Abstract
Acmella oleracea L. (Asteraceae), commonly known as the toothache plant, is a tropical herb with significant therapeutic potential. Traditionally, it has been used for its analgesic and sialagogue properties, and it is known for its distinctive yellow flower crest. This review addresses current gaps in the understanding of its pharmacological properties and aims to provide a comprehensive analysis of its therapeutic potential. A thorough literature review was conducted using databases such as Google Scholar, ScienceDirect, and PubMed/MedLine. The findings indicate that A. oleracea contains a variety of phytochemicals, including spilanthol, which exhibit diverse pharmacological activities such as antioxidant, anti-inflammatory, immunomodulatory, antinociceptive, pronociceptive, antibacterial, antifungal, antidiabetic, diuretic, and vasorelaxant effects. The results highlight the significant medicinal potential of A. oleracea while also identifying areas for further research, particularly regarding its toxicological impacts on humans and animals. This review aims to guide future studies on the health benefits and safety of this plant.
Article Highlights
-
Acmella oleracea exhibits significant pain-relieving properties, interacting with pain receptors effectively.
-
The plant's extracts show strong antimicrobial effects, useful against various bacterial infections.
-
Acmella oleracea demonstrates potent antioxidant and antifungal activities, promoting health and disease prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The South American herb Acmella oleracea L., also known as Spilanthes acmella Murr. or Spilanthes oleracea L. [1], has a variety of pharmacological qualities, including those that are anti-malarial, anesthetic, and anti-inflammatory. Extensive research has been carried out on this topic as a result of the rising demand for natural medications and their acceptability on the global market due to their beneficial medicinal properties and therapeutic potential [2,3,4]. To get these medications on the market, however, research must be done to analyze, standardize, and evaluate them for their potency, safety, and efficacy. A. oleracea is an important herb that is found across the world's tropical and subtropical areas and is an important source of several medicinal compounds [5]. The ability of A. oleracea to enhance the production of saliva (sialagogue), as well as its numbing and tingling effects, led to its common name, "toothache plant"[6]. Jambu, agrio do pará, akmaella, spot flower, bredemafane, and paracress are additional names for A. oleracea. The plant is well-known in Brazilian (jambu) cuisine because of its numbing and spiced properties [7]. The leafy parts and flowers of the medicinal herb A. oleracea have traditionally been applied locally to alleviate toothaches. On the other hand, stomatitis and the common cold are both cured by A. oleracea [8]. Multiple investigations have demonstrated that A. oleracea possesses a wide range of intriguing pharmacological features, such as vasorelaxant, analgesic, local anesthetic, diuretic, and antioxidant [9,10,11,12]. Due to the anesthetic action of the plant's additional metabolites, tongue-tingling occurs when its flowers, leaves, and stems are ingested [6, 13, 14]. In addition, mouth ulcers, herpes, and throat-related infections are treated with it in conventional medicine as a strong analgesic and regional anesthesia. According to several studies[15,16,17,18], it is also employed as an insecticide and fungicide and exhibits fungistatic properties. Lipophilic alkylamides or alkamides, such as spilanthol, are the most extensively studied compounds from A. oleracea, in spite of the fact that there have been several biologically active substances identified in this plant [19]. These compounds also appear to be the result of the plant's antioxidant and anti-inflammatory properties. In addition, triterpenoids, coumarin, and phenolic compounds have all been discovered to be present in S. acmella [8]. Additionally, rhamnogalacturonan, a kind of polysaccharide with gastro-protective action, has been extracted from the leafy parts of A. oleracea and showed positive impacts on acute gastric ulcers brought on by ethanol in rats [20]. This review endeavors to illustrate the most comprehensive and updated research on the pharmacological and phytochemical properties of A. oleracea. Unlike previous reviews that have focused primarily on the phytochemistry and specific pharmacological effects of related species such as Portulaca oleracea and Brassica oleracea var. acephala, this review provides a detailed analysis encompassing a wider range of pharmacological activities and potential therapeutic applications of Acmella oleracea. It specifically addresses the gaps in understanding its toxicological impacts and aims to offer a more integrative perspective on its medicinal potential and safety profile, which have been less emphasized in prior works.
2 Methodology
The following resources were searched: PubMed/MedLine, Science Direct, and Google Scholar. The term "Acmella oleracea L." was combined with "phytochemicals," "pharmacological activities/effects," or "biological activities/effects." The research and data collection for this review were conducted over a period from January 2020 to December 2022.The constraints of language were not taken into account. The data collected from all the papers outlining the concentration or dose (way of administration), experimental models, results, or potential mechanisms underlying the action of the plant or its constituents was evaluated. The following lists the inclusion and exclusion criteria for articles located in databases.
2.1 Inclusion criteria
-
1.
Research done on humans, human tissues, and human cells in vitro, ex vivo, or in vivo with or without employing laboratory animals.
-
2.
Research using A. oleracea and its raw extracts, isolated chemicals, or prepared derivatives.
-
3.
Tests with or without a suggested mechanism(s) that might account for certain outcome.
-
4.
Studies using extracts that demonstrate biological action but lack phytochemical evaluations.
2.2 Exclusion criteria
-
Duplicate information and headings and/or abstracts that don't match the requirements for inclusion standards.
-
Studies involving A. oleracea that do not address the current subject.
-
Studies involving a different plant genus or species
The taxonomy of the plant has been validated with World Flora Online, and chemical formulas with PubChem.
3 Botanical data, phytochemistry and ethnopharmacology
3.1 Botany
3.1.1 Taxanomy of A. oleracea
The taxonomy of A. oleracea is given below:
Kingdom: Plantae
Phylum: Tracheophyta
Class: Magnoliopsida
Order: Asterales
Family: Asteraceae
Subfamily: Mimosoideae
Genus: Acmella
Species: Acmella oleraceac L.
