Abstract
Introduction
This retrospective study investigated major adverse limb events (MALE) and mortality outcomes in critical limb-threatening ischemia (CLTI) patients with tissue loss after an endovascular revascularization-first (EVR-1st) strategy.
Methods
MALE and mortality were assessed in 157 consecutive patients with CLTI and tissue loss from June 2019 to June 2022 at the Eric Williams Medical Sciences Complex, Trinidad and Tobago.
Results
157 patients underwent the EVR-1st strategy, of whom 20 were pivoted to immediate surgical revascularization (SR). Of the remaining 137 patients, successful EVR was achieved in 112, giving a procedural success of 82% and an all-comer overall success of 71%. The mortality and MALE rates were 2.7% and 8.9% at 2 years, respectively. Males and patients with previous major amputations were at significantly higher risk for MALE (p values of 0.016 and 0.018, respectively). There was a statistically significant difference in successful EVR for both Rutherford–Baker (RB) 5 (minor) and RB 6 (major) classifications: 63 (56%) vs. 5 (20%) and 49 (44%) vs. 20 (80%), both with a p value of 0.01. There were no differences in successful EVR amongst Wound, Ischemia, Foot Infection (WIfI) clinical stages. There were no differences in successful EVR amongst the Trans-Atlantic Inter-Society Consensus (TASC II) classifications.
Conclusions
This study may prove clinically informative and applicable for an EVR-1st management strategy for high-risk patients with CLTI in a limited-resource, Caribbean setting.
Trial registration number: NCT05547022 (retrospectively registered).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Currently, there is a paucity of data that assess endovascular revascularization (EVR) outcomes in patients with critical limb-threatening ischemia (CLTI) within the Caribbean region. |
Successful EVR was achieved in 112 patients, giving a procedural success of 82%; however, there was an all-comer overall success of 71%, factoring in the initial patients routed to surgical revascularization (SR). |
There was a statistically significant difference in successful EVR vs. unsuccessful EVR for both Rutherford–Baker (RB) 5 (minor) and RB 6 (major) classifications: 63 (56%) vs. 5 (20%) and 49 (44%) vs. 20 (80%), both with a p value of 0.01. |
What was learned from this study? |
An endovascular revascularization-first (EVR-1st) strategy in high-risk patients with critical limb-threatening ischemia (CLTI) was a feasible management strategy in our limited-resource setting. |
Further large-scale, multicentric studies are required to confirm these exploratory findings. |
Introduction
Critical limb-threatening ischemia (CLTI) affects nearly 200 million persons globally [1,2,3]. The prevalence of PAD is higher in low-income countries, and a substantial disparity exists between low- and high-income countries, with the former displaying a markedly accentuated incidence.
CLTI is the most severe form of PAD. Furthermore, nearly half of the patients without intervention require a major amputation, and one-fifth have a significant mortality risk within 6 months [4,5,6]. Conventional risk factors such as diabetes, metabolic syndrome, and tobacco use are steadily increasing in Trinidad [7,8,9]. Additionally, it was recently reported that the loss of independent mobilization and lack of rehabilitation in Trinidad significantly impacts the quality of life for amputees [10, 11].
Surgical revascularization (SR) is considered more optimal in low-risk patients and is the gold standard, evidenced by improved durability, patency of grafts, and amputation-free survival [12, 13]. Since the advent of minimally invasive endovascular techniques, revascularization outcomes have been comparable with attenuated morbidity [14,15,16]. An endovascular revascularization-first (EVR-1st) strategy is now the treatment of choice for CLTI patients, as amputation-free survival is prolonged with a future surgical option despite unsuccessful EVR [17, 18]. Studies have demonstrated limb salvage rates approaching 90%, lending credence to EVR being the primary therapeutic modality for this subpopulation [13, 18].
This retrospective study investigated major adverse events (MALE) and mortality outcomes in CLTI patients with tissue loss after an EVR-1st strategy.
Methods
The study complied with the Declaration of Helsinki, International Conference on Harmonization, Good Clinical Practice, and was approved by the Campus Research Ethics Committee (CREC) of the University of the West Indies, St. Augustine (UWI STA), Trinidad (CEC175/04/17). The retrospective study reviewed consecutive patients diagnosed with CLTI for 3 years (June 2019–June 2022). These patients had peripheral lower-extremity angiography performed at the Eric Williams Medical Sciences Complex, Trinidad and Tobago, a monocentric, tertiary academic medical center. They were then stratified to either an EVR-1st strategy or SR based on their American Society of Anesthesiology (ASA) score, with higher-risk patients undergoing the transcatheter technique. Data were collected via case report forms by a postgraduate surgery resident and clinical research associate assessing demographic data, which included the patient’s medical and procedural history and cardiovascular medications. Peripheral angiograms were quality checked and assured by both vascular surgery consultants. Incentives were not offered for participating in the study. All data were secured in the Department of Clinical Surgical Sciences (DCSS) office at the UWI STA to maintain confidentiality. Computer data were accessible only to the research team, password protected, and database encrypted. During the data collection and analysis, no patient identifiers were included. Inclusion criteria comprised patients with Rutherford–Baker (RB) class 5–6 who underwent EVR [19]. Exclusion criteria included patients with RB class 0–4. Procedural success was defined as an angiographically acceptable result without mortality or MALE. Successful limb salvage was defined as patients who did not experience MALE post-EVR (see Fig. 1).
