Abstract
Background
Psoriasis imposes a substantial burden on patients’ social, emotional, physical, and family life. Although psoriasis has no complete cure, various treatments are available to control its symptoms and improve a patients’ quality of life.
Objective
We aimed to compare the effectiveness of biologic versus non-biologic treatments on health-related quality of life among patients with psoriasis in Malaysia.
Methods
This retrospective cross-sectional study evaluated data of adult patients diagnosed with psoriasis during 2007–18 from the Malaysian Psoriasis Registry. Baseline demographics, disease, and treatment characteristics were described. For a subset of patients treated with biologics and non-biologics who had baseline and 6-month follow-up data available, changes in the mean Dermatology Life Quality Index scores and the proportion of patients with a clinically relevant improvement (≥ 4 points) post-treatment were assessed.
Results
Overall, 15,238 adult patients with psoriasis from the Malaysian Psoriasis Registry were included in the analysis. Patients receiving biologics showed a statistically significant reduction in the mean Dermatology Life Quality Index scores after 6 months compared with those receiving non-biologic treatment (− 5.7 vs − 0.8%; p < 0.001). The proportion of patients who achieved a ≥ 4-point improvement in Dermatology Life Quality Index scores was approximately two times greater in the biologic-treated group versus the non-biologic-treated group (56.4 vs 27.7%).
Conclusions
Biologic treatment showed a greater reduction in the Dermatology Life Quality Index scores of patients with psoriasis versus non-biologic treatment. These results highlight the importance of early treatment with more efficacious treatment options, such as biologic therapies, to improve the overall health-related quality of life of patients with psoriasis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Psoriasis imposes a substantial burden on patients’ social, emotional, physical, and family life. Various treatments are available to control its symptoms and improve patients’ quality of life. |
Data from the Malaysian Psoriasis Registry were reviewed to assess the effect of biologic and non-biologic treatments on the health-related quality of life of patients with psoriasis. |
Treatment with biologics showed a greater reduction in the Dermatology Life Quality Index scores of patients with psoriasis, indicating better health-related quality of life compared with non-biologic treatment. |
1 Introduction
Psoriasis is more prevalent in northern Europe than in other geographic regions [1, 2]. In the USA, the prevalence of psoriasis in adults ranges from 0.5% [3] to 3.1% [4], whereas in Europe, it ranges from 1.3% [5] to 11.4% [1]. In Asia, the prevalence ranges from 0.1% [6] to 1.5% [7]. Although there is a lack of population-based epidemiological studies on psoriasis in the Malaysian population, a hospital-based study reported that psoriasis is common in Malaysia, accounting for approximately 9.5% of the dermatological conditions [8].
In both clinical practice and clinical trials, measurements such as body surface area (BSA), Physician’s Global Assessment, and the Psoriasis Area and Severity Index (PASI) are used to guide the categorization of psoriasis severity as mild, moderate, and severe [9]. To account for patients with limited cutaneous disease but substantial physical, psychosocial, and functional impairments owing to the involvement of special areas (face, palms, soles, genitalia, and scalp), the International Psoriasis Council redefined psoriasis severity classification to only two categories (candidates requiring topical therapy or those requiring systemic therapy) [10].
Patients with psoriasis experience a substantial emotional, social, physical, and family burden on their lives. In addition to physical discomfort due to the condition, patients also report sleep disturbances, stigmatization, low self-esteem, suicidal thoughts, and negative feelings of shame, embarrassment, worry, and anger [11,12,13,14]. Consequently, these patients experience a considerable psychological burden that profoundly influences their personal and social life [15]. Furthermore, the physical and emotional consequences associated with psoriasis frequently interfere with workplace performance, in addition to causing absenteeism [16, 17]. Psoriasis is also associated with a significant burden of comorbid conditions such as cardiometabolic comorbidities (hyperlipidemia, hypertension, type 2 diabetes mellitus, and obesity), cardiovascular diseases (stroke, myocardial infarction, and death), depression, and inflammatory bowel diseases [18,19,20,21,22,23]. These comorbidities are associated with significant impairments in patients’ health-related quality of life (HRQoL) [24, 25].
