Abstract
The increasing burden of type 2 diabetes (T2D), in relation to alarming rise in the prevalence; challenges in the diagnosis, prevention, and treatment; as well as the substantial impact of disease on longevity and quality of life, is a major concern in healthcare worldwide. Sulfonylureas (SUs) have been a cornerstone of T2D pharmacotherapy for over 60 years as oral antidiabetic drugs (OADs), while the newer generation SUs, such as gliclazide modified release (MR), are known to be associated with low risk of hypoglycemia in addition to the cardiovascular neutrality. This scoping review aimed to specifically address the current position of gliclazide MR among other SUs in the contemporary treatment paradigm for T2D and to provide a practical guidance document to assist clinicians in using gliclazide MR in real-life clinical practice. The main topics addressed in this paper include the role of early and sustained glycemic control and use of SUs in T2D management, the properties of gliclazide MR in relation to its effectiveness and safety, the use of gliclazide therapy in special populations, and the place of SUs as a class and gliclazide MR specifically in the current T2D treatment algorithm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Sulfonylureas (SUs) have been a cornerstone of type 2 diabetes (T2D) pharmacotherapy for over 60 years as oral antidiabetic drugs (OADs), due to their ability to stimulate insulin release from pancreatic β cells, ameliorating the effect of β cell dysfunction and efficacy in achieving target glycemic control in patients who failed on metformin monotherapy, as well as their tolerability over time and their affordability. |
Newer generation SUs are important in this regard, given that they are preferred over other SUs in terms of lower risk of hypoglycemia and cardiovascular neutrality. |
Gliclazide MR is proven to offer high efficacy for glucose lowering, low risk of hypoglycemia, weight neutrality, cardiovascular safety, delay of secondary treatment failure, and favorable renal outcomes in patients with chronic kidney disease. |
Accordingly, guidelines should consider the prioritization of newer generation SUs such as gliclazide in the treatment algorithm instead of combined assessment of all SUs in the same category. |
Introduction
Type 2 diabetes (T2D) is a complex disease influenced by multiple genetic and environmental risk factors, while the failure of β cells is considered to be one of the core events in the etiopathogenesis [1, 2]. The natural history of adaptation of β cells to diabetes proceeds through the phases of susceptibility (genetic factors, fetal environment, and postnatal nutrition), initial adaptation (temporary β cell mass expansion in response to insulin resistance), and eventual failure [2].
Accordingly, given the association of prolonged hyperglycemia with vicious circle of continuous deterioration of β cell function and an increased risk of diabetes-related complications and mortality [3,4,5], effective early glycemic control is considered critical to achieve sustained and long-term reductions in diabetes-related complications and thereby to reduce mortality [6, 7].
Sulfonylureas (SUs) have been a cornerstone of T2D pharmacotherapy for over 60 years as oral antidiabetic drugs (OADs), due to their ability to stimulate insulin release from pancreatic β cells, ameliorating the effect of β cell dysfunction and efficacy in achieving target glycemic control in patients who failed on metformin monotherapy, as well as their tolerability over time and their affordability [8,9,10,11]. Nonetheless, despite their well-established efficacy, there is no consensus among global experts and modern guidelines regarding the priority of SUs in relation to other second-line therapeutic options, particularly due to concerns regarding the hypoglycemia risk and cardiovascular safety [8, 12].
Newer generation SUs, such as gliclazide modified release (MR), are important in this regard, given that they are preferred over other SUs in terms of lower risk of hypoglycemia as well as the cardiovascular neutrality [13,14,15,16,17]. In fact, gliclazide is also involved along with metformin and insulin in the World Health Organization (WHO) model list of essential medicines 2021 under medicine for diabetes [18]. However, few guidelines on diabetes management specifically suggest gliclazide as the second-line treatment, instead of SUs as a class, despite it offering a low risk of hypoglycemia, weight neutrality, cardiovascular safety, delay of secondary treatment failure, and favorable renal outcomes in patients with chronic kidney disease (CKD) [19, 20].
This scoping review aimed to specifically address the current position of gliclazide MR among other SUs in the contemporary treatment paradigm for T2D and to provide a practical guidance document to assist clinicians in using gliclazide in real-life clinical practice.
Methods
This scoping review was prepared by an expert panel of endocrinology specialists. The current position of SUs as a class, and gliclazide MR specifically, with respect to changing treatment algorithms in the contemporary management of T2D was discussed, as supported by scientific evidence and expert clinical opinion. The literature search involved PubMed/Medline, Scopus, and Web of Science from inception to June 2023 for studies that assessed the gliclazide and sulfonylureas in the management of type 2 diabetes, using the keywords: “sulfonylurea” OR “gliclazide MR” AND “type 2 diabetes mellitus” in addition to “mechanism of action,” “oral antidiabetics,” “metformin,” “DPP-4 inhibitors,” “SGLT2 inhibitor,” “GLP-1 receptor agonist,” “thiazolidinedione,” “insulin,” “monotherapy,” “combination therapy”, glycemic control”, “sustainability”, “glycemic variability”, “hypoglycemia risk,” “weight gain,” “antioxidant,” “macrovascular complications,” “cardiac mortality,” “morbidity,” “efficacy,” “safety,” “elderly,” “renal disease,” “hepatic disease,” “Ramadan fasting,” and “monogenic diabetes.” In accordance with the scope of an expert panel-based narrative review, the inclusion of studies was limited to provide the scientific evidence within the context of topics addressed in the paper. Following four meetings (three online and the final face-to-face meeting), the panel critically analyzed recommendations from international guidelines, systemic reviews, and meta-analyses; results of randomized control trials (RCTs); and studies focusing on the efficacy/effectiveness and safety of gliclazide MR, and agreed on a series of statements supported by scientific evidence and expert clinical opinion. The main topics addressed in this paper include (a) early and sustained glycemic control in T2D management, (b) SUs in T2D management (history, general mechanisms of action, diabetes treatment), (c) gliclazide MR (new formulation allowing once daily dosing, low hypoglycemia risk, antioxidant effects, cardiovascular neutrality, low risk of secondary failure, and the longer sustainability of glycemic control), (d) clinical studies on efficacy/effectiveness of gliclazide MR (as monotherapy or in combination with metformin, compared with other SUs or OADs, as add-on to insulin therapy), (e) clinical studies on safety of gliclazide (all-cause and cardiac mortality and macrovascular complications, hypoglycemia risk, weight gain), (f) use of gliclazide therapy in special populations (patients with renal or hepatic diseases, elderly, during Ramadan fasting, in monogenic diabetes), and (g) place of SUs as a class and gliclazide MR specifically in the current T2D treatment algorithm.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Critical Role of Early and Long-Term Glycemic Control in the Management of Type 2 Diabetes
Given the causal link between prolonged glucose load, diabetes-related complications, and premature deaths, attainment of effective early glycemic control and its long-term maintenance are of critical importance in the management of patients with T2D [4,5,6,7, 21].
As per several guidelines and consensus reports, the standard treatment for T2D involves use of metformin in conjunction with lifestyle changes as first-line glucose lowering therapy, while early and prompt transition to combination therapies is recommended in case of failure to achieve glycemic control [22,23,24]. The rationale behind these recommendations is the data from clinical trials in the diabetes setting that indicated the association of achieving early intensive glycemic control [hemoglobin A1c (HbA1c) levels < 7%] with the decrease in microvascular complications, as well as the sustained microvascular favorable outcome and emergent macrovascular benefits in the long term [4, 21, 25, 26].
In the United Kingdom Prospective Diabetes Study (UKPDS) conducted in newly diagnosed patients with T2D, intensive treatment (insulin and SU-based therapies) was associated with significant reduction in microvascular complications by 25% (p = 0.0099) and a nearly significant decrease in macrovascular complications by 16% (p = 0.052) compared with conventional treatment at 1 year [27]. Over 10 years, intensive treatment was found to yield a lower HbA1c (median 7.0% versus 7.9%) than the conventional treatment [27]. Between-group differences in HbA1c was lost 1 year after the completion of study. After a median 10 years of extended follow-up, relative risk reduction of 24% persisted for microvascular complications, while risk reduction for macrovascular endpoints became apparent with significantly lower rates of myocardial infarction (MI) by 15% (p = 0.01) and mortality by 13% (p = 0.007) than the conventional treatment [7].
The efficacy of early glycemic control in reducing diabetes-related complications was also reported in the 10-year follow-up of the Veterans Affairs Diabetes Trial (VADT) for microvascular and macrovascular complications [28], in the ADVANCE study and 10-year extension ADVANCE–ON studies for microvascular complications (nephropathy in particular) [29, 30], and in the STENO-2 trial in terms of decrease in cardiovascular and all-cause mortality at 13.2 years follow-up [31] and 7.9 years of gain of life at 21.2 years of follow-up [32].
The sustained benefits of earlier intensive multifactorial intervention demonstrated in the STENO-2 trial are considered to reflect a direct consequence of early treatment intensification in patients at lower absolute risk rather than a later treatment intensification in patients with already established vascular damage [31]. Likewise, in a cohort study with 271,174 patients with T2D and 1,355,870 age- and gender-matched controls, an HbA1c level outside the target range was reported to be the strongest predictor of stroke and acute MI in patients with T2D [33]. Also, data from the UKPDS 35 study revealed the association of each 1% reduction in mean HbA1c with a 37%, 43%, 21%, 14%, and 12% reduction in microvascular complications; amputation and fatal peripheral artery disease; and deaths related to diabetes, MI, and stroke, respectively [25]. Furthermore, in patients with newly diagnosed T2D, having HbA1c levels ≥ 6.5% for the first year after the diagnosis was reported to be associated with higher rate of microvascular and macrovascular complications within the 10-year period [34].
Achievement of optimal glycemic control via provision of the intensive treatment in the early period of diabetes is suggested to induce a metabolic memory, enabling maintenance of reduced risk of complications in the following years, even in the case of failure to continue intensive therapy [26, 35]. Notably, recent studies have reported a legacy effect (better long-term outcomes) associated with early achievement of glycemic control glucose in terms of macrovascular and mortality outcomes [34, 36, 37]. Also, in the new analysis of UKPDS 44-year follow-up data of 1525 patients (mean follow-up 18.6 years), benefits of early intensive glycemic control with SU/insulin versus conventional glucose control on all endpoints were found to be maintained over the 44-years follow-up, including any diabetes-related endpoints (10% RR, HR 0.90, p = 0.016), MI (15% RR, HR 0.85, p = 0.0074), microvascular disease (26% RR, HR 0.74, p < 0.0001), and all-cause mortality (11% RR, HR 0.89, p = 0.0093), confirming the continuum of the legacy effect [38].
