Abstract
Sociality is an instinctive property of organisms that live in relation to others and is a complex characteristic of higher order brain functions. However, the evolution of the human brain to acquire higher order brain functions, such as sociality, and the neural basis for executing these functions and their control mechanisms are largely unknown. Several studies have attempted to evaluate how human sociality was acquired during the course of evolution and the mechanisms controlling sociality from a neurodevelopment viewpoint. This review discusses these findings in the context of human brain evolution and the pathophysiology of autism spectrum disorder (ASD). Comparative genomic studies of postmortem primate brains have demonstrated human-specific regulatory mechanisms underlying higher order brain functions, providing evidence for the contribution of oligodendrocytes to human brain function. Functional analyses of the causative genes of ASD in animal models have demonstrated that the neural basis of social behavior is associated with layer 6 (L6) of the neocortex and oligodendrocytes. These findings demonstrate that both neurons and oligodendrocytes contribute to the neural basis and molecular mechanisms underlying human brain evolution and social functioning. This review provides novel insights into sociability and the corresponding neural bases of brain disorders and evolution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sociality is a higher order brain function that is essential for living within a community. Sociality is formed through relationships with others, and accumulating social experience leads to higher levels of sociality (Kappeler et al. 2015; Sachser et al. 2013; Tomasello 2020). Sociability begins with the formation of attachment to the caregiver after birth, and advanced sociability is acquired during different stages of growth from childhood (Tomasello 2020; Cascio et al. 2019; Jethava et al. 2022; Vivanti and Nuske 2017). Impairment in attachment formation during childhood negatively affects children’s social development (Teicher et al. 2016). Sociality and brain development are closely related (Fig. 1), and brain developmental disorders during the prenatal period increase the risk of postnatal neurodevelopmental and psychiatric disorders (Usui et al. 2023; Li et al. 2023; Doi et al. 2022a, b).
Developmental trajectories of the brain and plasticity. The pink bars at the top indicate social plasticity. The attachment was the first social bond formed with caregiver. Sociality develops through interactions with others in a social environment. Middle yellow bars indicate brain functional plasticity events. The sensory pathway includes functions associated with the visual, auditory, and somatosensory cortices such as vision, hearing, and touch, respectively. Language and speech include functions associated with the angular gyrus and Broca’s area. Amygdala-dependent learning includes cued conditioning and implicit learning. Hippocampus-dependent learning involves learning objects, places, spaces, and work. Higher cognitive function is associated with the prefrontal cortex (PFC). The bottom blue bars indicate developmental brain events in human and mouse. After neural tube formation in the ectoderm (neurulation), brain vesicles are formed, and neuroepithelial cells produce neural stem cell progenitors and neurons (neurogenesis). Neural progenitor cells also produce glia, such as astrocytes and oligodendrocytes (gliogenesis). Developing neurons migrate, differentiate into specific subtypes, form synapses, and become myelinated. GW gestational week
Previous studies demonstrated that several brain regions are associated with sociality. However, sociality is related to various factors, such as emotion, anxiety, and cognition, and cannot be explained by a specific circuit because of its complexity. Clinical studies on autism spectrum disorder (ASD), a neurodevelopmental disorder (NDD), have identified various brain regions associated with social interactions and communication (Amaral et al. 2008). For example, the orbitofrontal cortex, anterior cingulate cortex, and amygdala mirror neuron regions have been reported to be responsible for social interaction, while the inferior frontal gyrus, superior temporal sulcus, and basal ganglia are responsible for communication (Amaral et al. 2008; Hadjikhani et al. 2006). Moreover, studies using animals with modifications in genes responsible for ASD have reported developmental abnormalities in various brain regions involved in social behavior (de la Torre-Ubieta et al. 2016; Wang et al. 2014; Tebbenkamp et al. 2014; Doi et al. 2022a, b; Quesnel-Vallières et al. 2019; Willsey et al. 2022). Many genes related to human genetic risks associated with brain development and disorders, such as ASD and schizophrenia (SCZ), have been identified to understand the molecular mechanisms underlying social behavior (Doi et al. 2022a, b; Doan et al. 2018).
Humans have built a more advanced civilization than other primates. In this process, various events such as biological evolution, the evolution of the living environment and civilization, and population increases have led to the acquisition of high-level and complex functions (Russo and Nestler 2013; Usui et al. 2014). Previous studies on primates reported a positive correlation between the size of social groups and the neocortex capacity (Silk 2007; Dunbar and Shultz 2007). Language has evolved as a tool for social communication during the course of evolution (Russo and Nestler 2013; Usui et al. 2014). However, acquiring higher order brain functions is often accompanied by the risk of developing disorders that can impair these functions, such as NDDs, psychiatric disorders, and cognitive impairment (Usui et al. 2014; Irie et al. 2022; Pattabiraman et al. 2020; Kappeler et al. 2015; Vanderhaeghen and Polleux 2023).
Previous studies show that the acquisition and development of sociality could be closely related to brain development and influenced by the individual’s genes and the growing environment (Fig. 1). This review discusses the neural basis underlying sociality from the relevant studies of human brain evolution and disorders that impair sociality such as ASD and SCZ. In addition, I will also discuss the prospects of research aimed at elucidating neural circuits related to sociality, and research aimed at elucidating the functions of human-specific gene control mechanisms in the acquisition of higher brain functions in humans.
Human-specific genes and their functions in human brain evolution
The human brain has the largest proportion of body size among mammals. Various findings have been reported in the evolution of the human brain. Human neurons have larger cell bodies, thicker dendrites, and axons, with increased numbers and density of spines in pyramidal cells (Defelipe 2011; Elston et al. 2001; Duan et al. 2003). A recent connectome study comparing the cerebral cortices of humans, macaque monkeys, and mice reported that humans have a threefold increase in the number of interneurons and tenfold expansion of the interneuron-to-interneuron network compared with mice (Loomba et al. 2022).
Human-specific genes and their regulation machinery helped evolve the modern human brain (Usui et al. 2014; Irie et al. 2022; Florio et al. 2017). SRGAP2 gene has been duplicated thrice during human evolution, subsequently evolving as SRGAP2B, SRGAP2C, and SRGAP2D (Geschwind and Konopka 2012; Charrier et al. 2012; Dennis et al. 2012). Human-specific SRGAP2C increases spine density and delays maturation in the neocortex (Geschwind and Konopka 2012; Charrier et al. 2012), suggesting that SRGAP2C contributes to cortical expansion and increased spine numbers in humans. ARHGAP11B arose from the duplication of ARHGAP11A in humans and is expressed in the human apical and basal radial glia (Florio et al. 2015). ARHGAP11B promotes the generation and self-renewal of basal progenitors in the mouse cortex and increases cortical size and folding in the neocortex of marmosets (Florio et al. 2015; Heide et al. 2020). ARHGAP11B also regulates the mitochondrial Ca2+ concentration and induces glutaminolysis, which is required for human basal progenitor proliferation (Namba et al. 2020).
In addition, NOTCH2NL, human-specific paralogs of the NOTCH2 receptor, plays a role in the expansion of the human cortex by promoting cortical progenitor maintenance to generate higher neuronal outputs through inhibition of cis Delta/Notch interactions (Suzuki et al. 2018). Another group also reported that different alleles of NOTCH2NL enhance Notch signaling by directly interacting with NOTCH receptors to maintain the proliferation (Fiddes et al. 2018). In addition, NOTCH2NL located in 1q21.1 where distal deletions are associated with microcephaly and schizophrenia, and duplications are associated with macrocephaly and ASD, respectively (Fiddes et al. 2018). Hominini-specific regulation of CBLN2 having species differences in level of expression and laminar distribution promotes PFC dendritic spine formation (Shibata et al. 2021). CROCCP2 expressed in human fetal cortex also promotes proliferation of cortical progenitors by acting as a human-specific modifier to cilia dynamics and mTOR signaling (Van Heurck et al. 2023). Primate-specific gene TMEM14B expressed in outer radial glia cells also plays a role in cortical expansion and folding by interaction with IQGAP1 (Liu et al. 2017).
Moreover, the molecular evolution of FOXP2 identified from the KE family associated with language disorders is important for human signatures (Lai et al. 2001). Human FOXP2 has two amino acid mutations, T303N and N325S, compared to chimpanzees (Enard et al. 2002; Konopka et al. 2009), which induce functional changes in the FOXP2 transcription factor, resulting in changes in the motor and craniofacial development necessary for language and speech functions (Konopka et al. 2009), as well as diversity in vocalization (Enard et al. 2009).