3.1.2 Morphology of A. oleracea
The annual herb A. oleracea has stems that are either erect or decumbent. The stems are usually reddish in color, hairless, and prostrate or erect. According to Favoreto and Gilbert (2010), the leaves of the plant are in opposite directions, simple, generally ovate to triangle-shaped, length 5–11 cm, width 4–8 cm, with a tapered to slightly attenuated base, a sharp to briefly angled top, and a dentate margin. Inflorescence: discoid head up to 2.5 cm, 1.5 cm; involucral bracts 15–18, 3-seriate, up to 8 mm, 1 mm, apex acute; receptacular bractlet straw-colored, frequently tinted purple–red, up to 6 mm, 1 mm; Corolla 5-merous, yellow, up to 3.5 mm long, disk blooms 400–620. Achene has two bristles on the pappus, measuring 2–2.5 mm × 1 mm [13]. Different parts of A. oleracea are shown in Fig. 1.
3.2 Phytoconstituents of Acmella oleracea—overview of the main chemical compounds identified
The chemical composition of Acmella oleracea has been extensively studied, revealing a rich variety of phytochemicals. Spilanthol (N-isobutyl-2E, 6Z, and 8E-decatrienamide), an insecticide and a key bioactive compound, is one of the primary components identified. Additionally, butylated hydroxytoluene is another significant compound found in the plant. The ethanolic extract of A. oleracea shows a GCMS chromatogram with 48 peaks, suggesting the presence of at least 48 phytochemical components, of which 15% are essential oils. The most common substances identified in the plant include 9,12,15-Octadecatrienoic acid, (Z,Z,Z)- (14%), 3,7,11,15-Tetramethylhexadec-2-En-1-Ol (16%), Cedrene (10.65%), Pentadecanoic acid (8%), 1-Butanol, 3-methyl-, acetate (5.65%), Stigmast-5-en-3-Ol, (3-beta)—(1.79%), Ethyl (9Z,12Z)-9,12-Octadecadienoate (1.35%), Phytol, acetate (2.67%), 1,3-Bis(Cinnamoyloxy-methyl) adamantane (2.13%), Beta-elemen (1.90%), DL-alpha-tocopherol (1.84%), N-Isobutyl-(2E,4Z,8Z,10E)-dodeca-tetraenamide (1.29%), Trans (Beta)-Caryophyllene (3.28%), and Ethyl linoleate (2.85%) [21, 22]. The main chemical classes found in the plant include sesquiterpenes, steroidals, terpene alcohols, polyenoic fatty acids, palmitic acid ethyl esters, and hydrocarbons. Fatty acid amides, such as spilanthol, are particularly notable for their taste-active properties and involvement in saliva-inducing mechanisms. These compounds are significant in products like Jambu oleoresin, an intense preparation from A. oleracea. [14, 23]. The chemical structures of these phytoconstituents are shown in Fig. 2.
3.3 Traditional uses of Acmella oleracea
A. oleracea, commonly known as the toothache plant, has a rich history of use in traditional medicine across various cultures, particularly in tropical and subtropical regions. The plant is well-known for its analgesic and sialagogue properties, which have been utilized by indigenous populations for centuries[24]. In South America, especially in Brazil, A. oleracea (locally known as jambu) is widely used to alleviate toothache and other oral health issues [25]. The leaves and flowers of the plant are chewed to produce a numbing and tingling sensation in the mouth, which is primarily attributed to the bioactive compound spilanthol[25]. This compound is known to interact with sensory receptors, providing relief from pain and discomfort. Beyond its use for toothache, A. oleracea has been employed to treat a variety of ailments. Traditional medicine practitioners use the plant to treat gum infections, mouth ulcers, and stomatitis. Its antimicrobial properties are harnessed to combat throat infections and as a general antiseptic [24]. In addition to its oral applications, A. oleracea is utilized in Brazilian cuisine for its unique flavor and sensory effects. The plant is an essential ingredient in regional dishes, adding a distinctive numbing sensation that enhances the culinary experience [7]. The traditional uses of A. oleracea extend to its application as a diuretic and anti-inflammatory agent. The plant has been used to treat fever, rheumatism, and to boost the immune system. Studies have documented its effectiveness in enhancing saliva production, reducing inflammation, and providing pain relief, supporting its traditional use in these areas [25]. The extensive traditional uses of A. oleracea provide a valuable foundation for modern scientific research. Investigating these traditional practices can offer insights into the plant's pharmacological properties and potential therapeutic applications, bridging the gap between ethnomedicine and contemporary biomedical research.
4 Pharmacological activities of Acmella oleracea: mechanistic insights
4.1 Antioxidant effect
The formation of free radicals in the body creates an imbalance between oxidative and antioxidative mechanisms, known as oxidative stress. Free radicals are molecules that destroy proteins through oxidative reactions and are mainly generated by mitochondria, potentially leading to various ailments [26, 27]. These damaging oxygen radicals are absorbed and compensated by antioxidants, which decrease the damage to the body [28, 29]. Beer's antioxidant activity was substantially enhanced by adding Acmella oleracea extract using supercritical extraction. Applying the ABTS, DPPH, and ORAC techniques, the antioxidant activity was determined, and an important compound, spilanthol, was identified by utilizing the Gas Chromatography-Flame Ionization Detection (GC-FID), Thin-Layer Chromatography (TLC), and Fourier Transform Infrared-Attenuated Total Reflectance (FTIR-ATR) techniques. This study demonstrated that jambu flower extract has the potential to be used as an antioxidant addition to beer [10]. Plant powders have been extracted with 1% HCl in a 90% methanol solvent for two hours in order to measure the amounts of phenolic compounds and determine the antioxidant effect of Acmella oleracea. Then, DPPH and ABTS tests were used to evaluate the antioxidant potential of the extracts. Each extract's ability to scavenge radicals was measured using Trolox equivalents (TE; μmol/g dry weight) and compared to a Trolox standard. Both tests showed that, in comparison, Acmella oleracea's leaves, stems, and flowers have the highest antioxidant properties [30]. In this investigation, the antioxidant activity of the methanol extract of A. oleracea was evaluated using both a hydrogen peroxide- and hydroxyl radical-scavenging assessment. The plant extract demonstrated free radical scavenging capabilities in both assays in a dose-dependent way. The MTT test was used to determine its cytotoxic properties, and the results suggested that it may have a cytotoxic impact on cancerous cells such as the HeLa and V79 cancer cell lines [31]. The most potent superoxide detoxification activity was found in the methanol extract of the stem of S. acmella, while the most powerful DPPH scavenging activity was found in the leaves of this plant [32].
