Abstract
Fires are becoming increasingly frequent, intense, severe and prolonged worldwide, and such situation is worsening. As a result, extreme fire conditions will increase, with consequences for wildlife, including increased mass mortality and changes in trophic relationships in natural communities. This intensification is expected to be particularly pronounced in the Mediterranean ecosystems. In this scoping review, we summarized current knowledge and gaps in understanding the effects of fires on wildlife, focusing on predator–prey interactions. These interactions play a critical role in animal communities and their understanding is fundamental for appropriate management and conservation. Mammals were chosen as a model group because of their remarkable ecological role. We grouped and analysed the post-wildfire changes in the predator–prey relationships into three-time intervals: immediate, short- and long-term effects. This is relevant as vegetation restoration, by altering cover and habitat structure, may affect hunting strategies and anti-predatory behaviour. Our review showed that studies generally had several limitations, the most common of which were the lack of replication, the strong geographical bias, and the focus on few target species. Nevertheless, we could formally describe how fire affects predator–prey relationships in Mediterranean ecosystems through processes that exert different cascading effects at different times after the fire event. We encourage long-term studies on communities, including as many components of the food chain as possible, using an interdisciplinary approach, and prioritising investigations in high-risk ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the greatest global threats to wildlife is the alteration of fire regimes (Flannigan et al. 2013; Bowman et al. 2020). In fact, fires are becoming increasingly intense and frequent worldwide, even in less flammable environments, such as arctic circle and equatorial rainforests (Mariani et al. 2018). Consequently, the twenty-first century has been designated as “the age of the megafire” (Stephens et al. 2014). Recent examples include megafires in Chile, California, Portugal, Amazon, and Australia, with ~ 15 million hectares burned (Gómez-González et al. 2018; Williams et al. 2019; Brando et al. 2020; Nolan et al. 2020; Geary et al. 2022). There are likely several causes of increased fire risk, such as abandonment of agricultural land, biomass accumulation, inadequate woodland management, depopulation of the countryside and expansion of cities (Moreira and Russo 2007; Pausas et al. 2008; Seijo and Gray 2012; Fernandes et al. 2013). In addition, there is increasing evidence that global warming played a key role (Bradstock et al. 2014; Abatzoglou and Williams 2016; Jones et al. 2020).
Consistently with the fire risk increase, climate models suggest that, under all scenarios, more frequent, intense, severe, and extended fires, and, ultimately, more “megafires”, may occur (Jolly et al. 2015; Bowman et al. 2020; Geary et al. 2022). Consequently, extreme fire conditions will be more common, with consequences for wildlife, including higher mass mortality, changes in trophic relationships among species and potentially cascade effects on animal populations (Williams 2013; Nolan et al. 2020; Nimmo et al. 2021). For instance, at least 1071 animal and plant species are classified as threatened by fire regime alteration (IUCN 2019; Kelly et al. 2020).
Mediterranean regions (Box 1) are particularly prone to fires, and intensification of fire regimes have particularly increased in recent decades (Moreira and Russo 2007; Turco et al. 2018; Geary et al. 2022). Furthermore, they are likely to be increasingly at risk in the future in southern Europe (Turco et al. 2018; Abatzoglou et al. 2019; Ancillotto et al. 2021). Considering the potentially increasingly catastrophic effects of fires on Mediterranean ecosystems, they need to be fully understood.
With this scoping review, we aimed to summarize knowledge regarding effects of fire on wildlife in Mediterranean ecosystems. However, considering the broad field of study, we set specific objectives. We focused on the effects of fire on predator–prey interactions (Box 2) with the purpose to identify existing knowledge gaps, particularly the influence of successional patterns in post-fire vegetation recovery on the shaping of the predator–prey relationships. Understanding how fires affect these relationships is essential for a more detailed comprehension of the role of fires in shaping biocoenoses (Doherty et al. 2022). Predator–prey interactions have critical roles in animal communities and understanding them is fundamental for appropriate management and conservation of species (Sih et al. 1998; Matter and Mannan 2005). In the age of megafire, these aspects will become increasingly crucial for biodiversity conservation (Geary et al. 2022). Nevertheless, knowledge on this issue is very fragmented, particularly in some areas of the Mediterranean-type ecosystems, such as the Mediterranean basin, an area expected to become increasingly prone to fires (Bowman et al. 2017; Turco et al. 2018). This was an additional reason to focus the study of the effects of fires on predator–prey interactions in Mediterranean ecosystems. Mammals were chosen as the model group, given their important ecological role (Clutton-Brock 2009; Lacher et al. 2019).
Materials and methods
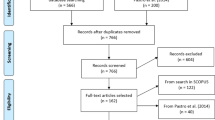
This review was carried out in accordance with the PRISMA protocol (Moher et al. 2009), following standardized steps (Fig. 2).
PRISMA flow diagram, from (Moher et al. 2009)
In April 2021 Scopus and Web of Science were scanned to identify existing papers, using the following string:
((prey* OR predator* OR predator–prey OR predation OR (predator PRE/0 prey)) AND (vertebrate* OR mammal*) AND (*fire* OR burn*)).
These sources were supplemented by further references that were considered relevant but not retrieved in the initial search. All identified papers were processed according to the PRISMA protocol, and 118 papers and books were selected.
In addition, the database was updated in December 2023 using the same approach and 70 studies were added to write this review. A total of 188 papers and books were then comprehensively analysed for this review.
Discussion
Fires and their effects on Mediterranean biocoenoses
Structure and dynamics of natural communities can be significantly impacted by various disturbances (Sousa 1984; Johnson and Miyanishi 2007). Natural disturbances include flooding (Brum and Souza 2020), insect outbreaks (Flower et al. 2013), trampling (Pellerin et al. 2006), and wildfires (Bowman et al. 2009; Pausas and Keeley 2009). Among them, wildfires are particularly relevant because they alter structure and functions of ecosystems worldwide (Bowman et al. 2009). Fires exert both direct and indirect effects on wildlife (Engstrom 2010; Puig-Gironès et al. 2018). Direct effects include injury or death, although the magnitude of the effect varies greatly among species (Doherty et al. 2015). By contrast, indirect effects mainly are changes in soil properties, vegetation structure and composition (Monamy and Fox 2000; Banks et al. 2011; Bento-Gonçalves et al. 2012), affecting habitat elements like soil fertility and structure, availability of food, shelter, and microclimate. These environmental shifts can affect prey and predator abundance, distribution, and behaviour in many ways (Letnic et al. 2004; Zwolak et al. 2012; Jorge et al. 2020), with the potential to modify predator–prey relationships (Paine 1980; Hradsky et al. 2017). In turn, predator–prey relationships are highly context-dependent relationships, which resulted mainly affected by the time elapsed since the fire and ecosystem type (Cherry et al. 2018; Doherty et al. 2022). An example is the reciprocal effects of predators on prey in terms of habitat use, abundance and/or activity (Hradsky et al. 2017; Puig-Gironès and Pons 2020; Doherty et al. 2022; Puig-Gironès 2023). For example, in a recently burned area, the stone marten Martes foina has been found to influence the foraging activity of small mammals (Puig-Gironès 2023).
In Mediterranean ecosystems, fires are a natural occurrence exerting an adapting force that has shaped the evolution of biocoenoses for millennia (Bowman et al. 2009; Pausas and Keeley 2009; Pausas and Parr 2018). As a result, flora and fauna are generally adapted to fires (Engstrom 2010; Pausas and Keeley 2014; Pausas and Parr 2018). However, implications of recent altered fire regimes (i.e. megafires) on the trophic network are unknown and could be disastrous, leading to biodiversity loss and resource depletion (Kelly et al. 2020; Geary et al. 2022; Nimmo et al. 2022). To understand the effects of fires on the predator–prey relationship in these ecosystems, it is necessary to fully understand some key elements such as their impact on the ecosystem and post-fire successional stages. The impacts of fires on animal populations, as well as on their interactions, depend mostly on the vegetation succession, which in turn is related to the time passed since the disturbance (Monamy and Fox 2000; Keeley et al. 2005). In fact, the effects of fires may persist over time, even after the flames are extinguished. Hence, as the vegetation recovery is a crucial factor to understanding the impacts of wildfires on predator–prey relationships, we summarised the known effects by grouping them into 3 phases: (i) immediate effects, i.e., from extinguishing the fire to the first few months afterwards (Lees et al. 2022), when there is lack of vegetation (Moreno and Oechel 1994; Pausas and Keeley 2009); (ii) short-term, i.e., within a few years after the fire (Spencer et al. 2022), when the undergrowth is usually dense, but the arboreal cover is lacking (Trabaud 1994; Keeley et al. 2005); and (iii) long-term, i.e., many years after the fire (Soyumert et al. 2020), when the arboreal cover is also restored (Pausas et al. 2008; Keeley et al. 2011a). In the literature, these intervals do not have distinct thresholds and may vary according to specific ecological contexts. However, in this review we have used the phenological phases of the Mediterranean vegetation following a wildfire to define the limits of the intervals. A conceptual diagram of these effects is depicted in Fig. 3.
The immediate effects
By burning vegetation, a fire may exert many different effects, depending by factors like ecosystem type and fire regime. For instance, a mixed-severity fire essentially increases landscape heterogeneity, whereas a high-severity fire decreases landscape heterogeneity, leading the burnt area to a former successional stage (Pausas and Keeley 2009). However, many plants species in Mediterranean fire-prone areas are defined “fire-adapted” species and can survive fires through various strategies, including serotiny (i.e. an induced seed release system, common in Pinus spp.) and resprouting (i.e. post-fire activation of dormant vegetative buds to regrowth) (Trabaud 1994; Konstantinidis et al. 2005; Paula et al. 2009; Keeley et al. 2011b; Pausas and Keeley 2014). The effectiveness of these strategies depends on specific conditions, such as those defined by the fire regime (Keeley et al. 2011b). Although recurrent fires can, under certain circumstances, stimulate seed germination and reduce competition (Pausas 2004; García et al. 2016), they can also be particularly deleterious, because they can affect plants at earlier life stages, which cannot reach reproductive maturity and produce a seed bank (Díaz-Delgado et al. 2002; Baeza et al. 2007; Pausas et al. 2008). These alterations influence the ensuing ecological succession, with potential to affect predator–prey relationships of species that enter it (Monamy and Fox 2000; Torre et al. 2022).
