Abstract
Introduction
This study assessed the safety, tolerability, and PK/PD of HSK7653 tablets in Chinese patients with type 2 diabetes mellitus (T2DM).
Methods
This was a Phase IIa, multicenter, randomized, double-blind, placebo-controlled, and dose-increasing study with 48 Chinese diabetes patients. Subjects were randomly assigned to placebo and 10/25/50 mg dose groups, and they received oral administration once every two weeks for a total of six times. Safety and tolerability were assessed throughout this study, and PK/PD parameters were analyzed using non-compartment model with WinNonlin.
Results
The three doses of HSK7653 were well tolerated, and the incidence of TEAE and ADR was not significantly increased compared with the placebo group. Cmax increased linearly with the increasing dose, and the mean t1/2 was 64.0–87.0 h. The first dose and last dose PK parameters were similar. After oral administration of 10–50 mg HSK7653 every two weeks, the average Rac_Cmax and Rac_AUC were 0.9–1.0 and 1.0–1.1 respectively; therefore, HSK7653 was not accumulated in vivo. All three doses significantly inhibited DPP-4 activity and increased plasma GLP-1 level and serum insulin levels. When the plasma concentration of HSK7653 was ≥ 20.0 ng/mL, the DPP-4 inhibition rate in all subjects was maintained at > 80.0%. In 10 and 25 mg dose groups, the HbA1c levels maintained a downward trend compared with the placebo group.
Discussion
HSK7653 showed desirable pharmacokinetic and pharmacodynamic properties with good safety and tolerability in Chinese T2DM patients. DPP-4 inhibition rate and plasma GLP-1 levels were higher in each dose group than in placebo group.
Trial Registration Number
CTR20182505 (Drug Clinical Trial Registration and Information Disclosure Platform, www.chinadrugtrials.org.cn).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
China has the largest number of adults with diabetes mellitus (140.9 million), and more than 90% of them have T2DM. Long-acting DPP-4 inhibitors, including Omarigliptin (Marizev®) and Trelagliptin (Zafatek®), once a week, were approved for marketing in Japan in 2015, but not yet approved in China. |
HSK7653 is a long-acting DPP-4 inhibitor that improves glycemic control by increasing GLP-1 and GIP plasma concentrations, promoting insulin secretion in a glucose concentration-dependent manner. |
This is the first Phase IIa study to evaluate the safety and PK/PD of biweekly HSK7653 in Chinese patients with T2DM. |
What was learned from the study? |
The PK characteristics of HSK7653 were similar for single versus multiple dosing, with Cmax and AUC increasing linearly with dosage. HSK7653 showed the DPP-4 inhibition rate in most subjects could reach more than 80% and was well tolerated in all dose groups. Plasma GLP-1 levels were significantly higher in each HSK7653 dose group than in the placebo group and were dose-dependent. |
HSK7653 is a long-acting inhibitor of DPP-4 and can be administered once every two weeks. The 10 mg and 25 mg doses can significantly reduce the level of HbA1c compared with baseline, and have a good hypoglycemic benefit. It is recommended to enter the subsequent clinical trial as the recommended dose. |
Introduction
Diabetes mellitus is a chronic metabolic disease characterized by hyperglycemia, which includes type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM). T1DM is caused by absolute insulin deficiency, and T2DM is caused by relative insulin deficiency and insulin resistance. Complications of diabetes include damage to the heart, blood vessels, eyes, and kidneys, which lead to a high disability rate and early death and require ongoing medical treatment [1]. There are 536.6 million adults with diabetes worldwide in 2021. China has the largest number of adults with diabetes (140.9 million) [2], and more than 90.0% of them have T2DM [3]. By 2045, adults with diabetes are expected to reach 783.0 million globally and 174.4 million in China [2].
Dipeptidyl peptidase-4 (DPP-4) inhibitors or “gliptins” are emerging oral hypoglycemic agents, which inhibit the activity of DPP-4 and increase the levels of glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). They boost insulin secretion in a glucose concentration-dependent manner, inhibit glucagon secretion, delay gastric emptying, and protect islet β-cell function, all contributing to their hypoglycemic effect [4,5,6]. DPP-4 inhibitors are glucose-dependent agents with low incidence of hypoglycemia and good safety and tolerability, and they can be administered alone or in combination with other hypoglycemia drugs. HSK7653 ((2R,3S,5R,6S)-2-(2,5-difluorophenyl)-5-[2-(methylsulfonyl)-2,6-dihydropyrrolo[3,4-c]pyrazol-5(4H)-yl]-6-(trifluoromethyl)-tetrahydro-2H-pyran-3-amine) (Supplementary Fig. S1) [7] is an oral hypoglycemic agent of DPP-4 inhibitor class, treatment of T2DM developed by Sichuan Haisco Pharmaceutical Co., Ltd. The main components of HSK7653 are structurally similar to Omarigliptin in that they are both long-acting DPP-4 inhibitors and can be administered biweekly. HSK7653 is relatively rapidly absorbed orally, and a mass balance study (CTR20201559) showed that the elimination process is dominated by renal excretion (68.1%), followed by hepatic metabolism (metabolites in urine 9.7% and feces 5.7%) and fecal excretion (7.4%). Thus, the kidney is the major organ for eliminating HSK7653, and glomerular filtration is the main route of excretion. This study evaluated the safety, tolerability, and PK/PD of HSK7653 tablets in Chinese patients with T2DM.