Although there is no complete cure for psoriasis, various treatments are available to control its symptoms and improve patients’ HRQoL. The Dermatology Life Quality Index (DLQI) is a validated tool to measure the impact of psoriasis on patients’ HRQoL [26, 27]. Furthermore, changes in the DLQI scores correlate well with variations in patients’ clinical status measured using the PASI scores [28]. In clinical practice, DLQI measurement was also found to be useful for assessing patients’ concerns that cannot be identified by PASI alone while initiating biologics or switching from other biologics [29].
Therefore, this study aimed to compare the effectiveness of biologic and non-biologic treatments on HRQoL assessed using the DLQI among patients with psoriasis in Malaysia.
2 Methods
2.1 Study Design
This was a retrospective cross-sectional study that evaluated data from the Malaysian Psoriasis Registry (MPR).
2.2 Data Source
The MPR is a prospective ongoing database that collects data on patients with psoriasis from 32 government-funded public hospitals and two private hospitals in Malaysia. The registry has data on patients’ demographic characteristics, such as age, sex, and ethnicity; clinical characteristics, such as the type and clinical presentation of psoriasis; comorbidities; disease severity (assessed by BSA); HRQoL (assessed by the DLQI); and type of treatment (biologic vs non-biologic treatment).
2.3 Patient Population
Adult patients (aged ≥ 18 years) diagnosed with psoriasis by a registered dermatologist or a medical practitioner (under the supervision of a dermatologist) during 2007–18 were included in the study.
2.4 Outcomes
The baseline demographic, clinical, and treatment characteristics were described by severity levels (BSA < 5%; BSA 5–10%; BSA > 10–90%; and BSA > 90%). For a subset of patients who had baseline and 6-month follow-up data available, the following outcomes were assessed: change in the mean DLQI scores, proportion of patients reporting a negative impact on DLQI domains, and proportion of patients with a clinically relevant improvement in DLQI scores (defined as minimal clinically important difference of 4 points in the DLQI score) [30]. These outcomes were compared between patients treated with biologic and non-biologic treatments.
2.5 Analysis
Descriptive statistics were prepared, with results presented as frequencies and percentages for the categorical variables (sex, ethnicity, sites affected, comorbidities, and treatment history) and as mean and standard deviation for the continuous variables (age, psoriasis duration, and DLQI scores). The difference in the mean DLQI scores of patients treated with biologic versus non-biologic therapy was tested using the one-tailed Welch’s t test. A p value of < 0.05 was considered to indicate statistical significance. Statistical analyses were performed using SPSS version 21.0 (IBM Corporation, Armonk, NY, USA) and R software, version 4.1.3.
This study was approved by the Malaysian Research Ethics Committee (NMRR-20-1170-54885) and was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. All subjects provided informed consent to participate in the study. No identifying information is included in this article.
3 Results
Table 1 presents the baseline characteristics of the patients identified from the MPR. A total of 15,238 adult patients with psoriasis with complete BSA involvement data from the MPR were included in this analysis. The mean age of the patients was 44.7 years, with a male-to-female ratio of 1:0.8 and an average disease duration of 9.3 years (Table 1). Plaque psoriasis was the most common phenotype (92.7%), followed by guttate (3.1%), erythrodermic (2.6%), and others (1.7%).
The proportion of patients with face, scalp, and nail involvement appeared to increase in tandem with a higher disease severity based on BSA. Obesity (52.4%) was the most frequent comorbidity observed in these patients followed by hypertension (25.3%), hyperlipidemia (17.5%), diabetes (16.9%), and psoriatic arthritis (14.5%). The mean DLQI score for all patients was 9.5, which increased with disease severity. Most patients were being treated with topical agents, either alone or in combination with other therapies (94.0%), whereas a few were receiving phototherapy (2.9%), non-biologic systemic agents (16.2%), and biologics (0.8%; Table 1). Over the last 12 years, the non-biologic systemic agents used included methotrexate, acitretin, corticosteroids, ciclosporin, hydroxyurea, and dapsone, whereas the biologics used were infliximab, etanercept, adalimumab, ustekinumab, certolizumab pegol, efalizumab, and secukinumab.