Statistical Analysis
The sample size was calculated as 128 patients, based on a quantitative study to find an association, an alpha (α) value of 0.05, a power of 80%, and an absolute delta (Δ) of 25% between surgical revascularization (SR) and EVR procedural success. Statistical analysis was performed using SPSS® 28.0.1 (IBM, New York City, New York, USA). The parameters and outcomes were investigated using descriptive statistics. Percentages represented categorical data and means with standard deviations (SDs) were used for continuous data. The comparisons between samples for categorical data used chi-squared tests; however, comparisons between groups for continuous data were made using Student’s t-test. Univariate analysis was performed to identify parameters associated with EVR. All p values were two-tailed; any value < 0.05 was considered statistically significant.
Results
Of the 157 patients, 20 failed the EVR-1st strategy and underwent surgical revascularization (SR) (Tables 1, 2). Successful EVR was achieved in 112 patients, giving a procedural success of 82%; however, there was an all-comer overall success of 71% when factoring in the initial patients routed to SR. There were 157 peripheral interventions in 157 patients, with 20 failed limb interventions in 20 patients. MALE occurred in 28 of 157 limbs (18%) due to both failed EVR (10) and eventual amputation despite successful EVR (18). Those with an immediate EVR failure and eventual failed EVR went on to have SR (34 of 45), resulting in 11 amputations. Of the 34 limbs revascularized with SR, 7 underwent amputations. Thus, there were 18 amputations in these 45 patients.
80% of the patients requiring SR had Trans-Atlantic Inter-Society Consensus (TASC II) classification D lesions, 70% had Rutherford-Baker (RB) class 6 tissue loss, and 75% had Wound, Ischemia, Foot Infection (WIfI) clinical stage 4 wounds. A total of 137 patients were treated with EVR; however, 112 and 25 patients underwent successful and unsuccessful revascularization, respectively. Occlusion or failed recanalization accounted for 9% (15 of 157 cases) of the technical failures. Diabetes, hypertension, dyslipidemia, and tobacco use prevalence ranged from 56 to 82%, 92 to 94%, 31 to 34%, and 32 to 45%, respectively. Aspirin, clopidogrel or ticagrelor, and statin use ranged from 92 to 96%, 68 to 72%, and 72 to 79%, respectively. Males and patients with previous major amputations were at significantly higher risk for unsuccessful EVR (p values 0.016 and 0.018, respectively). Patients with complete occlusions which were not successfully treated transitioned to SR (Table 3).
Of the 137 patients, 68 had minor (RB 5) and 69 had major (RB 6) tissue loss. There was a statistically significant difference in successful EVR vs. unsuccessful EVR for both RB 5 (minor) and RB 6 (major) classifications: 63 (56%) vs. 5 (20%) and 49 (44%) vs. 20 (80%), both with a p value of 0.01. However, in the unsuccessful EVR group, 80% had major tissue loss (RB 6), 64% had WIfI clinical stage 4, and 36% had a TASC II D classification. There were no differences in successful EVR amongst the WIfI groups. There were no differences in successful EVR amongst the TASC II classifications (Table 4).
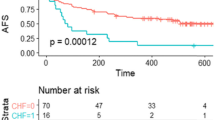
The overall mortality (3 deaths) and MALE rate (10 amputations) were 2.7% and 8.9% at 2 years, respectively. The three patient deaths were not procedure-related and were attributed to acute coronary syndromes (ACS), cardiogenic shock (CS), and heart failure (HF) (Table 5).
