Abstract
In the COVID-19 pandemic era, anti-SARS-CoV-2 vaccination is considered to be the most efficient way to overtake the COVID-19 scourge. Like all medicines, vaccines are not devoid of risks and can in rare cases cause some various side effects. The objective of this case report is to highlight this unusual presentation of Henoch-Schönlein purpura following an anti-COVID-19 vaccination in a 62-year-old adult. The 62-year-old patient admitted to the emergency room for a petechial purpuric rash, sloping, occurring within hours, involving both legs and ascending. The clinical signs also included polyarthralgia and hematuria. Reported in the history the notion of an anti-COVID-19 vaccination 8 days prior to the onset of symptomatology. In the case of our patient, we retain the diagnosis of rheumatoid purpura based on the EULAR/PRINTO/PReS diagnostic criteria. Corticosteroid therapy (prednisone) was started, resulting to a rapid regression of clinical and laboratory symptoms, few days after the treatment. Patient was asymptomatic on subsequent visits. The low number of published cases of post-vaccine vasculitis does not question the safety of vaccines, but knowledge of such complications deserves to be known in order to avoid new immunizations that could have more serious consequences, and to avoid aggravating or reactivating a pre-existing vasculitis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vasculitides are a group of heterogeneous diseases characterized by inflammation of the vascular wall. Their pathophysiological mechanism remains complex and little known. A combined role of a particular genetic background and environmental factors (particular exposures to toxins or infections such as the hepatitis B or C virus) can explain the diseases [1].
Among these factors, we find vaccines. Several cases of post-vaccine vasculitis have been described in the literature.
We report here a case of post-COVID-19 vaccine rheumatoid purpura diagnosed within the internal medicine department, at the Cheikh Khalifa International University Hospital in Casablanca, Morocco.
Observation
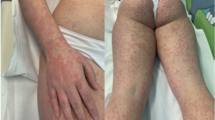
This is a 62-year-old patient admitted to the emergency room for a petechial purpuric rash, sloping, occurring within hours, involving both legs and ascending (Fig. 1).
The patient also reports bilateral polyarthralgia of the knees and ankles which had occurred a day before.
His history includes osteosarcoma of the left tibia at the age of 5 treated with surgery and chemotherapy, intercostal shingles at the age of 16, tonsillectomy at the age of 20, and COVID-19 infection in October 2020 without notable complications. We also note in its medical history the notion of first dose anti-COVID-19 vaccination [Oxford-AstraZeneca COVID19 = ChaAdOx1 nCoV-19 vaccine (AZD1222)] 8 days before the onset of its symptomatology.
Investigations
The biological assessment carried out in the emergency department found an inflammatory syndrome (CRP at 25.64 mg/L) and D-dimers at 1490 ng/mL. In addition, no anomaly was found in the blood count.
An autoimmune assessment was performed in the internal medicine consultation the next day and found positive ANA at 640 and a rheumatoid factor at 215 IU/mL.
A cytobacteriological urine test was performed and showed microscopic hematuria at 2000 RBCs/mL.
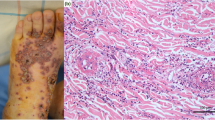
Corticosteroid therapy (prednisone) was started at a dose of 40 mg per day for 7 days, leading to a rapid regression of clinical and laboratory symptoms: regression/healing of the lesions (Fig. 2), a negativation of ANA, a decrease in rheumatoid factor to 127 IU/mL, D-dimers to 360 ng/mL and CRP to 4.5 mg/L.
Discussion
To our knowledge, up to date, this is the second case of post-vaccine vasculitis described in the literature [2], as part of the SARS-CoV-2 vaccine in general, and first AZD1222 (ChAdOx1 nCoV-19) in particular. And the first post-COVID-19 vaccine Henoch-Schönlein vasculitis was described.
Henoch-Schönlein disease, or rheumatoid purpura, is vasculitis affecting capillaries in the blood as a result of IgA deposition in the vessel walls [3, 4].
The clinical signs associated with Henoch-Schönlein disease are a change in general condition and fever, but also other more specific signs [3, 5]:
-
Symmetrical vascular purpura, not erased by diascopy, most often present in areas of pressure with the primary lesion of petechia or even bruising, or necrosis or hemorrhagic bubbles in adults. The skin lesions disappear in about 15 days.
-
Arthralgia localized to the knees and ankles in more than ¾ of cases.
-
Diffuse abdominal pain of significant intensity in 60% of the cases identified.
-
Intussusception is possible in children and is most often accompanied by vomiting and upper or lower gastrointestinal bleeding.
-
Glomerular renal impairment in 40% of cases, which results in proteinuria and/or hematuria, without the presence of renal failure in most cases.
The exact pathophysiological mechanism in post-vaccine vasculitis is still poorly understood. The most commonly discussed hypothesis is immunological, related to the presence of immune complexes starting from the vaccine antigen. The pathophysiology is similar to that linking periarteritis nodosa to HBV Ag-Ac immune complexes. However, in the majority of vasculitis, the causative agent is not known [6].
