Abstract
Coal due to its relatively large quantities and wide distribution worldwide has generated renewed interest in research and development with the aim of establishing coal conversion technologies that are technically reliable, environmentally and economically feasible. It has proved to be a prominent energy source in emerging markets with increasing energy demand by accounting for the largest increase in the demand of energy amongst all other energy sources. Furthermore, with its higher mesophase content, coal tar is an appropriate raw material for precursors in the production of carbon fiber. However, whenever a material is put to use, it is important to be able to associate its properties to the behavioral characteristics during a conversion process so as to have a basis for opting for the material in a given process or adjusting the operating conditions in order to optimize the material utilization. Therefore, as with any other material, it is important to be able to relate the properties of coal to its utilization. A review was carried out on the influence of coal properties on four main utilization technologies: gasification, carbonization, liquefaction and carbon fibre production. Among several properties rank, type, mineral matter content, distribution of trace elements, structural composition and pore structure were found to be most influential on the behavior of coal during conversion processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Coal is a combustible sedimentary rock formed from ancient vegetation which has been compressed between other rock strata. The vegetation was accumulated under conditions that either limited or inhibited its decay. Although it is a rock with one evolution process, it is very heterogeneous in nature and the heterogeneity is brought about by the kind of vegetation deposited (coal type), degree of coalification (coal rank) and range of impurities (coal grade) (Edgar 1983; Warwick 2002; Suarez-Ruiz and Crelling 2008). The rank, type and grade are highly influential on the structural composition of coal which can be described as a cross-linked macromolecule comprising of heterocyclic monomers joined by covalent and non-covalent bonds (Marzec 2002). The structure is made up of aromatic layers consisting of aromatic rings ranging from one to three rings and linked by aliphatic chains, as shown in Fig. 1. During coalification, the chemical structure gradually changes to an aromatized structure, acquiring more stability and compactness as the methyl carbons present in the aliphatic chain changes to aromatic carbons thus reducing the spacing between the aromatic units (Blayden et al. 1944; Hirsch 1954; Cartz and Hirsch 1960; Manoj and Kunjomana 2012; Manoj 2014, 2016; Ahamed et al. 2019). As this happens moisture and volatiles are lost while carbon and the energy given out per unit weight of coal increases resulting in different classes of coal, as shown in Table 1.
Clusters of aromatic layers linked to the aliphatic structures (Ahamed et al. 2019)
The chemical constituents present in coal structure impact greatly on its mechanical behavior. The composition of organic matter and its degree of decomposition led to varying coal strength and modulus of elasticity of different coals (Pan et al. 2013; Singh and Mrityunjay 2018b; Ahamed et al. 2019) as the chemical structure was altered. Coals of low rank have a high concentration of aliphatic carbon (Cal–Cal) bonds while those of high rank are highly concentrated with the aromatic—aliphatic carbon (Car–Cal) and aromatic carbon (Car–Car) bonds. These aliphatic and aromatic carbon bonds have different bond energies with the aromatic carbon bonds possessing high bond energies to a maximum of 450 kJ/mol while that of aliphatic carbons is 300 kJ/mol (Li et al. 2015; Ahamed et al. 2019). The increase in bond energies affects the structural skeleton strength and hence coal strength increases as coal matures. Also, the reduction in the aliphatic side compounds and the inter-layer space between the aromatic carbons resulting in reduced space between the aromatic units contribute to the level of condensation and solid form of the chemical structure (Manoj and Kunjomana 2012; Manoj 2016). It is this densely packed coal matrix together with the enhanced bond energies that result in improved coal mechanical properties.
Calcite minerals found in cleats or fracture infillings reduces the inter-granular stresses between particles and this affects the frictional and cohesion strength of coal by increasing coal porosity and permeability (Ward 2002; 2016; Susilawati and Ward 2006; Lebedev et al. 2014). However, under dry conditions calcite minerals improve Young Modulus of Elasticity of coal because of its high resistance. They have resistance higher than that of coal material which tends to improve the overall stiffness of coal material (Gao and Kang 2017).
Clay minerals normally found in cleats, fractures or pore spaces are susceptible to swelling with water adsorption leading to fracturing either on the mineral or maceral phase. The fracturing uses up the elastic energy stored within the coal structure and its reduction causes a decrease in the mechanical strength of coal (Faraj et al. 1996; Ward 2016; Xue et al. 2017).
The complexity of coal and variance in properties has necessitated the need to understand these properties and appreciate their influence on different coal utilization processes (Sprunk 1942; Durie 1982). Not only do they vary across different ranks but also in iso ranks. Properties of coal play a significant role on the behaviour of coal during conversion processes as they define its nature. They are also important in establishing its value for different end-use applications (Sprunk 1942; Mitchell 1997; Taylor et al. 1998; Holuszko and Mastalerz 2015). The article conducted a review of numerous relevant literature material, inclusive of those reported since 1942 to the recent material as an endeavor to capture an update on coal properties in relation to its usage.
2 The role played by coal properties on coal utilization technologies
2.1 Carbonization
Coke is vital during metallurgical processing as it acts as a fuel source, spacer in the blast furnace, reducing agent and carburizes the reduced iron. These roles are quite challenging as coke gets exposed to severe thermal, physical and chemical conditions that no other material is capable of (Devasahayam and Sahajwalla 2013). As a result of this, it is important to control the quality of coke through proper coal selection since carbonization is highly influenced by coal properties. Several coal properties are essential for the accomplishment of the carbonization process.
2.1.1 Organic composition
The organic composition of coals comprises of macerals which are organic substances derived from plant material. The degree at which the macerals have metamorphosed during coal formation era affects the properties of the produced coke (Jasienko et al. 1981; Jee 2012; Devasahayam and Sahajwalla 2013) as it impacts on the ability of coal to melt when subjected to increasing temperatures and forming a coherent residue on cooling. The comparative proportions of both reactive and inert constituents in macerals are influential on the strength of the final carbonized product. Some of the maceral components will melt upon heating making coal to act as a pseudo-liquid material at various temperature ranges thus affecting the strength of coke and coking operations (Patrick 1974). Knowing the coal rank is important because as the quality of coal improves, it molecular structure gets ordered with an increase in aromatic carbon structure while the aliphatic carbon structure decreases (Dı´ez et al. 2002; Jee 2012; Devasahayam and Sahajwalla 2013). The ordered structure means more of the unreactive structure as the aromatics are more stable and will need improved conditions for reactions. Due to this, high-rank coals are inappropriate for the carbonizing process as it requires more reactive material while low-rank coals with more aliphatic structures are preferable.
2.1.2 Chemical composition
The content of volatile matter together with fixed carbon determines the yield of tar, oil, and coke. High volatile content will result in coal yielding large amounts of oil and tar. The increased amount of volatile matter is due to the presence of resins and spores with unusually high volatile compounds. Coals considered good for coking contain volatile matter in the range of 15% to 45% (Sprunk 1942; Jasienko et al. 1981; Devasahayam and Sahajwalla 2013). Increased quantities of fixed carbon in coal will yield large quantities of coke. The production of coke is directly related to the amount of fixed carbon regardless of the type of coal being carbonized.
Coal with high calorific value, carbon and ash content as well as high fusion temperatures results in the production of coke that is slightly harder and less friable (Gupta et al. 2005). While increased oxygen content results in low quantities of tar being distilled as compared to those distilled from coals containing low oxygen with the same amounts of volatile matter mainly because oxygen tends to act as an inert material.
Coals with high moisture, nitrogen, hydrogen, and oxygen content result in the distillation of high volumes of coke gas. It can therefore be concluded that the yield of coking gas increases with an increase in the volatile matter. However, the coke produced from these properties have the tendency to expand and it is this expansion factor that should be given detailed/comprehensive attention during coal selection since high expanding coals can cause damage to coke ovens (Sprunk 1942; Patrick 1974). Low moisture content leads to an increase in bulk density of coal and the degree of rocking attained inside the oven chamber thus contributing to the enhancement of coke quality. Dry coal charging processes capable of reducing moisture in coal by a maximum of 5%–6% has been developed (Nomura et al. 2004). This has resulted in less heat being used during carbonization, increased productivity and improved coke quality being achieved.
Small amounts of sulphur and phosphorus in coal is vital for metallurgical processing as both components are highly deleterious affecting the quality of products. Sulphur impacts negatively on the mechanical characteristics of steel by inducing brittleness during the heated state (red-shortness). While phosphorus tends to increase the development of brittleness in a material at reduced temperatures (cold-short) (Devasahayam and Sahajwalla 2013).
2.1.3 Inorganic composition
Great attention needs to be given to the composition of ash, most particularly the presence of Al2O3, SiO3 and alkalis. Silica strongly affects the silicon content of hot metal while alumina affects the fluidity of slag (Ward 2002; 2016; Devasahayam and Sahajwalla 2013). Attempts have been made to decrease the melting point of coal and coke as well as the silicon content of molten metal by simultaneously injecting pulverized coal and dolomite (Yamagata et al. 1990). This was to address the inactiveness of the deadman resulting from the formation of material with high melting temperatures outside the raceway. The presence of alkalis in ash leads to their sublimation in areas of high temperatures in the furnace. They then re-condense in areas of reduced temperatures decreasing the mechanical strength of coke at the bottom part of the furnace, forming scaffolds and attacking furnace lining refractory (Ward 2002; Matjie et al. 2011; Creelman et al. 2013).
2.1.4 Mineral matter content
Ash content increases the fuel consumption of the blast furnace by affecting the heat balance in the furnace as the carbon content and calorific value of coal are reduced by the requirement of additional fluxes essential for removing ash as slag (Oshnock 1995; Devasahayam and Sahajwalla 2013). Also experienced is the negative effect on the stability of the blast furnace operation because of the catalytic influence on the reactivity of coke and the alkali attacks (Berkowitz 1979; van Krevelen 1993; Ward 2002; Miller 2017).