Discussion
Patients with CLTI are at an elevated risk of MALE and major adverse cardiovascular events (MACE). The 2016 American Heart Association/American College of Cardiology guideline on managing patients with lower extremity PAD specifies that the key objectives in managing CLI are to (1) minimize tissue loss, (2) completely heal wounds, and (3) preserve a functional foot [20]. With the intrinsically less invasive EVR and its attendant signal for decreased morbidity and lower morbidity, this modality has now supplanted vascular surgery as the preferred initial revascularization strategy [21,22,23,24]. Lin et al. reported that EVR-1st management in CLTI patients is associated with improved amputation-free survival [25]. The SPINACH (Surgical Reconstruction Versus Peripheral Intervention in Patients With Critical Limb Ischemia) study also found that 3-year amputation-free survival and other outcomes were equivocal when comparing surgical reconstruction against EVR in this subpopulation [26]. The BEST-CLI trial demonstrated that SR with a great saphenous vein (GSV) conduit was superior to EVR in reducing MALE and mortality; however, outcomes were similar if no GSV was available [12].
Our study displayed several interesting results. The patients initially referred to SR (20) had complex PAD (long segment occlusions) with poor inflow and were triaged by the attending vascular surgeon. The patients with unsuccessful EVR (25) who were then referred to SR had procedural failure, with the most cited reason being a failure to cross the lesion. Patients were primarily stratified using the Rutherford–Baker (RB) classification, which describes the most severe forms as stage 5 (RB 5—minor) and stage 6 (RB 6—major) [19]. Traditionally, SR was the initial treatment of choice for patients with tissue loss [27]. There was a statistically significant difference between successful EVR and unsuccessful EVR for both RB 5 (minor) and RB 6 (major) classifications: 63 (56%) vs. 5 (20%) and 49 (44%) vs. 20 (80%), both with p values of 0.01. However, this trend was not seen when a comparative analysis was performed with the WIfI clinical stage or TASC II classification, which is likely due to the study being inadequately powered to assess these relationships, although this was not pre-specified. The Society for Vascular Surgery (SVS) staging system, known as the Wound, Ischemia, and Foot Infection (WIfI) classification system, stratifies patients’ disease severity and need for revascularization of the threatened limb [18, 28]. Darling et al. assessed the predictive capability of the WIfI grading for infra-popliteal EVR outcomes such as the 1-year amputation rate and amputation-free survival [23, 29]. Although there was a relatively high proportion of patients with WIfI grade 3 and 4 disease in our study, there was no statistically significant association between EVR and WIfI grade (43% and 48% in the successful EVR group vs. 36% and 64% in the unsuccessful EVR group). TASC II divides the anatomic distribution of lesions into aortoiliac and femoral-popliteal, and lesion patterns are grouped into A–D lesions [30, 31]. Males and patients with previous major amputations were at significantly higher risk for unsuccessful EVR (p values of 0.016 and 0.018, respectively). Previous studies have recognized these risk factors as negative prognosticators for procedural success [13, 26]. Our study demonstrated a relatively high preponderance of advanced PAD with respect to the RB, WIfI, and TASC II staging—for example, multivessel, small-caliber disease, diffuse, calcified lesions, and chronic total occlusions—which may have accounted for the unsuccessful EVR in these subgroups [32]. Smaller-caliber vessels afflicted with severe PAD have a negative impact on long-term patency [33]. Almost all patients underwent stenting instead of “plain old balloon angioplasty,” as the former displayed superior immediate outcomes [34]. Although Romiti et al. conducted a meta-analysis comparing infragenicular angioplasty versus bypass surgery, which established that SR resulted in greater longevity, EVR still displayed comparable outcomes with respect to limb salvage [35]. In our study, technical failures of EVR included a failure to cross lesions in addition to perforations. This was attributed to our minimal chronic total occlusion (CTO) armamentarium, and hence the “one-balloon, one-wire technique” was primarily adopted in our resource-poor setting [36, 37]. Endovascular techniques included native vessel recanalization, retrograde access, subintimal arterial flossing with antegrade and retrograde intervention (SAFARI), and the controlled antegrade and retrograde technique (CART). However, re-entry, atherectomy, and intravascular lithotripsy devices are not available in our setting and thus were not utilized.
Trinidad and Tobago’s economy is predominantly petrochemical based, with an estimated gross domestic product per capita of 15,000 dollars (USD) [38]. The population is approximately 1.5 million persons and is considered multi-ethnic. Trinidad has experienced both socioeconomic and cultural changes influenced mainly by Western-developed societies [7,8,9]. In the Caribbean, EVR is in its nascency, and the most common method used to treat CLTI is SR, which is traditionally associated with greater longevity; however, this is now being challenged by an evolving paradigm [17]. In the SR group, 25% of patients were classified as American Society of Anesthesiology (ASA) class 4. As operating theater time and personnel are at a premium, alternative and innovative strategies to pivot these high-risk cases to the cardiac catheterization laboratory became a real-time necessity [39]. Given their high-risk status, an EVR-1st strategy was conceptualized as an attempt to attenuate peri-operative MACE and downstream resource utilization, such as cardiac care and intensive care units [40].
