Abstract
Background and Objective
Dual human epidermal growth factor receptor 2 (HER2) blockade with trastuzumab and pertuzumab combined with taxane-based chemotherapy (Cht) has been the standard first-line treatment for HER2-positive metastatic breast cancer (mBC) for years, due to the impressive results of the CLEOPATRA study. Real-world (RW) studies have become critical for assessing treatment effectiveness and safety in real-life circumstances. The aim of this study was to analyze the treatment outcomes of first-line therapy for HER2-positive mBC in RW clinical practice, specifically focusing on the use of maintenance endocrine therapy (ET) in hormone receptor positive (HR-positive) patients.
Methods
This retrospective analysis included 106 HER2-positive mBC patients treated with trastuzumab and pertuzumab combined with taxane-based Cht from October 2015 to December 2020 at the University Hospital Centre Zagreb.
Results
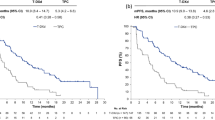
At a median follow-up of 30 months, median progression-free survival (PFS) was 25 months for the total population (95% confidence interval [CI] 16 - not analyzed). Patients with de novo mBC had longer median PFS than patients with recurrent disease (not reached vs. 18 months; hazard ratio 1.99; 95% CI 0.69–3.64, p<0.022). Age, hormone receptor positivity, visceral involvement, number of Cht cycles and previous adjuvant trastuzumab did not impact PFS. Most HR-positive patients (N=55, 88.7%) received maintenance ET after induction Cht.
Conclusion
This retrospective study provides additional data on patient characteristics, treatment and outcomes of RW HER2-positive mBC patients treated with pertuzumab and trastuzumab as first-line therapy. In our institution, maintenance ET after induction Cht has become standard clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Since maintenance ET after induction Cht was not permitted by the CLEOPATRA trial protocol, such an approach is not without controversy and differences in clinical practice can be observed. |
This study provides additional data on treatment outcomes with dual HER2 blockade in RW clinical practice, consistent with previously published RW studies. |
Maintenance ET after taxane-based induction Cht is a valid option for HR-positive patients that should be implemented in clinical practice. |
1 Introduction
Breast cancer (BC) is the most common malignancy in women worldwide. This heterogeneous disease is commonly classified into four major surrogate subtypes: luminal A-like, luminal B-like, human epidermal growth factor receptor 2 (HER2) enriched (non-luminal), and triple-negative subtype [1, 2]. The HER2 is overexpressed or amplified in approximately 15-20% of breast cancers and is considered a poor prognostic factor [3]. The invention of trastuzumab, the first monoclonal anti-HER2 antibody, significantly improved the outcomes of patients with HER2-positive early and metastatic BC (mBC) [4,5,6,7].
Up to 10% of HER2-positive BC patients present with metastatic disease at the time of diagnosis, and approximately 20% of patients will develop distant metastases after the primary treatment of early-stage disease [8,9,10]. Dual HER2 blockade with trastuzumab and pertuzumab combined with chemotherapy (Cht) has become the standard first-line treatment for HER2-positive mBC after the impressive results of the CLEOPATRA trial [11]. In the multicentric phase III CLEOPATRA trial, 808 patients were randomized to receive docetaxel + trastuzumab + pertuzumab or docetaxel + trastuzumab + placebo at a 1:1 ratio. The results showed a meaningful improvement in both progression-free survival (PFS) and overall survival (OS) with the addition of pertuzumab (6.3 months, hazard ratio (HR) 0.69; 95% confidence interval (CI) 0.59–0.81, and 16.3 months, HR 0.68, 95% CI 0.56–0.84; P < 0.001, respectively) [11, 12]. Since the use of maintenance endocrine therapy (ET) after induction Cht in HER2-positive hormone receptor-positive (HR-positive) patients was not permitted by the registrational trial protocol, such an approach is not without controversy. Although guidelines recommend using ET as a maintenance strategy, there is a lack of real-world (RW) data on such a treatment approach and its impact on disease outcomes [13]. Clinical trial populations are specific due to restrictive eligibility criteria and may not faithfully represent patients' characteristics in the RW. Therefore, RW studies are vital to provide evidence of treatment effectiveness and safety in daily clinical practice.
This study aimed to retrospectively analyze the outcomes of patients treated with dual HER2 blockade as first-line therapy for the HER2-positive mBC in RW clinical practice, specifically focusing on the use of maintenance ET in HR-positive patients after discontinuation of induction Cht.
