Abstract
Background
In the last 5 years since our last systematic review, a significant number of articles have been published on the technical aspects of muscle near-infrared spectroscopy (NIRS), the interpretation of the signals and the benefits of using the NIRS technique to measure the physiological status of muscles and to determine the workload of working muscles.
Objectives
Considering the consistent number of studies on the application of muscle oximetry in sports science published over the last 5 years, the objectives of this updated systematic review were to highlight the applications of muscle oximetry in the assessment of skeletal muscle oxidative performance in sports activities and to emphasize how this technology has been applied to exercise and training over the last 5 years. In addition, some recent instrumental developments will be briefly summarized.
Methods
Preferred Reporting Items for Systematic Reviews guidelines were followed in a systematic fashion to search, appraise and synthesize existing literature on this topic. Electronic databases such as Scopus, MEDLINE/PubMed and SPORTDiscus were searched from March 2017 up to March 2023. Potential inclusions were screened against eligibility criteria relating to recreationally trained to elite athletes, with or without training programmes, who must have assessed physiological variables monitored by commercial oximeters or NIRS instrumentation.
Results
Of the identified records, 191 studies regrouping 3435 participants, met the eligibility criteria. This systematic review highlighted a number of key findings in 37 domains of sport activities. Overall, NIRS information can be used as a meaningful marker of skeletal muscle oxidative capacity and can become one of the primary monitoring tools in practice in conjunction with, or in comparison with, heart rate or mechanical power indices in diverse exercise contexts and across different types of training and interventions.
Conclusions
Although the feasibility and success of the use of muscle oximetry in sports science is well documented, there is still a need for further instrumental development to overcome current instrumental limitations. Longitudinal studies are urgently needed to strengthen the benefits of using muscle oximetry in sports science.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Near-infrared spectroscopy (NIRS), on which commercially available oximeters are based, is a useful complementary method for non-invasively assessing, with good sensitivity, skeletal muscle oxygen delivery and utilization in response to different exercise modes, training interventions and ergogenic aids. |
NIRS instrumentation can assess a large panel of muscles at single measurement sites. |
NIRS is a functional tool in which skeletal muscle oxygenation data can be viewed in ‘real time’, complementing external power and heart rate data, allowing coaches and physical trainers to make better-informed decisions to guide training or recovery processes. |
Multi-modal techniques and sophisticated multi-channel NIRS instrumentations are required to provide a more detailed evaluation of muscle activity data during active exercise for a better understanding of muscle function. |
1 Introduction
Muscle oximetry, based on near-infrared spectroscopy (NIRS), is able to provide, non-invasively, information about the changes in oxygenation of haemoglobin (Hb), present primarily in smal vessels (< 1 mm in diameter) such as the capillary, arteriolar and venular bed, and myoglobin (Mb), a cytoplasmic protein of the striated muscles [1,2,3]. Considering that Mb oxygenation is expected to remain almost unchanged during exercise, any alteration of the [Hb + Mb] signal reflects mainly changes in Hb. NIRS has been implemented in three main modalities that differ from each other based on the temporal characteristics of the employed light: continuous-wave NIRS (CW-NIRS: light with constant intensity), frequency-domain NIRS (FD-NIRS: modulated light intensity) and time-domain NIRS (TD-NIRS: pulsed light intensity). CW-NIRS, based on constant tissue illumination, measures only the light attenuation through the muscle. FD-NIRS, which illuminates the muscle with intensity-modulated light, measures both the attenuation and the phase delay of the emerging light. TD-NIRS, by illuminating the muscle with short pulses of light, detects the shape of the pulse after propagation through the tissue. The quantification of muscle oxygenation depends on the NIRS technology adopted. Since the nineties, oximeters (utilizing spatially resolved CW-NIRS) have been made available for monitoring brain and muscle oxygen (O2) saturation (SmO2). The market launch of commercial relatively low-cost portable wireless muscle oximeters dates back to 2006, and our 2018 systematic review [4] highlighted the application of muscle oximetry in the assessment of skeletal muscle oxidative performance in sports activities. Fifty-seven studies published over 11 years were included in Ref. [4], emphasizing the application of this technology to physical exercise and training.
Afterwards, in 2019, Barstow [1] summarized the most common methodologies of skeletal muscle NIRS, their strengths and limitations, and discussed some of the potential confounding factors that may affect the quality and reproducibility of NIRS data in skeletal muscle. Recommendations to reduce variability and errors in data collection, analysis and interpretation were also provided. Salvatore et al. [5] summarized the effects of aging in healthy individuals and muscle O2 utilization; aging reduces SmO2 at rest, and during submaximal and maximal exercise, and extends the timeframe for restoration of SmO2 following exercise. In 2021, Cornelis et al. [6], summarizing the results of 11 clinical trials on the impact of exercise therapy on lower limb SmO2 evaluated by NIRS in patients with lower-extremity artery disease, showed that exercise training improved the de-oxygenation and re-oxygenation patterns. Recently, Tuesta et al. [7] reviewed 18 clinical trials that evaluated the effects of physical exercise on SmO2 in subjects with different pathologies. Muscle oximetry made it possible to observe changes in muscle oxygenation/deoxygenation parameters such as SmO2, oxyhaemoglobin (O2Hb), total haemoglobin (tHb) and deoxyhaemoglobin (HHb) upon exercise interventions in patients with chronic diseases and in healthy active subjects. NIRS is currently considered to be a particularly promising wearable biosensor. This technology enables the continuous monitoring of physiological signals at the muscle site, thereby facilitating more accurate diagnoses and follow-up examinations pertaining to local exercise metabolism and adaptation in skeletal muscle performance.
In the last 5 years, several articles have been published focusing on the technical aspects of muscle NIRS, the interpretation of the signals and the benefits of utilizing NIRS technique to measure the physiological status of muscles and to determine how the working muscles are being used [1, 3, 8, 9]. Within the field of sports science field and beyond, NIRS monitoring has a huge potential that is often ignored in an applied exercise environment [9].
Considering the consistent number of studies on the application of muscle oximetry in sports science published in the last 5 years, the objectives of this updated systematic review were to highlight the most recent applications of muscle oximetry in the assessment of skeletal muscle oxidative performance in different sports activities and to emphasize how this technology has been applied to exercise and training involving different interventions in sporting environments. In addition, some recent instrumental developments are briefly summarized.
2 Methods
2.1 Literature Search Methodology
A thorough systematic search of the research literature was performed conforming to the Preferred Reporting Items for Systematic Reviews (PRISMA) statement [10]. Our search of the literature began in December 2022 and continued through to March 2023. A search of electronic databases was conducted to identify all publications which utilized muscle oximetry in sport science published from March 2017 up to March 2023. As a prerequisite, all studies should have been performed in healthy sports populations including both adolescents and adults. Three databases (Scopus, MEDLINE/PubMed, and SPORTDiscus via the EBSCOHost) were searched electronically from inception using the terms ‘‘near infrared spectroscopy’’ OR “NIRS”, OR “oximetry” OR ‘‘muscle oxygenation”, AND with the term ‘‘sports”. Additionally, these four terms were combined (AND) with terms of different sports (“athletics” OR “badminton” OR “baseball” OR “basketball” OR “biathlon” OR “bicycling” OR “boxing” OR “canoeing” OR “climbing” OR “cricket” OR “croquet” OR “cross country” OR “cycling” OR “decathlon” OR “diving” OR “field hockey” OR “football” OR “futsal” OR “golf” OR “gymnastics” OR “handball” OR “high jump” OR “hockey” OR “ice hockey” OR “ice skating” OR “inline skates” OR “judo” OR “jumping” OR “karate” OR “kayaking” OR “kickball” OR “lacrosse” OR “long jump” OR “martial arts” OR “Nordic skiing” OR “paddling” OR “pentathlon” OR “ping pong” OR “polo” OR “racquetball” OR “rafting” OR “rock climbing” OR “roller skating” OR “rowing” OR “rugby” OR “running” OR “sailing” OR “skiing” OR”sledding” OR “snowboarding” OR “soccer” OR “softball” OR “speed skating” OR “squash” OR “Sumo wrestling” OR “surfing” OR “swimming” OR “synchronized swimming” OR “table tennis” OR “taekwondo” OR “tennis” OR “triathlon” OR “triple jump” OR “ultramarathon” OR “volley ball” OR “water polo” OR “waterskiing” OR”weightlifting” OR “white water rafting” OR “windsurfing”. Each database automatically uses its own term mapping. The results were screened to identify relevant studies, first by title, then by abstract, and finally by full text. Non-relevant titles and abstracts were omitted. full texts were screened for inclusion criteria and were only included in the review if they met all criteria. Differences in search outcomes were verified and consensus for inclusion was reached. We also extended the search scope to include “related articles”. Reference lists of fully evaluated publications were also examined for studies not found in the online database searches. Authors of published papers were also contacted directly if crucial data were not reported in the original paper.
2.2 Inclusion and Exclusion Criteria
The following inclusion criteria were used to select articles to be included in the systematic review:
-
1.
Only full articles published in English in peer-reviewed journals were considered. Book chapters and Proceedings were not included.
-
2.
Studies performed in healthy sports populations including both adolescents (over 15 years) and adults were included.
-
3.
Only studies performed using commercial oximeters or NIRS instruments that measure SmO2, and/or O2Hb and HHb changes utilizing the modified Beer–Lambert law were included.
-
4.
Muscle studies in which physiological variables were monitored in recreationally trained to elite athletes with or without training programme were included.
-
5.
NIRS muscle studies that did not report the oxygenation data units were discarded.
2.3 Study Selection and Data Extraction
Regarding the study eligibility, titles and abstracts of potentially relevant articles were screened independently by two reviewers (M.F. and V.Q.). The explicit rule was to select studies that could possibly meet the inclusion criteria. Title duplicate publications were removed, and articles which did not meet the inclusion criteria were excluded. Full texts were assessed for eligibility by the three authors, and any articles that were ambiguous regarding inclusion were independently assessed against the eligibility criteria. Disagreements regarding inclusion of ambiguous articles were discussed and a consensus was agreed. A pre-designed data extraction form was used to collate data from individual studies, including country/setting, study design, characteristics of participants, representativeness of the study sample and results. Data for each included study were extracted by two reviewers (M.F. and V.Q.) and were checked by a third reviewer (S.P.). For each article, a standardized document form was used to extract the following relevant information from the selected papers: the type of sport(s), authors, publication data, sample size, participant characteristics (age, sex, body mass, training status), exercise protocol, NIRS instrumentation and related-measured variables, outcome measures with training intervention if any, muscle(s) assessed, and a summary of main findings. All these data are included in the Table S1 of the Electronic Supplementary Material [ESM] available in the online version; the Table S1 includes 191 studies [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179,180,181,182,183,184,185,186,187,188,189,190,191,192,193,194,195,196,197,198,199,200,201]. We decided to not assess risk of bias due to several limitations of existing tools for assessing risk of reporting biases in systematic reviews [202].
3 Results
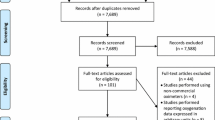
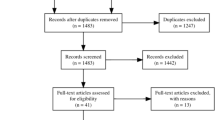
Figure 1 shows the flow of information through the systematic reviewing process. Of the total 12,018 (Scopus), 4176 (PubMed) and 1288 (SPORTDiscus) articles retrieved, 5408 were excluded as title duplicate publications (5293) and irrelevant topic (115). Other 7964 records were also excluded from the remaining 12,074 because they were not pertinent. A further 3798 records were excluded from the remaining 4110 after screening of the titles and the content of the abstracts; the remaining articles were 312. Nine studies that did not report oxygenation data units and 119 studies did not relate to sport were excluded from the 312 articles; the remaining articles were 184. Further 7 articles were identified from the reference list of other articles and were added to the 184 articles. Therefore, 191 studies were retained for inclusion in the final stage of this review. Study characteristics are summarized in Table S1.
3.1 Sporting Disciplines and Characteristics
For the purposes of reporting and analysis, the 191 included studies with 3435 participants were grouped into 37 sport disciplines (Table 1). The included studies examined recreational to elite athletes from a broad range of individual sports (e.g. cycling, rowing, skiing) and team-based sports (e.g. basketball, football, rugby). Of note, a total of eight studies included more than three sports [185,186,187,188,189,190,191,192] and a total of nine studies did not report the sporting discipline in their design [193,194,195,196,197,198,199,200,201]. The most common sports activities used in the 191 studies are cycling (n = 55), running (n = 23), and climbing (n = 11). Of the 191 studies, the majority utilized male participants (n = 119), 64 studies included males and females, and 8 studies included females only. Twenty-one studies reported a training intervention. A total of 53 studies (Table 1) included elite or highly trained participants, while 29 studies included well-trained participants, 51 studies included trained participants, and moderately trained participants were included in 58 studies. Of note, 16 studies were targeted especially on elite athletes, 3 studies compared young and old participants [66, 195, 197] and 7 studies investigated junior athletes (≤ 18 years) [15, 18, 60, 68, 70, 101, 161].
A total of 20 muscle sites (Fig. 2), covering both the lower and upper body, were measured by muscle oximetry during exercise. The vastus lateralis muscle (n = 138) was used in the majority of the 191 studies for the lower limbs, and the biceps brachii and brachioradialis muscles (n = 12 each) were used for the upper limbs. The adipose tissue thickness was not measured in 128 out of 191 studies using skinfold caliper or ultrasound or computed tomography (CT) or magnetic resonance imaging (MRI).
