ABSTRACT
Various liver diseases result in terminal hepatic failure, and liver transplantation, cell transplantation and artificial liver support systems are emerging as effective therapies for severe hepatic disease. However, all of these treatments are limited by organ or cell resources, so developing a sufficient number of functional hepatocytes for liver regeneration is a priority. Liver regeneration is a complex process regulated by growth factors (GFs), cytokines, transcription factors (TFs), hormones, oxidative stress products, metabolic networks, and microRNA. It is well-known that the function of isolated primary hepatocytes is hard to maintain; when cultured in vitro, these cells readily undergo dedifferentiation, causing them to lose hepatocyte function. For this reason, most studies focus on inducing stem cells, such as embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), hepatic progenitor cells (HPCs), and mesenchymal stem cells (MSCs), to differentiate into hepatocyte-like cells (HLCs) in vitro. In this review, we mainly focus on the nature of the liver regeneration process and discuss how to maintain and enhance in vitro hepatic function of isolated primary hepatocytes or stem cell-derived HLCs for liver regeneration. In this way, hepatocytes or HLCs may be applied for clinical use for the treatment of terminal liver diseases and may prolong the survival time of patients in the near future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Viral hepatitis, fatty liver disease, drug-induced liver injury, liver cirrhosis, hepatic carcinoma, and other liver diseases can cause acute or chronic liver failure. Approximately 10% of patients with liver disease succumb to their condition while waiting for liver sources each year (Kim et al., 2006).
Liver transplantation was once the only therapeutic option for patients with end stage liver diseases, and its clinical use was limited due to limited donor availability, surgical injuries, a high incidence of surgical complications and the high cost of the treatment (Duan et al., 2013). Later, cell transplantation and artificial liver support emerged as effective methods for compensation of lost liver function and increased the survival rate of patients; however, these two methods are also limited by the availability of effective cell sources and equipment. The inability of hepatocytes to proliferate in vitro and the severely inadequate supply of hepatocytes due to donor shortage are still the main problems for primary human hepatocyte-based treatments. Stem cells have been proposed as an ideal cell source because they have potent self-renewal, low immunogenicity, and the capacity to differentiate into various cell types. Furthermore, they can generate unlimited hepatocytes with incomplete function (Sancho-Bru et al., 2009) that are generally defined as hepatocyte-like cells (HLCs). HLCs can be derived from multiple stem cell types, such as embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), hepatic progenitor cells (HPCs), and mesenchymal stem cells (MSCs). Therefore, it is crucial to develop robust methods for differentiating stem cells into mature hepatocytes in vitro for clinical use.
Here, we present an overview of isolated primary hepatocytes and stem cell-derived HLCs used for liver regeneration and describe how the in vitro environment in which they are cultured is continuously being optimized to mimic in vivo conditions and maintain hepatic function. The main disadvantages, histologic origin, 3D, and co-culture environment for in vitro culture of isolated hepatocytes or stem cell-derived hepatocytes were demonstrated in Table 1. Optimization of in vitro culturing of functional hepatocytes will solve the issues of limited cell numbers and limited function, and sufficient numbers of functional hepatocytes will be used to promote liver regeneration directly or indirectly.
NATURE OF LIVER REGENERATION
The liver serves as a major storage site of glycogen and vitamin A and is one of only a few organs in adults that are capable of regeneration. Normal mature hepatocytes and cholangiocytes stay in the G0 phase of the cell cycle, exhibit a quiescent phenotype and show minimal turnover, but in response to partial hepatectomy (PH), they can undergo cell proliferation to compensate for the lost cells, a process called liver regeneration. However, severe damage caused by liver diseases can significantly diminish the proliferative ability of these cells and, thus, their liver regeneration ability. When that is the case, liver tissue transplantation may be required (Samuel et al., 2011).
Spontaneous liver tissue regeneration (Fujiyoshi and Ozaki, 2011) is achieved by a complex interactive network consisting of liver cells (hepatocytes, kupffer cells, sinusoidal endothelial cells, hepatic stellate cells, and stem cells) and extrahepatic organs (the thyroid gland, adrenal glands, pancreas, duodenum, and autonomic nervous system). Growth factors (GFs), transcription factors (TFs), cytokines, hormones, oxidative stress products, metabolic networks, and microRNA are essential for liver regeneration to proceed in an optimal manner to gain adequate hepatic mass (Mao et al., 2014). Mitogenic GFs override the G1 restriction point and promote hepatocytes to transit into S phase. The restoration of liver volume depends on hepatocyte proliferation, which includes initiation, proliferation, and termination phases.
After PH, more than 100 immediate early genes are activated by TFs that are latent in the quiescent liver. Interleukin (IL)-6 (Li et al., 2001), lipopolysaccharide (Cornell et al., 1990), C3a, and C5a (Strey et al., 2003) can initiate the cytokine cascade and trigger liver regeneration. Nuclear factor-kB (Deng et al., 2009), calcitonin gene-related peptide (Mizutani et al., 2013), caspase recruitment domain-containing protein 11, zinc finger protein 490 (Nygard et al., 2012), and heat shock protein 70 (Wolf et al., 2014) contribute to the early phase of successful liver regeneration. Pituitary hormone prolactin administration directly or indirectly increases the number of proliferating cells during the priming stage of hepatectomy, which causes an increase in the binding activity of several TFs involved in cell proliferation, liver-specific differentiation, and the maintenance of energetic metabolism (Olazabal et al., 2009). Furthermore, the process of liver regeneration involves a general reduction of the levels of many coagulation cascade proteins (Tatsumi et al., 2011). Auxiliary mitogens include norepinephrine, vascular endothelial growth factor, insulin, bile acids, serotonin, complement, leptin, estrogens, fibroblast growth factor (FGF)-1, FGF-2, and IL-4 are also indispensable for the hepatocyte cell cycle (DeAngelis et al., 2012; Michalopoulos, 2010). Vitamin D3 upregulated protein-1 regulates proliferative signaling during liver regeneration by altering the activation of genes involved in the extracellular signal-regulated kinase 1/2 and AKT signaling pathways (Kwon et al., 2011). Growth hormone signaling molecules (Zerrad-Saadi et al., 2011) and the transcription factor E2F2 (Delgado et al., 2011) are also key regulators of the cell cycle. Activation of Notch, a signaling pathway that mediates lineage segregation during liver development, is sufficient to reprogram hepatocytes into biliary epithelial cells (Yanger et al., 2013). FGF15 is an essential mediator of the liver growth promoting effects of bile acids and is necessary to maintain liver growth homeostasis (Uriarte et al., 2013). P-element-induced wimpy testis interacting RNAs exert regulatory functions on the cell genome and transcriptome (Rizzo et al., 2014). Hepatic non-parenchymal cells play a time-dependent regulatory role (Nejak-Bowen et al., 2013). Nogo-B, also known as reticulon 4B, promotes liver fibrosis and cirrhosis by facilitating the TGF-b signaling pathway in activated hepatic stellate cells (HSCs) and facilitates hepatocyte proliferation and liver regeneration (Gao et al., 2013). The termination of liver regeneration is a complex process that is affected by integrin-mediated signaling. The return of HGF and TGF-b to their baseline levels (Michalopoulos, 2010) and activation of mitogen-activated protein kinase kinase-4 (Wuestefeld et al., 2013) can cause complete termination of liver regeneration.
IN VITRO CELLS WITH HEPATIC FUNCTION
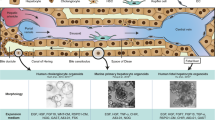
Liver regeneration can proceed through two different mechanisms: replacement of lost tissue with cell types of phenotypic fidelity; and replacement of tissue by activation of transdifferentiation pathways originating from facultative stem cells. Liver regeneration is a rapid and well-coordinated process that requires contributions from multiple cell populations (Fig. 1).
Primary hepatocytes
The liver is primarily composed of two epithelial cell lineages, namely hepatocytes and cholangiocytes, which originate from hepatoblasts during fetal development. Hepatocytes are the predominant cell type in the liver under nonpathological conditions. Isolated primary human hepatocytes are currently the gold standard for in vitro drug screening because they express the entire complement of hepatic drug metabolizing enzymes and transporters. In spite of their prolific growth ability in vivo, attempts to proliferate adult hepatocytes in vitro have been less successful. It has taken a long time to optimize the hepatocyte culture conditions to allow them to grow steadily in vitro.
