Abstract
Purpose
Postoperative atrial fibrillation (POAF) has an incidence of 20–60% in cardiac surgery. The Society of Cardiovascular Anesthesiologists and the European Association of Cardiothoracic Anaesthesiology Practice Advisory have recommended postoperative beta blockers and amiodarone for the prevention of POAF. By employing quality improvement (QI) strategies, we sought to increase the use of these agents and to reduce the incidence of POAF among our patients undergoing cardiac surgery.
Methods
This single-centre QI initiative followed the traditional Plan, Do, Study, Act (PDSA) cycle scientific methodology. A POAF risk score was developed to categorize all patients undergoing cardiac surgery as either normal or elevated risk. Risk stratification was incorporated into a preprinted prescribing guide, which recommended postoperative beta blockade for all patients and a postoperative amiodarone protocol for patients with elevated risk starting on postoperative day one (POD1). A longitudinal audit of all patients undergoing cardiac surgery was conducted over 11 months to track the use of prophylactic medications and the incidence of POAF.
Results
Five hundred and sixty patients undergoing surgery were included in the QI initiative from 1 December 2020 to 1 November 2021. The baseline rate of POAF across all surgical subtypes was 39% (198/560). The use of prophylactic amiodarone in high-risk patients increased from 13% (1/8) at the start of the project to 41% (48/116) at the end of the audit period. The percentage of patients receiving a beta blocker on POD1 did fluctuate, but remained essentially unchanged throughout the audit (34.8% in December 2020 vs 46.7% in October 2021). After 11 months, the overall incidence of POAF was 29% (24.9% relative reduction). Notable reductions in the incidence of POAF were observed in more complex surgical subtypes by the end of the audit, including multiple valve replacement (89% vs 56%), aortic repair (50% vs 33%), and mitral valve surgery (45% vs 33%).
Conclusions
This single-centre QI intervention increased the use of prophylactic amiodarone by 28% for patients at elevated risk of POAF, with no change in the early postoperative initiation of beta blockers (46.7% of patients by POD1). There was a notable reduction in the incidence of POAF in patients at elevated risk undergoing surgery.
Résumé
Objectif
Il y a une incidence de 20 à 60 % de fibrillation auriculaire postopératoire (FAPO) en chirurgie cardiaque. Dans un avis de pratique, la Society of Cardiovascular Anesthesiologists et l’European Association of Cardiothoracic Anaesthesiology ont recommandé l’utilisation de bêtabloquants et d’amiodarone en postopératoire pour la prévention du FAPO. En employant des stratégies d’amélioration de la qualité (AQ), nous avons cherché à augmenter l’utilisation de ces agents et à réduire l’incidence de FAPO chez nos patient·es bénéficiant d’une chirurgie cardiaque.
Méthode
Cette initiative d’AQ monocentrique a suivi la méthodologie scientifique traditionnelle du cycle Plan, Do, Study, Act (PDSA), soit Planifier, Réaliser, Étudier, Agir. Un score de risque de FAPO a été mis au point pour catégoriser toute la patientèle bénéficiant d’une chirurgie cardiaque comme présentant un risque normal ou élevé. La stratification du risque a été intégrée dans un guide de prescription préimprimé, qui recommandait des bêtabloquants en période postopératoire pour tou·tes les patient·es et un protocole postopératoire d’amiodarone pour celles et ceux présentant un risque élevé et débutant à partir du premier jour postopératoire (JPO1). Une vérification longitudinale de toute la patientèle bénéficiant d’une chirurgie cardiaque a été menée sur une période de 11 mois afin de suivre l’utilisation de médicaments prophylactiques et l’incidence de FAPO.
Résultats
Cinq cent soixante personnes opérées ont été incluses dans l’initiative d’AQ entre le 1er décembre 2020 et le 1er novembre 2021. Le taux initial de FAPO pour tous les sous-types chirurgicaux était de 39 % (198/560). L’utilisation d’amiodarone prophylactique chez les patient·es à risque élevé est passée de 13 % (1/8) au début du projet à 41 % (48/116) à la fin de la période de vérification. Le pourcentage de patient·es recevant un bêtabloquant au JPO1 a fluctué, mais est resté fondamentalement inchangé tout au long de la période de vérification (34,8 % en décembre 2020 vs 46,7 % en octobre 2021). Après 11 mois, l’incidence globale de FAPO était de 29 % (réduction relative de 24,9 %). Des réductions notables de l’incidence de FAPO ont été observées dans des sous-types chirurgicaux plus complexes à la fin de la vérification, y compris le remplacement de plusieurs valves (89 % vs 56 %), la réparation aortique (50 % vs 33 %) et la chirurgie valvulaire mitrale (45 % vs 33 %).