A range of commercially available muscle NIRS oximeters (Table 2) were used to assess muscle oxygenation during exercise in the 191 selected studies, during either local (small muscle mass) or global (whole body mass) exercise with increased load (isometric or dynamic contraction) on the primary active muscles. A total of 130 studies (68%, Table 1) used wearable devices to assess muscle oxygenation during exercise. Among these studies, a total of 40% used the PortaMon device (Artinis Medical System, the Netherlands) and a total of 24% studies used the Moxy device (Fortiori Design LLC, USA). Both are CW-NIRS wearable systems with wireless data transmission. The remaining 12 NIRS devices (Table 2) utilized different techniques, and several are no more commercially available. In most of the studies (n = 179) CW-NIRS was employed. The other two NIRS techniques were FD-NIRS [47, 56, 61, 62, 65, 66, 148, 149, 160, 197] or TD-NIRS [40]. These latter technologies could measure the O2Hb and HHb in absolute quantitative units in micromoles/litre, as well as the tHb.
3.2 Main Findings
Over the last 5 years, there has been a significant growth in the use of NIRS technique to study muscle oxygenation in conjunction with physical exercise and training as an intervention (Table 1). Regarding the sample size, in a total of 52 studies 20 and more participants were recruited, and in a total of 139 studies less than 20 participants were recruited. Several NIRS use and applications in sport sciences research can be found among the 191 studies reported in detail in Table S1.
3.2.1 Variables in Evaluating Muscle Oxygenation
The variables reported in the review for evaluating muscle oxygenation are:
1.NIRS-derived muscle O2 saturation (rSO2/SmO2/StO2/SO2/TOI/TSI named according to the different instruments);
2.muscle O2 consumption (mVO2), evaluated by measuring changes in (O2Hb + O2Mb) during arterial occlusion manoeuvres;
3.blood flow changes evaluated as tHb changes;
4.absolute blood flow evaluated by using venous occlusion manoeuvres;
5.the rate of reoxygenation after submaximal, maximal and brief high-intensity exercise.
Absolute blood flow was measured in a total of 35 studies. Specifically, 6 studies assessed muscle blood flow and mVO2 [41, 117, 138, 167, 169, 190], while 8 studies measured muscle blood flow [18, 41, 90, 117, 138, 167, 169, 190], and 25 studies quantified mVO2 [26, 39, 41, 53, 54, 58, 82, 98, 102, 116, 117, 121, 126, 132, 134, 138, 167, 169, 175, 184,185,186, 190, 195, 201]. Finally, the rate of reoxygenation after submaximal, maximal and brief high-intensity exercise are among key indicators for assessing muscle oxidative capability [26, 27, 95, 132, 138, 144, 178, 185, 195, 201].
3.2.2 Delineating the Exercise Intensity Domains
Specific physiological breakpoints have been assessed using NIRS technique in many muscle groups [73, 79, 91, 115, 145, 179] during ramp incremental exercise testing to better understand how distinct working muscles are differently affected by exercise intensity or training dose. These data were compared with other well-known physiological markers that delineate exercise intensity zones, such as the measurements of the blood lactate [73, 79] and ventilatory [91, 115, 145, 179] thresholds.
3.2.3 Examining the Impact of Ergogenic Aids on Muscle Oxygenation Responses
Over the last 5 years, the NIRS technique has received increasing attention in the world of sports and exercise science as a way to examine the physiological effects that different various ergogenic aids may have on changes in muscle oxygenation (e.g. as a means of enhancing muscle performance or recovery). The following situations were compared with some control scenarios to evaluate the muscle oxygenation responses and the potential benefits of the tested product or intervention: the use of a tracksuit jacket with heating elements [162], nonivamide-nicoboxil cream [180], sports compression garment (n = 3 for lower limb: tights, calf sleeve, socks; n = 1 for upper limb, forearm) [18, 19, 31, 107], core and skin cooling [136], undergoing thigh cooling by a water-circulating pad [85] and oral supplementation with dietary inorganic nitrate-rich and placebo beetroot juice [32, 33, 40, 43, 63, 75, 76, 131, 146, 158, 183, 190, 201], supplementation with pomegranate extract and co-supplementation with N-acetylcysteine [39], protein powder [134], pre-exercise ingestion of a drink with higher dissolved O2 [135], anthocyanin-rich New Zealand blackcurrant supplementation [27], 1 h after a single dose of mango leaf extract rich in mangiferin and lecithin, or mango leaf extract rich in mangiferin and quercetin, phospholipids addition [53,54,55], red spinach extract [121], citrulline drink/citrulline malate [190, 196], l-arginine [141, 143], Montmorency cherry polyphenols [67], eicosapentaenoic acid and docosahexaenoic acid (fish oil) [198], caffeine intake [189] and dark chocolate [81].
The extent of notable changes in the muscle oxygenation levels measured with NIRS indicators in response to various physiological stressors (hypoxia, exercise intensity, exercise profile, training) was investigated in the following cases: repeated sprint training [14, 15, 34, 107, 157, 165, 168, 172, 188, 191, 192], high-intensity interval training [15, 25, 43, 137, 157, 164, 184], voluntary hypoventilation at low lung volume versus normal breathing [17, 34, 88, 89, 151], inspiratory muscle training/pre-activation [45, 97, 100, 112, 113, 140, 152, 181, 187], blood flow restriction [12, 14, 147, 149, 191, 192], intermittent bilateral cuff inflation of lower limbs with three 5/10-min ischemia–reperfusion cycles [53], hypoxia (normo- and hypo-baric) [14, 35, 59, 72, 76, 77, 90, 108, 123, 129, 130, 137, 142, 148,149,150, 168, 188, 191, 192, 200], ischemic preconditioning [38, 46, 57, 111, 112, 120, 138, 153, 169, 172, 173, 199], water immersions [21, 85, 94, 95], breathing atmospheric air at 1.35 atmospheres [94, 95], muscle heating [46], cold face immersion while breath-holding [93], active passive recovery at different air temperature (20–40 °C) and/or simulated altitudes (400–3800 m) [86, 166], photobiomodulation therapy [105], hyperoxic conditions/preconditioning [11, 56, 95, 165, 194], muscle electrical stimulation [138, 144, 173, 184, 197] and stretching [193].
4 Discussion
This updated systematic review of studies on the application of muscle oximetry in sports sciences published over the last 5 years, aimed at synthesizing data on the use of muscle oximetry in evaluating oxidative skeletal muscle performance in 37 major sporting disciplines from included 191 studies.
4.1 Methodological Considerations
Some points need to be made about the NIRS devices and measured NIRS variables used in the studies included in this systematic review. Table 2 presents the 14 models of NIRS devices/instruments, belonging to nine manufacturers, used in the 191 selected studies. Only three CW-NIRS instruments (Humon Hex, Moxy and PortaMon), dedicated to muscle measurements only, are lightweight, compact and wearable and have wireless data transmission. The reliability and validity of these oximeters have been reported [203,204,205]. A study comparing SmO2 measurements from Moxy and PortaMon devices provided physiologically credible SmO2 measures at rest and during exercise [206]. However, absolute values obtained during exercise were generally not comparable between devices unless corrected by physiological calibration after arterial occlusion. This indicates that further efforts should be made to standardize all muscle oximeters, for example, by using tissue-simulating phantoms [207] and following the guidelines of ISO 80601-2-85:2021 (Medical electrical equipment—Part 2–85: particular requirements for the basic safety and essential performance of cerebral tissue oximeter equipment). The cumbersome instruments, developed for brain oximetry measurements, can be utilized for muscle studies. The light sources used are laser diodes (in eight devices) or light-emitting diodes (LEDs) (in six devices), with the latter being much less expensive. Only one wireless instrument (PortaMon) includes a six-axis motion sensor, a built-in gyroscope and accelerometer, that can be used to acquire real-time position and orientation movement data and synchronize it with NIRS data. To the best of our knowledge, data from the motion sensor have never been included in a publication. This monitoring of movement combined with machine learning may be useful outside the laboratory for both athletes and coaches in sports applications such as performance enhancement, technical analysis and injury risk mitigation [208]. In addition, movement, or any compression on the area near the NIRS optodes may cause changes in local blood flow that are reflected in the NIRS signals. Therefore, some adaptive filtering with this additional reference signal can be used to control any non-physiological alterations in the NIRS signals that may interfere with proper interpretation.
Most of the instruments (n = 13) are capable of measuring SmO2 and ~ 85% of the studies reported SmO2 measurement or equivalent obtained by spatially resolved spectroscopy method aiming to correct for light scattering. One study utilized a TD-NIRS oximeter, while nine studies utilized an FD-NIRS oximeter. FD-NIRS in skeletal muscle is less sensitive to superficial layer while TD-NIRS can disentangle scattering from absorption information. As a result, both methods improve the accuracy of the muscle haemodynamics assessment. The other CW-NIRS studies, assuming constant path length, potentially lead to errors in estimating of muscle metabolic changes due to incorrect assumptions about tissue scattering. This in turn underestimates actual muscle oxygenation/deoxygenation as compared with measurements obtained by real-time path-length determination by either TD-NIRS or FD-NIRS [209, 210]. Therefore, the implementation of TD- and FD-NIRS approaches on dedicated wearable sensors remains to be achieved. This may be an evolving topic for the next generation of NIRS devices.
It is well known that the relatively high attenuation of the near-infrared light in muscle measurements is due to (i) the two main chromophores (Hb and Mb); (ii) light scattering; and (iii) other molecules (mainly skin melanin, water, lipids of the adipose tissue, intramuscular lipids and cytochrome c oxidase) [2]. Adipose tissue greatly attenuates the signals; correcting for its attenuation has been suggested on the basis of the strength of the relationship between NIRS-derived measurements and the adipose tissue thickness [211, 212]. It remains difficult to discriminate between Hb and Mb spectra as they are very similar in the near-infrared range [4]. Therefore, the contribution of Mb desaturation to the NIRS signal during exercise remains unclear [213]. Subjects with darker skin tones have significantly larger and more concentrated melanosomes, which increase the absorption cross-section of melanin, resulting in enhanced light absorption. Some of the studies included in Table S1 reported the impossibility to perform oximetry measurements on subjects with a dark pigmented skin. More recently, the robustness of two commercial CW-NIRS oximeters (using four or five wavelengths) to variations in skin pigmentation was evaluated using a tissue-simulating phantom; unexpectedly increasing melanin content decreased the O2 saturation values displayed by both devices [214]. Differences in melanin content must be taken into account when measuring SmO2 values [215]. In addition to the topics included in Table 3 entitled “Pressing issues to improve the quality in muscle oximetry for sports science” of our 2018 review [4], we suggest reporting skin colour using the Fitzpatrick skin type classification scale.
4.2 Meaningful and Promising Applications of Muscle Oxygenation Measures
Monitoring muscle oxygenation during exercise, as assessed by wearable NIRS, is becoming a common physiological marker of internal burden [9]. Muscle O2 saturation measured by wearable NIRS was found to have similar reliability to O2 uptake and heart rate, across exercise intensities, suggesting that it is suitable for daily use as a non-invasive method of monitoring internal burden alongside other regular systemic physiological variables [216]. Thus, NIRS can become one of the primary monitoring tools in practice, such as heart rate or mechanical power monitoring during endurance exercise. Muscle oxygenation offers a distinct viewpoint on the physiological response of the muscle site being investigated in conjunction with, or in comparison with, other systemic physiological responses observed in different exercise contexts and through different types of training. Currently, wearable NIRS measurements using either PortaMon or Moxy, demonstrated moderate-to-excellent relative reliability scores, and CV as low as 10% for SmO2 [217, 218]. Hence, NIRS is a functional tool in which skeletal muscle oxygenation data can be viewed in ‘real time’, complementing external power and heart rate data, allowing coaches to make more informed decisions [4]. Real-time potential can guide athletes’ muscle performance during training and competition, by providing real-time feedback on the metabolic status of the working muscle groups during exercise. This information can be advantageous for handling large datasets by utilizing machine learning models in predicting internal burden [219] and may be suitable for diagnosing muscle fatigue during long-term monitoring. There is probably also a need to establish individual reference values of muscle oxygenation during standardized tests, such as during incremental exercise to exhaustion and an isometric contraction test inducing total arterial occlusion. These two tests performed regularly according to the training programme make it possible to assess the relative evolution of the metabolic state of the trained muscles.
VO2max, ventilatory/lactate thresholds and maximum lactate steady state are basic physiological evaluations related to endurance performance. The minimum level of oxygenation and the magnitude of O2 extraction in the biceps brachii, latissimus dorsi and vastus lateralis muscles were found to be more predictive of canoe kayak performance than VO2max [156] in a short endurance event (200 m). While measuring muscle oxygenation appears to be useful, this finding needs to be replicated and tested in other sports involving different muscle groups. Submaximal to maximal exercise intensities are prescribed to optimize training and improve cardiovascular fitness and endurance using specific intensity zones (moderate, heavy and severe). Typically, this is done by identifying ventilatory or blood lactate thresholds, and critical power/speed can be used. The determination of thresholds or zones of exercise intensity domains using muscle oxygenation variables as an alternative to pulmonary gas exchange or blood lactate methods has been extensively tested in the last 5 years [73, 79, 91, 115, 145, 179]. However, the proximity of the deoxygenation breakpoint to the respiratory compensation point remains controversial [220,221,222]. Yogev et al. [222] reported that the deoxygenation breakpoint derived from a wearable NIRS sensor over the vastus lateralis did not differ from the respiratory compensation point in a group of trained male and female cyclists with heterogenous fitness. A recent systematic review and meta-analysis on 15 studies indicated that the reliability (based on intraclass correlation coefficient) between the first ventilatory or lactate threshold and the first muscle oxygenation threshold was 0.53 (based on data from 3 studies), while the second threshold was 0.80 [223]. These moderate to good reliability values for the determination of the second ventilatory and lactate thresholds with the NIRS device are likely due to significant variations between the methods of determination, the ability to detect the first threshold and other factors to be investigated (e.g. muscle region, adipose tissue influence). This appears similar to the decades-long debate surrounding the ventilatory profile reported with two inflection points during graded exercise testing [224]. Concerns about using these indices interchangeably are raised by the considerable individual variability. Apart from the measurement of metabolic breakpoints, contextual observations of muscle oxygenation responses and their repeatability may still provide practitioners with pertinent data to comprehend a specific athlete's response to endurance exercise. Muscle oxygenation provides athletes with a targeted measurement of muscle performance during exercise.