2.1.5 Coal grindability and handleability
Coal grindability determines the amount of power and throughput required to crush coal to the targeted size by a system used for preparing coal. This property is directly associated with the composition of macerals. It is also influenced by the rank of coal and the amount of mineral matter contained in coal. Low volatile metallurgical coals having a grindability index of 80, get easily milled thus requiring less power for a given coal throughput. Also, they allow for an increase in milling capacity hence an increase in rates of injection without added capital expenses on the added milling capacity (Anon 2 2001).
As the injection rates increase the handleability of coal gets increasingly important because the injection system at high injection rates gets easily affected by coals with poor flow and handleability characteristics. Poor coal handleability in most cases leads to transfer lines getting blocked for the dense phase in the transport system (Bennett and Holcombe 1994) as well as jeopardizing the capability of the system to ignite. In order to assess the handleability, experiments have been conducted (Black et al. 2005) to predict the characteristic behavior of bulk material in hoppers through assessment of characteristics such as friction of walls, compressibility, permeability and cohesion.
2.2 Carbon fibre production
Carbon fibre can be produced through the distillation of carbonaceous natural resources like coal and petroleum to attain pitch as a precursor. Pitch extracted from coal, a by-product of the carbonization process during the production of metallurgical coke, constitutes a valuable aromatic hydrocarbon source essential as a raw material for manufacturing different carbon materials. The carbon materials are derived from the thermal transformation of its highly distributed macromolecules containing high aromatic carbon content and aliphatic constituents substituted by large polycyclic aromatic hydrocarbons (PAH) (Kim et al. 2016a, b; Yang et al. 2016; Apicella et al. 2017; Hiremath et al. 2017). However, scarce information on the chemical structural composition of the precursor pitches which is influential on the yield, quality of the resultant carbon material and that of final products attained from their thermal decomposition/treatment makes it a challenge in manufacturing carbon materials. Several studies have been conducted on carbon fibre production using coal-tar pitch as raw material (Table 2).
Successful production of CFs is dependent on the spinning process which in-turn is influenced by coal parameters such as chemical structural composition, structural molecular weight, and proximate properties.
2.2.1 Chemical structural composition
The oxygen bonding structure is important in the production of CFs due to its effect on the composition of pitch used in CF production. Oxygen atoms in coal extract with high oxygen ratios are not directly bonded to the aromatic carbons, which are the major constituents of the pitch. Rather, they are bonded to hydrogen atoms when the energy available for a reaction is relatively low. The oxygen atom positioned at the CH-chain tail such as in the structure formation of low ranked coals makes the coal increasingly reactive and it is this high reactivity that makes it extremely difficult to spin fibres from this type of coal (Fathollahi et al. 2005; Takanohashi et al. 2008; Lee et al. 2019). Also of importance is the double bond of the carbon–carbon atoms. A strong double bond is indicative of a strongly formed aromatic structure that is highly stable. However, this high aromatic structure with solid contents only tends to make it challenging for coal to attain the structural chain required for a pitch with spinnable behavior (Cooke et al. 1986; Odeh 2015; Kim et al. 2014, 2016a, b; Lee et al. 2019).
The aliphatic low molecular components i.e. the methyl groups are essential for pitch spinnability. The carbons connecting the two aromatic rings like a bridge result in the production of CFs with thinner diameter because of the improved linearity of the raw material (Kim et al. 2014, 2016a, b). It is important though that they remain in the structure without bridging because they turn into volatile material during spinning since they decompose easily at relatively low temperatures making it difficult to maintain the shape of the fibre (Yoshida et al. 1982; Okuyama et al. 2004; Takanohashi et al. 2008; Lee et al. 2019).
Minimal aliphatic structures present in coal, in particular, high-rank coal like bituminous coal and considered appropriate for carbonization, renders it a challenge spinning CF since high content of aliphatic carbons results in a linear structure which is more spinnable. Additional hindrance to the spinnability process is high content of aromatic carbons (Kim et al. 2016a, b; Lee et al. 2019). However, CFs with uniform and clear surfaces can be successfully manufactured from coal with high aromaticity and low aliphatic carbons by producing pitch with relatively high aliphatic content, high oxygen content and yet relatively low reactivity through chemical refining technique (Wang et al. 1998; Watanabe et al. 1999; Okuyama et al. 2004; Rahman et al. 2013; Yang et al. 2016; Kim et al. 2016a, b; Lee et al. 2019). Blending coal tar pitch with other precursors such as petroleum pitch and subjecting the coal tar pitch to solution or thermal treatment can improve the quality of the resultant precursor pitch (Tables 3, 4).
The aliphatic content was improved by blending and treating the coal tar pitch. Initially, the coal tar pitch had low aliphatic content, high aromaticity and oxygen content. Subjecting it to solvent/heat treatment and blending improved the aliphatic structures while the aromaticity and oxygen content was reduced thus rendering the resultant pitch appropriate for carbon fibre spinning. The morphology of the resultant CFs also was improved (Fig. 2).
Carbon fibres produced from different spinnable pitches had a smooth and uniform structure. The fibres showed no sign of fusing, an indication that the stabilization process was a success (Yang et al. 2016). However, the resultant CFs differed in diameters. Coal tar pitch yielded CF with a bigger diameter (15 µm), followed by a pitch from hyper coal at 9 µm and a blend of NCO and coal tar pitch at 7.6 µm. The tensile strength of hyper coal-based CFs was 1150 MPa while that of NCO/CT based CFs was 1800 MPa (Kim et al. 2016a, b; Yang et al. 2016).
Although blending of coal tar pitch with other precursors has been done to achieve the desired results, it can have adversarial effects on the quality of the resultant pitch. Studies conducted in the past (Giray et al. 2013; Hiremath et al. 2017; Zabihi et al. 2019) indicated that adding coal tar pitch to other precursors can drop their charge density consequently leading to a reduction in the repulsion forces hence a decrement in the conductivity of electricity. It also leads to an increase in pitch viscosity and this can be detrimental in achieving dense and smooth fibres. It has been revealed that the addition of more coal tar pitch to polyacrylonitrile (Huson 2017) had a negative impact on the produced carbon fibres (Fig. 3). There was a change in the colour of the fibre mats to brownish and the packing of the carbon fibres got thicker. Also affected was the morphology of the carbon fibers as it got uneven with a surface that has lost its smooth appearance.
Carbon fibers from Polyacrylonitrile precursor with 0% coal tar a, 25% coal tar b, 50% coal tar c (Modified after Zabihi et al. 2019)
The defects on the quality of the resultant CF has also been shown through the blending of coal tar and pyrolysis fuel oil (PFO) (Lee et al. 2019), as shown in Fig. 4. When the amount of pyrolysis fuel oil (PFO) was more in the blend, the thermal stability of the pitch was lowered resulting in fibre breakage during spinning, Fig. 4a. Although the pitch blend of 300% coal tar did not result in the breakage of the fibres, the surface of the CFs was affected, Fig. 4c. The effect on the shape of the fibres could have been due to the uneven distribution of molecular weight (Baumgarten 1971; Deitzel et al. 2001; Okuyama et al. 2004; Takanohashi et al. 2008; Zabihi 2012; Hiremath et al. 2017; Huson 2017; Lee et al. 2019) as the highly aromatic carbons in coal tar pitch mixed with and overpowered the aliphatic carbons in polyacrylonitrile/petroleum pitches hence poor spinnability.
Carbon fibers from pyrolysis fuel oil with 100% coal tar a, 200% coal tar b, 300% coal tar c (Modified after Lee et al. 2019)
2.2.2 Molecular weight distribution
Molecular weight influenced by aromaticity, is important in CF production. High carbon-hydrogen mole ratio indicative of high aromaticity results in large molecular weight due to the polycyclic structure. And because of this high molecular weight distillation is rendered ineffective during the preparation of spinnable pitches. The distillation temperature would, therefore, need to be increased in order to have improved yields of spinnable pitches (Kim et al. 2016a; Lee et al. 2019). On the other hand, the high content of low molecular weight compounds results in easy breakage of the fibers because of the volatile matter given out during the spinning process. Also, an inconstant molecular weight distribution results in the thickness of the fibre being non-uniform due to partial presentation of high molecular weight material (Lee et al. 2019).
2.2.3 Fluidity
Coal fluidity is essential during the preparation of the precursor pitch. A detailed attention is vital in controlling the development of mesophase (presence of liquid crystal in an isotropic phase and tends to increase pitch viscosity, Fig. 5). Existence of partial mesophase development can be detrimental to fibre spinning process because of variances in the density and viscosity of the co-existing isotropic and anisotropic phases (Park and Mochida 1989; Kim et al. 2016a, b; Yang et al. 2018). The viscosity of the pitch can adversely affect the initial shape of the droplet, path of the jet and fiber diameter. When the viscosity increases the fibre diameter increases thus negatively affecting the pitch spinnability (Deitzel et al. 2001; Zabihi 2012; Hiremath et al. 2017; Huson 2017).
Isotropic phase a, formation of mesophase in an isotropic phase b, anisotropic phase c (Modified after Huson 2017)
Coal tar pitch presents a viable precursor alternative in that it facilitates the process of stabilization by increasing the heat that is released during the process thus lowering the energy consumed for stabilization of fibres i.e. minimized stabilization temperatures with high heat of reactions are essential for an efficient thermal stabilization process (Kissinger 1957; Zabihi 2012). In addition, its high aromaticity is indicative of high thermal stability, a quality essential for the steady spinning of the pitch possessing high softening point since melt spinning is normally carried out at a temperature 50 °C higher than the softening point (Kim et al. 2013, 2014; Lee et al. 2019).
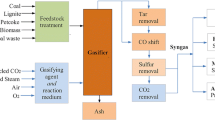
2.3 Coal gasification
Coal gasification involves coal reaction with air, oxygen, steam or their mixture to produce a gaseous fuel termed synthesis gas. The principal controlling properties of coal during the gasification process are its reactivity, ash and slag properties, particle size and coal caking and swelling.
2.3.1 Coal reactivity
Coal reactivity is one fundamental property essential for the success of the coal gasification process (Ozer et al. 2017). It is influenced by the degree of coalification, organic composition (macerals), mineral contents, coal porosity and surface area.
