Abstract
Background
Evidence on associations between drug–drug interactions (DDIs) and health outcomes in the older community-dwelling population is limited.
Objective
We estimate potentially clinically important DDI prevalence and examine the association between DDIs and (1) adverse drug events (ADEs), (2) emergency hospital attendance and (3) health-related quality of life (HRQoL) in an older community-dwelling population in Ireland.
Methods
This is a prospective cohort study of community-dwelling older adults (N = 904) aged ≥ 70 years from 15 general practices in Ireland recruited in 2010 (wave-1) and followed-up over 2 years (wave-2; 2012–2013), with linked national pharmacy claims data. Individuals dispensed two or more drugs (wave-1: N = 842; wave-2: N = 763) were included. DDI prevalence at baseline, follow-up and 6 months prior to each health outcome was estimated. Multi-level regression was used to model the association between DDI-exposure and health outcomes at follow-up. DDI prevalence, adjusted incidence-rate ratios (aIRR), adjusted odds ratios (aOR), β coefficients and robust standard error (RSE) from multi-level regression analyses, and 95% confidence intervals (CIs) are reported.
Results
At wave-1, n = 196 (23.3% [95% CI 20.5–26.3]), individuals were potentially exposed to ≥ 1 DDI, increasing to n = 345 (45.2% [41.7–48.9]) at wave-2. At 2-year follow-up, the median number of ADEs was 3 (interquartile range [IQR 2–5]); 229 (30.1%) had ≥ 1 emergency hospital attendance, and the mean EQ-5D was 0.74 (± 0.23). Evidence for the association between DDI-exposure and emergency hospital attendance at follow-up was lacking (aOR = 1.38 [0.42–4.53]). DDI-exposure was associated with an increasing number of ADEs (aIRR = 1.26 [1.03–1.55]), and decreasing EQ-5D utility (β = − 0.07, [−0.11 to −0.04], RSE = 0.02). Aspirin–warfarin, clarithromycin–prednisolone, amiodarone–furosemide, clarithromycin–salbutamol, rosuvastatin–warfarin, amiodarone–bisoprolol, and aspirin–nicorandil were common DDIs 6 months preceding these health outcomes.
Conclusions
We found a two-fold increase in DDI prevalence between wave 1 and 2. DDI exposure was associated with increasing ADEs and declining HRQoL at 2-year follow-up. Common DDIs involved anticoagulants, cardiovascular and antimicrobial drugs, which should be targeted for medicine optimisation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Using real-world data, we found that drug-drug interactions (DDIs) are associated with an increasing number of adverse drug events in the older community-dwelling population. |
DDI exposure was predictive of lower health-related quality of life in this older population. |
Common DDIs preceding these health outcomes frequently involved anticoagulants, cardiovascular drugs, and antimicrobial drugs; which should be targeted for medicine optimisation. |
1 Introduction
Medication-related harm continues to be a major concern in the older population, where around one in two older adults are reported to have an adverse health outcome related to potential drug exposure, 35–59% of which is reported to be preventable [1]. Drug–drug interactions (DDIs) are an example of an often avoidable cause of medication-related harm [2]. A DDI occurs when the effect of one drug is altered by the use of another drug [3], which may lead to adverse drug events (ADEs) and/or adverse drug reactions (ADRs), potentially requiring hospitalisation [4, 5]. In the older community-dwelling population, potentially clinically important DDIs are common, with estimates in Europe, Australia and the USA ranging from 0.8 to 54.3% [6]. Moreover, approximately 5% of hospitalisations in the older (≥ 65 years) population are reportedly due to DDIs [4, 7]; DDI exposure, specifically DDIs which increase bleeding risk, has also been shown to increase an older individual’s likelihood of an ADR-related hospital admission [8]. ADEs are also common in the older community-dwelling population [9]. An ADE, which, unlike an ADR, does not have a causal relationship with the drug, is defined as ‘an injury resulting from the use of a drug’ [10]. Unfortunately, these terms are often used interchangeably in the literature, making interpretation and comparison of studies challenging [11]. A 2011 systematic review estimates ADE prevalence in the older community-dwelling population to be between 5.5 and 34.7% [9]. Approximately one in four older community-dwelling individuals experiences a preventable ADE, with DDIs reported to be responsible for 13.3% of these [12].
In the current literature, studies have mainly examined the effect of potentially inappropriate prescribing (PIP) criteria (such as STOPP and Beers) on an older individual’s risk of experiencing an ADE [13,14,15]. These studies have generally reported the effect at the overall PIP level; though the utility, validity, and clinical relevance of such PIP criteria for real-world clinical practice has been questioned [16, 17]. More recently, researchers in the Netherlands published a study describing preventable ADEs caused by three high risk potential DDIs in patients admitted to intensive care units [18]. However, the effect of DDI exposure on ADE risk in the older community-dwelling population is less well understood. Further, since Becker and colleagues published their literature review in 2007 [4], studies examining DDIs and hospital admission in this older population are limited [19] and mainly descriptive [20,21,22]. Moreover, to date, there is a paucity of evidence on the effect of DDI exposure on health-related quality of life (HRQoL) in the older population [8]. The aim of this study is to estimate the prevalence of ‘severe’ potentially clinically important DDIs and to examine the association between these DDIs and adverse health outcomes (ADEs, emergency hospital attendance and HRQoL) in a prospective cohort of older community-dwelling adults from 15 general practices (GP) in Ireland.
2 Methods
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) and the Reporting of Studies Conducted using Observational Routinely Collected Data for Pharmacoepidemiological Research (RECORD-PE) guidelines were used in the reporting of this study [23, 24].