Nonetheless, given the growing data on the impressive reduction in cardiovascular outcomes with use of antidiabetic agents that offer cardiorenal protection independent of the glycemic control, both glucocentric and cardiocentric approaches seem to be equally important in reducing the risk of complications to the greatest extent possible [35, 38, 39].
Sulfonylureas in Type 2 Diabetes Treatment
History of SUs
The history of SUs began in 1937 with the discovery of the hypoglycemic activity of synthetic sulfur containing compounds by Ruiz et al. [40, 41]. Their efficacy on stimulation of β cell release of insulin was acknowledged in 1946, and within a decade they were introduced as the first OADs in the treatment of diabetes [8, 42, 43]. Starting from 1950s with tolbutamide and followed by the introduction of the other first-generation agents (chlorpropamide, acetohexamide, and tolazamide), more potent second-generation agents [gliclazide, glipizide, gliquidone, and glibenclamide (glyburide)] and the third-generation agents [glimepiride, gliclazide MR, and glipizide MR/extended release (XL)], SUs have become the most widely distributed OADs in mainstream clinical practice for more than 60 years [8, 14, 40,41,42,43] (Table 1).
SUs can also be classified either according to their hierarchy of development (conventional and newer-generation SUs) or based on the duration of action (short-, intermediate-, and long-acting) [13]. Conventional SUs include tolbutamide, chlorpropamide, glibenclamide, and glipizide, while newer-generation safer and more potent SUs include glimepiride, gliclazide, gliclazide MR, and glipizide MR/XL [13] (Table 1).
Mechanisms of Sulfonylurea Action
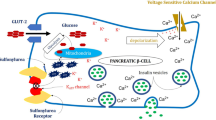
In basal physiological conditions, insulin release from β cells is considered to be minimal and the β cell membrane remains hyperpolarized. Upon glucose entry into the pancreatic β cells through the glucose transporter GLUT2, a cascade of events starts that finally leads to insulin secretion, including metabolization of intracellular glucose, increase in intracellular ATP levels, inhibition of the ATP-sensitive potassium (KATP) channels, depolarization of the β cell membrane, Ca2+ influx via opening of voltage-gated Ca2+ channels, and a rise in cytosolic Ca2+, which stimulates the exocytosis of insulin-containing secretory granules [11, 44].
SUs, by binding to the sulfonylurea receptor (SUR) subunit of the KATP channel, also induce channel closure and membrane depolarization, independent of the intracellular glucose entry, followed by a similar cascade of intracellular events until insulin secretion [11, 44, 45] (Fig. 1).
The KATP channel is a hetero-octameric complex of two different types of protein subunits, including an inwardly rectifying K+ channel (Kir6.x: Kir6.1, Kir6.2) and a SUR (SUR1, SUR2A, SUR2B) [44, 45]. KATP channels, with different subunit composition, are also found in cardiac, skeletal, and smooth muscle. Opening of KATP channels in response to metabolic stress leads to inhibition of electrical activity in all these tissues [11]. Basically, Kir6.2 serves as the pore-forming subunit and associates with SUR1 in pancreatic β cells and with SUR2A in cardiomyocytes and skeletal muscle, while Kir6.1 associates with SUR2B in vascular smooth muscle [11]. This variation in the subunit composition of the KATP channel accounts for the different metabolic and drug sensitivities of KATP channels in different cells [11]. SUs (i.e., tolbutamide, gliclazide, glimepiride) close KATP channels by binding with high affinity to SUR and also interact with Kir6.2, but with low affinity [11].
In fact, while SUs are categorized as a drug class, they substantially differ in terms of individual SUR subtype specificity and affinity, determining their hypoglycemia risk and effects on ischemic preconditioning [14, 43]. Hence, individual SU expresses a different selectivity for SUR1 (β cell type), SUR2A (myocardial), and SUR2B (vascular smooth muscle) receptors [14, 43]. Gliclazide and glipizide specifically bind and block channels containing SUR1 (β cell type), but not SUR2 (cardiac, smooth muscle types), whereas glibenclamide and glimepiride can nonspecifically bind and block both types of SUR1- and SUR2-containing channels [11, 14, 43]. Accordingly, both glimepiride and glibenclamide carry the potential to mediate effects both at the level of heart and smooth muscles. However, only glibenclamide possesses the characteristics to abolish ischemic preconditioning, a cardioprotective phenomenon limiting ischemic injury or infarct size after repeated episodes of ischemia, via blocking the cardiac myocyte KATP channels [11, 14, 43] (Fig. 1).
Different selectivity for SUR1 (β cell type), SUR2A (myocardial), and SUR2B (vascular smooth muscle) receptors expressed by individual SUs. Gliclazide and glipizide specifically bind and block channels containing SUR1 (β cell type), but not SUR2 (cardiac, smooth muscle types). Glibenclamide (abolishes ischemic preconditioning via blocking cardiac myocyte KATP channels) and glimepiride (no effect on cardiac myocyte KATP channels or ischemic preconditioning) can nonspecifically bind and block both types of SUR1- and SUR2-containing channels and carry the potential to mediate effects both at the level of heart and smooth muscles.
SUs also inhibit glucagon secretion from pancreatic α cells via direct stimulation of somatostatin release through a similar mechanism as well as with the paracrine effect of increased insulin [43]. In this way, they provide glycemic control by increasing insulin release, by decreasing the hepatic glucose output via decreased glucagon release, and by reducing the hepatic uptake and the metabolic clearance rate of insulin [43].
SUs in Diabetes Treatment
The glucose-lowering efficacy of SUs (mean HbA1c reduction of 1.0–1.5%) is similar to metformin and glucagon-like peptide-1 (GLP-1) receptor agonists, and better than newer agents such as sodium-glucose cotransporter-2 (SGLT2) inhibitors (mean HbA1c reduction of 0.5–1.0%) and dipeptidyl peptidase-4 (DPP-4) inhibitors (mean HbA1c reduction of 0.4–0.9%) [13, 20, 39, 46,47,48].
Newer generation SUs (glimepiride and gliclazide MR) are considered effective and safe as second-line agents in patients with metformin failure, and also as first-line agents if used in combination with lifestyle modification and metformin in patients with a baseline HbA1c ≥ 7.5% [13, 40, 47]. They also offer glucose-lowering efficacy with minimal side effects, when used as dual or triple therapy in combination with other antidiabetic agents [13, 39, 46, 49].
Notably, all SUs are not similar in terms of their efficacies, adverse effects, and tolerability. Newer generation SUs such as gliclazide MR offer more favorable efficacy and safety profiles. However, clinical utility of SUs and their place in diabetes treatment are being surpassed by newer, more expensive therapies; however, SUs are valuable treatment option for many patients with T2D, particularly in the resource-limited settings [10, 13, 39, 46, 50].
Gliclazide MR: Distinguishing Pharmacological Properties
New Formulation Allowing Once Daily Dosing
Gliclazide MR is a gliclazide formulation with a hydrophilic matrix allowing once daily dosing regimen taken at lower dose (30–120 mg/day) than gliclazide (80–320 mg/day, twice daily) to be effective over 24 h [16, 17, 51, 52]. In fact, gliclazide MR 60 mg is currently the only long-acting once-daily OAD that is scored and breakable into two halves while retaining the long-acting once-daily characteristic, which is considered to further improve the patient compliance besides once-daily dosing [53].
Gliclazide MR is considered a SU of reference due to its efficacy in achieving glycemic control in monotherapy or in combination with metformin without increasing the risk of hypoglycemia or cardiovascular mortality [12, 14, 16, 17, 53,54,55].
Antioxidant Effects
Oxidative stress due to excess free radical activity and glycation plays a pivotal role in the development of microvascular and macrovascular diabetes complications [56].
Gliclazide MR also differs from other SUs in terms of having antioxidant effects both in vessel wall and β cells, which is explained by its azabicyclo-octyl ring serving as a free radical scavenger [56,57,58]. Hence, gliclazide is considered likely to offer β cell protection by causing a reduction in glucose-dependent mitochondrial stress and apoptosis in β cells, preventing hydrogen peroxide-mediated oxidative damage, as well as by improving the nitric oxide-mediated vasodilation and ameliorating the endothelial stress not only in atherosclerotic vessels but also in the retinal and renal vasculature [58,59,60,61,62]. Consistent with the anti-atherogenic effect, gliclazide was reported to significantly and independently reduce the progression of carotid artery intima-media thickness (IMT) as a result of its antioxidant properties compared with glibenclamide monotherapy [63]. Also, in a study assessing the effects of 10 months of oral gliclazide therapy on oxidative parameters in 44 type 2 diabetic patients, gliclazide but not other SUs (glibenclamide, glimepiride, glipizide, or tolbutamide), was found to inhibit low-density lipoprotein oxidation and enhanced the total plasma antioxidant capacity [64].
Indeed, some studies also reported beneficial effects of gliclazide (i.e., stimulation of endothelial prostacyclin synthesis, restoration of endothelial function and reduction of platelet reactivity) on the hemorheological abnormalities seen in diabetic vascular disease [63, 65]. These effects, which are independent of glycemic control and not seen with other SUs, have been related to a reduction in oxidative stress [65], and suggested to mediate the effect of gliclazide on the carotid artery IMT and the attenuation of the progression of atherosclerosis in patients with T2D [63].
Glycation and oxidation are inextricably linked, while the increased glycation and oxidation are central to the pathogenesis of diabetic vascular disease [63, 65]. In this regard, acting both as a blood glucose lowering agent and a free radical scavenger, gliclazide may be more effective than other SUs in preventing diabetes-related macrovascular complications [63, 65].