Overall, 7031 patients with follow-up treatment data at 6 months were included for further analysis. Patients receiving biologic treatment (n = 78) had a mean (standard deviation) baseline DLQI score of 13.9 (7.2), whereas patients receiving non-biologic treatment (n = 6953) had a score of 8.9 (6.7).
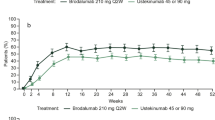
Patients receiving biologic treatment showed a statistically significant reduction in the mean DLQI scores after 6 months compared with those receiving non-biologic treatment (overall patient population: − 5.7% vs − 0.8%; nail [all patients with nail involvement with or without concurrent face/scalp involvement]: − 5.2 vs − 0.8%; scalp [concurrent nail/face or solely scalp involvement]: − 5.5 vs −0.8%; face [concurrent nail/scalp or solely face involvement]: − 5.4 vs − 1.0%; Fig. 1). Similarly, the proportion of patients who achieved a ≥ 4-point improvement in DLQI scores was approximately two times greater in the biologic-treated group compared with the non-biologic-treated group (56.4 vs 27.7%; Fig. 2).
Figure 3 represents the reduction in the proportion of patients reporting a negative impact on various DLQI domains after 6 months of treatment. The proportion of patients reporting a negative impact on various DLQI domains after 6 months of treatment was lower (for all domains except “leisure activities” and “work and school”) for the biologic-treated group compared with the non-biologic-treated group.
4 Discussion
The greater improvement in DLQI scores at 6 months observed in this study for patients treated with biologics compared with those treated with non-biologics corroborated the observations of other studies [31, 32]. More than half of the patients treated with biologics showed a ≥ 4-point reduction in DLQI scores, whereas only approximately one-quarter of the non-biologic-treated patients achieved this magnitude of DLQI score reduction. In a study conducted in Japan, treatment with biologics led to a marked improvement in DLQI scores compared with any other treatment modality [31]. Another Australian study demonstrated similar results, with biologics showing a significant reduction in DLQI scores at 24 weeks of treatment compared with other systemic and topical treatments [32]. It was interesting to note that biologic treatment conferred a higher magnitude of reduction in all domains of DLQI, except for “leisure activities” and “work and school,” compared with non-biologic treatment. This finding may warrant further investigation in our patient population.
In the USA, approximately 37.0% of patients with moderate-to-severe psoriasis received biologics [3]. A survey among the rheumatologists and dermatologists in North America and the European Union 5 countries revealed that 19.6% of patients with moderate-to-severe psoriasis received biologics [33]. Data from the Adelphi 2007 Psoriasis Disease Specific Program demonstrated that the percentages of patients with moderate-to-severe psoriasis receiving biologics were 27.8, 9.7, and 9.3% in Spain, France, and the UK, respectively [34]. Our study revealed a much lower access to biologics (0.8%) among patients with moderate-to-severe psoriasis in Malaysia compared with these countries. One of the reasons for this might be the more stringent criteria set by the Malaysian Ministry of Health clinical practice guidelines, where only patients with a high disease severity (BSA > 30%, PASI or DLQI > 20) would be considered for biologic treatment. High biologic cost and contraindications in patients with psoriasis with tuberculosis could be other contributing factors for a lower access to biologics in Malaysia [35]. By contrast, the British Association of Dermatologists guidelines consider patients with BSA > 10% and DLQI > 10 as candidates for biologic therapy, which results in a lower hurdle to treatment access. Furthermore, in our study, a substantial proportion of patients with moderate-to-severe psoriasis had face (49.0%), scalp (78.5%), or nail (58.9%) involvement, which may further impact their quality of life owing to a high visibility of the lesions. As the conventional systemic therapies are known to be less effective and slower acting compared with biologics, in patients with nail and scalp involvement, timely access to biologics is crucial [36].