Once plated in a monolayer, primary hepatocytes typically undergo progressive dedifferentiation, which is reflected at the level of the drug transporters and the dramatic loss in the phenotypic characteristics of the cells. Specifically, hepatocytes and liver sinusoidal endothelial cells dedifferentiate within 72 h when cultured as monolayers in vitro. Conventional approaches to counteract this dedifferentiation aim at reestablishing the natural hepatocyte microenvironment in vitro and include reintroduction of an extracellular matrix (ECM) backbone (Skardal et al., 2012), addition of differentiation promoting soluble compounds to the culture medium, and boosting of homotypic hepatocyte interactions or cocultivation of hepatocytes with other cell types. However, the use of this approach is limited by the availability of reproducible sources of hepatocytes.
The ability of EGF to induce DNA synthesis in primary hepatocytes was first demonstrated in 1976. Thereafter, many researchers have tried to determine the essential factors for triggering hepatic regeneration. E-cadherin is required for hepatocyte spheroid formation and may be responsible for protecting hepatocytes from a novel form of caspase-independent cell death (Luebke-Wheeler et al., 2009). Culturing of rat liver sinusoidal endothelial cells in a layered three-dimensional configuration, with the layers separated by a chitosan-hyaluronic acid polyelectrolyte multilayer, resulted in enhanced heterotypic cell-cell interactions, which led to improvements in hepatocyte function (Kim and Rajagopalan, 2010). PuraMatrix, a well-defined synthetic peptide that can self-assemble into an interweaving nanofiber scaffold to form a hydrogel, is an attractive system for generating hepatocyte spheroids that quickly restore liver function after seeding (Wang et al., 2008). More recently, hepatocytes sandwiched between matrigel layers were reported to have stable function. Despite their advantages, collagen and matrigel sandwich cultures do not provide the complex multi-cellular environment found in vivo, and co-cultures do not mimic the layered liver architecture (Sellaro et al., 2010). Current techniques that maintain hepatocellular function in vitro with different biomaterials and geometries exhibit a low cell density and functional capacity per unit volume.
Stem cell-derived hepatocytes
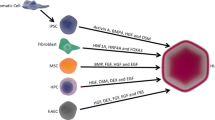
Stem cells have the potential for numerous biomedical applications, including therapeutic cell replacement to repair damaged body organs, as tools for studying genetic defects and testing drugs, and as models for studying cell differentiation and early development. HLCs can be derived from ESCs (Subramanian et al., 2014), iPSCs (Amimoto et al., 2011), HPCs (Wang et al., 2011), and MSCs (Piryaei et al., 2011), which have the capacity for unlimited proliferation and multilineage differentiation. However, there are certain requirements for the generation of HLCs: (a) an unlimited source of initial cell material is needed to ensure routine large-scale generation of the required cells; (b) the generated cultures should be reproducible in terms of their hepatic-like functionality; and (c) the established system should allow for highly efficient selection of hepatocytes.
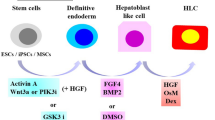
Embryonic stem cell-derived hepatocytes (ESC-Heps)
ESCs are derived from the inner cell mass of the fertilized egg, which is pluripotent, and can be cultured indefinitely in an undifferentiated state and have the potential to differentiate into three germ layer cell types. They have been shown to give rise to functional hepatocytes that effectively integrate into and replace injured parenchyma in many devastating liver diseases. Definitive endoderm (DE) cells are the precursors of both the liver and pancreas, and they have to be induced to undergo hepatic and pancreatic differentiation (Murry and Keller, 2008). Moreover, the expression of alpha fetal protein (AFP), albumin (ALB), and a biliary molecular marker appear sequentially, suggesting the differentiation of ESCs recapitulates the normal developmental processes of the liver. There are some issues with these the current differentiation protocols, including spontaneous differentiation, low yield, the presence of undefined and xenogenetic compounds, necessity of cell sorting for specific cell lineages, considerable enzymatic stress during repeated culture (Haque et al., 2011) and cellular heterogeneity in the culture (Nagaoka et al., 2008). Terminally specified cells of a certain lineage represent only a minor cell fraction of the differentiating ESCs in culture. Therefore, large-scale production of highly purified cell lineages of interest is the principle task of the ESC-based approach to regenerative medicine. Potential therapeutic applications of ESC-Heps are limited by their relatively low output in differentiating ESC cultures and by the danger of contamination with tumorigenic, undifferentiated ESCs. Furthermore, the use of ESCs typically causes ethical and immunorejection issues, and the elimination of these issues is critical for stem cell transplantation therapies to be effective.
Many studies have made substantial contributions to the differentiation of ESCs into hepatocytes by continuously improving inducers of differentiation and optimizing their combinations and sequences (Li et al., 2010b; Liu et al., 2010b; Wang et al., 2012; Zhang et al., 2013b). Most induction methods try to imitate the routine embryonic developmental process of the liver within a few days of in vitro culture. Most of the induction processes are ineffective, complicated, time-consuming, expensive, and limited by the difficulties involved in scaling up the procedures. Cytochrome P (CYP) activity in human ESC-Heps was much lower than in human primary hepatocytes cultured for 4 h and was stable or increased for at least one week in culture, which contrasts the observation of rapid loss of CYP activity in cultured human primary hepatocytes (Ulvestad et al., 2013). However, ESC-Heps are unable to function as efficiently as hepatoblasts or primary hepatocytes upon transplantation in liver repopulation models. Clearly, improvement of in vitro hepatic differentiation protocols and a better understanding of the molecular mechanisms underlying liver development are needed. Therefore, it can be speculated that there are certain specific TFs, or a combination of TFs, that can facilitate the differentiation of ESCs. Exposure of adherent human ESCs in culture to activin A treatment followed by various GFs, including dexamethasone, oncostatin M, and HGF, results in the production of ESC-Heps that possess hepatocyte-specific functions (Moore and Moghe, 2009). The signaling molecules bone morphogenic protein (BMP) and FGF that have been implicated in hepatic differentiation during normal embryonic development and have been shown to play pivotal roles in generating hepatic cells from DE cells derived from ESCs. Using a set of human adult markers, including CAAT/enhancer binding protein (C/EBPalpha), hepatocyte nuclear factor 4/7 ratio (HNF4alpha1/HNF4alpha7), CYP7A1, CYP3A4 and constitutive androstane receptor, and fetal markers, including AFP, CYP3A7, and glutathione S-transferase P1, by 21 days of differentiation, ESC-Heps have the characteristics of fetal hepatocytes at less than 20 weeks of gestation, but extending the differentiation to 4 weeks does not improve cell maturation (Funakoshi et al., 2011). Li et al. (Li et al., 2011a) established an efficient method for the induction of mouse ESC-derived DE cells in suspension embryonic body culture. The chemical activation of the canonical Wnt signaling pathway synergized with the activin A-mediated nodal signaling pathway to promote the induction of DE cells, and inhibition of BMP4 signaling by Noggin and activin A further improved the efficiency of DE cell differentiation. A combined treatment with Wnt3a and BMP4 efficiently differentiated human ESCs (Kim et al., 2013); after co-culture with STO feeder cells, human ESCs were able to differentiate into HLCs and cholangiocyte-like cells (Zhao et al., 2009). Forkhead box A2 (Liu et al., 2013) and synthesized basement membrane components (Shiraki et al., 2011) significantly increased the hepatic differentiation of ESCs.
