Abstract
Background and Objective
All latanoprost formulations currently available for the treatment of glaucoma or ocular hypertension contain the same concentration of latanoprost (0.005%) but differ in excipients, which may affect corneal drug permeability or stability. This study aimed at comparing corneal penetration of three marketed latanoprost solutions with different excipient formulations in in vitro and in vivo drug permeability studies.
Methods
Three latanoprost formulations were tested under good laboratory practice conditions: a formulation containing benzalkonium chloride (BAK) but no surfactant (Preserved latanoprost); the same formulation except preservative-free (PF) without BAK or surfactant (SF) (PF SF latanoprost); and a different formulation without BAK but containing a non-ionic surfactant (MGHS 40 at 5%) combined with thickening agents (Carbomer 974P, Macrogol 4000) (PF latanoprost). Corneal permeation of latanoprost acid (LAT) was first determined in vitro using a reconstructed human corneal epithelium tissue. Then, in vivo pharmacokinetic studies were performed on pigmented rabbits, for which LAT concentration was measured in the aqueous humour (AH) and iris–ciliary body (ICB).
Results
In vitro, the cumulative transport of LAT was linear between 1 h and 4 h for preserved latanoprost and PF SF latanoprost, and LAT concentrations matched exactly at each timepoint. By contrast, the permeation of PF latanoprost was linear between 2 h and 12 h and was significantly lower than that of preserved latanoprost and PF SF latanoprost at 4 and 8 h (p < 0.001). In rabbits, the concentrations of LAT in AH and ICB were not statistically different between preserved latanoprost and PF SF latanoprost at each timepoint, except at 1 h in ICB (p = 0.005). By comparison, the LAT concentration of PF latanoprost was statistically (p < 0.05) lower than that of preserved latanoprost and PF SF latanoprost in AH and ICB from 0.5 to 3 h.
Conclusion
BAK did not influence the corneal penetration of latanoprost in in vitro and in vivo studies. The formulation containing a non-ionic surfactant resulted in lower and slower ocular penetration compared with preserved or PF SF formulations. This raises questions about the relevance of BAK and some surfactants in enhancing corneal penetration of ocular formulations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The presence of BAK in the formulation did not influence the corneal penetration of latanoprost. |
The formulation containing a non-ionic surfactant resulted in lower and slower ocular penetration when compared with preservative-containing or preservative-free formulations without surfactant. |
The in vitro permeation model of latanoprost is a good predictor of its in vivo performance. This model could be further used to compare and discriminate formulations in terms of eye kinetic, allowing an ethical alternative to in vivo studies. |
1 Introduction
Glaucoma is the name given to a group of eye diseases that lead progressively to retinal ganglion cells death and optic neuropathy. Consequently, it can lead to severe irreversible visual field defect and ultimately loss of vision [1]. Intraocular pressure (IOP) and age are the major risk factors for the development of glaucoma. Because there is currently no curative therapy, treatment aiming at lowering IOP is nowadays the only alternative to slow glaucoma progression and prevent irreversible blindness. Ophthalmic prostaglandin F2 alpha analogues (latanoprost, bimatoprost, travoprost and tafluprost) are recommended as the first-choice treatment for glaucoma [2], along with beta-blockers. They selectively stimulate prostaglandin F receptors leading to the relaxation of the trabecular meshwork and ciliary muscle cells, and to the rapid decrease in outflow resistance mainly through the uveoscleral pathway, thus reducing IOP in glaucomatous patients [3, 4]. Side effects are mainly topical and include conjunctival hyperaemia, induced iris darkening, periocular skin pigmentation and eyelash changes [5]. Meibomian gland dysfunction, conjunctival goblet cell dropout, tear films alterations and pseudodendritic keratitis are overlooked conditions that may also result from prostaglandin analogues therapy but seem more frequent in benzalkonium chloride (BAK)-containing and multiple anti-glaucoma medications [6].
Latanoprost is an isopropyl ester prodrug rapidly absorbed in the cornea and then converted to latanoprost free acid (active metabolite) by corneal esterase enzymes [7]. Several latanoprost formulations are currently available on the market, all of which contain the same concentration of latanoprost (50 μg/mL). However, they differ in the type or amount of excipients, i.e. preservatives, stabilizers, thickeners and/or solubilizing agents. These additives are important components since they can affect corneal drug permeability and/or ocular surface toxicity [8]. Ocular side effects, including ocular discomfort, eye irritation or ocular surface disease in long-term use may affect adherence to the treatment and thus compromise treatment efficacy [9]. Thus, the corneal permeability of anti-glaucomatous drugs is generally optimized to improve ocular bioavailability and efficacy without increasing side effects.
BAK is a quaternary ammonium molecule that has a strong detergent effect on lipids and cell membranes. BAK is commonly used in ophthalmic preparations primarily for its antimicrobial activity [9]. In vitro and in vivo animal studies have suggested that BAK could enhance the corneal permeability of lipophilic products, although results in the literature are controversial [9]. Moreover, BAK is well known for its deleterious effect on the ocular surface, recurrently assessed in vitro and in animal and human studies [10,11,12].