-
(1)
Degree of coalification
Degree of coalification is reliant on the organic composition of coal (macerals), chemical structural composition, mineral matter content and pore structure (Jones et al. 1985; Alonso et al. 2001; Méndez et al. 2003; Choudhury et al. 2008). Coal has been classified using the carbon content of its macerals into a low rank containing carbon content less than 80% and a high rank containing more than 80% carbon. The catalytic effect of coal minerals has been found to be the chief controlling factor of the reactivity for coals in the low rank while the reactivity for high ranked coals is mainly controlled by the available active sites found in the coal matrix (Miura et al. 1989). Low ranked coals contain high volatile material and more hetero-atoms inside its organic structure. Their structure also has more open pores and increased oxygen-functional groups as compared to high ranked coals. Furthermore, low-rank coals comprise of a poorly aligned aromatic structure as opposed to high-rank coals composed of better aligned carbon atoms. The amorphous and more open pore structure of low-rank coals makes it more reactive with oxygen and steam (Ozer et al. 2017).
The highly dispersed catalytic minerals, carboxylic acids as well as phenolic-functional groups also contribute to the high reactivity of low ranked coals (Radovic et al. 1983). Also accountable for this high reactivity is the highly concentrated oxygen-functional groups, increased amount of macropores and the distribution of the catalytic in-organic constituents. The functional groups provide the cations with reaction zones hence improving on the reactivity of coal due to catalysts (Ye et al. 1998). They can also act as active zones as well as exchange zones on which alkali elements can be mounted as exchangeable cations (Takarada et al. 1985). Previous studies (Irfan et al. 2011) revealed that once alkaline elements are removed from low ranked coal, its reactivity would be the same as the reactivity of high ranked coal.
The high moisture content of low ranked coals reduces the temperature generated in the combustion area thus decreasing the thermal efficiency of the gasification process. As a result of this, coal feed rates of low ranked coals need to be more than those of high ranked coals in-order to improve the quality of syngas by increasing the heating value (Durie 1982; Shadle et al. 2002; Ozer et al. 2017; Fletcher 2017).
Low rank coals contain high volatile matter as compared to high-rank coals. This characteristic tends to enhance coal reactivity because the activation energy would be lowered (Hakvoort et al. 1989; Küçükbayrak et al. 2001). It also improves on the carbon burnout, ease of ignition and has been used to predict the number of hydrocarbons that can be given out during the gasification process (Hattingh et al. 2011).
-
(2)
Organic composition (coal macerals)
The organic structure of coal is made up of three maceral groups; vitrinite, inertinite, and liptinite/exinite. It is the composition of these constituents that is influential in the transformation of coal structure during the conversion process since maceral groups react differently (Suarez-Ruiz and Crelling 2008; Suarez-Ruiz 2012).
Behavioural characteristics of macerals during the coal gasification process are as followed:
-
①
Vitrinite—the most abundant maceral group derived from woody tissues with qualities and the reactivity varying with the rank of coal. Their high hydrogen content has resulted in their capability to melt, swell and soften after coal has been carbonized. The mobile carbon undergoes nanoscale re-arrangements to align layers that will easily coalesce when subjected to heat treatment leading to the formation of extended layers as well as the loss of edge sites and that of reactivity (Beeley et al. 1996; Zhuo et al. 2000; Zhang et al. 2008). Coals rich in vitrinite produce char with high porous structure and large amounts of mineral matter mainly calcium and potassium improving its catalytic activity. Infact, vitrinite-rich char has high gasification reactivity regardless of the presence of minerals (Megaritis et al. 1999; Sun et al. 2004).
-
②
Liptinite/exinite—this maceral group originates from hydrogen-rich sources like spores, leaf cuticles, plant algae and is very reactive. Although it fails to swell or agglomerate, it has the capacity to melt during heating and hence most of its mass proportion is lost during pyrolysis (Zhuo et al. 2000; Mahagaokar 2004).
-
③
Inertinite—the most unreactive maceral group rich in carbon and originates from biodegraded wood. Inertinite fails to melt when subjected to heating and would lose only a slight quantity of its mass through pyrolysis and as a result it is a poor gasification agent due to its unreactivity towards CO2. Coals rich in inertinite possess a structure that is highly oriented with a high degree of crystalline packing (Sharma et al. 2000; Van Niekerk et al. 2008). They are also more aromatic with more polycondensed contents and have been found to be appropriate for combustion and gasification technologies (Sun et al. 2003; van Niekerk et al. 2008).
Inertinite and vitrinite are responsible for the carbon structure conversion of coal during gasification while the change in the mineral matter has been attributed to the presence of inertinite (Gupta et al. 2005; Matjie et al. 2011).
-
①
-
(3)
Porosity of coal and surface area
Coals of low rank have a highly open porous structure with increased active sites for oxygen-steam reaction during the gasification process. A correlation has been established between the reactivity of coal and its micro porosity basing on the active surface area that is directly proportional to the entire surface area (Irfan et al. 2011). Reactions of gasification mainly take place on the surface of macropores instead of micro pores due to the highly concentrated active zones in the former than in the latter. In fact, macro pores have been found to materialize in crystallite edges or sites that have contact with organic impurities catalytically active whereas the micropores were found to be made up of less reactive basal planes (Hurt et al. 1998).
Also, there is a relation between active sites concentration or surface area reaction and the accessibility of reactants in the gaseous state to the active sites, indicating that the structure of pores can be linked to charcoal reactivity (Slaghuis et al. 1993; Ng et al. 1988; Kajitani et al. 2006; Çakal et al. 2007). When a critical value (70%–80%) for char porosity is attained fragmentation into finely grounded particles occurs improving the reactivity hence char conversion (Jayaraman and Gokalp 2015). Coals with low surface area and porosity have low reactivity (Adschiri et al. 1986; Arenillas et al. 2004; Wu et al. 2009; Ozer et al. 2017).
-
(4)
Mineral matter in coal
The most common minerals found in coals used for gasification comprise mainly of kaolinite with small quantities of quartz, illite, dolomite, calcite, pyrite and small traces rutile and phosphate (Ward 2002, 2016; Matjie et al. 2011). Calcite together with dolomite appear within vitrinite macerals while Ca and Ti appears as inorganic elements that associate with the organic matter. Sodium, chlorine and sulfur were also found in some coals.
-
(5)
Effect of catalytic behaviour of coal minerals
The interaction of mineral matter with the carbon matrix in coal has a great effect on coal reactivity. Mineral matter can be discrete grains, embedded within or attached to the carbon matrix as is the case with high-rank coals. The embedded mineral matter has a high specific heat capacity and absorb more heat leading to temperature decrease and subsequently low coal reactivity (Wigley et al. 1997; Hattingh et al. 2011). Mineral matter occurring as dissolved salts trapped inside the pores and/or are attached chemically to the carboxylic acids and the phenolic groups in the form of exchangeable ions have strong effect on char reactivity of low ranked coals than that of high ranked coals (Quann and Sarofim 1986; Huggins and Huffman 1995; Ballantyne et al. 2005).
Also of great importance on coal reactivity and hence coal gasification rate are the inorganic metal ions such as K2+, Na2+, Ca2+ Mg2+, and Fe2+ as they get to trade ions with hydrogen ions (H+) making up the carboxylic acids or phenolic groups. Ca2+ influences the syngas composition by promoting the water–gas shift reaction while Ca and CaO organically bound by calcite decomposition impacts on the catalytic effect during coal gasification (Radovic et al. 1983; Wigley et al. 1997; Skodras and Sakellaropoulos 2002; Chodankar et al. 2007; Hattingh et al. 2011; Ozer et al. 2017).
2.3.2 Properties of ash and slag
-
(1)
Mineral matter conversion into ash
The fusing, melting and partial crystallization on cooling of mineral matter results in the formation of cristobalite, magnetite, mullite, diopside and anorthite. These new formed phases and their properties (chemical composition, fusion temperatures and slag viscosity) need to be taken into account to reduce risk of clinkering, ash deposits and agglomeration, fouling and corrosion. The occurrence of any would lower the efficiency and capacity of the gasification process resulting in an increase on the power cost (Zevenhoven-Onderwater et al. 2001). Coal components formed at temperatures lower than the ash melting temperature such as magnesia (MgO), ferric oxide (Fe2O3), silica (SiO2), lime (CaO), alumina (Al2O3), titanium oxide (TiO2), alkali compounds (K2O, Na2O), some sulphur and chloride compounds may react with each other under high gasification temperatures forming several different compounds with interstitial vesicular glass of calcium to iron-rich calcium (Hu et al. 2006; Kosminski et al. 2006a, b, c; Gupta et al. 2007; Bai et al. 2010).
-
(2)
Ash fusion temperature
Ash fusion temperature defines the melting behavior of ash during the gasification process. When gasifying low-rank coals, the operating temperature needs to be less than the temperature at which the ash fuses to avoid molten fused deposits forming whereas for high rank coals the operating temperature needs to be higher than the temperature at which fusion of ash occurs. This would lessen challenges related to slag formation by allowing the melted heated ash (slag) to flow down the reactor walls and drain easily from the reactor (Li 2010). Ash fusion temperature was found to reduce with increasing SiO2 wt % signifying its fluxing potential on ash and it increased with increasing Al2O3 content (Gupta et al. 1998).
-
(3)
Slag viscosity
Coals with similar ash fusion temperature can have differing slag viscosities and portray different slagging behavioural traits because the ash fusion temperature is dependent on ash mineral composition. Therefore, ash having varying compositions may exhibit similar ash fusion temperatures but dissimilar slag viscosities (Gupta et al. 1998; Lolja et al. 2002; Trapp et al. 2004; Liu et al. 2013). Slag viscosity is highly influenced by the temperature and chemical composition of ash. When temperatures are low the slag gets more viscous with the poor erratic flow while high temperatures lead to fast refractory wear (Groen et al. 1998). Slag viscosity increases with increasing alumina and silica but reduces in the presence CaO, FeO, MnO2 and MgO (Wang and Massoudi 2013).
