Abstract
Introduction
Physicians need an accurate understanding of diabetic retinopathy (DR) severity to optimally manage patients. The aim of this prospective study is to correlate the severity of macular and peripheral retinal vascular abnormalities seen on widefield (WF) optical coherence tomography angiography (OCTA) with DR grading based on WF fundus photography.
Methods
The study included 150 eyes from 82 patients with treatment-naïve DR. All patients were imaged with WF fundus photography and swept-source WF OCTA. Quantitative and qualitative analyses of the foveal avascular zone (FAZ) size and shape, and measurement of capillary nonperfusion (CNP) areas, were performed from the OCTA images. The mixed-effects model was used to compare the DR grading from WF photography with the vascular changes seen on WF-OCTA, and Bonferroni correction was applied to the gradings.
Results
The mean [± standard deviation (SD)] age of patients was 55.5 (± 9.4) years. The WF-OCTA showed that an increasing size of the FAZ (from 0.442 (± 0.059) µm to 0.933 (± 0.086) µm) correlated with increasing severity of the DR (as determined with WF photography). The deep capillary plexus, FAZ size, and CNP areas in eyes with proliferative diabetic retinopathy (PDR) differed from those with mild nonproliferative diabetic retinopathy (NPDR) (p < 0.001). Most eyes with severe nonproliferative DR were found to have CNP in four quadrants [superficial capillary plexus (SCP) 60%, deep capillary plexus (DCP) 50%]. The WF-OCTA detected subtle neovascularization of the disc (NVD) in 7 eyes (10%) and neovascularization elsewhere (NVE) in 13 eyes (18%) that had been diagnosed with only moderate NPDR on WF photography.
Conclusions
FAZ and CNP areas as measured by WF-OCTA correlate with DR severity. WF-OCTA can also detect subtle NVE and NVD that cannot be seen with fundus photography.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The study provides insights into the quantitative and qualitative metrics derived from widefield optical coherence tomography angiography (WF-OCTA) across various stages of diabetic retinopathy (DR). |
It establishes a correlation between the severity of DR, as determined by widefield (WF) color fundus photography, and capillary perfusion measurements obtained from WF-OCTA. |
Measuring foveal avascular zone size and capillary nonperfusion areas at the levels of the superficial and deep capillary plexuses can predict the stage of DR. |
Neovascularization elsewhere and neovascularization of the disc that cannot be seen on WF fundus photography can often be seen with WF-OCTA. |
Introduction
Optimal management of patients with diabetic retinopathy (DR) requires that physicians be able to accurately characterize the disease severity. Several grading systems are used to classify DR, including the widely used Early Treatment of Diabetic Retinopathy severity scale, and most rely on the evaluation of seven-field color fundus photography [1]. This method has its limitations, including difficulty in differentiating between severe nonproliferative diabetic retinopathy (NPDR) and early proliferative diabetic retinopathy (PDR), particularly in the presence of media opacities, such as cataracts, hemorrhage, corneal scarring, and asteroid hyalosis. When confronted with photographs that are difficult to read, physicians frequently use fundus fluorescein angiography (FFA), an invasive imaging modality that is better equipped to identify neovascularization and that provides more gradable images when photographed through media opacities [2, 3].
Optical coherence tomography angiography (OCTA) is a noninvasive imaging technique that identifies retinal circulatory patterns as well as retinal and choroidal neovascularization without the need for intravenous dye. While FFA produces a two-dimensional image of the fundus circulation without differentiating between superficial and deep capillary plexuses, OCTA produces a highly detailed three-dimensional image that extends from the inner retinal surface to the choriocapillaris. Unlike FFA, OCTA can differentiate between the superficial and deep retinal capillary networks, and it is superior to FFA for detecting sight-threatening changes, such as foveal avascular zone (FAZ) distortion, areas of capillary nonperfusion (CNP), macular ischemia, and neovascularization [4,5,6]. The limitations of conventional OCTA include imaging artifacts, the learning curve of the operator, the cooperation of the patient, and a limited field of view (3 × 3 mm, 6 × 6 mm, or 9 × 9 mm) with conventional scanners [6, 7].
