Abstract
Introduction
Poor metabolic control and excess body weight are frequently present in people with type 2 diabetes (PwT2D).
Methods
A systematic literature review was conducted to identify observational studies reporting clinical, economic, and health-related quality of life (HRQoL) outcomes associated with poor metabolic (according to HbA1c, blood pressure [BP] and low density lipoprotein cholesterol [LDL-C] levels) and/or weight control (defined by a body mass index [BMI] ≥ 30 kg/m2) in adults with T2D in Spain, including articles published in either Spanish or English between 2013 and 2022 and conference abstracts from the last 2 years.
Results
Nine observational studies were included in the analysis. Poor glycemic control (HbA1c ≥ 7%) was associated with cardiovascular disease (CVD), increased requirements for antidiabetic medications, higher and more frequent weight gain, a greater probability of hypoglycemia and dyslipidemia, and worse health-related quality of life (HRQoL). Uncontrolled BP in PwT2D was related with the presence of CVD, worse metabolic control, and higher BMI and abdominal perimeter values. Poor LDL-C control or dyslipidemia was associated with CVD, hypoglycemia, and elevated HbA1c and triglycerides levels. The presence of a BMI ≥ 30 kg/m2 was related to CVD and hypoglycemia, a higher prevalence of metabolic syndrome and worse BP control. Direct medical costs were found to be higher in PwT2D when coexisting with HbA1c levels ≥ 7%, uncontrolled BP or obesity. Increased total costs, including productivity losses, were also detected in those who presented uncontrolled BP and a BMI ≥ 30 kg/m2, and when poor weight control existed together with HbA1c ≥ 8% and poorly controlled BP.
Conclusion
Gathered evidence supports the high clinical, economic and HRQoL burden of poor metabolic and/or weight control in PwT2D in Spain and reinforces the importance of prioritizing its control to reduce the associated burden, at both the individual and healthcare system levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Despite existing evidence demonstrating that the presence of poor metabolic control and excess body weight negatively impact T2D, we have no knowledge of a systematic literature review that aggregates and summarizes its clinical, economic and quality of life burden in the Spanish population with T2D |
The present systematic literature review identifies and describes available evidence reporting quantitative data regarding clinical, economic and quality of life outcomes in Spanish adults with T2D and poor metabolic control or obesity compared to those controlled and with normal weight |
Available evidence supports a high clinical, economic and HRQoL burden associated with poor metabolic and/or weight control in adults with T2D in Spain |
Addressing metabolic control and excess weight would reduce the burden that T2D imposes on individuals and the healthcare system |
Digital Features
This article is published with digital features, including infographics, to facilitate understanding of the article. To view digital features for this article, go to https://doi.org/10.6084/m9.figshare.24420694.
Introduction
Diabetes mellitus (DM) is one of the most prevalent chronic diseases worldwide, with type 2 diabetes (T2D) accounting for approximately 90% of total diagnosis [1]. The prevalence of T2D in Spain is the second-highest in Europe [2], and the country ranks third in diabetes-related health expenditure [3].
Micro- and macrovascular complications are the major cause of morbidity and mortality in people with T2D (PwT2D) [4, 5] and are also responsible for most of medical costs incurred by patients [6]. Accordingly, a holistic, person-centered treatment approach is now being advocated with the goal of avoiding or delaying complications in the long term while maintaining PwT2D’s quality of life. This includes the management of blood glucose levels (HbA1c), weight, cardiovascular risk factors and comorbidities [7].
The achievement of glycemic control early in the course of the disease has proven to confer protection against the onset and progression of microvascular and macrovascular complications [8,9,10]. In line with this, a general objective of HbA1c < 7 is recommended, although more stringent targets are reasonable if they can be achieved safely, mainly in PwT2D with longer life expectancy [7].