In this study population, approximately 18.0% of patients with moderate-to-severe psoriasis (BSA > 10%) had psoriatic arthritis. Psoriatic arthritis imposes a considerable additional burden on the HRQoL and functional abilities of these patients [37, 38]. Biologic treatments are more effective in treating psoriatic arthritis compared with conventional systemic therapies [39,40,41,42] and can slow the disease progression when initiated early in the disease course [40, 43, 44]. Therefore, this subgroup of patients can benefit from early treatment initiation with biologics.
Greater skin clearance in patients with moderate-to-severe psoriasis was associated with increased productivity at the workplace and a reduction in annual indirect costs [45]. With a greater improvement in the PASI score, the mean overall work productivity loss decreased correspondingly. Notably, patients with a less than 50.0% improvement from baseline (PASI < 50) lost 429 work hours/year, whereas patients with a 90.0% or more improvement from baseline (PASI ≥ 90) lost only 93 work hours/year. Similarly, the indirect costs associated with PASI ≥90 ($3125) were 74.0% lower than those associated with PASI < 50 ($11,906) [45]. In addition, improvements in PASI strongly correlated with improvements in HRQoL. For example, the probability of achieving a DLQI score of 0/1 was increased by approximately 50.0% when PASI 75 improved to PASI ≥ 90 [46, 47]. However, a PASI response of ≥ 90 is more likely to be achieved with biologic treatment than with conventional systemic therapies [48]. Therefore, timely treatment escalation to better treatment options, such as biologics, which are the current gold-standard treatment for moderate-to-severe psoriasis, would be beneficial in improving their clinical parameters (reductions in PASI and BSA) and decreasing indirect costs, eventually contributing to the well-being of patients, their families, and society.
The chronic elevation of systemic inflammatory cytokines in patients with psoriasis increases the risk of cardiometabolic comorbidities (diabetes, hyperlipidemia, hypertension, and obesity) [49]. In our study, patients with psoriasis reported a higher prevalence of cardiometabolic comorbidities consistent with the findings from other studies, which implies that the disease manifestations go beyond the skin [50,51,52]. Therefore, early and effective treatment is crucial for better disease control, which can potentially modify the risk of comorbidities, thereby enhancing HRQoL and life expectancy [53,54,55,56,57].
The limitations of this study include the relatively smaller number of patients in the biologic treatment group compared with the other treatments, as well as the unavailability of PASI scores in the registry data, which prevented a better assessment of the disease severity.
5 Conclusions
Patients treated with biologics had greater reductions in the DLQI scores, indicating better HRQoL compared with those treated with non-biologic treatment. These results highlight the importance of early treatment with more efficacious treatment options, such as biologic therapies, to improve the overall HRQoL of patients with psoriasis.
References
Danielsen K, Olsen AO, Wilsgaard T, Furberg AS. Is the prevalence of psoriasis increasing? A 30-year follow-up of a population-based cohort. Br J Dermatol. 2013;168(6):1303–10.
Bø K, Thoresen M, Dalgard F. Smokers report more psoriasis, but not atopic dermatitis or hand eczema: results from a Norwegian population survey among adults. Dermatology. 2008;216(1):40–5.
Takeshita J, Gelfand JM, Li P, et al. Psoriasis in the US Medicare population: prevalence, treatment, and factors associated with biologic use. J Invest Dermatol. 2015;135(12):2955–63.
Helmick CG, Lee-Han H, Hirsch SC, Baird TL, Bartlett CL. Prevalence of psoriasis among adults in the US: 2003–2006 and 2009–2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47(1):37–45.
O’Neill P, Kelly P. Postal questionnaire study of disability in the community associated with psoriasis. BMJ. 1996;313(7062):919–21.
Shao CG, Zhang GW, Wang GC. Distribution of psoriasis in China: a nationwide screening. Proc Chin Acad Med Sci Peking Union Med Coll. 1987;2(2):59–65.
Ding X, Wang T, Shen Y, et al. Prevalence of psoriasis in China: a population-based study in six cities. Eur J Dermatol. 2012;22(5):663–7.
Heah SK, Mohd Noor N, Johar A. Prevalence of skin diseases in dermatology outpatient clinic, Hospital Kuala Lumpur. Malays J Dermatol. 2017;38:19–24.
