Abstract
The valorization of phosphate mine waste (PMW) in different fields has recently become attractive to many researchers. This review paper examines the potential use of PMW as a raw material in different applications toward sustainable management strategies. Dewatering, recovery of phosphate and heavy metals, building materials and ceramics applications have been captured in this paper. Based on the origin of deposit and the treatment during ore beneficiation, the PMW shows different mineral compositions and various percentage of chemical elements which make it suitable for most of the studied applications. The objective of this paper is to show the potential of the phosphate mine waste valorization and to identify the knowledge gap.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Human civilization demands mineral extraction for the basic amenities of a comfortable daily life. Mining played an essential role in the evolution and vigorous development of modern technology (Graedel et al. 2015; Jowitt et al. 2020). However, this advancement comes with harmful effects, frequently reported as socio-ecological hazards, such as heavy metal leaching, soil contamination, radioactive elements, acid mine drainage, and so on (The U.S. Department of Agriculture (USDA) 1998; Haddaway et al. 2019). Among various mining-related issues, managing and handling of the mine residues (mainly tailings) have become a considerable burden to the extractive industries due to the large volume being stored in dammed ponds near the mining sites, posing risks to the environment and community in the proximity (Bowker and Chambers 2017; Lyu et al. 2019). Tailings are part of the material extracted from the earth crust and discarded during mineral separation processing with "no economic value" (Kiventerä et al. 2020). Accumulation of tailings increases every year due to the increase in demand for raw materials and the lack of technology to efficiently handle such mining wastes. In recent days, mining of low-grade ores further accelerated this scenario and environmental regulatory norms on "mining waste storage practices" pressurizes the mining industry to explore alternatives for tailings disposal facilities (The European Parliament and Council of the EU 2006).
Tailings have untapped potential in several proven applications such as metal recovery (Evdokimov and Evdokimov 2015; Falagán et al. 2017; Alcalde et al. 2018), alkali activated Materials (Moukannaa et al. 2019; Dabbebi et al. 2020; Perumal et al. 2020, 2021b) and several other applications (Solismaa et al. 2018; Wang et al. 2019; Perumal et al. 2021a; Ramanathan et al. 2021). Properties of mine tailings vary with the mineralogy of the ore deposit extracted and the method of refinement (or) processing adopted during mineral separation (Kinnunen et al. 2018; Perumal et al. 2019). In most cases, tailings need to undergo several treatments before being declared potential raw materials for any secondary applications (Morone et al. 2014; Wang et al. 2014; Perumal et al. 2019; Niu et al. 2020b). Furthermore, a suitable application for a particular tailing may not be feasible with others due to their non-uniform physical and chemical nature. For instance, Perumal et al. (2019) studied thermal treatment on three different tailings to utilize those tailings in alkali activated materials. However, with similar process conditions, only impure kaolinite and phosphate tailings responded to the thermal treatment, whereas lithium tailings were identified to be unsuitable for application in alkali activated materials (Perumal et al. 2019). This proves that each tailing from a different mineralogical origin should be considered an independent material, to tap their complete reuse potential.
There are several review articles about the reuse potential and alternative strategies for mine tailings management (Edraki et al. 2014; Kinnunen et al. 2018; Kiventerä et al. 2020; Eskanlou and Huang 2021; Xiaolong et al. 2021). Nevertheless, it is valuable to analyze the tailings based on their mineralogy and origin, which would help in developing a well-defined management strategy. Hence, in this paper, an attempt is made to outline the wastes that originated exclusively from phosphate mining and their utilization potential in different applications. Phosphate mines mainly operate on the extraction of P2O5 and leave out other oxides that contain silicon (Si), calcium (Ca), aluminum (Al) and traces of phosphorus (P) in the form of fluorapatite, aluminosilicate, quartz and carbonates as wastes. Due to the intensive processing and treatment involved in phosphorus extraction, the waste generated in different processing levels varies in its physical and chemical properties. Phosphate mine wastes (PMWs) include slimes, sludge, clay, tailings and waste rocks (Abdel-Khalek et al. 2000a; Loutou et al. 2017; Moukannaa et al. 2018; Eskanlou and Huang 2021). However, these materials that are considered as "zero-value" waste have enormous potential as raw materials in secondary applications. Such research studies conducted on the industrial mining wastes are proven to be beneficial to the mining industry and the environment (Edraki et al. 2014). The motivation of this review is to understand the possibilities of utilizing the PMWs in secondary application for better environmental, social and economic benefits. Focus is made to emphasize the variation in the properties of phosphate mine waste generated from different locations worldwide and to envision several beneficial actions toward potential utilization of PMWs. This comprehensive review of literature aims to throw light on the area of current research and to identify the knowledge gaps.
The paper is structured into three main sections. Where the first section is about phosphate mining process and wastes generated. The following two parts are dedicated to the properties of the PMWs and sustainable management strategies, including dewatering, metal recovery, ceramics and building materials applications.
Review methodology
The review is based on the literature accumulated using studies reported in peer reviewed journals, patents and books. Papers from 1970 to 2021 were reviewed to investigate the concepts used for the phosphate tailing management. The publications were sourced from keyword research on databases and publisher websites "Google Scholar, Scopus, Springer link, Elsevier Sciences Direct …" Keywords such as valorization, management, recycling, utilizing, characterization with phosphate slime, phosphate sludge, phosphate washing waste, phosphate tailing, phosphate mine tailing and phosphatic clay were used in the literature research. The review also included other publications based on "phosphate processing, phosphate production, phosphate generation and utilization.
Phosphate mining
The phosphate ore is mined by different countries in the world processed into an estimated 23 million tons of marketable product in 2020. According to industry analysts from different countries, which are reported as a pioneer (Krekeler et al. 2008; Cánovas et al. 2018) for instance, USA, Brazil, Morocco and Tunisia, the mine production capacity was approximately 240 million tons in 2019 and is expected to highly increase (Survey 2020). The phosphate production has known an increasing evolution with time, as shown in Fig. 1. The phosphate production has been increased from 1994 to 2020 by ~ 87%, from 128 to 240 million tons. The marketable product refers to beneficiated phosphate rock with phosphorus pentoxide (P2O5) content suitable for phosphoric acid or elemental phosphorus production (El Zrelli et al. 2018). The beneficiation techniques of phosphate ores generally depend on the type of associated gangue minerals present in the mined phosphate rock (Abouzeid 2008).
Production data for large mines only from 1993 to 2020 based on USGS.gov | Science for a changing world. https://www.usgs.gov/. Accessed 7 Dec 2021
Mining process
Mining is the first operation in the commercial exploitation of a mineral resource. It is defined as the extraction of material from the ground in order to recover one or more component parts of the mined material. The remaining mineral gangue is generally rejected as a waste (Evdokimov and Evdokimov 2015; Alcalde et al. 2018).