Most studies demonstrating hepatic differentiation from ESCs have been based on embryoid body (EB) formation, aggregated colony formation in static culture. It has been shown that dynamic three-dimensional perfusion culture is superior to other culture systems for inducing maturation of ESCs into fetal hepatocytes and prolonging the maintenance of the hepatic functions of those cells. Ten pathways that were significantly upregulated in cells differentiated in a bioreactor compared to cells grown in static culture were shown to be highly related to liver functions (Sivertsson et al., 2013). The differentiated phenotype was sustained for more than 2 weeks in the three-dimensional spheroid culture system, which is significantly longer than in monolayer culture (Subramanian et al., 2014). EB-derived cells grown in a rotating bioreactor exhibited higher levels of liver-specific functions than those in static culture (Zhang et al., 2013a). The hollow fiber/organoid culture method allows for cultured ESCs to form an organoid, and the differentiating ESCs reach a level of functionality comparable to or better than that of primary mouse hepatocytes (Amimoto et al., 2011). Differentiated cells grown on a biodegradable polymer scaffold and a rotating bioreactor also exhibit morphologic traits and biomarkers characteristic of liver cells (Wang et al., 2012). Significantly upregulated hepatic gene expression was observed in the hepatic differentiation hollow fiber-based three-dimensional perfusion bioreactors with integral oxygenation culture group (Miki et al., 2011). However, most of the current three-dimensional differentiation configurations involve interruptive operations during the multistaged differentiation process, which might impose unwanted influence on cellular differentiation. Off-the-shelf micro-stencil arrays were developed to generate adherent multilayered colonies composed of human ESC-derived cells; the microscaled multilayered colonies with uniform and defined sizes constrained within the microwells are composed of more homogenous and mature HLCs with significantly lowered AFP expression and elevated hepatic functions (Yao et al., 2014). A combination of co-culture with non-parenchymal liver cells and optimal GF stimulation was found to induce endoderm and hepatic phenotypes earlier and to a much greater extent than GF arrays or micropatterned co-cultures used individually (Tuleuova et al., 2010). When coculturing ESCs with MLSgt20 cells, which are derived from mesenchymal cells residing in murine fetal livers, human ESC-derived AFP-producing cells displayed higher hepatocyte functions (Ishii et al., 2010). A similar effect was observed in mouse ESCs co-cultured with mouse HSCs as feeder cells in basal medium without additional hepatocyte growth factors (Chan et al., 2013).
Induced pluripotent stem cell-derived hepatocytes (iPSC-Heps)
iPSCs were initially generated from terminally differentiated adult cells using viral vectors for specific TFs, and later using the non-integrating methods of treating cells with small molecules that affect methylation or acetylation, mimic the Wnt-signaling pathway, or modulate the TGF-b pathway (Li et al., 2009). These methods raised few ethical concerns because of their derivation from somatic cells and, thus, are powerful tools for studying basic developmental biology. iPSCs are now considered to have the same level of pluripotency as ESCs (Jia et al., 2010), and the mitochondria of iPSCs were shown to act like those of ESCs by measuring oxygen concentration and pH in hepatic induction medium, which indicate the oxygen consumption rate and extracellular acidification rate, respectively (Tamai et al., 2011). iPSCs can be differentiated into neural, osteogenic, cardiac, adipogenic, pancreatic, vascular, hematopoietic, and endothelial cells. In addition, they are potential sources of hepatocytes for applications in regenerative medicine and drug development (Noto et al., 2014). iPSCs from mouse embryonic fibroblasts and human fibroblasts can be differentiated into hepatic lineages with four reprogramming factors (Oct-4/Sox2/Klf-4/c-Myc) (Yamanaka and Blau, 2010). Li et al. (Li et al., 2011b) demonstrated that iPSCs could be differentiated into iPSC-Heps with biological functions without being treated with c-Myc, and those iPSC-Heps reduced the hepatic necrotic area and improved liver function. However, iPSCs in vivo are able to create chimeras by germ line transmission and tetraploid complementation and form teratomas containing various cell types from three embryonic germ layers.
Liu et al. (Liu et al., 2010a) were the first to reprogram primary hepatocytes to pluripotency, and these hepatocyte-derived iPSCs were able to directly differentiate into DE, HPCs, and mature hepatocytes. The derivation of iPSCs from somatic cells of patients with liver diseases, including tyrosinemia, glycogen storage disease, progressive familial hereditary cholestasis, and Crigler-Najjar syndrome (two siblings), through retroviral transduction of Yamanaka’s factors in serum and feeder-free culture conditions has been shown (Ghodsizadeh et al., 2010). Furthermore, those iPSCs were efficiently differentiated into functional HLCs (Ghodsizadeh et al., 2010). iPSCs are specified to primitive streak/mesendoderm/definitive endoderm by sequential stimulation with liver development-related cytokines, resulting in differentiated cells with characteristics of HLCs. CYP activities in iPSC-Heps were stable or increased for at least one week in culture, which contrasts with the rapid loss of CYP activities in cultured human primary hepatocytes between 4 h and 48 h after plating (Ulvestad et al., 2013). iPSC-Heps can be directed to differentiate into HLCs by mimicking embryonic and fetal liver development (Hannan et al., 2013; Kajiwara et al., 2012; Takebe et al., 2013), but most differentiated cells co-express AFP and ALB, suggesting incomplete cell maturation. Although progress in developing differentiation procedures has been made, it remains challenging to generate iPSC-derived mature hepatocytes. Umeda et al. (Umeda et al., 2013) performed knock-in of a monomeric Kusabira orange (mKO1) cassette into the ALB gene in iPSCs with the use of a helper-dependent adenovirus vector, and the ALB/mKo1 knock-in iPSCs are valuable resources, as they show enhanced in vitro hepatic differentiation function.
Compared with the in vivo environment of the liver, culture conditions are relatively artificial, and this is likely to impact the function of iPSC-Heps (Si-Tayeb et al., 2010). Activin has the opposing effects of promoting differentiation into endoderm and maintaining pluripotency by regulating the expression of Nanog (Shin et al., 2011). Dexamethasone and insulin-transferrin-selenium are used to maintain the in vitro functions of hepatocytes. In consideration of the tedious differentiation work and complex combination of cytokines, many investigators have focused on developing concise and rapid methods for differentiating iPSCs into HLCs. Chen et al. (Chen et al., 2012) established a rapid and efficient three-step differentiation protocol with HGF, activin A, and Wnt3a that is able to generate functional HLCs from human iPSCs. Then, Takata et al. (Takata et al., 2011) described a two-step protocol for directing human iPSCs to differentiate into hepatic cells using only two cytokines and a short incubation time; furthermore, the differentiation efficiency of the two-step protocol was comparable to that of the three-step protocol, and the induced hepatic cells were functional. In the latest study by Tomizawa et al. (Tomizawa et al., 2013), a single-step protocol for the differentiation of iPSCs into hepatocytes was designed and involved exposure to FoxA2, GATA4, HEX, and C/EBPα and culturing with OERDITS supplementation. This protocol has the potential to induce the differentiation of iPSCs into HLCs within 8 days. Kondo et al. (Kondo et al., 2014) developed a simple method of differentiation of human iPSCs into functional HLCs with small-molecule compounds, which are convenient and inexpensive to obtain for large scale production and do not have the potential to be contaminated with exogenous viruses or cells. However, Zhang et al. (Zhang et al., 2014) showed that human iPSC-derived mature HLCs hardly ever proliferated in vitro, and in contrast, human iPSC-derived hepatic endoderm cells exhibited a marked proliferative capability.
All of the protocols discussed above are static culture protocols for iPSCs, and three-dimensional culturing protocols can significantly enhance the function of HLCs. Hollow fiber/organoid culture of mouse iPSCs to induce expression of liver-specific genes and functions allows for spontaneous differentiation with cell proliferation and self-organization, high cell density, and the induction of differentiation in a large number of cells (Amimoto et al., 2011). The micro-cavitary hydrogel (MCG) system enhances nutrient exchange, permits greater living space for the encapsulated pluripotent stem cells to rapidly grow into colonies and results in significantly greater production of endoderm markers, hepatic markers, urea, and ALB by iPSCs compared to the typical non-MCG system, or monolayer culture (Lau et al., 2013). Zhang et al. (Liu et al., 2013) reported three-dimensional clump culture collagen matrices compatible with high throughput screening resulted in significantly increased functional maturation of iPSC-Heps towards an adult phenotype when compared to conventional culture systems. Additionally, this approach spontaneously results in the presence of polarized structures necessary for drug metabolism and improves the functional longevity when culturing in vitro over 75 days. Chiang et al. (Chiang et al., 2015) reprogrammed human dental pulp-derived fibroblasts into iPSCs, developed an injectable carboxymethyl-hexanoyl chitosan hydrogel (CHC) with sustained HGF release and investigated the hepatoprotective activity of HGF-CHC-delivered iPSC-Heps in vitro. Compared with PBS-delivered iPSC-Heps, the HGF-CHC-delivered iPSC-Heps exhibited higher antioxidant and anti-apoptotic activities, which resulted in a reduction of the hepatic necrotic area. Chien et al. (Chien et al., 2015) demonstrated that embedment of miR122 complexed with a polyurethane-graft-short-branch polyethylenimine copolymer in nanostructured amphiphatic carboxymethyl-hexanoyl chitosan led to dramatically enhanced miR122 delivery into human dental pulp-derived iPSCs and facilitated these cells to differentiate into iPSC-Heps with mature hepatocyte functions.