Low Hypoglycemia Risk
SUs, as a general class, are considered to yield an increased risk of hypoglycemic events than the potentially nonhypoglycemic antidiabetic agents (i.e., DPP-4 inhibitors, SGLT2i and TZD) [48, 66, 67]. However, studies of gliclazide indicate its association with a significantly lower risk of hypoglycemic events than other SUs, and a risk similar to other insulinotropic agents [54, 55, 68, 69].
The absence of the benzamide structure in the gliclazide molecule, which exists in all other SUs, enables the gliclazide to bind the SUR1 receptor reversibly [57, 70, 71]. Gliclazide also differs from other SUs in terms of its effect on insulin release pattern by enabling restoration of early insulin peak in response to glucose stimulation, which translates into reduced pancreatic stimulation and a reduced incidence of hypoglycemia [14, 15, 72]. The restoration of the early insulin peak in response to glucose stimulation and the stronger reversible binding of gliclazide MR to the β cell’s SUR1 receptor compared with other SUs are considered to enable lesser pancreatic overstimulation and the lower hypoglycemia risk [13,14,15, 72].
Gliclazide is also the only SU that does not bind or activate exchange protein directly activated by cAMP (Epac2), a stimulating factor for insulin exocytosis from the readily releasable granule pool, which may also confer a lower risk of hypoglycemia [13, 43, 50].
Cardiovascular Neutrality
The cardiovascular neutrality of gliclazide represents another distinct advantage over other SUs [14]. Compared with other SUs, gliclazide binds more selectively to SUR1 (β cell type) and shows reversible binding (due to lack of benzamide moiety), along with its lower affinity to SUR2A (myocardial) and SUR2B (vascular smooth muscle) receptors [19].
The SUR1 specificity of gliclazide MR is important in terms of cardioprotective efficacy, since the affinity for extra-pancreatic SURs is hypothesized to inhibit ischemic preconditioning that translates into varying degrees of adverse hypoglycemia and cardiovascular outcomes with some SUs [8, 11, 73,74,75]. In a population-based T2D study, SUs with high versus low cardiac mitochondrial KATP channel affinity were associated with a significantly increased risk of three-point major adverse cardiovascular events (MACE), ischemic stroke, and cardiovascular death [76].
Hence, in contrast to other SUs inhibiting the opening of KATP channels and thus increasing the risk of cardiovascular ischemia due to their high affinity for SUR2 receptors, gliclazide as a pancreas-selective SU with low SUR2 binding does not confer an increased risk of cardiovascular events or mortality [8, 19, 77, 78]. Lower incidence of hypoglycemic events and less weight gain on gliclazide compared with other SUs are also proposed as an explanation for a possible favorable cardiovascular safety profile [14].
Low Risk of Secondary Failure and the Longer Sustainability of Glycemic Control
The oxidative stress and subsequent β cell apoptosis are associated with a secondary failure and thus less effective glycemic control over time [79]. While the effects of SUs, as a general class, on β cell exhaustion have always been a major concern, gliclazide is considered to enable a lower rate of secondary failure and thus longer sustainability of glycemic control than other SUs due to its antioxidant and anti-apoptotic properties, as well as its higher physiological action in β cell insulin release [79, 80].
Accordingly, the secondary failure rate over 5 years in patients with T2D was reported to be lowest with gliclazide (7%) compared with glibenclamide (17.9%) and glipizide (25.6%) [81], while gliclazide was also reported to be more protective against secondary β cell failure than glibenclamide, delaying the initiation of insulin by 7 years (8 years for gliclazide and 15 years for glibenclamide) [80]. These findings suggest that gliclazide as a potent SU with low rate of secondary failure and a low incidence of side effects may represent a better choice in long-term SU therapy [80].
Studies on the Efficacy/Effectiveness of Gliclazide MR
Adequate glycemic control with gliclazide, regardless of baseline HbA1c, has been consistently reported [17, 29, 53, 81, 82]. A summary of gliclazide efficacy trials is provided in Table 2.
Monotherapy or in Combination with Metformin
In the real-world EASYDia trial in 7170 patients with T2D with a broad range of HbA1c, body mass index (BMI), and background glucose-lowering therapy, 6-month use of 30–120 mg of gliclazide MR 60 mg as first-line, second-line, or switch therapy was reported to be associated with a 1.98%, 1.74%, and 1.61% reduction in HbA1c levels, respectively [81]. Also, 65.3% of the overall study population and nearly half of the treatment-naïve patients successfully achieved an HbA1c ≤ 7.0% target at 3 months with gliclazide MR [81].
In the Xrise study with patients with T2D uncontrolled on diet alone or metformin monotherapy, 4-month use of 60–120 mg of gliclazide MR 60 mg as monotherapy or in combination with metformin was reported to yield a 1.5% reduction in HbA1c levels, while FPG levels were decreased more by the increasing doses of gliclazide MR (78.3 mg/dL, 80.1 mg/dL, and 106.5 mg/dL for 60 mg, 90 mg, and 120 mg daily doses, respectively) [53].
In a study with 507 patients with T2D (40% aged over 65 years and 20% with mild-to-moderate renal impairment), gliclazide MR alone or combined with other OADs, was associated with a significant reduction in HbA1c (by −0.46% total, −0.95% in diet-failed patients, −0.34% in OAD-pretreated patients, −0.43% in monotherapy, and −0.51% in combination therapy), even in the elderly patients and in those with impaired renal function [17].
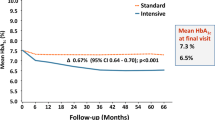
In the ADVANCE study, standard glucose control versus intensive glucose control (gliclazide MR plus other drugs as required to achieve a glycated hemoglobin ≤ 6.5%) was assessed in 11,140 patients with T2D [29]. After a median of 5 years of follow-up, the mean HbA1c level was reported to be lower in the intensive-control versus standard-control group (6.5% versus 7.3%) along with maintenance of ≤ 7% HbA1c target by 80% of patients in the intensive group [32].
In an epidemiologic surveillance protocol study of 60 days in 759 patients with T2D, one to two tablets of gliclazide 60 mg + metformin 500 mg treatment was associated with the achievement of the primary outcome (FPG of 90–130 mg/dL) by 62.5% of patients and the frequency of hypoglycemia was 0.7%, while the FPG decreased from baseline by 48.7 mg/dL, 71.3 mg/dL, and 86.3 mg/dL with 1 tablet, 0.5 tablet, and 2 tablets, respectively [82].
Compared with Other SUs or OADs
In a double-blind, randomized, active-controlled study on the efficacy and safety of 2-year monotherapy with vildagliptin 50 mg bid versus gliclazide up to 320 mg/day in 546 drug-naïve patients with T2D, gliclazide and vildagliptin revealed similar HbA1c levels (8.7% and 8.5%, respectively) and similar HbA1c reduction from baseline to week 104 (0.6% and 0.5%, respectively), while vildagliptin had significant benefits in terms of less weight gain (0.8 versus 1.6 kg) and less hypoglycemia (0.7% versus 1.6%) [83].
In a real-word UK CPRD study among 1986 patients with T2D who received either gliclazide MR or sitagliptin as second-line treatment added to metformin for up to 9 years, superiority of gliclazide MR over sitagliptin was reported in terms of rapid achievement of target [hazard ratio (HR) 1.35 for HbA1c < 7.0% and HR 1.51 for HbA1c ≤ 6.5%] [55]. Moreover, gliclazide MR was associated with a 35% higher probability of target attainment within just 3 months (HR 1.35), while gliclazide MR and sitagliptin had comparable treatment persistence and durability of more than 2.5 years [55]. The rapid reduction of HbA1c levels was considered favorable to prevent long-term risk of macrovascular complications in relation to legacy effect, while the comparable durability and persistence offered by gliclazide MR was also emphasized given the concerns regarding the effect of SUs, as a general class, on β cell exhaustion and poor durability [55, 79, 80].
In a study among 96 patients with T2D treated concurrently for 1 year with five different SUs, gliclazide was reported to reveal significantly better HbA1c improvement than chlorpropamide, gliquidone, and glipizide, indicating the likelihood of differences between different SUs, which may offer a clinical advantage in certain patients [84].
In a 13-week, open-label RCT comparing repaglinide (4 mg tid) versus gliclazide (160 mg bid) plus bedtime NPH insulin in 80 patients with T2D inadequately controlled on OADs, glycemic control improved similarly in both gliclazide/insulin and repaglinide/insulin groups (by 1.0% and 0.9%, respectively), along with similar number of hypoglycemic episodes per patient (2.95 versus 2.3, respectively) and similar weight gain (4.1 versus 3.4 kg, respectively) [85].
In the European GUIDE study with a double-blind, 27-week, parallel-group design, 845 patients with T2D were randomized to either gliclazide MR 30–120 mg daily or glimepiride 1–6 mg daily as monotherapy or in combination with metformin or an AGI [68]. Gliclazide MR was found to be at least as effective as glimepiride, either as monotherapy or in combination, with similarly decreased HbA1c (from 8.4% to 7.2% on gliclazide MR and from 8.2% to 7.2% on glimepiride) in both groups, while the gliclazide MR revealed 50% fewer confirmed hypoglycemic episodes (3.7% versus 8.9% of patients) compared with glimepiride [68].
Cross-sectional analysis of data from the Joint Asia Diabetes Evaluation (JADE) registry in 62,512 patients with T2D from 11 Asian countries/regions revealed that among SU users (59.4% of treated patients), gliclazide was most commonly prescribed (46.7%), followed by glimepiride (40.0%) and glibenclamide (8.1%) [86]. Also, gliclazide users more commonly reported HbA1c levels < 7% than non-gliclazide SU users (OR 1.09, 95% CI 1.02–1.17) and less frequent self-reported hypoglycemia in the 3 months before registration (OR 0.81, 95% CI 0.72–0.92), indicating good glycemic control and safety profile of gliclazide therapy [86].
In a prospective, 24-week randomized add-on therapy study in 42 women with T2D on metformin therapy comparing vildagliptin and gliclazide MR treatment revealed similarly significant reductions in glycemic variability (20% decrease in each group) and in HbA1c (−0.56% versus −0.80%, respectively) [54]. Both treatments yielded slightly changed body weight (−0.3 ± 7.7 kg versus −0.5 ± 3.5 kg), no case of major hypoglycemia, and a similar frequency of minor hypoglycemia events (seven and two events, respectively) [54]. However, only gliclazide MR enables significantly increase in the time spent in target blood glucose range of 70–180 mg/dL and significant decrease in the time spent in hyperglycemia (> 180 mg/dL) [54]. The authors suggest that gliclazide MR can be an exception in the SU class relative to its profile in glycemic variability, offering results comparable to those of a DPP-4 inhibitor [54].