-
(4)
Ash fouling, deposition and agglomeration
Some mineral constituents can get entrained within the stream of the synthesis gas being deposited on the cool reactor surfaces or wall tubes causing corrosion, slagging and fouling. They can even negatively impact on the devices for emission control and other equipment (Vamvuka et al. 2008). The deposition of inorganics affects the transfer of heat within the reactor thus altering the operation and capacity of the reactor hence the system efficiency. Fouling and deposition challenges are promoted by the existence of the alkali metals (alkaline earth metals, chloride, silicon, Na) and sulphur in ash (Ward 2002, 2016; Creelman et al. 2013; Ozer et al. 2017). Furthermore, vapours due to alkali metals lead to the final end-use equipment being corroded. The dominant alkali species are sodium and potassium hydroxides as well as chlorides. Small amounts of chlorine contained in some coals during the gasification process produce gaseous HCl that interact with metal compounds accumulating in syngas coolers leading to deposits of chlorides like FeCl2, NaCl, and CaCl (Kosminski et al. 2006a). Ash agglomeration results in channel burning. It also leads to challenges related to pressure drop in the reactor and the reactor performance being unstable hence cut-backs of the gasification loading (van Dyk and Waanders 2007).
2.3.3 Coal particle size
Large sized particles have a tendency to reduce the reactivity of coal and hence poor syngas quality. Also experienced in relation to large sized particles are challenges with transporting the large particles in the reactor and start-up difficulties of the equipment. Gasifying low rank coals using large particle sizes can lead to the formation of clinker and channeling (Speight 2014). Fine coal particles, on the other hand, reduce carbon conversion thereby decreasing syngas production (Dittus and Johnson 2001).
2.3.4 Coal caking and swelling
Coal caking and swelling apply in particular to bituminous coal and it leads to particles adhering to one another and forming clusters to create a phase known as metaplast. The forming agglomerate phase can interrupt flow patterns of gas and thus reduce the thermal efficiency. However, the tendency of coal to cake reduces as the volatile matter content falls below 20 wt % (Shadle et al. 2002). The swelling of coal affects the reactivity as the operation of the reactor gets unstable due to fluctuations in pressure drop and channeling. Also, the size of coal particles and density of the produced char are affected (Jayaraman and Gokalp 2015). In addition, caking coals tend to form tars with high molecular weight which if not controlled can lead to blocking and fouling of lines, heat exchangers and filters thus tampering with the system sufficiency.
2.4 Liquefaction
Coal liquefaction involves the conversion of coal into liquids. There are two methods for liquefying coal; direct liquefaction, also known as hydrogenation involves the production of liquid fuels directly from coal using a hydrogen donor solvent and indirect liquefaction. Indirect liquefaction process coal is liquefied through the Fischer–Tropsch (FTS) process. The FTS process uses synthesis gas from the gasification process and converts it into liquidous hydrocarbons and associating oxygenated compounds using different catalysts from Group VIII. The product mixture depends on the carbon monoxide to hydrogen ratio (Berkowitz 1979; Durie 1982; Höök and Aleklett 2009; Khan 2011; Vasireddy et al. 2011; Heydari et al. 2016; Miller 2017).
Coal consists mainly of solid organic matter with an atomic hydrogen to carbon ratio ranging from 0.1 to 4. Therefore to produce liquids from coal requires breaking down the chemical structure into compounds with a low molecular weight that is in a fluid state under ambient conditions, eliminating heteroatoms in particular O, N and S and increasing the hydrogen to carbon ratio. These generate saturated aromatic compounds and stabilizes the resultant liquids (Durie 1982; Gibson 1983).
Different coal properties have varying significant impact on the coal liquefaction process. Comprehensive studies have been carried out on several chemical and physical characteristics of coal and their importance during the conversion of coal into liquid products (Table 5).
2.4.1 Degree of coalification
The degree of coalification is the most crucial coal parameter in liquefaction. Coal rank through vitrinite reflectance can be used to determine the potential of a coal seam for producing hydrocarbons. A model by Tissot and Welte (1984) was used to predict the primary hydrocarbon potential through the evolution stages of coal (Fig. 6). Coals which have passed the evolution phase of diagenesis (a phase when coal significantly loses oxygen as carbon dioxide and water) and reached to the catagenesis stage (when hydrogen and carbon are lost due to oil and gas generation when the organic matter under-goes thermal decomposition as temperature increases with burial in the sedimentary basins) have great potential for generation of hydrocarbons (Tissot and Welte 1984; Suarez-Ruiz and Crelling 2008; Suarez-Ruiz 2012; Singh and Mrityunjay 2018a, b).
Main stages of maturity relating coal organic composition and rank (Singh and Mrityunjay 2018a)
A high liquefaction yield is obtained near the transition from brown coal to bituminous coal i.e. coals with carbon content in the range of 80% to 85% (Randolph 1944; Vasireddy et al. 2011). When the carbon content is between 87% and 89% (medium volatile bituminous coal), syncrude yield declines sharply. Coals of high rank with a carbon content greater than 90% (low volatile bituminous coal and anthracite coal) are mostly unreactive (Vasireddy et al. 2011). The yield of oil decreases and that of moisture and carbon dioxide increases with a decrease in the carbon content of the bright coal hydrogenated (Randolph 1944). Hydrocarbon yield is limited by high oxygen content approaching 30 wt% dmf. The oxygen reacts with carbon and hydrogen to form water and carbon dioxide during oxygen elimination processes. In addition, moisture content needs to below as it has to be removed during the hydrogenation (Durie 1982; Vasireddy et al. 2011).
-
(1)
Chemical structure
The differences in the yield of syncrude from different coal ranks can be attributed to the chemical configuration of coal with the potential to influence the nature of its chemical reactivity (Zhou et al. 2016). Low ranked coals have an increased content of aliphatic carbon (Cal–Cal) bonds whereas high ranked coals have an increased concentration of aromatic-aliphatic carbon (Car–Cal) bonds and aromatic carbon (Car–Car) bonds. An enhancement in the carbon content results in an increased number of aromatic carbon bonds (Shi et al. 2013; Zhou et al. 2016).
The bond energies of the aliphatic carbons and the aromatic carbons are different with the aromatic carbons having high bond energy (Fig. 7) (Shi et al. 2013; Li et al. 2015; Ahamed et al. 2019). The increased bond energy reduces the reactivity of coal as more energy will be required to breakdown the aromatic structure hence the low syncrude yield by coals of high rank. Coal liquefaction is favoured by the simple skeletal structure that comprises mainly of simple aromatic rings which are less likely to be removed from the lignin basing on the building block for phenyl propane (Durie 1982).
Fig. 7 Aliphatic and aromatic carbon bonds in relation to carbon content a, bonding energies for major covalent bonds in coal b (Ahamed et al. 2019)
-
(2)
Porosity
Coal porosity is most relevant in relation to direct liquefaction. Determining the surface area and pore volume is essential for establishing the correlation between the surface area and chemical reactivity with regard to the access of gases and liquids to the interior of coal particles (Vasireddy et al. 2011). Coals from different ranks have a structure made up of varying pores being the macropores having diameters greater than 50 nm, mesopores with pores ranging from 2 nm to 50 nm and the micropores with pores less than 2 nm. The macropores reduce with an increase of coal rank while the mesopores and micropores increase with an increase in the rank of coal (Zhu et al. 2016).
2.4.2 Organic composition
The organic composition of coal is directly related to the oil chemistry and potential of coal to generate hydrocarbon compounds hence its importance in the liquefaction process. Organic constituents in coal differ greatly in ease of liquefaction and yield of the various products. The type of maceral usually with the highest degree of capacity for liquefaction is fusain. Fusain is a mixture of opaque fibers termed fusinite and translucent or semi-opaque material ranging from 10% to 30%. The yield of liquefaction is dependent primarily on the amount and nature of the translucent material. The translucent material consisting of constituents such as leaves, spores, resins, cuticles are usually considered good for hydrogenation (Sprunk 1942; Randolph 1944).
The estimated oil yields based on weight that can be obtained from clean macerals are 78%–80% for liptinite, 6% to 10% for vitrinite while the inertinite yields 0%–2% of liquefaction products. The vitrinite macerals readily dissolve and as such assist in conversion of coal into liquid products through their natural thermoplastic characteristics. The high yields by liptinites is due to the presence of aliphatic structures while the inertinites simply remain inert during a conversion process (Saxby and Shibaoka 1986; Bertrand 1989; Hunt 1991; Newman et al. 1997; Peters et al. 2000; Sykes 2001; Suarez-Ruiz and Crelling 2008; Singh and Mrityunjay 2018a, b). Generally, for coal to be considered suited for liquefaction, its organic matter should be 10% to 20% Type I kerogen or 20% to 30% with Type II kerogen (Powell and Boreham 1994). Type I kerogen is from a different precursor with high aliphatic content from various sedimentary environments whereas Type II is related to the planktonic organic matter found in open marine as well as freshwater from lacustrine environments (Suarez-Ruiz 2012).
The liquid yield and the nature of solid residue from coals of constant coal rank condition is highly dependent on the maceral composition (Kalkreuth et al. 1986). Coals rich in vitrinite and exinite have high hydrogen content and increased hydrogen has been found to increase the products of liquefaction (Singh et al. 2013; Mishra et al. 2018).
2.4.3 Size of coal particle
Particle size is deemed a significant parameter during coal liquefaction conversion. Small-sized particles have reduced distances of diffusion by the solvent molecule in order to access the core of the coal matrix for the extraction of liquid products. Large-sized particles result in a diffusing component being consumed quicker than its diffusion into the particle due to the long distance it has to travel to reach the core of the particle. This leads to a reaction in zones near the center of particle which in turn will lead to an increased level of mixing of free radicals. The free radicals result in products with stable high molecular weight being formed which is undesirable during liquefaction (Schlosberg 1985; McMillen and Malhotra 2006; Li et al. 2008a, b). Extraction yields have been found to be more for small sized particles as they provide increased coal surface area and enhance the ability of the solvent to easily penetrate into the coal structure hence an increment of yields (Giri and Sharma 2000; Heydari et al. 2016).