4.2 Antinociceptive properties
In pain management, understanding antinociceptive properties is vital due to varying pain perceptions among individuals[33]. Temporal Summation of Pain (TSP) helps differentiate people based on heightened or reduced pain sensations; recognizing these profiles is fundamental for effective treatment strategies, as some patients require intense treatments while others respond to standard therapies.[34]. An expeditiously expanding body of studies into painful visceral syndromes can compete with significant research into neuropathic or inflammatory pain by exploring specific groups of patients with pain disorders [35]. By using in vivo and in silico experiments, the antinociceptive properties of A. oleracea methanolic extracts can be determined. The antinociceptive activity of alkylamides was confirmed by the extracts' inhibition of the neurogenic and inflammatory aspects during the formalin exposure experiment. Additionally, the findings using molecular docking revealed that A. oleracea alkylamides are associated with the CB1/CB2 and TRPV1 receptors. In addition, the flower's crude methanol extraction did not result in any of the anticipated negative effects associated with the traditional cannabinoid tetrad, such as seizures and hyperlocomotion. In conclusion, this study validates the potential of A. oleracea 's alkylamides as antinociceptive drugs and indicates, for the first time in history, a correlation between their impact and those of the endocannabinoid and vanilloid signaling pathways [36]. The mechanisms for the antinociceptive (0.1μg) and pronociceptive (30μg) effects of hexanic fraction (HF) and isobutylalkyl amide (IBA) have been investigated in chemical and sensory assays after intraplantar injection into Swiss male mice. In the formalin experiment towards glutamate-induced nociception, HF and IBA with 0.1 μg enhanced analgesic alleviation both in neurogenic and inflammatory stages. The effect depended on TRPV1 (Transient Receptor Potential Vanilloid 1) regulation and was independent of activation of the endogenous opioid system. Additionally, compound 48/80-induced pain and mast cell breakdown in the hind paw were both reduced by high-frequency stimulation (HF). However, according to Dallazen et al. (2018), both drugs enhanced nociception, which was mediated by TRPA1[37].
4.3 Antimicrobial effects
Antimicrobials are medicinal compounds used for preventing or treating infections and include antiseptics, fungicides, antibacterials, antivirals, and antiparasitics[38]. They focus on essential cellular metabolic events such as cell wall and membrane configurations, mutations in genes, the activity of enzymes, and the synthesis of macromolecular substances [39]. A disc diffusion in vitro assay was used to test the antibacterial activity of a methanol extract of Acmella oleracea leaves against four different gram-positive and gram-negative bacteria. Zones of inhibition ranged from 10 to 26 mm, indicating that the extracts demonstrated mild to average antibacterial properties against both gram-negative and gram-positive bacteria. The maximum degree of activity was observed in Vibrio mimicus bacterial strains, with a growth inhibition zone diameter of 23 mm. These results suggest that Acmella oleracea is a potential source of antibacterial agents [40]. Acmella oleracea's aqueous extract and hydroethanolic extract have been chosen for the antimicrobial tests, both of which showed a minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of 125 g/ml against S. mutans. A great amount of spilanthol concentration was found in the leaves of the Pará variety (EELAO-PA) as well as the stem of the Amapá variation (EESAO-AP), at 99.97% and 99.17%, respectively. EELAO-PA at 500 g/ml (4xMIC) demonstrated a decrease in activity of 50.89% with a single dose in the antibiofilm experiment, comparable to that discovered following exposure with chlorhexidine gluconate 0.12%. As a result, extracts from A. oleracea containing significant spilanthol indicated significant antibacterial action in combating the cariogenic S. mutans and may potentially be employed as a coadjuvant in low-cost dental treatments for the prevention and treatment of dental decay [41]. Through the use of several drying methods and solvent extractions, the study examined the antibacterial characteristics of Acmella flower pods that had been preserved. Three distinct solvent extracts (ethanol extracts (EE), water extracts (WE), and pet ether extracts (PEE)) have been assessed for extraction production, phytochemical screening, and in vitro antimicrobial efficacy. Zones of inhibition (ZOI) for Staphylococcus aureus, Bacillus subtilis, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans were greater in CWD-dried WE, measuring 18.8, 14.0, 12.0, 20.2, and 17.3 mm, respectively. The antimicrobial activity of CWD-dried pod extracts against bacteria and fungi was greater than that of SD (sun-drying) extracts [42]. The research conducted in vitro explores the ethanol extract and fractions of A. oleracea 's antimicrobial activity against infectious microbes in the skin and digestive tract of domesticated livestock. The entire extract and fragments have been assessed against both fungi and bacteria at a variety of concentrations (1000, 500, 250, 125, 62.5, 31.25, 15.62, and 7.81 mg/mL). Spilanthol was discovered by phytochemical study, perhaps through thin-layer chromatography with Dragendorff reagent. The discovery and development of A. oleracea e as an herbal supplement were made possible by the chloroform fraction's ability to reduce Salmonella typhi's multiplication with MIC conc at 31.25 mg/mL [43]. In three identical wells, 50 μl of the A. oleracea extract at a concentration of 100 mg/ml were added. After one hour at ambient temperature, transfer the extract to the agar plates. The agar plates were allowed to incubate at 37°C for about 18 h. By employing the well diffusion procedure, the antimicrobial efficacy of A. oleracea was examined in relation to various isolates of bacterial species, including Pseudomonas aeruginosa, Streptococcus salivarius, Streptococcus viridans, Streptococcus mutants, Bacillus megaterium, and Neisseria catarrhalis. While A. oleracea 's chloroform extract had a substantial impact on S. viridans, its extract of acetone indicated great efficacy against N. catarrhalis and S. mutans. Conversely, methanol extract demonstrated a potent effect against B. megaterium and P. aeruginosa. There was no suppressive effect of the water extract on the subject species [44].