2.1 Study Design
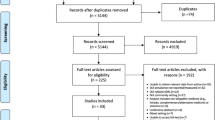
This is a secondary analysis of a prospective (2010–2012) cohort study of community-dwelling older adults (N = 904) aged ≥ 70 years from 15 general practices in the Republic of Ireland. At baseline (wave-1, 2010), participants were recruited over a 5-month period, using proportionate stratified random sampling, and were followed up over 2 years (wave 2, 2012–2013) [13, 14]. Full details on the inclusion and exclusion criteria have previously been described [13]. Study participants were in receipt of a General Medical Services (GMS) card, which permitted linkage of their pharmacy claims dispensing data from the Health Service Executive-Primary Care Reimbursement Service (HSE-PCRS) GMS database with their self-reported and GP medical record data. The HSE-PCRS GMS scheme (means tested since January 2009) provides free health services, including subsidised medications, to eligible individuals in Ireland [25]. As of 2013, approximately 90% of the general population aged ≥ 70 years was eligible for a GMS card [26]. In the HSE-PCRS GMS database, pharmacy claims data are coded using the World Health Organization (WHO) Anatomical Therapeutic Chemical (ATC) classification system. To be included in this study, participants were required to have been dispensed at least two medicines (distinct ATC codes) on the same day (i.e. co-dispensed) or within 7 days of each other (i.e. concomitant) [27] during wave-1 baseline and wave-2 follow-up.
2.2 Drug–Drug Interaction Exposure
The exposure of interest was any potentially clinically important DDI versus none. We have recently published a list of 28,225 unique DDIs classified as being both ‘severe’ (i.e. the result may be a life-threatening event or have a permanent detrimental effect) per the British National Formulary 84 (September 2022–March 2023) and also as being ‘a life-threatening or contraindicated combination’ or ‘dosage adjustment or close monitoring is needed’ per Stockley’s Drug Interactions, as of Q4 2022 [28]. Further details on the methodology used to generate the DDI lists have previously been described [8]. Using these DDI lists, HSE-PCRS-GMS pharmacy claims data for study participants co-dispensed ≥ 2 medications at wave-1 baseline (n = 842) and at wave-2 follow-up (N = 763), overall and in the 6 months prior to (1) ADE interview date (n = 563), (2) emergency hospital attendance date (n = 745) and (3) administration of postal questionnaire for HRQoL (n = 621) at wave-2 were examined for potentially clinically important DDIs (Fig. 1). In addition, we also report DDI prevalence per drugs dispensed within 7 days of each other (i.e. concomitant DDI prevalence [27]; n=839 at wave-1 baseline and n=761 at wave-2 follow-up had at least two medicines dispensed within 7 days of each other and were included for analysis) and the number of acute DDIs (DDI dispensed for less than three consecutive monthly pharmacy claims) and chronic DDIs (DDI continued to be dispensed for three or more consecutive monthly pharmacy claims) for each dispensing pattern. Unless otherwise stated, any reference herein to a DDI refers to those classified as ‘severe’ potentially clinically important.
2.3 Health outcomes
2.3.1 Adverse drug event
At wave-2 follow-up, patient interviews were conducted to identify patient-reported ADEs that had occurred in the previous 6 months; GP medical records were also reviewed to verify patient-reported ADEs and to identify additional ADEs occurring during this 6-month time period [13, 14]. A validated ADE identification method was used, where only ADEs with a clinical reviewer consensus rating of ≥ 50% likelihood were included [29]. A detailed explanation of the ADE identification method has previously been described [13, 14, 29].
2.3.2 Emergency hospital attendance
GP medical record data were reviewed to identify episodes (dates) of emergency hospital attendance, defined as any emergency (non-elective) admission(s) or emergency department (ED) visit(s) occurring during the 2-year follow-up period (2012–2013) [14].
2.3.3 Health-related quality of life (EQ-5D Utility)
HRQoL was measured using the EQ-5D, which estimates health states across five domains: mobility, self-care, usual activities, pain/discomfort and anxiety/depression [30]. We used the three-level rating system (EQ-5D-3L), administered via postal questionnaire (sent July–August 2012), which classifies each domain into three levels: no problems, some/moderate problems, and extreme problems [31]. A single utility value was calculated for each patient from a set of utility values derived from a UK population, using the time trade-off technique [32]. EQ-5D values range from 0 to 1, where 1 represents full health and 0 corresponds to a health state as bad as being dead and < 0 to worse than death [30]. The EQ-5D-3L has previously been validated to measure health status in the older population in 15 European countries [33].
2.4 Potential Confounders
For each health outcome, potential confounders were determined a priori based on current literature [13, 34,35,36], and included the confounders at baseline from GP medical record (age, gender, Charlson Comorbidity Index, and prior healthcare utilisation), postal questionnaire (social class [skilled or unskilled], deprivation score [based on participant’s address], education [basic, upper and post-secondary], social support [low, medium, or high], Lubben social network scale [number of social contacts]), and medication-related confounders from linked HSE-PCRS pharmacy claims data (polypharmacy, defined as the number of regular medicines at ATC level 3 dispensed per participant in at least three consecutive months between baseline [wave-1] and the 2-year follow-up period [wave-2]).
