Abstract
Introduced in the late 1950s, polyenes represent the oldest family of antifungal drugs. The discovery of amphotericin B and its therapeutic uses is considered one of the most important scientific milestones of the twentieth century . Despite its toxic potential, it remains useful in the treatment of invasive fungal diseases owing to its broad spectrum of activity, low resistance rate, and excellent clinical and pharmacological action. The well-reported and defined toxicity of the conventional drug has meant that much attention has been paid to the development of new products that could minimize this effect. As a result, lipid-based formulations of amphotericin B have emerged and, even keeping the active principle in common, present distinct characteristics that may influence therapeutic results. This study presents an overview of the pharmacological properties of the different formulations for systemic use of amphotericin B available for the treatment of invasive fungal infections, highlighting the characteristics related to their chemical, pharmacokinetic structures, drug–target interactions, stability, and others, and points out the most relevant aspects for clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Amphotericin B (AMB) is still considered one of the most important antifungals of the last 60 years. |
We present an overview of the pharmacological properties of the different formulations for systemic use of AMB available for the treatment of invasive fungal infections. |
The study highlights its chemical characteristics, pharmacokinetic, structures, drug–target interactions, stability, bioequivalence, and others, and points out the most relevant aspects for clinical practice. |
The indications for the different formulations of AMB are based on the latest consensus and guidelines, and studies on their toxicity are based on the main clinical trials conducted in humans. |
A timeline presents the main scientific milestones for AMB over the decades. |
An updated list of the last 2 years of clinical trials that seek to improve the use of AMB in different situations is also provided. |
Digital Features
This article is published with digital features, including a summary slide, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13325681.
Introduction
Licensed in 1959 [1], amphotericin B (AMB) was initially designed for the treatment of local mycotic infections and later approved for the treatment of progressive and potentially fatal fungal infections [2]. After 60 years, it is still an important option in the treatment of fungal diseases.
Traditionally, the drug is administered as a formulation of deoxycholate amphotericin B (D-AMB) capable of forming micelles in aqueous solution [3]. Besides being a long-known medication, AMB has important side effects, such as nephrotoxicity, which have limited its indiscriminate use [4,5,6]. Most of the time, patients who need intervention with D-AMB are severely compromised because of their underlying diseases and comorbidities, and therefore, they end up becoming vulnerable to the reported toxic effects, especially when combined with other drugs.
To overcome this impasse, new systemic therapeutic options have been proposed: amphotericin B lipid complex (ABLC) and liposomal amphotericin B (L-AMB). There was a third lipid formulation known as AMB colloidal dispersion (ABCD), presented as uniform disk-shaped particles, that was discontinued in 2011 because of its high rate of infusion-related events and it is no longer manufactured [7, 8]. Although ABLC and L-AMB have the same active principle in common, their pharmacological characteristics distinguish them and may influence the final therapeutic results. Their chemical structures, pharmacokinetics, drug–target interactions, stability, bio and therapeutic equivalences share similarities but also present peculiarities, notably when compared to the conventional formulation.
This study presents an overview of the pharmacological and biopharmaceutical properties of the different systemic formulations of AMB available for the treatment of invasive fungal infections and highlights the most relevant aspects in 60 years for clinical practice. This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Why the Gold Standard?
Amphotericin B is a life-saving drug in the treatment of serious systemic fungal infections and is still the most widely used antifungal in and intensive care, despite the development of a series of new antifungal agents, especially the second-generation triazoles and the echinocandins.
These 60 years of clinical experience have proven that AMB is a reliable antifungal agent. At the time of its introduction and for decades, doctors had few other therapeutic options, and they learned and adapted to use it in order to minimize its toxicities.
The new therapeutic options also offer great prospects for treatment. Such options include improved azole antifungal agents, the echinocandin class, in addition to constant studies in search of new lipid formulations of AMB itself. However, it has been in recent years that these agents have proven their worth in a variety of clinical settings, providing high rates of effectiveness with minimal safety-related problems. Therefore, over time, it is natural that the use of conventional AMBs and even lipid formulations of amphotericin B (LFABs) may have limited use, as the evidence with the new agents, with new combination schemes, will show improvements in patient care and its benefits will be increasingly noticed.
Despite these advances, AMB remains in use both in medical practice and in clinical trials owing to the wide possibility of licensed indications. In addition, AMB remains the treatment of choice for many serious fungal infections in vulnerable hosts owing to its excellent spectrum of activity and its low resistance rates. To date, it continues to be the agent with the widest spectrum of action and the lowest resistance potential of any known antifungal agent [9].
Some characteristics that maintain its status as the gold standard are the low cost of conventional AMB therapy, the high acceptance of this formulation in continuous use by neonates, the improvement of toxicity rates with the arrival of the LFABs, and its intrathecal use in Coccidioides immitis meningitis [10, 11]. It is also noteworthy that there are individuals who actually tolerate conventional therapy better than advanced formulations [12]. Basically, these are the fundamentals that make the medical community consider the use of AMB as a therapeutic standard in addition to a standard comparator for clinical trials among antifungal agents.
With the new pharmaceutical forms and formulations, such as the possibility of the long-awaited AMB for oral use and the production of a generic version, for example, the cost of LFABs may start to decrease and its wide access will be offset by reduced rates of toxicity.
Finally, the newest treatment guidelines still mention its use as first-line therapy in certain defined situations, which reinforces AMB as the official holder of the gold standard title in the treatment of serious invasive fungal diseases.
Historical Findings, Characteristics, and Structures
Amphotericin B belongs to the class of polyene macrolides which also comprises amphotericin A and nystatin, the latter being considered the first antifungal agent developed for the treatment of mycoses [13], despite its production as a systemic agent being avoided because of serious toxicities.
The drug was discovered in 1956 by Donovick, Gold, Pagano, and Stout [14] following the fermentation of the actinomycete Streptomyces nodosus, originally identified as M-4575, isolated from a soil sample collected in the Orinoco River region, in Venezuela. As a therapeutic agent, it was licensed in 1959, on the basis of available and non-comparative data [1], and became accessible commercially in 1960 as Fungizone® (Bristol-Myers-Squibb, USA), a colloidal suspension of AMB.
Currently AMB is certified for the treatment of various fungal and potentially fatal infections such as opportunistic mycoses, e.g., aspergillosis, candidiasis, cryptococcosis, fusariosis, mucormycosis, hyalohyphomycosis, and phaohyphomycosis, as well as severe and widespread forms of endemic mycoses, e.g., histoplasmosis, paracoccidioidomycosis, blastomycosis, coccidioidomycosis, sporotrichosis, talaromycosis (Talaromyces marneffei, formally Penicillium marneffei), and emergomycosis [15,16,17,18,19]. Lipotrophic molecules such as deoxycholate, liposomes, and lipid complexes were added in the intravenous formulations for systemic use because of the insolubility of the standard form.
AMB comprises a 38-membered macrocyclic ring formed by lactonization and it has a chain of unsubstituted conjugated double bonds (heptaene) (Fig. 1a). On the opposite side, a polyhydroxylated chain with seven free hydroxyl groups guarantees it an amphipathic characteristic. A mycosamine residue (lactone) rests at one end of the molecule, with a free amino group, forming a side chain [20]. The conventional formulation contains approximately 41 mg of sodium deoxycholate and 20.2 mg of phosphate buffer [21]. Sodium deoxycholate increases the solubility of amphotericin B in water, because, although AMB has an amphiphilic region, its solubility in water is low. Sodium deoxycholate also stabilizes the micellar suspension formed [22, 23]. The hydrophobic part of the molecule binds to ergosterol, the main sterol in the cytoplasmic membrane of fungi. As a result of this connection, pores and channels are formed in the plasma membrane that allow the extravasation of electrolytes from the intracellular medium such as potassium, ammonium, and phosphate in addition to carbohydrates and proteins, thus causing cell death [24,25,26,27,28].
This mechanism of action (Fig. 1b), added to an induction of oxidative damage in the fungal cell [28], guarantees its fungicidal characteristic. Nevertheless, the success of this interaction depends on the concentration of the drug in body fluids and on the fungal specimen’s susceptibility to it.
Besides its affinity for the fungal ergosterol, AMB also stands out as a molecule with affinity for cholesterol present in mammalian cells (Fig. 1c). This characteristic per se explains why kidneys, heart, and blood cells are damaged during some therapeutic schemes [25, 29]. Despite that, there is no other antifungal medication that combines so many positive characteristics. Its potent fungicidal activity, broad spectrum of action, and rare induction of resistance guarantees it as an extremely effective option among other chemotherapeutic possibilities [30]. Studies have been conducted to improve lipid preparations as vehicles for new formulations such as liposomes [31,32,33,34,35,36,37], lipid complexes [38,39,40], emulsions [41,42,43,44,45,46], nanoparticles with dimercaptosuccinic acid [47], cationic lipid–polymer hybrids targeting macrophages [48], and Pluronic F127 micelles [49]. Figure 2 illustrates the main historical events of the last 60 years, maintaining AMB as the gold standard for the treatment of most invasive fungal infections (IFIs).
The current lipid formulations of amphotericin B (LFABs) available for clinical use differ in pharmacological characteristics such as structure, shape, size, composition, and toxicity—when compared to the conventional formulation (Table 1). ABLC and L-AMB allow the administration of higher doses and vary in efficacy and toxicity depending on the preparation and the species of fungus. Both benefits were goals for the development of LFABs, and the approval of these formulations was based on their comparison with conventional amphotericin B in clinical trials that are also cited in Table 1 for any further reading.
Although licensing decisions in the USA for LFABs have been based primarily on data from open non-comparative studies, there is now more available data that supports the effectiveness and safety of these compounds in the treatment of systemic fungal infections. The use of higher concentrations of AMB in less toxic lipid formulations is of great importance owing to its high clinical tolerability. However, undesirable effects such as fever, chills, stiffness, drowsiness, slight elevation in liver function tests, renal dysfunction, and cardiopulmonary toxicity have been documented even in patients who received those liposomal subtypes [58]; therefore, studies are still being carried out to find ways to reduce these events further. Recent research presented results regarding the development of nanoparticles, signaling superiority of these compounds to the conventional preparations [46,47,48, 60].
Infusion-related toxicity is a side effect which was initially attributed to the conventional formulation, as a result of the pro-inflammatory response to cytokines that manifests during the first minutes of administration. The symptoms are well controlled with antihistamines, analgesics, and corticosteroids [106].
It was the study by Gigliotti et al. [107] that postulated the chills and fever produced by an infusion of AMB were mediated through prostaglandin E2 synthesis. After this understanding new findings started bringing attention to the use of premedication as new way to better prevent these side effects. Some trials—even decades before that—revealed that hydrocortisone could be effective in the prevention of infusion-related reactions because of its cytokine transcription inhibitory property [108]. There are also studies about the use of opioids (mainly represented by meperidine IV in bolus) as a good option in ameliorating the infusion reactions. Authors argue that meperidine can eliminate these reactions more effectively and more rapidly than simply discontinuing the AMB [109]. Acetaminophen and metamizole are also commonly reported as drugs used for premedication.
In short, reactions related to the infusion of conventional therapy are possibly treatable. For patients who develop undesirable reactions, switching to an LFAB can also be a solution.
However, in 2003, Roden et al. [110] stated that the infusion of L-AMB could result in an idiosyncratic reaction manifested as a triad of chest pain and/or discomfort, flank and/or abdominal pain and dyspnea. This reaction is credited more to the liposome than the active drug itself [111], although Wade et at. [112] ensured that the toxicity related to the infusion of L-AMB is consistently less that other formulations of polyenes, including ABLC.
For the lipid complex of AMB there are recommendation in case of adverse events, including the infusion-related ones. The document suggests as optimal premedication the use of hydrocortisone and chlorphenamine (antihistamine agent) given 15–30 min before the infusion. Other advice includes the minimum infusion time of 2 h and adequate hydration before and after dosing for renal function improvement [113].