The mining industries produce phosphate rock with 28–31% of P2O5 content. The phosphate ore is generally separated from the minerals of granges in different steps using several processes. After mining, the phosphate ore underwent a screening process on-site to separate the phosphate rock from the bulky mine wastes (on-site sand waste, Fig. 2). Then, the beneficiation process consisting of a washing treatment to remove the unwanted minerals and concentrate the ore (Evdokimov and Evdokimov 2015; Falagán et al. 2017). During this process, the phosphate rock is separated from the gangue by size classification and chemical separation, first by sieves during the washing were the primary phosphate mine slimes are generated. Then, by hydro- cyclones to separate phosphate particles that goes directly to the stock of concentrate phosphate from the fine particles that underwent a flotation process and the very fine part that constitutes the first part of the phosphate sludge. During the flotation step, the phosphate grains are separated from the gangue minerals such as silicates, carbonates and clays, which constitute the second part of phosphate sludge. The two parts of phosphate sludge are deposited together or separately in tailing pounds. Overall, during the mining and beneficiation processes of phosphate ore, 4 different types of wastes are generated: sand waste, primary phosphate mine slimes, phosphate tailing and secondary phosphate mine slimes Fig. 2.
This process generates a huge volume of waste named tailing, which is discharged in tailing ponds or discarded to rivers and oceans. The inefficient management of this mine waste presents severe environmental and economic and environmental problems. This material is classified as industrial mining waste, which causes different problems.
To date, several studies have investigated the phosphate mine wastes using different terminologies such as phosphate sub-group (Perumal et al. 2020, 2021b), phosphate slimes (Moukannaa et al. 2019; Dabbebi et al. 2020; Ramanathan et al. 2021), phosphate sludge (Tabrika et al. 2020) and phosphate tailing (Solismaa et al. 2018; Wang et al. 2019; Perumal et al. 2021a).
Characteristics of phosphate mine waste
Different studies have characterized and investigated the properties of the Phosphate mine tailing (PMW), mainly the chemical and mineralogical properties and physical properties as particle size distribution.
Chemical composition
The chemical composition of PMW depends on the process used in the P2O5 production and on the origin of the deposits. Based on different studies summarized in Table.1, the main elements present in phosphate wastes are SiO2, CaO, P2O5 and Al2O3. Different other elements are also reported to be present in the PWS as minor elements such as MgO, Na2O, K2O, Fe2O3 and SO3.
According to Graedel et al. (2015), phosphate wastes can be divided into 2 categories Fig. 2. The First type, called phosphate mine slime PMS, characterized by its high clay fraction and very fine particle size. The percentage of P2O5 in this waste is a low grade (5–20%), making the Recovery of phosphate from it very difficult because of the high content of clay minerals. While the second category is named phosphate tailing (PT), marked by the low percentage of P2O5 < 2%, but it presents a much higher percentage of Mg and Ca. Mainly, those two wastes are mixed in ponds.
Mineralogical composition
The mineralogical characteristics of PMW can be determined using X-Ray diffraction and scanning microscopy with energy dispersive spectroscopy (EDS). The mineralogical composition of phosphate wastes is variable depending on the stratigraphy of the deposit and the kinds of minerals of gangue as well as the kind of the generated waste. Numerous studies have reported the mineralogy of phosphate wastes that mainly consists of 4 mineralogical groups: fluorapatite minerals, aluminosilicates which are mostly clays and layered clay, and zeolites in some cases carbonate minerals, quartz, and other minor phases such as gypsum and pyrite Table 2.
Waste management strategies
The mining industry that is one of the essential fields contributing to the economic development of countries, is knowing a continuous evolution worldwide, which leads to the amplification of the quantities of wastes and by-products generated by this activity. Consequently, Enormous environmental and economic issues related to the disposal of these wastes are engendered. Hence, the study and development of efficient management methods of mine wastes in general, among them phosphate mine wastes, have attracted the attention of researchers. The most investigated management methods for phosphate washing wastes are reprocessing, reuse, and recycling.
Dewatering
During the phosphate beneficiation processes of washing and flotation, large quantities of water are used and discharged with the phosphate washing wastes in the ponds. Hence, the Recovery of the used water from the slime ponds is necessary. In fact, different recovery or dewatering methods have been investigated. The recovered water is generally recycled to the mining, mineral processing or other operations, treated further or discharged to the environment.
In the area of washing which is one of the most important industrial unit operations, the water is recovered from phosphate waste differently. The physical separation methods like centrifugation with chemical treatments (using flocculant and coagulants or by the electrokinetic method) are the most used dewatering methods.
Research on dewatering has been mainly carried out on Florida phosphate slime. Since 1971, many researchers have studied the dewatering of phosphate waste using different methods as presented in Table 3. The enhanced settling and densification method was used by Liu and Stern (1972). Polyacrylamide flocculation agent was used with other additives like "quartz or Gypsum" with and without agitation. The results of the experiments performed using phosphate slime show that the different densification conditions such as the agitation speed, the number of flocculation stages, flocculation additives and the type of the porous filter media have a huge impact on the settling percentage. Overall, different processes for the separation of solids from an aqueous liquid suspension were developed and tested on phosphate washing wastes. Also, in this investigation, the authors stated that this process is applicable for the treatment of any suspension containing suspended solid matter. Ettoumi et al. (2020) investigated water Recovery using different anionic and cationic flocculent and optimized the flocculant concentration, the agitation speed and the settling time. The results showed that fast settling (81.3%) after just 15 s could be archived using one of the anionic investigated flocculent. The water recovery has been improved with the tested anionic flocculants to reach 79.4–84.1% compared to 58.1% found using the slim flocculent floc used previously by the company. Yap (1999) also used X polymer as a flocculating agent to facilitate solid slime formation. They then separated the flocculated slime from water employing flotation. Caswell and Puddington (1980) used a different method consisting of agglomerating the solid adding a hydrophobic organic bridging liquid and conditioner liquid then agitating to form a dispersion of solid particles agglomerates.
Rahman (2000) studied the dewatering of the Egyptian phosphate clay waste using the flocculation methods by polyacrylamides as commercial and starches as naturel flocculant. In this study, the settling rate of phosphate waste was increased 37 times with commercial flocculant compared to natural settling. Also, Ibrahim and Abdel-Khalek (1992) studied the use of different natural corn starches as flocculants and the effect of multiple parameters (pH, temperature, pulp density) on the settling of the phosphate slime. The work shows that the type of the starches and the different parameters affect the settling, e.g., the range of temperature from 60 to 75 °C improve the settling rate. The use of other additives as fly ash for the dewatering of the clay slime was investigated by Raba Jr et al. (1985) and Mewes et al. (1976) using silane as coupling reagent to which is attached a polyacrylamide flocculent.
Palmer and Gaynor (1984) stated in his patent that the dewatering of phosphate slime can be achieved by the incorporation of hydratable calcium sulphate from the waste of phosphogypsum in the suspension. For the invention of Nair and Somasundaran (1986), the dewatering of phosphate slime was obtained with the addition of different chemicals and different combinations were used as (CaSO4.1/2 H2O + CaO), (CaSO4.1/2 H2O + Phosphogypsum + CaO), (CaSO4.1/2 H2O + Phosphogypsum) and (CaSO4.1/2 H2O), and Sulfuric acid was used in this combination to adjust pH.
Smelley et al. (1981) investigated the dewatering of the slime by using the sufficient amount of flocculating agent to form flocs and then, the mechanical manipulation (rotating cylindrical screen or vibrating screen) and the filtration, respectively, to recover the used water during the phosphate beneficiation. Silva et al. (2015) also explored the addition of flocculant to the slime, and then, different vacuums filtration were used for dewatering the waste.
