Abstract
The post-operative abdomen can be challenging and knowledge of normal post-operative anatomy is important for diagnosing complications. The aim of this pictorial essay is to describe a few selected common, major gastrointestinal surgeries, their clinical indications and depict their normal post-operative computed tomography (CT) appearance. This essay provides some clues to identify the surgeries, which can be helpful especially when surgical history is lacking: recognition of the organ(s) involved, determination of what was resected and familiarity with the type of anastomoses used.
Similar content being viewed by others
Introduction
The post-operative abdomen can be challenging. Familiarity with normal post-operative anatomy can be helpful in diagnosing complications. The introduction of mechanical stapling devices using radio-opaque metallic sutures has greatly facilitated the identification of surgical anastomoses. However, occasionally some or part of anastomoses can be hand-sewn and if no radio-opaque markers are left in place, this is occult on computed tomography (CT), rendering the radiologist’s task difficult in identifying an anastomosis. There are three main types of anastomoses [1]. These include: (1) end to end—used when there is adequate luminal diameter on both sides; (2) end to side—used when there is a size discrepancy between the parts to be joined; (3) side to side—used in creation of a large anastomosis in the setting of a narrow lumen (Fig. 1).
A typical staple line has either two or three staggered rows of staples which cannot be individually resolved by CT. In addition there are three basic types of staplers: (1) circular staplers that produce a continuous ring of staples, often for end-to-end or end-to-side anastomoses; (2) linear staplers that produce one staple line, often used to transect a lumen, resulting in a stapled blind end and an open end which is resected; (3) linear staplers that produce a double staple line and include a knife to cut between the lines, used for side-to-side anastomoses, and to transect a lumen but sealing both sides simultaneously. This explains the finding on CT of two long linear staple lines at the site of a gastro-jejunostomy.
The standard practice at our institution is to image patients in the early postoperative period only when there is clinical suspicion of postoperative complications. Follow-up imaging is generally performed at 3- to 6-month intervals. At our institution, oral contrast is used on a per patient basis, according to suspicion of anastomotic leak by the clinician. When needed, we administer 750 ml of water-soluble oral contrast agent beginning approximately 1 h prior to imaging, with an additional 250 ml just prior to the start of CT. Additionally, 120 ml of a non-ionic contrast agent is administered intravenously at a rate of 3 ml/s, unless contraindicated. Multidetector CT scanning is started 70 s after the contrast agent injection. Parameters include a section thickness of 2.5 mm; pitch 1.25; 120 kVp; auto-milliamperage.
Gastric surgeries
Billroth 1 and 2
These surgeries are indicated for gastric malignancy and refractory peptic ulcer disease. Billroth 1 surgery entails a partial gastrectomy (antrectomy) with enteric continuity restored by an end-to-end anastomosis between the remnant stomach and duodenum—a gastroduodenostomy [2]. The entire incised end of the stomach may be used for anastomosis (Polya procedure) or only a part of the lumen may be used and the remainder closed (Hofmeister modification) (Figs. 2 and 3). In Billroth 2 surgery, the anastomosis is made between the remnant stomach and the jejunum—a gastrojejunostomy—with the duodenal stump closed [2]. Variations may include the type of anastomosis (Polya or Hofmeister modification), the type of gastrojejunostomy (Roux-en-Y or Loop-type) and the placement of anastomotic loop (ante- or retro-colic), depending on the clinical situation and surgeon’s preference. A retrocolic gastrojejunostomy is the preferred method, as it creates a shorter afferent loop, resulting in less postoperative retrostomal herniation and better nutritional status compared with the antecolic gastrojejunostomy (Figs. 4 and 5). A Roux-en-Y anastomosis typically implies a proximal segment of small bowel and a distal segment of small bowel (which is often a blind end) anastomosed together, representing the two upper limbs of the “Y” and the distal small bowel (beyond the anastomosis) representing the lower part of the “Y”.