Conclusion
Cette intervention monocentrique d’amélioration de la qualité a augmenté l’utilisation de l’amiodarone prophylactique de 28 % chez les patient·es présentant un risque élevé de FAPO, sans changement dans l’amorce postopératoire précoce des bêtabloquants (46,7 % des patient·es au JPO1). Il y a eu une réduction notable de l’incidence de FAPO chez les patient·es à risque élevé bénéficiant d’une intervention chirurgicale.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Postoperative atrial fibrillation (POAF) affects 20–40% of patients after coronary artery bypass grafting (CABG) and up to 60% of patients following a combined CABG and valvular repair/replacement procedure.1,2 Patient- and surgery-specific risk factors for POAF have been extensively studied and include age greater than 75, history of atrial fibrillation (AF) and mitral valve disease, and mitral valve surgery.1 The peak incidence of POAF usually occurs between 48 and 72 hr after surgery.3 Onset of POAF is associated with a prolonged hospital stay, increased cardiac surgery intensive care unit (CSICU) readmission rate, and an increased likelihood of prolonged mechanical ventilation.4 It is also associated with an eight-fold risk increase of developing long-term atrial fibrillation and a higher risk of long-term mortality.5,6 Previous quality improvement (QI) initiatives have aimed to identify patients at elevated risk and initiate prophylactic treatment against the development of POAF.7
According to the 2019 Practice Advisory by the Society of Cardiovascular Anesthesiologists and the European Association of Cardiothoracic Anesthetists (SCA/EACTA), the perioperative use of oral beta blockers for the prevention of POAF is a class-I recommendation. There is also a class-IIa recommendation for the use of perioperative amiodarone in patients at elevated risk.1 In 2018, the SCA/EACTA performed a survey examining the use of POAF prophylaxis in patients undergoing cardiac surgery. This yielded over 640 responses and revealed that, while the treatment of POAF with both rate and rhythm modulators is commonplace, the prophylactic routine use of these agents is not. Surveys suggested that this was due to concerns regarding the side effects of amiodarone, including hypotension, bradycardia, heart block, and liver and thyroid dysfunction.1 Nevertheless, while short-term hemodynamic side effects are common, most noncardiac side effects are generally associated with higher doses and longer treatment periods that are generally not seen in the perioperative period.8
The goals of this QI project were to evaluate our current use of beta blockers and of amiodarone as prophylaxis in patients at elevated risk for developing POAF, improve the timely administration of postoperative beta blockers and, where indicated, amiodarone, and reduce the incidence of POAF in patients undergoing cardiac surgery at our institution. We sought to achieve this using a multidisciplinary, collaborative approach that would encourage long lasting institutional change that could be generalized to other centres caring for patients undergoing cardiac surgery.
Methods
Our QI project was conducted at Vancouver General Hospital (VGH), Vancouver, Canada, a tertiary referral centre performing approximately 700–800 cardiac surgical cases annually, between December 2020 and November 2021. At VGH, postoperative care of patients undergoing cardiac surgery takes place initially in the CSICU by anesthesiologists, CSICU nurses, and cardiac surgeons, and later on the cardiac surgery ward by cardiac surgeons, nurse practitioners, and ward nurses. Our initial QI team was composed of individuals with an interest in QI work from anesthesia, nursing, and pharmacy.
Postoperative AF was defined as “clinically important POAF,” defined as AF or flutter that results in angina, heart failure, or symptomatic hypotension, or that requires treatment with a rate-controlling drug, antiarrhythmic drug, or electrical cardioversion. The baseline rate of POAF in patients undergoing cardiac surgery at VGH was established using data collected by Heart Services British Columbia, a central database that collects outcome data for cardiac surgery in the province of British Columbia, and retrospective analysis of discharge records from the year preceding our study—1 January 2019 to 31 December 2019. According to Heart Services BC data, the rate of POAF in patients undergoing cardiac surgery at VGH was 33.0%.
A Project Ethics Community Consensus Initiative Screening Tool was completed, and this QI project was deemed minimal risk to patients.Footnote 1 As per the Tri-Council Policy Statement 29 and endorsed by the Vancouver Coastal Health Research Institute, a formal research ethics board approval was therefore not required for this QI initiative.
Prior to implementation, the goals of our project were discussed among members of our cardiac anesthesia, cardiac surgery, pharmacy, and cardiac nursing teams. All of these stakeholders were supportive. The plan was to develop a stepwise preprinted order set (PPO) that would support the clinician in identifying patients at elevated risk of POAF and then prescribe appropriate POAF prophylaxis. The first step of the PPO would risk stratify the patient using an evidence-based POAF risk scoring system, after which the second step would employ a protocolized prescription of beta blockers and/or amiodarone based on the patient’s calculated risk. Buy-in from clinicians was achieved by making the action of prescribing prophylaxis for POAF as easy as possible. We did this by making the PPO as clear and concise as possible to ensure a high completion rate. We also asked all bedside nurses in the CSICU to ensure that the PPO was completed for every patient to ensure a high compliance rate.