Methods
Study Design and Participants
A multicenter, randomized, double-blind, dose-increasing design was used in this study, which was carried out at the Department of Endocrinology of People's Hospital of Peking University, Phase I Clinical Ward of Tianjin People's Hospital and Chinese People’s Liberation Army General Hospital. The study protocols were approved by the Independent Ethics Committees of the three participating hospitals, and registered at drug clinical trial registration and information disclosure platform, CTR20182505. The studies followed the Declaration of Helsinki (2013) and local applicable laws and regulations, and all subjects provided written informed consents before participated in the studies.
Forty-eight Chinese T2DM patients were randomly divided into 10, 25, and 50 mg dose groups and placebo group. Patient admission criteria included: diagnosed with T2DM for at least 3 months according to 1999 WHO diagnostic criteria and classification; hypoglycemia agents are allowed in the past year but not allowed in the past three months, blood sugar can be controlled through diet and exercise; and within one year before three months, not allowed using hypoglycemia agents continuously more than 4 weeks, as for the insulin treatment is not allowed except pregnancy. The qualifying patients entered the 6-week double-blind treatment (called initial study period with oral HSK7653 tablets or placebo), and subjects who had successfully completed the initial study continued into the extended study period. In the extended study period, subjects continued oral HSK7653 or placebo for 6 weeks, and a safety follow-up was performed for 2 weeks. The test drug and placebo will be indistinguishable by shape, appearance and packaging. Until the end of the study, the investigator and their staff, the subjects, monitors and sponsors will remain blinded.
Study Drug
HSK7653 and placebo tablets were provided by Sichuan Haisco Pharmaceutical Co., LTD. The drug or placebo was administered with 240.0 mL water, fasted on day 1 and day 29. And dosing regimens were once every 2 weeks for 12 consecutive weeks. Subjects should swallow the entire tablet, not chew it. Unless required for a test such as Electrocardiograph (ECG) or vital sign, subjects did not lie down within 2.0 h of taking drugs.
Safety and Efficacy Assessment
Safety evaluation included the occurrence of adverse events (AE) during the clinical study, physical examination, laboratory examination (blood routine, urine routine, blood biochemistry, amylase, lipase), vital signs, 12-ECG, and occurrence of hypoglycemic events (including incidence, occurrence time, duration, clinical symptoms related to hypoglycemia, etc.).
The main efficacy parameters included: plasma DPP-4 inhibition rate, plasma active GLP-1 level, fasting plasma glucose, insulin, C-peptide, glucagon, glycosylated albumin (GA), and glycosylated hemoglobin (HbA1c) and other PD parameters.
PK Parameters and PK/PD Analysis
In the study, 2.0 mL blood samples were collected during screening, the day − 2, before the first and third dosing (within 0.5 h) and at 0.25, 0.5, 1, 3, 6, 9, 12, 24, 72, and 168 h (day 8 and 29) post-dose, within 0.5 h before the second and fourth dosing (day 15 and 43), before dosing at day 57 and 71, post the last dosing (day 85). Analyzed in Suzhou Haike Medical Technology Co., Ltd. An established and validated high-performance liquid chromatography-tandem mass spectrometry (HPLC–MS/MS) method was used to determine the HSK7653 concentrations in plasma samples. First and last doses PK parameters included but not limited to: peak concentration (Cmax), peak time (Tmax), area under the drug-time curve from 0 to the last measurable concentration time point (AUC0–last), area under the drug-time curve calculated from 0 to infinity (AUC0–inf), percentage area under the extrapolated drug-time curve (AUC_%Extrap), elimination rate constant (λz), elimination half-life (t1/2), apparent clearance (CL/F), observed volume of distribution (Vz/F). The pharmacokinetic and pharmacodynamic parameters of HSK7653 were calculated with the actual sampling time using non-compartmental model analysis (NCA) by WinNonlin (version 8.2.0).