The direct effects of fires on wildlife are attributed to burning, asphyxiation, heat or physiological stress (Smith 2000; Michel et al. 2023). Specific fire events have variable impacts on wildlife, due to properties of the fire-regime, including intensity, severity, scale, season and time since fire, as well as habitat types and taxa affected (Engstrom 2010; Birtsas et al. 2012; Geary et al. 2020; Nimmo et al. 2021). However, while estimating direct effects of fire on fauna is complex and can be highly variable (Pausas and Parr 2018; Tomas et al. 2021), Jolly et al. (2022) in their systematic review have suggested that direct mortality is generally low (estimated as 3%).
In addition to the features of the fire regime, two key features that determine impacts of fire on various mammalian species are vagility and the kind of shelter employed (Smith 2000; Chia et al. 2015; Jolly et al. 2022). For instance, among small mammals, the most decisive impact is on taxa that are not very vagile and take refuge on the ground; some individuals of these species are usually killed by fires (Simons 1991). By contrast, individuals belonging to highly vagile species and/or those that shelter underground are more likely to escape or survive fires (Robinson et al. 2013; Jolly et al. 2022). Large predators for example, such as the wolf Canis lupus, are often regarded as having some degree of resistance to fire (Lino et al. 2019).
The effect of shelter location is particularly clear for small mammal assemblage. Although small mammals are generally considered very susceptible to fire, certain burrowing species suffer low direct mortality (Letnic et al. 2005). As expected, by contrast, mortality is usually higher in species that shelter above the ground (Simons 1991). The mortality of strictly arboreal species is poorly documented, especially in the Mediterranean ecosystem, but could be reduced due to their vagility (Koprowski et al. 2006).
For an herbivore, a first effect of fire is depletion in food resources (i.e., burnt vegetation). In turn, this decline can trigger a series of cascading effects, such as starvation, decreased reproductive success leading to population decline and emigration to unburnt areas, richer in food resources (Morris et al. 2011a, 2011b). However, the extent of this depends on the features of the fire regime and the trophic specialisation of the prey. After a fire, populations of specialist species, such as insectivorous, may be most affected by a decline in food availability since their main food resources may have been burnt (Recher and Christensen 1981; Sutherland and Dickman 1999; Driscoll 2007).
Another immediate effect on prey can be a reduction in cover and refugia availability, through the simplification of vegetation structure (Derrick et al. 2010; Robinson et al. 2013; Chia et al. 2015). This could lead to increased predation risk (Green and Sanecki 2006; Conner et al. 2011), then increased mortality, when prey vulnerability depends on habitat structure (Verdolin 2006). This is certainly relevant for small ground-based mammals; there is strong evidence that their mortality is positively correlated with increasing post-fire habitat simplification (Conner et al. 2011; González et al. 2022).
In the Mediterranean ecosystems, the period of highest prey mortality tends to occur shortly after the fire, especially during the first months after a high severity fire (Conner et al. 2011; Morris et al. 2011a; Leahy et al. 2016). However, specific fire regimes can also benefit certain species, based on their food habits and anti-predatory behaviour (Bleich et al. 2008; Jaffe and Isbell 2009; Bond 2015). For instance, the fox squirrel Sciurus niger seems to benefit from fires of low intensity, through stimulation of fungi and cone generation and creation of open foraging areas, that simplify squirrels movement (Karmacharya et al. 2013). Formation of open spaces also increases visibility, which can favour species that need to detect predators (e.g., bighorn sheep Ovis canadensis; Bleich et al. (2008) and vervet monkeys Cercopithecus aethiops; Jaffe & Isbell (2009).
Like their prey, predators can also become direct victims of fires, especially those of high severity and/or large size (Jolly et al. 2022). However, the impact on predatory mammals varies, and while it may be minimal for large, highly mobile species, such as large carnivores (Engstrom 2010; Lino et al. 2019), smaller predators like the stone marten may also face significant challenges (Birtsas et al. 2012). In addition, there appears to be a positive relationship between the home range size of predators and their resistance to fire. Indeed, extensive movements make them less dependent on local resources, which may be less or no longer available. Furthermore, it also allows them to exploit both resources available in the burnt area and the nearby unburnt area (Nimmo et al. 2019).
There are indications that the predator response to fire is highly species-specific as well, and it depends on factors such as environmental variables, progression of secondary succession, trophic and non-trophic interactions, and population dynamics (Smith 2018). For example, in a Mediterranean area in Macedonia, stone martens were not recorded in burned areas even three years after a fire (Birtsas et al. 2012), whereas in study areas in Spain this species was common even few months after fires (Puig-Gironès and Pons 2020; Puig-Gironès 2023), suggesting that differences even among areas of the same ecosystem type can strongly influence the predator response. Fires can increase habitat suitability for predators that could benefit from a reduction in vegetation, facilitating hunting (Conner et al. 2011; Leahy et al. 2016). In fact, several studies suggest that predators could increase their occurrence in newly burnt areas due to increased predation success (Leahy et al. 2016; Nimmo et al. 2019; Geary et al. 2020). However, if the predator species needs cover to hunt, as in the case of ambush predators, predators tend to avoid burnt areas (Eby et al. 2013; Doherty et al. 2022).
The choice of prey species also depends on their availability. Therefore, a change in availability due to fire could influence the choice of prey species towards more available species (Green and Sanecki 2006; Hradsky et al. 2017). However, even non-trophic interactions such as intra-guild competitive interactions can also shape responses of predator species to fires (Geary et al. 2020). In fact, a usual apex predators’ effect is the suppression of less competitive species (i.e. mesopredators), through competitive interactions (Palomares and Caro 1999; Geary et al. 2018). As a result, mesopredators tend to adopt prey-like behaviour in such scenarios, showing spatial and temporal avoidance of apex predators (Durant 1998; Vanak et al. 2013). Fires, by the creation of open habitat, can affect the habitat use and selection of apex predators. Consequently, mesopredators may avoid these environments, where they may also be more vulnerable to apex predators (Schuette et al. 2014; Geary et al. 2018, 2020). However, such responses may not always occur. For example, there is evidence that in certain situations mesocarnivores may not be competitively affected by apex predators, but rather may be facilitated, by capitalizing on the enhanced feeding opportunities resulting from carcasses left by apex predators (Ferretti et al. 2021; Rossa et al. 2021). The combined effects of fire, predation and competition are therefore difficult to generalise, then further research is needed.
Short-term effects
After a fire, plants gradually develop towards more mature conditions following their recovery processes (e.g., serotiny, resprouting, Keeley et al. 2011b; Pausas and Keeley 2014) and the natural stages of secondary succession (Trabaud 1994; Keeley et al. 2011a). In Mediterranean ecosystems, usually the first phase of plant recovery involves establishment of a dense undergrowth, with herbs and grasses often blooming in recently burnt vegetation (Gill and McMahon 1986; Moreno and Oechel 1994), although plant recovery differs among biomes due to biotic factors, e.g., plant community as well abiotic factors (topography, soil, rainfall and fire severity; Keeley et al. 2005; Puig-Gironès et al. 2017). The process is particularly quick in Mediterranean areas, where climatic conditions and adaptive traits of the plant species promote a rapid post-fire recovery (Trabaud 1994; Paula et al. 2009). Consequently, a few years after fire, the undergrowth could become even more dense in burnt than in unburnt areas (Torre and Díaz 2004).
For mammals, a widely accepted post-fire successional model is the Habitat Accommodation model (HAM; Fox 1982; Monamy and Fox 2000, 2010; Torre and Díaz 2004; Swan et al. 2015; Torre et al. 2022), derived from facilitation, tolerance, and inhibition models (Connell and Slatyer 1977). The HAM can be used to predict the successional order of mammalian community following a fire, considering the essential habitat requirements of each species. This model proposes that an early species enters the post-fire succession and reaches maximum density when the habitat is at an optimal level for that species in terms of vegetation. As post-fire succession proceeds, vegetation will change, becoming less suitable for early species, which decrease in density, whereas other more suitable seral species will enter succession (Monamy and Fox 2000). For instance, in a pioneering study conducted in Lakes National Park (Australia), species like the New Holland mouse Pseudomys novaehollandiae, the eastern chestnut mouse P. gracilicaudatus, the house mouse Mus domesticus, and the slender-tailed dunnart Sminthopsis murina reached higher abundances in the early post-fire stages, when their habitat requirements were fulfilled, and they were then considered early successional species (Fox 1982; Monamy and Fox 2000). In contrast, species that required advanced forest maturity, such as squirrels, are considered late successional species (Koprowski et al. 2006; Mazzamuto et al. 2020). The key point of HAM is therefore the change in vegetation, and particularly its structure. Habitat Accommodation model has had strong experimental support in the small mammal assemblage (Torre and Díaz 2004; Swan et al. 2015; Torre et al. 2022), a fundamental prey group for many predators (Hanski et al. 2001; Posłluszny et al. 2007; Castañeda et al. 2022). Usually, the earliest small mammals are ground-foraging species, with herbivorous and/or granivorous food habits (Haim and Izhaki 1994, 2000), well suited to early-successional vegetation.
