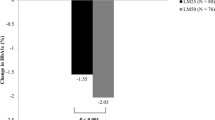
Abstract
Introduction
Increased postprandial glucose (PPG) is associated with high glycated haemoglobin levels and is an independent risk factor for cardiovascular diseases. The aim of this study was to compare PPG increments in Asian versus non-Asian adults with type 2 diabetes (T2D), who were insulin-naïve or insulin-experienced, from the phase 3 insulin degludec/insulin aspart (IDegAsp) clinical trials.
Methods
This was a post hoc analysis of data from 13 phase 3, randomised, parallel-group, open-label IDegAsp trials in patients with T2D. The pooled baseline clinical data were analysed for insulin-naïve and insulin-experienced groups; and each group was split into subgroups of Asian and non-Asian patients, respectively, and analysed accordingly. Baseline self-monitored blood glucose (SMBG) values at breakfast, lunch and the evening meal (before and 90 min after each meal) were used to assess PPG increments. The estimated differences in baseline SMBG increment between the Asian and non-Asian subgroups were analysed.
Results
Clinical data from 4750 participants (insulin-naïve, n = 1495; insulin-experienced, n = 3255) were evaluated. In the insulin-naïve group, the postprandial SMBG increment was significantly greater in the Asian versus the non-Asian subgroup at breakfast (estimated difference 28.67 mg/dL, 95% confidence interval [CI] 18.35, 38.99; p < 0.0001), lunch (17.34 mg/dL, 95% CI 6.47, 28.21; p = 0.0018) and the evening meal (16.19 mg/dL, 95% CI 5.04, 27.34; p = 0.0045). In the insulin-experienced group, the postprandial SMBG increment was significantly greater in the Asian versus non-Asian subgroup at breakfast (estimated difference 13.81 mg/dL, 95% CI 9.19, 18.44; p < 0.0001) and lunch (29.18 mg/dL, 95% CI 24.22, 34.14; p < 0.0001), but not significantly different at the evening meal.
Conclusion
In this post hoc analysis, baseline PPG increments were significantly greater in Asian participants with T2D than in their non-Asian counterparts at all mealtimes, with the exception of the evening meal in insulin-experienced participants. Asian adults with T2D may benefit from the use of regimens that control PPG excursions.
Clinical Trial Numbers
NCT02762578, NCT01814137, NCT01513590, NCT01009580, NCT01713530, NCT02648217, NCT01045447, NCT01365507, NCT01045707, NCT01272193, NCT01059812, NCT01680341, NCT02906917.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Differences in genetic factors, ethnicity, culture and diet can influence the insulin requirement and therefore alter the risk of postprandial hyperglycaemia in specific populations. |
This post hoc analysis of 13 randomised clinical trials compared the risk of postprandial hyperglycaemia in insulin-naïve or insulin-experienced Asian and non-Asian adults with type 2 diabetes (T2D). |
What was learned from this study? |
Self-monitored blood glucose-based postprandial glucose excursions were statistically significantly greater in Asian versus non-Asian adults with T2D at all mealtimes, with the exception of the evening meal in insulin-experienced adults. |
Intensified insulin treatment using premixed or novel insulin co-formulations, such as IDegAsp, which target both fasting and postprandial hyperglycaemia, might be beneficial in Asian populations with T2D. |
Introduction
The prevalence of type 2 diabetes (T2D) has increased worldwide in the last three decades [1] and is predicted to continue to rise. Two regions in which this increase is particularly notable are South-East Asia and the Western Pacific [2]. It has estimated that in 2019 there were approximately 88 million adults (aged 20–79 years) with diabetes in South-East Asia and 163 million in the Western Pacific region, and that by 2045 these figures are estimated to increase to 153 million and 212 million, respectively [2, 3]. China is the country with the highest prevalence of diabetes, followed by India, with Pakistan, Bangladesh and Indonesia also among the top ten countries globally in terms of diabetes prevalence [2]. The increasing prevalence of diabetes is also a significant concern among non-Asian countries [2], and a better understanding of the effect of race and ethnicity on glycaemic variables to effectively manage T2D is required [4].
Glycaemic fluctuations or acute changes in blood glucose levels should be addressed in the management of T2D, as these play a vital role in the development of vascular complications [5]. An increase in postprandial glucose (PPG) levels is considered to be a major contributor to higher glycated haemoglobin (HbA1c) levels and is an independent risk factor for cardiovascular disease [6]. Self-monitored blood glucose (SMBG) is a convenient and simple method for assessing glycaemic control that can be used in addition to HbA1c [7], and can be used to provide information on glucose indices in response to exercise, meals, daily events, medications and illness.
