Abstract
Biochar has a large specific surface area, well-developed pore structure, abundant surface functional groups, and superior nutrient supply capacity, which is widely available and environmentally friendly with its advantages in waste resource utilization, heavy metal(loid) remediation, and carbon storage. This review focuses on the interactions between biochar (including raw biochar, functional biochar (modified/ engineered/ designer biochar), and composite biochar) and rhizosphere during the remediation of soil contaminated with heavy metal(loid)s (Pb, As, Cd, Hg, Co, Cu, Ni, Zn, Cr, etc.) and the effects of these interactions on the microbial communities and root exudates (enzymes and low-molecular-weight organic acids (LMWOAs)). In terms of microorganisms, biochar affects the composition, diversity, and structure of microbial communities through the supply of nutrients, provision of microbial colonization sites, immobilization of heavy metal(loid)s, and introduction of exogenous microorganisms. With regard to root exudates, biochar provides electron transfer support between the microorganisms and exudates, regulates the secretion of enzymes to resist the oxidative stress stimulated by heavy metal(loid)s, ameliorates rhizosphere acidification caused by LMWOAs, and promotes the activity of soil enzymes. The roles and mechanisms of biochar on rhizosphere soils are discussed, as well as the challenges of biochar in the remediation of heavy metal(loid)-contaminated soils, and the issues that need to be addressed in future research are foreseen.
Graphical Abstract

Highlights
-
The interactions between biochar and rhizosphere in the remediation of soil with heavy metal(loid)s have been focused
-
Biochar affects rhizosphere microorganisms by changing population diversity and community structure
-
Biochar regulates root exudates secretion and promotes soil enzyme activity in contaminated soil
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Heavy metals (e.g., lead (Pb), cadmium (Cd), chromium (Cr), zinc (Zn), mercury (Hg), copper (Cu), cobalt (Co), and nickel (Ni)) and metalloids (e.g., arsenic (As)), henceforth collectively referred to as heavy metal(loid)s, are caused by both natural activities and human-induced activities (such as mining and smelting, agricultural activity, as well as urban and industrial emissions) (Yuan et al. 2023a; Hou et al. 2020; Narayanan and Ma 2022). The leaching of large amounts of heavy metal(loid)s into soil and water can accumulate in living organisms through the food chain, and cause irreversible damage to the human body due to the non-biodegradable properties of heavy metal(loid)s (Gao et al. 2022; O’Connor et al. 2020). In addition, once the content of heavy metal(loid)s in the soil exceeds the self-purification capacity of the soil, the ecological balance of the soil will be disrupted, such as the death of beneficial organisms and microorganisms will lead to the reduction of soil biota, and severe heavy metal(loid) contamination will lead to the loss of soil production capacity and agricultural use value (Lin et al. 2022). Therefore, appropriate remediation of heavy metal(loid)-contaminated soils is imperative. For this purpose, a number of remediation strategies, such as bioremediation, physical remediation, and chemical remediation, have indeed been developed based on the mechanism of mobilization or immobilization of heavy metal(loid)s (Xiang et al. 2022). However, these strategies are usually expensive and require large amounts of energy and chemicals, as well as potential secondary contamination (Ghosh and Maiti 2021; Wang et al. 2020d). Therefore, nature-based remediation substances are emerging, which ought to be cost-effective and environmentally beneficial in remediation (Yuan et al. 2023b; Narayanan and Ma 2022).
Remediation of heavy metal(loid)s in soils with biochar is one such viable approach that is cost-effective and environmentally beneficial because biochar can be obtained from abundant and widely available waste biomass resources, which mainly include agricultural and garden waste, forest waste, industrial waste, and livestock waste (Feng et al. 2021; Khan et al. 2020; Li et al. 2019c; Novak et al. 2019). Furthermore, the 196 parties to the United Nations Framework Convention on Climate Change have agreed to achieve a balance in the anthropogenic greenhouse-gas (GHG) budget between 2050 and 2100, and this objective necessitates that both agriculture and industry strive towards achieving zero emissions or actively removing GHGs from the atmosphere (Williamson 2016). Yang et al. (2021a) showed that Converting 1 ton of agricultural waste into biochar can sequester over 920 kg CO2-equivalent. Each hectare of soil amended with biochar saves 91 metric tons of CO2-equivalent through the soil conservation effect, with 13 metric tons of CO2-equivalent being sequestered in the soil (Lan et al. 2021; Latawiec et al. 2019). Biochar application as a soil amendment is a critical technology that combines zero carbon emissions with co-benefits for both the soil environment and agricultural yields (Buss et al. 2022).
The carbon (C), nitrogen (N), potassium (K), calcium (Ca), phosphorus (P), magnesium (Mg), and other organic matter contained in biochar can increase soil fertility, which provides nutrients for the growth of plants, soil animals, and microorganisms (Chen et al. 2016; El-Naggar et al. 2019; Wang et al. 2020b). The excellent pore structure of biochar has the capacity to adsorb soil heavy metal(loid)s, improve soil physicochemical properties, retain soil moisture, and provide sites for microbial colonization (El-Naggar et al. 2019; Wang et al. 2021a). Moreover, the surface functional groups of biochar are abundant and capable of transforming free heavy metal(loid) ions into less bioavailable forms through precipitation, coordination, and complexation (Alam et al. 2018; Chen et al. 2016; Yuan et al. 2021b). In addition to raw biochar, functional (modified/engineered/designer) biochar, which is commonly used to indicate application-oriented, result -based synthesis or modification of biochar with specific functions, can have more specific properties attached to it through various modifications (e.g., nano-, iron (Fe)-, aluminum (Al)-, manganese (Mn)-, nitrogen (N)-, sulfur (S)-, and acid/base-modification) that can enhance the immobilization of heavy metal(loid)s in soil (Hazrati et al. 2021; Liu et al. 2020b, 2022b; Zhu et al. 2020). Furthermore, co-composite biochar, which works synergistically through biochar and some remediation materials, such as biochar-compost, biochar-microorganisms, biochar-biomass waste, biochar-cement, and biochar-zeolite, has also been used to remediate the soil contaminated with heavy metal(loid) (Chuaphasuk and Prapagdee 2019; Risueno et al. 2021; Simiele et al. 2021; Zhu et al. 2022b). These biochar amendments (raw, functional, and co-composite biochar) enable the pre-remediation of heavily heavy metal(loid) contaminated soils that are difficult to remediate by bioremediation and provide favorable conditions for the survival and colonization of soil organisms, thus contributing to the establishment and recovery of soil ecosystems (Gao et al. 2022). Thus, using waste biomass to create biochar can remediate soil contaminated with heavy metal(loid)s while achieving multiple goals such as immobilizing heavy metal(loid)s, utilizing waste resources, and achieving net zero.
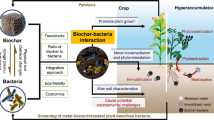
The rhizosphere system is the basis for plant-environment interactions, plant growth, and crop production (Sun et al. 2020). Microorganisms, including bacteria and fungi, as well as root exudates (such as enzymes, amino acids, organic acids, and sugars), are crucial for plant-soil interactions and rhizosphere function, performing important functions such as mobilizing and uptaking soil nutrients, exchanging information, and transferring energy (Gargallo-Garriga et al. 2018). The chemical and biological processes in the rhizosphere of heavy metal(loid) contaminated soil differ significantly from those in the bulk soil (non-rhizosphere soil) because of the influence of rhizosphere microorganisms and exudates on the consumption, accumulation, or immobilization of heavy metal(loid)s (Sterckeman et al. 2005; Wang et al. 2002; Zhang et al. 2022). The rhizosphere, as the first interface between the soil and the root, is the major battlefield where biochar plays its role in soils contaminated with heavy metal(loid)s (Zhang et al. 2022). The interactions between biochar and the rhizosphere include (1) direct interactions, such as the direct contact between biochar and plant roots, the enrichment of rhizosphere microorganisms on biochar, and the adsorption of root exudates on biochar; and (2) indirect interactions, where biochar causes changes in soil physicochemical properties that result in the immobilization of heavy metal(loid)s, changes in microbial communities and activities, enzyme species and activities, and types and concentrations of root exudates (Jones et al. 2009; Lehmann et al. 2015; Prendergast-Miller et al. 2014). In addition, rhizosphere-biochar interactions are also influenced by soil properties such as water-holding capacity, pH, bulk density, minerals, and heavy metal(loid)s (Jones et al. 2012).
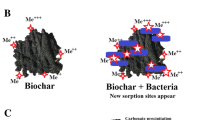
Biochar has been shown to regulate the rhizosphere environment of plants. To obtain a comprehensive overview of the present application of biochar in soil contaminated with heavy metal(loid)s and its effects on the rhizosphere, we collected and succinctly evaluated a total of 1069 papers from the “Web of Science” database using the keywords (topic) “heavy metal or trace element or mercury or lead or arsenic or chromium or cadmium or zinc or cobalt or copper or nickel” and “soil” and “remediat* or immobili* or stabili*” and “root or rhizosphere or inter-root” and “biochar” in the last 15 years since 2009. The number of papers on the effect of biochar on the rhizosphere increased from 1 in 2009 to 243 in 2022, which represented an explosion of attention to biochar and rhizosphere in the soil contaminated with heavy metal(loid)s (Fig. 1a). Scientometric visualization of the top 443 keywords showed that (a) the reduction of bioavailability of heavy metal(loid)s through the immobilization and accumulation of biochar has been extensively studied in soil remediation, and (b) studies on biochar and rhizosphere have focused on microbial activity and abundance, enzymes, exudates, and nutrients in the soil contaminated with heavy metal(loid)s (Fig. 1b). Although a very large number of research articles have examined the remediation of heavy metal contaminated soils by biochar and the effects on rhizosphere microorganisms and secretions, there is a lack of a systematic summary. Therefore, this review systematically summarizes the following aspects of the interactions between biochar amendments and the rhizosphere in heavy metal(loid)-contaminated soils: 1) the biochar amendments and its effect on heavy metal(loid) contaminated soil; 2) the effects of biochar on rhizosphere microorganisms; and 3) the interactions between biochar and root exudates. The roles and mechanisms of biochar on rhizosphere soils were discussed, the effects of biochar in the rhizosphere were critically analyzed, and the challenges of biochar in the remediation of heavy metal(loid) contaminated soils and the issues that need to be addressed in future research were foreseen.
a The number of published studies on the effect of biochar on the rhizosphere in the remediation of heavy metal(loid)-contaminated soils, according to Web of Science-Clarivate Analytics, 15.03.2023; search from 2009 to 2022. b Scientometric visualization of the top 443 keywords of 1069 publications from 2009 to 2023. The search content Web of Science is “(((((((((((((((((ALL = ((heavy metal*))) OR ALL = ((trace element*))) OR ALL = (arsenic)) OR ALL = (mercury)) OR ALL = (chromium)) OR ALL = (cadmium)) OR ALL = (zinc)) OR ALL = (cobalt)) OR ALL = (copper)) OR ALL = (nickel)) AND ALL = ((Soil*))) AND ALL = ((remediat*))) OR ALL = ((immobili*))) OR ALL = ((stabili*))) AND ALL = (root)) OR ALL = (rhizosphere)) OR ALL = (inter-root)) AND ALL = (biochar)”. The data was collected on 15.03.2023. Using the Web of Science Core Collection as the database, the collected data was visualized using VOSviewer, where each circle represents a keyword and its size corresponds to the frequency of appearance for pairs of keywords in the publication
2 Biochar amendments and their modifications
Theoretically, almost all types of organic matter can be converted into biochar, and it has been found that agricultural/garden waste, forest waste, industrial waste, and livestock waste have been mainly used as feedstock to obtain biochar in the present studies (Ghosh and Maiti 2021; Lu et al. 2022; Wang et al. 2021b). Biochar from different feedstock displays various physical and chemical properties such as pH, functional groups, specific surface area, pore structure, cation exchange capacity, and mineral element concentrations (Sizmur et al. 2017; Zhang et al. 2018b). In addition, in order to obtain specific remediation functions for the target contaminants and to improve the remediation effect of raw biochar, functional biochar and co-composite biochar have been developed for application in remediation of soil contaminated with heavy metal(loid)s (e.g., the Fe-loaded biochar has a significant remediation effect on As, and the OH-modified biochar significantly improves the remediation effect on Pb and Cd) (Gasco et al. 2019; Lebrun et al. 2018; Martins et al. 2018; Su et al. 2021). In this study, the modifications of functional biochar are classified as non-metallic modification (e.g., N, P, S, and thiol modification), single/multi-metallic modification (e.g., Fe-, Mn-, Co-, Ni-, Fe-/Zn-, Fe-/Mn/Ce-modification), non-metallic/metallic modification (e.g., S/Fe-modification), acid and base modification, and other modifications (e.g., nanomodification, zeolite modification). Moreover, co-composite biochar includes biochar combined with compost, biochar combined with sewage sludge, biochar combined with lime and fertilizer, and biochar combined with microorganisms, etc.
Non-metallic modification of biochar, such as grafting of N-, P-, and S-containing functional groups, is an effective strategy for enhancing heavy metal(loid) adsorption capacity. Some studies have found that thiol groups have high affinity for heavy metal(loid)s (e.g., Hg2+, Pb2+, Cd2+, Cu2+, and Zn2+) (Chai et al. 2010; Zhang et al. 2012). Xia et al. (2019) modified biochar with sulfhydryl groups using 3-mercaptopropyltrimethoxysilane and found that thiol-modified biochar showed a significant increase in adsorption capacity of Hg2+. Fan et al. (2020) modified rice husk biochar with thiol groups by β-mercaptoethanol esterification reaction, and found that thiol-modified biochar effectively removed Pb2+ and Cd2+ from soil by surface complexation.
Single-metallic modification of biochar with Fe, Mn, Co, or Ni results in biochar that obtains magnetic properties and can be easily recovered by external magnetic fields (Wang et al. 2020c). Some studies have found that the introduction of iron oxides into biochar promotes the formation of inner-sphere complexes, which leads to a decrease in the mobility and bioavailability of heavy metal(loid)s and promotes plant growth (Wan et al. 2020; Wu et al. 2020). In addition, multi-metallic modification of biochar can form layered double hydroxides on the surface of biochar, which is a type of ionic layered compound with positively charged metal hydroxide layers and exchangeable negative ions with strong anion exchange capacity, and such materials have strong remediation effects on anionic heavy metal(loid)s (such as AsO43−, H2AsO3−) in soils (Meili et al. 2019). Furthermore, the effect of metal-modified biochar on the soil rhizosphere may be due to the fact that these metals provide essential elements for the growth of rhizosphere plants and microorganisms (Lu et al. 2018).
Biochar modified by acidification usually has higher specific surface area and pore volume as well as more acid-binding sites for adsorption of contaminants and hydrophobic groups (e.g., lactone, phenolic, and carbonyl functional groups) (Ghorbani et al. 2022; Li et al. 2014). In addition, acid modified biochar can increase the concentration of water-soluble K, Ca, Mg, and Fe, which contribute to soil nutrients and promotes plant growth (Wang et al. 2020c). However, when the biochar itself contains heavy metal(loid)s, acid modification may not be a suitable modification method because the heavy metals will be washed out during the acid modification process, increasing the bioavailability of the heavy metals (Wang et al. 2020c). Base modification of biochar can increase the specific surface area and the content of oxygenated functional groups (e.g., hydroxyl, carboxyl, carbonyl, and ether) of raw biochar (Wang and Wang 2019). Wang et al. (2020a) found that low concentrations of KOH-modified biochar were beneficial for the stabilization of Pb and Cd in soil. However, acid- and alkali-modified biochar ought to be neutralized by a large amount of water after application to the soil in order to avoid excessive acid and base that may cause negative impacts on the environment.
In addition, co-composite biochar contributes to play a better role of biochar itself and synergizes with microorganisms, lime, compost, and other materials to significantly improve the biological, chemical, and physical characteristics of soil (Haider et al. 2021). For example, in order to improve the properties and minimize the defects of the remediation amendments, Forján et al. (2018) combined compost with biochar to increase the pH and total carbon of soil, and thus reduce the concentration of available Cu, Ni, Pb, and Zn. Furthermore, contaminated soil cannot support the rapid growth of microorganisms with remediation capacity for soil due to the biotoxicity of heavy metals, and nutrient deprivation and competition from native microorganisms have a negative impact on the growth and proliferation of exogenous microorganisms (Ma et al. 2020). Therefore, the combined application of microorganisms with biochar enables microorganisms with remediation capacity for heavy metals to have higher bioactivity, greater population density, better regeneration capacity, and higher resistance to environmental disturbances (Wei et al. 2022).
3 Effect of biochar amendment on the physicochemical properties of heavy metal(loid)-contaminated soil
3.1 The effect of biochar amendments on heavy metal(loid)s
Since heavy metals and metalloids exist as both anionic and cationic forms, the behavior of heavy metal(loid)s is closely related to the anionic and cationic charges present on the surface of biochar (Fijałkowska et al. 2021). When added to soil, biochar with a positive charge holds anionic heavy metal(loid)s (e.g., arsenate and arsenite) while biochar with a negative charge binds to the cationic fraction (e.g., Pb2+, Zn2+, and Cd2+) (Gupta et al. 2021). For cations, the mechanisms of biochar remediation of heavy metal(loid)s are mainly surface precipitation, adsorption, and surface complexation with reactive functional groups, while anionic heavy metal(loid)s (including Cr, Se, and As) are more mobile in alkaline soils than cationic heavy metal(loid)s, and they are less likely to be adsorbed by negatively charged soils (Gupta et al. 2021; Narayanan and Ma 2022). Therefore, improvements in soil properties by biochar, such as reduced or increased soil pH caused by the use of acid- or base-modified biochar, can further reduce the bioavailability of heavy metal(loid)s. In addition, the mobility of heavy metal(loid)s in soil is also affected by their redox potential, which is influenced by the oxidation-reduction state of the soil (Narayanan and Ma 2022). The study of Dong et al. (2017) showed that the presence of oxygen-containing active functional groups on the surface of the biochar allowed the conversion of Cr6+ to the less mobile Cr3+. The physicochemical properties of biochar depend on the feedstock, prepare and modify biochar, so it is necessary to screen the appropriate feedstock, preparation and modification of biochar according to the properties of heavy metal(loid)s in the soil, thus enabling biochar amendments to remediate specific types of soils. Table 1 summarizes the significant effects of biochar amendments on the remediation of heavy metal(loid)s in soil, and in general, the remediation mechanisms (Fig. 2b) of biochar amendments for heavy metal(loid)s mainly involve (1) electrostatic adsorption by π-π electron donor-acceptor interactions or electrostatic attraction between biochar amendments and cations/anions of heavy metals(loid)s (Yang et al. 2019); (2) ion exchange between cations on the surface of biochar (Ca2+, Mg2+, etc.) and heavy metal(loid) cations (Qiu et al. 2021), or anions on the surface of biochar (e.g., OH−, CO32− in layered double hydroxide-modified biochar) and heavy metal(loid) anions (AsO43−, H2AsO3−, etc.) (Dai et al. 2022); (3) complexation between abundant oxygen-containing functional groups (-COOH, C–OH, and -OH, etc.) on the surface of biochar amendments and heavy metal(loid) ions to form stable complexes (Vithanage et al. 2017); and (4) precipitation of heavy metal(loid)s with minerals on the surface of biochar to form insoluble precipitates (Pb(OH)2, Cd(OH)2, and As-bearing Fe mineral, etc.) (Ghosh and Maiti 2021; Palansooriya et al. 2022; Zhang et al. 2020b).