In Europe, 50.9–98.6% of PwT2Ds have obesity [11]. There is a high correlation between excess body weight and T2D, with men and women with obesity having a 7- and 12-fold higher risk of developing T2D, respectively, than those with a normal body weight [12]. However, obesity is not only an important environmental factor involved in the etiopathogenesis of T2D but is also highly associated with the development of its complications [13]. Weight gain increases the risk of cardiometabolic complications [12], and weight loss has been demonstrated to improve risk factors for cardiometabolic disease and PwT2D’s quality of life [14]. Current treatment guidelines positioned weight loss as a primary target in the management of T2D in many cases and advocate for a minimum weight loss of 5 to 10% to obtain metabolic improvements [15]. In general, outcomes are better with greater weight loss, and reductions of higher magnitude (10–15%) have shown disease-modifying effects leading to diabetes remission [16].
Despite recommendations, keeping adequate levels of HbA1c, blood pressure (BP) and lipids is still challenging, and PwT2Ds often have poor metabolic control, with no significant improvements observed over the past few years. A cross-sectional study using yearly clinical data from 2007 to 2018 revealed that almost half of the Spanish population with T2D in Catalonia was under HbA1c targets. The proportion of PwT2D with HbA1c < 7% exhibited minimal changes over the years, barely shifting from 54.9% in 2007 to 55.9% in 2018. Additionally, the simultaneous attainment of all three targets (HbA1c, BP and lipids) showed improvement from 12.5 to 20.1% until 2013, but remained constant thereafter and was found to be insufficient [17]. Although lifestyle modifications, such as diet and increased physical activity, are recommended for PwT2D at all disease stages, they are challenging and often fail to achieve glycemic targets and weight loss goals; therefore, glucose-lowering medication with weight loss efficacy is often required [18, 19].
Evidence reporting outcomes associated with poor metabolic and/or weight control in T2D in our country exists, but studies focused on clinical, economic or HRQoL variables and no comprehensive systematic literature review summarizing all these aspects is available. Therefore, we set out the present systematic literature review to gather all available evidence describing the clinical, economic and HRQoL burden of poor metabolic and/or weight control in PwT2D in Spain.
Methods
Search Strategy
A comprehensive search strategy was carried out in international (MEDLINE/PubMed) and Spanish (Medicina en Español, Índice Bibliográfico Español en Ciencias de la Salud) databases and complemented with an extensive search of the gray literature. Terms used for this purpose are included in Supplementary Text 1.
Ethical approval was not required since this article is based on previously conducted research and does not contain any new studies involving human participants or animals.
Eligibility Criteria
Observational studies reporting clinical, economic and HRQoL outcomes based on the degree of metabolic/weight control in Spanish PwT2D were selected. Poor metabolic control was defined following the American Diabetes Association (ADA) as poor glycemic control (HbA1c ≥ 7%[53 mmol/mol]), and/or poorly controlled BP and/or hypertension (BP ≥ 140/90 mmHg) and/or poorly controlled low-density lipoprotein cholesterol (LDL-C) or dyslipidemia (LDL-C ≥ 100 mg/dl) [20]. Articles using alternative definitions for hypertension and dyslipidemia were also evaluated and included in the review. Poor weight control was defined as the presence of obesity, determined by a body mass index (BMI) ≥ 30 kg/m2.
Studies published in English and/or Spanish in the last 10 years (2013–2022) which were conducted in Spain or in several countries but provide data specific for the Spanish population with T2D were included. Conference abstracts from the last 2 years (2020–2022) were also considered in the review.
Clinical trials, economic evaluations, opinion articles, letters to the editor and narrative or systematic literature reviews were excluded as well as observational studies focusing on specific drugs.
Study Selection
The review was performed using search filters and standardized terms and followed the "Preferred Reporting Items of Systematic Reviews and Meta-analysis: The PRISMA Statement" guidelines [21]. Two reviewers independently screened all identified articles for the full-publication review. Discrepancies were resolved by consensus or with the involvement of a third team member.
Assessment of Study Quality
The quality of included publications was assessed using Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) quality score [22], with 22 points (100%) being the maximum score.
Data Extraction
Data were directly extracted from each study, and no formal statistical analysis was performed. Study variables analyzed in this review included: design (observational [case series or case study]; analytical [cross-sectional; case-control; retrospective and/or prospective cohorts]), sample size, main objectives and a summary of the most relevant results obtained by variable analyzed (clinical [macro- and microvascular complications, CVD, mortality, etc.], HRQoL and economic outcomes [healthcare resource use/associated medical costs/loss of productivity]).