2.5 Statistical Analysis
Baseline characteristics of the population are reported as medians (interquartile range [IQR]) for continuous variables and as number of study participants (percent) for categorical variables. DDI prevalence and 95% confidence intervals (CIs), estimated using the Agresti–Coull method, are presented for the overall prevalence at wave-1 and wave-2 follow-up and the top ten most prevalent DDIs at wave-2. For overall DDI prevalence estimates, the denominator used was the number of individuals co-dispensed (or concomitantly dispensed) at least two distinct drugs. To account for clustering by general practice, multi-level regression models were used to model the association between co-dispensed DDI exposure and health outcomes at wave-2 follow-up: multi-level negative binomial regression (incident rate ratio) for ADE, multi-level logistic regression (odds ratio) for emergency hospital attendance and multi-level linear regression (β coefficient) for EQ-5D utility. Negative binomial regression was used due to overdispersion of ADE data at wave-2 follow-up. Multicollinearity was assessed using the variance inflation factor and tolerance statistics [37]. To avoid immortal time bias and other time-related exposure misclassification [38], the start of DDI exposure was defined based on pharmacy dispensing date in the 6-months prior to the outcome of interest, and each study participant was followed from the start of DDI exposure until the date of the outcome of interest (Fig. 1). Adjusted incidence rate ratios (aIRR), adjusted odds ratios (aOR), adjusted β coefficients, robust standard errors (RSE) and 95% CIs are reported. The data were analysed using SAS version 9.4 statistical package and STATA version 14.0 (StataCorp, College Station, TX).
3 Results
3.1 Study Population
Of the 904 participants who completed wave-1 (2010), n = 791 (88%) completed wave-2 follow-up [14]. In total, 763 of these individuals were co-dispensed two or more drugs during wave-2 follow-up and were included for analysis. At wave-2 follow-up, among eligible study participants using two or more drugs, 563 had ADE data, 745 had emergency hospital attendance data and 621 responded to the postal questionnaire (Fig. 1); full details on participants lost to follow-up have previously been described [14]. Table 1 provides a summary of study participants by characteristics and DDI exposure at wave-2 follow-up. A comparison of study participant characteristics, including medications, between wave-1 baseline and wave-2 follow-up is presented in Supplementary Table 1.
3.2 DDI Prevalence
3.2.1 DDI Prevalence Involving Co-dispensed Medications
Overall, n = 196 (23.3% [95% CI 20.5–26.3%]) individuals were potentially exposed to at least one (range 1–11) potentially clinically important DDI at wave-1 baseline (2010). During the 2-year follow-up period (2012–2013), n = 345 (45.2% [95% CI 41.7–48.8]) study participants were potentially exposed to at least one (range 1–10) DDI; n = 296 (38.8% [95% CI 35.4–42.3]) in the year (2011–2012) preceding wave-2 follow-up. Figure 2 presents a comparison of DDI prevalence over the study observation period. In total, n = 347 unique DDIs were identified in the study population during 2-year follow-up. The median number of DDIs per study participant was two (IQR 1–3). Among these individuals, n = 341 (44.7%) were potentially exposed to at least one (range 1–9) DDI requiring ‘dosage adjustment or close monitoring’, and n = 42 (5.5%) were potentially exposed to at least one (range 1–3) DDI involving a ‘life-threatening or contraindicated combination’. Antimicrobial (22.3%) and cardiovascular (21.5%) DDIs, and DDIs which carry an increased risk of hypokalaemia (28.6%), torsade de pointes (27.8%) and bleeding (13.9%) were most common in the population. Clarithromycin–prednisolone (n = 69, 20.0%) was the most prevalent DDI, followed by aspirin–warfarin (n = 45, 13.0%). Table 2 presents a summary of the top ten most prevalent DDIs during wave-2 follow-up and corresponding interaction effect and clinical guidance per the BNF and Stockley’s drug interactions. The prevalence of all n = 347 DDIs identified in the study population during the 2-year follow-up period (2012–2013) is reported in Supplementary Table 2.
3.2.2 Acute and Chronic DDIs Involving Co-dispensed Medications
Among the n = 345 study participants with a co-dispensed DDI at wave-2 follow-up, n = 187 (54.2%) participants were dispensed an acute DDI, and n = 158 (45.8%) participants were dispensed a chronic DDI. Aspirin–warfarin was the most frequently co-dispensed chronic DDI (n = 26 participants), followed by amiodarone–furosemide (n = 11), aspirin–nicorandil (n = 10), allopurinol–ramipril (n = 9) and amiodarone–bisoprolol (n = 8). Clarithromycin–salmeterol was the most frequently co-dispensed acute DDI involving ‘a life-threatening or contraindicated combination’ (n = 10), and domperidone–escitalopram was the most frequent co-dispensed acute (n < 5) and chronic (n < 5) DDI involving ‘a life-threatening or contraindicated combination’. The prevalence of all acute and chronic DDIs co-dispensed during wave-2 follow-up is presented in Supplementary Table 3.1–3.2.
3.2.3 DDI Prevalence Involving Concomitant (± 7 day) Medications
At baseline, n = 226 (26.9% [95% CI 24.0–30.0]) study participants were potentially exposed to at least one concomitant DDI (range 1–11). Aspirin–warfarin (n = 25) was the most prevalent concomitant DDI. At wave-2 follow-up, n = 348 (45.7% [95% CI 42.2–49.3]) study participants were potentially exposed to at least one concomitant DDI (range 1–10). Clarithromycin–prednisolone (n = 50) was the most prevalent concomitant DDI, followed by aspirin–warfarin (n = 45). The prevalence of all concomitant DDIs at wave-2 follow-up is presented in Supplementary Table 4.
3.2.4 Acute and Chronic DDIs Involving Concomitant (± 7 day) Medications
At wave-2 follow-up, among the n = 348 study participants with a concomitant DDI, n = 205 (58.9% [95% CI 53.7–64.0]) were dispensed an acute DDI, and n = 143 (41.1% [95% CI 36.0–46.3]) were dispensed a chronic DDI. Aspirin–warfarin (n = 24) was the most frequent chronic concomitantly dispensed DDI, followed by allopurinol–ramipril (n = 9). The prevalence of all acute and chronic DDIs concomitantly dispensed during wave-2 follow-up is presented in Supplementary Table 5.