Furthermore, we demonstrated that characteristic human gene expression networks are important for human brain evolution and brain disorders. A comparative genomic study among primates has shown that CLOCK is specifically upregulated in the human prefrontal cortex (PFC) and has identified it as a hub gene involved in cognitive function rather than in controlling circadian rhythms (Konopka et al. 2012). We found that human CLOCK regulates gene expression networks involved in cognitive functions, ASD, intellectual disability, as well as neurodevelopment, such as neuronal migration (Fontenot et al. 2017).
These findings suggest that human-specific gene functions are involved in the expansion of the human brain and the increase in synapses, contributing to neurodevelopment and acquisition of higher order brain functions (Fig. 2).
Schematic diagram of human-specific genes and its functions in human brain evolution. The blue color and pink color indicate the mouse genes and human genes, respectively. Human-specific genes accelerate evolution and expansion of human brain and its functions. These genes contribute to progenitor proliferation, expansion, increased numbers of synapses, and acquisition of higher order brain functions in human brain. Briefly, ARHGAP11B regulates basal progenitor proliferation. CROCCP2 promotes cortical progenitor proliferation. NOTCH2NL promotes cortical progenitor maintenance and cortical expansion. TMEM14B promotes cortical expansion. CLOCK also regulates neurodevelopmental gene expressions and neuronal migration. CBLN2 promotes dendritic spine formation. SRGAP2C increases spine density. FOXP2 regulates expression of genes associated with motor and craniofacial development
Oligodendrocytes contributions to human brain evolution
It has been argued that the human brain has an increased number of glia compared to neurons. The ratio of neurons to glia is said to increase from 1:1 in mice to 1:10 in humans. However, this signature has not been proven, and several studies have reported negative results regarding the tenfold increase. In fact, it has been reported that the ratio is 1:1 or slightly more glia in humans (Sherwood et al. 2006; Herculano-Houzel 2009, 2012, von Bartheld et al. 2016), but the actual ratio remains unknown because quantification in the whole brain has not been performed.
Glia were once thought to be supportive cells that fill gaps in the brain, but the importance and function of glia in the brain is becoming better understood. Glia in the central nervous system include astrocytes, oligodendrocytes, and microglia. Astrocytes physically support neurons, interact with brain blood vessels, reuptake of glutamate between synapses, and regulate extracellular ion concentrations (Volterra and Meldolesi 2005; Abbott et al. 2006). Oligodendrocytes form myelin sheaths, which increase the conduction velocity of action potentials by saltatory conduction and play a role in the metabolism of neurons and astrocytes (Richardson et al. 2006; de Faria et al. 2021). Microglia control immunity in the central nervous system through phagocytosis, release humoral factors, and performs synaptic pruning and interactions (Butovsky and Weiner 2018; Li and Barres 2018). Single-cell transcriptome studies have revealed an enormous diversity of glia that play a role in brain development (Fan et al. 2020; Darmanis et al. 2015; Polioudakis et al. 2019; Huang et al. 2020). Furthermore, the engraftment of human astrocytes into the mouse brain has been reported to alter synaptic plasticity and improve learning ability in mice (Han et al. 2013). These studies suggest that human neurons and glia contribute more to higher order brain functions than other primates and rodents.
We also conducted comparative genomics on primates and identified human-specific gene expression network modules in both neurons and oligodendrocytes (Berto et al. 2019; Mendizabal et al. 2019). Interestingly, a human-specific oligodendrocyte module was found to be involved in transcription and histone methylation (Berto et al. 2019). In addition, these module genes are enriched in risk variants such as NDDs, psychiatric disorders, and cognition (Berto et al. 2019). These findings suggest that not only neurons but also oligodendrocytes contributed to the acquisition of higher order brain functions in humans during evolution, and oligodendrocyte impairment is also a risk factor for NDDs and cognitive disorders. Thus, oligodendrocytes play essential roles in human brain evolution and brain functions such as sociality and cognition.
Regarding oligodendrocyte development, neural stem cells generate glia progenitor cells, which differentiate into oligodendrocyte progenitor cells (OPCs). OPCs further differentiate into immature, mature, and myelinated oligodendrocytes (Fields 2015). In human, the frontal cortex is still myelinating beyond late adolescence (Miller et al. 2012). Furthermore, complete myelination occurs only at sexual maturity in chimpanzees, whereas in humans, myelination is expanded compared to that of other primates (Miller et al. 2012), suggesting that oligodendrocytes and their myelination play a role in the development of higher order brain functions and cognition. Like neurogenesis, oligodendrogenesis occurs in the adult brain, and dead oligodendrocytes undergo periodic turnover in human (Rivers et al. 2008). Oligodendrocyte production and myelination peak at the age of 5 in humans and decline yearly after that, and the lost oligodendrocytes are replaced by neogenesis (Yeung et al. 2014). Recently, it has been reported that outer radial glial cells produce EGFR-expressing pre-OPCs in the subventricular zone (SVZ) and that pre-OPCs proliferate to increase the number of mitotic OPCs in the SVZ of human (Huang et al. 2020). They also reported that pre-OPCs proliferate and differentiate into early and late OPCs in the SVZ and subplate of human, respectively (Huang et al. 2020).
However, the human-specific functions of oligodendrocytes remain largely unknown. The human brain undergoes white matter volume expansion (Rilling and van den Heuvel 2018; Donahue et al. 2018). We identified human-specific transcriptome signatures of oligodendrocytes in postmortem brains (Berto et al. 2019); thus, analyses of these gene functions and/or networks may uncover human oligodendrocyte function. Interestingly, the human-specific upregulated neuronal module significantly overlapped with genes in the neuronal module dysregulated in SCZ and ASD in data from the PsychENCODE Consortium (Berto et al. 2019; Gandal et al. 2018). In addition, the human-specific downregulated oligodendrocyte module also overlaps with the genes in the oligodendrocyte module dysregulated in SCZ, ASD, and bipolar disorder in data from the PsychENCODE Consortium (Berto et al. 2019; Gandal et al. 2018). Another study also reported that hominin-specific gene regulatory elements such as enhancers and promoters function as transcriptional units selectively emerged in human oligodendrocytes-lineages; however, these elements were disrupted in patients with ASD (Castelijns et al. 2020). These results suggest that human-specific characteristics of gene expression networks and oligodendrocytes contribute to human brain evolution and sociality.
Development and impairment of sociality
In addition to normal brain development, the postnatal environment is an important factor in the development of sociality (Fig. 1). The environment influences optimal growth and health, including developmental aspects related to the social, cognitive, and immune systems in children (Consiglio and Brodin 2020; Ferguson et al. 2013; Mackes et al. 2020; Sonuga-Barke et al. 2017). The childhood environment significantly impacts brain structure, synaptic plasticity, and mental development (Miguel et al. 2019; Takesian and Hensch 2013). The period immediately after birth through adolescence, which is characterized by the development and acquisition of various senses, is called the critical period (Fig. 1) (Reh et al. 2020; Alberini and Travaglia 2017). The critical period peaks early in life, during which the nervous system is highly plastic and dynamically regulated throughout life (Takesian and Hensch 2013).
Attachment is the first social and emotional bond fostered between the caregiver and the child in human. Studies show that the critical period for attachment formation is up to the postnatal day (P) 16 in mice (Landers and Sullivan 2012), but the actual critical period has not been established. Impaired attachment formation during childhood increases the risk for attachment disorders in human. Attachment disorders are classified into reactive attachment disorders and disinhibited social engagement disorders (American Psychiatric Association 2013). Odors and touch stimuli are thought to be important for attachment formation in animals (Landers and Sullivan 2012; Sakano 2020). Oxytocin, a well-known social and affective hormone, plays an important role in rodent imprinting (Roth et al. 2013). Oxytocin-mediated imprinting has a critical period in mice (Inoue et al. 2021; Sakano 2020), suggesting that maternal odor during the neonatal period is important for attachment formation in mice. However, the neural basis underlying attachment and social formation remains largely unknown. At present, we have successfully generated a mouse model for attachment disorders and are attempting to elucidate the underlying mechanisms of attachment formation and its neural basis by studying the pathogenesis of attachment disorders.