Results
A total of 2282 potentially relevant titles were initially recovered. After duplicate removal (n = 170) and eliminating articles for other reasons, such as being outside of the defined time limit (n = 460) or not containing Spanish specific data (n = 164), 1488 titles were selected. Following analysis of the titles and abstracts, 1465 publications did not include information related to the search objective and 23 articles were considered for full-text review, 9 of which met study selection criteria and were finally included (Fig. 1).
Description of Studies Included in the Review
Of the nine selected observational studies, four had cross-sectional [23,24,25,26] and four retrospective [27,28,29,30] designs. Study design was not specified for one of the selected publications [31].
There were five studies that met ADA criteria for poor metabolic control [23,24,25, 28, 31]. Additionally, one defined the presence of poor BP control as PwT2D on antihypertensive therapy or with systolic/diastolic BP values ≥ 130/85 mmHg [27], one considered PwT2D with dyslipidemia as those on lipid-lowering therapy or with LDL-C values > 160 mg/dl [26], and an additional one did not provide a definition for dyslipidemia [27].
Regarding the correct communication of the information, two of the nine studies had 90.1% of STROBE items adequately reported [27, 29], followed by three others with 81.8% [23,24,25] and two additional one with 77.3% [26, 28]. Lower STROBE percentages (68.2%) were found for two of the selected publications [30, 31] because of the absence or lack of clarity around essential study aspects, such as study design, variables, statistical methods or sample size (Table 1).
Poor Metabolic Control in T2D
Poor Glycemic Control
Six of the nine selected studies compared clinical (n = 4), economic (n = 4) and HRQoL (n = 1) variables between adults with T2D and HbA1c levels < 7% with those who had poor glycemic control (HbA1c ≥ 7%) [23, 24, 26,27,28, 31] (Table 2 and S1).
Poor glycemic control in individuals with T2D was found to be associated with the presence of CVD (OR = 2.8) [27], hypoglycemia (OR = 1.6 in PwT2D 7% < HbA1c ≤ 8% vs. HbA1c < 7%) [31] or dyslipidemia (OR = 1.7) [26] (Fig. 2). Individuals with T2D and uncontrolled HbA1c levels had higher insulin needs (77.0% vs. 23.0%) and received combined therapy more often, with 70.5% and 77.4% requiring three or four antidiabetic medications, respectively, compared with 29.5% and 22.6% in those HbA1c < 7% [24]. Annual changes in body weight were significantly different between HbA1c controlled and not controlled groups, with those above glycemic control targets experiencing weight gains of higher magnitude (3.7 kg vs. 3.1 kg) and more frequently compared to HbA1c < 7% ones [24]. Available evidence also indicates poorer HRQoL, according to the Analysis of Diabetes-related Quality of Life (ADDQoL), reduced treatment satisfaction (Diabetes Treatment Satisfaction Questionnaire [DTSQ]) and increased fear of hypoglycemia (Hypoglycemia Fear Subscale [HFS-II]) in PwT2D with HbA1c > 7% [23].
Risk of CVD, hypoglycemia and dyslipidemia in uncontrolled vs. controlled normal weight individuals with T2D in Spain. *Results for 8% ≥ HbA1c > 7% group compared to HbA1c controlled individuals with T2D (HbA1c < 7%). Additional comparisons also available: 9% ≥ HbA1c > 8% vs. HbA1c < 7%, OR = 1.5; HbA1c > 9% vs. HbA1c < 7%, OR = 2.2. † Risk of dyslipidemia in those with a BMI ≥ 30 kg/m2. Orozco Beltrán et al. 2022 found a higher risk of dyslipidemia (OR = 1.6) in those of normal weight (BMI < 30 kg/m2)
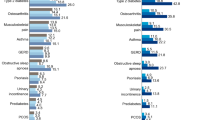
Total direct medical costs were significantly higher in PwT2D who were HbA1c ≥ 7% compared to those HbA1c < 7% with an additional cost per person-year ranging from + 251€ to + 712€ [27, 31] (Fig. 3). Hospitalization and medication costs were the main variables contributing to the yearly cost increment [28], which was observed to rise with poorer HbA1c control [31]. In addition, a higher proportion of patients required medical attention because of hypoglycemia episodes during the year prior to the analysis among those with poor glycemic control (8.9% vs. 4.6%) [24].
