Abstract
Atherosclerosis with its manifestations and associated diseases is a main cause of morbidity and mortality in industrial countries. The pathomechanisms underlying atherosclerosis are complex and comprise exogenous factors as well as genetic predisposition. Beyond the well-defined risk factors for the development of atherosclerosis, obstructive sleep apnoea (OSA) merits more and more attention. A growing body of evidence has associated OSA with vascular pathologies. Although the exact mechanisms involved are not known, the occurrence of intermittent hypoxia typical for OSA may lead to oxidative stress, inflammation, metabolic and neural changes which in turn are responsible for vessel dysfunction underlying atherosclerosis. It has been demonstrated that therapy with continuous positive airway pressure (CPAP) plays a vasoprotective role. This review summarises data resulting from epidemiological and clinical studies with emphasis on the possible mechanisms linking OSA with atherosclerosis, predictive biomarkers helping identify OSA patients at high cardiovascular risk and personalised treatment approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atherosclerosis is an inflammatory and systemic disease [1–3]. A key process that initiates the development of atherosclerosis is an endothelial dysfunction, followed by lipid accumulation, the recruitment of macrophages and T-lymphocytes as well as the migration and proliferation of smooth muscle cells in the vascular wall. Additionally, the regeneration potential of endothelial progenitor cells is diminished because of their reduced number and impaired function [2]. The resultant impaired vasorelaxation and the expansion of the vascular wall impede the laminar blood stream, which promotes the thrombus formation. The consequent plaque rupture and the activated coagulation system may lead to complete stenosis of an affected vessel with consecutive infarction of the correspondent tissue [3]. Besides known main risk factors for development of atherosclerosis such as hypertension, diabetes mellitus, hypercholesterolemia, nicotine abuse, obesity, age and male sex, sleep apnoea syndrome have been reported to have an independent negative impact on the vascular structure and function.
The obstructive sleep apnoea (OSA) is characterised by a repetitive partial or complete collapse of upper airways during sleep terminated by arousals with consecutive repetitive hypoxia/reoxygenation phases. In contrast to central apnoea syndrome, the obstructive one is associated with inefficient breath efforts [4–9]. The obstructive sleep apnoea syndrome is defined as more than five apnoeas (no breath flow for more than 10 s)/hypopnoea (reduced breath flow) per hour (so called apnoea/hypopnoea index: AHI) in polysomnography accompanied by the following symptoms: loud snoring, breathing stoppage, nocturia, headache, excessive daytime sleepiness and deficits in memory and attention with subsequent risk of accident by day. According to the actual data, about 20% and 7% of adults suffer from mild OSA (5/h < AHI < 15/h) and moderate to severe OSA (AHI > 15/h), respectively. The nocturnal hyperpressure treatment, so-called CPAP (continuous positive airway pressure) ventilation is the gold-standard therapy of OSA. It consists in creating of pneumatic track that avoids the collapse of the upper airways. However, a relevant number of patients does not tolerate CPAP therapy [10], which implicates necessity of searching for alterative therapy options. Moreover, it is not clear, which group of OSA patients should be treated. It seems that the genetic predisposition which influences the extent of vascular responses to OSA-related pathological changes and coexistence of independent cardiovascular risk factors determine patients at particularly high cardiovascular risk. Because of that, the identification of such patients is one of the most important issues when deciding about OSA therapy. The estimation of individual risk constellation should be a basis for an optimal personalised OSA treatment.
Possible pathomechanisms linking OSA with atherosclerosis
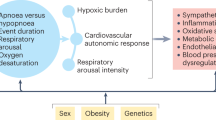
A growing body of evidence associates OSA with development of atherosclerosis [4–9]. A key phenomenon which is believed to initiate the atherosclerotic vessel changes is the intermittent hypopnoea-related disturbance of vascular micromilieu. Because OSA is often attended by other comorbidities which are also risk factors for atherosclerosis formation, it is sometimes difficult to understand the exclusive impact of OSA on vascular diseases. Nevertheless, a lot of controlled, methodically excellent studies have demonstrated that different neural, vascular, mechanic, inflammatory and metabolic mechanisms discussed below may be activated in OSA (Fig. 1):
-
Neural activity: stimulation of sympathetic nerves with consequent release of catecholamine substances in the blood plasma and the urine [11].
-
Vascular changes: overproduction of vasoconstrictors like endothelin in relation to reduced release of vasorelaxants, such as NO [12–14]; detection of elevated oxidative stress [15], reduced endothelium-dependent vasodilatation due to impaired function of endothelial cells [16].
-
Mechanical factors: fluctuation of intrathoracic pressure via increase in blood pressure may enhance shear stress in the vessel wall with subsequent diastolic dysfunction [17].
-
Inflammatory mediators: cytokines and adhesion molecules attract leukocytes to the endothelial layer, which promotes atherosclerosis in OSA [18–20]. Elevated inflammatory markers, such as C-reactive protein (CRP), serum amyloid A were demonstrated to be negative predictor markers for cardiovascular endpoints [21, 22]; the CRP levels were increased among children with OSA, irrespective of body weight [23]; other study showed high CRP concentration only in obese OSA patients [24].
