Abstract
Probiotics are widely recognized for their ability to prevent and therapy antibiotic-associated diarrhea (AAD). This study was designed to evaluate Lactiplantibacillus plantarum ELF051 ability to prevent colon inflammation and its effect on gut microbial composition in a mouse model of AAD. The mice were intragastrically administered triple antibiotics for 7 days and then subjected to L. plantarum ELF051 for 14 days. The administration of L. plantarum ELF051 ameliorated the pathological changes in the colon tissue, downregulated interleukin (IL)-1β and tumor necrosis factor (TNF)-α, and upregulated IL-10, and increased the intestinal short-chain fatty acids (SCFAs) level. Lactiplantibacillus plantarum ELF051 also regulated the Toll-like receptor/myeloid differentiation primary response 88/nuclear factor kappa light chain enhancer of activated B cells (TLR4/MyD88/NF-κB) and the phosphatidylinositol 3-kinase/protein kinase B/ NF-κB (PI3K/AKT/ NF-κB) inflammatory signaling pathways. 16S rRNA analyses showed that L. plantarum ELF051 increased the abundance and diversity of gut bacteria, restoring gut microbiota imbalance. A Spearman’s rank correlation analysis showed that lactobacilli are closely associated with inflammatory markers and SCFAs. This work demonstrated that L. plantarum ELF051 can attenuate antibiotic-induced intestinal inflammation in a mouse AAD model by suppressing the pro-inflammatory response and modulating the gut microbiota.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antibiotic-associated diarrhea (AAD) is defined as otherwise unexplained diarrhea that occurs in association with the administration of antibiotics. Most known antibiotics could induce AAD, with ampicillin, cephalosporins, and clindamycin being the most serious offenders, leading to an incidence of AAD ranging from 5 to 30% when any of the above antibiotics are used [1, 2]. Antibiotic therapy may induce AAD by disrupting the gut microbiota, altering the intestinal short-chain fatty acids (SCFAs) content, and changing the intestinal structure and barrier function [3]. Untreated AAD may result in pseudomembranous enteritis which has 15–24% mortality [4]. Treatments for AAD include antibiotic withdrawal, targeted antibiotic therapy, and oral microbiotics. However, none of these is very effective, and some of them might even be harmful [5]. Therefore, a new and effective AAD treatment is urgently required.
Probiotics have been studied for their health promoting effects, including influencing the composition and function of the gut microbiota, modulating the immune response, exhibiting antimutagenic effect and anti-cancer properties, and lowering serum cholesterol [6]. In recent years, increasing evidence has shown that probiotics also have beneficial impact on alleviating symptoms of AAD. Numerous randomized, double-blind, controlled clinical trials have provided evidence that probiotics, including Bifidobacterium lactis, B. animalis, Lactobacillus casei, L. plantarum, and L. rhamnosus, exert positive effects on modulating the intestinal microbiota and preventing and treating AAD [7,8,9]. Animal studies have also demonstrated that consumption of a mixture of Lactobacilli species (JUP-Y4) not only promoted recovery from antibiotic-induced gut dysbiosis but also enhanced the function of the gut barrier, and lowered levels of circulating endotoxin in mice [10]. The specific molecular mechanisms underlying the treatment and prevention of AAD by probiotics primarily involve the following aspects: (i) Modulating the gut microbiome. As direct evidence, many results show that AAD in mice is related to the changes in normal intestinal microbiota, which is mainly manifested by the reduction of beneficial bacteria and the increase of potential pathogens. The administration of probiotics has been proved to regulate the disorder of gut microbiota [11]. (ii) Enhancing the intestinal immune response. Probiotics have been proved to enhance humoral immunity response by increasing the cells secreting IgG, IgM, and IgA [12, 13]. (iii) Improving intestinal barrier function. Probiotics can increase mucin secretion, upregulate ZO-1 and occludin protein synthesis, thus repairing intestinal permeability caused by AAD [14]. (iv) Maintaining normal levels of SCFAs. Antibiotic-mediated gut microbiome remodeling results in significant alterations to intestinal metabolomes. Probiotic supplementation can significantly increase SCFAs, contributing to the improvement of intestinal health [15].
AAD is often accompanied by systemic inflammation, characterized by an increase in pro-inflammatory cytokines and a decrease in anti-inflammatory cytokines, indicating that anti-inflammatory may be a potential mechanism for probiotics to alleviate AAD [16]. Previous studies have confirmed that gastric perfusion of L. plantarum 2–33 into AAD mice can significantly increase the levels of anti-inflammatory factors IL-4 and IL-10 and reduce the levels of pro-inflammatory factors TNF-α and IFN-γ. Similar results were observed with probiotics B. animalis subsp. lactis XLTG11 [15] and Bacillus subtilis DU-106 [17] in regulating cytokine levels in diarrhea mice. However, while existing research has primarily focused on cytokine levels, a deeper understanding of the mechanisms underlying intestinal inflammation in AAD is lacking. Therefore, this study is mainly to carry out the protective effect of probiotics on intestinal inflammation in the development of AAD.