Successful EVR was achieved in 112 patients, giving a procedural success of 82%. The overall mortality (3 deaths) and MALE rate (10 amputations) were 2.7% and 8.9% at 2 years, respectively. The limb salvage rate in this study was similar to that in a recent study by Verwer et al. which evaluated outcomes 5 years post-EVR [41, 42]. Elbadawi et al. assessed procedural volume in relation to EVR for CLTI in the United States, and although it did not positively impact mortality, there were improvements in several other parameters, such as short-term limb salvage [43]. Despite the overall lack of mortality benefit and MALE amongst a spectrum of US volumes, EVR remains a practical alternative in low- to moderate-volume centers. Other metrics should be heavily weighed, such as decreased in-hospital major amputation, minor amputation, and time to discharge to the nursing facility [44]. Future, long studies analyzing post-EVR outcomes across a continuum of volumes in CLTI patients are imperative. It is also pivotal to institute a multidisciplinary team approach to optimize management strategies in these patients [45].
The recently concluded BEST-CLI trial demonstrated that SR with a great saphenous vein (GSV) conduit was superior to EVR in reducing MALE and mortality; however, outcomes were similar if no GSV was available [12]. BASIL, a previous revascularization strategy trial, also demonstrated nearly equivalent outcomes [13]. As a result, there has been much consternation with respect to which approach remains superior from both a limb salvage and a morbidity/mortality perspective. Given the aforementioned issues for pre-operative GSV imaging and with respect to proceeding to immediate SR in a limited-resource setting, an EVR-1st strategy may prove to be a viable alternative.
Study Limitations
This study has several limitations. While a pilot study should provide insightful information about whether a full-scale study is feasible and specific enhancements, there is a caveat that this small pilot study is monocentric and situated in Trinidad. Although it reflects clinically pertinent findings, it may not be generalizable or applicable to other subpopulations. As previously stated, this is a single-center study, although that center is the only tertiary referral academic medical center for patients presenting with CLTI in the entire country. Although this study was adequately powered for clinical outcomes, less than 200 CLTI patients were enrolled. Trinidad could be considered a low-volume, resource-limited setting; however, this series represents the largest EVR CLTI study in the English-speaking Caribbean.
Observer, reporting, and misclassification bias and inter-observer variability could potentially have impacted the accurate interpretation of peripheral angiography despite the supervision and adjudication of two board-certified endovascular surgeons as a risk mitigation measure; for example, a TASC II C lesion could have been reported and analyzed as a TASC II D lesion. As there are no electronic medical records, data collection and its respective entry by the study team (a postgraduate surgery resident and clinical research associate) could have been compromised when they were fatigued, although the data were cleaned and quality checked by an experienced biostatistician.
Despite the relatively high prevalence of T2DM in this study (77% overall) compared with other local studies (bordering 50%), no adjusted analyses were performed to assess the effects of this endocrinopathy on EVR success. As aforementioned, T2DM is implicated in negative remodeling and may result in smaller-caliber vasculature and allude to heterogeneous results [46]. This study also assessed baseline cardiovascular medications—such as antiplatelets, anticoagulants, vasodilators, high-intensity statins, oral hypoglycemics, and insulin—which may attenuate both MACE and MALE by modulating critical pathways of inflammation, vascular dysfunction, and metabolic derangements [47,48,49]. Novel diabetes and lipid-lowering agents such as sodium-glucose co-transporter 2 inhibitors (SGLT2i) and proprotein convertase subtilisin-kexin type 9 inhibitors (PCSK9i) were not available in Trinidad during this study. While this study shared similarities with respect to the demographic data and comorbidities of cardiovascular studies performed in Trinidad, such as CAD and T2D, there is the added concern of suboptimal cardiovascular literacy and adherence to medical therapies, which may contribute to these CLTI patients who are presenting with complex PAD [7,8,9, 50]. Additionally, it has been demonstrated that Trinidadian South Asian patients have accentuated platelet reactivity and a smaller caliber of coronary arteries, which may contribute to a higher incidence of CLTI [7]. The team also did not record peripheral artery segment dimensions. The overwhelming majority of PAD CLTI studies are predominantly based on Caucasian populations, which may not be applicable if ethnic-related disparities occur with respect to the prevalence of comorbidities and vascular dimensions [51]. Other procedural aspects, such as the duration of the procedure, radiation exposure metrics, and contrast media volume, were not recorded.
