/10.1016/j.carpath.2011.10.001.
Caforio ALP, Pankuweit S, Arbustini E, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34:2636–48. https://doi.org/10.1093/eurheartj/eht210.
Tschöpe C, Ammirati E, Bozkurt B, et al. Myocarditis and inflammatory cardiomyopathy: current evidence and future directions. Nat Rev Cardiol. 2021;18:169–93.
Krejci J, Mlejnek D, Sochorova D, Nemec P. Inflammatory Cardiomyopathy: A Current View on the Pathophysiology, Diagnosis, and Treatment. BioMed Res Int. 2016:1–11. https://doi.org/10.1155/2016/4087632.
Korosoglou G, Giusca S, Montenbruck M, et al. Fast Strain-Encoded Cardiac Magnetic Resonance for Diagnostic Classification and Risk Stratification of Heart Failure Patients. JACC Cardiovasc Imaging. 2021;14:1177–88. https://doi.org/10.1016/j.jcmg.2020.10.024.
Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007;357:2153–65. https://doi.org/10.1056/NEJMra071714.
Okada DR, Bravo PE, Vita T, et al. Isolated cardiac sarcoidosis: A focused review of an under-recognized entity. J Nucl Cardiol. 2018;25:1136–46. https://doi.org/10.1007/s12350-016-0658-1.
Birnie DH, Sauer WH, Bogun F, et al. HRS Expert Consensus Statement on the Diagnosis and Management of Arrhythmias Associated With Cardiac Sarcoidosis. Heart Rhythm. 2014;11:1304–23. https://doi.org/10.1016/j.hrthm.2014.03.043.
Terasaki F, Azuma A, Anzai T, et al. JCS 2016 Guideline on Diagnosis and Treatment of Cardiac Sarcoidosis. Circ J. 2019;83:2329–88. https://doi.org/10.1253/circj.CJ-19-0508.
Kusano KF, Satomi K. Diagnosis and treatment of cardiac sarcoidosis. Heart (British Cardiac Society). 2016;102:184–90. https://doi.org/10.1136/heartjnl-2015-307877.
McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599–726. https://doi.org/10.1093/eurheartj/ehab368.
• Kouranos V, Tzelepis GE, Rapti A, et al. Complementary Role of CMR to Conventional Screening in the Diagnosis and Prognosis of Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2017;10:1437–47. https://doi.org/10.1016/j.jcmg.2016.11.019. This paper demonstrates, that CMR is the most valuable diagnostic test compared to electrocardiography and echogardiography to diagnose CS and provides prognostic information.
Ferreira VM, Schulz-Menger J, Holmvang G, et al. Cardiovascular Magnetic Resonance in Nonischemic Myocardial Inflammation: Expert Recommendations. J Am College Cardiol. 2018:72. https://doi.org/10.1016/J.JACC.2018.09.072.
Aitken M, Chan MV, Urzua Fresno C, et al. Diagnostic Accuracy of Cardiac MRI versus FDG PET for Cardiac Sarcoidosis: A Systematic Review and Meta-Analysis. Radiology. 2022;304:566–79. https://doi.org/10.1148/radiol.213170.
Waki H, Eguchi K, Toriumi S, et al. Isolated Cardiac Sarcoidosis Mimicking Arrhythmogenic Right Ventricular Cardiomyopathy. Int Med (Tokyo, Japan). 2018;57:835–9. https://doi.org/10.2169/internalmedicine.9395-17.
Narula N, Iannuzzi M. Sarcoidosis: Pitfalls and Challenging Mimickers. Front Med. 2020;7:594275. https://doi.org/10.3389/fmed.2020.594275.
Oyama-Suzuki K, Fukushima K, Egi R, et al. Cardiac Sarcoidosis Mimicking Anterior Myocardial Infarction. Ann Nucl Cardiol. 2021;7:73–6. https://doi.org/10.17996/anc.21-00136.
• Pöyhönen P, Nordenswan H-K, Lehtonen J, et al. Cardiac magnetic resonance in giant cell myocarditis: a matched comparison with cardiac sarcoidosis. Eur Heart J - Cardiovasc Imaging. 2023;24:404–12. https://doi.org/10.1093/ehjci/jeac265. This paper indicates that CS can mimic other cardiac diseases espacially giant cell myocarditis.