Annual direct medical costs in uncontrolled vs. controlled normal weight individuals with T2D in Spain. Studies reporting total direct costs are included in this graph. The studies exhibited heterogeneity in the variables considered for calculating total direct cost. Alonso Morán et al. (2014) also analyzed the economic burden of uncontrolled HbA1c showing additional costs per person-year in different groups vs. HbA1c < 7% population: 7% < HbA1c ≤ 8%, + 251.5€; 8% < HbA1c ≤ 9%, + 561.8€; HbA1c > 9%, + 447.5€. Differences between controlled and not controlled subgroups were significant
Infographic 1

Poor Blood Pressure Control
Three studies investigated clinical variables associated with poor BP control [25,26,27], one of which also examined its economic burden [27] (Table 3 and S2).
An association was identified between suboptimal BP control and the presence of CVD (OR = 1.5) or dyslipidemia (OR = 1.7) in individuals diagnosed with T2D [26, 27]. In addition, PwT2D and uncontrolled BP had worse metabolic control, higher BMI (30.9 vs. 30.2 kg/m2) and superior abdominal perimeter values (104.1 cm vs. 102.3 cm) [25].
The presence of poor BP was linked to significantly higher direct and total medical costs, which also accounted for productivity losses [27]. Total annual direct costs in PwT2D and uncontrolled BP were + 554€ higher (Fig. 3) compared to BP controlled ones, mainly driven by primary care cost (2831€ vs. 2238€).
Poor LDL Cholesterol or Dyslipidemia
The clinical burden of poor LDL-C control or dyslipidemia in PwT2D was evaluated in two of the selected publications [26, 27] (Table 4 and S3). Findings revealed that poorly controlled LDL-C or dyslipidemia was associated with the presence of CVD (OR = 1.7) and hypoglycemia (OR = 1.5) [27] (Fig. 2). Additionally, higher HbA1c levels (7.0 vs. 6.7) and triglycerides values (145.8 vs. 90.2) were observed in PwT2D who had dyslipidemia [26].
Poor weight control in T2D
A total of four studies analyzed clinical outcomes in PwT2D and poor weight control compared to those of normal weight [25,26,27, 30] (Table 5 and S4). The presence of a BMI ≥ 30 kg/m2 in PwT2D was linked to CVD disease (OR = 1.8) and hypoglycemia (OR = 1.7) (Fig. 2). A higher proportion of PwT2D were found to have metabolic syndrome (75.8% vs. 31.2%) [27] and suboptimal BP control (53.0% vs. 47.6%) [25] among those with poor weight control. Accordingly, a BMI < 30 kg/m2 was associated with a reduced risk of inadequate BP control (OR = 0.6) [30]. Contradictory findings were observed for the relationship between body weight and dyslipidemia. One study revealed higher likelihood of dyslipidemia in people with obesity (OR = 7.7) [26], whereas another study suggested that those with normal weight (BMI < 30 kg/m2) are more likely to have uncontrolled LDL-C values (OR = 1.6) [30].
Economic outcomes associated with poor weight control were assessed in one of the studies [27], which revealed significantly higher direct (3159€ vs. 2527€) and total medical costs (4915€ vs. 3431€) in PwT2D and a BMI ≥ 30 kg/m2, mostly due to increased primary care costs (2794€ vs. 2238€) [27] (Fig. 3). In addition, using a linear regression model, poor weight control was found to be associated with increased direct costs [27].
Infographic 2

Additional Findings
Poor Glycemic and Weight Control
Healthcare resource use and associated direct and total medical costs were higher in PwT2D HbA1c ≥ 8% with a BMI ≥ 30 kg/m2 than in those HbA1c < 7% with normal weight, mainly driven by an increase in primary care medical visits, medicines and hospitalization costs (Fig. 3 and table S5) [29].