Lactiplantibacillus plantarum ELF051 is a potential probiotic isolated from Kimchi. Our previous research has proved that L. plantarum ELF051 significantly inhibits the growth of Clostridium difficile (data not shown), indicating its potential therapeutic effect on AAD. However, the precise mechanism underlying of L. plantarum ELF051 exerts effects in AAD mice remains unclear. Therefore, the present study was conducted to investigate the potential mechanisms of L. plantarum ELF051 in alleviating the progression in a AAD mouse model, by reducing colonic pathological changes, restoring SCFAs levels, modulating inflammatory cytokines, regulating inflammatory signaling pathways, and correcting gut microbiota imbalance.
Materials and Methods
Bacterial Strains and Culture Methods
Lactiplantibacillus plantarum ELF051 was incubated in De Man, Rogosa, and Sharpe (MRS) medium at 37 °C for 17 h, centrifuged at 3000 × g for 15 min, washed thrice with phosphate-buffered saline (PBS; pH 7.4), and resuspended in saline and the viable count of the bacterial solution was adjusted to 1.0 × 109 CFU/mL, and kept at 4 °C.
Animal Experiment Design
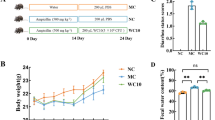
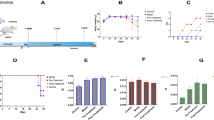
Male C57BL/6 mice (20 ± 2 g, n = 30) were obtained from Liaoning Changsheng Biotechnology Co., Ltd. (Benxi, China). All mice were adaptively fed under standard conditions over 7 days. Mice were randomly divided into three groups (n = 10/group): control, model, and ELF051 groups. From day 1 to day 7, the control group received physiological saline via gavage. Meanwhile, the model and ELF051 groups were administered a mixed antibiotic solution consisting of 500 mg/kg amoxicillin (Henan Puxin Biotechnology Co., Ltd; Henan, China), 400 mg/kg clindamycin (Anhui Lianyi Pharmaceutical Co. Ltd; Fuyang, Anhui, China), and 350 mg/kg streptomycin (Chongqing Xinjiheng Pharmaceutical Co. Ltd; Chongqing, China) via gavage with a volume of 0.2 mL [17]. From day 8 to day 21, the ELF051 group received L. plantarum ELF051 at a dosage of 0.1 mL/10 g via gavage, while both the control and model groups received an equal volume of saline. All animal experiments complied with the institutional animal care regulations of Jilin Academy of Agricultural Sciences (approval number: SCXΚ2020-0001).
After the final treatment, the mice were fasted for 12 h, and then anesthetized with ether. Subsequently, blood, feces, cecum, and colon samples were collected. The mice’s feces, cecal contents, and a portion of the colon were promptly frozen in liquid nitrogen and stored at − 80 °C for subsequent analysis. The remaining colon samples were preserved in Cano’s fixative for further investigations.
Histological Analysis
The fixed colons were dehydrated with ethanol, embedded in paraffin, cut into sections 4 μm thick, and stained with hematoxylin and eosin (H&E) [18]. Pathological changes in colonic tissues were detected by light microscopy (Nikon, Tokyo, Japan) at × 200.
Biochemical Analysis
The mouse colon tissue was mixed with PBS in a weight (g)/volume (mL) ratio of 1:9, and thoroughly ground to obtain colon tissue homogenate. The homogenate was then centrifuged at 10,000 × g for 8 min, and the supernatant was collected for further analysis [19]. The levels of TNF-α, IL-1β, and IL-10 in the homogenate supernatant were determined using an ELISA kit manufactured by Jiangsu Meibiao Biotechnology Co. Ltd (Jiangsu, China). The specific operation was conducted by instruction manual, and the optical densities were determined on a Bio-Rad microplate reader (Bio-Rad, USA) at a wavelength of 450 nm. The levels of TNF-α, IL-1β, and IL-10 were calculated according to the standard curve.
SCFAs Analysis
The cecal contents were placed in a 2-mL centrifuge tube containing 50 μL of 15% (v/v) phosphoric acid, 100 μL of 125 μg/mL internal standard (4-methylpentanoic acid), and 400 μL ether, centrifuged for 1 min, and centrifuged again at 10,000 × g and 4 °C for 10 min. The supernatant was transferred to gas-phase vials and analyzed by gas chromatography-mass spectrometry (GC–MS) (Thermo Fisher Scientific, USA) [20]. For the GC, the samples were injected and then separated using He as the carrier gas (flow rate: 1.0 mL/min, split ratio: 10:1), the inlet temperature was 250 °C. The initial temperature is 90 °C, and the temperature was raised to 120 °C at an increment of 10 °C/min; then, the temperature was raised to 150 °C at an increment of 5 °C/min, raised to 250 °C at an increment of 25 °C/min, and held at 250 °C for 2 min. The MS was scanned in SIM mode at 70 eV [21].