-
Circulating endothelial progenitor cells as regeneration capacity marker are reduced in OSA patients [25]. In contrast, the number of circulating apoptotic endothelial cells increases as indicator for endothelial dysfunction [26, 27]. The last one correlates positively with the AHI degree [28].
-
Hypercoagulability: elevation of fibrinogen, haematocrit, plasminogen activator inhibitor-1 and platelet aggregation [7, 29].
-
Metabolic changes: obesity is closely associated with OSA. It was described as a cause of OSA, but it can also be worsen by OSA. The patients with new diagnosed OSA had a significant weight increase 1 year prior diagnosis of OSA compared to similar adipose patients without OSA [30]. Moreover, the CPAP therapy induces fat redistribution with reduction of subcutaneous and visceral fat [31]. Many studies have linked obesity to oxidative stress and inflammation underlying endothelial dysfunction. However, the presence of OSA in these studies was not systematically excluded. Recently, it has been shown that OSA rather than obesity may be predominantly responsible for increased oxidative stress, inflammation and endothelial impairment [32].
Pathophysiological interactions between OSA and atherosclerosis. The OSA-associated effects (top) initiate different pathomechanisms (left) that contribute to the development of atherosclerosis (down). Defined risk factors (right) augment the atherosclerotic changes underlying vascular diseases (down)
The metabolic deregulation in OSA may be connected to the impaired leptin secretion. The increased levels of leptin in OSA in comparison with weight-adjusted patients without OSA may influence the breath control centre. Additionally, leptin may enhance the rate of cardiovascular events via stimulation of sympathetic activity and platelet aggregation [33].
Moreover, the appetite and body fat regulators such as neuroleptid G and ghrelin are upregulated in OSA [34].
The OSA patients suffer often from metabolic syndrome that comprises obesity, hypertension, increased levels of triglycerides and glucose and decreased levels of HDL-cholesterol [35, 36]. Insulin resistance and diabetes mellitus, irrespective of obesity, are related to OSA degree [37, 38]. Additionally, hypoxia-induced insulin resistance has been demonstrated in an animal model [39]. Moreover, hypertension is widely accepted effect of OSA [4–9].
The most of the described pathomechanisms in OSA are positively influenced by CPAP therapy [4–9, 25, 28].
Epidemiological and clinical studies associating OSA with atherosclerotic disease
OSA is associated with coronary heart disease, myocardial infarction, ischaemic brain infarction, aorta dissection, hypertension, heart failure, erectile dysfunction, atrial fibrillation and sudden cardiac death. It is difficult to answer if OSA alone can act atherosclerotic, because the most OSA patients have additional atherosclerotic risk factors such as obesity, hypertension, hypercholesterolemia, insulin resistance and hyperglycaemia.
However, some animal and human studies attempted to demonstrate the direct link between OSA and atherosclerosis. Atherosclerosis could be induced in a mouse model of hypoxia on a cholesterol rich diet [40]. Moreover, the patients with OSA have increased intima-media thickness (IMT) of a carotid artery, enhanced pulse wave velocity (PWV) between carotid and femoralis artery [41] and increased coronary artery calcification in an electron beam tomography (EBCT) [42]. The above parameters are early markers of atherosclerosis. A small study could also show a reduction of IMT and PWV under CPAP therapy [43]. However, there are discrepant data concerning OSA and early atherosclerotic parameters, namely some studies could not demonstrate any association between OSA and IMT [44].
Impact of OSA on coronary artery disease/myocardial infarction
The prevalence of OSA in coronary artery disease is about two to three times higher compared to similar population without cardiovascular diseases. Oppositely, one can find OSA in 31–50% patients with angiographic documented coronary artery disease [4–9]. Although further studies have also demonstrated an association between OSA and coronary artery disease (Table 1a), their data are limited by study design with small patient number and lack of controls [45–52].
The most important prospective observational study by Marin et al. demonstrated that patients with an untreated severe OSA suffer more frequently from myocardial infarction and stroke than patients without OSA in a time frame of 10 years [48]. Additionally, the CPAP therapy could prevent the above vascular endpoints in the study. However, also this study must be interpreted carefully, because the results of non randomised behavioural studies can not always be confirmed in subsequent randomised studies. This fact depends for example on higher health awareness and/or better compliance of patients who accept CPAP therapy.
In contrast to the study of Martin et al., a community-based prospective cohort study of the cardiovascular consequences of OSA [53] demonstrated that the association of OSA with incident coronary heart disease after adjustment for other risk factors is much weaker and of no statistical significance. These discrepancies could be explained by older age of the cohort, as OSA-related cardiovascular risk decreases with age. Another finding of Gottlieb et al. was that OSA is a significant independent predictor of incident heart failure in men but not in women [53]. However, it is not clear, if sex differences may reflect a protective effect of a female sex against cardiovascular risk or may be only a result of a statistical underestimation because of lower prevalence of women with severe OSA and later age of onset of OSA in women than in men.