4.4 Antifungal effect
Both in developed and emerging nations, infections caused by fungi are the primary contributory factor to death rates. To combat these infections, a variety of potent natural anti-fungal drugs are available [45]. In an in vivo investigation, it was discovered that Spilanthol (AcO) was a fungal inhibitor against the planktonic and biofilm subtypes of a multidrug-resistant C. albicans variant, impacting the functioning of the yeast cell wall and membrane of cells and inhibiting the growth of fungus. Spilanthol decreased biofilm adhesion, growth, and the removal of DNA and carbohydrates from the biofilm structure. Spilanthol has been proposed as a viable alternative therapy for vulvovaginal candidiasis after infection of rats with vulvovaginal candidiasis demonstrated the alleviation of fungal infections and signs of inflammation [16]. In another in vitro experiment to evaluate Spilanthes acmella's effectiveness with Ca(OH)2, an impressive zone of inhibition was obtained against dental canal infection. It was discovered that the C. albicans zone of inhibition increased as concentrations rose. For E. faecalis, SPA demonstrated a zone of inhibition around 2–5% and 10%, while Ca(OH)2 produced a zone of inhibition merely at 10%. For Streptococcus sp. and S. aureus, respectively, Ca(OH)2 exhibited a considerable zone of inhibition at 5% and 10%, whereas SPA demonstrated a zone of inhibition only at 10%. Endodontic problems are frequently caused by typical root canal infectious agents, including E. faecalis and C. albicans, for which SPA has exceptional antibacterial and antifungal efficacy [46]. The minimum inhibitory concentration (MIC) of B. aegyptiaca and S. acmella alcoholic extracts against the assessed fungi was 3.05 to 24.0 g/ml and 1.53 to 49.0 µg/ml, respectively. The results of the study suggest that extracts of Balanites aegyptiaca and Spilanthes acmella have potent antifungal properties and may be used as significant therapy to treat a variety of possibly fatal fungal diseases caused by Candida species and Aspergillus species, particularly in immunodeficient individuals who might have complications with antifungal drug resistance [47].
4.5 Diuretic effect
The diuretic effect involves enhanced renal excretion of water and electrolytes, leading to increased urine output [48]. Diuretic agents are utilized in the management of conditions such as fluid retention, hypertension, heart failure, and cerebrovascular accidents. Fluid accumulation in tissues results from the kidneys' failure to adequately excrete sodium and its associated water [49]. The cold-water preparation of the A. olleracea flower remarkably enhanced urine production, urinary Na + and K + extents, and decreased the level of osmolarity of urine, indicating that it primarily functions like a loop diuretic. It could also suppress the secretion and function of the antidiuretic hormone (ADH) [50]. The urine production and elimination of Na + and K + were both considerably enhanced by the ethanol extract of A. olleracea leaves and it is significantly used in the treatment of hypertension [51].
4.6 Vasorelaxant effect: implications for hypertension and cardiovascular health
Hypertension, a critical regulator of various cardiovascular conditions including cardiac arrhythmia, heart failure, and atherosclerosis, is profoundly influenced by vascular tone. The modulation of vascular tone plays a pivotal role in the management of high blood pressure and its associated complications[52]. Effective regulation of vascular tone is not only essential for controlling hypertension, but also for preventing the progression of related cardiovascular diseases [53]. This understanding underscores the importance of identifying and developing therapeutic agents that can effectively induce vasorelaxation, thereby contributing to the broader strategy of cardiovascular health management [54]. The maximum vasorelaxation induced by Spilanthes acmella chloroform and ethylacetate extraction on phenylephrine is dose-dependent; however, it is smaller than the acetylcholine-enhanced nitric oxide (NO) vasorelaxation, and chloroform extract exhibited the most potent vasorelaxation effect and antioxidant properties [11].
4.7 Anti-hypercholesterolemic effects through pancreatic lipase inhibition
Pancreatic lipase plays a fundamental role in lipid metabolism by converting triglycerides into monoglycerides and free fatty acids, which are then absorbed and metabolized by the body and this process is important for the regulation of cholesterol levels in the bloodstream. By inhibiting this enzyme, the breakdown and subsequent absorption of dietary fats are reduced, leading to lower cholesterol levels in the body. This pathway offers a promising target for the development of anti-hypercholesterolemic therapies, focusing on the modulation of lipid digestion and absorption.[55]. In vitro experiments with ethanol extracts of S. acmella flowers they revealed a 40% suppression of pancreatic lipase function. It has been demonstrated that the action is inadequate when compared to Aframomum meleguetta (which showed 90% pancreatic lipase inhibition), although the 0.75 mg/mL extraction of the plant suppressed the greater pancreatic lipase [6, 56].
4.8 Immunomodulatory effects
Immunomodulatory medicines enhance or suppress the production of serum antibodies, improving the immune system's protection against ailments caused by infectious agents, malignancies, immunodeficient conditions, and antibody rejection. In contrast, immunosuppressive agents decrease immune reactions toward transplanted tissues and are used in the treatment of autoimmune disorders.[57]. Mice were used in the research to examine hematopoietic antibody (HA) synthesis and delayed-type hypersensitivity (DTH) reactions. For 7 days, the two groups of animals were orally administered an ethanolic extract of Spilanthes oleracea at doses of 200 mg/kg and 400 mg/kg, respectively. The mice were injected with 0.1 ml of 20% sheep red blood cell solution (SRBC) intraperitoneally every day for the next 7 days. On the seventh day post-vaccination, serum was extracted from each animal's bloodstream by taking samples from the retro-orbital plexus into small centrifuge tubes. The haemagglutination method was implemented for calculating the level of antibody. Stress-related conditions caused muscular weakness, which changed the levels of triglycerides, blood urea nitrogen, and lactate dehydrogenase. In mice, immediate and perpetual Spilanthes oleracea extract (SPE) therapy for 21 days dose-dependently alleviated all of this behavioral distress and related physiological adaptation, alleviating CFS symptoms because this extract has polyphenols along with flavonoids, which are thought to be accountable for its antioxidant and immunomodulatory properties [9].