Preimplementation planning phase
We developed a 16-point POAF risk stratification system to be applied to all cardiac surgery patients (Fig. 1). Our POAF scoring system was composed of both patient and surgical risk factors, as recommended by the SCA/EACTA Practice Advisory.1 We also performed a literature review comparing the POAF incidence according only to surgical subtype. The POAF risk scoring system was designed to categorize all patients as either “normal risk” or “elevated risk,” in accordance with the SCA/EACTA Practice Advisory.1 We also developed an amiodarone prescribing protocol for patients deemed to be at elevated risk for POAF. The risk scoring system and the amiodarone dosing protocol were developed following a literature review. This review highlighted 25 relevant studies, five of which referred to risk analysis for POAF and 19 of which addressed the question of amiodarone dosing and timing for POAF prophylaxis.10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32 After developing the PPO, we held focused group meetings with all stakeholders, including representation from anesthesiology, cardiac surgery, nursing, and pharmacy, to finalize the PPO. This collaborative approach was designed to maximize clinical staff buy-in. The dosing protocol, similar to that for POAF treatment, was reviewed and approved by the VGH institutional Pharmacy Safety Committee. Contraindications to the use of amiodarone were included in PPO as well as baseline blood work to monitor for hepatic complications from amiodarone administration.
Preprinted order set for prevention of postoperative atrial fibrillation in cardiac surgery. Page 1 contains the POAF risk stratification scoring template and prophylaxis prescribing tool. Reverse side of single page PPO (Page 1A) describes suggested dosing, contraindications, and reasons to discontinue prophylaxis.
bpm = beats per minute; COPD = chronic obstructive pulmonary disease; CSICU = cardiac surgery intensive care unit; eGFR = estimated glomerular filtration rate; HR = heart rate; LVEF = left ventricular ejection fraction; POAF = postoperative atrial fibrillation; POD = postoperative day; PPO = preprinted order set; TTE = transthoracic echocardiography
The dosing and timing of amiodarone administration was considered in the preplanning phase. Studies have shown that both pre- and postoperative administration of amiodarone were effective in preventing POAF after surgery.25,26 Therefore, the PPO indicated that amiodarone prophylaxis for elevated risk patients could begin within the first 24 hr of admission to the CSICU postoperatively. This would provide an opportunity for the anesthesiologist, surgeon, and CSICU clinicians to make collaborative serial assessments on the risk vs benefit of starting amiodarone for a particular patient. The amiodarone protocol consisted of an iv loading dose of amiodarone 150 mg iv over one hour followed by an amiodarone iv infusion at 1 mg·min–1 for six hours, then a 0.5 mg·min–1 iv infusion for 18 hr. After the 18-hr iv loading regimen, oral amiodarone 200 mg bid was prescribed for seven days (or until hospital discharge), to include the most high-risk period for developing POAF.
The PPO prompted the CSICU clinician to commence beta blocker therapy as soon as possible postoperatively for all cardiac surgery patients (Fig. 1). Clinicians were already familiar with prescribing postoperative beta blockers, as it was an informally established practice at our centre, although details about how consistently and how soon after surgery were unknown. Our PPO suggested continuing the patient’s own beta blocker if already on established therapy, otherwise metoprolol 12.5 mg po bid was recommended. An alternative more cardioselective beta blocker, bisoprolol, could be initiated if there were clinical concern for respiratory disease. All patients undergoing cardiac surgery at our CSICU are eventually prescribed beta blockers postoperatively where tolerated; however, it was unknown how soon postoperatively this was being initiated. The PPO therefore highlighted the importance of starting beta blockade as early as deemed tolerable, to provide maximum POAF prophylaxis. Relative contraindications to beta blockade were also listed for consideration by the prescribing clinician (Fig. 1).
Data collection/analysis
Data collection took place in the form of paper audit sheets attached to the patient’s chart and were completed in the CSICU by bedside nurses and on the postoperative cardiac surgery ward by one of two nurse practitioners prior to patient discharge. Paper audit sheets did not contain any identifying information for the patient and were stored in a secured area in the local research office. Patient data were then manually inputted to a secure online database, REDCap®Footnote 2 (Vanderbilt University, Nashville, TN, USA), for secure online storage.