2 Patients and Methods
2.1 Study Population
This was a retrospective, single-institution cohort study conducted at the University Hospital Centre (UHC) Zagreb. Data on 106 patients treated for HER2-positive mBC from October 2015 to December 2020 were collected and analyzed. All patients received dual HER2 blockade (pertuzumab and trastuzumab) combined with induction taxane-based Cht as first-line therapy for HER2-positive mBC.
Patients eligible for this study were 18 years of age and older, with confirmed metastatic and/or locally advanced inoperable HER2-positive BC, who received at least one cycle of dual HER2 blockade with pertuzumab and trastuzumab combined with taxane-based Cht. Data were retrieved from the electronic health record database and organized in an anonymous database. HER2 overexpression was defined as either 3+ by immunohistochemistry (IHC) or 2+ by IHC, and HER2 amplification by FISH according to ASCO/CAP guidelines [14, 15].
Patients received either intravenous (IV) trastuzumab 8 mg/kg loading dose followed by 6 mg/kg maintenance dose every three weeks, or subcutaneous (SC) trastuzumab 600 mg fixed dose, along with IV pertuzumab 840 mg loading dose, followed by 420 mg maintenance dose every 3 weeks. Cht backbone was administered as follows: docetaxel 75 mg/m2 every three weeks, weekly paclitaxel 80 mg/m2, or weekly vinorelbine 60–80 mg/m2.
After the discontinuation of the induction Cht, patients continued with dual HER2 blockade until disease progression or unacceptable toxicity. Most patients with HR-positive tumor also received maintenance ET of the physician's choice. The follow-up and data collection were completed in February 2021.
2.2 Objectives
The primary aim was to determine the outcomes of patients treated with dual HER2 blockade combined with Cht as a first-line therapy for HER2-positive mBC in terms of PFS and OS. The secondary aims were to analyze patient characteristics, RW treatment patterns (particularly focusing on maintenance ET), and cardiac safety profile of treatment with pertuzumab and trastuzumab, estimated by left ventricular ejection fraction (LVEF). PFS was defined as the time from the first dual HER2 blockade administration to disease progression or death from any cause. In patients with measurable disease, treatment efficacy was evaluated by contrast-enhanced CT scan of the thorax, abdomen, and pelvis every three months according to RECIST 1.1 criteria [16]. PET 18FDG/CT or MRI were not used in the evaluation of treatment response. Along with CT scans, bone scans and serum tumour biomarkers were used for response evaluation in cases of unmeasurable disease.
LVEF was evaluated by echocardiogram at baseline and every 3 months during treatment. OS was defined as the time from the first dual HER2 blockade administration to death from any cause.
2.3 Statistical Analysis
Categorical variables were presented as frequency and percentage, while continuous variables were reported as medians with interquartile range (IQR).
The distribution of PFS and OS (quantified using the median) were estimated using the Kaplan-Meier method [17]. The reverse Kaplan-Meier method was used to estimate the median follow-up durations. The Log-rank test and the Cox regression model were used to compare PFS differences in different patient subsets [18]. All the results were obtained and plotted using R v. 3.6.2 [19]. Kaplan Meier analysis was performed using the "survival" and "survminer" packages. Follow-up time distribution based on the Kaplan-Meier method was performed using the “prodlim,”. To summarise the Cox regression model, the package “gtsummary” was used. A threshold of p < 0.05 was considered significant in all analyses.
3 Results
3.1 Patient Characteristics
From January 2015 to December 2020, 109 patients were identified with National Health Insurance fund approval for first-line treatment with dual HER2 blockade with trastuzumab and pertuzumab for HER2-positive mBC. Due to various reasons, three patients did not receive at least one cycle of dual HER2 blockade and were excluded from further analyses (Fig. 1.).
The main patient demographic and clinical characteristics are listed in Table 1. Most patients were female (N=105, 99.05%). The median age at the beginning of therapy was 55.5 years (IQR 46–64.5). Almost half of the patients (N=46, 43.4%) had de novo metastatic disease, and 75 (70.8%) patients had HR-positive disease. At the time of the screening, 67 (63.2%) patients had visceral involvement, and 8 (7.5%) patients had central nervous system (CNS) involvement. Among patients who had recurrent metastatic disease, 29 (48.3%) received trastuzumab in the (neo)adjuvant setting, while none received (neo) adjuvant pertuzumab or adjuvant trastuzumab emtansine. In one-third of patients (N=20, 33.3%), there was a change in HER2 status from negative to positive at the time of disease recurrence.