Moeglich (1981) indicated that is possible to dewater the sludge by electrophoresis with minimum energy use in which the sludge is maintained between the electrodes. The invention also shows that the dewatered sludge can be used as fertilizer, since this method provides a water sufficiently free of heavy metal. Shang and Lo (1997) explored the dewatering of the phosphate slime by the electrokinetic technique indicating that the success of the treatment needs the study of different parameters as materials properties, and electrode configuration. Dizon and Orazem (2019) also used the electrokinetic dewatering of the phosphate clay waste. In this investigation, the authors used a single-stage and two-stage prototypes. The results demonstrate that the single-stage design approach presents better results dry clay waste production from an economic point of view.
Other authors used other simple processes as mixing phosphate tailing and phosphate slime to recover the water as reported by Cook and Haynsworth (1973). In contrast, (Gitchel and Kerr 1982) used the wet oxidation at 150–320 °C which make the solid rapidly settable.
Tao et al. (2010) investigated the Deep cone thickener in a pilot scale demonstration on the recovery of water. The authors studied the effect of the adding orders of the anionic and cationic flocculant on the recovery of the water from the phosphate clay waste. Other parameters where also studied like effect of the addition of sand, solid retention time and flocculant dosage. The used method where effective to recover 90%.
Heavy metal and phosphate recovery
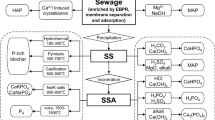
The presence of high amount of phosphorous in the tailings (Table 1) gives a motivation for the secondary recovery of this high value material. In previous studies, this was tried in several ways such as, acid leaching (Hazen et al. 1969), calcination (Ribas and Nickerson 1976), autoclave acidulation (Jha et al. 1985) (Zhang and Bogan 1995) and floatation (Abdel-Khalek et al. 2000b; Zhang et al. 2001; Alsafasfeh and Alagha 2017). Other than this, phosphate waste was also used as adsorbents to recover PO4 (Jellali et al. 2010, 2011; Jaradat et al. 2016). However, it is also clarified from the studies that secondary recovery of phosphate is a complicated process stating the physical and mineralogical characteristics of the tailing’s material. On the other hand, phosphate tailings are rich source of heavy metals and have potential for recovering the rare earth metals (Chen and Graedel 2015). Data from several studies have shown that heavy metals elements "Cd, Cr, Mn, Mo, Ni, Pb, U, V and Zn" and phosphorus are mined in phosphate slimes (Zhang et al. 2017). Studies shows that Zn and Ni can be successfully extracted with solvent method (Powell and Smith 1972), whereas floatation method seems to be efficient for maximum recovery of rare earth metals (Al-Thyabat and Zhang 2016).
Hazen et al. (1969) leached the slime with sulfuric acid and then, recovered P2O5 from the acid leachate with a mixture of amine and alcohol in a solvent extraction step. Ribas and Nickerson (1976) used the method of consolidation, calcination in the temperature range of 871–1093 °C, then digested the slime in mineral acid to produce phosphoric acid. However, the process was energy intensive. Jha et al. (1985) used the autoclave acidulation technic to recover the phosphate from the waste. At 175 °C, more than 90% of P2O5 was extracted. After acid treatment, the clay fraction consolidates and a longer leaching time led to the formation of phosphogypsum crystals. The problem faced in this process is the equipment investment and poor acid quality. Zhang and Bogan (1995) studied the standard acidulation process technology and the sizing using hydrocyclone, making the phosphate waste easy to manipulate. However, the selection of the flocculation, sizing followed by flotation, also bioleaching seems to be the best solutions on an excellent phosphate recovering. Abdel-Khalek et al. (2000b) investigate the phosphate slime of Sebya Egypt to recover the phosphate by column flotation method also using by changing parameters as the Superficial Air Velocity, froth concentration, column height and Superficial Wash water Velocity conditions (Zhang et al. 2001).mentioned that the most efficient method to recover phosphate from waste clay was cycloning flowed by flotation. The cycloning was made to reduce the fine and the unwanted components. Negm (2008) presented the production of different materials based on Egyptian phosphate waste. The phosphate recovering was investigated by studying different parameters such as the effects of liquid/solid ratio, type of froths and grinding. Jellali et al. (2010, 2011) used the phosphate slime as an adsorbent for phosphorus removal from aqueous solution. The performance of this adsorption process increases with increasing adsorbent dosage and temperature and decrease with increasing of the pH values. In addition, Jaradat et al. (2016) investigated the phosphate mine waste of Jordan to remove and recover PO4. The authors claimed that phosphate waste has a high potential sorption capacity for PO4 than other materials like palm fibers, wood particles and zeolites. The possibility of upgrading the P2O5 content in tailing using a direct froth flotation process with the investigation of different parameters: solid contents, pH and flotation time to optimize froth recovery and grade was investigated by Alsafasfeh and Alagha (2017). It could be concluded from most studies that it is technically impossible or economically restrictive because of the mineralogical and physical characteristics of slime, which cause difficulty in recovering the phosphate. The slime is characterized by 30%-50% of clay minerals and ultrafine particle size.
Earlier research has stated that phosphate waste can be a resource for heavy metal recovery. Powell and Smith (1972) studied the phosphate slime to recover the zinc and nickel by solvent extraction. According to a particular procedure, repeated extraction showed that three to five extraction was enough to extract the total of Zn and Ni. Rashed et al. (2015) explored the phosphate slime as cost effective adsorbent for heavy metal recovering from polluted water. Range of parameters was studied: pH, adsorbent dosage, contact time and initial metal concentration. The results show that the phosphate sludge can be used successfully for the removal of heavy metal. Marzougui et al. (2016) studied the sludges from 4 different Tunisian laundries to investigate the behavior and the mobility of trace metals. The finding shows that the trace metals were weakly mobilized, the pH and biological process affect some processes. So, it is mandatory to understand some reactions to control the release of trace elements. Al-Thyabat and Zhang (2016) mentioned that the shaking table or froth flotation can be used to concentrate the extraction of rare earth elements. The results show that the shaking table allows just 20% recovering of REE while froth flotation presents 63.5% of Recovery.