3.3 Health Outcomes
3.3.1 Adverse Drug Event
At 2-year follow-up, of the 563 eligible study participants with ADE data, n = 419 (74.4%) either reported or had one or more (range 1–20) ADE recorded in their GP medical record during the 6 months prior to wave-2 interview date. The median number of ADEs was three (IQR 2–5); 88 (16%) reported one ADE, 91 (16%) reported two, 63 (11%) reported three, 43 (8%) reported four and 134 (24%) reported five or more ADEs. Among those with an ADE, n = 102 (24.3%) were potentially exposed to at least one DDI during this time period. Prevalent DDIs included: aspirin–warfarin (n = 21); clarithromycin–prednisolone (n = 7); amiodarone–furosemide (n = 6); clarithromycin–salbutamol (n = 5); and rosuvastatin–warfarin (n = 5). A full list of all DDIs identified in the 6 months prior to ADE outcome is reported in Supplementary Table 6. In the adjusted multi-level negative binomial regression analysis, exposure to at least one DDI was associated with an increasing number of ADEs at follow-up (aIRR 1.26 [95% CI 1.03–1.55], p = 0.02). Polypharmacy (aIRR 1.78 [95% CI 1.39–2.29], p < 0.001) and major polypharmacy (aIRR 2.53 [95% CI 1.91–3.37], p < 0.001) were independently associated with a greater number of ADEs at follow-up.
3.3.2 Emergency Hospital Attendance
At 2-year follow-up, data on emergency hospital attendance (ED visits or emergency hospital admission) were available for n = 745 study participants using two or more drugs. Among these individuals, n = 229 (30.1%) had at least one emergency hospital attendance recorded; n = 87 (38.0%) were potentially exposed to at least one DDI in the 6 months prior to emergency hospital attendance date. Prevalent DDIs in the 6 months prior to emergency hospital attendance included: aspirin–warfarin (n = 15); clarithromycin–prednisolone (n = 12); clarithromycin–salbutamol (n = 7); amiodarone–bisoprolol (n = 5); and amiodarone–warfarin (n < 5). A full list of all DDIs identified in the 6 months prior to emergency hospital attendance date is reported in Supplementary Table 7. In the adjusted multi-level logistic regression analysis, evidence was lacking on the association between exposure to at least one DDI and any emergency hospital attendance at 2-year follow-up (aOR 1.38 [95% CI 0.42–4.53]), p = 0.60) (Table 3).
3.3.3 Health-Related Quality of Life (EQ-5D Utility)
EQ-5D utility data were available for n = 621 (81.4%) individuals using two or more drugs at follow-up. N = 138 (22.2%) of these individuals were potentially exposed to at least one DDI in the 6 months pre-administration of the EQ-5D questionnaire. Prevalent DDIs in the 6 months prior to completion of the EQ-5D included: aspirin–warfarin (n = 24); clarithromycin–prednisolone (n = 13); clarithromycin–salbutamol (n = 10); amiodarone–furosemide (n = 9); and amiodarone–bisoprolol (n = 8). A full list of all DDIs identified in the 6 months prior to EQ-5D assessment date is reported in Supplementary Table 8. The mean EQ-5D in the population was 0.74 (± 0.23). Individuals potentially exposed to at least one DDI had a lower EQ-5D utility (0.66 [± 0.25]) compared with those unexposed (0.77 [±0.22]). In the adjusted multi-level linear regression analysis, there was a negative association between DDI exposure and EQ-5D utility at 2-year follow-up, where a one-unit (100%) increase in DDI exposure was significantly associated with a decreased EQ-5D utility (β −0.07, [95% CI −0.11 to −0.04] RSE 0.02, p < 0.001) (Table 3).
4 Discussion
In a population of older community-dwelling adults we found an increasing (almost two-fold) trend in potentially clinically important DDI prevalence over a 2-year follow-up period. This change in DDI prevalence can be explained by an increase in the number and type of medications dispensed at wave-2, including an increase in the use of nonsteroidal anti-inflammatory drugs (NSAIDs) and antimicrobials (mainly beta-lactams and macrolides). Almost half (45.8%) of the study participants followed-up were dispensed the same DDI for a prolonged period of time (≥ 3 consecutive monthly pharmacy claims). Most study participants were potentially exposed to at least one DDI requiring ‘dosage adjustment or close monitoring’; DDIs involving antimicrobial drugs, predominantly the macrolide clarithromycin, DDIs involving cardiovascular drugs and DDIs which carry an increased risk of hypokalaemia, torsade de pointes and bleeding, were all common. Clarithromycin–prednisolone was the most prevalent DDI, with one-fifth of older community-dwelling individuals potentially exposed to this DDI. With regard to the health outcomes examined, evidence to demonstrate a precise association between DDI exposure and emergency hospital attendance at 2-year follow-up was lacking. However, DDI exposure was associated with an increasing number of ADEs, and a lower EQ-5D utility in the older community-dwelling population at 2-year follow-up. Aspirin–warfarin, clarithromycin–prednisolone, amiodarone–furosemide, clarithromycin–salbutamol, rosuvastatin–warfarin, amiodarone–bisoprolol, aspirin–nicorandil and atorvastatin–diltiazem were common DDIs identified among study participants in the 6-month period prior to the occurrence of these two specific health outcomes. These findings provide insight into some of the key DDIs which may potentially contribute to medication-related harm in the older community-dwelling population and which should be the focus of medication optimisation strategies.