However, in a multicenter retrospective study on the effectiveness of DPP4 inhibitor versus gliclazide XR (mainly 30–60 mg/day) in 2410 patients with T2D, after an average 6-months follow-up, patients starting DPP4 inhibitor versus those starting gliclazide were reported to experienced greater reductions in HbA1c (−0.6% versus −0.4%; p < 0.001), fasting glucose (−14.1 mg/dL versus −8.8 mg/dL; p = 0.007), and body weight (−0.4 kg versus −0.1 kg; p = 0.006), indicating that in routine clinical practice, starting a DPP4 inhibitor allows better glycemic control than starting low-dose gliclazide [87].
Notably, in a systematic review and meta-analysis of 19 RCTs on the safety and efficacy of gliclazide (3083 patients) compared with other OADs (3155 patients), the authors noted the poor methodological quality and a considerable amount of heterogeneity between and bias in studies, while gliclazide was considered to appear at least equally effective and safe (nonsevere hypoglycemic events 2.2% versus 1.8%) compared with other OADs and to be slightly more effective (−0.13%, 95% CI −0.25 to −0.02) than other OADs except metformin [88], while a systematic review showed that gliclazide MR has a significantly reduced risk of hypoglycemia in comparison with other SUs [69].
As Add-on to Insulin Therapy
Patients with T2D who are inadequately controlled on OADs can benefit from the addition of once-daily insulin glargine to the gliclazide-MR regimen [20, 89], or for those with preserved endogen insulin reserve, the use of gliclazide MR in combination with basal insulin may offer lower dose of insulin usage and better glycemic control [90].
In a parallel-group, randomized treat-to-target clinical trial, the efficacy and safety of insulin glargine plus gliclazide MR versus the premixed insulin injection was assessed in 110 patients with T2D inadequately controlled on OADs over 12-week treatment period [89]. Once-daily insulin glargine plus gliclazide MR regimen resulted in significantly more robust HbA1c decrease (−1.23 ± 0.92% versus −1.02 ± 1.04%; p = 0.031) and better improvement in FPG (−3.87 ± 2.09 versus −2.22 ± 2.67 mmol/L; p = 0.003) than the twice-daily premixed insulin regimen [89]. Also, albeit not significant, more patients in the combination therapy group were reported to achieve an HbA1c level of ≤ 6.5% (17.39% versus 9.62%) and ≤ 7% (30.43% versus 26.92%) along with lower incidence (45 versus 60 episodes) and rate (35.4% versus 52.8%) of hypoglycemic episodes [89].
In a study among 25 patients with T2D with secondary SU failure, three treatment phases (each lasting 2 months) were evaluated, including gliclazide (320 mg, phase 1), gliclazide (320 mg) plus bedtime NPH insulin (phase 2), and insulin as monotherapy (phase 3) [91]. The authors reported a 50% increase in insulin dose in phase 3, along with a significant reduction in all points of the glycemic curve during phase 2 compared with phases 1 and 3, and concluded that gliclazide plus bedtime insulin was more efficient in enabling a better glycemic response to a mixed meal than insulin alone for patients with T2D and secondary SU failure [91].
Studies on Safety of Gliclazide
Gliclazide differs from some other SUs in terms of lower risk of cardiovascular adverse effects, while it is unique in terms of offering a lowest risk of hypoglycemic events and weight gain [12, 14,15,16,17, 53,54,55, 72]. The summary of gliclazide safety trials is provided in Table 3.
All-Cause and Cardiac Mortality and Macrovascular Complications
Cardiac benefit of any antidiabetic drug is of critical importance, since the patients with versus without diabetes are twice as likely to develop cardiovascular disease (CVD), which is also the leading cause of mortality in these patients [20, 92]. Gliclazide, as a newer-generation pancreatic β cell-specific SU, seems to be advantageous in this regard, by not increasing the cardiovascular risk, not impairing the protective ischemic cardiac preconditioning, and having a low risk of hypoglycemia [8, 20, 39, 78, 93].
Notably, the cardiovascular risk pertaining to SUs as a general class has not been confirmed in the RCTs of newer generation SUs with more pancreatic selectivity action and less cardiovascular mortality, such as the Thiazolidinediones Or Sulfonylureas Cardiovascular Accidents Intervention Trial (TOSCA.IT) and CAROLINA studies [12, 13, 94, 95]. TOSCA-IT showed that a SU (mostly glimepiride and gliclazide) was noninferior to pioglitazone for preventing MACE and both SU and pioglitazone as add-on treatments to metformin represent widely available and affordable suitable options with respect to efficacy and adverse events [94]. The cardiovascular outcome trial data (CVOT) from the CAROLINA trial demonstrated that glimepiride was not associated with an increased cardiovascular risk when compared with a DPP-4 inhibitor (linagliptin) with established cardiovascular safety [95] The primary 3P-MACE endpoint occurred in 11.8% versus 12.0% of patients treated with linagliptin (2.1 per 100 person-years) and glimepiride (2.1 per 100 person-years), respectively, meeting the criterion for noninferiority [95].
A meta-analysis of RCTs demonstrated that SUs as add-ons to metformin were not associated with all-cause mortality [odds ratio (OR) 1.26, 95% CI 0.94–1.68] or cardiovascular mortality (OR 1.40, 95% CI 0.61–3.22) [96]. In fact, SUs have become the major second-line class of OADs used in large-scale trials (i.e., UKPDS and ADVANCE studies) that demonstrated the value of intensive glucose lowering in delaying the development of macrovascular events in patients with T2D [27, 29]. In the ADVANCE study, intensive treatment (gliclazide MR in 91% of patients) relative to a standard treatment (gliclazide MR in 2%) was associated with a 10% relative reduction in the combined outcome of major macrovascular and microvascular events, and a significant reduction in the rate of microvascular complications (14%, p = 0.01, mainly for nephropathy 21%, p = 0.006) but not in the rate of macrovascular events [29]. The 10-year extension study, ADVANCE-ON, revealed the similar HbA1c levels and macrovascular outcome (all-cause and cardiovascular mortality) between two treatment arms, whereas a continued decrease in nephropathy rates in the intensive treatment group (46%, p = 0.007) [30].
The STENO-2 study reported about a 50% reduction in CVD risk with gliclazide-based intensive treatment after the mean follow-up of 7.8 years [97], while at 13.3 years of follow-up, a significantly lower risk of death from cardiovascular causes (HR 0.43, 95% CI 0.19–0.94; p = 0.04) and cardiovascular events (HR 0.41, 95% CI 0.25–0.67; p < 0.001) was reported [31]. At 21.2 years of follow-up (7.8 years of intensified treatment), median time before first cardiovascular event after randomization was found to be 8.1 years longer along with a median of 7.9 years of gain of life in the intensive treatment group, as matched by time free from incident cardiovascular events [32, 98]. These findings seem notable given that life expectancy is considered to be reduced by 6 years in patients with diabetes and by 12 years in those with diabetes and CVD compared with the general population [39, 99].
In a retrospective observational cohort study of 568 patients with T2D on comparison of all-cause, cardiovascular and noncardiovascular mortality, and cardiac morbidity, between patients treated with glibenclamide (n = 378) and gliclazide (n = 190), glibenclamide versus gliclazide treatment was associated with significantly higher yearly mortality rate (4.3% versus 2.2%, respectively, p < 0.05) and a significant increase in all-cause mortality (OR 2.1, 95% CI 1.2–2.7, p < 0.05) and mortality for malignancies (OR 3.6, 95% CI 1.1–11.9, p < 0.05), while no significant difference was noted between treatment groups in terms of cardiovascular mortality [100].
In a population-based study among 107,806 patients with a median follow-up of 3.3 years, evaluation of several mortality endpoints with different insulin secretagogues versus metformin revealed that all-cause mortality was significantly higher with all SUs (glimepiride: HR 1.32; glipizide: HR 1.27; and glibenclamide: HR 1.19) except gliclazide (HR 1.05) in patients with or without a previous history of MI [101].
In a nationwide population-based follow-up study in 3930 SU users admitted with MI, 35.3% annual mortality rate was reported, while gliclazide monotherapy showed a trend toward lower mortality rate (adjusted HR 0.70, 95% CI 0.48–1.00) [102]. In a population-based registry in 64,188 patients, both gliclazide (HR 0.33, 95% CI 0.26–0.41) and glimepiride (HR 0.605, 95% CI 0.41–0.88) revealed lower total mortality rates than glibenclamide monotherapy, while gliclazide also resulted in lower cardiovascular mortality rate than glibenclamide (HR 0.29, 95% CI 0.21–0.38) [103].
In a nationwide population-based study on the real-world cardiovascular safety of second-line OADs in 29,518 patients with T2D including SUs initiators (87.2% with gliclazide, 8.3% with glimepiride, and 0.1% with glibenclamide) and non-SU (DPP4 inhibitor/TZD) initiators, multivariable Cox regression revealed that HRs for MACE and all-cause death were 1.00 (95% CI 0.91–1.09) and 1.03 (0.94–1.13), respectively [104]. Hence, second-line SUs for glucose lowering are considered unlikely to increase CV risk or all-cause mortality and thus SU should remain a part of the global diabetes treatment portfolio [104]. Notably, given that SUs with high-affinity versus low-affinity binding with cardiac mitochondrial KATP channels are associated with increased MACE risk [76], they also suggested the significant contribution of gliclazide as the SU of choice in their population (87.2% of second-line SU users) to the CV outcomes [104].
Nonetheless, in a population-based cohort study with 17,604 sulfonylurea initiators, the use of nonspecific long-acting sulfonylureas (glyburide and glimepiride) was not associated with an increased risk of AMI, ischemic stroke, cardiovascular death, or all-cause mortality, when evaluated with respect to pancreas-specific, short-acting sulfonylureas (tolbutamide, gliclazide, and glipizide, as reference group), contradicting the previous studies that emphasized the clinical implications of the lack of pancreas specificity of certain sulfonylureas [105].