3.2 Effect of biochar amendments on soil physicochemical properties
Biochar amendments significantly affect the biophysical and chemical properties of the rhizosphere in soil contaminated with heavy metal(loid)s (Fig. 2a). Several studies have demonstrated that biochar amendments could improve soil health, which is beneficial for arid and degraded soils, especially for mining soils with low organic matter, nutrient content, and water-holding capacity (Malyan et al. 2021; Rodriguez-Franco and Page-Dumroese 2021). The various physical and chemical properties of biochar have different effects on heavy metal(loid) remediation and the soil fertility improvement (Sizmur et al. 2017; Zhang et al. 2018b). For example, Chen et al. (2022) investigated the effects of agricultural waste (peanut shell, corn straw, and wheat straw) and forest waste (pine chips) biochars on Pb-contaminated soil, which revealed that agricultural waste biochar had higher specific surface area and abundant surface oxygen functional group contents compared to forest waste biochar, and the soil acid-soluble Pb contents reduced by 49.51%, 50.71%, and 52.71% after 150 days of remediation with peanut shell, corn straw, and wheat straw biochars, respectively, which showed a better remediation capacity than that of pine chip biochar (37.30%). Research indicates that as pyrolysis temperature increases, the absorption peaks of O-H, C-O-C, C-O, and -CH2- on biochar surface decrease, with only peaks of C-O and C-C remaining on the aromatic ring of high-temperature biochar (Chen and Chen 2009). And these functional groups have a large number of hydrophilic and hydrophobic groups, and therefore, they are closely related to the water-holding capacity which is closely relevant to the growth and health of soil rhizosphere plants and microorganisms in the soil (Gao et al. 2022). Additionally, the well-developed pore structure and large specific surface area of biochar amendments promote water-holding capacity and provide a favorable habitat for rhizosphere microorganisms (Malyan et al. 2021; Palansooriya et al. 2019; Tomczyk et al. 2020). Additionally, mineral element concentrations in biochar, such as Mg, Fe, Na, Ca, P, and K, increase with pyrolysis temperature and are influenced by the type of feedstock used (Tomczyk et al. 2020). Saletnik et al. (2016) concluded that the maximum C and N contents (73.6% and 1.9%, respectively) were found at a pyrolysis temperature of 400 °C, while the highest P, K, and Mg contents (4.3 g/kg, 9.9 g/kg, and 2.8 g/kg, respectively) were found at 500 °C. Thus, the contribution of biochar obtained in different conditions to the nutrients required by soil plants and microorganisms is different. Nair et al. (2017) reported that the biochar stimulated carbon sequestration and humification processes through the increased carbon content, reduced bulk density, and improved water-holding capacity. Furthermore, biochar amendments are usually alkaline, which can regulate the pH of the rhizosphere soil, especially in acidic soils or acidified soils due to organic acid production by roots, and biochar amendments can also improve the fertility of the rhizosphere soil due to the abundant elements of C, N, P, and K, all of which provide favorable conditions for the growth of soil plants and microorganisms (Bashir et al. 2020; Jain et al. 2020; Lebrun et al. 2018). There are a number of review articles summarizing the application methods, effects, mechanisms, and microbial impacts of various types of biochar in diverse types of soil. In general, soils are categorized into two main types, namely, those contaminated with anionic heavy metal(loid)s (As and Cr) and those contaminated with cationic heavy metal(loid)s (Pb and Cd, etc.), and the selection of biochar depends on the type of soil as well as the properties of the biochar (Batool et al. 2022; Gong et al. 2022). Similarly, the effect of biochar on the soil rhizosphere depends on the plant species and the microbial species of the rhizosphere. However, although there are a number of research articles on the effects of biochar on soil rhizosphere, there is no definitive summary of which type of biochar is best suited for rhizosphere in different soils, plants, and cultivation strategies, which is a target that should be pursued in subsequent studies.
4 Effect of biochar amendments on rhizosphere microorganisms in heavy metal(loid)-contaminated soil
Soil microorganisms include bacteria, fungi, actinomycetes, archaea, algae, and protozoa, which are crucial for the genetic diversity of earth and the functioning of soil ecosystems (Bandara et al. 2019; Zhang et al. 2018d). Some microorganisms are highly resistant to heavy metal(loid)s, such as Pseudomonas, Delftia, Enterobacter, Arthrobacter, and Bacillus, which can reduce the mobility and bioavailability of heavy metal(loid)s by precipitation or uptake of them (Ma et al. 2020). In addition, the existence of larger microbial biomass itself can enhance the remediation efficiency of heavy metal(loid)s in contaminated soils through direct adsorption of heavy metal(loid)s and participation in redox processes (Chen et al. 2018a; Wang et al. 2019). The heavy metal(loid)s, together with nutrients, water, electron acceptors, and colonization sites are important factors which affect the growth and activity of rhizosphere microorganisms (Abdu et al. 2016; Giller et al. 2009; Lehmann et al. 2011). The addition of biochar amendments has both positive and negative effects on soil rhizosphere microorganisms, depending on the type of biochar amendments, the soil environment, and the heavy metal(loid)s. Negatively, biochar amendments may release some toxic compounds (such as potentially toxic elements, volatile organic compounds, or environmentally persistent free radicals) or mobilize anionic heavy metal(loid)s (such as Cr, As, and Sb) from soils (Bandara et al. 2019). On the positive side, biochar amendments can immobilize heavy metal(loid)s from soils, thereby reducing the bioavailability of heavy metal(loid)s, and providing electron shuttles, nutrients, and habitats for soil microorganisms.
4.1 Physical structure support
The population diversity and community structure of rhizosphere microorganisms in soil are closely associated with soil ecosystems (Li et al. 2016). The well-developed pore structure and large specific surface area of biochar provide colonization sites for rhizosphere microorganisms in soil contaminated with heavy metal(loid)s, whichand increases the enhances the enrichment of microorganisms, and increases the microbial abundance (Chaudhary et al. 2023; Kolb et al. 2009). Liu et al. (2018a) found that hydrochloric acid pickling and ultrasonication effectively removed or dissolved impurities in coconut shell biochar, which significantly increased the specific surface area and pore size and volume of biochar, and the counts of bacteria and fungi (especially Actinomyces) significantly increased in multi-metal (Cd, Ni, and Zn) contaminated soils after the addition of modified coconut shell biochar than the control group. In addition, biochar has been found to promote the colonization and enrichment of specific microbial strains (Chen et al. 2019; Wu et al. 2019a). Tu et al. (2020) investigated the remediation of Cd and Cu contaminated soil using maize biochar loaded with heavy metal(loid)s-tolerant strain (Pseudomonas sp.) and observed in scanning electron microscope images that Pseudomonas sp. attached well to the biochar, and most cells scattered or aggregated on the pore and surface of the biochar. However, the potential of biochar to serve as a colonization carrier for microorganisms is contingent on the specific microbial groups, physicochemical properties of biochar, and the soil conditions (Zhu et al. 2017). Different microbial groups respond differently to biochar, and the two most common mycorrhizal fungi (ectomycorrhizal and arbuscular) are usually protected by biochar whose internal pore structure may protect the mycelium from predators (Warnock et al. 2007). Mycorrhizal response is usually assessed by the measurement of root colonization (i.e., the abundance of fungal tissue) (Lehmann et al. 2011). The tip number and the formation rate of ectomycorrhizal infection increased by 19–157% in larch seedling roots, and the colonization of arbuscular increased from 5% to 20% (control) to 20%–40% of wheat roots after the addition of biochar (Lehmann et al. 2011; Makoto et al. 2009). The study by Quilliam et al. (2013) observed that after being buried for three years, biochar had only sparse concentrations of attached microorganisms. This was likely due to high levels of mineral salts and contaminants such as polycyclic aromatic hydrocarbons in the soil, which may hinder microbial colonization.
4.2 Nutrient and micronutrient supply
The organic matter and mineral elements contained in biochar amendments provide nutrients such as C and N sources, and essential elements for the metabolism of cells, the composition of cellular proteins and nucleic acids, as well as the composition of cellular structures of rhizosphere microorganisms in soil contaminated with heavy metal(loid)s (Bruins et al. 2000). It has been reported that long-term exposure of soils to heavy metal(loid) contamination (As, Pb, Cd, Cr, Ni, Cu, V, and Zn) resulted in a decrease in soil basal respiration and microbial biomass carbon (Parelho et al. 2016). The application of biochar amendments reduced the microbial stress caused by high concentrations of heavy metal(loid)s and contributed to an increase in the number of soil bacteria and fungi in soil contaminated with heavy metal(loid)s (Parelho et al. 2016; Zornoza et al. 2016). Haddad and Lemanowicz (2021) studied the effect of biochar application on soybean rhizosphere microorganisms in Cd, Pb, and Ni contaminated soils and found that biochar overcame the inhibitory effect of heavy metal(loid)s on enzyme activities (arginase and urease), and provided more stable and degradable C sources, thereby promoting the growth of soil fungi (Vries et al. 2006; Orwin and Wardle 2004). The nutrient structure of biochar is significantly impacted by the pyrolysis temperature. Increasing the pyrolysis temperature of biochar generally results in an increase in its aromatic carbon structure and in the content of Mg, Fe, Na, Ca, P, and K, while the number of C-, O-, and N-functional groups decreases (Tomczyk et al. 2020). Therefore, the lower pyrolysis temperature of biochar may provide more organic matter and functional groups to rhizosphere microorganisms, while the higher pyrolysis temperature of biochar may provide more mineral elements, allowing for an increase in the abundance and activity of microbial communities in soil contaminated with heavy metal(loid)s (Halim et al. 2020; Kaplan et al. 2016; Tomczyk et al. 2020). Actinomycetes are hypersensitive to high concentrations of heavy metal(loid)s in soils (Hiroki 1992). Thus, their reduction is related to the concentration of heavy metal(loid)s in the soil (Frostegård et al. 1993). Igalavithana et al. (2017) observed that Actinomycetes were absent in soils contaminated with higher concentrations of Pb and As, whereas Actinomycetes were present in soils contaminated with lower concentrations of Pb and As, but the proliferation of Actinomycetes increased in both soils with higher and lower concentrations of Pb and As after the addition of vegetable waste and pine cone biochar. The abundance of Actinomycetes was higher with 200 °C biochar than with 500 °C biochar because biochar obtained at 200 °C provides more C sources for microorganisms in contaminated soils (Bandara et al. 2019; Igalavithana et al. 2017). In addition, Ahmad et al. (2016) and Igalavithana et al. (2017) observed that adding biochar amendments to heavy metal(loid) contaminated soil increased the population of Gram-negative bacteria. This is because Gram-negative bacteria are more tolerant to heavy metal(loid) stress in soil and can rapidly utilize a variety of C sources for growth, compared to Gram-positive bacteria (Moche et al. 2015).
In the rhizosphere, the interactions of C and N transformations between plants and microorganisms are reciprocal, as plants produce and release inorganic and organic matter while microorganisms take up CO2 and dissolve inorganic carbon, organic acids, sugars, and amino acids, but the mobility of C and N depends on the plant and environmental conditions (Kaplan et al. 2016; Rilling et al. 2019). The addition of biochar amendments to the soil increases the availability of micronutrients to plants and also promotes microbial growth and metabolism, which further enhances the chemical reactivity of the rhizosphere and affects the mobility of heavy metal(loid)s (Halim et al. 2020; Yin et al. 2020). Zhang and Guan (2022) found that the modified biochar (biochar loaded with polyacrylic acid grafted chitosan composite) reduced the bioavailability of heavy metal(loid)s such as Mn, Cu, Fe, Zn, Ni, Pb, Cr, and As in soil while inhibiting nitrification, enhancing nitrogen fixation, ammonification, and denitrification, which had a significant effect on improving soil N fertilizer utilization. The relative abundance of Firmicutes and Bacteroidetes increased, while that of Actinobacteria, Acidobacteria, and Planctomycetes decreased at the phylum level after the application of the modified biochar, but the proportions of Actinobacteria, Acidobacteria, and Planctomycetes for the total nodes within the networks increased significantly (Zhang and Guan 2022). Firmicutes and Bacteroidetes are widely recognized as groups with resistance characteristics to external stressors such as heavy metal(loid)s (Chen et al. 2018c; Li et al. 2017; Liu et al. 2018b; Zhao et al. 2020). Therefore, they can grow and multiply rapidly, taking over ecological niches normally occupied by other communities after the addition of biochar amendments (Liu et al. 2018b). Some studies have reported that Actinobacteria communities play important roles in soil N cycling, while Acidobacteria communities play roles in soil organic matter decomposition and denitrification (Kalam et al. 2020; Samaddar et al. 2019). This indicates that biochar amendments optimize the structure of rhizosphere microbial communities, resulting in a more complex and functionally specific symbiotic network of microbial communities (Yuan et al. 2021a; Zhang et al. 2018a; Zhang and Guan 2022).
4.3 Direct and intermediate electron transfer assistance
In biogeochemical redox reactions, some biochar amendments (e.g., modified with Fe2+, Fe3+, and Mn2+), which contain redox-active functional groups and electrical conductivity, electrically support the growth of rhizosphere microorganisms through at least four ways (Fig. 3): (1) as a terminal electron acceptor for microbial respiration; (2) as a source of electrons and/or energy for microbial growth; (3) as an electrical conductor to enhance electron transfer between cells of microorganisms of the identical and distinct species; and (4) as an electron storage material or battery for microbial metabolism (Klüpfel et al. 2014; Wu et al. 2017; Xu et al. 2016; Zhang et al. 2018c). Thus, biochar amendments serve to accelerate electron transfer between microorganisms in the rhizosphere, thereby increasing microbial activity and producing more surface attachment sites that are capable of responding to heavy metal(loid)s. Li et al. (2018) reported that the biochar not only enhanced the interspecies electron transfer, which promoted the detoxification of Cd, but also improved the interaction and stabilization of the microbial community in Cd-contaminated soil. The improvement in nutrient availability and electron transfer favored the supply of substrates and energy after the biochar application, increasing the relative abundance of some species of Clostridiales, which were involved in the substrate decomposition, and some species of Sphingobacteriales, which play a key role in sulfur metabolism (Cernava et al. 2017; Li et al. 2018; Siripornadulsil and Siripornadulsil 2013).
Electrical conductivity of biochar amendments for the growth of rhizosphere microorganisms in soil contaminated with heavy metal(loid)s. a As a terminal electron acceptor for microbial respiration; b As a source of electrons and/or energy for microbial growth; c As an electrical conductor to enhance electron transfer between cells of microorganisms of the identical and distinct species; d As an electron storage material or battery for microbial metabolism (Shi et al. 2016; Zhang et al. 2018c)
The interactions between electrons of microorganisms and biochar amendments facilitate the connection of metal ion redox with the oxidation of organic matter, the fixation of CO2 into organic compounds, and the reduction of nitrate (NO3−) and photosynthesis (Shelobolina et al. 2012). Biochar can serve as an “electron shuttle”, which is an alternative terminal electron acceptor to replace NO3− in microbial communities, thereby facilitating denitrification activities in soil (Cayuela et al. 2013). Liu et al. (2020a) demonstrated that compost provided a C source for denitrifying microorganisms and promoted microbial metabolism, thereby increasing O2 consumption, and biochar acted as an alternative terminal electron acceptor for the microbial community to replace NO3−, while metals on the biochar surface could act as catalysts, which created favorable conditions for denitrification and improved the activity and growth of denitrifying microorganisms. It was found that microorganisms had difficulty surviving and performing remediation functions in soils with high concentrations of Pb and Cd, and that the excellent electrical conductivity and nutrient adsorption capacity of biochar increased the microbial extracellular electron transfer rate, which further facilitated the mineralization and immobilization of Pb and Cd (Chen et al. 2023a). The active oxygen-containing functional groups on the biochar allowed the conversion of the more mobile Cr6+ to the less mobile Cr3+ by continuous electron transfer, resulting in the stabilization of heavy metal(loid)s (Dong et al. 2017). Biochar amendments not only act as electron mediators to accelerate oxidation-reduction reactions between soil organic matter and biochar, but also promote the immobilization of heavy metal(loid)s in soil, reducing their toxicity and ultimately promoting the growth and reproduction of soil organisms (Saquing et al. 2016; Xu et al. 2019).
4.4 Heavy metal(loid)s immobilization
Heavy metal(loid)s have a negative impact on the growth, morphology, and activity of microbial populations, ultimately resulting in a reduction of both biomass and diversity (Roane and Pepper 1999). Heavy metal(loid)s such as Hg, Pb, Cd, and As serve no necessary function in the life cycle of organisms and can damage cell membranes, alter the specificity of enzymes, disrupt cellular functions, and even damage DNA structures (Bruins et al. 2000; Göhre and Paszkowski 2006). The toxicity of heavy metal(loid)s occurs through the displacement of essential metals from their natural binding sites or ligand interactions, as well as alterations to oxidative phosphorylation and conformational structure in osmotic equilibrium (Bruins et al. 2000). Moreover, heavy metal(loid)s are harmful to microorganisms due to their long biological half-life, toxicity, and non-biodegradability (Abdu et al. 2016). The microbial abundance and community diversity of metal(loid) contaminated soils are significantly lower than those of clean soils due to heavy metal(loid) stress (Chang et al. 2019; Wan et al. 2022). The addition of biochar amendments to soil facilitates the stabilization of heavy metal(loid)s, reducing their bioavailability and mitigating stress to microorganisms, ultimately contributing to the growth and metabolism of microorganisms, and improving both their abundance and activity. Zhang et al. (2020a) discovered that the addition of Fe-Mn-Ce oxide biochar to As-contaminated soil not only reduced the bioavailable forms of As but also converted specifically and non-specifically bound forms of As to amorphous and crystalline hydrous oxide bound forms, created a microbial growth-friendly environment, and significantly impacted the relative abundance of Ascomycota, Gemmatimonadetes, and Acidobacteria at the phylum level. Figure 4 analyzes the changes in microbial population diversity and community structure after the addition of biochar amendments to soils contaminated with heavy metal(loid)s (mainly Pb, Cd, As, and Hg) in recent years.
The dominant bacterial communities in different heavy metal(loid)-contaminated soils at the a phylum level (a: As, Pb, Cd, and Hg; b: As, Pb, and Cd; c: As, Cd, and Hg; d: Pb and Cd) and b genus level (A: As, Pb, and Cd; B: Pb, Cd, and Hg; C (except Pb and Hg): As and Cd; D: Cd and Hg; E: As and Hg; F: Cd) after remediation with biochar amendments (Chang et al. 2019; Igalavithana et al. 2017; Irshad et al. 2022; Liu et al. 2020b, 2017b, 2022a; Ma et al. 2020; Qi et al. 2022; Tu et al. 2020; Wan et al. 2022; Wei et al. 2022; Wu et al. 2019a; Zhang et al. 2020a, 2021; Zhou et al. 2022)
In general, the relative abundance changes in dominant groups Acidobacteria, Actinobacteria, Bacteroidetes, Firmicutes, Gemmatimonadetes, and Proteobacteria at the phylum level are found in Pb, As, Cd, and Hg contaminated soils after the biochar amendments addition. Besides these dominant groups, the relative abundance changes in Chloroflexi and Patescibacteria are easily detected in Pb contaminated soils (Liu et al. 2022a; Wan et al. 2022), the relative abundance changes in Chloroflexi and Planctomycetes are easily monitored in As contaminated soils (Irshad et al. 2022; Liu et al. 2017b; Zhang et al. 2020a), the relative abundance changes in Chloroflexi, Planctomycetes, and Patescibacteria are easily observed in Cd contaminated soils (Liu et al. 2020b, 2022a; Zhou et al. 2022), and the relative abundance changes in Planctomycetes are easily measured in Hg contaminated soils after the biochar amendments addition (Chang et al. 2019). At the genus level, the relative abundance changes in Bacillus, Flavisolibacter, Sphingomonas, Clostridium, Nitrospira, and Nocardioides are generally observed in Pb-contaminated soils after biochar addition (Liu et al. 2022a; Wei et al. 2022; Zhang et al. 2020a), while the relative abundance changes in Bacillus, Flavisolibacter, Clostridium, Nitrospira, Nocardioides, and Gemmatimonas are generally observed in As contaminated soils (Irshad et al. 2022; Liu et al. 2017b; Zhang et al. 2020a), and the relative abundance changes in Bacillus, Flavisolibacter, Sphingomonas, Clostridium, Nitrospira, Nocardioides, Massilia, and Lysobacter are generally detected in Cd contaminated soils (Liu et al. 2022a; Qi et al. 2022; Wu et al. 2019a; Zhou et al. 2022), while the relative abundance changes in Sphingomonas, Clostridium, Nitrospira, Nocardioides, Massilia, and Gemmatimonas are generally detected in Hg contaminated soils (Chang et al. 2019).