An approach to appraise the determinants and limitations of endurance exercise performance is by identifying the work rate that corresponds to the highest steady-state metabolic rate. The critical power/speed model, and the work rate at maximal lactate steady state are widely accepted approaches for this purpose. Recent research [225] has found that, during whole-body exercise, dynamic muscle oxygenation profiles which describe the balance between muscle O2 supply and metabolic O2 demand are a valuable physiological surrogate for critical metabolic rate. This refers to the highest exercise intensity at which a plateau in the muscle oxygen saturation rate (zero-slope) is reached [224]. Critical oxygenation, as an alternative to critical power/speed, may provide insight into the causes of muscle performance and fatigue in various sporting activities, through its determination at different workloads and durations, based on a physiological framework [226]. It is worth noting that critical metabolic rate, determined by the balance between muscle O2 supply and metabolic demand in quadriceps and forearm muscle sites, predicts the time of exhaustion during continuous and intermittent exercise [227].
Deoxygenation breakpoint measurements [222, 228] may allow better categorization of the training stimulus into zones of exercise intensity (e.g. percentage of one-repetition maximum for the load prescription or percentage of maximum voluntary contraction) for specific muscle groups in resistance/strength training, thereby favouring the desired local muscle adaptations. Strength training research is frequently interested in how and when muscle is activated/recruited during movement, when muscle fatigue occurs and how different neuromuscular mechanisms contribute to force production. Surface electromyography (sEMG) is the most commonly used measurement for these purposes. However, evaluating the effects of resistance training on skeletal muscles might be characterized by a lower muscle oxygenation response due to a restriction in blood supply to the primary muscle in relation to the number of repetitions and the load, which induces increased intramuscular mechanical pressure. A recent systematic review [229] examined baseline and end-points values acquired by NIRS during resistance exercise in healthy persons. SmO2, the most studied variable with NIRS devices (Moxy and Portamon), decreases as an acute response to muscular strength exercise, according to the four included lower limb studies using squat-like exercise modalities.
Another relevant variable to discuss is that in some selected articles we have observed that data reported by NIRS during exercise or training interventions have been introduced by adding manipulations in the form of venous and arterial occlusion to assess both muscle blood flow and mVO2 [41, 117, 138, 167, 169, 190]. Muscle oxidative capacity is the maximum rate at which the muscle can utilize O2 to meet the energy demand of exercise [230]. With NIRS, the initial rate of muscle deoxygenation during transient arterial occlusion is a direct measure of mVO2, a reliable indicator of muscle oxidative capacity [2]. In the present review, 25 studies quantified mVO2 (see Table S1 and Results). Venous occlusion has also been used with NIRS to provide measurements of muscle blood flow during or after repeated high-intensity exercises [231]. Incorporating this simple test both before and after intervention [232] provides additional insight into changes in muscle haemodynamics and metabolic activity as a result of training. During and after exercise, repeated transient arterial occlusions can yield sequential mVO2 measurements [82, 232]. Therefore, for the purpose of determining changes in mitochondrial capacity, the recovery of mVO2 values after exercise provides information that is essentially the same as that obtained from the kinetics of phosphocreatine levels after exercise [233, 234]. These possibilities offered by NIRS may be useful for practitioners.
The NIRS tests, evaluating potential physiological responses that may contribute in some way to the ergogenic effects, are the final aspect of the data collected in the present review. Coaches and athletes are looking for effective strategies to enhance performance and speed up recovery. The NIRS device was employed to find out whether the ergogenic aid would affect the balance between local O2 delivery and muscle O2 utilization in skeletal muscle and thus improve performance. Ergogenic aids are any of the methods, including dietary, pharmacological and physiological ones, that can improve performance. Cherry polyphenols [67] and mango leaf extract rich in mangiferin [53,54,55] may be useful nutritional aids for improving muscular endurance performance. Contrarily, this does not appear to be the case for acute dietary nitrate supplementation [33, 63, 75, 76, 158, 183, 190], citrulline [190, 196], l-arginine, red spinach extract [121], dark chocolate [181] and caffeine [189]. Additionally, mechanical ergogenic aids like compression clothing [18, 19, 31, 107] tend to increase blood flow and muscle oxygenation, especially at rest. With regard to physiological ergogenic aids, techniques like blood flow restriction [12, 14, 147,148,149, 191, 192], voluntary hypoventilation at low lung volume [17, 34, 88, 89, 151], electrical muscle stimulation [138, 144, 173, 184, 197] and dynamic stretching [193] typically alter the muscle oxygenation response; however, the effects of other interventions like photobiomodulation therapy [105] or immersion in water [21, 85, 94, 95] are less certain. All these data, obtained using different protocols, show that the sensitivity of the NIRS technology is being study here to assess the true value of a particular ergogenic aid. It is interesting to note that they show how some ergogenic aids can modulate blood volume (strictly related to tHb) variations and hence the balance between O2 delivery and utilization (and hence O2 extraction) within the interrogated region.
4.3 Unanswered Questions and Future Research
Because the current NIRS technique only allows for the analysis of a small volume of muscle (superficially, with the average depth reaching only half the distance between the light source and the detector, e.g. 1.5–3 cm approximately), it is currently not indicative of what is happening in the other working muscles. We may develop NIRS equipment that can be integrated into sports clothing and can be utilized outside of the laboratory using energy harvesting technologies (solar batteries, sunlight as a light source, and bandpass filters). This equipment would be used to map whole-body activity. The surface electromyogram sensors should be utilized in conjunction with the NIRS equipment to provide an overall picture of changes in muscle function during exercise [235]. In addition to the haemodynamics and metabolic data provided by NIRS, we also gain knowledge about muscle-level activation using surface electromyography, and the effect of motion from accelerometers on these two signals.
4.4 Limitations and Strengths
Although this review was conducted according to the PRISMA guidelines updated in 2020 [10] and with standardized critical appraisal, several factors limit our ability to draw strong conclusions.
A common limitation of all systematic reviews is that some articles may be overlooked. To overcome this problem, we conducted an extensive search using sensitive search criteria and synonyms. Another limitation of this systematic review is that the quality of the study varied widely. For instance, details of participant selection were unclear in most articles. There was a large heterogeneity in the study populations examined. The number of participants was generally small (n < 20 in 72% of the studies), which limits the generalizability of the obtained results. Several studies did not follow a standardized research protocol. There was also considerable heterogeneity in study design and outcomes.
While the included studies mainly focused on athletes at the national/international level, differences between the included sports in terms of training, remuneration and other relevant parameters need to be taken into account. Although systematic reviews are generally considered to be of as “a high quality of evidence”, we believe that the reported findings are of moderate quality, taking into account the limitations mentioned above. A strength of the present review is that it provides an update of our previous systematic review of the literature on muscle oximetry in sports science [4]. Given the heterogeneous nature of the reported studies and the wide variation in the methods used, it is not possible to draw general conclusions about the role of muscle oximetry in sports science.
5 Conclusions and Prospects
There is no doubt that NIRS is a useful method for evaluating muscle adaptation effects in studies involving intermittent or continuous aerobic/anaerobic exercise and strength training, and it can therefore be utilized by physical trainers to guide training or recovery processes, and to test many potential interventions (the so-called ergogenic aids) favouring changes in the balance of O2 delivery and O2 utilization as a key factor in muscle performance.
The future of muscle oximetry in sports science is closely related to the instrumental development. In terms of miniaturization, CW-NIRS technology is the most convenient method. Different lightweight (about 20 g), compact, smartphone-controllable and wearable multi-distance CW-NIRS-based oximeters with Bluetooth connection up to 150 m and on-board data collection (up to 50 h) are commercially available [236]. The novel CW-NIRS oximeter (Train.Red Plus, Artinis Medical Systems) also includes very useful haptic feedback, so that the subject can feel a buzzer on the skin during the exercise [237]. The CW-NIRS technology has been incorporated into OctaMon M (Artinis Medical Systems), the only commercial imager dedicated to muscle studies (50 Hz sampling time; four source–detector distances in the range 25–40 mm) using eight measurement points [238].
Multi-modal techniques based on two types of sensors, sEMG and CW-NIRS, have been developed to provide a more detailed evaluation of muscle activity, as the information obtained by each sensor is based on different phenomena induced by muscle activity. The fusion of these wearable technologies in sporting garments can provide an objective assessment of the quality and the quantity of the muscle activity, as well as the continuous monitoring of exercise programs. A new wearable integrated quadriceps muscle oximetry/sEMG system adopting smart textiles for sEMG has been recently tested under resting and dynamic conditions (treadmill running and resistance exercise) [239]. More recently, the sEMG and CW-NIRS signals have been measured during isometric ramp contraction of the forearm and cycling exercise of the vastus lateralis muscle with stepped increments of the load using a wireless multi-layered sensor [240]. More complex devices using FD-NIRS and TD-NIRS allow the monitoring of both absorption and scattering and can provide more accurate signals under a wider range of conditions [2, 235]. The first commercial FD-NIRS and TD-NIRS systems were introduced in 1998 and 2003, respectively, but their high price limited the application. Recently, a commercial two-wavelength battery-operated wireless wearable TD-NIRS system, which fits into a backpack (3 kg) and performs measurements on the brain and muscle tissue of freely moving subjects using a 10-g optode, has been introduced [241]. This system provides SmO2 as well as absolute concentrations of O2Hb and HHb at 20 Hz.
Over the last 20 years, diffuse correlation spectroscopy (DCS) has emerged as a versatile, non-invasive method for the continuous measurement of microvascular blood flow as a tissue blood index [242]. DCS uses the temporal fluctuations of diffusely reflected light to quantify the motion of tissue scatterers (primarily the velocity of red blood cells). Application to ramp-incremental cycling exercise has been successfully demonstrated utilizing a complex and expensive commercial hybrid system equipped to employ FD-NIRS and DCS [243]. The technical limitations of this hybrid technology remain an important barrier to wider adoption.
NIRS technology continues to evolve, and the nature of this approach provides distinct advantages when studying human muscle during exercise. Despite current limitations, which are largely confined to limited penetration depth, low spatial resolution and interference from adipose tissue thickness [1], we believe that the feasibility and success of applying muscle oximetry in sports science have been well documented and encourage its routine use in sports science and medicine [4, 6, 7, 9].
References
Barstow TJ. Understanding near infrared spectroscopy and its application to skeletal muscle research. J Appl Physiol (1985). 2019;126(5):1360–76. https://doi.org/10.1152/japplphysiol.00166.2018.
Ferrari M, Muthalib M, Quaresima V. The use of near-infrared spectroscopy in understanding skeletal muscle physiology: recent developments. Philos Trans A Math Phys Eng Sci. 1955;2011(369):4577–90. https://doi.org/10.1098/rsta.2011.0230.
Hamaoka T, McCully KK. Review of early development of near-infrared spectroscopy and recent advancement of studies on muscle oxygenation and oxidative metabolism. J Physiol Sci. 2019;69(6):799–811. https://doi.org/10.1007/s12576-019-00697-2.
Perrey S, Ferrari M. Muscle oximetry in sports science: a systematic review. Sports Med. 2018;48(3):597–616. https://doi.org/10.1007/s40279-017-0820-1.
Salvatore SS, Zelenski KN, Perkins RK. Age-related changes in skeletal muscle oxygen utilization. J Funct Morphol Kinesiol. 2022;7(4):87. https://doi.org/10.3390/jfmk7040087.
Cornelis N, Chatzinikolaou P, Buys R, Fourneau I, Claes J, Cornelissen V. The use of near infrared spectroscopy to evaluate the effect of exercise on peripheral muscle oxygenation in patients with lower extremity artery disease: a systematic review. Eur J Vasc Endovasc Surg. 2021;61(5):837–47. https://doi.org/10.1016/j.ejvs.2021.02.008.
Tuesta M, Yáñez-Sepúlveda R, Verdugo-Marchese H, Mateluna C, Alvear-Ordenes I. Near-infrared spectroscopy used to assess physiological muscle adaptations in exercise clinical trials: a systematic review. Biology. 2022;11(7):1073. https://doi.org/10.3390/biology11071073.
Sanni AA, McCully KK. Interpretation of near-infrared spectroscopy (NIRS) signals in skeletal muscle. J Funct Morphol Kinesiol. 2019;4(2):28. https://doi.org/10.3390/jfmk4020028.
Perrey S. Muscle oxygenation unlocks the secrets of physiological responses to exercise: time to exploit it in the training monitoring. Front Sports Act Liv. 2022;4:864825. https://doi.org/10.3389/fspor.2022.864825.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. https://doi.org/10.1136/bmj.n71.
Morawetz D, Dünnwald T, Faulhaber M, Gatterer H, Höllrigl L, Raschner C, et al. Can hyperoxic preconditioning in normobaric hypoxia (3500 m) improve all-out exercise performance in highly skilled skiers? A randomized crossover study. Int J Sports Physiol Perform. 2020;15(3):346–53. https://doi.org/10.1123/ijspp.2019-0016.
Fortin JF, Billaut F. Blood-flow restricted warm-up alters muscle hemodynamics and oxygenation during repeated sprints in American football players. Sports (Basel). 2019;7(5):121. https://doi.org/10.3390/sports7050121.
Girard O, Brocherie F, Ihsan M. Does ischemic pre-conditioning during the recovery period between two successive matches preserve physical performance in badminton doubles players? Int J Racket Sports Sci. 2020;2(2):1–8.