Poor BP and weight control
PwT2D with uncontrolled BP and a BMI ≥ 30 kg/m2 showed a higher prevalence of metabolic syndrome, with percentages of people affected rising as BMI increase (only T2D group: 31.2%; T2D-high BP-overweight group: 78.7%; T2D-high BP-obesity group: 98.7%). In addition, a higher percentage of these PwT2D used antidiabetic medications or insulin compared to those controlled, following a similar trend (Table 6 and S5) [27].
Economic evaluations revealed an additional direct cost per person year of + 1239€ (Fig. 3) mostly driven by increased primary care costs (3355€ vs. 2238€) and significantly higher total costs (5201€ vs. 3431€) in PwT2D with poor BP and weight control [27].
Discussion
This systematic literature review provides a comprehensive summary of the influence of poor metabolic and weight control in Spanish adults with T2D using data from articles published within the last 10 years. A total of nine publications were identified, with seven of them exploring outcomes associated with the presence of poor metabolic control, while five addressed the burden of poor weight control in PwT2D. Retrieved article count was found to be low considering the extensive search period and the significance of the research question in the Spanish population with T2D. Data included in this review support that the presence of uncontrolled HbA1c levels, high BP, dyslipidemia and excess body weight in T2D pose a considerable burden on PwT2D and the healthcare system. This emphasizes the need to prioritize the management of these factors, considering the existing level of control in individuals with T2D in the Spanish setting. The fact that approximately half of the Spanish population with T2D remains outside control targets presents a clear chance for prevention strategies to mitigate the burden associated with this inadequate control [17].
Findings of this systematic literature review revealed an association between poor metabolic control and the presence of CVD, which aligns with existing literature across different research settings. Glycemic control has been described as a strong predictor of CVD and death in several long-term studies involving individuals with T2D [32,33,34].
Accordingly, data from the Swedish national Diabetes Register indicate considerable risk reductions for CVD and mortality associated with combined long-term improvement in HbA1c levels, systolic BP and ratio non-HDL: HDL in 13,477 PwT2D followed for a mean of 6.5 years. This study observed a 35% decrease in the risk of CVD linked to glycemic control, which rose to 56% when combined with BP control and further increased to 75% with the addition of non-HDL:HDL ratio control [35]. Therefore, data gathered in this review suggest that effective management of metabolic parameters could significantly reduce the risk of CVD and mortality among PwT2D.
The results of our study further support the well-established relationship between the presence of obesity in T2D and the development of CVD [36]. Accordingly, weight gain has been linked to an increased risk of cardiometabolic complications, and weight loss has been demonstrated to improve risk factors for CVD in a direct and linear fashion in individuals with T2D [37, 38]. Weight management should be a priority in the treatment of T2D and needs to be addressed early, preferably using weight-beneficial agents, with the aim of achieving weight loss goals ranging from 5 to 15% for many people [7, 19].
The identified studies reveal increased costs linked to poor metabolic and weight control, with increments per person-year in direct costs ranging from + 251 to + 1239€. Previous studies have already explored the economic burden of poor glycemic control in PwT2D, showing that delays in treatment intensification and the persistence of a suboptimal glycemic state translate into a significant increase in total annual costs [36, 39,40,41]. Liebl et al. reported that T2D costs are mainly driven by inpatient care for the treatment of complications (40–60% of total cost), with pharmacological therapy aimed at glycemic control accounting for 18% of the total cost. Therefore, early and strict glycemic control is required to prevent or delay these complications, promoting long-term health and reducing treatment costs [42]. Supporting the economic burden associated with the presence of excess body weight in T2D described in the present literature review, Karkare et al. found that weight loss was associated with significantly lower all-cause and T2D-related annual costs [43]. Weight control is crucial in T2D management programs, given its impact on people and the healthcare systems.
Our study presents some strengths, as the robust search strategy focused on collecting extensive and elaborated data from different databases. Therefore, most reviewed studies showed a high STROBE score, correctly reporting the information. Within the included studies, quantitative analyses of clinical, economic or HRQoL variables in controlled vs. uncontrolled PwT2D are presented, providing a comprehensive assessment of the extent to which poor control in T2D affects individuals and healthcare systems. Our study has limitations due to the observational nature of the investigations and the heterogeneity of the included studies (retrospective and cross-sectional designs). The definition of poor metabolic control among the studies included is inconsistent; therefore, the results may not be comparable. Thus, further high-quality prospective studies are necessary to accurately establish the consequences of poor metabolic control and/or weight control in Spanish adults with T2D.