The addition of biochar amendments immobilizes heavy metal(loid)s, thereby reducing toxicity and stress on metal-sensitive microorganisms, resulting in an increase in the abundance and activity of microbial communities (Zhou et al. 2022). Liu et al. (2020b) found that nanoscale zero-valent iron-loaded biochar reduced leachability of Cd toxicity characteristic leaching procedure by 42% compared to the control and converted the labile Cd into the stable fraction, which increased the relative abundance of Firmicutes and Bacteroidetes by 33–90% and 9–88% at the phylum level, respectively, through reduced heavy metal(loid)s toxicity. In addition, many studies have found that applying biochar amendments to reduce heavy metal(loid) toxicity increases the relative abundance of genus-level Bacillus, which can survive in adverse soil conditions and are tolerant to heavy metal(loid)s (Irshad et al. 2022; Ma et al. 2020; Qi et al. 2022; Zhou et al. 2022). Although biochar amendments can help reduce heavy metal(loid)s in contaminated soil, they can also negatively impact the abundance and activity of certain microorganisms. Several studies have found that even though Proteobacteria can tolerate toxic heavy metal(loid)s, the relative abundance of Proteobacteria decreases after biochar amendments addition due to the competition between Proteobacteria and other microorganisms which are sensitive to heavy metal(loid)s and can grow rapidly after heavy metal(loid) stress reduction (Liu et al. 2020b, 2017b, 2022a; Zhang et al. 2020a). Fifty papers on biochar remediation of heavy metal(loid)s since 2017 were summarized, fifteen of which describe changes in microbial population abundance, and Table 2 counts the changes in microorganisms that co-occur in two or more papers. It is known from the statistics that the response of microbial population abundance to biochar is not static, and the change can be influenced by the type of soil, biochar and plant. For example, as a result of the addition of biochar, at the phylum level, the number of papers showing an increase in the relative abundance of Bacteroidetes, Firmicutes, Actinobacteria, Acidobacteria, Gemmatimonadetes, Planctomycetes, and Bacillus is higher than the number of papers showing a decrease; the number of papers showing a decrease in the relative abundance of Proteobacteria, Cyanobacteria, and Patescibacteria is higher than the number of papers showing an increase. At the genus level, the number of papers showing an increase in the relative abundance of Bacillus. Sp, Lysobacter. Sp, and Nocardioides. Sp is higher than the number of papers showing a decrease; the number of papers showing a decrease in the relative abundance of Sphingomonas. sp, Flavisolibacter. Sp, and Massilia. sp is higher than the number of papers showing an increase. However, due to the limited number of data samples, the trends in microbial population abundance shown by this result may not be universally applicable and therefore this result is only informative.
4.5 Exogenous microorganisms import
The changes in microbial communities, abundance, and activity in soil contaminated with heavy metal(loid)s by biochar can be attributed to the incorporation of exogenous microorganisms carried by the biochar in addition to the physical, nutrient, and electronic support, and reduction in heavy metal(loid) toxicity that biochar provided to the microorganisms. Soil rhizosphere microorganisms usually do not grow well in soils contaminated with heavy metal(loid)s due to nutrient deficiency and metal stress (Reddy and Cutright 2003). Some specific strains can adapt to the local environment and immobilize heavy metal(loid)s, and perform well in various contaminated sites (Ma et al. 2020). Biochar is considered an ideal carrier to facilitate the effective colonization of microorganisms and can be combined with specific microbial strains with bioremediation capabilities (Chen et al. 2019; Wu et al. 2019a). Therefore, combining biochar with microorganisms is an effective strategy for the remediation of soils contaminated with heavy metal(loid)s, which can immobilize heavy metal(loid)s via the adsorption of biochar, precipitation through microbial cell walls or exudates, and uptake by microorganisms (Li et al. 2018; Liu et al. 2018d; Pramanik et al. 2018). Hazrati et al. (2021) investigated that sewage sludge-derived biochar supported diverse microbial communities, which enhanced microbial activity and immobilized heavy metal(loid)s in Zn, Pb, and Cd contaminated soils. Ma et al. (2020) found that loading a novel Cd-immobilized strain Bacillus sp. onto biochar significantly increased the number of rhizosphere microorganisms and the proportion of relative abundance of the Bacillus genus, resulting in an 11.34% reduction in acetic acid-extractable Cd compared to the control. Although some studies found that biochar combined with specific strains could increase the relative abundance of specific strains in soils contaminated with heavy metals, the Shannon index microbial α-diversity decreased slightly, which could be due to competition between the inoculated strains and native microorganisms for ecological niches in the soil. Interestingly, Acidobacteria is being utilized as an indicator microorganism in the combination of biochar and microorganisms to remediate the soils contaminated with heavy metal(loid)s. In studies by Zhang et al. (2020a), Irshad et al. (2022), and Liu et al. (2017b), the relative abundance of Acidobacteria increased in the rhizosphere following the application of biochar without microorganisms. However, in studies by Liu et al. (2022a) and Chang et al. (2019), the relative abundance of Acidobacteria decreased after the addition of biochar loaded with microorganisms. These findings suggest that changes in community structure are due to competition between exogenously introduced and indigenous microorganisms in heavy metal(loid) contaminated soil (Wu et al. 2019a).
In addition, introducing microorganisms via microorganisms-loaded biochar can enhance the abundance of beneficial microorganisms and decrease the abundance of harmful microorganisms, ultimately improving the ecological stability of rhizosphere systems in heavy metal(loid) contaminated soils. Qi et al. (2022) remediated U and Cd contaminated soils by loading microbial agents, which included Citrobacter sp., Bacillus cereus, and Bacillus subtilis on biochar. The study revealed a decrease in the relative abundance of pathogenic bacteria, such as Fusarium sp. and Alternaria sp., and an increase in ecologically beneficial bacteria, including Lysobacter sp., Bacillus sp., Nitrosomonas sp., and Nitrospira sp. in the rhizosphere soils of vegetables. Many Fusarium sp. can be parasitic on multiple plants, causing plant root and stem rot in plants, and Alternaria sp. can lead to brown spot and early blight diseases in crops, which is the dominant pathogen (Budde-Rodriguez et al. 2022; Yao et al. 2017). Reducing the abundance of Fusarium sp. and Alternaria sp. can effectively decrease the risk of crop diseases. Lysobacter sp. is an important biocontrol microorganism that is antagonistic to nematodes of phytopathogenic fungi (Christensen and Cook 1978). Bacillus sp. can produce antibacterial and antifungal substances, such as lipopeptide antibiotics, which can control plant phytopathogens (Lanna-Filho et al. 2017). Therefore, biochar combined with microorganisms plays important roles in the immobilization of heavy metal(loid)s, improvement of microbial community structure, and stabilization of rhizosphere ecosystems.
5 Interactions between biochar amendments and root exudates in heavy metal(loid)-contaminated soils
Root exudates, which are produced by plant roots, rhizosphere microorganisms, and animals, play a critical role in promoting root-microorganism-soil interactions in the rhizosphere system (Rovira 1969). They can be categorized as follows: low-molecular-weight compounds, which include organic acids, carbohydrates, and some secondary metabolites (such as terpenoids and alkaloids); and high-molecular-weight exudates, which include enzymes and mucilage, as well as other compounds (Grayston et al. 1997; Jones et al. 2009; Pausch and Kuzyakov 2018; Rovira 1969). Studies have shown that root exudates can impact the physicochemical properties of rhizosphere fluids, such as pH and organic matter content, thereby affecting the mobility and bioavailability of heavy metal(loid)s in the rhizosphere soil (Layet et al. 2017; Medyńska-Juraszek et al. 2020). The interaction between biochar amendments and root exudates occurs in two ways: biochar amendments stimulate the production of root exudates, and in turn, root exudates influence the behavior of biochar amendments. The interactions involve three processes: physical adsorption, where root exudates are attracted to biochar amendments via electrostatic attraction and surface functional groups; chemisorption, which occurs through esterification reactions between -OH and -COOH in root exudates and biochar amendments; and oxidation-reduction reactions between biochar amendments and root exudates. Studies have found that oxygen-containing functional groups in biochar amendments in the rhizosphere are more abundant than those in fresh biochar amendments due to the adsorption of root exudates (Qian and Chen 2014; Ren et al. 2016; Trigo et al. 2014). This section focuses on the interactions between biochar amendments and primary root exudates (enzymes and low-molecular-weight organic acids (LMWOAs)) in heavy metal(loid)-contaminated soil.
5.1 Rhizosphere enzymes
The rhizosphere is an important enzymatic hotspot (Kuzyakov and Blagodatskaya 2015). Soil enzymes act as natural catalysts for various soil processes related to the decomposition of organic matter, leading to the release and accessibility of minerals to the rhizosphere (Ali et al. 2019; Haddad and Lemanowicz 2021). Enzyme activity reflects the intensity of biological processes and serves as an indicator of soil health and the mineralization of organic nutrients, which are typically correlated with the physical and chemical properties of the soil. There are generally three types of enzymes based on their location and role in the rhizosphere: (1) intracellular enzymes, which are located in soil organisms (microorganisms, animals, or roots) and are mainly involved in intracellular reactions of soil organisms; (2) extracellular enzymes, which are produced and released by living soil organisms and mainly catalyze the decomposition of macromolecules or biopolymers in the environment, and (3) enzymes located outside of cells due to the death or damage of soil organisms, which are always immobilized in organic mineral complexes (Burns 1982; Igalavithana et al. 2017). Phosphatase and urease are hydrolytic enzyme systems that are closely associated with the N and P cycles in soil, respectively, with phosphatase activity being related to total phosphorus and urease acting as the catalyst for the hydrolysis of urea to carbon dioxide and water (Jain et al. 2016; Wang et al. 2018). Sucrase, functioning as a hydrolase, plays a vital role in the carbon cycle of the soil system by hydrolyzing sucrose to provide energy for soil organisms and has a significant relationship with the utilization of soil nutrients (Alam et al. 2018; Liu et al. 2015). Hydrogen peroxide causes the oxidation of sulfhydryl groups in protein molecules, disrupting the normal function of cells (Liu et al. 2020b). Catalase is an oxidoreductase enzyme that decomposes hydrogen peroxide in the soil, maintaining normal microbial activities, and increasing the ability of microorganisms to withstand harsh environments (Feng et al. 2022; Sun et al. 2016b). Dehydrogenase acts as a proton acceptor in oxidation reactions, and it mainly controls the bio-oxidation of organic matter in soil, thereby reflecting the overall microbial activity (Burns et al. 2013; Igalavithana et al. 2017).
Biochar amendments remediate heavy metal(loid) contaminated soil, enhance soil physicochemical and biological properties, and consequently improve soil enzyme activity (Moore et al. 2018). Higher enzyme activity indicates a higher soil quality, while lower enzyme activity indicates an unhealthy change in soil biological processes. The addition of biochar amendments to contaminated soil is an effective option for improving enzyme activity and has a positive impact on soil health, e.g., since biochar amendments have powerful adsorption properties, the adsorption of reaction substrates by biochar amendments contributes to enzymatic reactions; moreover, biochar amendments can also sorb a portion of enzymes, protecting them from other harmful environmental factors and increasing the retention time of enzymes (Jain et al. 2016; Ji et al. 2022). However, biochar amendments can also harm enzyme activity, e.g., the adsorption of organic and inorganic substances by biochar blocks enzyme reaction sites (Ali et al. 2020; Haddad and Lemanowicz 2021). Therefore, the adsorption properties of biochar amendments also increase the complexity of soil enzymes. The impact of biochar amendments on enzyme activity is diverse and depends on soil properties, contaminant characteristics and concentrations, biochar amendment dosage, and mineral composition. Jain et al. (2016) used principal component analysis to investigate the impact of biochar, pH, and mineral solubility on enzyme activity during mine tailings remediation by biochar-Fe complex, revealing that enzyme and biochar interaction primarily influenced the activities of β-glucosidase, alkaline phosphatase, phenol oxidase, and dehydrogenase, whereas pH controlled the activities of alkaline phosphatase, β-glucosidase, and cellulase urease, and mineral solubility affected the activities of acid phosphatase and aryl sulfatase. Therefore, the effects of biochar amendments on enzyme activities were discussed from different viewpoints, including counteracting heavy metal(loid)s toxicity to soil organisms, enhancing soil bio-nutrition, improving soil bio-activity, and providing shelter.
5.1.1 Reduction of heavy metal(loid) toxicity
Biochar amendments regulate the enzymes in contaminated soil by reducing the toxicity of heavy metal(loid)s . Stimulation of heavy metal(loid)s causes damage to electron transfer processes in soil organisms occurring at the substrate end of the respiratory chain in the inner mitochondrial membrane, which disrupts the balance between pro-oxidant and antioxidant homeostasis, resulting in oxidative stress with the release of large amounts of free radicals (reactive oxygen species such as hydroxyl radical (·OH), superoxide anion (·O2−) and hydrogen peroxide (H2O2); and reactive nitrogen species such as nitrogen dioxide (·NO2), nitric oxide (·NO) and peroxynitrite (·ONOO-)) (Fryzova et al. 2017; Khan et al. 2022; Paithankar et al. 2021; Stohs and Bagchi 1995). Excess free radicals damage transcription factors, growth factors, sugars, nucleic acids, proteins, and lipids while stimulating soil organisms to produce corresponding antioxidant enzymes that resist free radical damage, such as catalase, superoxide dismutase, and glutathione peroxidase (AbdElgawad et al. 2020; Rahman 2020). Therefore, the oxidative stress of soil organisms is an important factor in the effect of heavy metal(loid)s on the alteration of soil enzymes. Applying biochar amendments to heavy metal(loid) contaminated soil enhances antioxidant enzyme activity, which mitigates excessive oxidation induced by heavy metal(loid)s. Zhou et al. (2022) showed that biochar stimulated the production of indole acetic acid by specific bacterial taxa (Flavisolibacter and Bacillus spp.) and increased the activity of superoxide dismutase and catalase in Cd-contaminated soil, thereby reducing malondialdehyde and hydrogen peroxide contents and ameliorating the Cd-induced oxidative damage.
In addition, heavy metal(loid)s (especially Pb, Cd, As, and Hg) display a high affinity for sulfhydryl-containing proteins and enzymes responsible for functional cellular defense mechanisms (e.g., regulation of excess free radicals) (Aponte et al. 2020). The combination of heavy metal(loid)s with sulfhydryl groups of enzymes to affect the catalytic action or structural integrity is another mechanism of inhibition of heavy metal(loid)s on enzyme activity (Poole and Gadd 1989). Moreover, some essential metals, such as Co, Cu, and Ni, serve as micronutrients and are components of various enzymes, which can be replaced and compete with the other heavy metal(loid)s for binding to the thiol-containing groups of enzymes, causing changes in the conformational structure of proteins and nucleic acids, thus inactivating the enzymes (Bruins et al. 2000) (Fig. 5). Biochar amendments can mitigate heavy metal(loid) toxicity by preventing their binding to enzyme active sites, such as sulfhydryl groups in urease, and consequently inhibiting the formation of metal sulfides, thus preserving the activity of urease (Liu et al. 2018c). Furthermore, biochar amendments alleviate heavy metal(loid) stress in contaminated soil by immobilizing heavy metal(loid)s through adsorption or the interaction between heavy metal(loid)s and organic matter functional groups in biochar (e.g., carboxylic acids, phenolic and aliphatic hydroxyl groups, and amino groups) to form organic-heavy metal(loid)s complexes, thereby reducing their biological effects and ultimately enhancing soil enzyme activity via detoxification (Naeem et al. 2021; Shahbaz et al. 2019; Turan 2019).
The impact of biochar amendments on soil enzyme activity varies based on factors such as biochar amendments dose, pH, organic matter, temperature, and remediation time, as demonstrated by various studies (Elzobair et al. 2016). Ali et al. (2020) showed that low doses of biochar (1% and 2.5%) increased dehydrogenase activity in Zn, and Cd contaminated soils, while higher doses of biochar (5% and 10%) inhibited the enzyme activity. Enzyme activity can be promoted by low-dose biochar by immobilizing heavy metal(loid)s and providing a suitable environment for microbial growth while higher doses of biochar result in high soil pH, which results in the inhibition of enzyme activity (Ali et al. 2020; Gong et al. 2019). Tu et al. (2020) found that the urease and catalase activities gradually decreased on the 45th day and then increased at the end of incubation on the 75th day of remediation of Cd and Cu contaminated soils, which was due to the inhibitory effect of high Cd and Cu concentrations at the early stage and the immobilization of heavy metal(loid)s at the later stage. In addition, the positive effects of biochar remediation vary under different conditions, which are related to the type of enzyme, soil properties, etc. It was found that the activities of arginase and urease were increased by 25.5% and 105%, respectively, in clay soils contaminated with Pb, Cd, and Ni by using biochar, while their activities increased by 37.1% and 83.3% in sandy soils, respectively (Haddad and Lemanowicz 2021). However, the adsorption of inorganic and organic matter in biochar and the blocking of reaction sites by biochar result in the inhibition of some enzyme activities (Bailey et al. 2011; Lehmann et al. 2011). It was found that leucine aminopeptidase, alanine aminopeptidase, phosphomonoesterase, phoshodiesterase, β-N-acetyl-D-glucosaminidase, cellobiosidase, β-xylosidase, β-glucosidase, α-glucosidase, and arylsulfatase were poorly affected by biochar (Niemi et al. 2015).
5.1.2 Regulation of microorganism and plant root
Biochar amendments significantly alter the biomass and activity of soil microorganisms and root growth, which play important roles in regulating soil enzyme activity. Rhizosphere microorganisms and roots employ enzyme production and release as a nutrient acquisition strategy, with higher enzyme expression observed in nutrient-deficient conditions (Harder and Dijkhuizen 1983). However, when nutrients are abundant, rhizosphere microorganisms and roots regulate the exudate levels of different enzymes. For example, all living microbial cells secrete dehydrogenase enzymes which can be indicative of overall microbial activity (Moeskops et al. 2010). In the study by Igalavithana et al. (2017), the addition of biochar provided microbes with a readily available C source and a significant increase in the abundance of each microbial population was subsequently observed, as well as a significant improvement in the composition and structure of the microbial community, which ultimately led to a significant increase in dehydrogenase activity. Irshad et al. (2022) found that goethite-modified biochar resulted in a significant increase in soil enzyme activity by altering soil nutrients, particularly improving phosphorus and organic matter content, which led to alterations in the soil bacterial community. Pandey et al. (2022) investigated that the higher microbial activity, resulting from increased soil fertility due to the addition of higher C, P, and K contents of Lantana biochar, led to higher activities of glucosidase, urease, dehydrogenase, and alkaline phosphatase compared to lower C, P, and K contents of Parthenium biochar and the control group in heavy metal(loid) contaminated soil. As shown by another study, the application of woody biochar to Au and Zn/Pb contaminated soils improved soil urease and β-glucosidase activities by providing a source of energy and nutrients (C, N, P, and K) for microorganisms (Ali et al. 2019). These researches indicate that biochar amendments not only reduce heavy metal(loid) toxicity through immobilization but also enhance soil microorganism activity by providing colonization sites and increasing soil nutrient levels (e.g., C, N, P, K, and Mg), resulting in increased overall soil enzyme activity (Lehmann et al. 2011).