All subjects with evaluable PK and PD data were included in the PK/PD analysis. For the detection of PD indexes, the plasma DPP-4 inhibition rate and GLP-1 were determined by validated fluorescence and electrochemiluminescence methods in a central laboratory. Other PD indexes were measured following the standard operating procedure of each research unit. The exposure parameters [blood concentration, AUC, Cmax and Ctrough (blood concentration 336 h after the third dose)] of the drugs in each dose group were plotted with PD indicators (DPP-4 inhibition rate, AUECDPP-4 and AUECGLP-1) respectively, and the PK/PD relationship was analyzed.
DPP-4 Inhibition Rate
The mean DPP-4 inhibition rate was calculated as: mean DPP-4 inhibition rate = AUECDPP-4,0–tau/tau (for subjects with |Tlast-tau|< 10%*tau, select AUECDPP-4,0–last instead of AUECDPP-4,0–tau).
Statistical Analyses
Quantitative indicators will be descripted using mean, standard deviation (std), median, minimum, and maximum values using SAS 9.4. Other safety analysis and efficacy analysis were described in lists.
Results
Patients
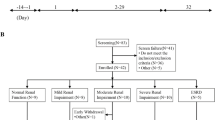
This study took place in 3 research centers and involved 48 subjects (Table 1). 243 subjects were screened after signing informed consent, and 195 did not meet the inclusion criteria. Forty-eight subjects were randomly assigned into three dose groups of HSK7653 (10, 25 and 50 mg) and placebo group using an interactive web response system. Forty-three subjects (89.6%, 43/48) completed 6 months of study, and drug compliance was 100% for the whole treatment period. In the initial study period, four subjects (2, 1 and 1 subject in the 10, 50 mg and placebo groups respectively) withdrew, 2 due to diabetic ketosis, and 2 for protocol violations in the extended study period, 1 subject (in the placebo group) withdrew due to poor blood glucose control and hyperglycemia (Fig. 1).
Safety
In this study, 38 subjects (79.2%, 38/48) experienced 137 incidences of treatment-emerged adverse events (TEAEs). The incidence of TEAEs in the 10, 25, 50 mg and placebo group were 58.3%, 91.7%, 83.3% and 83.3%, respectively. The highest incidence reported was positive urinary leukocyte (27.1%). Other TEAEs with an incidence > 5% included: prolonged QT interval (16.7%), abnormal T wave (12.5%), hypertriglyceridemia (10.4%), dyspepsia (10.4%), decreased T wave amplitude of ECG (8.3%), hyperlipidemia (8.3%), positive urine red blood cells (6.3%), hyperuricemia (6.3%), sinus bradycardia (6.3%), urinary tract infection (6.3%), hyperbilirubinemia (6.3%), dizziness (6.3%), and anemia (6.3%). Except one subject (25 mg dose group) with grade 3 upper respiratory tract infection, all the other TEAEs were grade 1 or 2, and 92.0% (126/137) of the TEAEs was classified as “symptom disappearance without sequelae”.
A total of 29 (60.4%, 29/48) subjects in this study experienced 93 adverse drug reactions (ADRs). The incidences of ADR in the 10, 25, 50 mg and the placebo group were 33.3%, 58.3%, 75.0% and 75.0%, respectively. The ADRs with HSK7653 or placebo incidence > 5.0% (HSK7653 dose group total vs placebo) included dyspepsia (11.1% vs 8.3%), hypertriglyceridemia (11.1% vs 0%),prolonged QT interval of ECG (11.1% vs 16.7%), white blood cells urine positive (8.3% vs 8.3%), electrocardiogram T wave abnormal (8.3% vs 8.3%), hyperlipidemia (8.3% vs. 8.3%), urinary tract infection (8.3% vs. 0%), decreased T wave amplitude of ECG (5.6% vs. 8.3%), positive urine red blood cells (5.6% vs. 8.3%), upper respiratory tract infection (5.6% vs. 0%), hyperbilirubinemia (5.6% vs. 8.3%), increased blood bilirubin (5.6% vs. 0%), anemia (5.6% vs. 8.3%) and dizziness (2.8% vs. 16.7%). Except for one subject with upper respiratory tract infection in the 25 mg dose group was classified as grade 3, and the rest of the ADRs were grades 1 or 2, 94.6% of the ADRs was classified as “symptom disappearance without sequelae”.
There were no serious adverse events/events, or adverse events requiring dose reduction, drug suspension or resumption, or posing a threat to life or death. In summary, HSK7653 was well-tolerated at doses of 10–50 mg, with no significant increase in TEAEs or ADRs compared to the placebo group (Supplementary Tables S1, S2).
Efficacy
HbA1c
HbA1c changes comparing to the baseline at each visit are shown in Table 2. Figure 2 shows that on day 85, the level of HbA1c in the HSK7653 dose groups were significantly lower than that at the baseline. Compared with the placebo group (the HbA1c level decreased by 0.3% from baseline), 10 and 25 mg dose groups showed more significant reductions (1.0% and 1.3% respectively), but 50 mg dose groups showed the lowest reductions (− 0.05).