To classify DR severity (consistent with what is seen on fundus photographs) using only macular circulatory findings, Wong et al. formulated an OCTA-based classification that used only 3 × 3 mm macular scans [6]. They showed that distortion of the FAZ on the OCTA scans correlated with progression of DR from mild to severe stages. The limited field of view used by this classification system, however, fails to incorporate important mid-peripheral and peripheral diabetic pathologies, such as the extent of CNP and neovascularization of the disc (NVD) and elsewhere (NVE).
Recently, a 12 × 12 mm widefield (WF) OCTA scan protocol was introduced, and the montage option can detect and quantify areas of peripheral CNP and retinal neovascularization [5, 8,9,10,11,12]. A small case series demonstrated that the WF-OCTA can identify peripheral DR lesions, but no large case series has used OCTA-derived quantitative and qualitative perfusion metrics to predict the severity of DR and compare these with assessments made with standard color fundus photography.
Our prospective study aims to correlate the severity of DR as determined by the quantitative and qualitative characteristics of WF-OCTA with grading made with WF fundus color photography.
Methods
A prospective study of consecutive patients presenting with treatment-naïve DR was performed at the LV Prasad Eye Institute. The study was approved by the local ethics committee (LEC 10–19-352), and it adhered to the tenets of the Declaration of Helsinki. Written informed consent was obtained from each participant prior to participation in the study.
Eligibility
Patients 18 years of age or older with treatment-naïve DR of any stage were included in the study. Pregnant women and patients with opacified media that precluded detailed fundus imaging, poor cooperation during imaging, poor scan quality (motion artifact score > 2, signal strength < 8), and decreased vision that interfered with fixation were excluded from the study.
Examination and Image Acquisition
All patients underwent a complete ophthalmologic examination, including measurement of best-corrected visual acuity (BCVA) and intraocular pressures (IOP), and examination with slit lamp biomicroscopy and fundoscopy. Fundus and OCTA images were acquired using WF fundus photography (CLARUS 500™, Carl Zeiss Meditec Inc., California, USA) and WF swept-source OCTA (Plex Elite 9000; Carl Zeiss Meditec). Sectioning of the OCTA into deep and superficial slabs was automated. The sections were verified by two research assistants for accuracy.Fundus photographs with DR and DME were classified according to the International Clinical DR Classification severity scale by two certified research assistants [13]. Any disagreement between the two graders was resolved by a fellowship-trained retina consultant.
Sample Size
We included 150 eyes from 82 patients with treatment-naïve diabetic retinopathy (DR) who met the study inclusion criteria. Based on a power analysis with a medium effect size (f = 0.25), a significance level (α) of 0.05, and a power (1 − β) of 0.80, the estimated required sample size was approximately 128 eyes. The study, conducted over 1 year, targeted 150 eyes to ensure statistical power, and provide reliable results for the study’s objectives.
OCTA
WF-OCTA images were analyzed quantitatively and qualitatively. Quantitative analyses of peripheral CNP (based on 12 × 12 mm montages) were performed manually with ImageJ software (National Institutes of Health, Bethesda, Maryland, USA) by two clinicians and two research assistants, and were adjusted for interobserver errors. FAZ area was analyzed manually using 3 × 3 mm scans, since these provided better resolution.
The severity of any FAZ abnormality was assessed qualitatively. Two observers used 3 × 3 mm scans to grade the FAZ, according to the technique by Bradley et al. with minor modifications.[14] Borders of the FAZ were graded as 0 (no disruption of the FAZ), 1 (outline is not smooth or oval, but changes are not clearly pathologic), 2 (outline of the FAZ is disrupted in two or fewer quadrants), or 3 (outline of the FAZ disrupted in three or more quadrants).