In addition to the vegetation structure, plant species also have important roles. For instance, black-footed rats Mesembriomys gouldii have been found to be particularly dependent on Cycas sp. in post-fire Australian forests, and the post-fire loss of these plants could lead to local black-footed rats extinction (Woinarski et al. 2005), with potential cascading effects on the whole ecosystem (Nimmo et al. 2019; Geary et al. 2020). Moreover, another hypothesis has been postulated, the Green Magnet Hypothesis (GMH; Archibald et al. 2005; Jorge et al. 2020), to explain why herbivores are inclined to visit recently burnt areas. According to GMH, herbivores are attracted to these areas because of the favourable feeding opportunities. However, this attraction takes perceived predation risk into account; consequently, herbivores do not always follow the GMH (Cherry et al. 2017, 2018). Thus, when the GHM is followed, density of certain prey species may become higher in burnt than in unburnt areas. In the Mediterranean basin, a good example is the small mammals assemblage (Torre and Díaz 2004; Puig-Gironès and Pons 2020). In a study in Catalonia (Spain) the authors reported that this assemblage was also particularly developed around boundaries of the burnt area (Puig-Gironès et al. 2018), where the habitat structure and cover were more elaborate and thicker than in the inner parts of the burnt area.
As predators tend to adapt to the availability and distribution of their prey, changes in prey abundance can trigger shifts in predator behaviour and foraging strategies (Nimmo et al. 2019; Doherty et al. 2022, 2023). Predators may concentrate their hunting efforts in burned areas with higher prey densities, resulting in locally increased predatory frequencies (Geary et al. 2020; Puig-Gironès and Pons 2020; Doherty et al. 2022). For example, Puig-Gironès and Pons (2020) report that 15 months after fires there was an increment of rodent foraging activity, and 6 months later, an increasing of red fox occurrence. Conversely, occurrence of the stone marten was not affected by small mammals’ abundance, presumably due to its generalist food-habits (Virgós et al. 2010; Puig-Gironès and Pons 2020). Shifts in predatory frequencies can have significant ecological effects. In fact, they may lead to a decline in prey populations, potentially triggering cascading effects throughout the food web. Changes in predatory behaviour might affect interspecific interactions, ultimately shaping community structure (Geary et al. 2018, 2020; Doherty et al. 2022). Furthermore, alteration of prey availability may also affect predators’ locomotory activity pattern, as reported for the red fox from 1 to 3 years after a fire (Birtsas et al. 2012; Hradsky 2020; Spencer et al. 2022).
Long-term effects
In Mediterranean ecosystems, in the years following a fire, that post-fire successional outcomes can be highly variable due to factors such as fire intensity and recurrence, plant reproductive strategy and seed maturation (Retana et al. 2002; Pausas et al. 2008; Keeley et al. 2011b; Pausas and Keeley 2014). High intensity fires can lead to massive community collapse, resulting in prolonged recovery periods. Conversely, low intensity fires can facilitate more rapid vegetation recovery, through both survival and reproductive mechanisms and nutrient supply (Moreno and Oechel 1994; Keeley et al. 2011a; Caon et al. 2014). On the other hand, recurrent fires could be particularly detrimental to forest regeneration, leading to prolongated forest recovery time, or even to its conversion into a successional shrubland (Díaz-Delgado et al. 2002; Baeza et al. 2007; Pausas et al. 2008). However, usually succession proceeds with increasing tree cover and decreasing herbaceous one (Trabaud 1994; Keeley et al. 2011a). Consequently, strictly terrestrial and herbivorous animal species will be at least partially replaced by arboreal, omnivorous and/or insectivorous species, or those that feed on trees grown in the further seral stages (Prodon et al. 1987; Arrizabalaga et al. 1993; Torre et al. 2022). These changes are consistent with Fox’s Habitat Accommodation Model, as their succession follows a predictable pattern of vegetation succession. For example, the response of meso/large mammals to a wildfire in Turkey was studied over several years (Soyumert et al. 2020). Sites burnt since more than 30 years had lower species richness than those burnt 13 years before, but supported species that are usually absent in the short term after fire, such as brown bears Ursus arctos and wild goats Capra aegagrus. Similar results for small mammals in terms of specific richness were found by Torre et al. (2022) in Spain. If the predicted successional scenario is respected, for prey, predation risk and pressure could theoretically increase until the local abundance of the prey return to pre-fire levels. However, experimental evidence is still limited (Torre and Díaz 2004; Torre et al. 2022).
Predators preferring forested habitats with high tree cover (e.g., Martes spp.) are expected to be more common in these late-successional systems (Bond 2015).
As ecological restoration proceeds, the system could return to the pre-fire stage, or to a newly evolved state that reflects the restoration of the ecosystem (Chakraborty and Li 2009; Keeley et al. 2011a; Holl 2020). However, long-term data are the least available, probably due to the challenges of maintaining consistent monitoring efforts over time and funding constraints.
Drawing general conclusions is challenging, because of the intricate nature of ecological processes. For instance, there is variability in the post-fire response depending on the type of ecosystem affected and the features of fire regime (Lindenmayer et al. 2016; Lewis et al. 2022). In addition, the potential occurrence of other disturbances over time (e.g. additional wildfires, establishment of human activities), and long-term cascading effects due to successional dynamics and trophic interactions increase the complexity of the analysis (Lindenmayer et al. 2016; Geary et al. 2018; Smith 2018).
Concluding remarks and future perspectives
Limitations of studies on post-fire effect
Despite the many studies on responses of wildlife to fires, our scoping review highlighted many limitations. Most studies focus on fire-responses of a single species or taxon (e.g., Jaffe and Isbell 2009; Eby et al. 2013; Sokos et al. 2016; Lino et al. 2019; Mazzamuto et al. 2020; Nalliah et al. 2022). This is a standard starting point for ecological studies. However, to fully comprehend the ecological effects of fires, a more comprehensive viewpoint that considers interactions among different taxa at the community level is necessary. The selection of target species is contingent upon the specific context, objectives and hypotheses formulated. Ideally, species from different functional groups should be included, e.g., at least one each of apex predator, mesopredator, and prey species.
Immediate and short-term studies can provide immediate insights about the fire effects on ecosystems, and long-term ones are needed to understand ecosystem dynamics over time. However, while the first ones abound (e.g., Hradsky et al. 2017; Puig-Gironès and Pons 2020; Spencer et al. 2022), the second ones are scarce (e.g., Sokos et al. 2016; Soyumert et al. 2020). Effects at one or two years after the fire are quite well-known for specific groups (e.g. rodents, Haim and Izhaki (1994); Puig-Gironès et al. (2018); Mazzamuto et al. (2020)), but long-term effects are poorly characterized. In order to mitigate the effects of fires, which will increasingly impact wildlife, it should be of primary importance to study the post-fire recovery period, which is highly variable depending on factors such as the ecosystem type and climate (Keeley et al. 2005, 2011a; Puig-Gironès et al. 2017).
Most of the studies were carried in North America and Australia (Geary et al. 2020; González et al. 2022). This geographical bias poses a significant challenge since it restricts the findings applicability to other continents. However, the studies on North American (e.g., Bleich et al. 2008; Schuette et al. 2014) and Australian (e.g., Hradsky 2020; Geary et al. 2022) Mediterranean ecosystems are useful for predicting the outcome of Mediterranean ecosystems on other continents, due to their shared temperate climate (Moreno and Oechel 1994; Keeley et al. 2011a). Nevertheless, it is imperative to underline the intrinsic diversity of the ecosystem’s biocoenosis of each geographical region. These differences highlight the need to conduct additional research on the unique ecological conditions of each region. Regretfully, there are very limited studies about mammalian post-fire responses in the Mediterranean basin (Haim et al. 1996; Soyumert et al. 2010, 2020; Birtsas et al. 2012), of which only three studies (Torre and Díaz 2004; Puig-Gironès and Pons 2020; Puig-Gironès 2023), conducted in Spain, included predator–prey relationships.
Another limitation in studying unpredictable phenomena is that many studies are not replicated under the same or even similar conditions. In fact, replicating the same fire regime features (e.g. spatial extent, fire intensity) and environmental conditions poses significant difficulties in achieving experimental control and standardisation. In addition, ethical considerations and safety concerns may limit the ability to conduct fire experiments. Consequently, as wildfire studies are often opportunistic and lack pre-treatment data, the conclusions that can be drawn are usually limited to that single system. By contrast, prescribed fires are manageable, allowing for study through appropriate sampling design (e.g. Before-After Control-Impact, BACI). Unlike wildfires, prescribed fires are less intense, severe and extensive (Whelan 1995; Fernandes et al. 2013), then their impact on wildlife is very different from that of wildfires (Pastro et al. 2011). As a result, studying the effects of prescribed fire cannot serve as a surrogate for understanding the effects of wildfire, although it depends by factors like ecosystem and taxa affected (Converse et al. 2006; Morris et al. 2011a; Hradsky et al. 2017).
Mechanisms to be disclosed
The actual mechanisms behind the observed effects is usually unknown (Kelly et al. 2012; Zwolak et al. 2012; Leahy et al. 2016). For instance, although it is frequently well recognised that animal abundance varies in response to fires (Torre and Díaz 2004; Zwolak et al. 2012; Jorge et al. 2020), the demographic drivers behind these changes are often not clear. In fire ecology, wildlife studies are typically disconnected dots, due to the wide range of fire characteristics and ecological contexts studied. For instance, each study has a distinct set of features (natural/prescribed fire, low/high severity and/or intensity, different scale, seasonality, biogeographical region, species investigated). In addition, responses of a certain taxon to a fire in a certain part of the world could be completely different to that of the same taxon in a different biogeographical region (Geary et al. 2020), or after a fire with different severity (Bond 2015; Chia et al. 2015; Lewis et al. 2022). For example, fire severity effects have been observed in taxa such as rodents (Diffendorfer et al. 2012), bats (Buchalski et al. 2013), and marsupials (Chia et al. 2015). Therefore, with such specific information it is difficult to draw a general picture from which to attempt to draw overarching conclusions about the effects of fires on wildlife and predict its effects on prey and predators (Hradsky et al. 2017; Puig-Gironès and Pons 2020). To predict effects of fires on animal populations driven by trophic relationships, e.g., predator–prey relationship, is challenging, also considering the complexity of the predator–prey relationship itself. However, fully identifying the effects of fires on these relationships will be crucial to understanding effects of fires on wildlife, given their role in shaping population dynamics.