The original latanoprost formulation (preserved latanoprost) has been very well studied in clinical trials and meta-analyses and was generally shown to be as effective as other prostaglandin analogues (bimatoprost, travoprost and tafluprost) and more effective than beta-blockers, carbonic anhydrase inhibitors and alpha 2-agonists [13]. It contains phosphate buffer, sodium hydrochloride and BAK at 0.02% and has been marketed worldwide for several decades. In 2013, another latanoprost formulation was approved. This solution without BAK contains a non-ionic surfactant [macrogolglycerol hydroxystearate 40 at 5% (MGHS 40)] with a combination of thickening agents (Carbomer 974P and Macrogol 4000) susceptible to increase stability and residence time on the ocular surface [14]. Recently, a new preservative-free (PF) latanoprost formulation without surfactant (SF) nor thickening agents (PF SF latanoprost) was also approved in several European countries. This new formula uses the refined formulation of the original product (preserved latanoprost) and does not contain BAK. These three formulations are comparable in terms of latanoprost concentration (0.005%) but are different in terms of excipient content (Table 1). Thus, the objective of our study was to compare the ocular penetration of these three formulations using in vitro reconstructed 3D corneal tissue and in vivo animal models, and to provide insights on the relevance of adding preservatives (BAK) or surfactants (MGHS 40) to improve ocular bioavailability.
2 Materials and Methods
The three formulations of latanoprost were used for in vitro and in vivo animal studies. They are all commercially available (Table 1).
2.1 In Vitro Experiments
In vitro experiments were subcontracted to Across Barriers GmbH, Saarbrucken, Germany. All experiments were performed in accordance with good laboratory practice (GLP) as defined by the Organization for Economic Co-operation and Development (OECD) guidelines [15] and the German “Chemikaliengesetz” (Bundesgesetzblatt (BGBI), Chemikaliengesetz-ChemG, 2013) [16].
2.1.1 In Vitro 3D Corneal Model
Human corneal-like epithelium tissues (EpiCorneal™) were provided from MatTek Life Sciences (Ashland, MA, USA). The commercial kit (Fig. 1) was composed of 3D corneal tissue grown in an individual microporous membrane cell culture inserts (COR-100, surface area 0.6 cm2) provided in sealed 24-well plates. The model has been previously fully standardized and validated for its relevance with native human corneal epithelium [17, 18], including tissue structures, thickness and barrier formation similar to the human cornea. Particularly, the apical epithelium layer expressed microvilli, tight junction proteins (ZO-1, occludin, claudin-1), desmosome and membrane-bound glycoprotein mucins as in excised human cornea. The model also showed a strong correlation with human corneal epithelium in terms of drug-metabolizing enzyme and transporter gene expression, including various esterases, responsible for the conversion of latanoprost ester to active latanoprost free acid [17]. This reconstructed 3D human corneal epithelial tissue was used for our in vitro permeation experiments.
EpiCornealTM tissue model. 3D human corneal tissues were grown in individual microporous membrane cell culture inserts (COR-100). The cell culture inserts were placed on a 12-well plate filled with 1 mL of culture medium [Hanks’ balanced salt solution (HBSS), pH 7.4]. Then, 100 μL of test formulations were applied in the donor compartment (topical exposure) and incubated at 37 °C. Samples were taken from the receiver compartment at different timepoints for latanoprost acid assay
2.1.2 In Vitro Corneal Permeability Studies
The tissue inserts were treated as defined and described by the manufacturer. The inserts were transferred to six-well plates filled with 1 mL of pre-warmed maintenance medium (COR-100-ASY, MatTek) under sterile conditions. The six-well plates were then incubated in a humidified incubator (37 ± 1 °C, 5 ± 0.5% CO2, 90 ± 10% Relative Humidity (RH)) overnight (18–24 h) before use for in vitro experiments.
For experiments, the COR-100 tissue inserts were transferred in 12-well plates prefilled with pre-warmed Hanks’ balanced salt solution (HBSS), pH 7.4 (Fig. 1). Then, 100 μL of the diluted test item (1:5, i.e. 10 μg/mL) or vehicle (HBSS) as negative control were applied in the donor compartment on a diffusion area of 0.6 cm2. Inserts were placed into the incubator (37 ± 1 °C, 5 ± 0.5% CO2, 90 ± 10% RH). Samples (150 μL) were then taken from the lower compartment (acceptor compartment) at different time intervals (0, 0.5, 1, 2, 3, 4, 5, 6, 8, 10 and 12 h). Indeed, a permeation study has been conducted previously by Gremilogianni et al. [19] on different latanoprost formulations in the same EpiCorneal model, allowing us the anticipation of latanoprost behaviour. After each sampling, the acceptor compartment was refilled with a pre-warmed receptor medium. The experiment was performed with six replicates per test item.
2.1.3 Transepithelial Electrical Resistance (TEER) Measurements
Preliminary experiments were performed to determine the highest tolerated concentration of the three tested formulations to be used for the permeation study. Tissue integrity was evaluated at the beginning (before test item application) and the end of the experiments using TEER measurement. TEER measurements were performed using the EVOMTM voltohmmeter with STX-2 or STX-3 electrodes (World Precision Instruments, Sarasota, FL, USA).
2.1.4 Analytical Method for Quantification of Latanoprost
Latanoprost acid and latanoprost ester were quantified using a validated high-performance liquid chromatography (HPLC)-UV method in accordance with the EMA Guideline for Bioanalytical method validation (EMA, Bioanalytical Method Validation, 2011). HPLC was conducted using the Acquity H-class Plus Arc apparatus (Waters Corporation, MA, USA). The separation was carried out on THERMO HYPERSILTM BDS Cyano reverse-phase column (250 × 4.6 mm, inner diameter 5 μm) (ThermoFisher Scientific, Rockford, IL, USA) using a mobile phase constituted of sodium phosphate buffer (pH 3.2) and acetonitrile/methanol 50:50 v/v % (64:36 v/v %) at a flow rate of 1.0–1.4 mL/min, with a run time of 55 min. The gradient starts after 25 min. The calibration curve in the HBSS medium was linear for latanoprost acid and latanoprost ester with an optimum detection at 210–260 nm. The lower limit of quantification (LLOQ) of the analytical method was 0.059 μg/mL for latanoprost acid and 0.150 μg/mL for latanoprost ester.