Childhood stress, also called early life stress, broadly refers to stress experienced before reaching adulthood and includes events such as neglect, physical and psychological abuse, sexual abuse, loss of a caregiver, relationship development, bullying, accidents, illnesses, natural disasters, and wars (Heim et al. 2003, 2004; Agid et al. 2000). Environment is a crucial factor in providing optimal growth and health conditions for children, including social, cognitive, and immune system-related aspects (Consiglio and Brodin 2020; Ferguson et al. 2013; Mackes et al. 2020; Sonuga-Barke et al. 2017). A previous study reported that postnatal socially isolated mice exhibited impairments in social interactions, working memory, and myelination in the PFC (Makinodan et al. 2012). We previously reported that longer periods of social isolation reduced social behaviors in mice, increased anxiety-like behavior, and reduced the number of neurons in the PFC of mice (Usui et al. 2021b). These studies suggest that postnatal environment and social experiences are critical for development of sociality.
Based on socially isolated mice studies, we identified Zbtb16 as a gene involved in sociality. Zbtb16 is the most significantly downregulated gene in the PFC of socially isolated mice (Usui et al. 2021b). ZBTB16 encodes a transcription factor that contains a BTB/POZ protein–protein interaction domain at its N-terminus and a C2H2-type zinc finger DNA-binding domain at its C-terminus, which plays key roles in stem cell maintenance, proliferation, differentiation, apoptosis, and chromatin remodeling Suliman et al. 2012; Šeda et al. 2017). Behavioral analysis was performed to investigate whether Zbtb16 regulates mouse social behaviors, and it was found that social behavior was impaired in Zbtb16 knockout (KO) mice (Usui et al. 2021a). Interestingly, this mouse had impaired oligodendrocyte development and differentiation and reduced myelination in the neocortex (Usui et al. 2021a). Oligodendrocyte development has also been reported to be impaired when normal development in mice childhood is disrupted by external factors such as early life stress (Kokkosis et al. 2022; Teissier et al. 2020). Taken together, these findings suggest that oligodendrocytes play an important role in social development and form the neural basis underlying sociality.
Cortical deep layers underlying sociality
To understand the neural basis of sociality, we focused on ASD as a disorder that impairs sociality. ASD is a heterogeneous NDD that causes pervasive abnormalities in social communication, repetitive restricted behaviors and interests, and hyperesthesia and hypesthesia (Lord et al. 2020, 2018; Khodosevich and Sellgren 2023). ASD pathogenesis is associated with complex genetic and environmental factors (Lord et al. 2020, 2018; Doi et al. 2022a, b; Usui et al. 2023, 2022a; Willsey et al. 2022). Over 1000 ASD-associated genes have been identified in individuals with ASD (Wilkinson et al. 2015; Toma et al. 2014; Griswold et al. 2015; Yao et al. 2015; Satterstrom et al. 2020; Wang et al. 2020; Iossifov et al. 2014; Willsey et al. 2022), which play essential roles in fetal brain development, particularly in neurodevelopment and synaptogenesis (de la Torre-Ubieta et al. 2016; Wang et al. 2014; Tebbenkamp et al. 2014; Doi et al. 2022a, b; Quesnel-Vallières et al. 2019; Willsey et al. 2022).
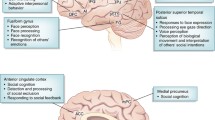
Studies in humans have identified various regions in the brain associated with social interactions and communication in individuals with ASD (Amaral et al. 2008). The orbitofrontal cortex, anterior cingulate cortex, and amygdala mirror neuron regions are responsible for social interactions (Fig. 3) (Amaral et al. 2008; Hadjikhani et al. 2006). The inferior frontal gyrus, superior temporal sulcus, and basal ganglia are responsible for communication (Fig. 3) (Amaral et al. 2008). In animal studies, we demonstrated that developmental defects in layer 6 (L6) neurons and oligodendrocytes, which are common in ASD model mice, are responsible for sociality (Fig. 3) (Co et al. 2020a, b; Usui et al. 2017a, 2021a). Studies also demonstrated that Purkinje cells in the cerebellum play a role in social communication (Fig. 3) (Usui et al. 2017b). These findings indicate that brain regions associated with social interaction and communication in humans are consistent with those impaired in mouse models of ASD-causing genes.
Schematic diagram of human and mouse neural circuits related to social behaviors. a, b Networks of the human (a) and mouse (b) brain regions involved in sociality. Brain regions associated with sociality have been identified in ASD studies (Amaral et al. 2008; Barak and Feng 2016; Gandhi and Lee 2020). In mice, we identified brain regions associated with sociality, as reported in previous studies, including ours. c Schematic diagram of neocortical phenotypes in ASD model mice. From the phenotypes of the multiple ASD model mice, we found decreased cortical thickness, L6 thickness, L6 neurons, impaired neuronal migration, and decreased oligodendrocytes. d Schematic diagram of the neural circuits centered at L6 of the neocortex, which are thought to be associated with social behavior. L6 neurons form a feedback loop with the thalamus and play a role in switching behavior by adjusting sensory information, such as visual and auditory. In addition to the thalamus, L6 neurons project to the claustrum, striatum, and ipsilateral cortex, which are associated with social behaviors. e Connectome of representative brain regions in mouse social behavior. Abbreviations of each brain region and their roles in social behavior and/or ASD characteristics as follow; PFC prefrontal cortex (social information processing), ACC anterior cingulate cortex (social cognition), OFC orbitofrontal cortex (social adjustment), TP temporal pole (language, theory of mind), FG fusiform gyrus (face perception), PMC premotor cortex (mirror system), IFG inferior frontal gyrus (frontal mirror area and emotional judgment), AI anterior insula (social cognition), AMG amygdala (social information judgment, emotion recognition, and theory of mind), STR striatum (serotonin and dopamine signals), STS superior temporal sulcus (responses to face expression, eye gaze direction, and voice perception), SG supramarginal gyrus (mirror system), TPJ temporoparietal junction (beliefs and theory of mind), IPS interparietal sulcus (eye gaze direction and social status judgment), IPC inferior parietal cortex (mirror system), CTX cortex, CLA claustrum (social behavior, multi-sensory processes, attention, and consciousness), HIP hippocampus (social memory and LTP), TH thalamus (social recognition and multi-sensory processes), HY hypothalamus (oxytocin release, social reward, and stress), OT olfactory tubercle (social response, multi-sensory processes, and reward), VTA ventral tegmental area (dopamine release and movement), SN substantia nigra (dopamine release and movement), CB cerebellum (vocal communication), SC spinal cord, L layer, IC ipsilateral cortex, CC contralateral cortex, SUB subcortical nucleus including the striatum, red nucleus, pontine nucleus, olive nucleus, and spinal cord
FOXP1 is a forkhead transcription factor that regulates cell proliferation and differentiation during development and is a high-confidence gene associated with ASD (Iossifov et al. 2014; Sanders et al. 2015; Stessman et al. 2017; Satterstrom et al. 2020; Willsey et al. 2022; Bacon and Rappold 2012; Siper et al. 2017; Hamdan et al. 2010; Lozano et al. 2015; O'Roak et al. 2011). Deletion of Foxp1 in the brain using a conditional knockout (cKO) (Nes-Cre; Foxp1flox/flox) has been reported to cause ASD-like behaviors, abnormalities in the striatum and hippocampal development, and reduced excitability of hippocampal CA1 neurons in adult mice (Bacon et al. 2015). Patient-relevant Foxp1 mouse studies have reported increased excitability of striatal medium spiny neurons, reduced neonatal ultrasonic vocalizations (USVs), and altered gene expression related to ASD in adult mice (Fig. 3) (Araujo et al. 2015). We have also demonstrated that forebrain-specific Foxp1 cKO (Emx1-Cre; Foxp1flox/flox) mice show impaired neonatal and adult USVs, global motor dysfunction, social impairment, hyperactivity, and anxiety-like behavior in postnatal and adult mice (Usui et al. 2017a; Araujo et al. 2017). In Foxp1 cKO mice, reduced neocortical thickness, particularly L6 thickness, and fewer L6 neurons were observed (Fig. 3) (Usui et al. 2017a). Analysis of Foxp1-transcriptome in the PFC and hippocampus revealed that Foxp1 regulates cell proliferation and differentiation, cell migration, synaptic transmission, axon ensheathment, and ASD-associated gene expression (Araujo et al. 2017; Usui et al. 2017a). These findings demonstrate that the L6, the striatum, and the hippocampus, including electrophysiological properties, are essential for the neural basis of sociality.