Glycated Albumin (GA) and Fasting Plasma Glucose
On day 43, GA levels in the 10 and 25 mg dose groups were significantly lower than the baseline (2.3% and 0.8%), while GA levels in the 50 mg and placebo group were significantly higher than the baseline (0.4% and 0.8%). With the increase of treatment time, the level of GA in 10 and 25 mg dose groups decreased continuously. The mean baseline GA value of 10 and 25 mg dose groups were 23.3% and 21.8%; on day 43 were 21.2% and 21.0%; on day 85 were 20.0% and 20.5%, which were lower than those in the 50 mg and placebo group. The baseline mean GA value in the 50 mg and placebo group were 23.2% and 23.7%; on day 43 were 23.6% and 24.5%; on day 85 were 24.2% and 22.8%.
On day 43, compared to baseline, fasting blood glucose levels were significantly lower in 10 and 25 mg dose groups (0.7 and 0.5 mmol/L); in 50 mg and placebo group were significantly higher (increased 0.8 mmol/L and 0.5 mmol/L). Both 10 and 25 mg dose groups showed significant hypoglycemic benefit at each checking point. The baseline mean fasting blood glucose values in 10 and 25 mg dose group were 10.1 and 8.7 mmol/L; on day 43 were 9.2 and 8.1 mmol/L; on day 85 were 9.2 and 8.5 mmol/L. The hypoglycemic effect lasted until day 85 after administration, which decreased by 0.7 and 0.2 mmol/L compared with the baseline.
PK/PD
Pharmacokinetics
After oral administration of HSK7653, drug absorption was relatively rapid, and the median Tmax of HSK7653 was about 1.0–3.0 h. In the dose range of 10–50 mg, PK characteristics of the first dose and the last dose were similar. Cmax and AUC of HSK7653 increased linearly with dose. The average CL/F, VZ/F, and t1/2 of HSK7653 were 0.7–0.8 L/h, 61.1–81.9 L, and 64.0–87.0 h, respectively. After oral administration of 10–50 mg HSK7653 every 2 weeks, the mean Rac_Cmax and CV% were 0.9–1.0 and 19.0–22.0%, respectively. The mean Rac_AUC and CV% were 1.0–1.1 and 10.0–18.0%, respectively. Under the current administration regimen, HSK7653 did not accumulated in body (Table 3). The average plasma concentration versus time curve is shown in Fig. 3.
Pharmacodynamics
Plasma DPP-4 Inhibition Rate
After oral administration of HSK7653 every two weeks, the mean inhibition rate of DPP-4 in each HSK7653 dose group was significantly higher than that in the placebo group (Table 4a). There was no significant difference in DPP-4 inhibition rate among the HSK7653 dose groups after first and last doses, as shown in Fig. 4. The plasma concentration scatters diagram of DPP-4 inhibition rate of HSK7653 is shown in Fig. 5, when the plasma concentration of HSK7653 was 20.0 ng/mL or above, the DPP-4 inhibition rate in most subjects (99.8%) could reach more than 80.0%. AUECDPP-4 in HSK7653 dose groups was significantly higher than that in the placebo group and increased slightly with dose increase (Table 4b). There was no significant difference in AUECDPP-4 in each dose group after first and last doses (Table 4b).
DPP-4 inhibition rate-time curves of each dose group after first dose and last dose of HSK7653. Left: first dose (Day 1–15), Right: last dose (Day 29–43). Nominal time after first dose: time after planned first dose. Each color line represents the mean DPP-4 inhibition rate-time curve for one dose group, and the error bars represent the SD
Plasma GLP-1 Levels
The MTT test was carried out on day − 1 (baseline), day 42 (1 day before the fourth administration) and day 85 (2 weeks after the sixth administration), and the time courses of plasma GLP-1 levels are shown in Fig. 6. Before treatment, there was a slight difference in the AUECGLP-1 between the HSK7653 dose groups and the placebo group; the mean baseline values (CV%) of AUECGLP-1 for 10, 25, 50 mg and placebo were 15.5 (25.0), 13.7 (45.0), 21.2 (79.0), and 11.0 (26.0) pg*h/mL, respectively. After treatment with HSK7653, AUCEGLP-1 in the treatment groups were significantly higher than the placebo group. And the values of all the dose groups were similar. With the increase of dose, the median value showed a certain increasing trend. In each dose group, AUCEGLP-1 of HSK7653 (i.e., on day 42 and 85) was about twice as high as that at baseline. The mean value (CV%) of AUECGLP-1 on day 42 for 10, 25, 50 mg and placebo were 31.4 (53.0), 30.7 (45.0), 44.6 (60.0), and 12.2 (44.0) pg*h/mL, respectively. The mean (CV%) value of AUECGLP-1 on day 85 for 10, 25, 50 mg and placebo were 35.5 (80.0), 38.4 (60.0), 42.6 (38.0), and 15.0 (35.0) pg*h/mL, respectively.