In 2010, EULAR/PRINTO/PReS defined criteria (Table 1) that have become the benchmark for the diagnosis of Henoch-Schönlein disease (HSP). The diagnosis of HSP is clinical. These criteria, applicable to adults, have a diagnostic sensitivity of 79.2% and a specificity of 86%, which encourages their use for any patient suffering from this pathology [10].
In the case of our patient, we retain the diagnosis of rheumatoid purpura in front of:
-
Petechial purpuric rash (mandatory criterion).
-
Polyarthralgia.
-
Hematuria at cytobacteriological urine test.
AZD1222 (ChAdOx1 nCoV-19) is a vaccine against SARS-CoV-2 which uses a non-replicating chimpanzee adenovirus as a vector (ChAdOx1) and is modified to induce the S protein of SARS-CoV-2. AZD1222 was developed by the University of Oxford and AstraZeneca [11].
Globally, as of 5:15 p.m. CEST on May 10, 2021, 157,973,438 confirmed cases of COVID-19, including 3,288,455 deaths, have been reported to the WHO. As of May 10, 2021, a total of 1,206,243,409 doses of vaccine have been administered [12].
In Morocco, as of January 3, 2020, at 3:52 p.m. CET, on May 10, 2021, there have been 513,864 confirmed cases of COVID-19 with 9072 deaths, reported to the WHO. As of May 10, 2021, a total of 9,325,597 doses of vaccine have been administered [13].
Morocco is one of the best-known countries in terms of anti-SARS-CoV-2 vaccine strategy in the world. The vaccination rate in Morocco (9.75%) accomplished in a single month, which slightly surpasses that of France (6.97%), Italy (7.12%), and Spain (7.71%) in 2 months of vaccination [14].
Priority is given to people on the front lines, such as health workers, teachers, security forces, law enforcement officials, elders, and people with underlying health conditions. Thereafter, vaccination will be extended to the general population by age group [14].
All medicines, including vaccines, have side effects that vary in severity (including death). However, despite the risks, it is necessary to be vaccinated given the clearly established benefits from a public health point of view: Vaccines prevent between 2 and 3 million deaths each year from infectious diseases [15].
Given the high incidence of infectious-starting autoimmune diseases in COVID-19 infection (Kawasaki diseases, Guillain-Barre syndromes, autoimmune myasthenia gravis). We tried through this work to propose a research hypothesis which aims to demonstrate a link between the particles of the anti-SARS-CoV-2 vaccine and Henoch-Schönlein vasculitis.
Considering the fact that our patient has a history of recent COVID-19 infection in October 2020, the anti-SARS-CoV-2 vaccine could have induced a “trained immunity” in already vulnerable patients (history of COVID-19), given the fact that the patient has already been sensitized to SARS-CoV-2 components, particularly the spike protein which is used in the COVID-19 vaccines [16].
In the same logic, a lot of infections including COVID-19 are associated with the release of cytokines, the primary immune system regulators. Some of these cytokines can dysregulate the immune system and contribute to an inflammatory state called the cytokine storm [17] that increases between day 5 and day 13 [18]. It is known in the literature that the cytokine storm can be the gateway to certain auto-immune diseases.
A case of a mild itchy erythematous macular morbilliform rash in the lower back 48 h after administration of the first dose of Pfizer-BioNtech COVID-19 mRNA vaccine has been described in the literature. Then, a more significant recurrent morbilliform rash has appeared on both flanks and flesh-colored papules on the right flank with itching and significantly increased bodily surface damage after 48 h of the second booster dose [19].
The association between the anti-SARS-CoV-2 vaccine and the development of HSP is notable. We cannot explain the immunopathogenic link between ChAdOx1 nCoV-19 vaccine and HSP, but we believe that practitioners should be aware of this possible complication and evaluate other cases of post-vaccine vasculitis with keeping causality in mind. However, HSP is rarely used with vaccines and is not expected to affect vaccine use.
Conclusion
Although the existence of post-vaccine vasculitis has yet to be formally demonstrated, we are adding this case of post-vaccine rheumatoid purpura to the already existing list.
The low number of published cases of post-vaccine vasculitis does not question the safety of vaccines, but knowledge of such complications deserves to be known in order to avoid new immunizations that could have more serious consequences, and to avoid aggravating or reactivating a pre-existing vasculitis.
Abbreviations
- SNFMI:
-
French National Society of Internal Medicine
- COVID-19:
-
coronavirus disease 2019
- SARS-CoV-2:
-
severe acute respiratory syndrome coronavirus 2
- CRP:
-
C-reactive protein
- ANA:
-
anti-nuclear antibodies
- IgA:
-
immunoglobulin A
- Ag-Ac:
-
antigen-antibody
- HBV:
-
hepatitis B virus
- EULAR:
-
European League Against Rheumatism
- PRINTO:
-
The Pediatric Rheumatology INternational Trials Organization
- PReS:
-
Pediatric Rheumatology European Society
- HSP:
-
Henoch-Schönlein purpura
- CET:
-
Central Europe Time
- WHO:
-
World Health Organization
- mRNA:
-
messenger ribonucleic acid
References
Vascularites systémiques | SNFMI. https://www.snfmi.org/node/56. Accessed 6 Jul 2021.
