Abstract
Geochemical signatures of groundwater in the coastal aquifers of Kadaladi block of Ramanathapuram District, Tamilnadu, India were used to identify the chemical processes that control hydrochemistry. The parameters such as pH, EC, TDS and major ion concentrations Na, K, Ca, Mg, Cl, HCO3, SO4, PO4 and NO3 of the groundwater were analyzed. Abundances of these ions are in the following order Na > Ca > Mg > K and Cl > HCO3 > SO4 > NO3 > PO4. Na–Cl is the dominant hydrochemical facies of the study area. Interpretation of the hydrochemical data suggests that hydrochemistry of the study area is controlled by mixing of fresh water with relict saline water, ion-exchange processes, silicate weathering and evaporation are responsible for the groundwater chemistry of the study area. Above statement is further supported by Gibbs plot where most of the samples fall within the evaporation zone. Good quality groundwater is restricted to coastal sand dunes running parallel to the coast and it is important to conserve these dunes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Groundwater is the backbone of Indian agriculture and drinking water security. It is a common pool resource, used by millions of farmers across the country. Due to the ever increasing demand for potable and irrigation water and inadequacy of available surface water, the importance of groundwater is increasing exponentially every day (World Bank 2010; Selvam and Sivasubramanian 2012). Groundwater remains the only drinking water source in most of Indian rural household. With an estimated 30 million groundwater structures, India is fast hurtling towards a serious crisis of groundwater overuse and quality deterioration (World Bank Report 2010). Groundwater is threatened by several factors related to its mismanagement. Degradation of groundwater quality in coastal region generally occurs due to natural processes such as saline water intrusion, wind driven sea spray and marine aerosols deposited on the top soil, evaporation and interaction of groundwater with brines and sedimentary formation (Sanford et al. 2007). Apart from the natural process, anthropogenic contaminations such as industrial effluents, agricultural fertilizers, municipal waste water, septic tank effluent and landfill are other major causes of water quality deterioration (Mondal et al. 2008; Mondal and Singh 2011; Selvam et al. 2013).
Seawater intrusion is one of the most common problems in almost all coastal aquifers around the globe (Melloul and Goldenberg 1997, 1998; Sivakumar and Elango 2008; Chidambaram et al. 2009; Mondal et al. 2010, 2011; Srinivasamoorthy et al. 2011). This phenomenon can be explained by the variety of conditions like gentle coastal hydraulic gradients, tidal and estuarine activity, sea level rises, low infiltration, excessive withdrawal and local hydrogeological conditions (Sarma et al. 1982; Longe et al. 1987; Rajmohan et al. 1997, 2000; Barret et al. 2002; Saxena et al. 2004; Kacimov et al. 2009). The common method for assessing seawater intrusion through an aquifer in coastal belts is a periodic analysis of groundwater (Todd 1980; Kim et al. 2009; Mondal et al. 2010, 2011 and Krishnakumar et al. 2012). Chemistry of groundwater is an important factor determining its use for domestic, irrigation and industrial purposes. Interaction of groundwater with aquifer minerals through which it flows greatly controls the groundwater chemistry. Hydrogeochemical processes that are responsible for altering the chemical composition of groundwater vary with respect to space and time. The hydrogeochemical processes of groundwater system help to obtain an insight into the contributions of rock/soil–water interaction (Senthilkumar and Elango 2013). Very few research works have been conducted with the aim of assessing the chemical composition of groundwater in the coastal regions of Tamilnadu. When considering the present study area, so far there is no scientific publication available related to groundwater quality. In this work, a detailed investigation has been carried out to evaluate the geochemical process regulating groundwater quality.