Conclusion
Our study highlights the clinical, economic and HRQoL burden of poor metabolic and/or weight control in Spanish adults with T2D and reinforces the importance of prioritizing its control to reduce its associated burden, at both the individual and healthcare system levels.
Data Availability
All data generated or analyzed during this study are included in this published article/supplementary information files.
References
Association AD. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2021. Diabetes Care. 2020;44(Supplement_1):S15–33.
Rojo-Martínez G, Valdés S, Soriguer F, Vendrell J, Urrutia I, Pérez V, et al. Incidence of diabetes mellitus in Spain as results of the nation-wide cohort di@bet.es study. Sci Rep. 2020;10(1):2765.
WWD. With 1 in 7 adults living with diabetes, Spain has second highest diabetes prevalence in Europe. 2021.
Arnold SV, Khunti K, Tang F, Chen H, Nicolucci A, Gomes MB, et al. Impact of micro- and macrovascular complications of type 2 diabetes on quality of life: insights from the DISCOVER prospective cohort study. Endocrinol Diabetes Metab. 2022;5(2): e00321.
Mohammedi K, Woodward M, Marre M, Colagiuri S, Cooper M, Harrap S, et al. Comparative effects of microvascular and macrovascular disease on the risk of major outcomes in patients with type 2 diabetes. Cardiovasc Diabetol. 2017;16(1):95.
Li R, Bilik D, Brown MB, Zhang P, Ettner SL, Ackermann RT, et al. Medical costs associated with type 2 diabetes complications and comorbidities. Am J Manag Care. 2013;19(5):421–30.
Davies MJ, Aroda VR, Collins BS, Gabbay RA, Green J, Maruthur NM, et al. Management of hyperglycemia in type 2 diabetes, 2022. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2022;45(11):2753–86.
Sun S, Hisland L, Grenet G, Gueyffier F, Cornu C, Jaafari N, et al. Reappraisal of the efficacy of intensive glycaemic control on microvascular complications in patients with type 2 diabetes: A meta-analysis of randomised control-trials. Therapie. 2022;77(4):413–23.
Rodríguez-Gutiérrez R, Montori VM. Glycemic control for patients with type 2 diabetes mellitus: our evolving faith in the face of evidence. Circ Cardiovasc Qual Outcomes. 2016;9(5):504–12.
Laiteerapong N, Ham SA, Gao Y, Moffet HH, Liu JY, Huang ES, et al. The legacy effect in type 2 diabetes: impact of early glycemic control on future complications (The Diabetes & Aging Study). Diabetes Care. 2019;42(3):416–26.
Colosia AD, Palencia R, Khan S. Prevalence of hypertension and obesity in patients with type 2 diabetes mellitus in observational studies: a systematic literature review. Diabetes Metab Syndr Obes. 2013;6:327–38.
Wilding JP. The importance of weight management in type 2 diabetes mellitus. Int J Clin Pract. 2014;68(6):682–91.
Chobot A, Górowska-Kowolik K, Sokołowska M, Jarosz-Chobot P. Obesity and diabetes-Not only a simple link between two epidemics. Diabetes Metab Res Rev. 2018;34(7): e3042.
Lingvay I, Sumithran P, Cohen RV, le Roux CW. Obesity management as a primary treatment goal for type 2 diabetes: time to reframe the conversation. The Lancet. 2022;399(10322):394–405.
Association AD. Standards of medical care in diabetes—2019 Abridged for primary care providers. Clin Diabetes. 2019;37(1):11–34.
Lingvay I, Sumithran P, Cohen RV, le Roux CW. Obesity management as a primary treatment goal for type 2 diabetes: time to reframe the conversation. Lancet. 2022;399(10322):394–405.