Valenzuela PL, Sánchez-Martínez G, Torrontegi E, Vázquez-Carrión J, González M, Montalvo Z, et al. Acute responses to on-court repeated-sprint training performed with blood flow restriction versus systemic hypoxia in elite badminton athletes. Int J Sports Physiol Perform. 2019;14(9):1280–7. https://doi.org/10.1123/ijspp.2018-0878.
Delextrat A, Gruet M, Bieuzen F. Effects of small-sided games and high-intensity interval training on aerobic and repeated sprint performance and peripheral muscle oxygenation changes in elite junior basketball players. J Strength Cond Res. 2018;32(7):1882–91. https://doi.org/10.1519/JSC.0000000000002570.
Guan S, Lin N, Yin Y, Liu H, Liu L, Qi L. The effects of inter-set recovery time on explosive power, electromyography activity, and tissue oxygenation during plyometric training. Sensors (Basel). 2021;21(9):3015. https://doi.org/10.3390/s21093015.
Lapointe J, Paradis-Deschênes P, Woorons X, Lemaître F, Billaut F. Impact of hypoventilation training on muscle oxygenation, myoelectrical changes, systemic [K+], and repeated-sprint ability in basketball players. Front Sports Act Liv. 2020;2:29. https://doi.org/10.3389/fspor.2020.00029.
O’Riordan SF, McGregor R, Halson SL, Bishop DJ, Broatch JR. Sports compression garments improve resting markers of venous return and muscle blood flow in male basketball players. J Sport Health Sci. 2023;12(4):513–22. https://doi.org/10.1016/j.jshs.2021.07.010.
Paulauskas R, Kamarauskas P, Nekriošius R, Bigwood NM. Physical and physiological response to different modes of repeated sprint exercises in basketball players. J Hum Kinet. 2020;31:72–919. https://doi.org/10.2478/hukin-2019-0100.
Baláš J, Kodejška J, Krupková D, Hannsmann J, Fryer S. Reliability of near-infrared spectroscopy for measuring intermittent handgrip contractions in sport climbers. J Strength Cond Res. 2018;32(2):494–501. https://doi.org/10.1519/JSC.0000000000002341.
Baláš J, Kodejška J, Krupková D, Giles D. Males benefit more from cold water immersion during repeated handgrip contractions than females despite similar oxygen kinetics. J Physiol Sci. 2020;70(1):13. https://doi.org/10.1186/s12576-020-00742-5.
Baláš J, Gajdošík J, Giles D, Fryer S, Krupková D, Brtník T, et al. Isolated finger flexor vs. exhaustive whole-body climbing tests? How to assess endurance in sport climbers? Eur J Appl Physiol. 2021;121(5):1337–48. https://doi.org/10.1007/s00421-021-04595-7.
Dindorf C, Bartaguiz E, Dully J, Sprenger M, Becker S, Fröhlich M, et al. In vivo monitoring of acute and intermittent fatigue in sport climbing using near-infrared spectroscopy wearable biosensors. Sports (Basel). 2023;11(2):37. https://doi.org/10.3390/sports11020037.
Feldmann AM, Erlacher D, Pfster S, Lehmann R. Muscle oxygen dynamics in elite climbers during finger-hang tests at varying intensities. Sci Rep. 2020;10:3040. https://doi.org/10.1038/s41598-020-60029-y.
Feldmann A, Lehmann R, Wittmann F, Wolf P, Balas J, Erlacher D. Acute effect of high-intensity climbing on performance and muscle oxygenation in elite climbers. J Sci Sport Exerc. 2021;4:145–55. https://doi.org/10.1007/s42978-021-00139-9.
Fryer S, Stone KJ, Sveen J, Dickson T, España-Romero V, Giles D, et al. Differences in forearm strength, endurance, and hemodynamic kinetics between male boulderers and lead rock climbers. Eur J Sport Sci. 2017;17(9):1177–83. https://doi.org/10.1080/17461391.2017.1353135.
Fryer S, Giles D, Bird E, Stone K, Paterson C, Baláš J, et al. New Zealand blackcurrant extract enhances muscle oxygenation during repeated intermittent forearm muscle contractions in advanced and elite rock climbers. Eur J Sport Sci. 2021;21(9):1290–8. https://doi.org/10.1080/17461391.2020.1827048.
Gajdošík J, Baláš J, Krupková D, Psohlavec L, Draper N. Effect of climbing speed on pulmonary oxygen uptake and muscle oxygen saturation dynamics in the finger flexors. Int J Sports Physiol Perform. 2022;17(2):176–84. https://doi.org/10.1123/ijspp.2021-0110.
Limmer M, de Marées M, Roth R. Effects of forearm compression sleeves on muscle hemodynamics and muscular strength and endurance parameters in sports climbing: a randomized, controlled crossover trial. Front Physiol. 2022;13:888860. https://doi.org/10.3389/fphys.2022.888860.
Nolan J, Mclennan PL, Peoples GE. Forearm isometric fatigue-resistance is enhanced in rock climbers compared to power lifters and aerobically-trained athletes. J Sports Med Phys Fitness. 2020;60(8):1057–64. https://doi.org/10.23736/S0022-4707.20.10542-5.
Antunes A, Domingos C, Diniz L, Monteiro CP, Espada MC, Alves FB, et al. The relationship between VO2 and muscle deoxygenation kinetics and upper body repeated sprint performance in trained judokas and healthy individuals. Int J Environ Res Public Health. 2022;19(2):861. https://doi.org/10.3390/ijerph19020861.
de Oliveira GV, Nascimento LADD, Volino-Souza M, Mesquita JS, Alvares TS. Beetroot-based gel supplementation improves handgrip strength and forearm muscle O2 saturation but not exercise tolerance and blood volume in jiu-jitsu athletes. Appl Physiol Nutr Metab. 2018;43(9):920–7. https://doi.org/10.1139/apnm-2017-0828.
de Oliveira GV, do Nascimento LAD, Volino-Souza M, do Couto Vellozo O, Alvares TS. A single oral dose of beetroot-based gel does not improve muscle oxygenation parameters, but speeds up handgrip isometric strength recovery in recreational combat sports athletes. Biol Sport. 2020;37(1):93–99. https://doi.org/10.5114/biolsport.2020.92518.
Woorons X, Dupuy O, Mucci P, Millet GP, Pichon A. Cerebral and muscle oxygenation during repeated shuttle run sprints with hypoventilation. Int J Sports Med. 2019;40(6):376–84. https://doi.org/10.1055/a-0836-9011.
Nybäck L, Glännerud C, Larsson G, Weitzberg E, Shannon OM, McGawley K. Physiological and performance effects of nitrate supplementation during roller-skiing in normoxia and normobaric hypoxia. Nitr Oxide. 2017;70:1–8. https://doi.org/10.1016/j.niox.2017.08.001.
Øfsteng S, Sandbakk Ø, van Beekvelt M, Hammarström D, Kristoffersen R, Hansen J, et al. Strength training improves double-poling performance after prolonged submaximal exercise in cross-country skiers. Scand J Med Sci Sports. 2018;28(3):893–904. https://doi.org/10.1111/sms.12990.
Stöggl T, Born DP. Near infrared spectroscopy for muscle specific analysis of intensity and fatigue during cross-country skiing competition—a case report. Sensors. 2021;21(7):2535. https://doi.org/10.3390/s21072535.
Aebi MR, Willis SJ, Girard O, Borrani F, Millet GP. Active preconditioning with blood flow restriction or/and systemic hypoxic exposure does not improve repeated sprint cycling performance. Front Physiol. 2019;10:1393. https://doi.org/10.3389/fphys.2019.01393.
Batterson PM, Norton MR, Hetz SE, Rohilla S, Lindsay KG, Subudhi AW, et al. Improving biologic predictors of cycling endurance performance with near-infrared spectroscopy derived measures of skeletal muscle respiration: E pluribus unum. Physiol Rep. 2020;8(2):e14342. https://doi.org/10.14814/phy2.14342.
Breese BC, Poole DC, Okushima D, Bailey SJ, Jones AM, Kondo N, et al. The effect of dietary nitrate supplementation on the spatial heterogeneity of quadriceps deoxygenation during heavy-intensity cycling. Physiol Rep. 2017;5(14):e13340. https://doi.org/10.14814/phy2.13340.
Broatch JR, Bishop DJ, Halson S. Lower limb sports compression garments improve muscle blood flow and exercise performance during repeated-sprint cycling. Int J Sports Physiol Perform. 2018;13(7):882–90. https://doi.org/10.1123/ijspp.2017-0638.
Brock K, Antonellis P, Black MI, DiMenna FJ, Vanhatalo A, Jones AM, et al. Improvement of oxygen-uptake kinetics and cycling performance with combined prior exercise and fast start. Int J Sports Physiol Perform. 2018;13(3):305–12. https://doi.org/10.1123/ijspp.2016-0557.
Broeder CE, Flores V, Julian B, Wojan F, Tauber R, Schubert L, et al. Nitric oxide enhancement supplement containing beet nitrite and nitrate benefits high intensity cycle interval training. Curr Res Physiol. 2021;4:183–91. https://doi.org/10.1016/j.crphys.2021.07.002.
Cayot TE, Robinson SG, Davis LE, Bender PA, Thistlethwaite JR, Broeder CE, et al. Estimating the lactate threshold using wireless near-infrared spectroscopy and threshold detection analyses. Int J Exerc Sci. 2021;14(4):284–94.
Cheng CF, Hsu WC, Kuo YH, Chen TW, Kuo YC. Acute effect of inspiratory resistive loading on sprint interval exercise performance in team-sport athletes. Respir Physiol Neurobiol. 2020;282:103531. https://doi.org/10.1016/j.resp.2020.103531.
Cocking S, Ihsan M, Jones H, Hansen C, Timothy Cable N, Thijssen DHJ, et al. Repeated sprint cycling performance is not enhanced by ischaemic preconditioning or muscle heating strategies. Eur J Sport Sci. 2021;21(2):166–75. https://doi.org/10.1080/17461391.2020.1749312.
Colosio AL, Caen K, Bourgois JG, Boone J, Pogliaghi S. Metabolic instability vs fibre recruitment contribution to the V̇O2 slow component in different exercise intensity domains. Pflugers Arch. 2021;473(6):873–82. https://doi.org/10.1007/s00424-021-02573-8.
Combes A, Dekerle J, Bougault V, Daussin FN. Physiological comparison of intensity-controlled, isocaloric intermittent and continuous exercise. Eur J Sport Sci. 2018;18(10):1368–75. https://doi.org/10.1080/17461391.2018.1491627.
Crum EM, Barnes MJ, Stannard SR. Multiday pomegranate extract supplementation decreases oxygen uptake during submaximal cycling exercise, but cosupplementation with N-acetylcysteine negates the effect. Int J Sport Nutr Exerc Metab. 2018;28(6):586–92. https://doi.org/10.1123/ijsnem.2017-0407.
Espinosa-Ramírez M, Moya-Gallardo E, Araya-Román F, Riquelme-Sánchez S, Rodriguez-García G, Reid WD, et al. Sex-differences in the oxygenation levels of intercostal and vastus lateralis muscles during incremental exercise. Front Physiol. 2021;12:738063. https://doi.org/10.3389/fphys.2021.738063.
Fennell CRJ, Hopker JG. The acute physiological and perceptual effects of recovery interval intensity during cycling-based high-intensity interval training. Eur J Appl Physiol. 2021;121(2):425–34. https://doi.org/10.1007/s00421-020-04535-x.
Formenti F, Dockerill C, Kankanange L, Zhang L, Takaishi T, Ishida K. The effect of pedaling cadence on skeletal muscle oxygenation during cycling at moderate exercise intensity. Int J Sports Med. 2019;40(5):305–11. https://doi.org/10.1055/a-0835-6286.
Gelabert-Rebato M, Wiebe JC, Martin-Rincon M, Gericke N, Perez-Valera M, Curtelin D, et al. Mangifera indica L. leaf extract in combination with luteolin or quercetin enhances VO2peak and peak power output, and preserves skeletal muscle function during ischemia–reperfusion in humans. Front Physiol. 2018;9:740. https://doi.org/10.3389/fphys.2018.00740.
Gelabert-Rebato M, Martin-Rincon M, Galvan-Alvarez V, Gallego-Selles A, Martinez-Canton M, Vega-Morales T, et al. A single dose of the mango leaf extract Zynamite® in combination with quercetin enhances peak power output during repeated sprint exercise in men and women. Nutrients. 2019;11(11):2592. https://doi.org/10.3390/nu11112592.
Gelabert-Rebato M, Wiebe JC, Martin-Rincon M, Galvan-Alvarez V, Curtelin D, Perez-Valera M, et al. Enhancement of exercise performance by 48 hours, and 15-day supplementation with mangiferin and luteolin in men. Nutrients. 2019;11(2):344. https://doi.org/10.3390/nu11020344.
Goulding RP, Roche DM, Marwood S. Effect of hyperoxia on critical power and V˙O2 kinetics during upright cycling. Med Sci Sports Exerc. 2020;52(5):1041–9. https://doi.org/10.1249/MSS.0000000000002234.
Griffin PJ, Ferguson RA, Gissane C, Bailey SJ, Patterson SD. Ischemic preconditioning enhances critical power during a 3 minute all-out cycling test. J Sports Sci. 2018;36(9):1038–43. https://doi.org/10.1080/02640414.2017.1349923.
Hopker JG, O’Grady C, Pageaux B. Prolonged constant load cycling exercise is associated with reduced gross efficiency and increased muscle oxygen uptake. Scand J Med Sci Sports. 2017;27(4):408–17. https://doi.org/10.1111/sms.12673.
Horiuchi M, Nishida A, Dobashi S, Koyama K. Comparisons between normobaric normoxic and hypoxic recovery on post-exercise hemodynamics after sprint interval cycling in hypoxia. Front Physiol. 2022;13:843574. https://doi.org/10.3389/fphys.2022.843574.