2.4.4 Inorganic matter
The heterogeneity of coal has resulted in mineral matter distribution of various forms, compositions and associations. These mineral matter constituents are not really inert during the conversion of coal into liquids and can affect the distribution and yields of liquid products. It has been reported to act beneficially as a catalyst for liquefaction. Pyrite and alumino-silicates exert a catalytic effect during coal hydrogenation. Furthermore, the mineral matter can be detrimental on the equipment used for liquefaction by formation of scale and deposits on the reactor walls and can lead to blockage problems due to formation of compounds like calcium carbonate adversely affecting the reactor capacity (Durie 1982; Heydari et al. 2016).
The ash content of coal should not exceed 10 wt % as constituents likely to form ash exist as cations associating with carboxylic acid compounds in coal. Their nature and quantity can have a significant effect on the behavior of coal as it undergoes thermal decomposition and aid expedite oxygen elimination. In addition, high ash content adversely affects the liquefaction process by negatively impacting the reactor capacity. Also, a ballasting impact relating to the recoverable yield of distillable oil can be experienced as the added catalyst gets deactivated by the presence of mineral matter (Durie 1982).
Coals with vitrinite reflectance ranging from 0.57% to 0.62%, hydrogen to carbon atomic ratio falling in the range of 0.73 to 0.87 with reactive maceral content of 72% to 96.8% and volatile matter content in the range of 29.4% to 57.1% are considered suitable for conversion into liquid products (Davis et al. 1976).
3 Discussion
Physical and chemical characteristics of coal are essential in determining coal usage. The level of coalification reached by coal can be deemed the primary factor as it influences all other coal parameters. Different ranks of coal vary in the composition of macerals, plasticity, porosity, reactivity, chemical structural composition, and the inorganic compounds, all these being the parameters essential in selecting the coal technology to implement for a particular coal. Also of importance is the organic/maceral composition of coal as it is responsible for most of the coal benefits such as its energy output during combustion, its role in metallurgical processing, its capacity for in situ methane absorption and its potential as an alternative hydrocarbon source (Sprunk 1942; Falcon and Snyman 1986; van Krevelen 1993; Nas 1994; Mitchell 1997; Holuszko and Mastalerz 2015). An understanding of the effect of maceral composition during coal conversion process is essential as the variation in its physical and chemical properties influences coal utilization technologies. High yields of volatile matter can be achieved from liptinite upon heating making it ideal for bitumen and tar production (Falcon and Ham 1988). Vitrinite found in bituminous coal of low rank is suitable for liquefaction process due to its high hydrogen content, more aliphatic structures and low aromaticity (Falcon and Ham 1988; Sun et al. 2003). Inertinite molecular structure characterized by low hydrogen and volatile matter content, higher thermal stability, high aromaticity and condensation in comparison to other maceral groups renders them the least reactive yielding low gas and tar during pyrolysis (Zhao et al. 2011).
The composition of macerals influences the composition of elements in coal which in turn impacts on the coal reactivity. Vitrinite contains less hydrogen than liptinite but more oxygen while inertinite has high carbon content and lower hydrogen than vitrinite but similar oxygen (Durie 1982; Suarez-Ruiz and Crelling 2008; Suarez-Ruiz 2012). It is the arrangement of these elements in a coal structure that determines the reactivity of different coals. These elements can exist either as aromatic or aliphatic structures. Coals consisting of more aromatic structures as compared to the aliphatic structures have low reactivity as more energy will be needed to breakdown the covalent bonds existing in the aromatic structures. Hence for easier coal conversion more aliphatic structures are essential.
However, because of the intimate association of macerals within coal, determining the effect of one maceral group can be a challenge. As a result, separation techniques such as chemical fractionation, density separation and centrifuging, have been used to better appreciate the structural properties and the reactivity of individual maceral groups (Maledi et al. 2017). These techniques may have an adversarial effect on the maceral composition hence misleading results.
The degree of coalification together with maceral composition impact on the mechanical behaviour of coal by altering its structure. Coal structure is made up of aromatic and aliphatic carbon structures. As coal matures the aliphatic carbon structures turn into aromatic carbon structures. The high bond energies for the aromatic structures are responsible for the low reactivity of high rank coals while low bond energies for the aliphatic carbon structures renders low rank coals high reactivity (Pan et al. 2013; Li et al. 2015). The functional groups making up the structure of coal are inclusive of methylene, methyl, phenolic hydroxyl and carboxyl groups. The methyl groups exist as substituent in the aromatic nuclei and are responsible for almost 20% of the hydrogen found in coal structure. While 60%–80% of organic oxygen is due to phenolic hydroxyl groups. An appreciable oxygen content found in low rank coals is due to the carboxyl groups (Durie 1982). These functional groups tend to impact negatively on coal strength. Although they hardly react with water, others act as hydrophilic sites attracting water molecules that fill up the capillary pores. The water adsorption leads to more pressure being applied on particles causing the relaxation of the intergranular stresses hence reduced frictional and cohesion strength (Kaji et al. 1986; Yu et al. 2013). An average molecular model have been proposed using vitrinite macerals to gain information on the structure of coal. However, this model was found to be misleading as it was suitable for the particular vitrinite used. It also included a mixture of low molecular weight entities (Durie 1982). As a result, understanding coal structure still remains a challenge because of its complex heterogeneous cross linked polymeric structural networks.
The mineral matter during a conversion process goes through thermal decomposition, disintegration, fusion and agglomeration (Chakravarty et al. 2015). These transformations are essential in providing information with regard to coal suitability for a particular technology (Matjie et al. 2011; Zhang et al. 2011). As the minerals react and undergo transformations there is formation of molten deposits (fouling and slagging) on the walls of conversion units resulting in great operational challenges during the process (Fig. 8).
Fouling deposits on tubes in convective pass (Ma et al. 2007)
The deposits decreases the absorption of heat in the unit thus decreasing the thermal performance as well as efficiency. Previous studies have been conducted where soot blowers using high pressurized air, steam or water jets were used to remove the deposits. However, the method did not prove effective enough to get rid of strongly adhered deposits since excessive blowing led to erosion damage of the equipment (Wee et al. 2005).
A model was devised to determine the usage of coal (Falcon and Snyman 1986). This model is based on the rank and maceral composition of coal. It uses the vitrinite reflectance and the percentage of the reactive macerals to determine the end-use application of coal (Fig. 9).
When applied to Indian coals from Argada and West Bokaro coals (Singh et al. 2015; Singh and Mrityunjay 2018b) the model indicated that the coals were best used as blending coking coals because of the high amount of reactive macerals found in them.
Also developed to assist in determining the use of coal was a Seyler diagram (Fig. 10) basing on the content of hydrogen and carbon (Singh et al. 2015).
Seyler diagram used for determining coal utilization basing on hydrogen and total carbon contents (Singh et al. 2015)
The use of coal in relation to hydrogen and carbon contents can be deduced from the diagram as shown in Table 6.
Although the models have proved applicable in determining the coal utilization technology to implement for those particular coals, they were specifically developed for coals rich in minerals hence difficult to beneficiate for coals varying greatly in maturity and composition of organic matter. Therefore a tool suitable for coals portraying dissimilar characteristics is needed. Furthermore, the properties important in coal utilization are inversely related (Durie 1982; Mishra et al. 2005; Yang et al. 2016) therefore a compromise is necessary for satisfactory performance of the utilization technology deemed applicable.
4 Summary and Conclusion
The degree of coal coalification is the most fundamental parameter in determining the end-use application of coal as it influences all the other coal properties and as such is applicable in all coal utilization technologies. Final influence of coal properties found to be critical on carbonization, carbon fiber production, gasification and liquefaction was summarized in Table 7.
References
Adschiri T, Shiraha T, Kojima T, Furusawa T (1986) Prediction of CO2 gasification rate of char in fluidized bed gasifier. Fuel 65:1688–1693
Ahamed MAA, Perera MSA, Matthai SK, Ranjith PG, Dong-yinc L (2019) Coal composition and structural variation with rank and its influence on the coal-moisture interactions under coal seam temperature conditions—a review article. J Pet Sci Eng 180:901–917
Alonso MJG, Borrego AG, Alvarez D, Parra JB, Menéndez R (2001) Influence of pyrolysis temperature on char optical texture and reactivity. J Anal Appl Pyrolysis 58–59:887–909
Anon 2 (2001) Queensland high energy coals for the PCI market—advantages for low volatile coal. Retrieved from http://mines.industry.qld.gov.au/assets/coal-pdf/hi_energy_
Apicella B, Tregrossi A, Stanzione F, Ciajolo A, Russo C (2017) Analysis of petroleum and coal tar pitches as large PAH. Chem Eng Trans 57:1–6
Arenillas A, Rubiera F, Pevida C, Ania CO, Pis JJ (2004) Relationship between structure and reactivity of carbonaceous materials. J Therm Anal Calorim 76:593–602
Bai J, Li W, Li C-Z, Bai Z, Li B (2010) Influences of minerals transformation on the reactivity of high temperature char gasification. Fuel Process Technol 91:404–409
Ballantyne TR, Ashman PJ, Mullinger PJ (2005) A new method for determining the conversion of low-ash coals using synthetic ash as a tracer. Fuel 84:1980–1985
Baumgarten PK (1971) Electrostatic spinning of acrylic microfibers. J Colloid Interface Sci 36:71–79
Beeley T, Crelling J, Gibbins J, Hurt R, Lunden M, Man C (1996) Transient high-temperature thermal deactivation of monomaceral-rich coal chars. Symp Int Combust 26(2):3103–3110
Bennett P, Holcombe D (1994) Commissioned study on PCI research and future directions. Coal Technology. http://coaltech.com.au/CommissionedStudyonPCIResearchandFutureDirections.html. Accessed 21 March 2019.
Berkowitz N (1979) An introduction to coal technology. Academic Press, New York
Bertrand PR (1989) Microfacies and petroleum properties of coals as revealed by a study of North Sea Jurassic coals. Int J Coal Geol 13(1–4):575–595. https://doi.org/10.1016/0166-5162(89)90107-9
Black N, Whitehouse M, Sullivan K (2005) Improved measurement of handleability for quality control of Australian coals in PCI and carbonisation applications. Australian Coal Research Limited. http://acarp.com.au/abstracts.aspx?repld=C11016. Accessed 12 May 2019.