Ceramics application
Numerous studies indicate that clay minerals account for 50–80% of the total phosphatic slime mass. Based on this composition, this material can be suitable to produce ceramic materials. However, the low content in aluminum and silica requires the adequate amendment of this waste to be used as a prime material for ceramics production. Different studies attempted to recycle phosphate mine wastes to produce ceramic pellets, ceramic bricks, filtration and ultrafiltration membranes. (Raba Jr et al. 1985) claimed that the beneficiation of phosphate ore generates different types of waste. In this patent, the authors used different formulations of ceramic products based on different types of phosphate waste: slime and slag. Using just one type of waste to produce ceramic is not possible, and the mix of two or more will serve to produce such materials. (Khemakhem et al. 2011) investigated the phosphate slime to produce low-cost membrane support and a composite ultra-filtration membrane with Zirconium. The results indicate that this membrane presents a good performance similar to those obtained using commercial ultrafiltration alumina membrane. The authors Khemakhem et al. (2015) investigated a membrane based on the phosphate slime to be used for the treatment of industrial textile wastewater, giving acceptable results. Loutou et al. (2016a) used the phosphate waste as a raw material mix with different percentages of clay to produce ceramic products. The mechanical and physical properties were studied, and the showed results were correlated with the firing cycle and the added clay. The authors (Loutou et al. 2017) studied the mix of clay and phosphate waste heated in the range of 900–1200 °C in the form of pellets and investigated the effect of processing factors by full factorial design. The finding in this work shows that the present phases contributed to the properties of the materials like the conductivity. The optimum physical properties of ceramic were obtained by T° = 1152 °C, soaking time 4 h and clay addition 5wt%. Yang et al. (2017) investigated the preparation of ceramics by the utilization of phosphate waste, soft interlayer and black shale as additive using the single factors experiment with which the authors studied the impact of different parameters: preheating temperature, preheating time, sintering temperature and sintering time. Amar et al. (2018) investigated a new asymmetric ultrafiltration membrane based on phosphate waste by deposition of only one layer using a slip casting process for the treatment of wastewater from the textile industry. The results showed that the parameters of this type of membrane present an increase in performance compared to the commercial alumina ultrafiltration membrane. Muliawan and Astutiningsih (2018) studied the attempt of utilization of phosphate waste mixed with kaolin for ceramic bricks. The mixtures were prepared with different compositions by the variation of the percentage of phosphate sludge. The results displayed that the best compressive strength was obtained with the specimens that present an addition of 25–50% of phosphate waste fired at 1200 °C. Harech et al. (2020) studied the thermal behavior of the phosphate sludge after addition of different amount of clay, showing the mineralogical changes due to the interaction between the phosphate waste and clay during the synthesis of ceramics products.
Building materials
The heterogeneous nature and variating composition of phosphate mine tailings represent a challenge to its recycling as a prime raw material for construction materials (Duxson et al. 2007). Although, different investigations proved its possible utilization in lightweight and conventional concrete, asphalt, lightweight aggregates, bricks and alkali activated materials manufacturing. In most studies, the reactivity of the phosphate mine waste represents a challenge toward it recycling especially in alkali activated materials. Hence, several pretreatments has been suggested to improve the aluminosilicate reactivity of such uncalcined wastes including, mechanical (Tchadjie and Ekolu 2018), chemical (Xu et al. 2010) and thermal (Bondar et al. 2011)activation methods. The thermal and thermo-chemical (alkaline fusion) treatments seem to be the most suitable methods for uncalcined wastes such as phosphate mine tailings (Gharzouni et al. 2018; Moukannaa et al. 2018; Tchakouté et al. 2015).
Vasan and Corporation (1971) studied the feasibility of producing material-based phosphate slimes waste of Florida industry. The work showed that the phosphate waste was feasible for the production of pelletized lightweight concrete. Katamine (2000) explored the deformation behavior of asphalt concrete mixed with slime and other 3 phosphate by-products. Compared to the standard mixture containing limestone filler, the mixture presents higher stability and flow values. However, the mixture with phosphate slime presents poor behavior due to their susceptibility to water, making it not recommended for use in wearing courses. Boyd et al. (2017) investigated the use of phosphate waste as an admixture to cement paste, mortar and concrete and studied the chemistry, workability and mechanical properties. However, the problem faced by this work was the difficulty to improve the mechanical properties such as strength and ductility simultaneously. El-Shall (2007) used the phosphate slime to develop lightweight aggregates. A process was used to dewater the slime and obtain 50% of solids with flocculent and fibers. The mix was extruded and then fired at 900 °C in a kiln. As mentioned in the previous part, (Negm 2008) used Egyptian phosphate waste in different fields, such as brick based on 90% of phosphate slime and 10% of shale, pottery making formed with the same composition of bricks and coarse aggregates for the concrete mix. Loutou et al. (2013) investigated the treated Moroccan phosphate sludge mixed with swelling clay up to 30 wt% as lightweight aggregate. The results showed that the addition of clay enhances the formation of gehlenite phase. The temperature was the parameter that controlled the characteristic of the material. The fluorapatite phases has also an effect on the microstructure and the density of the obtained material. Zheng et al. (2015) studied the effect of phosphate tailing used as filler in Portland cement. The outcomes show that the phosphate slime affects the properties of cement Portland like quartz. Furthermore, it affects also different properties like the setting time, while no significant influence on the strength was observed. Loutou et al. (2016b) investigated the phosphate waste with a varied percentage (0–40%) of raw clay and cement kiln dust to form lightweight aggregate. The results display that the temperature, the soaking time had little effect, and the addition of phosphate waste to clay had a small negative effect. Chen et al. (2017) used a combination of phosphate waste as aggregate with slag and/or Portland cement as a binder and CaO as an additive. The results showed that the phosphate waste could not be considered an ideal backfill material by itself due to its low strength and the amount of CO2 and SO2 gas generation. The optimum backfill mixture has a composition of phosphogypsum/phosphate waste 1:3, cement 4 and 2 wt% of CaO. Dizon and Orazem (2019) studied the effect of phosphate slime on the characteristic of alkali activated aluminosilicates to formulate cement mortars. The findings indicate that phosphate sludge as an admixture in the cement improves the properties like compressive strength and shrinkage. Moukannaa et al. (2018) recycled the phosphate sludge as a partial replacement of fly ash and metakaolin to produce geopolymers in a portion of 50%. The authors studied the effect of different factors (activator concentration, curing temperature and curing time) on the physical properties. The most influent factor in this work was the curing temperature, and the best condition of manufacturing was activator concentration 12.5 M, temperature of curing 83.33 °C and 14.5 as days of curing. The investigation of the geopolymer mortars based on phosphate waste was studied also by Moukannaa et al. (2019). The sludge was mixed with 10 and 20% of NaOH and fused at 550 and 800 °C. The results show that the mortars obtained at 550 °C and with 10% of NaOH present the higher compressive strength. Also, Wu et al. (2020) studied the thermal alkaline activation process with the addition of NaOH to the phosphate slime and treat the mix at 600 °C for 2 h with the addition of slag 0–40%. The reactions show that the presence of N–A–S–H and C–A–S–H in the mortars was enhanced by the increase in the percentage of slag in the mixture. Perumal et al. (2019) used the waste after thermal treatment at 750 °C and 900 °C to improve its reactivity as a binder for alkali activated materials. The results show that the alkali activation was successful without heat treatment also, while the calcination at 750 °C improved the compressive strength of the final products. Dabbebi et al. (2018a) investigated the effects of the calcination temperature and the mortar mix design on the microstructure and mechanical properties of alkali activated phosphate washing waste. The results revealed the presence of zeolites and geopolymeric gel in the final cured products. Also, the mechanical properties results (11 MPa at 28 days) proved the feasibility of using calcined washing phosphate waste as binder for construction application such as alkali activated bricks (Dabbebi et al. 2018a). Furthermore, Dabbebi et al. (2020) studied the reactivity and reaction products of the washing phosphate waste calcined at 2 different temperatures (600 and 700 °C). They proved the occurrence of 2 types of reactions during the alkali activation leading to the formation of 2 different gels, the N–A–S–H/(C,N)–A–S–H gel and the C–S–H, C–(A)–S–H gel. In addition, the participation of different original phases such as Heulandite, palygorskite and phlogopite to the alkali activation reactions was confirmed (Dabbebi et al. 2020).