Pharmacokinetics and Pharmacodynamics Properties
The understanding of the pharmacokinetic (PK) and pharmacodynamic (PD) properties of an antimicrobial agent is based on the exposure–response relationship between the drug and the pathogen. Such affinity can be established by integrating PK and PD parameters, such as both maximum concentration (Cmax) and the area under the curve (AUC), and the minimum inhibitory concentration (MIC), respectively. As a result, optimal drug regimens are achieved, and toxicity and resistance development are minimized [114, 115].
To determine the PK/PD index of an antifungal, in vitro and in vivo studies are performed. In vitro susceptibility tests are needed to determine MIC in reproducible conditions and PK studies to estimate population parameters (clearance and volume of distribution). At the end, there is a dose–response experiment to relate exposure to the antifungal effect using the drug fractionation [116]. For AMB, it is known that the PK/PD indices were previously determined and their results confirmed by clinical studies which show that optimizing the dose to reach the PD targets leads to greater clinical efficacy [117].
Among lipid formulations, the clinical correlation of potential differences in PD has been difficult to establish because of limitations in determining MIC and differences in models of PK/PD indexes [116]. But, the study by Hong et al. [118], which included nine children diagnosed with fungal infection and treated with L-AMB, found that while Cmax/MIC = 40 ± 13 produced a partial response, the complete response would need values of 67.9 ± 17 (p = 0.021). Considering the study by Andes et al. that reported D-AMB as five times more potent (Cmax/MIC = 10), the results with L-AMB could be considered consistent [117].
Anyway, the pharmacokinetics of amphotericin B varies substantially between D-AMB, L-AMB, and ABLC, and its parameters should not be used to predict the behavior of any other AMB formulation [119]. The drug is poorly absorbed by the gastrointestinal tract and must be administered parenterally to treat systemic fungal infections.
As a fungicidal, amphotericin B relies on its concentration to display its antifungal effect. As mentioned before, the ability to reach those concentrations will determine the success of an intervention [120, 121].
According to the current manufacturer of D-AMB, an initial intravenous infusion of 1–5 mg/day gradually increased by 0.4–0.6 mg/kg/day produces Cmax ranging from approximately 0.5 to 2 μg/mL, which stabilizes at about 0.5 μg/mL. D-AMB is highly bound to plasma proteins (> 90%) and has an initial half-life of 24 h and an elimination period of 15 days. About two-thirds of plasma concentrations are detected concomitantly in peritoneal, synovial, inflamed pleura, and aqueous fluids and rarely exceed 2.5% in cerebrospinal fluid. Highly resistant fungi may need higher interventions such as 1.5 mg/kg per day, with prolonged infusions over 6 h, compared with 4 h for susceptible species [122,123,124,125,126].
D-AMB concentrations in vitreous humor or normal amniotic fluid are negligible, whereas full details of its tissue distribution are not known. Excretion of the drug is slow though the kidneys, with less than 5% of the dose being eliminated in the active form. The accumulated urine output over a period of 7 days is equivalent to approximately 40% of the amount of drug infused [127].
Balancing the drug’s kinetics along with its collateral effects, continuous infusion became one of the main strategies for the treatment of fungal infections with AMB, enhancing tolerability and lowering mortality, whilst reducing infusion-related toxicity [128,129,130].
LFABs, on the other hand, present a varied pattern in their pharmacokinetics, with primarily data obtained from animal studies. In 1989, Gondal et al. [131] reported peak concentrations five times higher compared to the same dosage of the conventional formulation, after administering 1 mg/kg of L-AMB to mice.
Subsequently, another study also indicated increased L-AMB concentrations in blood, liver, and spleen, while decreased levels were reported in kidneys and lungs [132]. In human beings, results described by Tollemar and Ringdén [133] showed that a dose of 3 mg/kg of the same compound obtained an average Cmax of 24.3 μg/mL—in accordance with previous studies that reported even greater peaks, varying from 10 to 35 μg/mL [134, 135], but reaching lower concentrations than those produced by other LFABs in liver, spleen, lung, and kidneys—except for the central nervous system [50, 136]. After the administration of 5 mg/kg/day of liposomal amphotericin B, 90 µg/mL peak levels were measured, along with a half-life of 5–10 h [134].
It is presumed that the volume of distribution (Vd) of the liposome is limited by a decreased AMB interaction with membrane proteins and/or cholesterol, thus allowing significantly higher peak concentrations. However, the association of these higher concentrations to an increased antifungal action in vivo is still not determined [137]. Despite these data, the pharmacokinetics of L-AMB remains relatively unclear, but it is a fact that a liposome composition has a significant impact on the properties of such formulations [138].
The first detailed profile of ABLC’s disposition in human beings presented a broad interindividual variability, beyond large tissue distribution and a long-standing half-life time—similar to D-AMB. At standard doses, a Cmax of 2 µg/mL was registered for the lipid complex [52, 136, 139].
The lipid complex of amphotericin B has a nonlinear dose-dependent kinetics, and, in contrast to the usual pattern, an increased clearance and Vd according to the dosage administered. In multiple schemes, with an interval smaller than t1/2, there is little accumulation of the drug in the body [52]. As for tissue levels, different ratios are reported, according to the systems tracked: 0.2× (kidneys and brain), 2× (liver and lungs), and 5× (spleen), when compared to plasma concentrations [136].
Special populations such as pregnant women, elderly, and obese still lack pharmacokinetic studies on their activity [128]. As for neonates, preterm infants, and children, although there is extensive use on these age ranges, pharmacokinetic data and ideal dosage schemes are also scarce and limited, especially for infants under 10 kg [118, 140].
Structure–Activity Relationship and Drug–Target Interactions
As a general rule, the polyenes exert their effect by associating with sterols of the fungal membrane and interrupting their integrity. This association occurs because of the high affinity between the drug and fungal ergosterol, which, after forming pores in its membrane, spills ions out of the cells, resulting in their death. Nevertheless, this affinity is less for human cholesterol, which explains the drug’s greater effect on the pathogen than on host cells.
In 1988, Chéron et al. [141] were the pioneers in studying the correlation between AMB derivates and their biological activity. Such compounds represent a unique basis for the study of the antifungal structure–activity relationship and the understanding of its properties [142, 143]. Basically, the four axes that support its structure–activity relationship are (1) the derivation of a hydroxyl group at C-13; (2) the absence of a negative charge in the acid group; (3) the polyene itself; and (4) an ionizable nitrogen [144, 145].
The crucial role of mycosamine and the C-35-OH group in the antifungal activity of AMB has been demonstrated by Gray et al. [146]. They concluded that the antifungal mechanism of action of the drug is through a simple binding to the ergosterol of the fungi cells. However, a study by Tevyashova et al. [147] to evaluate several semi-synthetic derivates of AMB showed that those which contained the C-35-OH group and the mycosamine portion afforded low antifungal activity. This result was attributed to the decisive role of the hydroxyl group, especially its position in the region of C-7 to C-10, in the biological activity of AMB.
As for the hypothesis that the ergosterol binding is fundamental to the antifungal activity, a test performed with a derivate of AMB lacking the mycosamine portion suggested the capacity of the composition to bind to ergosterol, but not to form pores in the membrane. This research concluded that the direct interaction mediated by mycosamine between amphotericin B and ergosterol is necessary to form ion channels and cause the death of fungal cells [148]. Once again, Tevyashova et al. [147] obtained different results, suggesting that the mycosamine group does not play a critical role in the interaction with ergosterol. Therefore, the detailed mechanism of these interactions is not yet clear and needs to be investigated.
In its liposomal formulation, amphotericin B is integrated with the liposome membranes, forming a non-covalent complex between mycosamine (positively charge) and distearoylphosphatidylglycerol (DMPG) (negatively charged), as well as hydrophobic interactions. Liposomes accumulate at the site of infection, adhering to the surface of fungal cells, disintegrating and releasing AMB [149]. The amphotericin B lipid complex, on the other hand, depends on fungal lipases acting on the formulation to then induce drug release in tissues [150].
Mechanisms of Action and Immune Response
After 60 years of investigation, the mechanisms of antifungal action of are not fully elucidated. However, there are ample consensus and evidence that AMB affects cells in two ways: via ergosterol binding and via oxidative damage.
Ergosterol Binding
Basically, the drug interacts with the lipid bilayer of the membrane through its hydrophobic domains resulting in multimeric pores that increase the permeability of ions (K+, Ca2+, and Mg2+) and cause intracellular loss and consequent cell death.
The specific mechanisms of pore formation and the AMB membrane entrance remain very unclear. Baginski et al. [30] proposed two hypothetical pathways in which AMB–ergosterol binding can happen. The sequential mechanism assumes that the AMB monomers somehow enter the membrane and form binary complexes with the lipids found there, forming the channels. The one-step mechanism assumes that the AMB supramolecular complexes are first formed on the surface of the membrane and shortly after they enter the membrane, producing a reorganization towards the functional channels. Palacios et al. [151] also later described two different mechanisms for it: the sterol sequestration (membrane destabilization) and the membrane permeabilization (ion depletion).
It is clear that ergosterol is needed in a large number of cellular processes such as endocytosis, vacuole fusion, and stabilization of proteins, and that the formation of pores increases antifungal efficacy; however, it is not essential for the death of fungal cells, since the simple connection and sequestration of ergosterol to AMB is sufficient to damage cells because of the multiple cellular processes in which ergosterol is involved [152, 153]. Other studies corroborate this premise, demonstrating that not only is the formation of pores sufficient to produce cell death but that the chemical modifications in the AMB domains do not affect its antifungal activity [28, 154]. Finally, two studies argue that AMB is able to form channels even in the absence of sterols; however, both agree that the concentration required to form pores in these conditions is much higher than in the presence of sterols [155, 156].
Oxidative Damage
Early studies demonstrated that AMB induces oxidative stress in the cells [157, 158]. More recently, Liu et al. [159] confirmed this through genome-wide expression analysis showing that the drug induces the expression of stress genes. Many other independent studies have been performed but the precise role of AMB’s oxidative damage in its antifungal activity remains undetermined [160,161,162,163,164,165].
Among the possibilities, AMB could act directly as a pro-oxidant and induce the accumulation of reactive oxygen species, which leads to influence of its mitochondrial activity, contributing to the oxidative burst. Consequently, the accumulation of free radicals induces multiple deleterious effects on the essential components of the cell, resulting in cell death [28].
In 1996, Brajtburg and Bolard [166] reported a compilation of revised information about the immunostimulatory properties of AMB. At first the drug induces an immune response predominantly in a proper dose range. The example used came from their study in which AMB increased the immune response in most inbred strains of mice. In addition, its prophylactic use against fungal infections would come from these assumptions, from stimulating the immune system under the appropriate conditions [167]. The article also highlights that although there are experimental studies agreeing with the stimulating effects of AMB on cells of the immune system, the suppression of humoral and cell-mediated immunity, as well as the suppression of macrophage activation, has also been reported.
Thus, the immunomodulatory effect also has been related to the AMB-associated toxicity. Suschek et al. [168] demonstrated that AMB increases the expression of the inducible nitric oxide synthase (iNOS) isoform, producing an increase in nitric oxide (implicated in the processes of vasodilation and protection against pathogens). However, AMB also increases the induction of pro-inflammatory cytokines and it would therefore be related to the drug’s toxicity in the host [169].
What is known so far is that the drug interacts with Toll-like receptors (TLR2), inducing the release of pro-inflammatory cytokines including interleukin-6 (IL-6), IL-8, tumor necrosis factor (TNFα), and monocyte chemotactic protein 1 ((MCP-1). On the other hand, its interaction with TLR4 produces the release of IL-10, an anti-inflammatory cytokine [170]. In addition, the binding of AMB to sterols can activate membrane enzymes, such as NADPH oxidase, involved in the oxidative stress pathway, generating the accumulation of free radicals as previously described.
Spectrum of Action
Literature about AMB’s activity against different fungal specimens is conflicting. Despite being a well-known agent against a number of invasive infections, some clinical practice data support therapeutic failure in species like Candida albicans and Candida parapsilosis [171,172,173,174], previously considered to be fully susceptible [175, 176].