FOXP2 is a member of the forkhead gene family and is expressed in deep-layer subcortical projection neurons, including L6 corticothalamic projection neurons and L5 pyramidal tract neurons (Sorensen et al. 2015; Tasic et al. 2016; Kast et al. 2019; Willsey et al. 2013). Mutations of FOXP2 have been identified in patients with speech and language disorders, ASD, and attention-deficit/hyperactivity disorder (Co et al. 2020a, b; Lai et al. 2001; Demontis et al. 2019; Reuter et al. 2017; Satterstrom et al. 2020). We have previously shown that Foxp2 regulates vocal communication and motor functions in postnatal mice through the development of cerebellar Purkinje cells (Fig. 3) (Usui et al. 2017b). Moreover, cortex-specific Foxp2 cKO (Emx1-Cre; Foxp2flox/flox) mice showed decreased neonatal USVs, impaired cognitive flexibility and hyperactivity, and decreased cortical dopamine receptor D1 (DRD1) expression (Fig. 3) (Co et al. 2020a, b). These studies demonstrate that the neural basis of cerebellar Purkinje cells and L6 is important for sociality.
ZBTB16 transcription factor plays a role in cell proliferation and differentiation, apoptosis, chromatin remodeling, and other biological functions (Suliman et al. 2012; Šeda et al. 2017). In one study, a mutation (c.1319G > A; p.Arg440Gln) in ZBTB16 was identified in brothers with ASD (Bacchelli et al. 2019). We demonstrated that Zbtb16 KO mice exhibit ASD- and SCZ-like behaviors, including social impairments, repetitive behaviors, risk-taking behaviors, and cognitive deficits, with reduced neocortical thickness, particularly L6 thickness, and reduced numbers of L6 neurons (Fig. 3) (Usui et al. 2021a). In Zbtb16 KO mice, the numbers of oligodendrocyte progenitors (OPCs) and mature oligodendrocytes were reduced, and myelination of the neocortex was impaired (Fig. 3) (Usui et al. 2021a). The Zbtb16 transcriptome in the PFC revealed that Zbtb16 is involved in neurogenesis, nervous system development, cell localization, axon ensheathment, myelination, and the regulation of ASD- and SCZ-associated gene expression (Usui et al. 2021a). In addition, the length of the axonal initial segment of pyramidal neurons was reduced in the primary somatosensory cortex of Zbtb16 KO mice (Fig. 3) (Usui et al. 2022b), suggesting that the action potential of these neurons may be impaired. These studies demonstrate that L6 and cytoarchitecture, oligodendrocytes, and myelination are essential for the neural basis of sociality.
Abnormalities in L6, such as reductions in the thickness and number of neurons, are common phenotypes in ASD mouse models (Usui et al. 2017a, 2021a; Co et al. 2020a, b). Interestingly, ASD-associated genes have been reported to be enriched in the deep layers (Willsey et al. 2013; Tebbenkamp et al. 2014). In particular, high-confidence ASD and probable ASD risk genes converge to glutamatergic projection neurons in L5 and L6 of the human mid-fetal prefrontal and primary motor-somatosensory cortex (Willsey et al. 2013). Cortical glutamatergic projection neurons can be divided into major classes and subclasses that contribute to distinct functional subnetworks (Harris and Shepherd 2015). The major class consists of pyramidal tract neurons, which give rise to corticofugal pathways that target all subcortical regions (Fig. 3) (Mohan et al. 2023). Another class consists of intratelencephalic neurons that target other cortical and striatal regions, including the contralateral hemisphere (Fig. 3) (Mohan et al. 2023). The L6 pyramidal neurons also project to the claustrum, striatum, and ipsilateral cortex (Fig. 3) (Thomson 2010; Bertero et al. 2022; Baker et al. 2018). Another study demonstrated that the activation of L6 corticothalamic neurons suppresses excitatory neurons in L4 and generates EPSPs in pyramidal neurons in L5a, indicating that L6 corticothalamic neurons strongly activate the output layer of the cortex (Kim et al. 2014). Moreover, the corticothalamic pathways originate from L5 or L6 of the neocortex and are much more numerous than the ascending thalamocortical pathways (Antunes and Malmierca 2021). Interestingly, L6 corticothalamic neurons are also involved in behavioral switching between sound detection and discrimination (Antunes and Malmierca 2021; Guo et al. 2017). Accordingly, L6 neurons coordinate not only auditory information but also various sensory systems, such as visual information, and the integration of this sensory information has been suggested to contribute to the control of social behaviors.
FOXP2 and TBR1 are the L6 markers. TBR1 is a T-box brain transcription factor that was identified in patients with ASD and ID (Nambot et al. 2020; Sapey-Triomphe et al. 2020; Vegas et al. 2018), has been shown to regulate cortical lamina formation, differentiation of L6, dendritic patterning, and inhibitory synaptic density during development (Co et al. 2022; Fazel Darbandi et al. 2018; Bedogni et al. 2010). Tbr1 haploinsufficiency in mice resulted in axonal projection defects in amygdala neurons and impairments in social interaction, USVs, and cognitive flexibility (Huang et al. 2014). TBR1 chromatin immunoprecipitation sequencing studies have also reported that TBR1-bound regions adjacent to ASD genes are enriched in the developing mouse neocortex (Notwell et al. 2016). FOXP2 is expressed in deep-layer subcortical projection neurons, such as L6 corticothalamic projection neurons and L5 pyramidal tract neurons (Sorensen et al. 2015; Tasic et al. 2016; Kast et al. 2019; Willsey et al. 2013). FOXP2 is associated with speech and language disorders, ASD, and attention-deficit/hyperactivity disorder (Lai et al. 2001; Demontis et al. 2019; Reuter et al. 2017; Satterstrom et al. 2020). Foxp2 cKO mice exhibited decreased USVs, impaired cognitive flexibility, and decreased cortical DRD1 expression (Co et al. 2020a, b). These findings suggest that L6 neurons play essential roles in social behavior, cognition, and output of cortical neuronal activity.
On the other hand, the abnormal laminar cytoarchitecture and cortical disorganization of L2/3 thourt L5 neurons in focal patches in the cortex have been reported in children with ASD (Stoner et al. 2014). This patch phenotype has been reported in the dysgranular zone of the primary somatosensory cortex where showing excessive neuronal activities in maternal immune activation offspring mice model of ASD (Shin Yim et al. 2017). It has also reported that disorganization of cortical networks within L1 in the lateral prefrontal cortex of children with ASD (Trutzer et al. 2019). These reports were observed in the upper layers of the cortex, but excessive neural activity has been reported in not only the upper layers but also the deep layers of the cerebral cortex in mice with the patch phenotype. As our findings related to those, the length of the axonal initial segment of pyramidal neurons was reduced in the L2/3 primary somatosensory cortex of Zbtb16 KO mice (Usui et al. 2022b). These previous studies show that not only the deep layers, but also upper layers in the cortex.
Taken together, our findings demonstrate that developmental defects in L6 neurons and Purkinje cells which are common in ASD model mice, are responsible for sociality (Co et al. 2020a, b; Usui et al. 2021a, 2017b, 2017a).
Relationships between oligodendrocytes and sociality
Oligodendrocytes are linked to NDDs and psychiatric disorders (Berto et al. 2019; Nagy et al. 2020; Fessel 2022; Zhou et al. 2021; Miyata et al. 2015; Castelijns et al. 2020). In patients with ASD, age-related differences in white matter diffusion have been reported in the uncinate fasciculus, corticospinal tract, inferior longitudinal fasciculus, inferior fronto-occipital fasciculus, anterior thalamic radiation, superior longitudinal fasciculus, and forceps major (Thompson et al. 2020). Studies on ASD severity and white matter development have reported lower white matter development in early childhood than in typical development (Andrews et al. 2021). Consistent with findings in patients with ASD (Barnea-Goraly et al. 2004, AmaralSchumann and Nordahl 2008), impairments in oligodendrogenesis and myelination were found in ASD model mice (Fig. 4) (Usui et al. 2021a). Tcf4 mutant mice display impaired oligodendrocyte development and myelination, supporting the importance of oligodendrocytes in ASD pathogenesis (Phan et al. 2020). Chd8 heterozygous mice also exhibit abnormal social behaviors, anxiety-like behaviors, defective myelination, and slower action potential transmission (Kawamura et al. 2020).