In addition, the extent of the rhizosphere is determined by root hairs and root radius, which strongly influence enzyme activity within the rhizosphere. Root hairs significantly enhance root surface area, facilitating critical functions such as water and nutrient uptake, microorganism interactions, and regulation of rhizosphere properties and composition through exudate secretion (Ma et al. 2018). The growth, death, and release of exudates from root hairs stimulate microbial activity and further influence the enzyme dynamics such as accelerated substrate turnover (Asmar et al. 1994). Ma et al. (2018) showed that β-glucosidase in the hairy regions of wheat roots has a 1.5 times broader activity area and a 2 times higher substrate turnover rate than in hairless regions, and the extent of the rhizosphere in roots of plants with thin and long root hairs (wheat) was 1.5–2 times wider and had 2–8 times higher enzyme activity than plants without thin and long root hairs (maize, lentil, and lupine). The application of biochar in heavy metal(loid) contaminated soils had a significant effect on plant roots. It was found that in the application of biochar in soil contaminated with Cd, Pb and Ni, the higher the addition of biochar within the appropriate range, the more developed and widespread the plant root system, and the less inhibitory effect of heavy metal(loid)s on enzyme activities (urease and arginase) (Haddad and Lemanowicz 2021). Xiang et al. (2017) conducted a meta-analysis of 136 articles in 2018 and showed that the root volume, root biomass, root length, surface area, root diameter, and root tip number increased by 29%, 32%, 52%, 39%, 9.9%, and 17% after biochar amendment. Ibrahim et al. (2022) found that Mango, Casuarina, and Salix biochar all increased root biomass in soil contaminated with Co, Cd, Cu, Cr, Pb, Ni, and Zn. In the study by Zhu et al. (2022a), the combination of biofertilizer and biochar improved cotton rhizosphere resistance to Cd stress by regulating phenylalanine metabolism and ABC transporter pathways, which increased taproot and lateral root activity, as well as superoxide dismutase and peroxidase activities in Cd contaminated soils.
5.1.3 Adjustment of rhizosphere pH
The addition of biochar amendments, which forms anoxic microdomains and depletes H+ content through the presence of silicates, carbonates, and bicarbonates, has been suggested by some studies as a method for increasing enzyme activity by increasing soil pH (Harter et al. 2014; Liu et al. 2020a). In addition, the increase in precipitation of inorganic salts due to the increase in pH after the soil is remediated by biochar amendments, which reduces the toxicity of soluble salts (e.g., possibly creating reverse osmotic pressure that displaces water from microorganisms) produced by microbial decomposition of organic matter, and this is another reason why the increased pH of the soil can improve enzyme activity (Liu et al. 2020a). Chen et al. (2017a) found that the dairy manure and rice straw-derived biochars increased soil pH, denitrification reductase activity, and functional gene abundance while decreasing Cd bioavailability in Cd contaminated soil. Liu et al. (2020c) found that in Cd contaminated soil, the manure-derived biochar increased soil pH, decreased the acid-soluble Cr, and resulted in higher N-acetyl-glucosaminidase and β-glucosidase activities compared to the control. However, the decrease in enzyme activity at higher addition rates of biochar amendments may be attributed to either the toxicity of biochar or the excessive adsorption of substrates or enzymes on the porous structures. Bandara et al. (2019) reported that alkaline phosphatase and convertase activities decreased with increasing application rates of biochar and compost (up to 5% w/w). Additionally, the addition of 5% (w/w) biochar obtained from 700 °C carbonization to serpentine soil significantly reduced the activities of dehydrogenase, polyphenol oxidase, and catalase (Bandara et al. 2017). Furthermore, the difference in the effect of soil pH on enzymes may be attributed to the presence of dominant microorganisms under acidic and alkaline conditions (Turner and Haygarth 2005). Normally, an increase in soil pH generally enhances alkaline phosphatase activity and decreases acid phosphatase activity (George et al. 2002). Application of biochar amendments causing a change in soil pH from acidic to alkaline can result in a shift in the dominant microbial population, leading to a corresponding change in phosphatase activity.
5.2 Low-molecular-weight organic acids
Root exudates play a crucial role in sustaining the activity of the soil rhizosphere system and modifying the bioavailability and mobility of heavy metal(loid)s in soil, contributing to the matter cycle (Martin et al. 2014). Low-molecular-weight organic acids such as malic acid, acetic acid, citric acid, oxalic acid, tartaric acid, fumaric acid, uronic acid, etc., are typical root exudates that significantly contribute to the rhizosphere effects (Jones 1998; Liu et al. 2017a). Biochar amendments can adsorb LMWOAs, thereby increasing nutrient competition between plants and microorganisms and promoting the release of LMWOAs by regulating gene expression (Joseph et al. 2010). In addition, biochar amendments can influence the production of root exudates by immobilizing heavy metal(loid)s and reducing their bioavailability. Therefore, this section mainly discusses the interactions between biochar amendments, LMWOAs, and heavy metal(loid)s in rhizosphere soils.
5.2.1 Regulation of genes over-expression
Root exudates can bind and/or attach heavy metal(loid)s to the cell wall, restricting their entry into the cell and reducing their mobility in the soil (Ghori et al. 2019; Hossain et al. 2012). The LMWOAs can form complexes with heavy metal(loid)s and affect the bioavailability of heavy metal(loid)s in the soil (Chen et al. 2017b). Ligands present in root exudates such as oxalic acid, acetic acid, citric acid, tartaric acid, uronic acid, fumaric acid, and polysaccharides can form complexes with Pb, thereby affecting its mobility and bioavailability in soil (Ghori et al. 2019; Li et al. 2019d). Under specific stress conditions in the rhizosphere system, such as nutrient deficiency, heavy metal(loid) toxicity, and environmental changes, gene expression of the MATE (e.g., maize ZmMATE1 gene) and ALMAT (e.g., soybean GmALMT1 gene) protein families are upregulated in the root, promoting the exudation of citric acid, malic acid, and other organic acid compounds (Zhang et al. 2022). Moreover, biochar amendments act as a stress that can promote the production and efflux of LMWOAs from plant roots (Akhter et al. 2015; Bais et al. 2006). The composition of root exudates and consequently the microorganisms and their ability to biodegrade heavy metal(loid)s varied with the source, type, and concentration of biochar amendments (Akhter et al. 2015). Pei et al. (2020) found that the exudate levels of indole acetic acid, abscisic acid, and amino acid significantly increased with the biochar addition in rice roots. Cheng et al. (2018) reported that biochar resulted in the overexpression of the ZmMATE1 gene in maize, which increased the production of LMWOAs. The MATE protein mediates the transport of citrate and flavonoids, which is also involved in the detoxification of exogenous agents (e.g., heavy metal(loid)s) and endogenous secondary metabolites (Zhu et al. 2016). Sun et al. (2020) also found that the response to LMWOAs was associated with the transcript levels of the ZmMATE1 and ZmMATE2 genes, and the expression of the ZmMATE1 was upregulated by the addition of straw biochar, which positively correlated with increases in aconitic acid, fumarate, and malonate compared to the control. However, there are fewer studies targeting the direct rhizosphere relationship between biochar amendments, LMWOAs, and heavy metal(loid)s in contaminated soils, and how biochar amendments affect gene expression in the presence of heavy metal(loid)s is an area that needs further research.
5.2.2 Adjustment of soil pH
The changes in pH caused by root exudates, especially LMWOAs, are a critical mechanism affecting the solubility of heavy metal(loid)s in the rhizosphere (Houben et al. 2014). Physiologically, most plants regulate the toxicity of heavy metal(loid)s by root exudates (Choppala et al. 2014). In the phytoremediation process, tolerant plants dissolve heavy metal(loid) oxides and convert them to bioavailable forms for plants under high levels of heavy metal(loid) stress by secreting LMWOAs (Huang et al. 2017; Qiao et al. 2020). Lu et al. (2007) reported that under Cd stress, mangroves with high heavy metal(loid) retention capacity secreted more than double the number of LMWOAs by roots compared to the control, resulting in a reduction of 0.2–0.5 units in the pH of the rhizosphere soil compared to the bulk soil, and a positive correlation between the total amount of LMWOAs and the proportions of Cd bound to carbonate and exchangeable Cd. In addition, the application of biochar amendments alters the rhizosphere response to soil pH, soil nutrients, and microbial communities, which in turn affects the transformation, mobility, and bioavailability of heavy metal(loid)s (Houben and Sonnet 2015; Park et al. 2011). Achor et al. (2020) investigated the chemical behavior of heavy metal(loid)s in rhizosphere soils in the presence of biochar and LMWOAs, it was found that biochar partially inhibited the mobility of Cd, Cu, and Pb driven by LMWOAs (oxalic, citric, and malic acids), except for the absence of inhibition of Cd release by biochar under citric acid treatment. The liming effect of biochar amendments is the main mechanism for inhibiting heavy metal migration by LMWOAs, despite their ability to protonate the surface of biochar amendments, which can deactivate or reduce their adsorption capacity for heavy metal(loid)s (Achor et al. 2020). However, the mobility and bioavailability of heavy metal(loid)s in the soil can increase again if the alkalinity of the biochar amendments is not sufficient to neutralize the acidity of the LMWOAs (Wang et al. 2022a). The study by Houben and Sonnet (2015) found that 5% biochar significantly reduced exchangeable heavy metal(loid)s and facilitated the transition from the exchangeable to the carbonate bound state in Cd, Pb, and Zn contaminated soils, but the heavy metal(loid)s were remobilized due to the subsequent acidification of root-generated LMWOAs that neutralized the liming effect of biochar. This is consistent with the trend observed in several studies where liming of neutralizing LMWOAs produced by biochar increases with increased application of biochar, resulting in immobilization of metal(loid)s and reduced uptake of heavy metal(loid)s by plants (Al-Wabel et al. 2015; Kim et al. 2015; Rees et al. 2015).
5.2.3 As electron acceptor or donor
The electron transfer behavior of biochar amendments has been extensively studied in the geochemistry and redox reactions of heavy metal(loid) contaminants (Chen et al. 2018b; Kappler et al. 2014; Yuan et al. 2017). The electron transfer between biochar amendments and LMWOAs is generated by the repeated reduction and oxidation processes on the oxygen-containing functional groups (e.g., -OH and -C=O) of their surfaces, which can change the composition of surface groups of biochar amendments (Deng and Stone 1996; Klüpfel et al. 2014; Saquing et al. 2016). Xu et al. (2019) investigated the impact of seven LMWOAs on the interaction of peanut shell biochar and its electron shuttle effect on Cr(VI) reduction and found that the adsorption and interaction of biochar with LMWOAs resulted in redox reactions that modified the Cr(VI) reduction-related functional groups (-C=O and C-O) on the biochar surface. LMWOAs with more reducing functional groups (-OH) or lower redox potential may reduce biochar amendments (LMWOA-CH2OH + BC-COOH → LMWOA-C = O + BC-CH2OH + H2O), while LMWOAs with more oxidizing functional groups (-COOH) and higher redox potential may lead to oxidation processes (LMWOA-C = O + BC-CH2OH + H2O → LMWOA-CH2OH + BC-COOH) (Xu et al. 2019). Sugars, LMWOAs, and amino acids are the main sources of soil carbon, which not only provide abundantly available C sources for microorganisms but also serve as electron donors in soil electron transfer activities (Kamilova et al. 2006; Zhalnina et al. 2018). Therefore, biochar amendments have the ability to accept electrons from LMWOAs in soil and transfer them to the receptor (heavy metal(loid)s) (Fig. 3) (Deng and Stone 1996; Mu et al. 2018).
5.2.4 Interaction between LMWOAs and biochar
The adsorption mechanisms between LMWOAs and biochar amendments include the formation of internal and external spherical surface complexes, cation bridge bonding, and ligand exchange, which depend on the pH of the solution, and the molecular structure of the LMWOAs (Sokolova 2020). In acidic environments, large amounts of LMWOAs were adsorbed on the biochar, resulting in an increase in bridge (hydrogen) bonding interactions on the biochar surface and a decrease in interparticle repulsion. This is particularly evident in nano-biochar, where aggregation of nano-biochar occurs due to LMWOAs, reducing the mobility of nano-biochar in the rhizosphere soil (Li et al. 2019a; Wang et al. 2022b; Zhang et al. 2010). In addition, LMWOAs and the carboxyl groups (COO-) and ionized protons (H+) contained in LMWOAs can dissolve minerals (such as CaCO3, Ca2Mg(PO4)2, CaMg(CO3)2, AlPO4, Pb2P2O7, and K2Mg(PO3)4) contained in soil and biochar amendments, which promotes the release of plant nutrients (e.g., PO43−, SO42−, K+, Ca2+, Mg2+, and Fe3+) and significantly improves soil fertility (Cao et al. 2009; Liu et al. 2017a; Wang et al. 2015; Yuan et al. 2011). However, it can also dissolve and release toxic metals from rhizosphere soils (Jones 1998). Liu et al. (2017a) found that the erosion of biochar by acetic, malic, and citric acids in the rhizosphere increased the risk of heavy metal(loid)s being released to the environment by mobilizing them from biochar to the aqueous phase. In addition, the adsorption of LMWOAs on biochar amendments promotes the development of the specific surface area and pore structure of biochar amendments, but its effects are closely related to the original pore characteristics (Sun et al. 2016a). Deashing by LMWOAs improves the pore size of biochar amendments by releasing minerals that are dissolved in the biochar amendments (Lou et al. 2011). In particular, low-temperature biochar can release more organic and dissolved inorganic fractions, and the mechanical strength determines that low-temperature biochar undergoes easier physical erosion during washing, leading to more macropore formation (Braadbaart et al. 2009; Sun et al. 2016a). Liu et al. (2017a) found that LMWOAs also induced pore widening by deashing in high-temperature biochar, and caused fractures of the carbon skeleton in 300 °C and 600 °C biochar, which may be due to the physical stress (e.g., swelling) caused by water adsorption into the pores or interstices within the sheets on the biochar structure (Spokas et al. 2014).
6 Challenges and future perspectives
There are some challenges in the application of biochar in the remediation of heavy metal(loid) contaminated soils and its impact on the soil rhizosphere , such as, how to ensure the stability of the properties of biochar, the effect of overuse of biochar on the rhizosphere microorganisms and plants in the soil, and the effect of the toxic substances (such as heavy metal(loid)s) in biochar on the soil by the use of biochar. The properties of biochar are affected by the type of feedstock, preparation temperature, heating rate, residence time, and modification method (Wang et al. 2020d). The organic matter of different types of feedstocks varies widely, for example, agricultural and forest wastes consist mainly of cellulose, lignin, and hemicellulose, while manure contains not only cellulose and hemicellulose, but also fats, proteins, organic acids, and humic acids (Oliveira et al. 2017; Wang et al. 2017). Due to the different sources of raw materials, the properties of the resulting biochar vary considerably. In addition, even the biochar produced from the same raw material may have different correspondences depending on the nature of the soil; even if the same production equipment is used, it is difficult to maintain the same characteristics of biochar produced from different batches of the same raw material. In addition, preparation temperature, heating rate, and residence time also affect the specific surface area, pore size structure, pH, cation exchange capacity, surface functional groups, and mineral content of biochar. Therefore, the properties of biochar are largely influenced by anthropogenic and non-anthropogenic factors, which can lead to difficulties in standardizing the stability of biochar on soil heavy metal(loid)s as well as its effect on the rhizosphere, so specific methods should be discussed when investigating the remediation of heavy metal(loid) contaminated soils by biochar as well as its effect on the rhizosphere.
A large number of studies have appeared in recent years on the application of biochar to remediate heavy metal(loid) contaminated soils, and in some of these studies, the amount of biochar used was much higher than the potential realistic need. Wang et al. (2020d) summarized 73 recently published research papers on the remediation of heavy metal(loid)s in soils with biochar and found that although most of the articles had a biochar use rate between 1% and 5%, there were still 15.7% of the articles that had a biochar use rate greater than 10%. Appropriate amounts of biochar can improve soil physicochemical properties, such as increasing soil nutrient content and promoting plant growth, but overuse of biochar increases the risk of soil alkalization, and an imbalance in the nutrient content of biochar can reduce seed germination (Beesley et al. 2013; Liu et al. 2023). Therefore, overuse of biochar may have negative long-term effects on soil or rhizosphere plants and microorganisms. Thus, when remediating heavy metal(loid) contaminated soils with biochar, it is important to consider the effects of biochar dosage on the rhizosphere in the soil to avoid negative effects of over-application that are detrimental to plants and microorganisms.
Some straw harvested from metal(loid) contaminated farmland also contains heavy metal(loid)s, and animal manure from the overuse of additives in animal feed also contains high levels of heavy metal(loid)s, in addition, sewage sludge also contains large amounts of heavy metal(loid)s (Li et al. 2019b; Mierzwa-Hersztek et al. 2018; Shen et al. 2016). Therefore, the application of these biochar, which are enriched with a large number of heavy metal(loid)s in the raw materials, to soil remediation will not only exacerbate the release of heavy metal(loid)s, but also have an inhibitory effect on rhizosphere plants and microorganisms (Shen et al. 2016). Therefore, when considering soil remediation and its effects on the rhizosphere, the feedstock and the biochar modification process should be considered simultaneously to determine whether new contaminants will be introduced.
7 Conclusion
Achieving the UN’s SDGs and sustaining life on Earth requires the search for green and sustainable remediation. The application of biochar amendments to soil contaminated with heavy metal(loid)s is a highly appropriate solution that utilizes waste resources, improves soil health, reduces CO2 emissions, and generates positive social, economic, and environmental impacts (Fig. 6). Based on the utilization of waste resources, biomass wastes (including forest waste, livestock waste, agricultural and garden waste, and industrial waste) are recovered and carbonized to produce biochar, energy, and co-products (e.g., bio-oil, bio-gas). This review presents recent research on the application of biochar amendments in the remediation of soil contaminated with heavy metal(loid)s. Three types of biochar amendments, raw, functional, and co-composite, have been studied. Biochar amendments have been found to immobilize heavy metal(loid)s and regulate the physical-chemical properties of the rhizosphere, such as regulating soil pH, increasing soil fertility, water-holding capacity and soil biomass, and promoting the activity of soil organisms. Furthermore, unique physicochemical properties of biochar amendments have been shown to improve the rhizosphere microbial community structure and activity by providing colonization sites, nutrients, and electronic support. Interactions between biochar and root exudates, including enzymes and LMWOAs, have also been observed. These interactions have the potential metal(loid)s toxicity, promote rhizosphere biomass activity, regulate pH, provide physical protection, regulate gene expression, and support electron transfer from biochar to root exudates in soil. The application of biochar amendments has been found to restore the health of contaminated soils, reduce the bioavailability of heavy metal(loid)s, and increase the utilization value for plants. This sustainable cycle can also generate organic waste that can be returned to the cycle, providing social and economic benefits.