Susceptible
It is common sense that most yeasts and molds are susceptible to amphotericin B. Among the genus Candida, the species Candida tropicalis, Candida krusei, Candida kefyr, Candida famata, and Candida guilliermondii are all considered susceptible [171,172,173,174, 176]. In addition, Cryptococcus neoformans, Malassezia spp., Saccharomyces cerevisiae, Aspergillus nidulans, Aspergillus niger, and Penicillium marneffei are also designated as responsive [174, 176,177,178,179].
Intermediate
Aspergillus terreus, melanized fungi like Bipolaris spp., Exophiala spp., Cladophialophora spp., Fonsecaea spp. and Phialophora spp. among others, along with some Paecilomyces species are reported as intermediate in susceptibility [176,177,178,179,180,181,182,183].
Contradictory
Some inconsistent data is reported on Scedosporium apiospermum, Aspergillus fumigatus, Aspergillus flavus, Trichosporon beigelii, and Fusarium spp. regarding resistance in some isolates and treatment failures [176, 177, 180, 181, 184, 185]. On the other hand, Candida lusitaniae has been reported to be resistant; nevertheless, most strains were susceptible in the laboratory [171, 172, 174]. Additionally, difficult culture techniques or poor laboratory data also compromise the susceptibility appraisal, being reported for Malassezia spp. and Zygomycetes (Absidia corymbifera, Apophysomyces elegans, Cunninghamella bertholletiae, Mucor spp., Rhizomucor pusillus, Rhizopus spp., Saksenaea vasiformis) [180].
Resistant
Previously, Scedosporium prolificans and Sporothrix schenckii were reported as remarkable resistant species [181, 184, 185]. Nowadays, the most recent guidelines suggests caution with this therapeutic approach, because of other first-line options or limited data. The indication large remains for severe or disseminated disease or when the first-line treatment is unavailable [18, 19]. Among Candida species, Candida auris, Candida haemulonii, and Candida lusitaniae are considered resistant [186, 187].
Other Therapeutic Uses of AMB
In addition to its antifungal action, evidence also supports the clinical use of amphotericin B in other contexts, as already well established in the treatment of visceral leishmaniasis (L-AMB formulation), caused by the parasite Leishmania ssp. [188]. Its use on cutaneous and mucosal leishmaniasis, on the contrary, is still considered off-label by the US Food and Drug Administration (FDA) [188,189,190,191].
Evidence also points to antiprotozoal therapeutic applications with Trypanosoma cruzi epimastigotes and many reports with Naegleria fowleri, for example. Such reports also show that a mixed therapeutic approach could benefit the patient in both cases [157, 192, 193].
There are also studies that propose AMB as a promising new option as an antiviral agent, as it can affect the structure of cholesterol in viral envelopes and cell membranes, as well as in intracellular organelles. Data from experimental studies on the human immunodeficiency virus (HIV), Japanese encephalitis virus, as well as hepatitis B, herpes simplex, and rubella viruses have been reported [194,195,196,197].
Mechanisms of Resistance
Fungal resistance mechanisms against AMB are rare, but have been reported, portraying a status where the patient does not respond to a standard therapeutic approach [198]. In 2014 Anderson et al. [199], in order to explain the paucity of clinically relevant microbial resistance against AMB, expanded the classic ion channel model and presented a sterol sponge model, in which AMB exists primarily in the form of an extra-membranous aggregates that physically extracts ergosterol from lipid bilayers. According to those authors, once the molecule may simultaneously perturb all of the cellular processes that depend on membrane ergosterol, a number of mutations would be necessary to provoke a relevant alteration, thus causing resistance [199].
Generally, there are host and microbial factors that interfere directly or indirectly with the immune response against a pathogen that predict the success of an intervention [200]. Important host factors are, for example, the immune status of the patient and presence of indwelling materials and surgical devices—possible vehicles for contaminations and biofilm development that could prevent sufficient concentrations of the drug reaching the infection site [201,202,203].
As for its mechanism of action, decreases in either the amount of ergosterol in the cell membrane or a change in the target lipid could compromise AMB’s performance, as a result of decreasing binding sites [175, 198]. In the same way, any mutations in the ergosterol production pathway could affect the quality of this interaction, resulting in poor kinetics—such as ERG genes, required for ergosterol biosynthesis [200].
Candida albicans resistant to amphotericin B and fluconazole, for example, revealed upregulated ERG5, ERG6, and ERG25 genes when compared with the wild-type strain. These mutations lead to an accumulation of sterol intermediates and a reduced affinity for AMB [204]. ERG2 and ERG3 mutations were also related, carrying a low ergosterol content [205,206,207]. Promastigotes of Leishmania donovani also highlight the importance of sterols, once their absence is related to resistance to amphotericin B [208]. Similar results were obtained from cultures of C. tropicalis and Torulopsis glabrata of a hematopoietic stem cell transplantation population [209]. C. parapsilosis, C. lusitaniae, T. beigelii, Malassezia furfur, S. apiospermum, S. prolificans, Fusarium spp., and some strains of S. schenckii also demonstrate primary resistance against amphotericin B as a result of those implications [175].
Oxidation resistance through increased catalase activity and incubation under hypoxia were credited for some C. albicans, A. terreus, and protoplasts cells [163, 210,211,212]. Additionally, biofilm formation was reported with Candida spp. [207]. Fatty acid composition was also proposed to explain polyene resistance, suggesting that an increased membrane fluidity could interfere with the interaction with amphotericin B. Nevertheless, no significant differences between mutants and wild types were tracked [213, 214]. Alterations of cell wall constituents such as chitin (C. albicans, Kluyveromyces spp., and Schizosaccharomyces spp.) and binding factors like glucans (C. albicans, C. tropicalis, A. flavus) could also determine resistance because of their preliminary role in polyene kinetics; notwithstanding, these mechanisms are only partially understood [215,216,217,218,219].
Finally, as ergosterol plays an essential role in the yeast cell cycle, stationary-phase cells were related to increased resistance in the exponential phase, a fact that could be associated with reduced chitin synthase activity in the stationary growth phase [216, 220, 221].
Resistance against amphotericin B during therapy is not common and is attributed to an acquired resistance of the pathogen or co-infection with different species [175]. In particular, patients with severe neutropenia or compromised hematopoietic health are likely to face this impasse [222, 223]. Cases of C. albicans, Candida rugosa, C. lusitaniae, and C. guilliermondii were reported [224,225,226].
Stability
According to the literature, reports of amphotericin B’s instability when submitted to unfavorable conditions such as exposure to heat, light, and low pH are commonly found [227]; it is even among the 110 substances liable to degradation in tropical conditions (50 °C and 100% humidity) listed by the World Health Organization (WHO) [228]. Aqueous solutions of AMB, on the contrary, could be more stable for prolonged periods of air and light exposure, if maintained between pH 4 and 10 [23, 229].
A recent study showed insignificant degradation of AMB in the presence of water [230]; this fact could be related to AMB’s low solubility in aqueous and neutral pH vehicles. Exposure of AMB to ± 70 °C for up to 7 days did not provoke thermal degradation. In a photolysis experiment, degradation occurred within 7 min after exposition to light, in agreement with previous studies that assessed AMB’s stability in dark environments [231].
Wiest et al. [232] documented the stability of amphotericin B (100 µg/mL) in four different concentrations of dextrose injection (5%, 10%, 15%, and 20%), when stored for up to 24 h at 15–25 °C and protected from light. This study raised an important question regarding the administration of the drug in dextrose solutions, with concentrations greater than 5%, which would minimize nutritional deficits and glucose instability in neonates.
Regarding the LFABs, the liposome stability is guaranteed by their small size and the fact that cholesterol and DMPG exhibit a high transition temperature (55 °C), when the preparation naturally tends to collapse, releasing its content [128, 233]. The lipid complex, with its two phospholipids, distestylylphosphatidylcholine (DMPC) and DMPG, has a transition temperature of 23 °C (below body temperature), which suggests that the preparation may disintegrate before reaching the site of action [233].
Bioequivalance or Therapeutic Equivalance
Pharmacologically, two preparations are considered equivalent if they present the same qualitative and quantitative composition of active ingredients and the same pharmaceutical form. Two pharmaceutical equivalents are defined as bioequivalent when, after administration of the same dosage, their bioavailability does not differ statistically. Once two equivalent pharmaceutical forms are also bioequivalent, theoretically, they can be considered as therapeutically equivalent [233].
With the advent of lipid formulations and the fact that they are related to reduction of toxicity, it became necessary to control these drugs to guarantee the level of tolerance and effectiveness, since any change in manufacturing may affect drug performance [24]. It is known that the lipid composition, charge, and size of these preparations can vary considerably depending on the manufacturer [137]. For instance, the manufacturer of ABLC informs in the package insert that liposomal encapsulation or incorporation in a lipid complex can substantially affect the functional properties of the drug by differing in the chemical composition and physical form of the lipid component [234], which, therefore, already attests to its non-bioequivalence.
In vitro [235] and in vivo animal studies [236] sought to establish a therapeutic equivalence of the conventional formulation compared to that associated with liposomes, claiming that both had the same antifungal potency. However, it is still questioned whether such preclinical bioequivalence data can be extrapolated in humans. Heinemann et al. [137] discussed the need to demonstrate that a dosage of D-AMB of 1 mg/kg would in fact equate to the same antifungal activity as a dose of L-AMB of 1 mg/kg.
Recently, a study gathered evidence and confirmed that LFABs are not therapeutically equivalent [233]. L-AMB and ABLC data are exposed in relation to Cmax and area under the curve (AUC), showing evident differences between them (non-standard confidence interval of 90%), once again opposing the definition of bioequivalence.
Yet, recommendations of scientific associations and guidelines clearly state the differences between LFABs when presenting their evidence grid in the treatment of different fungal infections. The guidelines of the Infectious Diseases Society of America (IDSA) declare that L-AMB and ABLC have the same spectrum of activity as D-AMB; however, they have distinct pharmacological properties and frequencies of adverse events [237].
Finally, Cifani et al. [233] summarized why lipid formulations cannot be considered therapeutically equivalent. First, because the preparations are not bioequivalent. Second, because there are not enough controlled clinical trials that compare the effectiveness of the formulations in question. Last of all, because therapeutic equivalence is not supported by worldwide guidelines and consensus as different recommendations are attributed to lipid formulations of amphotericin B in their recommendations.
Aspects Relevant to Clinical Practice
In the daily routine, AMB is an important resource in severe fungal infections, available as a useful agent against virulent infections such as A. flavus and Scedosporium spp., often related to refractoriness [238].
Data suggests different applications for the different formulations of AMB, including primary and secondary prophylaxis and in refractory disease, when aspergillosis is suspected or confirmed. As a primary prophylaxis, data supports the use of AMB in hematological malignancies (acute myeloid leukemia with prolonged neutropenia [239,240,241,242,243,244], acute lymphoblastic leukemia [245], allogeneic hematopoietic stem cell transplantation, HSCT [240]), as a result of a high-risk neutropenic status; invasive infections of the central nervous system [63, 64, 76, 243], pulmonary and extrapulmonary disease [63].
As a secondary prophylaxis, patients with previous invasive infections and undergoing allogeneic HSCT or entering a risk period with non-resectable foci of Aspergillus disease benefit from L-AMB [246, 247]. Whereas patients with refractory hematological disease had an improved survival rate with L-AMB 3–5 mg/kg [58, 248, 249] and ABLC 5 mg/kg [238, 249,250,251].
Amphotericin B toxicity is the barrier that prevents its proper prescription, which can result in the spread of infections and therapy failure [136]. In addition, acute infusion-related reactions often imply the interruption of a complete course of the medication. In this sense, LFABs have brought a significant advance in the treatment of invasive fungal infections, allowing prolonged and higher dosage use when compared to D-AMB. Such formulations have often been used interchangeably, although constant vigilance is necessary given the possibility of significant differences in their effectiveness. One question that remains concerns access to such formulations, since the high cost significantly limits their use in developing countries.