Illustration of Billroth type I procedure. a A partial gastrectomy (antrectomy) (dashed lines) is performed. b An end-to-end anastomosis is created between the remnant stomach (S) and duodenum (D), a gastroduodenostomy (arrow). The illustration also depicts the Hofmeister modification (dashed arrow)
Billroth type 1 procedure. a Axial CT image with oral contrast in a 74-year-old woman with Billroth 1 and vagotomy for peptic ulcer disease shows metallic sutures (black arrow) at the end-to-end anastomosis between the remnant stomach (S) and duodenum (gastroduodenostomy). b Coronal CT image showing the gastroduodenostomy; duodenum (D) and stomach (S). Note the staples representing the vagotomy (arrowheads in a and b) and pneumoperitoneum and pneumoretroperitoneum (white arrow, a), (the patient presented with perforated sigmoid diverticulitis, not shown)
Illustration of Billroth type 2 procedure. a A side-to-side anastomosis between the two jejunal limbs may be created in a Billlroth 2 (arrowhead). b An anastomosis is created between the remnant stomach (S) and jejunum (J), a gastrojejunostomy (dashed arrow) and the duodenal stump (black arrow) is closed. A afferent limb, E efferent limb, white arrow Hofmeister modification
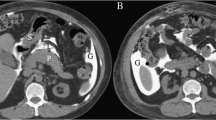
Billroth type 2 procedure. Axial contrast-enhanced CT images of a 75-year-old man with Billroth II antecolic loop gastrojejunostomy for a gastric ulcer. Images show a remnant stomach (S), and metallic suture material on the lesser curvature side indicating a Hofmeister modification (white arrow, a). The gastrojejunal anastomosis (dashed arrow, b), and the two jejunal limbs (A afferent limb, E efferent limb) are identified. The blind end of the afferent limb (A) is continuous with the third portion of the duodenum and the efferent limb (E) is brought up to the anastomosis anterior to the transverse colon (C). The duodenal stump (white arrow, c) is also noted
Bariatric surgery
Bariatric surgeries are a vast subject and may be classified as restrictive, malabsorptive or mixed. Restrictive procedures involve the creation of a small gastric pouch which induces weight loss through satiety. Examples include adjustable gastric banding and vertical-banded gastroplasty. Malabsorptive procedures bypass portions of the small bowel, limiting the digestive processes and absorption to a short segment of ileum. Examples of malabsorptive surgeries include the jejuno-ileal bypass, abandoned because of the severe malnutrition it induces, and biliopancreatic diversion with or without a duodenal switch, which has been slow to gain popularity. Mixed procedures are a combination of both [3].
The Roux-en-Y gastric bypass (RYGBP) is a common procedure that combines restrictive (small gastric pouch) and malabsorptive properties (Roux limb) [4, 5]. The standard set-up involves four steps: (1) the stomach is divided, thus creating a small gastric pouch and an excluded stomach; (2) the jejunum is divided distal to the ligament of Treitz by creating a bilio-pancreatic limb and a Roux limb; (3) the Roux limb is joined to the bilio-pancreatic limb via a side-to-side jejuno-jejunal anastomosis. Small bowel distal to this anastomosis is called the “common channel”. (4) The proximal Roux limb is brought up to the gastric pouch to form the gastro-jejunal anastomosis. The position of the Roux limb with regards to the transverse colon is variable: it may be antecolic (superior and anterior to the transverse colon) or retrocolic (tunneling through a surgically created defect in the transverse mesocolon). Its position with regards to the stomach may be antegastric (anterior to excluded stomach) or retrogastric (behind excluded stomach) (Figs. 6 and 7).
Illustration of Roux-en-Y gastric bypass (RYGBP) procedure. Gastrojejunal anastomosis (dashed black arrow) between gastric pouch (GP) and the Roux limb (R). In this case, the Roux limb is antegastric. S remnant stomach, BPL bilio-pancreatic limb, black arrow side-to-side jejuno-jejunal anastomosis, CC common channel distal to jejuno-jejunal anastomosis
Roux-en-Y gastric bypass (RYGBP) procedure. Axial contrast-enhanced CT images of a 44-year-old man who underwent a Roux-en-Y gastric bypass for morbid obesity. Images show a gastric pouch (dashed white arrow, a), an end-to-side gastrojejunal anastomosis (dashed black arrow, a) and the surgically separated remnant stomach (arrowhead, a). The Roux limb (white arrow, b) is in a retrocolic-antegastric position; colon (C, in b). The jejuno-jejunal anastomosis is identified (black arrow, c)
Pancreas
Whipple
Indications include the treatment of pancreatic cancer, periampullary neoplasms, symptomatic chronic pancreatitis and pancreatic trauma. Classically, it entails radical resection of the pancreatic head, duodenum and gastric antrum [6–8]. This procedure requires three anastomoses: (1) a gastrojejunostomy, or when feasible, a pylorus-preserving duodenojejunostomy, (2) a choledochojejunostomy and (3) an end-to-side pancreaticojejunostomy. Normal post-operative findings include: transient fluid collections (resolution within 4–6 weeks after surgery), perivascular cuffing, reactive lymphadenopathy and pneumobilia (Figs. 8 and 9).