Hepatic progenitor cell-derived hepatocytes (HPC-Heps)
The first HPCs were identified by Farber (1956) and were termed oval cells, a small bipotent cell type with a high nuclear-to-cytoplasmic ratio, but in human it was defined as HPCs. They have been shown to emerge in several human liver diseases, including primary biliary cirrhosis (Crosby et al., 1998), primary sclerosing cholangitis (Vessey and de la Hall, 2001), and hepatocellular adenoma (Libbrecht et al., 2001). PH-activated progenitor cells are hepatic stem-like cells that can be cultured in vitro for more than 3 months, with the number of cells doubling 100 times over that period (He et al., 2004). Human liver-derived stem cells can be isolated and expanded from donated livers unsuitable for transplantation and present a comparable morphology to that of HSCs, which express a-smooth muscle actin, vimentin, fibronectin, CD73, and CD90 in accordance with their mesenchymal origin (Berardis et al., 2014). They can be induced to differentiate into cells with morphological, phenotypic, and functional characteristics of mature hepatocytes (He and Feng, 2011). Moreover, HPCs have a greater regenerative capacity than adult hepatocytes and participate in liver tissue repair and reconstruction following injury. It has been reported that HPCs transplantation can be used as a substitute for liver transplantation, and HPCs have a definite therapeutic effect on patients with end-stage liver diseases (Hughes et al., 2012). The effect of different PHs and the duration of collagenase perfusion on hepatic stem cell proliferation and differentiation varies; optimal differentiation of hepatic stem cells to CK-18 and AFP-positive cells was observed when stem cells isolated from 83.4% PH rats were perfused with IV collagenase for 20 min (Gong et al., 2013). Thy1 and CD44 oval and progenitor cells are able to differentiate into hepatocytes, but the degree of maturation of the induced hepatocytes may not be equal to that of healthy resident hepatocytes (Ichinohe et al., 2013). Messenger RNA expression levels of CYP1A2, CYP2B1/2, and CYP3A1 were higher in cells of young rats, and the proliferation and differentiation potential of oval cells decreased with age (Czekaj et al., 2010). Wang et al. (Wang et al., 2010) demonstrated slight acceleration of proliferation of hepatic oval cells after the 50th passage, but the cells remained diploid with features of chromosomal stability. Furthermore, they did not acquire anchorage-independent growth capacity and did not develop into tumors in immunodeficient mice, suggesting that hepatic oval cells do not undergo spontaneous malignant transformation.
Current protocols for the differentiation of oval cells make use of multiple treatments of soluble signals and/or matrix factors and typically result in partial differentiation of oval cells to mature cells with under- or over-expression of adult tissue-specific genes. The activation of AKT, p70s6k, and ERK1/2 induced by HGF in OC/CDE22 rat oval cells was abolished by pretreatment with a phosphoinositide 3-OH kinase inhibitor and a mitogen-activated protein kinase/ERK kinase inhibitor, respectively (Okano et al., 2003). This finding suggested that this signaling pathway is responsible for the biological effect of HGF. LE/2 and LE/6 oval cells are non-tumorigenic cells that were derived from the livers of adult rats fed a choline-deficient diet containing 0.1% ethionin. After 4 weeks in a three-dimensional collagen gel matrix and a fibroblast feeder layer culture system, these cells acquired typical hepatocytic morphology; however in the absence of a feeder layer and in the presence of HGF and/or keratinocyte growth factor, the precursor cells formed ductal structures, suggestive of differentiation along the bile duct lineage (Lazaro et al., 1998). The hepatic stem cell lines HY1, HY2, and HY3, which were derived from healthy livers of adult rats, showed an expression pattern similar to oval cells and efficiently induced hepatic differentiation following sequential treatment with type I collagen, TGF-b1, and HGF or oncostatin M (Hirata et al., 2009). Human hepatic stem cells seeded onto liver biomatrix scaffolds in a hormonally defined medium tailored for adult liver cells lost stem cell markers and differentiated into mature, functional parenchymal cells and remained viable with stable mature cell phenotypes for more than 8 weeks (Wang et al., 2011).
Mesenchymal stem cell-derived hepatocytes (MSC-Heps)
MSCs are highly proliferative, adherent mesenchymal cells with a unique cell surface molecule expression profile. The differentiation potential of adult stem cells has long been believed to be limited to the tissue or germ layer of their origin. In addition to long-term self-renewal capability, MSCs possess versatile differentiation potential ranging from mesenchyme-related multipotency to neuroectodermal and endodermal competency. Furthermore, they have the ability to differentiate into a number of organ-specific cell types, including hepatocytes (Christ and Dollinger, 2011). Adult stem cells can be derived from different tissues, such as bone marrow, adipose, placenta, umbilical cord blood, menstrual blood, and synovial tissues, etc.
When MSCs are treated with an external factor, their differentiation ability may be improved. Cytokines may play a more important role in the differentiation of MSCs into hepatocytes. When HGF, nicotinamide, or dexamethasone was added to MSCs individually, incomplete hepatocyte differentiation was achieved; the obtained cell populations contained progenitors that expressed both hepatic (ALB) and biliary (CK19) markers as well as AFP. When all factors were added to the culture medium, the cells exhibited features that closely resembled human adult hepatocytes (Chivu et al., 2009). A combination of insulin-like growth factor-I and liver-specific factors supported the potential development of MSCs into primary hepatocytes (Ayatollahi et al., 2011), and the addition of dimethylsulfoxide enhanced their differentiation into hepatocytes (Seo et al., 2005). Alpha-1 antitrypsin (AAT) deficiency is a hereditary disorder characterized by a severe decrease in AAT plasma level, which leads to progressive liver dysfunction; however, human MSC-Heps can be AAT genetically modified as a novel paradigm of coupling cell therapy for this disease (Ghaedi et al., 2010). Acute hepatic failure-derived bone marrow mesenchymal stem cells (BMMSCs) have a hepatic transcriptional profile, express hepatocyte-specific genes early during differentiation, and possess greater hepatogenic potency in vitro compared to cells isolated from control animals (Li et al., 2010a). Pretreatment of MSCs with injured liver tissue in an in vitro model resulted in high expression of albumin, CK8, CK18, transaminase, and HNF1a compared to untreated MSCs, indicating that this pretreatment augmented the homing and hepatic differentiation abilities of MSCs (Mohsin et al., 2011). Culturing in hepatocyte-conditioned medium without any cytokines can induce the differentiation of BMMSCs into HLCs (Chen et al., 2007). Quiescent HSCs or culture-activated HSCs do not have the ability to modulate the differentiation of MSCs. Moreover, Kupffer cell-activated HSCs expressed HGF mRNA, and culture-activated HSCs did not (Deng et al., 2008).
MSCs grown on nanofibers showed enhanced differentiation into HLCs and maintained their function in long-term culture; hepatocyte markers ALB and HNF4α were elevated in a time-dependent manner, and CYP450 enzymes were significantly increased in the HLCs differentiated in vitro from MSCs grown on nanofibers at day 36 (Piryaei et al., 2011). BMMSCs cultured in alginate scaffolds in the presence of specific GFs display several liver-specific markers and functions (Lin et al., 2010). A three-dimensional co-culture system of porcine hepatocytes and BMMSCs was established in vitro, and the best hepatic function levels were achieved on day 2 and moderately decreased in the following co-culture days (Gu et al., 2009). Culturing human adipose-derived mesenchymal stem cells (ADMSCs) on top of HGF/Col spots (HGF co-printed with collagen I to create arrays of protein spots on glass) for 2 weeks can differentiate them into HLCs (Ghaedi et al., 2011). Floating culture efficiently induced human ADMSCs into functional HLCs in vitro (Okura et al., 2010).