CNP of the superficial capillary plexus (SCP) and deep capillary plexus (DCP) within the macula was graded qualitatively using 3 × 3 mm scans. The scans were divided into four equal-sized squares, each of which was considered a quadrant. The CNP was graded as 0 (no CNP), 1 (one quadrant with CNP), 2 (two quadrants with CNP), 3 (three quadrants with CNP), and 4 (four quadrants with CNP). Interobserver agreement was validated using the kappa value.
Statistical Analyses
The statistical analyses were performed with the software STATA v14.2 (StataCorp, College Station, TX, USA). A mixed-effects model using maximum likelihood estimation with random intercept at the level of the participant (right/left eye) was used to compare the various grades of DR. A p-value of < 0.05 was considered statistically significant. For pair-wise comparisons among gradings, Bonferroni correction was applied, and a p-value of < 0.017 was considered statistically significant. Interobserver agreement was validated using Cohen’s kappa coefficient.
Results
Demographics and Baseline Data
The study included 150 eyes from 82 participants. The mean age (± standard deviation) of the participants was 55.5 (± 9.4) years; 59 were male and 23 were female. The mean duration of DM was 10 years [interquartile range (IQR) 6–16.5 years], and 48 of 82 (58.5%) patients had coexisting systemic arterial hypertension. The mean BCVA was 0.33 (± 0.04) logMAR.
Based on grading of the fundus photography, eyes were divided into the following four severity groups: mild NPDR, moderate NPDR, severe NPDR, and PDR. Table 1 presents the proportions of participants with each DR severity as determined by assessments of WF fundus photography, and the corresponding macular and peripheral retinal vascular measurements obtained with WF-OCTA.
Macula OCT Analysis
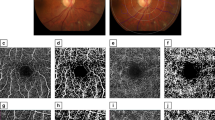
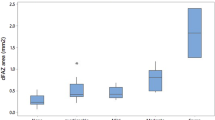
The DR severity grade correlated with the size of the FAZ, whether measured at the level of SCP (p = 0.07) or DCP (p = 0.01) (Fig. 1). The mean size of the FAZ (in µm) at the DCP level was greater than at the SCP level for each subgroup. After adjustment for the Bonferroni correction, multiple pair-wise comparisons showed that the size of the FAZ in the DCP of the PDR group was significantly different from the mild NPDR (p = 0.004) and moderate NPDR (p = 0.01) groups.
The figure shows the increasing size of the foveal avascular zone and capillary nonperfusion areas with an increase in the severity of diabetic retinopathy. a mild nonproliferative diabetic retinopathy (NPDR), b moderate NPDR, c severe NPDR, d proliferative diabetic retinopathy (PDR), e mild NPDR, f moderate NPDR, g severe NPDR, h PDR
According to the fundus photographs, eyes were categorized into four subgroups: no DME, mild DME, moderate DME, and severe DME. Increasing sizes of the FAZ were seen with increase in severity of DR in all the groups at both SCP and DCP levels (Table 1). The total CNP area increased in relation to the severity of DME in all groups; however, multiple pair-wise comparison after Bonferroni correction of the combinations (total CNP area versus severity of DME) was not significantly different (Table 2).
Normal qualitative FAZ morphology (grades 0 and 1) was observed in 55 (53%) eyes at the SCP level and 39 (43%) eyes at the DCP level. Most eyes with mild or moderate DR had normal FAZ morphology [SCP level 41/55 (75%), DCP level 35/42 (83%)] (Fig. 2). Abnormal FAZ morphology was observed at the SCP level in 27/52 (52%) eyes with moderate NPDR, 9/12 (75%) eyes with severe NPDR, and 7/18 (39%) eyes with PDR, and at the DCP level in 28/50 (56%) eyes with moderate NPDR, 10/11(91%) eyes with severe NPDR, and 12/18 (66%) eyes with PDR. Four-quadrant CNP was seen in most eyes with severe NPDR at the SCP (60%) and DCP levels (50%) (Table 3).