Establishing an international network that links together all available information from each future fire could be a possible solution. To date, information is still relatively scarce due to some of the limitations highlighted, but it is hoped that future studies will fill these gaps by elucidating the role of each factor in affecting the ecological system impacted by fire, as well as how these factors may interact and reverberate on the relationships among the different components of the system. For example, by clarifying the effects of individual components of the fire regime on vegetation regrowth, habitat availability, species mortality and community dynamics, taking into account differences among different biocoenoses, is essential to this understanding. By analysing the fine interactions among these factors, we could gain a thorough understanding of their combined influence on many ecosystem components, ultimately leading to a deeper knowledge of how fire affects ecosystems. With this understanding, or a sort of framework for the impact assessment, it may be possible to identify patterns that make the effects of fires more predictable. Given the current megafires outbreak and their devastating effects on ecosystems around the world (Nolan et al. 2020; Geary et al. 2022), the development of predictive models of megafire occurrence and behaviour should be a priority task. Combining data and knowledge from various ecosystems and areas, such a network could improve our knowledge of megafire dynamics and make it easier the development of effective megafire management plans.
Applying this strategy, we suggest that future studies should characterise in detail some key aspects of the fires and their potential effects (Whelan et al. 2002; Keeley et al. 2011a; Geary et al. 2020) (Table 1). Among these, the fire regime, including the extent of the impacted area, the severity of the various zones and the intensity of the fire should be prioritized (Keeley 2009; Lewis et al. 2022). Additional aspects such as soil type, climate zone, topography, other disturbance sources and affected biocoenoses should also be characterised. As there is strong evidence that effects of fire on vegetation have a key role in the entire trophic network (Moreno and Oechel 1994; Keeley et al. 2011a), effects of the fire on the vegetation should be studied in detail, to assess the potential impacts on the animal community through alteration of key components and structures such as food resources and cover (Monamy and Fox 2000; Geary et al. 2018; Torre et al. 2022). A interdisciplinary approach to fire studies is therefore recommended. This recommendation stems from the recognition that fires are multifaceted phenomena with far-reaching consequences. Each aspect considered—from the physical characteristics of the fire to its ecological consequences—requires expertise from different disciplines (Whelan 1995; Pausas and Keeley 2009; Moyo 2022). Studies on components such as soil and vegetation should be primarily carried out as key ecological drivers of primary productivity. In fact, wildfires, through the burning of organic matter can reduce soil fertility, triggering cascading effects on the vegetation recovery (Keeley et al. 2011a; Caon et al. 2014). For this purpose, remote sensing monitoring systems could complement field studies, by monitor changes in vegetation over time (Parks et al. 2014; Allison et al. 2016). On the other hand, animal studies should involve community studies, including as many components of the food chain as possible, e.g., primary and secondary consumers, plus meso- and apex-predators (Geary et al. 2020; Doherty et al. 2022). In fact, the effects of fires extend beyond individual species and may affect the entire ecological communities. By adopting a community perspective, researchers could investigate the dynamic interactions that occur among different species within an ecosystem and gain a broad overview that can be insightful in understanding post-fire dynamics (Whelan 1995; Morin 2011; Moyo 2022).
More emphasis should be placed also on changes in trophic and non-trophic relationships among species, which play key roles in shaping post-fire community dynamics (Geary et al. 2018; Smith 2018; Doherty et al. 2022). Trophic relationships, such as predator–prey dynamics, are known drivers of ecosystem structure and function (Estes et al. 2011; Terborgh and Estes 2013). Understanding how fire affects them is therefore essential for proper management and conservation of wildlife (Sih et al. 1998; Matter and Mannan 2005). In addition, non-trophic relationships, including competition, also have profound effects on community composition (Mayfield and Levine 2010; Morin 2011), although the effects of fire on these are still little investigated (Geary et al. 2018; McHugh et al. 2022). By prioritising the study of both trophic and non-trophic interactions, we can achieve a more comprehensive understanding of the multifaceted effects of fire on ecosystems, also encompassing the intricate web of relationships that collectively shape post-fire community dynamics (Leahy et al. 2016; Geary et al. 2018; Smith 2018; Doherty et al. 2022; Puig-Gironès 2023). Such research should follow ecological succession over time, to understand the actual impacts of a fire on wildlife, which most likely does not end within a few years of the event (Kelly et al. 2011; Soyumert et al. 2020). Thus, studies should begin soon after the fire and be continued for several years and possibly decades thereafter.
Finally, more attention should be paid to less studied but high-risk areas, such as the Mediterranean basin, where, although there has been considerable research on the effects of fires on several taxa (e.g., Santos and Cheylan 2013; Puig-Gironès et al. 2017, 2023; Ancillotto et al. 2021), there are surprisingly few studies investigating their effects on ecological interactions within mammalian communities (Torre and Díaz 2004; Puig-Gironès and Pons 2020; Puig-Gironès 2023).
As scientific research is often limited by economic resources, it may not always be possible to implement the proposed solutions. However, a more feasible alternative might be to choose one or more indicator taxa and study them intensively around the world (Geary et al. 2020). Studying the same taxa in different ecosystems allows data to be collected under a variety of environmental conditions. This may help to determine the role of specific factors and to generalise ecological patterns, thereby improving our comprehension of global ecosystem dynamics. A suitable indicator species could be the red fox, due to its extraordinary ecological plasticity (Geary et al. 2020; Garcês and Pires 2021). This species is widespread in almost the entire northern hemisphere, plus parts of Australia, including three of the five Mediterranean areas: the Mediterranean basin, Australia and California. The red fox is both autochthonous predator (northern hemisphere) and invasive predator (southern hemisphere), consequently this relevant conservation element can also be assessed. As a mesopredator, it can play the trophic role of predator or prey, and it can undergo competitive interactions with apex predators (Geary et al. 2018; Garcês and Pires 2021; Rossa et al. 2021). Finally, there are already data on relationships between red foxes and fires, e.g., in Australia (Hradsky 2020; Nalliah et al. 2022; Spencer et al. 2022; Doherty et al. 2023). As a highly opportunistic species, it tends to adapt very well to fire-prone areas (Hradsky 2020; Doherty et al. 2023).
However, this approach has its drawbacks. Variability in habitat types, species interactions and environmental factors across ecosystems can introduce complexity and limitations to data interpretation. In addition, the influence of other factors, such as human disturbance and habitat fragmentation (Sousa 1984; Baeza et al. 2007; Ancillotto et al. 2021), cannot be underestimated, as they can confound research results and make it difficult to extrapolate findings to larger geographic scales.
Together with the red fox, the small mammal assemblages, as a key prey group, could be studied. Small mammals are distributed worldwide and are among the taxa for which we know most about the effects of fires on recovery (Banks et al. 2011; Puig-Gironès et al. 2018; Hale et al. 2022) and dynamics (Arrizabalaga et al. 1993; Haim and Izhaki 1994; Torre et al. 2022). Furthermore, their specific composition varies greatly at geographical scale, and include from early to late successional species (Fox 1982; Monamy and Fox 2010).
In summary, post-fire community research must be given top priority in order to gain a more comprehensive understanding of the ecological effects of fires on wildfire in Mediterranean ecosystems. It is necessary to apply an interdisciplinary approach to the study of these communities and give emphasis on the comprehension of ecological interactions. In the case this is not feasible, we suggest investigating at least the red fox/small mammals system as soon as the fire occurs, and to continue the research for as long as possible.
We believe that a large-scale application of this framework would yield significant insights of how fire affects predator–prey relationships in Mediterranean ecosystems. Given the ongoing of climate change and intensification of fire regimes, this knowledge may prove valuable in the future for managing and predicting changes in mammal communities. In this way, researchers can lead the way for more effective approaches to mitigate the ecological impacts of wildfires and preserve the biodiversity of these threatened ecosystems for future generations.