Study area
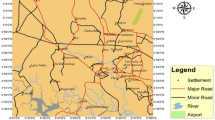
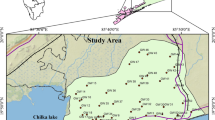
Kadaladi block of Ramanathapuram district, Tamilnadu, India lies between the latitude 9° 6′–9° 21′ N and longitude 78° 18′–78° 45′ E and covered by the Survey of India Toposheets 58 K/7, 58 K/8, 58 K/11 and 12 (Fig. 1). Total areal extent of the present study area is 623 km2. Presently, Ramanathapuram district is divided into 11 blocks and Kadaladi block is further divided into 60 Panchyat villages. Kadaladi block is mainly covered by sedimentary formation and about 70 % of the study area is covered with Fluvial, Fluvio-Marine, Aeolian and marine sediments of Quaternary age. The Fluvial deposits are made up of sand, silt and clay. The western side of the study area is exposed with Archaean rocks such as Garnetiferous Biotite Gneiss, Quartzite and Charnockites which are intruded later by Pink Granite. The Aeolian deposits are made up of red sands and they are in the form of ancient sand dunes. These dunes run ~10 km parallel to the coast and extend up to 3 km across. These sand dunes are separated by marshy deposits of black clays from the sea coast. The sands are underlain by calcareous hardpan. The study area enjoys a tropical climate. In the plains, the maximum and minimum temperatures recorded are 39 and 29 °C, respectively. Heat becomes intense in March to September and shoots further up unless interfered by rains. Humidity is relatively high as 79 % on an average and ranges between 80 and 90 % in coastal regions. Coastal areas are flanked by beach ridge complex, swales, swamps and backwater. The sand flat is another feature of the coast comprising clay, silt and often inundated by seawater and encrusted with salt. Major rivers flowing through the study area are Gundar River and Palar River. They flow from northwest to southeast within the study area and enter into Bay of Bengal. The drainage pattern, in general, is dendritic. Both the rivers are seasonal and flow only during monsoon season. In addition to this, a small Kadugusandai Odai also flows through the study area.
The district receives rain under the influence of both southwest and northeast monsoons. The northeast monsoon chiefly contributes to the rainfall in the district. Most of the precipitation occurs in the form of cyclonic storms caused due to the depressions in the Bay of Bengal. The southwest monsoon rainfall is highly erratic and summer rains are negligible. Rainfall data from two stations over the period 1901–2000 were utilized and a perusal of the data shows that the normal annual rainfall over the district is 827 mm and decreases toward inland. The general practice of people in the study area is to store rain water in tanks and use them for their needs throughout the year (Fig. 2). But most monsoons fail. To cater the need of the people, Tamilnadu Government has established a desalination plant at Naripayur located within the study area. This plant produces good quality drinking water from sea water and supplies them to the surrounding villages.
The alluvium with alternate layers of sand and clay with a thickness of 15–25 m and are characterized by floating freshwater lenses limited to a depth of 6–7 m below ground level. It can sustain a pumping of up to 2 h and can yield about 2–5 l/s. So people prefer dug wells of 3–6 m depth than the bore wells of 30 or 50 m and extract good quality groundwater which floats as lens on the saline water. Comparatively good quality of water is available in the narrow strip of land between red sand dunes running roughly 10 km parallel to the coast and the coastline (Fig. 3). Natural recharge from rainfall elevates the groundwater level and replenishes the fresh water lens of these sand dune areas. In the study area, there are no deep aquifer has been reported so far. During our reconnaissance survey, we have visited the drilling sites of Mineral Exploration Corporation Ltd (MECL) who were in search of lignite beds. They have not encountered any hard rock or lignite up to the depth of 400 m.
Materials and methods
Collection of groundwater samples
Totally 36 wells were identified during our reconnaissance survey for the collection of water samples to carry out chemical analysis. Due care was given to the distribution of wells throughout the study area during well selection. Dug well and bore well samples were collected during the pre-monsoon (PRM) period of 2012 (June 2012). Methods of collection and analysis of groundwater samples were essentially the same as given by Brow et al. (1983), APHA (1995). The samples were collected in 1 l capacity polythene bottles. Prior to the collection, bottles were thoroughly washed with diluted HNO3 acid, and then with distilled water in the laboratory before filling the bottle with the sample. Portable Eco-testers were used to measure TDS, EC and pH on-site. The precise locations of the sampling points were also determined in the field using GARMIN 12 Channel GPS. The well locations of the groundwater samples are given in Fig. 1.