Combination Antifungal Therapy
The application of combined antifungal therapy (CAF) is widely accepted to maximize the antifungal effect through the synergistic effect by attacking the same or different targets in fungal cells [252]. As advantages, in addition to the synergistic effect, the amplitude in the spectrum of action, less risk of toxicity owing to the reduction of the combined doses, and less probability of resistance or tolerance (even without evidence to support this statement) can be mentioned. As disadvantages, antagonistic adverse reactions should be considered, in addition to the higher costs and the possibility of systemic toxicity due to the accumulation of more than one antifungal in the body [253, 254].
Drug interactions (whether synergistic or antagonistic) depend on the type of preparations used in a CAF, on the genus and species of fungi, and, of course, on the timing of drug administration and their doses [252]. Table 2 summarizes the available data on the use of amphotericin B in combination therapy, currently recommended in clinical practice.
Future Perspectives
Despite the broad spectrum of fungicidal activity, limitations such as parenteral administration, reactions related to infusion, acute and chronic toxicity, and also the dosage limits end up harming the potential clinical use of AMB. Although LFABs exhibit a more favorable tolerability and toxicity profile, they are not free of side effects.
The development of non-invasive formulations of AMB is very challenging because of its low aqueous solubility in physiological pH, permeability through membranes, and tendency to self-aggregate, in addition to its low stability at high temperatures and acid pH [261].
Progress in the development of a new formulation of AMB has been described in the literature, with emphasis on encochleated amphotericin B (Coch-AmB). It is a new formulation composed of phospholipid bilayers precipitated with bivalent cations in a multilayer structure, wrapped in a spiral without internal watery space. Such a structure protects the molecule inside, which makes it more stable and allows its oral administration. The drug is released after the interaction of this new system with the target cells, which open in the presence of low concentrations of intracellular calcium [262].
This new possibility would bring numerous advantages to clinical practice including the avoidance of unnecessary patient hospitalization, expansion of antifungal therapy to developing countries where access to hospitals is difficult, prophylactic use of AMB, lack of side effects related to the infusion, and accessibility to treatment. Finally, with the slower release of the active ingredient, higher concentrations could be achieved in several organs. Table 3 displays the list of the clinical trials from the last 2 years that seek to improve the use of amphotericin B in different situations.
Conclusions
In the 60 years since it was first marketed, amphotericin B remains the gold standard for the treatment of invasive fungal infections while its lipid formulations have been developed to improve tolerability with a similar spectrum of activity and a more favorable safety profile. However, they have considerably different pharmacological characteristics. Both ABLC and L-AMB have a distinguished pharmacokinetic profile that determines their efficacy and toxicity. On the other hand, the amphotericin B lipid complex, with a larger particle size, is characterized by a rapid decline in the concentration of AMB after intravenous administration, followed by an extended elimination half-life which contrasts with the higher Cmax values and AUC, lower volume of distribution, and shorter elimination half-life of the liposomal version.
Even though some experimental tests have been published, guidelines for better bioequivalence studies are lacking since it is essential to characterize both the stability and the pharmacokinetic profile of LFABs and thus ensure that not only patients benefit from these formulations but that professionals are safe to use them.
The development and registration of new formulations that bring improvements in pharmacological and biopharmaceutical characteristics represent expensive and time-consuming tasks but are essential to reduce toxicity and improve drug tolerability. Promising clinical trials stimulate new possibilities for amphotericin B. The goal will be achieved when AMB can be widely distribute at a lower cost and in a non-parenteral version, resulting in numerous benefits for end users.
References̄
Dutcher JD, Gold W, Pagano JF, Vandeputte J. Amphotericin B, Its Production, and Its Salts, in United States Patent Office, U.S.P. Office, Editor. 1959, James D. Dutcher: United States of America. p. 11.
Al-Mohsen I, Hughes WT. Systemic antifungal therapy: Past, present and future. Ann Saudi Med. 1998;18(1):28–38.
Utz JP, Treger A, Mc CN, Emmons CW. Amphotericin B: intravenous use in 21 patients with systemic fungal diseases. Antibiot Annu. 1958;6:628–34.
Utz JP. Amphotericin B toxicity. General Side Effects. Ann Intern Med. 1964;61:340–3.
Maddux BD, Whiting RB. Toxic synergism of disopyramide and hyperkalemia. Chest. 1980;78(4):654–6.
Medoff G, Kobayashi GS. Strategies in the treatment of systemic fungal infections. N Engl J Med. 1980;302(3):145–55.
Timmers GJ, Zweegman S, Simoons-Smit AM, van Loenen AC, Touw D, Huijgens PC. Amphotericin B colloidal dispersion (Amphocil) vs fluconazole for the prevention of fungal infections in neutropenic patients: data of a prematurely stopped clinical trial. Bone Marrow Transpl. 2000;25(8):879–84.
Hamill RJ. Amphotericin B formulations: a comparative review of efficacy and toxicity. Drugs. 2013;73(9):919–34.
Chang Y-L, Yu S-J, Heitman J, Wellington M, Chen Y-L. New facets of antifungal therapy. Virulence. 2017;8(2):222–36.
Rowen JL, Tate JM. Management of neonatal candidiasis. Neonatal Candidiasis Study Group. Pediatr Infect Dis J. 1998;17(11):1007–11.
Stevens DA, Shatsky SA. Intrathecal amphotericin in the management of coccidioidal meningitis. Semin Respir Infect. 2001;16(4):263–9.
Bishara J, Weinberger M, Lin AY, Pitlik S. Amphotericin B–not so terrible. Ann Pharmacother. 2001;35(3):308–10.
Dismukes WE. Introduction to antifungal drugs. Clin Infect Dis. 2000;30(4):653–7.
Donovick R, Gold W, Pagano JF, Stout HA. Amphotericins A and B, antifungal antibiotics produced by a streptomycete. I. In vitro studies. Antibiot Annu. 1955;3:579–86.
Chen SC-A, Perfect J, Colombo AL et al. Global guideline for the diagnosis and management of invasive infections caused by emerging, uncommon or rare yeasts. 2020 [cited 2020 October 5th].; Available from: https://www.clinicalsurveys.net/uc/admin/5445/images/Rare_Yeasts_Guideline_Draft_Public_Consultation.pdf.
Cornely OA, Alastruey-Izquierdo A, Arenz D, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19(12):e405–21.
Hoenigl M, Gangneux J-P, Segal E, et al. Global guidelines and initiatives from the European Confederation of Medical Mycology to improve patient care and research worldwide: New leadership is about working together. Mycoses. 2018;61(11):885–94.
Hoenigl M, Salmanton-Garcia J, Walsh TJ, et al. Global guideline for the diagnosis and management of rare mold infections: An initiative of the ECMM in cooperation with TBD. 2020 [cited 2020 October 5th].; https://www.ecmm.info/news/global-guideline-for-the-diagnosis-andmanagement-of-rare-mold-infections-public-review/.
Thompson GR, Le T, Chindamporn A, et al. Global guideline for the diagnosis and management of the endemic mycoses 2020 [cited 2020 Sep 2].; https://www.ecmm.info/news/global-guideline-for-the-diagnosis-and-management-of-the-endemic-mycoses-an-initiative-of-the-ecmmwith-tbd/.
Filippin FB, Souza LC. Therapeutic efficacy of amphotericin B lipid formulations. Braz J Pharm Sci. 2006;42(2):27.
Almeida MVAd. Amphotericin B and its lipid formulations, in Faculty of Health Sciences. 2013, University Fernando Pessoa. p. 58.
Martinez R. An update on the use of antifungal agents. Braz J Pneumol. 2006;32(5):12.
O'Neil MJ. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals. 14 ed. Journal of the American Chemical Society. 2007: American Chemical Society, p. 2197.
Adler-Moore JP, Gangneux JP, Pappas PG. Comparison between liposomal formulations of amphotericin B. Med Mycol. 2016;54(3):223–31.
Bergold AMGS. New antifungic drugs: a review. Visão Acadêmica 2004;5(2):13.
Finkelstein A, Holz R. Aqueous pores created in thin lipid membranes by the polyene antibiotics nystatin and amphotericin B. Membranes. 1973;2:377–408.
Georgopapadakou NH. Antifungals: mechanism of action and resistance, established and novel drugs. Curr Opin Microbiol. 1998;1(5):547–57.
Mesa-Arango AC, Scorzoni L, Zaragoza O. It only takes one to do many jobs: Amphotericin B as antifungal and immunomodulatory drug. Front Microbiol. 2012;3:286.
Sidrim JJC, Rocha MFG. Micologia médica à luz de autores contemporâneos. 1 ed. Guanabara Koogan. p. 396.
Baginski M, Sternal K, Czub J, Borowski E. Molecular modelling of membrane activity of amphotericin B, a polyene macrolide antifungal antibiotic. Acta Biochim Pol. 2005;52(3):655–8.
Chopra R, Blair S, Strang J, Cervi P, Patterson KG, Goldstone AH. Liposomal amphotericin B (AmBisome) in the treatment of fungal infections in neutropenic patients. J Antimicrob Chemother. 1991;28 Suppl B:93–104.
Hospenthal D, Gretzinger K, Rogers A. Treatment of a murine model of systemic candidiasis with liposomal amphotericin B bearing antibody to Candida albicans. J Med Microbiol. 1989;30(3):193–7.
Hospenthal DR, Rogers AL, Mills GL. Development of amphotericin B liposomes bearing antibody specific to Candida albicans. Mycopathologia. 1988;101(1):37–45.
Jung SH, Lim DH, Jung SH, et al. Amphotericin B-entrapping lipid nanoparticles and their in vitro and in vivo characteristics. Eur J Pharm Sci. 2009;37(3–4):313–20.
Lopez-Berestein G, Mehta R, Hopfer RL, et al. Treatment and prophylaxis of disseminated infection due to Candida albicans in mice with liposomeencapsulated amphotericin B. J Infect Dis. 1983;147(5):939–45.
Moribe K, Maruyama K, Iwatsuru M. Molecular localization and state of amphotericin B in PEG liposomes. Int J Pharm. 1999;193(1):97–106.
Wasan KM, Brazeau GA, Keyhani A, Hayman AC, Lopez-Berestein G. Roles of liposome composition and temperature in distribution of amphotericin B in serum lipoproteins. Antimicrob Agents Chemother. 1993;37(2):246–50.
Balakrishnan AR, Easwaran KR. Lipid-amphotericin B complex structure in solution: a possible first step in the aggregation process in cell membranes. Biochemistry. 1993;32(15):4139–44.
Janoff AS, Boni LT, Popescu MC, et al. Unusual lipid structures selectively reduce the toxicity of amphotericin B. Proc Natl Acad Sci USA. 1988;85(16):6122–6.
Tadini MC, de Freitas Pinheiro AM, Carrão DB, et al. Method validation and nanoparticle characterization assays for an innovative amphothericin B formulation to reach increased stability and safety in infectious diseases. J Pharm Biomed Anal. 2017;145:576–85.
Chavanet P, Clement C, Duong M, et al. Toxicity and efficacy of conventional amphotericin B deoxycholate versus escalating doses of amphotericin B deoxycholate–-fat emulsion in HIV-infected patients with oral candidosis. Clin Microbiol Infect. 1997;3(4):455–61.
Chavanet PY, Garry I, Charlier N, et al. Trial of glucose versus fat emulsion in preparation of amphotericin for use in HIV infected patients with candidiasis. BMJ. 1992;305(6859):921–5.
Davis SS, Washington C, West P, et al. Lipid emulsions as drug delivery systems. Ann NY Acad Sci. 1987;507:75–88.
Kirsh R, Goldstein R, Tarloff J, et al. An emulsion formulation of amphotericin B improves the therapeutic index when treating systemic murine candidiasis. J Infect Dis. 1988;158(5):1065–70.
Miyazaki T, Kohno S, Yasuoka A, et al. A lipid emulsion formulation of ampiootericin B for the treatment of murine candidiasis and cryptococcosis. Chemotherapy. 1990;38(6):548–51.
Richter AR, Feitosa JPA, Paula HCB, Goycoolea FM, de Paula RCM. Pickering emulsion stabilized by cashew gum- poly-l-lactide copolymer nanoparticles: synthesis, characterization and amphotericin B encapsulation. Colloids Surf B Biointerfaces. 2018;164:201–9.