Qualitative analysis: The 3 × 3 mm central scan was divided into four equal-sized squares with a foveal avascular zone (FAZ) at the center; each square box was considered a quadrant. An outline of the FAZ border and capillary nonperfusion (CNP) areas around the FAZ were analyzed. This is a case of mild NPDR (left image, a) that showed a grade 0 FAZ border (no disruption of the FAZ) and grade 0 CNP areas (no CNP) around the FAZ. A case of severe NPDR (right image, b) shows a grade 3 FAZ border (outline of the FAZ disrupted in three or more quadrants) and grade 4 CNP areas (four quadrant CNP: red arrows) around the FAZ
Widefield-OCT Angiography Analysis
Higher grades of DR were generally found to have larger areas of CNP (p < 0.001) (Fig. 1), but the mean CNP area of the moderate NPDR group was larger than the severe NPDR group (Table 1). After adjusting for the Bonferroni correction, multiple pair-wise comparisons showed that the CNP area in the PDR group was significantly larger than that in the mild NPDR (p < 0.001), moderate NPDR (p = 0.008), and severe NPDR (p = 0.017) groups. Moreover, the mean CNP area in the moderate NPDR group was significantly larger than in the mild NPDR group (p < 0.001).
WF-OCTA detected subtle NVE and NVD in some eyes that were not seen with fundus photography. NVE was found with WF-OCTA in 13 eyes (18%) that had been graded as moderate NPDR on fundus photography (Fig. 3), and NVD was noted in 7 eyes (10%) that had been graded as moderate NPDR. The presence of NVD correlated significantly with the area of CNP (p = 0.04). The median CNP area in eyes with (n = 17) and without NVD (n = 133) was 29.87 mm2 (IQR 16.70–31.80) and 14.74 mm2 (IQR 0–30.50), respectively.
A case of moderate nonproliferative diabetic retinopathy (NPDR) based on fundus photography (a and c), corresponding widefield optical coherence tomography angiography images showing neovascularization elsewhere (white circles in b and d) and capillary nonperfusion areas (b), which establishes the diagnosis of proliferative diabetic retinopathy
The distance BCVA decreased in conjunction with increases in FAZ size and CNP area (from 0.13 ± 0.08 logMAR in the mild DR group to 0.66 ± 0.07 logMAR in the PDR group (Table 1). After adjusting for the Bonferroni correction, multiple pair-wise comparisons showed that the BCVA in the PDR group was significantly worse than in the mild NPDR (p < 0.001), moderate NPDR (p < 0.001), and severe NPDR (p < 0.001) groups (Table 1).
Intergrader Variability
Excellent agreement was seen between observers [the SCP–FAZ borders grading kappa (k) 0.78, standard error (SE) 0.10; DCP–FAZ border grading k 84, SE 0.09; grading of CNP around FAZ at SCP level k 0.88, SE 0.06; grading of CNP around FAZ at DCP level k 0.920, SE 0.04].
Discussion
Our study found that several WF-OCTA observations and measurements of the retinal microvascular correlate with DR severity as determined by fundus photography. The size of the FAZ at the DCP level was larger than at the SCP level in all grades of DR, and the size of the FAZ increased with the severity of DR (Fig. 1). Additionally, WF-OCTA is better able to diagnose subtle NVD and NVE.
Rodrigues et al. found that the size of the FAZ increased with the severity of NPDR, and they correlated the size of the FAZ with the degree of macular ischemia (not contiguous with the FAZ) [15]. Our study strengthens their findings and adds supporting results from the PDR cases. Lu et al. created an automated algorithm to measure the FAZ using OCTA to classify the severity of DR and predict BCVA [16]. Our study supported these findings by showing that a decline in BCVA correlated with an increase in FAZ size, and by demonstrating that macular ischemia correlates with both increasing grade of DR and decreasing BCVA.