The DDI prevalence which we identified at 2-year follow-up is at the upper-end of the range reported by other European studies (from 0.8 to 54.3%) for the older community dwelling population [6]. This could be due to differences between study populations, but more likely, it is related to the lack of a standardised DDI identification method [6]. Indeed, it may also be due to the large number of potentially clinically important DDIs (28,225) examined in our study. Interestingly, the most prevalent DDI (clarithromycin–prednisolone) in the population at 2-year follow-up has been described in a 2014 case-report, involving a case of mania induced by clarithromycin in an older patient, with no prior history of mood disorder, on low dose (5 mg od) prednisolone [39].
To date, few studies have evaluated DDI exposure and ADEs in the older community-dwelling population [12], with most research in this area restricted to long-term care (nursing home) facilities [40] and the intensive care unit [18]. In a 2003 cohort study of ambulatory Medicare patients (aged ≥ 65 years) in the USA, Gurwitz and colleagues found that DDIs were responsible for 13.3% of preventable ADEs [12]. Similar to our findings, the authors note that warfarin was commonly implicated in the DDIs identified. Since the introduction of direct oral anticoagulants in Ireland, there has been a decline in the use of warfarin [41]. However, for some individuals (e.g. those with atrial fibrillation and a mechanical heart valve or mitral stenosis), warfarin is the most suitable anticoagulant [42]; hence, awareness and knowledge of these DDIs remains relevant to current clinical practice. In current literature, a limited number of studies have also assessed DDI exposure and emergency department visits/hospital admissions in the older community-dwelling population [4, 7, 20]. In their 2007 literature review, Becker and colleagues report that 4.8% of admissions in the older population are due to DDIs, and often involve NSAIDs and cardiovascular drugs [4]. More recently, in a cross-sectional study of older (≥ 75 years) individuals admitted to hospital in France between 2016 and 2017, approximately 6% of admissions were considered to be DDI-related [20]. Similar to our study, many of these DDIs involved cardiovascular drugs (e.g. amiodarone); interestingly, the authors highlight that only about one-third (32%) of DDIs were identified by clinicians, suggesting gaps in awareness and knowledge of DDIs. Other studies have examined the association between specific DDIs and hospital admission in the older community-dwelling population [5, 19, 43, 44]. Gasse et al. used population data from the UK CPRD database to determine the risk of serious bleeding resulting in hospitalisation associated with exposure to DDIs involving warfarin and found a 3 to 4.5-fold increased risk [43]. In an older Canadian population, Juurlink and colleagues found an increased risk for hospital admission among those exposed to co-trimoxazole and glyburide, clarithromycin and digoxin, and potassium-sparing diuretics and angiotensin-converting enzyme (ACE)-inhibitors [5]. A large (~ 876,000) case–control study among older (≥ 65 years) individuals in Bologna examined the risk of hospitalisation associated with 10 DDIs and found that ACE-inhibitors/diuretics with glucocorticoids and fluoroquinolones with antidiabetic drugs were both associated with increased hospitalisation [19]. Compared with these studies, our study possibly lacks sufficient power to demonstrate equivalent and precise associations. Nonetheless, similar to our findings, DDIs involving cardiovascular and antimicrobial drugs were common across these studies.
Despite the high DDI prevalence reported for the older community-dwelling population [6], there is a paucity of research investigating the effect of DDI exposure on HRQoL [45]. In the present study, the estimated declining EQ-5D utility at 2-year follow-up suggests DDI exposure to be predictive of worse HRQoL in the older community-dwelling population. Similar to our findings, a previous study which included both primary care (70%) and non-primary care (30%) older (≥ 65 years) adults with type-2 diabetes in Portugal found that DDI exposure was predictive of lower EQ-5D utility (OR 1.34, 95% CI 0.73–2.48) [45]. It has previously been shown that a decline in EQ-5D utility of 0.05 is predictive of 5-year mortality in the older (≥ 65 years) population [46], which is similar in magnitude to that observed in the present study, further highlighting the potentially consequential harm arising from DDI exposure in this population.
4.1 Strengths and Limitations
We used real-world data from a 3-year prospective cohort study, a validated ADE identification method [29], and detailed pharmacy dispensing data derived from a large national population-based pharmacy claims database to explicitly examine associations between DDIs and multiple health outcomes (ADEs, emergency hospital attendance and HRQoL) in the older community-dwelling population. We used two compendia (the BNF and Stockley’s, the latter often referred to as the gold standard [47]) to identify a number of potentially clinically important DDIs from a list of approximately 28,225 DDIs. We used a multi-level model to account for residual clustering due to GP practices in our effect estimates. In addition, we considered time-related exposure misclassification (immortal time bias) in the design and analysis of this study, which is often not accounted for by observational studies [38]. However, our study has some limitations which should be considered. First, our analysis explores a ‘temporal association’, we do not affirm a causal relationship between DDI exposure and the outcomes examined. Medication adherence has previously been shown to contribute to preventable ADEs [12], which may have influenced our findings. However, a previous study that used the same cohort reports a median medication possession ratio > 80% at baseline and follow-up [14]. Indeed, medication non-adherence may have presented due to a DDI; however, this specific information was not available for assessment. We reviewed the dose of drugs dispensed involving DDIs subject to ‘dosage adjustment or close monitoring’ and found some variation during the study observational period; however, without supplementary clinical data to corroborate any dose changes, as well as indication, it is difficult to comment definitively on this. In addition, for DDIs involving antimicrobials (e.g. clarithromycin), we do not know if the individual study participant was advised by their pharmacist/doctor to hold the interacting drug during antibiotic treatment. There is uncertainty in the clinical relevance of some of our results (wide confidence interval for the effect of DDI exposure on emergency hospital attendance) due to study power limitations, which should be considered when interpreting these results. Although we examined a large number (28,225) of potentially clinically important DDIs, those with no severity rating in the BNF (e.g. opioids and gabapentin, amitriptyline and haloperidol) were not included, which suggests the BNF lacks some sensitivity in this respect.