Souza AC, Nascimento AL, de Vasconcelos NM, et al. Activity and in vivo tracking of Amphotericin B loaded PLGA nanoparticles. Eur J Med Chem. 2015;95:267–76.
Asthana S, Jaiswal AK, Gupta PK, Pawar VK, Dube A, Chourasia MK. Immunoadjuvant chemotherapy of visceral leishmaniasis in hamsters using amphotericin B-encapsulated nanoemulsion template-based chitosan nanocapsules. Antimicrob Agents Chemother. 2013;57(4):1714–22.
Shaarani S, Hamid SS, Mohd Kaus NH. The Influence of Pluronic F68 and F127 Nanocarrier on Physicochemical Properties, In vitro Release, and Antiproliferative Activity of Thymoquinone Drug. Pharmacognosy Res. 2017;9(1):12–20.
Groll AH, Giri N, Petraitis V, et al. Comparative efficacy and distribution of lipid formulations of amphotericin B in experimental Candida albicans infection of the central nervous system. J Infect Dis. 2000;182(1):274–82.
Pappas PG, Kauffman CA, Andes DR, et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62(4):e1–50.
Adedoyin A, Bernardo JF, Swenson CE, et al. Pharmacokinetic profile of ABELCET (amphotericin B lipid complex injection): combined experience from phase I and phase II studies. Antimicrob Agents Chemother. 1997;41(10):2201–8.
Martino R, Cortés M, Subirá M, Parody R, Moreno E, Sierra J. Efficacy and toxicity of intermediate-dose amphotericin B lipid complex as a primary or salvage treatment of fungal infections in patients with hematological malignancies. Leukemia Lymphoma. 2005;46(10):1429–35.
Oravcová E, Mistrík M, Sakalová A, et al. Amphotericin B lipid complex to treat invasive fungal infections in cancer patients: report of efficacy and safety in 20 patients. Chemotherapy. 1995;41(6):473–6.
Sharkey PK, Graybill JR, Johnson ES, et al. Amphotericin B lipid complex compared with amphotericin B in the treatment of cryptococcal meningitis in patients with AIDS. Clin Infect Dis. 1996;22(2):315–21.
Subirà M, Martino R, Gómez L, Martí JM, Estany C, Sierra J. Low-dose amphotericin B lipid complex vs. conventional amphotericin B for empirical antifungal therapy of neutropenic fever in patients with hematologic malignancies--a randomized, controlled trial. Eur J Haematol. 2004;72(5):342–7.
Walsh TJ, Finberg RW, Arndt C, et al. Liposomal amphotericin B for empirical therapy in patients with persistent fever and neutropenia. National Institute of Allergy and Infectious Diseases Mycoses Study Group. N Engl J Med. 1999;340(10):764–71.
Walsh TJ, Hiemenz JW, Seibel NL, et al. Amphotericin B lipid complex for invasive fungal infections: analysis of safety and efficacy in 556 cases. Clin Infect Dis. 1998;26(6):1383–96.
Wingard JR. Efficacy of amphotericin B lipid complex injection (ABLC) in bone marrow transplant recipients with life-threatening systemic mycoses. Bone Marrow Transpl. 1997;19(4):343–7.
Arrieta AC, Shea K, Dhar V, et al. Once-weekly liposomal amphotericin B as Candida prophylaxis in very low birth weight premature infants: a prospective, randomized, open-label, placebo-controlled pilot study. Clin Ther. 2010;32(2):265–71.
Bodhe PV, Kotwani RN, Kirodian BG, Kshirsagar NA, Pandya SK. Open label, randomised, comparative phase III safety and efficacy study with conventional amphotericin B and liposomal amphotericin B in patients with systemic fungal infection. J Assoc Phys India. 2002;50(5):662–70.
Cordonnier C, Pautas C, Maury S, et al. Empirical versus preemptive antifungal therapy for high-risk, febrile, neutropenic patients: a randomized, controlled trial. Clin Infect Dis. 2009;48(8):1042–51.
Cornely OA, Maertens J, Bresnik M, et al. Liposomal amphotericin B as initial therapy for invasive mold infection: a randomized trial comparing a high-loading dose regimen with standard dosing (AmBiLoad trial). Clin Infect Dis. 2007;44(10):1289–97.
Ellis M, Spence D, de Pauw B, et al. An EORTC international multicenter randomized trial (EORTC number 19923) comparing two dosages of liposomal amphotericin B for treatment of invasive aspergillosis. Clin Infect Dis. 1998;27(6):1406–12.
Jadhav MP, Shinde VM, Chandrakala S, et al. A randomized comparative trial evaluating the safety and efficacy of liposomal amphotericin B (Fungisome) versus conventional amphotericin B in the empirical treatment of febrile neutropenia in India. Indian J Cancer. 2012;49(1):107–13.
Johnson PC, Wheat LJ, Cloud GA, et al. Safety and efficacy of liposomal amphotericin B compared with conventional amphotericin B for induction therapy of histoplasmosis in patients with AIDS. Ann Intern Med. 2002;137(2):105–9.
Meunier F, Prentice HG, Ringdén O. Liposomal amphotericin B (AmBisome): safety data from a phase II/III clinical trial. J Antimicrob Chemother. 1991;28 Suppl B:83–91.
Penack O, Schwartz S, Martus P, et al. Low-dose liposomal amphotericin B in the prevention of invasive fungal infections in patients with prolonged neutropenia: results from a randomized, single-center trial. Ann Oncol. 2006;17(8):1306–12.
Ringdén O, Meunier F, Tollemar J et al. Efficacy of amphotericin B encapsulated in liposomes (AmBisome) in the treatment of invasive fungal infections in immunocompromised patients. J Antimicrob Chemother 1991;28 Suppl B:73–82.
Shah T, Lai WK, Gow P, Leeming J, Mutimer D. Low-dose amphotericin for prevention of serious fungal infection following liver transplantation. Transpl Infect Dis. 2005;7(3–4):126–32.
Sunakawa K, Tsukimoto I, Tsunematsu Y, et al. Evaluation of the safety and efficacy of liposomal amphotericin B (L-AMB) in children. J Infect Chemother. 2012;18(4):456–65.
de Lalla F, Pellizzer G, Vaglia A, et al. Amphotericin B as primary therapy for cryptococcosis in patients with AIDS: reliability of relatively high doses administered over a relatively short period. Clin Infect Dis. 1995;20(2):263–6.
Joly V, Aubry P, Ndayiragide A, et al. Randomized comparison of amphotericin B deoxycholate dissolved in dextrose or Intralipid for the treatment of AIDS-associated cryptococcal meningitis. Clin Infect Dis. 1996;23(3):556–62.
Pappas PG, Chetchotisakd P, Larsen RA, et al. A phase II randomized trial of amphotericin B alone or combined with fluconazole in the treatment of HIV-associated cryptococcal meningitis. Clin Infect Dis. 2009;48(12):1775–83.
Riley DK, Pavia AT, Beatty PG, et al. The prophylactic use of low-dose amphotericin B in bone marrow transplant patients. Am J Med. 1994;97(6):509–14.
Schwartz S, Ruhnke M, Ribaud P, Reed E, Troke P, Thiel E. Poor efficacy of amphotericin B-based therapy in CNS aspergillosis. Mycoses. 2007;50(3):196–200.
Techapornroong M, Suankratay C. Alternate-day versus once-daily administration of amphotericin B in the treatment of cryptococcal meningitis: a randomized controlled trial. Scand J Infect Dis. 2007;39(10):896–901.
Aguado JM, Lumbreras C, González-Vidal D. Assessment of nephrotoxicity in patients receiving amphotericin B lipid complex: a pharmacosurveillance study in Spain. Clin Microbiol Infect. 2004;10(9):785–90.
Cannon JP, Garey KW, Danziger LH. A prospective and retrospective analysis of the nephrotoxicity and efficacy of lipid-based amphotericin B formulations. Pharmacotherapy. 2001;21(9):1107–14.
Hasibi M, Jafari S, Manshadi SA, et al. Efficacy of Intralipid infusion in reducing amphotericin-B-associated nephrotoxicity in head and neck invasive fungal infection: a randomized, controlled trial. Ear Nose Throat J. 2017;96(2):E18–e22.
Sorkine P, Nagar H, Weinbroum A, et al. Administration of amphotericin B in lipid emulsion decreases nephrotoxicity: results of a prospective, randomized, controlled study in critically ill patients. Crit Care Med. 1996;24(8):1311–5.
Arsura EL, Ismail Y, Freedman S, Karunakar AR. Amphotericin B-induced dilated cardiomyopathy. Am J Med. 1994;97(6):560–2.
Bandeira AC, Filho JM, de Almeida Ramos K. Reversible cardiomyopathy secondary to Amphotericin-B. Med Mycol Case Rep. 2016;13:19–21.
Barcia JP. Hyperkalemia associated with rapid infusion of conventional and lipid complex formulations of amphotericin B. Pharmacotherapy. 1998;18(4):874–6.
Chung DK, Koenig MG. Reversible cardiac enlargement during treatment with amphotericin B and hydrocortisone. Report of three cases. Am Rev Respir Dis. 1971;103(6):831–41.
Craven PC, Gremillion DH. Risk factors of ventricular fibrillation during rapid amphotericin B infusion. Antimicrob Agents Chemother. 1985;27(5):868–71.
Danaher PJ, Cao MK, Anstead GM, Dolan MJ, DeWitt CC. Reversible dilated cardiomyopathy related to amphotericin B therapy. J Antimicrob Chemother. 2004;53(1):115–7.
Groot OA, Trof RJ, Girbes AR, Swart NL, Beishuizen A. Acute refractory hyperkalaemia and fatal cardiac arrest related to administration of liposomal amphotericin B. Neth J Med. 2008;66(10):433–7.
Kullab SM, Patel PD, Lewis PO. Non-occlusive ST-segment elevated myocardial infarction following the administration of liposomal amphotericin B in the treatment of cryptococcal meningitis. J Clin Pharm Ther. 2020.
Moyssakis I, Vassilakopoulos TP, Sipsas NV, et al. Reversible dilated cardiomyopathy associated with amphotericin B treatment. Int J Antimicrob Agents. 2005;25(5):444–7.
Rowles DM, Fraser SL. Amphotericin B lipid complex (ABLC)-associated hypertension: case report and review. Clin Infect Dis. 1999;29(6):1564–5.
Sanches BF, Nunes P, Almeida H, Rebelo M. Atrioventricular block related to liposomal amphotericin B. BMJ Case Rep. 2014;2014.
Soares JR, Nunes MC, Leite AF, Falqueto EB, Lacerda BE, Ferrari TC. Reversible dilated cardiomyopathy associated with amphotericin B therapy. J Clin Pharm Ther. 2015;40(3):333–5.
Bicanic T, Bottomley C, Loyse A, et al. Toxicity of Amphotericin B Deoxycholate-Based Induction Therapy in Patients with HIV-Associated Cryptococcal Meningitis. Antimicrob Agents Chemother. 2015;59(12):7224–31.
Arning M, Dresen B, Aul C, Schneider W. Influence of infusion time on the acute toxicity of amphotericin B: results of a randomized doubleblind study. Recent Results Cancer Res. 1991;121:347–52.
Ellis ME, al-Hokail AA, Clink HM et al. Double-blind randomized study of the effect of infusion rates on toxicity of amphotericin B. Antimicrob Agents Chemother. 1992;36(1):172–9.
Nicholl TA, Nimmo CR, Shepherd JD, Phillips P, Jewesson PJ. Amphotericin B infusion-related toxicity: comparison of two- and four-hour infusions. Ann Pharmacother. 1995;29(11):1081–7.
Wingard JR, White MH, Anaissie E, Raffalli J, Goodman J, Arrieta A. A randomized, double-blind comparative trial evaluating the safety of liposomal amphotericin B versus amphotericin B lipid complex in the empirical treatment of febrile neutropenia. L Amph/ABLC Collaborative Study Group. Clin Infect Dis. 2000;31(5):1155–63.