4.9 Local anesthetic effect
In research conducted in vitro and in vivo, Spilanthes acmella, a plant used as a topical anesthetic, has been demonstrated to be reliable and efficient. Plenty of studies have shown that the plant's roots, petals, and leaves could potentially function as topical anesthetics, and those assertions have been verified by much research [12]. In vitro research to assess the effectiveness of the new mucoadhesive film comprising S. acmella extract as an oral mucosal local anesthesia. The effectiveness of the mucoadhesive films made of chitosan with S. acmella extract (10% JB: 2.37 mg; 20% JB: 5.13 mg; 10% JBC: 3.14 mg) has been examined by the permeability coefficient of spilanthol across the pig esophageal mucosa. The mucoadhesive film formulation showed high S. acmella penetration and efficient anesthetic activity. Also, an in vivo observation claimed that the duration of the anesthetic effect was estimated by the increase in the time requisite for the tail to be withdrawn in the tail-flick test of mice with the mucoadhesive films, and the effective formulation exhibited an in vivo anesthetic effect of methanolic extract. The results of the study suggest that the safe and low-toxic S. acmella extract with the mucoadhesive film might be applied as a backup to the topical and oral anesthetic preparations available for dental ailments [17]. The purpose of this study was to evaluate the efficacy and safety of an ointment made from Acmella oleracea that acts as a local anesthetic for buccal mucosa. Buccal mucosa that had dried out to receive the topical treatment. After that, the small needles were instantly withdrawn after being injected into the mucobuccal space. Everyone who participated performed under their supervision. An optical analog index and a heart rate monitor were used to evaluate pain. There were hardly significant variations between the two topical preparations of Acmella oleracea strengths examined and correlated with the benzocaine 20% control when using a one-way analysis of difference and a Kruskal-Walli’s analysis. In the approach utilized in this study, Acmella oleracea ointment proved useful and secure in minimizing discomfort during the injection of the needle [58]. The most important pharmacological properties of Acmella Oleraceae are summarized in Fig. 3 and Table 1.
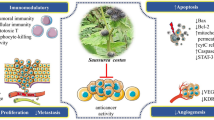
Illustrative scheme regarding pharmacological activities of Acmella oleracea. It exhibits a reduction in mitochondrial reactive oxygen species, signifying its potential to ameliorate oxidative damage. In the realm of pain relief, it acts as an antinociceptive by attenuating neurogenic inflammation and suppressing the activity of the pain-related ion channel TRPV1. It also possesses antimicrobial and antifungal characteristics, highlighting its efficacy against a spectrum of pathogenic microorganisms and fungi. In terms of immune response, Acmella oleracea serves an immunomodulatory role, curtailing the secretion of inflammatory cytokines such as TNF-α and IL-6. Addressing metabolic health, the plant demonstrates anti-obesity effects by impeding pancreatic lipase, which is in fat digestion, consequently influencing weight management. Its antihypertensive action is facilitated through the induction of vasorelaxation. Lastly, its diuretic capability is illustrated by its impact on ADH secretion, altering urine osmolality and the renal excretion of essential electrolytes like sodium, potassium, and chloride
5 Toxicological profile: safety and potential side effects of Acmella oleracea
The male reproductive system of Amblyomma cajennense sensu stricto (s.s.) ticks has been assessed in an experiment to see how different quantities of crude ethanolic extract of A. oleracea affect the tick's ability to reproduce. 100 males consumed food from host rabbits of identical body weight. There were five groups of ticks, each with ten animals. Treatments 1 through 3 were exposed by immersion to dosages of 6.2, 12.5, and 25 mg/mL of the ethanolic concentrate of A. oleracea, respectively, diluting with 50% ethanol and DMSO. Control 1 and Control 2 were treated with distilled water and ethanol 50% with dimethyl sulfoxide (DMSO) 1%, respectively. After exposure, the males were dissected for the removal of the reproductive system and subjected to routine histological analysis with HE staining and histochemical techniques (PAS for the detection of neutral polysaccharides and Bromo phenol blue to detect total proteins). After exposure, the males undergo standard histological study using HE staining and histochemical methods after being anatomized to detach reproductive organs. The glandular complex cells of those exposed exhibited modifications, and these modifications were dose-responsive and greater in severity with enhancing the extract concentration. The results of this research exhibited the ethanolic extract of A. oleracea 's cytotoxic properties on the reproductive organs of A. cajennenses at doses of 6.2, 12.5, and 25 mg/mL [59]. This study's goal was to assess whether providing zebrafish with both an oral and submerged dose of hydroethanolic extract of A. oleracea flowers would lead to acute toxicity in the animals. Gills, the liver, the gut, and the kidney were examined behaviorally and histopathologically. Within the histopathological investigation, it was demonstrated that oral EHFAo concentrations of 88.915, 199.53, and 281.83 mg/kg caused tissue damage that impaired the organism's regular function, with the gut being most severely impacted. The histological study of the therapy by immersion revealed that the most hazardous EHFAo concentrations were 300, 350, and 400 μg/L, which most severely afflicted the gill tissue. It is, therefore, possible to conclude that, based on the dose, the principal phytochemical substance of EHFAo, spilanthol, might cause histopathological lesions in the various organs mentioned throughout this research by taking into account the data observed and the medicinal value of this substance [60].
6 Limitations and challenges
While there are several studies on the pharmacological effects of Acmella oleracea, the majority are based on in vitro and animal models. There is a need for clinical trials to validate these findings in human populations to ensure safety and efficacy for medical use. The chemical composition of Acmella oleracea can vary significantly depending on geographical location, cultivation practices, and environmental factors, which may affect the consistency and predictability of therapeutic outcomes. Although generally considered safe, there are limited studies on the long-term toxicity and side effects of Acmella oleracea. Further research is needed to establish safe dosage guidelines and identify any potential adverse effects, especially with prolonged use. There is minimal information on how Acmella oleracea interacts with other medications or its contraindications. This is critical for integrating it into conventional treatment regimens without compromising patient safety. Ensuring the standardization of extracts and active compounds poses a significant challenge. Quality control measures must guarantee the therapeutic potency and purity of herbal formulations.