FUTURE DIRECTIONS
The liver regeneration process has been evaluated in PH models. Although it has been studied extensively, many important fundamental mechanisms remain undefined, such as the mechanisms of cellular hypertrophy, cell division, nuclear division, ploidy changes, and organ size control. The current shortage of donor organs available for live transplantation and the severe morbidity and mortality associated with this procedure underscores the need for alternatives to liver transplantation. In vivo hepatocytes proliferate to repair injured liver tissue, but this process is too complex to mimic in vitro to obtain functional hepatocytes. Culture methods for the enhancement of isolated primary hepatocytes or HLCs in vitro include the addition of GFs, TFs, and cytokines, the activation of signaling pathways, the use of an optimized matrix, and co-culture with various nonparenchymal cells. All of these methods are able to improve the function of primary hepatocytes or HLCs in vitro to a certain extent, but unfortunately, the amount of available mature hepatocytes have been insufficient for clinical use in the past several decades. And the lack of standardization of protocols for isolating specific cell types and the use of a variety of injury/disease models have made the interpretation of these results rather difficult and have left unanswered questions regarding the mechanisms through which these cells generate their beneficial effects.
The observed effects of cell therapies for liver regeneration have certainly generated reserved optimism toward their value for clinical use once the methodologies have become standardized and optimized. With clarification of the process of liver regeneration, hepatocytes or HLCs that can grow and proliferate persistently and functionally in vitro and in vivo will be able to be used to fulfill the needs of injured livers for regeneration, and cell transplantation and artificial liver support will completely replace liver transplantation.
REFERENCES
Amimoto N, Mizumoto H, Nakazawa K, Ijima H, Funatsu K, Kajiwara T (2011) Hepatic differentiation of mouse embryonic stem cells and induced pluripotent stem cells during organoid formation in hollow fibers. Tissue Eng Part A 17:2071–2078
Ayatollahi M, Soleimani M, Tabei SZ, Salmani MK (2011) Hepatogenic differentiation of mesenchymal stem cells induced by insulin like growth factor-I. World J Stem Cells 3:113–121
Berardis S, Lombard C, Evraerts J, El Taghdouini A, Rosseels V, Sancho-Bru P, Lozano JJ, van Grunsven L, Sokal E, Najimi M (2014) Gene expression profiling and secretome analysis differentiate adult-derived human liver stem/progenitor cells and human hepatic stellate cells. PLoS One 9:e86137
Chan KM, Fu YH, Wu TJ, Hsu PY, Lee WC (2013) Hepatic stellate cells promote the differentiation of embryonic stem cell-derived definitive endodermal cells into hepatic progenitor cells. Hepatol Res 43:648–657
Chen Y, Dong XJ, Zhang GR, Shao JZ, Xiang LX (2007) In vitro differentiation of mouse bone marrow stromal stem cells into hepatocytes induced by conditioned culture medium of hepatocytes. J Cell Biochem 102:52–63
Chen YF, Tseng CY, Wang HW, Kuo HC, Yang VW, Lee OK (2012) Rapid generation of mature hepatocyte-like cells from human induced pluripotent stem cells by an efficient three-step protocol. Hepatology (Baltimore, Md.) 55:1193–1203
Chiang CH, Wu WW, Li HY, Chien Y, Sun CC, Peng CH, Lin AT, Huang CS, Lai YH, Chiou SH et al (2015) Enhanced antioxidant capacity of dental pulp-derived iPSC-differentiated hepatocytes and liver regeneration by injectable HGF-releasing hydrogel in fulminant hepatic failure. Cell Transplant 24:541
Chien Y, Chang YL, Li HY, Larsson M, Wu WW, Chien CS, Wang CY, Chu PY, Chen KH, Lo WL et al (2015) Synergistic effects of carboxymethyl-hexanoyl chitosan, cationic polyurethane-short branch PEI in miR122 gene delivery: accelerated differentiation of iPSCs into mature hepatocyte-like cells and improved stem cell therapy in a hepatic failure model. Acta Biomater 13:228–244
Chivu M, Dima SO, Stancu CI, Dobrea C, Uscatescu V, Necula LG, Bleotu C, Tanase C, Albulescu R, Ardeleanu C et al (2009) In vitro hepatic differentiation of human bone marrow mesenchymal stem cells under differential exposure to liver-specific factors. Transl Res 154:122–132
Cho CH, Berthiaume F, Tilles AW, Yarmush ML (2008) A new technique for primary hepatocyte expansion in vitro. Biotechnol Bioeng 101:345–356
Christ B, Dollinger MM (2011) The generation of hepatocytes from mesenchymal stem cells and engraftment into the liver. Curr Opin Organ Transplant 16:69–75
Cornell RP, Liljequist BL, Bartizal KF (1990) Depressed liver regeneration after partial hepatectomy of germ-free, athymic and lipopolysaccharide-resistant mice. Hepatology (Baltimore, Md.) 11:916–922
Crosby HA, Hubscher S, Fabris L, Joplin R, Sell S, Kelly D, Strain AJ (1998) Immunolocalization of putative human liver progenitor cells in livers from patients with end-stage primary biliary cirrhosis and sclerosing cholangitis using the monoclonal antibody OV-6. Am J Pathol 152:771–779
Czekaj P, Bryzek A, Czekaj TM, Koryciak-Komarska H, Wiaderkiewicz A, Plewka D, Sieron AL (2010) Cytochrome P450 mRNA expressions along with in vitro differentiation of hepatocyte precursor cells from fetal, young and old rats. Folia Histochem Cytobiol 48:46–57
da No Y, Lee SA, Choi YY, Park D, Jang JY, Kim DS, Lee SH (2012) Functional 3D human primary hepatocyte spheroids made by co-culturing hepatocytes from partial hepatectomy specimens and human adipose-derived stem cells. PLoS One 7:e50723
DeAngelis RA, Markiewski MM, Kourtzelis I, Rafail S, Syriga M, Sandor A, Maurya MR, Gupta S, Subramaniam S, Lambris JD (2012) A complement-IL-4 regulatory circuit controls liver regeneration. J Immunol 188:641–648
Delgado I, Fresnedo O, Iglesias A, Rueda Y, Syn WK, Zubiaga AM, Ochoa B (2011) A role for transcription factor E2F2 in hepatocyte proliferation and timely liver regeneration. Am J Physiol Gastrointest Liver Physiol 301:G20–31
Deng X, Chen YX, Zhang X, Zhang JP, Yin C, Yue HY, Lin Y, Han ZG, Xie WF (2008) Hepatic stellate cells modulate the differentiation of bone marrow mesenchymal stem cells into hepatocyte-like cells. J Cell Physiol 217:138–144
Deng X, Li W, Chen N, Sun Y, Wei H, Jiang Y, He F (2009) Exploring the priming mechanism of liver regeneration: proteins and protein complexes. Proteomics 9:2202–2216
Du C, Narayanan K, Leong MF, Wan AC (2014) Induced pluripotent stem cell-derived hepatocytes and endothelial cells in multi-component hydrogel fibers for liver tissue engineering. Biomaterials 35:6006–6014
Duan BW, Lu SC, Wang ML, Liu JN, Chi P, Lai W, Wu JS, Guo QL, Lin DD, Liu Y et al (2013) Liver transplantation in acute-on-chronic liver failure patients with high model for end-stage liver disease (MELD) scores: a single center experience of 100 consecutive cases. J Surg Res 183:936–943
Farber E (1956) Similarities in the sequence of early histological changes induced in the liver of the rat by ethionine, 2-acetylamino-fluorene, and 3’-methyl-4-dimethylaminoazobenzene. Cancer Res 16:142–148
Farzaneh Z, Pakzad M, Vosough M, Pournasr B, Baharvand H (2014) Differentiation of human embryonic stem cells to hepatocyte-like cells on a new developed xeno-free extracellular matrix. Histochem Cell Biol 142:217–226