Chemical analysis of groundwater
The groundwater samples collected were analyzed for various physicochemical parameters as described by the American Public Health Association (APHA 1995). Calcium (Ca) and Magnesium (Mg) were determined titrimetrically using standard EDTA; Chloride(Cl) was determined by standard AgNo3 titration; bicarbonate (HCO3) was determined by titration with HCL; sodium (Na+) and potassium (K+) were determined by Flame Spectrometer; Sulphate (SO4) was determined by spectrophotometric turbidimetry. Majority of the analyzed samples show ion balance errors within ±5 %, indicating the results are generally acceptable. All concentrations are expressed in milligrams per liter (mg/l), except pH, EC and TDS. The EC concentrations were expressed in μS/cm and TDS concentrations were expressed in ppm. Concentration of chemical parameters of water samples are given in Table 1.
Results and discussion
General characteristics
The results of chemical analysis for the groundwater samples collected in June, 2012 are listed in Table 1. Groundwater in the study area is generally alkaline in nature with pH ranging from 7.2 to 8.2, with an average of 7.8. EC is an indirect measure of ionic strength and mineralization of natural water. EC of groundwater samples of the study area ranges from 190 to 47,600 μS/cm, with an average of 8,226 μS/cm. Total dissolved solids (TDS), which is generally the sum of dissolved ionic concentration varies between 116 and 33,320 mg/l with an average of 5,406 mg/l. Table 1 clearly shows that about 89 % of samples collected falls under brackish water category. Lower pH (7.2), EC (190 μS/cm) and TDS (116 ppm) has been recorded in the samples collected from Roachmanagar, Velayuthapuram, Vellapatti, M. Karisalkulam and Terku Narippaiyur. Higher pH (8.2), EC (47,600 μS/cm) and TDS (33,320 ppm) has been recorded from Valamppatti, T. M. Kottai, Peikulam, Enathi, Komboothi and Ervadi.
Among the cations, the minimum concentration of Na, Ca, Mg and K are 16, 11, 10 and 5 mg/l and the maximum concentrations are 6,500, 1,872, 1,838 and 310 mg/l respectively. The averages of these cations are 956, 284, 258 and 59 respectively. The order of abundance of these cations is Na > Ca > Mg > K.
Geochemical classification
Trilinear plotting systems are used to study water chemistry and quality (Piper 1944). On conventional trilinear diagrams sample values for three cations (Calcium, Magnesium and Alkali metals-Sodium and Potassium) and three anions (Bicarbonate, Chloride and Sulfate) are plotted relative to one another. These ions are generally the most common constitutions in unpolluted groundwater. Fundamental interpretations of the chemical nature of water samples are based on the location of the sample ion values.
Distinct zones within aquifers having defined water chemistry properties are referred to as hydrochemical facies (Vasanthavigar et al. 2010). Determining the nature and distribution of hydrochemical facies can provide insight into how groundwater quality changes within and between aquifers. In Fig. 4, the HCO3–Cl–SO4 anion triangle shows groundwater samples plotted between the bicarbonate type and chloride type end member and Sulfate (SO4) is not present in any significant proportion. In Ca–Mg–Na cation triangle samples clustered off-center towards Sodium type groundwater. These two triangles projected onto the main diamond field. If the groundwater is simply mixed with seawater, groundwater composition should be plotted on a straight line connecting the background freshwater and seawater. However, the groundwater in the study area shows a curved path (arrow) of hydrochemical evolution, which starts from the CaHCO3 type via the mixed CaMgCl type and ends to the NaCl type. This is a typical pattern which indicates the cation exchange reaction when seawater incursion occurs (Richter et al. 1993). This pattern suggests that the groundwater chemistry in the study area changes due to the cation exchange reaction as well as the simple mixing with seawater as a result of seawater incursion. Figure 5, showing the spatial distribution of groundwater type within the study areas reveals that groundwater available in the coastal sand dunes is comparatively fresh and come under CaHCO3 and mixed CaNaHCO3 type. This type of groundwater may have resulted due the recent recharge of the coastal sand dunes by rainwater. Majority of the groundwater within the study area comes under NaCl and mixed CaMgCl type. The higher salinity of the groundwater of the present study area may be due to the mixing of fresh water with relict saline water from a high stand of sea level in past geological time. Since the study area comes under a semi-arid climate, the absence of effective natural flushing activity enhances accumulation of salts and saline water (Cardona et al. 2004).