Louis et al. correlated visual acuity and macular ischemia with FAZ size and the circularity index. They concluded that the circularity index of the FAZ was the most sensitive indicator of ischemia and proposed that DCP abnormalities were associated with increasing grades of DR [17]. Our study also showed that DCP abnormalities are the earliest and most sensitive indicators (compared with SCP) of advanced DR, but unlike Louis et al., our study showed that FAZ area and circularity index are not good indicators for visual acuity changes.
Several studies have shown that OCTA can replace FFA in suitable cases. Bradley et al. showed a moderate agreement between OCTA and FFA in identifying diabetic macular ischemia [14]. We found an excellent agreement between graders when qualitatively assessing the borders of FAZ and the areas of CNP around the FAZ in 3 × 3 mm OCTA scans. Most eyes with moderate NPDR had disrupted FAZs, and larger CNP areas were seen in eyes with severe NPDR. Our study did not compare FFA with OCTA because previous studies have shown the superiority of OCTA over FFA in assessing macular circulations [8,9,10,11,12]. Our findings suggest, however, that eyes with moderate NPDR on fundus photography should have their FAZs evaluated by angiography (either OCTA or FFA) because many will have FAZ disruption and macular ischemia that may decrease BCVA and increase the risk of peripheral NV.
The value of WF-OCTA and ultra-widefield (UWF) OCTA in assessing peripheral ischemia in DR have been compared previously. A wider field of view (from 12 × 12 mm to 21 × 21 mm) decreases the resolution of the images [9, 18,19,20,21], but the montage protocol with 12 × 12 mm scans significantly broadens the field and enables an analysis of the peripheral retina with good resolution of the capillary layers [18]. In this study, we used WF-OCTA (12 × 12 mm) with the montage protocol to assess both the macular and peripheral perfusions. Tan et al. showed that WF-OCTA effectively detects capillary dropout areas in eyes with NPDR, but our study also included eyes with PDR. We also found that eyes with moderate NPDR had larger CNP areas (16.564 ± 1.678 mm2) compared with eyes with severe NPDR (14.481 ± 3.271 mm2). This could have been due to the limited PDR cohort size, but it highlights the fact that significant portions of nonperfusion areas are seen in moderate NPDR and suggests that these eyes should be observed for the development of PDR.
In our study, WF-OCTA detected subtle NVD in 7 (10%) eyes and subtle NVE in 13 (18%) eyes that had been diagnosed as moderate NPDR on fundus photography. Pichi et al. showed that WF-OCTA [compared with ultra-widefield color fundus photography (UWF-CP)] can identify NVE and NVD with comparable diagnostic accuracy in eyes with PDR. WF-OCTA is a faster and safer alternative to ultra-widefield fundus fluorescein angiography (UWF-FA). Similarly to our findings, Belenje et al. showed that WF-OCTA can detect NV that goes undetected during clinical examinations [18, 22].
Wang et al. used UWF-OCTA to classify DR by calculating the ratio of the areas of CNP and capillary perfusion. They concluded that the ratios were higher in severe grades of DR, and higher ratios were found in peripheral sectors on the WF-OCTA than in central sectors [23]. Our study found that WF-OCT evaluations of CNP in both posterior and peripheral correlate with the severity of DR, though some eyes will have normal FAZs and no CNP despite having severe NPDR and PDR. This suggests that peripheral examinations should be done carefully, even when macular pathology is not seen, because these eyes are at risk of vitreous hemorrhages from undiagnosed peripheral NVE.
FAZ abnormalities on OCTA are associated with macular ischemia, but they also correlate with peripheral ischemia. This association has been studied extensively in branch vein occlusions but infrequently in DR [24,25,26,27]. Capone et al. and Rabiolo et al. showed that peripheral and macular nonperfusion might share a common pathogenic mechanism, thereby suggesting an association between peripheral CNP and FAZ abnormalities [25, 26]. We correlated the size of the FAZ with macular and peripheral nonperfusion, and showed that a serial increase in FAZ size correlates with the extent of nonperfusion.