References
Abatzoglou JT, Williams AP (2016) Impact of anthropogenic climate change on wildfire across western US forests. PNAS 113:11770–11775
Abatzoglou JT, Williams AP, Barbero R (2019) Global emergence of anthropogenic climate change in fire weather indices. Geophys Res Lett 46:326–336
Allison RS, Johnston JM, Craig G, Jennings S (2016) Airborne optical and thermal remote sensing for wildfire detection and monitoring. Sensors 16:1310
Ancillotto L, Fichera G, Pidinchedda E, Veith M, Kiefer A, Mucedda M, Russo D (2021) Wildfires, heatwaves and human disturbance threaten insular endemic bats. Biodivers Conserv 30:4401–4416
Archibald S, Bond WJ, Stock WD, Fairbanks DHK (2005) Shaping the landscape: fire–grazer interactions in an African savanna. Ecol Appl 15:96–109
Arrizabalaga A, Montagud E, Fons R (1993) Post-fire succession in small mammal communities in the Montserrat Massif (Catalonia, Spain). In: Trabaud L, Prodon R (eds) Fire in Mediterranean ecosystems. Commission of the European Communities, Brussels, pp 281–291
Baeza MJ, Valdecantos A, Alloza JA, Vallejo VR (2007) Human disturbance and environmental factors as drivers of long-term post-fire regeneration patterns in Mediterranean forests. J Veg Sci 18:243–252. https://doi.org/10.1111/j.1654-1103.2007.tb02535.x
Banks SC, Dujardin M, McBurney L, Blair D, Barker M, Lindenmayer DB (2011) Starting points for small mammal population recovery after wildfire: recolonisation or residual populations? Oikos 120:26–37. https://doi.org/10.1111/j.1600-0706.2010.18765.x
Barbosa P, Castellanos I (2005) Ecology of predator-prey interactions. Oxford University Press, New York
Bento-Gonçalves A, Vieira A, Úbeda X, Martin D (2012) Fire and soils: key concepts and recent advances. Geoderma 191:3–13
Birtsas P, Sokos C, Exadactylos S (2012) Carnivores in burned and adjacent unburned areas in a Mediterranean ecosystem. Mammalia 76:407–415. https://doi.org/10.1515/mammalia-2011-0070
Bleich VC, Johnson HE, Holl SA, Konde L, Torres SG, Krausman PR (2008) Fire history in a chaparral ecosystem: implications for conservation of a native ungulate. Rangeland Ecol Manag 61:571–579
Bond ML (2015) Mammals and mixed- and high-severity fire. In: DellaSala D, Hanson C (eds) The ecological importance of mixed-severity fires: nature’s phoenix. Elsevier, pp 89–117
Bowman DMJS, Balch JK, Artaxo P, Bond WJ, Carlson JM, Cochrane MA, D’Antonio CM, DeFries RS, Doyle JC, Harrison SP, Johnston FH, Keeley JE, Krawchuk MA, Kull CA, Marston JB, Moritz MA, Prentice IC, Roos CI, Scott AC, Swetnam TW, van der Werf GR, Pyne SJ (2009) Fire in the Earth system. Science 324:481–484. https://doi.org/10.1126/science.1163886
Bowman DM, Williamson GJ, Abatzoglou JT, Kolden CA, Cochrane MA, Smith AM (2017) Human exposure and sensitivity to globally extreme wildfire events. Nat Ecol Evol 1:1–6
Bowman DM, Kolden CA, Abatzoglou JT, Johnston FH, van der Werf GR, Flannigan M (2020) Vegetation fires in the Anthropocene. Nat Rev Earth Environ 1:500–515
Bradstock R, Penman T, Boer M, Price O, Clarke H (2014) Divergent responses of fire to recent warming and drying across south-eastern Australia. Global Change Biol 20:1412–1428
Brando P, Macedo M, Silvério D, Rattis L, Paolucci L, Alencar A, Coe M, Amorim C (2020) Amazon wildfires: scenes from a foreseeable disaster. Flora 268:151609
Brockway DG, Gatewood RG, Paris RB (2002) Restoring fire as an ecological process in shortgrass prairie ecosystems: initial effects of prescribed burning during the dormant and growing seasons. J Environ Manag 65:135–152
Brown JS, Laundré JW, Gurung M (1999) The ecology of fear: optimal foraging, game theory, and trophic interactions. J Mammal 80:385–399
Brum HD, Souza AF (2020) Flood disturbance and shade stress shape the population structure of açaí palm Euterpe precatoria, the most abundant Amazon species. Botany 98:147–160
Buchalski MR, Fontaine JB, Heady PA III, Hayes JP, Frick WF (2013) Bat response to differing fire severity in mixed-conifer forest California, USA. PLoS ONE 8:e57884
Caon L, Vallejo VR, Ritsema CJ, Geissen V (2014) Effects of wildfire on soil nutrients in Mediterranean ecosystems. Earth Sci Rev 139:47–58
Castañeda I, Doherty TS, Fleming PA, Stobo-Wilson AM, Woinarski JC, Newsome TM (2022) Variation in red fox Vulpes vulpes diet in five continents. Mammal Rev 52:328–342
Chakraborty A, Li B-L (2009) Post-fire ecological succession: a theoretical modeling framework. Acta Ecol Sin 29:7–12
Cherry MJ, Warren RJ, Conner LM (2017) Fire-mediated foraging tradeoffs in white-tailed deer. Ecosphere 8:e01784
Cherry MJ, Chandler RB, Garrison EP, Crawford DA, Kelly BD, Shindle DB, Godsea KG, Miller KV, Conner LM (2018) Wildfire affects space use and movement of white-tailed deer in a tropical pyric landscape. For Ecol Manag 409:161–169
Chia EK, Bassett M, Nimmo DG, Leonard SW, Ritchie EG, Clarke MF, Bennett AF (2015) Fire severity and fire-induced landscape heterogeneity affect arboreal mammals in fire-prone forests. Ecosphere 6:1–14
Clutton-Brock T (2009) Structure and function in mammalian societies. Phil Trans R Soc B 364:3229–3242. https://doi.org/10.1098/rstb.2009.0120
Connell JH, Slatyer RO (1977) Mechanisms of succession in natural communities and their role in community stability and organization. Am Nat 111:1119–1144. https://doi.org/10.1086/283241
Conner LM, Castleberry SB, Derrick AM (2011) Effects of mesopredators and prescribed fire on hispid cotton rat survival and cause-specific mortality. J Wildl Manag 75:938–944. https://doi.org/10.1002/jwmg.110
Converse SJ, Block WM, White GC (2006) Small mammal population and habitat responses to forest thinning and prescribed fire. For Ecol Manag 228:263–273. https://doi.org/10.1016/j.foreco.2006.03.006
Cozzi G, Börger L, Hutter P, Abegg D, Beran C, McNutt JW, Ozgul A (2015) Effects of trophy hunting leftovers on the ranging behaviour of large carnivores: a case study on spotted hyenas. PLoS ONE 10:e0121471
Dawkins R, Krebs JR (1979) Arms races between and within species. Proc R Soc Ser B Biol Sci 205:489–511
Derrick AM, Conner LM, Castleberry SB (2010) Effects of prescribed fire and predator exclusion on refuge selection by Peromyscus gossypinus Le Conte (Cotton Mouse). Southeast Nat 9:773–780. https://doi.org/10.1656/058.009.0411
Di Castri F, Mooney HA (2012) Mediterranean type ecosystems: origin and structure. Springer-Verlag, New York
Díaz-Delgado R, Lloret F, Pons X, Terradas J (2002) Satellite evidence of decreasing resilience in Mediterranean plant communities after recurrent wildfires. Ecology 83:2293–2303
Diffendorfer J, Fleming GM, Tremor S, Spencer W, Beyers JL (2012) The role of fire severity, distance from fire perimeter and vegetation on post-fire recovery of small-mammal communities in chaparral. Int J Wildland Fire 21:436–448. https://doi.org/10.1071/WF10060
Doherty TS, Davis RA, van Etten EJB (2015) A game of cat-and-mouse: microhabitat influences rodent foraging in recently burnt but not long unburnt shrublands. J Mammal 96:324–331. https://doi.org/10.1093/jmammal/gyv034
Doherty TS, Geary WL, Jolly CJ, Macdonald KJ, Miritis V, Watchorn DJ, Cherry MJ, Conner LM, González TM, Legge SM, Ritchie EG, Stawski C, Dickman CR (2022) Fire as a driver and mediator of predator–prey interactions. Biol Rev 97:1539–1558. https://doi.org/10.1111/brv.12853
Doherty TS, Watchorn DJ, Miritis V, Pestell AJ, Geary WL (2023) Cats, foxes and fire: quantitative review reveals that invasive predator activity is most likely to increase shortly after fire. Fire Ecol 19:1–14
Driscoll DA (2007) The diverse impacts of grazing, fire and weeds: how ecological theory can inform conservation management. In: Lindenmayer DB, Hobbs RJ (eds) Managing and designing landscapes for conservation. Blackwell Publishing Ltd, Oxford, UK, pp 111–130
Durant SM (1998) Competition refuges and coexistence: an example from Serengeti carnivores. J Anim Ecol 67:370–386
Eby S, Mosser A, Swanson A, Packer C, Ritchie M (2013) The impact of burning on lion Panthera leo habitat choice in an African savanna. Curr Zool 59:335–339
Engstrom RT (2010) First-order fire effects on animals: review and recommendations. Fire Ecol 6:115–130. https://doi.org/10.4996/fireecology.0601115
Estes JA, Terborgh J, Brashares JS, Power ME, Berger J, Bond WJ, Carpenter SR, Essington TE, Holt RD, Jackson JBC, Marquis RJ, Oksanen L, Oksanen T, Paine RT, Pikitch EK, Ripple WJ, Sandin SA, Scheffer M, Schoener TW, Shurin JB, Sinclair ARE, Soulé ME, Virtanen R, Wardle DA (2011) Trophic downgrading of planet earth. Science 333:301–306. https://doi.org/10.1126/science.1205106
Fernandes PM, Davies GM, Ascoli D, Fernández C, Moreira F, Rigolot E, Stoof CR, Vega JA, Molina D (2013) Prescribed burning in southern Europe: developing fire management in a dynamic landscape. Front Ecol Environ 11:e4–e14. https://doi.org/10.1890/120298