While these results may be potentially informative, they may not be externally valid when applied to patients with other limb-threatening conditions, such as acute limb ischemia (ALI). The information gleaned from this study may prove valuable in ensuring a reliable supply chain with respect to consumables, such as drug-coated balloons for patients with CLTI in terms of quantity and caliber. With the onset of the recent coronavirus-19 (COVID-19) pandemic and its attendant sequelae for the hospital supply chain, the continuous availability of consumables such as catheters, guidewires, and balloons was inconsistent. Thus, patients with CLTI requiring EVR ultimately had to be deferred or routed to SR. In Trinidad and Tobago’s public healthcare sector, there are several technical, logistical, and personnel challenges in delivering a clinically safe and effective service for patients presenting with CLTI. For example, the Cardiac Catheterization Laboratory (CCL) is only available during weekdays and conventional business hours as there are insufficient specialists, nursing staff, cardiovascular technicians, radiographers, and ancillary staff to operate a “full-time, on-call” interventional suite. This CCL also has to be shared with the Cardiology Service, where a vast majority of cardiovascular patients require percutaneous coronary interventions (PCI). There is often a risk-stratification discussion between interventional cardiologists and endovascular surgeons regarding case priority. As such, the median time to EVR was approximately 3 days and is likely accountable for any increased MALE. Early revascularization is necessary as patients with tissue loss have a very high risk of limb loss [22]. As the adage states, “time is tissue” is imperative in achieving successful EVR to attenuate MALE [52]. Currently, the vascular surgery team comprises two board-certified surgeons capable of performing EVR; however, they lack the necessary adjunctive support to achieve optimal outcomes. Ideally, there should be a consensus amongst a multidisciplinary team consisting of an interventional cardiologist, interventional radiologist, vascular surgeon, and vascular medicine specialist to delineate an appropriate treatment strategy for these CLTI patients; however, these specialist roles are often unavailable for various reasons [25].
General surgeons perform most of the SR cases if the two EVR-trained vascular surgeons are unavailable. These are usually done in tertiary hospitals inadequately equipped for this vascular niche, with a high likelihood of limb amputation due to the added complexity of suboptimal primary care of multiple major comorbidities, such as T2DM, along with frequently late and severe presentations with evidence of tissue loss [11]. Generally, an EVR procedure costs at least 20,000 USD, as compared to SR, which is circa 50,000 USD. No economic benefit analyses were performed, as this study’s primary aim was to establish baseline EVR PAD data for Trinidad and, to a broader extent, the Caribbean basin. Despite constrained resources, it also illustrates that procedural success rates and clinical outcomes are nearly comparable in international regions, although our results were expected to be worse due to the aforementioned patient and logistic factors. Another critical factor when conducting this study was innovating a strategy to alleviate operating room time and personnel, “buying time” to streamline already depleted resources. The overarching rationale was that most patients with RB 6 disease would be stratified as high risk for SR; thus, revascularizing multiple angiosomes for complex PAD would attenuate peri-operative MACE. Although baseline health-related quality of life was poor in BEST-CLI patients, there was substantial improvement among patients undergoing EVR compared to SR [12]. Our study did not assess the quality-of-life-related outcomes, which would have also impacted our strategy.
Learning lessons and future directions for managing CLTI in Trinidad include implementing a “limb team” involving all the pertinent specialties, akin to the heart team [53], as well as developing an endovascular fellowship training program to increase the output of capable proceduralists to cope with the disease burden and prevent physician and nursing personnel burnout. Electronic medical record utilization may improve the efficiency of database and registry building. Finally, a dedicated endovascular suite with an inventory control system may prove valuable in the ongoing battle against limb loss, as this would mitigate against untimely days in an EVR-1st strategy. The study would have estimated the CLTI patient volume for 3 years, 2019–2022, and thus future planning to mitigate supply chain disruptions could be completed. Building a registry to capture long-term interval limb surveillance in both EVR-1st and SR groups is also imperative in maintaining high-fidelity outcomes and quality control and assurance with respect to clinical audits.
Conclusion
This study may prove clinically informative and applicable for an endovascular revascularization-first strategy for high-risk patients with CLTI in a limited-resource setting. Further large-scale, multicentric studies are required to confirm these exploratory findings.
References
Criqui MH, Matsushita K, Aboyans V, Hess CN, Hicks CW, Kwan TW, et al. Lower extremity peripheral artery disease: contemporary epidemiology, management gaps, and future directions: a scientific statement from the American Heart Association. Circulation. 2021. https://doi.org/10.1161/cir.0000000000001005.
Dieter RS, Dieter RA Jr, Dieter RA III, Nanjundappa A. Critical limb ischemia: acute and chronic. Berlin: Springer; 2016.
Criqui MH, Aboyans V. Epidemiology of peripheral artery disease. Circ Res. 2015;116:1509–26.