Blau IW, Fauser AA. Review of comparative studies between conventional and liposomal amphotericin B (Ambisome) in neutropenic patients with fever of unknown origin and patients with systemic mycosis. Mycoses. 2000;43(9–10):325–32.
Falci DR, da Rosa FB, Pasqualotto AC. Comparison of nephrotoxicity associated to different lipid formulations of amphotericin B: a real-life study. Mycoses. 2015;58(2):104–12.
Fleming RV, Kantarjian HM, Husni R et al. Comparison of amphotericin B lipid complex (ABLC) vs. ambisome in the treatment of suspected or documented fungal infections in patients with leukemia. Leuk Lymphoma. 2001;40(5–6):511–20.
Hooshmand-Rad R, Chu A, Gotz V, Morris J, Batty S, Freifeld A. Use of amphotericin B lipid complex in elderly patients. J Infect. 2005;50(4):277–87.
Jeon GW, Koo SH, Lee JH, et al. A comparison of AmBisome to amphotericin B for treatment of systemic candidiasis in very low birth weight infants. Yonsei Med J. 2007;48(4):619–26.
Leenders AC, Daenen S, Jansen RL, et al. Liposomal amphotericin B compared with amphotericin B deoxycholate in the treatment of documented and suspected neutropenia-associated invasive fungal infections. Br J Haematol. 1998;103(1):205–12.
Linder N, Klinger G, Shalit I, et al. Treatment of candidaemia in premature infants: comparison of three amphotericin B preparations. J Antimicrob Chemother. 2003;52(4):663–7.
Goodwin SD, Cleary JD, Walawander CA, Taylor JW, Grasela TH Jr. Pretreatment regimens for adverse events related to infusion of amphotericin B. Clin Infect Dis. 1995;20(4):755–61.
Gigliotti F, Shenep JL, Lott L, Thornton D. Induction of prostaglandin synthesis as the mechanism responsible for the chills and fever produced by infusing amphotericin B. J Infect Dis. 1987;156(5):784–9.
Saliba A, Beatty OA. Treatment of mycotic infections: hydrocortisone in the control of amphotericin-B toxicity. Dis Chest. 1962;41:214–9.
Burks LC, Aisner J, Fortner CL, Wiernik PH. Meperidine for the treatment of shaking chills and fever. Arch Intern Med. 1980;140(4):483–4.
Roden MM, Nelson LD, Knudsen TA, et al. Triad of acute infusion-related reactions associated with liposomal amphotericin B: analysis of clinical and epidemiological characteristics. Clin Infect Dis. 2003;36(10):1213–20.
Szebeni J, Baranyi L, Savay S, et al. Liposome-induced pulmonary hypertension: properties and mechanism of a complement-mediated pseudoallergic reaction. Am J Physiol Heart Circ Physiol. 2000;279(3):H1319–28.
Wade RL, Chaudhari P, Natoli JL, Taylor RJ, Nathanson BH, Horn DL. Nephrotoxicity and other adverse events among inpatients receiving liposomal amphotericin B or amphotericin B lipid complex. Diagn Microbiol Infect Dis. 2013;76(3):361–7.
Craddock C, Anson J, Chu P, et al. Best practice guidelines for the management of adverse events associated with amphotericin B lipid complex. Expert Opin Drug Saf. 2010;9(1):139–47.
Craig WA. Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin Infect Dis. 1998;26(1):1–10;quiz 11–2.
Drusano GL. Pharmacokinetics and Pharmacodynamics of Antimicrobials. Clin Infect Dis. 2007;45(Supplement_1):S89–95.
Gonzalez JM, Rodriguez CA, Agudelo M, Zuluaga AF, Vesga O. Antifungal pharmacodynamics: Latin America’s perspective. Braz J Infect Dis. 2017;21(1):79–87.
Andes D, Stamsted T, Conklin R. Pharmacodynamics of amphotericin B in a neutropenic-mouse disseminated-candidiasis model. Antimicrob Agents Chemother. 2001;45(3):922–6.
Hong Y, Shaw PJ, Nath CE, et al. Population pharmacokinetics of liposomal amphotericin B in pediatric patients with malignant diseases. Antimicrob Agents Chemother. 2006;50(3):935–42.
National Institutes of Health. Amphotericin B. PubChem 2006 September 22, 2022;2006.
Bellmann R, Smuszkiewicz P. Pharmacokinetics of antifungal drugs: practical implications for optimized treatment of patients. Infection. 2017;45(6):737–79.
Lepak AJ, Andes DR. Antifungal PK/PD considerations in fungal pulmonary infections. Semin Respir Crit Care Med. 2011;32(6):783–94.
Ayestarán A, López RM, Montoro JB, et al. Pharmacokinetics of conventional formulation versus fat emulsion formulation of amphotericin B in a group of patients with neutropenia. Antimicrob Agents Chemother. 1996;40(3):609–12.
Kan VL, Bennett JE, Amantea MA, et al. Comparative safety, tolerance, and pharmacokinetics of amphotericin B lipid complex and amphotericin B desoxycholate in healthy male volunteers. J Infect Dis. 1991;164(2):418–21.
Hoeprich PD. Elimination half-life of amphotericin B. J Infect. 1990;20(2):173–5.
Atkinson AJ Jr, Bennett JE. Amphotericin B pharmacokinetics in humans. Antimicrob Agents Chemother. 1978;13(2):271–6.
Maharom P, Thamlikitkul V. Implementation of clinical practice policy on the continuous intravenous administration of amphotericin B deoxycholate. J Med Assoc Thai. 2006;89(Suppl 5):S118–24.
National Institutes of Health. Amphotericin B. Clinical info HIV 2019 [cited 2020 September 22th]; https://clinicalinfo.hiv.gov/en/drugs/amphotericin-b/patient.
Stone NRH, Bicanic T, Salim R, Hope W. Liposomal amphotericin B (AmBisome(®)): a review of the pharmacokinetics, pharmacodynamics, clinical experience and future directions. Drugs. 2016;76(4):485–500.
Falci DR, Lunardi LW, Ramos CG, Bay MB, Aquino VR, Goldani LZ. Continuous infusion of amphotericin B deoxycholate in the treatment of cryptococcal meningoencephalitis: analysis of safety and fungicidal activity. Clin Infect Dis. 2010;50(5):e26–9.
Chabot GG, Pazdur R, Valeriote FA, Baker LH. Pharmacokinetics and toxicity of continuous infusion amphotericin B in cancer patients. J Pharm Sci. 1989;78(4):307–10.
Gondal JA, Swartz RP, Rahman A. Therapeutic evaluation of free and liposome-encapsulated amphotericin B in the treatment of systemic candidiasis in mice. Antimicrob Agents Chemother. 1989;33(9):1544–8.
Van Etten EW, Otte-Lambillion M, Van Vianen W, Ten Kate MT, Bakker-Woudenberg AJ. Biodistribution of liposomal amphotericin B (AmBisome) and amphotericin B-desoxycholate (Fungizone) in uninfected immunocompetent mice and leucopenic mice infected with Candida albicans. J Antimicrob Chemother. 1995;35(4):509–19.
Tollemar J, Ringdén O. Early pharmacokinetic and clinical results from a noncomparative multicentre trial of amphotericin B encapsulated in a small unilamellar liposome (AmBisome®). Drug Investig. 1992;4(3):232–8.
de Marie S, Janknegt R, Bakker-Woudenberg IA. Clinical use of liposomal and lipid-complexed amphotericin B. J Antimicrob Chemother. 1994;33(5):907–16.
Heinemann V, Kähny B, Debus A, Wachholz K, Jehn U. Pharmacokinetics of liposomal amphotericin B (AmBisome) versus other lipid-based formulations. Bone Marrow Transplant. 1994;14(Suppl 5):S8–9.
Ostrosky-Zeichner L, Marr KA, Rex JH, Cohen SH. Amphotericin B: time for a new “gold standard.” Clin Infect Dis. 2003;37(3):415–25.
Heinemann V, Bosse D, Jehn U, et al. Pharmacokinetics of liposomal amphotericin B (Ambisome) in critically ill patients. Antimicrob Agents Chemother. 1997;41(6):1275–80.
Bekersky I, Fielding RM, Dressler DE, Lee JW, Buell DN, Walsh TJ. Plasma protein binding of amphotericin B and pharmacokinetics of bound versus unbound amphotericin B after administration of intravenous liposomal amphotericin B (AmBisome) and amphotericin B deoxycholate. Antimicrob Agents Chemother. 2002;46(3):834–40.
Walsh TJ, Yeldandi V, McEvoy M, et al. Safety, tolerance, and pharmacokinetics of a small unilamellar liposomal formulation of amphotericin B (AmBisome) in neutropenic patients. Antimicrob Agents Chemother. 1998;42(9):2391–8.
Nath CE, McLachlan AJ, Shaw PJ, Coakley JC, Earl JW. Amphotericin B dose optimization in children with malignant diseases. Chemotherapy. 2007;53(2):142–7.
Chéron M, Cybulska B, Mazerski J, Grzybowska J, CzerwiŃski A, Borowski E. Quantitative structure-activity relationships in amphotericin B derivatives. Biochem Pharmacol. 1988;37(5):827–36.
Belakhov VV, Shenin YD. Synthesis and antifungal activity of N-benzyl derivatives of amphotericin B. Pharm Chem J. 2007;41(7):362–6.
Paquet V, Volmer AA, Carreira EM. Synthesis and in vitro biological properties of novel cationic derivatives of amphotericin B. Chem A Eur J. 2008;14(8):2465–81.
Bastos MM, Hoelz LVB, Boechat N, Oliveira Apd. Antileishmanial Chemotherapy: A Literature Review. Virtual de Quı´mica. 2016;8(6):32.
Cereghetti DM, Carreira E. Amphotericin B: 50 Years of Chemistry and Biochemistry. ChemInform. 2006;37.
Gray KC, Palacios DS, Dailey I, et al. Amphotericin primarily kills yeast by simply binding ergosterol. Proc Natl Acad Sci USA. 2012;109(7):2234–9.
Tevyashova AN, Olsufyeva EN, Solovieva SE, et al. Structure-antifungal activity relationships of polyene antibiotics of the amphotericin B group. Antimicrob Agents Chemother. 2013;57(8):3815–22.
Palacios DS, Dailey I, Siebert DM, Wilcock BC, Burke MD. Synthesis-enabled functional group deletions reveal key underpinnings of amphotericin B ion channel and antifungal activities. Proc Natl Acad Sci USA. 2011;108(17):6733–8.
Adler-Moore J, Proffitt RT. AmBisome: liposomal formulation, structure, mechanism of action and pre-clinical experience. J Antimicrob Chemother. 2002;49(Suppl 1):21–30.
Perkins WR, Minchey SR, Boni LT, et al. Amphotericin B-phospholipid interactions responsible for reduced mammalian cell toxicity. Biochim Biophys Acta. 1992;1107(2):271–82.
Palacios DS, Dailey I, Siebert DM, Wilcock BC, Burke MD. Synthesis-enabled functional group deletions reveal key underpinnings of amphotericin B ion channel and antifungal activities. Proc Natl Acad Sci. 2011;108(17):6733.
Gray KC, Palacios DS, Dailey I, et al. Amphotericin primarily kills yeast by simply binding ergosterol. Proc Natl Acad Sci. 2012;109(7):2234.
Zhang Y-Q, Gamarra S, Garcia-Effron G, Park S, Perlin DS, Rao R. Requirement for ergosterol in V-ATPase function underlies antifungal activity of azole drugs. PLoS Pathog. 2010;6(6):e1000939.
Palacios DS, Anderson TM, Burke MD. A post-PKS Oxidation of the amphotericin B skeleton predicted to be critical for channel formation is not required for potent antifungal activity. J Am Chem Soc. 2007;129(45):13804–5.
Cotero BV, Rebolledo-Antúnez S, Ortega-Blake I. On the role of sterol in the formation of the amphotericin B channel. Biochimica et Biophysica Acta (BBA) - Biomembranes. 1998;1375(1):43–51.