2.1.5 Cumulative Concentration, Apparent Permeability Coefficient and Flux Calculation
From the concentration of latanoprost ct [μg/mL] determined by HPLC at each timepoint, the amount sampled from the acceptor volume will be calculated. The corrected mass mcor (second fraction to last fraction) due to dilution of the acceptor comportment via sampling will be calculated according to the following equation, and then related to the exposed surface (0.6 cm2): \(m_{{{\text{cor}}}} = c_{t} \cdot V_{{{\text{acc}}}} + \sum\nolimits_{n}^{t - 1} {c_{i} \cdot V_{{{\text{sam}}}} ,}\) where Vacc is the acceptor volume (approximately 12 mL), ci the measured concentration of the several timepoints of the acceptor sample, and Vsam the sampling volume (0.32 mL). The cumulative concentration of latanoprost acid (μg/cm2) was plotted over time for each formulation.
The apparent permeation coefficients (Papp) were calculated using the following formula Papp = ΔQ/ΔT × 1/(A × C0), where ΔQ/ΔT (μg/s) is the permeability rate obtained by linear regression from the profile of the latanoprost in the acceptor compartment versus time, A is the exposed surface (0.6 cm2), and C0 is the initial latanoprost donor concentration (μg/cm2) in the apical compartment. The flux (μg/cm2/h) was calculated as the slope of the cumulated transport. The flux max (Jmax) was reported as the highest point of the flux curve over time.
2.2 Animal Studies
2.2.1 Approval and Guidelines
The ethical project application was submitted to and approved by the French Ministry of Research under the number #12827 on 8 January 2018. Animal experiments were performed by Iris Pharma, La Gaude, France, in accordance with the OECD Principles on GLP [15] and applicable facility Standard Operating Procedures (SOPs), Food and Drug Administration (FDA) and European Medicines Agency (EMA) requirements. All institutional and national guidelines for the care of the laboratory animals were followed; thus, all animals were treated according to the Directive 2010/63/UE European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes and to the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research.
2.2.2 In Vivo Study
Prior to experiments, the rabbits were housed by two in standard cages and allowed free access to food and water. The in vivo experiment ran from 29 November 2021 to 14 December 2021. A total of 126 female Dutch Belted pigmented rabbits (Envigo, Denver, PA, USA) aged 12–15 weeks were randomized into three study groups of 42 animals (group preserved latanoprost, average weight 1300 ± 110 g; group PF SF latanoprost, average weight 1307 ± 100 g; group PF latanoprost, average weight 1292 ± 111 g). Each rabbit received a single instillation of 50 μL of the test item in the upper corneo-limbic junction of the right eye using an appropriate micropipette. Rabbits were then euthanized at different times, i.e. 0.5, 1, 2, 3, 4, 8 and 24 h by intravenous injection of pentobarbital (Doléthal®, 0.6 mL/kg) following a pre-sedation (recommended methods for euthanasia by the European authorities).We relied on the data available in the literature, e.g. Sekine et al. [20] (pharmacokinetic in human eyes) and a latest animal pharmacokinetic study of Halder et al. [21], to choose wisely and according to the 3Rs principle [22] the most relevant timepoints. Each experiment was performed in six replicates in line with the GLP guidelines. Immediately after euthanasia, aqueous humour (AH) and iris–ciliary body (ICB) from the right eyes were carefully sampled in separate polypropylene tubes, weighed and stored at −23 ± 7 °C until extraction procedures and analysis.
2.2.3 Bio-analytic
The AH and ICB samples (50 µL AH or ½ ICB) containing the test item latanoprost free acid and the internal standard, i.e. latanoprost free acid-d4 (Cayman Chemical, Ann Arbor, MI, USA), at 50 ng/10 µL were thawed at room temperature and treated with 10 µL hydrochloric acid (1 M) and 1000 µL ethyl acetate. After centrifugation, the organic layer was evaporated under a nitrogen stream at about 40 °C, and the dry residue was dissolved in 100 µL of matrix solution [90 mL of solution phase (formic acid 0.1%, acetonitrile and MeOH) added to 10 mL of acetonitrile]. An analytical procedure using rapid resolution liquid chromatography (RRLC) with tandem mass spectrometry (MS/MS) detection (Agilent 1200 Triple Quad G6410B, Agilent Technologies, Les Ulys, France) (positive mode) was developed and validated for the quantification of latanoprost free acid in AH and ICB after ionisation using an electrospray interface (ESI). Separation was performed using a Halo C18 column (100 × 2.1 mm, 2.7 μm) at 40 °C. The mobile phase was composed of formic acid 0.1% (45%) and acetonitrile/methanol 35/20 (55%) at a flow of 0.2 mL/min. The LLOQ of latanoprost acid was 10 ng/mL of AH and 14 ng/g of ICB.
2.3 Statistics
For in vitro corneal permeation experiments, arithmetic means of cumulative transport of latanoprost acid (n = 6 single values at each timepoint) and permeation coefficients from 1 to 4 h and from 1 to 8 h were compared between formulations by two-sided unpaired t-tests with a significance level of 0.05. For all parametric tests, the following prerequisites should be met: (1) normal distribution of the two samples to be compared (Shapiro–Wilk test at a significance level of 0.05) and (2) equality of variance of the two samples to be compared (at a significance level of 0.05). The Welch modification was applied in case of unequal variances.