Mata-Cases M, Vlacho B, Real J, Puig-Treserra R, Bundó M, Franch-Nadal J, et al. Trends in the degree of control and treatment of cardiovascular risk factors in people with type 2 diabetes in a primary care setting in Catalonia during 2007–2018. Front Endocrinol (Lausanne). 2021;12: 810757.
Rise MB, Pellerud A, Rygg L, Steinsbekk A. Making and maintaining lifestyle changes after participating in group based type 2 diabetes self-management educations: a qualitative study. PLoS ONE. 2013;8(5): e64009.
Meneghini LF, Orozco-Beltran D, Khunti K, Caputo S, Damci T, Liebl A, et al. Weight beneficial treatments for type 2 diabetes. J Clin Endocrinol Metab. 2011;96(11):3337–53.
Standards of Medical Care in Diabetes-2016 Abridged for Primary Care Providers. Clin Diabetes. 2016;34(1):3–21.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71.
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61(4):344–9.
de Pablos-Velasco P, Salguero-Chaves E, Mata-Poyo J, DeRivas-Otero B, García-Sanchez R, Viguera-Ester P. Calidad de vida y satisfacción con el tratamiento de sujetos con diabetes tipo 2: resultados en España del estudio PANORAMA. Endocrinología y Nutrición. 2014; p. 18–26.
Perez A, Mediavilla JJ, Miñambres I, González-Segura D. Control glucémico en pacientes con diabetes mellitus tipo 2 en España. Revista Clínica Española. 2014; p. 429–36.
Barquilla García A, Llisterri Caro JL, Prieto Díaz MA, Alonso Moreno FJ, García Matarín L, Galgo Nafría A, et al. Control de la presión arterial en una población hipertensa diabética asistida en atención primaria: Estudio PRESCAP-Diabetes 2010. SEMERGEN - Medicina de Familia. 2015;41(1):13–23.
Díaz Vera AS, Abellán Alemán J, Segura Fragoso A, Martínez de Esteban JP, Lameiro Couso FJ, Golac Rabanal MDS, et al. The prevalence and risk factors associated with dyslipidemia in type 2 diabetic patients in the autonomous Region of Cantabria. Endocrinol Diabetes Nutr (Engl Ed). 2020;67(2):102–12.
Sicras-Mainar A, Navarro-Artieda R, Ibáñez-Nolla J. Características clínicas y económicas asociadas a la diabetes tipo 2. Revista Clínica Española. 2014; p. 121–30.
Mata-Cases M, Casajuana M, Franch-Nadal J, Casellas A, Castell C, Vinagre I, et al. Direct medical costs attributable to type 2 diabetes mellitus: a population-based study in Catalonia. Spain Eur J Health Econ. 2016;17(8):1001–10.
Díaz-Cerezo S, Romera I, Sicras-Mainar A, López-Simarro F, Dilla T, Artime E, et al. Resource use and costs in patients with poorly controlled type 2 diabetes mellitus and obesity in routine clinical practice in Spain. Curr Med Res Opin. 2020;36(9):1449–56.
Orozco-Beltrán D, Cinza-Sanjurjo S, Escribano-Serrano J, López-Simarro F, Fernández G, Gómez García A, et al. Adherence, control of cardiometabolic factors and therapeutic inertia in patients with type 2 diabetes in the primary care setting. Endocrinol Diabetes Metab. 2022;5(2): e00320.
Alonso-Morán E, Orueta JF, Nuño-Solinís R. Incidence of severe hypoglycaemic episodes in patients with type 2 diabetes in the Basque country: impact on healthcare costs. BMC Health Serv Res. 2015;15:207.
Rawshani A, Rawshani A, Franzén S, Sattar N, Eliasson B, Svensson AM, et al. Risk factors, mortality, and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2018;379(7):633–44.
Control G, Turnbull FM, Abraira C, Anderson RJ, Byington RP, Chalmers JP, et al. Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia. 2009;52(11):2288–98.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577–89.
Eeg-Olofsson K, Zethelius B, Gudbjornsdottir S, Eliasson B, Svensson AM, Cederholm J. Considerably decreased risk of cardiovascular disease with combined reductions in HbA1c, blood pressure and blood lipids in type 2 diabetes: Report from the Swedish National Diabetes Register. Diab Vasc Dis Res. 2016;13(4):268–77.