Hamdane et al. (2020) studied the effect of different modulus of NaOH and KOH as activators for phosphate slime and metakaolin precursors to synthesize geopolymer. The modulus of the NaOH and KOH highly affects the formed phases and, therefore, the materials' properties. Haddaji et al. (2021) used the phosphate mine tailing as a binder with the metakaolin for geopolymer composite production to investigate two types of synthetic fibers "Polypropylene and glass fiber." The results show that adding the fiber to the mixture enhances the flexure strength of the obtained materials.
The difference obtained in some works using the phosphate washing waste as the raw materials for the same application "dewatering, phosphate and heavy metal recovery, ceramic and building materials" can be first explained by the difference of the composition of this waste from a country and continent to another as presented in Table 1.
The phosphate mining waste is an industrial waste that can be classified as a by-product and used in different applications. Despite that, number of studies and results show that the composition and the mineralogy should be studied, to understand the effect of the present elements in the waste on the targeted applications. The following step for the researchers is to investigate the adequate management path for the large scale and focus also on the environmental impact of these works.
Conclusion
We commonly experience the benefits of minerals, starting from toothpaste to vehicles and buildings, however, before being presented to us as the final products. They underwent different treatments in a long mining process. Starting from the extraction to the beneficiation and transformation processes, large quantities of wastes are generated. The deposition of the mine wastes results in the occupation of large land areas, generating serious storage capacity problems and resulting in severe environmental issues if they are not chemically stable.
The phosphate industry is one of the active mining activities generating large quantities of wastes (Yang et al. 2017). The phosphate recovery chain can be divided into three processes. Starting with the extraction process where the phosphate rock is separated from the associated minerals of guange and sent to the beneficiation plant. In this second process, the phosphate rock is concentrated using physical and chemical separation methods. Then, the concentrate phosphate goes to the transformation plants to produce phosphoric acid and different kinds of fertilizers.
During the different steps of this process, several kinds of mine wastes are generated, among which is the phosphate slime/sludge or phosphate washing waste generated during the beneficiation process. Most of the time, they are deposited on the surface (Dabbebi et al. 2018a). Hence, finding a management solution for these mine wastes was the ultimate motivation of numerous investigations.
The present review summarized the most relevant studies on phosphate washing wastes management, reprocessing, recycling and valorization. One of the first strategic management actions taken for phosphate slime is reprocessing the large quantities used in the beneficiation process. The Recovery of water for further utilization in the processing operations needs the implementation of efficient dewatering processes. Different dewatering methods has been investigated including densification by flocculation (Ettoumi et al. 2020), a combination of flocculation and filtration or flotation, electrokinetic (Shang and Lo 1997; Dizon and Orazem 2019) including electrophoresis, dielectrophoresis and electro-osmosis, dewatering and wet oxidation at temperatures ranging from 150 to 320 °C. The flocculation method has been proven to be efficient and presented a high settling achieving 81.3% at short time 15 s when anionic flocculants are used Ettoumi et al. (2020). Also, polyacrylamide flocculent has been studied and proved to be a good flocculation agent increasing the settling 37 times compared to natural settling (Liu and Stern 1972). The electrokinetic dewatering could be an efficient technique; however, it is not commonly used in practice due to the adverse electrode reactions, energy consumption, and the different parameters to control such as electrode configurations and the current density. The use of efficient method of water recovery, can be a good way to reuse the water and recycle the solid waste of the phosphate mine.
Phosphate recovery could be performed by leaching in sulfuric acid (Hazen et al. 1969), consolidating or calcinating the slime at high temperatures 870–1093 °C combined with digestion in mineral acid (Ribas and Nickerson 1976), these methods seem to be energy consuming while producing low-quality acid. The physical separation by mean of cyclones followed by flotation was reported to be the most suitable for efficient phosphate recovery. Nonetheless, the clay lay characteristics of phosphate slime and its fine particle size represent a barrier to PO4 recovery, and further investigations are needed to develop efficient recovery methods.
Furthermore, recycling and utilization of phosphate washing waste as raw material for the preparation of ceramics or building materials could be considered as a valuable management method because of the thermal, chemical and mechanical properties of the obtained materials. The high amount of clay minerals in phosphate slime makes it a good candidate as ceramic raw material for membrane adsorbent (Loutou et al. 2019) or bricks manufacturing. In both cases, the optimization of phosphate slime addition rate, preheating temperature and time and firing cycle and high calcination temperatures are required.
In addition, broad investigations focused on recycling phosphate slime in building materials, to produce pelletized lightweight aggregate (Loutou et al. 2013), as an additive in cement paste (Zheng et al. 2015), for bricks manufacturing, for backfill materials (Chen et al. 2017), and as raw material for geopolymer synthesis (Dabbebi et al. 2018b; Moukannaa et al. 2019). In most of the applications, the dosage of phosphate addition as well as the preparation conditions such as the curing temperature and time, the calcination temperature for bricks and other parameters should be optimized depending on the chemical and mineralogical composition of the phosphate slime. The heterogeneity and variability of this waste depending on the phosphate deposit represent a challenge for its continuous recycling. in addition, the reutilization of phosphate slime for geopolymers manufacturing is a promising research pathway; however, an adequate pretreatment is required to enhance the reactivity of the phosphate slime and increase its potential incorporation rate in geopolymer manufacturing.
Overall, the valorization of phosphate washing waste has attracted the attention of many researchers who have made significant progresses in developing efficient recycling methods; however, none of the developed methods has been used in practice and in large scale so far due to the lack of durability, long term performance and reproducibility of the studies on the developed materials and sometimes the economic and practical viability of the methods. Scientists need to investigate the applications with the very well consideration of the starting materials, efficient process and reusable methods.
Availability of data and materials
Not applicable.