Criqui MH, Ninomiya JK, Wingard DL, Ji M, Fronek A. Progression of peripheral arterial disease predicts cardiovascular disease morbidity and mortality. J Am Coll Cardiol. 2008;52:1736–42.
Ploeg AJ, Lardenoye J-W, VranckenPeeters M-PFM, Breslau PJ. Contemporary series of morbidity and mortality after lower limb amputation. Eur J Vasc Endovasc Surg. 2005;29:633–7.
Falluji N, Mukherjee D. Critical and acute limb ischemia: an overview. Angiology. 2014;65:137–46.
Seecheran NA, Maharaj A, Boodhai B, Seecheran R, Seecheran V, Persad S, et al. Prevalence of clopidogrel “resistance” in a selected population of patients undergoing elective percutaneous coronary intervention at a tertiary cardiovascular centre in Trinidad: the POINT pilot study. Open Heart. 2019;6: e000841.
Seecheran N, Baldeo S, Balbosa K, Baksh S, Bethelmy S, Balkaran S, et al. Epidemiologic insight into Trinidad’s awareness and prevention of heart disease. The EPITAPH study—a Trinidadian cardiovascular health literacy survey. Int J Commun Med Public Health. 2017;4(4):903. https://doi.org/10.18203/2394-6040.ijcmph20171305.
Seecheran N, Nandlal N, Nankissoon S, Nancoo C, Nelson C, Nkemakolam A, et al. A Trinidadian cardiovascular medication adherence survey: the ADHERE TNT study. Int J Commun Med Public Health. 2017;4(10):3601. https://doi.org/10.18203/2394-6040.ijcmph20174218.
Pran L, Baijoo S, Harnanan D, Slim H, Maharaj R, Naraynsingh V. Quality of life experienced by major lower extremity amputees. Cureus. 2021;13: e17440.
Hambleton IR, Jonnalagadda R, Davis CR, Fraser HS, Chaturvedi N, Hennis AJ. All-cause mortality after diabetes-related amputation in Barbados: a prospective case–control study. Diabetes Care. 2009;32:306–7.
Farber A, Menard MT, Conte MS, Kaufman JA, Powell RJ, Choudhry NK, et al. Surgery or endovascular therapy for chronic limb-threatening ischemia. N Engl J Med. 2022;387:2305–16.
Bradbury AW, Adam DJ, Bell J, Forbes JF, Fowkes FGR, Gillespie I, et al. Bypass versus angioplasty in severe ischaemia of the leg (BASIL) trial: analysis of amputation free and overall survival by treatment received. J Vasc Surg. 2010;51:18S-31S.
Wiseman JT, Fernandes-Taylor S, Saha S, Havlena J, Rathouz PJ, Smith MA, et al. Endovascular versus open revascularization for peripheral arterial disease. Ann Surg. 2017;265:424–30.
Pande RL, Perlstein TS, Beckman JA, Creager MA. Secondary prevention and mortality in peripheral artery disease: National Health and Nutrition Examination Study, 1999 to 2004. Circulation. 2011;124:17–23.
Fernandez N, McEnaney R, Marone LK, Rhee RY, Leers S, Makaroun M, et al. Predictors of failure and success of tibial interventions for critical limb ischemia. J Vasc Surg. 2010;52:834–42.
DeRubertis BG, Faries PL, McKinsey JF, Chaer RA, Pierce M, Karwowski J, et al. Shifting paradigms in the treatment of lower extremity vascular disease. Ann Surg. 2007;246(3):415–24. https://doi.org/10.1097/sla.0b013e31814699a2.
Romiti M, Albers M, Brochado-Neto FC, Durazzo AES, Pereira CAB, De Luccia N. Meta-analysis of infrapopliteal angioplasty for chronic critical limb ischemia. J Vasc Surg. 2008;47(5):975-981.e1. https://doi.org/10.1016/j.jvs.2008.01.005.
Rutherford RB, Dennis Baker J, Ernst C, Wayne Johnston K, Porter JM, Ahn S, et al. Recommended standards for reports dealing with lower extremity ischemia: revised version. J Vasc Surg. 1997;26(3):517–38. https://doi.org/10.1016/s0741-5214(97)70045-4.
Gerhard-Herman MD, Gornik HL, Barrett C, Barshes NR, Corriere MA, Drachman DE, et al. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines. J Am Coll Cardiol. 2017;69:1465–508.
Almasri J, Adusumalli J, Asi N, Lakis S, Alsawas M, Prokop LJ, et al. A systematic review and meta-analysis of revascularization outcomes of infrainguinal chronic limb-threatening ischemia. J Vasc Surg. 2019;69(6):126S-136S. https://doi.org/10.1016/j.jvs.2018.01.071.