Vertut-Croquin A, Bolard J, Chabbert M, Gary-Bobo C. Differences in the interaction of the polyene antibiotic amphotericin B with cholesterol- or ergosterol-containing phospholipid vesicles. A circular dichroism and permeability study. Biochemistry. 1983;22(12):2939–44.
Haido RMT, Barreto-Bergter E. Amphotericin B-induced damage of Trypanosoma cruzi epimastigotes. Chem Biol Interact. 1989;71(1):91–103.
Sokol-Anderson ML, Brajtburg J, Medoff G. Amphotericin B-induced oxidative damage and killing of Candida albicans. J Infect Dis. 1986;154(1):76–83.
Liu TT, Lee RE, Barker KS, et al. Genome-wide expression profiling of the response to azole, polyene, echinocandin, and pyrimidine antifungal agents in Candida albicans. Antimicrob Agents Chemother. 2005;49(6):2226–36.
Sangalli-Leite F, Scorzoni L, Mesa-Arango AC, et al. Amphotericin B mediates killing in Cryptococcus neoformans through the induction of a strong oxidative burst. Microbes Infect. 2011;13(5):457–67.
Sharma M, Manoharlal R, Negi AS, Prasad R. Synergistic anticandidal activity of pure polyphenol curcumin I in combination with azoles and polyenes generates reactive oxygen species leading to apoptosis. FEMS Yeast Res. 2010;10(5):570–8.
Al-Dhaheri RS, Douglas LJ. Apoptosis in Candida biofilms exposed to amphotericin B. J Med Microbiol. 2010;59(Pt 2):149–57.
Blum G, Perkhofer S, Haas H, et al. Potential basis for amphotericin B resistance in Aspergillus terreus. Antimicrob Agents Chemother. 2008;52(4):1553–5.
Mousavi SAA, Robson GD. Oxidative and amphotericin B-mediated cell death in the opportunistic pathogen Aspergillus fumigatus is associated with an apoptotic-like phenotype. Microbiology (Reading). 2004;150(Pt 6):1937–45.
Phillips AJ, Sudbery I, Ramsdale M. Apoptosis induced by environmental stresses and amphotericin B in Candida albicans. Proc Natl Acad Sci. 2003;100(24):14327.
Brajtburg J, Bolard J. Carrier effects on biological activity of amphotericin B. Clin Microbiol Rev. 1996;9(4):512–31.
Vecchiarelli A, Verducci G, Perito S, Puccetti P, Marconi P, Bistoni F. Involvement of host macrophages in the immunoadjuvant activity of amphotericin B in a mouse fungal infection model. J Antibiot (Tokyo). 1986;39(6):846–55.
Suschek CV, Bonmann E, Kapsokefalou A, et al. Revisiting an old antimicrobial drug: amphotericin B induces interleukin-1-converting enzyme as the main factor for inducible nitric-oxide synthase expression in activated endothelia. Mol Pharmacol. 2002;62(4):936–46.
Shadkchan Y, Keisari Y, Segal E. Cytokines in mice treated with amphotericin B-intralipid. Med Mycol. 2004;42(2):123–8.
Bellocchio S, Gaziano R, Bozza S, et al. Liposomal amphotericin B activates antifungal resistance with reduced toxicity by diverting Toll-like receptor signalling from TLR-2 to TLR-4. J Antimicrob Chemother. 2005;55(2):214–22.
Arthington-Skaggs BA, Motley M, Warnock DW, Morrison CJ. Comparative evaluation of PASCO and national committee for clinical laboratory standards M27-A broth microdilution methods for antifungal drug susceptibility testing of yeasts. J Clin Microbiol. 2000;38(6):2254–60.
Pfaller MA, Arikan S, Lozano-Chiu M, et al. Clinical evaluation of the ASTY colorimetric microdilution panel for antifungal susceptibility testing. J Clin Microbiol. 1998;36(9):2609–12.
Pfaller MA, Bale M, Buschelman B, et al. Quality control guidelines for National Committee for Clinical Laboratory Standards recommended broth macrodilution testing of amphotericin B, fluconazole, and flucytosine. J Clin Microbiol. 1995;33(5):1104–7.
Davey KG, Holmes AD, Johnson EM, Szekely A, Warnock DW. Comparative evaluation of FUNGITEST and broth microdilution methods for antifungal drug susceptibility testing of Candida species and Cryptococcus neoformans. J Clin Microbiol. 1998;36(4):926–30.
Ellis D. Amphotericin B: spectrum and resistance. J Antimicrob Chemother. 2002;49(suppl_1):7–10.
Espinel-Ingroff A. In vitro activity of the new triazole voriconazole (UK-109,496) against opportunistic filamentous and dimorphic fungi and common and emerging yeast pathogens. J Clin Microbiol. 1998;36(1):198–202.
Arikan S, Lozano-Chiu M, Paetznick V, Nangia S, Rex JH. Microdilution susceptibility testing of amphotericin B, itraconazole, and voriconazole against clinical isolates of Aspergillus and Fusarium species. J Clin Microbiol. 1999;37(12):3946–51.
Espinel-Ingroff A, Bartlett M, Bowden R, et al. Multicenter evaluation of proposed standardized procedure for antifungal susceptibility testing of filamentous fungi. J Clin Microbiol. 1997;35(1):139–43.
Wildfeuer A, Seidl HP, Paule I, Haberreiter A. In vitro activity of voriconazole against yeasts, moulds and dermatophytes in comparison with fluconazole, amphotericin B and griseofulvin. Arzneimittelforschung. 1997;47(11):1257–63.
McGinnis MR, Pasarell L, Sutton DA, Fothergill AW, Cooper CR Jr, Rinaldi MG. In vitro activity of voriconazole against selected fungi. Med Mycol. 1998;36(4):239–42.
Johnson EM, Szekely A, Warnock DW. In vitro activity of Syn-2869, a novel triazole agent, against emerging and less common mold pathogens. Antimicrob Agents Chemother. 1999;43(5):1260–3.
Guarro J, Llop C, Aguilar C, Pujol I. Comparison of in vitro antifungal susceptibilities of conidia and hyphae of filamentous fungi. Antimicrob Agents Chemother. 1997;41(12):2760–2.
Aguilar C, Pujol I, Sala J, Guarro J. Antifungal susceptibilities of Paecilomyces species. Antimicrob Agents Chemother. 1998;42(7):1601–4.
Cuenca-Estrella M, Ruiz-Díez B, Martínez-Suárez JV, Monzón A, Rodríguez-Tudela JL. Comparative in-vitro activity of voriconazole (UK-109,496) and six other antifungal agents against clinical isolates of Scedosporium prolificans and Scedosporium apiospermum. J Antimicrob Chemother. 1999;43(1):149–51.
Espinel-Ingroff A, Dawson K, Pfaller M, et al. Comparative and collaborative evaluation of standardization of antifungal susceptibility testing for filamentous fungi. Antimicrob Agents Chemother. 1995;39(2):314–9.
Arendrup MC, Patterson TF. Multidrug-Resistant Candida: Epidemiology, Molecular Mechanisms, and Treatment. J Infect Dis. 2017;216(suppl_3):S445–51.
Falahati M, Nozari S, Makhdoomi A, Ghasemi Z, Nami S, Assadi M. Comparison of antifungal effect of nanosilver particles alone and in combination with current drugs on candida species isolated from women with recurrent vulvovaginal candidiasis. Eur J Exp Biol 2014;4.
Meyerhoff A. U.S. Food and Drug Administration approval of AmBisome (liposomal amphotericin B) for treatment of visceral leishmaniasis. Clin Infect Dis. 1999;28(1):42–8; discussion 49–51.
Solomon M, Pavlotsky F, Leshem E, Ephros M, Trau H, Schwartz E. Liposomal amphotericin B treatment of cutaneous leishmaniasis due to Leishmania tropica. J Eur Acad Dermatol Venereol. 2011;25(8):973–7.
Wortmann G, Zapor M, Ressner R, et al. Lipsosomal amphotericin B for treatment of cutaneous leishmaniasis. Am J Trop Med Hyg. 2010;83(5):1028–33.
Guery R, Henry B, Martin-Blondel G, et al. Liposomal amphotericin B in travelers with cutaneous and muco-cutaneous leishmaniasis: Not a panacea. PLoS Negl Trop Dis. 2017;11(11):e0006094.
Vargas-Zepeda J, Gómez-Alcalá AV, Vásquez-Morales JA, Licea-Amaya L, De Jonckheere JF, Lares-Villa F. Successful treatment of Naegleria fowleri meningoencephalitis by using intravenous amphotericin B, fluconazole and rifampicin. Arch Med Res. 2005;36(1):83–6.
Schuster FL, Visvesvara GS. Opportunistic amoebae: challenges in prophylaxis and treatment. Drug Resist Updat. 2004;7(1):41–51.
Kim H, Kim S-J, Park S-N, Oh J-W. Antiviral effect of amphotericin B on Japanese encephalitis virus replication. J Microbiol Biotechnol. 2004;14(1):121–7.
Jordan GW, Humphreys S, Zee YC. Effect of amphotericin B methyl ester on vesicular stomatitis virus morphology. Antimicrob Agents Chemother. 1978;13(2):340–1.
Konopka K, Guo LS, Düzgüneş N. Anti-HIV activity of amphotericin B-cholesteryl sulfate colloidal dispersion in vitro. Antiviral Res. 1999;42(3):197–209.
Kessler HA, Dixon J, Howard CR, Tsiquaye K, Zuckerman AJ. Effects of amphotericin B on hepatitis B virus. Antimicrob Agents Chemother. 1981;20(6):826–33.
Cowen LE, Sanglard D, Howard SJ, Rogers PD, Perlin DS. Mechanisms of antifungal drug resistance. Cold Spring Harb Perspect Med. 2014;5(7):a019752.
Anderson TM, Clay MC, Cioffi AG, et al. Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat Chem Biol. 2014;10(5):400–6.
White TC, Marr KA, Bowden RA. Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev. 1998;11(2):382–402.
Ben-Ami R, Lewis RE, Kontoyiannis DP. Immunocompromised hosts: immunopharmacology of modern antifungals. Clin Infect Dis. 2008;47(2):226–35.
Bonhomme J, d’Enfert C. Candida albicans biofilms: building a heterogeneous, drug-tolerant environment. Curr Opin Microbiol. 2013;16(4):398–403.
Ramage G, Mowat E, Jones B, Williams C, Lopez-Ribot J. Our current understanding of fungal biofilms. Crit Rev Microbiol. 2009;35(4):340–55.
Sanglard D, Ischer F, Parkinson T, Falconer D, Bille J. Candida albicans mutations in the ergosterol biosynthetic pathway and resistance to several antifungal agents. Antimicrob Agents Chemother. 2003;47(8):2404–12.
Kelly SL, Lamb DC, Kelly DE, Loeffler J, Einsele H. Resistance to fluconazole and amphotericin in Candida albicans from AIDS patients. Lancet. 1996;348(9040):1523–4.
Haynes MP, Chong PL, Buckley HR, Pieringer RA. Fluorescence studies on the molecular action of amphotericin B on susceptible and resistant fungal cells. Biochemistry. 1996;35(24):7983–92.
Baillie GS, Douglas LJ. Effect of growth rate on resistance of Candida albicans biofilms to antifungal agents. Antimicrob Agents Chemother. 1998;42(8):1900–5.
Pourshafie M, Morand S, Virion A, Rakotomanga M, Dupuy C, Loiseau PM. Cloning of S-adenosyl-L-methionine:C-24-Delta-sterolmethyltransferase (ERG6) from Leishmania donovani and characterization of mRNAs in wild-type and amphotericin B-Resistant promastigotes. Antimicrob Agents Chemother. 2004;48(7):2409–14.
Dick JD, Merz WG, Saral R. Incidence of polyene-resistant yeasts recovered from clinical specimens. Antimicrob Agents Chemother. 1980;18(1):158–63.
Sokol-Anderson M, Sligh JE Jr, Elberg S, Brajtburg J, Kobayashi GS, Medoff G. Role of cell defense against oxidative damage in the resistance of Candida albicans to the killing effect of amphotericin B. Antimicrob Agents Chemother. 1988;32(5):702–5.