References
Abdel-Khalek NA, Hassan F, Arafa MA (2000a) Separation of valuable fine phosphate particles from their slimes by column flotation. Sep Sci Technol 35:1077–1086. https://doi.org/10.1081/SS-100100212
Abouzeid A-ZM (2008) Physical and thermal treatment of phosphate ores—an overview. Int J Miner Process 85:59–84. https://doi.org/10.1016/j.minpro.2007.09.001
Alcalde J, Kelm U, Vergara D (2018) Historical assessment of metal recovery potential from old mine tailings: a study case for porphyry copper tailings, Chile. Miner Eng 127:334–338. https://doi.org/10.1016/j.mineng.2018.04.022
Alsafasfeh A, Alagha L (2017) Recovery of phosphate minerals from plant tailings using direct froth flotation. Minerals 7:145. https://doi.org/10.3390/min7080145
Al-Slaty FM (2018) Index properties of alkali-activated cement mortar affected by the addition of phosphatic clay. Jordan J Earth Environ Sci 9:63–66
Al-Thyabat S, Zhang P (2016) Extraction of rare earth elements from upgraded phosphate flotation tailings. M&MP 33:23–30. https://doi.org/10.19150/mmp.6464
Amar BEN, R, Oun A, Cerneaux S et al (2018) Decolorization of dyeing effluent by novel ultrafiltration ceramic membrane from low cost natural material. J Membr Sci Res 4:101–107
Andersen B, Somasundaran P (1993) Mechanisms determining separation of phosphatic clay waste by selective flocculation. Min, Metall Explor 10:200–205
Ayouch I, Barrak I, Kassab Z et al (2020) Improved recovery of cadmium from aqueous medium by alginate composite beads filled by bentonite and phosphate washing sludge. Colloids Surf: Physicochem Eng Asp 604:125305. https://doi.org/10.1016/j.colsurfa.2020.125305
Bondar D, Lynsdale CJ, Milestone NB et al (2011) Effect of heat treatment on reactivity-strength of alkali-activated natural pozzolans. Constr Build Mater 25:4065–4071. https://doi.org/10.1016/j.conbuildmat.2011.04.044
Bowker LN, Chambers DM (2017) In the dark shadow of the supercycle tailings failure risk & public liability reach all time highs. Environ—MDPI 4:1–21. https://doi.org/10.3390/environments4040075
Boyd AJ, Birgisson B, Beatty C, Zaman A (2017) Utilization of phosphatic clay waste in concrete 56
Cánovas CR, Macías F, Pérez-López R et al (2018) Valorization of wastes from the fertilizer industry: current status and future trends. J Clean Prod 174:678–690
Caswell BF, Puddington IE (1980) Dewatering of slimes from phosphate ores, U.S. Patent No. 4,229,293. Washington, DC: U.S. Patent and Trademark Office
Chen M, Graedel TE (2015) The potential for mining trace elements from phosphate rock. J Clean Prod 91:337–346. https://doi.org/10.1016/j.jclepro.2014.12.042
Chen Q, Zhang Q, Fourie A, Xin C (2017) Utilization of phosphogypsum and phosphate tailings for cemented paste backfill. J Environ Manage 201:19–27. https://doi.org/10.1016/j.jenvman.2017.06.027
Cook C, Haynsworth E (1973) PROCESS OF REMOVING WATER FROM SLIMES U.S. Patent No. 3,763,041. Washington, DC: U.S. Patent and Trademark Office
Dabbebi R, de Aguiar JB, Camões A, Samet B, Baklouti S (2018a) Effect of the calcinations temperatures of phosphate washing waste on the structural and mechanical properties of geopolymeric mortar. Constr Build Mater 185:489-498. https://doi.org/10.1016/j.conbuildmat.2018.07.045
Dabbebi R, Baklouti S, Barroso de Aguiar JL et al (2018b) Investigations of geopolymeric mixtures based on phosphate washing waste. Sci Technol Mater 30:1–5. https://doi.org/10.1016/j.stmat.2018.08.001
Dabbebi R, de Aguiar JLB, Samet B, Baklouti S (2019) Mineralogical and chemical investigation of Tunisian phosphate washing waste during calcination. J Therm Anal Calorim 137:1827–1840. https://doi.org/10.1007/s10973-019-08057-3
Dabbebi R, Barroso de Aguiar JL, Baklouti S (2020) Spectroscopic and microscopic study of alkali activated mortars based on Tunisian phosphate washing waste. Cem Concrete Compos 105:103449. https://doi.org/10.1016/j.cemconcomp.2019.103449
Dizon A, Orazem ME (2019) Efficient continuous electrokinetic dewatering of phosphatic clay suspensions. Electrochim Acta 298:134–141. https://doi.org/10.1016/j.electacta.2018.12.003
Duxson P, Fernández-Jiménez A, Provis JL et al (2007) Geopolymer technology: the current state of the art. J Mater Sci 42:2917–2933. https://doi.org/10.1007/s10853-006-0637-z
Edraki M, Baumgartl T, Manlapig E et al (2014) Designing mine tailings for better environmental, social and economic outcomes: a review of alternative approaches. J Clean Prod 84:411–420. https://doi.org/10.1016/j.jclepro.2014.04.079
El Zrelli R, Rabaoui L, Daghbouj N et al (2018) Characterization of phosphate rock and phosphogypsum from Gabes phosphate fertilizer factories (SE Tunisia): high mining potential and implications for environmental protection. Environ Sci Pollut Res 25:14690–14702. https://doi.org/10.1007/s11356-018-1648-4
El-Shall H (2007) Commercial products from phosphatic clays: a pre-feasibility study 55
Eskanlou A, Huang Q (2021) Phosphatic waste clay: origin, composition, physicochemical properties, challenges, values and possible remedies—a review. Miner Eng 162:106745. https://doi.org/10.1016/j.mineng.2020.106745
Ettoumi M, Jouini M, Neculita CM, Bouhlel S, Coudert L, Haouech I, Benzaazoua M (2020) Characterization of Kef Shfeir phosphate sludge (Gafsa, Tunisia) and optimization of its dewatering. J Environ Manage 254:109801. https://doi.org/10.1016/j.jenvman.2019.109801
Evdokimov SI, Evdokimov VS (2015) Metal recovery from old tailings. J Min Sci 50:800–808. https://doi.org/10.1134/S1062739114040206
Falagán C, Grail BM, Johnson DB (2017) New approaches for extracting and recovering metals from mine tailings. Miner Eng 106:71–78. https://doi.org/10.1016/j.mineng.2016.10.008
Gharzouni A, Ouamara L, Sobrados I, Rossignol S (2018) Alkali-activated materials from different aluminosilicate sources: effect of aluminum and calcium availability. J Non-Cryst Solids 484:14–25. https://doi.org/10.1016/j.jnoncrysol.2018.01.014
Gitchel WB, Kerr RK (1982) Treatment of clay slimes U.S. Patent No. 4,312,761. Washington, DC: U.S. Patent and Trademark Office
Graedel TE, Harper EM, Nassar NT et al (2015) Criticality of metals and metalloids. Proc Natl Acad Sci USA 112:4257–4262. https://doi.org/10.1073/pnas.1500415112
Haddaji Y, Majdoubi H, Mansouri S et al (2021) Effect of synthetic fibers on the properties of geopolymers based on non-heat treated phosphate mine tailing. Mater Chem Phys 260:124147. https://doi.org/10.1016/j.matchemphys.2020.124147
Haddaway NR, Cooke SJ, Lesser P et al (2019) Evidence of the impacts of metal mining and the effectiveness of mining mitigation measures on social-ecological systems in Arctic and boreal regions: a systematic map protocol. Environ Evid 8:1–11. https://doi.org/10.1186/s13750-019-0152-8
Hamdane H, Tamraoui Y, Mansouri S et al (2020) Effect of alkali-mixed content and thermally untreated phosphate sludge dosages on some properties of metakaolin based geopolymer material. Mater Chem Phys 248:122938. https://doi.org/10.1016/j.matchemphys.2020.122938