In addition to Piper diagram, Gibbs plots were also used to gain better insight into hydrochemical processes such as precipitation, rock water interaction and evaporation on groundwater chemistry in the study area. Gibbs (1970) demonstrated that if TDS is plotted against Na/(Na + Ca) and Cl/Cl + HCO3, this would provide information on the mechanism controlling the chemistry of waters. Figure 6a, b displays that groundwater samples were plotted mostly in the evaporation zone and few samples in the rock water interaction zone. Cation points are scattered. But the anion shows some linear trend because the increase in Cl and HCO3 may due to the level of mixing of sea water and evaporation. It is expected that as evaporation greatly increases the concentration of ions formed by chemical weathering of subsurface lithology, leading to higher salinity (Volker 1961; Stuyfzand P 1993). Both Piper and Gibbs suggest that groundwater chemistry is mainly regulated by mixing of high saline water and evaporation.
Geochemical process
The compositional relations among the dissolved ions can be used to assess the origin of solutes and the processes that results in water composition. The chemical data of groundwater samples are plotted on Na Versus Cl scatter plot. The Na/Cl relationship has often been used to identify the mechanism for acquiring salinity and saline intrusions. Sodium and Chloride show a good correlation (Fig. 7a), indicating groundwater are probably controlled by water–rock interaction, most likely derived from the weathering of calcium–magnesium silicates, chiefly from calcite, plagioclase, gypsum and feldspar (Srinivasamoorthy et al. 2011). However, increased concentration of HCO3 compared to Na concentration (Fig. 7b) in groundwater suggests that silicate weathering also occurs (Fisher and Mullican 1997).The evolution of the slopes of Ca and Mg with HCO3 gives valuable information about the sources of Ca and Mg in groundwater (Richter et al. 1993). Ca + Mg Versus HCO3 plot (Fig. 7c) shows a horizontal trend line indicating that Ca + Mg/HCO3 ratio does not change during the increase of HCO3. This indicates contribution of Ca + Mg and HCO3 are from different sources. Feldspars present in the sand and the clay minerals in the black soil may be the source for Ca and Mg. Higher HCO3 may because of the higher evaporation and the presence of Kankar. Enrichment of HCO3 and depletion of Ca + Mg may be due to cation exchange (Spears 1986). The plot of Ca + Mg versus total cations shows that the data falls on the 1:1 trend line, reflecting Na and K as the major contributor for the increase of TDS (Fig. 7d). From Fig. 7e, increase in Alkalinity corresponds to the simultaneous increase of Cl + SO4 suggesting a common source for these ions. The increased Na content which is an index of weathering suggests that ions result from silicate weathering or dissolution of soil salts, the excess of Na + K over Cl suggests silicate weathering and also suggests the higher concentration of Alkalis is from sources and precipitation (Singh et al. 2011). The Na + K versus Total cations scatter diagram (Fig. 7f) of the study area shows most of the sample points falling below the trend line. This suggests that the cations in the groundwater might have been derived from silicate weathering.
Conclusion
In this study, an attempt has been made to evaluate the geochemical processes regulating groundwater quality in the coastal region of Kadaladi block, Ramanathapuram district, Tamilnadu, India. For this study, 36 groundwater samples were collected and analyzed for pH, EC, TDS and major ions. The order of abundance of ions are Na > Ca > Mg > K and Cl > HCO3 > SO4 > NO3 > PO4. Geochemical facies of classification of groundwater shows most of the samples are Na–Cl type and the remaining samples are mixed Ca–Mg–Cl type. This suggests mixing of high saline water which generally indicates a strong sea water influence. The scatter plot Na/Cl shows good correlation indicating the groundwater chemistry is probably controlled by rock–water interaction. The plot of Ca + Mg/HCO3 shows enrichment of HCO3 than Ca + Mg suggesting both are from different sources. In addition with Piper diagram and Scatter plots, Gibbs plots were also used to understand the hydrochemical processes. In the Gibbs plots, most of the samples fall in evaporation dominance zone. Present study describes a wide range of salinity values which are noticed in the study area. This is due to the wide range of sediment permeability that occurs in the coastal plain may result in the incomplete flushing of the relict salinity. Higher salinity of groundwater distributed within the study area may be due to mixing of fresh water with relict saline water due to a higher stand of sea level in past geological time. In addition to saline incursion, hydrochemistry is also controlled by rock–water interaction followed by the concentration of these ions by evaporation. The coastal sand dunes running parallel to coast for roughly 30 km are recharged with recent meteoric water. Hence, good quality of groundwater is restricted only to coastal dune within the study area and it is important to conserve these dunes. This is well reflected in the spatial distribution of hydrochemical facies map (Fig. 5). To further pin point the origin of the salinity, trace element and isotope studies are warranted.