In a systematic review of studies on the SUs and the risk of ventricular arrhythmias (VA), SUs as a class were found to be associated with a higher risk of arrhythmia versus DPP-4 inhibitors [adjusted hazard ratio (aHR) 1.52, 95% CI 1.27–1.80] and of VA versus metformin (aHR 1.52, 95% CI 1.10–2.13) [106]. However, two studies with gliclazide-specific data in the review revealed inconclusive results for the risk of VA with gliclazide relative to glyburide due to wide 95% CIs (OR 1.20, 95% CI 0.60–2.30) [107], and a reduced out-of-hospital cardiac arrest risk with SUs compared with metformin with gliclazide having a lower risk than glimepiride (ORadj 0.5, 95% CI 0.3–0.9) [108].In a systematic review and network meta-analysis of 18 studies on the mortality risk among SUs, gliclazide was associated with a significantly lower risk of cardiovascular mortality [relative risk (RR) 0.60, 95% CI 0.45–0.84] and all-cause mortality (RR 0.65, 95% CI 0.53–0.79) compared with glibenclamide and others [105]. Hence, as associated with the lowest risk of cardiovascular-related mortality (followed by glimepiride), as well as the lowest risk of severe hypoglycemia among all SUs, the relative safety of a gliclazide makes it a preferred SU among the SUs [12, 13, 30, 105, 109].
Hypoglycemia Risk
Accumulating evidence indicate that, among SU drugs, gliclazide poses the lowest risk for episodes of major hypoglycemia, which is comparable to other second-line OADs [8, 17, 30, 54, 55, 68, 81]. Notably, to cause one severe hypoglycemic event, at least 232 patients and 333 patients would need to be treated with a SU and gliclazide MR, respectively [12].
A recent meta-analysis demonstrated that the relative risk of hypoglycemia with gliclazide versus placebo (3.9) is more similar to the risk with metformin (2.0) and lower than the risk with other SUs such as glimepiride (8.9), glyburide (10.2), or glipizide (13.9) [110].
In addition to the higher compliance (OR 3.50) and adherence (OR 3.07) rates attributable to once-daily dosing of the long-acting MR formulation [111], properties such as metabolization into inactive metabolites and a gradual increase in drug concentrations are considered to enable gliclazide MR to offer a lower risk of hypoglycemia (RR 0.47) compared with other SUs [19, 20, 69].
In a multicenter prospective observational study on hypoglycemia detected by continuous glucose monitoring in 94 patients with T2D treated with OADs (50 were treated with SU, primarily gliclazide, 84%), gliclazide treatment was not found to be a risk factor for hypoglycemia regardless of cut-off value [112]. However, the subgroup analysis of patients with hypoglycemia indicated the likelihood of other possible risk factors for hypoglycemia (i.e., prolonged overnight fasting, physical activity, alcohol consumption, and concomitant antihypertensive agents), emphasizing the careful consideration of other potential factors related to concomitant therapy, nutrition, and daily habits in patients experiencing hypoglycemia under SU therapy [112].
In the GUIDE study, the safety of gliclazide MR was reported to be significantly better, demonstrating approximately 50% fewer hypoglycemic episodes than glimepiride [68]. In fact, the incidence of hypoglycemia with gliclazide MR was particularly low in patients at higher risk for hypoglycemia [moderately elevated HbA1c (≤ 7%) at baseline and/or decreased HbA1c (< 6.5%) on treatment], suggesting that gliclazide MR can be used following current recommendations of aggressive treatment to obtain HbA1c targets of 6.5–7% [68].
Weight Gain
Gliclazide MR is considered an exception among the class of SUs that it does not promote more weight gain than DPP-4 inhibitors [29, 54, 94]. There was no weight gain with gliclazide MR (0.1 kg) in the ADVANCE study over a 5-year period [29], while data from TOSCA.IT showed a moderate weight gain (less than 2 kg, on average) in both pioglitazone and gliclazide groups [94]. In fact, gliclazide’s effect on weight was considered even more beneficial during short than long treatment periods, as demonstrated by the EASYDia trial [weight loss of 0.9–2.2 kg by patients with a baseline body mass index (BMI) ≥ 25 kg/m2) over a 6-month period] [81] and by the DIA-RAMADAN study (mean weight loss of 0.5 kg over a 14–18-week period) [113].
Use of Gliclazide in Special Populations
Patients with Renal or Hepatic Diseases
In mild-to-moderate renal impairment, newer-generation SUs may be used, preferably at lower doses [13]. Long-term intensive use of gliclazide was reported to prevent end-stage renal disease (ESRD) in ADVANCE and ADVANCE-ON studies [29, 30], while the GUIDE study also indicated that even mild impairment of renal function increases the incidence of hypoglycemia on glimepiride but not on gliclazide MR [68].
Gliclazide, as eliminated mainly by the kidneys (60–70%) and mostly excreted as unchanged drug or inactive metabolite, is suggested to produce less hypoglycemia in patients with renal impairment and therefore can be used with proper dose adjustment and monitoring in patients with renal insufficiency [13, 50]. Nonetheless, while dose adjustment is not necessary for gliclazide in case of impaired renal function, it is contraindicated in patients with an estimated glomerular filtration rate (eGFR) of < 30 ml/min/1.73 m2 due to hypoglycemia risk, and the dose should be reduced in those with eGFR of 30–60 ml/min/1.73 m2 [8, 14, 20, 114].
SUs should be cautiously used in patients with liver diseases. As most of the SUs are inactivated in the liver, they may accumulate in the body during liver dysfunction and ultimately cause hypoglycemia [13]. Reductions of SU dose and/or longer intervals between dosing are recommended in patients with mild-to-moderate hepatic impairment, and gliclazide can be used in mild-to-moderate hepatic failure but not in severe hepatic failure [13].
Elderly Population
Elderly populations with T2D are considered to be at a higher risk of hypoglycemia as well as the adverse consequences of hypoglycemia (i.e., serious acute vascular events in case of severe hypoglycemia or morbidity with physical and cognitive dysfunction, frailty, and disability in case of recurrent episodes of hypoglycemia) due to multiple comorbidities, poor nutritional status, and polypharmacy [115, 116]. Newer-generation SUs (gliclazide MR and glimepiride) can be used in elderly patients as they are associated with a low risk of hypoglycemia [13]. Use of gliclazide MR alone or in combination with another OAD over 2 years was reported to be an effective and safe option in the elderly and in patients with impaired renal function [17]. In the GUIDE study, gliclazide MR was reported to significantly improved HbA1c levels (from 8.4% to 7.2% at 27 weeks) in the subgroup of patients aged > 65 years [68].
During Ramadan Fasting
In general, while patients with diabetes are not recommended to undertake the fasting component of Ramadan, some decide to fast despite the associated difficulties such as hypoglycemia (almost 7.5-fold increase in risk) and other health-related complications due to suboptimal healthcare during prolonged intervals of oral intake [8, 13, 50, 113].
Although, there is insufficient evidence regarding the use of glicazide MR during Ramadan fasting, the newer-generation SUs are considered to be safe and tolerable options in such circumstances [8, 117,118,119]. The recent IDF-DAR practical guideline indicates that patients continuing second-generation SUs can fast safely during Ramadan [120], while RCTs reported that gliclazide had the lowest risk of hypoglycemia among all SUs during Ramadan, almost similar to DPP-4 inhibitors [117, 118]. In the randomized STEADFAST study, patients treated with metformin plus gliclazide during Ramadan had an adjusted mean reduction in HbA1c (0.03%), and body weight (1.1 kg) and hypoglycemic episodes were similar between vildagliptin and gliclazide (both add-ons to metformin) [121]. In the DIA-RAMADAN study among 1214 patients across nine countries on the efficacy and safety of gliclazide MR (as monotherapy in 41%, and with other OADs in 59%) during Ramadan, no patients reported severe hypoglycemia along with a significant reduction in HbA1c (−0.3%), FPG (−9.7 mg/dL), and body weight (−0.5 kg) [119]. When changing to an evening dosing regimen during Ramadan, mean HbA1c was reported to be decreased by 0.8% and mean body weight increased by 0.4 kg in gliclazide MR-treated patients [122]. Hence, patients with T2D treated with gliclazide MR during Ramadan are considered to be able to maintain HbA1c and body weight values along with having a low risk of hypoglycemia while fasting [119].
In Monogenic Diabetes
Hepatocyte nuclear factor 1alpha (HNF-1A) and HNF4A gene mutations are common causes of maturity-onset diabetes of the young (MODY) and patients with HNF-MODY diabetes show greater response to SUs, particularly to gliclazide (5.2-fold greater than metformin and 3.9-fold greater than patients with T2D) [123, 124]. Hence, these patients can be successfully treated with first-line gliclazide before metformin since they do not have insulin resistance and due to the higher efficacy of gliclazide over metformin in lowering FPG and increasing β cell function in patients with MODY3 versus patients with T2D [12, 123, 124].
Place of SUs as A Class and Gliclazide MR as A Newer Generation SU in The Current Type 2 Diabetes Treatment Algorithm
Guidelines and consensus reports, available for diabetes management, almost universally recommend metformin, in conjunction with lifestyle changes, as the first-line glucose-lowering treatment in patients with newly diagnosed T2D, but they vary in terms of the recommendations on second-line agents for add-on therapies in case of failure of metformin monotherapy [8, 12, 23, 24, 50, 125,126,127,128,129,130].
A summary of guidelines and consensus report recommendations on the use of SUs and gliclazide MR in the second-line T2D treatment is provided in Table 4.
While SUs are considered as second-line treatment in most of the guidelines, there is a discrepancy related to the place of SUs as a class, and gliclazide MR specifically, and some recommend the use of SUs in later steps of treatment or only in special conditions [8, 12, 23, 128, 129]. This discrepancy among the guidelines seems to be related to the differences in priorities considered for certain parameters such as cost, efficacy, safety (short term or long term), practicability, access to treatment (absence of gliclazide in the USA), target physician population (general practitioner, specialist), prioritization of comorbidities (those with or without CVD, CKD, heart failure), or drug molecules (newer-generation SUs such as gliclazide MR).