The positive influence of CPAP therapy on cardiovascular endpoints was shown in further studies [54–58 Table 1b]. It was demonstrated that an effective CPAP therapy can reduce nocturnal cardiac ischemia/ST-segment elevations by cardiologic patients [59].
Unfortunately, OSA appears to be strongly under diagnosed in patients with myocardial infarction, which probably results from persistent low awareness of OSA as a potential risk factor in cardiovascular patients [60].
OSA as a risk factor for ischaemic brain infarction
Sleep-associated disorders can be found in approximately 2/3 cases of ischaemic brain infarction; while obstructive sleep apnoea may be responsible for onset of ischaemic brain infarction, the central one can be an effect of stroke [61]. The genesis of OSA-associated ischaemic insult is probably the result of multiple factors: atherosclerotic plaques in brain vessels, hypertension, thrombembolic events based on atrial fibrillation, paradoxical embolism via enhanced right-left shunts by persistent foramen ovale. Moreover, OSA may disturb the cerebral autoregulation [62]. Lately, the association between microembolisms detected in MRT and OSA has been demonstrated [63]. A prospective study based on the questionnaire showed that snoring may be an independent risk factor for ischaemic insult [64]. Other studies were mostly cross-sectional or case-controlled that included only those patients who survived the cerebral insult [50–52, 56 Table 1]. Yaggi et al. demonstrated increased stroke rates in OSA patients in a longitudinal study, but in contrast to Marin et al. [48], they could not show any positive effects of CPAP therapy on the reduction of stroke incidence [50]. The explanation of this fact could be a low CPAP tolerance and reduced patients’ compliance. Finally, another study concerning OSA patients after stroke event showed that CPAP therapy reduced the cerebral re-infarction rate as compared to untreated patients.
Potential diagnostic biomarkers for prediction and prevention of OSA-associated atherosclerosis
Identification of OSA patients at a significant risk for the development of atherosclerosis is mandatory in order to prevent or at least to delay the occurrence of the vessel disease. It is known that similar degree of OSA severity may induce wide spectrum of atherosclerotic changes in different patients. It can be explained by interindividual differences in response to intermittent hypoxia which may mirror congenital or acquired disposition to disease or protective and regenerative potential. In order to anticipate if protective or deleterious mechanisms possibly predominate, several determinants have been proposed to serve as predictive biomarkers of the disease (Fig. 2). Besides direct changes in vessel structure and elasticity indicated by increase in arterial stiffness, intima/media thickness and atherosclerotic plaque volume which are already detectable in only minimal symptomatic OSA patients and are considered to be early markers of atherosclerosis [43, 65, 66], alternations in number and function of different blood cells and cytokines have been examined for their utility as biomarkers of atherosclerosis. An example of such a biomarker may be leukotriene B4 which production is upregulated in OSA indicating its role as an incipient mediator of atherosclerosis [67]. A number of further proinflammatory transcription factors and agents such as nuclear factor kappa B (NFkB), CRP, interleukin (il)-6, tumour necrosis factor (TNF)-alpha, intracellular adhesion molecule (ICAM)-1, vascular adhesion molecule (VCAM)-1, L-selectin, E-selectin, P-selectin and CD40 ligand as well as antioxidant genes [68–79] are elevated in OSA and play an important role in various stages of atherosclerosis. Moreover, decrease in anti-inflammatory acting interleukin-10 plasma levels predisposes to progression of atherosclerosis [80, 81]. Early inflammatory responses underlying atherosclerosis may be partly triggered by lipid accumulation in arterial wall as a result of decreased lipoprotein lipase activity found in OSA [82]. Conversely, inflammation und oxidative stress lead to metabolic disorders accelerating atherosclerotic changes underlying end organ damage [83–85]. Another OSA-associated disorder that contributes to the progression of vessel lesions is the unbalance between vasoactive substances such as nitric oxide (NO) and endothelin (ET)-1 [86], partly as a consequence of the overexpression of asymmetric dimethylarginine (ADMA) - endogenous inhibitor of NO production [87] observed in OSA. Furthermore, some studies suggested OSA-related upregulation of hypoxia-inducible factor (HIF) 1 alpha followed by the exaggerated release of proatherogenic substances like vascular endothelial growth factor (VEGF) and ET-1 [86, 88]. Additionally, serum amyloid A and myeloid-related protein 8/14 which are linked to augmented risk for cardiovascular morbidity may be also elevated in OSA [22, 89]. Likewise, cellular changes in blood may indicate later atherosclerotic vessel alternations in OSA. For instance, an attenuated apoptotic index of neutrophils with subsequent disturbance of injury limiting mechanisms demonstrated in OSA may help identify patients prone to cardiovascular pathologies [90]. Beyond that, OSA-induced hypoxic stress enhances invasive ability of monocytes, which may provide evidence for a raised risk for atherosclerosis development [91]. Another phenomenon occurring in OSA which accelerates atherosclerosis is the increased lipid accumulation in macrophages [92]. Lymphocyte activation with augmented cytotoxicity and cytokine imbalance in CD4 and CD8 T cells in OSA may also modulate some atherogenic pathways [93]. Higher numbers of circulating endothelial (Annexin-V positive), platelet and leukocyte apoptotic microparticles combined with diminished endothelial progenitor cell count indicating disturbed endothelial repair mechanisms are believed to be further biomarkers for atherosclerosis in OSA [94–97]. Perspectively, molecular studies on the telomere length, which is thought to reflect cumulative burden of oxidative stress and inflammation in OSA, may contribute to improvement of cardiovascular risk estimation in OSA patients [98]. Summarising, the risk stratification for the development of atherosclerosis in OSA by means of different biomarkers enables better monitoring of disease dynamics and, if required, early treatment initialisation.