Racial and cultural differences, such as dietary habits, greatly impact glycaemic indices and lead to differences in glycaemic load [8]. The Asian population (Asian Indian, South-East Asian and East Asian) tends to have higher PPG and lower insulin sensitivity compared with European and Arabic Caucasian populations, when consuming the same food [9]. Hence, it is important to assess PPG control and PPG excursions in people with T2D from both Asian and non-Asian populations.
The American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) recommend the use of a basal insulin, with further intensification using bolus insulin if glycaemic targets are not met [10]. The International Diabetes Federation (IDF) recommends the use of specific PPG-lowering agents, such as rapid-acting and biphasic (premixed) human insulins/analogues, among other pharmacologic agents [11]. Guidelines for the management of T2D vary among countries. Several non-Western countries have guidelines that are generally consistent with international guidelines [12, 13]; however, these differ according to the respective populations. A majority of these guidelines make provision for PPG control while recommending treatment options; the addition of a rapid-acting insulin to a basal insulin regimen is the most common recommendation [14].
Basal insulin, when administered alone, is often associated with slow initiation and intensification [15]. In addition, intensification of basal insulin without consideration of fast-acting/bolus options can contribute to poor glycaemic control in the long term [16]. Separate administration of a bolus insulin may be considered inconvenient and may negatively impact treatment adherence. Insulin degludec/insulin aspart (IDegAsp; 70% insulin degludec [degludec] and 30% insulin aspart [IAsp]) is a fixed-ratio co-formulation of degludec, a long-acting basal insulin, and IAsp, a rapid-acting insulin targeting PPG [17]. The basal degludec component provides a stable and long-lasting glucose-lowering effect while the bolus IAsp component provides rapid-onset and a peak glucose-lowering effect [18].
The safety and efficacy of IDegAsp have been evaluated over a series of phase 3 clinical trials across several countries involving Asian and non-Asian participants [19,20,21,22,23,24,25,26,27,28,29,30,31]. These trials recorded various glycaemic parameters, including fasting plasma glucose (FPG), SMBG and PPG increment. Pooling data from the phase 3 clinical trial program provided the opportunity to evaluate the extent of meal-related dysglycaemia in Asian versus non-Asian participants with T2D.
This post hoc analysis compared PPG increments in Asian versus non-Asian adults with T2D, who were insulin-naïve or insulin-experienced, using pooled baseline data from IDegAsp phase 3 clinical trials. The results obtained may help assess the probable impact of ethnicity on treatment outcomes and the need for customising treatment approaches based on PPG excursions.
Methods
Study Design
This was a post hoc analysis of pooled baseline data from 13 IDegAsp phase 3 clinical trials [19,20,21,22,23,24,25,26,27,28,29,30,31]. These phase 3, randomised, parallel-group, open-label trials were conducted between 2010 and 2018 and compared the safety and efficacy of IDegAsp with active comparators, such as biphasic insulin aspart 30, insulin glargine U100 (glargine), degludec + IAsp basal–bolus therapy (BBT), glargine + IAsp BBT and other oral antidiabetic drugs (OADs) in adults (aged ≥ 18 years) with T2D. The trial designs have been reported previously and are summarised in Electronic Supplementary Material (ESM) Table S1.
Baseline clinical data were pooled from insulin-naïve participants of four trials [19,20,21,22], while baseline data related to insulin-experienced participants were pooled from nine separate trials [23,24,25,26,27,28,29,30,31]. The pooled baseline data included participants with T2D from different countries who were categorised into Asian and non-Asian, based predominantly on geographical location. Data specific to ethnicity were not recorded. For the purpose of this analysis, countries in the Asian group included China, Hong Kong, India, Japan, Korea, Lebanon, Malaysia, Thailand and Taiwan. Countries in the non-Asian group included (predominantly Caucasian participants) Algeria, Australia, Austria, Bulgaria, Croatia, Czech Republic, Denmark, Finland, France, Mexico, Norway, Poland, Romania, Russian Federation, Serbia, Slovakia, South Africa, Spain, Sweden, Turkey, Ukraine and the USA.
Outcomes
The SMBG values and demographic information (age, sex, blood glucose parameters, duration of diabetes and ongoing antidiabetic medications) from the pooled baseline data were reported for insulin-naïve and insulin-experienced groups in the current analysis. The baseline SMBG values from the pooled baseline data were analysed for insulin-naïve and insulin-experienced groups; each group was also analysed by Asian and non-Asian subgroup. SMBG increments were calculated for breakfast, lunch and dinner (evening meal) using values from before and 90 min after each meal. Baseline FPG was also assessed.