Perone F, Pingitore A, Conte E, Halasz G, Ambrosetti M, Peruzzi M, et al. Obesity and cardiovascular risk: systematic intervention is the key for prevention. Healthcare. 2023;11(6):902.
Chobot A, Gorowska-Kowolik K, Sokolowska M, Jarosz-Chobot P. Obesity and diabetes-Not only a simple link between two epidemics. Diabetes Metab Res Rev. 2018;34(7): e3042.
Ryan DH, Yockey SR. Weight loss and improvement in comorbidity: differences at 5%, 10%, 15%, and over. Curr Obes Rep. 2017;6(2):187–94.
Lindvig A, Tran MP, Kidd R, Tikkanen CK, Gæde P. The economic burden of poor glycemic control associated with therapeutic inertia in patients with type 2 diabetes in Denmark. Curr Med Res Opin. 2021;37(6):949–56.
Ali SN, Dang-Tan T, Valentine WJ, Hansen BB. Evaluation of the clinical and economic burden of poor glycemic control associated with therapeutic inertia in patients with type 2 diabetes in the United States. Adv Ther. 2020;37(2):869–82.
Hellgren M, Svensson A-M, Franzén S, Ericsson Å, Gudbjörnsdottir S, Ekström N, et al. The burden of poor glycaemic control in people with newly diagnosed type 2 diabetes in Sweden: a health economic modelling analysis based on nationwide data. Diabetes Obes Metab. 2021;23(7):1604–13.
Liebl A, Khunti K, Orozco-Beltran D, Yale JF. Health economic evaluation of type 2 diabetes mellitus: a clinical practice focused review. Clin Med Insights Endocrinol Diabetes. 2015;8:13–9.
Karkare S, Fridman M, Dang-Tan T, Lu J, Smolarz B, DeKoven M, et al. Effect of weight change on economic outcomes among persons with type 2 diabetes mellitus in the United States: beyond glycemic control. J Manag Care Spec Pharm. 2019;25:1–12.
Acknowledgements
The authors thank Marta Comellas, Neus Vidal and Patricia Felip, who received honoraria for their assistance in the project and medical writing and data analysis tasks.
Funding
Lilly SAU sponsored this study and the publication of this article. The Rapid Service Fee was also funded by Lilly SAU.
Author information
Authors and Affiliations
Contributions
Irene Romera, Miriam Rubio-de Santos and Silvia Díaz-Cerezo contributed to the conception of the work. Jennifer Redondo-Antón, Irene Romera, Luis Lizán, Miriam Rubio-de Santos and Silvia Díaz-Cerezo contributed to the design and data analysis. Antonio Pérez, Jennifer Redondo-Antón, Irene Romera, Luis Lizán, Miriam Rubio-de Santos, Silvia Díaz-Cerezo, and Domingo Orozco-Beltrán contributed to data interpretation and manuscript writing. All authors participated in the critical revision of the manuscript and approved the final submitted version.
Corresponding author
Ethics declarations
Conflict of Interest
Antonio Pérez reports honoraria from Sanofi, Boehringer Ingelheim, Menarini, Novo Nordisk, Lilly, Pfizer, Amarin, Daiichi Sankyo, AstraZeneca, Almirall, Novartis, Merck Sharp & Dohme, Amgen, AMARIN and Esteve outside of the submitted work.
Jennifer Redondo-Antón, Irene Romera, Miriam Rubio-de Santos and Silvia Díaz-Cerezo are employees and minor shareholders of Eli Lilly. Luis Lizán works for an independent scientific consultancy (Outcomes’10) that has received honoraria for conducting the systematic literature review and writing the manuscript for conducting the systematic literature review and manuscript writing tasks. Domingo Orozco-Beltrán reports honoraria from MSD, Lilly and Novo Nordisk outside of the submitted work.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Pérez, A., Redondo-Antón, J., Romera, I. et al. Disease and Economic Burden of Poor Metabolic and Weight Control in Type 2 Diabetes in Spain: A Systematic Literature Review. Diabetes Ther 15, 325–341 (2024). https://doi.org/10.1007/s13300-023-01503-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01503-4