Blayden H, Gibson J, Riley H (1944) Ultra-fine structure of coals and cokes. In: BCURA conference proceedings, vol 153, p 697
Çakal GÖ, Yücel H, Gürüz AG (2007) Physical and chemical properties of selected Turkish lignites and their pyrolysis and gasification rates determined by thermogravimetric analysis. J Anal Appl Pyrolysis 80:262–268
Cartz L, Hirsch PB (1960) A contribution to the structure of coals from X-ray diffraction studies. Philos Trans R Soc Lond 252(1019):557–602
Chakravarty S, Mohanty A, Banerjee A, Tripathy R, Mandal GK, Basariya R, Sharma M (2015) Composition, mineral matter characteristics and fusion behavior of some Indian coals. Fuel3 150:96–101
Chodankar C, Feng B, Klimenko A (2007) Gasification kinetics of Australian bituminous coal in CO2 environment: unification approach of reactivity. Proc Aust Combust Symp 2007:82–85
Choudhury N, Biswas S, Sarkar P, Kumar M, Ghosal S, Mitra T (2008) Influence of rank and macerals on the burnout behaviour of pulverized Indian coal. Int J Coal Geol 74:145–153
Cooke NE, Fuller OM, Gaikwad RP (1986) FTIR spectroscopic analysis of coals and coal extracts. Fuel 65:1254–1260
Creelman RA, Ward CR, Schumacher G, Juniper L (2013) Relation between coal mineral matter and deposit mineralogy in PF furnances. Energy Fuel 27:5714–5724
Davis A, Spackman W, Given PH (1976) The influence of the properties of coals on their conversion into clean fuels. Energy Sources 3:55–81. https://doi.org/10.1080/00908317608945968
Deitzel JM, Kleinmeyer J, Harris D, Tan NCB (2001) The effect of processing variables on the morphology of electrospun nanofibers and textiles. Polymer 42:261–272
Devasahayam S, Sahajwalla V (2013) Evaluation of coal for metallurgical applications. In: Osborne D (ed) The coal handbook: towards cleaner production, vol 2. Woodhead Publishing Limited, Sawston, pp 352–386
Dı´ez MA, Alvarez RC, Barriocanal R (2002) Coal for metallurgical coke production: predictions of coke quality and future requirements for cokemaking. Int J Coal Geol 50:389–412
Dittus M, Johnson D (2001) The hidden value of lignite coal. In: Gasification technologies conference, proceedings, pp 1–8
Durie RA (1982) Coal properties and their importance in the production of liquid fuels. Fuel 61(10):883–886. https://doi.org/10.1016/0016-2361(82)90084-9
Edgar TF (1983) Coal deposits and properties. In: Coal processing and pollution control, 1st edn. https://che.utexas.edu/course/che3599385/lecture_notes/topic3/chapter2
Falcon R, Ham AJ (1988) The characteristics of Southern African coals. J S Afr Inst Min Metall 88(5):145–161
Falcon RMS, Snyman CP (1986) An introduction to coal petrography: atlas of petrographic constituents in the bituminous coals of Southern Africa. Geol Soc South Afr 2:1–27
Faraj BS, Fielding CR, Mackinnon ID (1996) Cleat mineralization of upper Permian Baralaba/Rangal coal measures, Bowen Basin, Australia. Geol Soc 109:151–164
Fathollahi B, Jones B, Chau PC, White J (2005) Injection and stabilization of mesophase pitch in the fabrication of carbonecarbon composites. Part III: mesophase stabilization at low temperatures and elevated oxidation pressures. Carbon 43:143–151
Fletcher TH (2017) Gasification fundamentals. In: Wang T, Stiegel GJ (eds) Integrated gasification combined cycle (IGCC) technologies. Woodhead Publishing, Sawston, pp 223–256. https://doi.org/10.1016/B978-0-08-100167-7.00006-8
Gao F, Kang H (2017) Experimental study on the residual strength of coal under low confinement. Rock Mech Rock Eng 50:285–296
Gibson J (1983) Liquid fuels from coal. J R Soc Arts 132(5329):41–51
Giray D, Balkan T, Dietzel B, Sarac AS (2013) Electrochemical impedance study on nanofibers of poly(m-anthranilic acid)/polyacrylonitrile blends. Eur Polym J 49(9):2645–2653
Giri CC, Sharma DK (2000) Mass-transfer studies of solvent extraction of coals in N-methyl-2-pyrrolidone. Fuel 79(5):577–585
Groen J, Brooker D, Welch P, Oh M (1998) Gasification slag rheology and crystallization in titanium-rich, iron–calcium–aluminosilicate glasses. Fuel Process Technol 56:103–127
Gupta SK, Wall TF, Creelman RA, Gupta RP (1998) Ash fusion temperatures and the transformations of coal ash particles to slag. Fuel Process Technol 56(1–2):33–43. https://doi.org/10.1016/S0378-3820(97)00090-8
Gupta S, Sahajwalla V, Chaubal P, Youmans T (2005) Carbon structure of coke at high temperatures and its influence on coke fines in blast furnace dust. Metall Mater Trans B 36:385–394
Gupta S, Dubikova M, French D, Sahajwalla V (2007) Effect of CO2 gasification on the transformations of coke minerals at high temperatures. Energy Fuels 21:1052–1061
Hakvoort G, Schouten J, Valkenburg P (1989) The determination of coal combustion kinetics with thermogravimetry. J Therm Anal 35:335–346
Hattingh BB, Everson RC, Neomagus HWJP, Bunt JR (2011) Assessing the catalytic effect of coal ash constituents on the CO2 gasification rate of high ash, South African coal. Fuel Process Technol 92:2048–2054
Heydari M, Rahman M, Rajender G (2016) Effect of initial coal particle size on coal liquefaction conversion. Int J Oil Gas Coal Technol 12(1):63–80
Hiremath N, Mays J, Bhat G (2017) Recent Developments in carbon fibers and carbon nanotube-based fibers: a review. Polym Rev 57:339–368
Hirsch PB (1954) X-ray scattering from coals. Proc R Soc 226(1165):143–169
Holuszko ME, Mastalerz MD (2015) Coal maceral chemistry and its implications for selectivity in coal flotability. Int J Coal Prep Utlil 35:99–110
Höök M, Aleklett K (2009) A review on coal-to-liquid fuels and its coal consumption. Int J Energy Res 34(10):848–864
Hu G, Dam-Johansen K, Wedel S, Hansen JP (2006) Decomposition and oxidation of pyrite. Prog Energy Combust Sci 32(3):295–314
Huang X (2009) Fabrication and properties of carbon fibers: review. Materials 2:2369–2403
Huggins FE, Huffman GP (1995) Chlorine in coal: an XAFS spectroscopic investigation. Fuel 74:556–569
Hunt JM (1991) Generation of gas and oil from coal and other terrestrial organic matter. Org Geochem 17(6):673–680
Hurt R, Sun J-K, Lunden M (1998) A kinetic model of carbon burnout in pulverized coal combustion. Combust Flame 113(1–2):181–197
Huson MG (2017) High performance pitch based carbon fibers. In: Bhat G (ed) Structure and properties of high performance fibers. Woodhead Publishing, Victoria, pp 31–78. https://doi.org/10.1016/B978-0-08-100550-7.00003-6
Irfan MF, Usman MR, Kusakabe K (2011) Coal gasification in CO2 atmosphere and its kinetics since 1948: a brief review. Energy 36:12–40
Jasienko S, Kimber GM, Patrick JW (1981) Coal as raw material for carbon production: some new aspects [and discussion]. Philos Trans R Soc Lond Ser A Math Phys Sci 300(1453):171–182
Jayaraman K, Gokalp I (2015) Effect of char generation method on steam, CO2 and blended mixture gasification of high ash Turkish coals. Fuel 153:320–327
Jee K (2012) The effect of coal properties on carbonization behaviour and strength of coke blends. Dissertation. University of New South Wales
Jones RB, Mccourt CB, Morley C, King K (1985) Maceral and rank influences on the morphology of coal char. Fuel 64:1460–1467
Kaji R, Muranaka Y, Otsuka K, Hishinuma Y (1986) Water absorption by coals: effects of pore structure and surface oxygen. Fuel 65(2):288–291
Kajitani S, Suzuki N, Ashizawa M, Hara S (2006) CO2 gasification rate analysis of coal char in entrained flow coal gasifier. Fuel 85:163–169
Kalkreuth W, Roy C, Hebert M (1986) Vaccume pyrolysis of Canadian PrinceMine coal—chemical and petrological analyses of feed coal and solid residues. Fuel 64:213–222
Khan MR (2011) Advances in clean hydrocarbon fuel processing. Sci Technol 19:14–27
Kim BJ, Kil H, Watanabe N, Seo MH, Kim BH, Yang KS, Kato O, Miyawaki J, Mochida I, Yoon SH (2013) Preparation of novel isotropic pitch with high softening point and solvent solubility for pitch-based electrospun carbon nanofiber. Curr Org Chem 17(13):1463–1468
Kim BJ, Eom Y, Kato O, Miyawaki J, Kim BC, Mochida I, Yoon SH (2014) Preparation of carbon fibers with excellent mechanical properties from isotropic pitches. Carbon 77:747–755
Kim B, Kotegawa T, Eom Y, An J, Hong I, Kato O, Nakabayashi K, Miyawaki J, Kim BC, Mochida I, Yoon S (2016a) Enhancing the tensile strength of isotropic pitch-based carbon fibers by improving the stabilization and carbonization properties of precursor pitch. Carbon 99:649–657
Kim J, Im U, Lee B, Peck D, Yoon S, Jung D (2016b) Pitch-based carbon fibers from coal tar or petroleum residue under the same processing condition. Carbon Lett 19:72–78
Kissinger HE (1957) Reaction kinetics in differential thermal analysis. Anal Chem 29(11):1702–1706
Kosminski A, Ross D, Agnew J (2006a) Influence of gas environment on reactions between sodium and silicon minerals during gasification of low-rank coal. Fuel Process Technol 87:953–962