Harech M, Mesnaoui M, Abouliatim Y, et al (2020) Effect of temperature and clay addition on the thermal behavior of phosphate sludge. Española de Cerámica y Vidrio 60(3):194-204. https://doi.org/10.1016/j.bsecv.2020.03.002
Hazen WC, Henrickson AV, Hadzeriga P (1969) Recovery of phosphate values from phosphatic slimes U.S. Patent No. 3,425,799. Washington, DC: U.S. Patent and Trademark Office
Ibrahim SS, Abdel-Khalek NA (1992) The action of different types of corn starch on the flocculation of phosphate slimes 10
Jaradat DMM, Ghrair AM, Alhesan JSA et al (2015) An efficient sorbent for phosphate removal from wastewater: a new application of phosphate mine wastes from Ruseifa City—Jordan. Desalin Water Treat 57:9914–9924. https://doi.org/10.1080/19443994.2015.1033472
Jaradat DMM, Ghrair AM, Alhesan JSA et al (2016) An efficient sorbent for phosphate removal from wastewater: a new application of phosphate mine wastes from Ruseifa City—Jordan. Desalin Water Treat 57:9914–9924. https://doi.org/10.1080/19443994.2015.1033472
Jellali S, Wahab MA, Anane M et al (2010) Phosphate mine wastes reuse for phosphorus removal from aqueous solutions under dynamic conditions. J Hazard Mater 184:226–233. https://doi.org/10.1016/j.jhazmat.2010.08.026
Jellali S, Wahab MA, Hassine RB et al (2011) Adsorption characteristics of phosphorus from aqueous solutions onto phosphate mine wastes. Chem Eng J 169:157–165. https://doi.org/10.1016/j.cej.2011.02.076
Jha MC, Lussiez GW, Beckstead LW (1985) Autoclave Acidulation of Phosphate Slimes. AMAX Extractive Research & Development
Jowitt SM, Mudd GM, Thompson JFH (2020) Future availability of non-renewable metal resources and the influence of environmental, social, and governance conflicts on metal production. Commun Earth Environ 1:1–8. https://doi.org/10.1038/s43247-020-0011-0
Katamine NM (2000) Phosphate waste in mixtures to improve their deformation. J Transp Eng 126:382–389. https://doi.org/10.1061/(ASCE)0733-947X(2000)126:5(382)
Khemakhem M, Khemakhem S, Ayedi S, Amar RB (2011) Study of ceramic ultrafiltration membrane support based on phosphate industry subproduct: application for the cuttlefish conditioning effluents treatment. Ceram Int 37:3617–3625. https://doi.org/10.1016/j.ceramint.2011.06.020
Khemakhem M, Khemakhem S, Ayedi S et al (2015) Development of an asymmetric ultrafiltration membrane based on phosphates industry sub-products. Ceram Int 41:10343–10348. https://doi.org/10.1016/j.ceramint.2015.05.101
Kinnunen P, Ismailov A, Solismaa S et al (2018) Recycling mine tailings in chemically bonded ceramics—a review. J Clean Prod 174:634–649. https://doi.org/10.1016/j.jclepro.2017.10.280
Kiventerä J, Perumal P, Yliniemi J, Illikainen M (2020) Mine tailings as a raw material in alkali-activation—a review. Int J Miner Metall Mater 27:1009. https://doi.org/10.1007/s12613-020-2129-6
Krekeler MPS, Morton J, Lepp J et al (2008) Mineralogical and geochemical investigation of clay-rich mine tailings from a closed phosphate mine, Bartow Florida, USA. Environ Geol 55:123–147. https://doi.org/10.1007/s00254-007-0971-8
Liu T, Stern DR (1972) Process for the treatment of slimes and waste solids U.S. Patent No. 3,680,698. Washington, DC: U.S. Patent and Trademark Office
Liu X, Zhang Y, Liu T et al (2017) Characterization and separation studies of a fine sedimentary phosphate ore slime. Minerals 7:94. https://doi.org/10.3390/min7060094
Loutou M, Hajjaji M, Mansori M et al (2013) Phosphate sludge: Thermal transformation and use as lightweight aggregate material. J Environ Manage 130:354–360. https://doi.org/10.1016/j.jenvman.2013.09.004
Loutou M, Hajjaji M, Mansori M et al (2016a) Heated blends of clay and phosphate sludge: microstructure and physical properties. J Asian Ceram Soc 4:11–18. https://doi.org/10.1016/j.jascer.2015.10.003
Loutou M, Hajjaji M, Mansori M et al (2016b) Heated blends of phosphate waste: microstructure characterization, effects of processing factors and use as a phosphorus source for alfalfa growth. J Environ Manage 177:169–176. https://doi.org/10.1016/j.jenvman.2016.04.030
Loutou M, Hajjaji M, Babram MA et al (2017) Phosphate sludge-based ceramics: microstructure and effects of processing factors. J Build Eng 11:48–55. https://doi.org/10.1016/j.jobe.2017.04.002
Loutou M, Misrar W, Koudad M et al (2019) phosphate mine tailing recycling in membrane filter manufacturing: microstructure and filtration suitability. Minerals 9:318. https://doi.org/10.3390/min9050318
Lyu Z, Chai J, Xu Z, et al (2019) A comprehensive review on reasons for tailings dam failures based on case history. Adv Civil Eng. https://doi.org/10.1155/2019/4159306
Marzougui S, Sdiri A, Rekhiss F (2016) Heavy metals’ mobility from phosphate washing effluents discharged in the Gafsa area (southwestern Tunisia). Arab J Geosci 9:599. https://doi.org/10.1007/s12517-016-2613-5
Mewes AR, Styron RW, Smith MH (1976) Process for the treatment of mineral slimes
Moeglich K (1981) SLIME AND SLUDGE DEWATERING, U.S. Patent No. 4,244,804. Washington, DC: U.S. Patent and Trademark Office
Morone M, Costa G, Polettini A et al (2014) Valorization of steel slag by a combined carbonation and granulation treatment. Miner Eng 59:82–90. https://doi.org/10.1016/j.mineng.2013.08.009
Moukannaa S, Loutou M, Benzaazoua M et al (2018) Recycling of phosphate mine tailings for the production of geopolymers. J Clean Prod 185:891–903. https://doi.org/10.1016/j.jclepro.2018.03.094
Moukannaa S, Nazari A, Bagheri A et al (2019) Alkaline fused phosphate mine tailings for geopolymer mortar synthesis: thermal stability, mechanical and microstructural properties. J Non-Cryst Solids 511:76–85. https://doi.org/10.1016/j.jnoncrysol.2018.12.031
Muliawan J, Astutiningsih S (2018) Preparation and characterization of phosphate-sludge kaolin mixture for ceramics bricks. Int J Technol 9:317. https://doi.org/10.14716/ijtech.v9i2.1119
Nair KV, Somasundaran P (1986) Method for dewatering phosphate slimes
Negm AA (2008) Utilization of solid wastes from phosphate processing plants
Niu H, Abdulkareem M, Sreenivasan H et al (2020a) Recycling mica and carbonate-rich mine tailings in alkali-activated composites: a synergy with metakaolin. Miner Eng 157:106535. https://doi.org/10.1016/j.mineng.2020.106535
Niu H, Kinnunen P, Sreenivasan H et al (2020b) Structural collapse in phlogopite mica-rich mine tailings induced by mechanochemical treatment and implications to alkali activation potential. Miner Eng 151:106331. https://doi.org/10.1016/j.mineng.2020.106331
Palmer JW, Gaynor JC (1984) Method for solidifying waste slime suspensions
Perumal P, Piekkari K, Sreenivasan H, Kinnunen P (2019) One-part geopolymers from mining residues—Effect of thermal treatment on three different tailings. Miner Eng 144:106026. https://doi.org/10.1016/j.mineng.2019.106026
Perumal P, Niu H, Kiventerä J et al (2020) Upcycling of mechanically treated silicate mine tailings as alkali activated binders. Miner Eng 158:106587. https://doi.org/10.1016/j.mineng.2020.106587
Perumal P, Hasnain A, Luukkonen T et al (2021a) Role of surfactants on the synthesis of impure kaolin-based alkali-activated, low-temperature porous ceramics. Open Ceram 6:100097. https://doi.org/10.1016/j.oceram.2021.100097