Langley RG, Ellis CN. Evaluating psoriasis with Psoriasis Area and Severity Index, Psoriasis Global Assessment, and Lattice System Physician’s Global Assessment. J Am Acad Dermatol. 2004;51(4):563–9.
Strober B, Ryan C, van de Kerkhof P, et al. Recategorization of psoriasis severity: Delphi consensus from the International Psoriasis Council. J Am Acad Dermatol. 2020;82(1):117–22.
Pariser D, Schenkel B, Carter C, et al. A multicenter, non-interventional study to evaluate patient-reported experiences of living with psoriasis. J Dermatol Treat. 2016;27(1):19–26.
Møller AH, Erntoft S, Vinding GR, Jemec GB. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167–77.
Yang H-J, Yang K-C. Impact of psoriasis on quality of life in Taiwan. Dermatol Sin. 2015;33(3):146–50.
Bhosle MJ, Kulkarni A, Feldman SR, Balkrishnan R. Quality of life in patients with psoriasis. Health Qual Life Outcomes. 2006;4:35.
Baker CS, Foley PA, Braue A. Psoriasis uncovered: measuring burden of disease impact in a survey of Australians with psoriasis. Australas J Dermatol. 2013;54(Suppl. 1):1–6.
Schaefer CP, Cappelleri JC, Cheng R, et al. Health care resource use, productivity, and costs among patients with moderate to severe plaque psoriasis in the United States. J Am Acad Dermatol. 2015;73(4):585-93.e3.
Ko WC, Tsai TF, Tang CH. Health state utility, willingness to pay, and quality of life among Taiwanese patients with psoriasis. Dermatol Sin. 2016;34(4):185–91.
Shah K, Mellars L, Changolkar A, Feldman SR. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77(2):287-92.e4.
Brazzelli V, Maffioli P, Bolcato V, Ciolfi C, D’Angelo A, Tinelli C, Derosa G. Psoriasis and diabetes, a dangerous association: evaluation of insulin resistance, lipid abnormalities, and cardiovascular risk biomarkers. Front Med (Lausanne). 2021;8: 605691.
Gisondi P, Bellinato F, Girolomoni G, Albanesi C. Pathogenesis of chronic plaque psoriasis and its intersection with cardio-metabolic comorbidities. Front Pharmacol. 2020;11:117.
Boehncke WH. Systemic inflammation and cardiovascular comorbidity in psoriasis patients: causes and consequences. Front Immunol. 2018;9:579.
Bu J, Ding R, Zhou L, Chen X, Shen E. Epidemiology of psoriasis and comorbid diseases: a narrative review. Front Immunol. 2022;13: 880201.
Kang JS, Balogh EA, Feldman SR. Psoriasis comorbidities and clinical implications when using biologics. Dermatol Rev. 2021;2:236–44.
Sanchez-Carazo JL, López-Estebaranz JL, Guisado C. Comorbidities and health-related quality of life in Spanish patients with moderate to severe psoriasis: a cross-sectional study (Arizona study). J Dermatol. 2014;41(8):673–8.
Feldman SR, Gottlieb AB, Bala M, et al. Infliximab improves health-related quality of life in the presence of comorbidities among patients with moderate-to-severe psoriasis. Br J Dermatol. 2008;159(3):704–10.
Badia X, Mascaró JM, Lozano R. Measuring health-related quality of life in patients with mild to moderate eczema and psoriasis: clinical validity, reliability and sensitivity to change of the DLQI. The Cavide Research Group. Br J Dermatol. 1999;141(4):698–702.
Shikiar R, Bresnahan BW, Stone SP, Thompson C, Koo J, Revicki DA. Validity and reliability of patient reported outcomes used in psoriasis: results from two randomized clinical trials. Health Qual Life Outcomes. 2003;1:53.
Mazzotti E, Picardi A, Sampogna F, et al. Sensitivity of the Dermatology Life Quality Index to clinical change in patients with psoriasis. Br J Dermatol. 2003;149(2):318–22.
Imafuku S, Kanai Y, Murotani K, Nomura T, Ito K, Ohata C, Yamazaki F, et al. Utility of the Dermatology Life Quality Index at initiation or switching of biologics in real-life Japanese patients with plaque psoriasis: results from the ProLOGUE study. J Dermatol Sci. 2021;101(3):185–93.