Western Blotting
The total protein was extracted from the mouse colon tissue according to the method of Wang et al. [22]. The protein concentration was measured by the bicinchoninic acid (BCA) assay, and the same concentration was used in all subsequent experiments. After electrophoresis, the sample was transferred to a membrane made of polyvinylidene fluoride (PVDF). These were then blocked with 3% bovine serum albumin (BSA) at 25 °C for 1 h. The rabbit antibodies used included anti-β-actin (Solarbio, China), anti-TLR4 (No. bs-20595R; Bioss Antibodies Inc., Woburn, MA, USA), anti-MyD88 (No. GTX112987; GeneTex In c., Irvine, CA, USA), anti-PI3K (No. ab40776; Abcam, Cambridge, UK), anti-p-PI3K (No. ab278545; Abcam), anti-Akt (No. N3C2; GeneTex Inc.), anti-Akt (No. GTX128414; GeneTex Inc.), anti-NF-κB (No. ab16502; Abcam), and anti-IκBα (No. ab32518; Abcam). The membrane was incubated at 4 °C overnight, subjected to horseradish peroxidase (HRP)-coupled secondary antibody, and kept in the dark for 1 h. The bands were detected with Cytiva ImageQuant™ LAS 4000 (Cytiva Life Sciences, Marlborough, MA, USA), and the proteins were expressed as a percentage of β-actin.
Gut Microbial Analysis
Total fecal DNA was extracted with an OMEGA Soil DNA Kit (ONorcross, GA, USA), and 16S rRNA of bacteria was amplified using 338F primer (5′-ACTCCTACGGGAGGCAGCA-3′) and 806R primer (5′-GGACTACHVGGGTWTCTAAT-3′), which were region-specific primers of V3-V4 bacteria. QIIME2 (https://qiime2.org/) and R v. 3.2.0 (Vienna, Austria) were used to analyze the diversity and abundance of the gut microbiota based on relative operational taxonomic units (OTU) at 97% similarity levels. The α-diversity was analyzed using the Chao1, Shannon, and Simpson indices. The β-diversity was evaluated by principal coordinate analysis (PCoA). The relative abundance of the phylum level was used to determine the bacterial community structure, and the difference in genus levels among groups was compared via a heat map.
Statistical Analysis
The data were presented as means ± standard deviation (SD). They were analyzed with SPSS v. 20.0 (IBM Corp., Armonk, NY, USA), and the graphs were plotted with Origin v. 8.0 (OriginLab, Northampton, MA, USA). Spearman analysis was conducted to investigate the correlation among SCFAs, inflammatory factors, and microorganisms to evaluate the relationship between the gut microbiota and SCFAs and anti-inflammation. Statistically significant differences were evaluated by one-way analysis of variance (ANOVA) followed by the post hoc Bonferroni correction. Significance was defined at the p < 0.05 level.
Results
L. plantarum ELF051 Ameliorated the Pathological Changes in the Colon
H&E staining of the colon tissues revealed distinct structural differences among the experimental groups (Fig. 1). In the control group, the mucosal layer appeared structurally intact with well-preserved mucosal epithelial cells and organized intestinal glands. In contrast, the model group exhibited significant damage, characterized by shedding of mucosal epithelial cells, loosely arranged intestinal glands, and indications of slight edema and inflammatory cell infiltration. However, following the administration of the L. plantarum ELF501, the degree of inflammatory cell infiltration decreased and edema was alleviated. Additionally, the loosely arranged intestinal gland structures became tightly arranged. Therefore, L. plantarum ELF501 mitigated the intestinal mucosal structural damage caused by antibiotics.
Effect of L. plantarum ELF051 on the pathological changes of colon tissue in AAD mice. H&E staining were used to observe the morphological changes in the colon. Black arrow: margin of the intestinal gland was widened; red arrow: connective tissue loose (40 ×, scale bars: 500 µm; 200 ×, scale bars: 100 µm)
L. plantarum ELF051 Regulates Inflammatory Cytokines in Serum
We measured the TNF-α, IL-1β, and IL-10 levels to determine the impact of L. plantarum ELF051 on inflammation (Fig. 2). The TNF-α and IL-1β levels were higher, and the IL-10 level was markedly lower in the model group than in the control group. The TNF-α and IL-1β levels were significantly (p < 0.05) lower, while the IL-10 level was higher in the ELF051 group than in the model group.
L. plantarum ELF051 Increased the SCFAs Content
The effect of L. plantarum ELF051 on the content of SCFAs in mouse cecal contents was evaluated using GC–MS method. Based on the data presented in Table 1, the model group exhibited significantly lower levels of acetic acid, propionic acid, butyric acid, and total SCFAs showing a significant decrease compared to the control group (p < 0.05). After administration of L. plantarum ELF051 via gavage in AAD mice, the levels of acetic acid, propionic acid, butyric acid, hexanoic acid, and total SCFAs were increased. Among them, the levels of propionic acid, butyric acid, and total SCFAs showed significant difference when compared to the model group (p < 0.05).
L. plantarum ELF051 Alleviated Inflammation by Inhibiting the Expression of Key Proteins in the TLR4/MyD88/NF-κB Signaling Pathway
In order to elucidate the anti-inflammatory effect of L. plantarum ELF051 in AAD mice model, we employed Western blot analysis to assess the expression levels of key proteins in the TLR4/MyD88/NF-κB signaling pathway in colonic tissues. Figure 3 shows that TLR4, MyD88, NF-κB, and IκBα were markedly upregulated in the model group compared to the control group but significantly downregulated in the ELF051 group (17.00%, 18.53%, 21.90%, and 14.37%, respectively) compared to the model group. Thus, it can be observed that L. plantarum ELF051 can alleviate the inflammatory response in AAD mice by inhibiting the activation of the TLR4/MyD88/NF-κB signaling pathway.