Illustration of Whipple procedure. Depending on the extent of disease, the Whipple procedure can be modified. a When feasible, a pylorus-preserving procedure is performed, where the duodenum is not resected, creating a duodenojejunostomy rather than the classic gastrojejunostomy (as illustrated in b). b Classic Whipple anatomy with gastrojejunostomy (dashed black arrow), choledochojejunostomy (black arrow), and an end-to-side pancreaticojejunostomy (white arrow). S stomach, J jejunum
Whipple procedure. a Axial contrast-enhanced CT image 10 days post-operative of a 60-year-old man who underwent a Whipple procedure for a mixed intraductal papillary mucinous tumor (IPMT) shows the distal common bile duct approaching the choledochojejunostomy (black arrow) and a mildly thickened pancreaticojejunostomy (white arrow) with some mild adjacent fat stranding (normal finding in the early post-operative period). Atrophy of the residual pancreatic gland. Follow-up CT scan, obtained 1 month later: (b) axial CT image depicting the gastrojejunostomy (black dashed arrow) and resolution of the pancreaticojejunostomy edema (white arrow). Note the dilation of the main pancreatic duct, which was present pre-operatively. c, d Coronal reformatted CT images showing the choledochojejunostomy (black arrow) and pancreaticojejunostomy (white arrow
Puestow
This procedure is indicated in patients with intractable chronic pancreatitis associated with pancreatic duct dilation beyond 6 mm [9]. The pancreas is filleted, incised longitudinally, exposing the main pancreatic duct from neck to tail with extraction of ductal calculi. A Roux-en-Y loop of jejunum is brought up to create a lateral side-to-side pancreaticojejunostomy with direct drainage of the pancreatic duct into the jejunum (Figs. 10 and 11).
Puestow procedure. Axial CT image of normal anatomy post-Puestow procedure for chronic pancreatitis of a 60-year-old man showing the Roux-en-Y jejunal loop anastomosed to the anterior aspect of the pancreatic head (black arrow). Note the pancreatic calcifications consistent with chronic pancreatitis
Small bowel
Ileal pouch-anal anastomosis
The ileal pouch-anal anastomosis (IPAA) preserves fecal continence in patients who require a proctocolectomy for chronic ulcerative colitis and familial adenomatous polyposis [10, 11]. Multiple configurations exist for the ileal reservoir, with the J pouch being the most common. The surgical steps include: (1) performing a colectomy and proctectomy; (2) stapling the distal end of ileum to form a blind end “stump”; (3) folding the distal ileum on itself and creating an apical enterotomy; (4) creating a side-to-side anastomosis by cutting the ileal walls centrally and connecting the two ileal loops to form a large pouch with two parallel staple lines; (5) creating an anastomosis between the anus and enterotomy with a circular stapling device passed trans-anally; (6) performing a temporary loop ileostomy which is usually reversed 6–12 weeks later. Normal CT findings include: a staple line at the blind end of the distal ileum, parallel staple lines along the ileal pouch and a circular staple line at the ileal pouch-anal anastomosis within the sling of the levator ani muscles (Figs. 12 and 13).
Ileal pouch-anal anastomosis (IPAA). Axial, contrast-enhanced CT images of a 50-year-old woman with proctocolectomy and ileal pouch-anal anastomosis for ulcerative colitis showing: a a circular staple line (black arrow) at the level of the pouch-anal anastomosis within the sling of levator ani muscles (white arrowheads); b ileal pouch with two rows of parallel suture lines (white arrows) and staple line at blind end of ileum (dashed black arrow) are identified
Colorectal
Hartmann procedure
The Hartmann procedure is indicated in patients with complicated diverticulitis, obstructive or perforated sigmoid cancer or penetrating trauma operated on an emergent basis [12, 13]. It is performed instead of a primary re-anastomosis because of the risk of anastomotic rupture secondary to inadequate bowel preparation and/or complicating peritonitis. After a partial colectomy or sigmoidectomy, it involves the creation of a temporary diverting colostomy and a blind-ending rectal or colonic stump closed by suture (Hartmann’s pouch). The colostomy can then be taken down in the second stage of the procedure, 4–6 months later, after the peritoneal inflammation subsides (Figs. 14 and 15).