Statistical Analyses
Statistical analyses were carried out for SMBG values only (SAS 9.4M5 software with encoding Latin1; Copenhagen, Denmark). Baseline SMBG increments were analysed using a linear mixed model adjusted for age, sex, duration of diabetes and ongoing antidiabetic medications at baseline. Age and duration of diabetes were used as covariates, while sex and ongoing antidiabetic medications at baseline were used as factors. An additional analysis in which baseline body mass index (BMI) was used as a covariate (in addition to age and duration of diabetes) was also performed. After fitting the model, the estimated differences in SMBG increment between the Asian and non-Asian groups were assessed with 95% confidence intervals (CIs). Cases with data missing at specific time points were not included in the analysis of the respective time points.
Ethics Statement
The individual trials considered for this post hoc analysis were approved by health authorities according to the corresponding local regulations and by the local independent ethics committees. These trials were conducted in accordance with the Declaration of Helsinki [32] and Good Clinical Practice Guidelines. All participants provided written informed consent prior to enrolment into the respective trials [19,20,21,22,23,24,25,26,27,28,29,30,31].
Results
Demographics
Overall, data from 1495 participants in the insulin-naïve group and 3255 participants in the insulin-experienced group were included in the analysis (Table 1). In the insulin-naïve group, the majority of participants were non-Asian (68.2%), whereas the distribution was relatively even in the insulin-experienced group. Country-specific data are presented in ESM Table S2. The majority of participants in the Asian subgroup were from Japan (insulin-naïve, n = 296; insulin-experienced, n = 178) or China (insulin-naïve, n = 0; insulin-experienced, n = 543), and a majority of the non-Asian participants were from the USA (insulin-naïve, n = 291; insulin-experienced, n = 617). Females comprised 41% of Asian participants and 50% of non-Asian participants in the insulin-naïve group, and 47% of Asian participants and 45% of non-Asian participants in the insulin-experienced group.
Baseline Characteristics
Baseline characteristics, including age and HbA1c, were generally comparable among Asian and non-Asian participants in the insulin-naïve and insulin-experienced groups, respectively (Table 1). However, weight and BMI were numerically smaller in the Asian subgroup compared with the non-Asian subgroup in both the insulin-naïve and the insulin-experienced groups.
Pooled baseline data on the use of antidiabetic agents indicated variations between Asian and non-Asian subgroups (Table 1). In the insulin-naïve group, fewer participants in the Asian subgroup used metformin plus a sulfonylurea (SU) (0.0%) or metformin monotherapy (1.7%) than in the non-Asian subgroup (28.3% and 12.8%, respectively), whereas more than half of participants in the Asian subgroup (52.6%), but no participants in the non-Asian subgroup (0.0%), used SU and/or glinides Moreover, in the insulin-naïve group, a smaller proportion of participants in the Asian subgroup used metformin in combination with SU or with glinides versus the non-Asian subgroup (19.2 vs. 35.7%).
A greater proportion of the insulin-experienced participants in the Asian subgroup used premix/self-mix insulin with or without metformin (25.7 and 20.9%, respectively) compared with the non-Asian subgroup (0.0% for both). However, the use of basal insulin (alone and in combinations) was lower in Asian participants compared with non-Asian participants (27.4 vs. 47.1%). Furthermore, the use of once-daily basal insulin was also lower in the Asian subgroup (2.5%) versus the non-Asian subgroup (23.5%).
Postprandial Glucose Excursions
The PPG increment based on baseline SMBG was higher in Asian versus non-Asian participants for all meals (breakfast, lunch, and evening meal) in both insulin-naïve and insulin-experienced subgroups (Table 2; ESM Table S3).
In the insulin-naïve group, the estimated difference in the PPG increment (SMBG) was significantly higher in the Asian versus non-Asian subgroup at breakfast (estimated difference 28.67 mg/dL, 95% CI 18.35, 38.99; p < 0.0001), lunch (17.34 mg/dL, 95% CI 6.47, 28.21; p = 0.0018) and the evening meal (16.19 mg/dL, 95% CI 5.04, 27.34; p = 0.0045). The additional analysis, adjusting for baseline BMI, gave similar findings, although the differences between the Asian and non-Asian subgroups in PPG increment were slightly diminished, and statistical significance was not met for the lunchtime increment (estimated difference 11.05, 95% CI –0.62, 22.72; p = 0.0635) (ESM Table S3).