3.2 Treatment Patterns
Most patients (N=104, 98.1%) received taxane, and only two (1.9%) patients received vinorelbine as Cht backbone. Among the taxane-treated patients, 88 (83.0%) received docetaxel, 8 (7.6%) received paclitaxel, 7 (6.5%) received docetaxel followed by paclitaxel, and one patient received docetaxel followed by vinorelbine. Patients received a median of nine cycles of Cht (IQR 6–11).
Most patients (N=83, 78.3%) received IV trastuzumab, and 23 (21.7%) patients received SC trastuzumab. Most HR-positive patients (N=55, 88.7%) received maintenance ET after induction Cht, mainly non-steroidal aromatase inhibitors (AI) (N=38, 69.1%), but also tamoxifen (N=10, 18.2%), exemestane (N=5, 9.1%) or fulvestrant (N=2, 3.6%). Of those patients who received maintenance ET (N=55), 14 were premenopausal. Among them, 5 received AI + Luteinizing Hormone-Releasing Hormone (LHRH) agonist, 3 AI + surgical castration, 3 tamoxifen + LHRH agonists, 1 patient received tamoxifen + surgical castration, and 2 patients received tamoxifen alone.
3.3 Clinical Outcomes
By data cut-off, 51 (48.1%) patients had disease progression or died. The median follow-up time was 30 months. The median PFS was 25 months for the total population (95% CI 16–not analyzed [NA]), shown in Fig. 2. Patients with de novo mBC had longer median PFS than patients with recurrent disease (not reached [NR] vs. 18 months; HR 1.99; 95% CI 0.69–3.64, p< 0.022), shown in Fig.3. Age, hormone receptor positivity, visceral or CNS involvement, number of Cht cycles, and previous adjuvant trastuzumab, did not impact PFS (Table 2., Supplement Figs. 5–9).
Overall, 27 (25.5%) patients have died at data lock, and the 30-month OS rate was 74.5%. Median OS was not reached (95% CI 48-NR) (Fig.4). Left ventricular systolic dysfunction (LVSD) was reported in 4 (3.7%) patients. Among them, 3 patients had asymptomatic LVSD, and one patient had dyspnoea. Two patients experienced LVEF recovery, and in two patients, LVEF was permanently reduced. None of the patients who had LVSD continued dual HER2 blockade therapy.
4 Discussion
In this retrospective data analysis, RW outcomes of patients receiving first-line dual HER2 blockade for HER2-positive mBC were evaluated regarding patient clinical and demographic characteristics, treatment patterns, and cardiac-related adverse events.
The median PFS was 25 months in the overall study population, which is longer than in the pivotal phase III CLEOPATRA trial (median PFS 18.5 months), although no direct comparison was made [11]. Patients with de novo mBC had significantly longer median PFS than patients with recurrent disease (NR vs. 18 months). LVSD in our RW cohort was slightly lower than in the CLEOPATRA trial (3.7% vs 4.4%) [11]. Demographic and clinical characteristics of our RW cohort were concordant with the CLEOPATRA trial population (median age 55.5 yrs vs. 54 yrs, de novo metastatic disease 43.4% vs. 54.2%). However, unlike patients in the CLEOPATRA trial, most patients in our RW cohort had HR-positive disease (70.8% vs. 47%), and more than twice as many patients had received prior adjuvant trastuzumab (27.4% vs. 12%). The rate of subtype conversion from HER2-negative to HER2-positive after (neo)adjuvant therapy was 33.3% in the recurrent disease group. No such data are available from the CLEOPATRA trial [11]. Contrary to the CLEOPATRA trial, our study included patients with baseline brain metastases (7.5% in RW cohort). ET maintenance therapy combined with dual HER2 blockade after Cht discontinuation in our RW cohort was administered according to guidelines and applied routinely.
The usage of the maintenance ET along with dual HER2 blockade was discussed for the first time at the 4th International Consensus Conference for Advanced Breast Cancer (ABC4) in November 2017. The same ABC4 version adopted the statement that for patients with HR-positive HER2-positive mBC, for whom Cht with dual HER2 was chosen as first-line treatment and proven to be effective, it is reasonable to use ET and dual HER2 blockade as a maintenance strategy after ChT discontinuation [20]. Our institution adopted the combined approach, with 89% of HR-positive HER2-positive patients from this study receiving ET with dual HER2 blockade as maintenance.