Takemura K, Nakamura R, Shimazu K, et al. A case of cardiac sarcoidosis mimicking cardiac amyloidosis on cardiovascular magnetic resonance. ESC Heart Fail. 2018;5:306–10. https://doi.org/10.1002/ehf2.12263.
Bobbio E, Bollano E, Oldfors A, et al. Phenotyping of giant cell myocarditis versus cardiac sarcoidosis using cardiovascular magnetic resonance. Int J Cardiol. 2023;387:131143. https://doi.org/10.1016/j.ijcard.2023.131143.
Mahrholdt H, Wagner A, Judd RM, et al. Delayed enhancement cardiovascular magnetic resonance assessment of non-ischaemic cardiomyopathies. Eur Heart J. 2005;26:1461–74. https://doi.org/10.1093/eurheartj/ehi258.
Smedema J-P, Snoep G, van Kroonenburgh MPG, et al. Evaluation of the Accuracy of Gadolinium-Enhanced Cardiovascular Magnetic Resonance in the Diagnosis of Cardiac Sarcoidosis. J Am Coll Cardiol. 2005;45:1683–90. https://doi.org/10.1016/j.jacc.2005.01.047.
Flamée L, Symons R, Degtiarova G, et al. Prognostic value of cardiovascular magnetic resonance in patients with biopsy-proven systemic sarcoidosis. Eur Radiol. 2020;30:3702–10. https://doi.org/10.1007/s00330-020-06765-1.
Greulich S, Deluigi CC, Gloekler S, et al. CMR imaging predicts death and other adverse events in suspected cardiac sarcoidosis. JACC Cardiovasc Imaging. 2013;6:501–11. https://doi.org/10.1016/j.jcmg.2012.10.021.
Agoston-Coldea L, Kouaho S, Sacre K, et al. High mass (>18g) of late gadolinium enhancement on CMR imaging is associated with major cardiac events on long-term outcome in patients with biopsy-proven extracardiac sarcoidosis. Int J Cardiol. 2016;222:950–6. https://doi.org/10.1016/j.ijcard.2016.07.233.
Magnocavallo M, Vetta G, Polselli M, et al. “Function follows form”: Role of cardiac magnetic resonance for ventricular arrhythmia risk stratification in patients with cardiac sarcoidosis. J Cardiovasc Electrophysiol. 2023;34:1781–4. https://doi.org/10.1111/jce.16020.
•• Stevenson A, Bray JJH, Tregidgo L, et al. Prognostic Value of Late Gadolinium Enhancement Detected on Cardiac Magnetic Resonance in Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2023;16:345–57. https://doi.org/10.1016/j.jcmg.2022.10.018. In this meta-analysis of Stevenson et al. late gadolinum enhancement results in increased odds of ventricular arrhythmia and all-cause mortality with additional prognostic information in case of biventricular involvement.
• Gowani Z, Habibi M, Okada DR, et al. Utility of Cardiac Magnetic Resonance Imaging Versus Cardiac Positron Emission Tomography for Risk Stratification for Ventricular Arrhythmias in Patients With Cardiac Sarcoidosis. Am J Cardiol. 2020;134:123–9. https://doi.org/10.1016/j.amjcard.2020.08.007. This work showed that late gadolinum enhancement is one of the most important features to predict ventricular arrhythmia and is superior to inflammation in 1818FDG PET-CT.
Velangi PS, Chen K-HA, Kazmirczak F, et al. Right Ventricular Abnormalities on Cardiovascular Magnetic Resonance Imaging in Patients With Sarcoidosis. JACC Cardiovasc Imaging. 2020;13:1395–405. https://doi.org/10.1016/J.JCMG.2019.12.011.