Ferretti F, Pacini G, Belardi I, Ten Cate B, Sensi M, Oliveira R, Rossa M, Burrini L, Lovari S (2021) Recolonizing wolves and opportunistic foxes: interference or facilitation? Biol J Linn Soc 132:196–210
Flannigan M, Cantin AS, De Groot WJ, Wotton M, Newbery A, Gowman LM (2013) Global wildland fire season severity in the 21st century. For Ecol Manag 294:54–61
Flower CE, Knight KS, Gonzalez-Meler MA (2013) Impacts of the emerald ash borer (Agrilus planipennis Fairmaire) induced ash (Fraxinus spp.) mortality on forest carbon cycling and successional dynamics in the eastern United States. Biol Invasions 15:931–944
Fox BJ (1982) Fire and mammalian secondary succession in an Australian coastal heath. Ecology 63:1332–1341. https://doi.org/10.2307/1938861
Garcês A, Pires I (2021) Secrets of the astute red fox (Vulpes vulpes, Linnaeus, 1758): an inside-ecosystem secret agent serving one health. Environments 8:103
García Y, Castellanos MC, Pausas JG (2016) Fires can benefit plants by disrupting antagonistic interactions. Oecologia 182:1165–1173
Geary WL, Ritchie EG, Lawton JA, Healey TR, Nimmo DG (2018) Incorporating disturbance into trophic ecology: fire history shapes mesopredator suppression by an apex predator. J Appl Ecol 55:1594–1603. https://doi.org/10.1111/1365-2664.13125
Geary WL, Doherty TS, Nimmo DG, Tulloch AIT, Ritchie EG (2020) Predator responses to fire: a global systematic review and meta-analysis. J Anim Ecol 89:955–971. https://doi.org/10.1111/1365-2656.13153
Geary WL, Buchan A, Allen T, Attard D, Bruce MJ, Collins L, Ecker TE, Fairman TA, Hollings T, Loeffler E, Muscatello A, Parkes D, Thomson J, White M, Kelly E (2022) Responding to the biodiversity impacts of a megafire: acase study from south-eastern Australia’s Black Summer. Divers Distrib 28:463–478. https://doi.org/10.1111/ddi.13292
Gill AM, McMahon A (1986) A postfire chronosequence of cone, follicle and seed production in Banksia ornata. Aust J Bot 34:425–433
Gómez-González S, Ojeda F, Fernandes PM (2018) Portugal and Chile: longing for sustainable forestry while rising from the ashes. Environ Sci Policy 81:104–107
González TM, González-Trujillo JD, Muñoz A, Armenteras D (2022) Effects of fire history on animal communities: a systematic review. Ecol Processes 11:1–11
Green K, Sanecki G (2006) Immediate and short-term responses of bird and mammal assemblages to a subalpine wildfire in the Snowy Mountains, Australia. Austral Ecol 31:673–681. https://doi.org/10.1111/j.1442-9993.2006.01629.x
Haim A (2002) Fire size and location in forest restoration: the use of small mammal community structure for bioindication. In: Trabaud L, Prodon R (eds) Fire and biological processes. Bachuys, Brussels-Luxembourg, pp 249–254
Haim A, Izhaki I (1994) Changes in rodent community during recovery from fire: relevance to conservation. Biodivers Conserv 3:573–585. https://doi.org/10.1007/BF00114202
Haim A, Izhaki I (2000) The effect of different treatments on the community composition of small mammals in a post-fire pine forest. J Mediterr Ecol 1:249–257
Haim A, Izhaki I, Golan A (1996) Rodent species diversity in pine forests recovering from fire. Isr J Ecol Evol 42:353–359
Hale S, Mendoza L, Yeatman T, Cooke R, Doherty T, Nimmo D, White JG (2022) Evidence that post-fire recovery of small mammals occurs primarily via in situ survival. Divers Distrib 28(3):404–416. https://doi.org/10.1111/ddi.13283
Halle S (2000) Ecological relevance of daily activity patterns. In: Halle S, Stenseth NC (eds) Activity patterns in small mammals. Springer, New York, pp 67–90
Hanski I, Henttonen H, Ki EK, Oksanen L, Turchin P (2001) Small-rodent dynamics and predation. Ecology 82:16
Holl K (2020) Primer of ecological restoration. Island Press, Washington, D.C., USA
Hradsky BA (2020) Conserving Australia’s threatened native mammals in predator-invaded, fire-prone landscapes. Wildl Res 47:1–15. https://doi.org/10.1071/WR19027
Hradsky BA, Mildwaters C, Ritchie EG, Christie F, Di Stefano J (2017) Responses of invasive predators and native prey to a prescribed forest fire. J Mammal 98:835–847. https://doi.org/10.1093/jmammal/gyx010
International Union for the Conservation of Nature (IUCN) (2019) The IUCN red list of threatened species, version 2019–3. www.iucnredlist.org
Jaffe KE, Isbell LA (2009) After the fire: benefits of reduced ground cover for vervet monkeys (Cercopithecus aethiops). Am J Primatol 71:252–260. https://doi.org/10.1002/ajp.20644
Johnson EA, Miyanishi K (2007) Plant disturbance ecology: the process and the response. Elsevier/AP, Amsterdam Boston
Jolly WM, Cochrane MA, Freeborn PH, Holden ZA, Brown TJ, Williamson GJ, Bowman DM (2015) Climate-induced variations in global wildfire danger from 1979 to 2013. Nat Commun 6:1–11
Jolly CJ, Dickman CR, Doherty TS, Eeden LM, Geary WL, Legge SM, Woinarski JCZ, Nimmo DG (2022) Animal mortality during fire. Global Change Biol 28:2053–2065. https://doi.org/10.1111/gcb.16044
Jones MW, Smith A, Betts R, Canadell JG, Prentice IC, Le Quéré C (2020) Climate change increases the risk of wildfires. ScienceBrief 116:117
Jorge MH, Garrison EP, Conner LM, Cherry MJ (2020) Fire and land cover drive predator abundances in a pyric landscape. For Ecol Manag 461:117939. https://doi.org/10.1016/j.foreco.2020.117939
Karmacharya B, Hostetler JA, Conner LM, Morris G, Oli MK (2013) The influence of mammalian predator exclusion, food supplementation, and prescribed fire on survival of Glaucomys volans. J Mammal 94:672–682. https://doi.org/10.1644/12-MAMM-A-071.1
Kats LB, Dill LM (1998) The scent of death: chemosensory assessment of predation risk by prey animals. Écoscience 5:361–394. https://doi.org/10.1080/11956860.1998.11682468
Keeley JE (2009) Fire intensity, fire severity and burn severity: a brief review and suggested usage. Int J Wildland Fire 18:116–126. https://doi.org/10.1071/WF07049
Keeley JE, Fotheringham CJ, Baer-Keeley M (2005) Determinants of postfire recovery and succession in Mediterranean-climate shrublands of California. Ecol Appl 15:1515–1534
Keeley JE, Bond WJ, Bradstock RA, Pausas JG, Rundel PW (2011a) Fire in Mediterranean ecosystems: ecology, evolution and management. Cambridge University Press, Cambridge
Keeley JE, Pausas JG, Rundel PW, Bond WJ, Bradstock RA (2011b) Fire as an evolutionary pressure shaping plant traits. Trends Plant Sci 16:406–411. https://doi.org/10.1016/j.tplants.2011.04.002
Kelly LT, Nimmo DG, Spence-Bailey LM, Haslem A, Watson SJ, Clarke MF, Bennett AF (2011) Influence of fire history on small mammal distributions: insights from a 100-year post-fire chronosequence. Divers Distrib 17:462–473
Kelly LT, Nimmo DG, Spence-Bailey LM, Taylor RS, Watson SJ, Clarke MF, Bennett AF (2012) Managing fire mosaics for small mammal conservation: a landscape perspective. J Appl Ecol 49:412–421
Kelly LT, Giljohann KM, Duane A, Aquilué N, Archibald S, Batllori E, Bennett AF, Buckland ST, Canelles Q, Clarke MF (2020) Fire and biodiversity in the Anthropocene. Science 370:1–10
Konstantinidis P, Tsiourlis G, Galatsidas S (2005) Effects of wildfire season on the resprouting of kermes oak (Quercus coccifera L.). For Ecol Manag 208:15–27
Koprowski JL, Leonard KM, Zugmeyer CA, Jolley JL (2006) Direct effects of fire on endangered mount graham red squirrels. Southwest Nat 51:59–63. https://doi.org/10.1894/0038-4909(2006)51[59:DEOFOE]2.0.CO;2
Kronfeld-Schor N, Dayan T (2003) Partitioning of time as an ecological resource. Annu Rev Ecol Evol Syst 34:153–181
Kuijper DPJ, De Kleine C, Churski M, Van Hooft P, Bubnicki J, Jędrzejewska B (2013) Landscape of fear in Europe: wolves affect spatial patterns of ungulate browsing in Białowieża Primeval Forest, Poland. Ecography 36:1263–1275. https://doi.org/10.1111/j.1600-0587.2013.00266.x
Lacher TE Jr, Davidson AD, Fleming TH, Gómez-Ruiz EP, McCracken GF, Owen-Smith N, Peres CA, Vander Wall SB (2019) The functional roles of mammals in ecosystems. J Mammal 100:942–964
Laundré JW (2010) Behavioral response races, predator–prey shell games, ecology of fear, and patch use of pumas and their ungulate prey. Ecology 91:2995–3007
Lawes MJ, Murphy BP, Fisher A, Woinarski JCZ, Edwards AC, Russell-Smith J (2015) Small mammals decline with increasing fire extent in northern Australia: evidence from long-term monitoring in Kakadu National Park. Int J Wildland Fire 24:712–722. https://doi.org/10.1071/WF14163
Leahy L, Legge SM, Tuft K, McGregor HW, Barmuta LA, Jones ME, Johnson CN (2016) Amplified predation after fire suppresses rodent populations in Australia’s tropical savannas. Wildl Res 42:705–716. https://doi.org/10.1071/WR15011
Lees DM, Watchorn DJ, Driscoll DA, Doherty TS (2022) Microhabitat selection by small mammals in response to fire. Aust J Zool 69:67–79
Letnic M, Dickman CR, Tischler MK, Tamayo B, Beh C-L (2004) The responses of small mammals and lizards to post-fire succession and rainfall in arid Australia. J Arid Environ 59:85–114