Quality improvement projects that aim to make a process or system produce better results benefit from using “graphical methods of analysis [that] reveal the trajectory of system behaviour over time” such as control charts (or Shewart charts).33 Control charts allow identification of variation due to special causes (as opposed to common causes, which result in random variation in a process or system) through the use of rules.33 By reviewing the incidence of POAF over time in consecutive groups of ten patients, we could observe whether our PDSA cycles were making a difference by following the data chronologically. The control chart for POAF incidence over time was built using an Excel software add on (QI charts, Process Improvement Products, pipproducts.com, San Antonio, TX, USA) according to previously published QI methodology.34 The data were grouped in subgroups of ten consecutive observations to smoothen the variation in the data (e.g., variation in volume of daily cardiac cases over time), while also allowing a reasonable number of observations to analyze. According to previously published QI methodology, five rules were used to analyze the control chart and identify special causes and process changes.34
Plan-Do-Study-Act cycles
Plan-Do-Study-Act cycle 1: 1 December 2020 to 28 February 2021—implementation phase
The first Plan-Do-Study-Act (PDSA) cycle involved a multidisciplinary collaboration to educate clinicians about the importance of POAF prophylaxis, and to review the PPO and gain multidisciplinary approval. Cardiac surgeons and anesthesiologists were informed of our QI initiative and feedback was solicited. An educational drive was conducted with CSICU nursing staff to increase familiarity with the PPO and optimize completion and accuracy of data collection. This was essential in capturing patients in whom prophylactic amiodarone would be suitable. Our initial POAF score threshold for patients at elevated risk, who should receive amiodarone prophylaxis, was 6 or greater during this phase.
Plan-Do-Study-Act cycle 2: 28 February 2021 to 1 June 2021—elevated risk threshold decreased
Upon completion of PDSA cycle 1, a preplanned review of our preliminary data at three months revealed that very few patients met the POAF risk criteria of six points or greater on the risk scoring system, and therefore very few patients qualified for amiodarone prophylaxis (8/150 patients). It also highlighted that, when patients did meet the threshold, the amiodarone protocol was rarely adhered to. A preliminary analysis of the POAF risk scoring system during PDSA cycle 1 revealed a consistent increase in the incidence of POAF to greater than 40% in patients with a POAF score of 2 or greater (Fig. 2). We therefore decreased the POAF risk score to redefine elevated risk as a score of 3 or greater. Improved staff engagement was achieved by discussing the QI project at the Cardiac Anesthesia Divisional Meetings and further nurse education at the bedside. We also posted POAF infographics in clinical areas describing the incidence of POAF, clinical consequences of POAF, and recommendations for initiating amiodarone prophylactically (Electronic Supplementary Material [ESM] eAppendix 1).
Plan-Do-Study-Act cycle 3: June 2021 to November 2021—improving early postoperative beta blocker prescription
Summative data analysis at the preplanned six-month time point (i.e., at the end of PDSA cycle 2) showed increased use of prophylactic amiodarone but also highlighted a lack of improvement with early beta blocker administration postoperatively (see Results section below). To understand why, the data collection tool was modified so clinicians would document a reason for withholding beta blockers beyond postoperative day (POD) 1. Interventions intended to expedite beta blocker prescription included a nurse-led bedside checklists for readiness for initiation of beta blockade, and a new CSICU ward round mandate to review each patient's readiness for beta blocker initiation every 12 hr (ESM eAppendix 2). Also, the clinical pharmacist was made responsible for asking the question whether beta blocker restart was appropriate during every patient review on CSICU ward rounds.
Results
There were 560 patients included in this study encompassing a wide range of cardiac surgeries—isolated CABG (55%, 310/560), valve surgery (27%, 152/560), combined valve and CABG (5%, 27/560), aortic surgery (11%, 61/560), and other surgeries (2%, 10/560). The incidence of POAF across all surgical types in the first PDSA cycle was 36.7% (55/150).
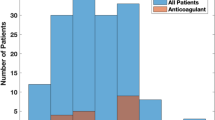
The POAF risk scoring tool was evaluated by comparing POAF rates across patients with increasing POAF risk scores during PDSA cycle 1 (Fig. 2). Patients with a POAF risk score of 0 and 1 had an incidence of POAF of 17% (26/149) and 33% (53/159), respectively. Postoperative AF risk scores of ≥ 2 are associated with an incidence of POAF > 45%. These data were used to inform our lowering of the threshold for elevated risk patients to a score of 3 or greater in PDSA cycle 2 (Fig. 2).
The change in incidence of POAF over the course of the QI initiative was assessed by using a control chart and was analyzed for special causes and process changes (Fig. 3). It was observed that the data points after observation 33 (coinciding with the start of PDSA cycle 3) were clustering at lower overall values and with a shorter dispersion. Therefore, the mean and control limits were recalculated and the mean incidence of POAF was observed to fall from 39% in PDSA cycles 1 and 2 (points 1–32) to 30% in PDSA cycle 3 (points 33–56). This process change represented a 25% relative reduction in POAF after PDSA cycle 3. Only one special cause was observed for data point 28 (POAF incidence of 0 for these ten patients), which fell outside of two standard deviations of the lower control limit. This data point was investigated and it was recognized that no special cause existed, i.e., these ten patients all either had pre-existing chronic AF or did not develop new POAF.