Association between oligodendrocytes and sociability. The red bars indicate the normal function of oligodendrocytes. Oligodendrocytes form myelin and play important roles in modulating neuronal activity, axonal protection, white matter expansion, and maintaining brain functions through metabolism supports. In contrast, the blue bars indicate the associated dysfunction of oligodendrocytes in patients with ASD and in ASD model mice. Dyslipidemia is associated with sociality in children with ASD. Lipids are the major components of myelin, and dyslipidemia in ASD adversely affects myelin structure. Oligodendrocytes are the major components of the white matter, and decreased white matter volume has been reported in patients with ASD, suggesting oligodendrocytes play a role in sociability and other higher order brain functions
Lipids are the primary components of the myelin sheath formed by oligodendrocytes. Dyslipidemia phenotypes in children with ASD have been reported previously (Fig. 4) (Usui et al. 2020), with these children showing positive correlations between clinical scores for social interactions and a specific decrease in very-low-density lipoprotein (VLDL) levels, as well as increases in fatty acid levels (Usui et al. 2020). Furthermore, the activity of lipoprotein lipase, a VLDL-degrading enzyme, is higher in children with ASD (Hirai et al. 2020), suggesting that VLDL is specifically degraded during ASD pathogenesis. Interestingly, VLDL and low-density lipoprotein receptors are specifically expressed in mature myelinating oligodendrocytes at postnatal stages but are progressively downregulated after P15 (Zhao et al. 2007), suggesting that VLDL and low-density lipoprotein play important roles in myelination. These studies suggest that oligodendrocytes contribute to higher order brain functions in humans, while oligodendrocyte impairment is a risk factor for disorders such as NDDs and cognitive disorders and plays an essential role in sociality (Fig. 4).
Conclusion
This review focused primarily on studies aimed at understanding the neural basis of sociality. Studies have also characterized the molecular mechanisms by which sociality is acquired by focusing on human brain evolution. Moreover, focusing on brain disorders that impair sociality like ASD as a disorder characterized by social impairment, studies have demonstrated many sociability-related brain phenotypes. A combination of these two approaches may uncover the neural circuits and human-specific gene expression networks that are suggested to be related to sociality. From these methods, we suggested that layer 6 of the cerebral cortex and oligodendrocytes are related to the neural basis of human sociality.
However, unlike specific genes, it is difficult to actually study the complete function of the gene networks themselves identified from studies focused on brain evolution. Therefore, it is possible to analyze the function of highly important genes such as “hub genes” on the network instead of expressing the genes network themselves. In order to elucidate the functions of human genes, studies that expresses human genes in model animals has provided many findings (Geschwind and Konopka 2012; Charrier et al. 2012; Dennis et al. 2012). On the other hand, it is also true that model animals have the problem that they do not have the same genomic background as humans.
To avoid such problems, there are methods using human cells or human brain organoids (Doi et al. 2022a, b; Gordon and Geschwind 2020). Similarly, to elucidate the function of human oligodendrocytes, there are options to use cultured human oligodendrocytes, cultured brain organoids, or even engraftment of human oligodendrocytes in the model animal brain such as mouse to analyze their effects at the individual level. We will elucidate the function of these human hub genes and human oligodendrocytes in the future studies. Particularly, we are particularly interested in determining how the capabilities and functions of human oligodendrocytes differ from those of other primates and rodents. The latest study shows that OPCs are specifically increased in human posterior cingulate cortical tissue, while mature oligodendrocytes are decreased, suggesting OPCs accelerated human brain evolution (Caglayan et al. 2023). Moreover, we will investigate the involvement of oligodendrocytes and their myelination in the development and regulation of sociality, which is relevant to the pathogenesis of ASD and human brain evolution.
In addition, it is important to elucidate how neural circuits centered on cortical L6 control sociality, a common phenotype observed in several ASD mouse models. In the L6 of the cerebral cortex, genes related to ASD and sociality, and genes such as FOXP2 related to language are expressed, indicating functional importance for sociality. Interestingly, the latest study also shows FOXP2 expression is specifically increased in two excitatory subtypes (L4-6_RORB_2 and L5-6_THEMIS_1 subtypes) of posterior cingulate cortex, compared to other primates (Caglayan et al. 2023). Therefore, we will focus on the output from L6 and input to L6 in the cerebral cortex, and will investigate the neural circuits related to sociality including the interaction with oligodendrocytes using transgenic animals, optogenetics, and chemogenetics.
In close, we will continue to study these topics to elucidate a complete picture of the neural basis underlying human sociality for providing the insight into the neural basis of sociality.
References
Abbott NJ, Rönnbäck L, Hansson E (2006) Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci 7:41–53
Agid O, Kohn Y, Lerer B (2000) Environmental stress and psychiatric illness. Biomed Pharmacother 54:135–141
Alberini CM, Travaglia A (2017) Infantile Amnesia: A Critical Period of Learning to Learn and Remember. The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 37:5783–5795
Amaral DG, Schumann CM, Nordahl CW (2008) Neuroanatomy of autism. Trends Neurosci 31:137–145
American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Washington, DC
Andrews DS, Lee JK, Harvey DJ et al (2021) A longitudinal study of white matter development in relation to changes in autism severity across early childhood. Biol Psychiatry 89:424–432
Antunes FM, Malmierca MS (2021) Corticothalamic pathways in auditory processing: recent advances and insights from other sensory systems. Front Neural Circ 15:721186
Araujo DJ, Anderson AG, Berto S et al (2015) FoxP1 orchestration of ASD-relevant signaling pathways in the striatum. Genes Dev 29:2081–2096
Araujo DJ, Toriumi K, Escamilla CO et al (2017) Foxp1 in forebrain pyramidal neurons controls gene expression required for spatial learning and synaptic plasticity. J Neurosci 37:10917–10931
Bacchelli E, Loi E, Cameli C et al (2019) Analysis of a sardinian multiplex family with autism spectrum disorder points to post-synaptic density gene variants and identifies CAPG as a functionally relevant candidate gene. J Clin Med 8:212
Bacon C, Rappold GA (2012) The distinct and overlapping phenotypic spectra of FOXP1 and FOXP2 in cognitive disorders. Hum Genet 131:1687–1698
Bacon C, Schneider M, Le Magueresse C et al (2015) Brain-specific Foxp1 deletion impairs neuronal development and causes autistic-like behaviour. Mol Psychiatry 20:632–639
Baker A, Kalmbach B, Morishima M, Kim J, Juavinett A, Li N, Dembrow N (2018) Specialized subpopulations of deep-layer pyramidal neurons in the neocortex: bridging cellular properties to functional consequences. J Neurosci 38:5441–5455
Barak B, Feng G (2016) Neurobiology of social behavior abnormalities in autism and Williams syndrome. Nat Neurosci 19:647–655
Barnea-Goraly N, Kwon H, Menon V, Eliez S, Lotspeich L, Reiss AL (2004) White matter structure in autism: preliminary evidence from diffusion tensor imaging. Biol Psychiatry 55:323–326
Bedogni F, Hodge RD, Elsen GE et al (2010) Tbr1 regulates regional and laminar identity of postmitotic neurons in developing neocortex. Proc Natl Acad Sci USA 107:13129–13134
Bertero A, Verrillo L, Apicella AJ (2022) A novel layer 4 corticofugal cell type/projection involved in thalamo-cortico-striatal sensory processing. J Neurosci 42:1383–1405