Hovorka M, Prinz B, Simon D, Zöger M, Rumpl C, Nimmerichter A. Longitudinal alterations of pulmonary V̇O2 on-kinetics during moderate-intensity exercise in competitive youth cyclists are related to alterations in the balance between microvascular O2 distribution and muscular O2 utilization. Front Sports Act Liv. 2022;4:982548. https://doi.org/10.3389/fspor.2022.982548.
Iannetta D, Qahtani A, Mattioni Maturana F, Murias JM. The near-infrared spectroscopy-derived deoxygenated haemoglobin breaking-point is a repeatable measure that demarcates exercise intensity domains. J Sci Med Sport. 2017;20(9):873–7. https://doi.org/10.1016/j.jsams.2017.01.237.
Inglis EC, Iannetta D, Keir DA, Murias JM. Training-induced changes in the respiratory compensation point, deoxyhemoglobin break point, and maximal lactate steady state: evidence of equivalence. Int J Sports Physiol Perform. 2020;15(1):119–25. https://doi.org/10.1123/ijspp.2019-0046.
Kent GL, Dawson B, Cox GR, Abbiss CR, Smith KJ, Croft KD, et al. Effect of dietary nitrate supplementation on thermoregulatory and cardiovascular responses to submaximal cycling in the heat. Eur J Appl Physiol. 2018;118(3):657–68. https://doi.org/10.1007/s00421-018-3809-z.
Jo E, Fischer M, Auslander AT, Beigarten A, Daggy B, Hansen K, et al. The effects of multi-day vs. single pre-exercise nitrate supplement dosing on simulated cycling time trial performance and skeletal muscle oxygenation. J Strength Cond Res. 2019;33(1):217–24. https://doi.org/10.1519/JSC.0000000000001958.
Mattioni Maturana F, Peyrard A, Temesi J, Millet GY, Murias JM. Faster V̇O2 kinetics after priming exercises of different duration but same fatigue. J Sports Sci. 2018;36(10):1095–102. https://doi.org/10.1080/02640414.2017.1356543.
McLay KM, Murias JM, Paterson DH. Similar pattern of change in V̇O2 kinetics, vascular function, and tissue oxygen provision following an endurance training stimulus in older and young adults. Am J Physiol Regul Integr Comp Physiol. 2017;312(4):R467–76. https://doi.org/10.1152/ajpregu.00399.2016.
Morgan PT, Barton MJ, Bowtell JL. Montmorency cherry supplementation improves 15-km cycling time-trial performance. Eur J Appl Physiol. 2019;119(3):675–84. https://doi.org/10.1007/s00421-018-04058-6.
Nimmerichter A, Breese BC, Prinz B, Zoeger M, Rumpl C, Williams CA. Test–retest reliability of pulmonary oxygen uptake and muscle deoxygenation during moderate- and heavy-intensity cycling in youth elite-cyclists. J Sports Sci. 2020;38(21):2462–70. https://doi.org/10.1080/02640414.2020.1792115.
O’Grady C, Passfield L, Hopker JG. Variability in submaximal self-paced exercise bouts of different intensity and duration. Int J Sports Physiol Perform. 2021;16(12):1824–33. https://doi.org/10.1123/ijspp.2020-0785.
Oueslati F, Boone J, Tabka Z, Ahmaidi S. Respiratory and locomotor muscle implications on the VO2 slow component and the VO2 excess in young trained cyclists. Respir Physiol Neurobiol. 2017;239:1–9. https://doi.org/10.1016/j.resp.2017.01.012.
Porter MS, Reed K, Jones B. The use of acute oxygen supplementation upon muscle tissue saturation during repeat sprint cycling. J Hum Sport Exerc. 2020;17(1):93–104. https://doi.org/10.14198/jhse.2022.171.10.
Raberin A, Elmer J, Willis SJ, Richard T, Vernillo G, Iaia FM, et al. The oxidative-glycolytic balance influenced by sprint duration is key during repeated sprint in hypoxia. Med Sci Sports Exerc. 2023;55(2):245–54. https://doi.org/10.1249/MSS.0000000000003042.
Raleigh C, Donne B, Fleming N. Association between different non-invasively derived thresholds with lactate threshold during graded incremental exercise. Int J Exerc Sci. 2018;11(4):391–403.
Rasica L, Inglis EC, Iannetta D, Soares RN, Murias JM. Fitness level- and sex-related differences in macrovascular and microvascular responses during reactive hyperemia. Med Sci Sports Exerc. 2022;54(3):497–506. https://doi.org/10.1249/MSS.0000000000002806.
Rokkedal-Lausch T, Franch J, Poulsen MK, Thomsen LP, Weitzberg E, Kamavuako EN, et al. Chronic high-dose beetroot juice supplementation improves time trial performance of well-trained cyclists in normoxia and hypoxia. Nitr Oxide. 2019;85:44–52. https://doi.org/10.1016/j.niox.2019.01.011.
Rokkedal-Lausch T, Franch J, Poulsen MK, Thomsen LP, Weitzberg E, Kamavuako EN, et al. Multiple-day high-dose beetroot juice supplementation does not improve pulmonary or muscle deoxygenation kinetics of well-trained cyclists in normoxia and hypoxia. Nitr Oxide. 2021;111–112:37–44. https://doi.org/10.1016/j.niox.2021.03.006.
Rupp T, Saugy JJ, Bourdillon N, Millet GP. Brain–muscle interplay during endurance self-paced exercise in normobaric and hypobaric hypoxia. Front Physiol. 2022;13: 893872. https://doi.org/10.3389/fphys.2022.893872.
Saito A, Goda M, Yamagishi T, Kawakami Y. Riding posture affects quadriceps femoris oxygenation during an incremental cycle exercise in cycle-based athletes. Physiol Rep. 2018;6(16):e13832. https://doi.org/10.14814/phy2.13832.
Salas-Montoro JA, Mateo-March M, Sánchez-Muñoz C, Zabala M. Determination of second lactate threshold using near-infrared spectroscopy in elite cyclists. Int J Sports Med. 2022;43(8):721–8. https://doi.org/10.1055/a-1738-0252.
Shastri L, Alkhalil M, Forbes C, El-Wadi T, Rafferty G, Ishida K, et al. Skeletal muscle oxygenation during cycling at different power output and cadence. Physiol Rep. 2019;7(3):e13963. https://doi.org/10.14814/phy2.13963.
Shaw K, Singh J, Sirant L, Neary JP, Chilibeck PD. Effect of dark chocolate supplementation on tissue oxygenation, metabolism, and performance in trained cyclists at altitude. Int J Sport Nutr Exerc Metab. 2020;30(6):420–6. https://doi.org/10.1123/ijsnem.2020-0051.
Skovereng K, Ettema G, van Beekvelt M. The effect of cadence on shank muscle oxygen consumption and deoxygenation in relation to joint specific power and cycling kinematics. PLoS ONE. 2017;12(1):e0169573. https://doi.org/10.1371/journal.pone.0169573.
Stöcker F, Von Oldershausen C, Paternoster FK, Schulz T, Oberhoffer R. End-exercise ΔHHb/ΔVO2 and post-exercise local oxygen availability in relation to exercise intensity. Clin Physiol Funct Imaging. 2017;37(4):384–93. https://doi.org/10.1111/cpf.12314.
van der Zwaard S, van der Laarse WJ, Weide G, Bloemers FW, Hofmijster MJ, Levels K, et al. Critical determinants of combined sprint and endurance performance: an integrative analysis from muscle fiber to the human body. FASEB J. 2018;32(4):2110–23. https://doi.org/10.1096/fj.201700827R.
Wakabayashi H, Osawa M, Koga S, Li K, Sakaue H, Sengoku Y, et al. Effects of muscle cooling on kinetics of pulmonary oxygen uptake and muscle deoxygenation at the onset of exercise. Physiol Rep. 2018;6(21):e13910. https://doi.org/10.14814/phy2.13910.
Willis SJ, Alvarez L, Millet GP, Borrani F. Changes in muscle and cerebral deoxygenation and perfusion during repeated sprints in hypoxia to exhaustion. Front Physiol. 2017;8:846. https://doi.org/10.3389/fphys.2017.00846.
Willis SJ, Alvarez L, Borrani F, Millet GP. Oxygenation time course and neuromuscular fatigue during repeated cycling sprints with bilateral blood flow restriction. Physiol Rep. 2018;6(19):e13872. https://doi.org/10.14814/phy2.13872.
Woorons X, Mucci P, Aucouturier J, Anthierens A, Millet GP. Acute effects of repeated cycling sprints in hypoxia induced by voluntary hypoventilation. Eur J Appl Physiol. 2017;117(12):2433–43. https://doi.org/10.1007/s00421-017-3729-3.
Woorons X, Millet GP, Mucci P. Physiological adaptations to repeated sprint training in hypoxia induced by voluntary hypoventilation at low lung volume. Eur J Appl Physiol. 2019;119(9):1959–70. https://doi.org/10.1007/s00421-019-04184-9.
Yamaguchi K, Sumi D, Hayashi N, Ota N, Ienaga K, Goto K. Effects of combined hot and hypoxic conditions on muscle blood flow and muscle oxygenation during repeated cycling sprints. Eur J Appl Physiol. 2021;121(10):2869–78. https://doi.org/10.1007/s00421-021-04738-w.
Yogev A, Arnold J, Clarke D, Guenette JA, Spores BC, Koehle MS. Comparing the respiratory compensation point with muscle oxygen saturation in locomotor and non-locomotor muscles using wearable NIRS spectroscopy during whole-body exercise. Front Physiol. 2022;13:818733. https://doi.org/10.3389/fphys.2022.818733.
Zurbuchen A, Lanzi S, Voirol L, Trindade CB, Gojanovic B, Kayser B, et al. Fat oxidation kinetics is related to muscle deoxygenation kinetics during exercise. Front Physiol. 2020;11:571. https://doi.org/10.3389/fphys.2020.00571.
Costalat G, Coquart J, Castres I, Joulia F, Sirost O, Clua E, et al. The oxygen-conserving potential of the diving response: a kinetic-based analysis. J Sports Sci. 2017;35(7):678–87. https://doi.org/10.1080/02640414.2016.1183809.
Myers CM, Kim JS, Musilli M, McCully K, Florian JP. Effects of resting, consecutive, long-duration water immersions on neuromuscular endurance in well-trained males. Front Physiol. 2018;9:977. https://doi.org/10.3389/fphys.2018.00977.
Myers CM, Kim JS, McCuly KK, Florian JP. Effects of repeated, long-duration hyperoxic water immersions on neuromuscular endurance in well-trained males. Front Physiol. 2019;10:858. https://doi.org/10.3389/fphys.2019.00858.
Abaïdia AE, Cosyns S, Dupont G. Muscle oxygenation induced by cycling exercise does not accelerate recovery kinetics following exercise-induced muscle damage in humans: a randomized cross-over study. Respir Physiol Neurobiol. 2019;266:82–8. https://doi.org/10.1016/j.resp.2019.05.005.
Archiza B, Andaku DK, Caruso FCR, Bonjorno JC Jr, Oliveira CR, Ricci PA, et al. Effects of inspiratory muscle training in professional women football players: a randomized sham-controlled trial. J Sports Sci. 2018;36(7):771–80. https://doi.org/10.1080/02640414.2017.1340659.
Bonilla AAV, Timon R, Camacho-Cardeñosa A, Camacho-Cardeñosa M, Guerrero S, Olcina G. Fatigue increases in resting muscle oxygen consumption after a women’s soccer match. Int J Sports Med. 2020;41(13):e2–8. https://doi.org/10.1055/a-1177-0849.
Bujalance-Moreno P, Latorre Román P, Ramirez-Campillo R, Martínez-Amat A, Pinillos F. Acute responses to 4 vs. 4 small-sided games in football players. Kinesiology. 2020;52:46–53. https://doi.org/10.26582/k.52.1.7.
Caruso FR, Archiza B, Andaku DK, Trimer R, Bonjorno-Junior JC, de Oliveira CR, et al. Effects of acute inspiratory loading during treadmill running on cerebral, locomotor and respiratory muscle oxygenation in women soccer players. Respir Physiol Neurobiol. 2020;281:103488. https://doi.org/10.1016/j.resp.2020.103488.
Michailidis Y, Chatzimagioglou A, Mikikis D, Ispirlidis I, Metaxas T. Maximal oxygen consumption and oxygen muscle saturation recovery following repeated anaerobic sprint test in youth soccer players. J Sports Med Phys Fitness. 2020;60(3):355–60. https://doi.org/10.23736/S0022-4707.19.10162-4.
Vasquez-Bonilla AA, Brazo-Sayavera J, Timon R, Olcina G. Measurement of resting muscle oxygenation and the relationship with countermovement jumps and body composition in soccer players. Cult Cienc y Deporte. 2021;16(48):215–23. https://doi.org/10.12800/ccd.v16i48.1726.
Vasquez-Bonilla AA, Camacho-Cardeñosa A, Timón R, Martínez-Guardado I, Camacho-Cardeñosa M, Olcina G. Muscle oxygen desaturation and re-saturation capacity limits in repeated sprint ability performance in women soccer players: a new physiological interpretation. Int J Environ Res Public Health. 2021;18(7):3484. https://doi.org/10.3390/ijerph18073484.
Vasquez-Bonila AA, Rojas-Valverde D, Timone R, Olcina G. Influence of fat percentage on muscle oxygen uptake and metabolic power during repeated-sprint ability of footballers. Apunts Sports Med. 2016;2022(57):100395. https://doi.org/10.1016/j.apunsm.2022.100395.
dos Santos IAD, Lemos MP, Coelho VHM, Zagatto AM, Marocolo M, Soares RN, et al. Acute photobiomodulation does not influence specific high-intensity and intermittent performance in female futsal players. Int J Environ Res Public Health. 2020;17(19):7253. https://doi.org/10.3390/ijerph17197253.