In patients with established atherosclerotic CVD, recent guidelines consistently recommend second-line treatment with a SGLT-2 inhibitor or a GLP-1 receptor agonist based on their favorable cardiac safety data [12, 23, 128, 129],
For those without established CVD, the recommendations for add-on glucose-lowering therapy differ among guidelines [12]. Australian, Indian, and WHO guidelines specify SUs as the agents of choice for second-line treatment [50, 126, 127], while guidelines from the National Institute for Health and Care Excellence (NICE) in the UK and the International Diabetes Federation (IDF) include SUs as one of the preferred options, along with a DPP-4 inhibitor and either pioglitazone (NICE-UK) or a SGLT-2 inhibitor (IDF) [24, 125]. The Society of Endocrinology and Metabolism of Türkiye (SEMT) 2022 guidelines also include SUs as one of the preferred options, along with a glinide, DPP4i, AGI, GLP-1 receptor agonist, thiazolidinedione (TZD), or SGLT2 [130]. In contrast, one of the most widely used consensus report from the American Diabetes Association (ADA) and European Association for the Study of Diabetes (EASD) as well as joint guidelines from the EASD and the European Society of Cardiology (ESC) recommends SUs as second-line therapy only if cost is a compelling issue and place SUs as a fifth-line treatment after other classes of OADs have been tried [23, 129] (Table 1).
Although unique properties such as low risk of hypoglycemia, weight neutrality, cardiovascular safety, favorable renal outcomes, and delay in secondary treatment failure appear to be the reasons for preference of gliclazide MR over other SUs [12, 14,15,16,17, 53, 55, 72, 79, 80], few guidelines on diabetes management specifically suggest gliclazide, instead of SUs as a class, as the second-line treatment [12, 14, 19, 20]. Accordingly, given the accumulating evidence regarding the association of newer generation SUs (glimepiride, gliclazide, and glipizide) with much lower risk of hypoglycemia and weight gain and a more favorable cardiac safety profile than conventional SUs, guidelines should consider the prioritization of newer-generation SUs such as gliclazide in the treatment algorithm instead of combined assessment of all SUs in the same category [20].
Conclusions
As shown by many studies, maintaining early and sustained glycemic control prevents the development of microvascular and macrovascular complications. Patient- and drug-specific characteristics should be considered when deciding on a treatment to control blood glucose.
Sulfonylureas have been used for many years in different stages of diabetes treatment in many parts of the world. Among sulfonylureas, gliclazide MR is distinguished by its specific pharmacological structure and molecular properties. In addition to its strong efficacy, gliclazide MR is different with its features such as low hypoglycemia risk, weight neutrality, protection of beta cells with its antioxidant ring, and is differentiated by some guidelines.
With its extensive clinical experience and strong efficacy and safety profile, gliclazide MR can be included in every step of diabetes treatment algorithm.
References
Defronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes. 2009;58(4):773–95.
Alejandro EU, Gregg B, Blandino-Rosano M, Cras-Méneur C, Bernal-Mizrachi E. Natural history of β-cell adaptation and failure in type 2 diabetes. Mol Aspects Med. 2015;42:19–41.
International Diabetes Federation. IDF Diabetes Atlas, 10th Ed. Brussels, Belgium : International Diabetes Federation; 2021. https://diabetesatlas.org/ Accessed 10 Jan 2023
Mannucci E, Dicembrini I, Lauria A, Pozzilli P. Is glucose control important for prevention of cardiovascular disease in diabetes? Diabetes Care. 2013;36(Suppl 2):S259–63.
Cernea S, Dobreanu M. Diabetes and beta cell function: from mechanisms to evaluation and clinical implications. Biochem Med (Zagreb). 2013;23(3):266–80.
Baxter M, Hudson R, Mahon J, et al. Estimating the impact of better management of glycaemic control in adults with Type 1 and Type 2 diabetes on the number of clinical complications and the associated financial benefit. Diabet Med. 2016;33:1575–81.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577–89.
Al-Saleh Y, Sabico S, Al-Furqani A, et al. Sulfonylureas in the current practice of type 2 diabetes management: Are they all the same? Consensus from the Gulf Cooperation Council (GCC) Countries Advisory Board on Sulfonylureas. Diabetes Ther. 2021;12(8):2115–32.
Amod A. The place of sulfonylureas in guidelines: why are there differences? Diabetes Ther. 2020;11(Suppl 1):5–14.
Mohan V, Khunti K, Chan SP, et al. Management of type 2 diabetes in developing countries: balancing optimal glycaemic control and outcomes with affordability and accessibility to treatment. Diabetes Ther. 2020;11(1):15–35.
Proks P, Reimann F, Green N, Gribble F, Ashcroft F. Sulfonylurea stimulation of insulin secretion. Diabetes. 2002;51(Suppl 3):S368–76.
Khunti K, Hassanein M, Lee MK, Mohan V, Amod A. Role of gliclazide MR in the management of type 2 diabetes: report of a symposium on real-world evidence and new perspectives. Diabetes Ther. 2020;11(Suppl 2):33–48.
Kalra S, Bahendeka S, Sahay R, et al. Consensus recommendations on sulfonylurea and sulfonylurea combinations in the management of type 2 diabetes mellitus—International Task Force. Indian J Endocrinol Metab. 2018;22(1):132–57.
Singh AK, Singh R. Is gliclazide a sulfonylurea with difference? A review in 2016. Expert Rev Clin Pharmacol. 2016;9:839–51.
Gribble FM, Reimann F. Differential selectivity of insulin secretagogues: mechanisms, clinical implications, and drug interactions. J Diabetes Complicat. 2003;17:11–5.
Drouin P. Diamicron MR once daily is effective and well tolerated in type 2 diabetes: a double-blind, randomized, multinational study. J Diabetes Complicat. 2000;14(4):185–91.
Drouin P, Standl E, Diamicron MR, Study Group. Gliclazide modified release: results of a 2-year study in patients with type 2 diabetes. Diabetes Obes Metab. 2004;6(6):414–21.
WHO model list of essential medicines - 22nd list, 2021. https://www.who.int/publications/i/item/WHO-MHP-HPS-EML-2021.02 Accessed 10 Jan 2023
Colagiuri S, Matthews D, Leiter LA, Chan SP, Sesti G, Marre M. The place of gliclazide MR in the evolving type 2 diabetes landscape: a comparison with other sulfonylureas and newer oral antihyperglycemic agents. Diabetes Res Clin Pract. 2018;143:1–14.
Kalra S, Unnikrishnan AG, Bantwal G, Das S, Polavarapu NK, Gaurav K. The position of gliclazide in the evolving landscapes and disease continuum of T2DM: a collaborative Delphi survey-based consensus from India. Diabetes Ther. 2021;12(3):679–95.
Nathan DM. The diabetes control and complications trial/epidemiology of diabetes interventions and complications study at 30 years: overview. Diabetes Care. 2014;37:9–16.
American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S90–102.
Buse JB, Wexler DJ, Tsapas A, et al. 2019 update to: Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia. 2020;63(2):221–8.
National Institute for Health and Care Excellence (NICE). Type 2 diabetes in adults: management (NICE Guideline 28). 2015 (last updated 2020). www.nice.org.uk/guidance/NG28. Accessed 10 Jan 2023
Stratton IM, Adler AI, Neil HA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321(7258):405–12.
Boye KS, Thieu VT, Lage MJ, Miller H, Paczkowski R. The association between sustained hba1c control and long-term complications among individuals with type 2 diabetes: a retrospective study. Adv Ther. 2022;39(5):2208–21.
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–53.
Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129–39.
Patel A, MacMahon S, ADVANCE Collaborative Group, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358(24):2560–72.
Zoungas S, Chalmers J, Neal B, et al. ADVANCE-ON Collaborative Group. Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med. 2014;371(15):1392–406.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358(6):580–91.
Gæde P, Oellgaard J, Carstensen B, et al. Years of life gained by multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: 21 years follow-up on the Steno-2 randomised trial. Diabetologia. 2016;59(11):2298–307.
Rawshani A, Rawshani A, Franzén S, et al. Risk factors, mortality, and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2018;379(7):633–44.
Laiteerapong N, Ham SA, Gao Y, et al. The legacy effect in Type 2 diabetes: impact of early glycemic control on future complications (the diabetes & aging study). Diabetes Care. 2019;42(3):416–26.
Caramelli B, Gualandro DM. Both glucocentric and cardiocentric approaches are necessary for a resilient disease such as diabetes. Rev Assoc Med Bras (1992). 2018;64(3):212–3.
Paul SK, Klein K, Thorsted BL, Wolden ML, Khunti K. Delay in treatment intensification increases the risks of cardiovascular events in patients with type 2 diabetes. Cardiovasc Diabetol. 2015;14:100.
Cefalu WT, Rosenstock J, LeRoith D, Blonde L, Riddle MC. Getting to the “heart” of the matter on diabetic cardiovascular disease: “thanks for the memory.” Diabetes Care. 2016;39(5):664–7.
Adler AI, Holman RR, Clarke PM, Whiteley WN. UKPDS 44 year Follow up: Do the legacy effects persist. Symposium on 58th Annual Meeting of the European Association for the Study of Diabetes (EASD) in Stockholm, Sweden, September 2022.
Yu M. The place of sulfonylureas in the evolving landscape of combination therapy. Diabetes Ther. 2020;11(Suppl 1):23–8.
Levine R. Sulfonylureas: background and development of the field. Diabetes Care. 1984;7(Suppl. 1):3–7.
Quianzon CC, Cheikh IE. History of current non-insulin medications for diabetes mellitus. J Community Hosp Intern Med Perspect. 2012;2(3):19081.
White JR Jr. A Brief history of the development of diabetes medications. Diabetes Spectr. 2014;27(2):82–6.
Thulé PM, Umpierrez G. Sulfonylureas: a new look at old therapy. Curr Diab Rep. 2014;14(4):473.
Ashcroft FM, Rorsman P. Electrophysiology of the pancreatic beta-cell. Prog Biophys Mol Biol. 1989;54:87–143.
Ashcroft FM, Gribble FM. ATP-sensitive K+ channels and insulin secretion: their role in health and disease. Diabetologia. 1999;42:903–19.