Perumal P, Kiventerä J, Illikainen M (2021b) Influence of alkali source on properties of alkali activated silicate tailings. Mater Chem Phys. https://doi.org/10.1016/j.matchemphys.2021.124932
Powell HE, Smith LL (1972) Recovery of zinc and nickel from waste phosphate liquor
Raba Jr CF, Smith RL, Huang FY (1984) Composition and process for the treatment of clay slimes
Raba Jr CF, Smith RL, Huang FY (1985) Process for the treatment of clay slimes
Rahman MKA-E (2000) Dewatering of phosphatic clay waste by flocculation. Chem Eng Technol 23:457–461. https://doi.org/10.1002/(SICI)1521-4125(200005)23:5%3c457::AID-CEAT457%3e3.0.CO;2-P
Ramanathan S, Perumal P, Illikainen M, Suraneni P (2021) Mechanically activated mine tailings for use as supplementary cementitious materials. RILEM Tech Lett 6:61–69. https://doi.org/10.21809/rilemtechlett.2021.143
Rashed MN, Mohamed AR, Awadallah MA (2015) Chemically activated phosphate slime as adsorbent for heavy metals removal from polluted water
Ribas RS, Nickerson JD (1976) Process for the manufacture of phosphoric acid with minimized environmental effects
Shang JQ, Lo KY (1997) Electrokinetic dewatering of a phosphate clay. J Hazard Mater 55:117–133. https://doi.org/10.1016/S0304-3894(97)00018-6
Silva AC, Silva EMS, da Silva Junior ÂP et al (2015) Mineral paste production from phosphate rock tailings. Rem: Rev Escola Minas 68:103–108. https://doi.org/10.1590/0370-44672015680103
Smelley AG, Montgomery RW, Hamner BJ (1981) Dewatering of slimes. U.S. Patent No. 4,303,532. Washington, DC: U.S. Patent and Trademark Office
Solismaa S, Ismailov A, Karhu M et al (2018) Valorization of finish mining tailings for use in the ceramics industry. Bull Geol Soc Finland 90:33–54. https://doi.org/10.17741/bgsf/90.1.002
Survey UG (2020) Mineral commodity summaries 2020. US Geological Survey Washington
Tabrika I, Mayad EH, Zaafrani M et al (2020) Optimization of solid phosphate sludge composting by integration of horticultural waste. Org Agr. https://doi.org/10.1007/s13165-020-00302-6
Tao D, Parekh BK, Zhao Y, Zhang P (2010) Pilot-scale demonstration of deep cone™ paste thickening process for phosphatic clay/sand disposal. Sep Sci Technol 45:1418–1425. https://doi.org/10.1080/01496391003652783
Tchadjie LN, Ekolu SO (2018) Enhancing the reactivity of aluminosilicate materials toward geopolymer synthesis. J Mater Sci 53:4709–4733. https://doi.org/10.1007/s10853-017-1907-7
Tchakouté HK, Kong S, Djobo JNY et al (2015) A comparative study of two methods to produce geopolymer composites from volcanic scoria and the role of structural water contained in the volcanic scoria on its reactivity. Ceram Int 41:12568–12577. https://doi.org/10.1016/j.ceramint.2015.06.073
The European Parliament and Council of the EU (2006) Directive 2006/21/EC of the European Parliament and of the Council of 15 March 2006 on the Management of Waste from Extractive Industries and Amending Directive 2004/35/EC
The U.S. Department of Agriculture (USDA) (1998) Drainage from abandoned mines in remote areas
USGS.gov|Science for a Changing World. https://www.usgs.gov/. Accessed 7 Dec 2021
Vasan S, Corporation IM (1971) Utilization of phosphate slimes. Environmental Protection Agency, Water Quality Office
Wang C, Harbottle D, Liu Q, Xu Z (2014) Current state of fine mineral tailings treatment: a critical review on theory and practice. Miner Eng 58:113–131. https://doi.org/10.1016/j.mineng.2014.01.018
Wang G, Ning, Lu X, X et al (2019) Effect of sintering temperature on mineral composition and heavy metals mobility in tailings bricks. Waste Manage 93:112–121. https://doi.org/10.1016/j.wasman.2019.04.001
Wu J, Li J, Rao F, Yin W (2020) Mechanical property and structural evolution of alkali-activated slag-phosphate mine tailings mortars. Chemosphere 251:126367. https://doi.org/10.1016/j.chemosphere.2020.126367
Xiaolong Z, Shiyu Z, Hui L, Yingliang Z (2021) Disposal of mine tailings via geopolymerization. J Clean Prod 284:124756. https://doi.org/10.1016/j.jclepro.2020.124756
Xu H, Li Q, Shen L et al (2010) Low-reactive circulating fluidized bed combustion (CFBC) fly ashes as source material for geopolymer synthesis. Waste Manage 30:57–62. https://doi.org/10.1016/j.wasman.2009.09.014
Yang Y, Wei Z, Chen Y et al (2017) Utilizing phosphate mine tailings to produce ceramisite. Constr Build Mater 155:1081–1090. https://doi.org/10.1016/j.conbuildmat.2017.08.070
Yap SN (1999) Phosphate beneficiation process using polymers as slime flocculants, U.S. Patent No. 5,858,214. Washington, DC: U.S. Patent and Trademark Office
Zhang P, Bogan M (1995) Recovery of phosphate from Florida beneficiation slimes I. Re-identifying the problem. Miner Eng 8:523–534. https://doi.org/10.1016/0892-6875(95)00014-H
Zhang P, Snow R, Yu Y, Bogan M (2001) Recovery of phosphate from Florida phosphatic clays. Final report, FIPR Publication 02-096
Zhang P, Liang H, Jin Z, DePaoli D (2017) The ultimate mineral processing challenge: recovery of rare earths, phosphorus and uranium from Florida phosphatic clay. Miner Metall Process 34:183–188. https://doi.org/10.19150/mmp.7858
Zheng K, Zhou J, Gbozee M (2015) Influences of phosphate tailings on hydration and properties of Portland cement. Constr Build Mater 98:593–601. https://doi.org/10.1016/j.conbuildmat.2015.08.115
Acknowledgements
The authors would like to kindly thank Prof. Mirja Illikainen and Dr. Patrick Zhang, for their helpful comments and advice.
Funding
Open Access funding provided by University of Oulu including Oulu University Hospital. The authors have no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Contributions
The first draft and data collection of the manuscript were written by Rawia Dabbebi. Samira Moukannaa and Priyadharshini Perumal commented and added introduction and summary. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors did not receive support from any organization for the submitted work, and they have no financial interests. The authors have no competing interests to declare that are relevant to the content of this article. We declare that this manuscript is original, has not been published before and is not currently being considered for publication elsewhere. We know of no conflicts of interest associated with this publication, and as corresponding author, I confirm that the manuscript has been read and approved for submission to the journal by the entire named author.
Ethical approval
The authors declare that this paper not considered for publication in any other journal. The submitted work is original and not have been published elsewhere in any form or language.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Editorial responsibility: Lifeng Yin.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dabbebi, R., Perumal, P. & Moukannaa, S. Management and valorization of phosphate beneficiation slime: a critical review. Int. J. Environ. Sci. Technol. 20, 11763–11776 (2023). https://doi.org/10.1007/s13762-023-04901-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-023-04901-0