Matzka M, Zinner C, Kunz P, Holmberg HC, Sperlich B. Comparison of physiological parameters during on-water and ergometer kayaking and their relationship to performance in sprint kayak competitions. Int J Sports Physiol Perform. 2021;16(7):958–64. https://doi.org/10.1123/ijspp.2019-0912.
Paulauskas R, Nekriošius R, Dadelienė R, Sousa A, Figueira B. Muscle oxygenation measured with near-infrared spectroscopy following different intermittent training protocols in a world-class kayaker—a case study. Sensors. 2022;22(21):8238. https://doi.org/10.3390/s22218238.
Yamaguchi K, Kasai N, Sumi D, Yatsutani H, Girard O, Goto K. Muscle oxygenation during repeated double-poling sprint exercise in normobaric hypoxia and normoxia. Front Physiol. 2019;10:743. https://doi.org/10.3389/fphys.2019.00743.
Kojima C, Yamaguchi K, Ito H, Kasai N, Girard O, Goto K. Acute effect of repeated sprint exercise with blood flow restriction during rest periods on muscle oxygenation. Front Physiol. 2021;12:665383. https://doi.org/10.3389/fphys.2021.665383.
Piucco T, Soares R, Diefenthaeler F, Millet GY, Murias JM. Oxygen uptake and muscle deoxygenation kinetics during skating: comparison between slide-board and treadmill skating. Int J Sports Physiol Perform. 2018;13(6):783–8. https://doi.org/10.1123/ijspp.2017-0440.
Richard P, Billaut F. Combining chronic ischemic preconditioning and inspiratory muscle warm-up to enhance on-ice time-trial performance in elite speed skaters. Front Physiol. 2018;9:1036. https://doi.org/10.3389/fphys.2018.01036.
Richard P, Billaut F. Time-trial performance in elite speed skaters after remote ischemic preconditioning. Int J Sports Physiol Perform. 2018;13(19):1308–16. https://doi.org/10.1123/ijspp.2018-0111.
Richard P, Billaut F. Effects of inspiratory muscle warm-up on locomotor muscle oxygenation in elite speed skaters during 3000 m time trials. Eur J Appl Physiol. 2019;119(1):191–200. https://doi.org/10.1007/s00421-018-4015-8.
Contreras-Briceño F, Espinosa-Ramirez M, Hevia G, Llambias D, Carrasco M, Cerda F, et al. Reliability of NIRS portable device for measuring intercostal muscles oxygenation during exercise. J Sports Sci. 2019;37(23):2653–9. https://doi.org/10.1080/02640414.2019.1653422.
Contreras-Briceño F, Espinosa-Ramírez M, Moya-Gallardo E, Fuentes-Kloss R, Gabrielli L, Araneda OF, et al. Intercostal muscles oxygenation and breathing pattern during exercise in competitive marathon runners. Int J Environ Res Public Health. 2021;18(16):8287. https://doi.org/10.3390/ijerph18168287.
Jones S, D’Silva A, Bhuva A, Lloyd G, Manisty C, Moon JC, et al. Improved exercise-related skeletal muscle oxygen consumption following uptake of endurance training measured using near-infrared spectroscopy. Front Physiol. 2017;8:1018. https://doi.org/10.3389/fphys.2017.01018.
Kerhervé HA, Samozino P, Descombe F, Pinay M, Millet GY, Pasqualini M, et al. Calf compression sleeves change biomechanics but not performance and physiological responses in trail running. Front Physiol. 2017;8:247. https://doi.org/10.3389/fphys.2017.00247.
Paredes-Ruiz M, Jodar-Reverte M, Ferrer-Lopez V, Martínez-González-Moro I. Quadriceps muscle oxygenation during a maximum stress test in middle-aged athletes. Monten J Sports Sci Med. 2020;9(2):43–9. https://doi.org/10.26773/mjssm.200908.
Guardado IM, Guerra AM, Pino BS, Camacho GO, Andrada RT. Acute responses of muscle oxygen saturation during different cluster training configurations in resistance-trained individuals. Biol Sport. 2021;38(3):367–76. https://doi.org/10.5114/biolsport.2021.99701.
Halley SL, Marshall P, Siegler JC. Effect of ischemic preconditioning and changing inspired O2 fractions on neuromuscular function during intense exercise. J Appl Physiol (1985). 2019;127(6):1688–97. https://doi.org/10.1152/japplphysiol.00539.2019.
Haynes JT 4th, Townsend JR, Aziz MA, Jones MD, Littlefield LA, Ruiz MD, Johnson KD, Gonzalez AM. Impact of red spinach extract supplementation on bench press performance, muscle oxygenation, and cognitive function in resistance-trained males. Sports. 2021;9(6):77. https://doi.org/10.3390/sports9060077.
Marshall PW, Metcalf E, Hagstrom AD, Cross R, Siegler JC, Enoka RM. Changes in fatigue are the same for trained men and women after resistance exercise. Med Sci Sports Exerc. 2020;52(1):196–204. https://doi.org/10.1249/MSS.0000000000002103.
Scott BR, Slattery KM, Sculley DV, Lockhart C, Dascombe BJ. Acute physiological responses to moderate-load resistance exercise in hypoxia. J Strength Cond Res. 2017;31(7):1973–81. https://doi.org/10.1519/JSC.0000000000001649.
Seeberg TM, Kocbach J, Danielsen J, Noordhof DA, Skovereng K, Meyer F, et al. Physiological and biomechanical responses to cross-country skiing in varying terrain: low- vs. high-intensity. Front Physiol. 2021;12:741573. https://doi.org/10.3389/fphys.2021.741573.
Klusiewicz A, Rębiś K, Ozimek M, Czaplicki A. The use of muscle near-infrared spectroscopy (NIRS) to assess the aerobic training loads of world-class rowers. Biol Sport. 2021;38(4):713–9.
Possamai LT, de Aguiar RA, Borszcz FK, do NascimentoSalvador PC, de Lucas RD, Turnes T. Muscle oxidative capacity in vivo is associated with physiological parameters in trained rowers. Res Q Exerc Sport. 2022. https://doi.org/10.1080/02701367.2022.2100862.
Turnes T, de Aguiar RA, de Oliveira Cruz RS, Salvador AF, Lisbôa FD, Pereira KL, et al. Impact of ischaemia-reperfusion cycles during ischaemic preconditioning on 2000-m rowing ergometer performance. Eur J Appl Physiol. 2018;118(8):1599–607. https://doi.org/10.1007/s00421-018-3891-2.
Vasquez-Bonilla AA, Tomas-Carus P, Brazo-Sayavera J, Malta J, Folgado H, Olcina G. Muscle oxygenation is associated with bilateral strength asymmetry during isokinetic testing in sport teams. Sci Sports. 2023;38(4):426.e1-e9. https://doi.org/10.1016/j.scispo.2022.03.014.
Pramkratok W, Yimlamai T. Effects of acute hypoxia on psycho-physiological response and muscle oxygenation during incremental running exercise. J Exerc Physiol Online. 2021;24(3):44–54.
Pramkratok W, Songsupap T, Yimlamai T. Repeated sprint training under hypoxia improves aerobic performance and repeated sprint ability by enhancing muscle deoxygenation and markers of angiogenesis in rugby sevens. Eur J Appl Physiol. 2022;122(3):611–22. https://doi.org/10.1007/s00421-021-04861-8.
Balsalobre-Fernández C, Romero-Moraleda B, Cupeiro R, Peinado AB, Butragueño J, Benito PJ. The effects of beetroot juice supplementation on exercise economy, rating of perceived exertion and running mechanics in elite distance runners: a double-blinded, randomized study. PLoS ONE. 2018;13(7):e0200517. https://doi.org/10.1371/journal.pone.0200517.
Bellinger PM, Sabapathy S, Craven J, Arnold B, Minahan C. Overreaching attenuates training-induced improvements in muscle oxidative capacity. Med Sci Sports Exerc. 2020;52(1):77–85. https://doi.org/10.1249/MSS.0000000000002095.
Born DP, Stöggl T, Swarén M, Björklund G. Near-infrared spectroscopy: more accurate than heart rate for monitoring intensity in running in hilly terrain. Int J Sports Physiol Perform. 2017;12(4):440–7. https://doi.org/10.1123/ijspp.2016-0101.
de Aguiar RA, Turnes T, Borszcz FK, Raimundo JAG, Caputo F. Near-infrared spectroscopy-derived muscle V̇O2 kinetics after moderate running exercise in healthy males: reliability and associations with parameters of aerobic fitness. Exp Physiol. 2022;107(5):476–88. https://doi.org/10.1113/EP090105.
Fleming N, Vaughan J, Feeback M. Ingestion of oxygenated water enhances lactate clearance kinetics in trained runners. J Int Soc Sports Nutr. 2017;14:9. https://doi.org/10.1186/s12970-017-0166-y.
Gagnon DD, Peltonen JE, Rintamäki H, Gagnon SS, Herzig KH, Kyröläinen H. The effects of skin and core tissue cooling on oxygenation of the vastus lateralis muscle during walking and running. J Sports Sci. 2017;35(20):1995–2004. https://doi.org/10.1080/02640414.2016.1245436.
Hobbins L, Gaoua N, Hunter S, Girard O. Psycho-physiological responses to perceptually-regulated interval runs in hypoxia and normoxia. Physiol Behav. 2019;209:112611. https://doi.org/10.1016/j.physbeh.2019.112611.
Jeffries O, Waldron M, Pattison JR, Patterson SD. Enhanced local skeletal muscle oxidative capacity and microvascular blood flow following 7-day ischemic preconditioning in healthy humans. Front Physiol. 2018;9:463. https://doi.org/10.3389/fphys.2018.00463.
Manchado-Gobatto FB, Marostegan AB, Rasteiro FM, Cirino C, Cruz JP, Moreno MA, et al. New insights into mechanical, metabolic and muscle oxygenation signals during and after high-intensity tethered running. Sci Rep. 2020;10(1):6336. https://doi.org/10.1038/s41598-020-63297-w.
Marostegan AB, Gobatto CA, Rasteiro FM, Hartz CS, Moreno MA, Manchado-Gobatto FB. Effects of different inspiratory muscle warm-up loads on mechanical, physiological and muscle oxygenation responses during high-intensity running and recovery. Sci Rep. 2022;12(1):11223. https://doi.org/10.1038/s41598-022-14616-w.
Meirelles CM, Matsuura C, Silva RS Jr, Guimarães FF, Gomes PSC. Acute effects of L-Arginine supplementation on oxygen consumption kinetics and muscle oxyhemoglobin and deoxyhemoglobin during treadmill running in male adults. Int J Exerc Sci. 2019;12(2):444–55.
Osawa T, Arimitsu T, Takahashi H. Hypoxia affects tissue oxygenation differently in the thigh and calf muscles during incremental running. Eur J Appl Physiol. 2017;117(10):2057–64. https://doi.org/10.1007/s00421-017-3696-8.
Pawlak-Chaouch M, Boissière J, Munyaneza D, Tagougui S, Gamelin FX, Cuvelier G, et al. Plasma asymmetric dimethylarginine concentrations are not related to differences in maximal oxygen uptake in endurance trained and untrained men. Exp Physiol. 2019;104(2):254–63. https://doi.org/10.1113/EP087398.
Roberson PA, Romero MA, Mumford PW, Osburn SC, Haun CT, Vann CG, et al. Protein supplementation throughout 10 weeks of progressive run training is not beneficial for time trial improvement. Front Nutr. 2018;5:97. https://doi.org/10.3389/fnut.2018.00097.
Rodrigo-Carranza V, González-Mohíno F, Turner AP, Rodriguez-Barbero S, González-Ravé JM. Using a portable near-infrared spectroscopy device to estimate the second ventilatory threshold. Int J Sports Med. 2021;42(10):905–10. https://doi.org/10.1055/a-1343-2127.
Rowland SN, Da Boit M, Tan R, Robinson GP, O’Donnell E, James LJ, et al. Dietary nitrate supplementation enhances performance and speeds muscle deoxyhaemoglobin kinetics during an end-sprint after prolonged moderate-intensity exercise. Antioxidants. 2023;12(1):25. https://doi.org/10.3390/antiox12010025.
Salzmann K, Sanchez AMJ, Borrani F. Effects of blood flow restriction on O2 muscle extraction and O2 pulmonary uptake kinetics during heavy exercise. Front Physiol. 2021;12:722848. https://doi.org/10.3389/fphys.2021.722848.
Solsona R, Berthelot H, Borrani F, Sanchez AMJ. Mechanical, cardiorespiratory, and muscular oxygenation responses to sprint interval exercises under different hypoxic conditions in healthy moderately trained men. Front Physiol. 2021;12:773950. https://doi.org/10.3389/fphys.2021.773950.
Solsona R, Deriaz R, Borrani F, Sanchez AMJ. Muscle deoxygenation rates and reoxygenation modeling during a sprint interval training exercise performed under different hypoxic conditions. Front Physiol. 2022;13:864642. https://doi.org/10.3389/fphys.2022.864642.
Sweeting AJ, Billaut F, Varley MC, Rodriguez RF, Hopkins WG, Aughey RJ. Variations in hypoxia impairs muscle oxygenation and performance during simulated team-sport running. Front Physiol. 2017;8:80. https://doi.org/10.3389/fphys.2017.00080.
Woorons X, Billaut F, Lamberto C. Running exercise with end-expiratory breath holding up to the breaking point induces large and early fall in muscle oxygenation. Eur J Appl Physiol. 2021;121(12):3515–25. https://doi.org/10.1007/s00421-021-04813-2.
Yáñez-Sepúlveda R, Verdugo-Marchese H, Duclos-Bastías D, Tuesta M, Alvear-Ordenes I. Effects of inspiratory muscle training on muscle oxygenation during vascular occlusion testing in trained healthy adult males. Int J Environ Res Public Health. 2022;19(24):16766. https://doi.org/10.3390/ijerph192416766.