Letnic M, Tamayo B, Dickman CR (2005) The responses of mammals to La Niña (El Niño Southern Oscillation)–associated rainfall, predation, and wildfire in central Australia. J Mammal 86:689–703
Lewis JS, LeSueur L, Oakleaf J, Rubin ES (2022) Mixed-severity wildfire shapes habitat use of large herbivores and carnivores. For Ecol Manag 506:119933. https://doi.org/10.1016/j.foreco.2021.119933
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640
Lindenmayer DB, Burton PJ, Franklin JF (2012) Salvage logging and its ecological consequences. Island Press, Washington
Lindenmayer DB, Blanchard W, MacGregor C, Barton P, Banks SC, Crane M, Michael D, Okada S, Berry L, Florance D, Gill M (2016) Temporal trends in mammal responses to fire reveals the complex effects of fire regime attributes. Ecol Appl 26:557–573. https://doi.org/10.1890/15-0575
Linkie M, Ridout MS (2011) Assessing tiger–prey interactions in Sumatran rainforests. J Zool 284:224–229
Lino S, Sillero N, Torres J, Santos X, Álvares F (2019) The role of fire on wolf distribution and breeding-site selection: insights from a generalist carnivore occurring in a fire-prone landscape. Landscape Urban Plann 183:111–121
Macdonald DW (1983) The ecology of carnivore social behaviour. Nature 301:379–384
Mandelik Y, Jones M, Dayan T (2003) Structurally complex habitat and sensory adaptations mediate the behavioural responses of a desert rodent to an indirect cue for increased predation risk. Evol Ecol Res 5:501–515
Mariani M, Holz A, Veblen TT, Williamson G, Fletcher M-S, Bowman DM (2018) Climate change amplifications of climate-fire teleconnections in the Southern Hemisphere. Geophys Res Lett 45:5071–5081
Matter WJ, Mannan RW (2005) How do prey persist? J Wildl Manag 69:1315–1320
Mayfield MM, Levine JM (2010) Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecol Lett 13:1085–1093. https://doi.org/10.1111/j.1461-0248.2010.01509.x
Mazzamuto MV, Mazzella MN, Merrick MJ, Koprowski JL (2020) Fire impacts on a forest obligate: Western gray squirrel response to burn severity. Mamm Biol 100:295–303
McHugh D, Goldingay RL, Letnic M (2022) Occupancy and co-occurrence patterns of endemic mammals and introduced predators across a broad geographical gradient in eastern Australia. Anim Biodivers Conserv 31:989–1021. https://doi.org/10.1007/s10531-022-02374-0
Michel A, Johnson JR, Szeligowski R, Ritchie EG, Sih A (2023) Integrating sensory ecology and predator-prey theory to understand animal responses to fire. Ecol Lett 26:1050–1070
Moher D, Liberati A, Tetzlaff J, Altman DG, Group* P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097
Monamy V, Fox BJ (2000) Small mammal succession is determined by vegetation density rather than time elapsed since disturbance. Austral Ecol 25:580–587
Monamy V, Fox BJ (2010) Responses of two species of heathland rodents to habitat manipulation: vegetation density thresholds and the habitat accommodation model. Austral Ecol 35:334–347. https://doi.org/10.1111/j.1442-9993.2009.02042.x
Monterroso P, Alves PC, Ferreras P (2013) Catch me if you can: diel activity patterns of mammalian prey and predators. Ethology 119:1044–1056. https://doi.org/10.1111/eth.12156
Moreira F, Russo D (2007) Modelling the impact of agricultural abandonment and wildfires on vertebrate diversity in Mediterranean Europe. Landscape Ecol 22:1461–1476. https://doi.org/10.1007/s10980-007-9125-3
Moreno JM, Oechel WC (1994) The role of fire in Mediterranean-type ecosystems. Springer, New York, New York, NY
Morin PJ (2011) Community ecology, 2nd edn. John Wiley & Sons, Oxford, United Kingdom
Morris G, Hostetler JA, Conner LM, Oli MK (2011a) Effects of prescribed fire, supplemental feeding, and mammalian predator exclusion on hispid cotton rat populations. Oecologia 167:1005–1016. https://doi.org/10.1007/s00442-011-2053-6
Morris G, Hostetler JA, Oli MK, Conner LM (2011b) Effects of predation, fire, and supplemental feeding on populations of two species of Peromyscus mice. J Mammal 92:934–944. https://doi.org/10.1644/10-MAMM-A-419.1
Moyo S (2022) Community responses to fire: a global meta-analysis unravels the contrasting responses of fauna to fire. Earth 3:1087–1111
Nalliah R, Sitters H, Smith A, Di Stefano J (2022) Untangling the influences of fire, habitat and introduced predators on the endangered heath mouse. Anim Conserv 25:208–220. https://doi.org/10.1111/acv.12731
Nimmo DG, Avitabile S, Banks SC, Bird RB, Callister K, Clarke MF, Dickman CR, Doherty TS, Driscoll DA, Greenville AC, Haslem A, Kelly LT, Kenny SA, Lahoz-Monfort JJ, Lee C, Leonard S, Moore H, Newsome TM, Parr CL, Ritchie EG, Schneider K, Turner JM, Watson S, Westbrooke M, Wouters M, White M, Bennett AF (2019) Animal movements in fire-prone landscapes. Biol Rev 94:981–998. https://doi.org/10.1111/brv.12486
Nimmo DG, Carthey AJ, Jolly CJ, Blumstein DT (2021) Welcome to the Pyrocene: animal survival in the age of megafire. Global Change Biol 27:5684–5693
Nimmo DG, Jolly CJ, Carthey AJ (2022) Megafire: the Darwinian guillotine? Aust Zool 42:217–222
Nolan RH, Boer MM, Collins L, Resco de Dios V, Clarke HG, Jenkins M, Kenny B, Bradstock RA (2020) Causes and consequences of eastern Australia’s 2019–20 season of mega-fires. Global Change Biol 26:1039–1041
Paine RT (1980) Food webs: linkage, interaction strength and community infrastructure. J Anim Ecol 49:667–685
Palmer MS, Fieberg J, Swanson A, Kosmala M, Packer C (2017) A ‘dynamic’ landscape of fear: prey responses to spatiotemporal variations in predation risk across the lunar cycle. Ecol Lett 20:1364–1373
Palomares F, Caro TM (1999) Interspecific killing among mammalian carnivores. Am Nat 153:492–508
Parks SA, Dillon GK, Miller C (2014) A new metric for quantifying burn severity: the relativized burn ratio. Remote Sens 6:1827–1844
Pastro LA, Dickman CR, Letnic M (2011) Burning for biodiversity or burning biodiversity? Prescribed burn vs. wildfire impacts on plants, lizards, and mammals. Ecol Appl 21:3238–3253. https://doi.org/10.1890/10-2351.1
Paula S, Arianoutsou M, Kazanis D, Tavsanoglu Ç, Lloret F, Buhk C, Ojeda F, Luna B, Moreno JM, Rodrigo A (2009) Fire-related traits for plant species of the Mediterranean Basin. Ecology 90:1420–1420
Pausas JG (2004) Changes in fire and climate in the eastern Iberian Peninsula (Mediterranean basin). Clim Change 63:337–350
Pausas JG, Keeley JE (2009) A burning story: the role of fire in the history of life. Bioscience 59:593–601. https://doi.org/10.1525/bio.2009.59.7.10
Pausas JG, Keeley JE (2014) Evolutionary ecology of resprouting and seeding in fire-prone ecosystems. New Phytol 204:55–65. https://doi.org/10.1111/nph.12921
Pausas JG, Parr CL (2018) Towards an understanding of the evolutionary role of fire in animals. Evol Ecol 32:113–125. https://doi.org/10.1007/s10682-018-9927-6
Pausas JG, Llovet J, Rodrigo A, Vallejo R (2008) Are wildfires a disaster in the Mediterranean basin? - a review. Int J Wildland Fire 17:713–723. https://doi.org/10.1071/WF07151
Peckarsky BL, Abrams PA, Bolnick DI, Dill LM, Grabowski JH, Luttbeg B, Orrock JL, Peacor SD, Preisser EL, Schmitz OJ (2008) Revisiting the classics: considering nonconsumptive effects in textbook examples of predator–prey interactions. Ecology 89:2416–2425
Pellerin S, Huot J, Côté SD (2006) Long-term effects of deer browsing and trampling on the vegetation of peatlands. Biol Conserv 128:316–326
Penteriani V, Kuparinen A, del Mar DM, Palomares F, López-Bao JV, Fedriani JM, Calzada J, Moreno S, Villafuerte R, Campioni L (2013) Responses of a top and a meso predator and their prey to moon phases. Oecologia 173:753–766
Posłluszny M, Pilot M, Goszczyński J, Gralak B (2007) Diet of sympatric pine marten (Martes martes) and stone marten (Martes foina) identified by genotyping of DNA from faeces. Ann Zool Fenn 44:269–284
Prodon R, Fons R, Athias-Binche R (1987) The impact of fire on animal communities in Mediterranean area. In: Trabaud L (ed) The role of fire in ecological systems. SPB Academic Publishers, The Hague, pp 121–157
Prugh LR, Golden CD (2014) Does moonlight increase predation risk? Meta-analysis reveals divergent responses of nocturnal mammals to lunar cycles. J Anim Ecol 83:504–514
Puig-Gironès R (2023) Can predators influence small rodent foraging activity rates immediately after wildfires? Int J Wildland Fire 32:1391–1403
Puig-Gironès R, Pons P (2020) Mice and habitat complexity attract carnivorans to recently burnt forests. Forests 11:855. https://doi.org/10.3390/f11080855
Puig-Gironès R, Brotons L, Pons P (2017) Aridity influences the recovery of vegetation and shrubland birds after wildfire. PLoS ONE 12:e0173599
Puig-Gironès R, Clavero M, Pons P (2018) Importance of internal refuges and the external unburnt area in the recovery of rodent populations after wildfire. Int J Wildland Fire 27:425–436. https://doi.org/10.1071/WF17102
Puig-Gironès R, Santos X, Bros V (2023) Temporal differences in snail diversity responses to wildfires and salvage logging. Environ Conserv 50:40–49