The median PFS in our study is consistent with other retrospective studies looking at the outcomes of dual HER2 blockade as first-line therapy for HER2-positive mBC (median PFS 27.5 months in the Italian RW trial (N=155), 28.5 months in Turkish Oncology Group RW trial (N=317), 21.7 months in SONABRE registry subanalysis (N=123), RW trial done by Gamuchi et al. (N=264), and 24 months in the retrospective/prospective GIM/BIOMETA trial (N=251), respectively) [21,22,23,24,25]. Although no direct comparison can be made with the CLEOPATRA reference trial, the use of maintenance ET could be one of the reasons for longer PFS seen in RW studies. This observation is in line with the Turkish RW subanalysis, recently published retrospective study done by Lofe et al. (N=147) and RW trial done by Gamuchi et al. demonstrating significantly better PFS with the addition of maintenance ET to dual HER2 blockade after Cht discontinuation [22, 24, 26].
Although the Turkish RW study and the study done by Lofe et al. showed OS improvement, in a study done by Gamucci et al. however, no significant difference was seen in median OS between those who received maintenance ET and those who did not [24]. Due to the small number of patients who did not receive maintenance ET, it was not possible to make such a comparison in our study.
Indirect evidence suggesting that maintenance ET improves PFS comes from two retrospective studies in which maintenance ET was not used. Both, the Danish and Korean studies had similar median PFS to the CLEOPATRA trial (mPFS 15.8 months and 19.8 months, respectively), which was shorter than the PFS seen in RW studies, in which maintenance ET was used [27, 28]. Interestingly, in a recently published United States (US) RW study (N=615) in which maintenance ET was frequently used (59%), the median PFS was 17 months, and median OS was 58 months, but no subgroup analysis was performed [29].
In terms of other factors affecting PFS, both the Korean and Turkish RW studies showed longer OS in patients who received docetaxel for more than six cycles. Additionally, the study by Gamucci et al. and the Korean RW study showed longer OS in trastuzumab-naïve patients. Furthermore, the Turkish RW study and study by Gamucci et al. demonstrated better PFS and OS in patients without brain metastasis at diagnosis, and the Turkish study also showed better outcomes in patients with single-organ visceral involvement. [22, 24, 28].
Data from several randomized trials in which maintenance ET was used as standard treatment for HER2-positive HR-positive mBC show that combining dual HER2 blockade with ET effectively extends PFS and OS. The PERTAIN study, for example, showed benefit of adding pertuzumab to trastuzumab and ET combination as first-line therapy for HER2-positive HR-positive mBC. Although this was not its primary aim, the study provided clear evidence to support the efficacy of adding ET to dual HER2 blockade [30]. The phase 3 study, PERUSE, which compared the efficacy of different taxanes in combination with pertuzumab and trastuzumab in the first-line treatment of HER2-positive mBC, allowed maintenance ET in HR-positive patients. Although PFS was similar in both HR-positive and HR-negative subgroups, OS was longer in the HR-positive group [31]. In the recently published phase 3 SYSUCC 2 trial, ET plus trastuzumab showed longer PFS to Cht plus trastuzumab in first-line treatment (19.2 months vs 14.8 months, HR 0.88), with less toxicity. This study found non-inferiority of trastuzumab plus ET compared to trastuzumab plus Cth [32]. Given that pertuzumab was not used in the study, dual anti-HER2 therapy in combination with Cht remains the standard first-line treatment. Nevertheless, ET combined with dual anti-HER2 therapy (Cht-free regimen) seems to be a safe and efficient option for frail patients unfit for Cht [33].
It should be noted that in the retrospective studies, there was a higher percentage of HR-positive patients than in the CLEOPATRA trial (70.8% in our RW cohort, 70% in the Italian RW cohort, 68.1% in the Turkish RW cohort, 70.2% in the US RW cohort vs 47% in the Cleopatra cohort, respectively).
HER2-positive HR-positive breast cancer confers low sensitivity to ET due to bidirectional cross-talk between estrogen receptor signaling and HER2 signaling pathways, playing a role in both intrinsic and acquired resistance to ET [34,35,36]. Constitutively activated HER2 signaling augments estrogen receptor function, and HER2-directed therapies lead to modest antitumor activity [37]. There is a strong rationale for targeting both signaling pathways to optimize endocrine therapy and overcome resistance. Both signaling pathways converge to the Cyclin-D1/CDK4/6 complex as a key regulator of the cell cycle progression [38, 39].