Based on the current understanding of the interactions between biochar amendments, heavy metal(loid)s, and the rhizosphere in soil remediation, we propose further research as follows: (1) investigating the long-term stability and rhizosphere microbial dynamics of biochar in heavy metal(loid) contaminated soil remediation; (2) studying the variations in interactions between biochar and rhizosphere microorganisms under different heavy metal(loid)s; (3) examining the direct interaction between biochar and LMWOAs in heavy metal(loid) contaminated soil; and (4) exploring the mechanism by which microorganisms combined with biochar affect the relative abundance of specific species (such as Acidobacteria) in heavy metal(loid) contaminated soil.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
AbdElgawad H, Zinta G, Hamed BA, Selim S, Beemster G, Hozzein WN, Wadaan MA, Asard H, Abuelsoud W (2020) Maize roots and shoots show distinct profiles of oxidative stress and antioxidant defense under heavy metal toxicity. Environ Pollut 258:113705. https://doi.org/10.1016/j.envpol.2019.113705
Abdu N, Abdullahi AA, Abdulkadir A (2016) Heavy metals and soil microbes. Environ Chem Lett 15:65–84. https://doi.org/10.1007/s10311-016-0587-x
Achor S, Aravis C, Heaney N, Odion E, Lin C (2020) Response of organic acid-mobilized heavy metals in soils to biochar application. Geoderma 378:114628. https://doi.org/10.1016/j.geoderma.2020.114628
Ahmad M, Ok YS, Rajapaksha AU, Lim JE, Kim B-Y, Ahn J-H, Lee YH, Al-Wabel MI, Lee S-E, Lee SS (2016) Lead and copper immobilization in a shooting range soil using soybean stover-and pine needle-derived biochars: Chemical, microbial and spectroscopic assessments. J Hazard Mater 301:179–186. https://doi.org/10.1016/j.jhazmat.2015.08.029
Akhter A, Hage-Ahmed K, Soja G, Steinkellner S (2015) Compost and biochar alter mycorrhization, tomato root exudation, and development of Fusarium oxysporum f. sp. lycopersici. Front Plant Sci 6:529. https://doi.org/10.3389/fpls.2015.00529
Al-Wabel MI, Usman AR, El-Naggar AH, Aly AA, Ibrahim HM, Elmaghraby S, Al-Omran A (2015) Conocarpus biochar as a soil amendment for reducing heavy metal availability and uptake by maize plants. Saudi J Biol Sci 22:503–511. https://doi.org/10.1016/j.sjbs.2014.12.003
Alam MS, Gorman-Lewis D, Chen N, Flynn SL, Ok YS, Konhauser KO, Alessi DS (2018) Thermodynamic analysis of Nickel(II) and Zinc(II) adsorption to biochar. Environ Sci Technol 52:6246–6255. https://doi.org/10.1021/acs.est.7b06261
Ali A, Guo D, Arockiam Jeyasundar PGS, Li Y, Xiao R, Du J, Li R, Zhang Z (2019) Application of wood biochar in polluted soils stabilized the toxic metals and enhanced wheat (Triticum aestivum) growth and soil enzymatic activity. Ecotoxicol Environ Saf 184:109635. https://doi.org/10.1016/j.ecoenv.2019.109635
Ali A, Shaheen SM, Guo D, Li Y, Xiao R, Wahid F, Azeem M, Sohail K, Zhang T, Rinklebe J, Li R, Zhang Z (2020) Apricot shell- and apple tree-derived biochar affect the fractionation and bioavailability of Zn and Cd as well as the microbial activity in smelter contaminated soil. Environ Pollut 264:114773. https://doi.org/10.1016/j.envpol.2020.114773
Aponte H, Meli P, Butler B, Paolini J, Matus F, Merino C, Cornejo P, Kuzyakov Y (2020) Meta-analysis of heavy metal effects on soil enzyme activities. Sci Total Environ 737:139744. https://doi.org/10.1016/j.scitotenv.2020.139744
Asmar F, Eiland F, Nielsen NE (1994) Effect of extracellular-enzyme activities on solubilization rate of soil organic nitrogen. Biol Fertil Soils 17:32–38
Bailey VL, Fansler SJ, Smith JL, Bolton H (2011) Reconciling apparent variability in effects of biochar amendment on soil enzyme activities by assay optimization. Soil Biol Biochem 43:296–301. https://doi.org/10.1016/j.soilbio.2010.10.014
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266. https://doi.org/10.1146/annurev.arplant.57.032905.105159
Bandara T, Franks A, Xu J, Bolan N, Wang H, Tang C (2019) Chemical and biological immobilization mechanisms of potentially toxic elements in biochar-amended soils. Crit Rev Environ Sci Technol 50:903–978. https://doi.org/10.1080/10643389.2019.1642832
Bandara T, Herath I, Kumarathilaka P, Seneviratne M, Seneviratne G, Rajakaruna N, Vithanage M, Ok YS (2017) Role of woody biochar and fungal-bacterial co-inoculation on enzyme activity and metal immobilization in serpentine soil. J Soils Sediments 17:665–673. https://doi.org/10.1007/s11368-015-1243-y
Bashir S, Hussain Q, Jun Z, Qingling F, Houben D, Hongqing H (2020) Efficiency of KOH-modified rice straw-derived biochar for reducing cadmium mobility, bioaccessibility and bioavailability risk index in red soil. Pedosphere 30:874–882. https://doi.org/10.1016/S1002-0160(20)60043-1
Batool M, W-u-D Khan, Hamid Y, Farooq MA, Naeem MA, Nadeem F (2022) Interaction of pristine and mineral engineered biochar with microbial community in attenuating the heavy metals toxicity: a review. Appl Soil Ecol 175:104444. https://doi.org/10.1016/j.apsoil.2022.104444
Beesley L, Marmiroli M, Pagano L, Pigoni V, Fellet G, Fresno T, Vamerali T, Bandiera M, Marmiroli N (2013) Biochar addition to an arsenic contaminated soil increases arsenic concentrations in the pore water but reduces uptake to tomato plants (Solanum lycopersicum L.). Sci Total Environ 454–455:598–603. https://doi.org/10.1016/j.scitotenv.2013.02.047
Bhaduri AM, Fulekar M (2012) Antioxidant enzyme responses of plants to heavy metal stress. Rev Environ Sci Biotechnol 11:55–69. https://doi.org/10.1007/s11157-011-9251-x
Braadbaart F, Poole I, van Brussel AA (2009) Preservation potential of charcoal in alkaline environments: an experimental approach and implications for the archaeological record. J Archaeol Sci 36:1672–1679. https://doi.org/10.1016/j.jas.2009.03.006
Bruins MR, Kapil S, Oehme FW (2000) Microbial resistance to metals in the environment. Ecotoxicol Environ Saf 45:198–207. https://doi.org/10.1006/eesa.1999.1860
Budde-Rodriguez S, Pasche JS, Mallik I, Gudmestad NC (2022) Sensitivity of Alternaria spp. from potato to pyrimethanil, cyprodinil, and fludioxonil. Crop Prot. 152:105855. https://doi.org/10.1016/j.cropro.2021.105855
Burns R (1982) Enzyme activity in soil location and a possible role inmicrobial ecology. Soil Biol Biochem 14:423–427. https://doi.org/10.1016/0038-0717(82)90099-2
Burns RG, DeForest JL, Marxsen J, Sinsabaugh RL, Stromberger ME, Wallenstein MD, Weintraub MN, Zoppini A (2013) Soil enzymes in a changing environment: current knowledge and future directions. Soil Biol Biochem 58:216–234. https://doi.org/10.1016/j.soilbio.2012.11.009
Buss W, Wurzer C, Manning DA, Rohling EJ, Borevitz J, Mašek O (2022) Mineral-enriched biochar delivers enhanced nutrient recovery and carbon dioxide removal. Commun Earth Environ 3:67. https://doi.org/10.1038/s43247-022-00394-w
Cao X, Ma L, Gao B, Harris W (2009) Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ Sci Technol 43:3285–3291. https://doi.org/10.1021/es803092k
Cayuela ML, Sánchez-Monedero MA, Roig A, Hanley K, Enders A, Lehmann J (2013) Biochar and denitrification in soils: when, how much and why does biochar reduce N2O emissions? Sci Rep 3:1–7. https://doi.org/10.1038/srep01732
Cernava T, Erlacher A, Aschenbrenner IA, Krug L, Lassek C, Riedel K, Grube M, Berg G (2017) Deciphering functional diversification within the lichen microbiota by meta-omics. Microbiome 5:1–13. https://doi.org/10.1186/s40168-017-0303-5
Chai L, Li Q, Zhu Y, Zhang Z, Wang Q, Wang Y, Yang Z (2010) Synthesis of thiol-functionalized spent grain as a novel adsorbent for divalent metal ions. Biores Technol 101:6269–6272. https://doi.org/10.1016/j.biortech.2010.03.009
Chang J, Yang Q, Dong J, Ji B, Si G, He F, Li B, Chen J (2019) Reduction in Hg phytoavailability in soil using Hg-volatilizing bacteria and biochar and the response of the native bacterial community. Microb Biotechnol 12:1014–1023. https://doi.org/10.1111/1751-7915.13457
Chaudhary DK, Park J-H, Kim P-G, Ok YS, Hong Y (2023) Enrichment cultivation of VOC-degrading bacteria using diffusion bioreactor and development of bacterial-immobilized biochar for VOC bioremediation. Environ Pollut 320:121089. https://doi.org/10.1016/j.envpol.2023.121089
Chen B, Chen Z (2009) Sorption of naphthalene and 1-naphthol by biochars of orange peels with different pyrolytic temperatures. Chemosphere 76:127–133. https://doi.org/10.1016/j.chemosphere.2009.02.004
Chen D, Liu W, Wang Y, Lu P (2022) Effect of biochar aging on the adsorption and stabilization of Pb in soil. J Soils Sediments 22:56–66. https://doi.org/10.1007/s11368-021-03059-x
Chen G, Zhang Z, Zhang Z, Zhang R (2017a) Influence of biochar addition on the denitrification process and N2O emission in Cd-contaminated soil. Water Air Soil Pollut 228:1–11. https://doi.org/10.1007/s11270-016-3228-x
Chen H, Min F, Hu X, Ma D, Huo Z (2023) Biochar assists phosphate solubilizing bacteria to resist combined Pb and Cd stress by promoting acid secretion and extracellular electron transfer. J Hazard Mater 452:131176. https://doi.org/10.1016/j.jhazmat.2023.131176
Chen H, Zhang J, Tang L, Su M, Tian D, Zhang L, Li Z, Hu S (2019) Enhanced Pb immobilization via the combination of biochar and phosphate solubilizing bacteria. Environ Int 127:395–401. https://doi.org/10.1016/j.envint.2019.03.068
Chen J, Dong J, Chang J, Guo T, Yang Q, Jia W, Shen S (2018a) Characterization of an Hg(II)-volatilizing Pseudomonas sp. strain, DC-B1, and its potential for soil remediation when combined with biochar amendment. Ecotoxicol Environ Saf 163:172–179. https://doi.org/10.1016/j.ecoenv.2018.07.071
Chen J, Wang C, Pan Y, Farzana SS, Tam NF-Y (2018b) Biochar accelerates microbial reductive debromination of 2, 2′, 4, 4′-tetrabromodiphenyl ether (BDE-47) in anaerobic mangrove sediments. J Hazard Mater 341:177–186. https://doi.org/10.1016/j.jhazmat.2017.07.063
Chen X, Fan G, Zhu X, Li H, Li Y, Li H, Xu X (2023) The remediation of hexavalent chromium-contaminated soil by nanoscale zero-valent iron supported on sludge-based biochar. J Soils Sediments 23:1607–1616. https://doi.org/10.1007/s11368-023-03433-x
Chen Y-T, Wang Y, Yeh K-C (2017b) Role of root exudates in metal acquisition and tolerance. Curr Opin Plant Biol 39:66–72. https://doi.org/10.1016/j.pbi.2017.06.004
Chen Y, Jiang Y, Huang H, Mou L, Ru J, Zhao J, Xiao S (2018c) Long-term and high-concentration heavy-metal contamination strongly influences the microbiome and functional genes in Yellow River sediments. Sci Total Environ 637:1400–1412. https://doi.org/10.1016/j.scitotenv.2018.05.109
Chen Z, Wang Y, Xia D, Jiang X, Fu D, Shen L, Wang H, Li QB (2016) Enhanced bioreduction of iron and arsenic in sediment by biochar amendment influencing microbial community composition and dissolved organic matter content and composition. J Hazard Mater 311:20–29. https://doi.org/10.1016/j.jhazmat.2016.02.069
Cheng N, Peng Y, Kong Y, Li J, Sun C (2018) Combined effects of biochar addition and nitrogen fertilizer reduction on the rhizosphere metabolomics of maize (Zea mays L.) seedlings. Plant Soil 433:19–35. https://doi.org/10.1007/s11104-018-3811-6
Choppala G, Saifullah BN, Bibi S, Iqbal M, Rengel Z, Kunhikrishnan A, Ashwath N, Ok YS (2014) Cellular mechanisms in higher plants governing tolerance to cadmium toxicity. Crit Rev Plant Sci 33:374–391. https://doi.org/10.1080/07352689.2014.903747
Christensen P, Cook F (1978) Lysobacter, a new genus of nonfruiting, gliding bacteria with a high base ratio. Int J Syst Evol Microbiol 28:367–393. https://doi.org/10.1099/00207713-28-3-367
Chuaphasuk C, Prapagdee B (2019) Effects of biochar-immobilized bacteria on phytoremediation of cadmium-polluted soil. Environ Sci Pollut Res 26:23679–23688. https://doi.org/10.1007/s11356-019-05661-6
Dai X, Yi W, Yin C, Li K, Feng L, Zhou Q, Yi Z, Zhang X, Wang Y, Yu Y, Han X, Zhang Y (2022) 2D–3D magnetic NiFe layered double hydroxide decorated diatomite as multi-function material for anionic, cationic dyes, arsenate, and arsenite adsorption. Appl Clay Sci 229:106664. https://doi.org/10.1016/j.clay.2022.106664
De Vries FT, Hoffland E, van Eekeren N, Brussaard L, Bloem J (2006) Fungal/bacterial ratios in grasslands with contrasting nitrogen management. Soil Biol Biochem 38:2092–2103. https://doi.org/10.1016/j.soilbio.2006.01.008
Deng B, Stone AT (1996) Surface-catalyzed chromium (VI) reduction: reactivity comparisons of different organic reductants and different oxide surfaces. Environ Sci Technol 30:2484–2494. https://doi.org/10.1021/es950780p
Dong H, Deng J, Xie Y, Zhang C, Jiang Z, Cheng Y, Hou K, Zeng G (2017) Stabilization of nanoscale zero-valent iron (nZVI) with modified biochar for Cr(VI) removal from aqueous solution. J Hazard Mater 332:79–86. https://doi.org/10.1016/j.jhazmat.2017.03.002
Dos Santos FH, Soares MB, Alleoni LRF (2022) Pristine and biochar-supported nano zero-valent iron to immobilize As, Zn and Pb in soil contaminated by smelting activities. J Environ Manage 321:116017. https://doi.org/10.1016/j.jenvman.2022.116017
El-Naggar A, Lee SS, Rinklebe J, Farooq M, Song H, Sarmah AK, Zimmerman AR, Ahmad M, Shaheen SM, Ok YS (2019) Biochar application to low fertility soils: a review of current status, and future prospects. Geoderma 337:536–554. https://doi.org/10.1016/j.geoderma.2018.09.034
Elzobair KA, Stromberger ME, Ippolito JA, Lentz RD (2016) Contrasting effects of biochar versus manure on soil microbial communities and enzyme activities in an Aridisol. Chemosphere 142:145–152. https://doi.org/10.1016/j.chemosphere.2015.06.044
Fan J, Cai C, Chi H, Reid BJ, Coulon F, Zhang Y, Hou Y (2020) Remediation of cadmium and lead polluted soil using thiol-modified biochar. J Hazard Mater 388:122037. https://doi.org/10.1016/j.jhazmat.2020.122037
Feng Q, Wang B, Chen M, Wu P, Lee X, Xing Y (2021) Invasive plants as potential sustainable feedstocks for biochar production and multiple applications: A review. Resour Conserv Recycl 164:105204. https://doi.org/10.1016/j.resconrec.2020.105204
Feng S, Zhang P, Hu Y, Jin F, Liu Y, Cai S, Song Z, Zhang X, Nadezhda T, Guo Z, Lynch I, Dang X (2022) Combined application of biochar and nano-zeolite enhanced cadmium immobilization and promote the growth of Pak Choi in cadmium contaminated soil. NanoImpact 28:100421. https://doi.org/10.1016/j.impact.2022.100421
Fijałkowska G, Wiśniewska M, Szewczuk-Karpisz K, Jędruchniewicz K, Oleszczuk P (2021) Comparison of lead(II) ions accumulation and bioavailability on the montmorillonite and kaolinite surfaces in the presence of polyacrylamide soil flocculant. Chemosphere 276:130088. https://doi.org/10.1016/j.chemosphere.2021.130088
Forján R, Rodríguez-Vila A, Cerqueira B, Covelo E (2018) Comparison of compost with biochar versus technosol with biochar in the reduction of metal pore water concentrations in a mine soil. J Geochem Explor 192:103–111. https://doi.org/10.1016/j.gexplo.2018.06.007
Frostegård Å, Tunlid A, Bååth E (1993) Phospholipid fatty acid composition, biomass, and activity of microbial communities from two soil types experimentally exposed to different heavy metals. Appl Environ Microbiol 59:3605–3617. https://doi.org/10.1128/aem.59.11.3605-3617.1993
Fryzova R, Pohanka M, Martinkova P, Cihlarova H, Brtnicky M, Hladky J, Kynicky J (2017) Oxidative stress and heavy metals in plants. Rev Environ Contam Toxicol 245:129–156. https://doi.org/10.1007/398_2017_7
Gao Y, Wu P, Jeyakumar P, Bolan N, Wang H, Gao B, Wang S, Wang B (2022) Biochar as a potential strategy for remediation of contaminated mining soils: mechanisms, applications, and future perspectives. J Environ Manage 313:114973. https://doi.org/10.1016/j.jenvman.2022.114973
Gargallo-Garriga A, Preece C, Sardans J, Oravec M, Urban O, Peñuelas J (2018) Root exudate metabolomes change under drought and show limited capacity for recovery. Sci Rep 8:1–15. https://doi.org/10.1038/s41598-018-30150-0
Gasco G, Alvarez ML, Paz-Ferreiro J, Mendez A (2019) Combining phytoextraction by Brassica napus and biochar amendment for the remediation of a mining soil in Riotinto (Spain). Chemosphere 231:562–570. https://doi.org/10.1016/j.chemosphere.2019.05.168
George T, Gregory P, Wood M, Read D, Buresh R (2002) Phosphatase activity and organic acids in the rhizosphere of potential agroforestry species and maize. Soil Biol Biochem 34:1487–1494. https://doi.org/10.1016/S0038-0717(02)00093-7
Ghorbani M, Konvalina P, Kopecký M, Kolář L (2022) A meta-analysis on the impacts of different oxidation methods on the surface area properties of biochar. Land Degrad Dev 34:299–312. https://doi.org/10.1002/ldr.4464
Ghori N-H, Ghori T, Hayat M, Imadi S, Gul A, Altay V, Ozturk M (2019) Heavy metal stress and responses in plants. Int J Environ Sci Technol 16:1807–1828. https://doi.org/10.1007/s13762-019-02215-8
Ghosh D, Maiti SK (2021) Biochar assisted phytoremediation and biomass disposal in heavy metal contaminated mine soils: a review. Int J Phytorem 23:559–576. https://doi.org/10.1080/15226514.2020.1840510
Giller KE, Witter E, McGrath SP (2009) Heavy metals and soil microbes. Soil Biol Biochem 41:2031–2037. https://doi.org/10.1016/j.soilbio.2009.04.026
Göhre V, Paszkowski U (2006) Contribution of the arbuscular mycorrhizal symbiosis to heavy metal phytoremediation. Planta 223:1115–1122. https://doi.org/10.1007/s00425-006-0225-0
Gong H, Zhao L, Rui X, Hu J, Zhu N (2022) A review of pristine and modified biochar immobilizing typical heavy metals in soil: applications and challenges. J Hazard Mater 432:128668. https://doi.org/10.1016/j.jhazmat.2022.128668