Diabetic macular edema has been correlated with peripheral ischemia, probably because peripheral ischemia upregulates vascular endothelial growth factor (VEGF), breaks down the blood–retinal barrier, and produces edema [28, 29]. The role of laser treatment to peripheral ischemic areas has been studied, but targeted treatment has not been shown to affect DME [30,31,32]. We found that higher severity of DME is associated with larger areas of nonperfusion, but the association was not statistically significant.
The strengths of our study include the prospective design, exclusion of previously treated eyes, subgroup classification of DR grades using fundus photography, assessment of the FAZ at both the SCP and DCP levels, detection of NVD and NVE with WF-OCTA, subgroup correlation of FAZ parameters with peripheral CNP areas and BCVA, and the qualitative assessment of FAZ morphology and distribution of CNP areas around the FAZ. The limitations of the study include the unequal number of eyes in each DR severity group and the lack of FFA data to correlate with OCTA findings.
Conclusions
Our study highlights the ability of WF-OCTA to identify macular and peripheral retinal vascular changes that correlate with DR severity. We have shown that the size and shape of the FAZ and the amount of macular nonperfusion correlates with overall retinal ischemia. Future studies are needed to validate the correlation of posterior pole vascular abnormalities with peripheral perfusion and neovascularization, and to better determine the relationship between DME severity and peripheral retinal findings. In addition, future studies are also necessary to investigate the effects of anti-VEGF and laser photocoagulation therapies on FAZ parameters and CNP areas. Our WF-OCTA study does not provide sufficient data to accurately predict DR severity (as is done with fundus photography), but additional confirmatory data from future studies may enable the creation of an OCTA-based classification system that is as robust (if not more so) than can currently be done with fundus photography.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Group ETDRSR. Grading diabetic retinopathy from stereoscopic color fundus photographs—an extension of the modified Airlie House classification: ETDRS report number 10. Ophthalmology. 1991;98(5):786–806.
Tavakoli M, Shahri RP, Pourreza H, Mehdizadeh A, Banaee T, Toosi MHB. A complementary method for automated detection of microaneurysms in fluorescein angiography fundus images to assess diabetic retinopathy. Pattern Recogn. 2013;46(10):2740–53.
Ding X, Xiao-hu Z, Zhe S, Qing-yu L, Fang W. Evaluating fundus photography and fundus fluorescein angiography in the grading of diabetic retinopathy. Chin J Exp Ophthalmol. 2013;31(8):779–82.
Arya M, Sorour O, Chaudhri J, Alibhai Y, Waheed NK, Duker JS, Baumal CR. Distinguishing intraretinal microvascular abnormalities from retinal neovascularization using optical coherence tomography angiography. Retina. 2019;40:1686–95.
Mastropasqua R, D’Aloisio R, Di Antonio L, Erroi E, Borrelli E, Evangelista F, et al. Widefield optical coherence tomography angiography in diabetic retinopathy. Acta Diabetol. 2019. https://doi.org/10.1007/s00592-019-01410-w.
Ting DSW, Tan GSW, Agrawal R, Yanagi Y, Sie NM, Wong CW, et al. Optical coherence tomographic angiography in type 2 diabetes and diabetic retinopathy. JAMA Ophthalmol. 2017;135(4):306–12.
Rani PK, Prajapati RC. Role of OCT angiography in the detection of retinal vascular and macular abnormalities in subjects with asteroid hyalosis. Semin Ophthalmol. 2018. https://doi.org/10.1080/08820538.2018.1551497.
Pellegrini M, Cozzi M, Staurenghi G, Corvi F. Comparison of wide field optical coherence tomography angiography with extended field imaging and fluorescein angiography in retinal vascular disorders. PLoS ONE. 2019;14(4): e0214892.