The ongoing phase 3, placebo-controlled PATINA study, that aims to evaluate the efficacy and safety of adding palbociclib to the standard dual HER2 blockade and AI combination, will reveal whether the impressive efficacy of CDK 4/6 inhibitors also translates into HER2-positive HR-positive disease [40].
Our study has several limitations typical for retrospective studies, such as a lack of random treatment assignment and standardized disease progression assessment. The main advantage of this study is that it was a single-institution study, which allowed all patients to be treated according to the same clinical practice pattern. Even though this RW data cannot replace the results of randomized clinical trials (RCT), they may provide more information on questions not answered by the RCT.
While we are awaiting the results of the study comparing the effectiveness of the current first-line standard treatment with novel options like trastuzumab deruxtecan, alone or in combination with pertuzumab, we must take advantage of the good tolerability and excellent quality of life offered to patients by dual anti-HER2 maintenance therapy in combination with ET [41, 42].
Optimizing the maintenance treatments to extend the duration of first-line treatment and maximize patient quality of life is crucial [43]. According to the available RW data, the addition of ET maintenance consistently prolongs PFS, and indirect data from the PERUSE study show that it also improves OS. [22, 24, 26, 31].
5 Conclusion
This retrospective study provides additional data on patient characteristics, treatment patterns and outcomes of RW HER2-positive mBC patients treated with dual HER2 blockade with pertuzumab and trastuzumab as first-line therapy for mBC. In most cases, our hospital adopted a strategy of using maintenance ET after taxane-based induction Cht in HER2-positive HR-positive mBC patients.
Data Availability
The data analyzed during this study are available from the corresponding author upon reasonable request.
Code Availability
Not applicable.
Abbreviations
- AI:
-
Aromatase inhibitors
- BC:
-
Breast cancer
- Cht:
-
Chemotherapy
- CI:
-
Confidence interval
- CNS:
-
Central nervous system
- CT:
-
Computed tomography
- ET:
-
Endocrine therapy
- HER2:
-
Human epidermal growth factor receptor 2
- HR:
-
Hazard ratio
- HR-positive:
-
Hormon receptor positive
- IHC:
-
Immunohistochemistry
- IQR:
-
Interquartile range
- IV:
-
Intravenous
- LHRH:
-
Luteinizing hormone-releasing hormone
- LVEF:
-
Left ventricular ejection fraction
- LVSD:
-
Left ventricular systolic dysfunction
- mBC:
-
Metastatic breast cancer
- NA:
-
Not analyzed
- NR:
-
Not reached
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- RCT:
-
Randomized clinical trials
- RW:
-
Real-world
- SC:
-
Subcutaneous
- UHC:
-
University Hospital Centre
- US:
-
United States
References
Toss A, Cristofanilli M. Molecular characterization and targeted therapeutic approaches in breast cancer. Breast Cancer Res. 2015;17:60. https://doi.org/10.1186/s13058-015-0560-9.
Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thürlimann B, Senn JH, et al. Strategies for subtypes-dealing with the diversity of breast cancer: highlights of the St Gallen international expert consensus on the primary therapy of early breast cancer 2011. Ann Oncol. 2011;22(8):1736–47. https://doi.org/10.1093/annonc/mdr304.
Cronin KA, Harlan LC, Dodd KW, Abrams JS, Ballard-Barbash R. Population-based estimate of the prevalence of HER-2 positive breast cancer tumors for early stage patients in the US. Cancer Invest. 2010;28(9):963–8. https://doi.org/10.3109/07357907.2010.496759.
Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353:1673–84. https://doi.org/10.1056/NEJMoa052122.
Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659–72. https://doi.org/10.1056/NEJMoa052306.
Joensuu H, Kellokumpu-Lehtinen PL, Bono P, et al. Adjuvant docetaxel or vinorelbine with or without trastuzumab for breast cancer. N Engl J Med. 2006;354:809–20. https://doi.org/10.1056/NEJMoa053028.
Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344:783–92. https://doi.org/10.1056/NEJM200103153441101.
SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program, National Cancer Institute; 2023 Apr 19. [updated: 2023 Jun 8; cited 2023 Sep 27]. Available from: https://seer.cancer.gov/statistics-network/explorer/. Data source(s): SEER Incidence Data, November 2022 Submission (1975-2020), SEER 22 registries.