• Wang J, Zhang J, Hosadurg N, et al. Prognostic Value of RV Abnormalities on CMR in Patients With Known or Suspected Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2023;16:361–72. https://doi.org/10.1016/j.jcmg.2022.11.012. This work highlights the prognostic importance to monitor and assess RV abnormalities
Moon JC, Messroghli DR, Kellman P, et al. Myocardial T1 mapping and extracellular volume quantification: a Society for Cardiovascular Magnetic Resonance (SCMR) and CMR Working Group of the European Society of Cardiology consensus statement. J Cardiovasc Magn Reson. 2013;15:92. https://doi.org/10.1186/1532-429X-15-92.
Haaf P, Garg P, Messroghli DR, et al. Cardiac T1 Mapping and Extracellular Volume (ECV) in clinical practice: a comprehensive review. J Cardiovasc Magn Reson. 2016;18:89. https://doi.org/10.1186/s12968-016-0308-4.
Messroghli DR, Moon JC, Ferreira VM, et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imagi. J Cardiovasc Magn Reson. 2017;19:75. https://doi.org/10.1186/s12968-017-0389-8.
Greulich S, Ferreira VM, Dall’Armellina E, Mahrholdt H. Myocardial Inflammation-Are We There Yet? Curr Cardiovasc Imaging Rep. 2015;8:6. https://doi.org/10.1007/s12410-015-9320-6.
O’Brien AT, Gil KE, Varghese J, et al. T2 mapping in myocardial disease: a comprehensive review. J Cardiovasc Magn Reson. 2022;24:33. https://doi.org/10.1186/s12968-022-00866-0.
Spieker M, Katsianos E, Gastl M, et al. T2 mapping cardiovascular magnetic resonance identifies the presence of myocardial inflammation in patients with dilated cardiomyopathy as compared to endomyocardial biopsy. Eur Heart J - Cardiovasc Imaging. 2018;19:574–82. https://doi.org/10.1093/ehjci/jex230.
Pezel T, Viallon M, Croisille P, et al. Imaging Interstitial Fibrosis, Left Ventricular Remodeling, and Function in Stage A and B Heart Failure. JACC Cardiovasc Imaging. 2021;14:1038–52. https://doi.org/10.1016/j.jcmg.2020.05.036.
Moustafa A, Khan MS, Alsamman MA, et al. Prognostic significance of T1 mapping parameters in heart failure with preserved ejection fraction: a systematic review. Heart Fail Rev. 2021;26:1325–31. https://doi.org/10.1007/s10741-020-09958-4.
•• Puntmann VO, Isted A, Hinojar R, et al. T1 and T2 Mapping in Recognition of Early Cardiac Involvement in Systemic Sarcoidosis. Radiology. 2017;285:63–72. https://doi.org/10.1148/radiol.2017162732. In this work of Puntmann et al. the diagnostic importance of tissue characterization by T1- and T2-Mapping in early disease stages is highlighted.
Greulich S, Kitterer D, Latus J, et al. Comprehensive Cardiovascular Magnetic Resonance Assessment in Patients with Sarcoidosis and Preserved Left Ventricular Ejection Fraction. Circulation: Cardiovasc Imaging 2016:9. https://doi.org/10.1161/CIRCIMAGING.116.005022.
Crouser ED, Ono C, Tran T, et al. Improved Detection of Cardiac Sarcoidosis Using Magnetic Resonance with Myocardial T2 Mapping. Am J Respir Crit Care Med. 2014;189:109–12. https://doi.org/10.1164/rccm.201309-1668LE.
Yang Y, Zavodni A, Roifman I, et al. Myocardial extracellular space volume in patients with systemic sarcoidosis: quantitative measurement using a T1 mapping technique. J Cardiovasc Magn Reson. 2014;16:P279. https://doi.org/10.1186/1532-429X-16-S1-P279.
Greulich S, Mahrholdt H. Prognosis of Myocardial Damage in Sarcoidosis Patients With Preserved Left Ventricular Ejection Fraction: Always Look at the Bright Side of Cardiovascular Magnetic Resonance. Circ Cardiovasc Imaging. 2016;9:e004417. https://doi.org/10.1161/CIRCIMAGING.115.004417.