4.2 Implications
Our study confirms that potentially clinically important DDIs are associated with a number of adverse health outcomes (ADEs and lower HRQoL) in an older community-dwelling population prospectively followed-up over a 2-year period. Greater care is therefore needed when prescribing and dispensing medications for this older population to mitigate potential DDI-related adverse health outcomes. In general, based on our findings and those of previous studies, in clinical practice it would seem prudent to direct attention to anticoagulants (e.g. warfarin), cardiovascular drugs (e.g. amiodarone), and antimicrobial drugs.
DDI mitigation strategies currently employed in clinical practice are mainly reliant upon clinical decision support systems; however, the often poor validity of these systems and well-established problem of ‘alert fatigue’ [48], casts doubt on their utility. From a clinical practice point of view, it is clear that a patient-centred approach is needed to mitigate DDI-related harm and to ensure the safe and rational use of medicines in the older population. In recent years, deprescribing strategies have been effectively utilised to reduce harm from polypharmacy and potentially inappropriate medications in the older population [49, 50]. Similar DDI-orientated strategies could also be implemented to facilitate ‘medication without harm’ [51] in the older population; although, research in this area is currently lacking. Indeed, prioritising medication safety in the older population is perhaps also a question of sensible and efficient use of available resources. In other countries, for example, pharmacists have been integrated into general practice to perform medication reviews and reduce hazardous prescribing [52, 53]. A similar approach could be adopted in Ireland; although, as noted in a recent realist review which examined interventions to reduce ADEs in general practice, there is a need to identify which healthcare professional is responsible for conducting a medication review, and formal policy and legislation is needed to support GP–pharmacist collaboration [54].
Ultimately, prescribers and pharmacists caring for the older population must be cognisant of the gaps in the current evidence base and age-related changes in physiological reserve affecting drug pharmacokinetic and pharmacodynamic responses, all of which mediate the risk of DDI-related adverse health outcomes [55]. To that end, continuous monitoring of DDIs at the population-level is necessary to proactively identify and mitigate DDI-related harm. Further research, using longitudinal data, is warranted to examine which specific DDIs pose the greatest risk to the older community-dwelling population. Ireland’s health service has recently implemented a preferred antibiotics initiative to improve antimicrobial prescribing in primary care and minimise the use of hazardous antibacterial agents [56]. This policy provides a good opportunity to examine population-level, longitudinal data to compare trends in DDIs involving common precipitant agents, such as macrolides. For DDIs which are subject to ‘dosage adjustment or close monitoring’, further research is needed to assess the extent to which this is performed in current clinical practice. Research is also needed to understand healthcare professional’s awareness and knowledge of potentially clinically important DDIs and how they manage these DDIs in current practice. Finally, DDI-orientated deprescribing strategies should be the focus of future research.
5 Conclusions
Potentially clinically important DDIs are associated with ADEs and worse HRQoL in the older community-dwelling population. Prevalent and common DDIs frequently involved anticoagulants, cardiovascular drugs and antimicrobial drugs, all of which should be targeted by medicine optimisation interventions in routine clinical practice.
References
Parekh N, Ali K, Page A, Roper T, Rajkumar C. Incidence of medication-related harm in older adults after hospital discharge: a systematic review. J Am Geriatr Soc. 2018;66(9):1812–22.
Magro L, Moretti U, Leone R. Epidemiology and characteristics of adverse drug reactions caused by drug–drug interactions. Expert Opin Drug Saf. 2012;11(1):83–94.
Preston CL. Stockley’s drug interactions: a source book of interactions, their mechanisms, clinical importance and management. 12th ed. London: Pharmaceutical Press; 2019.
Becker ML, Kallewaard M, Caspers PW, Visser LE, Leufkens HG, Stricker BH. Hospitalisations and emergency department visits due to drug–drug interactions: a literature review. Pharmacoepidemiol Drug Saf. 2007;16(6):641–51.
Juurlink DN, Mamdani M, Kopp A, Laupacis A, Redelmeier DA. Drug-drug interactions among elderly patients hospitalized for drug toxicity. JAMA. 2003;289(13):1652–8.
Hughes JE, Waldron C, Bennett KE, Cahir C. Prevalence of drug–drug interactions in older community-dwelling individuals: a systematic review and meta-analysis. Drugs & Aging. 2023 2023/02/01;40(2):117-34.
Jankel CA, Fitterman LK. Epidemiology of drug-drug interactions as a cause of hospital admissions. Drug Saf. 1993;9(1):51–9.
Hughes JE, Moriarty F, Bennett KE, Cahir C. Drug‐drug interactions and the risk of adverse drug reaction‐related hospital admissions in the older population. British journal of clinical pharmacology. 2023 2023/20/11.
Taché SV, Sönnichsen A, Ashcroft DM. Prevalence of adverse drug events in ambulatory care: a systematic review. Ann Pharmacother. 2011;45(7–8):977–89.
Nebeker JR, Barach P, Samore MH. Clarifying adverse drug events: a clinician’s guide to terminology, documentation, and reporting. Ann Intern Med. 2004;140(10):795–801.
Lavan AH, Gallagher P. Predicting risk of adverse drug reactions in older adults. Therap Adv Drug Saf. 2016;7(1):11–22.
Gurwitz JH, Field TS, Harrold LR, Rothschild J, Debellis K, Seger AC, et al. Incidence and preventability of adverse drug events among older persons in the ambulatory setting. JAMA. 2003;289(9):1107–16.
Cahir C, Bennett K, Teljeur C, Fahey T. Potentially inappropriate prescribing and adverse health outcomes in community dwelling older patients. Br J Clin Pharmacol. 2014;77(1):201–10.