Zinner C, Born DP, Sperlich B. Ischemic preconditioning does not alter performance in multidirectional high-intensity intermittent exercise. Front Physiol. 2017;8:1029. https://doi.org/10.3389/fphys.2017.01029.
Rębiś K, Sadowska D, Starczewski M, Klusiewicz A. Usefulness of portable device to establish differences in muscle oxygenation between the Wingate test and graded exercise test: effect of gender on anaerobic and aerobic capacity in speed skaters. Front Physiol. 2022;13:809864. https://doi.org/10.3389/fphys.2022.809864.
Yamaguchi K, Imai T, Yatsutani H, Goto K. A combined hot and hypoxic environment during maximal cycling sprints reduced muscle oxygen saturation: a pilot study. J Sports Sci Med. 2021;20(4):684–9. https://doi.org/10.52082/jssm.2021.684.
Paquette M, Bieuzen F, Billaut F. Muscle oxygenation rather than VO2max as a strong predictor of performance in sprint canoe-kayak. Int J Sports Physiol Perform. 2018;13(10):1299–307. https://doi.org/10.1123/ijspp.2018-0077.
Paquette M, Bieuzen F, Billaut F. Sustained muscle deoxygenation vs. sustained high VO2 during high-intensity interval training in sprint canoe-kayak. Front Sports Act Liv. 2019;1:6. https://doi.org/10.3389/fspor.2019.00006.
Paquette M, Bieuzen F, Billaut F. Effect of a 3-weeks training camp on muscle oxygenation, V̇O2 and performance in elite sprint kayakers. Front Sports Act Liv. 2020;2:47. https://doi.org/10.3389/fspor.2020.00047.
Dalamitros AA, Semaltianou E, Toubekis AG, Kabasakalis A. Muscle oxygenation, heart rate, and blood lactate concentration during submaximal and maximal interval swimming. Front Sports Act Liv. 2021;3:759925. https://doi.org/10.3389/fspor.2021.759925.
Grossman KJ, Lim DJ, Murias JM, Belfry GR. The effect of breathing patterns common to competitive swimming on gas exchange and muscle deoxygenation during heavy-intensity fartlek exercise. Front Physiol. 2021;12:723951. https://doi.org/10.3389/fphys.2021.723951.
Jones B, Parry D, Cooper CE. Underwater near-infrared spectroscopy can measure training adaptations in adolescent swimmers. PeerJ. 2018;6:e4393. https://doi.org/10.7717/peerj.4393.
McGowan CJ, Pyne DB, Thompson KG, Raglin JS, Osborne M, Rattray B. Elite sprint swimming performance is enhanced by completion of additional warm-up activities. J Sports Sci. 2017;35(15):1493–9. https://doi.org/10.1080/02640414.2016.1223329.
Pratama AB, Yimlamai T. Effects of active and passive recovery on muscle oxygenation and swimming performance. Int J Sports Physiol Perform. 2020;159(9):1289–96. https://doi.org/10.1123/ijspp.2019-0537.
Chang SC, Adami A, Lin HC, Lin YC, Chen CPC, Fu TC, et al. Relationship between maximal incremental and high-intensity interval exercise performance in elite athletes. PLoS ONE. 2020;15(5):e0226313. https://doi.org/10.1371/journal.pone.0226313.
Cyr-Kirk S, Billaut F. Hyperoxia improves repeated-sprint ability and the associated training load in athletes. Front Sports Act Living. 2022;4:817280. https://doi.org/10.3389/fspor.2022.817280.
Dennis MC, Goods PSR, Binnie MJ, Girard O, Wallman KE, Dawson B, et al. Increased air temperature during repeated-sprint training in hypoxia amplifies changes in muscle oxygenation without decreasing cycling performance. Eur J Sport Sci. 2023;23(1):66–72. https://doi.org/10.1080/17461391.2021.2003868.
Fryer S, Stone K, Dickson T, Wilhelmsen A, Cowen D, Faulkner J, et al. The effects of 4 weeks normobaric hypoxia training on microvascular responses in the forearm flexor. J Sports Sci. 2019;37(11):1235–41. https://doi.org/10.1080/02640414.2018.1554177.
Gatterer H, Menz V, Salazar-Martinez E, Sumbalova Z, Garcia-Souza LF, Velika B, et al. Exercise performance, muscle oxygen extraction and blood cell mitochondrial respiration after repeated-sprint and sprint interval training in hypoxia: a pilot study. J Sports Sci Med. 2018;17(3):339–47.
Jeffries O, Evans DT, Waldron M, Coussens A, Patterson SD. Seven-day ischaemic preconditioning improves muscle efficiency during cycling. J Sports Sci. 2019;37(24):2798–805. https://doi.org/10.1080/02640414.2019.1664537.
Contreras-Briceño F, Espinosa-Ramirez M, Keim-Bagnara V, Carreño-Román M, Rodríguez-Villagra R, Villegas-Belmar F, et al. Determination of the respiratory compensation point by detecting changes in intercostal muscles oxygenation by using near-infrared spectroscopy. Life. 2022;12(3):444. https://doi.org/10.3390/life12030444.
Olcina G, Perez-Sousa MÁ, Escobar-Alvarez JA, Timón R. Effects of cycling on subsequent running performance, stride length, and muscle oxygen saturation in triathletes. Sports. 2019;7(5):115. https://doi.org/10.3390/sports7050115.
Paradis-Deschênes P, Joanisse DR, Mauriège P, Billaut F. Ischemic preconditioning enhances aerobic adaptations to sprint-interval training in athletes without altering systemic hypoxic signaling and immune function. Front Sports Act Liv. 2020;2:41. https://doi.org/10.3389/fspor.2020.00041.
Paradis-Deschênes P, Lapointe J, Joanisse DR, Billaut F. Similar recovery of maximal cycling performance after ischemic preconditioning, neuromuscular electrical stimulation or active recovery in endurance athletes. J Sports Sci Med. 2020;19(4):761–71.
Triska C, Hopker J, Wessner B, Reif A, Tschan H, Karsten B. A 30-min rest protocol does not affect W’, critical power, and systemic response. Med Sci Sports Exerc. 2021;53(2):404–12. https://doi.org/10.1249/MSS.0000000000002477.
Giovanelli N, Biasutti L, Salvadego D, Alemayehu HK, Grassi B, Lazzer S. Changes in skeletal muscle oxidative capacity after a trail running race. Int J Sports Physiol Perform. 2020;15(2):278–84. https://doi.org/10.1123/ijspp.2018-0882.
Kerhervé HA, McLean S, Birkenhead K, Parr D, Solomon C. Influence of exercise duration on cardiorespiratory responses, energy cost and tissue oxygenation within a 6 hour treadmill run. PeerJ. 2017;5:e3694. https://doi.org/10.7717/peerj.3694.
Vernillo G, Brighenti A, Limonta E, Trabucchi P, Malatesta D, Millet GP, et al. effects of ultratrail running on skeletal-muscle oxygenation dynamics. Int J Sports Physiol Perform. 2017;12(4):496–504. https://doi.org/10.1123/ijspp.2015-0745.
Meyer PH, Missao ET, McCully K. Muscle oxidative capacity in the arms and legs of various types of endurance trained athletes. Med Res Archiv. 2021;9(7):1–15. https://doi.org/10.18103/mra.v9i7.2505.
Feldmann A, Ammann L, Gächter F, Zibung M, Erlacher D. Muscle oxygen saturation breakpoints reflect ventilatory thresholds in both cycling and running. J Hum Kinet. 2022;83:87–97. https://doi.org/10.2478/hukin-2022-0054.
Schörkmaier T, Wahl Y, Brinkmann C, Bloch W, Wahl P. No influence of nonivamide-nicoboxil on the peak power output in competitive sportsmen. Int J Sports Med. 2021;42(12):1092–7. https://doi.org/10.1055/a-1403-2701.
Tong TK, Baker JS, Zhang H, Kong Z, Nie J. Effects of specific core re-warm-ups on core function, leg perfusion and second-half team sport-specific sprint performance: a randomized crossover study. J Sports Sci Med. 2019;18(3):479–89.
Anthierens A, Olivier N, Thevenon A, Mucci P. Trunk muscle aerobic metabolism responses in endurance athletes, combat athletes and untrained men. Int J Sports Med. 2019;40(7):434–9. https://doi.org/10.1055/a-0856-7207.
Pawlak-Chaouch M, Boissière J, Munyaneza D, Gamelin FX, Cuvelier G, Berthoin S, et al. Beetroot juice does not enhance supramaximal intermittent exercise performance in elite endurance athletes. J Am Coll Nutr. 2019;38(8):729–38. https://doi.org/10.1080/07315724.2019.1601601.
Green ES, Williams ER, Feito Y, Jenkins NT. Physiological and anthropometric differences among endurance, strength, and high-intensity functional training participants: a cross-sectional study. Res Q Exerc Sport. 2023;94(1):131–42. https://doi.org/10.1080/02701367.2021.1947468.
Lagerwaard B, Keijer J, McCully KK, de Boer VCJ, Nieuwenhuizen AG. In vivo assessment of muscle mitochondrial function in healthy, young males in relation to parameters of aerobic fitness. Eur J Appl Physiol. 2019;119(8):1799–808. https://doi.org/10.1007/s00421-019-04169-8.
Lagerwaard B, Janssen JJE, Cuijpers I, Keijer J, de Boer VCJ, Nieuwenhuizen AG. Muscle mitochondrial capacity in high- and low-fitness females using near-infrared spectroscopy. Physiol Rep. 2021;9(9):e14838. https://doi.org/10.14814/phy2.14838.
Rodriguez RF, Townsend NE, Aughey RJ, Billaut F. Muscle oxygenation maintained during repeated-sprints despite inspiratory muscle loading. PLoS ONE. 2019;14(9):e0222487. https://doi.org/10.1371/journal.pone.0222487.
Rodriguez RF, Townsend NE, Aughey RJ, Billaut F. Respiratory muscle oxygenation is not impacted by hypoxia during repeated-sprint exercise. Respir Physiol Neurobiol. 2019;260:114–21. https://doi.org/10.1016/j.resp.2018.11.006.
Ruiz-Moreno C, Gutiérrez-Hellín J, Lara B, Del Coso J. Effect of caffeine on muscle oxygen saturation during short-term al-out exercise: a double-blind randomized crossover study. Eur J Nutr. 2022;61(6):3109–17. https://doi.org/10.1007/s00394-022-02875-2.
Trexler ET, Keith DS, Lucero AA, Stoner L, Schwartz TA, Persky AM, et al. Effects of citrulline malate and beetroot juice supplementation on energy metabolism and blood flow during submaximal resistance exercise. J Diet Suppl. 2020;17(6):698–717. https://doi.org/10.1080/19390211.2019.1650866.
Willis SJ, Borrani F, Millet GP. Leg- vs arm-cycling repeated sprints with blood flow restriction and systemic hypoxia. Eur J Appl Physiol. 2019;119(8):1819–28. https://doi.org/10.1007/s00421-019-04171-0.
Willis SJ, Peyrard A, Rupp T, Borrani F, Millet GP. Vascular and oxygenation responses of local ischemia and systemic hypoxia during arm cycling repeated sprints. J Sci Med Sport. 2019;22(10):1151–6. https://doi.org/10.1016/j.jsams.2019.05.001.
Brodeur ZR, Paustian MJ, Monteleone-Haught DA, Lamm RA, Pagano AG, Ellis CE. The effects of static and dynamic stretching on muscle oxygen saturation in the rectus femoris. Int J Exerc Sci. 2022;15(3):702–8.
Kojima Y, Fukusaki C, Ishii N. Effects of hyperoxia on dynamic muscular endurance are associated with individual whole-body endurance capacity. PLoS ONE. 2020;15(4):e0231643. https://doi.org/10.1371/journal.pone.0231643.
Lagerwaard B, Nieuwenhuizen AG, de Boer VCJ, Keijer J. In vivo assessment of mitochondrial capacity using NIRS in locomotor muscles of young and elderly males with similar physical activity levels. Geroscience. 2020;42:299–310. https://doi.org/10.1007/s11357-019-00145-4.
Le Roux-Mallouf T, Vallejo A, Pelen F, Halimaoui I, Doutreleau S, Verges S. Synergetic effect of NO precursor supplementation and exercise training. Med Sci Sports Exerc. 2020;52(11):2437–47. https://doi.org/10.1249/MSS.0000000000002387.
Lin TY, Chen JJ, Lin LL, Ou Yang WT, Chen MY, Tsai YC. Effects of multicomponent exercise training on muscle oxygenation in young and older adults. J Exerc Sci Fit. 2023;21(1):138–46. https://doi.org/10.1016/j.jesf.2022.12.002.
Morishima T, Tsuchiya Y, Ueda H, Ochi E. Muscular endurance and muscle metabolic responses to 8 weeks of omega-3 polyunsaturated fatty acids supplementation. Physiol Rep. 2020;8(16):e14546. https://doi.org/10.14814/phy2.14546.
Mota GR, Rightmire ZB, Martin JS, McDonald JR, Kavazis AN, Pascoe DD, et al. Ischemic preconditioning has no effect on maximal arm cycling exercise in women. Eur J Appl Physiol. 2020;120(2):369–80. https://doi.org/10.1007/s00421-019-04281-9.
Nell HJ, Castelli LM, Bertani D, Jipson AA, Meagher SF, Melo LT, et al. The effects of hypoxia on muscle deoxygenation and recruitment in the flexor digitorum superficialis during submaximal intermittent handgrip exercise. BMC Sports Sci Med Rehabil. 2020;12:16. https://doi.org/10.1186/s13102-020-00163-2.