In the insulin-experienced group, the postprandial PPG increment (SMBG) was significantly greater in the Asian versus non-Asian subgroup at breakfast (estimated difference 13.81 mg/dL, 95% CI 9.19, 18.44; p < 0.0001) and lunch (29.18 mg/dL, 95% CI 24.22, 34.14; p < 0.0001), but not significantly different at the evening meal (estimated difference 3.71 mg/dL, 95% CI –1.59, 9.01; p = 0.1695). The additional analysis, adjusting for baseline BMI, again produced similar results, with the differences between the Asian and non-Asian subgroups in PPG increment slightly diminished, but remaining statistically significant at breakfast and lunch (ESM Table S3).
Fasting Plasma Glucose Levels
In the insulin-naïve group, baseline FPG was 159.29 mg/dL in the Asian subgroup and 183.58 mg/dL in the non-Asian subgroup (Table 1). The corresponding values for baseline FPG in the insulin-experienced group were 154.12 mg/dL and 157.48 mg/dL in the Asian and non-Asian subgroups, respectively. No further analyses were conducted for this parameter.
Discussion
In this post hoc analysis of baseline data from 13 randomised controlled trials, PPG increments based on SMBG measurements were generally greater in Asian versus non-Asian participants with T2D. The estimated differences in PPG increment were statistically significant at all mealtimes for both insulin-naïve and insulin-experienced participants, except for at the evening meal in insulin-experienced participants. As well as being an independent risk factor for cardiovascular disease, PPG is a significant contributor to HbA1c levels [6]. HbA1c is strongly correlated with the risk of diabetes complications [33] and so reducing PPG excursions is an important part of diabetes management, for both Asian and non-Asian adults with T2D [11]. Modern insulin treatments that include the use of fixed-ratio co-formulations of basal and bolus insulin may be particularly beneficial to Asian adults with T2D in controlling PPG excursions.
Differences in genetic factors, ethnicity, culture and diet can all impact insulin sensitivity and PPG excursions [8]. In people of East Asian descent, T2D tends to have an earlier onset and starts to develop at a lower mean BMI than in people of European descent [34]. Furthermore, Asian individuals, particularly those from China, South Asia and East Asia, tend to have higher amounts of visceral adiposity compared with Caucasian individuals, which is strongly associated with the risk of diabetes, at any BMI [34,35,36,37]. Given the increased risks of cardiometabolic disease at lower BMI values in Asian populations, the World Health Organization (WHO) has suggested that lower cutoff values could be used for the definitions of overweight and obesity in Asian cohorts [38]. Although these cutoff points are not routinely used in mixed-cohort clinical studies, there are increasing calls for the definition of overweight to be lowered to a BMI of ≥ 23 kg/m2, at least in the screening for Asian people at risk of T2D [39, 40]. In our study, we adjusted the data for the potential confounding influence of BMI in an additional analysis. In this analysis, the difference in PPG increment between Asian and non-Asian patients was diminished slightly, although the trends were unaffected. It should be noted, however, that we did not catgeorise BMI differently for Asian and non-Asian participants.
T2D in Asian individuals is characterised by early beta-cell dysfunction, leading to insulin resistance and the need for early insulin treatment [34]. Early beta-cell dysfunction in Asian individuals with T2D is also likely to be a factor influencing greater PPG increments in this patient population versus their non-Asian counterparts [41,42,43,44,45]. Dietary habits, including the proportion of carbohydrates consumed per meal, overall amount of food consumed and cooking methods can also influence PPG excursions. Over the past two decades, there has been a shift in Asian countries towards higher fat intake with lower carbohydrate content in the diet [46]. White rice, a staple food, has a high glycaemic load and has been positively associated with the risk of developing T2D, particularly in East Asian (Chinese and Japanese) populations [47]. Overall, there are several factors that are likely to have resulted in the increasing incidence of T2D in non-Western countries in recent decades. Due to this increase, there is a need to assess differences in meal-related glycaemic fluctuations between different populations in order to guide effective management of T2D in Asian populations.
The greater risk of PPG excursions reported in this post hoc analysis in Asian versus non-Asian participants highlights the importance of treatment regimens that target meal-related glucose excursions via a prandial insulin component in Asian adults with T2D. In a retrospective pooled analysis of injection-naïve adults with T2D, baseline SMBG excursions were significantly higher for East Asian patients than for Caucasian patients at breakfast (72.5 vs. 46.6 mg/dL), lunch (60.7 vs. 25.7 mg/dL) and dinner (56.9 vs. 31.3 mg/dL) (p < 0.001 adjusted analyses) [37]. These values are comparable with those in the current analysis.
Compared with non-Asian participants, Asian participants had a statistically significantly greater baseline PPG increment at all meal times in the insulin-naïve subgroup. In this subgroup, the difference in baseline FPG values between Asian and non-Asian subgroups was similar to the difference in baseline PPG increments between subgroups. Therefore, the lower baseline FPG in the Asian versus the non-Asian insulin-naïve subgroups may have been a contributing factor to the greater absolute increase in PPG (SMBG) in the Asian subgroup.