In animal experiments, pharmacokinetic parameters were calculated from Microsoft Excel® software (version 16.50). Mean latanoprost free acid concentrations in ocular tissues were compared between groups at each timepoint using the two-way ANOVA (not repeated measures) Fisher’s least significant difference test without correction for multiple comparisons. The significant threshold was fixed at 0.05. In this animal study, ocular sampling could not be a “repeated sampling” on the same animal. Thus, a given animal could provide only one value of latanoprost free acid concentration for each structure at a given timepoint. Consequently, pharmacokinetic parameters, i.e. maximal concentration (Cmax), time to maximal concentration (Tmax) and area under the curve (AUC), were calculated using the mean value of the group, and no standard deviation could be determined for Tmax and AUC as only one value per group at each timepoint of the kinetic was available. AUC were calculated with linear trapezoidal method using linear interpolation between data points. The analysis was performed using the software GraphPad Prism (version 6.0h) (GraphPad Software, San Diego, CA, USA).
3 Results
3.1 Corneal Permeation
As shown in Fig. 2, the transport profile of latanoprost showed radically different shapes between preserved latanoprost or PF SF latanoprost and PF latanoprost. The cumulative transport of latanoprost acid for preserved latanoprost and PF SF latanoprost matched exactly and showed a linear increase between 1 h and 4 h, followed by a steady-state phase. By contrast, the transport of latanoprost acid for PF latanoprost increased linearly between 2 h and 12 h. The cumulative transport of latanoprost acid after 240 min (4 h) was significantly lower for PF latanoprost (0.475 ± 0.018 μg/cm2) than for preserved latanoprost (1.021 ± 0.048 μg/cm2, p < 0.001) or PF SF latanoprost (1.011 ± 0.047 μg/cm2, p < 0.001). After 480 min (8 h), the cumulative transport of latanoprost was still significantly lower for PF latanoprost (1.004 ± 0.025 μg/cm2) compared with preserved latanoprost (1.227 ± 0.060 μg/cm2, p < 0.001) or PF SF latanoprost (1.224 ± 0.057 μg/cm2, p < 0.001). After 720 min (12 h), the cumulative transport of latanoprost acid was similar for all formulations: 1.283 ± 0.065 μg/cm2 for preserved latanoprost; 1.279 ± 0.061 μg/cm2 for PF SF latanoprost; and 1.311 ± 0.034 μg/cm2 for PF latanoprost (p > 0.05, all comparisons). There was no statistically significant difference between preserved latanoprost and PF SF latanoprost at any timepoint (p > 0.05).
Cumulative transport of latanoprost acid from different formulations. The latanoprost acid in the acceptor compartment was quantified, and cumulative amounts per cm2 were plotted over time. Data are arithmetic means ± standard deviation of six independent experiments; *p < 0.001 (unpaired two-sided t-test) PF latanoprost versus preserved latanoprost or PF SF latanoprost. PF preservative-free, SF surfactant free
The permeation coefficient (Papps) of the linear part of the transport curves was significantly lower (p < 0.001) for PF latanoprost (3.14 ± 0.14 × 10−6 cm/s) compared with preserved latanoprost (8.47 ± 0.40 × 10−6 cm/s) or PF SF latanoprost (8.52 ± 0.47 × 10−6 cm/s). Tmax was higher for PF latanoprost (3 h) compared with preserved latanoprost or PF SF latanoprost (2 h).
Regarding latanoprost ester at the end of the transport experiment, it was detected in the donor compartment for PF latanoprost (2.241 μg/mL), but not for preserved latanoprost or PF SF latanoprost.
A tolerance test was performed by TEER, which is a sensitive method to evaluate tissue barrier damage (Fig. 3). TEER values at the end of the incubation remained above 900 ohms cm2, similar to the negative control (HBSS, pH 7.4), indicating that the tissues possessed a functional barrier [17].
3.2 Ocular Pharmacokinetic Parameters in Rabbits
As shown in Fig. 4A, the kinetics profiles of preserved latanoprost and PF SF latanoprost in the AH were quite similar without statistically significant differences at any timepoint (p > 0.05). Latanoprost free acid concentrations were significantly lower for PF latanoprost compared with preserved latanoprost-treated animals at the 0.5 h timepoint [mean difference (MD) of 42.4 ng/mL, p = 0.0018], 1 h (MD: 60.5 ng/mL, p < 0.0001), 2 h (MD: 80.1 ng/mL, p < 0.0001) and 3 h (MD: 61.7 ng/mL, p < 0.0001). Similarly, concentrations of latanoprost free acid were significantly lower for PF latanoprost-treated animals compared with PF SF latanoprost-treated animals at the 0.5 h timepoint (MD: 54.7 ng/mL, p < 0.0001), 1 h (MD: 86.6 ng/mL, p < 0.0001), 2 h (MD: 97.5 ng/mL, p < 0.0001) and 3 h (MD: 42.3 ng/mL, p = 0.0018). AUC0.5–24h was quite similar for preserved latanoprost (470 ng·h/mL) and PF SF latanoprost (521 ng·h/mL), but at least twice as low for PF latanoprost (210 ng·h/mL), indicating that latanoprost free acid penetration in AH was higher for preserved latanoprost and PF SF latanoprost than for PF latanoprost treatments. Tmax was shorter for preserved latanoprost and PF SF latanoprost (2 h) compared with PF latanoprost (3 h). Cmax was two to three times higher for preserved latanoprost (128 ng/mL) and PF SF latanoprost (145 ng/mL) compared with PF latanoprost (48 ng/mL).