Wallace E, McDowell R, Bennett K, Fahey T, Smith SM. Impact of potentially inappropriate prescribing on adverse drug events, health related quality of life and emergency hospital attendance in older people attending general practice: a prospective cohort study. J Gerontol Ser A Biomed Sci Med Sci. 2017;72(2):271–7.
Pérez T, Moriarty F, Wallace E, McDowell R, Redmond P, Fahey T. Prevalence of potentially inappropriate prescribing in older people in primary care and its association with hospital admission: longitudinal study. BMJ. 2018;363.
Parodi López N, Belfrage B, Koldestam A, Lönnbro J, Svensson SA, Wallerstedt SM. Revisiting the inter-rater reliability of drug treatment assessments according to the STOPP/START criteria. Br J Clin Pharmacol. 2023;89(2):832–42.
Parodi López N, Svensson SA, Wallerstedt SM. Clinical relevance of potentially inappropriate medications and potential prescribing omissions according to explicit criteria—a validation study. Eur J Clin Pharmacol. 2022;78(8):1331–9.
Klopotowska JE, Leopold JH, Bakker T, Yasrebi‐de Kom I, Engelaer FM, de Jonge E, et al. Adverse drug events caused by three high‐risk drug‐drug interactions in patients admitted to intensive care units: a multicentre retrospective observational study. British journal of clinical pharmacology. 2023.
Swart F, Bianchi G, Lenzi J, Iommi M, Maestri L, Raschi E, et al. Risk of hospitalization from drug-drug interactions in the elderly: real-world evidence in a large administrative database. Aging (Albany NY). 2020;12(19):19711.
Letinier L, Pujade I, Duthoit P, Evrard G, Salvo F, Gil-Jardine C, et al. Emergency department admissions induced by drug–drug interactions in the elderly: a cross-sectional study. Clin Transl Sci. 2022;15(6):1472–81.
Zerah L, Henrard S, Wilting I, O’Mahony D, Rodondi N, Dalleur O, et al. Prevalence of drug-drug interactions in older people before and after hospital admission: analysis from the OPERAM trial. BMC Geriatr. 2021;21(1):1–11.
Očovská Z, Maříková M, Vlček J. Potentially clinically significant drug-drug interactions in older patients admitted to the hospital: a cross-sectional study. Front Pharmacol. 2023;14.
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med. 2007;147(8):573–7.
Langan SM, Schmidt SA, Wing K, Ehrenstein V, Nicholls SG, Filion KB, et al. The reporting of studies conducted using observational routinely collected health data statement for pharmacoepidemiology (RECORD-PE). Bmj. 2018;363.
Sinnott S-J, Bennett K, Cahir C. Pharmacoepidemiology resources in Ireland—an introduction to pharmacy claims data. Eur J Clin Pharmacol. 2017;73(11):1449–55.
Central Statistics Office. Women and Men in Ireland 2013: persons with a medical card. [cited Date Accessed: 10 September, 2023]; Available from: https://www.cso.ie/en/releasesandpublications/ep/p-wamii/womenandmeninireland2013/healthlist/health/#d.en.65619
Tobi H, Faber A, Van Den Berg PB, Drane JW, de Jong‐van den Berg LT. Studying co‐medication patterns: the impact of definitions. Pharmacoepidemiol Drug Saf. 2007;16(4):405-11.
Hughes JE. Drug-drug interaction list for pharmacoepidemiology research. 2023 [cited; Available from: https://osf.io/y3gmx/
Cahir C, Wallace E, Cummins A, Teljeur C, Byrne C, Bennett K, et al. Identifying adverse drug events in older community-dwelling patients. Ann Fam Med. 2019;17(2):133–40.
Devlin N, Parkin D, Janssen B, Devlin N, Parkin D, Janssen B. An introduction to EQ-5D instruments and their applications. Methods Anal Report EQ-5D Data. 2020:1–22.
Rabin R, Charro Fd. EQ-SD: a measure of health status from the EuroQol Group. Ann Med. 2001;33(5):337–43.
Dolan P. Modeling valuations for EuroQol health states. Med Care. 1997;35(11):1095–108.
Buchholz I, Marten O, Janssen MF. Feasibility and validity of the EQ-5D-3L in the elderly Europeans: a secondary data analysis using SHARE (d) data. Qual Life Res. 2022;31(11):3267–82.
Field TS, Gurwitz JH, Harrold LR, Rothschild J, DeBellis KR, Seger AC, et al. Risk factors for adverse drug events among older adults in the ambulatory setting. J Am Geriatr Soc. 2004;52(8):1349–54.
Siqeca F, Yip O, Mendieta MJ, Schwenkglenks M, Zeller A, De Geest S, et al. Factors associated with health-related quality of life among home-dwelling older adults aged 75 or older in Switzerland: a cross-sectional study. Health Qual Life Outcomes. 2022;20(1):166.
Wallace E, Stuart E, Vaughan N, Bennett K, Fahey T, Smith SM. Risk prediction models to predict emergency hospital admission in community-dwelling adults: a systematic review. Med Care. 2014;52(8):751.
Kim JH. Multicollinearity and misleading statistical results. Korean J Anesthesiol. 2019;72(6):558.
Suissa S, Dell’Aniello S. Time-related biases in pharmacoepidemiology. Pharmacoepidemiol Drug Saf. 2020;29(9):1101–10.
Liu EY, Vasudev A. Mania induced by clarithromycin in a geriatric patient taking low-dose prednisone. Prim Care Compan CNS Disord. 2014;16(3):27457.
Gurwitz JH, Field TS, Judge J, Rochon P, Harrold LR, Cadoret C, et al. The incidence of adverse drug events in two large academic long-term care facilities. Am J Med. 2005;118(3):251–8.