Fujiyoshi M, Ozaki M (2011) Molecular mechanisms of liver regeneration and protection for treatment of liver dysfunction and diseases. J Hepato-Biliary-Pancreatic Sci 18:13–22
Funakoshi N, Duret C, Pascussi JM, Blanc P, Maurel P, Daujat-Chavanieu M, Gerbal-Chaloin S (2011) Comparison of hepatic-like cell production from human embryonic stem cells and adult liver progenitor cells: CAR transduction activates a battery of detoxification genes. Stem Cell Rev 7:518–531
Gao L, Utsumi T, Tashiro K, Liu B, Zhang D, Swenson ES, Iwakiri Y (2013) Reticulon 4B (Nogo-B) facilitates hepatocyte proliferation and liver regeneration in mice. Hepatology (Baltimore, Md.) 57:1992–2003
Ghaedi M, Lotfi AS, Soleimani M (2010) Establishment of lentiviral-vector-mediated model of human alpha-1 antitrypsin delivery into hepatocyte-like cells differentiated from mesenchymal stem cells. Tissue Cell 42:181–189
Ghaedi M, Tuleuova N, Zern MA, Wu J, Revzin A (2011) Bottom-up signaling from HGF-containing surfaces promotes hepatic differentiation of mesenchymal stem cells. Biochem Biophys Res Commun 407:295–300
Ghodsizadeh A, Taei A, Totonchi M, Seifinejad A, Gourabi H, Pournasr B, Aghdami N, Malekzadeh R, Almadani N, Salekdeh GH et al (2010) Generation of liver disease-specific induced pluripotent stem cells along with efficient differentiation to functional hepatocyte-like cells. Stem Cell Rev 6:622–632
Gong P, Wang Y, Zhang J, Wang Z (2013) Differential hepatic stem cell proliferation and differentiation after partial hepatectomy in rats. Mol Med Rep 8:1005–1010
Gu J, Shi X, Zhang Y, Chu X, Hang H, Ding Y (2009) Establishment of a three-dimensional co-culture system by porcine hepatocytes and bone marrow mesenchymal stem cells in vitro. Hepatol Res 39:398–407
Hannan NR, Segeritz CP, Touboul T, Vallier L (2013) Production of hepatocyte-like cells from human pluripotent stem cells. Nat Protoc 8:430–437
Haque A, Hexig B, Meng Q, Hossain S, Nagaoka M, Akaike T (2011) The effect of recombinant E-cadherin substratum on the differentiation of endoderm-derived hepatocyte-like cells from embryonic stem cells. Biomaterials 32:2032–2042
He Z, Feng M (2011) Activation, isolation, identification and culture of hepatic stem cells from porcine liver tissues. Cell Prolif 44:558–566
He ZP, Tan WQ, Tang YF, Zhang HJ, Feng MF (2004) Activation, isolation, identification and in vitro proliferation of oval cells from adult rat livers. Cell Prolif 37:177–187
Hirata M, Amano K, Miyashita A, Yasunaga M, Nakanishi T, Sato K (2009) Establishment and characterization of hepatic stem-like cell lines from normal adult rat liver. J Biochem 145:51–58
Hughes RD, Mitry RR, Dhawan A (2012) Current status of hepatocyte transplantation. Transplantation 93:342–347
Ichinohe N, Tanimizu N, Ooe H, Nakamura Y, Mizuguchi T, Kon J, Hirata K, Mitaka T (2013) Differentiation capacity of hepatic stem/progenitor cells isolated from D-galactosamine-treated rat livers. Hepatology (Baltimore, Md.) 57:1192–1202
Ishii T, Yasuchika K, Fukumitsu K, Kawamoto T, Kawamura-Saitoh M, Amagai Y, Ikai I, Uemoto S, Kawase E, Suemori H et al (2010) In vitro hepatic maturation of human embryonic stem cells by using a mesenchymal cell line derived from murine fetal livers. Cell Tissue Res 339:505–512
Javed MS, Yaqoob N, Iwamuro M, Kobayashi N, Fujiwara T (2014) Generation of hepatocyte-like cells from human induced pluripotent stem (iPS) cells by co-culturing embryoid body cells with liver non-parenchymal cell line TWNT-1. J Coll Phys Surg-Pak 24:91–96
Jia F, Wilson KD, Sun N, Gupta DM, Huang M, Li Z, Panetta NJ, Chen ZY, Robbins RC, Kay MA et al (2010) A nonviral minicircle vector for deriving human iPS cells. Nat Methods 7:197–199
Kajiwara M, Aoi T, Okita K, Takahashi R, Inoue H, Takayama N, Endo H, Eto K, Toguchida J, Uemoto S et al (2012) Donor-dependent variations in hepatic differentiation from human-induced pluripotent stem cells. Proc Natl Acad Sci USA 109:12538–12543
Kim Y, Rajagopalan P (2010) 3D hepatic cultures simultaneously maintain primary hepatocyte and liver sinusoidal endothelial cell phenotypes. PLoS One 5:e15456
Kim WR, Therneau TM, Benson JT, Kremers WK, Rosen CB, Gores GJ, Dickson ER (2006) Deaths on the liver transplant waiting list: an analysis of competing risks. Hepatology (Baltimore, Md.) 43:345–351
Kim SE, An SY, Woo DH, Han J, Kim JH, Jang YJ, Son JS, Yang H, Cheon YP (2013) Engraftment potential of spheroid-forming hepatic endoderm derived from human embryonic stem cells. Stem Cells Dev 22:1818–1829
Kim SJ, Park MH, Moon HJ, Park JH, du Ko Y, Jeong B (2014) Polypeptide thermogels as a three dimensional culture scaffold for hepatogenic differentiation of human tonsil-derived mesenchymal stem cells. ACS Appl Mater Interfaces 6:17034–17043
Kondo Y, Iwao T, Yoshihashi S, Mimori K, Ogihara R, Nagata K, Kurose K, Saito M, Niwa T, Suzuki T et al (2014) Histone deacetylase inhibitor valproic acid promotes the differentiation of human induced pluripotent stem cells into hepatocyte-like cells. PLoS One 9:e104010
Kwon HJ, Won YS, Yoon YD, Yoon WK, Nam KH, Choi IP, Kim DY, Kim HC (2011) Vitamin D3 up-regulated protein 1 deficiency accelerates liver regeneration after partial hepatectomy in mice. J Hepatol 54:1168–1176
Lau TT, Ho LW, Wang DA (2013) Hepatogenesis of murine induced pluripotent stem cells in 3D micro-cavitary hydrogel system for liver regeneration. Biomaterials 34:6659–6669
Lazaro CA, Rhim JA, Yamada Y, Fausto N (1998) Generation of hepatocytes from oval cell precursors in culture. Cancer Res 58:5514–5522
Lee JY, Tuleuova N, Jones CN, Ramanculov E, Zern MA, Revzin A (2009) Directing hepatic differentiation of embryonic stem cells with protein microarray-based co-cultures. Integr Biol 1:460–468
Li W, Liang X, Leu JI, Kovalovich K, Ciliberto G, Taub R (2001) Global changes in interleukin-6-dependent gene expression patterns in mouse livers after partial hepatectomy. Hepatology (Baltimore, Md.) 33:1377–1386
Li W, Wei W, Zhu S, Zhu J, Shi Y, Lin T, Hao E, Hayek A, Deng H, Ding S (2009) Generation of rat and human induced pluripotent stem cells by combining genetic reprogramming and chemical inhibitors. Cell Stem Cell 4:16–19
Li J, Tao R, Wu W, Cao H, Xin J, Guo J, Jiang L, Hong X, Demetriou AA, Farkas D et al (2010a) Transcriptional profiling and hepatogenic potential of acute hepatic failure-derived bone marrow mesenchymal stem cells. Differentiation 80:166–174
Li W, Wang D, Qin J, Liu C, Zhang Q, Zhang X, Yu X, Lahn BT, Mao FF, Xiang AP (2010b) Generation of functional hepatocytes from mouse induced pluripotent stem cells. J Cell Physiol 222:492–501
Li F, He Z, Li Y, Liu P, Chen F, Wang M, Zhu H, Ding X, Wangensteen KJ, Hu Y et al (2011a) Combined activin A/LiCl/Noggin treatment improves production of mouse embryonic stem cell-derived definitive endoderm cells. J Cell Biochem 112:1022–1034
Li HY, Chien Y, Chen YJ, Chen SF, Chang YL, Chiang CH, Jeng SY, Chang CM, Wang ML, Chen LK et al (2011b) Reprogramming induced pluripotent stem cells in the absence of c-Myc for differentiation into hepatocyte-like cells. Biomaterials 32:5994–6005
Libbrecht L, De Vos R, Cassiman D, Desmet V, Aerts R, Roskams T (2001) Hepatic progenitor cells in hepatocellular adenomas. Am J Surg Pathol 25:1388–1396
