Abstract
The present study was demonstrated with simple and rapid synthesis of silver (Ag) nanoparticles using marine seaweed, Gracilaria corticata. The visibility of prominent color change at 60 °C within 20 min indicates the formation of Ag nanoparticles. The synthesized Ag nanoparticles were well characterized by UV–vis spectrum, Fourier infrared spectroscopy (FTIR), transmission electron microscopy (TEM) and dynamic light scattering measurements (DLS). Prominent FTIR peaks were obtained corresponding to phenolic compounds, amide I group and aromatic rings which involved in the stabilization of Ag nanoparticles. G. corticata resulted in spherical shaped nanospheres of 18–46 nm as revealed by TEM. The average size distributions of Ag nanoparticles were 51.82 nm and are fairly stable with a zeta potential value of −26.2 mV. The result showed that, biosynthesized Ag nanoparticles from G. corticata have an effective antifungal activity against Candida albicans and C. glabrata.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Candida spp. are ubiquitous fungi and are most arguable opportunistic pathogen of immunocompromised hosts. They produce a wide spectrum of diseases, ranging from superficial mucocutaneous disease to invasive illnesses, such as hepatosplenic candidiasis, peritonitis and systemic candidiasis (Singhi and Deep 2009). Treating infection caused by Candida spp. have become a hectic problem due to serious side effects like renal and liver dysfunction associated with the polyenes (i.e., amphotericin B, nystatin) (Hoeharner et al. 2010). In addition, a significant increase in resistance to the less toxic azole drugs (i.e., fluconoazole) has occurred (White et al. 2002). Considering all these possibilities, there remains a pressing need for progress in the development of reliable diagnostic technology with improved antifungal activity.
The metallic nanoparticles are most promising and remarkable biomedical agents. Due to their large surface volume ratio, they govern interest of researchers on microbial resistance. Among the developed nanoparticles, silver (Ag) nanoparticles are pertaining to have a wide range of application in the fields of physical, chemical and biological science. In the past decade, several kinds of the biological organisms like microbes, plants and seaweeds have been employed and well studied for the synthesis of Ag nanoparticles (Ramanathan et al. 2011; Ahmad et al. 2003; Shankar et al. 2003; Mohanpuria et al. 2008; Kumar et al. 2012a). Of them, utilization of plants for the synthesis of Ag nanoparticle is advantageous over other biological methods. The rate of biosynthesis of Ag nanoparticles from plants is cost effective and does not use toxic chemicals, temperature and high pressure (Parashar et al. 2009).
During the past three decades, seaweed research has been increased considerably for the search of new and effective medicines of natural origin. Several compounds include primary and secondary metabolites synthesized by seaweeds are promising source for both industrial and biotechnological applications (Renn 1997). Keeping the above information in mind, we report on the seaweed-mediated reduction of silver into Ag nanoparticles using Gracilaria corticata for its antifungal activity against Candida spp. G. corticata is a predominant red seaweed species found in coastal regions of Indian subcontinent, belonging to the family Gracilariacea. It possess several biomedical properties such as antibacterial, antiviral, antifungal, antiprotozoal, anti-inflammatory, anti-oxidant, cytotoxic, contraception, gastrointestinal, cardiovascular, hypoglycemia, anti-enzymes, spasmolytic and allelophatic effects (Almeida et al. 2011).
Materials and methods
Preparation of seaweed extract
Seaweed (G. corticata) was collected from mandapam, southeast coast of India and its aqueous seaweed extract was prepared by the methodology previously reported with minor modifications (Kumar et al. 2012a). In brief, G. corticata were repeatedly surface sterilized with sterile seawater followed by distilled water to remove extraneous materials and salt content. The materials were shade dried for a period of 2 weeks and powdered with mixer grinder. 0.25 % of extract was prepared using deionized water by heating at 60 °C for 20 min. The extract was filtered using Whatmann No. 1 filter paper and refrigerated at −4 °C until further analysis.
Synthesis of silver (Ag) nanoparticles
In a critical experiment, 100 μl of 1-M silver nitrate (AgNO3, Sigma Aldrich) solution was prepared and added to 99.9 ml of seaweed extract in a conical flask. The extract was gradually heated to 60 °C for 20 min in a heating mantle for the reduction of metal ions to occur. Then, mixture was cooled to room temperature, centrifuged at 8,000 rpm and then redispersed with same aliquot of deionised water.
Characterization of Ag nanoparticles
The bioreduction of Ag nanoparticles were monitored by an UV–visible spectrophotometer (Shimadzu, 2450). The presence of bioactive functional groups responsible for the reduction of silver ions were recorded by Perkin-Elmer FTIR spectrum—RX-1 spectrophotometer at a resolution of 2 cm−1 ranging from 4,000 to 400 cm−1 in KBr pellet using diffuse reflectance mode. Dynamic light scattering (DLS) studies of Ag nanoparticles on dilution with deionized water were measured with Malvern—Zetasizer (Nano-Z590) instrument. Transmission electron microscopic (TEM) images were acquired on Philips Tecnai 10 instrument operated at an accelerating voltage of 80 kV.