Treiber J, Novak D, Fischer-Rasokat U, et al. Regional extracellular volume within late gadolinium enhancement-positive myocardium to differentiate cardiac sarcoidosis from myocarditis of other etiology: a cardiovascular magnetic resonance study. J Cardiovasc Magn Reson. 2023;25:8. https://doi.org/10.1186/s12968-023-00918-z.
Bohnen S, Radunski UK, Lund GK, et al. Tissue characterization by T1 and T2 mapping cardiovascular magnetic resonance imaging to monitor myocardial inflammation in healing myocarditis. Eur Heart J Cardiovasc Imaging. 2017;18. https://doi.org/10.1093/EHJCI/JEX007.
Lota AS, Gatehouse PD, Mohiaddin RH. T2 mapping and T2* imaging in heart failure. Heart Fail Rev. 2017;22:431. https://doi.org/10.1007/S10741-017-9616-5.
•• Chamberlin JH, Kocher MR, Aquino G, et al. Quantitative myocardial T2 mapping adds value to Japanese circulation society diagnostic criteria for active cardiac sarcoidosis. Int J Cardiovasc Imaging. 2023;1–12. https://doi.org/10.1007/s10554-023-02863-5. This group found that incorporating T2-mapping into the diagnostic process increases the accuracy of identifying active inflammation when compared to relying solely on the JCS diagnostic criteria.
Lurz P, Eitel I, Adam J, et al. Diagnostic performance of CMR imaging compared with EMB in patients with suspected myocarditis. JACC Cardiovasc Imaging. 2012;5:513–24. https://doi.org/10.1016/j.jcmg.2011.11.022.
Panopoulos S, Mavrogeni S, Vlachopoulos C, Sfikakis PP. Cardiac magnetic resonance imaging before and after therapeutic interventions for systemic sclerosis-associated myocarditis. Rheumatology (Oxford, England). 2023;62:1535–42. https://doi.org/10.1093/rheumatology/keac504.
Crouser ED, Ruden E, Julian MW, Raman SV. Resolution of abnormal cardiac MRI T2 signal following immune suppression for cardiac sarcoidosis. J Investig Med. 2016;64:1148–50. https://doi.org/10.1136/jim-2016-000144.
Slart RHJA, Glaudemans AWJM, Lancellotti P, et al. A joint procedural position statement on imaging in cardiac sarcoidosis: from the Cardiovascular and Inflammation & Infection Committees of the European Association of Nuclear Medicine, the European Association of Cardiovascular Imaging, and the American Society of Nuclear Cardiology. J Nucl Cardiol. 2018;25:298–319. https://doi.org/10.1007/s12350-017-1043-4.
Manabe O, Yoshinaga K, Ohira H, et al. The effects of 18-h fasting with low-carbohydrate diet preparation on suppressed physiological myocardial 18F-fluorodeoxyglucose (FDG) uptake and possible minimal effects of unfractionated heparin use in patients with suspected cardiac involvement sarcoidosis. J Nucl Cardiol. 2016;23:244–52. https://doi.org/10.1007/s12350-015-0226-0.
Atterton-Evans V, Turner J, Vivanti A, Robertson T. Variances of dietary preparation for suppression of physiological 18F-FDG myocardial uptake in the presence of cardiac sarcoidosis: A systematic review. J Nucl Cardiol. 2020;27:481–9. https://doi.org/10.1007/s12350-018-1379-4.
Youssef G, Leung E, Mylonas I, et al. The use of 18F-FDG PET in the diagnosis of cardiac sarcoidosis: a systematic review and metaanalysis including the Ontario experience. J Nucl Med. 2012;53:241–8. https://doi.org/10.2967/jnumed.111.090662.
Chareonthaitawee P, Beanlands RS, Chen W, et al. Joint SNMMI–ASNC expert consensus document on the role of 18F-FDG PET/CT in cardiac sarcoid detection and therapy monitoring. J Nucl Cardiol. 2017;24:1741–58. https://doi.org/10.1007/s12350-017-0978-9.
Ahmadian A, Pawar S, Govender P, et al. The response of FDG uptake to immunosuppressive treatment on FDG PET/CT imaging for cardiac sarcoidosis. J Nucl Cardiol. 2017;24:413–24. https://doi.org/10.1007/s12350-016-0490-7.