Although speculative, the observed differences in FPG and PPG increment between the subgroups may have been influenced by the different treatments used at baseline in the Asian and non-Asian populations. However, the increased used of SU and/or glinides in Asian participants would be expected to better target PPG, and yet higher PPG levels were experienced in Asian participants. Similarly, in the insulin-experienced group, the Asian subgroup tended to use prandial as well as basal insulin (either in the form of premix or basal/bolus regimens) with or without metformin, whereas non-Asian participants tended to use once-daily basal insulin. Baseline FPG was similar between the Asian and non-Asian insulin-experienced subgroups, but again PPG was increased in the former despite the higher rate of use of prandial insulin. Differences in PPG increment may also be due to higher levels of insulin resistance in Asian populations than non-Asian populations [34]. In a recent consensus report, IDegAsp was recommended as an initial insulin treatment for individuals with T2D in whom postprandial hyperglycaemia is a concern [16]. Furthermore, intensification to once-daily IDegAsp is recommended for those with a low BMI and beta-cell insufficiency, which may better target the postprandial dysglycaemia of the Asian population [16]. However, SUs may need to be discontinued before initiating IDegAsp, and this is clinically relevant given the high level of SU use in the insulin-naïve Asian population [16].
Differences in glycaemic responses between Asian and non-Asian populations have been previously reported [37, 48]. In a separate study, Venn et al. compared postprandial capillary blood glucose concentrations between Asian and Caucasian adults by calculating the mean difference in 2-h incremental areas under the curve (iAUCs), following the consumption of a glucose beverage and a breakfast cereal [48]. The mean difference in iAUC was 29% (95% CI 10, 51) and 63% (95% CI 32, 102) higher in the Asian compared with the Caucasian group, following the consumption of a glucose beverage and a breakfast cereal, respectively [48].
There are several limitations to this post hoc analysis. Specific dietary information, including carbohydrate content and glycaemic indices of the main meals eaten in different countries, was not recorded, and these differences may have contributed to the increased PPG increment observed in the Asian versus non-Asian subgroups. The potential association between PPG excursions and the timing of prandial insulin administration relative to meals was not evaluated. The trials from which the data were used in this analysis were not primarily powered to assess PPG differences between populations. A larger sample size of Asian participants may be required to study PPG excursions in further detail. Importantly, strict ethnicity-based segregation of participants was not performed.
The strengths of this analysis included the large data set used. This was a pooled analysis of baseline data from 13 multinational randomised controlled trials. This pooled analysis adds to the body of evidence showing a trend for greater risk of PPG excursions in Asian versus non-Asian populations.
Conclusions
Control of PPG excursions may be an unmet need of diabetes management in Asian people. In this post hoc analysis of pooled baseline data from randomised controlled trials, PPG increments based on SMBG measurements were greater in Asian versus non-Asian adults with T2D. Asian people with T2D may therefore benefit from a therapy regimen that includes an element designed to limit PPG excursions (as well as a basal insulin), as they tend to show greater PPG excursions than non-Asian people.
References
Khan MAB, Hashim MJ, King JK, Govender RD, Mustafa H, Al KJ. Epidemiology of type 2 diabetes—global burden of disease and forecasted trends. J Epidemiol Glob Health. 2020;10:107–11.
International Diabetes Federation (IDF). IDF diabetes atlas, 9th edition 2019. 2019. https://www.idf.org/aboutdiabetes/what-is-diabetes/facts-figures.html. Accessed 25 Feb 2021.
Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. 2019;157:107843.
McBean AM, Li S, Gilbertson DT, Collins AJ. Differences in diabetes prevalence, incidence, and mortality among the elderly of four racial/ethnic groups: whites, blacks, hispanics, and asians. Diabetes Care. 2004;27:2317–24.
Dandona P. Minimizing glycemic fluctuations in patients with type 2 diabetes: approaches and importance. Diabetes Technol Ther. 2017;19:498–506.
Hershon KS, Hirsch BR, Odugbesan O. Importance of postprandial glucose in relation to A1C and cardiovascular disease. Clin Diabetes. 2019;37:250–9.
Parsons SN, Luzio SD, Harvey JN, et al. Effect of structured self-monitoring of blood glucose, with and without additional TeleCare support, on overall glycaemic control in non-insulin treated type 2 diabetes: the SMBG Study, a 12-month randomized controlled trial. Diabet Med. 2019;36:578–90.