Antifungal activity
Pure culture of Candida albicans (NCIM 3074) and C. glabrata (NCIM 3226) were procured from National Collection of Industrial Microorganisms (NCIM), Pune, India. Stock cultures were prepared and maintained in Sabouraud Dextrose Agar (SDA) slants at 4 °C. Prior to experiment, SDA broth was prepared, inoculated and incubated with pure culture at 25 °C for 3 days. A disc diffusion method was adopted to evaluate the antifungal activity of Ag nanoparticles (Perez et al. 1990). About 150 CFU/ml of innoculum was swabbed onto SDA plates uniformly and allowed it to dry in a sterile environment. Sterile disc of 6 mm (HIMEDIA) were loaded with 30 μl of test solution (Seaweed extract, 1 mM AgNO3 and Ag nanoparticles). Nystatin (HIMEDIA—100 units) was used as positive control. The plates were incubated at 25 °C for 3 days to measure Zone of Inhibition (ZoI). Mean was calculated by performing the experiments in triplicates.
Results and discussion
Phyco-synthesis of Ag nanoparticles
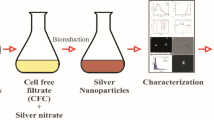
The reduction of AgNO3 in aqueous seaweed extract by heating at 60 °C showed visible color change whereas no color change was observed in Ag+ solution as shown in Fig. 1. The color of solution gradually intensified on heating, which indicates the synthesis of Ag nanoparticles. The change in color is an attribute to excitation of surface plasmon vibrations of Ag nanoparticles (Mulvaney 1996). Notably, even after 24 h there is no remarkable deepening of color indicating the saturation of reaction (data not shown). This reflects that the particles may be well dispersed in the solution with mild agglomeration (Nabikhan et al. 2010).
UV–visible spectroscopy
UV–vis spectroscopy is a convenient, preliminary and indirect method for characterization of Ag nanoparticles based on optical properties called surface plasmon resonance (SPR) (Kuber and D’Souza 2006). The color of the seaweed extract becomes turbid after the addition of aqueous AgNO3 solution signifying the initiation of reaction. The silver SPR band occurs at 420 nm with progressive increase in absorbance upon increasing time until 20 min as shown in Fig. 2. The observed band in this range has been associated with Ag nanoparticles confirming the synthesis of spherical Ag nanoparticles with narrow size distribution (Henglein 1993; Kumar et al. 2012b). Elevation in temperature results in formation of spherical- and octahedral-shaped nanoparticles of size 5–200 nm (Lengke et al. 2007). Similarly, shape-controlled Ag nanoparticles can be also synthesized in biological route by varying temperature (Bansal et al. 2012). Hence, a possible mechanism to control particle size and shape with reference to SPR peak has been addressed.
FTIR spectrum
Major FTIR peaks were observed at 3,430, 1,637 and 685 cm−1 indicating the possible interaction of biomolecules for capping, and stabilization of Ag nanoparticles is presented in Fig. 3. We also observed peaks in control solutions such as 1-mM AgNO3 solution (3,431, 1,634 and 694 cm−1) and seaweed extract (3,428, 1,637 and 668 cm−1), respectively. The shift in peak clearly attributes the reduction of silver ions into their respective nanoparticles as shown in Fig. 3. A strong absorption peak at 3,430 cm−1 indicates the presence of phenols and alcohols with free O–H group. The peak at 1,637 cm−1 represents the presence of amide I group and may well arise due to carbonyl stretch in proteins (Kumar et al. 2012b). The peak at 685 cm−1 may be assigned due to the stretching vibration of aromatic ring that might be attached to the free O–H group. Several species of Gracilaria have been reported to contain abundant of amino acids, fatty acids, vitamins, minerals, phenolic compounds and carbohydrates (Yang et al. 2012). Of which, phenolic compounds especially polyphenols and tannin have reported to have antimicrobial, anti-carcinogenic and anti-oxidant properties (Arunkumar et al. 2010). Satyavani et al. 2011 have reported that silver ions may possibly bind to phenolic compounds with one or more aromatic ring resulting in the formation of Ag nanoparticles.
Electron microscopy studies
Morphology of Ag nanoparticles observed using TEM indicates nanopheres ranging from 18 to 46 nm as given in Fig. 4. This may be due to weak SPR vibrations produced by Ag nanoparticles at 420 nm (Sathishkumar et al. 2009). Similarly, Ag nanoparticles synthesized from Acanthophora spicifera were analogous in structure and this may perhaps be due to the presence of similar reducing agents like phenolic compounds with free O–H group (Kumar et al. 2012a).