Ahmadian A, Brogan A, Berman J, et al. Quantitative interpretation of FDG PET/CT with myocardial perfusion imaging increases diagnostic information in the evaluation of cardiac sarcoidosis. J Nucl Cardiol. 2014;21:925–39. https://doi.org/10.1007/s12350-014-9901-9.
Blankstein R, Osborne M, Naya M, et al. Cardiac positron emission tomography enhances prognostic assessments of patients with suspected cardiac sarcoidosis. J Am Coll Cardiol. 2014;63:329–36. https://doi.org/10.1016/j.jacc.2013.09.022.
Dweck MR, Abgral R, Trivieri MG, et al. Hybrid Magnetic Resonance Imaging and Positron Emission Tomography With Fluorodeoxyglucose to Diagnose Active Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2018;11:94–107. https://doi.org/10.1016/j.jcmg.2017.02.021.
•• Greulich S, Gatidis S, Gräni C, et al. Hybrid Cardiac Magnetic Resonance/Fluorodeoxyglucose Positron Emission Tomography to Differentiate Active From Chronic Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2022;15:445–56. https://doi.org/10.1016/J.JCMG.2021.08.018. The hybride approach of CMR and PET-CT provides additional information for the detection of active disease status.
• Niemelä M, Uusitalo V, Pöyhönen P, et al. Incidence and Predictors of Atrial Fibrillation in Cardiac Sarcoidosis: A Multimodality Imaging Study. JACC Cardiovasc Imaging. 2022;15:1622–31. https://doi.org/10.1016/j.jcmg.2022.02.025. The group has identified that patients with atrial inflammation detected through PET-CT scans are at risk of developing atrial fibrillation in the future.
Stievenart J, Le Guenno G, Ruivard M, et al. Cardiac sarcoidosis: systematic review of the literature on corticosteroid and immunosuppressive therapies. Eur Respir J. 2022:59. https://doi.org/10.1183/13993003.00449-2021.
Ning N, Guo HH, Iagaru A, et al. Serial Cardiac FDG-PET for the Diagnosis and Therapeutic Guidance of Patients With Cardiac Sarcoidosis. J Cardiac Fail. 2019;25:307–11. https://doi.org/10.1016/j.cardfail.2019.02.018.
Osborne MT, Hulten EA, Singh A, et al. Reduction in 18F-fluorodeoxyglucose uptake on serial cardiac positron emission tomography is associated with improved left ventricular ejection fraction in patients with cardiac sarcoidosis. J Nucl Cardiol. 2014;21:166–74. https://doi.org/10.1007/s12350-013-9828-6.
Ishizuka M, Kashimura T, Watanabe M, et al. Optimal Timing of Serial 18F-Fluoro-2-Deoxyglucose Positron Emission Tomography after Prednisolone Treatment Introduction for Cardiac Sarcoidosis. Int Heart J. 2023;64:196–202. https://doi.org/10.1536/ihj.22-406.
• Arps K, Doss J, Geiger K, et al. Incidence and Predictors of Relapse After Weaning Immune Suppressive Therapy in Cardiac Sarcoidosis. Am J Cardiol. 2023;204:249–56. https://doi.org/10.1016/j.amjcard.2023.07.088. In this study, Arps et al. discovered that the extent of late gadolinium enhancement serves as a predictor for relapse following the cessation of immunosuppressive therapy.
Waller AH, Blankstein R. Quantifying myocardial inflammation using F18-fluorodeoxyglucose positron emission tomography in cardiac sarcoidosis. J Nucl Cardiol. 2014;21:940–3. https://doi.org/10.1007/s12350-014-9921-5.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
JT and AR wrote the main manuscript text
JT prepared figure 1-5
All authors reviewed the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Treiber, J., Hamm, C.W., Sossalla, S.T. et al. Insights of Inflammatory Heart Involvement in Cardiac Sarcoidosis – A Systemic Review. Curr Cardiovasc Imaging Rep 17, 73–83 (2024). https://doi.org/10.1007/s12410-024-09594-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12410-024-09594-8