Hu Y, Block G, Sternfeld B, Sowers M. Dietary glycemic load, glycemic index, and associated factors in a multiethnic cohort of midlife women. J Am Coll Nutr. 2009;28:636–47.
Dickinson S, Colagiuri S, Faramus E, Petocz P, Brand-Miller JC. Postprandial hyperglycemia and insulin sensitivity differ among lean young adults of different ethnicities. J Nutr. 2002;132:2574–9.
Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2018;41:2669–701.
International Diabetes Federation Guideline Development Group. Guideline for management of postmeal glucose in diabetes. Diabetes Res Clin Pract. 2014;103:256–68.
Araki E, Goto A, Kondo T, et al. Japanese clinical practice guideline for diabetes 2019. Diabetol Int. 2020;11:165–223.
Weng J, Ji L, Jia W, et al. Standards of care for type 2 diabetes in China. Diabetes Metab Res Rev. 2016;32:442–58.
Home P, Haddad J, Latif ZA, et al. Comparison of national/regional diabetes guidelines for the management of blood glucose control in non-Western countries. Diabetes Ther. 2013;4:91–102.
Dale J, Martin S, Gadsby R. Insulin initiation in primary care for patients with type 2 diabetes: 3-year follow-up study. Prim Care Diabetes. 2010;4:85–9.
Mehta R, Chen R, Hirose T, et al. Practical use of insulin degludec/insulin aspart in a multinational setting: beyond the guidelines. Diabetes Obes Metab. 2020;22:1961–75.
Novo Nordisk A/S. Ryzodeg summary of product characteristics. https://www.ema.europa.eu/en/documents/product-information/ryzodeg-epar-product-information_en.pdf. Accessed 25 Feb 2021.
Haahr H, Fita EG, Heise T. A review of insulin degludec/insulin aspart: pharmacokinetic and pharmacodynamic properties and their implications in clinical use. Clin Pharmacokinet. 2017;56:339–54.
Franek E, Haluzik M, Canecki Varzic S, et al. Twice-daily insulin degludec/insulin aspart provides superior fasting plasma glucose control and a reduced rate of hypoglycaemia compared with biphasic insulin aspart 30 in insulin-naive adults with type 2 diabetes. Diabetic Med. 2016;33:497–505.
Park SW, Bebakar WM, Hernandez PG, Macura S, Hersløv ML, de la Rosa R. Insulin degludec/insulin aspart once daily in type 2 diabetes: a comparison of simple or stepwise titration algorithms (BOOST(®): SIMPLE USE). Diabetic Med. 2017;34:174–9.
Kumar A, Franek E, Wise J, Niemeyer M, Mersebach H, Simo R. Efficacy and safety of once-daily insulin degludec/insulin aspart versus insulin glargine (U100) for 52 weeks in insulin-naive patients with type 2 diabetes: a randomized controlled trial. PLoS One. 2016;11:e0163350.
Onishi Y, Ono Y, Rabøl R, Endahl L, Nakamura S. Superior glycaemic control with once-daily insulin degludec/insulin aspart versus insulin glargine in Japanese adults with type 2 diabetes inadequately controlled with oral drugs: a randomized, controlled phase 3 trial. Diabetes Obes Metab. 2013;15:826–32.
Kumar S, Jang HC, Demirağ NG, Skjøth TV, Endahl L, Bode B. Efficacy and safety of once-daily insulin degludec/insulin aspart compared with once-daily insulin glargine in participants with type 2 diabetes: a randomized, treat-to-target study. Diabetic Med. 2017;34:180–8.
Fulcher GR, Christiansen JS, Bantwal G, et al. Comparison of insulin degludec/insulin aspart and biphasic insulin aspart 30 in uncontrolled, insulin-treated type 2 diabetes: a phase 3a, randomized, treat-to-target trial. Diabetes Care. 2014;37:2084–90.
Bebakar WM, Chaykin L, Hersløv ML, Rasmussen S. Intensification of IDegAsp twice daily (adding insulin aspart vs. switching to basal-bolus): exploratory randomized trial in type 2 diabetes. Diabetes Ther. 2017;8:197–205.
Philis-Tsimikas A, Astamirova K, Gupta Y, et al. Similar glycaemic control with less nocturnal hypoglycaemia in a 38-week trial comparing the IDegAsp co-formulation with insulin glargine U100 and insulin aspart in basal insulin-treated subjects with type 2 diabetes mellitus. Diabetes Res Clin Pract. 2019;147:157–65.
Rodbard HW, Cariou B, Pieber TR, Endahl LA, Zacho J, Cooper JG. Treatment intensification with an insulin degludec (IDeg)/insulin aspart (IAsp) co-formulation twice daily compared with basal IDeg and prandial IAsp in type 2 diabetes: a randomized, controlled phase III trial. Diabetes Obes Metab. 2016;18:274–80.