L. plantarum ELF051 inhibits the expression of key proteins in the TLR4/MyD88/NF-κB signaling pathway. Protein bands were shown in (A), TLR4/β-actin ratio (B), MyD88/β-actin ratio (C), IκBα/β-actin ratio (D), and NF-κB/β-actin ratio (E). #p < 0.05, ##p < 0.01 vs. control; *p < 0.05, **p < 0.01 vs. model. Data were presented as the means ± SD, n = 6
L. plantarum ELF051 Alleviated Inflammation by Inhibiting the Expression of Key Proteins in the PI3Κ/AΚT/NF-κB Signaling Pathway
We investigated whether the PI3Κ/AΚT/NF-κB pathway is involved in the anti-inflammatory mechanism of L. plantarum ELF051 in AAD mice. For this purpose, we compared the expression levels of key proteins in the PI3K/AKT/NF-κB signaling pathway in colon tissue homogenates from different groups. Figure 4 shows that the key proteins of p-PI3Κ, p-AΚT, and NF-κB were significantly upregulated in the model group compared to the control group (p < 0.05 or p < 0.01). In contrast, the p-PI3Κ/PI3Κ, p-AΚT/AΚT, and NF-κB levels were 29.08%, 15.62%, and 21.90% lower in the ELF051 group than in the model group (p < 0.05 or p < 0.01). Due to the inhibitory effect of L. plantarum ELF051 on the phosphorylation and activation of the PI3K/AKT/NF-κB signaling pathway in colon tissue samples, it is inferred that L. plantarum ELF051 exhibits a positive anti-inflammatory effect in AAD mice.
L. plantarum ELF051 inhibits the expression of key proteins in the PI3K/AT/NF-κB signaling pathway. Protein bands were shown in (A), p-PI3K/PI3Κ ratio (B), p-AKT/AΚT ratio (C), and NF-κB/β-actin ratio (D). Data were analyzed by one-way ANOVA: #p < 0.05, ##p < 0.01 vs. control; *p < 0.05, **p < 0.01 vs. model (n = 6 per group). mean ± SD. The β-actin was used as a standard control
L. plantarum ELF051 Improved Gut Microbiota Dysbiosis in AAD Mice
Fecal analysis by 16S rRNA gene sequencing of the V3-V4 region disclosed that L. plantarum ELF051 significantly altered gut microbiota richness and diversity compared to the model group. The Chao 1 index indicated that gut microbiota species richness was markedly higher in the ELF051 group than in the model group (Fig. 5A). The Shannon and Simpson indices (Fig. 5B and C) revealed that gut microbiota diversity was higher in the ELF051 group than in the model group. To assess the similarity of gut microbiota in fecal samples, we utilized β-diversity analysis through PCoA. In the PCoA analysis (Fig. 5E), we observed a distinct separation of the model group from the other two groups; the ELF051 group displayed a closer proximity to the control group. The main gut bacterial phyla in all three groups were Bacillota, Bacteroidota, and Pseudomonadota (Fig. 5F). In comparison to the control group, antibiotic treatment resulted in a significant decrease in the relative abundance of Bacillota, while increasing the relative abundance of Bacteroidota and Pseudomonadota. However, following treatment with L. plantarum ELF051, the levels of Bacteroidota and Pseudomonadota were decreased, while the abundance of Bacillota showed a significant increase compared to the model group. According to Fig. 5D, treatment with L. plantarum ELF051 resulted in a decrease in the abundance of Allobaculum, Desulfovibrio, and Akkermansia compared to the model group. Conversely, the abundance of Lactobacillus, Prevotella, and Oscillospira bacteria showed a significant increase following L. plantarum ELF051 treatment. These findings suggest that treatment with L. plantarum ELF051 may potentially improve gut microbiota dysbiosis in AAD mice.
Effects of L. plantarum ELF051 on the diversity and composition of the gut microbiota in AAD mice. Chao1, Shannon and Simpson indexes (A, B, and C). Heat map at the genus level (D). PCoA analysis based on weighted UniFrac phylogenetic distance matrices (E). Species compositions at the phylum level (F)
The Correlation Between Microbiota and Inflammatory Factors and Metabolites
Spearman’s rank correlation test generated a heatmap that intuitively visualized and disclosed strong associations among microbial species, inflammatory factors, inflammation-related proteins, SCFAs, and others (Fig. 6). Lactobacillus abundance was negatively but non-significantly correlated with TNF-α and IL-1β. Conversely, Lactobacillus abundance was positively related to IL-10 (R = 0.68; p = 0.0018). Lactobacillus abundance was negatively related to TLR4 (R = − 0.509; p = 0.031), MyD88 (R = − 0.719; p = 0.00077), NF-κB (R = − 0.742; p = 0.0004), IκBα (R = − 0.664; p = 0.0027), p-PI3K/PI3K (R = − 0.682; p = 0.0018), and p-Akt/Akt (R = − 0.474; p = 0.047). Lactobacillus was also positively related to the acetic acid content (R = 0.496; p = 0.036). SCFAs detected were closely associated with Lactobacillus, Oscillospira, and Prevotella. It can be inferred from the foregoing data that L. plantarum ELF051 may downregulate the pro-inflammatory factors, increase the inflammation-related protein content, and elevate the SCFAs concentrations by regulating the composition of the intestinal microbiota in AAD mice.