Cameron D, Piccart-Gebhart MJ, Gelber RD, Procter M, Goldhirsch A, de Azambuja E, Castro G Jr, Untch M, Smith I, Gianni L, Baselga J, Al-Sakaff N, Lauer S, McFadden E, Leyland-Jones B, Bell R, Dowsett M, Jackisch C; Herceptin Adjuvant (HERA) Trial Study Team. 11 years' follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the HERceptin Adjuvant (HERA) trial. Lancet. 2017 Mar 25;389(10075):1195-1205. https://doi.org/10.1016/S0140-6736(16)32616-2.
Piccart M, Procter M, Fumagalli D, de Azambuja E, Clark E, Ewer MS, Restuccia E, Jerusalem G, Dent S, Reaby L, Bonnefoi H, Krop I, Liu TW, Pieńkowski T, Toi M, Wilcken N, Andersson M, Im YH, Tseng LM, Lueck HJ, Colleoni M, Monturus E, Sicoe M, Guillaume S, Bines J, Gelber RD, Viale G, Thomssen C; APHINITY Steering Committee and Investigators. Adjuvant Pertuzumab and Trastuzumab in Early HER2-Positive Breast Cancer in the APHINITY Trial: 6 Years' Follow-Up. J Clin Oncol. 2021 May 1;39(13):1448-1457. https://doi.org/10.1200/JCO.20.01204.
Baselga J, Cortés J, Kim SB, et al. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med. 2012;366(2):109–19. https://doi.org/10.1056/NEJMoa1113216.
Swain SM, Miles D, Kim SB, Im YH, Im SA, Semiglazov V, Ciruelos E, Schneeweiss A, Loi S, Monturus E, Clark E, Knott A, Restuccia E, Benyunes MC, Cortés J; CLEOPATRA study group. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA): end-of-study results from a double-blind, randomised, placebo-controlled, phase 3 study. Lancet Oncol. 2020 Apr;21(4):519-530. https://doi.org/10.1016/S1470-2045(19)30863-0.
Gennari, A. et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Annals of Oncology, Volume 32, Issue 12, 1475–1495. https://doi.org/10.1016/j.annonc.2021.09.019.
Wolff AC, Hammond ME, Hicks DG, et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. Arch Pathol Lab Med. 2014;138(2):241–56. https://doi.org/10.5858/arpa.2013-0953-SA.
Wolff AC, Hammond MEH, Allison KH, et al. Human epidermal growth factor receptor 2 testing in breast cancer: American society of clinical oncology/college of American pathologists clinical practice guideline focused update. J Clin Oncol. 2018;36(20):2105–22. https://doi.org/10.1200/JCO.2018.77.8738.
Schwartz LH, Litière S, de Vries E, et al. RECIST 1.1-Update and clarification: From the RECIST committee. Eur J Cancer. 2016;62:132-137. https://doi.org/10.1016/j.ejca.2016.03.08117.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assn1958;53:457e81.
Peto R, Peto J. Asymptotically efficient rank invariant test procedures. J Royal Stat Soc A1972;135(2):185e2.
R Core Team. R: a language and environment for statistical computing. Vienna, Austria R Foundation for Statistical Computing; 2017. https://www.R-project.org/.
Cardoso F, Senkus E, Costa A, et al. 4th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 4)†. Ann Oncol. 2018;29(8):1634–57. https://doi.org/10.1093/annonc/mdy192.
De Placido S, Giuliano M, Schettini F, et al. Human epidermal growth factor receptor 2 dual blockade with trastuzumab and pertuzumab in real life: Italian clinical practice versus the CLEOPATRA trial results. Breast. 2018;38:86–91. https://doi.org/10.1016/j.breast.2017.12.012.
Esin E, Oksuzoglu B, Bilici A, et al. Turkish Oncology Group. Pertuzumab, trastuzumab and taxane-based treatment for visceral organ metastatic, trastuzumab-naïve breast cancer: real-life practice outcomes. Cancer Chemother Pharmacol. 2019 Jan;83(1):131-143. https://doi.org/10.1007/s00280-018-3712-7.
Ibragimova KIE, Geurts SME, Meegdes M, et al. Outcomes for the first four lines of therapy in patients with HER2-positive advanced breast cancer: results from the SONABRE registry. Breast Cancer Res Treat. 2023;198(2):239–51. https://doi.org/10.1007/s10549-022-06832-9.