Hassanein M, Echtay AS, Malek R, et al. Original paper: Efficacy and safety analysis of insulin degludec/insulin aspart compared with biphasic insulin aspart 30: a phase 3, multicentre, international, open-label, randomised, treat-to-target trial in patients with type 2 diabetes fasting during Ramadan. Diabetes Res Clin Pract. 2018;135:218–26.
Yang W, Ma J, Hong T, et al. Efficacy and safety of insulin degludec/insulin aspart versus biphasic insulin aspart 30 in Chinese adults with type 2 diabetes: a phase III, open-label, 2:1 randomized, treat-to-target trial. Diabetes Obes Metab. 2019;21:1652–60.
Kaneko S, Chow F, Choi DS, et al. Insulin degludec/insulin aspart versus biphasic insulin aspart 30 in Asian patients with type 2 diabetes inadequately controlled on basal or pre-/self-mixed insulin: a 26-week, randomised, treat-to-target trial. Diabetes Res Clin Pract. 2015;107:139–47.
Gerety G, Bebakar WM, Chaykin L, et al. Treatment intensification with insulin degludec/insulin aspart twice daily: randomized study to compare simple and step-wise titration algorithms. Endocr Pract. 2016;22:546–54.
World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310:2191–4.
Skyler JS, Bergenstal R, Bonow RO, et al. Intensive glycemic control and the prevention of cardiovascular events: implications of the ACCORD, ADVANCE, and VA diabetes trials: a position statement of the American Diabetes Association and a scientific statement of the American College of Cardiology Foundation and the American Heart Association. Diabetes Care. 2009;32:187–92.
Ma RC, Chan JC. Type 2 diabetes in East Asians: similarities and differences with populations in Europe and the United States. Ann N Y Acad Sci. 2013;1281:64–91.
Lear SA, Humphries KH, Kohli S, Chockalingam A, Frohlich JJ, Birmingham CL. Visceral adipose tissue accumulation differs according to ethnic background: results of the Multicultural Community Health Assessment Trial (M-CHAT). Am J Clin Nutr. 2007;86:353–9.
Hayashi T, Boyko EJ, Leonetti DL, et al. Visceral adiposity and the risk of impaired glucose tolerance: a prospective study among Japanese Americans. Diabetes Care. 2003;26:650–5.
Zhang XM, Li PF, Hou JN, Ji LN. Blood glucose profiles in East Asian and Caucasian injection-naive patients with type 2 diabetes inadequately controlled on oral medication: a pooled analysis. Diabetes Metab Res Rev. 2018;34:e3062.
WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–63.
Jih J, Mukherjea A, Vittinghoff E, et al. Using appropriate body mass index cut points for overweight and obesity among Asian Americans. Prev Med. 2014;65:1–6.
Hsu WC, Araneta MR, Kanaya AM, Chiang JL, Fujimoto W. BMI cut points to identify at-risk Asian Americans for type 2 diabetes screening. Diabetes Care. 2015;38:150–8.
Narayan KMV, Kanaya AM. Why are South Asians prone to type 2 diabetes? A hypothesis based on underexplored pathways. Diabetologia. 2020;63:1103–9.
Yabe D, Seino Y. Type 2 diabetes via beta-cell dysfunction in east Asian people. Lancet Diabetes Endocrinol. 2016;4:2–3.
Ohn JH, Kwak SH, Cho YM, et al. 10-year trajectory of beta-cell function and insulin sensitivity in the development of type 2 diabetes: a community-based prospective cohort study. Lancet Diabetes Endocrinol. 2016;4:27–34.
Moller JB, Dalla Man C, Overgaard RV, et al. Ethnic differences in insulin sensitivity, beta-cell function, and hepatic extraction between Japanese and Caucasians: a minimal model analysis. J Clin Endocrinol Metab. 2014;99:4273–80.
Kang ES, Kim HJ, Ahn CW, et al. Relationship of serum high sensitivity C-reactive protein to metabolic syndrome and microvascular complications in type 2 diabetes. Diabetes Res Clin Pract. 2005;69:151–9.
Huang L, Wang Z, Wang H, et al. Nutrition transition and related health challenges over decades in China. Eur J Clin Nutr. 2020;75:247–52.
Hu EA, Pan A, Malik V, Sun Q. White rice consumption and risk of type 2 diabetes: meta-analysis and systematic review. BMJ. 2012;344:e1454.
Venn BS, Williams SM, Mann JI. Comparison of postprandial glycaemia in Asians and Caucasians. Diabetic Med. 2010;27:1205–8.