Discussion
AAD has a complex pathogenesis. Antibiotic misuse or overuse can alter the gut microbiota and SCFAs profile, induce inflammation, and cause AAD [23]. In the present study, a mouse AAD model was constructed through intragastric amoxicillin, clindamycin, and streptomycin administration. This treatment altered the intestinal structure and microbiota and changed the gut SCFAs content. It also upregulated pro-inflammatory and downregulated anti-inflammatory cytokines and lowered the inflammation-related protein content in the intestine. Intragastric administration of the probiotic L. plantarum ELF051 improved AAD symptoms. Therefore, ELF051 could be efficacious in AAD therapy.
SCFAs are important end products of gut microbiota metabolism, and they are generated through the fermentation of indigestible carbohydrates. SCFAs may have potential anti-inflammatory and reparative effects on intestinal damage [24]. The present study showed that administration of L. plantarum ELF051 increased the levels of SCFAs in the intestine. Liu et al. found that Bacillus coagulans upregulated the levels of SCFAs in mice with colitis, and the increase in SCFAs played an anti-inflammatory role by regulating intestinal immune activity, and enhancing the function of the epithelial barrier [25]. Therefore, we infer that the restoration of intestinal pathological damage and a further alleviation of AAD-related symptoms by L. plantarum ELF051 are related to the increase of SCFAs levels. In addition, we observed a positive correlation between the amount of Bacteroidota and Bacillota and the concentrations of propionic and butyric acid in the gut (Fig. 5F). It is reported that butyric acid is produced by Bacillota and acetic acid is produced by Bacteroidota [26]. Research indicates that butyric acid is predominantly produced by Bacillota in the intestine and plays a vital role in maintaining intestinal health. Butyric acid has been shown to enhance intestinal permeability and promote intestinal health, contributing to the alleviation of diarrhea [27]. Treatment with L. plantarum has been shown to increase the levels of butyric acid. Shi et al. showed that treatment with L. casei CGMCC 12435 increased butyric acid concentration [28]. Therefore, L. plantarum ELF051 has the potential to improve AAD by enhancing the production of SCFAs.
Inflammation is a natural defense mechanism against harmful stimuli. Pro-inflammatory cytokines in general, and TNF-α in particular, are produced in abundance during this process. TNF-α activates the NF-κB pathway and upregulates other pro-inflammatory cytokines, thereby exacerbating the inflammatory response [29]. The present work demonstrated that the probiotic L. plantarum ELF051 significantly downregulated TNF-α and IL-1β while upregulating IL-10 in the mouse colons. These findings were consistent with those reported in previous studies. L. plantarum HNU082 and B. adolescentis also attenuated the inflammatory response in a DSS-induced colitis by downregulating TNF-α and IL-1β while upregulating IL-10 [30, 31]. Hence, the ability of L. plantarum ELF051 to decrease pro-inflammatory and increase anti-inflammatory factors may explain its therapeutic efficacy against AAD. A growing body of evidence indicates that the TLR4/MyD88/NF-κB pathway is implicated in the amelioration of diarrhea. The TLR4 signaling pathway is subdivided into the MyD88-dependent and MyD88-independent pathways [32]. Tong et al. showed that L. rhamnosus GG play a key role in the DSS-induced inflammatory response through its downregulation of the TLR4-MyD88 axis [33]. The MyD88-independent signaling pathway generates abundant interferon (IFN)-γ which is also associated with colonic inflammatory activity. Thus, we analyzed the MyD88-dependent pathway to elucidate the mechanism by which L. plantarum ELF051 ameliorates AAD. L. plantarum ELF051 significantly downregulated TLR4, MyD88, NF-κB, and IκBα. An earlier experiment demonstrated that L. rhamnosus GG Effector Protein HM0539 substantially mitigated inflammation and downregulated IL-6, IL-1β, and TNF-α in murine colitis. IL-6 and TNF-α are important mediators of the TLR4/MyD88/NF-κB pathway [34]. Another study showed that L. plantarum AR113 alleviated dextran sulfate sodium (DSS)–induced colitis in mice by downregulating the TLR4/MyD88/NF-κB pathway in the colon [35]. The results of this study indicated that in AAD mice, there was an increase in the expression of PI3K and AKT proteins, activating NF-κB, leading to an increase in the levels of phosphorylated IκBα and p65, thereby promoting the secretion of cytokines IL-1β, IL-6, and TNF-α. These cytokines have pro-inflammatory effects and contribute to the development of diarrhea by causing intestinal inflammation [36, 37]. However, after treatment with L. plantarum ELF051, NF-κB activation was inhibited, and regulated the PI3K/AKT/NF-κB signaling pathway, leading to a reduction in the secretion of inflammatory cytokines. Consist with previous study, gavage with the probiotic powder VSL#3 also exerted anti-inflammatory effects by inhibiting the PI3K/AKT and NF-κB pathways in the DSS induced rat colitis model [38].