Ocular bioavailability of latanoprost acid from different formulations in rabbits. Concentrations of latanoprost acid were determined at different timepoints per millilitre of aqueous humour (A) and per gram of iris–ciliary body (B) after a single administration of 50 μL of each test item formulation. Data are arithmetic means ± standard deviation of six independent experiments. *p < 0.05, PF latanoprost versus preserved latanoprost or PF SF latanoprost; ‡ p < 0.05 PF SF latanoprost versus preserved latanoprost. AH aqueous humour, ICB iris–ciliary body, PF preservative-free, SF surfactant free
In ICB (Fig. 4B), there was no significant difference between preserved latanoprost and PF SF latanoprost in latanoprost acid concentration in the ICB except at the 1 h timepoint (MD: 32.5 ng/g, p = 0.0053). Latanoprost free acid concentrations in ICB were significantly lower for PF latanoprost-treated animals compared with preserved latanoprost at the 0.5 h timepoint (MD: 45.9 ng/g, p = 0.0001), 1 h (MD: 48.3 ng/g, p < 0.0001), 2 h (MD: 45.0 ng/g, p = 0.0001) and 3 h (MD: 26.2 ng/g, p = 0.0236). Similarly, latanoprost free acid concentrations were significantly lower for PF latanoprost-treated animals compared with PF SF latanoprost-treated animals at the 0.5 h timepoint (MD: 64.5 ng/g, p < 0.0001), 1 h (MD: 80.8 ng/mL, p < 0.0001), 2 h (MD: 54.0 ng/mL, p < 0.0001) and 3 h (MD: 27.7 ng/g, p = 0.0171). AUC0.5–24h was quite similar between preserved latanoprost (269 ng·h/mL) and PF SF latanoprost (300 ng·h/mL) and more than twice lower for PF latanoprost (97 ng·h/mL), indicating that latanoprost free acid penetration in ICB was higher for preserved latanoprost and PF SF latanoprost than for PF latanoprost treatments. Cmax were three to four times higher for preserved latanoprost (72 ng/g) and PF SF latanoprost (104 ng/g) compared with PF latanoprost (25 ng/mL). Tmax was 1 h for preserved latanoprost and PF SF latanoprost and 0.5 h for PF latanoprost.
3.3 Bio-analytic
The method for the determination of latanoprost free acid in rabbit AH and ICB using HPLC with MS/MS detection has been fully validated with respect to selectivity, linearity, precision, accuracy, matrix effect, extraction efficiency and dilution integrity, spanning a theoretical concentration range of 10–2000 ng/mL and 100–20,000 ng/mL for AH and 13–2594 ng/g and 130–25,940 ng/g for ICB (Figs. 5 and 6). Stability evaluations in AH and ICB and solutions have also met acceptance criteria, demonstrating insignificant degradation of latanoprost free acid and latanoprost free acid-d4 over the specified storage durations and conditions.
Bio-analytical validation—selectivity in AH and ICB. Assay selectivity was assessed by employing six independent sources of AH and ICB and verifying for the presence of interference, compared with the LLOQ, at the retention times and mass transitions of latanoprost free acid or internal standard. No significant interference was observed in the six blanks AH and ICB. Representative chromatograms of extracted blank AH and ICB samples are presented (A and B, respectively). MRM conditions of latanoprost acid and latanoprost acid d4 are shown in tabular form (C). AH aqueous humour, ICB iris–ciliary body, LLOQ lower limit of quantification, MRM multiple reaction monitoring, RT retention time
Bio-analytical validation—sensitivity in AH and ICB. Assay sensitivity was assessed at a LLOQ concentration for latanoprost free acid. The LLOQ signals were > 5 times the height of the baseline noise. Representative chromatograms of an extracted LLOQ and ULOQ sample are presented for AH (A and B, respectively) and ICB (C and D, respectively). AH aqueous humour, ICB iris–ciliary body, LLOQ lower limit of quantification, MRM multiple reaction monitoring, RT retention time, ULOQ upper limit of quantification
4 Discussion
In this study, we compared the corneal penetration of latanoprost from three different ophthalmic formulations containing or not a presumed permeation enhancer, i.e. BAK or a non-ionic surfactant, i.e. MGHS 40. Using an in vitro model of human corneal epithelial tissue, we showed that BAK had no significant influence on corneal permeability of latanoprost as both formulations with (preserved latanoprost) or without (PF SF latanoprost) BAK produced the same corneal penetration of latanoprost. By comparison, the formulation containing a non-ionic surfactant combined with thickening agents (PF latanoprost) had the slowest and lowest permeability.
These results were confirmed in vivo by the ocular pharmacokinetic of latanoprost acid in the AH and ICB following a single dose administration in pigmented rabbits. The 3D corneal in vitro tissue model used in this study proved to be functional and highly reproducible in several other studies [17, 18]. Evidence has shown that it possesses similar tissue structure, barrier properties, and expression of cornea-specific markers to the in vivo human cornea. This in vitro model was found valuable to determine corneal permeation and corneal toxicity of ophthalmic preparations as an alternative to in vivo tests which required sacrificing a number of animals. Our study also showed consistent ocular penetration of latanoprost between in vitro and in vivo experiments in rabbits, demonstrating that the in vitro permeation model is a good predictor of the in vivo behaviour of latanoprost. Such in vitro model may be useful to compare and discriminate ophthalmic formulations in terms of eye kinetics and could be an ethical alternative to in vivo studies.