Standard and innovative therapy options
Once OSA has been diagnosed, the decision of therapy start and treatment options depends on the polysomnography results, including severity of OSA, clinical symptoms, comorbidities and patients’ compliance. General therapy recommendations comprise common and sleep hygienic means, body weight reduction, physical activity, avoidance of alcohol and nicotine, training of sleep position, intraoral protrusion splints and operative methods. The gold standard is CPAP therapy, however the threshold value of AH-Index for therapy start is not known. Figure 3 shows therapeutic procedures in OSA that have not been tested in randomised trials and which must be optimised for individual patients. In particular, OSA patients at elevated risk for cardiovascular diseases benefit mostly from early diagnosis and appropriate treatment. However, the recognition of such patients is not easy as there are no standardised criteria for cardiovascular risk stratification in OSA. Many biomarkers described above have been proposed to be a useful tool for anticipation of a vessel disease. Further randomised studies are needed to prove their clinical relevance.
Although the mainstay of therapy in OSA is a treatment with CPAP, the targeted therapy of concomitant, independent of OSA cardiovascular risk factors is at least of the same importance. Therefore, the primary and secondary prevention of the known predisposing factors for the development of atherosclerosis such as hypertension, diabetes, dyslipidemia by accurate screening, efficient therapy and frequent controls improves the effectiveness of CPAP therapy in minimising of the atherosclerosis risk.
Since the adherence to CPAP therapy for OSA is poor [10] and CPAP treatment does not eliminate all hypoxia-associated risk factors for atherosclerosis [99], many study designs have focused on the development of alternative therapies. Besides the introduction of improved forms of positive airway pressure therapies [100] and innovative devices such as electrical stimulator of the hypoglossal nerve [101], different pharmacological interventions have been tested. Based on the hypothesis that OSA mediates its proatherogenic effects via induction of oxidative stress, the antioxidant strategies such as the intravenous infusion of antioxidant vitamin C in OSA subjects have been demonstrated to improve endothelial function [102]. Investigations in the field of the involvement of the endothelin system in cardiovascular pathologies associated with OSA showed the beneficial cardiovascular effects of bosentan, a mixed endothelin antagonist, in OSA [88]. In a subgroup of patients with both OSA and hyperlipidemia, targeting of a sterol regulatory element binding protein (SREBP)-1, a transcription factor for lipid biosynthesis, which is upregulated in OSA, could improve lipid metabolism and in consequence cardiovascular prognosis [103]. Future studies are mandatory to prove attractive therapy concepts such as administration of anti-inflammatory substances, restoration of endothelial repair capacity via treatment with endothelial progenitor cells or molecular approaches taking into consideration genetically defined polymorphisms for prooxidant and antioxidant enzymes determining individual susceptibility to cardiovascular changes in OSA.
Perspectives
Despite coexistence of not homogenous study results and problems by establishment of a good study design because of confusing factors, there are many convincing data demonstrating that obstructive sleep apnoea may influence negatively the development of atherosclerosis. Therefore, higher awareness in recognising of OSA, early diagnosis and effective therapy of OSA are indispensible elements of an appropriate medical care, particularly in patients at cardiovascular risk. Future directions comprise further studies on the mechanisms of cardiovascular damage in OSA in order to find new biomarkers of atherosclerosis characterised by their simplicity, noninvasivness and combined high sensitivity and specificity as well as to develop alternative therapy modalities. The knowledge of individual risk factors and different possible cardiovascular responses to the intermittent hypoxia will help improve personalised treatment.
References
Skowasch D, Boenner G, Nickenig G. Obstructive sleep apnea as a risk factor of atherosclerosis. Somnologie. 2009;13:215–20.
Werner N, Kosiol S, Schiegl T, et al. Circulating endothelial progenitor cells and cardiovascular outcomes. N Engl J Med. 2005;353:999–1007.
Andrié R, Bauriedel G, Braun P, Höpp HW, Nickenig G, Skowasch D. Increased expression of C-reactive protein and tissue factor in acute coronary syndrome lesions. Atherosclerosis. 2009;202:135–43.
Podszus T, Seevers H, Mayer G, Baumann G. Positionspapier “Schlafmedizin in der Kardiologie”. Kardiologe. 2009;3:9–15.
Caples SM, Garcia-Touchard A, Somers VK. Sleep-disordered breathing and cardiovascular risk. Sleep. 2007;30:291–304.
Lüthje L, Andreas S. Obstructive sleep apnea and coronary artery disease. Sleep Med Rev. 2008;12:19–31.