Antibiotic use may disrupt the gut microbiota, leading to an increase in the abundance of harmful bacterial pathogens such as Actinomycetota and Pseudomonadota. Genera within these phyla are known to cause various types of infections in humans, animals, and plants, and in severe cases, affect host immunity [39,40,41]. In this study, the administration of L. plantarum ELF051 resulted in notable alterations in the abundance and diversity of the intestinal microbiota in mice. Specifically, the abundance of Pseudomonadota, Bacteroidota, and Actinomycetota decreased, while the abundance of Bacillota exhibited a reversal. These findings align with previous published reports [17]. At the genus level, the administration of L. plantarum ELF051 resulted in an increase in the abundance of Lactobacillus, Oscillospira, and Prevotella, while decreasing the relative abundance of Allobaculum, Desulfovibrio, and Akkermansia, compared to the model group. Guo et al. reported that antibiotic-associated diarrhea was induced by ceftriaxone in mice, the relative abundance of Lactobacillus and Prevotella was increase after gavage with apple juice fermented by L. plantarum CICC21809, which is consistent with the results of this study [42]. A Pearson’s correlation analysis disclosed the associations among microbiota and inflammatory mediators and SCFAs (Fig. 6), and the results indicated a close correlation between Lactobacillus and inflammatory mediators and SCFAs. Our findings suggest that increasing abundances of Lactobacillus spp. and the content of lactic acid and the other SCFAs they produced help establish and maintain homeostasis in the gut microbiota environment and attenuate the intestinal inflammatory response.
Conclusion
In conclusion, the current research findings suggest that the probiotic L. plantarum ELF051 has positive effects on AAD. L. plantarum ELF051 by improving the tissue morphology, normalizing the levels of inflammatory cytokines, increasing the production of SCFAs in the colon, and modulating the gut microbiota. In addition, L. plantarum ELF051 regulates the TLR4/MyD88/NF-κB and PI3K/AKT/NF-κB signaling pathways, thereby alleviating inflammation. These findings may provide a novel therapeutic approach for the prevention and treatment of AAD.
Data Availability
All 16 s rRNA Illumina amplicon sequencing data provided in this study can be publicly obtained in the Sequence Read Archive (SRA) of The National Center for Biotechnology Information (NCBI) under the accession number SRP PRJNA973244.
References
Agamennone V, Krul CAM, Rijkers G, Kort R (2018) A practical guide for probiotics applied to the case of antibiotic-associated diarrhea in The Netherlands. BMC Gastroenterol 18:103. https://doi.org/10.1186/s12876-018-0831-x
Silverman MA, Konnikova L, Gerber JS (2017) Impact of antibiotics on necrotizing enterocolitis and antibiotic-associated diarrhea. Gastroenterol Clin North Am 46:61–76. https://doi.org/10.1016/j.gtc.2016.09.010
Cote GA, Buchman AL (2006) Antibiotic-associated diarrhoea. Expert Opin Drug Saf 5:361–372. https://doi.org/10.1517/14740338.5.3.361
Vangay P, Ward T, Gerber JS, Knights D (2015) Antibiotics, pediatric dysbiosis, and disease. Cell Host Microbe 17:553–564. https://doi.org/10.1016/j.chom.2015.04.006
Bartlett JG (1996) Management of Clostridium difficile infection and other antibiotic-associated diarrhoeas. Eur J Gastroenterol Hepatol 8:1054–1061. https://doi.org/10.1097/00042737-199611000-00005
Simon E, Călinoiu LF, Mitrea L, Vodnar DC (2021) Probiotics, prebiotics, and synbiotics: implications and beneficial effects against irritable bowel syndrome. Nutrients. https://doi.org/10.3390/nu13062112
Ma Y, Yang JY, Peng X, Xiao KY, Xu Q, Wang C (2020) Which probiotic has the best effect on preventing Clostridium difficile-associated diarrhea? A systematic review and network meta-analysis. J Dig Dis 21:69–80. https://doi.org/10.1111/1751-2980.12839
Lukasik J, Dierikx T, Besseling-van der Vaart I, de Meij T, Szajewska H (2022) Multispecies probiotic for the prevention of antibiotic-associated diarrhea in children: a randomized clinical trial. JAMA Pediatr 176:860–866. https://doi.org/10.1001/jamapediatrics.2022.1973
Merenstein D, Fraser CM, Roberts RF, Liu T, Grant-Beurmann S, Tan TP, Smith KH, Cronin T, Martin OA, Sanders ME, Lucan SC, Kane MA (2021) Bifidobacterium animalis subsp. lactis BB-12 protects against antibiotic-induced functional and compositional changes in human fecal microbiome. Nutrients. https://doi.org/10.3390/nu13082814