The role of BAK to enhance corneal permeability has long been suggested for drugs with poor ocular bioavailability such as beta-blockers [23]. However, the addition of this preservative has been controverted since the enhancer effect is clearly associated with significant corneal toxicity through disruption of epithelial tight junction, especially at concentrations above 0.005% [10].
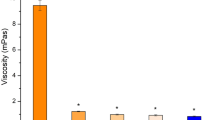
In our in vitro study, the permeation coefficient of the diluted formulation containing BAK at a final concentration of 0.004% (preserved latanoprost) was not statistically different compared with the same formulation without BAK (PF SF latanoprost). We used a diluted formulation since preliminary results (Fig. 7) showed that the non-diluted formulation containing BAK (0.02%) or a half-diluted formulation (BAK 0.01%) induced a significant drop in TEER compared to the control (culture medium) indicating a toxic effect on the corneal epithelial tissue [17].
In rabbits after one single dose (50 μL) administration of a non-diluted tested product, we found no relevant difference in pharmacokinetic parameters of latanoprost acid in the AH and ICB between the formulation containing BAK 0.02% (preserved latanoprost) and the same formulation without BAK (PF SF latanoprost), thus suggesting that BAK at high concentration does not affect the corneal penetration of latanoprost in these tissues. Consistent results suggesting no effect of BAK on the ocular bioavailability of latanoprost in rabbits have been published by others. Zhou et al. [24] showed no difference in latanoprost concentrations in AH after single dosing between the reference BAK-preserved formulation and a new PF formulation (composition not known). Halder et al. showed similar latanoprost pharmacokinetic parameters in AH after one single dose administration in rabbits between the reference formulation containing BAK 0.02% and a BAK-free latanoprost formulation containing potassium sorbate as a preservative [21]. Since BAK is considered to be toxic for the ocular surface [10,11,12], latanoprost eyedrop formulations should be exempted from this preservative.
These in vitro and in vivo results are consistent with the latanoprost penetration in human eyes. In patients undergoing phacoemulsification, Sekine et al. [20] compared the effect of various preserved and unpreserved latanoprost formulations on latanoprost acid penetration into the AH. They reported minimal pharmacokinetic differences between formulations, including BAK-preserved latanoprost, and independently of the excipients. Regarding the formulation containing the non-ionic surfactant MGHS 40 (PF latanoprost), we showed significantly lower latanoprost corneal permeability compared with the two other tested formulations without surfactant (preserved latanoprost and PF SF latanoprost, p < 0.001). Similar results were reported by Romaniuk et al. using a different human corneal epithelium culture model [25]. The lower permeation is not due to the absence of BAK, but rather to other excipients in the formulation which may limit the corneal penetration or the conversion rate of latanoprost ester to latanoprost acid by corneal epithelial cells. MGHS 40, Macrogol 4000 and carbomer 974P are used in this formulation to ensure the stability of latanoprost in solution [26]. The carbomer (thickening agent) is indeed widely used in artificial tears to enhance retention time [27]. The Macrogol 4000 (co-thickening and co-solubilizer agent) aims to ensure the desired level of viscosity and potentiates the solubilization of the active ingredient [28]. Although this excipient combination should prolong the residence time on the ocular surface, the ocular bioavailability of latanoprost is not improved compared with PF SF latanoprost and preserved latanoprost. Indeed, in our rabbit model, the latanoprost acid (active form) concentrations for PF latanoprost were decreased at the action site (ICB) and reservoir (AH) level at all timepoints. Thus, we assume that the stabilizing effect lowered the conversion rate of latanoprost ester in the reconstituted corneal epithelial tissue model. Indeed, our results showed that the latanoprost transport through the corneal epithelium tissue model was not concentration dependent for PF latanoprost formulation (not diluted, dilution 1:1, dilution 1:4), contrary to PF SF latanoprost and preserved latanoprost. This may be caused by a decreased metabolism of latanoprost ester to latanoprost acid likely due to the entrapment of the active substance into the excipients matrix. Altogether, the in vivo studies in rabbits confirmed lower ocular bioavailability (Cmax and AUC) in the non-ionic surfactant formulation compared with the two other formulations.
The in vitro EpiCorneal model was previously validated by Kaluzhny et al. [17]. Their results confirmed that 3D corneal tissues possess the functional esterase and amidase activity necessary for metabolic conversion of ester and amide prodrugs and can be utilized for in vitro evaluation of transcorneal permeability of prodrug candidates. Moreover, to evaluate model performance, a system suitability test was performed with the reference compound Latanoprost acid in one concentration within the linear range of the analytical method in six-fold determination previous to our experiment, to monitor the recovery percentage. The set specification was fulfilled, and TEER values clearly indicated that the tissues possessed a functional barrier throughout the experiment. Thus, we considered that no positive control was needed.