Lower anterior resection
Lower anterior resection is an anal-sparing procedure indicated for lesions of the recto-sigmoid junction and proximal rectum that are over 5 cm from the anal border [13]. It involves resection of the distal left colon, sigmoid and proximal rectum with a low colo-rectal anastomosis or resection including the entire rectum with a colo-anal anastomosis (Figs. 16 and 17).
Lower anterior resection. a Axial contrast-enhanced CT images of a 59-year-old man with lower anterior resection for rectal cancer 13 cm from the anal border shows the low colo-rectal anastomosis (white arrow). There is preservation of the distal rectum (with rectal tube) in the pre-coccygeal area (b). The anus is also spared (not shown)
Abdomino-perineal resection
This resection is indicated for lesions of the distal rectum, less than 5 cm from the anal border, and involves resection of the distal portion of the descending colon, sigmoid, rectum and entire anus (anus, sphincters, levator ani muscles) with creation of a permanent end colostomy (Figs. 18 and 19) [13].
Abdomino-perineal resection. Axial contrast-enhanced CT images of a 78-year-old woman with abdomino-perineal resection for cancer of the lower rectum located 2.5 cm from the anal border. Note the absence of rectum in the pre-coccygeal space in (a). The anus, anal sphincters and levator ani muscles were all resected and patient had a permanent end colostomy in left iliac fossa (white arrow)
Conclusion
The post-operative abdomen can be complex and challenging. This pictorial essay describes the appearance of a few selected common gastrointestinal surgeries. Familiarity with normal post-operative anatomy can aid in the detection of complications.
References
Nandakumar G, Stein SL, Michelassi F (2009) Anastomoses of the lower intestinal tract. Nat Rev Gastroenterol Hepatol 6:709–716
Kim KW, Choi BI, Han JK (2002) Postoperative anatomic and pathologic findings at CT following gastrectomy. Radiographics 22:323–336
Chandler RC, Srinivas G, Chintapalli KN, Schwesinger WH, Prasad SR (2008) Imaging in bariatric surgery: a guide to postsurgical anatomy and common complications. AJR Am J Roentgenol 190:122–135
Scheirey CD, Scholz FJ, Shah PC, Brams DM, Wong BB, Pedrosa M (2006) Radiology of the laparoscopic Roux-en-Y gastric bypass procedure: conceptualization and precise interpretation of results. Radiographics 26:1355–1371
Yu J, Turner MA, Cho SR (2004) Normal anatomy and complications after gastric bypass surgery: helical CT findings. Radiology 231:753–760
Lepanto L, Gianfelice D, Déry R, Dagenais M, Lapointe R, Roy A (1994) Postoperative changes, complications, and recurrent disease after Whipple’s operation: CT features. AJR Am J Roentgenol 163:841–846
Johnson PT, Curry CA, Urban BA, Fishman EK (2002) Spiral CT following the Whipple procedure: distinguishing normal postoperative findings from complications. J Comput Assist Tomogr 26:956–961
Mortelé KJ, Lemmerling M, de Hemptinne B, De Vos M, De Bock G, Kunnen M (2000) Postoperative findings following the Whipple procedure: determination of prevalence and morphologic abdominal CT features. Eur Radiol 10:123–128
Sandrasegaran K, Maglinte DD, Howard TJ, Lappas JC (2005) Surgery for chronic pancreatitis: cross-sectional imaging of postoperative anatomy and complications. AJR Am J Roentgenol 184:1118–1127
Alfisher MM, Scholz FJ, Roberts PL, Counihan T (1997) Radiology of ileal pouch-anal anastomosis: normal findings, examination pitfalls, and complications. Radiographics 17:81–98
Broder JC, Tkacz JN, Anderson SW, Soto JA, Gupta A (2010) Ileal pouch-anal anastomosis surgery: imaging and intervention for post-operative complications. Radiographics 30:221–233
Cherukuri R, Levine MS, Maki DD, Rubesin SE, Laufer I, Rosato EF (1998) Hartmann’s pouch: radiographic evaluation of postoperative findings. AJR Am J Roentgenol 171:1577–1582
Scardapane A, Brindicci D, Fracella MR, Angelelli G (2005) Post colon surgery complications: imaging findings. Eur J Radio 53:397–409
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Terrone, D.G., Lepanto, L., Billiard, JS. et al. A primer to common major gastrointestinal post-surgical anatomy on CT—a pictorial review. Insights Imaging 2, 631–638 (2011). https://doi.org/10.1007/s13244-011-0121-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13244-011-0121-4