7 Conclusion and future directions
Acmella oleracea, commonly known as the toothache plant, is a prominent herb in traditional medicine, particularly in India and South America, where it is most commonly used to treat toothaches. This plant demonstrates a diverse range of pharmacological activities, including antioxidant, anti-inflammatory, antinociceptive, antimicrobial, antifungal, diuretic, vasorelaxant, and immunomodulatory properties. These effects are largely attributed to key bioactive compounds such as spilanthol, phenolics, flavonoids, coumarins, and triterpenoids. Numerous studies have shown that the whole plant and its various extracts possess significant therapeutic potential. Despite these promising findings, current research on A. oleracea remains largely preclinical. Conducting well-designed clinical trials is imperative to validate the therapeutic efficacy and safety of A. oleracea in human populations. These trials should explore the plant's effects on various health conditions and assess optimal dosages and formulations. Developing standardized extraction methods is crucial to ensure consistent quality and potency of the bioactive compounds in A. oleracea extracts. Standardization will facilitate the reproducibility of research findings and the development of reliable therapeutic products. Further research is needed to elucidate the molecular mechanisms underlying the pharmacological effects of A. oleracea. This includes identifying the specific pathways and receptors involved in its diverse biological activities. Understanding these mechanisms will provide deeper insights into the plant's therapeutic potential and help optimize its use in medical applications. Comprehensive toxicological evaluations are necessary to assess the long-term safety and potential side effects of A. oleracea extracts and compounds. These studies should include assessments of chronic toxicity, genotoxicity, and carcinogenicity. Investigating the impact of environmental factors on the phytochemical composition of A. oleracea is important. Variations in growing conditions, soil types, and climatic factors can affect the concentration of bioactive compounds. Understanding this variability will enable the development of cultivation and harvesting strategies that maximize the medicinal properties of the plant. Research should also focus on developing various formulations of A. oleracea extracts, such as capsules, topical ointments, and oral solutions, to enhance their bioavailability and therapeutic efficacy. These formulations should be tested for their pharmacokinetics, stability, and effectiveness in delivering the desired therapeutic outcomes. Exploring the potential synergistic effects of A. oleracea with other medicinal plants or conventional drugs could provide new insights into its use in combination therapies. Additionally, investigating its applications in various fields, such as dentistry, dermatology, and infectious diseases, could expand its therapeutic scope. By addressing these future research directions, A. oleracea can be more effectively integrated into modern medicine, bridging traditional knowledge with contemporary biomedical science. This comprehensive approach will ensure the development of safe, effective, and standardized therapeutic products derived from A. oleracea, ultimately benefiting public health. The rationale behind conducting this comprehensive review is to convey all relevant scientific information to researchers and promote further studies to validate the beneficial impacts of this plant.
Data availability
No datasets were generated or analysed during the current study.
References
Martins C, Melo M, Honório I, D’Ávila V, Carvalho Júnior W. Morphological and agronomic characterization of Jambu (Spilanthes oleracea L.) accessions under the conditions of North Minas Gerais State. Brazil Revista Brasileira de Plantas Medicinais. 2012;14:410–3.
Šamec D, Urlić B, Salopek-Sondi B. Kale (Brassica oleracea var. acephala) as a superfood: Review of the scientific evidence behind the statement. Crit Rev Food Sci Nutr. 2019;59(15):2411–22. https://doi.org/10.1080/10408398.2018.1454400.
Zhou Y-X, Xin H-L, Rahman K, Wang S-J, Peng C. Portulaca oleracea L.: a review of phytochemistry and pharmacological effects. BioMed Res Int. 2015;1:925631. https://doi.org/10.1155/2015/925631.
Dogara AM, Al-Zahrani AA. Phytochemistry and pharmacological activities of Adansonia digitata L.: a review. S Afr J Bot. 2024;171:368–88. https://doi.org/10.1016/j.sajb.2024.06.026.
Uthpala T, Navaratne S. Acmella oleracea plant; identification, applications and use as an emerging food source–review. Food Rev Intl. 2021;37(4):399–414.
Dubey S, Maity S, Singh M, Saraf SA, Saha S. Phytochemistry, pharmacology and toxicology of Spilanthes acmella: a review. Adv Pharmacol Pharmaceut Sci. 2013;5:693.
Silveira N, Sandjo LP, Biavatti MW. Spilanthol-containing products: A patent review (1996–2016). Trends Food Sci Technol. 2018;74:107–11.
Prachayasittikul V, Prachayasittikul S, Ruchirawat S, Prachayasittikul V. High therapeutic potential of Spilanthes acmella: a review. EXCLI J. 2013;12:291.
Nipate SS, Tiwari AH. Antioxidant and immunomodulatory properties of Spilanthes oleracea with potential effect in chronic fatigue syndrome infirmity. Journal of Ayurveda and Integrative Medicine. 2020;11(2):124–30.
da Silva SP, Fernandes JAL, Santos AS, Ferreira NR. Jambu flower extract (Acmella oleracea) increases the antioxidant potential of beer with a reduced alcohol content. Plants. 2023;12(8):1581.
Wongsawatkul O, Prachayasittikul S, Isarankura-Na-Ayudhya C, Satayavivad J, Ruchirawat S, Prachayasittikul V. Vasorelaxant and antioxidant activities of Spilanthes acmella Murr. Int J Mol Sci. 2008;9(12):2724–44.
Shivananda S, Doddawad VG, Vidya C, Sanjay C, Shetty A, Kalgeri SH, Bhat HHK. The use of spilanthes acmella extracts as a local anesthetic agent in dentistry: a review. Int J Nutr Pharmacol Neurol Dis. 2023;13(1):3–8.
Gilbert B, Favoreto R (2010) Acmella oleracea (L.) RK Jansen (Asteraceae) Jambu.
Ramsewak RS, Erickson AJ, Nair MG. Bioactive N-isobutylamides from the flower buds of Spilanthes acmella. Phytochemistry. 1999;51(6):729–32.
Gouvêa SM, Carvalho GA, Fidelis EG, Ribeiro AV, Farias ES, Picanço MC. Effects of paracress (Acmella oleracea) extracts on the aphids Myzus persicae and Lipaphis erysimi and two natural enemies. Ind Crops Prod. 2019;128:399–404.
Fabri RL, Freitas JC, Lemos AS, Campos LM, Diniz IO, Pinto NC, Silva TP, Palazzi C, Marchesini P, Monteiro C. Spilanthol as a promising antifungal alkylamide for the treatment of vulvovaginal candidiasis. Med Mycol. 2021;59(12):1210–24.
Santana de Freitas-Blanco V, Franz-Montan M, Groppo FC, de Carvalho JE, Figueira GM, Serpe L, Oliveira Sousa IM, Guilherme Damasio VA, Yamane LT, de Paula E. Development and evaluation of a novel mucoadhesive film containing Acmella oleracea extract for oral mucosa topical anesthesia. PLoS ONE. 2016;11(9):e0162850.