Berto S, Mendizabal I, Usui N et al (2019) Accelerated evolution of oligodendrocytes in the human brain. Proc Natl Acad Sci USA 116:24334–24342
Butovsky O, Weiner HL (2018) Microglial signatures and their role in health and disease. Nat Rev Neurosci 19:622–635
Caglayan E, Ayhan F, Liu Y et al (2023) Molecular features driving cellular complexity of human brain evolution. Nature 620:145–153
Cascio CJ, Moore D, Mcglone F (2019) Social touch and human development. Dev Cogn Neurosci 35:5–11
Castelijns B, Baak ML, Timpanaro IS et al (2020) Hominin-specific regulatory elements selectively emerged in oligodendrocytes and are disrupted in autism patients. Nat Commun 11:301
Charrier C, Joshi K, Coutinho-Budd J et al (2012) Inhibition of SRGAP2 function by its human-specific paralogs induces neoteny during spine maturation. Cell 149:923–935
Co M, Anderson AG, Konopka G (2020a) FOXP transcription factors in vertebrate brain development, function, and disorders. Wiley Interdiscip Rev Dev Biol 9:e375
Co M, Hickey SL, Kulkarni A, Harper M, Konopka G (2020b) Cortical Foxp2 supports behavioral flexibility and developmental dopamine D1 receptor expression. Cereb Cortex 30:1855–1870
Co M, Barnard RA, Jahncke JN et al (2022) Shared and distinct functional effects of patient-specific Tbr1 mutations on cortical development. J Neurosci 42:7166–7181
Consiglio CR, Brodin P (2020) Stressful beginnings with long-term consequences. Cell 180:820–821
Darmanis S, Sloan SA, Zhang Y et al (2015) A survey of human brain transcriptome diversity at the single cell level. Proc Natl Acad Sci USA 112:7285–7290
De Faria Jr. O, Pivonkova H, Varga B, Timmler S, Evans KA, Káradóttir RT (2021) Periods of synchronized myelin changes shape brain function and plasticity. Nat Neurosci 24:1508–1521
De La Torre-Ubieta L, Won H, Stein JL, Geschwind DH (2016) Advancing the understanding of autism disease mechanisms through genetics. Nat Med 22:345–361
Defelipe J (2011) The evolution of the brain, the human nature of cortical circuits, and intellectual creativity. Front Neuroanat 5:29
Demontis D, Walters RK, Martin J et al (2019) Discovery of the first genome-wide significant risk loci for attention deficit/hyperactivity disorder. Nat Genet 51:63–75
Dennis MY, Nuttle X, Sudmant PH et al (2012) Evolution of human-specific neural SRGAP2 genes by incomplete segmental duplication. Cell 149:912–922
Doan RN, Shin T, Walsh CA (2018) Evolutionary changes in transcriptional regulation: insights into human behavior and neurological conditions. Annu Rev Neurosci 41:185–206
Doi M, Li M, Usui N, Shimada S (2022a) Genomic strategies for understanding the pathophysiology of autism spectrum disorder. Front Mol Neurosci 15:930941
Doi M, Usui N, Shimada S (2022b) Prenatal environment and neurodevelopmental disorders. Front Endocrinol (lausanne) 13:860110
Donahue CJ, Glasser MF, Preuss TM, Rilling JK, Van Essen DC (2018) Quantitative assessment of prefrontal cortex in humans relative to nonhuman primates. Proc Natl Acad Sci USA 115:E5183-e5192
Duan H, Wearne SL, Rocher AB, Macedo A, Morrison JH, Hof PR (2003) Age-related dendritic and spine changes in corticocortically projecting neurons in macaque monkeys. Cereb Cortex 13:950–961
Dunbar RI, Shultz S (2007) Evolution in the social brain. Science (new York, N.y.) 317:1344–1347
Elston GN, Benavides-Piccione R, Defelipe J (2001) The pyramidal cell in cognition: a comparative study in human and monkey. J Neurosci 21:Rc163
Enard W, Przeworski M, Fisher SE et al (2002) Molecular evolution of FOXP2, a gene involved in speech and language. Nature 418:869–872
Enard W, Gehre S, Hammerschmidt K et al (2009) A humanized version of Foxp2 affects cortico-basal ganglia circuits in mice. Cell 137:961–971
Fan X, Fu Y, Zhou X et al (2020) Single-cell transcriptome analysis reveals cell lineage specification in temporal-spatial patterns in human cortical development. Sci Adv 6:eaaz2978
Fazel Darbandi S, Robinson Schwartz SE, Qi Q et al (2018) Neonatal Tbr1 dosage controls cortical layer 6 connectivity. Neuron 100:831-845.e7
Ferguson KT, Cassells RC, Macallister JW, Evans GW (2013) The physical environment and child development: an international review. Int J Psychol 48:437–468
Fessel J (2022) Abnormal oligodendrocyte function in schizophrenia explains the long latent interval in some patients. Transl Psychiatry 12:120
Fiddes IT, Lodewijk GA, Mooring M et al (2018) Human-specific NOTCH2NL genes affect notch signaling and cortical neurogenesis. Cell 173:1356-1369.e22
Fields RD (2015) A new mechanism of nervous system plasticity: activity-dependent myelination. Nat Rev Neurosci 16:756–767
Florio M, Albert M, Taverna E et al (2015) Human-specific gene ARHGAP11B promotes basal progenitor amplification and neocortex expansion. Science (new York, N.y.) 347:1465–1470
Florio M, Borrell V, Huttner WB (2017) Human-specific genomic signatures of neocortical expansion. Curr Opin Neurobiol 42:33–44
Fontenot MR, Berto S, Liu Y et al (2017) Novel transcriptional networks regulated by CLOCK in human neurons. Genes Dev 31:2121–2135
Gandal MJ, Zhang P, Hadjimichael E et al (2018) Transcriptome-wide isoform-level dysregulation in ASD, schizophrenia, and bipolar disorder. Science 362:eaat8127
Gandhi T, Lee CC (2020) Neural mechanisms underlying repetitive behaviors in rodent models of autism spectrum disorders. Front Cell Neurosci 14:592710
Geschwind DH, Konopka G (2012) Neuroscience: genes and human brain evolution. Nature 486:481–482
Gordon A, Geschwind DH (2020) Human in vitro models for understanding mechanisms of autism spectrum disorder. Molecular Autism 11:26
Griswold AJ, Dueker ND, Van Booven D et al (2015) Targeted massively parallel sequencing of autism spectrum disorder-associated genes in a case control cohort reveals rare loss-of-function risk variants. Mol Autism 6:43
Guo W, Clause AR, Barth-Maron A, Polley DB (2017) A corticothalamic circuit for dynamic switching between feature detection and discrimination. Neuron 95:180-194.e5
Hadjikhani N, Joseph RM, Snyder J, Tager-Flusberg H (2006) Anatomical differences in the mirror neuron system and social cognition network in autism. Cereb Cortex 16:1276–1282
Hamdan FF, Daoud H, Rochefort D et al (2010) De novo mutations in FOXP1 in cases with intellectual disability, autism, and language impairment. Am J Hum Genet 87:671–678
Han X, Chen M, Wang F et al (2013) Forebrain engraftment by human glial progenitor cells enhances synaptic plasticity and learning in adult mice. Cell Stem Cell 12:342–353
Harris KD, Shepherd GM (2015) The neocortical circuit: themes and variations. Nat Neurosci 18:170–181
Heide M, Haffner C, Murayama A et al (2020) Human-specific ARHGAP11B increases size and folding of primate neocortex in the fetal marmoset. Science (new York, N.y.) 369:546–550
Heim CP, Meinlschmidt GMS, Nemeroff CBMDP (2003) Neurobiology of early-life stress. Psychiatr Ann 33:18–26
Heim C, Plotsky PM, Nemeroff CB (2004) Importance of studying the contributions of early adverse experience to neurobiological findings in depression. Neuropsychopharmacology 29:641–648
Herculano-Houzel S (2009) The human brain in numbers: a linearly scaled-up primate brain. Front Hum Neurosci 3:31
Herculano-Houzel S (2012) The remarkable, yet not extraordinary, human brain as a scaled-up primate brain and its associated cost. Proc Natl Acad Sci USA 109(Suppl 1):10661–10668
Hirai T, Usui N, Iwata K et al (2020) Increased plasma lipoprotein lipase activity in males with autism spectrum disorder. Res Autism Spectrum Disord 77:101630
Huang TN, Chuang HC, Chou WH, Chen CY, Wang HF, Chou SJ, Hsueh YP (2014) Tbr1 haploinsufficiency impairs amygdalar axonal projections and results in cognitive abnormality. Nat Neurosci 17:240–247
Huang W, Bhaduri A, Velmeshev D et al (2020) Origins and proliferative states of human oligodendrocyte precursor cells. Cell 182:594-608.e11
Inoue N, Nishizumi H, Ooyama R, Mogi K, Nishimori K, Kikusui T, Sakano H (2021) The olfactory critical period is determined by activity-dependent Sema7A/PlxnC1 signaling within glomeruli. eLife 10:e65078