Control chart of incidence of postoperative atrial fibrillation over time among groups of ten consecutive patients undergoing cardiac surgery. Mean incidence of postoperative atrial fibrillation and standard deviation (σ) were calculated over Plan, Do, Study, Act (PDSA) cycles 1 and 2 from intervals 1–32 (39%) and PDSA cycle 3 from intervals 33–56 (30%).
In the planned exploratory analysis of POAF according to surgical subtype, we compared POAF incidence during PDSA cycle 1 with POAF incidence during PDSA cycles 2 and 3 (Fig. 4). A notable reduction in POAF incidence was observed in high-risk surgical types, including aortic repair (50% vs 33%), mitral valve surgery (45% vs 33%), and multiple valve replacement (89% vs 56%) (Fig. 4).
Adherence to prescribing amiodarone to patients at elevated risk was analyzed during PDSA cycle 1 and compared with that in PDSA cycles 2 and 3. Amiodarone prescription for patients at elevated risk increased from 13% (1/8) to 41% (48/116) (Fig. 5). Clinician-reported barriers to prescribing amiodarone prophylaxis were predominantly bradycardia in 32% (24/75), hypotension in 17% (13/75), complete cardiac block in 13% (10/75), and chronic AF in 8% (5/75) (Fig. 6).
In PDSA cycle 1, we observed that POD1 postoperative beta blocker initiation was 35% (15/43), 50% (27/54), and 46% (23/50) in December, January, and February, respectively. Despite initiatives to improve time to beta blocker initiation, no significant changes in POD1 beta blocker initiation were observed over the course of the audit, including after PDSA cycle 3 (Fig. 7). The most common reasons reported for failure to initiate postoperative beta blockade were hypotension in 65% (90/138), bradycardia in 28% (39/138), heart block in 3% (4/1,398), and other reasons in 4% (5/138) (Fig. 6).
Discussion
The 2019 SCA/EACTA Practice Advisory on POAF1 highlighted the importance of identifying patients undergoing cardiac surgery at elevated risk of POAF and implementing evidence-based recommendations to prevent POAF in these patients. Institutions were encouraged to develop initiatives to address the lack of emphasis clinicians place on POAF prophylaxis, through educational initiatives and clinical tools to support decision-making. This QI initiative showed how a multidisciplinary approach using several QI techniques can improve adherence to POAF prophylaxis recommendations in cardiac surgery and reduce the incidence of POAF in high-risk surgical subtypes.
The locally developed POAF risk scoring system used in our PPO performed well with an increasing POAF score correlating with an increasing incidence of POAF. Determining the threshold for “elevated risk” was a challenge, as our local data on POAF incidence prior to starting the initiative did not include all of the risk factors in our scoring system. An initial threshold score of 6 was found to be too high, with too few patients meeting this threshold, which was unfortunate as much of the momentum for preventing POAF generated after our educational sessions was lost. A QI initiative designed to make an intervention (i.e., prophylactic amiodarone) that poses a side-effect risk and represents deviation from usual clinical practice relies on generating momentum to get traction with clinicians and stakeholders. Perhaps there would have been even greater use of prophylactic amiodarone had we established the elevated risk threshold earlier in our initiative. More studies are necessary to identify and standardize a scoring system that can be recommended for widespread use. Certain patient risk factors, such as age and a history of paroxysmal AF, and some surgical risk factors, such as mitral valve surgery, are consistently identified as high-risk features, whereas other risk factors are less consistent in predicting POAF. For this reason, the current SCA/EACTA Practice Advisory acknowledges that developing a hospital-specific risk score locally, as we have done, would be more effective as patient populations and surgical types vary from hospital to hospital.
Prophylactic amiodarone use increased over the course of our QI initiative. A fixed standard dose of amiodarone was used, as opposed to a weight-based dose, to improve ease of prescription for clinicians and to alleviate concerns with hemodynamic side effects of higher doses. Weight-based iv amiodarone dosing or higher doses of oral amiodarone (i.e., 400 mg po bid) could potentially have been more effective at preventing POAF. We did not assess how frequently prophylactic amiodarone was discontinued; however, no serious adverse events related to the prescription of prophylactic amiodarone were reported. We did not measure whether the application of prophylactic amiodarone delayed beta blocker restart time; however, this is a concern as both agents have similar hemodynamic side effects.