Khalid H, Schwartz R, Nicholson L, Huemer J, El-Bradey MH, Sim DA, et al. Widefield optical coherence tomography angiography for early detection and objective evaluation of proliferative diabetic retinopathy. Br J Ophthalmol. 2020. https://doi.org/10.1136/bjophthalmol-2019-315365.
Mastropasqua R, D’Aloisio R, Di Antonio L, Erroi E, Borrelli E, Evangelista F, et al. Widefield optical coherence tomography angiography in diabetic retinopathy. Acta Diabetol. 2019;56(12):1293–303.
Motulsky EH, Liu G, Shi Y, Zheng F, Flynn HW, Gregori G, Rosenfeld PJ. Widefield swept-source optical coherence tomography angiography of proliferative diabetic retinopathy. Ophthalmic Surg Lasers Imaging Retina. 2019;50(8):474–84.
Eastline M, Munk MR, Wolf S, Schaal KB, Ebneter A, Tian M, et al. Repeatability of wide-field optical coherence tomography angiography in normal retina. Trans Vis Sci Technol. 2019;8(3):6.
Wilkinson C, Ferris FL III, Klein RE, Lee PP, Agardh CD, Davis M, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology. 2003;110(9):1677–82.
Bradley PD, Sim DA, Keane PA, Cardoso J, Agrawal R, Tufail A, Egan CA. The evaluation of diabetic macular ischemia using optical coherence tomography angiography. Invest Ophthalmol Vis Sci. 2016;57(2):626–31.
Rodrigues TM, Marques JP, Soares M, Simão S, Melo P, Martins A, et al. Macular OCT-angiography parameters to predict the clinical stage of nonproliferative diabetic retinopathy: an exploratory analysis. Eye. 2019;33(8):1240–7.
Lu Y, Simonett JM, Wang J, Zhang M, Hwang T, Hagag AM, et al. Evaluation of automatically quantified foveal avascular zone metrics for diagnosis of diabetic retinopathy using optical coherence tomography angiography. Invest Ophthalmol Vis Sci. 2018;59(6):2212–21.
Louis LM, Fayed AE, Helal MS, Habib AM, Hashem MH, Embabi SN. Correlation between visual acuity and diabetic macular ischemia using optical coherence tomography angiography. Egyptian Retina Journal. 2019;6(2):33.
Pichi F, Smith SD, Abboud EB, Neri P, Woodstock E, Hay S, et al. Wide-field optical coherence tomography angiography for the detection of proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol. 2020;258:1901–9.
Couturier A, Rey P-A, Erginay A, Lavia C, Bonnin S, Dupas B, et al. Widefield OCT-angiography and fluorescein angiography assessments of nonperfusion in diabetic retinopathy and edema treated with anti–vascular endothelial growth factor. Ophthalmology. 2019;126(12):1685–94.
Tan B, Chua J, Lin E, Cheng J, Gan A, Yao X, et al. Quantitative microvascular analysis with wide-field optical coherence tomography angiography in eyes with diabetic retinopathy. JAMA Netw Open. 2020;3(1):e1919469-e.
Khalid H, Schwartz R, Nicholson L, Huemer J, El-Bradey MH, Sim DA, et al. Widefield optical coherence tomography angiography for early detection and objective evaluation of proliferative diabetic retinopathy. Br J Ophthalmol. 2021;105(1):118–23.
Belenje A. Role of wide-angle optical coherence tomography angiography in the detection of clinically non-apparent neovascularisation in proliferative diabetic retinopathy. BMJ Case Rep. 2020;13(9):e236836.
Wang F, Saraf SS, Zhang Q, Wang RK, Rezaei KA. Ultra-widefield protocol enhances automated classification of diabetic retinopathy severity with OCT angiography. Ophthalmol Retin. 2020;4(4):415–24.
Cabral D, Coscas F, Glacet-Bernard A, Pereira T, Geraldes C, Cachado F, et al. Biomarkers of peripheral nonperfusion in retinal venous occlusions using optical coherence tomography angiography. Trans Vis Sci Technol. 2019;8(3):7.