Shi Y, Zhao X, Zhao J, Zhang H, Zhai Q, Narbad A, Chen W (2018) A mixture of Lactobacillus species isolated from traditional fermented foods promote recovery from antibiotic-induced intestinal disruption in mice. J Appl Microbiol 124:842–854. https://doi.org/10.1111/jam.13687
Shao H, Zhang C, Xiao N, Tan Z (2020) Gut microbiota characteristics in mice with antibiotic-associated diarrhea. BMC Microbiol 20:313. https://doi.org/10.1186/s12866-020-01999-x
Guo S, Yan W, McDonough SP, Lin N, Wu KJ, He H, Xiang H, Yang M, Moreira MA, Chang YF (2015) The recombinant Lactococcus lactis oral vaccine induces protection against C. difficile spore challenge in a mouse model. Vaccine 33:1586–1595. https://doi.org/10.1016/j.vaccine.2015.02.006
Sullivan A, Nord CE (2005) Probiotics and gastrointestinal diseases. J Intern Med 257:78–92. https://doi.org/10.1111/j.1365-2796.2004.01410.x
Li W, Zhang S, Wang Y, Bian H, Yu S, Huang L, Ma W (2023) Complex probiotics alleviate ampicillin-induced antibiotic-associated diarrhea in mice. Front Microbiol 14:1156058. https://doi.org/10.3389/fmicb.2023.1156058
Xu B, Liang S, Zhao J, Li X, Guo J, Xin B, Li B, Huo G, Ma W (2022) Bifidobacterium animalis subsp. lactis XLTG11 improves antibiotic-related diarrhea by alleviating inflammation, enhancing intestinal barrier function and regulating intestinal flora. Food Funct 13:6404–6418. https://doi.org/10.1039/d1fo04305f
Hu JS, Huang YY, Kuang JH, Yu JJ, Zhou QY, Liu DM (2020) Streptococcus thermophiles DMST-H2 promotes recovery in mice with antibiotic-associated diarrhea. Microorganisms. https://doi.org/10.3390/microorganisms8111650
Huang D, Chen Y, Chen H, Deng X, Huang J, Lu S, Li P, Du B (2022) Supplementation of Bacillus sp. DU-106 alleviates antibiotic-associated diarrhea in association with the regulation of intestinal microbiota in mice. Probiotics Antimicrob Proteins 14:372–383. https://doi.org/10.1007/s12602-022-09906-8
Yang L, Zhang Q, Huang J, Liu D, Lan Y, Yuan L, Chen Q (2021) Xianglian Pill ameliorates antibiotic-associated diarrhea by restoring intestinal microbiota and attenuating mucosal damage. J Ethnopharmacol 264:113377. https://doi.org/10.1016/j.jep.2020.113377
Bie N, Duan S, Meng M, Guo M, Wang C (2021) Regulatory effect of non-starch polysaccharides from purple sweet potato on intestinal microbiota of mice with antibiotic-associated diarrhea. Food Funct 12:5563–5575. https://doi.org/10.1039/d0fo03465g
Han X, Guo J, You Y, Yin M, Ren C, Zhan J, Huang W (2018) A fast and accurate way to determine short chain fatty acids in mouse feces based on GC-MS. J Chromatogr B Analyt Technol Biomed Life Sci 1099:73–82. https://doi.org/10.1016/j.jchromb.2018.09.013
Hsu YL, Chen CC, Lin YT, Wu WK, Chang LC, Lai CH, Wu MS, Kuo CH (2019) Evaluation and optimization of sample handling methods for quantification of short-chain fatty acids in human fecal samples by GC-MS. J Proteome Res 18:1948–1957. https://doi.org/10.1021/acs.jproteome.8b00536
Wang L, Zhao Z, Zhao L, Zhao Y, Yang G, Wang C, Gao L, Niu C, Li S (2022) Lactobacillus plantarum DP189 reduces α-SYN aggravation in MPTP-induced Parkinson’s disease mice via regulating oxidative damage, inflammation, and gut microbiota disorder. J Agric Food Chem 70:1163–1173. https://doi.org/10.1021/acs.jafc.1c07711
Mekonnen SA, Merenstein D, Fraser CM, Marco ML (2020) Molecular mechanisms of probiotic prevention of antibiotic-associated diarrhea. Curr Opin Biotechnol 61:226–234. https://doi.org/10.1016/j.copbio.2020.01.005
Guo C, Wang Y, Zhang S, Zhang X, Du Z, Li M, Ding K (2021) Crataegus pinnatifida polysaccharide alleviates colitis via modulation of gut microbiota and SCFAs metabolism. Int J Biol Macromol 181:357–368. https://doi.org/10.1016/j.ijbiomac.2021.03.137
Liu Z, Jiang Z, Zhang Z, Liu T, Fan Y, Liu T, Peng N (2022) Bacillus coagulans in combination with chitooligosaccharides regulates gut microbiota and ameliorates the DSS-induced colitis in mice. Microbiol Spectr 10:e0064122. https://doi.org/10.1128/spectrum.00641-22
Louis P, Flint HJ (2017) Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 19:29–41. https://doi.org/10.1111/1462-2920.13589
Rivière A, Selak M, Lantin D, Leroy F, De Vuyst L (2016) Bifidobacteria and butyrate-producing colon bacteria: importance and strategies for yheir stimulation in the human gut. Front Microbiol 7:979. https://doi.org/10.3389/fmicb.2016.00979
Shi Y, Kellingray L, Le Gall G, Zhao JX, Zhang H, Narbad A, Zhai QX, Chen W (2018) The divergent restoration effects of Lactobacillus strains in antibiotic-induced dysbiosis. J Funct Foods 51:142–152. https://doi.org/10.1016/j.jff.2018.10.011
Hayden MS, Ghosh S (2014) Regulation of NF-κB by TNF family cytokines. Semin Immunol 26:253–266. https://doi.org/10.1016/j.smim.2014.05.004