Acknowledgements
The authors would like to thank all participants and researchers who took part in the individual trials.
Funding
This study and the journal’s Rapid Service Fee were funded by Novo Nordisk.
Medical Writing and Editorial Assistance
Medical writing and editorial support for the development of this manuscript, under the direction of the authors, was provided by Matthew Robinson, Chinnappa AB, and Malgorzata Urbacz of Ashfield MedComms, an Ashfield Health company, and funded by Novo Nordisk.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICJME) criteria for authorship of this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Balamurali Kalyanam and Shahid Akhtar contributed to the conception and design of this study. Soumitra Kar performed the data analysis. Martin Haluzík, Edward Franek and Wenying Yang were involved in data acquisition. Ted Wu, Dilek Gogas Yavuz, Ambika Gopalakrishnan Unnikrishnan, Takahisa Hirose, Martin Haluzík, Edward Franek, Wenying Yang, Balamurali Kalyanam and Shahid Akhtar contributed to data interpretation. All authors critically revised the draft of the manuscript and approved the final report.
Disclosures
Wenying Yang has attended advisory boards for Novo Nordisk; received investigator-initiated trial research funds from AstraZeneca; been a speaker for Novo Nordisk, Bayer, Sanofi Aventis, Merck Sharp & Dohme China, AstraZeneca, Eli Lilly, Boehringer Ingelheim and Servier; and received honorarium and travel support as an advisory board member from Merck & Co., Inc. Shahid Akhtar, Balamurali Kalyanam and Soumitra Kar are employees of Novo Nordisk A/S. Edward Franek has participated in advisory panels for AstraZeneca, Bioton, Boehringer Ingelheim and Novo Nordisk; and has received honoraria for serving on speakers' bureaus for AstraZeneca, Bioton, Boehringer Ingelheim, Eli Lilly, Merck, Merck Sharp & Dohme, Novo Nordisk and Servier. Martin Haluzík has served on an advisory panel for Eli Lilly, Novo Nordisk, Sanofi, AstraZeneca and Mundipharma; has served as a consultant for Eli Lilly, Novo Nordisk, Sanofi, AstraZeneca and Mundipharma; has received research support from AstraZeneca, Eli Lilly and Bristol‐Meyers Squibb; and has received honoraria or consulting fees from Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Johnson & Johnson, Novartis, Novo Nordisk, Pfizer, Medtronic and Sanofi. Takahisa Hirose has received honoraria from Sanofi K.K., Eli Lilly Japan K.K., Novo Nordisk Pharma Ltd., Takeda Pharmaceutical Company, Ltd., MSD K.K., Sumitomo Dainippon Pharma Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., Ono Pharmaceutical Co., Ltd. and AstraZeneca K.K.; has received research funding from Mitsubishi Tanabe Pharma Corp. and AstraZeneca K.K.; and has received subsidies or donations from Sumitomo Dainippon Pharma Co., Ltd., Novartis Pharma K.K., MSD K.K., Mitsubishi Tanabe Pharma Corp., Daiichi Sankyo Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., Ono Pharmaceutical Co., Ltd., Novo Nordisk Pharma Ltd., Soiken, Inc. and Takeda Pharmaceutical Company. Ted Wu has served as a consultant, speaker and/or advisory board member for AstraZeneca, Boehringer Ingelheim, Eli Lilly, MSD, Novartis, Novo Nordisk and Sanofi Aventis. Dilek Gogas Yavuz has served as consultant, speaker and/or advisory board member for Novo Nordisk, Sanofi-Aventis, Novartis, Eli Lilly, Boehringer Ingelheim, Astra Zeneca, Amgen and Pfizer. Ambika Gopalakrishnan Unnikrishnan has been part of speaker panels for Novo Nordisk, Eli Lilly, Sanofi, Merck Sharp & Dohme, AstraZeneca and Boehringer Ingelheim; and has received research funding support from Novo Nordisk, Sanofi, Eli Lilly and Janssen.
Compliance with Ethics Guidelines
The individual trials considered for this post hoc analysis were approved by health authorities according to the corresponding local regulations and by the local independent ethics committee. All procedures performed in the trials considered in this analysis were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the studies considered for this post hoc analysis.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Yang, W., Akhtar, S., Franek, E. et al. Postprandial Glucose Excursions in Asian Versus Non-Asian Patients with Type 2 Diabetes: A Post Hoc Analysis of Baseline Data from Phase 3 Randomised Controlled Trials of IDegAsp. Diabetes Ther 13, 311–323 (2022). https://doi.org/10.1007/s13300-021-01196-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-021-01196-7