Basra MK, Salek MS, Camilleri L, Sturkey R, Finlay AY. Determining the minimal clinically important difference and responsiveness of the Dermatology Life Quality Index (DLQI): further data. Dermatology. 2015;230(1):27–33.
Takahashi H, Iinuma S, Tsuji H, Honma M, Iizuka H. Biologics are more potent than other treatment modalities for improvement of quality of life in psoriasis patients. J Dermatol. 2014;41(8):686–9.
Norris D, Photiou L, Tacey M, et al. Biologics and dermatology life quality index (DLQI) in the Australasian psoriasis population. J Dermatol Treat. 2017;28(8):731–6.
van de Kerkhof PC, Reich K, Kavanaugh A, et al. Physician perspectives in the management of psoriasis and psoriatic arthritis: results from the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis survey. J Eur Acad Dermatol Venereol. 2015;29(10):2002–10.
Puig L, Fan T, Ding Q, Smith NE. Predictors of biologic treatment of psoriasis: a non-interventional study. Clinicoecon Outcomes Res. 2014;6:93–100.
Malaysia Health Technology Assessment Section (MaHTAS). Clinical practice guidelines: management of psoriasis vulgaris. 2013. Available from: https://www.moh.gov.my/moh/attachments/9004.pdf. Accessed July 2022.
Pasch MC. Nail psoriasis: a review of treatment options. Drugs. 2016;76(6):675–705.
Rosen CF, Mussani F, Chandran V, Eder L, Thavaneswaran A, Gladman DD. Patients with psoriatic arthritis have worse quality of life than those with psoriasis alone. Rheumatology (Oxford). 2012;51(3):571–6.
Uttjek M, Dufåker M, Nygren L, Stenberg B. Determinants of quality of life in a psoriasis population in northern Sweden. Acta Derm Venereol. 2004;84(1):37–43.
Kingsley GH, Scott DL. Assessing the effectiveness of synthetic and biologic disease-modifying antirheumatic drugs in psoriatic arthritis: a systematic review. Psoriasis (Auckl). 2015;5:71–81.
Wu D, Li C, Zhang S, et al. Effect of biologics on radiographic progression of peripheral joint in patients with psoriatic arthritis: meta-analysis. Rheumatology (Oxford). 2020;59(11):3172–80.
Lo Giudice L, Acosta Felquer ML, Galimberti ML, Mazzuoccolo L, Soriano E. SAT0426 Can biologics “prevent” the development of psoriatic arthritis in psoriasis patients? Data from a large university hospital cohort in Argentina. Ann Rheum Dis. 2020;79(Suppl. 1):1167–8.
Lininger EN, Siegel SA, Winthrop KL, Deodhar A, Kiwalkar S, Ortega-Loayza A. 471 Risk of psoriatic arthritis in psoriasis patients on biologics and methotrexate. J Invest Dermatol. 2020;140(7 Suppl.):S63.
McLaughlin M, Ostör A. Early treatment of psoriatic arthritis improves prognosis. Practitioner. 2014;258(1777):21–4 (3).
Kampylafka E, Simon D, d’Oliveira I, et al. Disease interception with interleukin-17 inhibition in high-risk psoriasis patients with subclinical joint inflammation: data from the prospective IVEPSA study. Arthritis Res Ther. 2019;21(1):178.
Feldman SR, Zhao Y, Gilloteau I, et al. Higher psoriasis skin clearance is associated with lower annual indirect costs in the United States: a post hoc analysis from the CLEAR study. J Manag Care Spec Pharm. 2018;24(7):617–22.
Elewski BE, Puig L, Mordin M, et al. Psoriasis patients with Psoriasis Area and Severity Index (PASI) 90 response achieve greater health-related quality-of-life improvements than those with PASI 75–89 response: results from two phase 3 studies of secukinumab. J Dermatol Treat. 2017;28(6):492–9.