Gamucci T, Pizzuti L, Natoli C, et al. A multicenter REtrospective observational study of first-line treatment with PERtuzumab, trastuzumab and taxanes for advanced HER2 positive breast cancer patients. RePer Study Cancer Biol Ther. 2019;20(2):192–200. https://doi.org/10.1080/15384047.2018.1523095.
Di Maio M, Bighin C, Schettini F, et al. Evolving treatments and outcomes in HER2-positive metastatic breast cancer: data from the GIM14/BIOMETA study. Breast. 2023;72: 103583. https://doi.org/10.1016/j.breast.2023.103583.
Loft M, Lok SW, De Boer R, Malik L, Greenberg S, Yeo B, Anton A, Nottage M, Wong V, Nott L, Collins IM, Torres J, Barnett F, Lombard JM, Gibbs P, Gately L. Addition of endocrine therapy to dual anti-HER2 targeted therapy in initial treatment of HER2 + /HR + metastatic breast cancer. Breast Cancer Res Treat. 2023;198(1):67–74. https://doi.org/10.1007/s10549-022-06856-124.
Christensen T, Berg T, Nielsen LB, Andersson M, Jensen MB, Knoop A. Dual HER2 blockade in the first-line treatment of metastatic breast cancer—a retrospective population-based observational study in Danish patients. Breast. 2020;51:34–9. https://doi.org/10.1016/j.breast.2020.03.002.
Lee YP, Lee MS, Kim H, et al. Real-world evidence of trastuzumab, pertuzumab, and docetaxel combination as a first-line treatment for korean patients with HER2-positive metastatic breast cancer. Cancer Res Treat. 2022. https://doi.org/10.4143/crt.2021.1103.10.4143/crt.2021.1103.
Polito L, Shim J, Hurvitz SA, et al. Real-world first-line use of pertuzumab with different taxanes for human epidermal growth factor receptor 2-positive metastatic breast cancer: a comparative effectiveness study using US electronic health records. JCO Oncol Pract. 2023;19(7):435–45. https://doi.org/10.1200/OP.22.00565.
Rimawi M, Ferrero JM, de la Haba-Rodriguez J, Poole C, De Placido S, Osborne CK, Hegg R, Easton V, Wohlfarth C, Arpino G; PERTAIN Study Group. First-Line Trastuzumab Plus an Aromatase Inhibitor, With or Without Pertuzumab, in Human Epidermal Growth Factor Receptor 2-Positive and Hormone Receptor-Positive Metastatic or Locally Advanced Breast Cancer (PERTAIN): A Randomized, Open-Label Phase II Trial. J Clin Oncol. 2018 Oct 1;36(28):2826-2835. https://doi.org/10.1200/JCO.2017.76.7863
Miles D, Ciruelos E, Schneeweiss A, et al. Final results from the PERUSE study of first-line pertuzumab plus trastuzumab plus a taxane for HER2-positive locally recurrent or metastatic breast cancer, with a multivariable approach to guide prognostication. Ann Oncol. 2021;32(10):1245–55. https://doi.org/10.1016/j.annonc.2021.06.024.
Hua X, Bi XW, Zhao JL, Shi YX, Lin Y, Wu ZY, Zhang YQ, Zhang LH, Zhang AQ, Huang H, Liu XM, Xu F, Guo Y, Xia W, Hong RX, Jiang KK, Xue C, An X, Zhong YY, Wang SS, Huang JJ, Yuan ZY. Trastuzumab plus endocrine therapy or chemotherapy as first-line treatment for patients with hormone receptor-positive and HER2-positive metastatic breast cancer (SYSUCC-002). Clin Cancer Res. 2022;28(4):637–45. https://doi.org/10.1158/1078-0432.CCR-21-3435.
Wang Y, Xu H, Han Y, Wu Y, Sa Q, Wang J. Identifying the optimal therapeutics for patients with hormone receptor-positive, HER2-positive advanced breast cancer: a systematic review and network meta-analysis. ESMO Open. 2023;8(3): 101216. https://doi.org/10.1016/j.esmoop.2023.101216.
Prat A, Baselga J. The role of hormonal therapy in the management of hormonal-receptor-positive breast cancer with co-expression of HER2. Nat Clin Pract Oncol. 2008;5:531–42.
Houston SJ, Plunkett TA, Barnes DM, Smith P, Rubens RD, Miles DW. Overexpression of c-erbB2 is an independent marker of resistance to endocrine therapy in advanced breast cancer. Br J Cancer. 1999;79:1220–6.