Smith A, Barry M. Oral anticoagulants-utilisation and expenditure under the community drugs schemes. Ir Med J. 2020;113(5):71.
Vahanian A, Beyersdorf F, Praz F, Milojevic M, Baldus S, Bauersachs J, et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease: developed by the Task Force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2022;43(7):561–632.
Gasse C, Hollowell J, Meier CR, Haefeli WE. Drug interactions and risk of acute bleeding leading to hospitalisation or death in patients with chronic atrial fibrillation treated with warfarin. Thromb Haemost. 2005;94(09):537–43.
Schelleman H, Bilker WB, Brensinger CM, Han X, Kimmel SE, Hennessy S. Warfarin with fluoroquinolones, sulfonamides, or azole antifungals: interactions and the risk of hospitalization for gastrointestinal bleeding. Clin Pharmacol Ther. 2008;84(5):581–8.
AL‐Musawe L, Torre C, Guerreiro JP, Rodrigues AT, Raposo JF, Mota‐Filipe H, et al. Polypharmacy, potentially serious clinically relevant drug‐drug interactions, and inappropriate medicines in elderly people with type 2 diabetes and their impact on quality of life. Pharm Res Perspect. 8(4):e00621.
Perera S, Studenski S, Chandler JM, Guralnik JM. Magnitude and patterns of decline in health and function in 1 year affect subsequent 5-year survival. J Gerontol A Biol Sci Med Sci. 2005;60(7):894–900.
Kheshti R, Aalipour M, Namazi S. A comparison of five common drug–drug interaction software programs regarding accuracy and comprehensiveness. J Res Pharm Pract. 2016;5(4):257.
Van De Sijpe G, Quintens C, Walgraeve K, Van Laer E, Penny J, De Vlieger G, et al. Overall performance of a drug–drug interaction clinical decision support system: quantitative evaluation and end-user survey. BMC Med Inform Decis Mak. 2022;22(1):1–11.
Battar S, Dickerson KRW, Sedgwick C, Cmelik T. Understanding principles of high reliability organizations through the eyes of VIONE: a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564.
Omuya H, Nickel C, Wilson P, Chewning B. A systematic review of randomised-controlled trials on deprescribing outcomes in older adults with polypharmacy. Int J Pharm Pract. 2023:riad025.
The World Health Organization. WHO Global Patient Safety Challenge: Medication Without Harm. 2017 [Cited Date Accessed: 12 May, 2021]; Available from: http://apps.who.int/iris/bitstream/handle/10665/255263/WHO-HIS-SDS-2017.6-eng.pdf?ua=1&ua=1?sequence=1
Avery AJ, Rodgers S, Cantrill JA, Armstrong S, Cresswell K, Eden M, et al. A pharmacist-led information technology intervention for medication errors (PINCER): a multicentre, cluster randomised, controlled trial and cost-effectiveness analysis. Lancet. 2012;379(9823):1310–9.
Tan EC, Stewart K, Elliott RA, George J. Pharmacist services provided in general practice clinics: a systematic review and meta-analysis. Res Social Adm Pharm. 2014;10(4):608–22.
Waldron C, Hughes J, Wallace E, Cahir C, Bennett K. Contexts and mechanisms relevant to General Practitioner (GP) based interventions to reduce adverse drug events (ADE) in community dwelling older adults: a rapid realist review. HRB Open Res. 2023;5(53):53.
Hilmer SN, McLachlan AJ, Le Couteur DG. Clinical pharmacology in the geriatric patient. Fund Clin Pharmacol. 2007;21(3):217–30.
Health Service Executive. Green Red antibiotic QI initiative for community prescribers. [Cited Date Accessed: 6 March, 2024]; Available from: https://www.hse.ie/eng/services/list/2/gp/antibiotic-prescribing/antibicrobial-stewardship-audit-tools/campaign-materials/
Acknowledgements
We acknowledge and thank the HSE-PCRS for providing the pharmacy claims dispensing data used in this study. We also wish to thank Emma Wallace and all researchers involved in the follow-up data collection. Finally we are grateful to the study participants and the 15 general practices who kindly gave their time to take part in this study. Finally, we wish to acknowledge the SPHeRE PhD programme.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Author contributions
J.H., C.C. and K.B. were involved in the concept and design of the study. J.H. led on the analysis and interpretation of the results. J.H. prepared the first draft of the manuscript and C.C. and K.B. provided feedback. All authors read and approved the final manuscript.
Funding
Open Access funding provided by the IReL Consortium. This research was supported by funding from the Health Research Board Research Leader Award (grant number RL-15-1579) and the Irish Research Council Government of Ireland Postgraduate Scholarship Programme Award to J.H. (grant number GOIPG/2021/1213). C.C. is supported by funding from the Health Research Board (SDAP-2021-020). The funding bodies had no part in the study design, the identification, analysis, and collection of the data, or preparation of the manuscript for publication.
Data availability
All data generated or analysed during this study are included in this article (and its Supplementary Information files). The master DDI list (which includes full details on interaction effect, evidence and action) is available to other researchers upon reasonable request.
Code availability
Not applicable.
Conflicts of interest
K.B. is in receipt of funding from IQVIA and Novartis for unrelated projects.
Ethics approval
Ethical approval for this study was obtained from the RCSI Research Ethics Committee (REC 462b).
Consent to participate
All participants gave informed consent before taking part in the study.
Consent for publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Hughes, J.E., Bennett, K.E. & Cahir, C. Drug–Drug Interactions and Their Association with Adverse Health Outcomes in the Older Community-Dwelling Population: A Prospective Cohort Study. Clin Drug Investig 44, 439–453 (2024). https://doi.org/10.1007/s40261-024-01369-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-024-01369-9