Lin N, Lin J, Bo L, Weidong P, Chen S, Xu R (2010) Differentiation of bone marrow-derived mesenchymal stem cells into hepatocyte-like cells in an alginate scaffold. Cell Prolif 43:427–434
Liu H, Ye Z, Kim Y, Sharkis S, Jang YY (2010a) Generation of endoderm-derived human induced pluripotent stem cells from primary hepatocytes. Hepatology (Baltimore, Md.) 51:1810–1819
Liu T, Zhang S, Chen X, Li G, Wang Y (2010b) Hepatic differentiation of mouse embryonic stem cells in three-dimensional polymer scaffolds. Tissue Eng Part A 16:1115–1122
Liu T, Zhang S, Xiang D, Wang Y (2013) Induction of hepatocyte-like cells from mouse embryonic stem cells by lentivirus-mediated constitutive expression of Foxa2/Hnf4a. J Cell Biochem 114:2531–2541
Luebke-Wheeler JL, Nedredal G, Yee L, Amiot BP, Nyberg SL (2009) E-cadherin protects primary hepatocyte spheroids from cell death by a caspase-independent mechanism. Cell Transplant 18:1281–1287
Mao SA, Glorioso JM, Nyberg SL (2014) Liver regeneration. Transl Res 163:352–362
Marekova D, Lesny P, Jendelova P, Michalek J, Kostecka P, Pradny M, Martinova L, Pantoflicek T, Ryska M, Sykova E (2013) Hepatocyte growth on polycapronolactone and 2-hydroxyethylmethacrylate nanofiber sheets enhanced by bone marrow-derived mesenchymal stromal cells. Hepato-gastroenterology 60:1156–1163
Michalopoulos GK (2010) Liver regeneration after partial hepatectomy: critical analysis of mechanistic dilemmas. Am J Pathol 176:2–13
Miki T, Ring A, Gerlach J (2011) Hepatic differentiation of human embryonic stem cells is promoted by three-dimensional dynamic perfusion culture conditions. Tissue Eng 17:557–568
Mizutani T, Yokoyama Y, Kokuryo T, Kawai K, Miyake T, Nagino M (2013) Calcitonin gene-related peptide regulates the early phase of liver regeneration. J Surg Res 183:138–145
Mobarra N, Soleimani M, Kouhkan F, Hesari Z, Lahmy R, Mossahebi-Mohammadi M, Arefian E, Jaafarpour Z, Nasiri H, Pakzad R et al (2014) Efficient differentiation of human induced pluripotent stem cell (hiPSC) derived hepatocyte-like cells on hMSCs Feeder. Int J Hematol-Oncol Stem Cell Res 8:20–29
Mohsin S, Shams S, Ali Nasir G, Khan M, Javaid Awan S, Khan SN, Riazuddin S (2011) Enhanced hepatic differentiation of mesenchymal stem cells after pretreatment with injured liver tissue. Differentiation 81:42–48
Moore RN, Moghe PV (2009) Expedited growth factor-mediated specification of human embryonic stem cells toward the hepatic lineage. Stem Cell Res 3:51–62
Murry CE, Keller G (2008) Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell 132:661–680
Nagamoto Y, Tashiro K, Takayama K, Ohashi K, Kawabata K, Sakurai F, Tachibana M, Hayakawa T, Furue MK, Mizuguchi H (2012) The promotion of hepatic maturation of human pluripotent stem cells in 3D co-culture using type I collagen and Swiss 3T3 cell sheets. Biomaterials 33:4526–4534
Nagaoka M, Hagiwara Y, Takemura K, Murakami Y, Li J, Duncan SA, Akaike T (2008) Design of the artificial acellular feeder layer for the efficient propagation of mouse embryonic stem cells. J Biol Chem 283:26468–26476
Nejak-Bowen KN, Orr AV, Bowen WC Jr, Michalopoulos GK (2013) Gliotoxin-induced changes in rat liver regeneration after partial hepatectomy. Liver Int 33:1044–1055
No da Y, Lee SA, Choi YY, Park D, Jang JY, Kim DS, Lee SH (2012) Functional 3D human primary hepatocyte spheroids made by co-culturing hepatocytes from partial hepatectomy specimens and human adipose-derived stem cells. PloS one 7:e50723
Noto FK, Determan MR, Cai J, Cayo MA, Mallanna SK, Duncan SA (2014) Aneuploidy is permissive for hepatocyte-like cell differentiation from human induced pluripotent stem cells. BMC Res Notes 7:437
Nygard IE, Mortensen KE, Hedegaard J, Conley LN, Kalstad T, Bendixen C, Revhaug A (2012) The genetic regulation of the terminating phase of liver regeneration. Comp Hepatol 11:3
Okano J, Shiota G, Matsumoto K, Yasui S, Kurimasa A, Hisatome I, Steinberg P, Murawaki Y (2003) Hepatocyte growth factor exerts a proliferative effect on oval cells through the PI3K/AKT signaling pathway. Biochem Biophys Res Commun 309:298–304
Okura H, Komoda H, Saga A, Kakuta-Yamamoto A, Hamada Y, Fumimoto Y, Lee CM, Ichinose A, Sawa Y, Matsuyama A (2010) Properties of hepatocyte-like cell clusters from human adipose tissue-derived mesenchymal stem cells. Tissue Eng 16:761–770
Olazabal IM, Munoz JA, Rodriguez-Navas C, Alvarez L, Delgado-Baeza E, Garcia-Ruiz JP (2009) Prolactin’s role in the early stages of liver regeneration in rats. J Cell Physiol 219:626–633
Paracchini V, Carbone A, Colombo F, Castellani S, Mazzucchelli S, Gioia SD, Degiorgio D, Seia M, Porretti L, Colombo C et al (2012) Amniotic mesenchymal stem cells: a new source for hepatocyte-like cells and induction of CFTR expression by coculture with cystic fibrosis airway epithelial cells. J Biomed Biotechnol 2012:575471
Piryaei A, Valojerdi MR, Shahsavani M, Baharvand H (2011) Differentiation of bone marrow-derived mesenchymal stem cells into hepatocyte-like cells on nanofibers and their transplantation into a carbon tetrachloride-induced liver fibrosis model. Stem cell Rev 7:103–118
Rizzo F, Hashim A, Marchese G, Ravo M, Tarallo R, Nassa G, Giurato G, Rinaldi A, Cordella A, Persico M et al (2014) Timed regulation of P-element-induced wimpy testis-interacting RNA expression during rat liver regeneration. Hepatology (Baltimore, Md.) 60:798–806
Samuel D, Colombo M, El-Serag H, Sobesky R, Heaton N (2011) Toward optimizing the indications for orthotopic liver transplantation in hepatocellular carcinoma. Liver Transplant 17(Suppl 2):S6–13
Sancho-Bru P, Najimi M, Caruso M, Pauwelyn K, Cantz T, Forbes S, Roskams T, Ott M, Gehling U, Sokal E et al (2009) Stem and progenitor cells for liver repopulation: can we standardise the process from bench to bedside? Gut 58:594–603
Sellaro TL, Ranade A, Faulk DM, McCabe GP, Dorko K, Badylak SF, Strom SC (2010) Maintenance of human hepatocyte function in vitro by liver-derived extracellular matrix gels. Tissue Eng 16:1075–1082
Seo MJ, Suh SY, Bae YC, Jung JS (2005) Differentiation of human adipose stromal cells into hepatic lineage in vitro and in vivo. Biochem Biophys Res Commun 328:258–264
Shin M, Alev C, Wu Y, Nagai H, Sheng G (2011) Activin/TGF-beta signaling regulates Nanog expression in the epiblast during gastrulation. Mech Dev 128:268–278
Shiraki N, Yamazoe T, Qin Z, Ohgomori K, Mochitate K, Kume K, Kume S (2011) Efficient differentiation of embryonic stem cells into hepatic cells in vitro using a feeder-free basement membrane substratum. PLoS One 6:e24228
Si-Tayeb K, Noto FK, Nagaoka M, Li J, Battle MA, Duris C, North PE, Dalton S, Duncan SA (2010) Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology (Baltimore, Md.) 51:297–305
Sivertsson L, Synnergren J, Jensen J, Bjorquist P, Ingelman-Sundberg M (2013) Hepatic differentiation and maturation of human embryonic stem cells cultured in a perfused three-dimensional bioreactor. Stem Cells Dev 22:581–594
Skardal A, Smith L, Bharadwaj S, Atala A, Soker S, Zhang Y (2012) Tissue specific synthetic ECM hydrogels for 3-D in vitro maintenance of hepatocyte function. Biomaterials 33:4565–4575