Sola D, Rossi L, Schianca GP, et al. State of the art paper Sulfonylureas and their use in clinical practice. Arch Med Sci. 2015;4(4):840–8.
Campbell KR, Cobble ME, Reid TS, Shomali MS. Glucose-lowering effects of incretin-based therapies. J Fam Pract. 2010;59(9 Suppl 1):S5–9.
Zhang Y, Hong J, Chi J, Gu W, Ning G, Wang W. Head-to-head comparison of dipeptidyl peptidase-IV inhibitors and sulfonylureas—a meta-analysis from randomized clinical trials. Diabetes Metab Res Rev. 2014;30(3):241–56.
Downes MJ, Bettington EK, Gunton JE, Turkstra E. Triple therapy in type 2 diabetes; a systematic review and network meta-analysis. PeerJ. 2015;3: e1461.
Kalra S, Aamir AH, Raza A, et al. Place of sulfonylureas in the management of type 2 diabetes mellitus in South Asia: a consensus statement. Indian J Endocrinol Metab. 2015;19(5):577–96.
Harrower A. Gliclazide modified release: from once-daily formulation to 24-hour blood glucose control. Metabolism. 2000;49:7–11.
Guillausseau PJ, Greb W. 24-hour glycemic profile in type 2 diabetic patients treated with gliclazide modified release once daily. Diabetes Metab. 2001;27:133–7.
Mohan V, Chopra V, Sanyal D, Jain S, Jayaprakashsai J. Treatment of type 2 diabetes with a breakable extended release gliclazide formulation in primary care: the Xrise study. J Assoc Physicians India. 2015;63(12):26–9.
Vianna AGD, Lacerda CS, Pechmann LM, Polesel MG, Marino EC, Faria-Neto JR. A randomized controlled trial to compare the effects of sulphonylurea gliclazide MR (modified release) and the DPP-4 inhibitor vildagliptin on glycemic variability and control measured by continuous glucose monitoring (CGM) in Brazilian women with type 2 diabetes. Diabetes Res Clin Pract. 2018;139:357–65.
Zaccardi F, Jacquot E, Cortese V, et al. Comparative effectiveness of gliclazide modified release versus sitagliptin as second-line treatment after metformin monotherapy in patients with uncontrolled type 2 diabetes. Diabetes Obes Metab. 2020;22(12):2417–26.
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107(9):1058–70.
Fava D, Cassone-Faldetta M, Laurenti O, De Luca O, Ghiselli A, De Mattia G. Gliclazide improves anti-oxidant status and nitric oxide-mediated vasodilation in Type 2 diabetes. Diabet Med. 2002;19(9):752–7.
Del Guerra S, Grupillo M, Masini M, et al. Gliclazide protects human islet beta-cells from apoptosis induced by intermittent high glucose. Diabetes Metab Res Rev. 2007;23:234–8.
Sena CM, Louro T, Matafome P, Nunes E, Monteiro P, Seica R. Antioxidant and vascular effects of gliclazide in type 2 diabetic rats fed high-fat diet. Physiol Res. 2009;58:203–9.
Renier G, Mamputu JC, Serri O. Benefits of gliclazide in the atherosclerotic process: decrease in monocyte adhesion to endothelial cells. Metabolism. 2003;52:13–8.
Mamputu JC, Renier G. Signalling pathways involved in retinal endothelial cell proliferation induced by advanced glycation end products: inhibitory effect of gliclazide. Diabetes Obes Metab. 2004;6:95–103.
Kimoto K, Suzuki K, Kizaki T, et al. Gliclazide protects pancreatic beta-cells from damage by hydrogen peroxide. Biochem Biophys Res Commun. 2003;303:112–9.
Katakami N, Yamasaki Y, Hayaishi-Okano R, et al. Metformin or gliclazide, rather than glibenclamide, attenuate progression of carotid intima-media thickness in subjects with type 2 diabetes. Diabetologia. 2004;47(11):1906–13.
O’Brien RC, Luo M, Balazs N, Mercuri J. In vitro and in vivo antioxidant properties of gliclazide. J Diabetes Complicat. 2000;14(4):201–6.
Jennings PE. Vascular benefits of gliclazide beyond glycemic control. Metabolism. 2000;49(10 Suppl 2):17–20.
Hirst JA, Farmer AJ, Dyar A, Lung TW, Stevens RJ. Estimating the effect of sulfonylurea on HbA1c in diabetes: a systematic review and meta-analysis. Diabetologia. 2013;56(5):973–84.
Yu O, Azoulay L, Yin H, Filion KB, Suissa S. Sulfonylureas as initial treatment for type 2 diabetes and the risk of severe hypoglycemia. Am J Med. 2018;131(3):317.
Schernthaner G, Grimaldi A, Di Mario U, et al. GUIDE study: double-blind comparison of once-daily gliclazide MR and glimepiride in type 2 diabetic patients. Eur J Clin Invest. 2004;34(8):535–42.
Chan SP, Colagiuri S. Systematic review and meta-analysis of the efficacy and hypoglycemic safety of gliclazide versus other insulinotropic agents. Diabetes Res Clin Pract. 2015;110(1):75–81.
Gribble FM, Ashcroft FM. Differential sensitivity of beta-cell and extrapancreatic K(ATP) channels to gliclazide. Diabetologia. 1999;42(7):845–8.
Engler RL, Yellon DM. Sulfonylurea KATP blockade in type II diabetes and preconditioning in cardiovascular disease. Time for reconsideration. Circulation. 1996;94(9):2297–301.
Hosker JP, Rudenski AS, Burnett MA, et al. Similar reduction of first and second-phase B-cell responses at three different glucose levels in type II diabetes and the effect of gliclazide therapy. Metabolism. 1989;38:767–72.
Abdelmoneim AS, Hasenbank SE, Seubert JM, Brocks DR, Light PE, Simpson SH. Variations in tissue selectivity amongst insulin secretagogues: a systematic review. Diabetes Obes Metab. 2012;14(2):130–8.
Khunti K, Chatterjee S, Gerstein HC, Zoungas S, Davies MJ. Do sulphonylureas still have a place in clinical practice? Lancet Diabetes Endocrinol. 2018;6(10):821–32.
Rahmi RM, Uchida AH, Rezende PC, et al. Effect of hypoglycemic agents on ischemic preconditioning in patients with type 2 diabetes and symptomatic coronary artery disease. Diabetes Care. 2013;36(6):1654–9.
Wang MT, Huang YL, Lai JH, et al. Association between specificity of sulfonylureas to cardiac mitochondrial katp channels and the risk of major adverse cardiovascular events in type 2 diabetes. Diabetes Care. 2022;45(5):1276–87.
Monami M, Luzzi C, Lamanna C, et al. Three-year mortality in diabetic patients treated with different combinations of insulin secretagogues and metformin. Diabetes Metab Res Rev. 2006;22(6):477–82.
Simpson SH, Majumdar SR, Tsuyuki RT, Eurich DT, Johnson JA. Dose-response relation between sulfonylurea drugs and mortality in type 2 diabetes mellitus: a population-based cohort study. CMAJ. 2006;174(2):169–74.
Kubota A, Yabe D, Kanamori A, et al. Factors influencing the durability of the glucose-lowering effect of sitagliptin combined with a sulfonylurea. J Diabetes Investig. 2014;5(4):445–8.
Satoh J, Takahashi K, Takizawa Y, et al. Secondary sulfonylurea failure: comparison of period until insulin treatment between diabetic patients treated with gliclazide and glibenclamide. Diabetes Res Clin Pract. 2005;70(3):291–7.
Leiter LA, Shestakova MV, Satman I. Effectiveness of gliclazide MR 60 mg in the management of type 2 diabetes: analyses from the EASYDia trial. Diabetol Metab Syndr. 2018;10:30.
Kalra S, Das AK. Epidemiologic surveillance of glycemic response to a scored, breakable, extended release, fixed dose combination of gliclazide and metformin in persons with type 2 diabetes. J Assoc Physicians India. 2017;65:38–41.
Foley JE, Sreenan S. Efficacy and safety comparison between the DPP-4 inhibitor vildagliptin and the sulfonylurea gliclazide after two years of monotherapy in drug-naive patients with type 2 diabetes. Horm Metab Res. 2009;41(12):905–9.
Harrower AD. Comparison of diabetic control in type 2 (non-insulin dependent) diabetic patients treated with different sulphonylureas. Curr Med Res Opin. 1985;9(10):676–80.
Furlong NJ, Hulme SA, O’Brien SV, Hardy KJ. Comparison of repaglinide vs. gliclazide in combination with bedtime NPH insulin in patients with Type 2 diabetes inadequately controlled with oral hypoglycaemic agents. Diabet Med. 2003;20(11):935–41.
Lim LL, Lau ESH, Cheung JTK, et al. Real-world usage of sulphonylureas in Asian patients with type 2 diabetes using the Joint Asia Diabetes Evaluation (JADE) register. Diabetes Obes Metab. 2023;25:208–21.
Fadini GP, Bottigliengo D, D’Angelo F, et al. DARWIN-T2D Network. Comparative effectiveness of DPP-4 inhibitors versus sulfonylurea for the treatment of type 2 diabetes in routine clinical practice: a retrospective multicenter real-world study. Diabetes Ther. 2018;9(4):1477–90.
Landman GW, de Bock GH, van Hateren KJ, et al. Safety and efficacy of gliclazide as treatment for type 2 diabetes: a systematic review and meta-analysis of randomized trials. PLoS One. 2014;9(2): e82880.
Zhou J, Zheng F, Guo X, et al. Glargine insulin/gliclazide MR combination therapy is more effective than premixed insulin monotherapy in Chinese patients with type 2 diabetes inadequately controlled on oral antidiabetic drugs. Diabetes Metab Res Rev. 2015;31(7):725–33.
Aschner P, Kattah W. Effects of the combination of insulin and gliclazide compared with insulin alone in type 2 diabetic patients with secondary failure to oral hypoglycemic agents. Diabetes Res Clin Pract. 1992;18(1):23–30.
Chazan AC, Gomes MB. Gliclazide and bedtime insulin are more efficient than insulin alone for type 2 diabetic patients with sulfonylurea secondary failure. Braz J Med Biol Res. 2001;34(1):49–56.