Serum Insulin
After multiple administration of HSK7653 (on day 85), the serum insulin levels were increased in each dose group compared with the placebo group. The mean AUECinsulin0–4 h and its change rate in each dose group were increased steadily with increasing treatment time. The AUECinsulin0–4 h values of the 10, 25, 50 mg and placebo groups on day − 1 were 73.1 ± 40.9, 96.3 ± 48.8, 65.9 ± 33.3, and 73.4 ± 35.8 mU*h/L, respectively; the AUECinsulin0–4 h values of the 10, 25, 50 mg and placebo groups on day 85 were 91.2 ± 41.8, 124.0 ± 107.0, 96.4 ± 34.7, and 91.1 ± 43.2 mU*h/L, respectively.
Serum C-peptide and Plasma Glucagon
Before administration, the level of plasma C-peptide in 25 mg dose group was the highest, followed by the 50 mg dose group and the placebo group, and the lowest in the 10 mg dose group. With the increase of treatment time, the level of C-peptide in the 10 mg dose group gradually increased to a similar level as the placebo group, while there was no significant change in the levels of C-peptide in the 25 and 50 mg dose groups compared with the placebo group.
Before administration, the level of the plasma glucagon in the 10 mg dose group was the highest, while in the 25 and 50 mg dose groups were lower than the placebo group. With the increase in treatment time, glucagon level in the 10 mg dose group was significantly higher than the placebo group at the beginning. It gradually decreased to below the level of the placebo group at 90–180 min after meal. The glucagon levels in both 25 and 50 mg dose groups were lower than the placebo group during treatment.
Discussion
To overcome the drawbacks of short half-lives, low bioavailability and adverse side effects of traditional hypoglycemic agents such as insulin, biguanides and sulfonylureas, new anti-diabetes drugs like DPP-4 inhibitors, GLP-1 receptor agonists, and sodium-glucose co-transporter 2 (SGLT2) inhibitors have been developed [8,9,10]. DPP-4 inhibitors act by directly inhibiting the DPP-4 enzyme, which prolong the half-life of GIP and GLP-1, providing an improved treatment option for T2DM at risk of hypoglycemia [11]. Currently, the short-acting DPP-4 inhibitors listed in China include sitagliptin, vildagliptin, saxagliptin, linagliptin and alogliptin, which require daily dosing. Omarigliptin and trelagliptin, as long-acting DPP-4 inhibitors, were approved in Japan in 2015 [12, 13]. These hypoglycemic agents enhance medication adherence, better glycemic control, prevent T2DM complications, and are dosed weekly [14, 15]. HSK7653 is a long-acting DPP-4 inhibitor that lasts longer than other inhibitors on the market. Studies have shown that introduction of a trifluoromethyl group at the 6-position of the (2R, 3S, 5R, 6S) tetrahydropyran ring of omarigliptin not only significantly improved the overall pharmacokinetic characteristics in mice, but also maintained considerably longer DPP-4 inhibitory activity. The plasma DPP-4 inhibition time of HSK7653 was about 4 times longer than that of omarigliptin [7].
We evaluated the safety, tolerability, and PK/PD of HSK7653 in Chinese patients with T2DM. There were no serious adverse events and death during the study period. The incidences of ADRs in the three dose groups of HSK7653 were not higher than the placebo group; and the incidences of ADRs in the 10 and 25 mg dose groups were significantly lower than the placebo group. Except for one case of ADR with grade 3, the CTCAE of other ADRs were grade 1 or 2. ADRs with an overall higher incidence in the HSK7653 dose groups included dyspepsia, hypertriglyceridemia and QT prolongation. Events of dyspepsia have also been reported in previous clinical studies of marketed DPP-4 inhibitors [16, 17].
Hypoglycemia was the expected ADR in this study. A significant advantage of DPP-4 inhibitors over sulfonylureas is the reduced risk of hypoglycemia, which is approximately tenfold greater comparing with DPP-4 inhibitors [18]. An observational study in Sweden demonstrated a lower risk of severe hypoglycemia compared with sulfonylureas [19]. Another RCT comparing DPP-4 inhibitors with α-glucosidase inhibitors suggested better glycemic control with DPP-4 inhibitors, including a lower risk of gastrointestinal adverse effects [20]. In this study, 2 subjects were reported with hypoglycemia; 1 subject in the placebo group (a total of 3 times) showed hypoglycemia; 1 subject (in the 10 mg dose group) in the HSK7653 dose group had a CTCAE grade of 1, and this subject recovered without taking any measures. Based on the above results, HSK7653 within the dose range of 10–50 mg was well tolerated and safe in Chinese T2DM subjects.