References
APHA (1995) Standard methods for the examinations of water and wastewater, 19th edn. American Public Association, Washington
Barret B, Heinson G, Hatch M, Telfer A (2002) Geophysical methods in saline groundwater studies: locating perched water tables and fresh-water lenses. Explor Geophys 33:115–121
Brow E, Skougstad MW, Fishmen MJ (1983) Method for collection and analyzing of water samples for dissolved minerals and gases. U.S. Govt, Washington, p 75
Cardona A, Carrillo-Rivera JJ, Huizar A’ lvarez R, Graniel-Castro E (2004) Salinization in coastal aquifers of arid zones: an example from Santo Domingo, Baja California Sur, Mexico. Environ Geol 45:350–366
Chidambaram S, Senthil Kumar G, Prasanna MV, John Peter A, Ramanthan AL, Srivasamoorthy K (2009) A study on the hydrogeology and hydrogeochemistry of groundwater from different depths in a coastal aquifer: Annamalai Nagar, Tamilnadu, India. Environ Geol 57:59–73
Fisher RS, Mullican FW (1997) Hydrochemical evolution of sodium-sulfate and sodium-chloride groundwater beneath the Northern Chihuahuan Desert, Trans- Pecos, Texas, USA. Hydrogeol J 10(4):455–474
Gibbs RJ (1970) Mechanisms controlling world water chemistry. Science 17:1088–1090
Kacimov AR, Sherif MM, Perret JS, Al-Mushikhi (2009) Control of sea-water intrusion by salt-water pumping: Coast of Oman. Hydrogeol J 17(3):541–558. doi:10.1007/s10040-008-0425-8
Kim KL, Park YS, Kim GP, Park KH (2009) Dynamic freshwater–saline water interaction in the coastal zone of Jeju Island, South Korea. Hydrogeol J 17:617–629
KrishnaKumar S, Chandrasekar N, Seralathan P, Prince S, Godson MNS (2012) Hydrogeochemical study of shallow carbonate aquifers, Rameswaram Island, India. Environ Monit Assess 184(7):4127–4138
Longe EO, Malomo S, Olorunniwo MA (1987) Hydrogeology of lagos metropolis. Afr J Earth Sci Technol 6(2):163–174
Melloul AJ, Goldenberg LC (1997) Monitoring of seawater intrusion in coastal aquifers: basics and local concerns. J Environ Manage 51:73–86
Melloul LC, Goldenberg AJ (1998) Early-indicator signals of groundwater contamination: the case study of seawater encroachment. Environ Geol 33(4):279–288
Mondal NC, Singh VP (2011) Hydrochemical analysis of salinization for a tannery belt in Southern India. J Hydro 405(2–3):235–247
Mondal NC, Saxena VK, Singh VS (2008) Occurrence of elevated nitrate in groundwaters of Krishna delta, India. African J Environ Sci Tech 2(9):265–271
Mondal NC, Singh VP, Singh VS, Saxena VK (2010) Determining the interaction between groundwater and saline water through groundwater major ions chemistry. J Hydro 25:100–111
Mondal NC, Singh VP, Singh VS (2011) Hydrochemical characteristic of coastal aquifer from Tuticorin, Tamilnadu, India. Environ Monit Assess 175:531–550. doi:10.1007/s10661-010-1549-6
Natarajan R (1997) Seasonal and spatial variation in magnesium and chloride concentration in groundwater of Nagai Quaid-E-Milleth district, Tamil Nadu, South India. Indian J Environ Prot 17(6):448–453
Piper AM (1944) A graphic procedure in the geochemical interpretation of water analysis. Trans Am Geophys Union 25:914–923
Rajmohan N, Elango L, Ramachandran S, Natrajan M (2000) Major ion correlation in groundwater of Kancheepuram Region, South India. Indian J Environ Prot 20(3):188–193