Dynamic light scattering studies
The average size distributions of Ag nanoparticle were found to be 51.82 nm and well dispersed in the colloidal solution (Fig. 5a). Similar observations have been studied in seaweed, Sargassum tenerrimum with a maximum peak intensity of 48 nm (Kumar et al. 2012b). The zeta potential value of Ag nanoparticle, −26.2 mV, indicates that the particles are highly stable as represented in Fig. 5b. This may be achieved by means of high repulsive and attractive forces exist between each nanoparticle (Lee et al. 2011). The observed result is well correlated with the TEM studies (Fig. 4).
Antifungal activity
The antifungal activity of Ag nanoparticles was tested against Candida spp. as shown in Fig. 6. 30 μl of the nanoparticle solution is well enough to impede Candida albicans (12 mm) and C. glabrata (11 mm) (Table 1). Moreover, Ag nanoparticles showed highest antifungal efficacy than other type of test solution (seaweed extract and 1-mM AgNO3 solution). But, the exact possible inhibitory mechanism displayed by Ag nanoparticles is unclear and requires further investigation. It is a well-known fact that antimicrobial activity of Ag nanoparticles is likely to be well correlated with its decreased size and shape owing to increased surface area with enhanced antimicrobial effect (Baker et al. 2005; Phaechamud et al. 2012). Presumably, we observed good antifungal activity as compared with standard reference antifungal drug (Nystatin). Although studies discloses, antibacterial activity of Ag nanoparticles was more distinct than antifungal activity (Nabikhan et al. 2010). This could possibly be due to the complexity of eukaryotic yeast cell in which it requires higher concentration of ionic silver (Panacek et al. 2009). Recently, Hwang et al. (2012) have reported that the increase in hydroxyl radicals by Ag nanoparticles causes apoptotic cell death in Candida albicans. Altogether, fungal cells when exposed to Ag nanoparticle solution results in formation of ‘pits’ on the surface of membrane resulting in destruction of its cell integrity (Nasrollahi et al. 2011).
Conclusions
To conclude, we have demonstrated the rapid synthesis of Ag nanoparticles using seaweed, G. corticata for its antifungal activity. We achieved biocompatible, stable Ag nanoparticles at a temperature of 60 °C within 20 min of reaction time without the use of toxic chemicals. The presence of functional bioactive compounds in seaweed extract is responsible for the formation of Ag nanoparticles as revealed by FTIR. The average size of spherical-shaped Ag nanoparticles ranges between 18 and 46 nm. This work also demonstrates the use of Ag nanoparticles as a potential antifungal agent against Candida spp.
References
Ahmad A, Mukherjee P, Senapati S, Mandal D, Khan MI, Kumar R, Sastry M (2003) Extracellular biosynthesis of silver nanoparticles using the fungus Fusarium oxysporium. Colloids Surf B Interface 28:313–318
Almeida CLF, De S, Falcao H, De M, Lima GR, De A, Montenegro C, Lira NS, De Athayde-Filho PF, Rodrigues LC, De Souza MFV, Barbosa Filho JM, Batista LM (2011) Bioactivities from marine algae of the Genus Gracilaria. Int J Mol Sci 12:4550–4573
Arunkumar K, Sivakumar SR, Rengasamy R (2010) Review on Bioactive potential in seaweeds (Marine Macroalgae): a special emphasis on biodiversity of seaweeds against plant pathogens. Asian J Plant Sci 9:227–240
Baker C, Pradhan A, Pakstis L, Pochan DJ, Shah SI (2005) Synthesis of antibacterial properties of silver nanoparticles. J Nanosci Nanotechnol 5:244–249
Bansal V, Bharde A, Ramanathan R, Bhargava SK (2012) Inorganic materials using ‘unusual’ microorganisms. Adv Colloid Interface Sci. doi:10.1016/j.cis.2012.06.013
Henglein A (1993) Physicochemical properties of small metal particles in solution: “microelectrode” reactions, chemisorptions, composite metal particles, and the atom-to-metal transition. J Phys Chem 97:5457–5471
Hoeharner CF, Cummings ED, Hilliard GM, Rogers PD (2010) Changes in the proteome of Candida albicans in response to azole, polyene and echinocandin. Antimicrob Agents Chemother 54:1655–1664
Hwang IS, Lee J, Hwang JH, Kim KJ, Lee DJ (2012) Silver nanoparticles induce apotoptic cell death in Candida albicans through the increase of hydroxyl radicals. FEBS J 279:1327–1328