There were 4 subjects (11.1%, 4/36) exhibiting ADRs with prolonged QT interval in the HSK7653 dose group. Two subjects in the 25 and 50 mg group respectively, reported 10 incidences, and 2 subjects in the placebo group reported 2 incidences. The incidence of ADRs with prolonged QT interval in the HSK7653 dose groups were lower than in the placebo group, suggesting that the prolonged QT interval may be related to other factors and not due to HSK7653 administration. After reviewing all EGC results obtained during the screening period for these subjects with QT interval prolongation, except for 1 subject (placebo group), the corrected QT (QTc) values of ECG in the other 5 subjects (2, 2, and 1 subject in 25, 50 mg and placebo groups respectively) were higher than the normal value during the screening period. Therefore, the occurrence of adverse reactions cannot be excluded from the association with their medical history.
In previous studies on the safety and tolerance of single administration of HSK7653 in Chinese healthy subjects, no adverse events of prolonged QT interval were found. The results of plasma concentration QT analysis of single dose in healthy subjects showed that there was no significant risk of QT prolongation of HSK7653 in the concentration range of 0–4190.0 ng/mL (to be published). Considering that when dosing 25 mg once every two weeks reached a steady state, the mean value of Cmax was 822.0 ng/mL (the mean value of Cmax after multiple oral administration was 411.0 ng/mL at 25 mg), there was no significant risk of prolongation of QT interval at the dose of 25 mg or less every two weeks. For all these reported adverse events in this study, 1 subject remained stable after the end of the study, and the other ADRs were “symptom disappearance without sequelae”. Overall, the QT interval prolongation reported in the studies had little effect on the safety of the subjects. For DPP-4 inhibitors currently in the market, linagliptin did not increase the QTc interval in the treatment group compared with placebo at doses of 5 and 100 mg [21]. In healthy subjects, 40–400 mg dose of saxagliptin and its metabolites had no significant effect on QTC [22]. The clinical dose of sitagliptin 100 mg does not correlate with an increase in QTc interval, while the super-therapeutic dose of 800 mg is generally well tolerated and the QT interval is prolonged, but the clinical significance is not significant [23]. Teneligliptin, which has been marketed in other countries, significantly prolonged the QT interval only at 4 times the normal dose (160.0 mg/day) [24].
Treatment with HSK7653 showed significant increases in DPP-4 inhibition rate, plasma GLP-1 levels and insulin levels compared to the placebo group, and showed dose dependence. This demonstrates that HSK7653 plays a dual hormone regulatory role by inhibiting GLP-1 degradation, increasing endogenous active GLP-1 concentration, improving insulin levels and inhibiting insulin glucagon section. It is consistent with the action mechanism of similar DPP-4 inhibitors.
The efficacy results indicated that compared with the placebo group, HbA1c in 10 and 25 mg dose groups decreased more significantly on day 85. On day 43 of this study, 10 and 25 mg dose groups significantly reduced fasting blood glucose and GA levels. By day 85, fasting blood glucose and GA levels still maintained a downward trend. However, there was no significant improvement above those indexes in 50 mg dose group.
On day 85, among the 11 subjects in the 50 mg dose group, HbA1c levels decreased significantly in 5 subjects, while 6 subjects were increased compared with baseline. The baseline data indicated that the subjects enrolled in the 50 mg dose group had the highest average age, relatively longer average duration of illness, a higher proportion of retinopathy than other dose groups, and the lowest serum insulin levels. Therefore, although the pharmacodynamic results of the 50 mg dose group suggested that this dose also had potential hypoglycemic effect, the efficacy and safety of this dose were affected to some extent by the small sample size, individual differences in baseline clinical characteristics, sampling error and other factors, which is also the limitation of this study. Moreover, it may be related to saturation inhibition in the 50 mg dose group. When Cthrough > 20.0 ng/mL, the amplitude of AUECDPP-4 increase was also significantly reduced, indicating that the DPP-4 inhibition rate was close to saturation at this time. This may also be a factor affecting the effectiveness of the 50 mg dose group.
The HbA1c levels after treatment with DPP-4 inhibitor drugs show a decrease of ~ 0.5–1.0% when used alone and a decrease of ~ 0.6–1.1% when used in combination with metformin, depending on the drug, treatment dose and starting HbA1c levels [25]. Therefore, DPP-4 inhibitor drugs show improved glycemic control when used in combination with metformin, sulfonylureas or insulin. At present, there are no direct comparative trials of DPP-4 inhibitors [26]. Our study demonstrated that HSK7653 when dosed at 10 and 25 mg once every two weeks effectively inhibited DPP-4 enzyme, increased GLP-1 levels, and improved insulin levels in patients with T2DM. HSK7653 is safe and effective for the hypoglycemic control of patients.