Arpino G, Wiechmann L, Osborne C, Schiif R. Crosstalk between the estrogen receptor and the HER tyrosine kinase receptor family: molecular mechanism and clinical implications for endocrine therapy resistance. Endocr Rev. 2008;29:217–33.
Giuliano M, Hu H, Wang YC, Fu X, Nardone A, Herrera S, et al. Upregulation of ER signaling as an adaptive mechanism of cell survival in HER2-positive breast tumors treated with anti-HER2 therapy. Clin Cancer Res. 2015;21:3995–4003.
Lukas J, Bartkova J, Bartek J. Convergence of mitogenic signalling cascades from diverse classes of receptors at the cyclin D-cyclin-dependent kinase-pRb-controlled G1 checkpoint. Mol Cell Biol. 1996;16:6917–25.
Prall OW, Rogan EM, Sutherland RL. Estrogen regulation of cell cycle progression in breast cancer cells. J Steroid Biochem Mol Biol. 1998;65:169–74.
Metzger O, Mandrekar S, Loibl S, Mundhenke C, Seiler S, Valagussa P, Lim E, Tripathy D, Winer EP, Huang C, Carey L, Francis P, Miller K, Goetz MP, Prat A, Loi S, Krop I, Gianni L, Delalogue S, Vaz-Luis I, Dockter T, Lanzillotti J, Ciruelos E, DeMichele AM. PATINA: A randomized, open label, phase III trial to evaluate the efficacy and safety of palbociclib + anti-HER2 therapy + endocrine therapy (ET) vs. anti-HER2 therapy + ET after induction treatment for hormone receptor positive (HR+)/HER2-positive metastatic breast cancer (MBC) [abstract]. In: Proceedings of the 2018 San Antonio Breast Cancer Symposium; 2018 Dec 4-8; San Antonio, TX. Philadelphia (PA): AACR; Cancer Res 2019;79(4 Suppl):Abstract nr OT3-02-07. https://doi.org/10.1093/annonc/mdy272.357
Trastuzumab Deruxtecan (T-DXd) With or Without Pertuzumab Versus Taxane, Trastuzumab and Pertuzumab in HER2-positive Metastatic Breast Cancer (DESTINY-Breast09) ClinicalTrials.gov ID NCT04784715
A Phase 1b/2 Study of T-DXd Combinations in HER2-positive Metastatic Breast Cancer (DB-07) ClinicalTrials.gov ID NCT04538742
Cortés J, Baselga J, Im YH, Im SA, Pivot X, Ross G, Clark E, Knott A, Swain SM. Health-related quality-of-life assessment in CLEOPATRA, a phase III study combining pertuzumab with trastuzumab and docetaxel in metastatic breast cancer. Ann Oncol. 2013;24(10):2630–5. https://doi.org/10.1093/annonc/mdt274.
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Study conception NDP. Study design MK and NDP. Data collection MK, MP, TS, NDP. Data analysis MK and DG. Data interpretation MK, MP, TS, DG, NDP. The first manuscript draft MK, MP, NDP. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
MK declares speaking honoraria from Roche and Servier and institutional research funding from Roche, Novartis, and Pierre Fabre outside the submitted work. MP declares consulting/speaking honoraria from Roche, Gilead, Novartis, Pfizer, Astra Zeneca and institutional research funding from Roche and Novartis outside the submitted work. TS declares consulting/speaking honoraria from Roche, Novartis, Pfizer, Eli Lilly, Astra Zeneca, Swixx BioPharma and institutional research funding from Roche, Novartis, and Pierre Fabre outside the submitted work. NDP declares consulting/speaking honoraria from Roche, Novartis, Pfizer, Ely Lilly, Astra Zeneca, MSD, Gilead, Swixx BioPharma and institutional research funding from Roche, Novartis, and Pierre Fabre outside the submitted work. DG has no conflicts of interest to disclose.
Ethics Approval
The Ethics Committee of University Hospital Centre Zagreb approved the study design. Approval number: 02/21 AG, Date: April 24, 2018.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Križić, M., Popović, M., Silovski, T. et al. Real-world Outcomes of Dual HER2 Blockade Therapy in Metastatic HER2-Positive Breast Cancer: from Induction to Maintenance. Drugs - Real World Outcomes 11, 413–423 (2024). https://doi.org/10.1007/s40801-024-00438-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40801-024-00438-x