Kosminski A, Ross D, Agnew J (2006b) Reactions between sodium and silica during gasification of a low-rank coal. Fuel Process Technol 87:1037–1049
Kosminski A, Ross D, Agnew J (2006c) Transformations of sodium during gasification of low-rank coal. Fuel Process Technol 87:943–952
Küçükbayrak S, Haykiri-Açma H, Ersoy-Meriçboyu A, Yaman S (2001) Effect of lignite properties on reactivity of lignite. Energy Convers Manag 42:613–626
Lebedev M, Wilson ME, Mikhaltsevitch V (2014) An experimental study of solid matrix weakening in water-saturated Savonnières limestone. Geophys Prospect 62(6):1253–1265
Lee SH, Lee SM, Im U, Kim S, Yoon S, Lee B, Peck D, Shul Y, Jung D (2019) Preparation and characterization of high-spinnability isotropic pitch from 1-methylnaphthalene-extracted low-rank. Carbon 155:186–194. https://doi.org/10.1016/j.carbon.2019.08.061
Li S (2010) Char–slag transition during pulverized coal gasification. Dissertation. University of Utah
Li X, Hu H, Jin L, Hu S, Wu B (2008a) Approach for promoting liquid yield in direct liquefaction of Shenhua coal. Fuel Process Technol 89(11):1090–1095
Li X, Hu H, Zhu S, Hu S, Wu B, Meng M (2008b) Kinetics of coal liquefaction during heating-up and isothermal stages. Fuel 87(4–5):508–513
Li K, Khanna R, Zhang J, Barati M, Xu T, Yang T, Sahajwalla V (2015) Comprehensive investigation of various structural features of bituminous coals using advanced analytical techniques. Energy Fuels 11(29):7178–7189. https://doi.org/10.1021/acs.energyfuels.5b02064
Liu B, He Q, Jiang Z, Xu R, Hu B (2013) Relationship between coal ash composition and ash fusion temperatures. Fuel 105:293–300
Lolja SA, Haxhi H, Dhimitri R, Drushku S, Malja A (2002) Correlation between ash fusion temperatures and chemical composition in Albanian coal ashes. Fuel 81:2257–2261
Ma Z, Iman F, Lu P, Sears R, Kong L, Rokanuzzaman AS, McCollar DP, Benson SA (2007) A comprehensive slagging and fouling prediction tool for coal-fired boilers and its validation/application. Fuel Process Technol 88:1035–1043
Mahagaokar U (2004) Coal conversion processes, gasification. In: Kirk–Othmer encyclopedia of chemical technology. https://doi.org/10.1002/0471238961.0701190913010801.a01.pub2
Maledi NB, Oladijo OP, Botef I, Ntsoane TP, Madiseng A, Moloisane L (2017) Influence of cold spray parameters on the microstructures and residual stress of Zn coatings sprayed on mild steel. Surf Coat Technol 318:106–113. https://doi.org/10.1016/j.surfcoat.2017.03.062
Manoj B (2014) Investigation of nanocrystalline structure in selected carbonaceous materials. Int J Miner Metall Mater 21(9):940–946
Manoj B (2016) A comprehensive analysis of various structural parameters of Indian coals with the aid of advanced analytical tools. Int J Coal Sci Technol 3(2):123–132. https://doi.org/10.1007/s40789-016-0134-1
Manoj B, Kunjomana A (2012) Study of stacking structure of amorphous carbon by Xray diffraction technique. Int J Electrochem Sci 7:3127–3134
Marzec A (2002) Towards an understanding of the coal structure: a review. Fuel Process Technol 77:25–32
Matjie RH, French D, Ward CR, Pistorius PC, Li Z (2011) Behaviour of coal mineral matter in sintering and slagging of ash during the gasification process. Fuel Process Technol 92(8):1426–1433
McMillen DF, Malhotra R (2006) Hydrogen transfer in the formation and destruction of retrograde products in coal conversion. J Phys Chem A 110(21):6757–6770
Megaritis A, Messenböck RC, Chatzakis IN, Dugwell DR, Kandiyoti R (1999) High-pressure pyrolysis and CO2-gasification of coal maceral concentrates: conversions and char combustion reactivities. Fuel 78:871–882
Méndez LB, Borrego AG, Martinez-Tarazona MR, Menéndez R (2003) Influence of petrographic and mineral matter composition of coal particles on their combustion reactivity. Fuel 82:1875–1882
Miller BG (2011) The chemical and physical characteristics of coal. In: Clean coal engineering technology, 2nd edn, pp 53–68. https://doi.org/10.1016/B978-1-85617-710-8.00002-9
Miller BG (2017) Introduction to coal utilization technologies. In: Clean coal engineering technology, 3rd edn, pp 147–229. https://doi.org/10.1016/b978-0-12-811365-3.00004-1
Mishra A, Saha M, Bhatia G, Aggarwal RK, Raman V, Yadav HS (2005) A comparative study on the development of pitch precursor for general purpose carbon fibers. J Mater Process Technol 168(2):316–320
Mishra A, Gautam S, Sharma T (2018) Effect of operating parameters on coal gasification. Int J Coal Sci Technol 5(2):113–125
Mitchell G (1997) Basics of coal and coal characteristics. Britannica, Warrendale
Miura K, Hashimoto K, Silveston PL (1989) Factors affecting the reactivity of coal chars during gasification, and indices representing reactvity. Fuel 68:1461–1475
Nas C (1994) Spatial variations in the thickness and quality coalof the Sangatta seam, Kutei Basin Kalimantan, Indonesia. Dissertation. University of Wollogong. http://ro.uow.edu.au/thesis/1409
Neavel RC, Knights CF, Schulz H (1981) Exxon donor solvent liquefaction process [and discussion]. Philos Trans R Soc Lond Ser A Math Phys Sci 300(1453):141–156
Newman J, Price LC, Johnston JH (1997) Hydrocarbon source potential and maturation in Eocene New Zealand vitrinite-rich coals. J Pet Geol 20(2):137–163. https://doi.org/10.1111/j.1747-5457.1997.tb00770.x
Ng SH, Fung DP, Kim SD (1988) Study of the pore structure and reactivity of Canadian coal-derived chars. Fuel 67:700–706
Nomura S, Arima T, Kato K (2004) Coal blending theory for dry coal charging process. Fuel 83:1771–1776
Odeh AO (2015) IR qualitative and quantitative ATR-FTIR analysis and its application to coal char of different ranks. J Fuel Chem Technol 43(2):129–137
Okuyama N, Komatsu N, Shigehisa T, Kaneko T, Tsuruya S (2004) Hyper-coal process to produce the ash-free coal. Fuel Process Technol 85:947–967
Orem WH, Finkelman RB (2003) Coal formation and geochemistry. US Geological Survey, US, Orem. https://doi.org/10.1016/B0-08-043751-6/07097-3
Oshnock TW (1995) Pulverised coal injection for blast furnace operation—part V: effect of coal properties on blast furnace performance. Iron Steel Maker 22:41–42
Ozer M, Basha OM, Stiegel G, Morsi B (2017) Effect of coal nature on the gasification process. In: Integrated gasification combined cycle (IGCC) technologies, pp 257–293. https://doi.org/10.1016/B978-0-08-100167-7.00007-X
Pan J, Meng Z, Hou Q, Ju Y, Cao Y (2013) Coal strength and Young’s modulus related to coal rank, compressional velocity and maceral composition. J Struct Geol 54:129–135. https://doi.org/10.1016/j.jsg.2013.07.008
Park YD, Mochida I (1989) A two-stage preparation of mesophase pitch from the vacuum residue of FCC decant oil. Carbon 27(6):925–929. https://doi.org/10.1016/0008-6223(89)90043-2
Patrick JW (1974) The coking of coal. Sci Prog 61(243):375–399
Peters KE, Snedden JW, Sulaeman A, Sarg JF, Enrico RJ (2000) A new geochemical-sequence stratigraphic model for the Mahakam Delta and Makassar Slope, Kalimantan, Indonesia. AAPG Bull 84:12–44
Powell TG, Boreham CJ (1994) Terrestrial sourced oils: Where do they exist and what are our limits of knowledge?—A geochemical perspective. In: Fleet AJ, Scott AC (eds) Coal and coal bearing strata as oil-prone source rocks. Geological Society of London, London, pp 11–29
Quann RJ, Sarofim AF (1986) A scanning electron microscopy study of the transformations of organically bound metals during lignite combustion. Fuel 65:40–46
Radovic LR, Walker PL, Jenkins RG (1983) Importance of catalyst dispersion in the gasification of lignite chars. J Catal 82(2):382–394
Rahman M, Samanta A, Gupta R (2013) Production and characterization of ash-free coal from low-rank Canadian coal by solvent extraction. Fuel Process Technol 115:88–98
Randolph J (1944) Gasoline from coal. Sci Am 170(2):73–86
Saxby JD, Shibaoka M (1986) Coal and coal macerals as source rocks for oil and gas. Appl Geochem 1:25–36. https://doi.org/10.1016/0883-2927(86)90035-1
Schlosberg RH (1985) Chemistry of coal conversion. Springer, New York
Shadle LJ, Berry DA, Syamlal M (2002) Coal conversion processes, gasification. In: Kirk–Othmer encyclopedia of chemical technology. https://doi.org/10.1002/0471238961.0701190913010801.a01.pub2
Sharma A, Kyotani T, Tomita A (2000) Direct observation of raw coals in lattice fringe mode using high-resolution transmission electron microscopy. Energy Fuels 14:1219–1225
Shi L, Liu Q, Guo X, Wu W, Liu Z (2013) Pyrolysis behavior and bonding information of coal—a TGA study. Fuel Process Technol 108:125–132. https://doi.org/10.1016/j.fuproc.2012.06.023
Singh AK, Mrityunjay KJ (2018a) Hydrocarbon potential of permian coals of south Karanpura coalfield, Jharkhand, India. Energy Sources Part A Recover Util Environ Effects 40(2):163–171
Singh AK, Mrityunjay KJ (2018b) Interrelation between mechanical and petrographic characteristics of coals of Argada B seam: implication to comminution and utilization. Int J Coal Prep Util. https://doi.org/10.1080/19392699.2017.1419207