Conclusion
HSK7653 in the dosing range of 10–50 mg once every two weeks showed good pharmacokinetic and pharmacodynamic results in Chinese patients with T2DM, with good safety and tolerability. HSK7653 did not accumulate in the body after multiple administrations. The DPP-4 inhibition rate and plasma GLP-1 levels of HSK7653 in each dose group were significantly higher than those in placebo group, and both showed dose correlation. All dose groups of HSK7653 increased the insulin levels of diabetes subjects, and 10 and 25 mg dose groups significantly reduced the HbA1c levels compared with baseline, which suggests that these two doses exhibited better hypoglycemic benefits. In summary, the 10 mg and 25 mg dose groups demonstrated superior efficacy and safety over the 50 mg dose group and the placebo group. Hence, the 10 and 25 mg doses are selected for subsequent clinical trials.
Data Availability
The data supporting this study's findings are available from the corresponding author upon reasonable request, at any time after publication.
References
Cole JB, Florez JC. Genetics of diabetes mellitus and diabetes complications. Nat Rev Nephrol. 2020;16(7):377–90.
Sun H, Saeedi P, Karuranga S, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183: 109119.
Chinese Elderly Type 2 Diabetes Prevention and Treatment of Clinical Guidelines Writing Group GEaMBoCGS, Geriatric Endocrinology and Metabolism Branch of Chinese Geriatric Health Care Society, Geriatric Professional Committee of Beijing Medical Award Foundation, & National Clinical Medical Research Center for Geriatric Diseases (PLA General Hospital), [Clinical guidelines for prevention and treatment of type 2 diabetes mellitus in the elderly in China (2022 edition)]. Zhonghua nei ke za zhi 2022;61(1), 12–50.
Deacon CF. Dipeptidyl peptidase 4 inhibitors in the treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2020;16(11):642–53.
Mulvihill EE, Varin EM, Gladanac B, et al. Cellular sites and mechanisms linking reduction of dipeptidyl peptidase-4 activity to control of incretin hormone action and glucose homeostasis. Cell Metab. 2017;25(1):152–65.
Luconi M, Cantini G, Ceriello A, et al. Perspectives on cardiovascular effects of incretin-based drugs: From bedside to bench, return trip. Int J Cardiol. 2017;241:302–10.
Zhang C, Ye F, Wang J, et al. Design, synthesis, and evaluation of a series of novel super long-acting DPP-4 inhibitors for the treatment of type 2 diabetes. J Med Chem. 2020;63(13):7108–26.
Ramzan S, Timmins P, Hasan SS, et al. Trends in global prescribing of antidiabetic medicines in primary care: a systematic review of literature between 2000–2018. Prim Care Diabetes. 2019;13(5):409–21.
Guo M, Gu J, Teng F, et al. The efficacy and safety of combinations of SGLT2 inhibitors and GLP-1 receptor agonists in the treatment of type 2 diabetes or obese adults: a systematic review and meta-analysis. Endocrine. 2020;67(2):294–304.
Deacon CF, Lebovitz HE. Comparative review of dipeptidyl peptidase-4 inhibitors and sulphonylureas. Diabetes Obes Metab. 2016;18(4):333–47.
Karagiannis T, Boura P, Tsapas A. Safety of dipeptidyl peptidase 4 inhibitors: a perspective review. Ther Adv Drug Saf. 2014;5(3):138–46.
Sheu WH, Gantz I, Chen M, et al. Safety and efficacy of omarigliptin (MK-3102), a novel once-weekly DPP-4 inhibitor for the treatment of patients with type 2 diabetes. Diabetes Care. 2015;38(11):2106–14.
Grimshaw CE, Jennings A, Kamran R, et al. Trelagliptin (SYR-472, Zafatek), novel once-weekly treatment for type 2 diabetes, inhibits dipeptidyl peptidase-4 (DPP-4) via a non-covalent mechanism. PLoS One. 2016;11(6): e0157509.
Scheen AJ. Once-weekly DPP-4 inhibitors: do they meet an unmet need? Lancet Diabetes Endocrinol. 2015;3(3):162–4.
Asche C, LaFleur J, Conner C. A review of diabetes treatment adherence and the association with clinical and economic outcomes. Clin Ther. 2011;33(1):74–109.
Tella SH, Rendell MS. DPP-4 inhibitors: focus on safety. Expert Opin Drug Saf. 2015;14(1):127–40.