Emerging Risk Factors Collaboration, Sarwar N, Gao P, Seshasai SR, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascu22lar disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010;375(9733):2215–22.
Francillard M, Frey N, Paraire M, Laveille C, Jochemsen R. Pharmacokinetics of Diamicron® modified release (MR) in 1007 type 2 diabetic patients (Abstract). J Nutr Health Aging. 2001;5:A14.
Vaccaro O, Masulli M, Nicolucci A, et al. Effects on the incidence of cardiovascular events of the addition of pioglitazone versus sulfonylureas in patients with type 2 diabetes inadequately controlled with metformin (TOSCA.IT): a randomised, multicentre trial. Lancet Diabetes Endocrinol. 2017;5(11):887–97.
Rosenstock J, Kahn SE, Johansen OE, et al. CAROLINA Investigators. Effect of linagliptin vs glimepiride on major adverse cardiovascular outcomes in patients with type 2 diabetes: the CAROLINA randomized clinical trial. JAMA. 2019;322(12):1155–66.
Rados DV, Pinto LC, Remonti LR, Leitão CB, Gross JL. Correction: The association between sulfonylurea use and all-cause and cardiovascular mortality: a meta-analysis with trial sequential analysis of randomized clinical trials. PLoS Med. 2016;13: e1002091.
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med. 2003;348(5):383–93.
Oellgaard J, Gæde P, Rossing P, et al. Reduced risk of heart failure with intensified multifactorial intervention in individuals with type 2 diabetes and microalbuminuria: 21 years of follow-up in the randomised Steno-2 study. Diabetologia. 2018;61(8):1724–33.
Emerging Risk Factors Collaboration, Di Angelantonio E, Kaptoge S, Wormser D, et al. Association of cardiometabolic multimorbidity with mortality. JAMA. 2015;314(1):52–60.
Monami M, Balzi D, Lamanna C, et al. Are sulphonylureas all the same? A cohort study on cardiovascular and cancer-related mortality. Diabetes Metab Res Rev. 2007;23(6):479–84.
Schramm TK, Gislason GH, Vaag A, et al. Mortality and cardiovascular risk associated with different insulin secretagogues compared with metformin in type 2 diabetes, with or without a previous myocardial infarction: a nationwide study. Eur Heart J. 2011;32(15):1900–8.
Horsdal HT, Johnsen SP, Søndergaard F, et al. Sulfonylureas and prognosis after myocardial infarction in patients with diabetes: a population-based follow-up study. Diabetes Metab Res Rev. 2009;25:515–22.
Khalangot M, Tronko M, Kravchenko V, et al. Glibenclamide-related excess in total and cardiovascular mortality risks: data from large Ukrainian observational cohort study. Diabetes Res Clin Pract. 2009;86:247–53.
Wang H, Cordiner RLM, Huang Y, et al. Scottish Diabetes Research Network Epidemiology Group. Cardiovascular safety in type 2 diabetes with sulfonylureas as second-line drugs: a nationwide population-based comparative safety study. Diabetes Care. 2023;46:967–77.
Douros A, Yin H, Yu OHY, Filion KB, Azoulay L, Suissa S. Pharmacologic differences of sulfonylureas and the risk of adverse cardiovascular and hypoglycemic events. Diabetes Care. 2017;40(11):1506–13.
Islam N, Ayele HT, Yu OHY, Douros A, Filion KB. Sulfonylureas and the risk of ventricular arrhythmias among people with type 2 diabetes: a systematic review of observational studies. Clin Pharmacol Ther. 2022;111(6):1248–57.
Davis TM, Parsons RW, Broadhurst RJ, Hobbs MS, Jamrozik K. Arrhythmias and mortality after myocardial infarction in diabetic patients. Relationship to diabetes treatment. Diabetes Care. 1998;21(4):637–40.
Eroglu TE, Jia L, Blom MT, et al. Sulfonylurea antidiabetics are associated with lower risk of out-of-hospital cardiac arrest: real-world data from a population-based study. Br J Clin Pharmacol. 2021;87(9):3588–98.
Simpson SH, Lee J, Choi S, Vandermeer B, Abdelmoneim AS, Featherstone TR. Mortality risk among sulfonylureas: a systematic review and network meta-analysis. Lancet Diabetes Endocrinol. 2015;3(1):43–51.
Maloney A, Rosenstock J, Fonseca V. A model-based meta-analysis of 24 antihyperglycemic drugs for type 2 diabetes: comparison of treatment effects at therapeutic doses. Clin Pharmacol Ther. 2019;105(5):1213–23.
Srivastava K, Arora A, Kataria A, Cappelleri JC, Sadosky A, Peterson AM. Impact of reducing dosing frequency on adherence to oral therapies: a literature review and meta-analysis. Patient Prefer Adher. 2013;7:419–34.
Baretić M, Bralić LV. Hypoglycemia in patients with type 2 diabetes treated with oral antihyperglycemic agents detected by continuous glucose monitoring: a multi-center prospective observational study in Croatia. BMC Endocr Disord. 2020;20(1):35.
Ibrahim M, Davies MJ, Ahmad E, et al. Recommendations for management of diabetes during Ramadan: update 2020, applying the principles of the ADA/EASD consensus. BMJ Open Diabetes Res Care. 2020;8(1): e001248.
Kidney Disease: Improving Global Outcomes (KDIGO) Diabetes Work Group. KDIGO 2020 clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int. 2020;98(4s):S1–115.
Abdelhafiz AH, Rodríguez-Mañas L, Morley JE, Sinclair AJ. Hypoglycemia in older people—a less well recognized risk factor for frailty. Aging Dis. 2015;6:156–67.
McAulay V, Frier BM. Hypoglycemia. In: Sinclair AJ, editor. Diabetes in old age. Chichester: John Wiley and Sons; 2009. p. 287–310.
Al Sifri S, Basiounny A, Echtay A, et al. 2010 Ramadan Study Group. The incidence of hypoglycaemia in Muslim patients with type 2 diabetes treated with sitagliptin or a sulphonylurea during Ramadan: a randomised trial. Int J Clin Pract. 2011;65(11):1132–40.
Aravind SR, Ismail SB, Balamurugan R, et al. Hypoglycemia in patients with type 2 diabetes from India and Malaysia treated with sitagliptin or a sulfonylurea during Ramadan: a randomized, pragmatic study. Curr Med Res Opin. 2012;28(8):1289–96.
Hassanein M, Al Sifri S, Shaikh S, et al. DIA-RAMADAN study investigators. A real-world study in patients with type 2 diabetes mellitus treated with gliclazide modifiedrelease during fasting: DIA-RAMADAN. Diabetes Res Clin Pract. 2020;163: 108154.
Hassanein M, Al-Arouj M, Hamdy O, et al. Diabetes and Ramadan: practical guidelines. Diabetes Res Clin Pract. 2017;126:303–16.
Hassanein M, Abdallah K, Schweizer A. A double-blind, randomized trial, including frequent patient-physician contacts and Ramadan-focused advice, assessing vildagliptin and gliclazide in patients with type 2 diabetes fasting during Ramadan: the STEADFAST study. Vasc Health Risk Manag. 2014;10:319–26.
Zargar AH, Siraj M, Jawa AA, Hasan M, Mahtab H. Maintenance of glycaemic control with the evening administration of a long acting sulphonylurea in male type 2 diabetic patients undertaking the Ramadan fast. Int J Clin Pract. 2010;64:1090–4.
Owen KR. Treating young adults with type 2 diabetes or monogenic diabetes. Best Pract Res Clin Endocrinol Metab. 2016;30(3):455–67.
Lachance CH. Practical aspects of monogenic diabetes: a clinical point of view. Can J Diabetes. 2016;40(5):368–75.
International Diabetes Federation. IDF clinical practice recommendations for managing type 2 diabetes in primary care. IDF. 2020. https://www.idf.org/e-library/guidelines/128-idf-clinicalpractice-recommendations-for-managing-type-2-diabetes-in-primary-care.html. Accessed 10 Jan 2023
Royal Australian College of General Practitioners (2020) General practice management of type 2 diabetes. 2016. https://www.racgp.org.au/FSDEDEV/media/documents/Clinical%20Resources/Guidelines/Diabetes/General-practice-management-of-type-2-diabetes_1.pdf. Accessed 10 Jan 2023
World Health Organization. Guidelines on second and third-line medicines and type of insulin for the control of blood glucose in non-pregnant adults with diabetes mellitus. Geneva: World Health Organization; 2021.
Diabetes Canada Clinical Practice Guidelines Expert Committee, Houlden RL. Clinical practice guidelines. Can J Diabetes. 2018;42(1):S1–325.
Cosentino F, Grant PJ, Aboyans V, et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2020;41(2):255–323.
Society of Endocrinology and Metabolism of Türkiye (SEMT). Diagnosis, treatment and follow-up guide of diabetes mellitus and its complication 2022 [July 2022]. https://file.temd.org.tr/Uploads/publications/guides/documents/diabetes-mellitus_2022.pdf. Accessed 20 Dec 2022
Medical Writing and Editorial Assistance
Kappa Training Consultancy and Research LLC, Izmir, Turkey provided the editorial support, funded by Servier Turkey.
Funding
This study was supported by Servier Turkey which played a role in organization of the expert panel meeting including the invitation of participants and compensation for the time and transport expenses of the experts. Servier Turkey had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Servier Affaires Médicales funded the Rapid Service Fee.
Author information
Authors and Affiliations
Contributions
All authors (Ibrahim Sahin, Okan Bakiner, Tevfik Demir, Ramazan Sari, Aysegul Atmaca) contributed equally to work. Ibrahim Sahin had primary responsibility for the manuscript preparation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Ibrahim Sahin, Okan Bakiner, Tevfik Demir, Ramazan Sari and Aysegul Atmaca declare that they have no conflicts of interest.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Sahin, I., Bakiner, O., Demir, T. et al. Current Position of Gliclazide and Sulfonylureas in the Contemporary Treatment Paradigm for Type 2 Diabetes: A Scoping Review. Diabetes Ther 15, 1687–1716 (2024). https://doi.org/10.1007/s13300-024-01612-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-024-01612-8