Prescribing practices for beta blockers did not change throughout our QI initiative. Clinicians reported that postoperative beta blockade was mainly held because of hypotension (65%; Fig. 6). At our institution, to be eligible for transfer from the CSICU to the cardiac surgery ward, a patient must maintain a systolic blood pressure of > 90 mm Hg for four hours while off vasopressor support. Therefore, patient flow out of the CSICU could be at risk if beta blockade contributes to hypotension. Other QI initiatives reported beta blocker initiation despite the use of low dose vasopressor support;13 however, we were unable to circumvent this barrier locally. Further efforts to implement early postoperative beta blockade, including reducing the initial metoprolol dose to 6.25 mg, using iv esmolol infusions that can be titrated and discontinued if hypotension occurs, or re-engaging stakeholders to reconsider different mean arterial pressure targets are being pursued.
In addition to the direct clinical impact of this QI project, there are several other encouraging outcomes. We have established a foundation to develop other QI initiatives in cardiac surgery by collaborating with leaders in QI from cardiac surgery, nursing, and pharmacy. There was a clear improvement in local understanding of the consequences of POAF and the importance of prophylaxis. After presenting our project to other local cardiac centres at regional meetings, there is interest in adding further POAF-preventing interventions to our pathway including colchicine, posterior pericardiotomy,21 or dual atrial chamber pacing.22 This PPO is now part of standard postoperative orders at our hospital, and a neighbouring institution has adopted this POAF prophylaxis pathway as well. We recently converted the PPO for use in electronic format (PowerChart™, Oracle Cerner, North Kansas City, MI, USA) and we are developing methods to track POAF scoring and amiodarone/beta blocker prescribing to perform future audits to ensure sustained change in clinical practice.
Conclusions
This QI project illustrates that educational initiatives and clinical tools (e.g., PPOs) can be used to help clinicians identify patients at risk of developing POAF, improving the prescription of recommended amiodarone prophylaxis, and lowering POAF rates in surgical subtypes with elevated risk. By adopting a multidisciplinary approach, we have generated sustained change in evidence-based prescribing practices at our institution, and established a stronger foundation for QI initiatives at our hospital. As per the SCA/EACTA Practice Advisory, our POAF prophylaxis PPO was tailored to our local patient population but has since been adopted at other cardiac centres in our region. Although we improved the local understanding of the importance of beta blockers in preventing POAF, further work to improve early postoperative beta blocker restarts and incorporate other pharmacologic and interventional POAF prevention strategies into our PPO is ongoing.
Notes
ARECCI. ARECCI ethics screening tool. Available from URL: https://arecci.albertainnovates.ca/ (accessed July 2023).
REDCap. Homepage. Available from URL: https://www.project-redcap.org (accessed July 2023).
References
Muehlschlegel JD, Burrage PS, Ngai JY, et al. Society of Cardiovascular Anesthesiologists/European Association of Cardiothoracic Anaesthetists practice advisory for the management of perioperative atrial fibrillation in patients undergoing cardiac surgery. Anesth Analg 2019; 128: 33–42. https://doi.org/10.1213/ane.0000000000003865
Aranki SF, Shaw DP, Adams DH, et al. Predictors of atrial fibrillation after coronary artery surgery. Current trends and impact on hospital resources. Circulation 1996; 94: 390–7. https://doi.org/10.1161/01.cir.94.3.390
Butt JH, Olesen JB, Gundlund A, et al. Long-term thromboembolic risk in patients with postoperative atrial fibrillation after left-sided heart valve surgery. JAMA Cardiol 2019; 4: 1139–47. https://doi.org/10.1001/jamacardio.2019.3649
Tseng YH, Ko HK, Tseng YC, Lin YH, Kou YR. Atrial fibrillation on intensive care unit admission independently increases the risk of weaning failure in nonheart failure mechanically ventilated patients in a medical intensive care unit: a retrospective case-control study. Medicine (Baltimore) 2016; 95: e3744. https://doi.org/10.1097/md.0000000000003744
Helgadottir S, Sigurdsson MI, Ingvarsdottir IL, Arnar DO, Gudbjartsson T. Atrial fibrillation following cardiac surgery: risk analysis and long-term survival. J Cardiothorac Surg 2012; 7: 87. https://doi.org/10.1186/1749-8090-7-87
Tamis JE, Steinberg JS. Atrial fibrillation independently prolongs hospital stay after coronary artery bypass surgery. Clin Cardiol 2000; 23: 155–9. https://doi.org/10.1002/clc.4960230305
Ebinger JE, Porten BR, Strauss CE, et al. Design, challenges, and implications of quality improvement projects using the electronic medical record: case study: a protocol to reduce the burden of postoperative atrial fibrillation. Circ Cardiovasc Qual Outcomes 2016; 9: 593–9. https://doi.org/10.1161/circoutcomes.116.003122
Vorperian VR, Havighurst TC, Miller S, January CT. Adverse effects of low dose amiodarone: a meta-analysis. J Am Coll Cardiol 1997; 30: 791–8. https://doi.org/10.1016/s0735-1097(97)00220-9
Canada Panel on Research Ethics. Tri-Council policy statement: ethical conduct for research involving humans – TCPS 2 (2022). Available from URL: https://ethics.gc.ca/eng/policy-politique_tcps2-eptc2_2022.html (accessed July 2023).