Regarding study limitations, there are major differences between latanoprost application in the in vitro corneal epithelial tissue model and clinical administration of latanoprost in humans, including the latanoprost concentration in the tissue culture, the volume and the duration of exposure. In vitro, extended exposure of the corneal epithelium for several hours may result in increased corneal hydration and affect corneal permeability; this is suggested in our study by a significant decrease of the TEER of about 25% in tissue cultured exposed only to the culture medium. The 3D human corneal epithelium tissue used for our experiments is also not fully equivalent to intact corneal tissue since the stroma and the endothelial layers are lacking in this model. Nevertheless, it is known that the corneal permeation is primarily limited to the hydrophobic epithelium barrier while the stroma is the less constrained filter layer for the passage of drugs, and the endothelium is a leaky lipophilic layer without a significant barrier for the ocular drug absorption [29]. In addition, when applied in humans, latanoprost is rapidly eliminated after application by various routes (i.e. tear film dilution, conjunctiva absorption and lacrimal duct) with a retention time of only several minutes [29]. Nevertheless, the in vitro results are well correlated to in vivo ocular bioavailability determined in rabbits. Thus, the in vitro model appears valuable to study drug permeability of latanoprost, especially in preliminary experiments. Finally, in rabbits’ experiments, the ocular pharmacokinetics was determined after one single dose administration, and thus the effect of BAK or the non-ionic surfactant of repeated doses is unknown. Nevertheless, Zhou et al. reported no difference in ocular pharmacokinetic parameters after multiple dose administration of a preserved (BAK) and a PF formulation once daily for 7 days [24].
5 Conclusions
Both in vitro and in vivo studies consistently show that all latanoprost formulations are not equivalent in terms of bioavailability. The new PF SF formulation of latanoprost has a quite similar corneal penetration compared with the reference BAK-preserved latanoprost. The addition of surfactant (MGHS 40 at 5%), combined with thickening agents (Carbomer 974P and Macrogol 4000) in PF latanoprost markedly impairs the corneal permeation and the ocular bioavailability of the active substance. Study assessments reported on latanoprost formula containing BAK or non-ionic surfactants show no related enhancement of corneal drug penetration. These excipients are moreover controversial as published data highlight corneal toxicity following their long-term use. Nowadays, preservative-free, surfactant-free formulations have been developed and seem to be the best available option to improve product tolerance without compromising efficacy. Such optimized formulations could better maintain long-term corneal integrity and thus facilitate adherence to treatment, even though this remains to be investigated in further studies.
References
Stein JD, Khawaja AP, Weizer JS. Glaucoma in adults-screening, diagnosis, and management: a review. JAMA. 2021;325(2):164–74.
European Glaucoma Society Terminology and Guidelines for Glaucoma, 5th edn. Br J Ophthalmol. 2021;105(Suppl 1):1–169.
Winkler NS, Fautsch MP. Effects of prostaglandin analogues on aqueous humor outflow pathways. J Ocul Pharmacol Ther. 2014;30(2–3):102–9.
Doucette LP, Walter MA. Prostaglandins in the eye: function, expression, and roles in glaucoma. Ophthal Genet. 2017;38(2):108–16.
Holló G. The side effects of the prostaglandin analogues. Expert Opin Drug Saf. 2007;6(1):45–52.
Ruiz-Lozano R, Azar N, Mousa H, Quiroga-Garza M, Komai S, Wheelock-Gutierrez L, et al. Ocular surface disease: a known yet overlooked side effect of topical glaucoma therapy. Front Toxicol. 2023;5:1067942. https://doi.org/10.3389/ftox.2023.1067942.
Basu S, Sjöquist B, Stjernschantz J, Resul B. Corneal permeability to and ocular metabolism of phenyl substituted prostaglandin esters in vitro. Prostaglandins Leukot Essent Fatty Acids. 1994;50(4):161–8.
Moiseev RV, Morrison PWJ, Steele F, Khutoryanskiy VV. Penetration enhancers in ocular drug delivery. Pharmaceutics. 2019;11(7):321.
Holló G, Katsanos A, Boboridis KG, Irkec M, Konstas AGP. Preservative-free prostaglandin analogs and prostaglandin/timolol fixed combinations in the treatment of glaucoma: efficacy, safety and potential advantages. Drugs. 2018;78(1):39–64.
Kusano M, Uematsu M, Kumagami T, Sasaki H, Kitaoka T. Evaluation of acute corneal barrier change induced by topically applied preservatives using corneal transepithelial electric resistance in vivo. Cornea. 2010;29(1):80–5.
Baudouin C, Labbé A, Liang H, Pauly A, Brignole-Baudouin F. Preservatives in eyedrops: the good, the bad and the ugly. Prog Retin Eye Res. 2010;29(4):312–34.
Baudouin C, Denoyer A, Desbenoit N, Hamm G, Grise A. In vitro and in vivo experimental studies on trabecular meshwork degeneration induced by benzalkonium chloride (an American Ophthalmological Society thesis). Trans Am Ophthalmol Soc. 2012;110:40–63.
Alm A. Latanoprost in the treatment of glaucoma. Clin Ophthalmol Auckl NZ. 2014;8:1967–85.
Chiambaretta F, Pouliquen P, Menerath JM, Pilotaz F, Rebika H, Rigal D. Efficacy and safety of a fluid carbomer gel versus a conventional carbomer gel in dry eye treatment. J Fr Ophtalmol. 2004;27(2):130–5.
Organization for Economic Co-operation and Development (OECD), ENV/MC/CHEM(98)17, OECD series on principles of good laboratory practice and compliance monitoring No. 1, OECD Principles on Good Laboratory Practice (as revised in 1997) (1999) OECD principles of good laboratory practice (OECD, Principles of GLP, 1999). https://read.oecd-ilibrary.org/environment/oecd-principles-on-good-laboratory-practice_9789264078536-en#page1. Accessed 5 Sept 2023.
Bundesgesetzblatt (BGBI). Gesetz zum Schutz vor gefährlichen Stoffen (Chemikaliengesetz-ChemG), 2013.