Lopez-Jimenez F, Kuniyoshi FHS, Gami A, Somers VK. Obstructive sleep apnea. Implications of cardiac and vascular disease. Chest. 2008;133:793–804.
Somers VK, White DP, Amin R, et al. Sleep apnea and cardiovascular disease. Circulation. 2008;118:1080–111.
Bradley TD, Floras JS. Obstructive sleep apnoea and its cardiovascular consequences. Lancet. 2009;373:82–9310.
Basner RC. Continuous positive airway pressure for obstructive sleep apnea. N Engl J Med. 2007;356:1751–8.
Somers VK, Dyken ME, Clary MP, Abboud FM. Sympathetic neural mechanisms in obstructive sleep apnea. J Clin Invest. 1995;96:1897–904.
Phillips BG, Narkiewicz K, Pesek CA, Haynes WG, Dyken ME, Somers VK. Effects of obstructive sleep apnea on endothelin-1 and blood pressure. J Hypertens. 1999;17:61–6.
Teramoto S, Kume H, Matsuse T, et al. Oxygen administration improves the serum level of nitric oxide metabolites in patients with obstructive sleep apnea syndrome. Sleep Med. 2003;4:403–7.
Kraiczi H, Caidahl K, Samuelsson A, Peker Y, Hedner J. Impairment of vascular endothelial function and left ventricular filling: association with the severity of apnea-induced hypoxemia during sleep. Chest. 2001;119:1085–91.
Lavie L, Lavie P. Molecular mechanisms of cardiovascular disease in OSAHS: the oxidative stress link. Eur Respir J. 2009;33:1467–84.
Duchna HW, Guilleminault C, Stoohs RA, et al. Obstructive sleep apnea syndrome: a cardiovascular risk factor? Z Kardiol. 2001;90:568–75.
Arias MA, Garcia-Rio F, Alonso-Fernandez A, Mediano O, Martínez I, Villamor J. Obstructive sleep apnea syndrome affects left ventricular diastolic function: effects of nasal continuous positive airway pressure in men. Circulation. 2005;112:375–83.
Ohga E, Tomita T, Wada H, Yamamoto H, Nagase T, Ouchi Y. Effects of obstructive sleep apnea on circulating ICAM-1, IL-8, and MCP-1. J Appl Physiol. 2003;94:179–84.
Zund G, Uezono S, Stahl GL, et al. Hypoxia enhances induction of endothelial ICAM-1: role for metabolic acidosis and proteasomes. Am J Physiol. 1997;273:C1571–80.
Dyugoskava L, Lavie P, Lavie L. Increased adhesion molecules expression and production of reactive oxygen species in leukocytes of sleep apnea patients. Am J Respir Crit Care Med. 2002;165:934–9.
Shamsuzzaman AS, Winnicki M, Lanfranchi P, et al. Elevated C-reactive protein in patients with obstructive sleep apnea. Circulation. 2002;105:1462–4.
Svatikova A, Wolk R, Shamsuzzaman AS, Kara T, Olson EJ, Somers VK. Serum amyloid A in obstructive sleep apnea. Circulation. 2003;108:1451–4.
Larkin EK, Rosen CL, Kirchner HL, et al. Variation of C-reactive protein levels in adolescents: association with sleep-disordered breathing and sleep duration. Circulation. 2005;111:1978–84.
Guillemiault C, Kirisoglu C, Ohayon MM. C-reactive protein and sleep-disordered breathing. Sleep. 2004;27:1507–11.
Jelic S, Padeletti M, Kawut SM, et al. Inflammation, oxidative stress and repair capacity of the vascular endothelium in obstructive sleep apnea. Circulation. 2008;117:2270–8.
Ip MS, Tse HF, Lam B, Tsang KW, Lam WK. Endothelial function in obstructive sleep apnea and response to treatment. Am J Respir Crit Care Med. 2004;169:348–53.
El Solh AA, Akkinnusi ME, Baddoura FH, Mankowski CR. Endothelial cell apoptosis in obstructive sleep apnea: a link to endothelial dysfunction. Am J Respir Crit Care Med. 2007;175:1186–91.
Nieto FJ, Herrington DM, Redline S, Benjamin EJ, Robbins JA. Sleep apnea and marker of vascular endothelial function in a large community sample of older adults. Am J Crit Care Med. 2004;169:354–60.
von Känel R, Loredo JS, Ancoli-Israel S, Mills PJ, Natarajan L, Dimsdale JE. Association between polysomnographic measures of disrupted sleep and prothrombotic factors. Chest. 2007;131:733–9.
Phillips BG, Hisel TM, Kato M, et al. Recent weight gain in patients with newly diagnosed obstructive sleep apnea. J Hypertens. 1999;17:1297–300.
Chin K, Shimizu K, Nakamura T, et al. Changes in intraabdominal visceral fat and serum leptin levels in patients with obstructive sleep apnea syndrome following nasal continuous sleep apnea pressure therapy. Circulation. 1999;100:706–12.