Cheng Y-B, Liu RH, Ho M-C, Wu T-Y, Chen C-Y, Lo I-W, Hou M-F, Yuan S-S, Wu Y-C, Chang F-R. Alkylamides of Acmella oleracea. Molecules. 2015;20(4):6970–7.
Rondanelli M, Fossari F, Vecchio V, Braschi V, Riva A, Allegrini P, Petrangolini G, Iannello G, Faliva MA, Peroni G. Acmella oleracea for pain management. Fitoterapia. 2020;140:104419.
Nascimento AM, de Souza LM, Baggio CH, Werner MFdP, Maria-Ferreira D, da Silva LM, Sassaki GL, Gorin PA, Iacomini M, Cipriani TR. Gastroprotective effect and structure of a rhamnogalacturonan from Acmella oleracea. Phytochemistry. 2013;85:137–42.
Payum T. Phytoconstituents of Spilanthes oleracea linn. shoot: a medicinal food herb used among tribal people of Arunchal Pradesh, India. Int J Pharm Sci Res. 2017;8(10):4381–7.
Ley J, Blings M, Krammer G, Reinders G, Schmidt C-O, Bertram H-J. Isolation and synthesis of acmellonate, a new unsaturated long chain 2-ketol ester from Spilanthes acmella. Nat Prod Res. 2006;20(9):798–804.
Lalthanpuii PB, Lalchhandama K. Chemical composition and broad-spectrum anthelmintic activity of a cultivar of toothache plant, Acmella oleracea, from Mizoram, India. Pharm Biol. 2020;58(1):393–9.
Rondanelli M, Fossari F, Vecchio V, Braschi V, Riva A, Allegrini P, Petrangolini G, Iannello G, Faliva MA, Peroni G, Nichetti M, Gasparri C, Spadaccini D, Infantino V, Mustafa S, Alalwan T, Perna S. Acmella oleracea for pain management. Fitoterapia. 2020;140:104419. https://doi.org/10.1016/j.fitote.2019.104419.
Spinozzi E, Ferrati M, Baldassarri C, Cappellacci L, Marmugi M, Caselli A, Benelli G, Maggi F, Petrelli R. A review of the chemistry and biological activities of Acmella oleracea (“jambù”, Asteraceae), with a view to the development of bioinsecticides and acaricides. Plants (Basel). 2022;11(20):25. https://doi.org/10.3390/plants11202721.
Mo Y-n, Cheng F, Yang Z, Shang X-f, Liang J-p, Shang R-f, Hao B-c, Wang X-h, Zhang H-j, Wali A. Antioxidant activity and the potential mechanism of the fruit from Ailanthus altissima swingle. Front Vet Sci. 2021;8:784898.
Azzini E, Peña-Corona SI, Hernández-Parra H, Chandran D, Saleena LAK, Sawikr Y, Peluso I, Dhumal S, Kumar M, Leyva-Gómez G, Martorell M, Sharifi-Rad J, Calina D. Neuroprotective and anti-inflammatory effects of curcumin in Alzheimer’s disease: targeting neuroinflammation strategies. Phytother Res. 2024;38(6):3169–89. https://doi.org/10.1002/ptr.8200.
Yashin A, Yashin Y, Xia X, Nemzer B. Antioxidant activity of spices and their impact on human health: a review. Antioxidants. 2017;6(3):70.
Sharifi-Rad J, Quispe C, Durazzo A, Lucarini M, Souto EB, Santini A, Imran M, Moussa AY, Mostafa NM, El-Shazly M, Sener B, Schoebitz M, Martorell M, Dey A, Calina D, Cruz-Martins N. Resveratrol’ biotechnological applications: enlightening its antimicrobial and antioxidant properties. J Herbal Med. 2022;2:32. https://doi.org/10.1016/j.hermed.2022.100550.
Weintraub L, Naftzger T, Parr T, Henning S, Soendergaard M. Antioxidant activity and antiproliferative effects of Acmella alba, Acmella oleracea, and Acmella calirrhiza. FASEB J. 2020;34(S1):1–1.
Lalthanpuii P, Lalruatfela B, Vanlaldinpuia K, Lalremsanga H, Lalchhandama K. Antioxidant and cytotoxic properties of Acmella oleracea. Med Plants-Int J Phytomed Related Ind. 2018;10(4):353–8.
Tanwer BS, Choudhary R, Vijayvergia R. In vitro and in vivo comparative study of primary metabolites and antioxidant activity in Spilanthes acmella Murr. Int J Biotechnol Biochem. 2010;6(5):819–25.
Afridi B, Khan H, Akkol EK, Aschner M. Pain perception and management: where do we stand? Curr Mol Pharmacol. 2021;14(5):678–88. https://doi.org/10.2174/1874467213666200611142438.
Cheng JC, Erpelding N, Kucyi A, DeSouza DD, Davis KD. Individual differences in temporal summation of pain reflect pronociceptive and antinociceptive brain structure and function. J Neurosci. 2015;35(26):9689–700.
Tracey I, Dunckley P. Importance of anti-and pro-nociceptive mechanisms in human disease. Gut. 2004;53(11):1553–5.
Yien RMK, Gomes ACC, Goetze Fiorot R, Miranda ALP, Neves GA, Andrade BdS, Costa FN, Tributino JLM, Simas NK. Alkylamides from Acmella oleracea: antinociceptive effect and molecular docking with cannabinoid and TRPV1 receptors. Nat Prod Res. 2023;37(18):3136–44.
Dallazen JL, Maria-Ferreira D, da Luz BB, Nascimento AM, Cipriani TR, de Souza LM, Glugoski LP, Silva BJG, Geppetti P, de Paula Werner MF. Distinct mechanisms underlying local antinociceptive and pronociceptive effects of natural alkylamides from Acmella oleracea compared to synthetic isobutylalkyl amide. Fitoterapia. 2018;131:225–35.
Global mortality associated with 33 bacterial pathogens in,. a systematic analysis for the Global Burden of Disease Study 2019 (2022). Lancet. 2019;400(10369):2221–48. https://doi.org/10.1016/s0140-6736(22)02185-7.