Recher HF, Christensen PE (1981) Fire and the evolution of the Australian biota. In: Keast A (ed) Ecological biogeography of Australia. Dr W Junk, The Hague, Boston, pp 135–162
Retana J, Maria Espelta J, Habrouk A, Luis OrdoÑEz J, de Solà-Morales F (2002) Regeneration patterns of three Mediterranean pines and forest changes after a large wildfire in northeastern Spain. Écoscience 9:89–97. https://doi.org/10.1080/11956860.2002.11682694
Robinson NM, Leonard SW, Ritchie EG, Bassett M, Chia EK, Buckingham S, Gibb H, Bennett AF, Clarke MF (2013) Refuges for fauna in fire-prone landscapes: their ecological function and importance. J Appl Ecol 50:1321–1329
Rossa M, Lovari S, Ferretti F (2021) Spatiotemporal patterns of wolf, mesocarnivores and prey in a Mediterranean area. Behav Ecol Sociobiol 75:1–13. https://doi.org/10.1007/s00265-020-02956-4
Rothermel RC, Deeming JE (1980) Measuring and interpreting fire behavior for correlation with fire effects. Intermountain Forest and Range Experiment Station, US Department of Agriculture, Forest Service
Ruxton GD (2009) Non-visual crypsis: a review of the empirical evidence for camouflage to senses other than vision. Phil Trans R Soc B 364:549–557. https://doi.org/10.1098/rstb.2008.0228
Santos X, Cheylan M (2013) Taxonomic and functional response of a Mediterranean reptile assemblage to a repeated fire regime. Biol Conserv 168:90–98. https://doi.org/10.1016/j.biocon.2013.09.008
Schuette PA, Diffendorfer JE, Deutschman DH, Tremor S, Spencer W (2014) Carnivore distributions across chaparral habitats exposed to wildfire and rural housing in southern California. Int J Wildland Fire 23:591–600
Seijo F, Gray R (2012) Pre-industrial anthropogenic fire regimes in transition: the case of Spain and its implications for fire governance in Mediterranean type biomes. Hum Ecol Rev 19:58–69
Sherratt TN, Beatty CD (2003) The evolution of warning signals as reliable indicators of prey defense. Am Nat 162:377–389. https://doi.org/10.1086/378047
Sih A, Englund G, Wooster D (1998) Emergent impacts of multiple predators on prey. Trends Ecol Evol 13:350–355
Simons LH (1991) Rodent dynamics in relation to fire in the Sonoran Desert. J Mammal 72:518–524
Smith AL (2018) Successional changes in trophic interactions support a mechanistic model of post-fire population dynamics. Oecologia 186:129–139
Smith JK (2000) Wildland fire in ecosystems: effects of fire on fauna. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Ft. Collins, CO
Sokos C, Birtsas P, Papaspyropoulos KG, Tsachalidis E, Giannakopoulosa A, Milis C, Spyrou V, Manolakou K, Valiakos G, Iakovakis C, Athanasiou LV, Sfougaris A, Billinis C (2016) Mammals and habitat disturbance: the case of brown hare and wildfire. Curr Zool 62:421–430. https://doi.org/10.1093/cz/zow020
Soulsbury CD, Iossa G, Baker PJ, White PC, Harris S (2011) Behavioral and spatial analysis of extraterritorial movements in red foxes (Vulpes vulpes). J Mammal 92:190–199
Sousa WP (1984) The role of disturbance in natural communities. Ann Rev Ecol Syst 15:353–391
Soyumert A, Tavsanoglu C, Macar O, Kainas BY, Gürkan B (2010) Presence of large and medium-sized mammals in a burned pine forest in southwestern Turkey. Hystrix 21:97–102
Soyumert A, Ertürk A, Tavşanoğlu Ç (2020) Fire-created habitats support large mammal community in a Mediterranean landscape. Mammal Res 65:323–330. https://doi.org/10.1007/s13364-019-00473-y
Spencer EE, Dickman CR, Wardle GM, Newsome TM, Greenville AC (2022) One year on: rapid assessment of fauna and red fox diet after the 2019–20 mega-fires in the Blue Mountains, New South Wales. Aust Zool 42:304–325
Stephens SL, Burrows N, Buyantuyev A, Gray RW, Keane RE, Kubian R, Liu S, Seijo F, Shu L, Tolhurst KG (2014) Temperate and boreal forest mega-fires: characteristics and challenges. Front Ecol Environ 12:115–122
Sutherland EF, Dickman CR (1999) Mechanisms of recovery after fire by rodents in the Australian environment: a review. Wildl Res 26:405–419. https://doi.org/10.1071/WR97045
Swan M, Christie F, Sitters H, York A, Di Stefano J (2015) Predicting faunal fire responses in heterogeneous landscapes: the role of habitat structure. Ecol Appl 25:2293–2305. https://doi.org/10.1890/14-1533.1
Taylor P, Swan M, Sitters H, Smith A, Di Stefano J (2023) Small mammals reduce activity during high moon illumination under risk of predation by introduced predators. Sci Rep 13:10532
Terborgh J, Estes JA (2013) Trophic cascades: predators, prey, and the changing dynamics of nature. Island press, Washington, DC
Tomas WM, Berlinck CN, Chiaravalloti RM, Faggioni GP, Strüssmann C, Libonati R, Abrahão CR, do Vallel Avarenga G, de Faria Bacellar AE, de Queiroz Batista FR (2021) Distance sampling surveys reveal 17 million vertebrates directly killed by the 2020’s wildfires in the Pantanal, Brazil. Sci Rep 11:23547
Torre I, Díaz M (2004) Small mammal abundance in Mediterranean post-fire habitats: a role for predators? Acta Oecol 25:137–142. https://doi.org/10.1016/j.actao.2003.10.007
Torre I, Jaime-González C, Díaz M (2022) Habitat suitability for small mammals in Mediterranean landscapes: how and why shrubs matter. Sustainability 14:1562. https://doi.org/10.3390/su14031562
Trabaud L (1994) Postfire plant community dynamics in the Mediterranean basin. In: Moreno JM, Oechel WC (eds) The role of fire in Mediterranean-type ecosystems. Springer, New York, NY, pp 1–15
Tsukada H (1997) A division between foraging range and territory related to food distribution in the red fox. J Ethol 15:27–37
Turco M, Rosa-Cánovas JJ, Bedia J, Jerez S, Montávez JP, Llasat MC, Provenzale A (2018) Exacerbated fires in Mediterranean Europe due to anthropogenic warming projected with non-stationary climate-fire models. Nat Commun 9:1–9
Vanak AT, Fortin D, Thaker M, Ogden M, Owen C, Greatwood S, Slotow R (2013) Moving to stay in place: behavioral mechanisms for coexistence of African large carnivores. Ecology 94:2619–2631
Verdolin JL (2006) Meta-analysis of foraging and predation risk trade-offs in terrestrial systems. Behav Ecol Sociobiol 60:457–464. https://doi.org/10.1007/s00265-006-0172-6
Virgós E, Cabezas-Díaz S, Lozano J, Mangas J (2010) Spatial distribution models in a frugivorous carnivore, the stone marten (Martes foina): is the fleshy-fruit availability a useful predictor? Anim Biol 60:423–436
Whelan RJ (1995) The ecology of fire. Cambridge University Press, Cambridge
Whelan RJ, Rodgerson L, Dickman CR (2002) Critical life processes of plants and animals: developing a process-based understanding of population changes in fire-prone landscapes. In: Bradstock RA, Williams JE, Gill AM (eds) Flammable Australia: the fire regimes and biodiversity of a continent. Cambridge University Press, Cambridge, pp 94–124
Williams J (2013) Exploring the onset of high-impact mega-fires through a forest land management prism. For Ecol Manag 294:4–10
Williams AP, Abatzoglou JT, Gershunov A, Guzman-Morales J, Bishop DA, Balch JK, Lettenmaier DP (2019) Observed impacts of anthropogenic climate change on wildfire in California. Earth’s Future 7:892–910
Woinarski JCZ, Williams RJ, Price O, Rankmore B (2005) Landscapes without boundaries: wildlife and their environments in northern Australia. Wildl Res 32:377–388
Zwolak R, Pearson DE, Ortega YK, Crone EE (2012) Mechanisms driving postfire abundance of a generalist mammal. Can J Zool 90:51–60
Acknowledgements
The authors would like to thank the University of Pisa Working group (Tavolo Tecnico) for the Monte Pisano fires, which is dedicated to support the local community to study the effects of fires and to promote restoration of burnt areas. Moreover, we would like to thank the University of Pisa and the Department of Biology for funding the PhD scholarship for Tomassini Orlando following the recommendation provided by the Tavolo Tecnico. In particular, we would like to thank Dimitri Giunchi, Gianni Bedini and Giulio Petroni for financial support, and John Kastelic for his writing assistance. Lastly, we are in debt with two anonymous reviewers whose comments helped to improve the quality of the manuscript.
Funding
Open access funding provided by Università di Pisa within the CRUI-CARE Agreement. Regione Toscana.
Author information
Authors and Affiliations
Contributions
Orlando Tomassini conducted the literature research and wrote the review. Alessandro Massolo conceptualized the study and reviewed and provided feedback on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Handling editor: Carlos Rouco.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tomassini, O., Massolo, A. From fire to recovery: temporal-shift of predator–prey interactions among mammals in Mediterranean ecosystems. Mamm Biol (2024). https://doi.org/10.1007/s42991-024-00439-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42991-024-00439-x