Jelic S, Lederer DJ, Adams T, et al. Vascular inflammation in obesity and sleep apnea. Circulation. 2010;121:1014–21.
Philipps BG, Kato M, Narkiewicz K, Choe I, Somers VK. Increases in leptin levels, sympathetic drive and weight gain in obstructive sleep apnea. Am J Physiol Heart Circ Physiol. 2000;279:H234–7.
Barceló A, Barbé F, Llompart E, et al. Neuropeptide Y and leptin in patients with obstructive sleep apnea syndrome: role of obesity. Am J Respir Crit Care Med. 2005;171:183–7.
Coughlin SR, Mawdyley L, Mugarza JA, Calverley PM, Wilding JP. Obstructive sleep apnea is independently associated with an increased prevalence of metabolic syndrome. Eur Heart J. 2004;25:735–41.
McArdle N, Hillman D, Beilin L, Watts G. Metabolic risk factors for vascular disease in obstructive sleep apnea: a link to endothelial dysfunction. Am J Respir Crit Care Med. 2007;175:190–5.
Ip MS, Lam B, Ng MM, Lam WK, Tsang KW, Lam KS. Obstructive sleep apnea is independently associated with insulin resistance. Am J Respir Crit Care Med. 2002;165:670–6.
Elmasry A, Lindberg E, Berne C, et al. Sleep-disordered breathing and glucose metabolism in hypertensive men: a population-based study. J Intern Med. 2001;249:153–61.
Iiyori N, Alonso LC, Li J, et al. Intermittent hypoxia causes insulin resistance in lean mice independent of autonomic activity. Am J Respir Crit Care Med. 2007;175:851–7.
Savransky V, Nanayakkara A, Li J, et al. Chronic intermittent hypoxia induces atherosclerosis. Am J Respir Crit Care Med. 2007;175:1290–2197.
Drager LF, Bortolotto LA, Lorenzi MC, Figueiredo AC, Krieger EM, Lorenzi-Filho G. Early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med. 2005;172:613–8.
Sorajja D, Gami AS, Somers VK, Behrenbeck TR, Garcia-Touchard A, Lopez-Jimenez F. Independent association between obstructive sleep apnea and subclinical coronary artery disease. Chest. 2008;133:927–33.
Drager LF, Bortolotto LA, Figueiredo AC, Krieger EM, Lorenzi GF. Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am Respir Crit Care Med. 2007;176:706–12.
Wattanakit K, Boland L, Punjabi NM, Shabar E. Relation of sleep-disordered breathing to carotid plaque and intima-media thickness. Atherosclerosis. 2008;197:125–31.
Schäfer H, Koehler U, Ewig S, Hasper E, Tasci S, Lüderitz B. Obstructive sleep apnea as risk factor in coronary artery disease. Cardiology. 1999;92:79–84.
Mooe T, Franklin KA, Holmström K, Rabben T, Wiklund U. Sleep-disordered breathing and coronary artery disease: long-term prognosis. Am J Respir Crit Care Med. 2001;164:1910–3.
Shahar E, Whitney DW, Redline S, et al. Sleep-disordered breathing and cardiovascular disease. Cross sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med. 2001;163:19–25.
Marin JM, Carrizo SJ, Vicente E, Augusti AGN. Long-term cardiovascular outcomes in men with obstructive sleep apnea-hypopnea with or without treatment with continuous positive airway pressure: an observational study. Lancet. 2005;365:1046–53.
Arzt M, Young T, Finn L, Skatrud JB, Bradley TD. Association of sleep-disordered breathing and the occurrence of stroke. J Respir Crit Care Med. 2005;172:1447–51.
Yaggi HK, Concato J, Kernan WN, Lichtman JH, Brass LM, Mohsenin V. Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med. 2005;353:2034–41.
Yumino D, Tsurumi Y, Takagi A, Suzuki K, Kasanuki H. Impact of obstructive sleep apnea on clinical and angiographic outcomes following percutaneous coronary intervention in patients with acute coronary syndrome. Am J Cardiol. 2007;99:26–30.
Valham F, Mooe T, Rabben T, Stenlund H, Wiklund U, Franklin KA. Increased risk of stroke in patients with coronary artery disease and sleep apnea: a 10-year follow-up. Circulation. 2008;118:995–60.
Gottlieb DJ, Yenokyan G, Newman AB, et al. Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the sleep heart health study. Circulation. 2010;122:352–60.
Doherty LS, Kiely JL, Swan V, McNicholas WT. Long-term effect of nasal continuous positive airway pressure therapy on cardiovascular outcomes in sleep apnea syndrome. Chest. 2005;127:2076–84.
Milleron O, Pilliere R, Foucher A, et al. Benefits of obstructive sleep apneas treatment in coronary artery disease: a long-term follow-up study. Eur Heart J. 2004;25:728–34.
Martinez-Garzia MA, Galiano-Blancart R, Roman-Sanchez P, et al. Continuous positive airway pressure treatment in sleep apnea prevents new vascular events after ischemic stroke. Chest. 2005;128:2123–9.