Singh PK, Singh MP, Singh AK, Arora M, Naik AS (2013) The prediction of the liquefaction behavior of the East Kalimantan coals of Indonesia: an appraisal through petrography of selected coal samples. Energy Sources Part A Recover Util Environ Effects 35(18):1728–1740
Singh AK, Singh PK, Singh MP, Banerjee PK (2015) Utilization of the Permian coal deposits of West Bokaro, India: a petrochemical evaluation. Energy Sources Part A Recover Util Environ Effects 37:1081–1088
Skodras G, Sakellaropoulos G (2002) Mineral matter effects in lignite gasification. Fuel Process Technol 77:151–158
Slaghuis JH, van der Halt TJ, Glasser D, Hildebrandt D (1993) Annomalies in gassification kinetic behaviour of some South African coal chars. In: International Pittsburgh coal conference, Pittsburgh, PA
Speight JG (2014) Gasification of unconventional feedstocks. https://doi.org/10.1016/C2013-0-14152-9
Speight JG (2015) Proximate analysis. In: Speight JG (ed) Handbook of coal analysis, 2nd edn. Wiley, New York, pp 1–28. https://doi.org/10.1002/9781119037699.ch5
Sprunk GC (1942) Influence of physical constitution of coal upon its chemical, hydrogenation and carbonization properties. J Geol 50(4):411–436
Suarez-Ruiz I. (2012) Organic petrology: an overview. In: Al-Juboury A (ed) Petrology—new perspectives and applications, pp 199–224. https://doi.org/10.5772/23431
Suarez-Ruiz I, Crelling JC (2008) The role of petrology in coal utilization. In: Applied coal petrology, 1st edn. Academic Press, New York. https://doi.org/10.1016/B978-0-08-045051-3-X0001-2
Sun Q, Li W, Chen H, Li B (2003) The variation of structural characteristics of macerals during pyrolysis. Fuel 82:669–676
Sun Q, Li W, Chen H, Li B (2004) The CO2-gasification and kinetics of Shenmu maceral chars with and without catalyst. Fuel 83:1787–1793
Susilawati R, Ward CR (2006) Metamorphism of mineral matter in coal from the Bukit Asam deposit, south Sumatra, Indonesia. Int J Coal Geol 68(3–4):171–195
Sykes R (2001) Depositional and rank controls on the petroleum potential of coaly source rocks. In: Eastern Australasian basins symposium, a refocused energy perspective for the future, Australia, pp 591–601
Takanohashi T, Shishido T, Kawashima H, Saito I (2008) Characterisation of hyper coals from coals of various ranks. Fuel 87(4–5):592–598
Takarada T, Tamai Y, Tomita A (1985) Reactivities of 34 coals under steam gasification. Fuel 64:1438–1442
Taylor GH, Teichmuller M, Davis A, Diessel CFK, Littke R, Robert P (1998) Organic petrology, 3rd edn. Schweizerbart, Stuttgart
Tissot BP, Welte D (1984) Petroleum formation and occurrence, 2nd edn. Springer, Berlin
Trapp B, Moock N, Denton D (2004) Coal gasification: ready for prime time. Power 148:42–50
Vamvuka D, Zografos D, Alevizos G (2008) Control methods for mitigating biomass ash-related problems in fluidized beds. Bioresour Technol 99(9):3534–3544
van Dyk J, Waanders F (2007) Manipulation of gasification coal feed in order to increase the ash fusion temperature of the coal enabling the gasifiers to operate at higher temperatures. Fuel 86:2728–2735
van Krevelen VW (1993) Typology, physics, chemistry, constitution, 3rd edn. Elsevier Science Publishers, Amsterdam
van Niekerk D, Pugmire RJ, Solum MS, Painter PC, Mathews JP (2008) Structural characterization of vitrinite-rich and inertinite-rich Permian-aged South African bituminous coals. Int J Coal Geol 76:290–300
Vasireddy SB, Morreale A, Cugini C, Song SJ (2011) Clean liquid fuels from direct coal liquefaction: chemistry, catalysis, technological status and challenges. Energy Environ Sci 2:311–345
Wang P, Massoudi M (2013) Slag behavior in gasifiers. Part I: influence of coal properties and gasification conditions. Energies 6:784–806
Wang C, Li M, Wu Y, Guo C (1998) Preparation and microstructure of hollow mesophase pitch-based carbon fibers. Carbon 36:1749–1754
Ward CR (2002) Analysis and significance of mineral matter in coal seams. Int J Coal Geol 50(1–4):135–168
Ward CR (2016) Analysis, origin and significance of mineral matter in coal: an updated review. Int J Coal Geol 165:1–27
Warwick PD (2002) Coal geology and resources. In: Geology. www.eolss.net/sample-chapters/c01/e6-15-08-02
Watanabe F, Ishida S, Korai Y, Mochida I, Kato I, Sakai Y, Kamatsu M (1999) Pitch-based carbon fiber of high compressive strength prepared from synthetic isotropic pitch containing mesophase spheres. Carbon 37:961–967
Wee HL, Wu H, Zhang D, French D (2005) The effect of combustion on mineral matter transformation and ash deposition in a utility boiler fired with a sub-bituminous coal. Proc Combust Inst 30:2981–2989
Wigley F, Williamson J, Gibb WH (1997) The distribution of mineral matter in pulverised coal particles in relation to burnout behaviour. Fuel 76:1283–1288
Wu Y, Wu S, Gu J, Gao J (2009) Differences in physical properties and CO2 gasification reactivity between coal char and petroleum coke. Process Saf Environ Prot 87(5):323–330
Xue Y, Ranjith PG, Gao F, Zhang D, Cheng H, Chong Z, Hou P (2017) Mechanical behaviour and permeability evolution of gas-containing coal from unloading confining pressure tests. J Nat Gas Sci Eng 40:336–346. https://doi.org/10.1016/j.jngse.2017.02.030
Yamagata C, Kajiwar Y, Suyama S, Sato K, Komatsu S (1990) Simultaneous injection of pulverized coal and dolomite into blast furnace tuyeres. ISIJ Int 30:362–371
Yang SJ, Nakabayashi K, Miyawaki J, Yoon SH (2016) Preparation of pitch based carbon fibers using hyper-coal as a raw material. Carbon 106:28–36
Yang J, Shi K, Li X, Yoon SH (2018) Preparation of isotropic spinnable pitch and carbon fiber from biomass tar through the co-carbonization with ethylene bottom oil. Carbon Lett 25:89–94
Ye DP, Agnew JB, Zhang DK (1998) Gasification of a South Australian low-rank coal with carbon dioxide and steam: kinetics and reactivity studies. Fuel 77:1209–1219
Yoshida T, Nakata Y, Yoshida R, Ueda S, Kanda N, Maekawa Y (1982) Elucidation of structural and hydroliquefaction characteristics of yallourn brown coal by C-13 Cp MAS Nmr spectrometry. Fuel 61(9):824–830
Yu J, Tahmasebi A, Han Y, Yin F, Li X (2013) A review on water in low rank coals: the existence, interaction with coal structure and effects on coal utilization. Fuel Process Technol 106:9–20. https://doi.org/10.1016/j.fuproc.2012.09.051
Zabihi O (2012) Modeling of phenomenological mechanisms during thermal formation and degradation of an epoxy-based nanocomposite. Thermochim Acta 543:239–245
Zabihi O, Shafel S, Fakhrhoseini SM, Ahmadi M, Nazarloo HA, Stanger R, Tran QA, Lucas J, Wall T, Naebe M (2019) Low-cost carbon fibre derived from sustainable coal tar pitch and polyacrylonitrile: fabrication and characterisation. Materials 12:1–14. https://doi.org/10.3390/ma12081281
Zevenhoven-Onderwater M, Backman R, Skrifvars BJ, Hupa M (2001) The ash chemistry in fluidised bed gasification of biomass fuels. Part I: predicting the chemistry of melting ashes and ash–bed material interaction. Fuel 80:1489–1502
Zhang SY, Lu JF, Zhang JS, Yue GX (2008) Effect of pyrolysis intensity on the reactivity of coal char. Energy Fuels 22:3213–3221
Zhang ZWX, Zhou T, Chen Y, Hou N, Piao G, Kobayashi N, Itaya Y, Mori S (2011) The effect of iron-bearing mineral melting behaviour on ash deposition during coal combustion. Proc Combust Inst 33(2):2853–2861
Zhao Y, Hu H, Jin L, He X, Wu B (2011) Pyrolysis behaviour of vitrinite and inertinite from Chinese Pingshuo coal by TG-MS and in a fixed bed reactor. Fuel Process Technol 92:780–786
Zhou B, Shi L, Liu Q, Liu Z (2016) Examination of structural models and bonding characteristics of coals. Fuel 184:799–807. https://doi.org/10.1016/j.fuel.2016.07.081
Zhu J, Liu J, Yang Y, Cheng J, Zhou J, Cen K (2016) Fractal characteristics of pore structures in 13 coal specimens: relationship among fractal dimension, pore structure parameter and slurry ability of coal. Fuel Process Technol 149:256–267
Zhuo Y, Messenbock R, Peterson N, Dugwell DR, Kandiyoti R (2000) High pressure gasification of coal in bench scale reactors; the effect pressure, gassifing medium and maceral content. In: International furnace and boilers, 5th European conference. INFUB, Rio Tonto, Portugal, 629–638
Acknowledgements
I would like to express my heart felt gratitude to the management, academic and support staff of Botswana International University of Science and Technology for providing access to the resources of the institution during the study carried out for this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Keboletse, K.P., Ntuli, F. & Oladijo, O.P. Influence of coal properties on coal conversion processes-coal carbonization, carbon fiber production, gasification and liquefaction technologies: a review. Int J Coal Sci Technol 8, 817–843 (2021). https://doi.org/10.1007/s40789-020-00401-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40789-020-00401-5