Kaluzhny Y, Kinuthia MW, Truong T, Lapointe AM, Hayden P, Klausner M. New human organotypic corneal tissue model for ophthalmic drug delivery studies. Invest Ophthalmol Vis Sci. 2018;59(7):2880–98.
Kaluzhny Y, Klausner M. In vitro reconstructed 3D corneal tissue models for ocular toxicology and ophthalmic drug development. In Vitro Cell Dev Biol Anim. 2021;57(2):207–37.
Gremilogianni AM, Kaluzhny Y, Tsoli C, Pantazopoulou S, Kinuthia M. Permeation of latanoprost eye drop formulations in epicorneal. Mat Tek Life Sci—Soc Toxicol 2016 Annu Meet New Orleans, USA, 2016.
Sekine Y, Shimada M, Satake S, Okubo M, Hisaka A, Hara T, et al. Pharmacokinetic analysis of intraocular penetration of latanoprost solutions with different preservatives in human eyes. J Ocul Pharmacol Ther Off J Assoc Ocul Pharmacol Ther. 2018;34(3):280–6.
Halder A, Khopade AJ. Pharmacokinetics and pharmacology of latanoprost 0.005% without benzalkonium chloride vs latanoprost 0.005% with benzalkonium chloride in New Zealand white rabbits and beagles. Curr Eye Res. 2021;46(7):1031–7.
EMA. Guideline on the principles of regulatory acceptance of 3Rs (replacement, reduction, refinement) testing approaches, 2016. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-principles-regulatory-acceptance-3rs-replacement-reduction-refinement-testing-approaches_en.pdf. Accessed 5 Sept 2023.
Fukuda M, Sasaki H. The transcorneal penetration of commercial ophthalmic formulations containing timolol maleate in rabbit eyes. J Ocul Pharmacol Ther Off J Assoc Ocul Pharmacol Ther. 2015;31(1):57–60.
Zhou T, Miao Y, Li Z, Lu P, Liang Z, Yang J, et al. A comparative ocular pharmacokinetics study of preservative-free latanoprost unit-dose eye drops and a benzalkonium chloride-preserved branded product following topical application to rabbits. J Ocul Pharmacol Ther Off J Assoc Ocul Pharmacol Ther. 2020;36(7):522–8.
Romaniuk D, Smagur J, Cisek A, Antonik J, Romaniuk W. Does preservative content affect drug permeability? The permeability analysis in vitro of 3 ophthalmic Latanoprost formulations in a human epithelial cell culture model. Klin Oczna. 2016;118(2):127–32.
Ochiai A, Danjo K. The stabilization mechanism of latanoprost. Int J Pharm. 2011;410(1–2):23–30.
Wilson CG, Zhu YP, Frier M, Rao LS, Gilchrist P, Perkins AC. Ocular contact time of a carbomer gel (GelTears) in humans. Br J Ophthalmol. 1998;82(10):1131–4.
Mercier F, inventor; Laboratoires Théa, assignee. Polymeric system for delivering a preservative-free prostaglandin-based nonviscous solution. European patent EP 2588078B1, 2014.
Mofidfar M, Abdi B, Ahadian S, Mostafavi E, Desai TA, Abbasi F, et al. Drug delivery to the anterior segment of the eye: a review of current and future treatment strategies. Int J Pharm. 2021;607: 120924.
Acknowledgments
We thank Dr Eleonore Haltner-Ukomadu and M. Manuel Sacha for technical support, data collection and analysis in the context of the permeation study. We also thank Iris Pharma for their expertise and the performance of the in vivo animal testing. We thank Thierry Radeau for editorial assistance in the preparation of this article (PhD Freelance Medical Writer (Epinay-sous-Sénart, France)). Support for this assistance was funded by Horus Pharma, France.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Author Contributions
Laure Chauchat and Camille Guerin contributed to study conception, design, and result discussion, and article outlines. All authors reviewed and approved the manuscript. All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole and have given their approval for this version to be published. The study was sponsored by Horus Pharma (Nice, France).
Conflict of Interest
Laure Chauchat was employed by Horus Pharma Company. Camille Guerin was employed by Horus Pharma Company. Yulia Kaluzhny was employed by MatTek Life Sciences, a manufacturer of the EpiCorneal tissue model used in the study.
Compliance with Ethics Guidelines
In vitro experiments were performed on the basis of OECD Guidelines for Testing of Chemicals and the German “Bundesgesetzblatt (BGBI) Chemikaliengesetz” (ChemG, 2013). Animal experiments were performed in accordance with the OECD Principles on good laboratory practice (GLP) and applicable facility Standard Operating Procedures (SOPs), Food and Drug Administration (FDA) and European Medicines Agency (EMA) requirement. All animals were treated according to the Directive 2010/63/UE European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes and to the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research. The registration number with the French Ministry of Research is #12827. This DAP (ethical project authorization request) was authorized by the Ministry on 8 January 2018 (and valid for 5 years). The ethics committee that approved this project is CEEA-087.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Prior Presentation
This work has been previously presented at the ARVO Annual Meeting, May 1–4, Denver, CO, 2022.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Code Availability
Not applicable.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Chauchat, L., Guerin, C., Kaluzhny, Y. et al. Comparison of In Vitro Corneal Permeation and In Vivo Ocular Bioavailability in Rabbits of Three Marketed Latanoprost Formulations. Eur J Drug Metab Pharmacokinet 48, 633–645 (2023). https://doi.org/10.1007/s13318-023-00853-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-023-00853-5