Buchner NJ, Sanner BM, Borgel J, Rump LC. Continuous positive airway pressure treatment of mild to moderate obstructive sleep apnea reduces cardiovascular risk. Am J Respir Crit Care Med. 2007;176:1274–80.
Cassar A, Morgenthaler TI, Lennon RJ, Rihal CS, Lerman A. Treatment of obstructive sleep apnea is associated with decreased cardiac death after percutaneous coronary intervention. J Am Coll Cardiol. 2008;50:1310–4.
Peled N, Abinader EG, Pillar G, Sharif D, Lavie P. Nocturnal ischemic events in patients with obstructive sleep apnea syndrome and ischemic heart disease: effects of continuous positive air pressure treatment. J Am Coll Cardiol. 1999;34:1744–9.
Konecny T, Kuniyoshi FH, Orban M, et al. Under-diagnosis of sleep apnea in patients after acute myocardial infarction. J Am Coll Cardiol. 2010;56:742–3.
Parra O, Arboix A, Bechich S, et al. Time course of sleep-related breathing disorders in first-ever stroke or transient ischemic attack. Am J Respir Crit Care Med. 2000;161:375–80.
Franklin KA. Cerebral haemodynamics in obstructive sleep apnea and Cheyne-Stokes respiration. Sleep Med Rev. 2002;6:429–41.
Minoguchi K, Yokoe T, Tazaki T, et al. Silent brain infarction and platelet activation in obstructive sleep apnea. Am J Respir Crit Care Med. 2007;1(7):612–7.
Hu FB, Willett WC, Manson JE, et al. Snoring and risk of cardiovascular disease in women. J Am Coll Cardiol. 2000;35:308–13.
Kohler M, Craig S, Nicoll D, Leeson P, Davies RJ, Stradling JR. Endothelial function and arterial stiffness in minimally symptomatic obstructive sleep apnea. Am J Respir Crit Care Med. 2008;178:984–8.
Turmel J, Sériès F, Boulet LP, et al. Relationship between atherosclerosis and the sleep apnea syndrome: an intravascular ultrasound study. Int J Cardiol. 2009;132:203–9.
Lefebvre B, Pépin JL, Baguet JP, et al. Leukotriene B4: early mediator of atherosclerosis in obstructive sleep apnoea? Eur Respir J. 2008;32:113–20.
Ryan S, Taylor CT, McNicholas WT. Systemic inflammation: a key factor in the pathogenesis of cardiovascular complications in obstructive sleep apnoea syndrome? Postgrad Med J. 2009;85:693–8.
Lavie L, Polotsky V. Cardiovascular aspects in obstructive sleep apnea syndrome - Molecular issues, hypoxia and cytokine profiles. Respiration. 2009;78:361–70.
Kheirandish-Gozal L, Capdevila OS, Tauman R, Gozal D. Plasma C-reactive protein in nonobese children with obstructive sleep apnea before and after adenotonsillectomy. J Clin Sleep Med. 2006;2:301–4.
Minoguchi K, Yokoe T, Tazaki T, et al. Increased carotid intima-media thickness and serum inflammatory markers in obstructive sleep apnea. Am J Respir Crit Care Med. 2005;172:625–30.
Minoguchi K, Tazaki T, Yokoe T, et al. Elevated production of tumor necrosis factor-alpha by monocytes in patients with obstructive sleep apnea syndrome. Chest. 2004;126:1473–9.
Ohga E, Nagase T, Tomita T, et al. Increased levels of circulating ICAM-1, VCAM-1, and L-selectin in obstructive sleep apnea syndrome. J Appl Physiol. 1999;87:10–4.
El-Solh AA, Mador MJ, Sikka P, Dhillon RS, Amsterdam D, Grant BJ. Adhesion molecules in patients with coronary artery disease and moderate-to-severe obstructive sleep apnea. Chest. 2002;121:1541–7.
Nácher M, Serrano-Mollar A, Farré R, Panés J, Seguí J, Montserrat JM. Recurrent obstructive apneas trigger early systemic inflammation in a rat model of sleep apnea. Respir Physiol Neurobiol. 2007;155:93–6.
Kobayashi K, Nishimura Y, Shimada T, et al. Effect of continuous positive airway pressure on soluble CD40 ligand in patients with obstructive sleep apnea syndrome. Chest. 2006;129:632–7.
Polotsky VY, Savransky V, Bevans-Fonti S, et al. Intermittent and sustained hypoxia induce a similar gene expression in the human aortic endothelial cells. Physiol Genomics. 2010. doi:10.1152/physiolgenomics.00091.2009.
Quercioli A, Mach F, Montecucco F. Inflammation accelerates atherosclerotic processes in obstructive sleep apnea syndrome (OSAS). Sleep Breath. 2010;14:261–9.
Arnaud C, Dematteis M, Pepin JL, Baguet JP, Lévy P. Obstructive sleep apnea, immuno-inflammation, and atherosclerosis. Semin Immunopathol. 2009;31:113–25.
Gozal D, Serpero LD, Sans Capdevila O, Kheirandish-Gozal L. Systemic inflammation in non-obese children with obstructive sleep apnea. Sleep Med. 2008;9:254–9.