Richter BC, Kreitler CW, Bledsoe BE (1993) Geochemical techniques for identifying sources of groundwater salinization. CRC, New York, 272p
Sanford W, Langevin C, Polemio M, Povinec P (2007) A new focus on groundwater-seawater interactions, vol 312. IAHS Publications. ISBN 978-1-901502-04-6
Sarma VVJ, Prasad NVBSS, Prasad R (1982) The geochemistry of groundwater along Visakhatnam–Bhimilipatnam coast with regard to their utility in drinking, domestic and irrigation purposes. J Explor Geophy 2(4):37–52
Saxena VK, Mondal NC, Singh VS (2004) Identification of sea-water ingress using strontium and boron in Krishna Delta, India. Curr Sci 86:586–590
Selvam S, Sivasubramanian P (2012) Groundwater potential zone identification using geoelectrical survey: a case study from Medak District, Andhra Pradesh, India. Inter J Geomat Geosci 3(1):55–62
Selvam S, Manimaran G, Sivasubramanian P (2013) Hydrochemical characteristics and GIS-based assessment of groundwater quality in the coastal aquifers of Tuticorin corporation, Tamilnadu, India. Appl Water Sci 3:145–159
Senthilkumar M, Elango L (2013) Geochemical processes controlling the groundwater quality in lower Palar river basin, Southern India. J Earth Syst Sci 122(2):419–432
Singh A, Tewary BK, Sinha A (2011) Hydrochemistry and quality assessment of groundwater in part of Noida metropolitan city, Uttarpradesh. J Geol Soc India 78:523–540
Sivakumar C, Elango L (2008) Assessment of water quality in kalpakkam region, Tamil Nadu. Nat Environ Poll Technol 7(4):687–691
Spears DA (1986) Mineralogical control of the chemical evolution of groundwater. In: Trudgill ST (ed) Solute processes. Wiley, Chichester, 512p
Srinivasamoorthy K, Vasanthavigar M, Chidambaram S, Anandhan P, Sarma VS (2011) Characterisation of groundwater chemistry in an eastern coastal area of Cuddalore district, Tamil Nadu. J Geo1 Soc India 78(6):549–558
Stuyfzand PJ (1993) Hydrochemistry and hydrology of the coastal dune area of the western Netherlands. Vrije Universiteit, Amsterdam
Todd DK (1980) Groundwater hydrology, 2nd edn. Wiley, New York
Vasanthavigar M, Srinivasamoorthy K, Vijayaragavan K, Rajiv Ganthi R, Chidambaram S, Anandhan P, Manivannan R, Vasudevan S (2010) Application of water quality index for groundwater quality assessment: Thirumanimuttar sub-basin, Tamilnadu, India. Environ Monit Assess 171:595–609. doi:10.1007/s10661-009-1302-1
Volker A (1961) Source of brackish ground water in pleistocene formations beneath the dutch polderland. Economic Geol 56:1045–1057
World Bank Report (2010) Deep wells and prudence: towards pragmatic action for addressing groundwater overexploitation in India. The World Bank, Washington Report No. 51676
Acknowledgments
The authors are thankful to the Department of Science and Technology, Ministry of Science and Technology, Government of India for providing the financial assistance to carry out the work under WAR for Water scheme. We extend our sincere thanks to anonymous reviewers for their valuable suggestions to improve the quality of the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Sivasubramanian, P., Balasubramanian, N., Soundranayagam, J.P. et al. Hydrochemical characteristics of coastal aquifers of Kadaladi, Ramanathapuram District, Tamilnadu, India. Appl Water Sci 3, 603–612 (2013). https://doi.org/10.1007/s13201-013-0108-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13201-013-0108-z