Gong X, Huang D, Liu Y, Zeng G, Chen S, Wang R, Xu P, Cheng M, Zhang C, Xue W (2019) Biochar facilitated the phytoremediation of cadmium contaminated sediments: metal behavior, plant toxicity, and microbial activity. Sci Total Environ 666:1126–1133. https://doi.org/10.1016/j.scitotenv.2019.02.215
Grayston S, Vaughan D, Jones D (1997) Rhizosphere carbon flow in trees, in comparison with annual plants: the importance of root exudation and its impact on microbial activity and nutrient availability. Appl Soil Ecol 5:29–56. https://doi.org/10.1016/S0929-1393(96)00126-6
Gupta A, Sharma V, Sharma K, Kumar V, Choudhary S, Mankotia P, Kumar B, Mishra H, Moulick A, Ekielski A, Mishra PK (2021) A review of adsorbents for heavy metal decontamination: growing approach to wastewater treatment. Materials 14:4702. https://doi.org/10.3390/ma14164702
Haddad SA, Lemanowicz J (2021) Benefits of corn-cob biochar to the microbial and enzymatic activity of soybean plants grown in soils contaminated with heavy metals. Energies 14:5763. https://doi.org/10.3390/en14185763
Haider FU, Coulter JA, Cheema SA, Farooq M, Wu J, Zhang R, Shuaijie G, Liqun C (2021) Co-application of biochar and microorganisms improves soybean performance and remediate cadmium-contaminated soil. Ecotoxicol Environ Saf 214:112112. https://doi.org/10.1016/j.ecoenv.2021.112112
Halim MA, Rahman MM, Megharaj M, Naidu R (2020) Cadmium immobilization in the rhizosphere and plant cellular detoxification: role of plant-growth-promoting rhizobacteria as a sustainable solution. J Agric Food Chem 68:13497–13529. https://doi.org/10.1021/acs.jafc.0c04579
Harder W, Dijkhuizen L (1983) Physiological responses to nutrient limitation. Annu Rev Microbiol 37:1–23. https://doi.org/10.1146/annurev.mi.37.100183.000245
Harter J, Krause H-M, Schuettler S, Ruser R, Fromme M, Scholten T, Kappler A, Behrens S (2014) Linking N2O emissions from biochar-amended soil to the structure and function of the N-cycling microbial community. ISME J 8:660–674. https://doi.org/10.1038/ismej.2013.160
Hazrati S, Farahbakhsh M, Cerda A, Heydarpoor G (2021) Functionalization of ultrasound enhanced sewage sludge-derived biochar: physicochemical improvement and its effects on soil enzyme activities and heavy metals availability. Chemosphere 269:128767. https://doi.org/10.1016/j.chemosphere.2020.128767
Hiroki M (1992) Effects of heavy metal contamination on soil microbial population. Soil Sci Plant Nutr 38:141–147. https://doi.org/10.1080/00380768.1992.10416961
Hossain MA, Piyatida P, da Silva JAT, Fujita M (2012) Molecular mechanism of heavy metal toxicity and tolerance in plants: central role of glutathione in detoxification of reactive oxygen species and methylglyoxal and in heavy metal chelation. J Botany. https://doi.org/10.1155/2012/872875
Hou D, O’Connor D, Igalavithana AD, Alessi DS, Luo J, Tsang DCW, Sparks DL, Yamauchi Y, Rinklebe J, Ok YS (2020) Metal contamination and bioremediation of agricultural soils for food safety and sustainability. Nat Rev Earth Environ 1:366–381. https://doi.org/10.1038/s43017-020-0061-y
Houben D, Sonnet P (2015) Impact of biochar and root-induced changes on metal dynamics in the rhizosphere of Agrostis capillaris and Lupinus albus. Chemosphere 139:644–651. https://doi.org/10.1016/j.chemosphere.2014.12.036
Houben D, Sonnet P, Tricot G, Mattielli N, Eo C, Opfergelt S (2014) Impact of root-induced mobilization of zinc on stable Zn isotope variation in the soil–plant system. Environ Sci Technol 48:7866–7873. https://doi.org/10.1021/es5002874
Huang D, Gong X, Liu Y, Zeng G, Lai C, Bashir H, Zhou L, Wang D, Xu P, Cheng M (2017) Effects of calcium at toxic concentrations of cadmium in plants. Planta 245:863–873. https://doi.org/10.1007/s00425-017-2664-1
Ibrahim EA, El-Sherbini MAA, Selim E-MM (2022) Effects of biochar on soil properties, heavy metal availability and uptake, and growth of summer squash grown in metal-contaminated soil. Sci Hortic 301:111097. https://doi.org/10.1016/j.scienta.2022.111097
Igalavithana AD, Lee SE, Lee YH, Tsang DCW, Rinklebe J, Kwon EE, Ok YS (2017) Heavy metal immobilization and microbial community abundance by vegetable waste and pine cone biochar of agricultural soils. Chemosphere 174:593–603. https://doi.org/10.1016/j.chemosphere.2017.01.148
Irshad MK, Ibrahim M, Noman A, Shang J, Mahmood A, Mubashir M, Khoo KS, Ng HS, Show PL (2022) Elucidating the impact of goethite-modified biochar on arsenic mobility, bioaccumulation in paddy rice (Oryza sativa L.) along with soil enzyme activities. Process Saf Environ Prot 160:958–967. https://doi.org/10.1016/j.psep.2022.02.069
Jain S, Khare P, Mishra D, Shanker K, Singh P, Singh RP, Das P, Yadav R, Saikia BK, Baruah B (2020) Biochar aided aromatic grass [Cymbopogon martini (Roxb.) Wats.] vegetation: a sustainable method for stabilization of highly acidic mine waste. J Hazard Mater 390:121799. https://doi.org/10.1016/j.jhazmat.2019.121799
Jain S, Mishra D, Khare P, Yadav V, Deshmukh Y, Meena A (2016) Impact of biochar amendment on enzymatic resilience properties of mine spoils. Sci Total Environ 544:410–421. https://doi.org/10.1016/j.scitotenv.2015.11.011
Ji M, Wang X, Usman M, Liu F, Dan Y, Zhou L, Campanaro S, Luo G, Sang W (2022) Effects of different feedstocks-based biochar on soil remediation: a review. Environ Pollut 294:118655. https://doi.org/10.1016/j.envpol.2021.118655
Jones DL (1998) Organic acids in the rhizosphere–a critical review. Plant Soil 205:25–44. https://doi.org/10.1023/A:1004356007312
Jones DL, Nguyen C, Finlay RD (2009) Carbon flow in the rhizosphere: carbon trading at the soil–root interface. Plant Soil 321:5–33. https://doi.org/10.1007/s11104-009-9925-0
Jones DL, Rousk J, Edwards-Jones G, DeLuca TH, Murphy DV (2012) Biochar-mediated changes in soil quality and plant growth in a three year field trial. Soil Biol Biochem 45:113–124. https://doi.org/10.1016/j.soilbio.2011.10.012
Joseph S, Camps-Arbestain M, Lin Y, Munroe P, Chia C, Hook J, Van Zwieten L, Kimber S, Cowie A, Singh B (2010) An investigation into the reactions of biochar in soil. Soil Research 48:501–515. https://doi.org/10.1071/SR10009
Kalam S, Basu A, Ahmad I, Sayyed R, El-Enshasy HA, Dailin DJ, Suriani NL (2020) Recent understanding of soil acidobacteria and their ecological significance: a critical review. Front Microbiol 11:580024. https://doi.org/10.3389/fmicb.2020.580024
Kamilova F, Kravchenko LV, Shaposhnikov AI, Azarova T, Makarova N, Lugtenberg B (2006) Organic acids, sugars, and L-tryptophane in exudates of vegetables growing on stonewool and their effects on activities of rhizosphere bacteria. Mol Plant Microbe Interact 19:250–256. https://doi.org/10.1094/MPMI-19-0250
Kaplan DI, Xu C, Huang S, Lin Y, Tolic N, Roscioli-Johnson KM, Santschi PH, Jaffe PR (2016) Unique organic matter and microbial properties in the rhizosphere of a wetland soil. Environ Sci Technol 50:4169–4177. https://doi.org/10.1021/acs.est.5b05165
Kappler A, Wuestner ML, Ruecker A, Harter J, Halama M, Behrens S (2014) Biochar as an electron shuttle between bacteria and Fe (III) minerals. Environ Sci Technol Lett 1:339–344. https://doi.org/10.1021/ez5002209
Khan AZ, Khan S, Khan MA, Alam M, Ayaz T (2020) Biochar reduced the uptake of toxic heavy metals and their associated health risk via rice (Oryza sativa L.) grown in Cr-Mn mine contaminated soils. Environ Technol Innov 17:100590. https://doi.org/10.1016/j.eti.2019.100590
Khan Z, Fan X, Khan MN, Khan MA, Zhang K, Fu Y, Shen H (2022) The toxicity of heavy metals and plant signaling facilitated by biochar application: implications for stress mitigation and crop production. Chemosphere 308:136466. https://doi.org/10.1016/j.chemosphere.2022.136466
Kim H-S, Kim K-R, Kim H-J, Yoon J-H, Yang JE, Ok YS, Owens G, Kim K-H (2015) Effect of biochar on heavy metal immobilization and uptake by lettuce (Lactuca sativa L.) in agricultural soil. Environ Earth Sci 74:1249–1259. https://doi.org/10.1007/s12665-015-4116-1
Klüpfel L, Keiluweit M, Kleber M, Sander M (2014) Redox properties of plant biomass-derived black carbon (biochar). Environ Sci Technol 48:5601–5611. https://doi.org/10.1021/es500906d
Kolb SE, Fermanich KJ, Dornbush ME (2009) Effect of charcoal quantity on microbial biomass and activity in temperate soils. Soil Sci Soc Am J 73:1173–1181. https://doi.org/10.2136/sssaj2008.0232
Kuzyakov Y, Blagodatskaya E (2015) Microbial hotspots and hot moments in soil: concept & review. Soil Biol Biochem 83:184–199. https://doi.org/10.1016/j.soilbio.2015.01.025
Lahori AH, Zhang Z, Shaheen SM, Rinklebe J, Guo Z, Li R, Mahar A, Wang Z, Ren C, Mi S (2019) Mono-and co-applications of Ca-bentonite with zeolite, Ca-hydroxide, and tobacco biochar affect phytoavailability and uptake of copper and lead in a gold mine-polluted soil. J Hazard Mater 374:401–411. https://doi.org/10.1016/j.jhazmat.2019.04.057
Lan Y, Du Q, Tang C, Cheng K, Yang F (2021) Application of typical artificial carbon materials from biomass in environmental remediation and improvement: a review. J Environ Manage 296:113340. https://doi.org/10.1016/j.jenvman.2021.113340
Lanna-Filho R, Souza RM, Alves E (2017) Induced resistance in tomato plants promoted by two endophytic bacilli against bacterial speck. Trop Plant Pathol 42:96–108. https://doi.org/10.1007/s40858-017-0141-9
Latawiec AE, Strassburg BB, Junqueira AB, Araujo E, de Moraes LFD, Pinto HA, Castro A, Rangel M, Malaguti GA, Rodrigues AF (2019) Biochar amendment improves degraded pasturelands in Brazil: environmental and cost-benefit analysis. Sci Rep 9:1–12. https://doi.org/10.1038/s41598-019-47647-x
Layet C, Auffan M, Santaella C, Chevassus-Rosset C, Montes M, Ortet P, Barakat M, Collin B, Legros S, Bravin MN (2017) Evidence that soil properties and organic coating drive the phytoavailability of cerium oxide nanoparticles. Environ Sci Technol 51:9756–9764. https://doi.org/10.1021/acs.est.7b02397
Lebrun M, Miard F, Nandillon R, Léger J-C, Hattab-Hambli N, Scippa GS, Bourgerie S, Morabito D (2018) Assisted phytostabilization of a multicontaminated mine technosol using biochar amendment: early stage evaluation of biochar feedstock and particle size effects on As and Pb accumulation of two Salicaceae species (Salix viminalis and Populus euramericana). Chemosphere 194:316–326. https://doi.org/10.1016/j.chemosphere.2017.11.113
Lehmann J, Kuzyakov Y, Pan G, Ok YS (2015) Biochars and the plant-soil interface. Plant Soil 395:1–5. https://doi.org/10.1007/s11104-015-2658-3
Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, Crowley D (2011) Biochar effects on soil biota—a review. Soil Biol Biochem 43:1812–1836. https://doi.org/10.1016/j.soilbio.2011.04.022
Li J, Chen J, Lu T, Wang Y, Zhang H, Shang Z, Li D, Zhou Y, Qi Z (2019a) Effects of low-molecular weight organic acids on the transport of graphene oxide nanoparticles in saturated sand columns. Sci Total Environ 666:94–102. https://doi.org/10.1016/j.scitotenv.2019.02.242
Li J, Huang B, Wang Q, Li Y, Fang W, Han D, Yan D, Guo M, Cao A (2017) Effects of fumigation with metam-sodium on soil microbial biomass, respiration, nitrogen transformation, bacterial community diversity and genes encoding key enzymes involved in nitrogen cycling. Sci Total Environ 598:1027–1036. https://doi.org/10.1016/j.scitotenv.2017.02.058
Li J, Xu Y, Wang L, Li F (2019b) Heavy metal occurrence and risk assessment in dairy feeds and manures from the typical intensive dairy farms in China. Environ Sci Pollut Res 26:6348–6358. https://doi.org/10.1007/s11356-019-04125-1
Li L, Wang S, Li X, Li T, He X, Tao Y (2018) Effects of Pseudomonas chenduensis and biochar on cadmium availability and microbial community in the paddy soil. Sci Total Environ 640–641:1034–1043. https://doi.org/10.1016/j.scitotenv.2018.05.287
Li X, Dong S, Yao Y, Shi W, Wu M, Xu H (2016) Inoculation of bacteria for the bioremediation of heavy metals contaminated soil by Agrocybe aegerita. RSC Adv 6:65816–65824. https://doi.org/10.1039/C6RA11767H
Li X, Song Y, Wang F, Bian Y, Jiang X (2019c) Combined effects of maize straw biochar and oxalic acid on the dissipation of polycyclic aromatic hydrocarbons and microbial community structures in soil: a mechanistic study. J Hazard Mater 364:325–331. https://doi.org/10.1016/j.jhazmat.2018.10.041
Li Y, Shao J, Wang X, Deng Y, Yang H, Chen H (2014) Characterization of modified biochars derived from bamboo pyrolysis and their utilization for target component (Furfural) adsorption. Energy Fuels 28:5119–5127. https://doi.org/10.1021/ef500725c
Li Z-R, Wang J-X, An L-Z, Tan J-B, Zhan F-D, Wu J, Zu Y-Q (2019d) Effect of root exudates of intercropping Vicia faba and Arabis alpina on accumulation and sub-cellular distribution of lead and cadmium. Int J Phytorem 21:4–13. https://doi.org/10.1080/15226514.2018.1523867
Lin H, Wang Z, Liu C, Dong Y (2022) Technologies for removing heavy metal from contaminated soils on farmland: a review. Chemosphere 305:135457. https://doi.org/10.1016/j.chemosphere.2022.135457
Liu G, Chen L, Jiang Z, Zheng H, Dai Y, Luo X, Wang Z (2017a) Aging impacts of low molecular weight organic acids (LMWOAs) on furfural production residue-derived biochars: porosity, functional properties, and inorganic minerals. Sci Total Environ 607–608:1428–1436. https://doi.org/10.1016/j.scitotenv.2017.07.046
Liu H, Dong N, Chai S, Wang F, Liu T, Jiang S (2015) Effects of eco-char on controlling wheat root-rot and the mechanism of renovating soil health. Acta Phytophylacica Sinica 42:504–509. https://doi.org/10.13802/j.cnki.zwbhxb.2015.04.004
Liu H, Liu L, Zhang K, Geng R (2023) Effect of combining biogeotextile and vegetation cover on the protection of steep slope of highway in northern China: a runoff plot experiment. Int J Sedim Res 38:387–395. https://doi.org/10.1016/j.ijsrc.2022.11.003
Liu H, Xu F, Xie Y, Wang C, Zhang A, Li L, Xu H (2018a) Effect of modified coconut shell biochar on availability of heavy metals and biochemical characteristics of soil in multiple heavy metals contaminated soil. Sci Total Environ 645:702–709. https://doi.org/10.1016/j.scitotenv.2018.07.115
Liu N, Liao P, Zhang J, Zhou Y, Luo L, Huang H, Zhang L (2020a) Characteristics of denitrification genes and relevant enzyme activities in heavy-metal polluted soils remediated by biochar and compost. Sci Total Environ 739:139987. https://doi.org/10.1016/j.scitotenv.2020.139987
Liu Q, Sheng Y, Wang W, Li C, Zhao G (2020b) Remediation and its biological responses of Cd contaminated sediments using biochar and minerals with nanoscale zero-valent iron loading. Sci Total Environ 713:136650. https://doi.org/10.1016/j.scitotenv.2020.136650
Liu S, Lu Y, Yang C, Liu C, Ma L, Dang Z (2017b) Effects of modified biochar on rhizosphere microecology of rice (Oryza sativa L.) grown in As-contaminated soil. Environ Sci Pollut Res 24:23815–23824. https://doi.org/10.1007/s11356-017-9994-1
Liu S, Pu S, Deng D, Huang H, Yan C, Ma H, Razavi BS (2020c) Comparable effects of manure and its biochar on reducing soil Cr bioavailability and narrowing the rhizosphere extent of enzyme activities. Environ Int 134:105277. https://doi.org/10.1016/j.envint.2019.105277
Liu X, Wang X, Xu T, Ma H, Xia T (2022a) The combined application of gamma-PGA-producing bacteria and biochar reduced the content of heavy metals and improved the quality of tomato (Solanum lycopersicum L.). Environ Sci Pollut Res 29:88938–88950. https://doi.org/10.1007/s11356-022-21842-2
Liu Y-R, Delgado-Baquerizo M, Bi L, Zhu J, He J-Z (2018b) Consistent responses of soil microbial taxonomic and functional attributes to mercury pollution across China. Microbiome 6:1–12. https://doi.org/10.1186/s40168-018-0572-7
Liu Y, Dai Q, Jin X, Dong X, Peng J, Wu M, Liang N, Pan B, Xing B (2018c) Negative impacts of biochars on urease activity: high pH, heavy metals, polycyclic aromatic hydrocarbons, or free radicals? Environ Sci Technol 52:12740–12747. https://doi.org/10.1021/acs.est.8b00672
Liu Y, Tie B, Li Y, Lei M, Wei X, Liu X, Du H (2018d) Inoculation of soil with cadmium-resistant bacterium Delftia sp. B9 reduces cadmium accumulation in rice (Oryza sativa L.) grains. Ecotoxicol Environ Saf 163:223–229. https://doi.org/10.1016/j.ecoenv.2018.07.081
Liu Z, Xu Z, Xu L, Buyong F, Chay TC, Li Z, Cai Y, Hu B, Zhu Y, Wang X (2022) Modified biochar: synthesis and mechanism for removal of environmental heavy metals. Carbon Res. https://doi.org/10.1007/s44246-022-00007-3
Lou L, Luo L, Wang L, Cheng G, Xu X, Hou J, Xun B, Hu B, Chen Y (2011) The influence of acid demineralization on surface characteristics of black carbon and its sorption for pentachlorophenol. J Colloid Interface Sci 361:226–231. https://doi.org/10.1016/j.jcis.2011.05.015
Lu H, Yan C, Liu J (2007) Low-molecular-weight organic acids exuded by Mangrove (Kandelia candel (L.) Druce) roots and their effect on cadmium species change in the rhizosphere. Environ Exp Bot 61:159–166. https://doi.org/10.1016/j.envexpbot.2007.05.007
Lu HP, Li ZA, Gascó G, Méndez A, Shen Y, Paz-Ferreiro J (2018) Use of magnetic biochars for the immobilization of heavy metals in a multi-contaminated soil. Sci Total Environ 622–623:892–899. https://doi.org/10.1016/j.scitotenv.2017.12.056
Lu Y, Cheng J, Wang J, Zhang F, Tian Y, Liu C, Cao L, Zhou Y (2022) Efficient remediation of cadmium contamination in soil by functionalized biochar: recent advances, challenges, and future prospects. Processes 10:1627. https://doi.org/10.3390/pr10081627