Blum G, Hörtnagl C, Jukic E, et al. New insight into amphotericin B resistance in Aspergillus terreus. Antimicrob Agents Chemother. 2013;57(4):1583–8.
Vahedi Shahandashti R, Lass-Flörl C. Antifungal resistance in Aspergillus terreus: A current scenario. Fungal Genet Biol. 2019;131:103247.
Broughton MC, Bard M, Lees ND. Polyene resistance in ergosterol producing strains of Candida albicans. Mycoses. 1991;34(1–2):75–83.
Pierce AM, Pierce HD Jr, Unrau AM, Oehlschlager AC. Lipid composition and polyene antibiotic resistance of Candida albicans mutants. Can J Biochem. 1978;56(2):135–42.
Seo K, Akiyoshi H, Ohnishi Y. Alteration of cell wall composition leads to amphotericin B resistance in Aspergillus flavus. Microbiol Immunol. 1999;43(11):1017–25.
Bahmed K, Bonaly R, Coulon J. Relation between cell wall chitin content and susceptibility to amphotericin B in Kluyveromyces, Candida and Schizosaccharomyces species. Res Microbiol. 2003;154(3):215–22.
Bahmed K, Bonaly R, Wathier M, Pucci B, Coulon J. Change of cell wall chitin content in amphotericin B resistant Kluyveromyces strains. FEMS Microbiol Lett. 2002;216(1):99–103.
Hammond SM, Kliger BN. Differential effects of monovalent and divalent ions upon the mode of action of the polyene antibiotic Candicidin. J Appl Bacteriol. 1976;41(1):59–68.
Mesa-Arango AC, Rueda C, Román E, et al. Cell wall changes in amphotericin B-resistant strains from Candida tropicalis and relationship with the immune responses elicited by the host. Antimicrob Agents Chemother. 2016;60(4):2326–35.
Gaber RF, Copple DM, Kennedy BK, Vidal M, Bard M. The yeast gene ERG6 is required for normal membrane function but is not essential for biosynthesis of the cell-cycle-sparking sterol. Mol Cell Biol. 1989;9(8):3447–56.
Gale EF, Ingram J, Kerridge D, Notario V, Wayman F. Reduction of amphotericin resistance in stationary phase cultures of Candida albicans by treatment with enzymes. J Gen Microbiol. 1980;117(2):383–91.
Kelly SL, Lamb DC, Taylor M, Corran AJ, Baldwin BC, Powderly WG. Resistance to amphotericin B associated with defective sterol delta 8–>7 isomerase in a Cryptococcus neoformans strain from an AIDS patient. FEMS Microbiol Lett. 1994;122(1–2):39–42.
Powderly WG, Kobayashi GS, Herzig GP, Medoff G. Amphotericin B-resistant yeast infection in severely immunocompromised patients. Am J Med. 1988;84(5):826–32.
Colombo AL, Melo AS, Crespo Rosas RF, et al. Outbreak of Candida rugosa candidemia: an emerging pathogen that may be refractory to amphotericin B therapy. Diagn Microbiol Infect Dis. 2003;46(4):253–7.
Krcmery V Jr, Oravcova E, Spanik S, et al. Nosocomial breakthrough fungaemia during antifungal prophylaxis or empirical antifungal therapy in 41 cancer patients receiving antineoplastic chemotherapy: analysis of aetiology risk factors and outcome. J Antimicrob Chemother. 1998;41(3):373–80.
Nolte FS, Parkinson T, Falconer DJ, et al. Isolation and characterization of fluconazole- and amphotericin B-resistant Candida albicans from blood of two patients with leukemia. Antimicrob Agents Chemother. 1997;41(1):196–9.
Hollister LE. AMA Drug Evaluations Annual 1991. JAMA. 1991;266(3):42.
World Health Organization. Pharmaceuticals, U. Accelerated stability studies of widely used pharmaceutical substances under simulated tropical conditions. 1986 [Geneva]: World Health Organization. World Health Organization: Geneva.
National Toxicology Program, Amphotericin B, in Reactivity profile I.o.E.H. Sciences, Editor. 1992, National Institutes of Health North Carolina.
Montenegro MB, Souza SPd, Leão RAC, Rocha HVA, Rezende CMd, Souza ROMAd. Methodology Development and Validation of Amphotericin B Stability by HPLC-DAD. J Braz Chem Soc. 2020;31: 916–26.
Hung CT, Lam FC, Perrier DG, Souter A. A stability study of amphotericin B in aqueous media using factorial design. Int J Pharm. 1988;44(1):117–23.
Wiest DB, Maish WA, Garner SS, el-Chaar GM. Stability of amphotericin B in four concentrations of dextrose injection. Am J Hosp Pharm. 1991;48(11):2430–3.
Cifani C, Costantino S, Massi M, Berrino L. Commercially available lipid formulations of amphotericin b: are they bioequivalent and therapeutically equivalent? Acta Biomed. 2012;83(2):154–63.
TEVA Pharmaceuticals Europe B.V, Package leaflet: Information for the user - Abelcet® Lipid Complex 5 mg/ml concentrate for dispersion for infusion, T. Pharmaceuticals, Editor. 2020:The Netherlands.
Anaissie E, Paetznick V, Proffitt R, Adler-Moore J, Bodey GP. Comparison of the in vitro antifungal activity of free and liposomeencapsulated amphotericin B. Eur J Clin Microbiol Infect Dis. 1991;10(8):665–8.
Adler-Moore JP, Chiang SM, Satorius A, et al. Treatment of murine candidosis and cryptococcosis with a unilamellar liposomal amphotericin B formulation (AmBisome). J Antimicrob Chemother. 1991;28 Suppl B:63–71.
Patterson TF, Thompson GR III, Denning DW, et al. Practice Guidelines for the Diagnosis and Management of Aspergillosis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;63(4):e1–60.
Ullmann AJ, Aguado JM, Arikan-Akdagli S, et al. Diagnosis and management of Aspergillus diseases: executive summary of the 2017 ESCMIDECMM-ERS guideline. Clin Microbi Infect. 2018;24:e1–38.
Jean E-C, Roberto C, Sabine F et al. Micafungin Versus Fluconazole Or Itraconazole For Prophylaxis Against Invasive Fungal Infections During Neutropenia In Patients Undergoing Haplo-Identical Hematopoietic Stem Cell Transplantation. Blood. 2013;122(21):4564.
Mattiuzzi GN, Kantarjian H, Faderl S, et al. Amphotericin B lipid complex as prophylaxis of invasive fungal infections in patients with acute myelogenous leukemia and myelodysplastic syndrome undergoing induction chemotherapy. Cancer. 2004;100(3):581–9.
Oren I, Rowe JM, Sprecher H, et al. A prospective randomized trial of itraconazole vs fluconazole for the prevention of fungal infections in patients with acute leukemia and hematopoietic stem cell transplant recipients. Bone Marrow Transplant. 2006;38(2):127–34.
Park S, Kim K, Jang JH, et al. Randomized trial of micafungin versus fluconazole as prophylaxis against invasive fungal infections in hematopoietic stem cell transplant recipients. J Infect. 2016;73(5):496–505.
Ullmann AJ, Sanz MA, Tramarin A, et al. Prospective study of amphotericin B formulations in immunocompromised patients in 4 European countries. Clin Infect Dis. 2006;43(4):e29–38.
Wingard JR, Carter SL, Walsh TJ, et al. Randomized, double-blind trial of fluconazole versus voriconazole for prevention of invasive fungal infection after allogeneic hematopoietic cell transplantation. Blood. 2010;116(24):5111–8.
Ullmann AJ, Lipton JH, Vesole DH, et al. Posaconazole or fluconazole for prophylaxis in severe graft-versus-host disease. N Engl J Med. 2007;356(4):335–47.
Eliashar R, Resnick IB, Goldfarb A, Wohlgelernter J, Gross M. Endoscopic surgery for sinonasal invasive aspergillosis in bone marrow transplantation patients. Laryngoscope. 2007;117(1):78–81.
Martino R, Parody R, Fukuda T, et al. Impact of the intensity of the pretransplantation conditioning regimen in patients with prior invasive aspergillosis undergoing allogeneic hematopoietic stem cell transplantation: a retrospective survey of the Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Blood. 2006;108(9):2928–36.
Huang X, Chen H, Han M, et al. Multicenter, randomized, open-label study comparing the efficacy and safety of micafungin versus itraconazole for prophylaxis of invasive fungal infections in patients undergoing hematopoietic stem cell transplant. Biol Blood Marrow Transplant. 2012;18(10):1509–16.
Oppenheim BA, Herbrecht R, Kusne S. The safety and efficacy of amphotericin B colloidal dispersion in the treatment of invasive mycoses. Clin Infect Dis. 1995;21(5):1145–53.
Denning DW, Marr KA, Lau WM, et al. Micafungin (FK463), alone or in combination with other systemic antifungal agents, for the treatment of acute invasive aspergillosis. J Infect. 2006;53(5):337–49.
Herbrecht R, Letscher V, Andres E, Cavalier A. Safety and efficacy of amphotericin B colloidal dispersion. An overview. Chemotherapy. 1999;45(Suppl 1):67–76.
Carrillo-Muñoz AJ, Finquelievich J, Tur-Tur C, et al. Combination antifungal therapy: a strategy for the management of invasive fungal infections. Rev Esp Quimioter. 2014;27(3):141–58.
Hatipoglu N, Hatipoglu H. Combination antifungal therapy for invasive fungal infections in children and adults. Expert Rev Anti Infect Ther. 2013;11(5):523–35.
Kontoyiannis DP, Lewis RE. Toward more effective antifungal therapy: the prospects of combination therapy. Br J Haematol. 2004;126(2):165–75.
Panackal AA, Parisini E, Proschan M. Salvage combination antifungal therapy for acute invasive aspergillosis may improve outcomes: a systematic review and meta-analysis. Int J Infect Dis. 2014;28:80–94.
Mihu CN, Kassis C, Ramos ER, Jiang Y, Hachem RY, Raad II. Does combination of lipid formulation of amphotericin B and echinocandins improve outcome of invasive aspergillosis in hematological malignancy patients? Cancer. 2010;116(22):5290–6.
Rex JH, Pappas PG, Karchmer AW, et al. A randomized and blinded multicenter trial of high-dose fluconazole plus placebo versus fluconazole plus amphotericin B as therapy for candidemia and its consequences in nonneutropenic subjects. Clin Infect Dis. 2003;36(10):1221–8.
Perfect JR, Dismukes WE, Dromer F, et al. Clinical Practice Guidelines for the Management of Cryptococcal Disease: 2010 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2010;50(3):291–322.
Forrest GN, Bhalla P, DeBess EE, et al. Cryptococcus gattii infection in solid organ transplant recipients: description of Oregon outbreak cases. Transpl Infect Dis. 2015;17(3):467–76.
Migone C, Ford N, Garner P, Eshun-Wilson I. Updating guidance for preventing and treating cryptococcal disease: how evidence and decisions interface. Cochrane Database Syst Rev. 2018;11:Ed000130.
Serrano DR, Ballesteros MP, Schätzlein AG, Torrado JJ, Uchegbu IF. Amphotericin B formulations—the possibility of generic competition. Pharm Nanotechnol. 2013;1(4):8.
Van Daele R, Spriet I, Wauters J et al. Antifungal drugs: What brings the future? Med Mycol. 2019;57(Supplement_3):S328–43.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Francelise Bridi Cavassin, João Luiz Baú Carneiro, Rogério Rodrigues Vilas Boas and Flávio de Queiroz-Telles declare that they have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Cavassin, F.B., Baú-Carneiro, J.L., Vilas-Boas, R.R. et al. Sixty years of Amphotericin B: An Overview of the Main Antifungal Agent Used to Treat Invasive Fungal Infections. Infect Dis Ther 10, 115–147 (2021). https://doi.org/10.1007/s40121-020-00382-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-020-00382-7




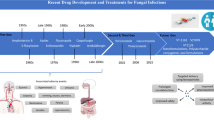



 Amphotericin B
Amphotericin B  Phospholipid
Phospholipid