Di Martino P. Antimicrobial agents and microbial ecology. AIMS Microbiol. 2022;8(1):1.
Alam MM, Akash SR (2023) In vitro pharmacological activities of methanol extract of acmella oleracea leaves: a variety grown in Dhaka, Bangladesh.
Peretti P, Rodrigues ET, De Souza Junior BM, Bezerra RM, Fernandez EG, de Sousa FFO, Pinheiro MT. Spilanthol content of Acmella oleracea subtypes and their bactericide and antibiofilm activities against Streptococcus mutans. S Afr J Bot. 2021;143:17–24.
Uthpala T, Munasinghe H, Peiris L, Navaratne S. Evaluation of antimicrobial potential and phytochemicals in Acmella (A. oleracea) flower pod extracts subjected to different drying techniques. J Food Process Preserv. 2021;45(6):e15570.
de Alcantara BN, Kobayashi YT, Barroso KF, da Silva IDR, de Almeida MB, Barbosa WLR. Pharmacognostic analyses and evaluation of the in vitro antimicrobial activity of Acmella oleracea (L.) RK Jansen (Jambu) floral extract and fractions. J Med Plants Res. 2015;9(4):91–6.
Thompson TS, Sukesh K, Singh D. A study on the antimicrobial effect of acmella oleraceae against dental caries bacteria. IJPSR. 2012;3(4):1194–7.
Murtaza G, Mukhtar M, Sarfraz A. A review: antifungal potentials of medicinal plants. J Bioresource Manag. 2015;2(2):4.
Sathyaprasad S, Jose BK, Chandra HS. Antimicrobial and antifungal efficacy of Spilanthes acmella as an intracanal medicament in comparison to calcium hydroxide: an in vitro study. Indian J Dent Res. 2015;26(5):528.
Khatoon R, Jahan N, Ahmad S, Shahzad A. In vitro evaluation of antifungal activity of aerial parts of medicinal plants Balanites aegyptiaca Del. and Spilanthes acmella Murr. J Appl Pharm Sci. 2014;4(1):123–7.
Amin R, Thalluri C, Docea AO, Sharifi-Rad J, Calina D. Therapeutic potential of cranberry for kidney health and diseases. eFood. 2022;3(5):e33.
Kehrenberg MC, Bachmann HS. Diuretics: a contemporary pharmacological classification? Naunyn Schmiedebergs Arch Pharmacol. 2022;395(6):619–27.
Ratnasooriya W, Pieris K, Samaratunga U, Jayakody J. Diuretic activity of Spilanthes acmella flowers in rats. J Ethnopharmacol. 2004;91(2–3):317–20.
Yadav R, Kharya MD, Yadav N, Savadi R. Diuretic activity of Spilanthes acmella Murr. leaves extract in rats. Int J Res Pharm Chem. 2011;1(1):57–61.
Gallo G, Volpe M, Savoia C. Endothelial dysfunction in hypertension: current concepts and clinical implications. Front Med (Lausanne). 2021;8:798958. https://doi.org/10.3389/fmed.2021.798958.
Ma J, Li Y, Yang X, Liu K, Zhang X, Zuo X, Ye R, Wang Z, Shi R, Meng Q, Chen X. Signaling pathways in vascular function and hypertension: molecular mechanisms and therapeutic interventions. Signal Transduct Target Ther. 2023;8(1):168. https://doi.org/10.1038/s41392-023-01430-7.
Adhikari D, Gong D-S, Oh SH, Sung EH, Lee SO, Kim D-W, Oak M-H, Kim HJ. Vasorelaxant effect of Boesenbergia rotunda and its active ingredients on an isolated coronary artery. Plants. 2020;9(12):1688.
Vangoori Y, Dakshinamoorthi A, Kavimani S. Prominent pancreatic lipase inhibition and free radical scavenging activity of a myristica fragrans ethanolic extract in vitro. Maedica. 2019;14(3):254.
Ekanem A, Wang M, Simon J, Moreno D. Antiobesity properties of two African plants (Afromomum meleguetta and Spilanthes acmella) by pancreatic lipase inhibition. Phytother Res. 2007;21(12):1253–5.
Bascones-Martinez A, Mattila R, Gomez-Font R, Meurman JH. Immunomodulatory drugs: ORAL and systemic adverse effects. Medicina Oral Patologia Oral y Cirugia Bucal. 2014;19(1):e24.
de Andrade LC, Rotta IW, Chaves SR, Uchida DT, Gazim ZC, Ferreira FBP, Jacomassi E, Boleta-Ceranto DdCF. Effectiveness of Acmella oleracea for topical anesthesia on buccal mucosa. Revista Odonto Ciência. 2013;28(3):61–5.
Anholeto LA, de Oliveira PR, Rodrigues RAF, Yamane LT, de Carvalho Castro KN, Ferreira ARF, Camargo-Mathias MI. Toxic action of Acmella oleracea extract on the male reproductive system of Amblyomma cajennense ticks. Vet Parasitol. 2017;244:164–71.
Custodio de Souza G, Ribeiro Dias, da Silva I, Duarte Viana M, Costa de Melo N, Sánchez-Ortiz BL, Rebelo Maia, de Oliveira M, Ramos Barbosa WL, Maciel Ferreira I, Tavares Carvalho JC. Acute toxicity of the hydroethanolic extract of the flowers of Acmella oleracea L. in zebrafish (Danio rerio): behavioral and histopathological studies. Pharmaceuticals. 2019;12(4):173.
Acknowledgements
The authors would like to express their gratitude to Dr. IZ, MD, RCP London, Basildon University Hospital UK, for providing professional English editing of this manuscript and for editorial support.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
MAA, MSB, SM, RC, CS, MAS, PR, ŽR, JS-R, DC, MAKS, MT made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas that is, revising or critically reviewing the article; giving final approval of the version to be published; agreeing on the journal to which the article has been submitted; and confirming to be accountable for all aspects of the work. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aktar, M.A., Bhuia, M.S., Molla, S. et al. Pharmacological and phytochemical review of Acmella oleracea: a comprehensive analysis of its therapeutic potential. Discov Appl Sci 6, 412 (2024). https://doi.org/10.1007/s42452-024-06108-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-024-06108-5