Dabrh AMA, Steffen MW, Asi N, Undavalli C, Wang Z, Elamin MB, et al. Bypass surgery versus endovascular interventions in severe or critical limb ischemia. J Vasc Surg. 2016;63(1):244-253.e11. https://doi.org/10.1016/j.jvs.2015.07.068.
Darling JD, McCallum JC, Soden PA, Korepta L, Guzman RJ, Wyers MC, et al. Results for primary bypass versus primary angioplasty/stent for lower extremity chronic limb-threatening ischemia. J Vasc Surg. 2017;66:466–75.
Biagioni RB, Brandão GD, Biagioni LC, Nasser F, Burihan MC, Ingrund JC. Endovascular treatment of TransAtlantic Inter-Society Consensus II D femoropopliteal lesions in patients with critical limb ischemia. J Vasc Surg. 2019;69:1510–8.
Lin JH, Brunson A, Romano PS, Mell MW, Humphries MD. Endovascular-first treatment is associated with improved amputation-free survival in patients with critical limb ischemia. Circ Cardiovasc Qual Outcomes. 2019;12: e005273.
Iida O, Takahara M, Soga Y, Kodama A, Terashi H, Azuma N, et al. Three-year outcomes of surgical versus endovascular revascularization for critical limb ischemia: the SPINACH study (Surgical Reconstruction Versus Peripheral Intervention in Patients With Critical Limb Ischemia). Circ Cardiovasc Interv. 2017. https://doi.org/10.1161/CIRCINTERVENTIONS.117.005531.
Uccioli L, Meloni M, Izzo V, Giurato L, Merolla S, Gandini R. Critical limb ischemia: current challenges and future prospects. Vasc Health Risk Manage. 2018;14:63–74. https://doi.org/10.2147/vhrm.s125065.
Ward R, Dunn J, Clavijo L, Shavelle D, Rowe V, Woo K. Outcomes of critical limb ischemia in an urban, safety net hospital population with high WIfI amputation scores. Ann Vasc Surg. 2017;38:84–9.
Beropoulis E, Stavroulakis K, Schwindt A, Stachmann A, Torsello G, Bisdas T. Validation of the wound, ischemia, foot Infection (WIfI) classification system in nondiabetic patients treated by endovascular means for critical limb ischemia. J Vasc Surg. 2016;64:95–103.
Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FGR. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg. 2007;45:5–67. https://doi.org/10.1016/j.jvs.2006.12.037.
Hardman RL, Jazaeri O, Yi J, Smith M, Gupta R. Overview of classification systems in peripheral artery disease. Semin Intervent Radiol. 2014;31:378–88.
Gamal WM, Askary ZM, Ibrahim M. Comparison of clinical outcomes of infragenicular angioplasty between diabetic and non-diabetic patients with peripheral arterial occlusive disease. Angiol Open Access. 2017. https://doi.org/10.4172/2329-9495.1000201.
Jude EB, Oyibo SO, Chalmers N, Boulton AJ. Peripheral arterial disease in diabetic and nondiabetic patients: a comparison of severity and outcome. Diabetes Care. 2001;24:1433–7.
Kasapis C, Henke PK, Chetcuti SJ, Koenig GC, Rectenwald JE, Krishnamurthy VN, et al. Routine stent implantation vs. percutaneous transluminal angioplasty in femoropopliteal artery disease: a meta-analysis of randomized controlled trials. Eur Heart J. 2009;30:44–55.
TASC Steering Committee, Jaff MR, White CJ, Hiatt WR, Fowkes GR, Dormandy J, Razavi M, et al. An update on methods for revascularization and expansion of the TASC lesion classification to include below-the-knee arteries: a supplement to the inter-society consensus for the management of peripheral arterial disease (TASC II). Catheter Cardiovasc Intervent. 2015;86(4):611–25. https://doi.org/10.1002/ccd.26122.
Manzi M, Palena LM. Treating calf and pedal vessel disease: the extremes of intervention. Semin Intervent Radiol. 2014;31:313–9.
Ferraresi R, Palena LM, Mauri G, Manzi M. Tips and tricks for a correct “endo approach.” J Cardiovasc Surg. 2013;54:685–711.
World Bank. GDP per capita (current US$)—Trinidad and Tobago. https://data.worldbank.org/indicator/NY.GDP.PCAP.CD?locations=TT. Cited 25 Oct 2022.
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status—historical perspectives and modern developments. Anaesthesia. 2019;74:373–9.
Hobbs SD, Yapanis M, Burns PJ, Wilmink AB, Bradbury AW, Adam DJ. Perioperative myocardial injury in patients undergoing surgery for critical limb ischaemia. Eur J Vasc Endovasc Surg. 2005;29:301–4.