Capone L, Rabiolo A, Cicinelli MV, Corbelli E, Baldin G, Carnevali A, et al. Correlation analysis between foveal avascular zone and peripheral ischemic index in diabetic retinopathy: a pilot study. Investig Ophthalmol Vis Sci. 2017;58(8):74.
Rabiolo A, Cicinelli MV, Corbelli E, Baldin G, Carnevali A, Lattanzio R, et al. Correlation analysis between foveal avascular zone and peripheral ischemic index in diabetic retinopathy: a pilot study. Ophthalmol Retin. 2018;2(1):46–52.
Huang J, Lu Y, Gu X, Zheng B, Chen T. Correlation between the nonperfusion area on ultra-widefield fluorescein angiography and nonflow area on optical coherence tomographic angiography in retinal vein occlusion. J Ophthalmol. 2021. https://doi.org/10.1155/2021/5581319.
Soliman AZ, Silva PS, Aiello LP, Sun JK. Ultra-wide field retinal imaging in detection, classification, and management of diabetic retinopathy. Semin Ophthalmol. 2012. https://doi.org/10.3109/08820538.2012.708812.
Selvam S, Sim DA, Keane PA, Rajendram R, Karampelas M, Fruttiger M, et al. Patterns of peripheral retinal and central macula Ischemia in diabetic retinopathy as evaluated by ultra widefield fluorescein angiography. Investig Ophthalmol Visual Sci. 2014;55(13):260.
Jin K, Pan X, You K, Wu J, Liu Z, Cao J, et al. Automatic detection of non-perfusion areas in diabetic macular edema from fundus fluorescein angiography for decision making using deep learning. Sci Rep. 2020;10(1):1–7.
Gardner TW, Eller AW, Friberg TR. Reduction of severe macular edema in eyes with poor vision after panretinal photocoagulation for proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol. 1991;229(4):323–8.
Sim DA, Keane PA, Rajendram R, Karampelas M, Selvam S, Powner MB, et al. Patterns of peripheral retinal and central macula ischemia in diabetic retinopathy as evaluated by ultra-widefield fluorescein angiography. Am J Ophthalmol. 2014;158(1):144–53.
Acknowledgments
We thank all the participants for their involvement in the study. None of the patients was involved in the study trial design or dissemination of results.
Funding
Hyderabad Eye Research Foundation funded this study. No funding or sponsorship was received for the publication of this article.
Author information
Authors and Affiliations
Contributions
The authors adhered to the guidelines for authorship as per ICMJE. Conception, design, data collection, manuscript preparation—Padmaja Kumari Rani and Deepika C. Parameswarappa. Data collection—Amelia Janis Langstang and Sanagavarapu Kavya. Design, statistical analysis—Ashik Mohamed. Conception, review—Padmaja Kumari Rani, Michael W. Stewart, and Deepika C Parameswarappa.
Corresponding author
Ethics declarations
Conflict of Interest
Deepika C. Parameswarappa, Amelia Janis, Langstang, Sanagavarapu Kavya, Ashik Mohamed, and Padmaja Kumari Rani confirm that they have no conflicts of interest to declare. Michael W. Stewart is an Editorial Board member of Ophthalmology and Therapy. Michael W. Stewart was not involved in the selection of peer reviewers for the manuscript nor any of the subsequent editorial decisions.
Ethical Approval
The study was approved by the local ethics committee (LEC 10–19-352). Written informed consent to participate in the study was obtained from each participant.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Parameswarappa, D.C., Langstang, A.J., Kavya, S. et al. The Role of Widefield Optical Coherence Tomography Angiography in Assessing the Severity of Diabetic Retinopathy. Ophthalmol Ther 13, 2369–2380 (2024). https://doi.org/10.1007/s40123-024-00995-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-024-00995-y