Dyugovskaya L, Lavie P, Lavie L. Phenotypic and functional characterization of blood gammadelta T cells in sleep apnea. Am J Respir Crit Care Med. 2003;168:242–9.
Iesato K, Tatsumi K, Saibara T, et al. Decreased lipoprotein lipase in obstructive sleep apnea syndrome. Circ J. 2007;71:1293–8.
Lam JC, Ip MS. Obstructive sleep apnea and the metabolic syndrome. Expert Rev Respir Med. 2009;3:177–86.
Tkacova R, Rizzo M, Berneis K. Therapy with noninvasive ventilation in patients with obstructive sleep apnoea: effects on atherogenic lipoprotein phenotype. Med Hypotheses. 2009;73:441–4.
Adeseun GA, Rosas SE. The impact of obstructive sleep apnea on chronic kidney disease. Curr Hypertens Rep. 2010;12:378–83.
Lavie L. Obstructive sleep apnoea syndrome- an oxidative stress disorder. Sleep Med Rev. 2003;7:35–51.
Barceló A, de la Peña M, Ayllón O, et al. Increased plasma levels of asymmetric dimethylarginine and soluble CD40 ligand in patients with sleep apnea. Respiration. 2009;77:85–90.
Belaidi E, Joyeux-Faure M, Ribuot C, Launois SH, Levy P, Godin-Ribuot D. Major role for hypoxia inducible factor-1 and the endothelin system in promoting myocardial infarction and hypertension in an animal model of obstructive sleep apnea. J Am Coll Cardiol. 2009;53:1309–17.
Kim J, Bhattacharjee R, Snow AB, Capdevila OS, Kheirandish-Gozal L, Gozal D. Myeloid-related protein 8/14 levels in children with obstructive sleep apnoea. Eur Respir J. 2010;35:843–50.
Dyugovskaya L, Polyakov A, Lavie P, Lavie L. Delayed neutrophil apoptosis in patients with sleep apnea. Am J Respir Crit Care Med. 2008;177:544–54.
Tamaki S, Yamauchi M, Fukuoka A, et al. Nocturnal hypoxic stress activates invasive ability of monocytes in patients with obstructive sleep apnoea syndrome. Respirology. 2009;14:689–94.
Lattimore JD, Wilcox I, Nakhla S, Langenfeld M, Jessup W, Celermajer DS. Repetitive hypoxia increases lipid loading in human macrophages-a potentially atherogenic effect. Atherosclerosis. 2005;179:255–9.
Dyugovskaya L, Lavie P, Lavie L. Lymphocyte activation as a possible measure of atherosclerotic risk in patients with sleep apnea. Ann NY Acad Sci. 2005;1051:340–50.
El Solh AA, Akinnusi ME, Baddoura FH, Mankowski CR. Endothelial cell apoptosis in obstructive sleep apnea: a link to endothelial dysfunction. Am J Respir Crit Care Med. 2007;175:1186–91.
El Solh AA, Akinnusi ME, Berim IG, Peter AM, Paasch LL, Szarpa KR. Hemostatic implications of endothelial cell apoptosis in obstructive sleep apnea. Sleep Breath. 2008;12:331–7.
Ayers L, Ferry B, Craig S, Nicoll D, Stradling JR, Kohler M. Circulating cell-derived microparticles in patients with minimally symptomatic obstructive sleep apnoea. Eur Respir J. 2009;33:574–80.
Jelic S, Lederer DJ, Adams T, et al. Endothelial repair capacity and apoptosis are inversely related in obstructive sleep apnea. Vasc Health Risk Manag. 2009;5:909–20.
Barceló A, Piérola J, López-Escribano H, et al. Telomere shortening in sleep apnea syndrome. Respir Med. 2010;104:1225–9.
Karkoulias K, Lykouras D, Sampsonas F, et al. The role of Endothelin-1 in obstructive sleep apnea syndrome and pulmonary arterial hypertension: pathogenesis and Endothelin-1 antagonists. Curr Med Chem. 2010;17:1059–66.
Antonescu-Turcu A, Parthasarathy S. CPAP and Bi-level PAP therapy: new and established roles. Respir Care. 2010;55:1216–29.
Vanderveken OM, Boudewyns A, Ni Q, et al. Cardiovascular implications in the treatment of obstructive sleep apnea. J Cardiovasc Transl Res. 2011;4:53–60.
Grebe M, Eisele HJ, Weissmann N, et al. Antioxidant vitamin C improves endothelial function in obstructive sleep apnea. Am J Respir Crit Care Med. 2006;173:897–901.
Li J, Nanayakkara A, Jun J, Savransky V, Polotsky VY. Effect of deficiency in SREBP cleavage-activating protein on lipid metabolism during intermittent hypoxia. Physiol Genomics. 2007;31:273–80.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Tuleta, I., Pabst, S., Juergens, U.R. et al. Obstructive sleep apnoea as a risk factor for atherosclerosis – implication for preventive and personalised treatment. EPMA Journal 2, 39–47 (2011). https://doi.org/10.1007/s13167-011-0070-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13167-011-0070-5