Arsenault KA, Yusuf AM, Crystal E, et al. Interventions for preventing post-operative atrial fibrillation in patients undergoing heart surgery. Cochrane Database Syst Rev 2013; 2013: CD003611. https://doi.org/10.1002/14651858.cd003611.pub3
Beaulieu Y, Denault AY, Couture P, et al. Perioperative intravenous amiodarone does not reduce the burden of atrial fibrillation in patients undergoing cardiac valvular surgery. Anesthesiology 2010; 112: 128–37. https://doi.org/10.1097/aln.0b013e3181c61b28
Akbarzadeh F, Kazemi-Arbat B, Golmohammadi A, Pourafkari L. Biatrial pacing vs. intravenous amiodarone in prevention of atrial fibrillation after coronary artery bypass surgery. Pak J Biol Sci 2009; 12: 1325–9. https://doi.org/10.3923/pjbs.2009.1325.1329
Roshanali F, Mandegar MH, Yousefnia MA, Alaeddini F, Saidi B. Prevention of atrial fibrillation after coronary artery bypass grafting via atrial electromechanical interval and use of amiodarone prophylaxis. Interact Cardiovasc Thorac Surg 2009; 8: 421–5. https://doi.org/10.1510/icvts.2008.191403
Alves RJ, Geovanini GR, de Brito G, Miguel GA, Glauser VA, Nakiri K. Prevention of atrial fibrillation with moderate doses of amiodarone in the postoperative period of cardiac surgery is safe and effective in patients with high risk for developing this arrhythmia. Arq Bras Cardiol 2007; 89: 22–7.
Turk T, Ata Y, Vural H, Ozkan H, Yavuz S, Ozyazicioglu A. Intravenous and oral amiodarone for the prevention of postoperative atrial fibrillation in patients undergoing off-pump coronary artery bypass surgery. Heart Surg Forum 2007; 10: E299–303. https://doi.org/10.1532/hsf98.20071060
Zebis LR, Christensen TD, Thomsen HF, et al. Practical regimen for amiodarone use in preventing postoperative atrial fibrillation. Ann Thorac Surg 2007; 83: 1326–31. https://doi.org/10.1016/j.athoracsur.2006.09.096
Alcalde RV, Guaragna JC, Bodanese LC, et al. High dose of amiodarone in a short-term period reduces the incidence of postoperative atrial fibrillation and atrial flutter. Arq Bras Cardiol 2006; 87: 236–40. https://doi.org/10.1590/s0066-782x2006001600002
Budeus M, Hennersdorf M, Perings S, et al. Amiodarone prophylaxis for atrial fibrillation of high-risk patients after coronary bypass grafting: a prospective, double-blinded, placebo-controlled, randomized study. Eur Heart J 2006; 27: 1584–91. https://doi.org/10.1093/eurheartj/ehl082
Mitchell LB, Exner DV, Wyse DG, et al. Prophylactic oral amiodarone for the prevention of arrhythmias that begin early after revascularization, valve replacement, or repair: PAPABEAR: a randomized controlled trial. JAMA 2005; 294: 3093–100. https://doi.org/10.1001/jama.294.24.3093
Mitchell LB, CCS Atrial Fibrillation Guidelines Committee. Canadian Cardiovascular Society Atrial Fibrillation guidelines 2010: prevention and treatment of atrial fibrillation following cardiac surgery. Can J Cardiol 2011; 27: 91–7. https://doi.org/10.1016/j.cjca.2010.11.005
Coletta MJ, Lis G, Clark P, Dabir R, Daneshvar F. Reducing new-onset atrial fibrillation after coronary artery bypass graft surgery. AACN Adv Crit Care 2019; 30: 249–58. https://doi.org/10.4037/aacnacc2019470
Onk OA, Erkut B. Is the preoperative administration of amiodarone or metoprolol more effective in reducing atrial fibrillation: after coronary bypass surgery? Medicine (Baltimore) 2015; 94: e1576. https://doi.org/10.1097/md.0000000000001576
Kerstein J, Soodan A, Qamar M, et al. Giving IV and oral amiodarone perioperatively for the prevention of postoperative atrial fibrillation in patients undergoing coronary artery bypass surgery: the GAP study. Chest 2004; 126: 716–24. https://doi.org/10.1378/chest.126.3.716
Raiten JM, Ghadimi K, Augoustides JG, et al. Atrial fibrillation after cardiac surgery: clinical update on mechanisms and prophylactic strategies. J Cardiothorac Vasc Anesth 2015; 29: 806–16. https://doi.org/10.1053/j.jvca.2015.01.001
Buckley MS, Nolan PE Jr, Slack MK, Tisdale JE, Hilleman DE, Copeland JG. Amiodarone prophylaxis for atrial fibrillation after cardiac surgery: meta-analysis of dose response and timing of initiation. Pharmacotherapy 2007; 27: 360–8. https://doi.org/10.1592/phco.27.3.360