Iossifov I, O’Roak BJ, Sanders SJ et al (2014) The contribution of de novo coding mutations to autism spectrum disorder. Nature 515:216–221
Irie K, Doi M, Usui N, Shimada S (2022) Evolution of the human brain can help determine pathophysiology of neurodevelopmental disorders. Front Neurosci 16:871979
Jethava V, Kadish J, Kakonge L, Wiseman-Hakes C (2022) Early attachment and the development of social communication: a neuropsychological approach. Front Psych 13:838950
Kappeler PM, Cremer S, Nunn CL (2015) Sociality and health: impacts of sociality on disease susceptibility and transmission in animal and human societies. Philos Trans R Soc Lond B Biol Sci 370:20140116
Kast RJ, Lanjewar AL, Smith CD, Levitt P (2019) FOXP2 exhibits projection neuron class specific expression, but is not required for multiple aspects of cortical histogenesis. eLife 8:e42012
Kawamura A, Katayama Y, Nishiyama M et al (2020) Oligodendrocyte dysfunction due to Chd8 mutation gives rise to behavioral deficits in mice. Hum Mol Genet 29:1274–1291
Khodosevich K, Sellgren CM (2023) Neurodevelopmental disorders-high-resolution rethinking of disease modeling. Mol Psychiatry 28:34–43
Kim J, Matney CJ, Blankenship A, Hestrin S, Brown SP (2014) Layer 6 corticothalamic neurons activate a cortical output layer, layer 5a. J Neurosci 34:9656–9664
Kokkosis AG, Madeira MM, Mullahy MR, Tsirka SE (2022) Chronic stress disrupts the homeostasis and progeny progression of oligodendroglial lineage cells, associating immune oligodendrocytes with prefrontal cortex hypomyelination. Mol Psychiatry 27:2833–2848
Konopka G, Bomar JM, Winden K et al (2009) Human-specific transcriptional regulation of CNS development genes by FOXP2. Nature 462:213–217
Konopka G, Friedrich T, Davis-Turak J et al (2012) Human-specific transcriptional networks in the brain. Neuron 75:601–617
Lai CS, Fisher SE, Hurst JA, Vargha-Khadem F, Monaco AP (2001) A forkhead-domain gene is mutated in a severe speech and language disorder. Nature 413:519–523
Landers MS, Sullivan RM (2012) The development and neurobiology of infant attachment and fear. Dev Neurosci 34:101–114
Li Q, Barres BA (2018) Microglia and macrophages in brain homeostasis and disease. Nat Rev Immunol 18:225–242
Li M, Usui N, Shimada S (2023) Prenatal sex hormone exposure is associated with the development of autism spectrum disorder. Int J Mol Sci 24:2203
Liu J, Liu W, Yang L et al (2017) The primate-specific gene TMEM14B marks outer radial glia cells and promotes cortical expansion and folding. Cell Stem Cell 21:635-649.e8
Loomba S, Straehle J, Gangadharan V et al (2022) Connectomic comparison of mouse and human cortex. Science (new York, N.y.) 377:eabo0924
Lord C, Elsabbagh M, Baird G, Veenstra-Vanderweele J (2018) Autism spectrum disorder. Lancet 392:508–520
Lord C, Ts B, Charman T et al (2020) Autism spectrum disorder. Nat Rev Dis Primers 6:5
Lozano R, Vino A, Lozano C, Fisher SE, Deriziotis P (2015) A de novo FOXP1 variant in a patient with autism, intellectual disability and severe speech and language impairment. Eur J Human Genet 23:1702–1707
Mackes NK, Golm D, Sarkar S et al (2020) Early childhood deprivation is associated with alterations in adult brain structure despite subsequent environmental enrichment. Proc Natl Acad Sci USA 117:641–649
Makinodan M, Rosen KM, Ito S, Corfas G (2012) A critical period for social experience-dependent oligodendrocyte maturation and myelination. Science (new York, N.y.) 337:1357–1360
Mendizabal I, Berto S, Usui N et al (2019) Cell type-specific epigenetic links to schizophrenia risk in the brain. Genome Biol 20:135
Miguel PM, Pereira LO, Silveira PP, Meaney MJ (2019) Early environmental influences on the development of children’s brain structure and function. Dev Med Child Neurol 61:1127–1133
Miller DJ, Duka T, Stimpson CD et al (2012) Prolonged myelination in human neocortical evolution. Proc Natl Acad Sci USA 109:16480–16485
Miyata S, Hattori T, Shimizu S, Ito A, Tohyama M (2015) Disturbance of oligodendrocyte function plays a key role in the pathogenesis of schizophrenia and major depressive disorder. Biomed Res Int 2015:492367
Mohan H, An X, Xu XH et al (2023) Cortical glutamatergic projection neuron types contribute to distinct functional subnetworks. Nat Neurosci 26:481–494
Nagy C, Maitra M, Tanti A et al (2020) Single-nucleus transcriptomics of the prefrontal cortex in major depressive disorder implicates oligodendrocyte precursor cells and excitatory neurons. Nat Neurosci 23:771–781
Namba T, Dóczi J, Pinson A et al (2020) Human-specific ARHGAP11B acts in mitochondria to expand neocortical progenitors by glutaminolysis. Neuron 105:867-881.e9
Nambot S, Faivre L, Mirzaa G et al (2020) De novo TBR1 variants cause a neurocognitive phenotype with ID and autistic traits: report of 25 new individuals and review of the literature. Eur J Human Genet EJHG 28:770–782
Notwell JH, Heavner WE, Darbandi SF et al (2016) TBR1 regulates autism risk genes in the developing neocortex. Genome Res 26:1013–1022
O’Roak BJ, Deriziotis P, Lee C et al (2011) Exome sequencing in sporadic autism spectrum disorders identifies severe de novo mutations. Nat Genet 43:585–589
Pattabiraman K, Muchnik SK, Sestan N (2020) The evolution of the human brain and disease susceptibility. Curr Opin Genet Dev 65:91–97
Phan BN, Bohlen JF, Davis BA et al (2020) A myelin-related transcriptomic profile is shared by Pitt-Hopkins syndrome models and human autism spectrum disorder. Nat Neurosci 23:375–385
Polioudakis D, De La Torre-Ubieta L, Langerman J et al (2019) A Single-cell transcriptomic atlas of human neocortical development during mid-gestation. Neuron 103:785-801.e8
Quesnel-Vallières M, Weatheritt RJ, Cordes SP, Blencowe BJ (2019) Autism spectrum disorder: insights into convergent mechanisms from transcriptomics. Nat Rev Genet 20:51–63
Reh RK, Dias BG, Nelson III CA et al (2020) Critical period regulation across multiple timescales. Proc Natl Acad Sci USA 117:23242–23251
Reuter MS, Riess A, Moog U et al (2017) FOXP2 variants in 14 individuals with developmental speech and language disorders broaden the mutational and clinical spectrum. J Med Genet 54:64–72
Richardson WD, Kessaris N, Pringle N (2006) Oligodendrocyte wars. Nat Rev Neurosci 7:11–18
Rilling JK, Van Den Heuvel MP (2018) Comparative primate connectomics. Brain Behav Evol 91:170–179
Rivers LE, Young KM, Rizzi M et al (2008) PDGFRA/NG2 glia generate myelinating oligodendrocytes and piriform projection neurons in adult mice. Nat Neurosci 11:1392–1401
Roth TL, Raineki C, Salstein L et al (2013) Neurobiology of secure infant attachment and attachment despite adversity: a mouse model. Genes Brain Behav 12:673–680
Russo SJ, Nestler EJ (2013) The brain reward circuitry in mood disorders. Nat Rev Neurosci 14:609–625
Sachser N, Kaiser S, Hennessy MB (2013) Behavioural profiles are shaped by social experience: when, how and why. Philos Trans R Soc Lond B Biol Sci 368:20120344
Sakano H (2020) Developmental regulation of olfactory circuit formation in mice. Dev Growth Differ 62:199–213
Sanders SJ, He X, Willsey AJ et al (2015) Insights into autism spectrum disorder genomic architecture and biology from 71 risk loci. Neuron 87:1215–1233
Sapey-Triomphe LA, Reversat J, Lesca G et al (2020) A de novo frameshift pathogenic variant in TBR1 identified in autism without intellectual disability. Hum Genom 14:32
Satterstrom FK, Kosmicki JA, Wang J et al (2020) Large-scale exome sequencing study implicates both developmental and functional changes in the neurobiology of autism. Cell 180:568-584.e23
Šeda O, Šedová L, Včelák J, Vaňková M, Liška F, Bendlová B (2017) ZBTB16 and metabolic syndrome: a network perspective. Physiol Res 66:S357-s365
Sherwood CC, Stimpson CD, Raghanti MA et al (2006) Evolution of increased glia-neuron ratios in the human frontal cortex. Proc Natl Acad Sci USA 103:13606–13611
Shibata M, Pattabiraman K, Muchnik SK et al (2021) Hominini-specific regulation of CBLN2 increases prefrontal spinogenesis. Nature 598:489–494
Shin Yim Y, Park A, Berrios J et al (2017) Reversing behavioural abnormalities in mice exposed to maternal inflammation. Nature 549:482–487