Fan L, Qi Y, Qu S, Chen X, Li A, Hendi M, Xu C, Wang L, Hou T, Si J, Chen S (2021) B. adolescentis ameliorates chronic colitis by regulating Treg/Th2 response and gut microbiota remodeling. Gut Microbes 13:1–17. https://doi.org/10.1080/19490976.2020.1826746
Wu Y, Jha R, Li A, Liu H, Zhang Z, Zhang C, Zhai Q, Zhang J (2022) Probiotics (Lactobacillus plantarum HNU082) supplementation relieves ulcerative colitis by affecting intestinal barrier functions, immunity-related gene expression, gut microbiota, and metabolic pathways in mice. Microbiol Spectr 10:e0165122. https://doi.org/10.1128/spectrum.01651-22
Lannoy V, Côté-Biron A, Asselin C, Rivard N (2023) TIRAP, TRAM, and Toll-like receptors: the untold story. Mediators Inflamm 2023:2899271. https://doi.org/10.1155/2023/2899271
Tong L, Zhang X, Hao H, Liu Q, Zhou Z, Liang X, Liu T, Gong P, Zhang L, Zhai Z, Hao Y, Yi H (2021) Lactobacillus rhamnosus GG derived extracellular vesicles modulate gut microbiota and attenuate inflammatory in DSS-induced colitis mice. Nutrients. https://doi.org/10.3390/nu13103319
Li Y, Yang S, Lun J, Gao J, Gao X, Gong Z, Wan Y, He X, Cao H (2020) Inhibitory effects of the Lactobacillus rhamnosus GG effector protein HM0539 on inflammatory response through the TLR4/MyD88/NF-кB axis. Front Immunol 11:551449. https://doi.org/10.3389/fimmu.2020.551449
Xia Y, Chen Y, Wang G, Yang Y, Ai L (2020) Lactobacillus plantarum AR113 alleviates DSS-induced colitis by regulating the TLR4/MyD88/NF-κB pathway and gut microbiota composition. J Funct Foods 67:103854. https://doi.org/10.1016/j.jff.2020.103854
Rahmani F, Asgharzadeh F, Avan A, Barneh F, Parizadeh MR, Ferns GA, Ryzhikov M, Ahmadian MR, Giovannetti E, Jafari M, Khazaei M, Hassanian SM (2020) Rigosertib potently protects against colitis-associated intestinal fibrosis and inflammation by regulating PI3K/AKT and NF-κB signaling pathways. Life Sci 249:117470. https://doi.org/10.1016/j.lfs.2020.117470
Jiang RH, Xu JJ, Zhu DC, Li JF, Zhang CX, Lin N, Gao WY (2020) Glycyrrhizin inhibits osteoarthritis development through suppressing the PI3K/AKT/NF-κB signaling pathway in vivo and in vitro. Food Funct 11:2126–2136. https://doi.org/10.1039/c9fo02241d
Dai C, Zheng CQ, Meng FJ, Zhou Z, Sang LX, Jiang M (2013) VSL#3 probiotics exerts the anti-inflammatory activity via PI3k/Akt and NF-κB pathway in rat model of DSS-induced colitis. Mol Cell Biochem 374:1–11. https://doi.org/10.1007/s11010-012-1488-3
Lv W, Liu C, Ye C, Sun J, Tan X, Zhang C, Qu Q, Shi D, Guo S (2017) Structural modulation of gut microbiota during alleviation of antibiotic-associated diarrhea with herbal formula. Int J Biol Macromol 105:1622–1629. https://doi.org/10.1016/j.ijbiomac.2017.02.060
Fatahi-Bafghi M (2019) Antibiotic resistance genes in the Actinobacteria phylum. Eur J Clin Microbiol Infect Dis 38:1599–1624. https://doi.org/10.1007/s10096-019-03580-5
Shin NR, Whon TW, Bae JW (2015) Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol 33:496–503. https://doi.org/10.1016/j.tibtech.2015.06.011
Guo X, Wang J, Niu RY, Li R, Wang JX, Fan XF, Wang XW, Sun ZL (2022) Effects of apple juice fermented with Lactobacillus plantarum CICC21809 on antibiotic-associated diarrhea of mice. J Funct Foods 99:15. https://doi.org/10.1016/j.jff.2022.105334
Funding
This work was financially supported by the Jilin Province Agricultural Science and Technology Innovation Project (CXGC2022RCY005) and the earmarked fund for CARS-36.
Author information
Authors and Affiliations
Contributions
Wei Liang: methodology resources, formal analysis, investigation, writing — original draft, writing — review and editing, supervision, funding acquisition. Yansong Gao: conceptualization, investigation, editing. Yujuan Zhao: conceptualization, investigation, writing — review and editing. Lei Gao: resources, data curation. Zijian Zhao: resources, investigation. Zhongmei He: resources, supervision. Shengyu Li: project administration, writing — review and editing, supervision, funding acquisition.
Corresponding authors
Ethics declarations
Ethics Approval
All animal studies complied with the institutional animal care regulations of Jilin Academy of Agricultural Sciences (approval number: SCXΚ2020-0001).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liang, W., Gao, Y., Zhao, Y. et al. Lactiplantibacillus plantarum ELF051 Alleviates Antibiotic-Associated Diarrhea by Regulating Intestinal Inflammation and Gut Microbiota. Probiotics & Antimicro. Prot. (2023). https://doi.org/10.1007/s12602-023-10150-x
Accepted:
Published:
DOI: https://doi.org/10.1007/s12602-023-10150-x