Kuber CB, D’Souza SF (2006) Extracellular biosynthesis of silver nanoparticles using the fungus Aspergillus fumigates. Colloids Surf B Interface 47:160–164
Kumar P, Senthamilselvi S, Lakshmipraba A, Premkumar K, Muthukumaran R, Visvanathan P, Ganeshkumar RS, Govindaraju M (2012a) Efficacy of biosynthesized silver nanoparticles using Acanthophora spicifera to encumber biofilm formation. Dig J Nanomater Bios 7:511–522
Kumar P, Senthamil Selvi S, Lakshmi Praba A, Prem Kumar K, Ganeshkumar RS, Govindaraju M (2012b) Synthesis of silver nanoparticles from Sargassum tenerrimum and screening phytochemcials for its anti-bacterial activity. Nano Biomed Eng 4:12–16
Lee J, Kim HY, Zhou H, Hwang S, Koh K, Han DW, Lee J (2011) Green synthesis of phytochemcial stabilized Au nanoparticles under ambient conditions and their biocompatibility and antioxidant activity. J Mater Chem 21:13316–13326
Lengke MF, Fleet ME, Southam G (2007) Biosynthesis of silver nanoparticles by filamentous cynaobacteria from a silver (I) nitrate complex. Langmuir 23:2694–2699
Mohanpuria P, Rana NK, Yadav SK (2008) Biosynthesis of nanoparticles: technological concepts and future applications. J Nanopart Res 10:507–517
Mulvaney P (1996) Surfaces plasmon spectroscopy of nanosized metal particles. Langmuir 12:788–800
Nabikhan A, Kandasamy K, Raj A, Alikunhi NM (2010) Synthesis of antimicrobial silver nanoparticles by callus and leaf extracts from saltmarsh plants, Sesuvium portulacastrum L. Colloids Surf B Interface 79:488–493
Nasrollahi A, Pourshamsian KH, Mansourkiaee P (2011) Antifungal activity of silver nanoparticles on some of fungi. Int J Nano Dim 1:233–239
Panacek A, Kolar M, Vecerova R, Prucek R, Soukapova J, Krystof V, Hamal P, Zboril R, Kvitek L (2009) Antifungal activity of silver nanoparticles against Candida spp. Biomaterials 30:6333–6340
Parashar UK, Saxena PS, Srivastava A (2009) Bioinspired synthesis of silver nanoparticles. Dig J Nanomater Bios 4:159–166
Perez C, Paul M, Bazerque P (1990) An antibiotic assay by the agar well diffusion method. Acta Biol Med Exp 15:113–115
Phaechamud T, Mahadlek J, Aroonrerk N, Choopun C, Charoenteeraboon J (2012) Antimicrobial activity of ZnO-doxycycline hyclate thermosensitive gel. Science Asia 38:64–74
Ramanathan R, O’Mullane AP, Parikh RY, Smooker PM, Bhargava SK, Bansal V (2011) Bacterial kinetics-controlled shape-directed biosynthesis of silver nanoplates using Morganella psychrotolerans. Langmuir 27(2):714–719
Renn D (1997) Biotechnology and the red seaweed polysaccharide industry: status, needs and prospects. Trends in Biotechnol 15:9–14
Sathishkumar M, Sneha K, Won SW, Cho CW, Kim S, Yun YS (2009) Cinnamon zeylanicum bark extract and powder mediated green synthesis of nano-crystalline silver particles and its bacterial activity. Colloids Surf B Interface 73:332–338
Satyavani K, Gurudeeban S, Ramanathan T, Balasubramanian T (2011) Biomedical potential of silver nanoparticles synthesized from calli cells of Citrullus colocynthis (L.). J Nanobiotechnology 9:43–50
Shankar SS, Ahmad A, Sastry M (2003) Geranium leaf assisted biosynthesis of silver nanoparticles. Biotechnol Prog 19:1627–1631
Singhi S, Deep A (2009) Invasive candidiasis in pediatric intensive care units. Indian J Pediatr 76:1033–1044
White TC, Holleman S, Dy F, Mirels LF, Stevens DA (2002) Resistance mechanism in clinical isolates of Candida albicans. Antimicrob Agents Chemother 46:1704–1713
Yang JI, Yeh CC, Lee JC, Yi SC, Huang HW, Tseng CN, Chang HW (2012) Aqueous extracts of the edible Gracilaria tenuistipitata are protective against H2O2-induced DNA damage, growth inhibition, and cell cycle arrest. Molecules 17:7241–7254
Acknowledgments
The authors are grateful to DST-NRDMS, Government of India, New Delhi for their financial assistance through major research project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Kumar, P., Senthamil Selvi, S. & Govindaraju, M. Seaweed-mediated biosynthesis of silver nanoparticles using Gracilaria corticata for its antifungal activity against Candida spp.. Appl Nanosci 3, 495–500 (2013). https://doi.org/10.1007/s13204-012-0151-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-012-0151-3