Papadopoulos S, Dipla K, Triantafyllou A, Nikolaidis MG, Kyparos A, Touplikioti P, et al. Beetroot increases muscle performance and oxygenation during sustained isometric exercise, but does not alter muscle oxidative efficiency and microvascular reactivity at rest. J Am Coll Nutr. 2018;37(5):361–72. https://doi.org/10.1080/07315724.2017.1401497.
Page MJ, McKenzie JE, Higgins JPT. Tools for assessing risk of reporting biases in studies and syntheses of studies: a systematic review. BMJ Open. 2018;8(3):e019703. https://doi.org/10.1136/bmjopen-2017-019703.
Cortese L, Zanoletti M, Karadeniz U, Pagliazzi M, Yaqub MA, Busch DR, et al. Performance assessment of a commercial continuous-wave near-infrared spectroscopy tissue oximeter for suitability for use in an international, multi-center clinical trial. Sensors (Basel). 2021;21(21):6957. https://doi.org/10.3390/s21216957.
Farzam P, Starkweather Z, Franceschini MA. Validation of a novel wearable, wireless technology to estimate oxygen levels and lactate threshold power in the exercising muscle. Physiol Rep. 2018;6(7):e13664. https://doi.org/10.14814/phy2.13664.
Feldmann A, Schmitz R, Erlacher D. Near-infrared spectroscopy-derived muscle oxygen saturation on a 0% to 100% scale: reliability and validity of the Moxy Monitor. J Biomed Opt. 2019;24(11):1–11. https://doi.org/10.1117/1.JBO.24.11.115001.
McManus CJ, Collison J, Cooper CE. Performance comparison of the MOXY and PortaMon near-infrared spectroscopy muscle oximeters at rest and during exercise. J Biomed Opt. 2018;23(1):1–14. https://doi.org/10.1117/1.JBO.23.1.015007.
Sudakou A, Wabnitz H, Liemert A, Wolf M, Liebert A. Two-layered blood-lipid phantom and method to determine absorption and oxygenation employing changes in moments of DTOFs. Biomed Opt Express. 2023;14(7):3506–31. https://doi.org/10.1364/BOE.492168.
Dorschky E, Camomilla V, Davis J, Federolf P, Reenalda J, Koelewijn AD. Perspective on “in the wild” movement analysis using machine learning. Hum Mov Sci. 2023;87:103042. https://doi.org/10.1016/j.humov.2022.103042.
Amendola C, Contini D, Re R, Spinelli L, Frabasile L, Levoni P, et al. Robustness of tissue oxygenation estimates by continuous wave space-resolved near infrared spectroscopy. J Biomed Opt. 2023;28(7):075002. https://doi.org/10.1117/1.JBO.28.7.075002.
Endo T, Kime R, Fuse S, Murase N, Kurosawa Y, Hamaoka T. Changes in optical path length reveal significant potential errors of muscle oxygenation evaluation during exercise in humans. Med Sci Sports Exerc. 2021;53(4):853–9. https://doi.org/10.1249/MSS.0000000000002530.
Craig JC, Broxterman RM, Wilcox SL, Chen C, Barstow TJ. Effect of adipose tissue thickness, muscle site, and sex on near-infrared spectroscopy derived total-[hemoglobin + myoglobin]. J Appl Physiol (1985). 2017;123(6):1571–8. https://doi.org/10.1152/japplphysiol.00207.2017.
Pirovano I, Porcelli S, Re R, Spinelli L, Contini D, Marzorati M, et al. Effect of adipose tissue thickness and tissue optical properties on the differential pathlength factor estimation for NIRS studies on human skeletal muscle. Biomed Opt Express. 2020;12(1):571–87. https://doi.org/10.1364/BOE.412447.
Koirala B, Concas A, Sun Y, Gladden LB, Lai N. Relationship between muscle venous blood oxygenation and near-infrared spectroscopy: quantitative analysis of the Hb and Mb contributions. J Appl Physiol. 2023;134(5):1063–74. https://doi.org/10.1152/japplphysiol.00406.2022.
Afshari A, Saager RB, Burgos D, Vogt WC, Wang J, Mendoza G, et al. Evaluation of the robustness of cerebral oximetry to variations in skin pigmentation using a tissue-simulating phantom. Biomed Opt Express. 2022;13(5):2909–28. https://doi.org/10.1364/BOE.454020.
Quaresima V, Scholkmann F, Ferrari M. Skin pigmentation bias in regional brain oximetry measurements? Crit Care. 2023;27(1):10. https://doi.org/10.1186/s13054-022-04295-4.
Yogev A, Arnold J, Nelson H, Clarke DC, Guenette JA, Sporer BC, et al. Comparing the reliability of muscle oxygen saturation with common performance and physiological markers across cycling exercise intensity. Front Sports Act Liv. 2023;5:1143393. https://doi.org/10.3389/fspor.2023.1143393.
Crum EM, O’Connor WJ, Van Loo L, Valckx M, Stannard SR. Validity and reliability of the Moxy oxygen monitor during incremental cycling exercise. Eur J Sport Sci. 2017;17(8):1037–43. https://doi.org/10.1080/17461391.2017.1330899.
van Hooff M, Meijer EJ, Scheltinga MRM, Savelberg HHCM, Schep G. Test-retest reliability of skeletal muscle oxygenation measurement using near-infrared spectroscopy during exercise in patients with sport-related iliac artery flow limitation. Clin Physiol Funct Imaging. 2022;42(2):114–26. https://doi.org/10.1111/cpf.12738.
Vallance E, Sutton-Charani N, Guyot P, Perrey S. Predictive modeling of the ratings of perceived exertion during training and competition in professional soccer players. J Sci Med Sport. 2023;26(6):322–7. https://doi.org/10.1016/j.jsams.2023.05.001.
Boone J, Barstow TJ, Celie B, Prieur F, Bourgois J. The interrelationship between muscle oxygenation, muscle activation, and pulmonary oxygen uptake to incremental ramp exercise: influence of aerobic fitness. Appl Physiol Nutr Metab. 2016;41(1):55–62. https://doi.org/10.1139/apnm-2015-0261.
Caen K, Bourgois JG, Stassijns E, Boone J. A longitudinal study on the interchangeable use of whole-body and local exercise thresholds in cycling. Eur J Appl Physiol. 2022;122(7):1657–70. https://doi.org/10.1007/s00421-022-04942-2.
Yogev A, Arnold J, Clarke D, Guenette JA, Sporer BC, Koehle MS. Comparing the respiratory compensation point with muscle oxygen saturation in locomotor and non-locomotor muscles using wearable NIRS spectroscopy during whole-body exercise. Front Physiol. 2022;13:818733. https://doi.org/10.3389/fphys.2022.818733.
Sendra-Pérez C, Sanchez-Jimenez JL, Marzano-Felisatti JM, Encarnación-Martínez A, Salvador-Palmer R, Priego-Quesada JI. Reliability of threshold determination using portable muscle oxygenation monitors during exercise testing: a systematic review and meta-analysis. Sci Rep. 2023;13(1):12649. https://doi.org/10.1038/s41598-023-39651-z.
Poole DC, Rossiter HB, Brooks GA, Gladden LB. The anaerobic threshold: 50+ years of controversy. J Physiol. 2021;599:737–67. https://doi.org/10.1113/JP279963.
Matthews IR, Heenan LJ, Fisher KG, Flood EF, Wehrman LW, Kirby BS, Wilkins BW. Identification of maximal steady-state metabolic rate by the change in muscle oxygen saturation. J Appl Physiol. 2023;134(6):1349–58. https://doi.org/10.1152/japplphysiol.00706.2022.
Feldmann A, Erlacher D. Critical oxygenation: can muscle oxygenation inform us about critical power? Med Hypoth. 2021;150:110575. https://doi.org/10.1016/j.mehy.2021.110575.
Kirby BS, Clark DA, Bradley EM, Wilkins BW. The balance of muscle oxygen supply and demand reveals critical metabolic rate and predicts time to exhaustion. J Appl Physiol. 2021;130:1915–27. https://doi.org/10.1152/japplphysiol.00058.2021.
Azevedo RA, Béjar Saona JE, Inglis EC, Iannetta D, Murias JM. The effect of the fraction of inspired oxygen on the NIRS-derived deoxygenated hemoglobin “breakpoint” during ramp-incremental test. Am J Physiol Regul Integr Comp Physiol. 2020;318(2):R399–409. https://doi.org/10.1152/ajpregu.00291.2019.
Miranda-Fuentes C, Chirosa-Ríos LJ, Guisado-Requena IM, Delgado-Floody P, Jerez-Mayorga D. Changes in muscle oxygen saturation measured using wireless near-infrared spectroscopy in resistance training: a systematic review. Int J Environ Res Public Health. 2021;18(8):4293. https://doi.org/10.3390/ijerph18084293.
Fennell CRJ, Mauger AR, Hopker JG. Reproducibility of NIRS-derived mitochondrial oxidative capacity in highly active older adults. Exp Gerontol. 2023;175:112156. https://doi.org/10.1016/j.exger.2023.112156.
Southern WM, Ryan TE, Reynolds MA, McCully K. Reproducibility of near-infrared spectroscopy measurements of oxidative function and postexercise recovery kinetics in the medial gastrocnemius muscle. Appl Physiol Nutr Metab. 2014;39(5):521–9. https://doi.org/10.1139/apnm-2013-0347.
Dennis JJ, Wiggins CC, Smith JR, Isautier JMJ, Johnson BD, Joyner MJ, et al. Measurement of muscle blood flow and O2 uptake via near-infrared spectroscopy using a novel occlusion protocol. Sci Rep. 2021;11(1):918. https://doi.org/10.1038/s41598-020-79741-w.
Ryan TE, Southern WM, Reynolds MA, McCully KK. A cross-validation of near-infrared spectroscopy measurements of skeletal muscle oxidative capacity with phosphorus magnetic resonance spectroscopy. J Appl Physiol. 2013;115(12):1757–66. https://doi.org/10.1152/japplphysiol.00835.2013.
Sako T, Hamaoka T, Higuchi H, Kurosawa Y, Katsumura T. Validity of NIR spectroscopy for quantitatively measuring muscle oxidative metabolic rate in exercise. J Appl Physiol. 2001;90(1):338–44. https://doi.org/10.1152/jappl.2001.90.1.338.
Scano A, Pirovano I, Manunza ME, Spinelli L, Contini D, Torricelli A, et al. Sustained fatigue assessment during isometric exercises with time-domain near infrared spectroscopy and surface electromyography signals. Biomed Opt Express. 2020;11(12):7357–75. https://doi.org/10.1364/BOE.403976.
da Mota Moreira I, Willigenburg TR, Kregting WJW, Van Steijn ERC, Floor-Westerdijk MJ, Colier WNJM. Assessing stability and accuracy of a novel commercial wearable near-infrared spectroscopy device. Proc. SPIE 12375. Biophotonics in Exercise Science, Sports Medicine, Health Monitoring Technologies, and Wearables IV. 2023. p. 123750A. https://doi.org/10.1117/12.2657434.
Thomas N, Miller AJ, Ayaz H, Brown JD. Haptic shared control improves neural efficiency during myoelectric prosthesis use. Sci Rep. 2023;13(1):484. https://doi.org/10.1038/s41598-022-26673-2.
Chen H, Liang J, Huang W, Yang A, Pang R, Zhao C, et al. Age-related difference in muscle metabolism patterns during upper limb’s encircling exercise: a near-infrared spectroscopy study. Biomed Opt Express. 2022;13(9):4737–51. https://doi.org/10.1364/BOE.462551.
Di Giminiani R, Cardinale M, Ferrari M, Quaresima V. Validation of fabric-based thigh-wearable EMG sensors and oximetry for monitoring quadriceps activity during strength and endurance exercises. Sensors. 2020;20(17):4664. https://doi.org/10.3390/s20174664.
Kimoto A, Fujiyama H, Machida M. A wireless multi-layered EMG/MMG/NIRS sensor for muscular activity evaluation. Sensors (Basel). 2023;23(3):1539. https://doi.org/10.3390/s23031539.
Lacerenza M, Buttafava M, Renna M, Mora AD, Spineli L, Zappa F, et al. Wearable and wireless time-domain near-infrared spectroscopy system for brain and muscle hemodynamic monitoring. Biomed Opt Express. 2020;11(10):5934–49. https://doi.org/10.1364/BOE.403327.
Carp SA, Robinson MB, Franceschini MA. Diffuse correlation spectroscopy: current status and future outlook. Neurophotonics. 2023;10(1):013509. https://doi.org/10.1117/1.NPh.10.1.013509.
Quaresima V, Farzam P, Anderson P, Farzam PY, Wiese D, Carp SA, et al. Diffuse correlation spectroscopy and frequency-domain near-infrared spectroscopy for measuring microvascular blood flow in dynamically exercising human muscles. J Appl Physiol. 2019;127(5):1328–37. https://doi.org/10.1152/japplphysiol.00324.2019.
Funding
Open access funding provided by Università degli Studi dell’Aquila within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
At no point was funding received by any of the authors for the writing of this manuscript.
Conflict of interest
Stéphane Perrey, Valentina Quaresima and Marco Ferrari declare that they have no conflicts of interest.
Data availability
All data and material reported in this systematic review are only from peer-reviewed publications. All the extracted data were included in Table S1 of the ESM, Tables 1 and 2, and in Fig. 2.
Author contributions
All authors conceptualized the review and criteria. Valentina Quaresima and Marco Ferrari performed the initial screening and data extraction of all data. Stephane Perrey completed the screening and data extraction within this manuscript. All authors created the tables and figures. All authors contributed to the writing of the manuscript. All authors reviewed, refined and approved the final manuscript.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Perrey, S., Quaresima, V. & Ferrari, M. Muscle Oximetry in Sports Science: An Updated Systematic Review. Sports Med 54, 975–996 (2024). https://doi.org/10.1007/s40279-023-01987-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-023-01987-x