Strey CW, Markiewski M, Mastellos D, Tudoran R, Spruce LA, Greenbaum LE, Lambris JD (2003) The proinflammatory mediators C3a and C5a are essential for liver regeneration. J Exp Med 198:913–923
Subramanian K, Owens DJ, Raju R, Firpo M, O’Brien TD, Verfaillie CM, Hu WS (2014) Spheroid culture for enhanced differentiation of human embryonic stem cells to hepatocyte-like cells. Stem Cells Dev 23:124–131
Takata A, Otsuka M, Kogiso T, Kojima K, Yoshikawa T, Tateishi R, Kato N, Shiina S, Yoshida H, Omata M et al (2011) Direct differentiation of hepatic cells from human induced pluripotent stem cells using a limited number of cytokines. Hepatol Int 5:890
Takebe T, Sekine K, Enomura M, Koike H, Kimura M, Ogaeri T, Zhang RR, Ueno Y, Zheng YW, Koike N et al (2013) Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499:481–484
Tamai M, Yamashita A, Tagawa Y (2011) Mitochondrial development of the in vitro hepatic organogenesis model with simultaneous cardiac mesoderm differentiation from murine induced pluripotent stem cells. J Biosci Bioeng 112:495–500
Tatsumi K, Ohashi K, Taminishi S, Sakurai Y, Ogiwara K, Yoshioka A, Okano T, Shima M (2011) Regulation of coagulation factors during liver regeneration in mice: mechanism of factor VIII elevation in plasma. Thromb Res 128:54–61
Tomizawa M, Shinozaki F, Sugiyama T, Yamamoto S, Sueishi M, Yoshida T (2013) Single-step protocol for the differentiation of human-induced pluripotent stem cells into hepatic progenitor-like cells. Biomed Rep 1:18–22
Tuleuova N, Lee JY, Lee J, Ramanculov E, Zern MA, Revzin A (2010) Using growth factor arrays and micropatterned co-cultures to induce hepatic differentiation of embryonic stem cells. Biomaterials 31:9221–9231
Ulvestad M, Nordell P, Asplund A, Rehnstrom M, Jacobsson S, Holmgren G, Davidson L, Brolen G, Edsbagge J, Bjorquist P et al (2013) Drug metabolizing enzyme and transporter protein profiles of hepatocytes derived from human embryonic and induced pluripotent stem cells. Biochem Pharmacol 86:691–702
Umeda K, Suzuki K, Yamazoe T, Shiraki N, Higuchi Y, Tokieda K, Kume K, Mitani K, Kume S (2013) Albumin gene targeting in human embryonic stem cells and induced pluripotent stem cells with helper-dependent adenoviral vector to monitor hepatic differentiation. Stem Cell Res 10:179–194
Uriarte I, Fernandez-Barrena MG, Monte MJ, Latasa MU, Chang HC, Carotti S, Vespasiani-Gentilucci U, Morini S, Vicente E, Concepcion AR et al (2013) Identification of fibroblast growth factor 15 as a novel mediator of liver regeneration and its application in the prevention of post-resection liver failure in mice. Gut 62:899–910
Vessey CJ, de la Hall PM (2001) Hepatic stem cells: a review. Pathology 33:130–141
Wang S, Nagrath D, Chen PC, Berthiaume F, Yarmush ML (2008) Three-dimensional primary hepatocyte culture in synthetic self-assembling peptide hydrogel. Tissue Eng 14:227–236
Wang P, Cong M, Liu TH, Yang AT, Cong R, Wu P, Tang SZ, Xu Y, Wang H, Wang BE et al (2010) Primary isolated hepatic oval cells maintain progenitor cell phenotypes after two-year prolonged cultivation. J Hepatol 53:863–871
Wang Y, Cui CB, Yamauchi M, Miguez P, Roach M, Malavarca R, Costello MJ, Cardinale V, Wauthier E, Barbier C et al (2011) Lineage restriction of human hepatic stem cells to mature fates is made efficient by tissue-specific biomatrix scaffolds. Hepatology (Baltimore, Md.) 53:293–305
Wang Y, Zhang Y, Zhang S, Peng G, Liu T, Li Y, Xiang D, Wassler MJ, Shelat HS, Geng Y (2012) Rotating microgravity-bioreactor cultivation enhances the hepatic differentiation of mouse embryonic stem cells on biodegradable polymer scaffolds. Tissue Eng 18:2376–2385
Wolf JH, Bhatti TR, Fouraschen S, Chakravorty S, Wang L, Kurian S, Salomon D, Olthoff KM, Hancock WW, Levine MH (2014) Heat shock protein 70 is required for optimal liver regeneration after partial hepatectomy in mice. Liver Transplant 20:376–385
Wuestefeld T, Pesic M, Rudalska R, Dauch D, Longerich T, Kang TW, Yevsa T, Heinzmann F, Hoenicke L, Hohmeyer A et al (2013) A Direct in vivo RNAi screen identifies MKK4 as a key regulator of liver regeneration. Cell 153:389–401
Yamanaka S, Blau HM (2010) Nuclear reprogramming to a pluripotent state by three approaches. Nature 465:704–712
Yang Y, Li J, Pan X, Zhou P, Yu X, Cao H, Wang Y, Li L (2013) Co-culture with mesenchymal stem cells enhances metabolic functions of liver cells in bioartificial liver system. Biotechnol Bioeng 110:958–968
Yanger K, Zong Y, Maggs LR, Shapira SN, Maddipati R, Aiello NM, Thung SN, Wells RG, Greenbaum LE, Stanger BZ (2013) Robust cellular reprogramming occurs spontaneously during liver regeneration. Genes Dev 27:719–724
Yao R, Wang J, Li X, Jung Jung D, Qi H, Kee KK, Du Y (2014) Hepatic differentiation of human embryonic stem cells as microscaled multilayered colonies leading to enhanced homogeneity and maturation. Small 10:4311–4323
Zerrad-Saadi A, Lambert-Blot M, Mitchell C, Bretes H, Collin de l’Hortet A, Baud V, Chereau F, Sotiropoulos A, Kopchick JJ, Liao L et al (2011) GH receptor plays a major role in liver regeneration through the control of EGFR and ERK1/2 activation. Endocrinology 152:2731–2741
Zhang S, Zhang Y, Chen L, Liu T, Li Y, Wang Y, Geng Y (2013a) Efficient large-scale generation of functional hepatocytes from mouse embryonic stem cells grown in a rotating bioreactor with exogenous growth factors and hormones. Stem Cell Res Ther 4:145
Zhang Z, Liu J, Liu Y, Li Z, Gao WQ, He Z (2013b) Generation, characterization and potential therapeutic applications of mature and functional hepatocytes from stem cells. J Cell Physiol 228:298–305
Zhang R, Takebe T, Sekine K, Koike H, Zheng Y, Taniguchi H (2014) Identification of proliferating human hepatic cells from human induced pluripotent stem cells. Transplant Proc 46:1201–1204
Zhao D, Chen S, Cai J, Guo Y, Song Z, Che J, Liu C, Wu C, Ding M, Deng H (2009) Derivation and characterization of hepatic progenitor cells from human embryonic stem cells. PLoS One 4:e6468
ABBREVIATIONS
AAT, alpha-1 antitrypsin; ADMSCs, adipose derived mesenchymal stem cells; AFP, alpha fetal protein; ALB, albumin; BMMSCs, bone marrow mesenchymal stem cells; BMP, bone morphogenic protein; CHC, carboxymethyl-hexanoyl chitosan hydrogel; CYP, cytochrome P; DE, definitive endoderm; EB, embryoid bodies; ECM, extracellular matrix; ESC, embryonic stem cell; ESC-Heps, embryonic stem cell-derived hepatocytes; FGF, fibroblast growth factor; GFs, growth factors; HLCs, hepatocyte-like cells; HPC, hepatic progenitor cell; HPC-Heps, hepatic progenitor cell-derived hepatocytes; HSCs, hepatic stellate cells; IL, interleukin; iPSC, induced pluripotent stem cell; iPSC-Heps, induced pluripotent stem cell-derived hepatocytes; MCG, micro-cavitary hydrogel; mKO1, monomeric Kusabira orange; MSC, mesenchymal stem cell; MSC-Heps, mesenchymal stem cell-derived hepatocytes; PH, partial hepatectomy; SHs, small hepatocyte-like progenitor cells; TFs, transcription factors.
COMPLIANCE WITH ETHICS GUIDELINES
Chenxia Hu and Lanjuan Li declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hu, C., Li, L. In vitro culture of isolated primary hepatocytes and stem cell-derived hepatocyte-like cells for liver regeneration. Protein Cell 6, 562–574 (2015). https://doi.org/10.1007/s13238-015-0180-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13238-015-0180-2