Ma H, Wei M, Wang Z, Hou S, Li X, Xu H (2020) Bioremediation of cadmium polluted soil using a novel cadmium immobilizing plant growth promotion strain Bacillus sp. TZ5 loaded on biochar. J Hazard Mater 388:122065. https://doi.org/10.1016/j.jhazmat.2020.122065
Ma X, Zarebanadkouki M, Kuzyakov Y, Blagodatskaya E, Pausch J, Razavi BS (2018) Spatial patterns of enzyme activities in the rhizosphere: effects of root hairs and root radius. Soil Biol Biochem 118:69–78. https://doi.org/10.1016/j.soilbio.2017.12.009
Makoto K, Tamai Y, Kim YS, Koike T (2009) Buried charcoal layer and ectomycorrhizae cooperatively promote the growth of Larix gmelinii seedlings. Plant Soil 327:143–152. https://doi.org/10.1007/s11104-009-0040-z
Malyan SK, Kumar SS, Fagodiya RK, Ghosh P, Kumar A, Singh R, Singh L (2021) Biochar for environmental sustainability in the energy-water-agroecosystem nexus. Renew Sustain Energy Rev 149:111379. https://doi.org/10.1016/j.rser.2021.111379
Martin BC, George SJ, Price CA, Ryan MH, Tibbett M (2014) The role of root exuded low molecular weight organic anions in facilitating petroleum hydrocarbon degradation: current knowledge and future directions. Sci Total Environ 472:642–653. https://doi.org/10.1016/j.scitotenv.2013.11.050
Martins GC, Penido ES, Alvarenga IFS, Teodoro JC, Bianchi ML, Guilherme LRG (2018) Amending potential of organic and industrial by-products applied to heavy metal-rich mining soils. Ecotoxicol Environ Saf 162:581–590. https://doi.org/10.1016/j.ecoenv.2018.07.040
Medyńska-Juraszek A, Rivier P-A, Rasse D, Joner EJ (2020) Biochar affects heavy metal uptake in plants through interactions in the rhizosphere. Appl Sci 10:5105. https://doi.org/10.3390/app10155105
Meili L, Lins PV, Zanta CLPS, Soletti JI, Ribeiro LMO, Dornelas CB, Silva TL, Vieira MGA (2019) MgAl-LDH/Biochar composites for methylene blue removal by adsorption. Appl Clay Sci 168:11–20. https://doi.org/10.1016/j.clay.2018.10.012
Mierzwa-Hersztek M, Gondek K, Klimkowicz-Pawlas A, Baran A, Bajda T (2018) Sewage sludge biochars management-Ecotoxicity, mobility of heavy metals, and soil microbial biomass. Environ Toxicol Chem 37:1197–1207. https://doi.org/10.1002/etc.4045
Moche M, Gutknecht J, Schulz E, Langer U, Rinklebe J (2015) Monthly dynamics of microbial community structure and their controlling factors in three floodplain soils. Soil Biol Biochem 90:169–178. https://doi.org/10.1016/j.soilbio.2015.07.006
Moeskops B, Sukristiyonubowo BD, Sleutel S, Herawaty L, Husen E, Saraswati R, Setyorini D, De Neve S (2010) Soil microbial communities and activities under intensive organic and conventional vegetable farming in West Java, Indonesia. Appl Soil Ecol 45:112–120. https://doi.org/10.1016/j.apsoil.2010.03.005
Moore F, González M-E, Khan N, Curaqueo G, Sanchez-Monedero M, Rilling J, Morales E, Panichini M, Mutis A, Jorquera M (2018) Copper immobilization by biochar and microbial community abundance in metal-contaminated soils. Sci Total Environ 616:960–969. https://doi.org/10.1016/j.scitotenv.2017.10.223
Moradi N, Karimi A (2021) Fe-Modified common reed biochar reduced cadmium (Cd) mobility and enhanced microbial activity in a contaminated calcareous soil. J Soil Sci Plant Nutr 21:329–340. https://doi.org/10.1007/s42729-020-00363-2
Mu Y, Jiang X, Ai Z, Jia F, Zhang L (2018) Mn2+ promoted Cr (VI) reduction with oxalic acid: The indispensable role of In-situ generated Mn3+. J Hazard Mater 343:356–363. https://doi.org/10.1016/j.jhazmat.2017.10.008
Munir MAM, Liu G, Yousaf B, Mian MM, Ali MU, Ahmed R, Cheema AI, Naushad M (2020) Contrasting effects of biochar and hydrothermally treated coal gangue on leachability, bioavailability, speciation and accumulation of heavy metals by rapeseed in copper mine tailings. Ecotoxicol Environ Saf 191:110244. https://doi.org/10.1016/j.ecoenv.2020.110244
Naeem I, Masood N, Turan V, Iqbal M (2021) Prospective usage of magnesium potassium phosphate cement combined with Bougainvillea alba derived biochar to reduce Pb bioavailability in soil and its uptake by Spinacia oleracea L. Ecotoxicol Environ Saf 208:111723. https://doi.org/10.1016/j.ecoenv.2020.111723
Nair VD, Nair PKR, Dari B, Freitas AM, Chatterjee N, Pinheiro FM (2017) Biochar in the agroecosystem-climate-change-sustainability nexus. Front Plant Sci 8:2051. https://doi.org/10.3389/fpls.2017.02051
Narayanan M, Ma Y (2022) Influences of biochar on bioremediation/phytoremediation potential of metal-contaminated soils. Front Microbiol 13:929730. https://doi.org/10.3389/fmicb.2022.929730
Niemi RM, Heiskanen I, Saarnio S (2015) Weak effects of biochar amendment on soil enzyme activities in mesocosms in bare or Phleum pratense soil. Boreal Environ Res 20:324–334
Novak JM, Ippolito JA, Ducey TF, Watts DW, Spokas KA, Trippe KM, Sigua GC, Johnson MG (2018) Remediation of an acidic mine spoil: Miscanthus biochar and lime amendment affects metal availability, plant growth, and soil enzyme activity. Chemosphere 205:709–718. https://doi.org/10.1016/j.chemosphere.2018.04.107
Novak JM, Ippolito JA, Watts DW, Sigua GC, Ducey TF, Johnson MG (2019) Biochar compost blends facilitate switchgrass growth in mine soils by reducing Cd and Zn bioavailability. Biochar 1:97–114. https://doi.org/10.1007/s42773-019-00004-7
O’Connor D, Hou D, Ok YS, Lanphear BP (2020) The effects of iniquitous lead exposure on health. Nature Sustainability 3:77–79. https://doi.org/10.1038/s41893-020-0475-z
Ojuederie OB, Babalola OO (2017) Microbial and plant-assisted bioremediation of heavy metal polluted environments: a review. Int J Environ Res Public Health 14:1504. https://doi.org/10.3390/ijerph14121504
Oliveira FR, Patel AK, Jaisi DP, Adhikari S, Lu H, Khanal SK (2017) Environmental application of biochar: current status and perspectives. Biores Technol 246:110–122. https://doi.org/10.1016/j.biortech.2017.08.122
Orwin KH, Wardle DA (2004) New indices for quantifying the resistance and resilience of soil biota to exogenous disturbances. Soil Biol Biochem 36:1907–1912. https://doi.org/10.1016/j.soilbio.2004.04.036
Paithankar JG, Saini S, Dwivedi S, Sharma A, Chowdhuri DK (2021) Heavy metal associated health hazards: an interplay of oxidative stress and signal transduction. Chemosphere 262:128350. https://doi.org/10.1016/j.chemosphere.2020.128350
Palansooriya KN, Li J, Dissanayake PD, Suvarna M, Li L, Yuan X, Sarkar B, Tsang DC, Rinklebe J, Wang X (2022) Prediction of soil heavy metal immobilization by biochar using machine learning. Environ Sci Technol 56:4187–4198. https://doi.org/10.1021/acs.est.1c08302
Palansooriya KN, Wong JTF, Hashimoto Y, Huang L, Rinklebe J, Chang SX, Bolan N, Wang H, Ok YS (2019) Response of microbial communities to biochar-amended soils: a critical review. Biochar 1:3–22. https://doi.org/10.1007/s42773-019-00009-2
Pandey B, Suthar S, Chand N (2022) Effect of biochar amendment on metal mobility, phytotoxicity, soil enzymes, and metal-uptakes by wheat (Triticum aestivum) in contaminated soils. Chemosphere 307:135889. https://doi.org/10.1016/j.chemosphere.2022.135889
Parelho C, Rodrigues A, Barreto M, Ferreira N, Garcia P (2016) Assessing microbial activities in metal contaminated agricultural volcanic soils–an integrative approach. Ecotoxicol Environ Saf 129:242–249. https://doi.org/10.1016/j.ecoenv.2016.03.019
Park JH, Lamb D, Paneerselvam P, Choppala G, Bolan N, Chung J-W (2011) Role of organic amendments on enhanced bioremediation of heavy metal (loid) contaminated soils. J Hazard Mater 185:549–574. https://doi.org/10.1016/j.jhazmat.2010.09.082
Pausch J, Kuzyakov Y (2018) Carbon input by roots into the soil: quantification of rhizodeposition from root to ecosystem scale. Glob Change Biol 24:1–12. https://doi.org/10.1111/gcb.13850
Pei J, Li J, Fang C, Zhao J, Nie M, Wu J (2020) Different responses of root exudates to biochar application under elevated CO2. Agr Ecosyst Environ 301:107061. https://doi.org/10.1016/j.agee.2020.107061
Penido ES, Martins GC, Mendes TBM, Melo LCA, do Rosário Guimarães I, Guilherme LRG (2019) Combining biochar and sewage sludge for immobilization of heavy metals in mining soils. Ecotoxicol Environ Saf 172:326–333. https://doi.org/10.1016/j.ecoenv.2019.01.110
R.K. Poole and G.M. Gadd, Eds (1989) Metal-Microbe Interactions. IRL Press, Oxford.
Pramanik K, Mitra S, Sarkar A, Maiti TK (2018) Alleviation of phytotoxic effects of cadmium on rice seedlings by cadmium resistant PGPR strain Enterobacter aerogenes MCC 3092. J Hazard Mater 351:317–329. https://doi.org/10.1016/j.jhazmat.2018.03.009
Prendergast-Miller MT, Duvall M, Sohi SP (2014) Biochar-root interactions are mediated by biochar nutrient content and impacts on soil nutrient availability. Eur J Soil Sci 65:173–185. https://doi.org/10.1111/ejss.12079
Qi X, Xiao S, Chen X, Ali I, Gou J, Wang D, Zhu B, Zhu W, Shang R, Han M (2022) Biochar-based microbial agent reduces U and Cd accumulation in vegetables and improves rhizosphere microecology. J Hazard Mater 436:129147. https://doi.org/10.1016/j.jhazmat.2022.129147
Qian L, Chen B (2014) Interactions of aluminum with biochars and oxidized biochars: implications for the biochar aging process. J Agric Food Chem 62:373–380. https://doi.org/10.1021/jf404624h
Qiao D, Lu H, Zhang X (2020) Change in phytoextraction of Cd by rapeseed (Brassica napus L.) with application rate of organic acids and the impact of Cd migration from bulk soil to the rhizosphere. Environ Pollut 267:115452. https://doi.org/10.1016/j.envpol.2020.115452
Qiu B, Tao X, Wang H, Li W, Ding X, Chu H (2021) Biochar as a low-cost adsorbent for aqueous heavy metal removal: a review. J Anal Appl Pyrolysis 155:105081. https://doi.org/10.1016/j.jaap.2021.105081
Quilliam RS, Glanville HC, Wade SC, Jones DL (2013) Life in the ‘charosphere’–Does biochar in agricultural soil provide a significant habitat for microorganisms? Soil Biol Biochem 65:287–293. https://doi.org/10.1016/j.soilbio.2013.06.004
Rahman Z (2020) An overview on heavy metal resistant microorganisms for simultaneous treatment of multiple chemical pollutants at co-contaminated sites, and their multipurpose application. J Hazard Mater 396:122682. https://doi.org/10.1016/j.jhazmat.2020.122682
Reddy K, Cutright T (2003) Nutrient amendment for the bioremediation of a chromium-contaminated soil by electrokinetics. Energy Sour 25:931–943. https://doi.org/10.1080/00908310390221318
Rees F, Germain C, Sterckeman T, Morel J-L (2015) Plant growth and metal uptake by a non-hyperaccumulating species (Lolium perenne) and a Cd-Zn hyperaccumulator (Noccaea caerulescens) in contaminated soils amended with biochar. Plant Soil 395:57–73. https://doi.org/10.1007/s11104-015-2384-x
Ren X, Sun H, Wang F, Cao F (2016) The changes in biochar properties and sorption capacities after being cultured with wheat for 3 months. Chemosphere 144:2257–2263. https://doi.org/10.1016/j.chemosphere.2015.10.132
Rilling J, Acuña J, Nannipieri P, Cassan F, Maruyama F, Jorquera M (2019) Current opinion and perspectives on the methods for tracking and monitoring plant growth-promoting bacteria. Soil Biol Biochem 130:205–219. https://doi.org/10.1016/j.soilbio.2018.12.012
Risueno Y, Petri C, Conesa HM (2021) A critical assessment on the short-term response of microbial relative composition in a mine tailings soil amended with biochar and manure compost. J Hazard Mater 417:126080. https://doi.org/10.1016/j.jhazmat.2021.126080
Roane T, Pepper I (1999) Microbial responses to environmentally toxic cadmium. Microb Ecol 38:358–364. https://doi.org/10.1007/s002489901001
Rodriguez-Franco C, Page-Dumroese DS (2021) Woody biochar potential for abandoned mine land restoration in the US: a review. Biochar 3:7–22. https://doi.org/10.1007/s42773-020-00074-y
Rovira AD (1969) Plant Root Exudates. Botanical Rev 35:35–57. https://doi.org/10.1007/BF02859887
Saletnik B, Bajcar M, Zaguła G, Czernicka M, Puchalski C (2016) Impact of the biomass pyrolysis parameters on the quality of biocarbon obtained from rape straw, rye straw and willow chips. Econtechmod Int Q J Econ Technol Model Process 5:129–134.
Samaddar S, Truu J, Chatterjee P, Truu M, Kim K, Kim S, Seshadri S, Sa T (2019) Long-term silicate fertilization increases the abundance of Actinobacterial population in paddy soils. Biol Fertil Soils 55:109–120. https://doi.org/10.1007/s00374-018-01335-6
Saquing JM, Yu Y-H, Chiu PC (2016) Wood-derived black carbon (biochar) as a microbial electron donor and acceptor. Environ Sci Technol Lett 3:62–66. https://doi.org/10.1021/acs.estlett.5b00354
Shahbaz AK, Ramzani PMA, Saeed R, Turan V, Iqbal M, Lewińska K, Abbas F, Saqib M, Tauqeer HM, Iqbal M (2019) Effects of biochar and zeolite soil amendments with foliar proline spray on nickel immobilization, nutritional quality and nickel concentrations in wheat. Ecotoxicol Environ Saf 173:182–191. https://doi.org/10.1016/j.ecoenv.2019.02.025
Shan R, Li W, Chen Y, Sun X (2020) Effects of Mg-modified biochar on the bioavailability of cadmium in soil. BioResources 15:8008. https://doi.org/10.15376/biores.15.4.8008-8025
Shelobolina E, Xu H, Konishi H, Kukkadapu R, Wu T, Blöthe M, Roden E (2012) Microbial lithotrophic oxidation of structural Fe (II) in biotite. Appl Environ Microbiol 78:5746–5752. https://doi.org/10.1128/AEM.01034-12
Shen X, Huang D-Y, Ren X-F, Zhu H-H, Wang S, Xu C, He Y-B, Luo Z-C, Zhu Q-H (2016) Phytoavailability of Cd and Pb in crop straw biochar-amended soil is related to the heavy metal content of both biochar and soil. J Environ Manage 168:245–251. https://doi.org/10.1016/j.jenvman.2015.12.019
Shi L, Dong H, Reguera G, Beyenal H, Lu A, Liu J, Yu HQ, Fredrickson JK (2016) Extracellular electron transfer mechanisms between microorganisms and minerals. Nat Rev Microbiol 14:651–662. https://doi.org/10.1038/nrmicro.2016.93
Simiele M, Sferra G, Lebrun M, Renzone G, Bourgerie S, Scippa GS, Morabito D, Scaloni A, Trupiano D (2021) In-depth study to decipher mechanisms underlying Arabidopsis thaliana tolerance to metal(loid) soil contamination in association with biochar and/or bacteria. Environ Exp Bot 182:104335. https://doi.org/10.1016/j.envexpbot.2020.104335
Siripornadulsil S, Siripornadulsil W (2013) Cadmium-tolerant bacteria reduce the uptake of cadmium in rice: potential for microbial bioremediation. Ecotoxicol Environ Saf 94:94–103. https://doi.org/10.1016/j.ecoenv.2013.05.002
Sizmur T, Fresno T, Akgul G, Frost H, Moreno-Jimenez E (2017) Biochar modification to enhance sorption of inorganics from water. Biores Technol 246:34–47. https://doi.org/10.1016/j.biortech.2017.07.082
Sokolova T (2020) Low-molecular-weight organic acids in soils: sources, composition, concentrations, and functions: a review. Eurasian Soil Sci 53:580–594. https://doi.org/10.1134/S1064229320050154
Spokas K, Novak J, Masiello C, Johnson M, Colosky E, Ippolito J, Trigo C (2014) Physical disintegration of biochar: an overlooked process. Environ Sci Technol Lett 1:326–332. https://doi.org/10.1021/ez500199t
Sterckeman T, Duquène L, Perriguey J, Morel J-L (2005) Quantifying the effect of rhizosphere processes on the availability of soil cadmium and zinc. Plant Soil 276:335–345. https://doi.org/10.1007/s11104-005-5087-x
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal ions. Free Radical Biol Med 18:321–336. https://doi.org/10.1016/0891-5849(94)00159-H
Su J, Weng X, Luo Z, Huang H, Wang W (2021) Impact of biochar on soil properties, pore water properties, and available cadmium. Bull Environ Contam Toxicol 107:544–552. https://doi.org/10.1007/s00128-021-03259-8
Sun B, Lian F, Bao Q, Liu Z, Song Z, Zhu L (2016a) Impact of low molecular weight organic acids (LMWOAs) on biochar micropores and sorption properties for sulfamethoxazole. Environ Pollut 214:142–148. https://doi.org/10.1016/j.envpol.2016.04.017
Sun C, Wang D, Shen X, Li C, Liu J, Lan T, Wang W, Xie H, Zhang Y (2020) Effects of biochar, compost and straw input on root exudation of maize (Zea mays L.): from function to morphology. Agri Ecosyst Environ 297:106952. https://doi.org/10.1016/j.agee.2020.106952
Sun Y, Sun G, Xu Y, Liu W, Liang X, Wang L (2016b) Evaluation of the effectiveness of sepiolite, bentonite, and phosphate amendments on the stabilization remediation of cadmium-contaminated soils. J Environ Manage 166:204–210. https://doi.org/10.1016/j.jenvman.2015.10.017
Ta Z, Chen HY, Ruan H (2018) Global negative effects of nitrogen deposition on soil microbes. ISME J 12:1817–1825. https://doi.org/10.1038/s41396-018-0096-y
Tan S, Narayanan M, Thu Huong DT, Ito N, Unpaprom Y, Pugazhendhi A, Lan Chi NT, Liu J (2022) A perspective on the interaction between biochar and soil microbes: a way to regain soil eminence. Environ Res 214:113832. https://doi.org/10.1016/j.envres.2022.113832
Tomczyk A, Sokołowska Z, Boguta P (2020) Biochar physicochemical properties: pyrolysis temperature and feedstock kind effects. Rev Environ Sci Biotechnol 19:191–215. https://doi.org/10.1007/s11157-020-09523-3
Trigo C, Spokas KA, Cox L, Koskinen WC (2014) Influence of soil biochar aging on sorption of the herbicides MCPA, nicosulfuron, terbuthylazine, indaziflam, and fluoroethyldiaminotriazine. J Agric Food Chem 62:10855–10860. https://doi.org/10.1021/jf5034398
Tu C, Wei J, Guan F, Liu Y, Sun Y, Luo Y (2020) Biochar and bacteria inoculated biochar enhanced Cd and Cu immobilization and enzymatic activity in a polluted soil. Environ Int 137:105576. https://doi.org/10.1016/j.envint.2020.105576