Cohen SR, Prussick L, Kahn JS, Gao DX, Radfar A, Rosmarin D. Leukocytoclastic vasculitis flare following the COVID-19 vaccine. Int J Dermatol. 2021:ijd.15623.
Purpura rhumatoïde. Wikipédia. 2021. https://fr.wikipedia.org/w/index.php?title=Purpura_rhumato%C3%AFde&oldid=179463092. Accessed 24 Apr
Pillebout E. Purpura rhumatoïde de l’adulte. 2008;:8.
Clinical update: Henoch-Schönlein purpura - The Lancet. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(07)60474-7/fulltext. Accessed 24 Apr 2021.
Saadoun D, Cacoub P, Mahoux D, Sbai A, Piette JC. Vascularites postvaccinales: à propos de trois observations. Rev Médecine Interne. 2001;22:172–6.
McPartland K, Wright G. Acute abdominal pain: Henoch–Schönlein purpura case in a young adult, a rare but important diagnosis. Clin Med. 2019;19:77–9.
Ozen S, Pistorio A, Iusan SM, Bakkaloglu A, Herlin T, Brik R, et al. EULAR/PRINTO/PRES criteria for Henoch–Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part II: Final classification criteria. Ann Rheum Dis. 2010;69:798–806.
Reamy BV, Servey JT, Williams PM. Henoch-Schönlein purpura (IgA vasculitis): rapid evidence review. Am Fam Physician. 2020;102:229–33.
Hetland L, Susrud K, Lindahl K, Bygum A. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol. 2017;97:1160–6.
Belete TM. <p>Review on up-to-date status of candidate vaccines for COVID-19 disease</p>. Infect Drug Resist. 2021;14:151–61.
WHO Coronavirus (COVID-19) Dashboard | WHO Coronavirus (COVID-19) Dashboard With Vaccination Data. https://covid19.who.int/. Accessed 13 Apr 2021.
Morocco: WHO Coronavirus Disease (COVID-19) Dashboard With Vaccination Data. https://covid19.who.int. Accessed 13 Apr 2021.
Drissi Bourhanbour A, Ouchetto O. Morocco achieves the highest COVID-19 vaccine rates in Africa in the first phase: what are reasons for its success? J Travel Med. 2021;28. https://doi.org/10.1093/jtm/taab040.
Cirillo N. Reported orofacial adverse effects of COVID-19 vaccines: the knowns and the unknowns. J Oral Pathol Med. https://doi.org/10.1111/jop.13165.
Luciani M, Bentivegna E, Spuntarelli V, Amoriello Lamberti P, Guerritore L, Chiappino D, et al. Coinfection of tuberculosis pneumonia and COVID-19 in a patient vaccinated with Bacille Calmette-Guérin (BCG): case report. SN Compr Clin Med. 2020;2:2419–22.
Najafi S, Rajaei E, Moallemian R, Nokhostin F. The potential similarities of COVID-19 and autoimmune disease pathogenesis and therapeutic options: new insights approach. Clin Rheumatol. 2020;39:3223–35.
Rowaiye AB, Okpalefe OA, Adejoke OO, Ogidigo JO, Oladipo OH, Ogu AC, et al. Attenuating the effects of novel COVID-19 (SARS-CoV-2) infection-induced cytokine storm and the implications. J Inflamm Res. 2021;14:1487–510.
Jedlowski PM, Jedlowski MF. Morbilliform rash after administration of Pfizer-BioNTech COVID-19 mRNA vaccine. Dermatol Online J. 2021;27 https://escholarship.org/uc/item/4xs486zg. Accessed 13 Apr 2021.
Availability of Data and Material
Not applicable
Code Availability
Not applicable
Author information
Authors and Affiliations
Contributions
• Abdelhamid Naitlho, Wahib Lahlou, and Abderrahim Bourial took part in taking care of the patient, taking photos, researching the bibliography, and writing the article.
• Hamza Rais participated in the bibliographic research.
• Imad Abousahfa and Lahcen Belyamani participated in the care of the patient.
• Nabil Ismaili participated in the design and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable
Consent to Participate
Orally
Consent for Publication
Orally
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on COVID-19
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Naitlho, A., Lahlou, W., Bourial, A. et al. A Rare Case of Henoch-Schönlein Purpura Following a COVID-19 Vaccine—Case Report. SN Compr. Clin. Med. 3, 2618–2621 (2021). https://doi.org/10.1007/s42399-021-01025-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42399-021-01025-9