Torii H, Sato N, Yoshinari T, Nakagawa H, Japanese Infliximab Study Investigators. Dramatic impact of a Psoriasis Area and Severity Index 90 response on the quality of life in patients with psoriasis: an analysis of Japanese clinical trials of infliximab. J Dermatol. 2012;39(3):253–9.
Sbidian E, Chaimani A, Garcia-Doval I, et al. Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis. Cochrane Database Syst Rev. 2017;12(12):CD011535.
Menter A, Krueger GG, Paek SY, Kivelevitch D, Adamopoulos IE, Langley RG. Interleukin-17 and interleukin-23: a narrative review of mechanisms of action in psoriasis and associated comorbidities. Dermatol Ther (Heidelb). 2021;11(2):385–400.
Langan SM, Seminara NM, Shin DB, et al. Prevalence of metabolic syndrome in patients with psoriasis: a population-based study in the United Kingdom. J Invest Dermatol. 2012;132(3 Pt 1):556–62.
Danielsen K, Wilsgaard T, Olsen AO, Furberg AS. Overweight and weight gain predict psoriasis development in a population-based cohort. Acta Derm Venereol. 2017;97(3):332–9.
Armstrong AW, Harskamp CT, Armstrong EJ. Psoriasis and the risk of diabetes mellitus: a systematic review and meta-analysis. JAMA Dermatol. 2013;149(1):84–91.
Ahlehoff O, Skov L, Gislason G, et al. Cardiovascular disease event rates in patients with severe psoriasis treated with systemic anti-inflammatory drugs: a Danish real-world cohort study. J Intern Med. 2013;273(2):197–204.
Wu JJ, Poon K-YT, Channual JC, Shen AY-J. Association between tumor necrosis factor inhibitor therapy and myocardial infarction risk in patients with psoriasis. Arch Dermatol. 2012;148(11):1244–50.
Prodanovich S, Kirsner RS, Kravetz JD, Ma F, Martinez L, Federman DG. Association of psoriasis with coronary artery, cerebrovascular, and peripheral vascular diseases and mortality. Arch Dermatol. 2009;145(6):700–3.
Prodanovich S, Ma F, Taylor JR, Pezon C, Fasihi T, Kirsner RS. Methotrexate reduces incidence of vascular diseases in veterans with psoriasis or rheumatoid arthritis. J Am Acad Dermatol. 2005;52(2):262–7.
Wu Y, Mills D, Bala M. Psoriasis: cardiovascular risk factors and other disease comorbidities. J Drugs Dermatol. 2008;7(4):373–7.
Acknowledgements
The authors thank the Director General of Health Malaysia for permission to publish this paper. They also thank Amit Pagada and Vidyasagar AEC (Novartis Healthcare, India) for their editorial assistance in preparing the article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The study was supported by Novartis Corporation (Malaysia) with funding provided for article preparation.
Conflict of interest
Shu Kee Eng, John Tiong and Harini Chinthapatla are employees of Novartis. Suganthy Robinson, Tang Min Moon, Tan Wooi Chiang, Teh Yeon Chiat, Latha Selvarajah, Tang Jyh Jong, and Suganthi Thevarajah have no conflicts of interest that are directly relevant to the content of this article.
Ethics approval
This study was approved by the Malaysian Research Ethics Committee (NMRR-20-1170-54885). This study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments.
Consent to participate
All subjects provided informed consent to participate in the study. No identifying information is included in this article.
Consent for publication
Not applicable.
Availability of data and material
The datasets generated and/or analyzed during the current study are not publicly available because of the privacy policies set by the Ministry of Health, Malaysia.
Code availability
Not applicable.
Author contributions
All authors contributed to the study conception, design, data collection, and analysis. Material preparation and writing were performed by SR and HC. All authors were involved in the review and correction of the manuscript. All named authors meet the International Committee of Medical Journal Editors criteria of authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Robinson, S., Moon, T.M., Eng, T.K. et al. Dermatology Life Quality Index in Patients with Psoriasis Treated with Biologic Versus Non-biologic Treatment in Malaysia: A Retrospective Cross-Sectional Study. Drugs - Real World Outcomes 10, 291–298 (2023). https://doi.org/10.1007/s40801-023-00359-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40801-023-00359-1