Chatterjee S, Sardar P, Mukherjee D, Lichstein E, Aikat S. Timing and route of amiodarone for prevention of postoperative atrial fibrillation after cardiac surgery: a network regression meta-analysis. Pacing Clin Electrophysiol 2013; 36: 1017–23. https://doi.org/10.1111/pace.12140
Dobrev D, Aguilar M, Heijman J, Guichard JB, Nattel S. Postoperative atrial fibrillation: mechanisms, manifestations and management. Nat Rev Cardiol 2019; 16: 417–36. https://doi.org/10.1038/s41569-019-0166-5
Piccini JP, Zhao Y, Steinberg BA, et al. Comparative effectiveness of pharmacotherapies for prevention of atrial fibrillation following coronary artery bypass surgery. Am J Cardiol 2013; 112: 954–60. https://doi.org/10.1016/j.amjcard.2013.05.029
Martin WJ 2nd, Rosenow EC 3rd. Amiodarone pulmonary toxicity. Recognition and pathogenesis (part I). Chest 1988; 93: 1067–75. https://doi.org/10.1378/chest.93.5.1067
Martino E, Bartalena L, Bogazzi F, Braverman LE. The effects of amiodarone on the thyroid. Endocr Rev 2001; 22: 240–54. https://doi.org/10.1210/edrv.22.2.0427
Gu S, Su PX, Liu Y, Yan J, Zhang XT, Wang TY. Low-dose amiodarone for the prevention of atrial fibrillation after coronary artery bypass grafting in patients older than 70 years. Chin Med J (Engl) 2009; 122: 2928–32.
Haddad M, Nair R, Hendry P, Coyle D, Mesana T. Peri-operative amiodarone for post-operative atrial fibrillation prophylaxis in valve surgery patients. J Surg Res 2010; 158: P226. https://doi.org/10.1016/j.jss.2009.11.154
Provost LP. Analytical studies: a framework for quality improvement design and analysis. BMJ Qual Saf 2011; 20: i92–6. https://doi.org/10.1136/bmjqs.2011.051557
Provost LP, Murray SK. The Health Care Data Guide: Learning from Data for Improvement. San Francisco: John Wiley & Sons, Inc; 2011.
Author contributions
Sinead Egan and Coilin Collins-Smyth prepared the manuscript. Shruti Chitnis was involved in the preimplementation planning phase of the study, PPO development, and data collection. Jamie Head assisted with data collection. Allison Chui supported QI educational material development and data analysis. Gurdip Bhatti assisted in design, implementation, data collection, and staff education and engagement. Sean R. McLean developed the QI initiative and prepared the manuscript.
Acknowledgements
This QI initiative was partially funded by the Physician Lead Quality Improvement Project at Vancouver Coastal Health. We are grateful to the cardiac anesthesiologists, cardiac surgeons, cardiac surgical intensive care unit staff, pharmacy staff, and postcardiac surgery ward nurses at Vancouver General Hospital for supporting this QI initiative. Specifically, Dr. Tiffany Chong, Ms. Patricia Choy (nurse practitioner), and Mr. Eric Chu (pharmacist) who provided outstanding support throughout. We also thank Heart Services BC for providing data pertaining to POAF at our institution, Dr. Enrique Fernandez Ruez for support with data analysis, and Dr. Hilary Grocott for reviewing and editing the manuscript.
Disclosures
None.
Funding statement
Partial funding for this project was provided by the Physician Lead Quality Improvement initiative at Vancouver General Hospital (Vancouver, BC, Canada).
Prior conference presentations
This work was presented at the Quality Forum 2022 (31 May–2 June 2022, Vancouver, BC, Canada) and at the 12th Annual Whistler Anesthesiology Summit 2022 (3–6 March, Whistler, BC, Canada).
Editorial responsibility
This submission was handled by Dr. Philip M. Jones, Deputy Editor-in-Chief, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is accompanied by an Editorial. Please see Can J Anesth 2023; https://doi.org/10.1007/s12630-023-02618-9.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Egan, S., Collins-Smyth, C., Chitnis, S. et al. Prevention of postoperative atrial fibrillation in cardiac surgery: a quality improvement project. Can J Anesth/J Can Anesth 70, 1880–1891 (2023). https://doi.org/10.1007/s12630-023-02619-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-023-02619-8