Verwer MC, Wijnand JGJ, Teraa M, Verhaar MC, de Borst GJ. Long term survival and limb salvage in patients with non-revascularisable chronic limb threatening ischaemia. Eur J Vasc Endovasc Surg. 2021;62:225–32.
Zayed H, Halawa M, Maillardet L, Sidhu PS, Edmonds M, Rashid H. Improving limb salvage rate in diabetic patients with critical leg ischaemia using a multidisciplinary approach. Int J Clin Pract. 2009;63:855–8.
Elbadawi A, Elgendy IY, Rai D, Mahtta D, Megaly M, Pershad A, et al. Impact of hospital procedural volume on outcomes after endovascular revascularization for critical limb ischemia. JACC Cardiovasc Interv. 2021;14:1926–36.
Mukherjee D, Chatterjee S. Endovascular revascularization and outcomes in critical limb ischemia: does hospital volume matter? JACC Cardiovasc Intervent. 2021;14(17):1937–9.
Mukherjee D, Lingam P, Chetcuti S, Grossman PM, Moscucci M, Luciano AE, et al. Missed opportunities to treat atherosclerosis in patients undergoing peripheral vascular interventions: insights from the University of Michigan Peripheral Vascular Disease Quality Improvement Initiative (PVD-QI2). Circulation. 2002;106:1909–12.
Spinetti G, Kraenkel N, Emanueli C, Madeddu P. Diabetes and vessel wall remodelling: from mechanistic insights to regenerative therapies. Cardiovasc Res. 2008;78:265–73.
Bhagirath VC, Nash D, Wan D, Anand SS. Building your peripheral artery disease toolkit: medical management of peripheral artery disease in 2022. Can J Cardiol. 2022;38:634–44.
Bonaca MP, Hamburg NM, Creager MA. Contemporary medical management of peripheral artery disease. Circ Res. 2021;128:1868–84.
Foley TR, Waldo SW, Armstrong EJ. Medical therapy in peripheral artery disease and critical limb ischemia. Curr Treat Options Cardiovasc Med. 2016;18:42.
Seecheran N, Jagdeo C-L, Seecheran R, Seecheran V, Persad S, Peram L, et al. Screening for depressive symptoms in cardiovascular patients at a tertiary centre in Trinidad and Tobago: investigation of correlates in the SAD CAT study. BMC Psychiatry. 2020;20:498.
Welch T, Rampersad F, Motilal S, Seecheran NA. Comparison of cardiac CT angiography coronary artery dimensions and ethnicity in Trinidad: the CADET pilot study. Open Heart. 2022;9: e001922. https://doi.org/10.1136/openhrt-2021-001922.
Setacci C. Time is tissue. J Endovasc Ther. 2012;19:515–6.
Holmes DR, Rich JB, Zoghbi WA, Mack MJ. The heart team of cardiovascular care. J Am Coll Cardiol. 2013;19(4):903–7. https://doi.org/10.1016/j.jacc.2012.08.1034.
Acknowledgements
We would like to thank the patients for their participation in the study.
Funding
No funding or sponsorship was received for this study or the publication of this article. The Rapid Service Fee was funded by the authors.
Author Contributions
Dave Harnanan and Sangeeta Parbhu conceptualized the study. Dave Harnanan and Sangeeta Parbhu designed the study. Dave Harnanan, Sangeeta Parbhu, Lemuel Pran, and Ilecia Baboolal conducted the study. Patrick Harnarayan and Vijay Naraynsingh reviewed the study. Dave Harnanan, Sangeeta Parbhu, Lemuel Pran, and Naveen Seecheran wrote the manuscript. Patrick Harnarayan, Vijay Naraynsingh, and Naveen Seecheran revised the manuscript. All authors approved the final manuscript.
Disclosures
Dave Harnanan, Sangeeta Parbhu, Lemuel Pran, Ilecia Baboolal, Patrick Harnarayan, Vijay Naraynsingh, and Naveen Seecheran have nothing to disclose.
Compliance with Ethics Guidelines
All participants provided verbal consent to participate in the study. Full ethical approval was granted by the campus research ethics committee (CREC) of the University of the West Indies, St. Augustine (UWI STA) (CEC175/04/17). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Harnanan, D., Parbhu, S., Pran, L. et al. Endovascular Revascularization and Outcomes of Critical Limb-Threatening Ischemia in Trinidad and Tobago: The EVENT Pilot Study—Challenges in a Limited-Resource, Caribbean Setting. Cardiol Ther 12, 511–524 (2023). https://doi.org/10.1007/s40119-023-00322-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40119-023-00322-9