Turan V (2019) Confident performance of chitosan and pistachio shell biochar on reducing Ni bioavailability in soil and plant plus improved the soil enzymatic activities, antioxidant defense system and nutritional quality of lettuce. Ecotoxicol Environ Saf 183:109594. https://doi.org/10.1016/j.ecoenv.2019.109594
Turner BL, Haygarth PM (2005) Phosphatase activity in temperate pasture soils: Potential regulation of labile organic phosphorus turnover by phosphodiesterase activity. Sci Total Environ 344:27–36. https://doi.org/10.1016/j.scitotenv.2005.02.003
Vithanage M, Herath I, Joseph S, Bundschuh J, Bolan N, Ok YS, Kirkham MB, Rinklebe J (2017) Interaction of arsenic with biochar in soil and water: a critical review. Carbon 113:219–230. https://doi.org/10.1016/j.carbon.2016.11.032
Wan X, Li C, Parikh SJ (2020) Simultaneous removal of arsenic, cadmium, and lead from soil by iron-modified magnetic biochar. Environ Pollut 261:114157. https://doi.org/10.1016/j.envpol.2020.114157
Wan Y, Devereux R, George SE, Chen J, Gao B, Noerpel M, Scheckel K (2022) Interactive effects of biochar amendment and lead toxicity on soil microbial community. J Hazard Mater 425:127921. https://doi.org/10.1016/j.jhazmat.2021.127921
Wang B, Ma Y, Lee X, Wu P, Liu F, Zhang X, Li L, Chen M (2021a) Environmental-friendly coal gangue-biochar composites reclaiming phosphate from water as a slow-release fertilizer. Sci Total Environ 758:143664. https://doi.org/10.1016/j.scitotenv.2020.143664
Wang J, Shi L, Zhai L, Zhang H, Wang S, Zou J, Shen Z, Lian C, Chen Y (2021b) Analysis of the long-term effectiveness of biochar immobilization remediation on heavy metal contaminated soil and the potential environmental factors weakening the remediation effect: a review. Ecotoxicol Environ Saf 207:111261. https://doi.org/10.1016/j.ecoenv.2020.111261
Wang J, Wang S (2019) Preparation, modification and environmental application of biochar: a review. J Clean Prod 227:1002–1022. https://doi.org/10.1016/j.jclepro.2019.04.282
Wang J, Liu J, Zhou J, Chang L, Shen Z, Shi L, Chen Y (2022a) Rhizosphere effect on physicochemical properties and immobilization performance of biochar. Water Air Soil Pollut 233:438. https://doi.org/10.1007/s11270-022-05902-4
Wang L, Bolan NS, Tsang DCW, Hou D (2020) Green immobilization of toxic metals using alkaline enhanced rice husk biochar: effects of pyrolysis temperature and KOH concentration. Sci Total Environ 720:137584. https://doi.org/10.1016/j.scitotenv.2020.137584
Wang L, O’Connor D, Rinklebe J, Ok YS, Tsang DC, Shen Z, Hou D (2020b) Biochar aging: mechanisms, physicochemical changes, assessment, and implications for field applications. Environ Sci Technol 54:14797–14814. https://doi.org/10.1021/acs.est.0c04033
Wang L, Ok YS, Tsang DCW, Alessi DS, Rinklebe J, Wang H, Mašek O, Hou R, O’Connor D, Hou D, Nicholson F (2020c) New trends in biochar pyrolysis and modification strategies: feedstock, pyrolysis conditions, sustainability concerns and implications for soil amendment. Soil Use Manag 36:358–386. https://doi.org/10.1111/sum.12592
Wang Q, Awasthi MK, Zhao J, Ren X, Li R, Wang Z, Wang M, Zhang Z (2017) Improvement of pig manure compost lignocellulose degradation, organic matter humification and compost quality with medical stone. Biores Technol 243:771–777. https://doi.org/10.1016/j.biortech.2017.07.021
Wang R, Wei S, Jia P, Liu T, Hou D, Xie R, Lin Z, Ge J, Qiao Y, Chang X (2019) Biochar significantly alters rhizobacterial communities and reduces Cd concentration in rice grains grown on Cd-contaminated soils. Sci Total Environ 676:627–638. https://doi.org/10.1016/j.scitotenv.2019.04.133
Wang Y, Liu Y, Zhan W, Zheng K, Wang J, Zhang C, Chen R (2020d) Stabilization of heavy metal-contaminated soils by biochar: challenges and recommendations. Sci Total Environ 729:139060. https://doi.org/10.1016/j.scitotenv.2020.139060
Wang Y, Wang C, Xiong J, Zhang Q, Shang J (2022) Effects of low molecular weight organic acids on aggregation behavior of biochar colloids at acid and neutral conditions. Biochar 4:20. https://doi.org/10.1007/s42773-022-00142-5
Wang Y, Xu Y, Li D, Tang B, Man S, Jia Y, Xu H (2018) Vermicompost and biochar as bio-conditioners to immobilize heavy metal and improve soil fertility on cadmium contaminated soil under acid rain stress. Sci Total Environ 621:1057–1065. https://doi.org/10.1016/j.scitotenv.2017.10.121
Wang Z, Liu G, Zheng H, Li F, Ngo HH, Guo W, Liu C, Chen L, Xing B (2015) Investigating the mechanisms of biochar’s removal of lead from solution. Biores Technol 177:308–317. https://doi.org/10.1016/j.biortech.2014.11.077
Wang Z, Shan X-q, Zhang S (2002) Comparison between fractionation and bioavailability of trace elements in rhizosphere and bulk soils. Chemosphere 46:1163–1171. https://doi.org/10.1016/S0045-6535(01)00206-5
Warnock DD, Lehmann J, Kuyper TW, Rillig MC (2007) Mycorrhizal responses to biochar in soil–concepts and mechanisms. Plant Soil 300:9–20. https://doi.org/10.1007/s11104-007-9391-5
Wei T, Li X, Li H, Gao H, Guo J, Li Y, Ren X, Hua L, Jia H (2022) The potential effectiveness of mixed bacteria-loaded biochar/activated carbon to remediate Cd, Pb co-contaminated soil and improve the performance of pakchoi plants. J Hazard Mater 435:129006. https://doi.org/10.1016/j.jhazmat.2022.129006
Williamson P (2016) Emissions reduction: scrutinize CO2 removal methods. Nature 530:153–155. https://doi.org/10.1038/530153a
Wu B, Wang Z, Zhao Y, Gu Y, Wang Y, Yu J, Xu H (2019a) The performance of biochar-microbe multiple biochemical material on bioremediation and soil micro-ecology in the cadmium aged soil. Sci Total Environ 686:719–728. https://doi.org/10.1016/j.scitotenv.2019.06.041
Wu C, Shi L, Xue S, Li W, Jiang X, Rajendran M, Qian Z (2019b) Effect of sulfur-iron modified biochar on the available cadmium and bacterial community structure in contaminated soils. Sci Total Environ 647:1158–1168. https://doi.org/10.1016/j.scitotenv.2018.08.087
Wu J, Li Z, Huang D, Liu X, Tang C, Parikh SJ, Xu J (2020) A novel calcium-based magnetic biochar is effective in stabilization of arsenic and cadmium co-contamination in aerobic soils. J Hazard Mater 387:122010. https://doi.org/10.1016/j.jhazmat.2019.122010
Wu S, Fang G, Wang Y, Zheng Y, Wang C, Zhao F, Jaisi DP, Zhou D (2017) Redox-Active oxygen-containing functional groups in activated carbon facilitate microbial reduction of ferrihydrite. Environ Sci Technol 51:9709–9717. https://doi.org/10.1021/acs.est.7b01854
Wu W, Liu Z, Azeem M, Guo Z, Li R, Li Y, Peng Y, Ali EF, Wang H, Wang S (2022) Hydroxyapatite tailored hierarchical porous biochar composite immobilized Cd (II) and Pb (II) and mitigated their hazardous effects in contaminated water and soil. J Hazard Mater 437:129330. https://doi.org/10.1016/j.jhazmat.2022.129330
Xia S, Huang Y, Tang J, Wang L (2019) Preparation of various thiol-functionalized carbon-based materials for enhanced removal of mercury from aqueous solution. Environ Sci Pollut Res 26:8709–8720. https://doi.org/10.1007/s11356-019-04320-0
Xiang L, Harindintwali JD, Wang F, Redmile-Gordon M, Chang SX, Fu Y, He C, Muhoza B, Brahushi F, Bolan N, Jiang X, Ok YS, Rinklebe J, Schaeffer A, Zhu YG, Tiedje JM, Xing B (2022) Integrating biochar, bacteria, and plants for sustainable remediation of soils contaminated with organic pollutants. Environ Sci Technol 56:16546–16566. https://doi.org/10.1021/acs.est.2c02976
Xiang Y, Deng Q, Duan H, Guo Y (2017) Effects of biochar application on root traits: a meta-analysis. GCB Bioenergy 9:1563–1572. https://doi.org/10.1111/gcbb.12449
Xu S, Adhikari D, Huang R, Zhang H, Tang Y, Roden E, Yang Y (2016) Biochar-facilitated microbial reduction of hematite. Environ Sci Technol 50:2389–2395. https://doi.org/10.1021/acs.est.5b05517
Xu Z, Xu X, Tao X, Yao C, Tsang DCW, Cao X (2019) Interaction with low molecular weight organic acids affects the electron shuttling of biochar for Cr(VI) reduction. J Hazard Mater 378:120705. https://doi.org/10.1016/j.jhazmat.2019.05.098
Yang Q, Mašek O, Zhao L, Nan H, Yu S, Yin J, Li Z, Cao X (2021a) Country-level potential of carbon sequestration and environmental benefits by utilizing crop residues for biochar implementation. Appl Energy 282:116275. https://doi.org/10.1016/j.apenergy.2020.116275
Yang T, Xu Y, Huang Q, Sun Y, Liang X, Wang L, Qin X, Zhao L (2021b) An efficient biochar synthesized by iron-zinc modified corn straw for simultaneously immobilization Cd in acidic and alkaline soils. Environ Pollut 291:118129. https://doi.org/10.1016/j.envpol.2021.118129
Yang X, Wan Y, Zheng Y, He F, Yu Z, Huang J, Wang H, Ok YS, Jiang Y, Gao B (2019) Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: a critical review. Chem Eng J 366:608–621. https://doi.org/10.1016/j.cej.2019.02.119
Yao Q, Liu J, Yu Z, Li Y, Jin J, Liu X, Wang G (2017) Three years of biochar amendment alters soil physiochemical properties and fungal community composition in a black soil of northeast China. Soil Biol Biochem 110:56–67. https://doi.org/10.1016/j.soilbio.2017.03.005
Yin D-X, Fang W, Guan D-X, Williams PN, Moreno-Jimenez E, Gao Y, Zhao F-J, Ma LQ, Zhang H, Luo J (2020) Localized intensification of arsenic release within the emergent rice rhizosphere. Environ Sci Technol 54:3138–3147. https://doi.org/10.1021/acs.est.9b04819
Yuan J-H, Xu R-K, Zhang H (2011) The forms of alkalis in the biochar produced from crop residues at different temperatures. Biores Technol 102:3488–3497. https://doi.org/10.1016/j.biortech.2010.11.018
Yuan MM, Guo X, Wu L, Zhang Y, Xiao N, Ning D, Shi Z, Zhou X, Wu L, Yang Y (2021a) Climate warming enhances microbial network complexity and stability. Nat Clim Chang 11:343–348. https://doi.org/10.1038/s41558-021-00989-9
Yuan X, Suvarna M, Low S, Dissanayake PD, Lee KB, Li J, Wang X, Ok YS (2021b) Applied machine learning for prediction of CO2 adsorption on biomass waste-derived porous carbons. Environ Sci Technol 55:11925–11936. https://doi.org/10.1021/acs.est.1c01849
Yuan X, Li J, Lim JY, Zolfaghari A, Alessi DS, Wang Y, Wang X, Ok YS (2023a) Machine learning for heavy metal removal from water: recent advances and challenges. ACS EST Water. https://doi.org/10.1021/acsestwater.3c00215
Yuan X, Cao Y, Li J, Patel AK, Dong C-D, Jin X, Gu C, Yip ACK, Tsang DCW, Ok YS (2023b) Recent advancements and challenges in emerging applications of biochar-based catalysts. Biotechnol Adv 67:108181. https://doi.org/10.1016/j.biotechadv.2023.108181
Yuan Y, Bolan N, Prévoteau A, Vithanage M, Biswas JK, Ok YS, Wang H (2017) Applications of biochar in redox-mediated reactions. Biores Technol 246:271–281. https://doi.org/10.1016/j.biortech.2017.06.154
Zhalnina K, Louie KB, Hao Z, Mansoori N, da Rocha UN, Shi S, Cho H, Karaoz U, Loqué D, Bowen BP (2018) Dynamic root exudate chemistry and microbial substrate preferences drive patterns in rhizosphere microbial community assembly. Nat Microbiol 3:470–480. https://doi.org/10.1038/s41564-018-0129-3
Zhang B, Zhang J, Liu Y, Shi P, Wei G (2018a) Co-occurrence patterns of soybean rhizosphere microbiome at a continental scale. Soil Biol Biochem 118:178–186. https://doi.org/10.1016/j.soilbio.2017.12.011
Zhang C, Shan B, Zhu Y, Tang W (2018b) Remediation effectiveness of Phyllostachys pubescens biochar in reducing the bioavailability and bioaccumulation of metals in sediments. Environ Pollut 242:1768–1776. https://doi.org/10.1016/j.envpol.2018.07.091
Zhang C, Sui J, Li J, Tang Y, Cai W (2012) Efficient removal of heavy metal ions by thiol-functionalized superparamagnetic carbon nanotubes. Chem Eng J 210:45–52. https://doi.org/10.1016/j.cej.2012.08.062
Zhang G, Liu X, Gao M, Song Z (2020a) Effect of Fe-Mn-Ce modified biochar composite on microbial diversity and properties of arsenic-contaminated paddy soils. Chemosphere 250:126249. https://doi.org/10.1016/j.chemosphere.2020.126249
Zhang J, Zhao W, Zhang H, Wang Z, Fan C, Zang L (2018c) Recent achievements in enhancing anaerobic digestion with carbon- based functional materials. Bioresour Technol 266:555–567. https://doi.org/10.1016/j.biortech.2018.07.076
Zhang L, Guan Y (2022) Microbial investigations of new hydrogel-biochar composites as soil amendments for simultaneous nitrogen-use improvement and heavy metal immobilization. J Hazard Mater 424:127154. https://doi.org/10.1016/j.jhazmat.2021.127154
Zhang M, Song G, Gelardi DL, Huang L, Khan E, Mašek O, Parikh SJ, Ok YS (2020b) Evaluating biochar and its modifications for the removal of ammonium, nitrate, and phosphate in water. Water Res 186:116303. https://doi.org/10.1016/j.watres.2020.116303
Zhang W, Niu J, Morales VL, Chen X, Hay AG, Lehmann J, Steenhuis TS (2010) Transport and retention of biochar particles in porous media: effect of pH, ionic strength, and particle size. Ecohydrology 3:497–508. https://doi.org/10.1002/eco.160
Zhang X, Wells M, Niazi NK, Bolan N, Shaheen S, Hou D, Gao B, Wang H, Rinklebe J, Wang Z (2022) Nanobiochar-rhizosphere interactions: implications for the remediation of heavy-metal contaminated soils. Environ Pollut 299:118810. https://doi.org/10.1016/j.envpol.2022.118810
Zhang X, Yu J, Huang Z, Li H, Liu X, Huang J, Zhuo R, Wu Z, Qin X, Gao Y, Wang M, Zhu Y (2021) Enhanced Cd phytostabilization and rhizosphere bacterial diversity of Robinia pseudoacacia L. by endophyte Enterobacter sp. YG-14 combined with sludge biochar. Sci Total Environ 787:147660. https://doi.org/10.1016/j.scitotenv.2021.147660
Zhao X, Dai J, Teng Z, Yuan J, Wang G, Luo W, Ji X, Hu W, Li M (2022) Immobilization of cadmium in river sediment using phosphate solubilizing bacteria coupled with biochar-supported nano-hydroxyapatite. J Clean Prod 348:131221. https://doi.org/10.1016/j.jclepro.2022.131221
Zhao X, Sun Y, Huang J, Wang H, Tang D (2020) Effects of soil heavy metal pollution on microbial activities and community diversity in different land use types in mining areas. Environ Sci Pollut Res 27:20215–20226. https://doi.org/10.1007/s11356-020-08538-1
Zhou X, Zhang X, Ma C, Wu F, Jin X, Dini-Andreote F, Wei Z (2022) Biochar amendment reduces cadmium uptake by stimulating cadmium-resistant PGPR in tomato rhizosphere. Chemosphere 307:136138. https://doi.org/10.1016/j.chemosphere.2022.136138
Zhu H, Wu J, Jiang Y, Jin J, Zhou W, Wang Y, Han G, Zhao Y, Cheng B (2016) Genomewide analysis of MATE-type gene family in maize reveals microsynteny and their expression patterns under aluminum treatment. J Genet 95:691–704. https://doi.org/10.1007/s12041-016-0686-2
Zhu X, Chen B, Zhu L, Xing B (2017) Effects and mechanisms of biochar-microbe interactions in soil improvement and pollution remediation: a review. Environ Pollut 227:98–115. https://doi.org/10.1016/j.envpol.2017.04.032
Zhu Y, Lv X, Li T, Zhong M, Song J, Wang H, Cui J (2022a) Cotton straw biochar and compound Bacillus biofertilizer reduce Cd stress on cotton root growth by regulating root exudates and antioxidant enzymes system. Front Plant Sci 13:1051935. https://doi.org/10.3389/fpls.2022.1051935
Zhu Y, Ma J, Chen F, Yu R, Hu G, Zhang S (2020) Remediation of soil polluted with Cd in a postmining area using thiourea-modified biochar. Int J Environ Res Public Health 17:7654. https://doi.org/10.3390/ijerph17207654
Zhu Y, Zhong M, Li W, Qiu Y, Wang H, Lv X (2022b) Cotton straw biochar and Bacillus compound biofertilizer decreased Cd migration in alkaline soil: Insights from relationship between soil key metabolites and key bacteria. Ecotoxicol Environ Saf 232:113293. https://doi.org/10.1016/j.ecoenv.2022.113293
Zornoza R, Acosta J, Faz A, Bååth E (2016) Microbial growth and community structure in acid mine soils after addition of different amendments for soil reclamation. Geoderma 272:64–72. https://doi.org/10.1016/j.geoderma.2016.03.007
Acknowledgements
Not applicable.
Funding
This work was funded by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA23010000). This work was funded by the State Scholarship Fund of China Scholarship Council (File No. 202104910265); This work was funded by the Fundamental Research Funds for the Central Universities. This work was carried out with the support of the Cooperative Research Program for Agriculture Science and Technology Development (Project No. PJ01475801) from Rural Development Administration, the Republic of Korea. This work was supported by the National Research Foundation of Korea (NRF) Grant funded by the Korea government (MSIT) (No. 2021R1A2C2011734). This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2021R1A6A1A10045235). This work was supported by the International Science & Technology Innovation Program of Chinese Academy of Agriculture Science (CAAS-CFSGLCA-IEDA-202302). This work was supported by the Young Scientist Exchange Program between the People’s Republic of China and the Republic of Korea.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by CF, YC, QZ, NY, XC, XY, SS, YC, and YSO. The first draft of the manuscript was written by CF and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships.
Additional information
Handling editor: Xing Yang
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fan, C., Cui, Y., Zhang, Q. et al. A critical review of the interactions between rhizosphere and biochar during the remediation of metal(loid) contaminated soils. Biochar 5, 87 (2023). https://doi.org/10.1007/s42773-023-00278-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-023-00278-y