Silk JB (2007) Social components of fitness in primate groups. Science (new York, N.y.) 317:1347–1351
Siper PM, De Rubeis S, Trelles MDP et al (2017) Prospective investigation of FOXP1 syndrome. Molecular Autism 8:57
Sonuga-Barke EJS, Kennedy M, Kumsta R et al (2017) Child-to-adult neurodevelopmental and mental health trajectories after early life deprivation: the young adult follow-up of the longitudinal English and Romanian Adoptees study. Lancet 389:1539–1548
Sorensen SA, Bernard A, Menon V et al (2015) Correlated gene expression and target specificity demonstrate excitatory projection neuron diversity. Cereb Cortex 25:433–449
Stessman HA, Xiong B, Coe BP et al (2017) Targeted sequencing identifies 91 neurodevelopmental-disorder risk genes with autism and developmental-disability biases. Nat Genet 49:515–526
Stoner R, Chow ML, Boyle MP et al (2014) Patches of disorganization in the neocortex of children with autism. N Engl J Med 370:1209–1219
Suliman BA, Xu D, WIlliams BR (2012) The promyelocytic leukemia zinc finger protein: two decades of molecular oncology. Front Oncol 2:74
Suzuki IK, Gacquer D, Van Heurck R et al (2018) Human-Specific NOTCH2NL genes expand cortical neurogenesis through delta/notch regulation. Cell 173:1370-1384.e16
Takesian AE, Hensch TK (2013) Balancing plasticity/stability across brain development. Prog Brain Res 207:3–34
Tasic B, Menon V, Nguyen TN et al (2016) Adult mouse cortical cell taxonomy revealed by single cell transcriptomics. Nat Neurosci 19:335–346
Tebbenkamp AT, Willsey AJ, State MW, Sestan N (2014) The developmental transcriptome of the human brain: implications for neurodevelopmental disorders. Curr Opin Neurol 27:149–156
Teicher MH, Samson JA, Anderson CM, Ohashi K (2016) The effects of childhood maltreatment on brain structure, function and connectivity. Nat Rev Neurosci 17:652–666
Teissier A, Le Magueresse C, Olusakin J et al (2020) Early-life stress impairs postnatal oligodendrogenesis and adult emotional behaviour through activity-dependent mechanisms. Mol Psychiatry 25:1159–1174
Thompson A, Shahidiani A, Fritz A et al (2020) Age-related differences in white matter diffusion measures in autism spectrum condition. Molecular Autism 11:36
Thomson AM (2010) Neocortical layer 6, a review. Front Neuroanat 4:13
Toma C, Torrico B, Hervás A et al (2014) Exome sequencing in multiplex autism families suggests a major role for heterozygous truncating mutations. Mol Psychiatry 19:784–790
Tomasello M (2020) The adaptive origins of uniquely human sociality. Philos Trans R Soc Lond B Biol Sci 375:20190493
Trutzer IM, García-Cabezas M, Zikopoulos B (2019) Postnatal development and maturation of layer 1 in the lateral prefrontal cortex and its disruption in autism. Acta Neuropathol Commun 7:40
Usui N, Co M, Konopka G (2014) Decoding the molecular evolution of human cognition using comparative genomics. Brain Behav Evol 84:103–116
Usui N, Araujo DJ, Kulkarni A et al (2017a) Foxp1 regulation of neonatal vocalizations via cortical development. Genes Dev 31:2039–2055
Usui N, Co M, Harper M, Rieger MA, Dougherty JD, Konopka G (2017b) Sumoylation of FOXP2 regulates motor function and vocal communication through purkinje cell development. Biol Psychiatry 81:220–230
Usui N, Iwata K, Miyachi T et al (2020) VLDL-specific increases of fatty acids in autism spectrum disorder correlate with social interaction. EBioMedicine 58:102917
Usui N, Berto S, Konishi A, Kondo M, Konopka G, Matsuzaki H, Shimada S (2021a) Zbtb16 regulates social cognitive behaviors and neocortical development. Transl Psychiatry 11:242
Usui N, Ono Y, Aramaki R, Berto S, Konopka G, Matsuzaki H, Shimada S (2021b) Early life stress alters gene expression and cytoarchitecture in the prefrontal cortex leading to social impairment and increased anxiety. Front Genet 12:754198
Usui N, Matsumoto-Miyai K, Koyama Y, Kobayashi Y, Nakamura Y, Kobayashi H, Shimada S (2022a) Social communication of maternal immune activation-affected offspring is improved by si-based hydrogen-producing agent. Front Psych 13:872302
Usui N, Tian X, Harigai W et al (2022b) Length impairments of the axon initial segment in rodent models of attention-deficit hyperactivity disorder and autism spectrum disorder. Neurochem Int 153:105273
Usui N, Kobayashi H, Shimada S (2023) Neuroinflammation and oxidative stress in the pathogenesis of autism spectrum disorder. Int J Mol Sci 24:5487
Van Heurck R, Bonnefont J, Wojno M et al (2023) CROCCP2 acts as a human-specific modifier of cilia dynamics and mTOR signaling to promote expansion of cortical progenitors. Neuron 111:65-80.e6
Vanderhaeghen P, Polleux F (2023) Developmental mechanisms underlying the evolution of human cortical circuits. Nat Rev Neurosci 24:213–232
Vegas N, Cavallin M, Kleefstra T et al (2018) Mutations in TBR1 gene leads to cortical malformations and intellectual disability. Eur J Med Genet 61:759–764
Vivanti G, Nuske HJ (2017) Autism, attachment, and social learning: Three challenges and a way forward. Behav Brain Res 325:251–259
Volterra A, Meldolesi J (2005) Astrocytes, from brain glue to communication elements: the revolution continues. Nat Rev Neurosci 6:626–640
Von Bartheld CS, Bahney J, Herculano-Houzel S (2016) The search for true numbers of neurons and glial cells in the human brain: a review of 150 years of cell counting. J Comp Neurol 524:3865–3895
Wang SS, Kloth AD, Badura A (2014) The cerebellum, sensitive periods, and autism. Neuron 83:518–532
Wang T, Hoekzema K, Vecchio D et al (2020) Large-scale targeted sequencing identifies risk genes for neurodevelopmental disorders. Nat Commun 11:4932
Wilkinson B, Grepo N, Thompson BL et al (2015) The autism-associated gene chromodomain helicase DNA-binding protein 8 (CHD8) regulates noncoding RNAs and autism-related genes. Transl Psychiatry 5:e568
Willsey AJ, Sanders SJ, Li M et al (2013) Coexpression networks implicate human midfetal deep cortical projection neurons in the pathogenesis of autism. Cell 155:997–1007
Willsey HR, Willsey AJ, Wang B, State MW (2022) Genomics, convergent neuroscience and progress in understanding autism spectrum disorder. Nat Rev Neurosci 23:323–341
Yao P, Lin P, Gokoolparsadh A, Assareh A, Thang MW, Voineagu I (2015) Coexpression networks identify brain region-specific enhancer RNAs in the human brain. Nat Neurosci 18:1168–1174
Ms Y, Zdunek S, Bergmann O et al (2014) Dynamics of oligodendrocyte generation and myelination in the human brain. Cell 159:766–774
Zhao S, Hu X, Park J et al (2007) Selective expression of LDLR and VLDLR in myelinating oligodendrocytes. Dev Dyn 236:2708–2712
Zhou B, Zhu Z, Ransom BR, Tong X (2021) Oligodendrocyte lineage cells and depression. Mol Psychiatry 26:103–117
Acknowledgements
I thank Dr. Sén Takeda and Selection committee of Encouragement Award of the Japanese Association of Anatomist for selection of Encouragement Award of the Japanese Association of Anatomist and providing writing opportunity. I also thank Drs. Shoichi Shimada, Hideo Matsuzaki, Genevieve Konopka, Hirohide Takebayashi, and Kazuhiro Ikenaka for supervising, Dr. Miyuki Doi for helping illustrations, and colleagues and collaborators for contributions.
Funding
Open access funding provided by Osaka University. This work was supported by the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (B) (23H02837); JSPS Grant-in-Aid for Scientific Research (C) (20K06872); JSPS Grant-in-Aid for Early-Career Scientists (18K14814); Uehara Memorial Foundation; Takeda Science Foundation; Naito Foundation; Mochida Memorial Foundation for Medical and Pharmaceutical Research; Inamori Foundation; SENSHIN Medical Research Foundation; Osaka Medical Research Foundation for Intractable Diseases.
Author information
Authors and Affiliations
Contributions
NU: conceptualization, writing—original draft, writing—review and editing, visualization, project administration, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Usui, N. Possible roles of deep cortical neurons and oligodendrocytes in the neural basis of human sociality. Anat Sci Int 99, 34–47 (2024). https://doi.org/10.1007/s12565-023-00747-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-023-00747-1