Wu S, Chai S, Yang J, et al. Gastrointestinal adverse events of dipeptidyl peptidase 4 inhibitors in type 2 diabetes: a systematic review and network meta-analysis. Clin Ther. 2017;39(9):1780–9 (e33).
Stoian AP, Sachinidis A, Stoica RA, et al. The efficacy and safety of dipeptidyl peptidase-4 inhibitors compared to other oral glucose-lowering medications in the treatment of type 2 diabetes. Metabolism. 2020;109: 154295.
Eriksson JW, Bodegard J, Nathanson D, et al. Sulphonylurea compared to DPP-4 inhibitors in combination with metformin carries increased risk of severe hypoglycemia, cardiovascular events, and all-cause mortality. Diabetes Res Clin Pract. 2016;117:39–47.
Li Z, Zhao L, Yu L, et al. Head-to-head comparison of the hypoglycemic efficacy and safety between dipeptidyl peptidase-4 inhibitors and alpha-glucosidase inhibitors in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Front Pharmacol. 2019;10:777.
Ring A, Port A, Graefe-Mody EU, et al. The DPP-4 inhibitor linagliptin does not prolong the QT interval at therapeutic and supratherapeutic doses. Br J Clin Pharmacol. 2011;72(1):39–50.
Boulton DW. Clinical pharmacokinetics and pharmacodynamics of saxagliptin, a dipeptidyl peptidase-4 inhibitor. Clin Pharmacokinet. 2017;56(1):11–24.
Bloomfield DM, Krishna R, Hreniuk D, et al. A thorough QTc study to assess the effect of sitagliptin, a DPP4 inhibitor, on ventricular repolarization in healthy subjects. J Clin Pharmacol. 2009;49(8):937–46.
Erande S, Sarwardekar S, Desai B. QT/QTc safety and efficacy evaluation of teneligliptin in Indian type 2 diabetes mellitus patients: the “thorough QT/QTc” study (Q-SET study). Diabetes Metab Syndr Obes. 2019;12:961–7.
Craddy P, Palin HJ, Johnson KI. Comparative effectiveness of dipeptidylpeptidase-4 inhibitors in type 2 diabetes: a systematic review and mixed treatment comparison. Diabetes Ther. 2014;5(1):1–41.
Munir KM, Lamos EM. Diabetes type 2 management: what are the differences between DPP-4 inhibitors and how do you choose? Expert Opin Pharmacother. 2017;18(9):839–41.
Acknowledgements
Sichuan Haisco Pharmaceutical Co., Ltd., China provided the study drug.
Medical Writing and Editorial Assistance
We thank Jeff Johnson of Scientific Writing Solutions (USA) for editorial assistance.
Authorship
All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship of this article, ensuring accuracy and completeness of the work, and have approved this version for publication.
Funding
This work was supported by the New Medicine Clinical Research Fund (4246Z512), the grant is used for the study and the Rapid Service Fee.
Author information
Authors and Affiliations
Contributions
Nan Bai contributed to the conception and design of study, laboratory data acquisition, data analysis and drafting of article. Jin Wang contributed to the design of study and critical revision. Wenxin Liang, Leili Gao, Wei Cui, Qinghe Wu and Fangqiang Li contributed to the laboratory data acquisition and data analysis. Linong Ji and Yun Cai contributed to the conception and design of study, analysis of data and drafting of article and critical revision.
Corresponding authors
Ethics declarations
Conflict of Interest
Nan Bai, Jin Wang, Wenxin Liang, Leili Gao, Wei Cui, Qinghe Wu, Fangqiong Li, Linong Ji, and Yun Cai declare no potential interest conflicts.
Ethical Approval
The study protocols were approved by the Independent Ethics Committees of the Department of Endocrinology of People's Hospital of Peking University (2019PHA001-001), Phase I Clinical Ward of Tianjin People's Hospital (in Chinese: (2018)  ) and Chinese People's Liberation Army General Hospital (C2019-016-01). The studies followed the Declaration of Helsinki (2013) and local applicable laws and regulations, and all subjects provided written informed consents before participated in the studies.
) and Chinese People's Liberation Army General Hospital (C2019-016-01). The studies followed the Declaration of Helsinki (2013) and local applicable laws and regulations, and all subjects provided written informed consents before participated in the studies.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Bai, N., Wang, J., Liang, W. et al. A Multicenter, Randomized, Double-Blind, Placebo-Controlled, and Dose-Increasing Study on the Safety, Tolerability and PK/PD of Multiple Doses of HSK7653 by Oral Administration in Patients with Type 2 Diabetes Mellitus in China. Diabetes Ther 15, 183–199 (2024). https://doi.org/10.1007/s13300-023-01496-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01496-0